Abstract
Microbes can mediate the precipitation of primary dolomite under surface conditions. Meanwhile, primary dolomite mediated by microbes often contains more Fe2+ than standard dolomite in modern microbial culture experiments. Ferroan dolomite and ankerite have been regarded as secondary products. This paper reviews the process and possible mechanisms of microbial mediated precipitation of primary ferroan dolomite and/or ankerite. In the microbial geochemical Fe cycle, many dissimilatory iron-reducing bacteria (DIRB), sulfate-reducing bacteria (SRB), and methanogens can reduce Fe3+ to Fe2+, while SRB and methanogens can also promote the precipitation of primary dolomite. There are an oxygen respiration zone (ORZ), an iron reduction zone (IRZ), a sulfate reduction zone (SRZ), and a methanogenesis zone (MZ) from top to bottom in the muddy sediment diagenesis zone. DIRB in IRZ provide the lower section with Fe2+, which composes many enzymes and proteins to participate in metabolic processes of SRB and methanogens. Lastly, heterogeneous nucleation of ferroan dolomite on extracellular polymeric substances (EPS) and cell surfaces is mediated by SRB and methanogens. Exploring the origin of microbial ferroan dolomite may help to solve the “dolomite problem”.
1. Introduction
Microorganisms represented by bacteria are the most widely distributed life forms on Earth, and various types of inorganic minerals are the basis of the inorganic world. Microorganisms and minerals are closely involved on multiple levels: microbial formation of minerals, microbial dissolution of minerals, and redox between microorganisms and minerals with polyvalent metallic elements [1,2,3]. Among these interactions, redox is considered the key pathway influencing elemental geochemical cycles on the Earth’s surface [2,4,5]. On the one hand, chemoautotroph microbes obtain electron energy though the oxidation of reduced minerals, such as sulfide minerals and divalent iron minerals. On the other hand, oxidized minerals act as terminal electron receptors for microbes in their own respiration [1]. For the microbial formation of minerals, researchers have found many examples in recent years. Microbial activity was revealed to have a catalytic effect on the clay mineral reaction of montmorillonite-illite [6]. In subsequent studies, other clay minerals and microbes such as bacteria and archaea were studied under medium-high temperature. Researchers simulated a complex diagenetic history, and analyzed microbial activity in the diagenetic process for the control of clay mineral phase transformations, the preservation mechanisms of organic matter in sedimentary rocks’ clay minerals, and the potential for hydrocarbon generation in later stages [7].
In the microbial geochemical Fe cycle, iron is one of the essential nutrients that is often limited by the formation of insoluble Fe3+ under oxidizing conditions [8]. It has been shown in numerous experiments that iron oxides such as goethite and haematite undergo dissimilatory reduction to form secondary minerals such as magnetite and siderite with S. oneidensis MR-1 [9,10,11]. Many studies demonstrate that the bioprecipitation mechanism plays an important role in the banded iron formation (BIF) [12,13], iron-manganese nodules [14,15,16], and cold seep carbonate [17,18,19,20,21,22,23,24,25,26,27]. In addition, carbonate rock is the most common microbialite, and its genesis [28,29,30,31,32] and macro-micro fabrics are greatly affected by microorganisms [33]. Ferrous carbonate minerals used to be considered of a secondary origin mostly [34,35], but now, there are more and more experiments indicating that ferrous carbonate minerals, such as siderite, precipitate under surface conditions [36]. Additionally, there are some Fe2+ in the more famous microbial dolomite [37,38], which is one of the main directions for studying the origin of primary dolomite [1,28,29,30,31,32,36,37,38,39,40,41,42,43,44].
This article points out that the primary dolomite obtained in microbial culture experiments contains an indeterminate amount of iron. Authors summarize the research of the microbial geochemical Fe cycle under a surface condition and solve the problem how sulfate-reducing bacteria (SRB) and methanogens get Fe2+. The origin mechanism for primary dolomite is referenced and discussed, especially for ferroan dolomite mediated by microbes.
2. Microbial Geochemical Fe Cycle
2.1. Valence Changes of Iron in Different Ecological Niches
Iron, which is the fourth most abundant metal element in the Earth’s crust, is a main redox active element in the environment [8], and is widely involved in a series of geochemical cycles [45]. There are two common redox states for iron in nature: reduced ferrous iron (Fe(II)) and oxidized trivalent iron (Fe(III)). Fe(III) usually forms some insoluble and/or poorly soluble minerals under oxygen conditions, such as goethite, hematite, and fibrite, which are often limited by chemical reactions [1,5,8,9,10,11].
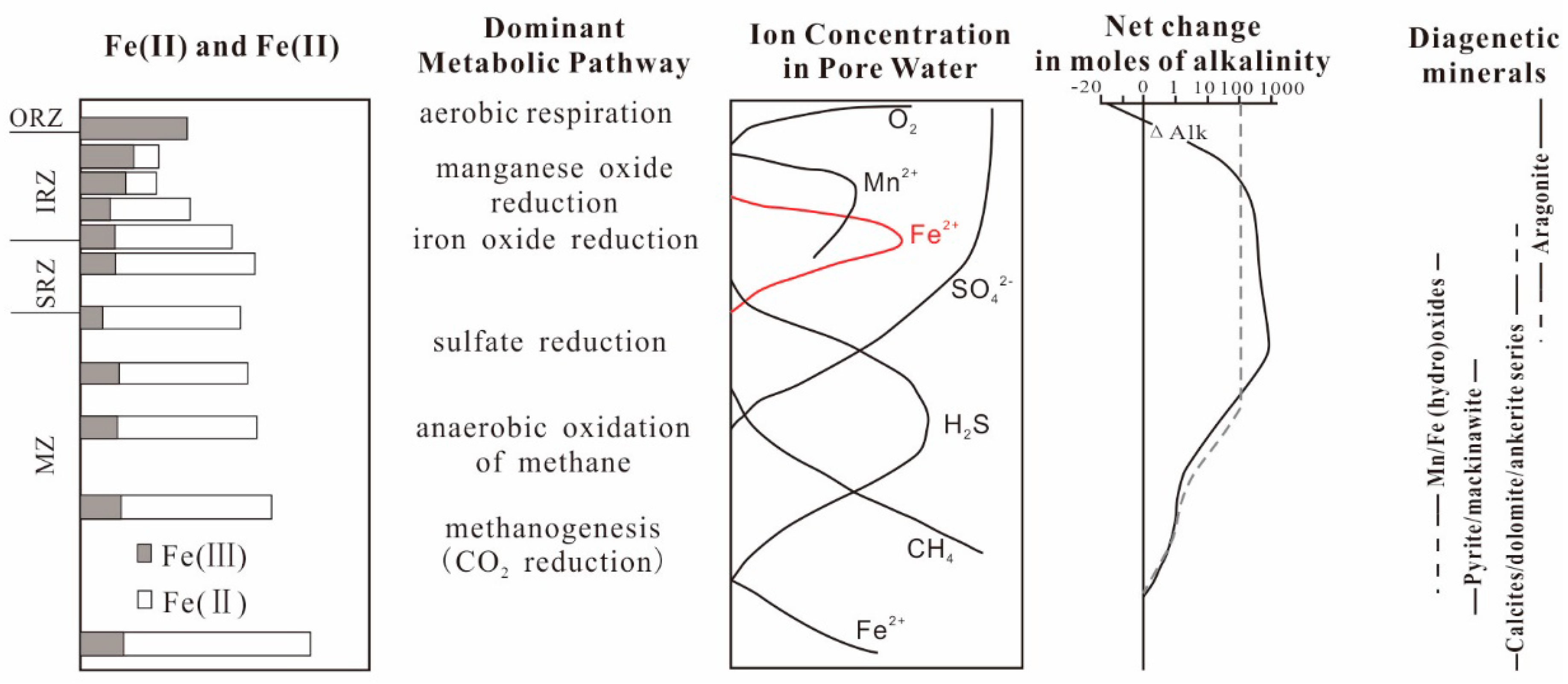
There are various microbial functional groups occupying different ecological niches, which have typical vertical zoning characteristics in the muddy sediment diagenesis zone (Figure 1) and distribute into an oxygen respiration zone (ORZ), an iron reduction zone (IRZ), a sulfate reduction zone (SRZ), and a methanogenesis zone (MZ) from top to bottom [46,47,48]. Additionally, there are certain transition intervals between these zones [48]. Therefore, Fe2+ obtained from reduction of Fe3+ in IRZ will migrate up and down at the same time. Those migrating upward are oxidized by oxygen and nitrate generally to form a new ferric oxide and then precipitate. The downward migration is often reacts with the H2S produced by the SRZ below it and, lastly, forms iron sulfide such as FeS and then is buried [49,50,51]. Moreover, the H2S produced in SRZ may also diffuse upward, directly reacting with the remaining iron oxides to form iron sulfide deposits, such as FeS [52,53].
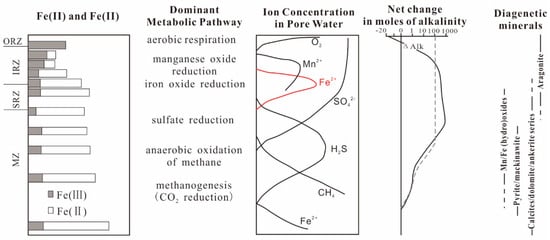
Figure 1.
Schematic diagram of a microbial functional group zoning and Fe(III) mineral distribution [46,57].
However, it is not true that there is no Fe(III) below IRZ [48]. Indirect evidence is that the iron oxides undergo multiple redox reactions before the sediment enters the burial stage [54,55], and direct evidence is that there is an unusual thermophilic bacterium capable of promoting Fe(III) reduction in deep subsurface sediments [56]. In addition, it has been confirmed that the main strains in different depths can reduce Fe3+ directly [57].
2.2. Microbial Fe(III) Respiration
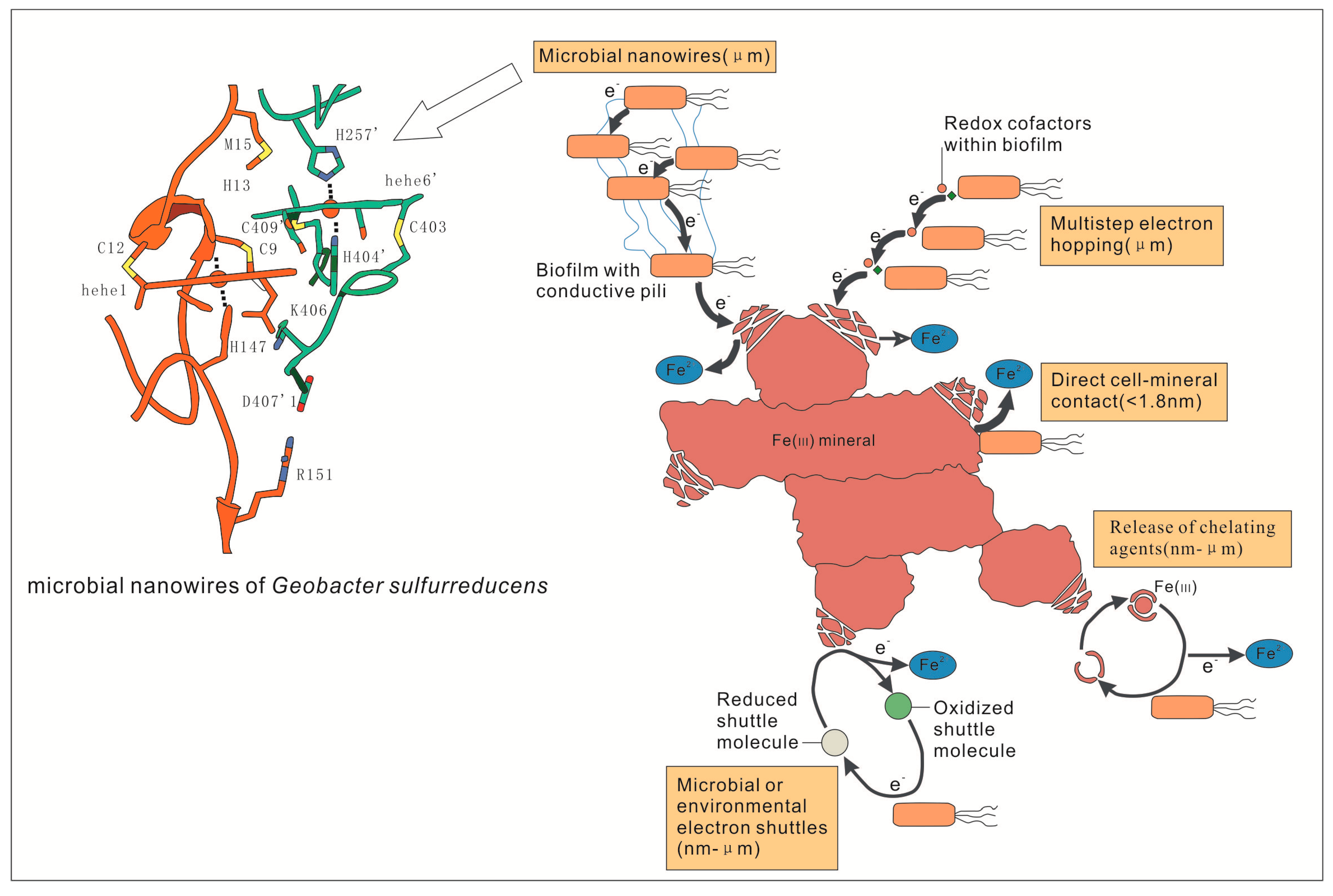
During the Fe cycle, the redox potential of Fe(III)-Fe(II) is located in the redox bonds formed by C, N, O, and S. Fe(III) is insoluble at ambient pH conditions even though it can still transfer electrons from cells to the surface of poorly soluble electron acceptors [11,58,59] (Figure 2). There are five ways to transfer electrons including: (1) increased dissolution of Fe(III) through the secretion and release of iron chelating agents, (2) adhesion between attachments and Fe(III) on the cell surface, (3) direct interaction of iron reductase and cytochrome with iron-containing minerals, (4) transfer of electrons from cells to Fe(III) assisted by electron shuttles, and (5) interactions with iron minerals relying on coenzymes in biofilms [8]. Most organisms can meet basic iron requirements through these five mechanisms [59,60], which are more important for microbes.
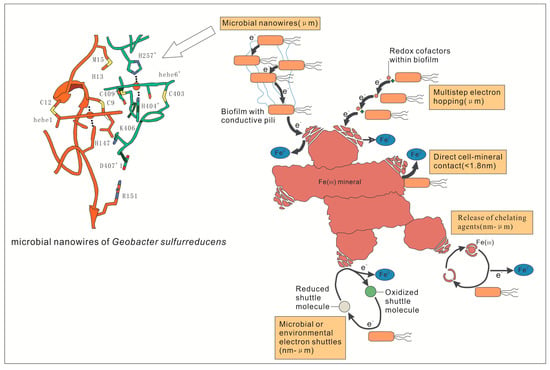
Figure 2.
Mechanisms of electron transfer from microorganisms to Fe(III) minerals [8,72].
Microbes participate in the geochemical Fe cycle involving both oxidation and reduction. Microbial-mediated iron oxidation processes mainly include: O2 oxidation of Fe(II), biomineralization, photochemical processes, and oxidation of nitrates [58]. It used to be thought that the reduction of Fe(III) was indirect (such as by changing Eh or pH), and only a few microorganisms were able to transfer electrons to Fe(III) during its fermentation [61]. However, it is now believed that dissimilatory iron-reducing bacteria (DIRB) can transfer electrons to Fe(III) during Fe(III) respiration [62,63]. DIRB is the most important driving force for the reduction of Fe(II) in IRZ [11,56,64,65,66,67,68,69,70].
In anaerobic environments, certain microbes are capable of utilizing Fe(III) as the electron receptor in microbial respiration, called Fe(III) respiration [61,62,63], which thrives in IRZ. This process is reducing and involves a multi-mechanism synergistic interaction between the Fe(III)-reducing bacteria and some other microbes at the community or group level, which affects Fe(III) reduction [58].
The normal symbiosis of microbes in the Fe cycle includes symbiosis not only between one bacterium and another bacterium but also between bacteria and archaea such as methanogens. The phylogenetic tree constructed by previous researchers shows that the pure strains with iron reduction ability are mainly types of Proteobacteria (α-, β-, γ-, δ-Proteobacteria, etc.), Firmicutes, Acidobacteria, and some archaea [5,71]. These symbiotic microorganisms have different physiological characteristics (psychrophilic, thermophilic, and/or facultative/obligate anaerobic, etc.), and Geobacter and Shewanella are two representative strains among them [1].
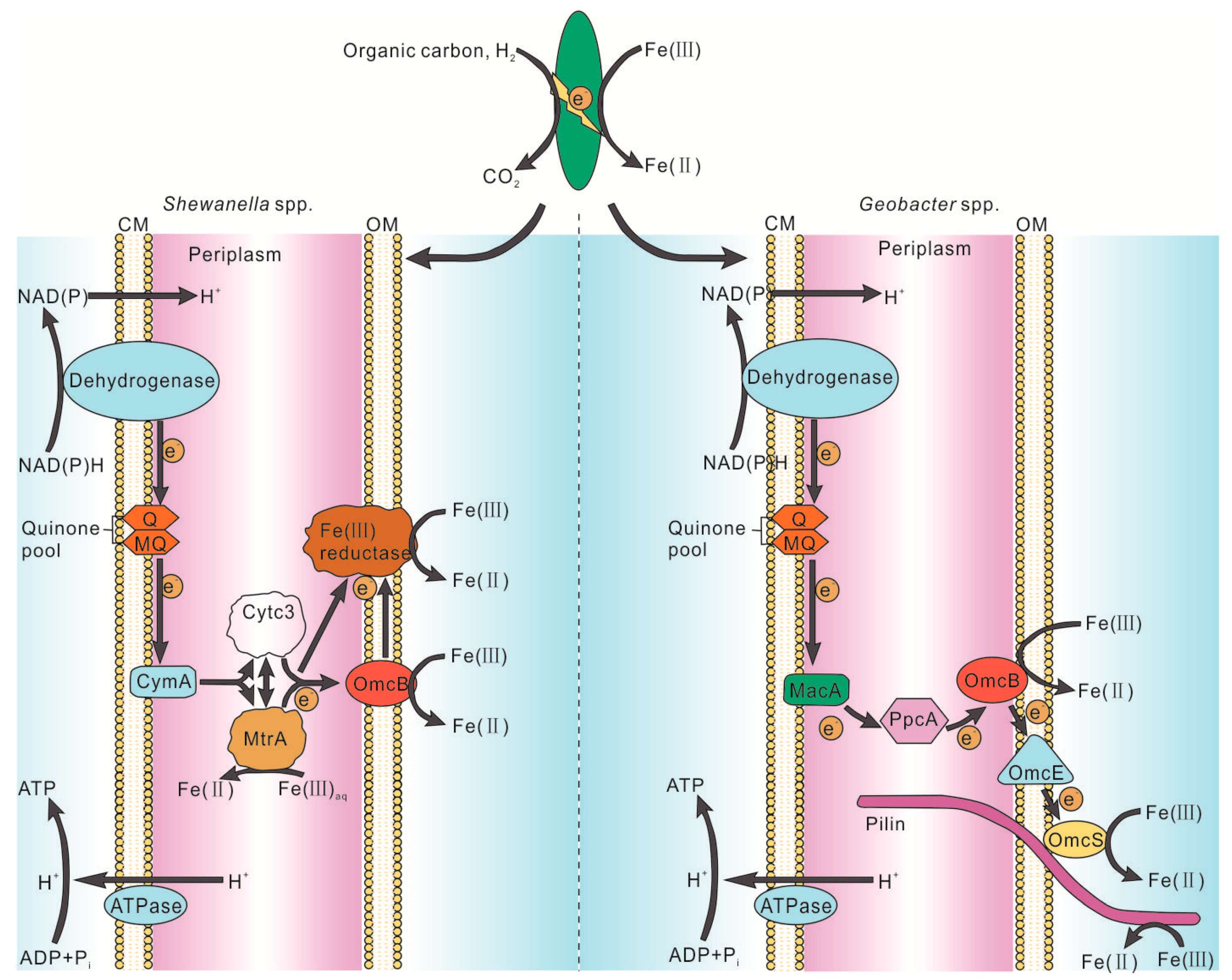
Due to the technically limited current, it is unable to visually observe the molecular motion at the microbial-mineral interface, and to understand the specific details of electron transfer during Fe(III) respiration. According to Figure 2, Fe2+ is obtained by means of direct contact, electron shuttle assist, and/or coenzymes on biofilms [8]. For example, electrons of reductase and/or cytochrome on the biofilms of cells are transferred to Fe(III) by typical DIRB, such as Geobacter and Shewanella (Figure 3). However, Wang et al. used cryo-electron microscopy (cyro-EM) to identify the extracellular Omcs poly filaments of wild-type Geobacter. sulfurreducens, which has a unique spatial characteristic (Figure 2) and is the molecular basis for the long-range electron transport mechanism of microorganisms [72]. This is consistent with the Shewanella oneidensis MR-1 nanowire introduced by Gorby et al. [68]. In addition to these, it has been demonstrated that SRB can reduce a variety of high-valent metal elements, instead of using sulfate as a terminal electron acceptor only [61,73,74,75]. For example, the SRB strain Desulfotomaculum, isolated from a variety of heavy metal contaminated sludge deposits, can take respiration with high valence metal elements as terminal electron acceptors [76]. It was also found that SRB can reduce Fe(III) in clay minerals [57]. However, it is still unclear how the specific process of reduction of Fe(III) was induced by SRB Fe(III) respiration. There may be two mechanisms: (1) indirect reduction, in which H2S, the reduction product, can be used as a reducing agent to reduce and precipitate iron sulfide, and (2) direct enzymatic action, which mainly focuses on the recognition of metal reductase on biofilm [73,74,75,76].
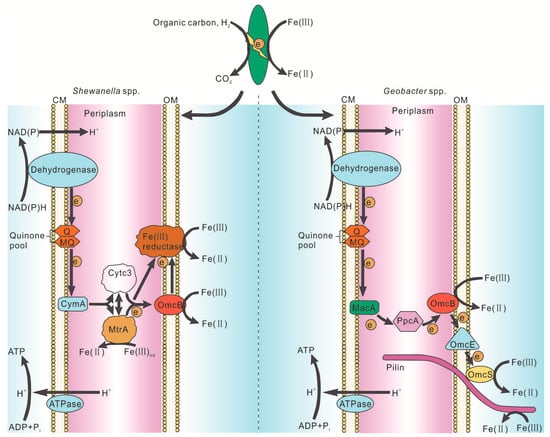
Figure 3.
Physiological model of the biochemistry involved in microbial Fe(III) reduction by Shewanella and Geobacter spp. [5].
3. Discussion: Microbial Mediation of Ferroan Dolomite Precipitation
3.1. Examples of Microbial-Mediated Precipitation of Ferroan Dolomite
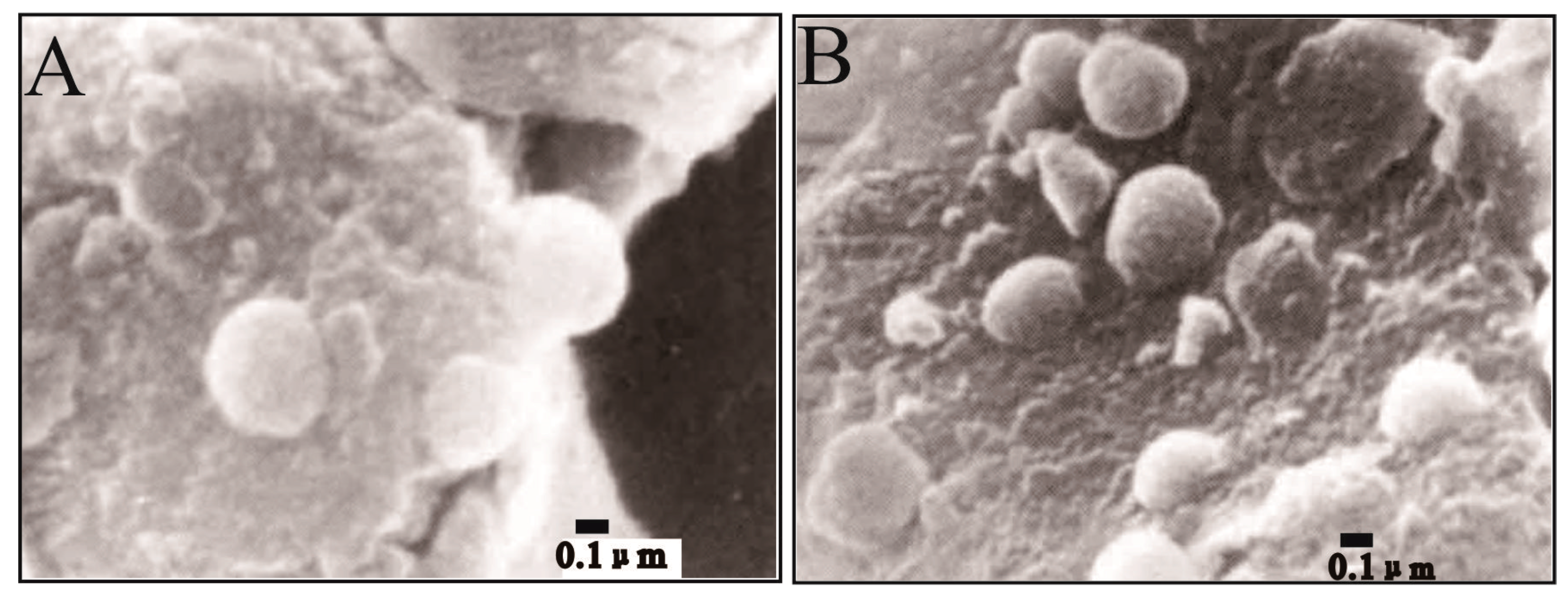
Vasconcelos et al. cultivated Desulfovibrio, which is a group of desulfurizing bacteria in the Lagoa Vermelha lagoon, Brazil, for one year at a constant temperature of −4 °C and precipitated primary dolomite with a high iron content [37]. In the experiment, some precipitation of FeS was observed after one week of cultivation under anaerobic conditions, and then 1 ml of supernatant was added to a 20 mL bottle of a substance-containing medium and a quartz sand substrate that had been previously cleaned with dilute hydrochloric acid to remove all original carbonates. This experiment was the first example of primary dolomite mediated by microbes (Figure 4).
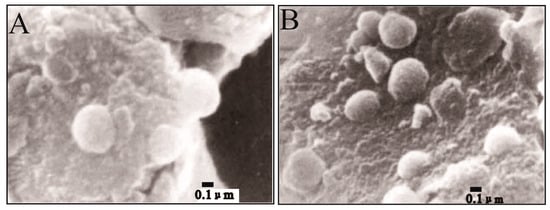
Figure 4.
SEM images of dolomite with high iron content in microbial culture experiments [37]. Both (A) and (B) are illustrating nanocrystals of the ferroan dolomite mediated by subpherical nanobacteria on the surface of the substrate.
At present, microbe-mediated pathways to precipitate primary dolomite found around the world can be divided into four major categories, which are dissimilatory sulfate reduction, aerobic heterotrophy, methanogenesis (anaerobic oxidation of methane), and chemotrophic sulfide oxidation. In addition, aerobic heterotrophy and chemotrophic sulfide oxidation promote the precipitation under aerobic conditions, while the other two work without oxygen [46]. The types of representative strains and mediating conditions are shown in Table 1. Desulfovibrio is a strain of SRB, and the precipitation of primary dolomite mediated by acetoclastic methanogens reported by Roberts et al. is an example of methanogenesis [37,38].

Table 1.
Microbial metabolism associated with dolomite formation [39,46].
It is difficult for dolomite to precipitate directly from solution under inorganic conditions at ambient temperature and pressure in the surface environment [77]. According to the International Mineralogical Association (IMA) guidelines, ankerite is obtained when Fe2+ substitutes for half of the Mg2+ in the dolomite crystal lattices, which is also not directly precipitated in the surface environment [78]. The standard dolomite and ankerite diffraction data in the International Center for Diffraction Data (ICDD) are shown in Table 2. Compared with the microbial-mediated dolomite d(hkl), with 102 and 122 peaks (Table 3), these data indicate that the experimental carbonate has a composition lying between dolomite and ankerite. There is no doubt that the mineral is a ferroan dolomite (Figure 4 and Figure 5). Although ankerite, in the strict sense, was not precipitated in these experiments, the microbial culture experiments revealed that iron would enter the dolomite crystal lattice during the microbial-mediated precipitation. A kind of calcium-rich, disordered ferroan dolomite was also found in n oil-contaminated aquifer near Bemidji, Minnesota, USA [38] (Figure 5), which was mediated by acetoclastic methanogens using Fe2+ produced by upper DIRB.

Table 2.
Standard dolomite and ankerite XRD data (ICDD data).

Table 3.
Comparison of the lattice parameters of dolomite synthesized with microbes and standard dolomite and ankerite [37].
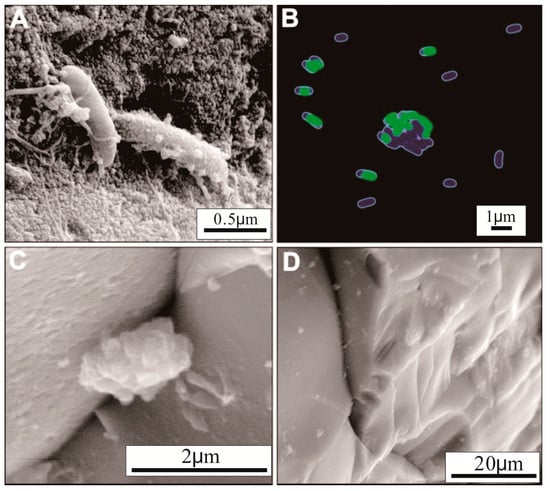
Figure 5.
Micrographs of calcium-rich, disordered ferroan dolomite [38]. (A) Scanning electron microscopy (SEM) image; (B) Scanning laser confocal microscopy image; (C) Environmental SEM (ESEM) micrograph; (D) ESEM micrograph of the basalt surface in the sterile control.
3.2. The Utilization of Fe2+ in Methanogans and SRB
Iron is an essential trace element for almost all organisms, and its morphology and bioavailability control the dynamics and function of ecosystems [8,69]. For example, the nanoparticles of Fe(III) minerals can adsorb other heavy metals that are unfavorable to organisms [70], and the redox process of Fe controls the concentration of toxic substance Arsenic in the environment [86]. For life activities, heterodisulfide reductase (a protein containing [Fe-S] cluster) in methanogens and the ruberdoxin (the simplest iron-sulfur protein) in the sulfate-reducing bacteria (SRB) are important enzymes in their metabolism [87,88]. Due to the redox activity of iron, it becomes a major player in the biogeochemical cycle. Fe2+ produced by organic matter reduction can supply microbial life activities in a reducing environment [2,5].
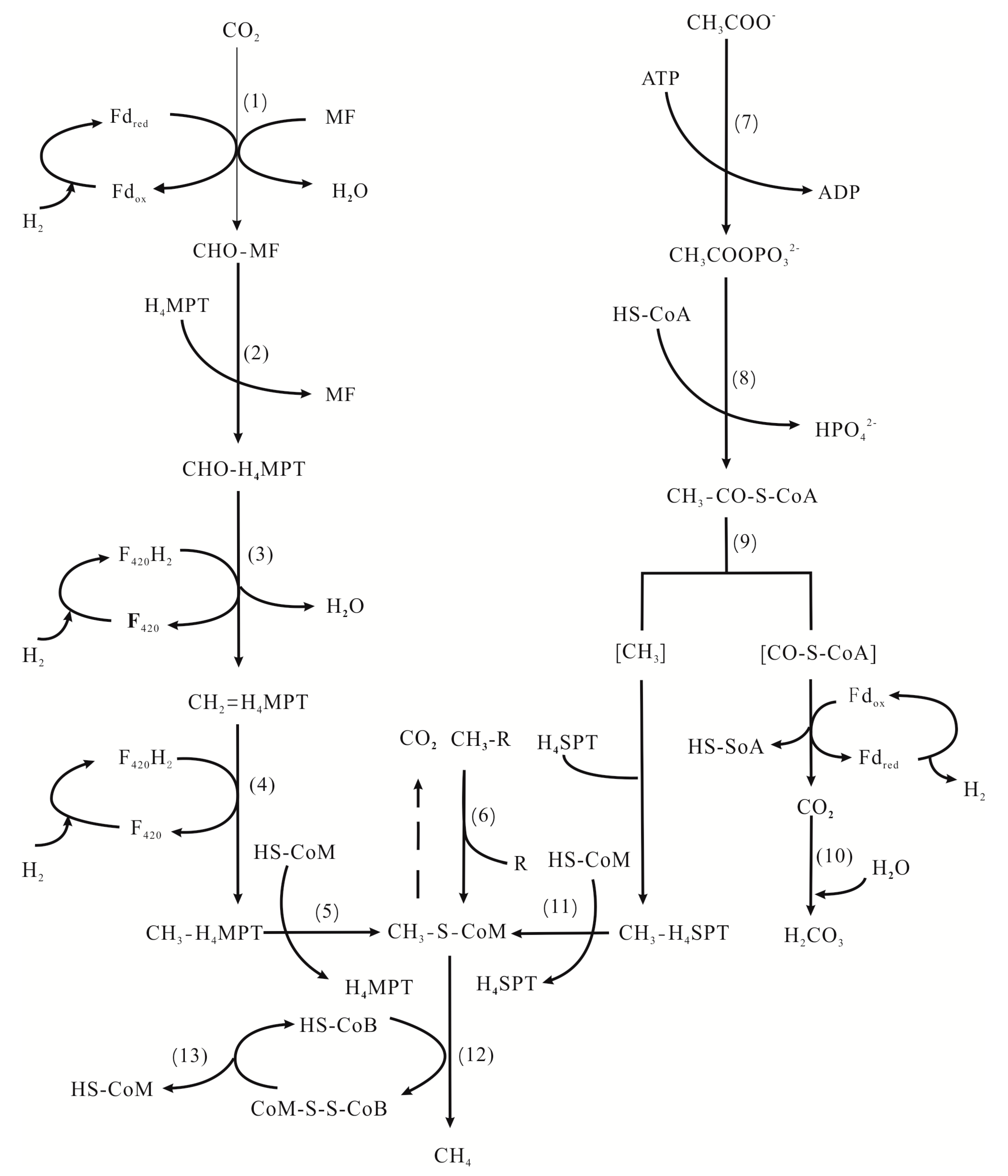
In the reaction in which anaerobic oxidation of methane (AOM) directly generates CH4, methyl-coenzyme M-reductase (Mcr) uses coenzyme B (HS–CoB), which is an electron donor, and reduces the terminal methyl carrier CH3–S–CoM to produce methane (Figure 6, Reaction (12)). In Reaction (13), heterodisulfide reductase (Hdr), as a key enzyme, can re-release coenzyme M and coenzyme B so that the methanogenesis process can be carried out continuously [87,89]. There are two types of Hdr [89,90]. One is necessary for methanogens using the reduced ferredoxin as electron donors. The other is to synthesize ATP by coupling with the reduction process of ferredoxin in the first step of the methanogenesis process. Fe2+, which is an essential component of ferredoxin, is important irreplaceably in methanogenesis. The Fe2+ produced by the DIRB in the Fe(III) respiration process compensates for the demand in MZ, and participates in various stages of metabolism with the enzyme.
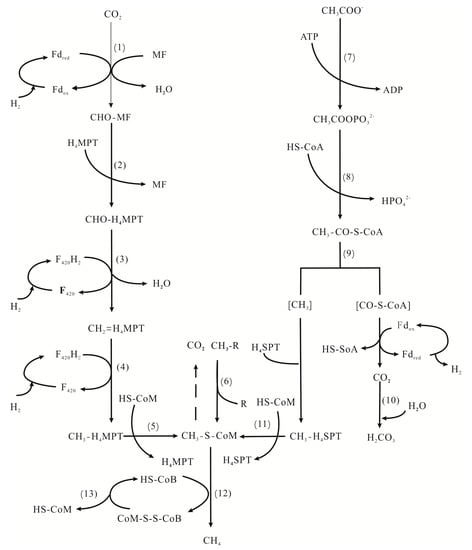
Figure 6.
Biochemical pathways of methanogenesis [87].
SRB can reduce a variety of high valence metals [61,73,74,75]. Although the SRZ is located below the IRZ, Fe(III) reduction still occurs in the sulfate reduction zone [48,57]. Moreover, Fe3+ in IRZ is reduced to Fe2+, and then Fe2+ can migrate downward, reacting with H2S generated in SRZ below and, lastly, forming iron sulfide such as FeS [49,50,51]. The combination of Fe2+ and H2S precipitates FeS, which eliminates the toxicity of H2S to SRB and promotes its metabolism [91]. In addition, the high Fe2+ concentration has no inhibitory effect on SRB growth, because there is also a large amount of protein containing Fe2+ to complete metabolism, such as rubredoxin and desulforedoxin [88,91].
3.3. Possible Process of Microbial-Mediated Nucleation
PH and carbonate alkalinity in the micro-environment can be changed in microbial metabolism, in which substances that promote nucleation are secreted to precipitate carbonate minerals [3,36,79,92,93,94,95]. There are a variety of groups with negatively charged ligands with different coordination geometries on the cell surface and extracellular polymeric substances (EPS), including sugars, amino acids, carboxylate, phosphate, and sulphate complexes, which bind divalent metal cations and promote carbonate minerals to nucleate and precipitate on the surface of organic matter [36,96,97,98,99,100,101,102,103,104,105]. This process is heterogeneous nucleation, which is closely related to the formation of ancient carbonate, including limestone and dolomite [42,43,106,107]. In the process, amorphous carbonate minerals (ACM) with lower order are precipitated first [97,108], and ACM can grow continuously then [105], even siderite [36].
In the precipitation of ferroan dolomite mediated by microbes, many functional groups exposed on cell surfaces and EPS, such as carboxylic acid (R–COOH), hydroxyl (R–OH), amino acid (R–NH2), sulfate (RO–SO3H), sulfonic acid (–SO3H), and thiol (–SH), bind metal ions like Fe2+, Ca2+, and Mg2+, and provide nucleation sites [39,42,43,44]. For the Mg2+ that are difficult to enter the dolomite lattice, it may be complexed by carboxyl groups on the cell surface, dehydrated into [Mg(H2O)5(R–COO)]+ [109], decreased the energy required to precipitate carbonate minerals, and directly precipitated by heterogeneous nucleation on the EPS and cell surface. The pH and carbonate alkalinity of the local micro-environment are increased throughout the microbial metabolism, including SRB [37,82] and DIRB [67], which overcomes the kinetic energy of dolomite formation [37,84], and an SRB removing sulfate, which is a nucleating inhibitor, at the same time, which uniformly nucleates dolomite in solution [110].
It has been confirmed that microbial factors are necessary for precipitation of primary dolomite under normal temperature conditions. Many enzymes containing Fe2+ participate microbial metabolism and metal cations bind on cell surfaces and EPS. However, there may be other factors affecting the process of Fe2+ entering the dolomite lattice, such as the ion radius (the Fe2+ radius common in a mineral crystal structure is 0.061 nm, which is less than the Mg2+ 0.072 nm [111]). The mediation of ferroan dolomite still needs more detailed experiments.
3.4. Ankerite in Cold Seeps
Cold seeps are widely distributed on the active and passive continental marginal slopes and transform boundaries, mainly in the form of mounds, pockmark, chimneys, cement, and small veins [23,24]. Many studies show that cold seep carbonate is associated with hydrocarbon leakage, mainly methane, in a large scale [23,25,112,113,114]. In the Gulf of Cadiz, which is a huge hydrocarbon seep, there are a large number of carbonate chimneys consisting of Fe-rich dolomite, ankerite, high magnesium calcite, quartz, feldspar, and clays [112,113,114,115].
The δ13C data suggests that the carbon source of these authigenic carbonates is derived from methane [114,116]. Many microfabrics induced by microbial mineralization are observed with SEM, including filamentous, rod-shaped, dumbbell-shaped, and cauliflower dolomite [114]. The evidence suggests that microbes play an essential role in this process. In the sulfate-methane transition zone (SMTZ or SMT) under anaerobic conditions, due to the synergistic effects of methane-oxidizing archaea and SRB, including Methanosarcina, Desulfobulbus and Desulfosarcina [25,26,27,28,29,30], anoxic oxidation of methane (AOM) and sulfate reduction (SR) produce a large amount of HCO3− and HS−. This causes an increase in environmental alkalinity, and results in authigenic carbonate minerals precipitation [23,25,112,113,114,115,116].
The Fe content of the primary dolomite in the Gulf of Cadiz is not significant [114], which has a texture similar to that of microbial-mediated dolomite in laboratories and surface lagoons [28,37,38,82,83,84,85]. However, oxygen isotope evidence indicates that their temperatures of formation are between 8.7 °C and 13.9 °C [112], which is lower than those in many laboratories and surface lagoons [28,38,82,83,84,85], but more than that achieved by Vasconcelos et al. (1995) [37]. The main source of iron in Fe-rich dolomite and ankerite in carbonate chimneys is sediment [113]. In other words, these Fe-rich dolomite and ankerite preserve evidence that some microbes mediate primary dolomite, but the microbes failed to directly enrich a large amount of iron into lattices of dolomite in the precipitation stage.
4. Conclusions and Future Directions
Fe(III), which is difficult to dissolve under surface conditions, cannot enter dolomite lattices, but insoluble Fe(III) and soluble Fe(II) are linked by microbes in the biogeochemical Fe cycle, especially Fe(III) respiration, which provides sufficient Fe2+ in anaerobic environments. Moreover, there are microbial cells and various functional groups on the surface adsorbing free metal cations. Fe2+ is involved in the metabolism of bacteria deeply as an important component of various enzymes. These functions make it possible for Fe2+ to enter dolomite lattices.
Future experiments can further be designed in both qualitative and quantitative aspects. Qualitative research studies can determine the process of Fe2+ entering the lattice and helps to understand the role of various organic groups such as EPS, ferredoxin, and iron-sulfur protein in the process. On the other hand, quantitative research studies explain how much Fe2+ precipitates in ferroan dolomite and then verify the yield of Fe2+ in the metabolic process of DIRB, methanogens, SRB, etc. Such research studies are beneficial for understanding organic dolomite precipitation. In addition, these studies are likely to provide new power to solve the “dolomite problem”.
Author Contributions
All the authors contributed to this paper. X.Y. and Q.L. concentrated on such phenomena. X.Y. discussed with F.X. and F.X. prepared and edited the manuscript. Y.L. drew some pictures. All the authors revised the manuscript.
Funding
The National Natural Science Foundation of China [grant number 41402102], and Fundamental Research Funds for the Central Universities [grant number 2652017132] financially supported this work.
Acknowledgments
We are grateful to the pioneers in this area for their experiments.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Dong, H.L. Mineral-microbe interactions: A review. Front. Earth Sci. PRC 2010, 4, 127–147. [Google Scholar] [CrossRef]
- Xie, S.C.; Yang, H.; Luo, G.M.; Huang, X.Y.; Liu, D.; Wang, Y.B.; Gong, Y.M.; Xu, R. Geomicrobial functional groups: A window on the interaction between life and environments. Chin. Sci. Bull. 2012, 57, 2–19. (In Chinese) [Google Scholar] [CrossRef]
- Krause, S.; Liebetrau, V.; Gorb, S.; Sánchez-Román, M.; McKenzie, J.A.; Treude, T. Microbial nucleation of Mg-rich dolomite in exopolymeric substances under anoxic modern seawater salinity: New insight into an old enigma. Geology 2012, 40, 987–990. [Google Scholar] [CrossRef]
- Konhauser, K.O.; Kappler, A.; Roden, E.E. Iron in microbial metabolisms. Elements 2011, 7, 89–93. [Google Scholar] [CrossRef]
- Weber, K.A.; Achenbach, L.A.; Coates, J.D. Microorganisms pumping iron: Anaerobic microbial iron oxidation and reduction. Nat. Rev. Microbiol. 2006, 4, 752–764. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Dong, H.L.; Seabaugh, J.; Newell, S.W.; Eberl, D.D. Role of microbes in the smectite-to-illite reaction. Science 2004, 303, 830–832. [Google Scholar] [CrossRef] [PubMed]
- Lian, B. Research progress of mineral-microbe interactions: A comments for geomicrobiology column. Bull. Mineral. Petrol. Geochem. 2014, 33, 759–763. (In Chinese) [Google Scholar]
- Melton, E.D.; Swanner, E.D.; Behrens, S.; Schmidt, C.; Kappler, A. The interplay of microbially mediated and abiotic reactions in the biogeochemical Fe cycle. Nat. Rev. Microbiol. 2014, 12, 797–808. [Google Scholar] [CrossRef]
- Ouyang, B.J.; Lu, X.C.; Liu, H.; Li, J.; Lu, J.J.; Guan, S.Q.; Wang, R.Y. Reduction of goethite by Shewanella oneidensis MR-1 and its geochemical implication. Acta Mineral. Sin. 2013, 33, 389–396, (In Chinese with English Abstract). [Google Scholar]
- Ding, H.R.; Li, Y.; Lu, A.H.; Wang, X.; Zeng, C.P.; Yan, Y.H.; Wang, C.Q. Electrochemical research on iron oxide minerals reduction by microorganisms. Bull. Mineral. Petrol. Geochem. 2011, 30, 299–303. [Google Scholar]
- Shi, L.; Rosso, K.M.; Zachara, J.M.; Fredrickson, J.K. Mtr extracellular electron-transfer pathways in Fe(III)-reducing or Fe(II)-oxidizing bacteria: A genomic perspective. Biochem. Soc. Trans. 2012, 40, 1261–1267. [Google Scholar] [CrossRef] [PubMed]
- Posth, N.R.; Hegler, F.; Konhauser, K.O.; Kappler, A. Alternating Si and Fe deposition caused by temperature fluctuations in Precambrian oceans. Nat. Geosci. 2008, 1, 703–708. [Google Scholar] [CrossRef]
- Konhauser, K.O.; Hamade, T.; Raiswell, R.; Morris, R.C.; Ferris, F.G.; Southam, G.; Canfield, D.E. Could bacteria have formed the precambrian banded iron formations? Geology 2002, 30, 1079–1082. [Google Scholar] [CrossRef]
- Han, X.Q.; Shen, H.T.; Chen, J.L.; Qian, J.C.; Lin, C.Y.; Bian, L.Z. Preliminary study on the biological genesis and bio-chemical binary mineralization mechanism of polymetallic nodules in the Pacific Ocean. Sci. China Earth Sci. 1997, 27, 349–353. (In Chinese) [Google Scholar]
- Hu, W.X.; Zhou, H.Y.; Gu, L.X.; Zhang, W.L.; Lu, X.C.; Pan, J.M.; Fu, Q.; Zhang, H.S. New evidence for microbial genesis of deep sea (iron) manganese nodules. Sci. China Earth Sci. 1999, 29, 362–366. (In Chinese) [Google Scholar]
- Dugolisky, B.K.; Margolis, S.K.; Dudly, M.C. Biogenic influence on growth of manganese nodules. J. Sediment. Pet. 1977, 47, 428–445. [Google Scholar]
- Suess, E. Marine cold seeps. In Handbook of Hydrocarbon and Lipid Microbiology; Timmis, K.N., Ed.; Springer: Heidelberg, Germany, 2010; pp. 187–203. [Google Scholar]
- Peckmanna, J.; Reimera, A.; Luth, U.; Luth, C.; Hansen, B.T.; Heinickes, C.; Hoefs, J.; Reitnera, J. Methane-derived carbonates and authigenic pyrite from the northwestern Black Sea. Mar. Geol. 2001, 177, 129–150. [Google Scholar] [CrossRef]
- Boetius, A.; Ravenschlag, K.; Schubert, C.J.; Rickert, D.; Widdel, F.; Gieseke, A.; Amann, R.; Jùrgensen, B.B.; Witte, U.; Pfannkuche, O. A marine microbial consortium apparently mediating anaerobic oxidation of methane. Nature 2000, 407, 623–626. [Google Scholar] [CrossRef]
- Orphan, V.J.; House, C.H.; Hinrichs, K.U.; McKeegan, K.D.; DeLong, E.F. Multiple archaeal groups mediate methane oxidation in anoxic cold seep sediments. Proc. Natl. Acad. Sci. USA 2002, 99, 7663–7668. [Google Scholar] [CrossRef]
- Knittel, K.; Lösekann, T.; Boetius, A.; Kort, R.; Amann, R. Diversity and distribution of methanotrophic archaea at cold seeps. Appl. Environ. Microb. 2005, 71, 467–479. [Google Scholar] [CrossRef]
- Valentine, D.L.; Reeburgh, W.S. New perspectives on anaerobic methane oxidation. Environ. Microb. 2000, 2, 477–484. [Google Scholar] [CrossRef]
- Lanoil, B.D.; Sassen, R.; La Duc, M.T.; Sweet, S.T.; Nealson, K.H. Bacteria and Archaea physically associated with Gulf of Mexico gas hydrates. Appl. Environ. Microb. 2001, 67, 5143–5153. [Google Scholar] [CrossRef]
- Guan, H.X.; Chen, D.F.; Song, Z.G. Biomarkers and bacterial processes in the sediments of gas seep site. Marin. Geol. Quat. Geol. 2007, 27, 75–83. [Google Scholar]
- Chen, Z.; Yang, H.P.; Huang, C.Y.; Yan, W.; Lu, J. Diagenetic environment and implication of seep carbonate precipitations from the southwestern Dongsha area, South China Sea. Geosciences 2008, 22, 382–389, (In English with Chinese Abstract). [Google Scholar]
- Yang, K.H.; Chu, F.Y.; Zhao, J.R.; Lei, J.J.; Ge, Q.; Fang, Y.X. The layer structure and its geological significanceof seep carbonates in the north of South China Sea. Marin. Geol. Quat. Geol. 2008, 28, 11–16, (In English with Chinese Abstract). [Google Scholar]
- Lu, H.F.; Chen, F.; Liu, J.; Zhou, Y.; Liao, Z.L. Mineralogies and stable isotopic compositions of methane-derived carbonates from the northeastern South China Sea. Marin. Geol. Quat. Geol. 2010, 30, 51–59. (In English) [Google Scholar] [CrossRef]
- Vasconcelos, C.; McKenzie, J.A. Microbial mediation of modern dolomite precipitation and diagenesis under anoxic conditions (Lagoa Vermelha, Rio de Janeiro, Brazil). J. Sediment. Res. 1997, 67, 378–390. [Google Scholar]
- Deng, S.C.; Dong, H.L.; Lv, G.; Jiang, H.C.; Yu, B.S.; Bishop, M.E. Microbial dolomite precipitation using sulfate reducing and halophilic bacteria: Results from Qinghai Lake, Tibetan Plateau, NW China. Chem. Geol. 2010, 278, 151–159. [Google Scholar] [CrossRef]
- Han, Z.Z.; Gao, X.; Zhao, H.; Tucker, E.M.; Zhao, Y.H.; Bi, Z.P.; Pan, J.T.; Wu, G.Z.; Yan, H.X. Extracellular and intracellular biomineralization induced by bacillus licheniformis DB1-9 at different Mg/Ca Molar Ratios. Minerals 2018, 8, 585. [Google Scholar] [CrossRef]
- Han, Z.Z.; Yu, W.W.; Zhao, H.; Zhao, Y.H.; Tucker, E.M.; Yan, H.X. The significant roles of Mg/Ca ratio, Cl− and SO42− in carbonate mineral precipitation by the halophile staphylococcus epidermis Y2. Minerals 2018, 8, 594. [Google Scholar] [CrossRef]
- Yang, R.C.; Fan, A.P.; Han, Z.Z.; Chi, N.J.; Han, Y. Characteristics and genesis of microbial lumps in the Maozhuang Stage (Cambrian Series 2), Shandong Province, China. Sci. China Earth Sci. 2013, 56, 494–503. [Google Scholar] [CrossRef]
- Riding, R. Microbialites, stromatolites, and thrombolites. In Encyclopedia of Geobiology; Reitner, J., Thiel, V., Eds.; Encyclopedia of Earth Science Series; Springer: Heidelberg, Germany, 2011; pp. 635–654. [Google Scholar]
- Dong, S.F.; Chen, D.Z.; Ling, H.R.; Zhou, X.Q.; Wang, D.; Guo, Z.H.; Jiang, M.S.; Qian, Y.X. Hydrothermal alteration of dolostones in the Lower Ordovician, Tarim Basin, NW China: Multiple constraints from petrology, isotope geochemistry and fluid inclusion microthermometry. Mar. Petrol. Geol. 2013, 46, 270–286. [Google Scholar] [CrossRef]
- Zhang, J.T.; He, Z.L.; Yue, X.J.; Sun, Y.P.; Jin, X.H.; Chen, X. Genesis of iron-rich dolostones in the 5th Member of the Majiagou Formation of the Ordovician in Ordos Basin. Oil Gas Geol. 2017, 38, 776–783, (In Chinese with English Abstract). [Google Scholar]
- Sánchez-Román, M.; Fernández-Remolar, D.; Amils, R.; Sánchez-Navas, A.; Schmid, T.; Martin-Uriz, P.S.; Rodríguez1, N.; McKenzie, J.A.; Vasconcelos, C. Microbial mediated formation of fe-carbonate minerals under extreme acidic conditions. Sci. Rep. 2014, 4, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, C.; Mckenzie, J.A.; Bernasconi, S.; Grujic, D.; Tiens, A.J. Microbial mediation as a possible mechanism for natural dolomite formation at low temperatures. Nature 1995, 377, 220–222. [Google Scholar] [CrossRef]
- Roberts, J.A.; Bennett, P.C.; González, L.A.; Macpherson, G.L.; Milliken, K.L. Microbial precipitation of dolomite in methanogenic groundwater. Geology 2004, 34, 277–280. [Google Scholar] [CrossRef]
- You, X.L.; Sun, S.; Zhu, J.Q.; Liu, L.; He, K. Progress in the study of microbial dolomite model. Front. Earth Sci. 2011, 18, 52–64, (In Chinese with English Abstract). [Google Scholar]
- You, X.L.; Sun, S.; Zhu, J.Q.; Li, Q.; Hu, W.X.; Dong, H.L. Microbially mediated dolomite in Cambrian stromatolites from the Tarim Basin, north-west China: Implications for the role of organic substrate on dolomite precipitation. Terra Nova 2013, 25, 387–395. [Google Scholar] [CrossRef]
- You, X.L.; Sun, S.; Zhu, J.Q. Significance of fossilized microbes from the Cambrian stromatolites in the Tarim Basin, Northwest China. Sci. China Earth Sci. 2014, 44, 1777–1790. (In Chinese) [Google Scholar] [CrossRef]
- You, X.L.; Lin, C.S.; Zhu, J.Q.; Tan, J.Y. Primary microbial dolomite precipitation in culture experiments and in stromatolite formations: Implications for the Dolomite Problem. Carpath. J. Earth Environ. 2015, 10, 197–206. [Google Scholar]
- You, X.L.; Sun, S.; Lin, C.S.; Zhu, J.Q. Microbial dolomite in the sabkha environment of the middle Cambrian in the Tarim Basin, NW China. Aust. J. Earth Sci. 2018, 65, 109–120. [Google Scholar] [CrossRef]
- You, X.L.; Jia, W.Q.; Xu, F.; Liu, Y. Mineralogical characteristics of ankerite and mechanisms of primary and secondary origins. J. Earth Sci. China 2018, 43, 4046–4055. [Google Scholar]
- Huang, Y.J.; Wang, C.S. Progress of the study of reactive iron cycling in the paleo-ocean and its applications to the genesis of Cretaceous anoxic-oxic sedimentary transition. Front. Earth Sci. 2009, 16, 172–179. [Google Scholar]
- Petrash, D.A.; Bialik, O.M.; Bontognali, T.R.R.; Vasconcelos, C.; Roberts, J.A.; McKenzie, J.A.; Konhauser, K.O. Microbially catalyzed dolomite formation: From near-surface to burial. Earth Sci. Rev. 2017, 171, 558–582. [Google Scholar] [CrossRef]
- Prauss, M.L. Availability of reduced nitrogen chemospecies in photic-zone waters as the ultimate cause for fossil prasinophyte prosperity. Palaios 2007, 22, 489–499. [Google Scholar] [CrossRef]
- Kappler, A.; Benz, M.; Schink, B.; Brune, A. Electron shuttling via humic acids in microbial iron(iii) reduction in a freshwater sediment. FEMS Microbiol. Ecol. 2004, 47, 85–92. [Google Scholar] [CrossRef]
- Berner, R.A. Early Diagenesis: A Theoretical Approach; Princeton University Press: Princeton, NJ, USA, 1980; p. 256. [Google Scholar]
- Froelich, P.N.; Klinkhammer, G.P.; Bender, M.L.; Luedtke, N.A.; Heath, G.R.; Cullen, D.; Dauphin, P.; Hammond, D.; Hartman, B.; Maynard, V. Early oxidation of organic matter in pelagic sediments of the Eastern Equatorial Atlantic: Suboxic diagenesis. Geochim. Cosmochim. Acta 1979, 43, 1075–1090. [Google Scholar] [CrossRef]
- Spiteri, C.; Regnier, P.; Slomp, C.P.; Charette, M.A. PH-Dependent iron oxide precipitation in a subterranean estuary. J. Geochem. Explor. 2006, 88, 399–403. [Google Scholar] [CrossRef]
- Raiswell, R.; Canfield, D.E. Sources of iron for pyrite formation in marine sediments. Am. J. Sci. 1998, 298, 219–245. [Google Scholar] [CrossRef]
- Meyers, S.R.; Sageman, B.B.; Lyons, T.W. Organic carbon burial rate and the molybdenum proxy: Theoretical framework and application to Cenomanian-Turonian Oceanic Anoxic Event 2. Paleoceanography 2005, 20, PA2002. [Google Scholar] [CrossRef]
- Beal, E.J.; House, C.H.; Orphan, V.J. Manganese-and iron-dependent marine methane oxidation. Science 2009, 325, 184–187. [Google Scholar] [CrossRef] [PubMed]
- Canfield, D.E.; Thamdrup, B.; Hansen, J.W. The anaerobic degradation of organic matter in danish coastal sediments: Iron reduction, manganese reduction, and sulfate reduction. Geochim. Cosmochim. Acta 1993, 57, 3867–3883. [Google Scholar] [CrossRef]
- Zhang, C.L.; Liu, S.; Logan, J.; Mazumder, R.; Phelp, T.J. Enhancement of Fe(III), Co(III), and Cr(VI) reduction at elevated temperatures and by a thermophilic bacterium. Appl. Biochem. Biotech. 1996, 57, 923–932. [Google Scholar] [CrossRef]
- Liu, D. Mineral Transformations Associated With Structural Fe(Iii) Reduction in Clay Minerals by Different Microbial Functional Groups. Ph.D. Thesis, China University of Geosciences, Wuhan, China, 2012. (In Chinese with English Abstract). [Google Scholar]
- Chen, L.; Zhang, H.X.; Li, Y.; Zheng, S.L.; Liu, F.H. The role of microorganisms in the geochemical iron cycle. Sci. Sin. Vitae 2016, 46, 1069–1078, (In Chinese with English Abstract). [Google Scholar]
- Roden, E.E.; Zachara, J.M. Microbial reduction of crystalline Iron(III) oxides: Influence of oxide surface area and potential for cell growth. Environ. Sci Technol. 1996, 30, 1618–1628. [Google Scholar] [CrossRef]
- Bolm, C. A new iron age. Nat. Chem. 2009, 1, 420. [Google Scholar] [CrossRef] [PubMed]
- Lovley, D.R. Dissimilatory Fe(III) and Mn(IV) reduction. Microbiol. Mol. Biol. Rew. 1991, 55, 259–287. [Google Scholar]
- Lovley, D.R.; Chapelle, F.H. Deep subsurface microbial processes. Rev. Geophys. 1995, 33, 365–381. [Google Scholar] [CrossRef]
- Lovley, D.R. Environmental Microbe-Metal Interactions; ASM Press: Washington, DC, USA, 2000; p. 408. [Google Scholar]
- Fredrickson, J.K.; Kostandarithes, H.M.; Li, S.W.; Plymale, A.E.; Daly, M.J. Reduction of Fe(III), Cr(VI), U(VI), and Tc(VII) by Deinococcus radiodurans R1. Appl. Environ. Microbiol. 2000, 66, 2006–2011. [Google Scholar] [CrossRef]
- Roden, E.E.; Lovley, D.R. Dissimilatory Fe(III) reduction by the marine microorganism Desulfuromonas acetoxidans. Appl. Environ. Microbiol. 1993, 59, 734–742. [Google Scholar]
- Roden, E.E. Fe(III) Oxide reactivity toward biological versus chemical reduction. Environ. Sci Technol. 2003, 37, 1319–1324. [Google Scholar] [CrossRef]
- Mckinley, J.P.; Stevens, T.O.; Fredrickson, J.K.; Zachara, J.M.; Colwell, F.S.; Wagnond, K.B.; Smith, S.C.; Rawson, S.A.; Bjornstad, B.N. Biogeochemistry of anaerobic lacustrine and paleosol sediments within an aerobic unconfined aquifer. Geomicrobiol. J. 1997, 14, 23–39. [Google Scholar] [CrossRef]
- Gorby, Y.A.; Yanina, S.; McLean, J.S.; Rosso, K.M.; Moyles, D.; Dohnalkova, A.; Beveridge, T.J.; Chang, I.S.; Kim, B.H.; Kim, K.S.; et al. Electrically conductive bacterial nanowires produced by Shewanella oneideasis strain MR-1 and other microorganisms. Proc. Natl. Acad. Sci. USA 2006, 103, 11358–11363. [Google Scholar] [CrossRef] [PubMed]
- Ilton, E.S.; Pacheco, J.S.L.; Bargar, J.R.; Shi, Z.; Liu, J.; Kovarik, L.; Engelhard, M.H.; Felmy, A.R. Reduction of U(VI) incorporated in the structure of hematite. Environ. Sci. Technol. 2012, 46, 9428–9436. [Google Scholar] [CrossRef] [PubMed]
- Waychunas, G.A.; Kim, C.S.; Banfield, J.F. Nanoparticulate iron oxide minerals in soils and sediments: Unique properties and contaminant scavenging mechanisms. J. Nanopart. Res. 2005, 7, 409–433. [Google Scholar] [CrossRef]
- Holmes, D.E.; O’Neil, R.A.; Vrionis, H.A.; N’Guessan, L.A.; Ortiz-Bernad, I.; Larrahondo, M.J.; Adams, L.A.; Ward, J.A.; Nicoll, J.S.; Nevin, K.P.; et al. Subsurface clade of Geobacteraceae that predominates in a diversity of Fe (III)-reducing subsurface environments. ISME J. 2007, 1, 663–677. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.B.; Gu, Y.Q.; O’Brien, J.P.; Yi, S.M.; Yalcin, S.E.; Srikanth, V.; Shen, C.; Vu, D.; Ing, N.L.; Hochbaum, A.I.; et al. Structure of microbial nanowires reveals stacked hemes that transport electrons over micrometers. Cell 2019, 177, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Vali, H.; Yang, J.; Phelps, T.; Zhang, C. Reduction of iron oxides enhanced by a sulfate-reducing bacterium and biogenic H2S. Geomicrobiol. J. 2006, 23, 103–117. [Google Scholar] [CrossRef]
- Michel, C.; Brugna, M.; Aubert, C.; Bernadac, A.; Bruschi, M. Enzymatic Reduction of Chromate: Comparative Studies Using Sulfate-Reducing Bacteria. Appl. Microbiol. Biotechnol. 2001, 55, 95. [Google Scholar] [CrossRef] [PubMed]
- Lovley, D.R. Dissimilatory metal reduction. Annu. Rev. Microbiol. 1993, 47, 263. [Google Scholar] [CrossRef]
- Tebo, B.M.; Obraztsova, A.Y. Sulfate-reducing bacterium grows with Cr(VI), U(VI), Mn(IV), and Fe(III) as electron acceptors. FEMS Microbiol. Lett. 1998, 162, 193–198. [Google Scholar] [CrossRef]
- Land, L.S. Failure to Precipitate Dolomite at 25 °C fromDilute Solution Despite 1000-Fold Oversaturation after 32 Years. Aquat. Geochem. 1998, 4, 361–368. [Google Scholar] [CrossRef]
- Gregg, J.M.; Bish, D.L.; Kaczmarek, S.E.; Machel, H.G. Mineralogy, nucleation and growth of dolomite in the laboratory and sedimentary environment: A review. Sedimentology 2015, 62, 1749–1769. [Google Scholar] [CrossRef]
- Sánchez-Román, M.; Vasconcelos, C.; Schmid, T.; Dittrich, M.; Mckenzie, J.A.; Zenobi, R.; Rivadeneyra, M.A. Aerobic microbial dolomite at the nanometer scale: Implications for the geologic record. Geology 2008, 36, 879–882. [Google Scholar] [CrossRef]
- Moreira, N.F.; Walter, L.M.; Vasconcelos, C.; McKenzie, J.A.; McCall, P.J. Role of sulfide oxidation in dolomitization: Sediment and pore-water geochemistry of a modern hypersaline lagoon system. Geology 2004, 32, 701–704. [Google Scholar] [CrossRef]
- van Gemerden, H. Microbial mats: A joint venture. Mar. Geol. 1993, 113, 3–25. [Google Scholar] [CrossRef]
- Warthmann, R.; van Lith, Y.; Vasconcelos, C.; Mckenzie, J.A.; Karpoff, A.M. Bacterially induced dolomite precipitation in anoxic culture experiments. Geology 2000, 28, 1091–1094. [Google Scholar] [CrossRef]
- Wright, D.T.; Wacey, D. Precipitation of dolomite using sulphate reducing bacteria from the Coorong Region, South Australia: Significance and implications. Sedimentology 2005, 52, 987–1008. [Google Scholar] [CrossRef]
- Moore, T.S.; Murray, R.W.; Kurtz, A.C.; Schrag, D.P. Anaerobic methane oxidation and the formation of dolomite. Earth Planet. Sci. Lett. 2004, 229, 141–154. [Google Scholar] [CrossRef]
- Kenward, P.A.; Goldstein, R.H.; González, L.A.; Roberts, J.A. Precipitation of low-temperature dolomite from an anaerobic microbial consortium: The role of methanogenic archaea. Geobiology 2010, 7, 556–565. [Google Scholar] [CrossRef]
- Tufano, K.J.; Reyes, C.; Saltikov, C.W.; Fendorf, S. Reductive processes controlling arsenic retention: Revealing the relative importance of iron and arsenic reduction. Environ. Sci. Technol. 2008, 42, 8283–8289. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Whitman, W.B. Metabolic, phylogenetic, and ecological diversity of the methanogenic archaea. Ann. N. Y. Acad. Sci. 2008, 1125, 171–189. [Google Scholar] [CrossRef] [PubMed]
- Moura, I.; Pereira, A.S.; Tavares, P.; Moura, J.J.G. Simple and complex iron-sulfur proteins in sulfate reducing bacteria. Adv. Inorg. Chem. 1999, 47, 361–419. [Google Scholar]
- Refai, S.; Berger, S.; Wassmann, K.; Deppenmeier, U. Quantification of methanogenic heterodisulfide reductase activity in biogas sludge. J. Biotechnol. 2014, 180, 66–69. [Google Scholar] [CrossRef] [PubMed]
- Thauer, R.; Kaster, A.; Seedorf, H.W.; Hedderich, R. Methanogenic archaea: Ecologically relevant differences in energy conservation. Nat. Rev. Microbiol. 2008, 6, 579–591. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y. Treatment of Acid Waste Water Containing Heavy Metal with Sulfate Reducing Bacteria and Fe0. Ph.D. Thesis, Tianjin University, Tianjin, China, 2004. (In Chinese with English Abstract). [Google Scholar]
- Aloisi, G.; Gloter, A.; Krüger, M.; Wallmann, K.; Guyot, F.; Zuddas, P. Nucleation of calcium carbonate on bacterial nanoglobules. Geology 2006, 34, 1017–1020. [Google Scholar] [CrossRef]
- Bontognali, T.R.; Vasconcelos, C.; Warthmann, R.; Dupraz, C.; Bernasconi, S.; McKenzie, J.A. Microbes produce nanobacteria-like structures, avoiding cell entombment. Geology 2008, 36, 663–666. [Google Scholar] [CrossRef]
- Ferris, F.G.; Fyfe, W.S.; Beveridge, T.J. Bacteria as nucleation sites for authigenic minerals. In Diversity of Environmental Biogeochemistry; Berthelin, J., Ed.; Dev. Geochem. Elsevier: Amsterdam, The Netherlands, 1991; pp. 319–326. [Google Scholar]
- Dupraz, C.; Visscher, P.T.; Baumgartner, L.K.; Reid, R.P. Microbe—Mineral interactions: Early carbonate precipitation in a hypersaline lake (Eleuthera Island, Bahamas). Sedimentology 2004, 51, 745–765. [Google Scholar] [CrossRef]
- Trichet, J.; Défarge, C. Non-biologically supported organomineralization. Bull. l’Institut Oceanogr. Monaco (Spec. Issue) 1995, 14, 203–226. [Google Scholar]
- Raz, S.; Weiner, S.; Addadi, L. Formation of high-magnesian calcites via an amorphous precursor phase: Possible biological implications. Adv. Mater. 2000, 12, 38–42. [Google Scholar] [CrossRef]
- Han, Y.J.; Aizenberg, J. Effect of magnesium ions on oriented growth of calcite on carboxylic acid functionalized self-assembled monolayer. J. Am. Chem. Soc. 2003, 125, 4032–4033. [Google Scholar] [CrossRef] [PubMed]
- Decho, A.W.; Visscher, P.T.; Reid, R.P. Production and cycling of natural microbial exopolymers (EPS) within a marine stromatolite. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2005, 219, 71–86. [Google Scholar] [CrossRef]
- Gilbert, P.; Abrecht, M.; Frazer, B.H. The organic-mineral interface in biominerals. Rev. Mineral. Geochem. 2005, 59, 157–185. [Google Scholar] [CrossRef]
- Visscher, P.T.; Stolz, J.F. Microbial mats as bioreactors: Populations, processes, and products. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2005, 219, 87–100. [Google Scholar] [CrossRef]
- Kwak, S.Y.; DiMasi, E.; Han, Y.J.; Aizenberg, J.; Kuzmenko, I. Orientation and mg incorporation of calcite grown on functionalized self-assembled monolayers: A synchrotron X-ray study. Cryst. Growth Des. 2005, 5, 2139–2145. [Google Scholar] [CrossRef]
- Braissant, O.; Decho, A.W.; Dupraz, C.; Glunk, C.; Przekop, K.M.; Visscher, P.T. Exopolymeric substances of sulfate-reducing bacteria: Interactions with calcium at alkaline pH and implication for formation of carbonate minerals. Geobiology 2007, 5, 401–411. [Google Scholar] [CrossRef]
- Braissant, O.; Decho, A.W.; Przekop, K.M.; Gallagher, K.L.; Glunk, C.; Dupraz, C.; Visscher, P.T. Characteristics and turnover of exopolymeric substances in a hypersaline microbial mat. FEMS Microbiol. Ecol. 2009, 69, 293–307. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Wallace, A.F.; De Yoreo, J.J.; Dove, P.M. Carboxylated molecules regulate magnesium content of amorphous calcium carbonates during calcification. Proc. Natl. Acad. Sci. USA 2009, 106, 21511–21516. [Google Scholar] [CrossRef] [PubMed]
- Bosak, T.; Newman, D.K. Microbial nucleation of calcium carbonate in the Precambrian. Geology 2003, 31, 577–580. [Google Scholar] [CrossRef]
- Allwood, A.C.; Grotzinger, J.P.; Knoll, A.H.; Burch, I.W.; Anderson, M.S.; Coleman, M.L.; Kanik, I. Controls on development and diversity of early archean stromatolites. Proc. Natl. Acad. Sci. USA 2009, 106, 9548–9555. [Google Scholar] [CrossRef]
- Aizenberg, B.J.; Lambert, G.; Addadi, L.; Weiner, S. Stabilization of amorphous calcium carbonate. Adv. Mater. 1996, 8, 222–226. [Google Scholar] [CrossRef]
- Xu, J.; Yan, C.; Zhang, F.; Konishi, H.; Xu, H.F.; Teng, H.H. Testing the cation-hydration effect on the crystallization of Ca-Mg-CO3 systems. Proc. Natl. Acad. Sci. USA 2013, 110, 17750–17755. [Google Scholar] [CrossRef] [PubMed]
- Baker, P.A.; Kastner, M. Constraints on the formation of sedimentary dolomite. Science 1981, 213, 214–216. [Google Scholar] [CrossRef] [PubMed]
- Shannon, R.D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Cryst. 1976, 32, 751–767. [Google Scholar] [CrossRef]
- Díaz-del-Río, V.; Somoza, L.; Martínez-Frias, J.; Mata, M.P.; Delgado, A.; Hernandez-Molina, F.J.; Lunar, R.; Martín-Rubí, J.A.; Maestro, A.; Fernández-Puga, M.C.; et al. Vast fields of hydrocarbon-derived carbonate chimneys related to the accretionary wedge/olistostrome of the Gulf of Cádiz. Mar. Geol. 2003, 195, 177–200. [Google Scholar] [CrossRef]
- González, F.J.; Somoza, L.; Lunar, R.; Martínez-Frías, J.; Rubí, J.M.; Torres, T.; Ortiz, J.E.; Díaz-del-Río, V.; Pinheiro, L.M.; Magalhães, V.H. Hydrocarbon-derived ferromanganese nodules in carbonate-mud mounds from the Gulf of Cadiz: Mud-breccia sediments and clasts as nucleation sites. Mar. Geol. 2009, 261, 64–81. [Google Scholar] [CrossRef]
- Magalhães, V.H.; Pinheiro, L.M.; Ivanov, M.K.; Kozlova, E.; Blinova, V.; Kolganova, J.; Vasconcelos, C.; McKenzie, J.A.; Bernasconi, S.M.; Kopf, A.J.; et al. Formation processes of methane-derived authigenic carbonates from the Gulf of Cadiz. Sediment. Geol. 2012, 243, 155–168. [Google Scholar] [CrossRef]
- Merinero, R.; Lunar, R.; Martínez-Frías, J.; Somoza, L.; Díaz-del-Río, V. Iron oxyhydroxide and sulphide mineralization in hydrocarbon seep-related carbonate submarine chimneys, Gulf of Cadiz (SW Iberian Peninsula). Mar. Petrol. Geol. 2008, 25, 706–713. [Google Scholar] [CrossRef]
- Niemann, H.; Duarte, J.; Hensen, C.; Omoregie, E.; Magalhães, V.H.; Elvert, M.; Pinheiro, L.M.; Kopf, A.; Boetius, A. Microbial methane turnover at mud volcanoes of the Gulf of Cadiz. Microbial methane turnover at mud volcanoes of the Gulf of Cadiz. Geochim. Cosmochim. Acta 2006, 70, 5336–5355. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).