Abstract
This paper presents an analysis of the results of the gold enrichment process from a gravel deposit using a technological installation equipped with a separator with an adhesive coating. In the separation process, natural waxy substances (patented by Andrzej Ciechulski—Golden Dust) were used as an adhesive coating. Practically, the SFE (Surface Free Energy) of the adhesive substances is the basis of an environmentally accepted enrichment process. Apart from gold, some critical raw materials (CRM) can also be processed from a gravel-type deposit located in the alluvial deposits of the recent rivers and old glacial valleys, which are usually lost in traditional processing of enrichment and purification of gravel and sand. In this work, a new technological installation is presented, equipped with a separator with an adhesive coating. The gold recovery results for the installation industrial operation test were discussed, during which approximately 17 Mg of raw material was processed, recovering over 3.999 g of fine-grained gold. Recovery based on quantitative bulk chemical analysis was calculated close to 90%–95%. The technology can achieve potential for separation of other valuable metals; however, only gold was verified during the tests. The described method is directed at gravel and gold companies dealing with fine, less than 1 mm gold grains, which usually go into clayey waste.
1. Introduction
The interest in gold began in the Chalcolithic period, that is, in the time between the Stone and Bronze Ages (about 4000 y. B.C.). Alluvial gold is easily available and has been recovered in Europe since prehistoric times. In Bulgaria, gold panning on the Zlatica River dates from the period of the Roman Empire. Such gold extraction techniques spread rapidly with the development of civilisation (Figure 1). It is estimated that up to several hundred million people are involved in gold mining on a casual and/or professional basis. Artisanal mining is a major concern for governments and environmentalists in many countries, especially when mercury is used to obtain metallic gold. Recently, analysis of the gold market indicated that up to 20% of the metal is obtained through illegal, environmentally destructive exploitation and extraction. It is enough to compare available satellite maps of rainforest areas to determine the scale of the problem. Artisanal miners identify gold macroscopically and use easily recognisable differences in weight to obtain it (Figure 1); however, gold particles below 100 μm in size become invisible and are discarded as waste. The proposed adhesive method allows the extraction of this gold, which in total can reach up to 50% of the resources of a given sedimentary system, and is usually lost. In the case of a primary deposit that contains refractory gold, using this method no longer makes sense. The use of this method to obtain visible gold from primary deposits that require crushing of the feed requires further experimentation. In these deposits, the presence of other minerals such as sulphides [1], tellurides, and carbonates, graphite creates complicated methods to obtain this metal and extends its residence time in the reactor [1,2,3,4,5,6]. Gravity leaching of concentrate using cyanide also causes significant gold loss due to the presence of similar components as in the primary deposits, and only up to 67.07% of gold can be obtained [7,8]. The fine gold adhesion method may prove to be a solution. Research in this area has been carried out in many countries, e.g., Russia [7], Japan [9], Australia [10], Finland, and Germany [11]. The option of chloride–hypochlorite leaching was proposed by the Russian Academy of Sciences [12]. Meanwhile, the primary method of extracting gold from various ores and concentrates is cyanidation [13,14]. Only a few companies, such as Barick, opt for the slower process with thiosulfates [15].

Figure 1.
Artisanal mining, gold recovery by gravity directly from the riverbed (a), gold grains recovered during manual panning from the Rhine River (b).
This paper involves the development of gold enrichment technology with the use of G-ECO™ adhesive substances (SFE—Surface Free Energy) and newly constructed separators. The G-ECO™ method for the separation of alluvial gold is a simplified process based on adhesion. The technology involves mainly mechanical operations: classification and gravity separation, which result in obtaining the most favourable streams for downstream separation by means of adhesion forces. Adhesive forces are well known and occur on the surfaces of two different substances. This phenomenon is used in many industries. In the case of gold extraction, lanolin was used, which, in general, is a product of fur animals such as sheep or llamas. The history of Jason’s “golden fleece” comes from the oldest accounts of Greek mythology [16], and sheepskins are still used in the Balkans and other places to this day. However, only a special separator design using prepared lanolin gives an extraordinary result of obtaining a rich concentrate from deposits with low gold content. This system is presented in this work.
In addition to the above-mentioned deposits, there are huge deposits of alluvial gold in the world [17] that cannot be recovered due to environmental protection laws. The biggest commercial attractiveness of the proposed solution is environmentally friendly methods that use only natural components and no chemical reagents. The introduction of such technology to the market may play an important role in the recovery of gold in the restricted area, unlocking access to many deposits.
Classic gravity methods have not changed much (Figure 1a,b). The classic pan made of wood is dominant not only in the Far East, where such devices can be found in every home. Other flotation-based methods are practically unattainable due to high investment costs. For the final gold recovery, mercury is used, which, unfortunately, brings environmental pollution and dangerous poisoning associated with it.
Alluvial gold has been recovered in Europe since prehistoric times. There are 70 active gravel pits in the area of SW Poland [18], with similar numbers in Bulgaria and other Central and Eastern European countries. All of these gravel pits contain traces of gold. Different sources of gold are well described in the monograph by Kozłowski and Mikulski entitled “Gold in Poland” [19]. Approximately 7,000,000–8,500,000 Mg of gravel is being extracted from these pits [18]. Gold concentrations have been defined as off an economic balance, usually below 0.15 ppm. Fine gold grains are dominant in sediments with a medium energy below the 0.250 mm fraction [20].
The total output of the five gravel pits analysed in Lower Silesia (PL) is 1,957,000 Mg of annual extraction (Institute 2025). Clay waste is usually 10% of the primary feed, which equates to 195,700 Mg of waste containing gold and heavy mineral fractions from REE. The Au content in gravels and sands varies from 0.05 to 15.0 ppm; the average is 0.1–0.2 ppm. In the screened waste, the fraction below 0.5 mm is present at the level of 1 ppm. The amount of Au to be obtained is at the level of 196 kg/year. In total, the results from all Lower Silesian gravel pits can give a result close to 1 ton. In Bulgarian and Romanian deposits, the values range from two to 10 times higher, depending on the location of the deposit.
2. Materials and Methods
Material from a gravel pit near Złotoryja was used for the first enrichment laboratory test. The feed used in this project was raw, unclassified material. The size of the supplied rock fragments reached 10 cm. The sand point of the feed varied between 40% and 60%. The feeds used in the other 2 projects were pre-screened on 20 mm sieves. In addition to quartz, the following minerals were identified in the sandy fraction using optical microscopy techniques: magnetite, ilmenite, zircon, monazite, rutile, hematite, garnet, pyroxene, amphibole, cassiterite, mica plates (muscovite, biotite, and chlorite), carbonates, native Au, and Pt–Fe. In addition, the volume of waste is also an important factor, because that is where the fine gold is concentrated. WD-XRF ZSX Primus II Rigaku spectrometer, (lamp Rh), Electron Micro Probe—Jeol SuperProbe JXA-8230, Japan, was used to complete bulk quantitative analysis of the obtained concentrate (Critical Elements Laboratory of the Faculty of Geology, Geophysics, and Environmental Protection of the AGH University of Science and Technology in Krakow).
For approximately 40 years, many inventors and companies have been working on developing adhesive technology to separate gold grains from natural ores. The first patent applications in this field date back to the 1990s. These applications involved coatings obtained from natural bitumen. To date, none of these methods has been successful. Another promising material with affinity for gold and other precious minerals is silicone, but silicone-based methods have also been unsuccessful. A material for the industrial adhesive method of gold separation in an aqueous environment must possess three properties in addition to permanent hydrophobicity: an affinity for valuable minerals, sufficiently high adhesive forces, and resistance to the anti-adhesive and erosive effects of the aqueous ore suspension flowing through industrial separators.
In 2017–2019, work on silicone coatings was conducted at the Industrial Chemistry Institute in Warsaw, where European silicones were created in 1945. Paweł Rościszewski, a parallel inventor of silicones in Europe [21,22,23] (along with the American James Wright), participated in the research of the G-ECO team for a year. During the work, over 500 syntheses were performed, resulting in a version of silicone characterised by high selectivity and adhesively binding to gold grains up to approximately 800 microns in size.
Unfortunately, the silicone did not withstand the anti-adhesive effect of water; the functional groups of the bonds underwent polarity reversal and released the gold grains after approximately 20 min.
In 2018, the AGH University of Kraków joined this work. The first materials have been published in conference materials.
In 2019, a prototype separator used lanolin (a protective wax found in sheep’s wool), which was then discovered to be useful for gold separation in industrial mining. It is well known in the pharmacy industry. In ancient times, the sheepskins were used in mountain streams in central Europe for gold recovery. However, the use of lanolin removed from wool and its application to an industrial mining separator occurred only in the G-ECO project. It turned out that the effectiveness of lanolin in the gold separation process is incomparably greater than that of other previously tested coatings. The most important properties of lanolin are high selectivity, very high adhesion forces, and appropriate rheological properties resulting in permanent binding of gold grains up to 2 mm in size to the coating, as well as resistance to the anti-adhesive and erosive effects of an aqueous ore suspension for over 100 h.
In the period of 2020–2021, research was conducted to determine the characteristics of an adhesive industrial separator with lanolin coatings. This work was primarily carried out using column separators. As a result of this work, the desired characteristics of flow turbulence, the magnitude of the forces and contact angles between the gold particles and the coating necessary for the grains to overcome the tension layers, as well as the operating temperature ranges of the coating, and the influence of these temperatures on the selectivity parameter and the adhesion forces (sizes of adsorbed grains) were documented.
In total, 3 tests were performed, one in the laboratory scale, and two next in active open pits. Semi-industrial tests were carried out in the Osetnica pit, 20 km north of Złotoryja, and the Byczeń pit near Kamień Ząbkowicki. Detailed descriptions of the tests are provided in Section 2.1. The final tests were carried out on 17 May 2024 (shown in Figure 2). The technological system is shown schematically in Figure 3.

Figure 2.
Adhesive separator in operating position: (a) one of the separating decks, (b) one of the decks used in gold separation.
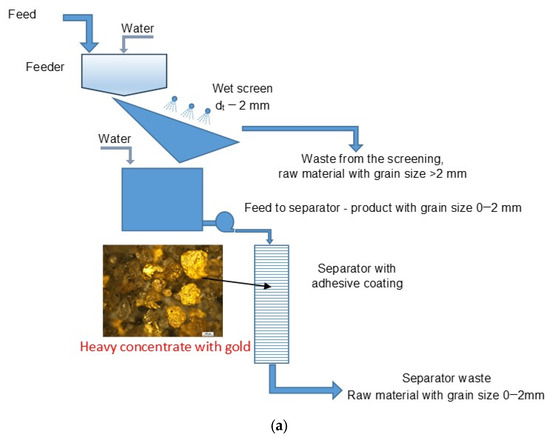

Figure 3.
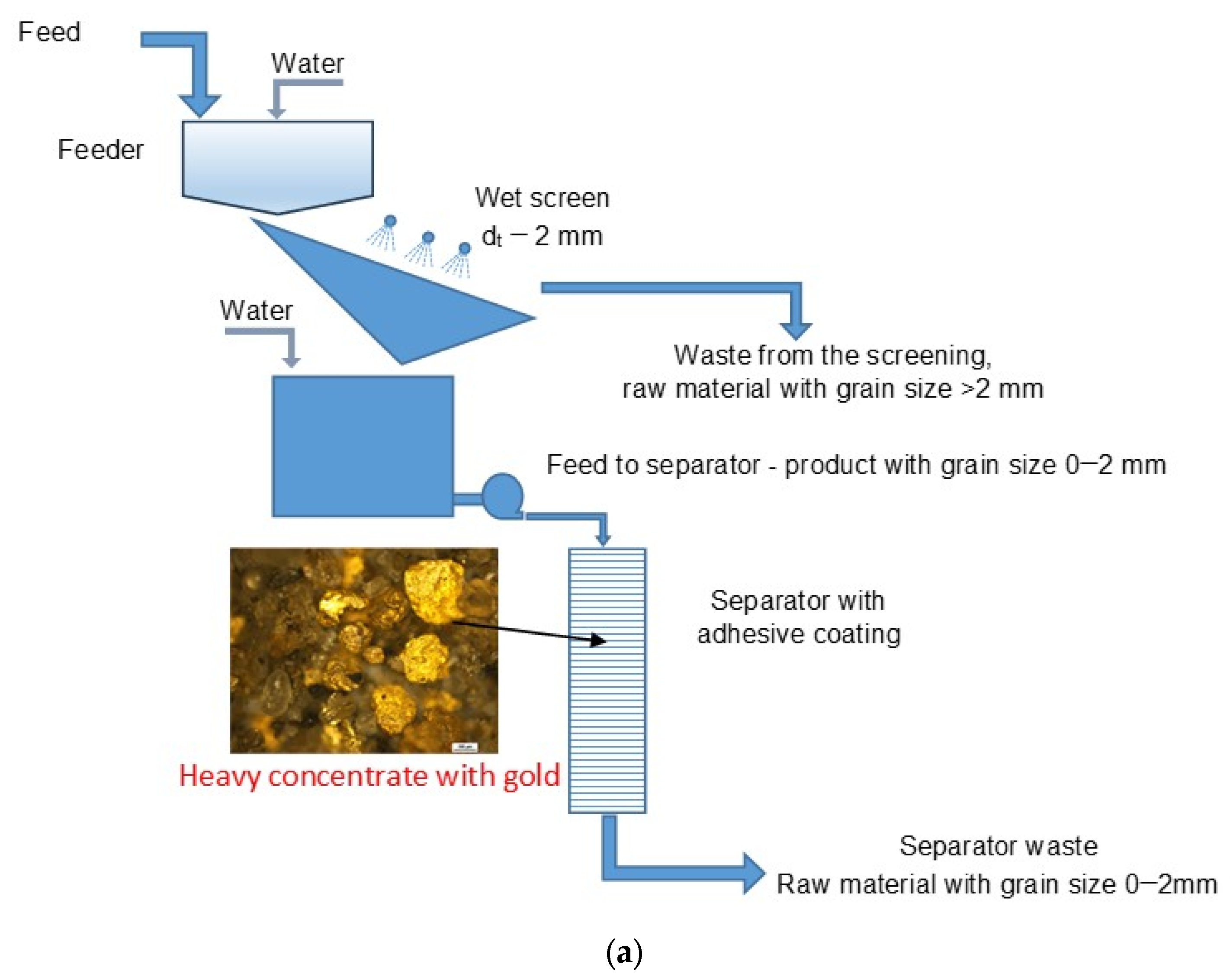
(a) Qualitative diagram of the technological system of the gold recovery installation equipped with an adhesive separator used in all tests. (b) The field system with a screen and a separator.
The material (test no 3) intended for enrichment came from a Byczeń gravel pit near the town of Kamieniec Ząbkowicki. The material collected on the heap, Figure 4, was fed for processing to the installation by a backhoe loader to a feeder, to which an appropriate amount of water was also directed to obtain the desired density of the feed to the separator.

Figure 4.
Sandy feed material from the Nysa Klodzka, Byczeń gravel deposit with the randomly oriented sampling points, 0.5 m deep.
The processing of raw materials in the installation was carried out from 7 a.m. to 12 p.m., i.e., for 5 h. The suspension flow measurements through the separator, which were taken every 60 min, were 17.40; 17.60; 18.44; 16.37; 17.36 m3/h, respectively, giving a volumetric suspension processing of 87.17 m3. The measured suspension density was 1117.3 kg/m3, which means a dry mass content in 1 m3 of suspension of approximately 190.5 kg, which gives a dry mass processing of the raw material by the separator during the test of 16.6 Mg, that is, a separator efficiency of approximately 3.3 Mg/h of dry mass. The specified efficiency of the separator is, of course, lower than that of the installation, which results from the fact that the 0–2 mm grain class is directed to the separator. The amount of material that was processed in the installation is approximately 17.17 Mg (value calculated on the basis of the determined output of the lower screen product that constitutes the feed to the separator).
Due to summer conditions, the temperature of the suspension directed to the installation was monitored. The water addition was controlled to obtain a suspension density of approximately 1100 g/dm3. The suspension temperature measurements directed to the separator were 16.5 °C (7:00 a.m.), 18.0 °C (10:30 a.m.), and 19.0 °C (12:00 p.m.). During the tests, a sample of the feed and waste was taken. The feed sample was taken pointwise from the accumulated heap (Figure 4), while the waste sample was taken directly under the philtre approximately every 60 min of the installation’s operation.
2.1. Open Pit near Złotoryja (First Test)
The material intended for enrichment came from a gravel pit near Złotoryja. The amount of material processed in the installation was calculated to be 8 Mg. According to the described quantitative assessment methodology, as a result of the processing of raw material from Złotoryja in the amount of approximately 8 Mg, 0.70072 g of gold was obtained. The gold content in the feed was close to 0.1 g/Mg, which means a recovery efficiency of approximately 90% (the amount of processed feed was approximately 8 Mg × 0.099 g/Mg = 0.792 g of gold in the processed feed). The determined bulk density of the raw material (feed for processing) was approximately 1.56 Mg/m3. The calculated Au content in the concentrate was 2.260388 g. The description of the calculations concerning the determination of the gold content in the processed material was corrected to 0.792 g; the recovered gold mass in the separator was 0.70072, which gives the yield = 100 × 0.70072/0.792 = 90%.
2.2. Open Pit in Osetnica (Second Test)
The amount of material feed processed in the installation was calculated to be 8 Mg. The first tests running with common silicon have brought surprisingly positive results and were the beginning of the search for a more efficient substance with adhesive properties; however, due to problems with its removal from the separator surface, it was rejected. The testing was focused on an industrial type of lanoline that can be easily removed from the separator using hot water.
2.3. Open Pit in Byczeń (Third Test)
The amount of material processed in the installation was 16.6 Mg. The feed used in this project was selected from the deposit section characterised with a high sand point greater than 97% (Figure 4).
After completion of the installation test, the coating, together with the recovered gold grains and other minerals were manually removed with a spatula from the individual separator decks due to time and simplified form, and the stage of separating the recovered grains from the coating was transferred to the AGH laboratory—Figure 5. This coating removal resulted in leaving a few percent of the extracted concentrate in the separator at the edges of the holes. Under industrial conditions, it is proposed to remove the concentrate from the separator decks using technological hot water (90 °C), which dissolves the adhesive substance.

Figure 5.
Removal of the coating with absorbed gold and other raw materials from the separator deck (a), close-up of a deck element (b).
Another important issue for the final design of the monolithic separator is determining the distance between the individual decks. During the test, the separator had partitions of two dimensions: 6 and 8 mm. The partitions were used alternately for the individual decks, i.e., between 1 and 2 the partition was 6 mm, between 2 and 3 the partition was 8 mm, between 3 and 4 the partition was 6 mm, etc.
The tests for the S1 sample to verify the results obtained were also carried out by WD-XRF ZSX analysis in the laboratory of the Faculty of Geology, Geophysics and Environmental Protection of the AGH University of Krakow. This analysis was performed for a selected representative sample S1P1 (approximately 5 g). The samples were fine powder. Equipment: WD-XRF spectrometer ZSX Primus II Rigaku (Rh lamp). Measurement methodology: The chemical composition of the samples was determined by the X-ray fluorescence method (WD-XRF). A qualitative analysis of the spectrum was performed by identifying spectral lines and determining their possible coincidences. On this basis, analytical lines were selected. A semi-quantitative analysis was developed using the SQX calculation programme (fundamental parameter method). The analysis was performed in the fluorine–uranium (F–U) range, and the contents of the elements determined were normalised to 100%.
Quantitative chemical analysis of gold was performed at Bureau Veritas, Canada, analysis coded F501 and FA330. For the feed, the analytical methodology was different (analysis coded FA330), specifically the Acme FA350-Au method (50 g feed). Geochemical sampling is widely discussed in publications [24].
Chemical analyses of gold at the spot were performed using a Jeol SuperProbe JXA-8230 microprobe at the Critical Elements Laboratory of the Faculty of Geology, Geophysics, and Environmental Protection of the AGH University of Science and Technology in Krakow. The analyses were performed in wavelength dispersion mode (WDS) at an accelerating voltage of 20 kV and a probe current of 40 nA, with a focused beam diameter of 1 μm. The following standards and measurement lines were used: AgLα (100%), AuMα (100%), SbLα (Sb2S3), SKα (FeS2), PbMα (PbS), HgMα (HgS), CuKα (CuFeS2), BiMα (10%), and AsLα (GaAs). Original Jeol ZAF procedures were used for the final correction of all the elements studied. Calculated statistical parameters, such as mean content, standard deviation, and coefficient of variation in WDS measurements, help in the interpretation of quantitative WDS data.
3. Results
3.1. Feed
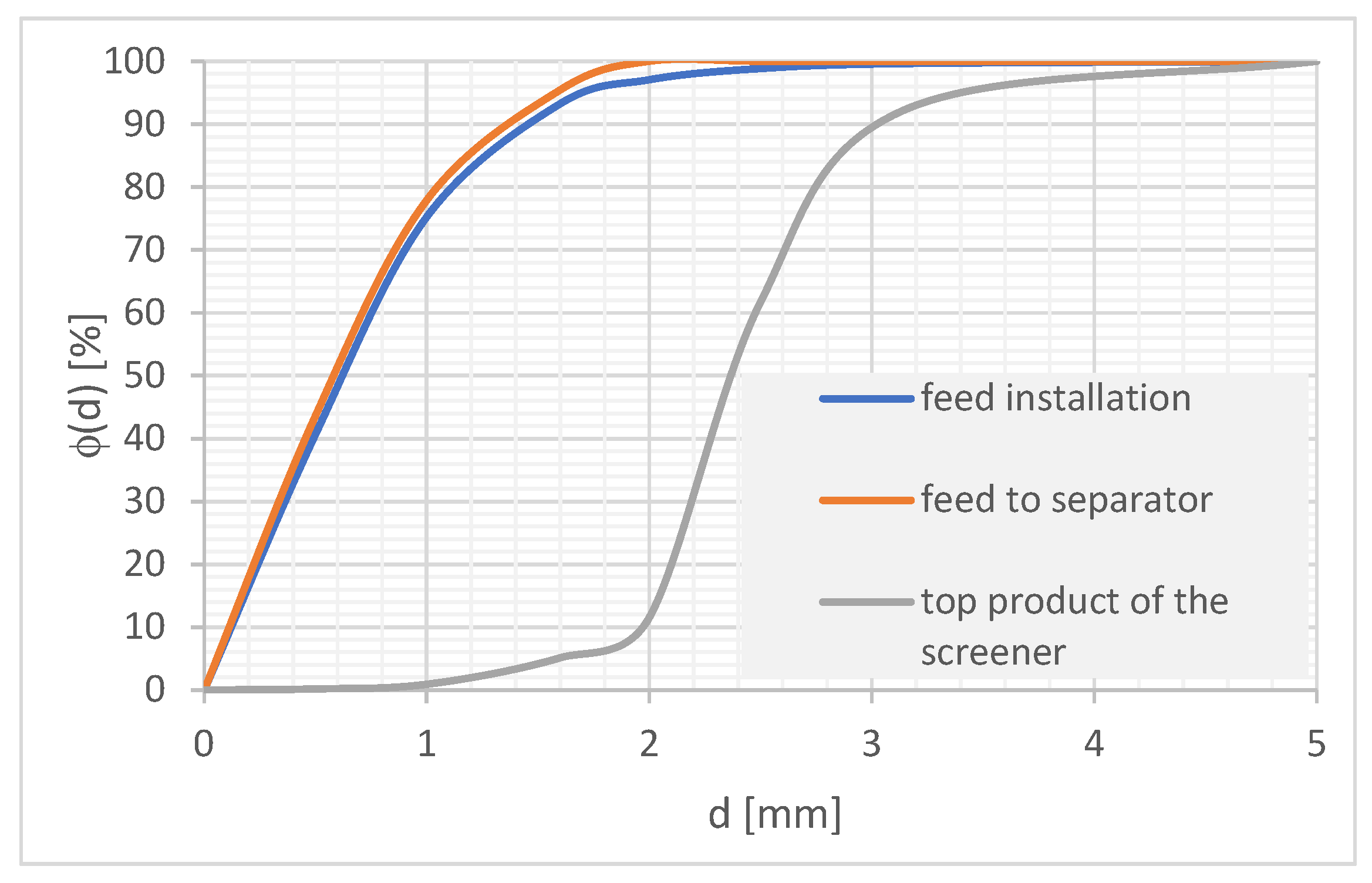
The feed to the separator was the lower product of the screen with a grain size of 0–2 mm. Ensure that an appropriate fine product is directed to the separator as a result of the need to ensure its patency. Larger grains can block the holes in the separator plates and the space between the plates, causing no flow of suspension. The grain composition of the feed and the upper product of the screen is shown in Figure 6. The content of the particles that constitute the stream directed to the separator in the feed was approximately 97%.
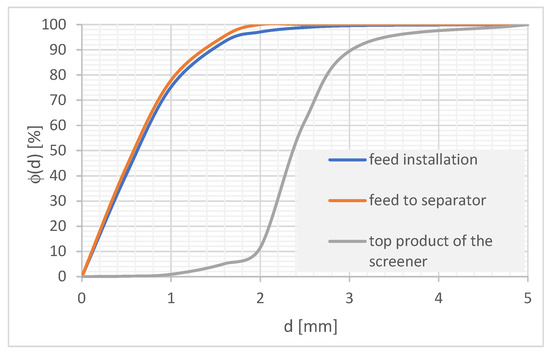
Figure 6.
Grain composition of the tested feed.
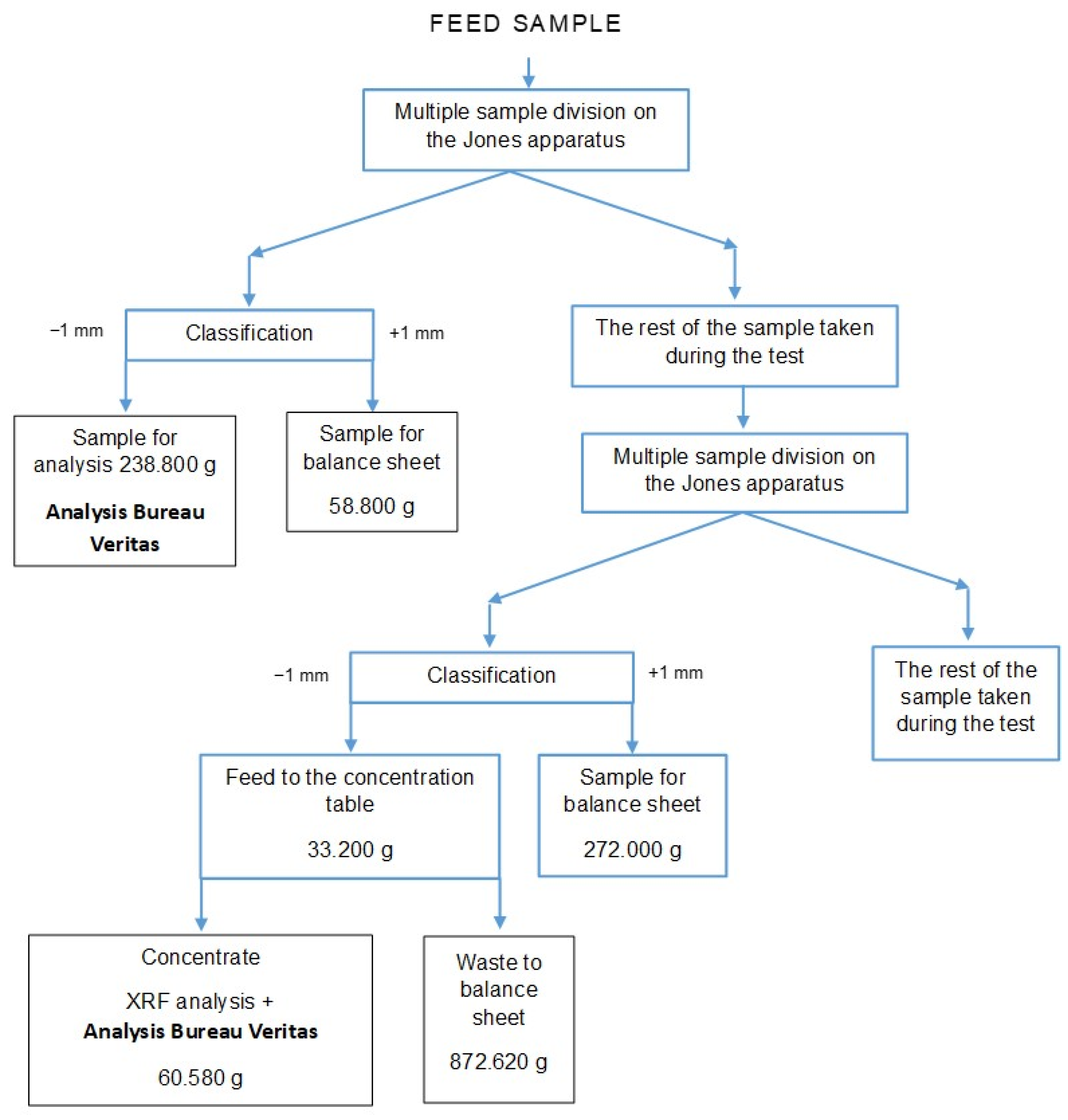
The results of the initial estimated gold evaluation by its concentration on the concentration table indicate its absence in the feed, which is not confirmed by the results obtained microscopically and in the accredited Bureau Veritas laboratory. The gold content in the feed sample obtained in the accredited Bureau Veritas laboratory in Canada indicates its very low content of 4 ppb. The determination performed was carried out for a separated part of the feed sample, i.e., grain class below 1 mm (238.8 g), taking into account the remaining balance part of the sample (58.8 g). The gold content in the feed material is 3.21 ppb (Figure 7).
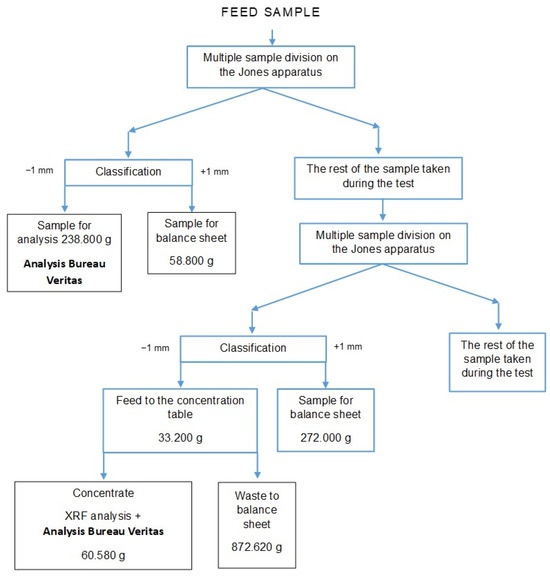
Figure 7.
Schematic diagram of sample isolation for analysis of the gold content in the feed to the installation.
In order to verify the representativeness of the sample taken and the methodology for determining the gold content in the feed, the concentrate obtained from the shaking table was sent to the accredited Bureau Veritas laboratory. The gold content in the 60.58 g sample was obtained at 3.907 ppm—Figure 7. Taking into account the remaining part of the balance sheet of the sample, i.e., 1144.62 g (872.62 g waste from the concentration table and 272.00 g grain class +1 mm) to assess the gold content in the feed according to the proposed methodology, we obtain a gold content in the feed of 0.19 ppm, i.e., 0.196 g/Mg, which gives a mass in the processed amount of raw material of 3.36532 g. The specific amount of gold in the feed using the proposed methodology is smaller than the recovered mass on the separator. Assuming that the concentration of gold on the concentration table has a specific efficiency (this results from the fact that each processing operation is carried out with a specific efficiency), the balance should also include, in addition to the gold content in the concentrate from the concentration table, the gold content in the waste, the amount of which depends on the efficiency/effectiveness of the operation). Assuming high gold concentration efficiency on the concentration table of 90%, this gives us a gold mass in the feed of 0.262985 mg (0.236686 mg concentrate + 0.0262985 mg waste from the shaking table), the gold content in the feed would then be 0.000021820819%, i.e., 0.21820819 g/Mg, which gives a mass in the processed amount of raw material, i.e., 17.170 Mg equal to 3.74663463 g, i.e., smaller than the mass recovered in the separator (Figure 7). For the assumed high gold concentration efficiency from the feed on the concentration table for the balance purposes, we obtain a separator efficiency of 98.14%. For the efficiency of concentration on the concentration table at a more realistic level of 82% (18% losses in waste from the table), the efficiency of the separator is already 97.26%. Assuming that the efficiency level of the concentration table was 80%, the gold recovery would be 94.89%, while for an efficiency of 78%, the percentage of gold recovered would be 92.51%.
The procedure carried out to determine the gold content in the feed sample shows that this is an extremely important issue that, if appropriate procedures are not developed, may result in erroneous results. Preliminary tests should provide information on the presence of gold in the gravel-sand deposit.
3.2. Concentrate
The concentrate obtained is composed of native gold (up to 10%), zircon (15%), ilmenite (15%), magnetite (up to 15%), monazite (up to 4%), quartz (up to 25%), feldspar, pyroxene, amphibolite, muscovite, chlorite, biotite, garnet, spinel, and carbonates. The bulk chemical composition of the concentrate of deck S1 (Table 1) shows an interesting composition of elements like Ti, Zr, In, Y, Yb, and Hf.

Table 1.
Chemical composition of concentrate received from deck S1, XRF analysis, sample 4.75 g, Byczeń concentrate.
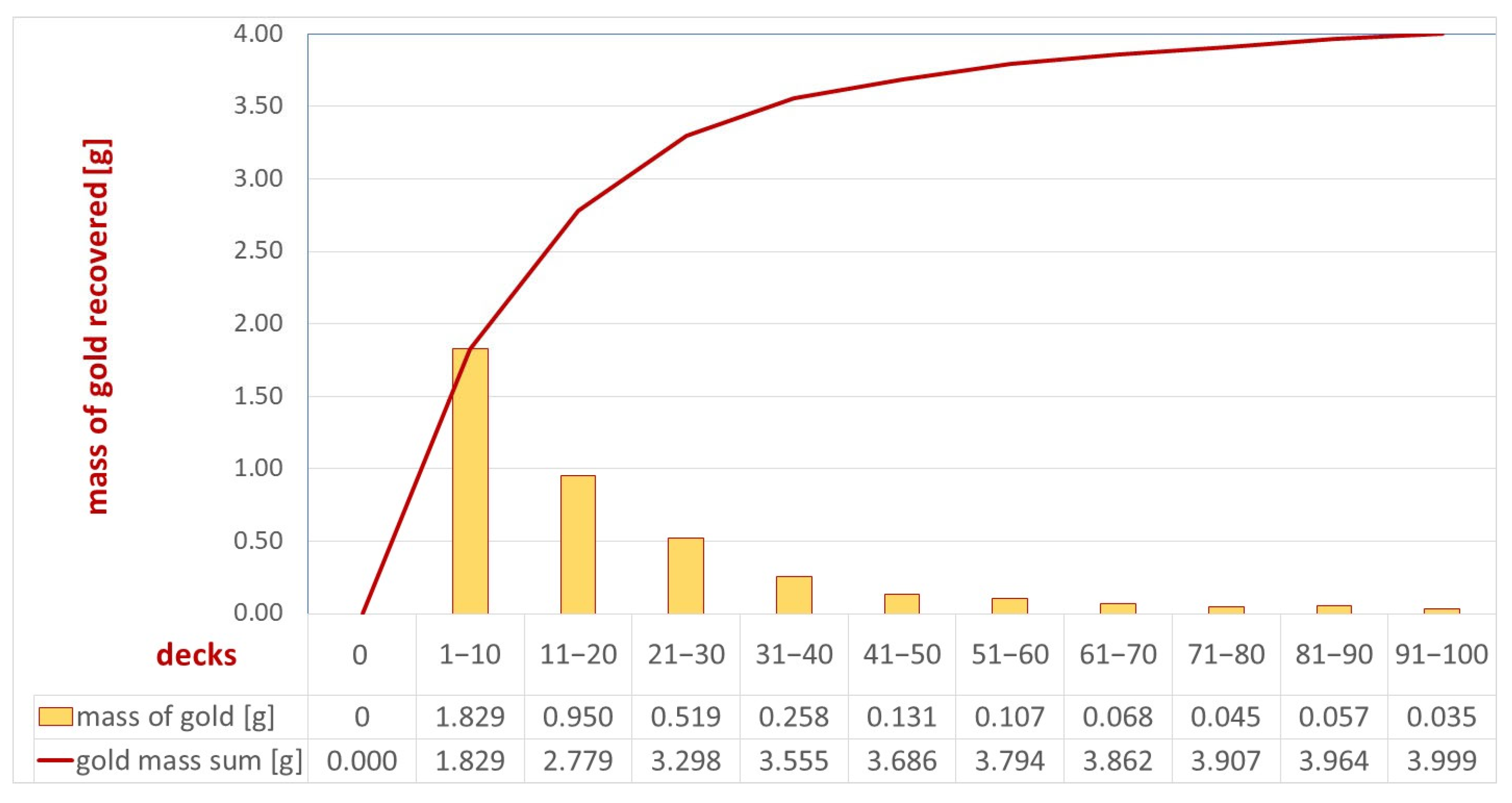
To enhance the recovery efficiency on individual separator decks, the coating was improved by combining ten decks into one product. The separator was equipped with 100 decks, resulting in ten concentrates, allowing the efficiency evaluation to depend on the number of decks installed. The products successively collected from the decks were designated S1—coating from decks 1–10; S2—coating from decks 11–20; S3—coating from decks 21–30; S4—coating from decks 31–40; S5—coating from decks 41–40; S6—coating from decks 51–60; S7—coating of decks 61–70; S8—coating from decks 71–80; S9—coating from decks 81–90; and S10—coating from decks 91–100. These were the separator sections. The material obtained was then fired in a muffle furnace at a temperature of 500 °C to remove adhesive substances. The solid phase concentrates obtained in this way (Figure 8 and Figure 9) were subjected to analytical tests, i.e., evaluation of both macroscopic and quantitative gold content according to the methodology described in this field. A cumulative curve of gold recovery is shown in Figure 10.

Figure 8.
Heavy concentration of S1 material from the first 10 docks with visible gold particles (yellow).

Figure 9.
Au grains (yellow) in the heavy concentrate, light reflected, Byczeń concentrate, level S1.
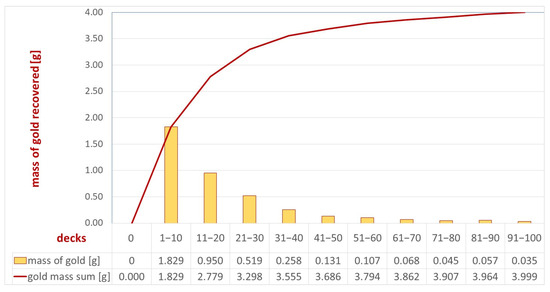
Figure 10.
Cumulative curve of gold grains recovered from the separator from the Byczeń gravel-sand deposit.
The composition of gold based on WDS point analysis in alluvial deposits is similar to that from other sources. This is related to the environment of sediment deposition and, of course, to the source. In addition to Permian deposits, the gold that occurs in the SOS system (Secondary Oxidation System) is of very high quality (98%–100% Au) [15]. For example, the average Ag content in the analysed gold grains is 8.789%, while the average Au content is at the level of 89%. The maximum silver content is 42.4% (electrum). The coefficient of variability is very high and amounts to 120% (for n = 51). In addition to Ag, Te, and Bi, admixtures have been found in gold. This corresponds to data from the literature on the occurrence of native Au in the Sudetes [20,25,26].
4. Discussion
The experiments carried out using the phenomenon of physicochemical adhesion, i.e., the force of adhesion of the surfaces of two different materials to each other, resulting from the presence of attractive intermolecular forces, can be used in the process of enriching alluvial gold deposits. The enrichment factors obtained were over 95% and are a good indicator of the positive economic factor (Table 2, Figure 10).

Table 2.
Overall gold balance after obtaining Canada’s results for combined samples S3–S6 (S3 + S4 + S5 + S6) and samples S7–S10 (S7 + S8 + S9 + S10).
It was also confirmed during optical examination of the concentrates obtained (Figure 8 and Figure 9). The presence of significant amounts of Zr, Hf, Ti, Sn, and Eu requires further research and testing. The presence of these elements and the possibility of obtaining their concentrates could bring additional economic benefits.
Based on the obtained results, the functional dependence of gold recovery as a function of the active surface of the separator was determined, enabling the calculation of the gold recovery efficiency depending on the separation surface for the purposes of design optimisation.
For the purpose of determining the recovery efficiency as a function of the separator surface area, it was assumed that the feed to be enriched contained 4.11 g with an average content 0.2395 g/Mg of gold, in accordance with the balance to determine the gold content in the feed at the assumed recovery efficiency of gold on the concentration table of 82% (as described in Section 3.2).
The calculations were performed using the Statistica non-linear modelling module. The modelling was performed using the non-linear least-squares method utilizing the Levenberg–Marquardt algorithm. When using the least-squares criterion, the Levenberg–Marquardt algorithm (for a detailed description of this algorithm, see Levenberg [27]; Marquardt [28]; and Moré [29]) is very efficient for estimating parameters in linear and non-linear regression models. For large datasets, when using the least-squares criterion, this method of fitting non-linear regression models is recommended. Using the user-specified loss of function [(observed-predicted)2], we may get differences in the results, but this difference is negligible, and the results are the same for 5–8 significant positions.
The general model equation is defined as
V1 = 100 × (1 − Exp(−b1 × V2)),
Dependent variable: recovery
Independent variables: one
Loss function: least squares method, Levenberg–Marquardt algorithm
(data of the non-linear modelling)
Final value: 80.60157455
Share of explained variance: 0.99100964 R = 0.99549467
According to Equation (1), the form of the estimated Equation (2) is
gold recovery (V1) = 100 × (1 − Exp(−b1 × separation surface [m2 of active decks] (V2)),
Dependent variable: gold recovery
Confidence level: 95.0% (alpha = 0.050)
The estimated value of the b1 coefficient is shown in Table 3.

Table 3.
Results of the estimated value of the b1 coefficient.
The standard error of estimation for the specified non-linear equation was SEE = 2.839.
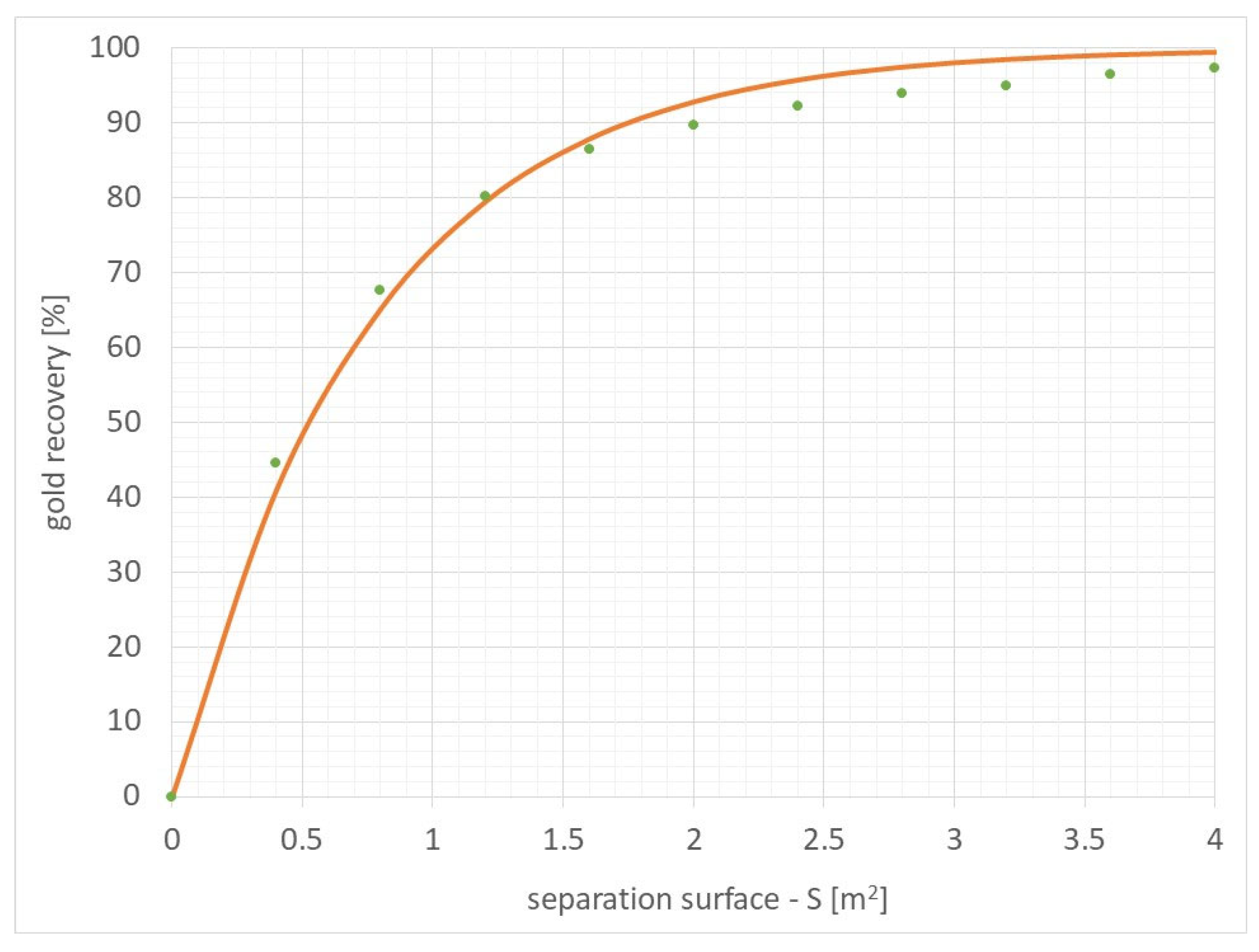
Figure 11 presents the results of the specific functional dependence of gold recovery as a function of the active surface of the separator, along with the measured points.
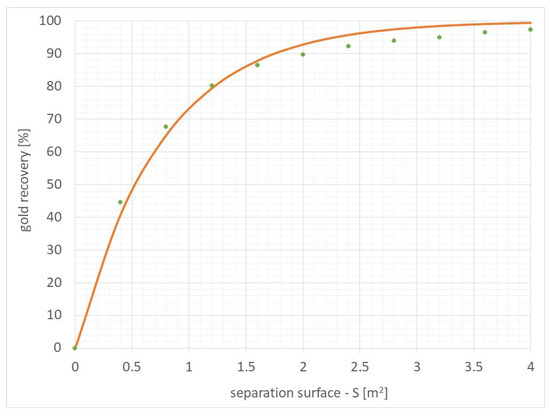
Figure 11.
The functional relationship of gold recovery as a function of the active surface of the separator (gold recovery = 100 × (1 − Exp(−1.320152 × separation surface [m2])) along with the measured points.
The determined functional relationship and the results obtained indicate that for the active surface of the separator of approximately 1.8–2 m2, a high level of gold recovery of approximately 90% is achieved.
Determining the separation efficiency requires developing a detailed methodology for sampling the feed and waste for testing due to the small amount of gold in the feed, i.e., a dozen or so/several hundredths of a gram per Mg of raw material. For the analysed feed, it was approximately 0.2446 g/Mg, which gives a much lower content in the waste of approximately 0.012 g/Mg with a recovery efficiency of 95% in the separator. The assumed high efficiency results from the analyses for these products conducted as part of this work and an attempt to establish an effective analytical methodology for determining the gold content in the sample. The results of the calculations of the sample mass for testing for such low contents, i.e., 0.25 g/Mg, indicate that, assuming the size of a single spherical gold grain of 0.1 mm, the number of gold grains in a 100 g sample would be about two grains. Therefore, ensuring an appropriate representative sample requires both an appropriate sampling methodology and conducting a test for a larger raw material volume.
The analysis of the gold recovery efficiency on individual separator decks (the tested separator was built of 100 decks) indicates a decreasing efficiency on subsequent decks. The highest efficiency is achieved in the initial decks; specifically, the first 30 decks accounted for more than 82% (3.298 g) of all gold recovered on the separator. Across the last 10 decks, less than 1% (0.035 g) was recovered, indicating that a further increase in the number of decks may not be economically viable. The number of decks should be determined by determining the durability of the coatings and their efficiency over time. In the event of a decrease in the efficiency of the first decks over time, the efficiency of the separator may be determined by subsequent layers.
Due to the need to direct a stream of specific granulation to the separator, the technology will use mechanical operations, mainly classification and gravity separation, in order to obtain the most advantageous streams for separation using adhesion forces. It requires clarification in this respect in order to select the most advantageous technological system depending on the specific efficiency of the separator in terms of adhesion forces, in particular with respect to the largest gold grains, depending on the separator operating parameters: suspension flow rate, thickness of the adhesive coating, durability of the coating over time, suspension temperature, as well as contact forces of gold grains with the coating, contact angles of grains with the coating preferably 90 degrees, flow turbulence causing contact of gold grains with the coating, one gold grain for several hundred thousand sand grains.
5. Conclusions
The results of the adhesive separator tests carried out for alluvial gold ore confirm the effectiveness in gold adsorption on the lanolin-based adhesive coating. The initial weight of the raw alluvial material from the Nysa Kłodzka River was 17.17 Mg. After screening on a 2 mm sieve, the feed weight was reduced to 16.6 Mg. This amount was used as the feed for the gold separator. The heavy fraction obtained in the separator with an adhesive surface contained 3.999 g of gold. This allowed the calculation of the gold content in the feed, which was 0.2446 g/Mg. The negligible amount of gold found in the waste indicated a very high efficiency of the adhesive separator, greater than 95%.
Gold recovery technology using G-ECO™ adhesive separators (SFE—Surface Free Energy) from the low-grade ores is a low operating cost method that requires further study in terms of application in industrial conditions, the flow stream parameters through the separator, and the design of the installation to remove the coating containing gold, and separate gold from the coating. The separator has no moving parts, and the preparation of the stream directed to gold recovery within the installation requires only a classification operation on the sieve screen and a pump, which can be eliminated if gravity feeding is utilized.
This technology is a remedy to two operational problems: the impossibility of recovering gold grains smaller than several dozen microns with grain-shape-dependent gravity-based technologies, and the prohibition on the use of chemical methods, e.g., cyanidation. The adhesive separator technology can also be applied to the separation of gold from waste as well as to detrital alluvial material with low gold content, making it more profitable than conventional methods.
Author Contributions
Conceptualization, D.F., A.P. and A.C.; methodology, D.F. and A.P.; validation, D.F. and A.P.; formal analysis, D.F.; investigation, D.F.; resources, A.C.; data curation, D.F.; writing—original draft preparation, D.F.; writing—review and editing, A.P.; visualisation, D.F.; supervision, A.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
The authors would like to express their sincere gratitude to the anonymous reviewers for their contribution and a number of valuable substantive comments. This study was partially financed by the AGH University of Science and Technology of Kraków through the State subsidy. The authors have reviewed and edited the output and take full responsibility for the content of this publication.
Conflicts of Interest
Author Andrzej Ciechulski was employed by the company Golden Dust. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Bas, A.D.; Koc, E.; Yazici, E.Y.; Deveci, H. Treatment of Copper-Rich Gold Ore by Cyanide Leaching, Ammonia Pretreatment and Ammoniacal Cyanide Leaching. Trans. Nonferrous Met. Soc. China 2015, 25, 597–607. [Google Scholar] [CrossRef]
- Ahtiainen, R.; Lundström, M.; Liipo, J. Preg-Robbing Verification and Prevention in Gold Chloride-Bromide Leaching. Miner. Eng. 2018, 128, 153–159. [Google Scholar] [CrossRef]
- Celep, O.; Serbest, V. Characterization of an Iron Oxy/Hydroxide (Gossan Type) Bearing Refractory Gold and Silver Ore by Diagnostic Leaching. Trans. Nonferrous Met. Soc. China 2015, 25, 1286–1297. [Google Scholar] [CrossRef]
- Owusu, C.; Mensah, S.; Ackah, K.; Amankwah, R.K. Reducing Preg-Robbing in Carbonaceous Gold Ores Using Passivative or Blanking Agents. Miner. Eng. 2021, 170, 106990. [Google Scholar] [CrossRef]
- Rees, K.L.; Van Deventer, J.S.J. The Role of Metal-Cyanide Species in Leaching Gold from a Copper Concentrate. Miner. Eng. 1999, 12, 877–892. [Google Scholar] [CrossRef]
- Tremolada, J.; Dzioba, R.; Bernardo-Sánchez, A.; Menéndez-Aguado, J.M. The Preg-Robbing of Gold and Silver by Clays during Cyanidation under Agitation and Heap Leaching Conditions. Int. J. Miner. Process. 2010, 94, 67–71. [Google Scholar] [CrossRef]
- Fedotov, P.K.; Senchenko, A.E.; Fedotov, K.V.; Burdonov, A.E. Study of enrichability of low-sulfide ores. Obogashchenie Rud 2020, 15–21. [Google Scholar] [CrossRef]
- Tsitos, I. Gold leaching: Can we replace cyanide at scale? Mining Magazine, September 2025; pp. 46–47. Available online: www.miningmagazine.com (accessed on 21 October 2025).
- Altansukh, B.; Haga, K.; Huang, H.-H.; Shibayama, A. Gold recovery from waste printed circuit boards by advanced hydrometallurgical processing. Mater. Trans. 2019, 60, 287–296. [Google Scholar] [CrossRef]
- Liddell, K.S.; Adams, M.D.; Smith, L.A.; Muller, B. Kell hydrometallurgical extraction of precious and base metals from flotation concentrates—Piloting, engineering, and implementation advances. J. South. Afr. Inst. Min. Metall. 2019, 119, 585–594. [Google Scholar] [CrossRef]
- Jung, J.; Keller, W. Process and cost improved agitator solutions for bioleach reactors. Solid State Phenom. 2017, 262, 315–318. [Google Scholar] [CrossRef]
- Prokhorov, K.V.; Burdonov, A.E. Chloride-hypochlorite leaching of gold from oxidized ores of the Malmyzhsky deposit. Gorn. Zhurnal 2018, 62–66. [Google Scholar] [CrossRef]
- Bas, A.D.; Larachi, F.; Laflamme, P. The Effect of Pyrite Particle Size on the Electrochemical Dissolution of Gold during Cyanidation. Hydrometallurgy 2018, 175, 367–375. [Google Scholar] [CrossRef]
- Celep, O.; Alp, I.; Deveci, H. Improved Gold and Silver Extraction from a Refractory Antimony Ore by Pretreatment with Alkaline Sulphide Leach. Hydrometallurgy 2011, 105, 234–239. [Google Scholar] [CrossRef]
- Piestrzyński, A.; Pieczonka, J.; Głuszek, A. Redbed-type gold mineralisation, Kupferschiefer, south-west Poland. Miner. Depos. 2002, 37, 512–528. [Google Scholar] [CrossRef]
- Parandowski, J. Mitologia Wierzenia i Podania Greków i Rzymian; Wydawnictwo Poznańskie: Poznań, Poland, 1989; 165p. [Google Scholar]
- McLeod, C.R.; Morison, S.R. Placer gold, platinum. In Geology of Canadian Mineral Deposit Types, Geology of Canada; Eckstrand, O.R., Sinclair, W.D., Thorpe, R.I., Eds.; Natural Resources Canada: Ottawa, ON, USA, 1995; pp. 23–32. [Google Scholar]
- National Geological Institute National Research Institute. The Balance of Mineral Resources in Poland as of 31 XII 2024 r; National Geological Institute National Research Institute: Warsaw, Poland, 2025; 515p, ISSN 2299-4459. Available online: www.pgi.gov.pl/images/surowce/2024/bilans_2024.pdf (accessed on 29 January 2026). (In Polish)
- Kozłowski, A.; Mikulski, S. (Eds.) Gold in Poland, Seria AM Monograph, Tom 2; MSC of the PAS; UW Warsaw and PGI Warsaw: Warsaw, Poland, 2011; 371p. [Google Scholar]
- Muszer, A.; Łuszczkiewicz, A. Mineralogical characteristics of accessory minerals from Osiecznica deposit, SW Poland. Physicochem. Probl. Miner. Process. 2006, 40, 77–88. [Google Scholar]
- Rościszewski, P.; Siek, Z.; Makosik, A. Sposób wytwarzania małocząsteczkowych, reaktywnych żywic silikonowych (Procedure for low molecular reactive silicone resins obtaining). Pat. Pol. 1985, 148, 395. [Google Scholar]
- Rościszewski, P.; Sobiczewski, Z. Synteza bezpośrednia metylochlorosilanów (Direct synthesis of methylchlorsilanes). Przem. Chem. 1954, 33, 515. [Google Scholar]
- Rościszewski, P.; Strojny, T.; Siemieńska, J.; Iwańska, S.; Kosińska, W. Synthesis and investigation of some physical and thermomechanical properties of butylmethylphenylsilicon resins, Abstr. of 6-th Intern. Symp. on Organosilicon Chem. Budapest 1981, s. 151. [Google Scholar]
- Dominy, S.C.; Glass, H.J. Geometallurgical Sampling and Testwork for Gold Mineralisation: General Considerations and a Case Study. Minerals 2025, 15, 370. [Google Scholar] [CrossRef]
- Grodzicki, A. Placer gold in Sudetes Mountains and their foreland. In Gold in Poland; Kozowski, A., Mikulski, S., Eds.; 2011; pp. 191–208. [Google Scholar]
- Wierchowiec, J. Placer gold of East Sudetes and its foreland, Poland. In Gold in Poland; Kozowski, A., Mikulski, S., Eds.; 2011; pp. 209–236. [Google Scholar]
- Levenberg, K. A method for the solution of certain non-linear problems in least squares. Q. Appl. Math. 1944, 2, 164–168. [Google Scholar] [CrossRef]
- Marquardt, D.W. An algorithm for least-squares estimation of non-linear parameters. J. Soc. Ind. Appl. Math. 1963, 11, 431–441. [Google Scholar] [CrossRef]
- Moré, J. The Levenberg-Marquardt Algorithm: Implementation and Theory; Watson, G.A., Ed.; Lecture Notes in Mathematics; Springer: Berlin, Germany, 1977; Volume 630, pp. 105–116. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.