Shrew Communities in Mediterranean Agro-Ecosystems of Central Greece: Associations with Crop Types, Land Uses, and Soil Parameters
Abstract
:1. Introduction
2. Materials and Methods
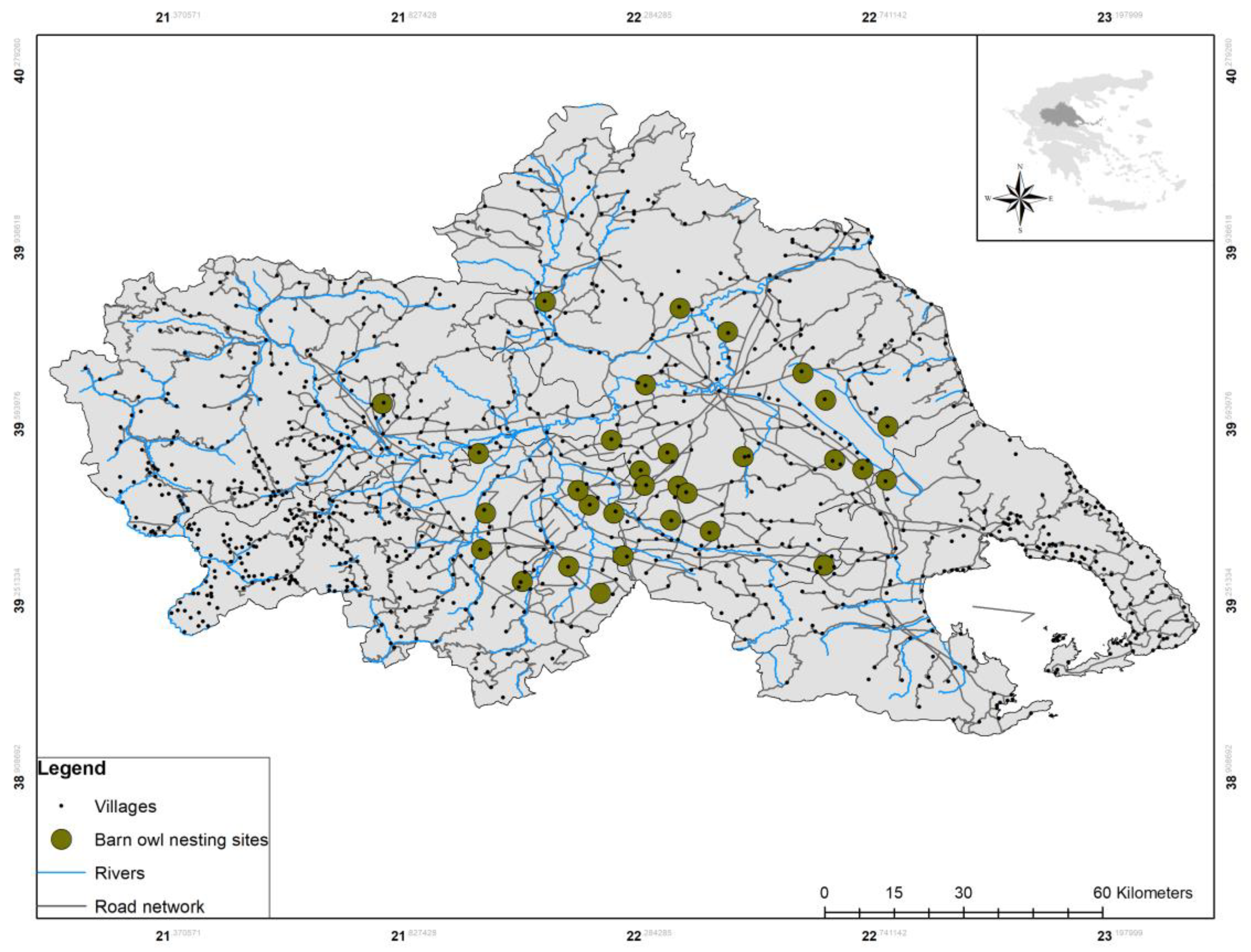
2.1. Field Work and Data Collection
2.2. Data Analysis
2.3. Datasets
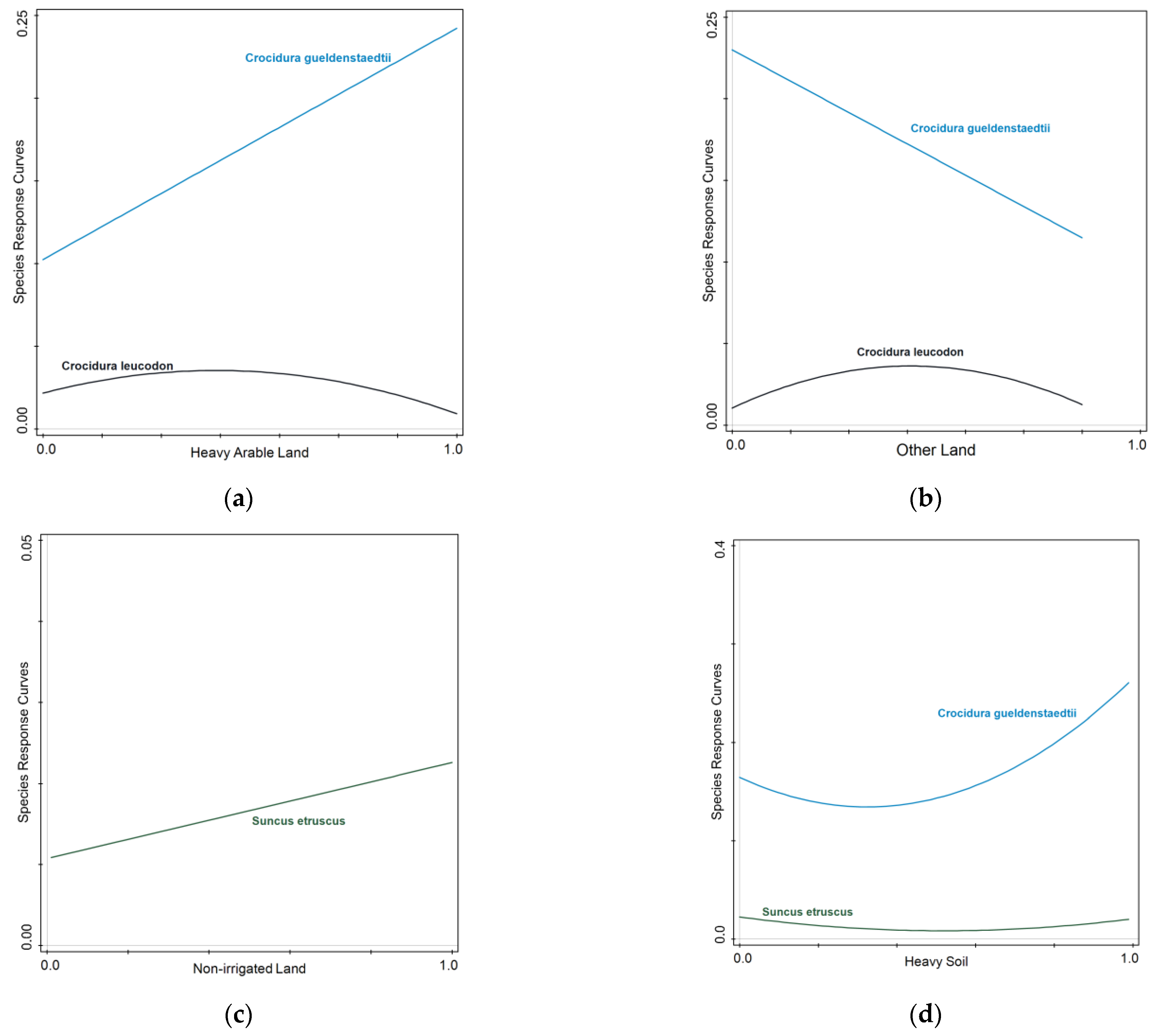
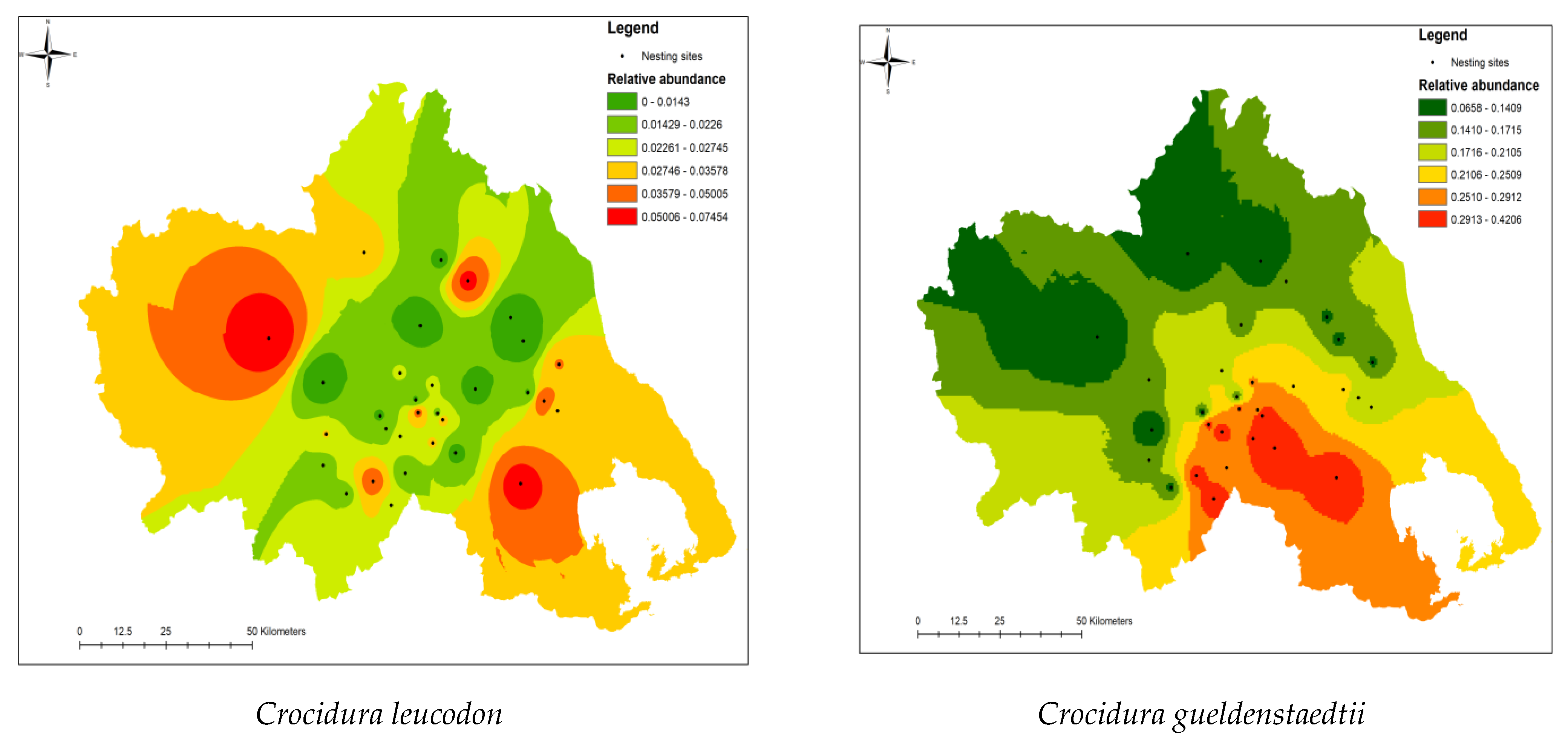
3. Results
4. Discussion
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bontzorlos, V. La Ecologia Trofica de la Lechuza Comun en los Ecosistemas Agricolas de Grecia Central: Su Aplicacion a la Distribucion y Abundancia de sus Presas. Ph.D. Thesis, University of Salamanca, Salamanca, Spain, 30 January 2009. [Google Scholar]
- Feldhamer, G.A. (Ed.) Mammalogy: Adaptation, Diversity, Ecology; Johns Hopkins University Press: Baltimore, ML, USA, 2015; p. 747. [Google Scholar]
- Stefke, K.; Landler, L. Long-term monitoring of rodent and shrew communities in a biodiversity hot-spot in Austria using barn owl (Tyto alba) pellets. Acta Oecologica 2020, 109, 103660. [Google Scholar] [CrossRef]
- Wilson, D.E.; Reeder, D.M. (Eds.) Mammal Species of the World: A Taxonomic and Geographic Reference, 3rd ed.; Johns Hopkins University Press: Baltimore, ML, USA, 2005; p. 2142. [Google Scholar]
- Neves, T.; Borda-de-Água, L.; Mathias, M.D.L.; Tapisso, J.T. The Influence of the Interaction between Climate and Competition on the Distributional Limits of European Shrews. Animals 2022, 12, 57. [Google Scholar] [CrossRef]
- Bontzorlos, V.; Vlachopoulos, K.; Xenos, A. Distribution of Four Vole Species through the Barn Owl Tyto alba Diet Spectrum: Pattern Responses to Environmental Gradients in Intensive Agroecosystems of Central Greece. Life 2023, 13, 105. [Google Scholar] [CrossRef]
- Burel, F.; Baudry, J.; Butet, A.; Clergeau, P.; Delettre, Y.; Le Coeur, D.; Dubs, F.; Morvan, N.; Paillat, G.; Petit, S.; et al. Comparative biodiversity along a gradient of agricultural landscapes. Acta Oecologica 1998, 19, 47–60. [Google Scholar] [CrossRef]
- Donald, P.F.; Green, R.E.; Heath, M.F. Agricultural intensification and the collapse of Europe’s farmland bird populations. Proc. R. Soc. Lond. B 2001, 268, 25–29. [Google Scholar] [CrossRef]
- Hazell, P.; Wood, S. Drivers of change in global agriculture. Philos. Trans. R. Soc. B 2008, 363, 495–515. [Google Scholar] [CrossRef]
- Gonthier, D.J.; Ennis, K.K.; Farinas, S.; Hsieh, H.-Y.; Iverson, A.L.; Batary, P.; Rudolphi, J.; Tscharntke, T.; Cardinale, B.J.; Perfecto, I. Biodiversity conservation in agriculture requires a multi-scale approach. Proc. R. Soc. Lond. B 2014, 281, 20141358. [Google Scholar] [CrossRef]
- Rowe, R.J.; Terry, R.C. Small mammal responses to environmental change: Integrating past and present dynamics. J. Mammal. 2014, 95, 1157–1174. [Google Scholar] [CrossRef]
- Balčiauskas, L.; Balčiauskienė, L. Small Mammal Diversity Changes in a Baltic Country, 1975–2021: A Review. Life 2022, 12, 1887. [Google Scholar] [CrossRef]
- Lomba, A.; Ferreiro da Costa, J.; Ramil-Rego, P.; Corbelle-Rico, E. Assessing the link between farming systems and biodiversity in agricultural landscapes: Insights from Galicia (Spain). J. Environ. Manag. 2022, 317, 115335. [Google Scholar] [CrossRef]
- Torre, I.; Bastardas-Llabot, J.; Arrizabalaga, A.; Diaz, M. Population dynamics of small endotherms under global change: Greater white-toothed shrews Crocidura russula in Mediterranean habitats. Sci. Total Environ. 2020, 705, 135799. [Google Scholar] [CrossRef]
- Torre, I.; Diaz, M. Assessing the Effects of Landscape Change on the Occupancy Dynamics of the Greater White-Toothed Shrew Crocidura russula. Life 2022, 12, 1230. [Google Scholar] [CrossRef]
- Mortellity, A.; Boitani, L. Distribution and coexistence of shrews in patchy landscapes: A field test of multiple hypotheses. Acta Oecologica 2009, 35, 797–804. [Google Scholar] [CrossRef]
- Torre, I.; Arrizabalaga, A.; Flaquer, C. Three Methods for Assessing Richness and Composition of Small Mammal Communities. J. Mammal. 2004, 85, 524–530. [Google Scholar] [CrossRef]
- Torre, I.; Fernández, L.; Arrizabalaga, A. Using barn owl Tyto alba pellet analyses to monitor the distribution patterns of the yellow-necked mouse (Apodemus flavicollis Melchior 1834) in a transitional Mediterranean mountain. Mammal Study 2015, 40, 133–142. [Google Scholar] [CrossRef]
- Paspali, G.; Oruci, S.; Koni, M.; Wilson, I.F.; Krystufek, B.; Bedo, F. Seasonal variation of small mammals in the diet of the barn owl (Tyto alba) in the Drinos River valley, southern Albania. Turk. J. Zool. 2013, 37, 97–105. [Google Scholar] [CrossRef]
- Heisler, L.M.; Somers, C.M.; Poulin, R.G.; Fisher, D. Owl pellets: A more effective alternative to conventional trapping for broad-scale studies of small mammal communities. Methods Ecol. Evol. 2016, 7, 96–103. [Google Scholar] [CrossRef]
- Milana, G.; Lai, M.; Maiorano, L.; Luiselli, L.; Amori, G. Geographic patterns of predator niche breadth and prey species richness. Ecol. Res. 2016, 31, 111–115. [Google Scholar] [CrossRef]
- Milana, G.; Luiselli, L.; Amori, G. Forty years of dietary studies on barn owl (Tyto alba) reveal long term trends in diversity metrics of small mammal prey. Anim. Biol. 2018, 68, 129–146. [Google Scholar] [CrossRef]
- Milana, G.; Battisti, C.; Luiselli, L.; Amori, G. Altitudinal variation of community metrics in Italian small mammal assemblages as revealed by Barn Owl (Tyto alba) pellets. Zool. Anz. 2019, 281, 11–15. [Google Scholar] [CrossRef]
- Balestrieri, A.; Gazzola, A.; Formenton, G.; Canova, L. Long-term impact of agricultural practices on the diversity of small mammal communities: A case study based on owl pellets. Environ. Monit. Assess. 2019, 191, 725. [Google Scholar] [CrossRef] [PubMed]
- Battisti, C.; Dodaro, G.; Di Bagno, E.; Amori, G. Reviewing an eco-biogeographic question at regional scale: The unexpected absence of a ubiquitous mammal species (Microtus savii, Rodentia) in coastal Southern Tuscany (central Italy). Rend. Lincei. Sci. Fis. E Nat. 2019, 30, 715–722. [Google Scholar] [CrossRef]
- Kiamos, N.; Lymberakis, P.; Rallis, G.; Poulakakis, N. Barn Owl (Tyto alba) prey in Evros (Greece) and the discovery of a new mammal for the Greek fauna. J. Nat. Hist. 2019, 53, 1691–1705. [Google Scholar] [CrossRef]
- Marti, C.D. Raptor food habit studies. In Raptor Management Techniques Manual; Pendleton, B.A., Millsap, B.A., Cleine, K.W., Bird, D.M., Eds.; Natural Wildlife Federation of Science Techniques: Washington, DC, USA, 1987; 420p. [Google Scholar]
- Yalden, D. The Analysis of Owl Pellets; Mammal Society: London, UK, 2003; 28p. [Google Scholar]
- Toschi, A.; Lanza, B. Mammalia. Generalita, Insectivora, Chiroptera; Edizioni Calderini: Bologna, Italy, 1959; 485p. [Google Scholar]
- Toschi, A. Mammalia: Lagomorpha, Rodentia, Carnivora, Artiodactyla, Cetacea; Edizioni Calderini: Bologna, Italy, 1965; 647p. [Google Scholar]
- Chaline, J.; Baudvin, H.; Jammot, D.; Saint-Girons, M.C. Les Proies des Rapaces. Petits Mammiferes et leur Environnement; Doin: Paris, France, 1974; 141p. [Google Scholar]
- Lawrence, M.J.; Brown, R.W. Mammals of Britain. Their Tracks, Trails and Signs; Blandford Press: Dorset, UK, 1974; 296p. [Google Scholar]
- Niethammer, J.; Krapp, F. Handbuch der Saugetiere Europas—Band 1, Nagetiere 1; Akademische Verlagsgesellschaft: Wiesbaden, Germany, 1977; 476p. [Google Scholar]
- Niethammer, J.; Krapp, F. Handbuch der Saugetiere Europas—Band 2/1, Nagetiere 2; Akademische Verlagsgesellschaft: Wiesbaden, Germany, 1982; 649p. [Google Scholar]
- Niethammer, J.; Krapp, F. Handbuch der Saugetiere Europas—Band 3/1, Insektenfresser—Herrentiere; AULA—Verlag: Wiesbaden, Germany, 1990; 523p. [Google Scholar]
- Krystufek, B.; Vohralik, V. Mammals of Turkey and Cyprus. Rodentia I: Sciuridae, Dipodidae, Gliridae, Arvicolinae; University of Primorska: Koper, Slovenia, 2005; 292p. [Google Scholar]
- Krystufek, B.; Vohralik, V. Mammals of Turkey and Cyprus. Rodentia II: Cricetinae, Muridae, Spalacidae, Calomyscidae, Capromyidae, Hystricidae, Castoridae; University of Primorska: Koper, Slovenia, 2009; 372p. [Google Scholar]
- Ter Braak, C.J.F.; Šmilauer, P. Canoco Reference Manual and User’s Guide: Software for Ordination, Version 5.0; Microcomputer Power: Ithaca, NY, USA, 2012; p. 496. [Google Scholar]
- Šmilauer, P.; Lepš, J. Multivariate Analysis of Ecological Data Using CANOCO 5; Cambridge University Press: Cambridge, UK, 2014; 362p. [Google Scholar]
- Acevedo, P.; Ruiz-Fons, F.; Estrada, R.; Marquez, A.L.; Miranda, M.A.; Gortazar, C.; Lucientes, J. A broad assessment of factors determining Culicoides imicola abundance: Modelling the present and forecasting its Future in climate change scenarios. PLoS ONE 2010, 5, e14236. [Google Scholar] [CrossRef]
- Barbosa, A.M.; Real, R.; Vargas, J.M. Transferability of environmental favourability models in geographic space: The case of the Iberian desman (Galemys pyrenaicus) in Portugal and Spain. Ecol. Model. 2009, 220, 747–754. [Google Scholar] [CrossRef]
- Bakaloudis, D.E.; Bontzorlos, V.A.; Kotsonas, E. Wildlife mortality on roads crossing a protected area: The case of Dadia-Lefkimi-Soufli National Park in north-eastern Greece. J. Nat. Conserv. 2023, 74, 126443. [Google Scholar] [CrossRef]
- Stojak, J.; Borowik, T.; Górny, M.; McDevitt, A.D.; Wójcik, J.M. Climatic influences on the genetic structure and distribution of the common vole and field vole in Europe. Mammal Res. 2019, 64, 19–29. [Google Scholar] [CrossRef]
- Horníková, M.A.; Marková, S.; Hayley, C.L.; Searle, J.B.; Kotlík, P. A dynamic history of admixture from Mediterranean and Carpathian glacial refugia drives genomic diversity in the bank vole. Ecol. Evol. 2021, 11, 8215–8225. [Google Scholar] [CrossRef]
- Barret, G.W.; Peles, J.D. Landscape Ecology of Small Mammals; Springer: New York, NY, USA, 1999; 372p. [Google Scholar]
- Manning, J.A.; Edge, W.D. Small mammals’ survival and downed wood at multiple scales in managed forests. J. Mammal. 2004, 85, 87–96. [Google Scholar] [CrossRef]
- Macdonald, D.; Barrett, P. Mammals of Europe; Princeton University Press: Princeton, NJ, USA, 1993; 312p. [Google Scholar]
- Libois, R.; Ramalhinho, M.G.; Fons, R. Crocidura suaveolens (Pallas, 1811). In Atlas of European Mammals; Mitchell-Jones, A.J., Amori, G., Bogdanowicz, W., Krystufek, B., Reijnders, P.J.H., Spitzenberger, F., Stubbe, M., Thissen, J.B.M., Vohralik, V., Zima, J., Eds.; Academic Press: London, UK, 1999; pp. 72–73. [Google Scholar]
- Palomo, L.; Krystufek, B.; Amori, G.; Hutterer, R. Crocidura suaveolens. The IUCN Red List of Threatened Species 2016: E.T29656A22296429. Available online: https://www.iucnredlist.org/species/29656/22296429 (accessed on 14 October 2023).
- Biedma, L.; Roman, J.; Godoy, J.A.; Calzada, J. Using owl pellets to infer habitat associations and clarify the regional distribution of a cryptic shrew. J. Zool. 2019, 308, 139–148. [Google Scholar] [CrossRef]
- Heisler, L.M.; Somers, C.M.; Wellicome, T.I.; Poulin, R.G. Landscape-scale features affecting small mammal assemblages on the northern Great Plains of North America. J. Mammal. 2013, 94, 1059–1067. [Google Scholar] [CrossRef]
- Davis, W.B.; Joeris, L. Notes on the Life-History of the Little Short-Tailed Shrew. J. Mammal. 1945, 26, 136–138. [Google Scholar] [CrossRef]
- Granjon, L.; Houssin, C.; Lecompte, E.; Angaya, M.; Cesar, J.; Cornette, R.; Dobigny, G.; Denys, C. Community ecology of the terrestrial small mammals of Zakouma National Park, Chad. Acta Theriol. 2004, 49, 215–234. [Google Scholar] [CrossRef]
- Sheppe, W.; Osborne, T. Patterns of use of a flood plain by Zambian mammals. Ecol. Monogr. 1971, 41, 179–205. [Google Scholar] [CrossRef]
- Mlyashimbi, E.C.M.; Broecke, B.V.; Mariën, J.; Kimaro, D.N.; Tarimo, A.J.P.; Machang’u, R.S.; Isabirye, M.; Makundi, R.H.; Massawe, A.W.; Hieronimo, P.; et al. Soil type influences population dynamics and survival of the Multimammate rat (Mastomys natalensis) in semi-arid areas in Tanzania. Crop Prot. 2019, 124, 104829. [Google Scholar] [CrossRef]
- Hillel, D. Encyclopedia of Soils in the Environment, 1st ed.; Elsiever Academic Press: Amsterdam, The Netherlands, 2005; 2200p. [Google Scholar]
- Libois, R.; Fons, R. Suncus etruscus (Savi, 1822). In Atlas of European Mammals; Mitchell-Jones, A.J., Amori, G., Bogdanowicz, W., Krystufek, B., Reijnders, P.J.H., Spitzenberger, F., Stubbe, M., Thissen, J.B.M., Vohralik, V., Zima, J., Eds.; Academic Press: London, UK, 1999; pp. 76–77. [Google Scholar]
- Spitzenberger, F. Suncus etruscus—Etruskerspitzmaus (Savi, 1822). In Handbuch der Saugetiere Europas. Band 3/I. Insectivora, Primates; Niethammer, J., Krapp, F., Eds.; AULA—Verlag: Wiesbaden, Germany, 1990; pp. 375–392. [Google Scholar]
- Bontzorlos, V.; Peris, S.J.; Vlachos, C.G.; Bakaloudis, D.E. New data on the distribution of three Insectivora species in Thessaly, central Greece. In Proceedings of the VIII Jornadas de la SECEM—Congress of the Spanish Society of Mammalogists, Huelva, Spain, 5–8 December 2007. [Google Scholar]
- Blanco, J.C. (Ed.) Mamíferos de España I. Insectívoros, Quirópteros, Primates y Carnívoros de la Península Ibérica, Baleares y Canarias; Planeta S.A.: Barcelona, Spain, 1998; p. 460. [Google Scholar]
- Lopez-Fuster, M.J. Suncus etruscus (Savi, 1822). In Atlas de los Mamíferos Terrestres de España; Palomo, L.J., Gisbert, J., Eds.; Ministerio de Medio Ambiente, Dirección General de la Conservación de la Naturaleza: Madrid, Spain, 2006; pp. 118–121. [Google Scholar]
- Aulagnier, S.; Hutterer, R.; Jenkins, P.; Bukhnikashvili, A.; Kryštufek, B.; Kock, D. Suncus etruscus. The IUCN Red List of Threatened Species 2017: E.T90389138A22288134. Available online: https://www.iucnredlist.org/species/90389138/22288134 (accessed on 15 October 2023).
- Barti, L. Unusual occurrence of the bicoloured white-toothed shrew (Crocidura leucodon, Soricomorpha: Soricidae) in a mature beech forest in the ciomad-puturosu mountains (Eastern Carpathians, Central Romania). Acta Siculica 2011, 2011, 119–124. [Google Scholar]
- Krystufek, B. Unusual occurrence of the Bicoloured White-toothed Shrew (Crocidura leucodon): Perspective from Slovenia (Soricomorpha: Soricidae). Lynx 2009, 40, 133–134. [Google Scholar]
- Leso, P.; Lesova, A.; Kropil, R. Unusual occurrence of the Bicoloured White-toothed Shrew (Crocidura leucodon) in a fir-beech forest in central Slovakia (Soricomorpha: Soricidae). Lynx 2008, 39, 191–194. [Google Scholar]
- Krapp, F. Crocidura leucodon (Hermann, 1780). In Atlas of European Mammals; Mitchell-Jones, A.J., Amori, G., Bogdanowicz, W., Krystufek, B., Reijnders, P.J.H., Spitzenberger, F., Stubbe, M., Thissen, J.B.M., Vohralik, V., Zima, J., Eds.; Academic Press: London, UK, 1999; pp. 64–65. [Google Scholar]



| Predictor Variables | Composition |
|---|---|
| Heavily Arable Land | Cereal Crops (wheat, barley, oat) Industrial Cultivations (cotton, tobacco, sugar beets) |
| Medium/Light Arable Land | Melon fields, Vegetables |
| Non Arable Land | Tree Cultivations, Vineyards |
| Other Land Uses | Fallow Land, Hills, Grassland, Pastures, Urban areas |
| Heavily Irrigated Land | Industrial Cultivations, Melon Fields, Vegetables |
| Medium/Lightly Irrigated Land | Cereals, Tree Cultivations, Vineyards |
| Non-Irrigated Land | remaining area after extracting the irrigated areas from total agricultural land |
| Alfisol Soil Type | - |
| Entisol Soil Type | - |
| Inceptisol Soil Type | - |
| Molllisol Soil Type | - |
| Vertisol Soil Type | - |
| Light Soils | Sandy Clay granulometric texture in the first 25 cm from soil surface/large particles and pore spaces |
| Heavy Soils | Argillaceous Clay granulometric texture in the first 25 cm from soil surface/fine clay particles difficult to manage but fertile when treated |
| River Length | linear measurement of rivers within each plot |
| Road Length | linear measurement of roads within each plot |
| Principal Component Analysis (PCA) | ||||
|---|---|---|---|---|
| Axes | 1 | 2 | 3 | 4 |
| Eigenvalues | 0.5313 | 0.2125 | 0.1044 | 0.0470 |
| Cumulative percentage variance of species data | 53.13 | 74.37 | 84.82 | 89.51 |
| Redundancy Analysis (RDA) | ||||
| Axes | 1 | 2 | 3 | 4 |
| Eigenvalues | 0.2299 | 0.0938 | 0.0397 | 0.0068 |
| Explained variation (cumulative) | 22.99 | 32.37 | 36.34 | 37.02 |
| Pseudo-canonical correlation | 0.6666 | 0.6582 | 0.6141 | 0.4943 |
| Explained fitted variation (cumulative) | 60.30 | 84.88 | 95.29 | 97.07 |
| Predictor Variable | Explains % | Contribution 100% | Pseudo-F | p Values |
|---|---|---|---|---|
| Non-irrigated Land | 11.4 | 30 | 15.7 | 0.0099 |
| Other Land | 4.2 | 10.9 | 6.0 | 0.0099 |
| Vertisol Soil | 4.7 | 12.4 | 7.1 | 0.0099 |
| Heavy Soil | 4.4 | 11.4 | 6.9 | 0.0099 |
| Heavy Arable Land | 2.1 | 5.4 | 3.3 | 0.0297 |
| Light Soil | 1.2 | 3.2 | 2.0 | 0.0351 |
| Entisol | 0.3 | 0.8 | 0.5 | 0.0459 |
| Model Selection | GLM Results | |||||
|---|---|---|---|---|---|---|
| R2 (%) | AIC | b0 + b1X | b0 + b1X + b2X2 | F | p | |
| Heavy Arable Land | ||||||
| Crocidura leucodon | 5.8 | 530.73 | √ | 3.7 | * | |
| Crocidura gueldenstaedtii | 7.9 | 182.27 | √ | 10.5 | ** | |
| Suncus etruscus | 2.8 | 637.42 | √ | 3.5 | 0.06423 | |
| Other Land | ||||||
| Crocidura leucodon | 5.5 | 530.38 | √ | 3.5 | * | |
| Crocidura gueldenstaedtii | 4.3 | 177.50 | √ | 5.5 | * | |
| Suncus etruscus | 1.9 | 636.26 | √ | 2.3 | 0.13039 | |
| Non-irrigated Land | ||||||
| Crocidura leucodon | 4.7 | 529.25 | √ | 3.0 | 0.05608 | |
| Suncus etruscus | 7.5 | 643.60 | √ | 9.9 | * | |
| Heavy Soil | ||||||
| Crocidura gueldenstaedtii | 17.9 | 194.38 | √ | 13.2 | ***** | |
| Suncus etruscus | 6.4 | 640.03 | √ | 4.2 | * | |
| Light Soil | ||||||
| Crocidura gueldenstaedtii | 19.8 | 197.34 | √ | 15.0 | ***** | |
| Suncus etruscus | 7.6 | 641.58 | √ | 5.0 | ** | |
| Vertisol | ||||||
| Crocidura gueldenstaedtii | 20.4 | 200.32 | √ | 31.2 | ***** | |
| Suncus etruscus | 18.5 | 659.30 | √ | 27.7 | ***** | |
| Entisol | ||||||
| Crocidura leucodon | 5.3 | 530.08 | √ | 3.4 | * | |
| Crocidura gueldenstaedtii | 9.8 | 182.71 | √ | 6.6 | ** | |
| Suncus etruscus | 7.7 | 643.80 | √ | 10.1 | ** | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bontzorlos, V. Shrew Communities in Mediterranean Agro-Ecosystems of Central Greece: Associations with Crop Types, Land Uses, and Soil Parameters. Life 2023, 13, 2248. https://doi.org/10.3390/life13122248
Bontzorlos V. Shrew Communities in Mediterranean Agro-Ecosystems of Central Greece: Associations with Crop Types, Land Uses, and Soil Parameters. Life. 2023; 13(12):2248. https://doi.org/10.3390/life13122248
Chicago/Turabian StyleBontzorlos, Vasileios. 2023. "Shrew Communities in Mediterranean Agro-Ecosystems of Central Greece: Associations with Crop Types, Land Uses, and Soil Parameters" Life 13, no. 12: 2248. https://doi.org/10.3390/life13122248
APA StyleBontzorlos, V. (2023). Shrew Communities in Mediterranean Agro-Ecosystems of Central Greece: Associations with Crop Types, Land Uses, and Soil Parameters. Life, 13(12), 2248. https://doi.org/10.3390/life13122248






