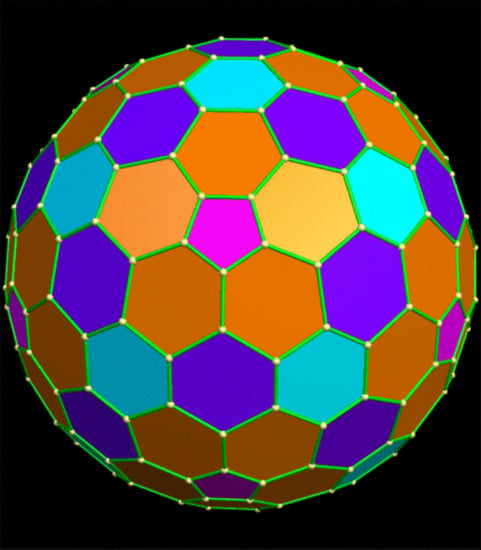
Combinatorics of Edge Symmetry: Chiral and Achiral Edge Colorings of Icosahedral Giant Fullerenes: C80, C180, and C240 †
Abstract
1. Introduction
2. Möbius Inversion Technique with Generalization of Sheehan’s Theorem to All Characters as Tools for the Combinatorics of Edge Groups and Edge Colorings of Giant Fullerenes
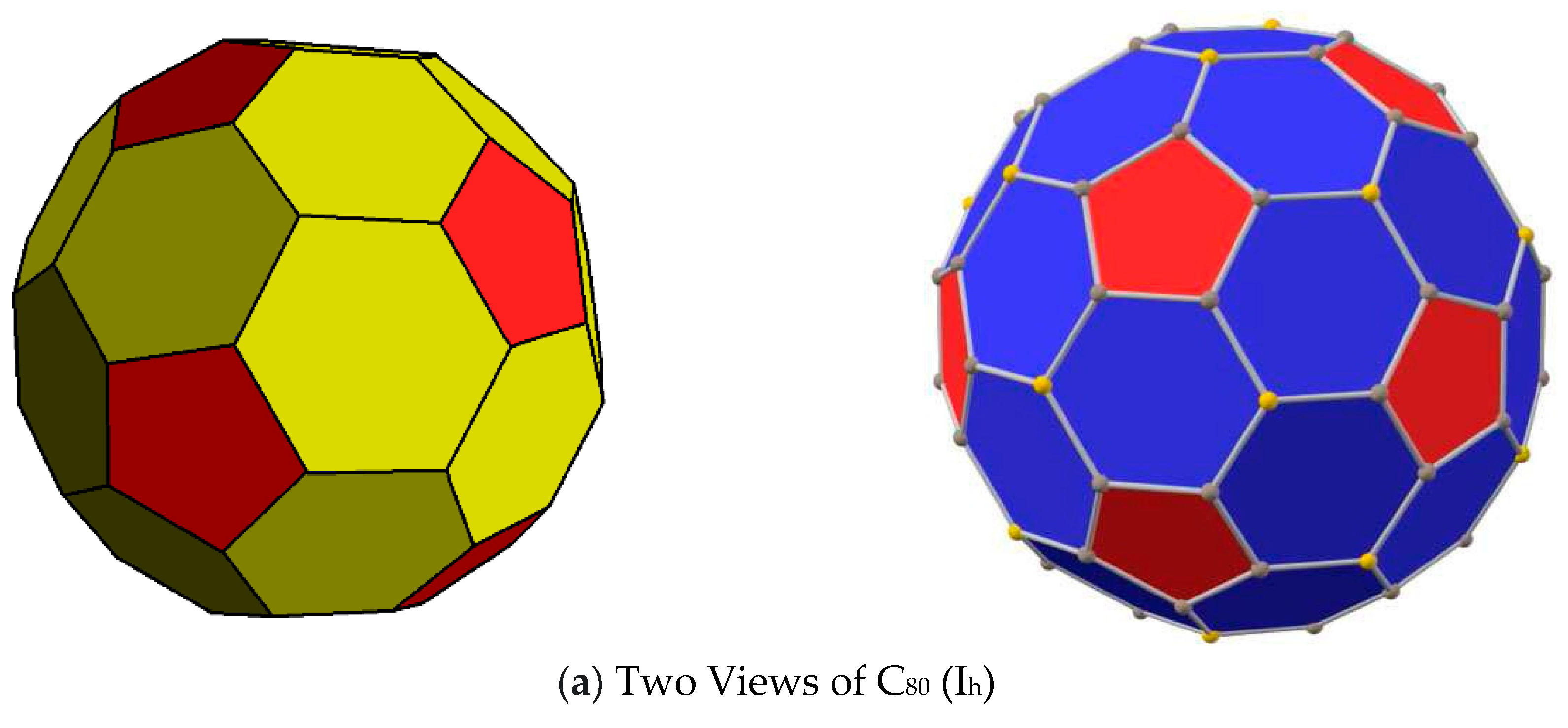
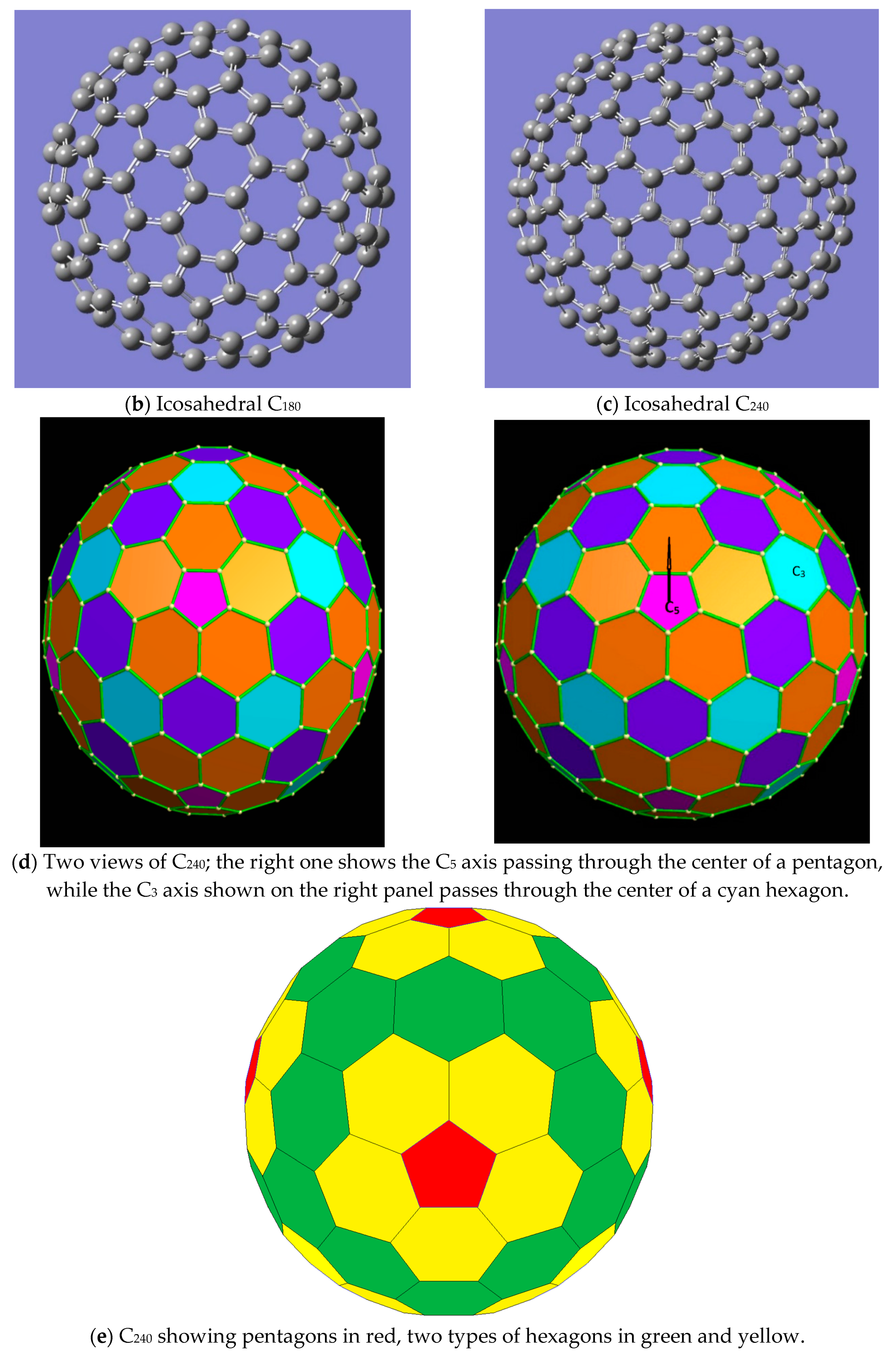
3. Results and Discussions
- (a)
- if all hex–hex edges of C60 are colored with one color (white) and pent–hex edges are colored with multiple colors;
- (b)
- (c)
- if both pent–hex and hex–hex edges are colored with multiple colors from a single set R of colors without making any distinction between the two sets of edges.
- (d)
- if all hex–hex edges are colored with colors from a single set, while all pent–hex edges are colored with colors chosen from a different set of colors, thus making a distinction between the pent–hex and hex–hex edges.
4. Conclusions
Funding
Conflicts of Interest
References
- Ghorbani, M.; Dehmer, M.; Emmert-Streib, F. Properties of Entropy-Based Topological Measures of Fullerenes. Mathematics 2020, 8, 740. [Google Scholar] [CrossRef]
- Ghorbani, M.; Dehmer, M.; Rajabi-Parsa, M.; Emmert-Streib, F.; Mowshowitz, A. Hosoya entropy of fullerene graphs. Appl. Math. Comput. 2019, 352, 88–98. [Google Scholar] [CrossRef]
- Ghorbani, M.; Matthias, D.; Rajabi-Parsa, M.; Mowshowitz, A.; Emmert-Streib, F. On properties of distance-based entropies on fullerene graphs. Entropy 2019, 21, 482. [Google Scholar] [CrossRef]
- Balasubramanian, K. Enumeration of stereo, position and chiral isomers of polysubstituted giant fullerenes: Applications to C180 and C240. Fuller. Nanotub. Carbon Nanostruct. 2020, 2810, 1–10. [Google Scholar] [CrossRef]
- Feng, L.; Hao, Y.; Liu, A.; Slanina, Z. Trapping Metallic Oxide Clusters inside Fullerene Cages. Acc. Chem. Res. 2019, 52, 1802–1811. [Google Scholar] [CrossRef]
- Yu, P.; Shen, W.; Bao, L.; Pan, C.; Slanina, Z.; Lu, X. Trapping an unprecedented Ti3C3 unit into the icosahedral C80 fullerene: A crystallographic survey. Chem. Sci. 2019, 10, 10925–10930. [Google Scholar] [CrossRef]
- Hu, S.; Liu, T.; Shen, W.; Slanina, Z.; Akasaka, T.; Xie, Y.; Uhlik, F.; Huang, W.; Lu, X. Isolation and Structural Characterization of Er@C2v (9)-C82 and Er@Cs(6)-C82: Regioselective Dimerization of a Pristine Endohedral Metallofullerene Induced by Cage Symmetry. Inorg. Chem. 2019, 58, 2177–2182. [Google Scholar] [CrossRef]
- Kroto, H.W.; Heath, J.R.; O’Brien, S.C.; Curl, R.F.; Smalley, R.E. C60: Buckminsterfullerene. Nature 1985, 318, 162–163. [Google Scholar] [CrossRef]
- Kroto, H.W.; Heath, J.R.; O’Brien, S.C.; Curl, R.F.; Smalley, R.E. Long carbon chain molecules in circumstellar shells. Astrophys. J. 1987, 314, 352–355. [Google Scholar] [CrossRef]
- Iijima, S. Helical microtubules of graphitic carbon. Nature 1991, 354, 56–58. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.; Jiang, D.; Katsnelson, M.I.; Grigorieva, I.; Irsov, A.A. Two-dimensional gas of massless Dirac fermions in graphene. Nature 2005, 438, 197. [Google Scholar] [CrossRef] [PubMed]
- Novoselov, K.S.; Jiang, D.; Schedin, F.; Booth, T.J.; Khotkevich, V.V.; Morozov, S.V.; Geim, A.K. Two-dimensional atomic crystals. Proc. Natl. Acad. Sci. USA 2005, 102, 10451–10453. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, K.; Nakahara, N.; Wakabayashi, T.; Suzuki, S.; Shiromaru, H.; Miyake, Y.; Saito, K.; Ikemoto, I.; Kainosho, M.; Achiba, Y. NMR Characterization of Isomers of C78, C82 and C84 Fullerenes. Nature 1992, 357, 142–145. [Google Scholar] [CrossRef]
- Diederich, F.; Whetten, R.L.; Thilgen, C.; Ettl, R.; Chao, I.T.O.; Alvarez, M.M. Fullerene isomerism: Isolation of C2v,-C78 and D3-C78. Science 1991, 254, 1768–1770. [Google Scholar] [CrossRef] [PubMed]
- Simeonov, K.S.; Amsharov, K.Y.; Jansen, M. Chlorinated Derivatives of C78-Fullerene Isomers with Unusually Short Intermolecular Halogen–Halogen Contacts. Chem. A Eur. J. 2008, 14, 9585–9590. [Google Scholar] [CrossRef]
- Simeonov, K.S.; Amsharov, K.Y.; Krokos, E.; Jansen, M. An epilogue on the C78-fullerene family: The discovery and characterization of an elusive isomer. Angew. Chem. Int. Ed. 2008, 47, 6283–6285. [Google Scholar] [CrossRef]
- Diederich, F.; Ettl, R.; Rubin, Y.; Whetten, R.L.; Beck, R.; Alvarez, M.; Anz, S.; Sensharma, D.; Wudl, F.; Khemani, K.C.; et al. The higher fullerenes: Isolation and characterization of C76, C84, C90, C94, and C70O, an oxide of D5h-C70. Science 1991, 252, 548–551. [Google Scholar] [CrossRef]
- Troyanov, S.I.; Yang, S.; Chen, C.; Kemnitz, E. Six IPR isomers of C90 fullerene captured as chlorides: Carbon cage connectivities and chlorination patterns. Chem. A Eur. J. 2001, 17, 10662–10669. [Google Scholar] [CrossRef]
- Haddon, R.C.; Smalley, R.E. C240—The most chemically inert fullerene? Chem. Phys. Lett. 1997, 272, 38–42. [Google Scholar] [CrossRef]
- Wang, B.C.; Chiu, Y.N. Geometry and orbital symmetry relationships of giant and hyperfullerenes, pp. C240, C540, C960 and C1500. Synth. Met. 1993, 56, 2949–2954. [Google Scholar] [CrossRef]
- Terrones, M.; Terrones, G.; Terrones, H. Structure, Chirality, and Formation of Giant Icosahedral Fullerenes and Spherical Graphitic Onions. In Science of Crystal Structures; Springer: Cham, Switzerland, 2015; pp. 101–112. [Google Scholar]
- Martin, J.W.; McIntosh, G.J.; Arul, R.; Oosterbeek, R.N.; Kraft, M.; Söhnel, T. Giant fullerene formation through thermal treatment of fullerene soot. Carbon 2017, 125, 132–138. [Google Scholar] [CrossRef]
- Hirsch, A.; Chen, Z.; Jiao, H. Spherical aromaticity in Ih symmetrical fullerenes: The 2 (N + 1) 2 rule. Angew. Chem. Int. Ed. 2000, 39, 3915–3917. [Google Scholar] [CrossRef]
- Dodziuk, H.; Ruud, K.; Korona, T.; Demissie, T.B. Chiral recognition by fullerenes: CHFClBr enantiomers in the C82 cage. Phys. Chem. Chem. Phys. 2016, 18, 26057–26068. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.B., III; Strongin, R.M.; Brard, L.; Furst, G.T.; Romanow, W.J.; Owens, K.G.; Goldschmidt, R.J.; King, R.C. Synthesis of prototypical fullerene cyclopropanes and annulenes. Isomer differentiation via NMR and UV spectroscopy. J. Am. Chem. Soc. 1995, 117, 5492–5502. [Google Scholar] [CrossRef]
- Thilgen, C.; Gosse, J.; Diederich, F. Chirality in fullerene chemistry. Top. Stereochem. 2003, 23, 1–124. [Google Scholar] [CrossRef]
- Yang, S.; Wang, S.; Kemnitz, E.; Troyanov, S.I. Chlorination of IPR C100 fullerene affords unconventional C96Cl20 with a nonclassical cage containing three heptagons. Angew. Chem. Int. Ed. 2014, 53, 2460–2463. [Google Scholar] [CrossRef]
- Wang, S.; Yang, S.; Kemnitz, E.; Troyanov, S.I. New giant fullerenes identified as chloro derivatives, pp. isolated-pentagon-rule C108 (1771) Cl12 C106 (1155) Cl24 as well as nonclassical C104Cl24. Inorg. Chem. 2016, 55, 5741–5743. [Google Scholar] [CrossRef]
- Ghorbani, M.; Songhori, M.; Ashrafi, A.R.; Graovac, A.A. Symmetry group of (3, 6)-fullerenes. Fuller. Nanotub. Carbon Nanostruct. 2015, 23, 788–791. [Google Scholar] [CrossRef]
- Balasubramanian, K. Enumeration of chiral and positional isomers of substituted fullerene cages (C20-C70). J. Phys. Chem. 1993, 97, 6990–6998. [Google Scholar] [CrossRef]
- Balasubramanian, K. Enumeration of isomers of substituted fullerene cages C20- C50. Chem. Phys. Lett. 1993, 202, 399–405. [Google Scholar]
- Ghorbani, M.; Jalali, M. Counting numbers of permutational isomers of hetero fullerenes. Dig. J. Nanomater. Biostruct. 2008, 3, 269–275. [Google Scholar]
- Balasubramanian, K. Enumeration of chiral and achiral edge and face substitutions of buckminsterfullerene. Chem. Phys. Lett. 1995, 237, 229–238. [Google Scholar] [CrossRef]
- Balasubramanian, K. Topological characterization of five C78 fullerene isomers. Chem. Phys. Lett. 1993, 206, 210–216. [Google Scholar] [CrossRef]
- Balasubramanian, K. Laplacians of Fullerenes (C42-C90). J. Phys. Chem. 1995, 99, 6509–6518. [Google Scholar] [CrossRef]
- Balasubramanian, K. Characteristic polynomials of fullerene cages. Chem. Phys. Lett. 1992, 198, 577–586. [Google Scholar] [CrossRef]
- Balasubramanian, K. Group theoretical analysis of vibrational modes and rovibronic levels of extended aromatic C48N12 azafullerene. Chem. Phys. Lett. 2004, 391, 64–68. [Google Scholar] [CrossRef]
- Balaban, A.T.; Liu, X.; Klein, D.J.; Babić, D.; Schmalz, T.G.; Seitz, W.A.; Randić, M. Graph invariants for fullerenes. J. Chem. Inf. Comput. Sci. 1995, 35, 396–404. [Google Scholar] [CrossRef]
- Fripertinger, H. The cycle index of the symmetry group of the fullerene C60. MATCH Commun. Math. Comput. Chem 1996, 33, 121–138. [Google Scholar]
- Poater, A.; Saliner, A.G.; Carbó-Dorca, R.; Poater, J.; Solà, M.; Cavallo, L.; Worth, A.P. Modeling the structure-property relationships of nanoneedles: A journey toward nanomedicine. J. Comput. Chem. 2009, 30, 275–284. [Google Scholar] [CrossRef]
- Carbo-Dorca, R.; Besalu, E. Construction of coherent nano quantitative structure–properties relationships (nano-QSPR) models and catastrophe theory. SAR QSAR Environ. Res. 2011, 22, 661–665. [Google Scholar] [CrossRef]
- Terrones, M.; Terrones, G.; Terrones, H. Structure, chirality, and formation of giant icosahedral fullerenes and spherical graphitic onions. Struct. Chem. 2002, 13, 373. [Google Scholar] [CrossRef]
- Aihara, J.I.; Makino, M.; Ishida, T.; Dias, J.R. Analytical study of superaromaticity in cycloarenes and related coronoid hydrocarbons. J. Phys. Chem. A 2013, 117, 4688–4697. [Google Scholar] [CrossRef] [PubMed]
- Makino, M.; Dias, J.R.; Aihara, J.I. Bond Resonance Energy Verification of σ-Aromaticity in Cycloalkanes. J. Phys. Chem. A 2020, 124, 4549–4555. [Google Scholar] [CrossRef] [PubMed]
- Aihara, J.I.; Hosoya, H. Aromaticity of multiply charged fullerene ions. Bull. Chem. Soc. Jpn. 1993, 66, 1955–1958. [Google Scholar] [CrossRef]
- Sakamoto, K.; Nishina, N.; Enoki, T.; Aihara, J.I. Aromatic character of nanographene model compounds. J. Phys. Chem. A 2014, 118, 3014–3025. [Google Scholar] [CrossRef]
- Ciesielski, A.; Krygowski, T.M.; Cyrański, M.K.; Dobrowolski, M.A.; Aihara, J.I. Graph–topological approach to magnetic properties of benzenoid hydrocarbons. Phys. Chem. Chem. Phys. 2009, 11, 11447–11455. [Google Scholar] [CrossRef]
- Balasubramanian, K. Combinatorial Enumeration of Isomers of Superaromatic Polysubstituted Cycloarenes and Coronoid Hydrocarbons with Applications to NMR. J. Phys. Chem. A 2018, 122, 8243–8257. [Google Scholar] [CrossRef]
- Dias, J.R. Handbook of Polycyclic Hydrocarbons. Part A: Benzenoid Hydrocarbons; Elsevier: Amsterdam, The Netherlands, 1987. [Google Scholar]
- Dias, J.R. Perimeter Topology of Benzenoid Polycyclic Hydrocarbons. J. Chem. Inf. Model. 2005, 45, 562–571. [Google Scholar] [CrossRef]
- Randić, M. Graph theoretical approach to π-electron currents in polycyclic conjugated hydrocarbons. Chem. Phys. Lett. 2010, 500, 123–127. [Google Scholar] [CrossRef]
- Aihara, J.I. Topological resonance energy, bond resonance energy, and circuit resonance energy. J. Phys. Org. Chem. 2008, 21, 79–85. [Google Scholar] [CrossRef]
- Aihara, J.I.; Hosoya, H. Spherical aromaticity of buckminsterfullerene. Bull. Chem. Soc. Jpn. 1988, 61, 2657–2659. [Google Scholar] [CrossRef]
- Sheehan, J. On Pόlya’s Theorem. Can. J. Math. 1967, 19, 792–799. [Google Scholar] [CrossRef]
- Balasubramanian, K. Computational Enumeration of Colorings of Hyperplanes of Hypercubes for all Irreducible Representations and Applications. J. Math. Sci. Model. 2018, 2, 158–180. [Google Scholar] [CrossRef]
- Balasubramanian, K. Computational Combinatorics of Colorings of hyperplanes of 7D-hypercube for all Irreducible Representation. J. Comput. Chem. 2020, 41, 653–686. [Google Scholar] [CrossRef]
- Balasubramanian, K. Nuclear Spin Statistics of Fullerene Cages C20-C40. J. Phys. Chem. 1993, 97, 4647–4658. [Google Scholar] [CrossRef]
- Balasubramanian, K. Group theory, nuclear spin statistics and tunneling splittings 1,3,5-triamino-2,4,6-trinitrobenzene. Chem. Phys. Lett. 2004, 398, 15–21. [Google Scholar] [CrossRef]
- Balasubramanian, K. Relativity and the Jahn–Teller, Berry pseudorotations of TBP clusters: Group theory, spin–orbit and combinatorial nuclear spin statistics of TBP Desargues–Levi isomerization graph. J. Math. Chem. 2018, 56, 2194–2225. [Google Scholar] [CrossRef]
- Babić, D.; Došlić, T.; Douglas, J.; Klein, D.J.; Misra, A. Kekulenoid addition patterns for fullerenes and some lower homologs. Bull. Chem. Soc. Jpn. 2004, 77, 2003–2010. [Google Scholar]
- Majumdar, D.; Roszak, S.; Balasubramanian, K.; Nitsche, H. Theoretical study of aqueous uranyl carbonate (UO2CO3) and its hydrated complexes: UO2CO3·nH2O (n = 1–3). Chem. Phys. Lett. 2003, 372, 232–241. [Google Scholar] [CrossRef]
- Schwaiger, L.K.; Parsons-Moss, T.; Hubaud, A.; Tueysuez, H.; Balasubramanian, K.; Yang, P.; Nitsche, H. Actinide and lanthanide complexation by organically modified mesoporous silica. Abstr. Pap. ACS 2010, 239, 98. [Google Scholar]
- Parsons-Moss, T.; Schwaiger, L.K.; Hubaud, A.; Hu, Y.J.; Tuysuz, H.; Yang, P.; Balasubramanian, K.; Nitsche, H. Plutonium complexation by phosphonate-functionalized mesoporous silica. Abstr. Pap. ACS 2011, 241, 48. [Google Scholar]
- Balasubramanian, K.; Ori, O.; Cataldo, F.; Ashrafi, A.R.; Putz, M.V. Face Colorings and Chiral Face Colorings of Icosahedral Giant Fullerenes: C80 to C240. Fuller. Nanotub. Carbon Nanostruct. 2020. (In Press)
- Balasubramanian, K. Nuclear spin statistics of extended aromatic C48N12 azafullerene. Chem. Phys. Lett. 2004, 391, 69–74. [Google Scholar] [CrossRef]
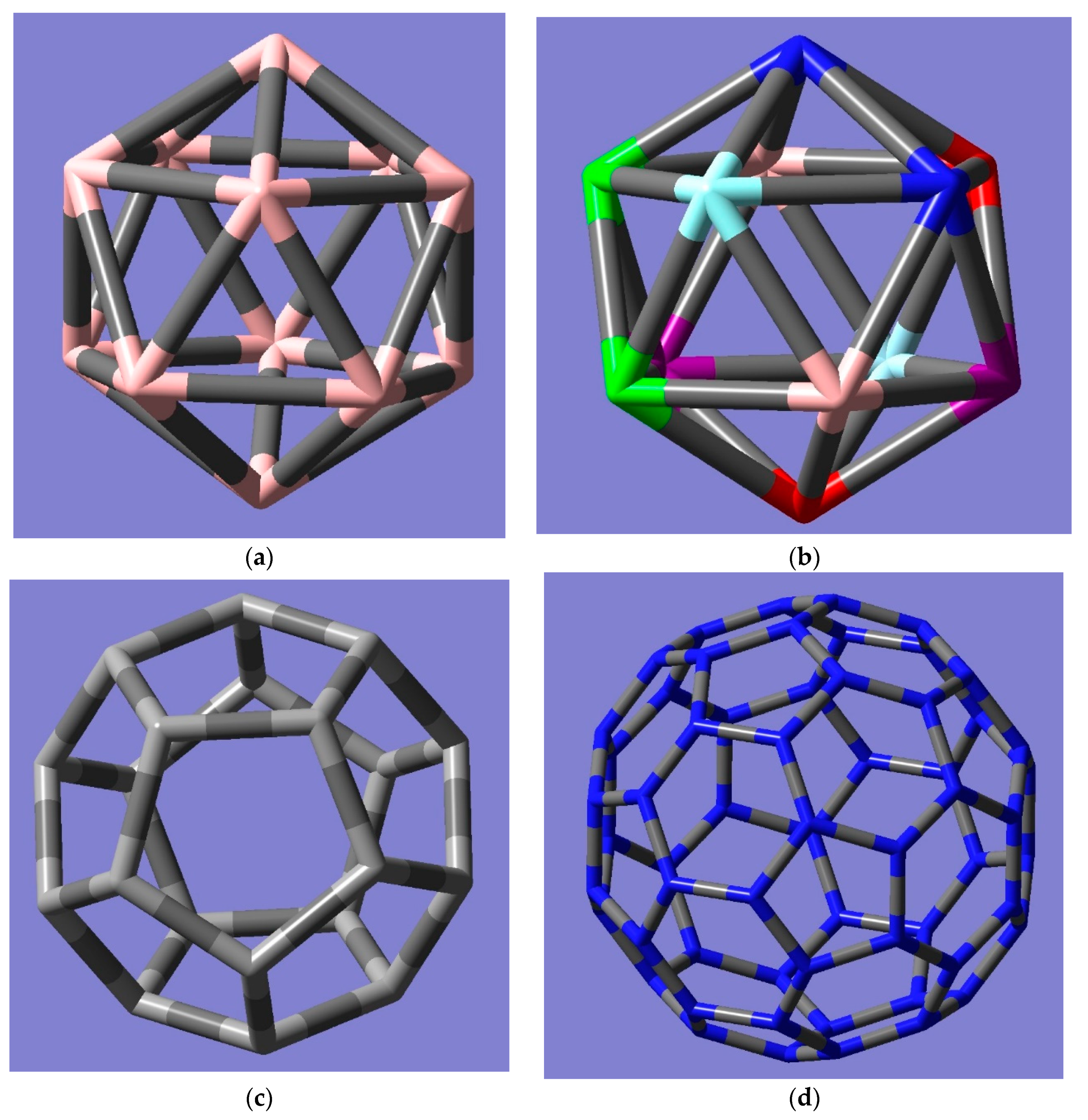
| CC | E | 15C2 | 12C5 | 12C52 | 20C3 | I | 12S10 | 12S103 | 20S6 | 15σd |
|---|---|---|---|---|---|---|---|---|---|---|
| C60 (30 hex–hex) | 130 | 12214 | 56 | 56 | 310 | 215 | 103 | 103 | 65 | 14213 |
| Icosahedron | ||||||||||
| (30 edges) | ||||||||||
| C60 (pent–hex edge) | 160 | 230 | 512 | 512 | 320 | 230 | 106 | 106 | 610 | 14 228 |
| C80 (hex–hex); | 160 | 230 | 512 | 512 | 320 | 230 | 106 | 106 | 610 | 14 228 |
| chamfered dodecahedron | ||||||||||
| C80 (pent–hex; edges) | 160 | 230 | 512 | 512 | 320 | 230 | 106 | 106 | 610 | 14 228 |
| C180 (hex–hex) | 1210 | 122104 | 542 | 542 | 370 | 2105 | 1021 | 1021 | 635 | 182101 |
| C180 (pent–hex) = C60 (pent–hex);Vertices: chamfered dodecahedron | 160 | 230 | 512 | 512 | 320 | 230 | 106 | 106 | 610 | 14 228 |
| C240 (hex–hex) | 1300 | 2150 | 562 | 562 | 3100 | 2150 | 1030 | 1030 | 650 | 1122144 |
| C240 (pent–hex) | 160 | 230 | 512 | 512 | 320 | 230 | 106 | 106 | 610 | 14 228 |
| C60 (pent–hex); chamfered truncated icosahedron | ||||||||||
| Ag | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| T1g | 3 | −1 | ω | ω * | 0 | 3 | ω | ω | 0 | −1 |
| T2g | 3 | −1 | ω * | ω | 0 | 3 | ω | ω * | 0 | −1 |
| Gg | 4 | 0 | −1 | −1 | 1 | 4 | −1 | −1 | 1 | 0 |
| Hg | 5 | 1 | 0 | 0 | −1 | 5 | 0 | 0 | −1 | 1 |
| Au | 1 | 1 | 1 | 1 | 1 | −1 | −1 | −1 | −1 | −1 |
| T1u | 3 | −1 | ω | ω * | 0 | −3 | −ω * | −ω | 0 | 1 |
| T2u | 3 | −1 | ω * | ω | 0 | −3 | −ω | −ω * | 0 | 1 |
| Gu | 4 | 0 | −1 | −1 | −1 | −4 | 1 | 1 | 1 | 0 |
| Hu | 5 | 1 | 0 | 0 | −1 | −5 | 0 | 0 | 1 | −1 |
| Color Partition | Ag | Au |
|---|---|---|
| 30 0 0 0 0 | 1 | 0 |
| 29 1 0 0 0 | 1 | 0 |
| 28 2 0 0 0 | 8 | 3 |
| 27 3 0 0 0 | 46 | 32 |
| 26 4 0 0 0 | 262 | 221 |
| 25 5 0 0 0 | 1257 | 1166 |
| 24 6 0 0 0 | 5113 | 4912 |
| 23 7 0 0 0 | 17,238 | 16,874 |
| 22 8 0 0 0 | 49,270 | 48,620 |
| 21 9 0 0 0 | 119,997 | 118,996 |
| 20 10 0 0 0 | 251,512 | 249,995 |
| 19 11 0 0 0 | 456,729 | 454,727 |
| 18 12 0 0 0 | 722,750 | 720,125 |
| 17 13 0 0 0 | 1,000,251 | 997,248 |
| 16 14 0 0 0 | 1,214,376 | 1,210,944 |
| 15 15 0 0 0 | 1,295,266 | 1,291,834 |
| 28 1 1 0 0 | 9 | 6 |
| 27 2 1 0 0 | 113 | 97 |
| 26 3 1 0 0 | 937 | 897 |
| 25 4 1 0 0 | 6019 | 5902 |
| 24 5 1 0 0 | 29,835 | 29,588 |
| 23 6 1 0 0 | 119,106 | 118,586 |
| 22 7 1 0 0 | 390,754 | 389,818 |
| 21 8 1 0 0 | 1,074,073 | 1,072,500 |
| 20 9 1 0 0 | 2,505,217 | 2,502,786 |
| 19 10 1 0 0 | 5,009,719 | 5,006,287 |
| 18 11 1 0 0 | 8,652,111 | 8,647,535 |
| 17 12 1 0 0 | 12,977,523 | 12,971,946 |
| 16 13 1 0 0 | 16,969,947 | 16,963,512 |
| 15 14 1 0 0 | 19,393,980 | 19,387,116 |
| 26 2 2 0 0 | 1438 | 1355 |
| 25 3 2 0 0 | 12,025 | 11,817 |
| 24 4 2 0 0 | 74,698 | 74,087 |
| 23 5 2 0 0 | 357,162 | 355,914 |
| 22 6 2 0 0 | 1,367,704 | 1,365,026 |
| 21 7 2 0 0 | 4,295,434 | 4,290,858 |
| 20 8 2 0 0 | 11,272,690 | 11,264,825 |
| 19 9 2 0 0 | 25,045,735 | 25,034,295 |
| 18 10 2 0 0 | 47,583,250 | 47,566,805 |
| 17 11 2 0 0 | 77,858,703 | 77,838,111 |
| 16 12 2 0 0 | 110,297,148 | 110,271,837 |
| 15 13 2 0 0 | 135,747,564 | 135,720,108 |
| 14 14 2 0 0 | 145,443,696 | 145,414,524 |
| 26 1 1 1 1 | 5484 | 5478 |
| 22 2 2 2 2 | 122,923,788 | 122,907,252 |
| 18 3 3 3 3 | 266,399,185,320 | 266,399,082,360 |
| 14 4 4 4 4 | 76,423,258,557,360 | 76,423,248,055,440 |
| 10 5 5 5 5 | 2,937,587,511,706,920 | 2,937,587,492,247,480 |
| 6 6 6 6 6 | 11,423,951,476,076,040 | 11,423,951,359,159,200 |
| [ ] | Ag | Au |
|---|---|---|
| 120 0 | 1 | 0 |
| 119 1 | 2 | 0 |
| 118 2 | 78 | 56 |
| 117 3 | 2410 | 2284 |
| 116 4 | 69,088 | 68,264 |
| 115 5 | 1,590,094 | 1,586.216 |
| 114 6 | 30,453,590 | 30,434,316 |
| 113 7 | 495,768,634 | 495,690,848 |
| 112 8 | 7,002,404,212 | 7,002,082,880 |
| 111 9 | 87,138,846,064 | 87,137,701,732 |
| 110 10 | 967,237,561,188 | 967,233,448,280 |
| 109 11 | 9,672,354,806,324 | 9,672,341,633,472 |
| 108 12 | 87,857,190,426,762 | 87,857,148,114,480 |
| 107 13 | 729,890,339,015,124 | 729,890,215,403,328 |
| 106 14 | 5,578,447,347,506,340 | 5,578,446,986,386,416 |
| 105 15 | 39,421,026,791,684,152 | 39,421,025,819,079,244 |
| 104 16 | 258,700,486,756,281,228 | 258,700,484,140,064,064 |
| 103 17 | 1,582,638,265,236,654,990 | 1,582,638,258,686,625,504 |
| 102 18 | 9,056,207,842,369,897,698 | 9,056,207,825,999,272,476 |
| 101 19 | 48,617,536,803,295,178,082 | 48,617,536,764,944,543,760 |
| 100 20 | 245,518,560,814,069,700,244 | 245,518,560,724,389,406,200 |
| 99 21 | 1,169,136,003,716,937,778,470 | 1,169,136,003,519,309,366,060 |
| 98 22 | 5,261,112,016,541,763,342,090 | 5,261,112,016,106,995,874,280 |
| 97 23 | 22,416,912,069,826,131,025,350 | 22,416,912,068,920,923,328,320 |
| 96 24 | 90,601,686,281,500,477,155,795 | 90,601,686,279,618,732,869,760 |
| 95 25 | 347,910,475,318,534,665,936,000 | 347,910,475,314,819,617,649,960 |
| 94 26 | 1,271,211,352,122,935,979,909,384 | 1,271,211,352,115,611,772,064,864 |
| 93 27 | 4,425,698,781,456,914,849,108,248 | 4,425,698,781,443,161,812,224,368 |
| 92 28 | 14,699,642,381,259,834,553,464,448 | 14,699,642,381,234,041,957,888,288 |
| 91 29 | 4,663,334,824,397,2017,281,896,744 | 4,663,334,824,3925,833,868,982,784 |
| 90 30 | 141,454,489,673,359,691,507,520,872 | 141,454,489,673,277,089,121,719,224 |
| 89 31 | 410,674,324,858,072,502,600,358,552 | 410,674,324,857,931,154,308,999,872 |
| 88 32 | 1,142,187,966,011,457,101,562,409,164 | 1,142,187,966,011,215,471,310,707,584 |
| 87 33 | 3,045,834,576,030,378,172,825,647,474 | 3,045,834,576,029,982,283,356,027,984 |
| 86 34 | 7,793,753,179,842,304,178,443,269,582 | 7,793,753,179,841,656,145,234,151,672 |
| 85 35 | 19,150,364,956,183,541,440,106,097,990 | 19,150,364,956,182,523,204,755,942,120 |
| 84 36 | 45,216,139,479,877,519,005,495,375,832 | 45,216,139,479,875,920,443,015,611,752 |
| 83 37 | 102,652,857,197,558,901,889,038,488,854 | 102,652,857,197,556,489,675,761,794,864 |
| 82 38 | 224,215,451,247,299,146,518,483,944,134 | 224,215,451,247,295,509,415,708,269,304 |
| 81 39 | 471,427,359,032,781,098,433,790,040,842 | 471,427,359,032,775,821,266,598,044,912 |
| 80 40 | 954,640,402,041,380,730,670,203,931,050 | 954,640,402,041,373,079,676,086,262,720 |
| 79 41 | 1,862,712,979,592,934,866,777,089,399,284 | 1,862,712,979,592,924,181,779,327,085,024 |
| 78 42 | 3,503,674,413,996,233,035,110,445,055,388 | 3,503,674,413,996,218,123,774,600,549,088 |
| 77 43 | 6,355,502,425,388,510,434,415,173,583,148 | 6,355,502,425,388,490,372,453,396,946,368 |
| 76 44 | 11,122,129,244,429,890,961,707,126,809,024 | 11,122,129,244,429,863,988,882,908,985,904 |
| 75 45 | 18,784,040,501,703,807,575,164,139,407,276 | 18,784,040,501,703,772,587,371,933,573,536 |
| 74 46 | 30,626,152,991,908,378,993,288,608,599,100 | 30,626,152,991,908,333,639,766,231,416,080 |
| 73 47 | 48,219,900,455,345,095,423,215,234,731,220 | 48,219,900,455,345,038,666,516,858,355,040 |
| 72 48 | 73,334,431,942,503,996,127,122,897,840,930 | 73,334,431,942,503,925,147,462,481,638,080 |
| 71 49 | 107,756,716,323,679,325,348,758,295,151,870 | 107,756,716,323,679,239,606,867,504,704,160 |
| 70 50 | 153,014,537,179,624,639,361,191,172,071,458 | 153,014,537,179,624,535,854,656,958,090,680 |
| 69 51 | 210,019,952,991,641,642,300,675,366,021,178 | 210,019,952,991,641,521,554,044,495,863,704 |
| 68 52 | 278,680,322,238,909,101,292,212,428,312,224 | 278,680,322,238,908,960,524,975,921,227,960 |
| 67 53 | 357,552,111,551,807,881,353,106,541,651,418 | 357,552,111,551,807,722,713,006,235,751,856 |
| 66 54 | 443,629,471,740,206,076,931,127,682,726,010 | 443,629,471,740,205,898,263,250,604,257,152 |
| 65 55 | 532,355,366,088,247,269,202,081,059,624,998 | 532,355,366,088,247,074,629,686,854,396,112 |
| 64 56 | 617,912,478,495,287,014,817,849,899,446,772 | 617,912,478,495,286,803,059,724,238148864 |
| 63 57 | 693,796,467,082,427,503,413,465,875,637,192 | 693,796,467,082,427,280,527,729,050,007,160 |
| 62 58 | 753,606,507,348,154,022,809,487,360,189,168 | 753,606,507,348,153,788,359,679,050,072,384 |
| 61 59 | 791,925,482,298,060,140,610,210,171,032,240 | 791,925,482,298,059,902,076,562,608,006,656 |
| 60 60 | 805,124,240,336,361,157,749,996,742,071,252 | 805,124,240,336,360,915,214,413,591,091,584 |
| [ ] | Ag | Au |
|---|---|---|
| 270 0 | 1 | 0 |
| 269 1 | 4 | 1 |
| 268 2 | 345 | 294 |
| 267 3 | 27,304 | 26,862 |
| 266 4 | 1,807,917 | 1,803,450 |
| 265 5 | 96,020,482 | 95,988,421 |
| 264 6 | 4,240,271,777 | 4,240,025,284 |
| 263 7 | 159,913,119,758 | 159,911,591,426 |
| 262 8 | 5,257,122,017,406 | 5,257,112,226,096 |
| 261 9 | 153,040,502,687,936 | 153,040,448,776,803 |
| 260 10 | 3,994,356,526,363,671 | 3,994,356,224,947,782 |
| 259 11 | 9,441,205,959,837,3881 | 9,441,205,809,631,1423 |
| 258 12 | 2,037,726,939,692,823,911 | 2,037,726,932,160,044,746 |
| 257 13 | 40,441,042,267,224,568,708 | 40,441,042,232,772,631,201 |
| 256 14 | 742,381,989,938,234,707,452 | 742,381,989,780,438,407,424 |
| 255 15 | 12,669,985,960,338,205,685,410 | 12,669,985,959,668,812,183,074 |
| 254 16 | 201,927,901,238,727,703,128,192 | 201,927,901,235,891,706,182,400 |
| 253 17 | 3,017,040,406,724,166,624,480,064 | 3,017,040,406,712,915,359,645,072 |
| 252 18 | 42,406,179,050,007,222,298,565,790 | 42,406,179,049,962,711,561,688,884 |
| 251 19 | 562,439,848,452,470,787,556,343,504 | 562,439,848,452,304,546,365,120,176 |
| 250 20 | 7,058,620,098,077,732,214,695,554,434 | 7,058,620,098,077,113,629,220,104,300 |
| 249 21 | 84,031,191,643,779,442,794,688,130,060 | 84,031,191,643,777,256,084,971,286,460 |
| 248 22 | 951,080,305,422,767,422,366,226,704,800 | 951,080,305,422,759,724,718,170,368,000 |
| 247 23 | 10,255,126,771,515,023,854,648,652,352,800 | 10,255,126,771,514,997,983,993,766,994,400 |
| 246 24 | 105,542,346,356,842,025,862,721,432,364,005 | 105,542,346,356,841,939,302,159,520,713,540 |
| 245 25 | 1,038,536,688,151,325,201,078,073,328,559,350 | 1,038,536,688,151,324,923,452,263,653,73,0150 |
| 244 26 | 9,786,211,099,887,486,562,268,711,079,585,360 | 9,786,211,099,887,485,675,909,620,716,272,160 |
| 243 27 | 88,438,352,161,946,171,819,410,469,089,895,264 | 88,438,352,161,946,169,097,774,234,297,660,864 |
| 242 28 | 767,518,556,262,604,268,829,301,580,115,605,632 | 767,518,556,262,604,260,510,851,755,374,513,280 |
| 241 29 | 6,404,810,021,225,870,079,832,897,841,141,394,432 | 6,404,810,021,225,870,055,311,800,035,487,058,944 |
| 240 30 | 51,451,973,837,181,156,242,454,272,224,708,534,272 | 51,451,973,837,181,156,170,504,106,998,886,072,320 |
| [ ] | Ag | Au |
|---|---|---|
| 360 0 | 1 | 0 |
| 359 1 | 5 | 1 |
| 358 2 | 599 | 523 |
| 357 3 | 64,695 | 63,867 |
| 356 4 | 5,742,088 | 5,732,528 |
| 355 5 | 408,394,963 | 408,310,967 |
| 354 6 | 24,161,376,447 | 24,160,630,719 |
| 353 7 | 1,221,853,155,773 | 1,221,847,572,889 |
| 352 8 | 53,914,173,386,252 | 53,914,131,840,160 |
| 351 9 | 2,108,642,343,606,824 | 2,108,642,069,551,032 |
| 350 10 | 74,013,342,528,379,652 | 74,013,340,742,388,172 |
| 349 11 | 2,354,969,960,511,184,484 | 2,354,969,949,897,381,076 |
| 348 12 | 68,490,376,233,711,549,198 | 68,490,376,171,529,817,312 |
| 347 13 | 1,833,434,686,063,143,560,788 | 1,833,434,685,724,940,441,732 |
| 346 14 | 45,442,988,287,129,208,605,684 | 45,442,988,285,316,727,072,676 |
| 345 15 | 1,048,218,263,136,994,898,032,224 | 1,048,218,263,127,866,791,758,384 |
| 344 16 | 22,602,206,298,818,610,054,452,936 | 22,602,206,298,773,304,345,806,944 |
| 343 17 | 457,362,292,163,918,680,987,994,882 | 457,362,292,163,705,502,108,517,978 |
| 342 18 | 8,715,292,567,344,302,910,553,154,422 | 8,715,292,567,343,313,881,950,682,458 |
| 341 19 | 156,875,266,212,189,736,812,649,026,446 | 156,875,266,212,185,357,989,454,884,534 |
| 340 20 | 2,674,723,288,917,808,199,876,682,916,172 | 2,674,723,288,91,778,907,356,818,542,3648 |
| 339 21 | 43,305,043,725,335,812,080,811,714,363,506 | 43,305,043,725,335,731,946,246,037,299,994 |
| 338 22 | 667,291,355,585,855,940,114,394,440,368,422 | 667,291,355,585,855,608,684,210,761,554,798 |
| 337 23 | 9,806,281,660,348,663,563,010,529,539,308,321 | 9,806,281,660,348,662,242,732,670,525,711,097 |
| 336 24 | 137,696,538,314,062,477,732,769,369,588,040,480 | 137,696,538,314,062,472,537,951,467,943,685,984 |
| 335 25 | 1,850,641,474,940,999,672,906,554,426,005,029,888 | 1,850,641,474,940,999,653,153,302,290,271,031,040 |
| 334 26 | 23,844,803,619,432,111,082,681,856,449,184,387,072 | 23,844,803,619,432,111,008,453,439,294,860,623,872 |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balasubramanian, K. Combinatorics of Edge Symmetry: Chiral and Achiral Edge Colorings of Icosahedral Giant Fullerenes: C80, C180, and C240 . Symmetry 2020, 12, 1308. https://doi.org/10.3390/sym12081308
Balasubramanian K. Combinatorics of Edge Symmetry: Chiral and Achiral Edge Colorings of Icosahedral Giant Fullerenes: C80, C180, and C240 . Symmetry. 2020; 12(8):1308. https://doi.org/10.3390/sym12081308
Chicago/Turabian StyleBalasubramanian, Krishnan. 2020. "Combinatorics of Edge Symmetry: Chiral and Achiral Edge Colorings of Icosahedral Giant Fullerenes: C80, C180, and C240 " Symmetry 12, no. 8: 1308. https://doi.org/10.3390/sym12081308
APA StyleBalasubramanian, K. (2020). Combinatorics of Edge Symmetry: Chiral and Achiral Edge Colorings of Icosahedral Giant Fullerenes: C80, C180, and C240 . Symmetry, 12(8), 1308. https://doi.org/10.3390/sym12081308