CarE1 and GST1 Are Involved in Beta-Cypermethrin Resistance in Field Populations of the Mirid Bug, Apolygus lucorum
Simple Summary
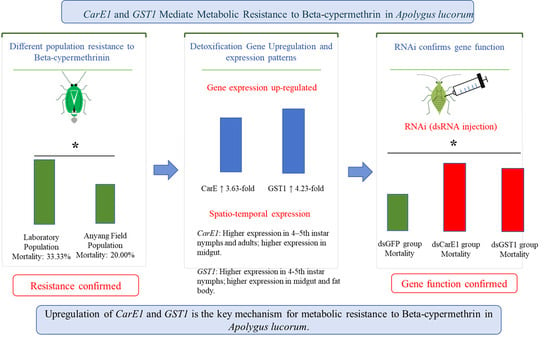
Abstract
1. Introduction
2. Materials and Methods
2.1. Insect
2.2. Toxicity Assessment of Apolygus lucorum
2.3. Determination of Detoxifying Enzyme Activity
2.3.1. Enzyme Preparation
2.3.2. CarE and GST Enzyme Activity Assays
2.4. Detoxification-Related Genes Expression Measured by qRT-PCR
2.5. RNA Interference
2.6. Data Analyses
3. Results
3.1. Detection of LC50 in Different Populations of Apolygus lucorum
3.2. Detection of Resistance Level of Field Population
3.3. Detection of CarE and GST Enzyme Activity
3.4. Detection of CarE1 and GST1 Gene Expression
3.5. Spatial and Temporal Expression Patterns of CarE1 and GST1 Genes in Apolygus lucorum
3.6. Effects of RNA Interference on the Mortality of Apolygus lucorum
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yang, L.; Chen, H.; Zheng, Q.; Luo, P.; Yan, W.; Huang, S.; Cheng, D.; Hong Xu, H.; Zhang, Z. A β-cyclodextrin-functionalized metal–organic framework enhances the insecticidal activity of indoxacarb by affecting amino acid metabolism in red imported fire ants. Chem. Eng. J. 2023, 458, 141417. [Google Scholar] [CrossRef]
- Lu, Y.H.; Qiu, F.; Feng, H.Q.; Li, H.B.; Yang, Z.C.; Wyckhuys, K.A.G.; Wu, K.M. Species composition and seasonal abundance of pestiferous plant bugs (Hemiptera: Miridae) on Bt Cotton in China. Crop Prot. 2008, 27, 465–472. [Google Scholar] [CrossRef]
- Bergé, J.B.; Ricroch, A.E. Emergence of minor pests becoming major pests in GE cotton in China: What are the reasons? What are the alternatives practices to this change of status? GM Crops 2010, 1, 214–219. [Google Scholar] [CrossRef]
- Li, G.; Feng, H.; McNeil, J.N.; Liu, B.; Chen, P.; Qiu, F. Impacts of transgenic Bt cotton on a non-target pest, Apolygus lucorum (Meyer-Dür) (Hemiptera: Miridae), in northern China. Crop Prot. 2011, 30, 1573–1578. [Google Scholar] [CrossRef]
- Lu, Y.; Wu, K.; Jiang, Y.; Xia, B.; Li, P.; Feng, H.; Wyckhuys, K.A.G.; Guo, Y. Mirid Bug Outbreaks in Multiple Crops Correlated with Wide-Scale Adoption of Bt Cotton in China. Science 2010, 328, 1151–1154. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Xiao, F.; Ojo, J.; Chao, W.H.; Ahmad, B.; Alam, A.; Abbas, S.; Abdelhafez, M.M.; Rahman, N.; Khan, K.A.; et al. Insect Resistance to Insecticides: Causes, Mechanisms, and Exploring Potential Solutions. Arch. Insect Biochem. 2025, 118, e70045. [Google Scholar] [CrossRef] [PubMed]
- Leigh, T.F.; Jackson, C.E.; Wynholds, P.F.; Cota, J.A. Toxicity of Selected Insecticides Applied Topically to Lygus hesperus. J. Econ. Entomol. 1977, 70, 42–44. [Google Scholar] [CrossRef]
- Hollingsworth, R.G.; Steinkraus, D.C.; Tugwell, N.P. Responses of Arkansas Populations of Tarnished Plant Bugs (Heteroptera: Miridae) to Insecticides, and Tolerance Differences Between Nymphs and Adults. J. Econ. Entomol. 1997, 90, 21–26. [Google Scholar] [CrossRef]
- Snodgrass, G.L.; Scott, W.P. Seasonal changes in pyrethroid resistance in tarnished plant bug (Heteroptera: Miridae) populations during a three-year period in the delta area of Arkansas, Louisiana, and Mississippi. J. Econ. Entomol. 2000, 93, 441–446. [Google Scholar] [CrossRef]
- Snodgrass, G.L.; Gore, J.; Abel, C.A.; Jackson, R. Acephate resistance in populations of the tarnished plant bug (Heteroptera: Miridae) from the Mississippi River Delta. J. Econ. Entomol. 2009, 102, 699–707. [Google Scholar] [CrossRef]
- Tan, Y.; Zhang, S.; Gao, X. Monitoring the insecticide resistance of the cotton bugs Apolygus lucorum and Adelphocoris suturalis. J. Appl. Entomol. 2012, 49, 348–358. [Google Scholar]
- Liu, J.; Li, T.; Huang, J.; Kang, Z.; Yang, Y.; Wu, Y.; Wu, S. Resistance to beta-cypermethrin and chlorpyrifos in populations of Apolygus lucorum from the Yellow and ChangjiangRiver Cotton Growing Areas of China. J. Appl. Entomol. 2015, 52, 616–622. [Google Scholar]
- Zhang, S.; Ma, Y.; Min, H.; Yu, X.; Li, N.; Rui, C.; Gao, X. Insecticide resistance monitoring and management demonstration of major insect pests in the main cotton-growing areas of northern China. Acta Entomol. Sin. 2016, 59, 1238–1245. [Google Scholar]
- Parys, K.A.; Luttrell, R.G.; Snodgrass, G.L.; Portilla, M.R. Patterns of Tarnished Plant Bug (Hemiptera: Miridae) Resistance to Pyrethroid Insecticides in the Lower Mississippi Delta for 2008–2015: Linkage to Pyrethroid Use and Cotton Insect Management. J. Insect Sci. 2018, 18, 29. [Google Scholar] [CrossRef]
- Bras, A.; Roy, A.; Heckel, D.G.; Anderson, P.; Karlsson Green, K. Pesticide resistance in arthropods: Ecology matters too. Ecol. Lett. 2022, 25, 1746–1759. [Google Scholar] [CrossRef]
- Coetzee, M.; Koekemoer, L.L. Molecular systematics and insecticide resistance in the major African malaria vector Anopheles funestus. Annu. Rev. Entomol. 2013, 58, 393–412. [Google Scholar] [CrossRef]
- Mugenzi, L.M.J.; Menze, B.D.; Tchouakui, M.; Wondji, M.J.; Irving, H.; Tchoupo, M.; Hearn, J.; Weedall, G.D.; Riveron, J.M.; Wondji, C.S. Cis-regulatory CYP6P9b P450 variants associated with loss of insecticide-treated bed net efficacy against Anopheles funestus. Nat. Commun. 2019, 10, 4652. [Google Scholar] [CrossRef]
- Tognarelli, J.; Moya, P.R.; González, C.R.; Collao-Ferrada, X. Global distribution and impact of knockdown resistance mutations in Aedes aegypti on pyrethroid resistance. Parasit. Vectors 2025, 18, 382. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Hu, X.; Hu, J.; Xu, W.; Wang, Q.; Liu, X.; Guo, L. UDP-glycosyltransferase confers anthranilic diamide resistance in Bemisia tabaci. J. Adv. Res. 2025; in press. [Google Scholar] [CrossRef]
- Li, L.; Yang, Q.; Liu, M.; Lin, S.; Hua, W.; Shi, D.; Yan, J.; Shi, X.; Hoffmann, A.A.; Zhu, B.; et al. Symbiotic bacteria mediate chemical-insecticide resistance but enhance the efficacy of a biological insecticide in diamondback moth. Curr. Biol. 2025, 35, 4494–4508.e4493. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.; Song, Y.; Zeng, R. The role of cytochrome P450-mediated detoxification in insect adaptation to xenobiotics. Curr. Opin. Insect Sci. 2021, 43, 103–107. [Google Scholar] [CrossRef]
- Kshatriya, K.; Gershenzon, J. Disarming the defenses: Insect detoxification of plant defense-related specialized metabolites. Curr. Opin. Plant Biol. 2024, 81, 102577. [Google Scholar] [CrossRef]
- Koirala, B.K.S.; Moural, T.; Zhu, F. Functional and Structural Diversity of Insect Glutathione S-transferases in Xenobiotic Adaptation. Int. J. Biol. Sci. 2022, 18, 5713–5723. [Google Scholar] [CrossRef]
- Wheelock, C.E.; Shan, G.; Ottea, J. Overview of Carboxylesterases and Their Role in the Metabolism of Insecticides. J. Pest Sci. 2005, 30, 75–83. [Google Scholar] [CrossRef]
- Dai, L.; Gao, H.; Ye, J.; Fu, D.; Sun, Y.; Chen, H. Isolation of CarE genes from the Chinese white pine beetle Dendroctonus armandi (Curculionidae: Scolytinae) and their response to host chemical defense. Pest Manag. Sci. 2019, 75, 986–997. [Google Scholar] [CrossRef]
- Tang, B.; Dai, W.; Qi, L.; Du, S.; Zhang, C. Functional Characterization of an α-Esterase Gene Associated with Malathion Detoxification in Bradysia odoriphaga. J. Agric. Food Chem. 2020, 68, 6076–6083. [Google Scholar] [CrossRef]
- Lu, K.; Li, Y.; Xiao, T.; Sun, Z. The metabolic resistance of Nilaparvata lugens to chlorpyrifos is mainly driven by the carboxylesterase CarE17. Ecotoxicol. Environ. Safe 2022, 241, 113738. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Liu, J.Y.; Wang, W.; Mota-Sanchez, D.; He, S.; Shi, Y.; Yang, X.Q. Glutathione S-Transferase Genes are Involved in Lambda-Cyhalothrin Resistance in Cydia pomonella via Sequestration. J. Agric. Food Chem. 2022, 70, 2265–2279. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.W.; Peng, M.L.; Chen, M.L.; Yuan, G.R.; Zheng, L.S.; Bai, W.J.; Smagghe, G.; Wang, J.J. A glutathione S-transferase (BdGSTd9) participates in malathion resistance via directly depleting malathion and its toxic oxide malaoxon in Bactrocera dorsalis (Hendel). Pest Manag. Sci. 2020, 76, 2557–2568. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Zhou, H.; Wan, F.; Liu, X.; Han, Z. Population dynamics and damage of Lygus lucorum in Bt cotton fields under two control measures. Chin. Bull. Entomol. 2005, 42, 424–428. [Google Scholar]
- Lu, K.; Wang, Y.; Chen, X.; Zhang, Z.; Li, Y.; Li, W.; Zhou, Q. Characterization and functional analysis of a carboxylesterase gene associated with chlorpyrifos resistance in Nilaparvata lugens (Stål). Comp. Biochem. Phys. C 2017, 203, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.C.; Yao, J.; Adamczyk, J.; Luttrell, R. Feeding toxicity and impact of imidacloprid formulation and mixtures with six representative pesticides at residue concentrations on honey bee physiology (Apis mellifera). PLoS ONE 2017, 12, e0178421. [Google Scholar] [CrossRef]
- Niu, L.; Liu, F.; Zhang, S.; Luo, J.; Zhang, L.; Ji, J.; Gao, X.; Ma, W.; Cui, J. Transgenic insect-resistant Bt cotton expressing Cry1Ac/CpTI does not affect the mirid bug Apolygus lucorum. Environ. Pollut. 2020, 264, 114762. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.; Luo, J.; Alariqi, M.; Xu, Z.; Wang, A.; Zafar, M.N.; Ren, J.; Wang, F.; Liu, X.; Xin, Y. Silencing of a LIM gene in cotton exhibits enhanced resistance against Apolygus lucorum. J. Cell. Physiol. 2021, 236, 5921–5936. [Google Scholar] [CrossRef] [PubMed]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative C(T) method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef]
- Cheng, T.; Wu, J.; Wu, Y.; Chilukuri, R.V.; Huang, L.; Yamamoto, K.; Feng, L.; Li, W.; Chen, Z.; Guo, H.; et al. Genomic adaptation to polyphagy and insecticides in a major East Asian noctuid pest. Nat. Ecol. Evol. 2017, 1, 1747–1756. [Google Scholar] [CrossRef]
- Song, X.W.; Zhong, Q.S.; Ji, Y.H.; Zhang, Y.M.; Tang, J.; Feng, F.; Bi, J.X.; Xie, J.; Li, B. Characterization of a sigma class GST (GSTS6) required for cellular detoxification and embryogenesis in Tribolium castaneum. Insect Sci. 2022, 29, 215–229. [Google Scholar] [CrossRef]
- Chen, H.; Wu, Y.; Gao, J.; Xu, X.; Wang, X.; Yang, J. Resistance and metabolic pathway of Spodoptera frugiperda population to beta cypermethrin in Sichuan. J. South China Agric. Univ. 2025, 46, 488–498. [Google Scholar]
- Li, X.; Schuler, M.A.; Berenbaum, M.R. Molecular mechanisms of metabolic resistance to synthetic and natural xenobiotics. Annu. Rev. Entomol. 2007, 52, 231–253. [Google Scholar] [CrossRef]
- Li, F.; Han, Z. Mutations in acetylcholinesterase associated with insecticide resistance in the cotton aphid, Aphis gossypii Glover. Insect Biochem. Mol. 2004, 34, 397–405. [Google Scholar] [CrossRef]
- Feng, Q.L.; Ladd, T.R.; Tomkins, B.L.; Sundaram, M.; Sohi, S.S.; Retnakaran, A.; Davey, K.G.; Palli, S.R. Spruce budworm (Choristoneura fumiferana) juvenile hormone esterase: Hormonal regulation, developmental expression and cDNA cloning. Mol. Cell. Endocrinol. 1999, 148, 95–108. [Google Scholar] [CrossRef]
- Huang, S.; Qin, W.; Chen, Q. Cloning, sequence analysis and expression levels of a carboxylesterase gene from Spodoptera litura (Fab.) (Lepidoptera: Noctuidae). Acta Entomol. Sin. 2010, 53, 29–37. [Google Scholar]
- Ranson, H.; Rossiter, L.; Ortelli, F.; Jensen, B.; Wang, X.; Roth, C.W.; Collins, F.H.; Hemingway, J. Identification of a novel class of insect glutathione S-transferases involved in resistance to DDT in the malaria vector Anopheles gambiae. Biochem. J. 2001, 359, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Zhang, S.; Lu, F.; Zhang, K.; Han, Q.; Ying, Q.; Zhang, X.; Zhang, C.; Zhou, S.; Chen, A. Cross-resistance, fitness costs, and biochemical mechanism of laboratory-selected resistance to tenvermectin A in Plutella xylostella. Pest Manag. Sci. 2021, 77, 2826–2835. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Zhu, Y.C.; Portilla, M.; Zhang, M.; Reddy, G.V.P. The mechanisms of metabolic resistance to pyrethroids and neonicotinoids fade away without selection pressure in the tarnished plant bug Lygus lineolaris. Pest Manag. Sci. 2023, 79, 3893–3902. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Xu, S.; Xu, Y.; Dong, J. Effects of Dinotefuran Stress on the Expression of Detoxifying Enzyme Systems and Transporter Genes in Bombyx mori Tissues Based on Transcriptome Sequencing Analysis. Acta Sericologica Sin. 2025, 51, 203–214. [Google Scholar]
- Brito, L.P.; Linss, J.G.; Lima-Camara, T.N.; Belinato, T.A.; Peixoto, A.A.; Lima, J.B.P.; Valle, D.; Martins, A.J. Assessing the effects of Aedes aegypti kdr mutations on pyrethroid resistance and its fitness cost. PLoS ONE 2013, 8, e60878. [Google Scholar]







| Gene | Orientation | Primer Sequences (5′–3′) | Application |
|---|---|---|---|
| β-actin | Forward | CGCCGACAGGATGCAGAAG | qRT-PCR |
| Reverse | CGAGGATGGAGCCACCGA | qRT-PCR | |
| RPL32 | Forward | CAAGCTCACGAGGAATTGGCG | qRT-PCR |
| Reverse | GAGGACTTTCCTGAAGCCGGT | qRT-PCR | |
| CarE1 | Forward | TCGCATCATCTACTCCGTTCACC | qRT-PCR |
| Reverse | GTCCACGTTCATCAGAGAGGTCA | qRT-PCR | |
| GST1 | Forward | CTGTTCAGGACCTACACTCTCGC | qRT-PCR |
| Reverse | TCACCGACATGTAAATGGTTATTCC | qRT-PCR | |
| CarE1 | Forward | gcgtaatacgactcactatagggGGATTGGACGGCATACAACC | RNAi |
| Reverse | gcgtaatacgactcactatagggGTTGCCTTTGTACTGCTCCC | RNAi | |
| GST1 | Forward | gcgtaatacgactcactatagggACTCATGAATGTTATCTCGCTGG | RNAi |
| Reverse | gcgtaatacgactcactatagggTAGCGGGTTGGGGAACGAC | RNAi |
| Strain | Toxicity Regression Equation | LC50 (mg/L) (95% CI) | RR | X2 Value | p Value |
|---|---|---|---|---|---|
| Laboratory Strain | y = 0.002x − 0.680 | 343.34 (267.24–433.52) | 1.0 | 528.47 | <0.01 |
| Ay field Strain | y = 0.001x − 0.771 | 700.45 (492.53–976.94) | 2.04 | 125.25 | <0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, H.; Song, W.; Wu, Q.; Xu, L.; Niu, L.; Tang, Q. CarE1 and GST1 Are Involved in Beta-Cypermethrin Resistance in Field Populations of the Mirid Bug, Apolygus lucorum. Insects 2026, 17, 66. https://doi.org/10.3390/insects17010066
Wang H, Song W, Wu Q, Xu L, Niu L, Tang Q. CarE1 and GST1 Are Involved in Beta-Cypermethrin Resistance in Field Populations of the Mirid Bug, Apolygus lucorum. Insects. 2026; 17(1):66. https://doi.org/10.3390/insects17010066
Chicago/Turabian StyleWang, Haojie, Weicheng Song, Qiyuan Wu, Liming Xu, Lin Niu, and Qingbo Tang. 2026. "CarE1 and GST1 Are Involved in Beta-Cypermethrin Resistance in Field Populations of the Mirid Bug, Apolygus lucorum" Insects 17, no. 1: 66. https://doi.org/10.3390/insects17010066
APA StyleWang, H., Song, W., Wu, Q., Xu, L., Niu, L., & Tang, Q. (2026). CarE1 and GST1 Are Involved in Beta-Cypermethrin Resistance in Field Populations of the Mirid Bug, Apolygus lucorum. Insects, 17(1), 66. https://doi.org/10.3390/insects17010066