Influence of Metal Ions on Model Protoamphiphilic Vesicular Systems: Insights from Laboratory and Analogue Studies
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Vesicle Preparation
2.2.2. Setting Up the Metal Ion Stability Experiments
2.2.3. Microscopic Analysis
2.2.4. Vesicle Re-Formation by the Addition of Magnesium Chelators
2.2.5. Vesicle Formation by NOG-Based Amphiphile Systems in Hot Spring Water Samples
- I.
- The collection of water samples from early Earth analogue hot spring sites: Water samples were directly collected from two hot spring pools at Hells Gate Geothermal Reserve, Tikitere (abbreviated as TIKB and TIKC, respectively). These samples were filtered with a 0.22 µm polyethersulfone membrane filter (rinsed with 20 mL of sample) and stored in acid-washed high-density polyethylene bottles until further use. Samples were untreated and unpreserved in the field.
- II.
- Geochemical analysis of hot spring water samples: The collected water samples were analysed for the identification and quantification of different ions present in them. These measurements were performed by following standard protocols as reported earlier [38]. Briefly, the concentrations of the major cations (Na+, K+, Ca2+, Mg2+, and NH4+) and anions (Cl−, F−,SO42−, and NO₃−) were measured using ion chromatography instrument Compact IC plus 882 (Metrohm, Herisau, Switzerland). Moreover, the alkalinity of the samples was measured using an auto-titrator Eco Titrator (Metrohm, Switzerland). The dissolved Silica concentrations were measured by conventional molybdenum-blue method using double beam UV-VIS Spectrophotometer (M.D.T. INTERNATIONAL, Ambala, Haryana, India). The accuracy and precision of these analyses were monitored regularly, which had an average value of ±4%. The net inorganic charge balance (NICB) for these samples was around 1, which indicates good data quality.
- III.
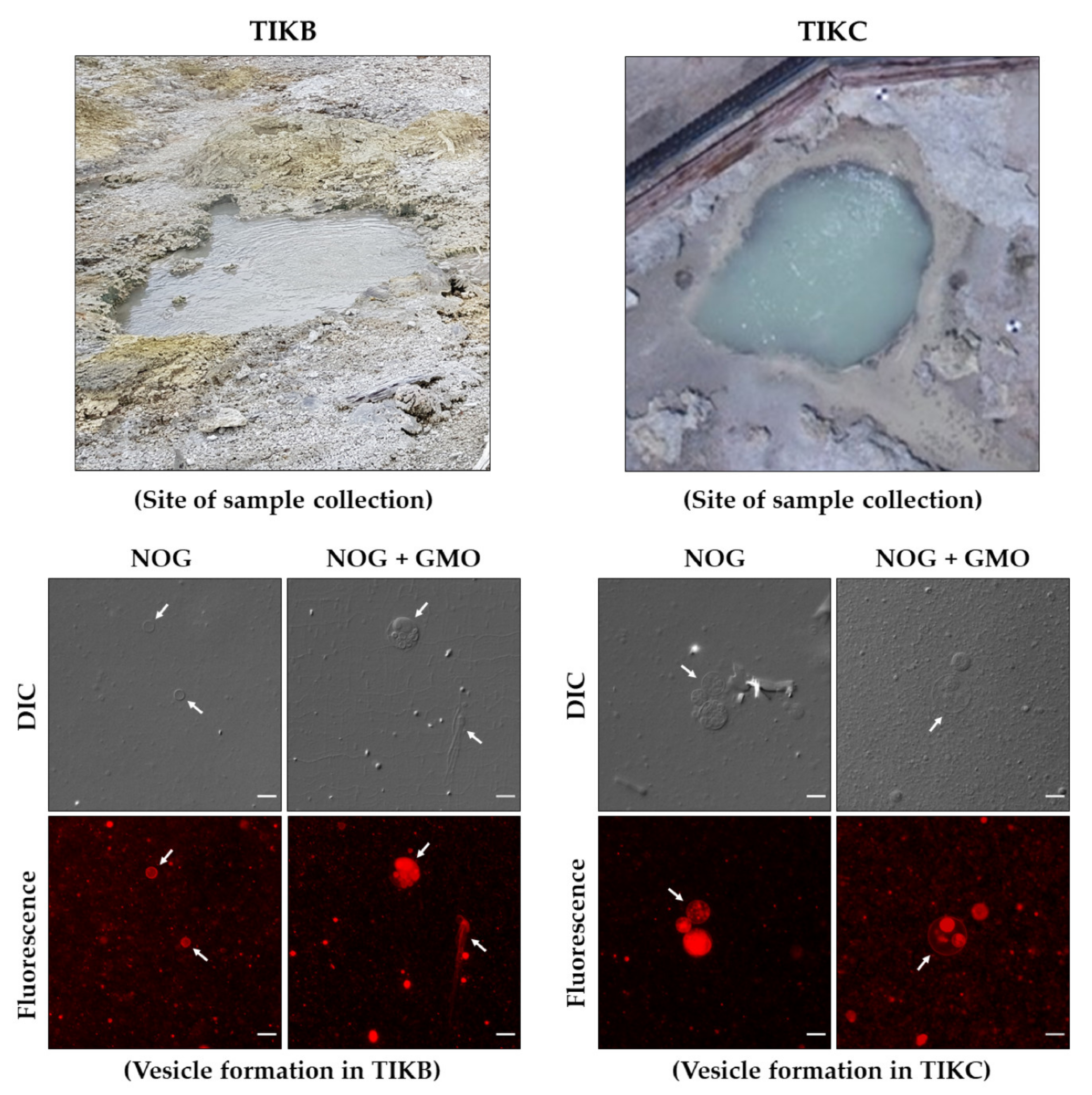
- Vesicle formation in hot spring water samples: Dry lipid films of pure NOG and mixed systems containing NOG + surfactant (GMO or OOH) in 2:1 ratio were prepared as previously described. These lipid films were hydrated with 50 µL of hot spring water sample (TIKB/TIKC) to get a total amphiphile concentration of 6 mM. The solution was further incubated at 60 °C for 1 h with constant shaking at 500 rpm, with intermittent mixing by vortexing and pipetting to facilitate the vesicle formation process. The initial pH of the hot spring water samples was 7–7.5, which decreased to 5–5.5 after the addition of the amphiphiles, likely because of the acidic nature of NOG. The formation of vesicles was checked by both DIC and epifluorescence microscopy as mentioned above.
3. Results
3.1. Effect of Mg2+ on the Stability of Pure NOG and NOG + GMO Mixed Systems
3.2. Effect of Na+ on the Stability of Pure NOG and NOG + GMO Mixed Systems
3.3. NOG-Based Amphiphile Systems Readily Form Vesicles in Hot Spring Water Samples
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, L.; Zou, Y.; Bhattacharya, A.; Zhang, D.; Lang, S.Q.; Houk, K.N.; Devaraj, N.K. Enzyme-free synthesis of natural phospholipids in water. Nat. Chem. 2020, 12, 1029–1034. [Google Scholar] [CrossRef]
- Gibard, C.; Bhowmik, S.; Karki, M.; Kim, E.K.; Krishnamurthy, R. Phosphorylation, oligomerization and self-assembly in water under potential prebiotic conditions. Nat. Chem. 2018, 10, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.; Eichberg, J.; Oró, J. Synthesis of phosphatidylethanolamine under possible primitive earth conditions. J. Mol. Evol. 1987, 25, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, W.R.; Mulvihill, S.J.; Deamer, D.W. Synthesis of phospholipids and membranes in prebiotic conditions. Nature 1977, 266, 78–80. [Google Scholar] [CrossRef] [PubMed]
- Bonfio, C.; Caumes, C.; Duffy, C.D.; Patel, B.H.; Percivalle, C.; Tsanakopoulou, M.; Sutherland, J.D. Length-Selective Synthesis of Acylglycerol-Phosphates through Energy-Dissipative Cycling. J. Am. Chem. Soc. 2019, 141, 3934–3939. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deamer, D. The Role of Lipid Membranes in Life’s Origin. Life 2017, 7, 5. [Google Scholar] [CrossRef]
- Mansy, S.S. Model protocells from single-chain lipids. Int. J. Mol. Sci. 2009, 10, 835–843. [Google Scholar] [CrossRef] [Green Version]
- Mansy, S.S. Membrane transport in primitive cells. Cold Spring Harb. Perspect. Biol. 2010, 2, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Sarkar, S.; Das, S.; Dagar, S.; Joshi, M.P.; Mungi, C.V.; Sawant, A.A.; Patki, G.M.; Rajamani, S. Prebiological Membranes and Their Role in the Emergence of Early Cellular Life. J. Membr. Biol. 2020, 253, 589–608. [Google Scholar] [CrossRef]
- Lawless, J.G.; Yuen, G.U. Quantification of monocarboxylic acids in the Murchison carbonaceous meteorite. Nature 1979, 282, 396–398. [Google Scholar] [CrossRef]
- McCollom, T.M.; Ritter, G.; Simoneit, B.R. Lipid synthesis under hydrothermal conditions by Fischer-Tropsch-type reactions. Orig. Life Evol. Biosph. 1999, 29, 153–166. [Google Scholar] [CrossRef]
- Joshi, M.P.; Sawant, A.A.; Rajamani, S. Spontaneous emergence of membrane-forming protoamphiphiles from a lipid–amino acid mixture under wet–dry cycles. Chem. Sci. 2021, 12, 2970–2978. [Google Scholar] [CrossRef] [PubMed]
- Mansy, S.S.; Szostak, J.W. Thermostability of model protocell membranes. Proc. Natl. Acad. Sci. USA 2008, 105, 13351–13355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rubio-Sánchez, R.; O’Flaherty, D.K.; Wang, A.; Coscia, F.; Petris, G.; Di Michele, L.; Cicuta, P.; Bonfio, C. Thermally Driven Membrane Phase Transitions Enable Content Reshuffling in Primitive Cells. J. Am. Chem. Soc. 2021, 143, 16589–16598. [Google Scholar] [CrossRef]
- Maurer, S. The Impact of Salts on Single Chain Amphiphile Membranes and Implications for the Location of the Origin of Life. Life 2017, 7, 44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walde, P.; Namani, T.; Morigaki, K.; Hauser, H.; Namani, T.; Morigaki, K.; Hauser, H. Formation and Properties of Fatty Acid Vesicles (Liposomes). In Liposome Technology; CRC Press: Boca Raton, FL, USA, 2018; pp. 23–42. [Google Scholar]
- Bonfio, C.; Godino, E.; Corsini, M.; Fabrizi de Biani, F.; Guella, G.; Mansy, S.S. Prebiotic iron–sulfur peptide catalysts generate a pH gradient across model membranes of late protocells. Nat. Catal. 2018, 1, 616–623. [Google Scholar] [CrossRef]
- Muchowska, K.B.; Varma, S.J.; Chevallot-Beroux, E.; Lethuillier-Karl, L.; Li, G.; Moran, J. Metals promote sequences of the reverse Krebs cycle. Nat. Ecol. Evol. 2017, 1, 1716–1721. [Google Scholar] [CrossRef]
- Bowman, J.C.; Lenz, T.K.; Hud, N.V.; Williams, L.D. Cations in charge: Magnesium ions in RNA folding and catalysis. Curr. Opin. Struct. Biol. 2012, 22, 262–272. [Google Scholar] [CrossRef]
- Inoue, A.; Takagi, Y.; Taira, K. Importance of magnesium ions in the mechanism of catalysis by a hammerhead ribozyme: Strictly linear relationship between the ribozyme activity and the concentration of magnesium ions. Magnes. Res. 2003, 16, 210–217. [Google Scholar]
- Attwater, J.; Wochner, A.; Holliger, P. In-ice evolution of RNA polymerase ribozyme activity. Nat. Chem. 2013, 5, 1011. [Google Scholar] [CrossRef] [Green Version]
- Horning, D.P.; Joyce, G.F. Amplification of RNA by an RNA polymerase ribozyme. Proc. Natl. Acad. Sci. USA 2016, 113, 9786–9791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bapat, N.V.; Rajamani, S. Templated replication (or lack thereof) under prebiotically pertinent conditions. Sci. Rep. 2018, 8, 15032. [Google Scholar] [CrossRef] [PubMed]
- Bowler, F.R.; Chan, C.K.W.; Duffy, C.D.; Gerland, B.; Islam, S.; Powner, M.W.; Sutherland, J.D.; Xu, J. Prebiotically plausible oligoribonucleotide ligation facilitated by chemoselective acetylation. Nat. Chem. 2013, 5, 383–389. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baross, J.A.; Hoffman, S.E. Submarine hydrothermal vents and associated gradient environments as sites for the origin and evolution of life. Orig. Life Evol. Biosph. 1985, 15, 327–345. [Google Scholar] [CrossRef]
- Damer, B.; Deamer, D. The hot spring hypothesis for an origin of life. Astrobiology 2020, 20, 429–452. [Google Scholar] [CrossRef] [Green Version]
- Milshteyn, D.; Damer, B.; Havig, J.; Deamer, D. Amphiphilic Compounds Assemble into Membranous Vesicles in Hydrothermal Hot Spring Water but Not in Seawater. Life 2018, 8, 11. [Google Scholar] [CrossRef] [Green Version]
- Monnard, P.-A.; Apel, C.L.; Kanavarioti, A.; Deamer, D.W. Influence of Ionic Inorganic Solutes on Self-Assembly and Polymerization Processes Related to Early Forms of Life: Implications for a Prebiotic Aqueous Medium. Astrobiology 2002, 2, 139–152. [Google Scholar] [CrossRef]
- Deamer, D.W.; Georgiou, C.D. Hydrothermal Conditions and the Origin of Cellular Life. Astrobiology 2015, 15, 1091–1095. [Google Scholar] [CrossRef] [PubMed]
- Joshi, M.P.; Samanta, A.; Tripathy, G.R.; Rajamani, S. Formation and Stability of Prebiotically Relevant Vesicular Systems in Terrestrial Geothermal Environments. Life 2017, 7, 51. [Google Scholar] [CrossRef] [Green Version]
- Deamer, D. Where Did Life Begin? Testing Ideas in Prebiotic Analogue Conditions. Life 2021, 11, 134. [Google Scholar] [CrossRef] [PubMed]
- Maurer, S.E.; Tølbøl Sørensen, K.; Iqbal, Z.; Nicholas, J.; Quirion, K.; Gioia, M.; Monnard, P.A.; Hanczyc, M.M. Vesicle Self-Assembly of Monoalkyl Amphiphiles under the Effects of High Ionic Strength, Extreme pH, and High Temperature Environments. Langmuir 2018, 34, 15560–15568. [Google Scholar] [CrossRef]
- Namani, T.; Deamer, D.W. Stability of Model Membranes in Extreme Environments. Orig. Life Evol. Biosph. 2008, 38, 329–341. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, S.; Dagar, S.; Verma, A.; Rajamani, S. Compositional heterogeneity confers selective advantage to model protocellular membranes during the origins of cellular life. Sci. Rep. 2020, 10, 1–11. [Google Scholar]
- Dalai, P.; Ustriyana, P.; Sahai, N. Aqueous magnesium as an environmental selection pressure in the evolution of phospholipid membranes on early earth. Geochim. Cosmochim. Acta 2018, 223, 216–228. [Google Scholar] [CrossRef]
- Jin, L.; Kamat, N.P.; Jena, S.; Szostak, J.W. Fatty Acid/Phospholipid Blended Membranes: A Potential Intermediate State in Protocellular Evolution. Small 2018, 14. [Google Scholar] [CrossRef]
- Toparlak, Ö.D.; Karki, M.; Egas Ortuno, V.; Krishnamurthy, R.; Mansy, S.S. Cyclophospholipids Increase Protocellular Stability to Metal Ions. Small 2019, 1903381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tripathy, G.R.; Goswami, V.; Singh, S.K.; Chakrapani, G.J. Temporal variations in Sr and 87 Sr/ 86 Sr of the Ganga headwaters: Estimates of dissolved Sr flux to the mainstream. Hydrol. Process. 2010, 24, 1159–1171. [Google Scholar] [CrossRef]
- Szostak, J.W. The eightfold path to non-enzymatic RNA replication. J. Syst. Chem. 2012, 3, 2. [Google Scholar] [CrossRef] [Green Version]
- Davey, S.G. Origin of Life: Cycling citrate sans enzymes. Nat. Rev. Chem. 2017, 1, 1. [Google Scholar] [CrossRef]
- Glusker, J.P. Citrate conformation and chelation: Enzymic implications. Acc. Chem. Res. 2002, 13, 345–352. [Google Scholar] [CrossRef]
- Adamala, K.; Szostak, J.W. Nonenzymatic template-directed RNA synthesis inside model protocells. Science (80-. ) 2013, 342, 1098–1100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dobson, M. Facies Mapping and Analysis of Diverse Hydrothermal Sedimentary Facies and Siliceous Spicular Sinter at Hell’s Gate, Tikitere Geothermal Field, Taupō Volcanic Zone, New Zealand. Honours Dissertation, University of Auckland, Auckland, New Zealand, 2018. [Google Scholar]
- Bonfio, C.; Russell, D.A.; Green, N.J.; Mariani, A.; Sutherland, J.D. Activation chemistry drives the emergence of functionalised protocells. Chem. Sci. 2020, 11, 10688–10697. [Google Scholar] [CrossRef] [PubMed]
- Izgu, E.C.; Björkbom, A.; Kamat, N.P.; Lelyveld, V.S.; Zhang, W.; Jia, T.Z.; Szostak, J.W. N-Carboxyanhydride-Mediated Fatty Acylation of Amino Acids and Peptides for Functionalization of Protocell Membranes. J. Am. Chem. Soc. 2016, 138, 16669–16676. [Google Scholar] [CrossRef]
- Maurer, S.E.; Nguyen, G. Prebiotic Vesicle Formation and the Necessity of Salts. Orig. Life Evol. Biosph. 2016, 46, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Mulkidjanian, A.Y.; Bychkov, A.Y.; Dibrova, D.V.; Galperin, M.Y.; Koonin, E.V. Origin of first cells at terrestrial, anoxic geothermal fields. Proc. Natl. Acad. Sci. USA 2012, 109, E821–E830. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dagar, S.; Sarkar, S.; Rajamani, S. Geochemical influences on nonenzymatic oligomerization of prebiotically relevant cyclic nucleotides. RNA 2020, 26, 756–769. [Google Scholar] [CrossRef]



| Hot Spring Sample | Major Cations | TZ⁺ | Major Anions | TZ− | TZ⁺/TZ− | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NH₄⁺ | Na⁺ | K⁺ | Ca2⁺ | Mg2⁺ | NO₃− | F− | Cl− | HCO₃− | SO₄2− | SiO₂ | ||||
| All Values in µM | µE | All Values in µM | µE | µM | ||||||||||
| TIKB † | 2756 | 743 | 285 | 98 | 31 | 4043 | 24 | 45 | 122 | 29 | 2550 | 5319 | 1128 | 0.76 |
| TIKC † | 3296 | 1007 | 301 | 167 | 27 | 4991 | 14 | 48 | 150 | 1458 | 2312 | 6294 | 1019 | 0.79 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joshi, M.P.; Steller, L.; Van Kranendonk, M.J.; Rajamani, S. Influence of Metal Ions on Model Protoamphiphilic Vesicular Systems: Insights from Laboratory and Analogue Studies. Life 2021, 11, 1413. https://doi.org/10.3390/life11121413
Joshi MP, Steller L, Van Kranendonk MJ, Rajamani S. Influence of Metal Ions on Model Protoamphiphilic Vesicular Systems: Insights from Laboratory and Analogue Studies. Life. 2021; 11(12):1413. https://doi.org/10.3390/life11121413
Chicago/Turabian StyleJoshi, Manesh Prakash, Luke Steller, Martin J. Van Kranendonk, and Sudha Rajamani. 2021. "Influence of Metal Ions on Model Protoamphiphilic Vesicular Systems: Insights from Laboratory and Analogue Studies" Life 11, no. 12: 1413. https://doi.org/10.3390/life11121413
APA StyleJoshi, M. P., Steller, L., Van Kranendonk, M. J., & Rajamani, S. (2021). Influence of Metal Ions on Model Protoamphiphilic Vesicular Systems: Insights from Laboratory and Analogue Studies. Life, 11(12), 1413. https://doi.org/10.3390/life11121413







