Impaired Interhemispheric Synchrony in Parkinson’s Disease with Fatigue
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Clinical Assessment
2.2. Image Acquisition
2.3. Data Preprocessing
2.4. Voxel-Mirrored Homotopic Connectivity
2.5. Statistical Analysis
3. Results
3.1. Demographic and Clinical Characteristics
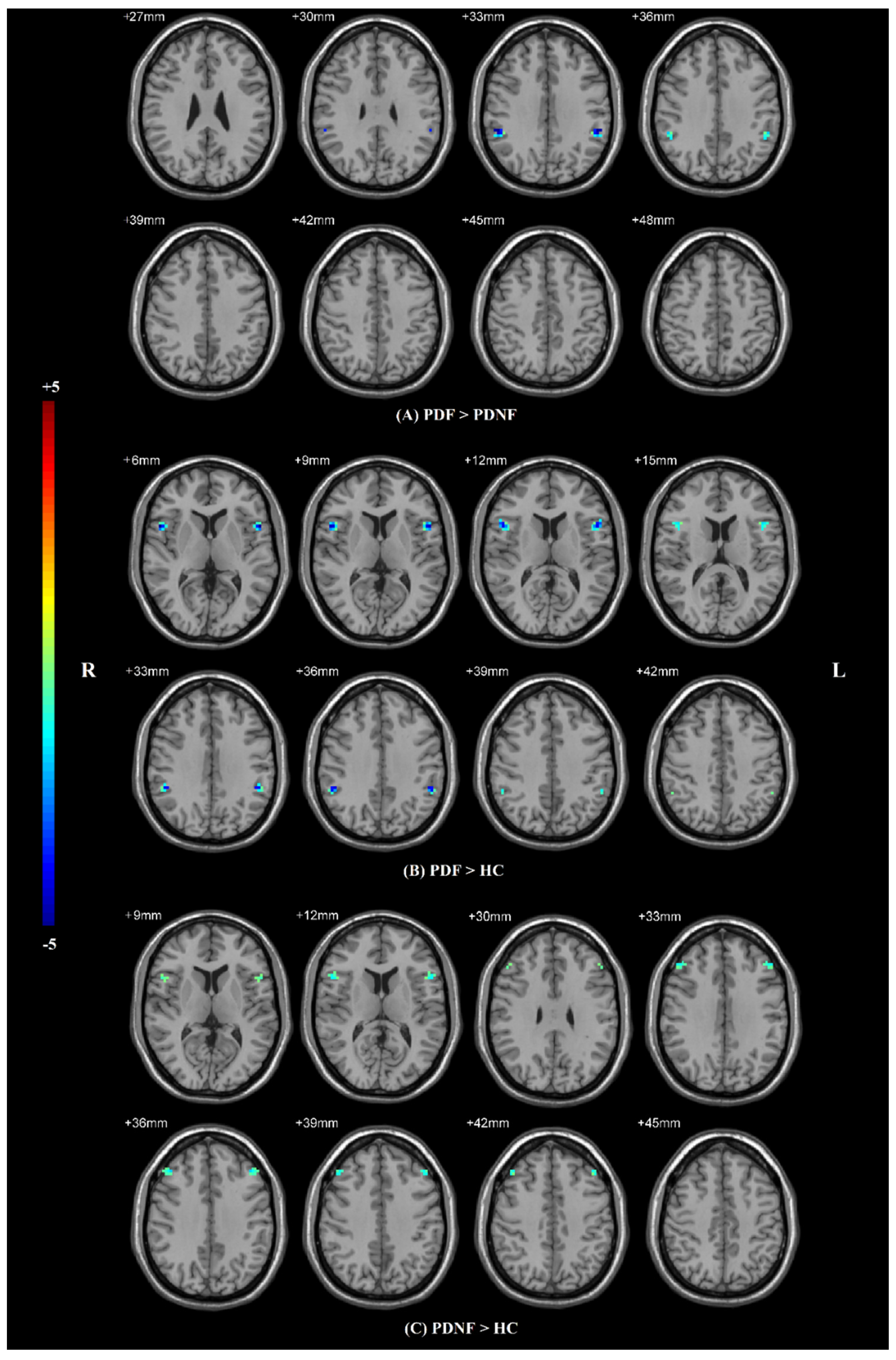
3.2. Voxel-Mirrored Homotopic Connectivity
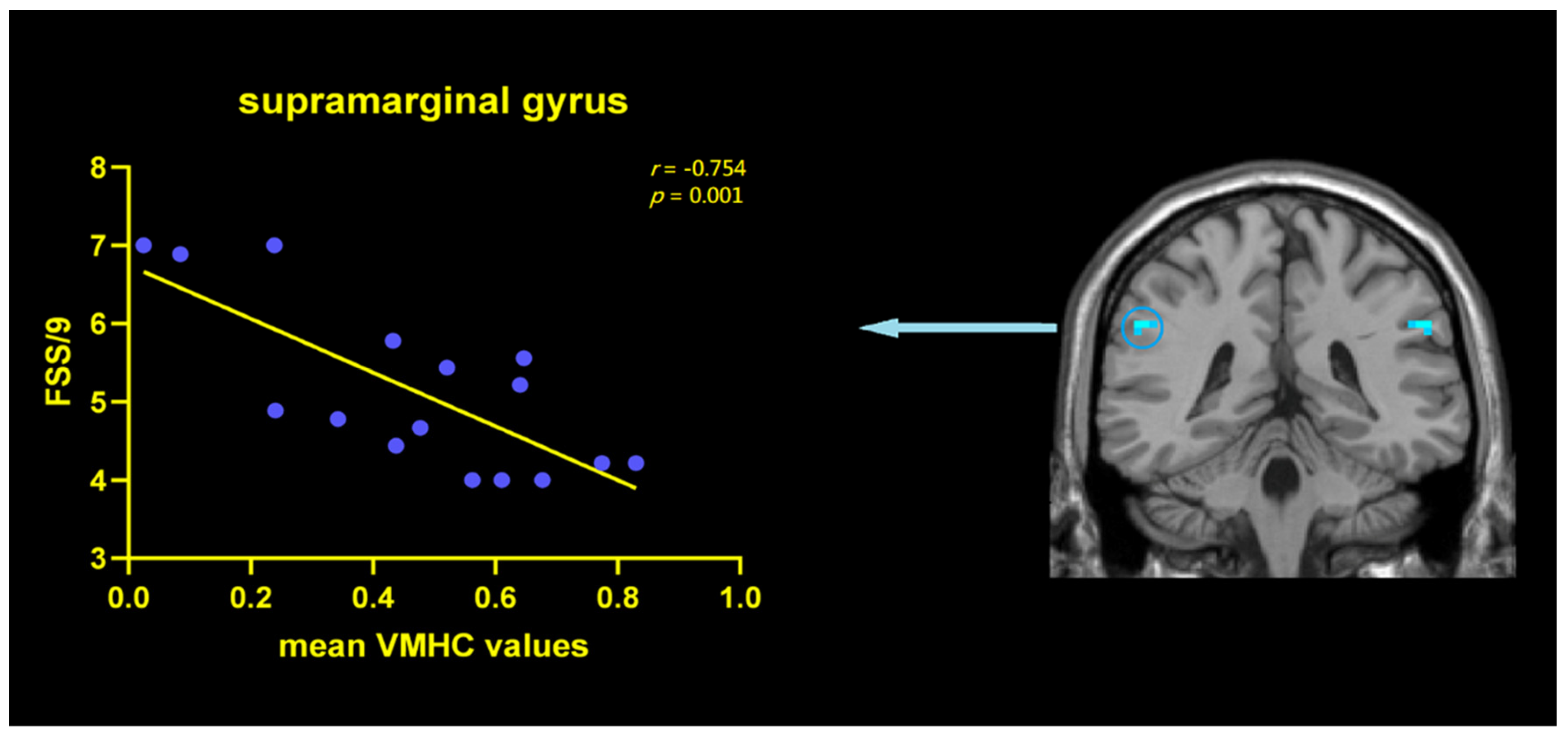
3.3. Correlation Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siciliano, M.; Trojano, L.; Santangelo, G.; De Micco, R.; Tedeschi, G.; Tessitore, A. Fatigue in Parkinson’s disease: A systematic review and meta-analysis. Mov. Disord. 2018, 33, 1712–1723. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, D.A.; Lees, A.J.; Schrag, A. What Are the Most Important Nonmotor Symptoms in Patients with Parkinson’s Disease and Are We Missing Them? Mov. Disord. 2010, 25, 2493–2500. [Google Scholar] [CrossRef] [PubMed]
- Beiske, A.G.; Loge, J.H.; Hjermstad, M.J.; Svensson, E. Fatigue in Parkinson’s disease: Prevalence and associated factors. Mov. Disord. 2010, 25, 2456–2460. [Google Scholar] [CrossRef] [PubMed]
- Miwa, H.; Miwa, T. Fatigue in patients with Parkinson’s disease: Impact on quality of life. Intern. Med. 2011, 50, 1553–1558. [Google Scholar] [CrossRef][Green Version]
- Abe, K.; Takanashi, M.; Yanagihara, T. Fatigue in patients with Parkinson’s disease. Behav. Neurol. 2000, 12, 103–106. [Google Scholar] [CrossRef]
- Pavese, N.; Metta, V.; Bose, S.K.; Chaudhuri, K.R.; Brooks, D.J. Fatigue in Parkinson’s disease is linked to striatal and limbic serotonergic dysfunction. Brain 2010, 133, 3434–3443. [Google Scholar] [CrossRef]
- Zhang, L.; Li, T.; Yuan, Y.; Tong, Q.; Jiang, S.; Wang, M.; Wang, J.; Ding, J.; Xu, Q.; Zhang, K. Brain metabolic correlates of fatigue in Parkinson’s disease: A PET study. Int. J. Neurosci. 2018, 128, 330–336. [Google Scholar] [CrossRef]
- Boissoneault, J.; Letzen, J.; Lai, S.; O’Shea, A.; Craggs, J.; Robinson, M.E.; Staud, R. Abnormal resting state functional connectivity in patients with chronic fatigue syndrome: An arterial spin-labeling fMRI study. Magn. Reson. Imaging 2016, 34, 603–608. [Google Scholar] [CrossRef]
- Li, J.; Yuan, Y.; Wang, M.; Zhang, J.; Zhang, L.; Jiang, S.; Ding, J.; Zhang, K. Alterations in regional homogeneity of resting-state brain activity in fatigue of Parkinson’s disease. J. Neural Transm. 2017, 124, 1187–1195. [Google Scholar] [CrossRef]
- Cho, S.S.; Aminian, K.; Li, C.; Lang, A.E.; Houle, S.; Strafella, A.P. Fatigue in Parkinson’s disease: The contribution of cerebral metabolic changes. Hum. Brain Mapp. 2017, 38, 283–292. [Google Scholar] [CrossRef]
- Tessitore, A.; Giordano, A.; De Micco, R.; Caiazzo, G.; Russo, A.; Cirillo, M.; Esposito, F.; Tedeschi, G. Functional connectivity underpinnings of fatigue in “Drug-Naïve” patients with Parkinson’s disease. Mov. Disord. 2016, 31, 1497–1505. [Google Scholar] [CrossRef] [PubMed]
- Kempster, P.A.; Gibb, W.R.; Stern, G.M.; Lees, A. Asymmetry of substantia nigra neuronal loss in Parkinson’s disease and its relevance to the mechanism of levodopa related motor fluctuations. J. Neurol. Neurosurg. Psychiatry 1989, 52, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yang, Q.X.; Sun, X.; Vesek, J.; Mosher, Z.; Vasavada, M.; Chu, J.; Kanekar, S.; Shivkumar, V.; Venkiteswaran, K.; et al. MRI evaluation of asymmetry of nigrostriatal damage in the early stage of early-onset Parkinson’s disease. Parkinsonism Relat. Disord. 2015, 21, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Siciliano, M.; De Micco, R.; Giordano, A.; Di Nardo, F.; Russo, A.; Caiazzo, G.; De Mase, A.; Cirillo, M.; Tedeschi, G.; Trojano, L.; et al. Supplementary motor area functional connectivity in “drug-naïve” Parkinson’s disease patients with fatigue. J. Neural Transm. 2020, 127, 1133–1142. [Google Scholar] [CrossRef] [PubMed]
- Krall, S.C.; Rottschy, C.; Oberwelland, E.; Bzdok, D.; Fox, P.T.; Eickhoff, S.B.; Fink, G.R.; Konrad, K. The role of the right temporoparietal junction in attention and social interaction as revealed by ALE meta-analysis. Brain Struct. Funct. 2015, 220, 587–604. [Google Scholar] [CrossRef]
- Zuo, X.N.; Kelly, C.; Di Martino, A.; Mennes, M.; Margulies, D.S.; Bangaru, S.; Grzadzinski, R.; Evans, A.C.; Zang, Y.F.; Castellanos, F.X.; et al. Growing together and growing apart: Regional and sex differences in the lifespan developmental trajectories of functional homotopy. J. Neurosci. 2010, 30, 15034–15043. [Google Scholar] [CrossRef]
- Hughes, A.J.; Daniel, S.E.; Kilford, L.; Lees, A.J. Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: A clinico-pathological study of 100 cases. J. Neurol. Neurosurg. Psychiatry 1992, 55, 181–184. [Google Scholar] [CrossRef]
- Friedman, J.H.; Alves, G.; Hagell, P.; Marinus, J.; Marsh, L.; Martinez-Martin, P.; Goetz, C.G.; Poewe, W.; Rascol, O.; Sampaio, C.; et al. Fatigue rating scales critique and recommendations by the Movement Disorders Society task force on rating scales for Parkinson’s disease. Mov. Disord. 2010, 25, 805–822. [Google Scholar] [CrossRef]
- Tomlinson, C.L.; Stowe, R.; Patel, S.; Rick, C.; Gray, R.; Clarke, C.E. Systematic review of levodopa dose equivalency reporting in Parkinson’s disease. Mov. Disord. 2010, 25, 2649–2653. [Google Scholar] [CrossRef]
- Gan, C.; Wang, M.; Si, Q.; Yuan, Y.; Zhi, Y.; Wang, L.; Ma, K.; Zhang, K. Altered interhemispheric synchrony in Parkinson’s disease patients with levodopa-induced dyskinesias. NPJ Parkinson’s Dis. 2020, 6, 14. [Google Scholar] [CrossRef]
- Cubo, E.; Martinez Martín, P.; Martin-Gonzalez, J.A.; Rodríguez-Blázquez, C.; Kulisevsky, J.; LEP Group Members. Motor laterality asymmetry and nonmotor symptoms in Parkinson’s disease. Mov. Disord. 2010, 25, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Tan, Y.Y.; Liu, D.Q.; Herzallah, M.M.; Lapidow, E.; Wang, Y.; Zang, Y.F.; Gluck, M.A.; Chen, S.D. Motor-symptom laterality affects acquisition in Parkinson’s disease: A cognitive and functional magnetic resonance imaging study. Mov. Disord. 2017, 32, 1047–1055. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Z.; Merkitch, D.; Karaman, M.M.; Zhang, J.; Sui, Y.; Goldman, J.G.; Zhou, X.J. High-Spatial-Resolution Diffusion MRI in Parkinson Disease: Lateral Asymmetry of the Substantia Nigra. Radiology. 2019, 291, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Kluger, B.M.; Zhao, Q.; Tanner, J.J.; Schwab, N.A.; Levy, S.A.; Burke, S.E.; Huang, H.; Ding, M.; Price, C. Structural brain correlates of fatigue in older adults with and without Parkinson’s disease. NeuroImage Clin. 2019, 22, 101730. [Google Scholar] [CrossRef]
- Bzdok, D.; Schilbach, L.; Vogeley, K.; Schneider, K.; Laird, A.R.; Langner, R.; Eickhoff, S.B. Parsing the neural correlates of moral cognition: ALE meta-analysis on morality, theory of mind, and empathy. Brain Struct. Funct. 2012, 217, 783–796. [Google Scholar] [CrossRef]
- Rubinstein, D.Y.; Camarillo-Rodriguez, L.; Serruya, M.D.; Herweg, N.A.; Waldman, Z.J.; Wanda, P.A.; Sharan, A.D.; Weiss, S.A.; Sperling, M.R. Contribution of left supramarginal and angular gyri to episodic memory encoding: An intracranial EEG study. NeuroImage 2021, 225, 117514. [Google Scholar] [CrossRef]
- Pauletti, C.; Mannarelli, D.; Locuratolo, N.; Currà, A.; Marinelli, L.; Fattapposta, F. Central fatigue and attentional processing in Parkinson’s disease: An event-related potentials study. Clin. Neurophysiol. 2019, 130, 692–700. [Google Scholar] [CrossRef]
- Luo, C.; Guo, X.; Song, W.; Zhao, B.; Cao, B.; Yang, J.; Gong, Q.; Shang, H.F. Decreased Resting-State Interhemispheric Functional Connectivity in Parkinson’s Disease. BioMed Res. Int. 2015, 2015, 692684. [Google Scholar] [CrossRef]
- Favre, P.; Kanske, P.; Engen, H.; Singer, T. Decreased emotional reactivity after 3-month socio-affective but not attention- or meta-cognitive-based mental training: A randomized, controlled, longitudinal fMRI study. NeuroImage 2021, 237, 118132. [Google Scholar] [CrossRef]
- Suo, X.; Ding, H.; Li, X.; Zhang, Y.; Liang, M.; Zhang, Y.; Yu, C.; Qin, W. Anatomical and functional coupling between the dorsal and ventral attention networks. NeuroImage 2021, 232, 117868. [Google Scholar] [CrossRef]
- Song, W.; Guo, X.; Chen, K.; Chen, X.; Cao, B.; Wei, Q.; Huang, R.; Zhao, B.; Wu, Y.; Shang, H.F. The impact of non-motor symptoms on the Health-Related Quality of Life of Parkinson’s disease patients from Southwest China. Parkinsonism Relat. Disord. 2014, 20, 149–152. [Google Scholar] [CrossRef] [PubMed]
- Tinaz, S.; Pillai, A.S.; Hallett, M. Sequence Effect in Parkinson’s Disease Is Related to Motor Energetic Cost. Front. Neurol. 2016, 7, 83. [Google Scholar] [CrossRef] [PubMed]
- Martino, D.; Tamburini, T.; Zis, P.; Rosoklija, G.; Abbruzzese, G.; Ray-Chaudhuri, K.; Pelosin, E.; Avanzino, L. An objective measure combining physical and cognitive fatigability: Correlation with subjective fatigue in Parkinson’s disease. Parkinsonism Relat. Disord. 2016, 32, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.J.; Ding, J.; Li, J.Y.; Wang, M.; Yuan, Y.S.; Zhang, L.; Jiang, S.M.; Wang, X.X.; Zhu, L.; Zhang, K.Z. Abnormal Resting-State Neural Activity and Connectivity of Fatigue in Parkinson’s Disease. CNS Neurosci. Ther. 2017, 23, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.Y.; Bang, M.; Hong, J.Y.; Oh, J.; Kim, J.S.; Han, Y.M.; Chang, S.K.; Lee, S.A.; Yoon, U.; Shin, N.Y. Neural and dopaminergic correlates of fatigue in Parkinson’s disease. J. Neural Transm. 2020, 127, 301–309. [Google Scholar] [CrossRef] [PubMed]
- DeLong, M.; Wichmann, T. Changing views of basal ganglia circuits and circuit disorders. Clin. EEG Neurosci. 2010, 41, 61–67. [Google Scholar] [CrossRef]
- Kann, S.J.; Chang, C.; Manza, P.; Leung, H.C. Akinetic rigid symptoms are associated with decline in a cortical motor network in Parkinson’s disease. NPJ Parkinson’s Dis. 2020, 6, 19. [Google Scholar] [CrossRef]
- Huckins, J.F.; Adeyemo, B.; Miezin, F.M.; Power, J.D.; Gordon, E.M.; Laumann, T.O.; Heatherton, T.F.; Petersen, S.E.; Kelley, W.M. Reward-related regions form a preferentially coupled system at rest. Hum. Brain Mapp. 2019, 40, 361–376. [Google Scholar] [CrossRef]
- Wildgruber, D.; Riecker, A.; Hertrich, I.; Erb, M.; Grodd, W.; Ethofer, T.; Ackermann, H. Identification of emotional intonation evaluated by fMRI. NeuroImage 2005, 24, 1233–1241. [Google Scholar] [CrossRef]
- Bernal, B.; Altman, N. Neural networks of motor and cognitive inhibition are dissociated between brain hemispheres: An fMRI study. Int. J. Neurosci. 2009, 119, 1848–1880. [Google Scholar] [CrossRef]
- van Eimeren, T.; Monchi, O.; Ballanger, B.; Strafella, P.A. Dysfunction of the Default Mode Network in Parkinson Disease A Functional Magnetic Resonance Imaging Study. Arch. Neurol. 2009, 66, 877–883. [Google Scholar] [CrossRef] [PubMed]
- Franciotti, R.; Pizzi, S.D.; Russo, M.; Carrarini, C.; Carrozzino, D.; Perfetti, B.; Onofrj, M.; Bonanni, L. Somatic symptoms disorders in Parkinson’s disease are related to default mode and salience network dysfunction. Neuroimage Clin. 2019, 23, 101932. [Google Scholar] [CrossRef] [PubMed]
- Boon, L.I.; Hepp, D.H.; Douw, L.; van Geenen, N.; Broeders, T.A.; Geurts, J.J.; Berendse, H.W.; Schoonheim, M.M. Functional connectivity between resting-state networks reflects decline in executive function in Parkinson’s disease: A longitudinal fMRI study. Neuroimage Clin. 2020, 28, 102468. [Google Scholar] [CrossRef] [PubMed]
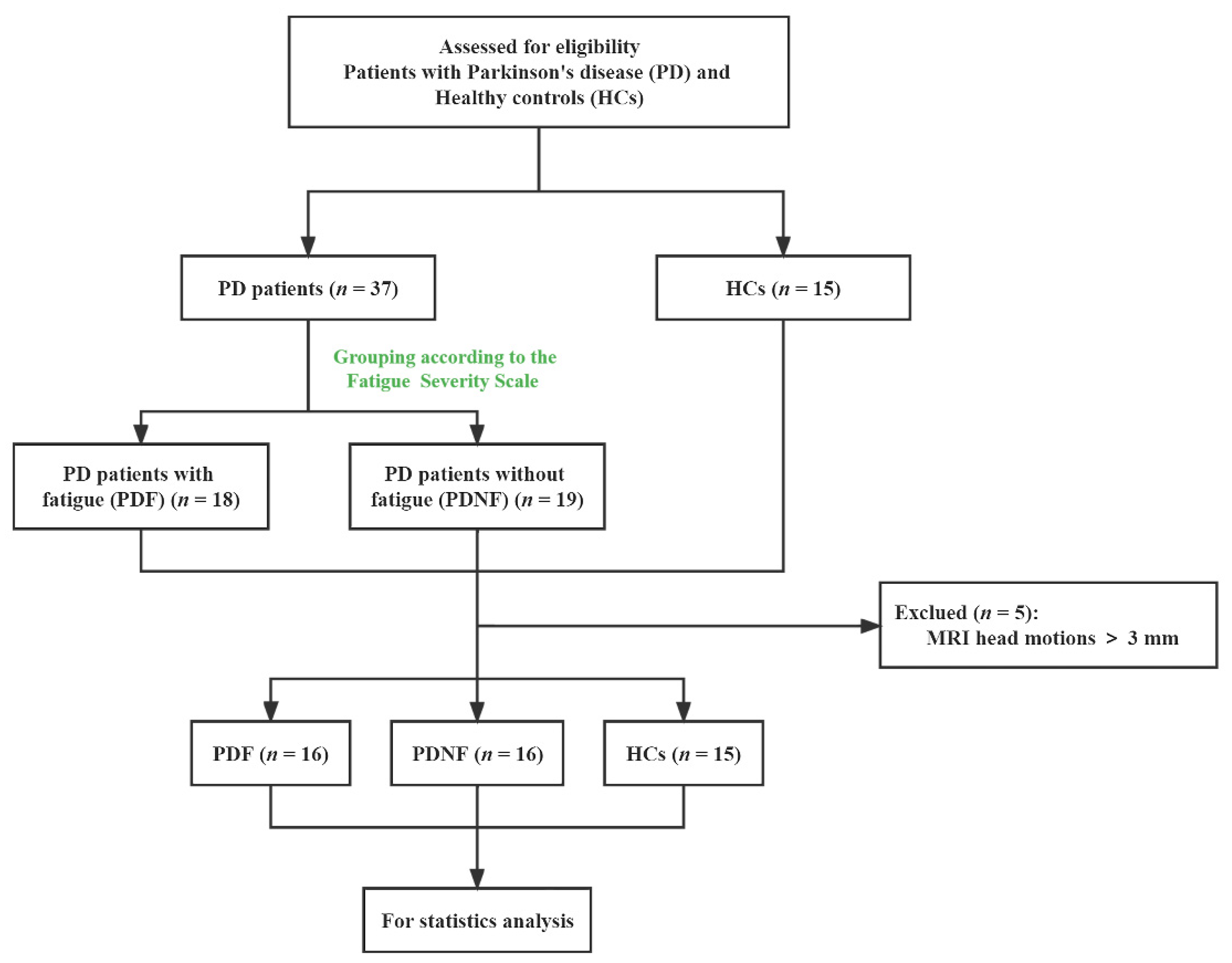
| Variables | PDF (n = 16) | PDNF (n = 16) | HCs (n = 15) | p Value |
|---|---|---|---|---|
| Age (y) a | 57.25 ± 13.98 | 63.37 ± 9.19 | 63.80 ± 5.72 | 0.147 |
| Sex (F/M) b | 8/8 | 4/12 | 5/10 | 0.326 |
| Education (y) c | 11.68 ± 3.43 | 11.06 ± 4.15 | 11.33 ± 3.45 | 0.922 |
| MMSE c | 28.25 ± 1.34 | 28.43 ± 1.20 | 28.93 ± 1.16 | 0.317 |
| Disease duration (y) d | 5.37 ± 3.52 | 6.50 ± 3.38 | NA | 0.296 |
| H&Y d | 2.34 ± 0.67 | 2.00 ± 0.60 | NA | 0.130 |
| UPDRS-III d | 29.00 ± 11.00 | 28.31 ± 11.72 | NA | 0.865 |
| LEDD (mg/day) d | 613.67 ± 248.89 | 659.68 ± 349.92 | NA | 0.671 |
| ESS d | 5.06 ± 3.67 | 4.06 ± 3.21 | NA | 0.419 |
| AS d | 10.06 ± 2.69 | 8.31 ± 3.51 | NA | 0.125 |
| HAMD e | 10.38 ± 4.98 | 7.94 ± 4.16 | NA | 0.143 |
| HAMA e | 10.56 ± 4.75 | 8.44 ± 5.42 | NA | 0.247 |
| FSS/9 a | 5.13 ± 1.06 | 2.07 ± 1.06 | 1.50 ± 0.42 | <0.001 * |
| Post hoc | PDF vs. PDNF | <0.001 * | ||
| PDF vs. HC | <0.001 * | |||
| PDNF vs. HC | 0.089 | |||
| Brain Regions (AAL) | Number of Voxels | MNI Coordinates | T Value | ||
|---|---|---|---|---|---|
| X | Y | Z | |||
| PDF > PDNF | |||||
| SMG | 15 | ±54 | −39 | 33 | −3.9081 |
| PDF > HCs | |||||
| IFG operc | 36 | ±54 | 12 | 6 | −4.7763 |
| SMG | 15 | ±51 | −42 | 36 | −4.4129 |
| PDNF > HCs | |||||
| IFG operc | 12 | ±54 | 15 | 12 | −3.6089 |
| MFG | 27 | ±42 | 33 | 36 | −3.8621 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, Y.-S.; Ji, M.; Gan, C.-T.; Sun, H.-M.; Wang, L.-N.; Zhang, K.-Z. Impaired Interhemispheric Synchrony in Parkinson’s Disease with Fatigue. J. Pers. Med. 2022, 12, 884. https://doi.org/10.3390/jpm12060884
Yuan Y-S, Ji M, Gan C-T, Sun H-M, Wang L-N, Zhang K-Z. Impaired Interhemispheric Synchrony in Parkinson’s Disease with Fatigue. Journal of Personalized Medicine. 2022; 12(6):884. https://doi.org/10.3390/jpm12060884
Chicago/Turabian StyleYuan, Yong-Sheng, Min Ji, Cai-Ting Gan, Hui-Min Sun, Li-Na Wang, and Ke-Zhong Zhang. 2022. "Impaired Interhemispheric Synchrony in Parkinson’s Disease with Fatigue" Journal of Personalized Medicine 12, no. 6: 884. https://doi.org/10.3390/jpm12060884
APA StyleYuan, Y.-S., Ji, M., Gan, C.-T., Sun, H.-M., Wang, L.-N., & Zhang, K.-Z. (2022). Impaired Interhemispheric Synchrony in Parkinson’s Disease with Fatigue. Journal of Personalized Medicine, 12(6), 884. https://doi.org/10.3390/jpm12060884





