Heart Valve Surgery in Antiphospholipid Syndrome Patients—Morbidity and Mortality
Abstract
1. Introduction
2. Patients and Methods
2.1. Patients
2.2. Methods
- Demographic and clinical data for each patient were collected from electronic case files, including APS manifestations, the aPL profile, other immunologic serologies (antinuclear antibodies, anti-double stranded DNA antibodies, complement levels) and periprocedural blood test results up to 60 days before surgery (blood count indices, creatinine, direct Coombs test). The presence of aCL and aB2GPI of the IgG and IgM isotypes were measured by enzyme-linked immunosorbent assay (ELISA) or by a multiplex system. All kits were commercial (ELISA—aB2GPI by AESKU Diagnosis, and aCL by Varelisa; Bioplex both aB2GPI and aCL by BIORAD). The results were considered positive if they were above the upper limit of normal (ULN) as specified for each kit according to the manufacturer’s instructions (IgG phospholipid units or IgM phospholipid units), or >99th percentile in a minimum of two tests performed at least 12 weeks apart. They were considered clinically significantly positive if above 40 MPL or GPL (which is 2–4 fold above the ULN), as specified by the Sapporo criteria [1]. LA activity was detected by coagulation assays in routine use at each center, and it was consistent with the International Society of Thrombosis and Hemostasis guidelines [23]. LA activity was measured by LA-responsive activated partial thromboplastin time (aPTT) aPL (by Stago, with confirms by Actin FS kit by Siemens) up to 2016, and by Silica Clotting time and dilute Russel Viper Venom time (kit by Werfen) after 2016. In the case of anticoagulation treatment or spontaneous INR > 1.5, the patient’s plasma was mixed with normal plasma in order to reduce false positivity. aPL positivity was defined as single, double, or triple positive according to the number of different positive tests obtained.
- Cardiac characteristics: LSE based upon the local echocardiographic report [24], coronary pathologies based upon coronary angiography findings and indications for surgery.
- Medical treatment before and after surgery: mode of anticoagulation (low molecular weight heparin [LMWH], vitamin K antagonists [VKA], direct oral anticoagulants [DOAC], aspirin, glucocorticoids [intravenous pulse therapy, oral prednisone or prednisone equivalent dosage]) and immunosuppressants.
- The valve replaced or repaired and the type of valve inserted (mechanical or biologic).
- New York Heart Association functional classification (NYHA) was reported for patients before surgery.
- Probability of pulmonary hypertension (PHTN) as assessed by echocardiography was graded as none when systolic pulmonary artery pressure estimation was below 30 mmHg, low if 30–39 mmHg, intermediate if 40–59 mmHg and high if above 60 mmHg [25].
- Adverse outcomes following surgery were defined as early (if they occurred up to 30 days post-surgery) or late (if they occurred later than 30 days post-surgery until the last follow-up visit).
- -
- Postoperative bleeding, with severe bleeding defined as the need for transfusion or hemodynamic instability secondary to bleeding;
- -
- Thromboembolic events, either venous (deep venous thrombosis [DVT], pulmonary embolism [PE]), or arterial (valve thrombosis, myocardial infarction), as well as cerebrovascular events (transient ischemic attack [TIA] or stroke, limb ischemia or other arterial thrombosis) were diagnosed clinically and corroborated by an appropriate imaging study (Doppler ultrasound, computed tomography, etc.);
- -
- Infections including endocarditis, bacteremia or other infection necessitating systemic antibiotic treatment;
- -
- Post-pericardiotomy syndrome (PPS);
- -
- Requirement for further operation;
- -
- Death.
Statistical Analysis
3. Results
3.1. Cardiac Pathologies in APS Patients Requiring Valve Surgery
3.2. Heart Valve Surgery Characteristics
3.3. Medical Treatment before and after Valve Surgery
3.4. Primary Outcomes: Severe Complications and Mortality
3.4.1. Severe Complications
3.4.2. Mortality
3.5. Variables Associated with Severe Complications
3.6. Variables Associated with Mortality
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Miyakis, S.; Lockshin, M.D.; Atsumi, T.; Branch, D.W.; Brey, R.L.; Cervera, R.; Derkesen, R.H.W.M.; de Groot, P.G.; Koike, T.; Meroni, P.L.; et al. International Consensus Statement on an Update of the Classification Criteria for Definite Antiphospholipid Syndrome (APS). J. Thromb. Haemost. 2006, 4, 295–306. [Google Scholar] [CrossRef]
- Cervera, R. Antiphospholipid Syndrome. Thromb. Res. 2017, 151 (Suppl. S1), S43–S47. [Google Scholar] [CrossRef]
- Kolitz, T.; Shiber, S.; Sharabi, I.; Winder, A.; Zandman-Goddard, G. Cardiac Manifestations of Antiphospholipid Syndrome with Focus on Its Primary Form. Front. Immunol. 2019, 10, 941. [Google Scholar] [CrossRef]
- Tincani, A.; Rebaioli, C.B.; Taglietti, M.; Shoenfeld, Y. Heart Involvement in Systemic Lupus Erythematosus, Anti-Phospholipid Syndrome and Neonatal Lupus. Rheumatology 2006, 45, iv8–iv13. [Google Scholar] [CrossRef]
- Lockshin, M.; Tenedios, F.; Petri, M.; McCarty, G.; Forastiero, R.; Krilis, S.; Tincani, A.; Erkan, D.; Khamashta, M.A.; Shoenfeld, Y. Cardiac Disease in the Antiphospholipid Syndrome: Recommendations for Treatment. Committee Consensus Report. Lupus 2016, 12, 518–523. [Google Scholar] [CrossRef] [PubMed]
- Abreu, M.M.; Danowski, A.; Wahl, D.G.; Amigo, M.C.; Tektonidou, M.; Pacheco, M.S.; Fleming, N.; Domingues, V.; Sciascia, S.; Lyra, J.O.; et al. The Relevance of “Non-Criteria” Clinical Manifestations of Antiphospholipid Syndrome: 14th International Congress on Antiphospholipid Antibodies Technical Task Force Report on Antiphospholipid Syndrome Clinical Features. Autoimmun. Rev. 2015, 14, 401–414. [Google Scholar] [CrossRef]
- Sciascia, S.; Amigo, M.-C.C.; Roccatello, D.; Khamashta, M. Diagnosing Antiphospholipid Syndrome: “extra-Criteria” Manifestations and Technical Advances. Nat. Rev. Rheumatol. 2017, 13, 548–560. [Google Scholar] [CrossRef]
- Kampolis, C.; Tektonidou, M.; Moyssakis, I.; Tzelepis, G.E.; Moutsopoulos, H.; Vlachoyiannopoulos, P.G. Evolution of Cardiac Dysfunction in Patients with Antiphospholipid Antibodies and/or Antiphospholipid Syndrome: A 10-Year Follow-up Study. Semin. Arthritis Rheum. 2014, 43, 558–565. [Google Scholar] [CrossRef]
- Hojnik, M.; George, J.; Ziporen, L.; Shoenfeld, Y. Heart Valve Involvement (Libman-Sacks Endocarditis) in the Antiphospholipid Syndrome. Circulation 1996, 93, 1579–1587. [Google Scholar] [CrossRef] [PubMed]
- Amigo, M.C. What Do We Know about the Cardiac Valve Lesion in the Antiphospholipid Syndrome (APS)? Lupus 2014, 23, 1259–1261. [Google Scholar] [CrossRef] [PubMed]
- Pardos-Gea, J.; Avegliano, G.; Evangelista, A.; Vilardell, M.; Ordi-Ros, J. Cardiac Manifestations Other than Valvulopathy in Antiphospholipid Syndrome: Long-Time Echocardiography Follow-up Study. Int. J. Rheum. Dis. 2015, 18, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Espínola-Zavaleta, N.; Vargas-Barrón, J.; Colmenares-Galvis, T.; Cruz-Cruz, F.; Romero-Cárdenas, A.; Keirns, C.; Amigo, M.C. Echocardiographic Evaluation of Patients with Primary Antiphospholipid Syndrome. Am. Heart J. 1999, 137, 973–978. [Google Scholar] [CrossRef] [PubMed]
- Turiel, M.; Sarzi-Puttini, P.; Peretti, R.; Bonizzato, S.; Muzzupappa, S.; Atzeni, F.; Rossi, E.; Doria, A. Five-Year Follow-up by Transesophageal Echocardiographic Studies in Primary Antiphospholipid Syndrome. Am. J. Cardiol. 2005, 96, 574–579. [Google Scholar] [CrossRef]
- Gorki, H.; Malinovski, V.; Stanbridge, R.D.L. The Antiphospholipid Syndrome and Heart Valve Surgery. Eur. J. Cardio-Thorac. Surg. 2008, 33, 168–181. [Google Scholar] [CrossRef] [PubMed]
- Nesher, G.; Ilany, J.; Rosenmann, D.; Abraham, A.S. Valvular Dysfunction in Antiphospholipid Syndrome: Prevalence, Clinical Features, and Treatment. Semin. Arthritis Rheum. 1997, 27, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Erdozain, J.G.; Ruiz-Irastorza, G.; Segura, M.I.; Amigo, M.C.; Espinosa, G.; Pomar, J.L.; Perez-Valero, I.; Ateka-Barrutia, O.; Khamashta, M.A. Cardiac Valve Replacement in Patients with Antiphospholipid Syndrome. Arthritis Care Res. 2012, 64, 1256–1260. [Google Scholar] [CrossRef]
- Moyssakis, I.; Tektonidou, M.G.; Vasilliou, V.A.; Samarkos, M.; Votteas, V.; Moutsopoulos, H.M. Libman-Sacks Endocarditis in Systemic Lupus Erythematosus: Prevalence, Associations, and Evolution. Am. J. Med. 2007, 120, 636–642. [Google Scholar] [CrossRef]
- Lee, J.L.; Naguwa, S.M.; Cheema, G.S.; Gershwin, M.E. Revisiting Libman-Sacks Endocarditis: A Historical Review and Update. Clin. Rev. Allergy Immunol. 2009, 36, 126–130. [Google Scholar] [CrossRef]
- Atisha-Fregoso, Y.; Espejo-Poox, E.; Carrillo-Maravilla, E.; Pulido-Ramírez, A.L.; Lugo Baruqui, D.; Hernández-Molina, G.; Cabral, A.R. Perioperative Management of Patients with Antiphospholipid Syndrome: A Single-Center Experience. Rheumatol. Int. 2017, 37, 1159–1164. [Google Scholar] [CrossRef]
- Raso, S.; Sciascia, S.; Kuzenko, A.; Castagno, I.; Marozio, L.; Bertero, M.T. Bridging Therapy in Antiphospholipid Syndrome and Antiphospholipid Antibodies Carriers: Case Series and Review of the Literature. Autoimmun. Rev. 2014, 14, 36–42. [Google Scholar] [CrossRef]
- Berkun, Y.; Elami, A.; Meir, K.; Mevorach, D.; Naparstek, Y. Increased Morbidity and Mortality in Patients with Antiphospholipid Syndrome Undergoing Valve Replacement Surgery. J. Thorac. Cardiovasc. Surg. 2004, 127, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Colli, A.; Mestres, C.A.; Espinosa, G.; Plasín, M.A.; Pomar, J.L.; Font, J.; Cervera, R. Heart Valve Surgery in Patients with the Antiphospholipid Syndrome: Analysis of a Series of Nine Cases. Eur. J. Cardiothorac. Surg. 2010, 37, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Brandt, J.T.; Barna, L.K.; Triplett, D.A. Laboratory Identification of Lupus Anticoagulants: Results of the Second International Workshop for Identification of Lupus Anticoagulants. Thromb. Haemost. 1995, 74, 1597–1603. [Google Scholar] [CrossRef]
- Roldan, C.A.; Qualls, C.R.; Sopko, K.S.; Sibbitt, W.L. Transthoracic versus Transesophageal Echocardiography for Detection of Libman-Sacks Endocarditis: A Randomized Controlled Study. J. Rheumatol. 2008, 35, 224–229. [Google Scholar] [PubMed]
- Frost, A.; Badesch, D.; Gibbs, J.S.; Gopalan, D.; Khanna, D.; Manes, A.; Oudiz, R.; Satoh, T.; Torres, F.; Torbicki, A. Diagnosis of Pulmonary Hypertension. Eur. Respir. J. 2019, 53, 1801904. [Google Scholar] [CrossRef]
- Tenedios, F.; Erkan, D.; Lockshin, M.D. Cardiac Involvement in the Antiphospholipid Syndrome. Lupus 2005, 14, 691–696. [Google Scholar] [CrossRef]
- Amital, H.; Langevitz, P.; Levy, Y.; Afek, A.; Goldberg, I.; Pras, M.; Livneh, A.; Shoenfeld, Y. Valvular Deposition of Antiphospholipid Antibodies in the Antiphospholipid Syndrome: A Clue to the Origin of the Disease. Clin. Exp. Rheumatol. 1999, 17, 99–102. [Google Scholar] [PubMed]
- Ziporen, L.; Goldberg, I.; Arad, M.; Hojnik, M.; Ordi-Ros, J.; Afek, A.; Blank, M.; Sandbank, Y.; Vilardell-Tarres, M.; de Torres, I.; et al. Libman-Sacks Endocarditis in the Antiphospholipid Syndrome: Immunopathologic Findings in Deformed Heart Valves. Lupus 1996, 5, 196–205. [Google Scholar] [CrossRef]
- Nishimura, R.A.; Otto, C.M.; Bonow, R.O.; Carabello, B.A.; Erwin, J.P.; Fleisher, L.A.; Jneid, H.; Mack, M.J.; McLeod, C.J.; O’Gara, P.T.; et al. 2017 AHA/ACC Focused Update of the 2014 AHA/ACC Guideline for the Management of Patients with Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2017, 135, e1159–e1195. [Google Scholar] [CrossRef]
- Zhou, S.; Egorova, N.; Moskowitz, G.; Giustino, G.; Ailawadi, G.; Acker, M.A.; Gillinov, M.; Moskowitz, A.; Gelijns, A. Trends in MitraClip, Mitral Valve Repair, and Mitral Valve Replacement from 2000 to 2016. J. Thorac. Cardiovasc. Surg. 2021, 162, 551–562.e4. [Google Scholar] [CrossRef]
- Arif, R.; Farag, M.; Seppelt, P.; Beller, C.J.; Ruhparwar, A.; Karck, M.; Kallenbach, K. Patients with Systemic Lupus Erythematosus and Antiphospholipid Syndrome Undergoing Cardiac Valve Surgery. J. Heart Valve Dis. 2015, 24, 228–235. [Google Scholar]
- Mishra, P.K.; Khazi, F.M.; Yiu, P.; Billing, J.S. Severe Antiphospholipid Syndrome and Cardiac Surgery: Perioperative Management. Asian Cardiovasc. Thorac. Ann. 2016, 24, 473–476. [Google Scholar] [CrossRef] [PubMed]
- Massoudy, P.; Cetin, S.M.; Thielmann, M.; Kienbaum, P.; Piotrowski, J.A.; Marggraf, G.; Specker, C.; Jakob, H. Antiphospholipid Syndrome in Cardiac Surgery—An Underestimated Coagulation Disorder? Eur. J. Cardiothorac. Surg. 2005, 28, 133–137. [Google Scholar] [CrossRef]
- Ciocca, R.G.; Choi, J.; Graham, A.M. Antiphospholipid Antibodies Lead to Increased Risk in Cardiovascular Surgery. Am. J. Surg. 1995, 170, 198–200. [Google Scholar] [CrossRef] [PubMed]
- Alkhouli, M.; Nishimura, R.A.; Alqahtani, F.; Simard, T.; Pislaru, S.; Schaff, H.V. Predictors of Use and Outcomes of Mechanical Valve Replacement in the United States (2008–2017). J. Am. Heart Assoc. 2021, 10, e019929. [Google Scholar] [CrossRef] [PubMed]
- Oku, K.; Atsumi, T.; Bohgaki, M.; Amengual, O.; Kataoka, H.; Horita, T.; Yasuda, S.; Koike, T. Complement Activation in Patients with Primary Antiphospholipid Syndrome. Ann. Rheum. Dis. 2009, 68, 1030–1035. [Google Scholar] [CrossRef]
- Chaturvedi, S.; Brodsky, R.A.; McCrae, K.R. Complement in the Pathophysiology of the Antiphospholipid Syndrome. Front. Immunol. 2019, 10, 449. [Google Scholar] [CrossRef]
- Nalli, C.; Lini, D.; Andreoli, L.; Crisafulli, F.; Fredi, M.; Lazzaroni, M.G.; Bitsadze, V.; Calligaro, A.; Canti, V.; Caporali, R.; et al. Low Preconception Complement Levels Are Associated with Adverse Pregnancy Outcomes in a Multicenter Study of 260 Pregnancies in 197 Women with Antiphospholipid Syndrome or Carriers of Antiphospholipid Antibodies. Biomedicines 2021, 9, 671. [Google Scholar] [CrossRef]
- Thakar, C.V.; Worley, S.; Arrigain, S.; Yared, J.P.; Paganini, E.P. Influence of Renal Dysfunction on Mortality after Cardiac Surgery: Modifying Effect of Preoperative Renal Function. Kidney Int. 2005, 67, 1112–1119. [Google Scholar] [CrossRef]
| APS Patients (n = 26) | |
|---|---|
| Female subjects n (%) | 20 (76.9) |
| Age, years mean ± SD | 45.6 ± 13.4 |
| SLE, n (%) | 11 (42.3) |
| APS disease duration, years mean ± SD | 16.1 ± 12.8 |
| Immunologic laboratory values | |
| aCL-IgM positive n (%) | 5 (19.2) |
| aCL-IgG positive n (%) | 20 (76.9) |
| aB2GPI-IgM positive n (%) | 5 (19.2) |
| aB2GPI-IgG positive n (%) | 18 (69.2) |
| LA positive (n = 24) n (%) | 22 (91.7) |
| Triple positive (n = 24) n (%) | 19 (79.2) |
| Direct Coombs positive n (%) | 5/24 (20.8) |
| C3 titer mean ± SD | 87.3 ± 19.3 |
| Low C3 < 85 n (%) | 10 (38.5) |
| C4 titer mean ± SD | 16.6 ± 7.1 |
| Low C4 < 15 n (%) | 7 (26.9) |
| APS manifestations n (%) | |
| Any venous thromboembolism | 10 (38.5) |
| Deep vein thrombosis | 10 (100) |
| Pulmonary embolism | 5 (50) |
| Arterial thrombosis | 22 (84.6) |
| Stroke | 12 (46.1) |
| Transient ischemic attack | 4 (15.4) |
| Myocardial infarction | 3 (11.5) |
| Peripheral arterial thrombosis (limb, spleen, etc.) | 11 (42.3) |
| Obstetric manifestations n = 20 | 6 (30) |
| Treatment | Chronic Medical Therapy before Surgery | Medical Therapy after Surgery |
|---|---|---|
| Aspirin | 12 (46.2) | 11 (42.3) |
| VKA | 10 (38.5) | 20 (76.9) |
| LMWH | 9 (34.6) | 4 (15.4) |
| DOAC (rivaroxaban) | 1 (3.8) | 0 |
| Fondaparinux | 2 (7.7) | 1 (3.8) |
| Heparin | 0 | 1 (3.8) |
| Immunotherapy | Before Hospitalization for Surgery Number/Total with Available Data (%) | Peri-Surgical Period, Number/Total with Available Data (%) |
|---|---|---|
| Pulse GC | 1/25 (4) | 3/24 (12.5) |
| Hydroxychloroquine | 8/25 (32) | 7/24 (29.2) |
| Azathioprine | 5/25 (20) | 4/24 (16.7) |
| Rituximab | 2/25 (8) | 2/24 (8.3) |
| Cyclophosphamide | 1/25 (8) | 1/24 (4.2) |
| Plasma exchange | 1/25 (4) | 3/24 (12.5) |
| IVIg | 2/25 (8) | 2/24 (8.3) |
| Mycophenolate mofetil | 0 | 0 |
| Belimumab | 1/25 (4) | 0 |
| Variable n(%) | Total Severe Complications (n = 14) | Uneventful Follow-Up (n = 11) | OR (95%CI) | p-Value |
|---|---|---|---|---|
| Female subjects | 11 (55) | 9 (45) | 1.222 (0.197–7.594) | >0.99 |
| Age at surgery (mean ± SD) | 47.86 ± 13.92 | 43 ± 12.842 | 0.367 | |
| APS with SLE | 6 (54.54) | 5 (45.45) | 1.05 (0.22–5.003) | >0.99 |
| Positive LA ** | 12 (54.5) | 10 (45.5) | 1.2 (0.066–21.723) | >0.99 |
| Triple positive aPL ** | 10 (52.6) | 9 (47.4) | 0.741 (0.1–5.49) | >0.99 |
| Low C3 | 8 (80) | 2 (20) | 6.667 (1.047–42.431) | 0.051 |
| Low C4 | 6 (85.7) | 1 (14.3) | 8.25 (0.823–82.665) | 0.081 |
| APS duration | 12.11 ± 13.36 | 20.79 ± 10.83 | 0.084 | |
| Platelet count, 10 × 103/µL | 127.07 ± 44.17 | 118.91 ± 60.73 * | 0.701 | |
| Hemoglobin, g/dL | 10.87 ± 1.53 | 11.81 ± 1.41 * | 0.127 | |
| Creatinine mg/dL | 1.88 ± 1.78 | 1.12 ± 0.4 *** | 0.146 | |
| eGFR ml/min/1.73 m (MDRD) | 57.14 ± 34.64 | 74 ± 36.39 *** | 0.277 | |
| APS manifestations | ||||
| VTE | 4 (40) | 6 (60) | 0.333 (0.063–1.752) | 0.241 |
| CVA/TIA | 8 (61.5) | 5 (38.5) | 1.6 (0.326–7.848) | 0.695 |
| Arterial APS | 12 (57.1) | 9 (42.9) | 1.333 (0.157–11.356) | >0.99 |
| Cardiac pathology | ||||
| LSE | 11 (73.3) | 4 (26.7) | 7.333 (1.272–42.294) | 0.045 |
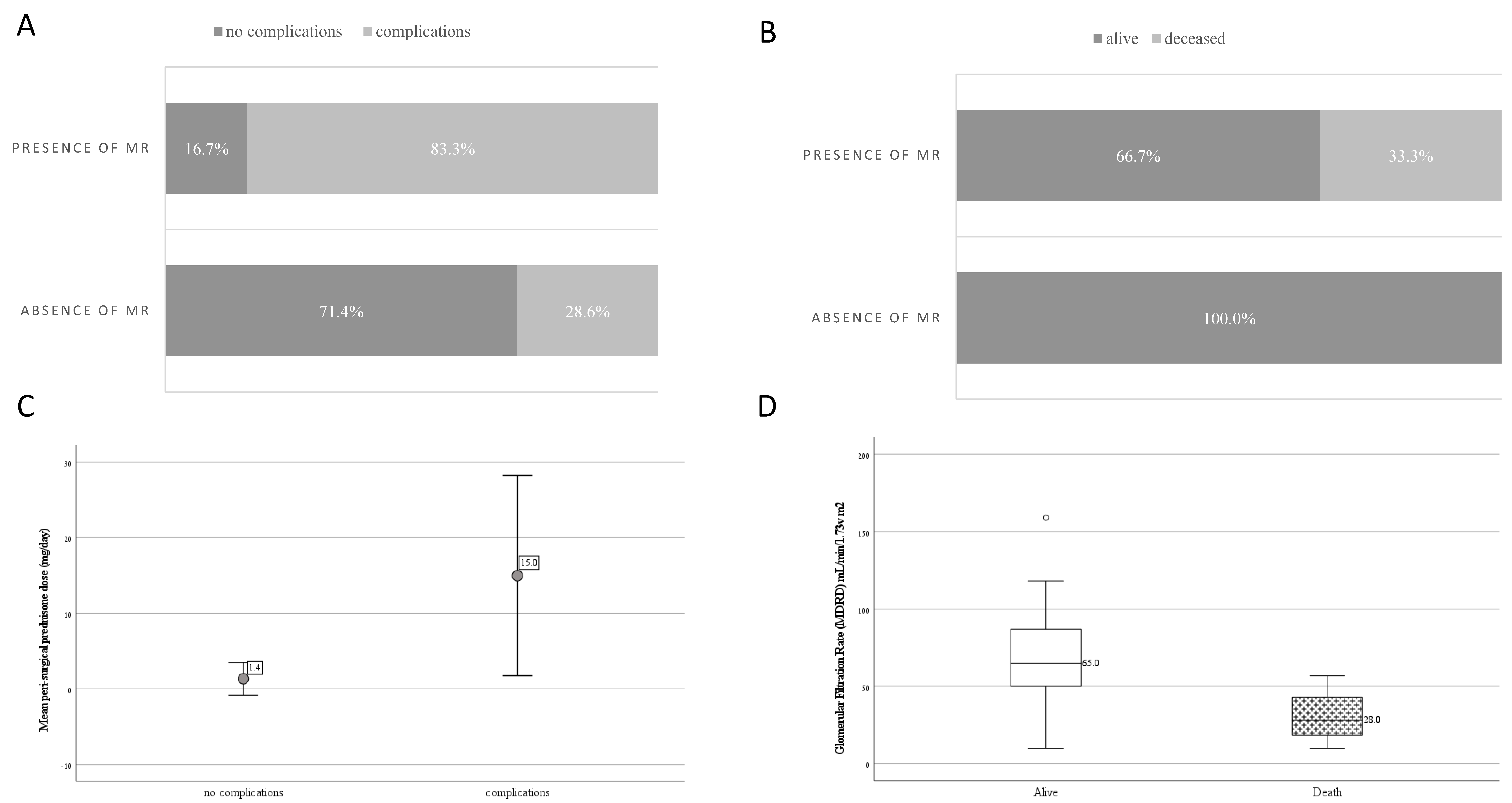
| Moderate-to-severe MR | 10 (83.2) | 2 (16.7) | 12.5 (1.85–84.442) | 0.008 |
| Moderate-to-severe AR | 3 (37.5) | 5 (62.5) | 0.382 (0.069–2.125) | 0.401 |
| Moderate-to-severe TR | 3 (75) | 1 (25) | 3 (0.269–33.487) | 0.598 |
| Intermediate-high probability of PHTN | 7 (36.4) | 4 (63.6) | 1.75 (0.329–9.298) | 0.680 |
| NYHA class | 2.21 ± 0.96 | 2.25 ± 0.866 | 0.923 | |
| Surgery year > 2015 | 8 (61.5) | 5 (38.5) | 1.867 (0.392–8.894) | 0.695 |
| Prosthetic valve | ||||
| Only repair | 1 (50) | 1 (50) | NA | 0.348 |
| Mechanic | 7 (43.8) | 9 (56.3) | ||
| Biologic | 6 (75) | 2 (25) | ||
| CABG | 1 (100) | 0 (0) | >0.99 | |
| Post-op redo | 3 (100) | 0 | 0.225 | |
| Medical treatment | ||||
| Background therapy | ||||
| VKA | 3 (30) | 7 (70) | 0.195 (0.035–1.084) | 0.105 |
| Aspirin | 7 (58.3) | 5 (41.7) | 1.4 (0.296–6.622) | 0.713 |
| LMWH | 7 (77.8) | 2 (22.2) | 5.0 (0.79–31.627) | 0.11 |
| Prednisone dose mg/day | 7.14 ± 15.78 | 1.36 ± 2.34 * | 0.243 | |
| Peri-surgical prednisone dose mg/day | 15 ± 21.89 * | 1.36 ± 3.23 * | 0.046 | |
| Post-surgical therapy | ||||
| VKA | 10 (50) | 10 (50) | 0.5 (0.074–3.378) | 0.652 |
| Aspirin | 6 (54.5) | 5 (45.5) | 1.05 (0.22–5.003) | >0.99 |
| LMWH | 3 (75) | 1 (25) | 3 (0.269–33.487) | 0.598 |
| Prednisone dose mg/day | 14.62 ± 23.32 * | 1.36 ± 3.23 * | 0.065 |
| Variable n(%) | Total Mortality n = 4 | No Mortality n = 22 | OR (95%CI) | p-Value |
|---|---|---|---|---|
| Female subjects | 2 (10) | 18 (90) | 0.222 (0.024–2.086) | 0.218 |
| Age at surgery | 48.25 ± 6.95 | 45.14 ± 14.32 | 0.678 | |
| APS with SLE | 3 (27.3) | 8 (72.7) | 5.25 (0.465–59.286) | 0.279 |
| Positive LA ** | 2 (9.1) | 20 (90.9) | 0.1 (0.004–2.287) | 0.239 |
| Triple positive aPL ** | 1 (5.3) | 18 (94.7) | 0.083 (0.006–1.232) | 0.099 |
| Low C3 | 2 (20) | 8 (80) | 1.75 (0.205–14.931) | 0.625 |
| Low C4 | 1 (14.3) | 6 (85.7) | 0.889 (0.077–10.3) | >0.99 |
| Platelet count, 103/µL | 121.5 ± 55.82 | 123.86 ± 51.62 * | 0.935 | |
| Hemoglobin, g/dL | 10.75 ± 1.7 | 11.38 ± 1.51 * | 0.46 | |
| Creatinine mg/dL | 2.65 ± 1.55 | 1.36 ± 1.35 | 0.105 | |
| eGFR ml/min/1.73 m (MDRD) | 30.75 ± 19.47 | 70.68 ± 34.44 | 0.038 | |
| APS duration | 14.75 ± 17.35 | 16.36 ± 12.32 | 0.822 | |
| APS manifestations | ||||
| VTE | 3 (30) | 7 (70) | 6.0 (0.524–68.719) | 0.267 |
| CVA/TIA | 3 (23.1) | 10 (76.9) | 3.3 (0.294–37.103) | 0.593 |
| Arterial APS | 4 (19) | 17 (81) | NA | >0.99 |
| Cardiac pathology | ||||
| LSE | 4 (26.7) | 11 (73.3) | NA | 0.113 |
| Moderate-to-severe MR | 4 (33.3) | 8 (66.7) | NA | 0.033 |
| Moderate-to-severe AR | 1 (12.5) | 7 (87.5) | 0.714 (0.063–8.15) | >0.99 |
| Moderate-to-severe TR | 2 (50) | 2 (50) | 10.0 (0.871–114.746) | 0.099 |
| Intermediate-high probability of PHTN | 2 (18.2) | 9 (81.8) | 1.11 (0.129–9.605) | >0.99 |
| NYHA class | 2.25 ± 1.5 | 2.23 ± 0.81 | 0.978 | |
| Surgery year > 2015 | 1 (7.7) | 12 (92.3) | 0.278 (0.025–3.104) | 0.593 |
| Prosthetic valve | ||||
| Only repair | 0 (0) | 2 (100) | NA | 0.228 |
| Mechanic | 4 (25) | 12 (75) | ||
| Biologic | 0 (0) | 8 (100) | ||
| CABG | 1 (100) | 0 (0) | NA | 0.154 |
| Medical treatment | ||||
| Background therapy | ||||
| VKA | 1 (10) | 9 (90) | 0.481 (0.043–5.401) | >0.99 |
| Aspirin | 3 (25) | 9 (75) | 4.33 (0.386–48.61) | 0.306 |
| LMWH | 3 (33.3) | 6 (66.7) | 8 (0.69–92.703) | 0.104 |
| Prednisone dose mg/day | 18.75 ± 27.8 | 1.9 ± 3.35 * | 0.312 | |
| Peri-surgical prednisone dose mg/day | 20 ± 27.39 | 6.5 ± 14.7 ** | 0.161 | |
| Post-surgical therapy | ||||
| VKA | 3 (15) | 17 (85) | 0.882 (0.074–10.464) | >0.99 |
| Aspirin | 2 (18.2) | 9 (81.8) | 1.444 (0.171–12.232) | >0.99 |
| LMWH | 1 (25) | 3 (75) | 2.111 (0.162–27.582) | 0.511 |
| Postsurgical prednisone dose mg/day | 32.5 ± 32.02 | 3.75 ± 9.85 ** | 0.17 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eviatar, T.; Niznik, S.; Elkayam, O.; Ben-Gal, Y.; Shavit, R.; Raanani, E.; Agmon-Levin, N.; Paran, D. Heart Valve Surgery in Antiphospholipid Syndrome Patients—Morbidity and Mortality. Life 2023, 13, 891. https://doi.org/10.3390/life13040891
Eviatar T, Niznik S, Elkayam O, Ben-Gal Y, Shavit R, Raanani E, Agmon-Levin N, Paran D. Heart Valve Surgery in Antiphospholipid Syndrome Patients—Morbidity and Mortality. Life. 2023; 13(4):891. https://doi.org/10.3390/life13040891
Chicago/Turabian StyleEviatar, Tali, Stanley Niznik, Ori Elkayam, Yanai Ben-Gal, Ronen Shavit, Ehud Raanani, Nancy Agmon-Levin, and Daphna Paran. 2023. "Heart Valve Surgery in Antiphospholipid Syndrome Patients—Morbidity and Mortality" Life 13, no. 4: 891. https://doi.org/10.3390/life13040891
APA StyleEviatar, T., Niznik, S., Elkayam, O., Ben-Gal, Y., Shavit, R., Raanani, E., Agmon-Levin, N., & Paran, D. (2023). Heart Valve Surgery in Antiphospholipid Syndrome Patients—Morbidity and Mortality. Life, 13(4), 891. https://doi.org/10.3390/life13040891







