A Clinically Relevant Classification and Staging System for Chronic Rhinosinusitis with Nasal Polyposis: A Cross-Sectional Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Study Size and Participants
2.3. Variables and Data Sources/Measurements
2.4. Endoscopy Procedure
2.5. Bias
- All eligible patients were included consecutively, with exclusion limited to those lacking essential data.
- Three independent specialists reviewed all CT scans and endoscopic videos to reduce inter-observer bias.
- Reviewers were blinded to the clinical classification of patients during image assessment.
- Clinical data were retrieved from electronic medical records to minimize recall bias.
- Confounders, such as comorbidities, were incorporated into the analysis.
2.6. Statistical Methods
2.7. Ethical Considerations
3. Results
3.1. General Findings
3.2. Comparative Analysis Among Phenotypes
3.3. Classification of CRSwNP
- Non-Eosinophilic CRSwNP (NECRSwNP)
- Central Compartment Atopic Disease (CCAD)
- Eosinophilic CRSwNP (ECRSwNP)
3.4. Subtype Profiles
- NECRSwNP: This phenotype accounted for 476 patients (23.1%), predominantly female (60.1%), with a median age of 41 years (IQR 7). Patients presented mainly with nasal obstruction and postnasal drip, often without atopy. Asthma was reported in 5.7%, while serum IgE and blood eosinophil levels were generally low. Histopathology confirmed neutrophilic infiltration without tissue eosinophilia. Radiologically, disease was limited, showing incomplete sinus opacification, with a median GOSS of 3 (IQR 1) and E:M ratio of 0.75 (IQR 0.30). The median SNOT-22 score was 41 (IQR 20.5).
- CCAD: There were 500 patients (24.3%), with near-equal sex distribution (51% male) and a median age of 24 years (IQR 5). All met the criteria for allergic rhinitis and had positive skin-prick tests (perennial allergens only). Asthma occurred in 18.8%. Histology demonstrated marked tissue eosinophilia (≥10 per HPF) despite low peripheral eosinophil counts. CT imaging typically showed central-compartment predominance (“black halo sign”), with sparing of lateral sinus walls and no pansinusitis or neo-osteogenesis. The median GOSS was 5 (IQR 4) and E:M ratio was 2.00 (IQR 0.40), consistent with localized allergic inflammation. The median SNOT-22 score was 43 (IQR 22).
- ECRSwNP: This was the most frequent subtype (1084 patients, 52.6%), mostly male (58.1%), with a median age of 32 years (IQR 6). Symptoms included nasal obstruction and anosmia ± snoring. Asthma was present in 36%, AERD in 7%, and allergic rhinitis in 32%. Nearly all patients had elevated serum IgE, positive multi-allergen skin-prick tests, and blood eosinophilia (>0.24 × 109/L). Histopathology confirmed tissue eosinophilia >10 per HPF, frequently >100 per HPF in advanced cases. Radiologically, the disease was more extensive, with diffuse sinus opacification, osteitis, and occasionally neo-osteogenesis. The median GOSS was 25 (IQR 16) and E:M ratio 1.20 (IQR 0.40), indicating high ethmoidal involvement relative to maxillary disease. The median SNOT-22 score was 69 (IQR 31).
3.5. Inter-Observer Agreement
- •
- ECRSwNP: κ = 0.938 (95% CI, 0.905–0.971; z = 16.0; p < 0.01);
- •
- NECRSwNP: κ = 0.946 (95% CI, 0.912–0.980; z = 11.6; p < 0.01);
- •
- CCAD: κ = 0.891 (95% CI, 0.845–0.937; z = 10.9; p < 0.01).
4. Discussion
4.1. General Evaluation
4.2. Radiologic and Endoscopic Signs of Eosinophilia
4.2.1. Endoscopic Features
- •
- •
- •
4.2.2. Radiological (CT) Features
- •
- •
- Central compartment opacity: Frequently referred to as the black halo sign, this finding, characterized by central mucosal thickening with peripheral sinus sparing, is common in both CCAD and ENP [33].
4.3. Clinical Features and Patient Demographics
4.4. Endoscopic and Radiologic Staging Insights
4.5. Study Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AERD | Aspirin-Exacerbated Respiratory Disease |
| ALP | Alkaline Phosphatase |
| CCAD | Central Compartment Atopic Disease |
| CRS | Chronic Rhinosinusitis |
| CRSsNP | Chronic Rhinosinusitis without Nasal Polyps |
| CRSwNP | Chronic Rhinosinusitis with Nasal Polyps |
| CT | Computed Tomography |
| ECRSwNP | Eosinophilic Chronic Rhinosinusitis with Nasal Polyps |
| ENP | Eosinophilic Nasal Polyposis |
| EPOS | European Position Paper on Rhinosinusitis and Nasal Polyps |
| GOSS | Global Osteitis Scoring Scale |
| HPF | High-Power Field |
| IgE | Immunoglobulin E |
| IQR | Interquartile Range |
| JESREC | Japanese Epidemiological Survey of Refractory Eosinophilic Chronic Rhinosinusitis |
| NECRSwNP | Non-Eosinophilic Chronic Rhinosinusitis with Nasal Polyps |
| OMC | Osteomeatal Complex |
| SNOT-22 | Sino-Nasal Outcome Test-22 |
| STROBE | Strengthening the Reporting of Observational Studies in Epidemiology |
| TGF-β1 | Transforming Growth Factor Beta 1 |
| Th2 | T-helper type 2 |
References
- Agache, I.; Song, Y.; Alonso-Coello, P.; Vogel, Y.; Rocha, C.; Solà, I.; Santero, M.; Akdis, C.A.; Akdis, M.; Canonica, G.W.; et al. Efficacy and safety of treatment with biologicals for severe chronic rhinosinusitis with nasal polyps: A systematic review for the EAACI guidelines. Allergy 2021, 76, 2337–2353. [Google Scholar] [CrossRef] [PubMed]
- Fokkens, W.; Lund, V.; Mullol, J.; Hopkins, C.; Hellings, P.W.; Kern, R.; Reitsma, S.; Toppila-Salmi, S.; Bernal-Sprekelsen, M.; Alobid, I.; et al. European Position Paper on Rhinosinusitis and Nasal Polyps 2020. Rhinol. Online 2020, 58, 1–464. [Google Scholar] [CrossRef] [PubMed]
- La Mantia, I.; Ciprandi, G.; Varricchio, A.; Ragusa, M.; Cipolla, F.; Andaloro, C. When rhinosinusitis is not just rhinosinusitis: Clinical characteristics and phenotypes of patients with type 2 chronic rhinosinusitis with nasal polyps. PubMed 2022, 93, e2022240. [Google Scholar] [CrossRef]
- Hellings, P.W.; Alobid, I.; Anselmo-Lima, W.T.; Bernal-Sprekelsen, M.; Bjermer, L.; Caulley, L.; Chaker, A.; Constantinidis, J.; Conti, D.M.; De Corso, E.; et al. EUFOREA/EPOS2020 statement on the clinical considerations for chronic rhinosinusitis with nasal polyps care. Allergy 2023, 79, 1123–1133. [Google Scholar] [CrossRef]
- Omer, G.L. Predictive significance of the blood eosinophilia for chronic sinusitis with nasal polyp recurrence: A systematic review and meta-analysis. Cell. Mol. Biol. 2024, 70, 168–173. [Google Scholar] [CrossRef]
- Parrino, D.; Brescia, G.; Zanotti, C.; Tealdo, G.; Giacomelli, L.; Barion, U.; Sfriso, P.; Marioni, G. Non-eosinophilic chronic rhinosinusitis with nasal polyps: Eosinophil, basophil, and neutrophil blood counts before and after surgery. Ann. Otol. Rhinol. Laryngol. 2018, 128, 233–240. [Google Scholar] [CrossRef]
- Kim, D.K.; Jin, H.R.; Eun, K.M.; Mutusamy, S.; Cho, S.H.; Oh, S.; Kim, D.W. Non-Eosinophilic nasal polyps shows increased epithelial proliferation and localized disease pattern in the early stage. PLoS ONE 2015, 10, e0139945. [Google Scholar] [CrossRef]
- Davies, C.; Wu, F.; Huang, E.Y.; Takashima, M.; Rowan, N.R.; Ahmed, O.G. Central compartment atopic disease as a pathophysiologically distinct subtype of chronic rhinosinusitis: A scoping review. Sinusitis 2023, 7, 12–26. [Google Scholar] [CrossRef]
- Fujieda, S.; Imoto, Y.; Kato, Y.; Ninomiya, T.; Tokunaga, T.; Tsutsumiuchi, T.; Yoshida, K.; Kidoguchi, M.; Takabayashi, T. Eosinophilic chronic rhinosinusitis. Allergol. Int. 2019, 68, 403–412. [Google Scholar] [CrossRef]
- Tokunaga, T.; Sakashita, M.; Haruna, T.; Asaka, D.; Takeno, S.; Ikeda, H.; Nakayama, T.; Seki, N.; Ito, S.; Murata, J.; et al. Novel scoring system and algorithm for classifying chronic rhinosinusitis: The JESREC Study. Allergy 2015, 70, 995–1003. [Google Scholar] [CrossRef]
- Rubel, K.E.; Lubner, R.J.; Lopez, A.A.; Li, P.; Huang, L.C.; Sheng, Q.; Wu, J.; Wise, S.K.; DelGaudio, J.M.; Chandra, R.K.; et al. Inflammatory characteristics of central compartment atopic disease. Int. Forum. Allergy Rhinol. 2023, 13, 2133–2143. [Google Scholar] [CrossRef]
- Sit, A.; Alvarado, R.; Earls, P.; Rimmer, J.; Kalish, L.; Campbell, R.; Sewell, W.; Harvey, R.J. CCAD or eCRS: Defining eosinophilic subpopulations in chronic rhinosinusitis. Am. J. Rhinol. Allergy 2023, 37, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Marambaia, P.P.; Lima, M.G.; Santos, K.P.; De Machado Gomes, A.; De Sousa, M.M.; De Macedo Marques, M.E. Evaluation of the quality of life of patients with chronic rhinosinusitis by means of the SNOT-22 questionnaire. Braz. J. Otorhinolaryngol. 2013, 79, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Poznanovic, S.A.; Kingdom, T.T. Total IGE levels and peripheral eosinophilia. Arch. Otolaryngol. 2007, 133, 701. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.; Earls, P.; Harvey, R.J. Systemic biomarkers of eosinophilic chronic rhinosinusitis. Curr. Opin. Allergy Clin. Immunol. 2019, 20, 23–29. [Google Scholar] [CrossRef]
- Grayson, J.W.; Cavada, M.; Harvey, R.J. Clinically relevant phenotypes in chronic rhinosinusitis. J. Otolaryngol. Head Neck Surg. 2019, 48. [Google Scholar] [CrossRef]
- Li, F.; Wang, S.; Cha, X.; Li, T.; Xie, Y.; Wang, W.; Ren, W.; Liao, J.; Liu, H. Blood eosinophil percentage and improved sinus CT score as diagnostic tools for ECRS. OTO Open 2024, 8, e106. [Google Scholar] [CrossRef]
- Georgalas, C.; Videler, W.; Freling, N.; Fokkens, W. Global Osteitis Scoring Scale and chronic rhinosinusitis: A marker of revision surgery. Clin. Otolaryngol. 2010, 35, 455–461. [Google Scholar] [CrossRef]
- Bernstein, J.A.; Bernstein, J.S.; Makol, R.; Ward, S. Allergic rhinitis. JAMA 2024, 331, 866. [Google Scholar] [CrossRef]
- Lee, R.U.; Stevenson, D.D. Aspirin-Exacerbated Respiratory Disease: Evaluation and management. Allergy Asthma Immunol. Res. 2010, 3, 3. [Google Scholar] [CrossRef]
- Masaki, M.; Tanaka, Y. Nasal polyps in the olfactory cleft. Laryngoscope 1998, 108, 1243–1246. [Google Scholar] [CrossRef] [PubMed]
- Min, Y.G.; Kim, Y.J.; Yun, Y.S. Distribution of Eosinophil Granule Proteins in Nasal Mucosa of Atopic Patients with Nasal Polyposis. ORL J. Otorhinolaryngol. Relat. Spec. 1996, 58, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Takasaka, T.; Kurihara, A.; Suzuki, H.; Kikuchi, T.; Hozawa, K.; Wataya, H. The differentiation of polyps and their mucosal ultrastructure. Am. J. Rhinol. Allergy 1990, 4, 159–162. [Google Scholar] [CrossRef]
- Bachert, C.; Gevaert, P.; Holtappels, G.; Cuvelier, C.; Van Cauwenberge, P. Nasal polyposis: From cytokines to growth. Am. J. Rhinol. Allergy 2000, 14, 279–290. [Google Scholar] [CrossRef]
- Saitoh, T.; Kusunoli, T.; Yao, T.; Kawano, K.; Kojima, Y.; Miyahara, K.; Onoda, J.; Yokoi, H.; Ikeda, K. Relationship between epithelial damage or basement membrane thickness and eosinophilic infiltration in nasal polyps with chronic rhinosinusitis. Rhinol. Online 2009, 47, 275–279. [Google Scholar] [CrossRef]
- Mortuaire, G.; Leroy, X.; Gengler, I.; Chevalier, D.; Prin, L.; Picry, A. Histopathological classification of refractory chronic rhinosinusitis with nasal polyps. PubMed 2015, 30, 1447–1454. [Google Scholar] [CrossRef]
- Bachert, C.; Hicks, A.; Gane, S.; Peters, A.T.; Gevaert, P.; Nash, S.; Horowitz, J.E.; Sacks, H.; Jacob-Nara, J.A. The interleukin-4/interleukin-13 pathway in type 2 inflammation in chronic rhinosinusitis with nasal polyps. Front. Immunol. 2024, 15, 1356298. [Google Scholar] [CrossRef]
- Pawankar, R. Nasal polyposis: An update. Curr. Opin. Allergy Clin. Immunol. 2003, 3, 1–6. [Google Scholar] [CrossRef]
- Zhang, Z.; Zuo, Q.; Du, Y.; Jiang, H.; Ma, F.; Zhang, Y. Correlation of Eosinophils and Type 2 Inflammatory Mediators with Osteitis in Chronic Rhinosinusitis with Nasal Polyps. J. Inflamm. Res. 2024, 17, 4055–4064. [Google Scholar] [CrossRef]
- Zhu, C.M.; Liu, H.B.; Wu, W.X.; Xu, C. To explore the role of CT scan in the diagnosis of eosinophilic chronic rhinosinusitis. PubMed 2018, 32, 944–948. [Google Scholar] [CrossRef]
- Zhang, Q.; Ji, J.F.; Zhang, T.; Xue, F.; Wu, M.H.; Wang, Z.Y.; Xu, L.; Cheng, Y. Different phenotypes in patients with chronic sinusitis with bilateral nasal polyps. PubMed 2018, 32, 1774–1778. [Google Scholar] [CrossRef]
- Jo, S.; Lee, S.H.; Jo, H.; Weon, S.; Jeon, C.; Park, M.K.; Kim, T.-H.; Cho, S.H. Eosinophil-derived TGFβ1 controls the new bone formation in chronic rhinosinusitis with nasal polyps. Rhinol. Online 2023, 61, 340–350. [Google Scholar] [CrossRef]
- Abdullah, B.; Vengathajalam, S.; Daud, M.K.M.; Mohammad, Z.W.; Hamizan, A.; Husain, S. The Clinical and Radiological Characterizations of the Allergic Phenotype of Chronic Rhinosinusitis with Nasal Polyps. J. Asthma Allergy 2020, 13, 523–531. [Google Scholar] [CrossRef]
- Kim, D.H.; Kim, S.W.; Basurrah, M.A.; Hwang, S.H. Clinical and Laboratory Features of Various Criteria of Eosinophilic Chronic Rhinosinusitis: A Systematic Review and Meta-Analysis. Clin. Exp. Otorhinolaryngol. 2022, 15, 230–246. [Google Scholar] [CrossRef]
- Sun, C.; Ouyang, H.; Luo, R. Distinct characteristics of nasal polyps with and without eosinophilia. Braz. J. Otorhinolaryngol. 2016, 83, 66–72. [Google Scholar] [CrossRef]
- McHugh, T.; Levin, M.; Snidvongs, K.; Banglawala, S.M.; Sommer, D.D. Comorbidities associated with eosinophilic chronic rhinosinusitis: A systematic review and meta-analysis. Clin. Otolaryngol. 2020, 45, 574–583. [Google Scholar] [CrossRef]
- Tsuda, T.; Takeda, K.; Terada, R.; Tanaka, S.; Waki, S.; Akama, T.; Nishimura, H. Osteitis in eosinophilic chronic rhinosinusitis. Ear Nose Throat J. 2022, 103, NP741–NP748. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kim, D.H.; Kim, S.W.; Im, Y.H.; Park, C.S.; Kim, D.H.; Alkhars, Z.; Kim, S.W. Diagnostic criteria for eosinophilic chronic rhinosinusitis: Comparative analysis and novel scoring system. Int. Forum. Allergy Rhinol. 2024, 14, 1746–1756. [Google Scholar] [CrossRef] [PubMed]




| Variables | NECRSwNP | CCAD | ECRSwNP | Total | |
|---|---|---|---|---|---|
| Age (Year [IQR]) | 41 (IQR 7) | 24 (IQR 5) | 32 (IQR 6) | 29.8 ± 11 | |
| Sex | Male | 190 (39.9%) | 255 (51%) | 630 (58.1%) | 1075 (52.2%) |
| Female | 286 (60.1%) | 245 (49%) | 454 (41.9%) | 985 (47.8%) | |
| Presenting Complaint | Nasal Obstruction, Anosmia ± Snoring | 0 | 0 | 1084 (100%) | 1084 (52.6%) |
| Nasal Obstruction, Postnasal Drip ± Cough | 476 (100%) | 0 | 0 | 476 (23.1%) | |
| Nasal Obstruction, Rhinorrhea, and Sneezing | 0 | 500 (100%) | 0 | 500 (24.3%) | |
| Associated Disease | Asthma | 27 (5.7%) | 94 (18.8%) | 390 (36%) | 511 (24.8%) |
| Aspirin-Exacerbated Respiratory Disease | 0 | 0 | 76 (7%) | 76 (3.7%) | |
| Allergic Rhinitis | 0 | 500 (100%) | 346 (32%) | 846 (41.1%) | |
| Total Serum IgE Count | More than 100 IU/mL | 27 (5.7%) | 0 | 1035 (95.5%) | 1062 (51.6%) |
| 100 IU/mL or less | 449 (94.3%) | 500 (100%) | 49 (4.5%) | 998 (48.4%) | |
| Blood Eosinophils | Less than or equal to 0.24 × 109 cells/L | 476 (100%) | 500 (100%) | 0 | 976 (47.4%) |
| More than 0.24 × 109 cells/L | 0 | 0 | 418 (38.6%) | 418 (20.3%) | |
| More than 0.45 × 109 cells/L | 0 | 0 | 666 (61.4%) | 666 (32.3%) | |
| Skin Prick Test | Positive test (perennial allergen only) | 0 | 500 (100%) | 0 | 500 (24.3%) |
| Positive test (multiple allergens/nonspecific) | 0 | 0 | 1084 (100%) | 1084 (52.6%) | |
| Tissue Histopathology | Neutrophil Infiltration | 476 (100%) | 0 | 0 | 476 (23.1%) |
| Eosinophil Count 10–100 per HPF | 0 | 437 (76.4%) | 418 (38.6%) | 855 (41.5%) | |
| Eosinophil Count >100 per HPF | 0 | 63 (24.6%) | 666 (61.4%) | 729 (35.4%) | |
| SNOT-22 § (Median [IQR]) | 41 (IQR 20.5) | 43 (IQR 22) | 69 (IQR 31) | 42.5 (IQR 35) | |
| GOSS * (Median [IQR]) | 3 (1) | 5 (4) | 25 (16) | 12 (9) | |
| E:M Ratio † (Median [IQR]) | 0.75 (0.30) | 2.00 (0.40) | 1.20 (0.40) | 1.20 (0.50) | |
| Total | 476 (23.1%) | 500 (24.3%) | 1084 (52.6%) | 2060 (100%) | |
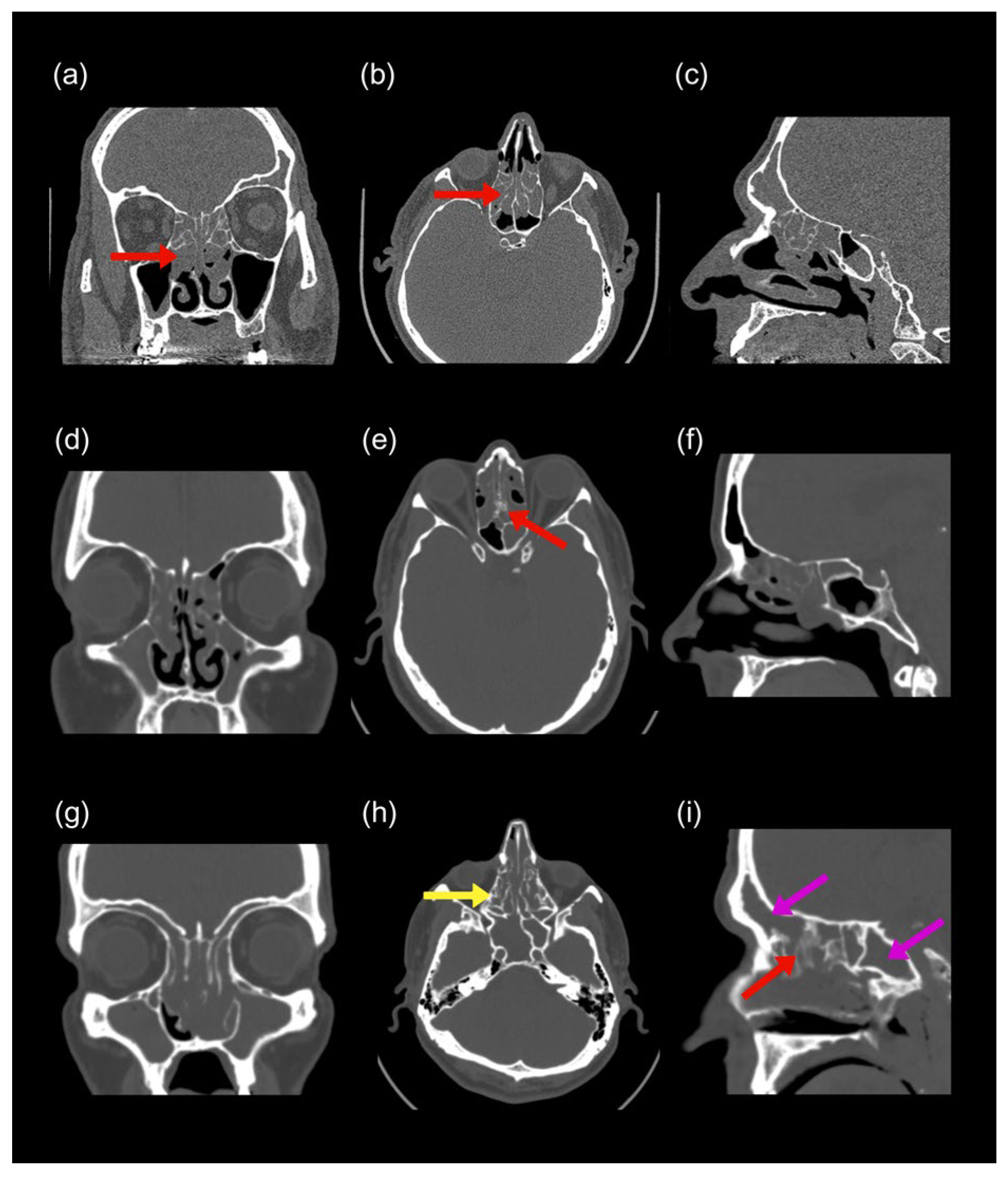
| Group 1: NECRSwNP (Non-Eosinophilic Chronic Rhinosinusitis with Nasal Polyposis) | |||
|---|---|---|---|
| Stages | Endoscopy Findings | CT Findings | Notes |
| Stage 1 |
| Incomplete opacification of the sinuses, especially the maxillary sinus (low E:M score †) |
|
| Stage 2 |
|
| |
| Group 2: CCAD (Central Compartment Atopic Disease) | |||
| Stages | Endoscopy Findings | CT Findings | Notes |
| Stage 1 |
|
|
|
| Stage 2 |
|
| |
| Group 3: ECRSwNP (Eosinophilic Chronic Rhinosinusitis with Nasal Polyposis) | |||
| Stages | Endoscopy Findings | CT Findings | |
| Stage 1 | Small multiple polyps matted together, from: Middle meatus, lateral surface of middle turbinate, and/or may extend to the nasal cavity |
| |
| Stage 2 |
|
| |
| Stage 3 |
|
| |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Omer, G.L.; Di Girolamo, S.; Ali, S.S.; Maurizi, R.; Viola, S.; De Donato, G. A Clinically Relevant Classification and Staging System for Chronic Rhinosinusitis with Nasal Polyposis: A Cross-Sectional Study. Diagnostics 2025, 15, 3197. https://doi.org/10.3390/diagnostics15243197
Omer GL, Di Girolamo S, Ali SS, Maurizi R, Viola S, De Donato G. A Clinically Relevant Classification and Staging System for Chronic Rhinosinusitis with Nasal Polyposis: A Cross-Sectional Study. Diagnostics. 2025; 15(24):3197. https://doi.org/10.3390/diagnostics15243197
Chicago/Turabian StyleOmer, Goran Latif, Stefano Di Girolamo, Sahand Soran Ali, Riccardo Maurizi, Sveva Viola, and Giuseppe De Donato. 2025. "A Clinically Relevant Classification and Staging System for Chronic Rhinosinusitis with Nasal Polyposis: A Cross-Sectional Study" Diagnostics 15, no. 24: 3197. https://doi.org/10.3390/diagnostics15243197
APA StyleOmer, G. L., Di Girolamo, S., Ali, S. S., Maurizi, R., Viola, S., & De Donato, G. (2025). A Clinically Relevant Classification and Staging System for Chronic Rhinosinusitis with Nasal Polyposis: A Cross-Sectional Study. Diagnostics, 15(24), 3197. https://doi.org/10.3390/diagnostics15243197






