Microtubule Minus-End Binding Proteins in Cancer: Advances
Abstract
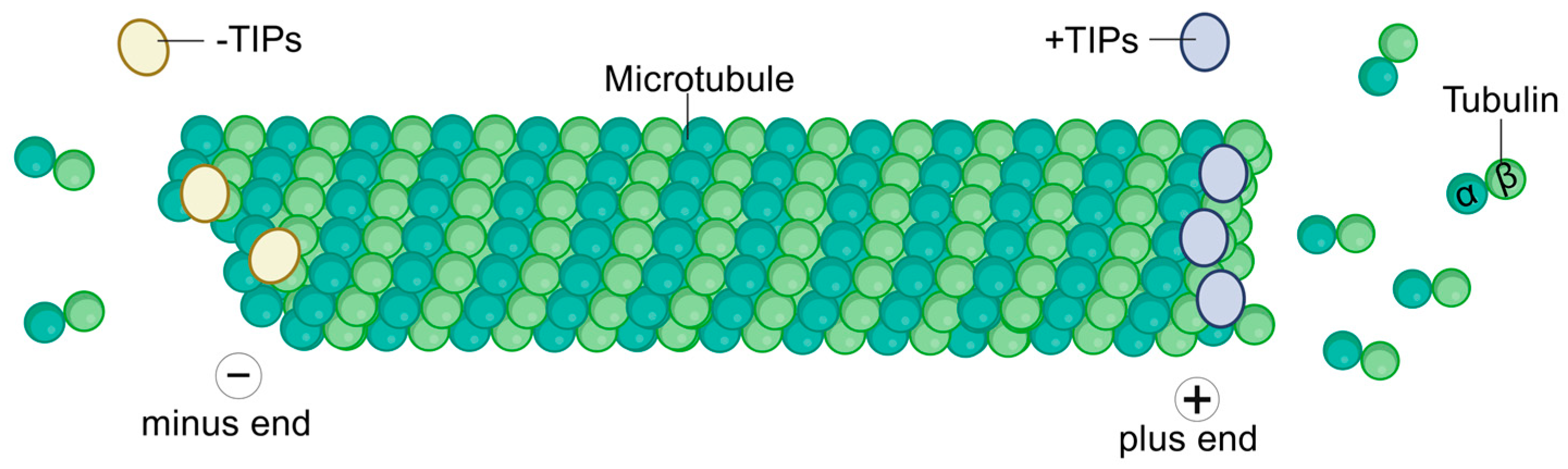
1. Introduction
2. Classification and Role of −TIPs
3. Characterization of the Domains of −TIPs
4. Relationship Between −TIPs and Tumor Prognosis
5. Mechanisms of −TIPs in Cancer Promotion
6. Translational Studies of −TIPs
7. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CAMSAPs | calmodulin-regulated spectrin-associated proteins |
| γ-TuRC | γ-tubulin ring complex |
| ASPM | abnormal spindle microtubule assembly |
| KANSL3 | KAT8 regulatory complex subunit 3 gene |
| SCLC | small-cell lung cancer |
| LIHC | liver hepatocellular carcinoma |
| CRC | colorectal cancer |
| NPC | nasopharyngeal carcinoma |
| HCC | hepatocellular carcinoma |
| NSCLC | non-small-cell lung cancer |
| LC | lung cancer |
| LUAD | lung adenocarcinoma |
| LSCC | Lung squamous cell carcinoma |
| GC | gastric cancer |
| PRCC | papillary renal cell carcinoma |
| ASPM-i1 | ASPM isoform 1 |
| ATC | anaplastic thyroid carcinoma |
| EMT | epithelial–mesenchymal transition |
| GRAMD1A | GRAM Domain Containing 1A |
| JNK | c-Jun N-terminal kinase |
| MMP-1 | Matrix Metalloproteinase-1 |
| CircSOD2 | circRNA SOD2 |
| TGF-β | transforming growth factor-β |
| RASAL2 | Ras protein activator-like 2 |
| ERK | extracellular signal-regulated kinase |
| Rac1 | Ras-related C3 botulinum toxin substrate 1 |
| HDAC6 | histone deacetylases 6 |
| HMGB1 | high mobility group box 1 |
| TSA | trichostatin A |
| NCL | nucleolin |
| HIF-1α | hypoxia-inducible factor-1α |
| CDK4 | cyclin dependent kinases 4 |
| GLI1 | glioma-associated oncogene homolog 1 |
| FOXM1 | Forkhead box protein M1 |
| Dvl-3 | disheveled-3 |
| Smo | Smoothened |
| KIF11 | kinesin family member 11 |
| METTL3 | methyltransferase-like 3 |
| m6A | N6-methyladenosine |
References
- Aher, A.; Urnavicius, L.; Xue, A.; Neselu, K.; Kapoor, T.M. Structure of the γ-tubulin ring complex-capped microtubule. Nat. Struct. Mol. Biol. 2024, 31, 1124–1133. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Gu, L.; Chen, W.; Tanaka, N.; Zhou, Z.; Xu, H.; Xu, T.; Ji, W.; Liang, X.; Meng, W. CAMSAP2 and CAMSAP3 localize at microtubule intersections to regulate the spatial distribution of microtubules. J. Mol. Cell Biol. 2024, 15, mjad050. [Google Scholar] [CrossRef] [PubMed]
- Wenger, A.; Karlsson, I.; Kling, T.; Carén, H. CRISPR-Cas9 knockout screen identifies novel treatment targets in childhood high-grade glioma. Clin. Epigenet. 2023, 15, 80. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhang, J.; Wang, Y.; Xu, Y.; Zhang, S. Non-coding ribonucleic acid-mediated CAMSAP1 upregulation leads to poor prognosis with suppressed immune infiltration in liver hepatocellular carcinoma. Front. Genet. 2022, 13, 916847. [Google Scholar] [CrossRef]
- Hendershott, M.C.; Vale, R.D. Regulation of microtubule minus-end dynamics by CAMSAPs and Patronin. Proc. Natl. Acad. Sci. USA 2014, 111, 5860–5865. [Google Scholar] [CrossRef]
- Goodwin, S.S.; Vale, R.D. Patronin Regulates the Microtubule Network by Protecting Microtubule Minus Ends. Cell 2010, 143, 263–274. [Google Scholar] [CrossRef]
- Li, D.; Ding, X.; Xie, M.; Huang, Z.; Han, P.; Tian, D.; Xia, L. CAMSAP2-mediated noncentrosomal microtubule acetylation drives hepatocellular carcinoma metastasis. Theranostics 2020, 10, 3749–3766. [Google Scholar] [CrossRef]
- Razuvaeva, A.V.; Graziadio, L.; Palumbo, V.; Pavlova, G.A.; Popova, J.V.; Pindyurin, A.V.; Bonaccorsi, S.; Somma, M.P.; Gatti, M. The Multiple Mitotic Roles of the ASPM Orthologous Proteins: Insight into the Etiology of ASPM-Dependent Microcephaly. Cells 2023, 12, 922. [Google Scholar] [CrossRef]
- Meunier, S.; Shvedunova, M.; Van Nguyen, N.; Avila, L.; Vernos, I.; Akhtar, A. An epigenetic regulator emerges as microtubule minus-end binding and stabilizing factor in mitosis. Nat. Commun. 2015, 6, 7889. [Google Scholar] [CrossRef]
- Rai, D.; Song, Y.; Hua, S.; Stecker, K.; Monster, J.L.; Yin, V.; Stucchi, R.; Xu, Y.; Zhang, Y.; Chen, F.; et al. CAMSAPs and nucleation-promoting factors control microtubule release from γ-TuRC. Nat. Cell Biol. 2024, 26, 404–420. [Google Scholar] [CrossRef]
- Liu, P.; Zupa, E.; Neuner, A.; Böhler, A.; Loerke, J.; Flemming, D.; Ruppert, T.; Rudack, T.; Peter, C.; Spahn, C.; et al. Insights into the assembly and activation of the microtubule nucleator γ-TuRC. Nature 2020, 578, 467–471. [Google Scholar] [CrossRef]
- Létard, P.; Drunat, S.; Vial, Y.; Duerinckx, S.; Ernault, A.; Amram, D.; Arpin, S.; Bertoli, M.; Busa, T.; Ceulemans, B.; et al. Autosomal recessive primary microcephaly due to ASPM mutations: An update. Hum. Mutat. 2018, 39, 319–332. [Google Scholar] [CrossRef]
- Wang, X.; Liu, Y.; Ding, Y.; Feng, G. CAMSAP2 promotes colorectal cancer cell migration and invasion through activation of JNK/c-Jun/MMP-1 signaling pathway. Sci. Rep. 2022, 12, 16899. [Google Scholar] [CrossRef]
- Singharajkomron, N.; Yodsurang, V.; Limprasutr, V.; Wattanathamsan, O.; Iksen, I.; Hayakawa, Y.; Pongrakhananon, V. CAMSAP2 enhances lung cancer cell metastasis by mediating RASAL2 degradation. Life Sci. 2024, 338, 122391. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Huang, E.; Wang, W.; Xu, L.; Xu, W.; Zheng, T.; Rui, M. Patronin regulates presynaptic microtubule organization and neuromuscular junction development in Drosophila. iScience 2024, 27, 108944. [Google Scholar] [CrossRef] [PubMed]
- Panzade, S.; Matis, M. The Microtubule Minus-End Binding Protein Patronin Is Required for the Epithelial Remodeling in the Drosophila Abdomen. Front. Cell Dev. Biol. 2021, 9, 682083. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Van Beem, L.; Snel, B.; Hoogenraad, C.C.; Harterink, M. PTRN-1 (CAMSAP) and NOCA-2 (NINEIN) are required for microtubule polarity in Caenorhabditis elegans dendrites. PLoS Biol. 2022, 20, e3001855. [Google Scholar] [CrossRef]
- Gong, T.; Yan, Y.; Zhang, J.; Liu, S.; Liu, H.; Gao, J.; Zhou, X.; Chen, J.; Shi, A. PTRN-1/CAMSAP promotes CYK-1/formin-dependent actin polymerization during endocytic recycling. EMBO J. 2018, 37, e98556. [Google Scholar] [CrossRef]
- Zhou, Z.; Xu, H.; Li, Y.; Yang, M.; Zhang, R.; Shiraishi, A.; Kiyonari, H.; Liang, X.; Huang, X.; Wang, Y.; et al. CAMSAP1 breaks the homeostatic microtubule network to instruct neuronal polarity. Proc. Natl. Acad. Sci. USA 2020, 117, 22193–22203. [Google Scholar] [CrossRef]
- Hu, W.; Zhang, R.; Xu, H.; Li, Y.; Yang, X.; Zhou, Z.; Huang, X.; Wang, Y.; Ji, W.; Gao, F.; et al. CAMSAP1 role in orchestrating structure and dynamics of manchette microtubule minus-ends impacts male fertility during spermiogenesis. Proc. Natl. Acad. Sci. USA 2023, 120, e2313787120. [Google Scholar] [CrossRef]
- Yi, Y.; Qiu, Z.; Yao, Z.; Lin, A.; Qin, Y.; Sha, R.; Wei, T.; Wang, Y.; Cheng, Q.; Zhang, J.; et al. CAMSAP1 Mutation Correlates With Improved Prognosis in Small Cell Lung Cancer Patients Treated With Platinum-Based Chemotherapy. Front. Cell Dev. Biol. 2022, 9, 770811. [Google Scholar] [CrossRef]
- Wan, Z.; Yang, X.; Liu, X.; Sun, Y.; Yu, P.; Xu, F.; Deng, H. M2 macrophage-derived exosomal microRNA-411-5p impedes the activation of hepatic stellate cells by targeting CAMSAP1 in NASH model. iScience 2022, 25, 104597. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.; Wang, D.; Yan, G.; Yang, Z.; Tang, C. MiR-411-5p Promotes Vascular Smooth Muscle Cell Phenotype Switch by Inhibiting Expression of Calmodulin Regulated Spectrin-Associated Protein-1. Int. Heart J. 2024, 65, 557–565. [Google Scholar] [CrossRef]
- Ho, K.-H.; Jayathilake, A.; Yagan, M.; Nour, A.; Osipovich, A.B.; Magnuson, M.A.; Gu, G.; Kaverina, I. CAMSAP2 localizes to the Golgi in islet β-cells and facilitates Golgi-ER trafficking. iScience 2023, 26, 105938. [Google Scholar] [CrossRef]
- Zhou, Z.; Yang, X.; Mao, A.; Xu, H.; Lin, C.; Yang, M.; Hu, W.; Shao, J.; Xu, P.; Li, Y.; et al. Deficiency of CAMSAP2 impairs olfaction and the morphogenesis of mitral cells. EMBO Rep. 2024, 25, 2861–2877. [Google Scholar] [CrossRef]
- Zhang, S.; Kwan, P.; Baum, L. The potential role of CAMSAP1L1 in symptomatic epilepsy. Neurosci. Lett. 2013, 556, 146–151. [Google Scholar] [CrossRef]
- Wattanathamsan, O.; Chetprayoon, P.; Chantaravisoot, N.; Wongkongkathep, P.; Chanvorachote, P.; Pongrakhananon, V. CAMSAP3 depletion induces lung cancer cell senescence-associated phenotypes through extracellular signal-regulated kinase inactivation. Cancer Med. 2021, 10, 8961–8975. [Google Scholar] [CrossRef] [PubMed]
- Mitsuhata, Y.; Abe, T.; Misaki, K.; Nakajima, Y.; Kiriya, K.; Kawasaki, M.; Kiyonari, H.; Takeichi, M.; Toya, M.; Sato, M. Cyst formation in proximal renal tubules caused by dysfunction of the microtubule minus-end regulator CAMSAP3. Sci. Rep. 2021, 11, 5857. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Saito, H.; Kawasaki, M.; Takeichi, M. CAMSAP3 is required for mTORC1-dependent ependymal cell growth and lateral ventricle shaping in mouse brains. Development 2021, 148, dev195073. [Google Scholar] [CrossRef]
- Saito, H.; Matsukawa-Usami, F.; Fujimori, T.; Kimura, T.; Ide, T.; Yamamoto, T.; Shibata, T.; Onoue, K.; Okayama, S.; Yonemura, S.; et al. Tracheal motile cilia in mice require CAMSAP3 for the formation of central microtubule pair and coordinated beating. Mol. Biol. Cell 2021, 32, ar12. [Google Scholar] [CrossRef]
- Toya, M.; Kobayashi, S.; Kawasaki, M.; Shioi, G.; Kaneko, M.; Ishiuchi, T.; Misaki, K.; Meng, W.; Takeichi, M. CAMSAP3 orients the apical-to-basal polarity of microtubule arrays in epithelial cells. Proc. Natl. Acad. Sci. USA 2016, 113, 332–337. [Google Scholar] [CrossRef]
- Mogensen, M.M.; Malik, A.; Piel, M.; Bouckson-Castaing, V.; Bornens, M. Microtubule minus-end anchorage at centrosomal and non-centrosomal sites: The role of ninein. J. Cell Sci. 2000, 113, 3013–3023. [Google Scholar] [CrossRef]
- Karoutas, A.; Szymanski, W.; Rausch, T.; Guhathakurta, S.; Rog-Zielinska, E.A.; Peyronnet, R.; Seyfferth, J.; Chen, H.-R.; De Leeuw, R.; Herquel, B.; et al. The NSL complex maintains nuclear architecture stability via lamin A/C acetylation. Nat. Cell Biol. 2019, 21, 1248–1260. [Google Scholar] [CrossRef]
- Sheikh, B.N.; Guhathakurta, S.; Tsang, T.H.; Schwabenland, M.; Renschler, G.; Herquel, B.; Bhardwaj, V.; Holz, H.; Stehle, T.; Bondareva, O.; et al. Neural metabolic imbalance induced by MOF dysfunction triggers pericyte activation and breakdown of vasculature. Nat. Cell Biol. 2020, 22, 828–841. [Google Scholar] [CrossRef] [PubMed]
- Chander, A.; Mager, J. Loss of KANSL3 leads to defective inner cell mass and early embryonic lethality. Mol. Reprod. Devel. 2024, 91, e23760. [Google Scholar] [CrossRef] [PubMed]
- Bolhuis, D.L.; Dixit, R.; Slep, K.C. Crystal structure of the Arabidopsis SPIRAL2 C-terminal domain reveals a p80-Katanin-like domain. PLoS ONE 2023, 18, e0290024. [Google Scholar] [CrossRef] [PubMed]
- Guan, C.; Hua, S.; Jiang, K. The CEP170B-KIF2A complex destabilizes microtubule minus ends to generate polarized microtubule network. EMBO J. 2023, 42, e112953. [Google Scholar] [CrossRef]
- Atherton, J.; Luo, Y.; Xiang, S.; Yang, C.; Vemu, A.; Stangier, M.; Cook, A.; Wang, S.; Jiang, K.; Steinmetz, M.; et al. Structural Model for Preferential Microtubule Minus End Binding by CAMSAP CKK Domains. Biophys. J. 2019, 116, 251a. [Google Scholar] [CrossRef]
- Meng, W.; Mushika, Y.; Ichii, T.; Takeichi, M. Anchorage of Microtubule Minus Ends to Adherens Junctions Regulates Epithelial Cell-Cell Contacts. Cell 2008, 135, 948–959. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, R.; Ren, J.; Chen, W.; Zhou, Z.; Xu, H.; Li, D.; Cheng, H.; Xie, Q.; Ji, W.; et al. CAMSAP3 forms dimers via its α-helix domain that directly stabilize non-centrosomal microtubule minus ends. J. Cell Sci. 2024, 137, jcs263609. [Google Scholar] [CrossRef]
- Ning, W.; Yu, Y.; Xu, H.; Liu, X.; Wang, D.; Wang, J.; Wang, Y.; Meng, W. The CAMSAP3-ACF7 Complex Couples Noncentrosomal Microtubules with Actin Filaments to Coordinate Their Dynamics. Dev. Cell 2016, 39, 61–74. [Google Scholar] [CrossRef]
- Rujano, M.A.; Sanchez-Pulido, L.; Pennetier, C.; Le Dez, G.; Basto, R. The microcephaly protein Asp regulates neuroepithelium morphogenesis by controlling the spatial distribution of myosin II. Nat. Cell Biol. 2013, 15, 1294–1306. [Google Scholar] [CrossRef]
- Jiang, K.; Rezabkova, L.; Hua, S.; Liu, Q.; Capitani, G.; Altelaar, A.F.M.; Heck, A.J.R.; Kammerer, R.A.; Steinmetz, M.O.; Akhmanova, A. Microtubule minus-end regulation at spindle poles by an ASPM–katanin complex. Nat. Cell Biol. 2017, 19, 480–492. [Google Scholar] [CrossRef]
- Ito, A.; Goshima, G. Microcephaly protein Asp focuses the minus ends of spindle microtubules at the pole and within the spindle. J. Cell Biol. 2015, 211, 999–1009. [Google Scholar] [CrossRef]
- Yao, M.; Wakamatsu, Y.; Itoh, T.J.; Shoji, T.; Hashimoto, T. Arabidopsis SPIRAL2 promotes uninterrupted microtubule growth by suppressing the pause state of microtubule dynamics. J. Cell Sci. 2008, 121, 2372–2381. [Google Scholar] [CrossRef]
- Ohno, M.; Higuchi, Y.; Yamai, K.; Fuchigami, S.; Sasaki, T.; Oda, Y.; Hayashi, I. Structural analysis of microtubule binding by minus-end targeting protein Spiral2. Biochim. Et Biophys. Acta (BBA)—Mol. Cell Res. 2024, 1871, 119858. [Google Scholar] [CrossRef]
- Jiang, K.; Hua, S.; Mohan, R.; Grigoriev, I.; Yau, K.W.; Liu, Q.; Katrukha, E.A.; Altelaar, A.F.M.; Heck, A.J.R.; Hoogenraad, C.C.; et al. Microtubule Minus-End Stabilization by Polymerization-Driven CAMSAP Deposition. Dev. Cell 2014, 28, 295–309. [Google Scholar] [CrossRef]
- Fan, Y.; Burkart, G.M.; Dixit, R. The Arabidopsis SPIRAL2 Protein Targets and Stabilizes Microtubule Minus Ends. Curr. Biol. 2018, 28, 987–994.e3. [Google Scholar] [CrossRef]
- Leong, S.Y.; Yamada, M.; Yanagisawa, N.; Goshima, G. SPIRAL2 Stabilises Endoplasmic Microtubule Minus Ends in the Moss Physcomitrella patens. Cell Struct. Funct. 2018, 43, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; Bai, X.; Wang, X.; Cao, Y.; Zhang, L.; Li, W.; Wang, S. Insight on the hub gene associated signatures and potential therapeutic agents in epilepsy and glioma. Brain Res. Bull. 2023, 199, 110666. [Google Scholar] [CrossRef] [PubMed]
- Dráber, P.; Dráberová, E. Dysregulation of Microtubule Nucleating Proteins in Cancer Cells. Cancers 2021, 13, 5638. [Google Scholar] [CrossRef]
- Zhou, C.; Liu, H.; Wang, F.; Hu, T.; Liang, Z.; Lan, N.; He, X.; Zheng, X.; Wu, X.; Xie, D.; et al. circCAMSAP1 Promotes Tumor Growth in Colorectal Cancer via the miR-328-5p/E2F1 Axis. Mol. Ther. 2020, 28, 914–928. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Lu, L.; Tang, Q.; Wei, W.; Chen, P.; Chen, Y.; Pu, J.; Wang, J. CircCAMSAP1 promotes hepatocellular carcinoma progression through miR-1294/GRAMD1A pathway. J. Cell. Mol. Med. 2021, 25, 3793–3802. [Google Scholar] [CrossRef]
- Wang, Y.; Yan, Q.; Mo, Y.; Liu, Y.; Wang, Y.; Zhang, S.; Guo, C.; Wang, F.; Li, G.; Zeng, Z.; et al. Splicing factor derived circular RNA circCAMSAP1 accelerates nasopharyngeal carcinoma tumorigenesis via a SERPINH1/c-Myc positive feedback loop. Mol. Cancer 2022, 21, 62. [Google Scholar] [CrossRef]
- Wang, J.; Mi, J.-Q.; Debernardi, A.; Vitte, A.-L.; Emadali, A.; Meyer, J.A.; Charmpi, K.; Ycart, B.; Callanan, M.B.; Carroll, W.L.; et al. A six gene expression signature defines aggressive subtypes and predicts outcome in childhood and adult acute lymphoblastic leukemia. Oncotarget 2015, 6, 16527–16542. [Google Scholar] [CrossRef]
- Lugen, Z.; Lian, W.; Zi, Y.; Junjie, L.; Wenfeng, W.; Jing, L.; Yueyue, W.; Xue, S.; Xiaofeng, Z.; Zhijun, G. High expression of CAMSAP2 promotes invasion and metastasis of gastric cancer cells by upregulating TGF-β signaling. J. South Med. Univ. 2023, 43, 1460–1468. [Google Scholar]
- Lv, C.; Hu, Y.; Zhou, X.; Zhu, Y.; Wang, J.; Zhou, F. CircRNA SOD2 motivates non-small cell lungs cancer advancement with EMT via acting as microRNA-2355-5p’s competing endogenous RNA to mediate calmodulin regulated spectrin associated proteins-2. Bioengineered 2022, 13, 5756–5768. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Jiang, J.; Ni, C.; Xu, Q.; Ye, S.; Wu, J.; Ge, F.; Han, Y.; Mo, Y.; Huang, D.; et al. HBV Integration-mediated Cell Apoptosis in HepG2.2.15. J. Cancer 2019, 10, 4142–4150. [Google Scholar] [CrossRef] [PubMed]
- Pongrakhananon, V.; Wattanathamsan, O.; Takeichi, M.; Chetprayoon, P.; Chanvorachote, P. Loss of CAMSAP3 promotes EMT via the modification of microtubule–Akt machinery. J. Cell Sci. 2018, 131, jcs216168. [Google Scholar] [CrossRef]
- Seephan, S.; Sasaki, S.-I.; Wattanathamsan, O.; Singharajkomron, N.; He, K.; Ucche, S.; Kungsukool, S.; Petchjorm, S.; Chantaravisoot, N.; Wongkongkathep, P.; et al. CAMSAP3 negatively regulates lung cancer cell invasion and angiogenesis through nucleolin/HIF-1α mRNA complex stabilization. Life Sci. 2023, 322, 121655. [Google Scholar] [CrossRef]
- Singharajkomron, N.; Seephan, S.; Iksen, I.; Chantaravisoot, N.; Wongkongkathep, P.; Hayakawa, Y.; Pongrakhananon, V. CAMSAP3-mediated regulation of HMGB1 acetylation and subcellular localization in lung cancer cells: Implications for cell death modulation. Biochim. Et Biophys. Acta (BBA)—Gen. Subj. 2024, 1868, 130614. [Google Scholar] [CrossRef]
- Mao, X.; Tang, X.; Ye, J.; Xu, S.; Wang, Y.; Liu, X.; Wu, Q.; Lin, X.; Zhang, M.; Liu, J.; et al. Multi-omics profiling reveal cells with novel oncogenic cluster, TRAP1low/CAMSAP3low, emerge more aggressive behavior and poor-prognosis in early-stage endometrial cancer. Mol. Cancer 2024, 23, 127. [Google Scholar] [CrossRef]
- Wang, L.; Hu, X.; Li, S.; Liang, X.; Ren, L.; Lv, S. ASPM facilitates colorectal cancer cells migration and invasion by enhancing β-catenin expression and nuclear translocation. Kaohsiung J. Med. Sci. 2022, 38, 129–138. [Google Scholar] [CrossRef]
- Chen, X.; Huang, L.; Yang, Y.; Chen, S.; Sun, J.; Ma, C.; Xie, J.; Song, Y.; Yang, J. ASPM promotes glioblastoma growth by regulating G1 restriction point progression and Wnt-β-catenin signaling. Aging 2020, 12, 224–241. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.-J.; Sun, Y.; Gao, R.; Yin, Z.; Yuan, Z.; Xu, L.-M. Abnormal spindle-like microcephaly-associated protein (ASPM) contributes to the progression of Lung Squamous Cell Carcinoma (LSCC) by regulating CDK4. J. Cancer 2020, 11, 5413–5423. [Google Scholar] [CrossRef]
- Fang, Q.; Li, Q.; Qi, Y.; Pan, Z.; Feng, T.; Xin, W. ASPM promotes migration and invasion of anaplastic thyroid carcinoma by stabilizing KIF11. Cell Biol. Int. 2023, 47, 1209–1221. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Chen, X.; Li, D.; Yang, L.; Jiang, J. METTL3-mediated m6A methylation of ASPM drives hepatocellular carcinoma cells growth and metastasis. Clin. Lab. Anal. 2021, 35, e23931. [Google Scholar] [CrossRef] [PubMed]
- Ala, U.; Fagoonee, S. RNA-binding protein transcripts as potential biomarkers for detecting Primary Sclerosing Cholangitis and for predicting its progression to Cholangiocarcinoma. Front. Mol. Biosci. 2024, 11, 1388294. [Google Scholar] [CrossRef]
- Khosravani, H.; Ataee Disfani, R.; Farhadi, B.; Tohidian, M.; Garrosi, L.; Shirvani, P.; Zabihi, M.R.; Akhoondian, M.; Norouzkhani, N.; Farzan, R. Esophageal chemical burns as a risk factor for esophageal malignancies: In-silico analyses-experimental research. Ann. Med. Surg. 2024, 86, 5170–5178. [Google Scholar] [CrossRef]
- JagadeeswaraRao, G.; SivaPrasad, A. Identification of potential biomarkers for pancreatic ductal adenocarcinoma: A bioinformatics analysis. Comput. Methods Biomech. Biomed. Eng. 2024, 28, 2049–2063. [Google Scholar] [CrossRef]
- Saleh, A.A.; Gohar, S.F.; Hemida, A.S.; Elgharbawy, M.; Soliman, S.E. Evaluation of ASPM and TEF Gene Expressions as Potential Biomarkers for Bladder Cancer. Biochem. Genet. 2020, 58, 490–507. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Chu, J.; Hu, K.; Zhang, H.; Li, N.; Chu, J.; Hu, K.; Zhang, H. ASPM overexpression enhances cellular proliferation and migration and predicts worse prognosis for papillary renal cell carcinoma. J. Biosci. 2023, 48, 17. [Google Scholar] [CrossRef]
- Deng, T.; Liu, Y.; Zhuang, J.; Tang, Y.; Huo, Q. ASPM Is a Prognostic Biomarker and Correlates With Immune Infiltration in Kidney Renal Clear Cell Carcinoma and Liver Hepatocellular Carcinoma. Front. Oncol. 2022, 12, 632042. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Wang, Y.; Xue, F. ASPM, CDC20, DLGAP5, BUB1B, CDCA8, and NCAPG May Serve as Diagnostic and Prognostic Biomarkers in Endometrial Carcinoma. Genet. Res. 2022, 2022, 3217248. [Google Scholar] [CrossRef]
- Cheng, L.-H.; Hsu, C.-C.; Tsai, H.-W.; Liao, W.-Y.; Yang, P.-M.; Liao, T.-Y.; Hsieh, H.-Y.; Chan, T.-S.; Tsai, K.K. ASPM Activates Hedgehog and Wnt Signaling to Promote Small Cell Lung Cancer Stemness and Progression. Cancer Res. 2023, 83, 830–844. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.-C.; Liao, W.-Y.; Chang, K.-Y.; Chan, T.-S.; Huang, P.-J.; Chiang, C.-T.; Shan, Y.-S.; Cheng, L.-H.; Liao, T.-Y.; Tsai, K.K. A multi-mode Wnt- and stemness-regulatory module dictated by FOXM1 and ASPM isoform I in gastric cancer. Gastric Cancer 2021, 24, 624–639. [Google Scholar] [CrossRef]
- Khalaf-Nazzal, R.; Fasham, J.; Inskeep, K.A.; Blizzard, L.E.; Leslie, J.S.; Wakeling, M.N.; Ubeyratna, N.; Mitani, T.; Griffith, J.L.; Baker, W.; et al. Bi-allelic CAMSAP1 variants cause a clinically recognizable neuronal migration disorder. Am. J. Hum. Genet. 2022, 109, 2068–2079. [Google Scholar] [CrossRef]
- Hubert, T.; Perdu, S.; Vandekerckhove, J.; Gettemans, J. γ-Tubulin localizes at actin-based membrane protrusions and inhibits formation of stress-fibers. Biochem. Biophys. Res. Commun. 2011, 408, 248–252. [Google Scholar] [CrossRef]
- Martin, M.; Veloso, A.; Wu, J.; Katrukha, E.A.; Akhmanova, A. Control of endothelial cell polarity and sprouting angiogenesis by non-centrosomal microtubules. eLife 2018, 7, e33864. [Google Scholar] [CrossRef]

| Member | Primary Function | Expression Localization | Phenotypes Upon KO/Knockdown/Mutation | Associated Diseases/Phenotypes |
|---|---|---|---|---|
| CAMSAP1 | Dynamically tracks microtubule minus-ends and maintains structural integrity. | Neurons [19], spermatozoa [20], lung [21], liver [22], vascular smooth muscle cells et al. [23] | Formation of multiple axon-bearing neurons; deformed sperm nuclei and flagella; activation of anti-tumor immunity; inhibition of hepatic stellate cell activation; enhanced proliferation, migration, and phenotypic switching of vascular smooth muscle cells. | Cortical development malformations; oligoasthenozoospermia; lung cancer; liver fibrosis; in-stent restenosis. |
| CAMSAP2 | Co-decorates and stabilizes growing microtubule minus-ends alongside CAMSAP3. | Pancreatic β-cells [24], liver [7], lung [14], mitral valve cells [25], neurons et al. [26] | Impaired glucose-stimulated insulin secretion; transition of non-centrosomal microtubule arrays to a centrosomal pattern; suppression of lung cancer cell motility in vitro and metastasis in vivo; impaired classic morphology of mitral valve cells; increased neurite outgrowth. | Diabetes; hepatocellular carcinoma; lung cancer; olfactory defects; epilepsy. |
| CAMSAP3 | Co-decorates and stabilizes growing microtubule minus-ends alongside CAMSAP2. | Lung [27], kidney [28], neurons [29], trachea [30], intestine et al. [31] | Promotion of cellular senescence in lung cancer; cyst formation in kidneys; aberrant narrowing or fusion of the boundary between the striatum and septum; failure of multi-cilia to undergo synchronous beating; impaired positioning of nuclei and Golgi apparatus, and mitochondrial shaping. | Lung cancer; proximal renal tubule cyst formation; abnormally narrow lateral ventricles; tracheal ciliary dyskinesia; organ growth retardation and impaired physiological function. |
| Molecular Name | Cancer Type | OS | Invasion (↑/↓) | Migration/ Metastasis (↑/↓) | Proliferation (↑/↓) | Key Findings and Mechanisms | Experimental System |
|---|---|---|---|---|---|---|---|
| Mutated CAMSAP1 [21] | SCLC | Long | — | — | — | Mutant activates anti-tumor immunity, mediates tumor cell apoptosis, and inhibits EMT. | Patient Sample Analysis |
| CAMSAP1 [4] | LIHC | Short | — | — | — | The AC145207.5/LINC01748-miR-101-3p-CAMSAP1 axis promotes tumor progression. | Bioinformatics Analysis |
| circCAMSAP1 [52] | CRC | Short | — | — | ↑ | Acts as a molecular sponge for miR-328-5p, relieving its repression of the transcription factor E2F1, thereby driving cell cycle progression and proliferation. | Patient Samples, In Vitro, In Vivo |
| circCAMSAP1 [53] | HCC | — | ↑ | ↑ | ↑ | The circCAMSAP1/miR-1294/GRAMD1A axis promotes oncogenic phenotypes. | Patient Samples, In Vitro, In Vivo |
| circCAMSAP1 [54] | NPC | — | ↑ | ↑ | ↑ | Forms a positive feedback loop with SERPINH1 and c-Myc to sustain tumor growth and invasion. | Patient Samples, In Vitro, In Vivo |
| CAMSAP2 [7] | HCC | Short | ↑ | ↑ | — | Suppresses HDAC6 via the Rac1/JNK/c-Jun pathway, promoting non-centrosomal microtubule acetylation, which enhances stability and facilitates metastasis. | Patient Samples, In Vitro, In Vivo |
| CAMSAP2 [13] | CRC | Short | ↑ | ↑ | → | Activates the JNK/c-Jun signaling axis, upregulating Matrix Metalloproteinase-1 (MMP-1) expression to promote extracellular matrix degradation and invasion. | Patient Samples, In Vitro, In Vivo |
| CAMSAP2 [56] | GC | Short | ↑ | ↑ | — | Promotes EMT potentially via upregulation of the TGF-β signaling pathway. | Patient Samples, In Vitro, In Vivo |
| CAMSAP2 [57] | NSCLC | — | ↑ | ↑ | ↑ | The CircSOD2/miR-2355-5p/CAMSAP2 axis contributes to malignant behaviors. | Patient Samples, In Vitro |
| CAMSAP2 [14] | NSCLC | Short | — | ↑ | → | Promotes RASAL2 degradation, leading to subsequent activation of the ERK signaling pathway and enhanced cell migration. | Patient Samples, In Vitro, In Vivo |
| CAMSAP3 [60] | LUAD | Long | ↓ | ↓ | — | Binds NCL to negatively regulate HIF-1α mRNA stability, thereby inhibiting tumor angiogenesis and invasion. | Patient Samples, In Vitro, In Vivo |
| CAMSAP3 [27] | NSCLC | — | — | ↓ | ↑ | Depletion inhibits ERK phosphorylation and Cyclin D1 expression, inducing G1 cell cycle arrest and senescence-like phenotypes. | In Vitro, In Vivo |
| CAMSAP3 [61] | LC | Long | — | — | — | Interacts with acetylated HMGB1 to enhance autophagic cell death upon TSA treatment. | Patient Samples, In Vitro |
| ASPM-i1 [75] | SCLC | Short | ↑ | — | — | Stabilizes key Hedgehog signaling components (GLI1, DVL3, SMO), enhancing cancer stemness and progression. | Patient Samples, In Vitro, In Vivo |
| ASPM-i1 [76] | GC | Short | ↑ | — | — | Cooperates with FOXM1 and β-catenin to potentiate Wnt/β-catenin signaling. | Patient Samples, In Vitro, In Vivo |
| ASPM [63] | CRC | Short | ↑ | ↑ | → | Promotes β-catenin nuclear translocation, activating Wnt/β-catenin signaling to drive EMT, migration, and invasion. | Patient Samples, In Vitro, In Vivo |
| ASPM [65] | LSCC | Short | — | ↑ | — | Contributes to progression potentially by regulating CDK4 and cell cycle progression. | Patient Samples, In Vitro |
| ASPM [72] | PRCC | Short | ↑ | ↑ | ↑ | Promotes malignant phenotypes, partially through activation of the Wnt/β-catenin signaling pathway. | Patient Samples, In Vitro, In Vivo |
| ASPM [66] | ATC | — | ↑ | ↑ | — | Binds to and stabilizes kinesin KIF11 by inhibiting its ubiquitin-mediated degradation, thereby promoting cell motility. | Patient Samples, In Vitro, In Vivo |
| ASPM [67] | LIHC | Short | ↑ | ↑ | ↑ | The METTL3-mediated m6A methylation enhances ASPM mRNA stability and expression, forming a pro-oncogenic axis. | Patient Samples, In Vitro, In Vivo |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Q.; Li, X.; Xie, M.; Ding, X.; Li, D. Microtubule Minus-End Binding Proteins in Cancer: Advances. Diagnostics 2025, 15, 3116. https://doi.org/10.3390/diagnostics15243116
Wang Q, Li X, Xie M, Ding X, Li D. Microtubule Minus-End Binding Proteins in Cancer: Advances. Diagnostics. 2025; 15(24):3116. https://doi.org/10.3390/diagnostics15243116
Chicago/Turabian StyleWang, Qingwen, Xiuling Li, Meng Xie, Xiangming Ding, and Dongxiao Li. 2025. "Microtubule Minus-End Binding Proteins in Cancer: Advances" Diagnostics 15, no. 24: 3116. https://doi.org/10.3390/diagnostics15243116
APA StyleWang, Q., Li, X., Xie, M., Ding, X., & Li, D. (2025). Microtubule Minus-End Binding Proteins in Cancer: Advances. Diagnostics, 15(24), 3116. https://doi.org/10.3390/diagnostics15243116







