Towards More Realistic Leaf Shapes in Functional-Structural Plant Models
Abstract
1. Introduction
2. Materials and Methods
2.1. Virtual Plant Model
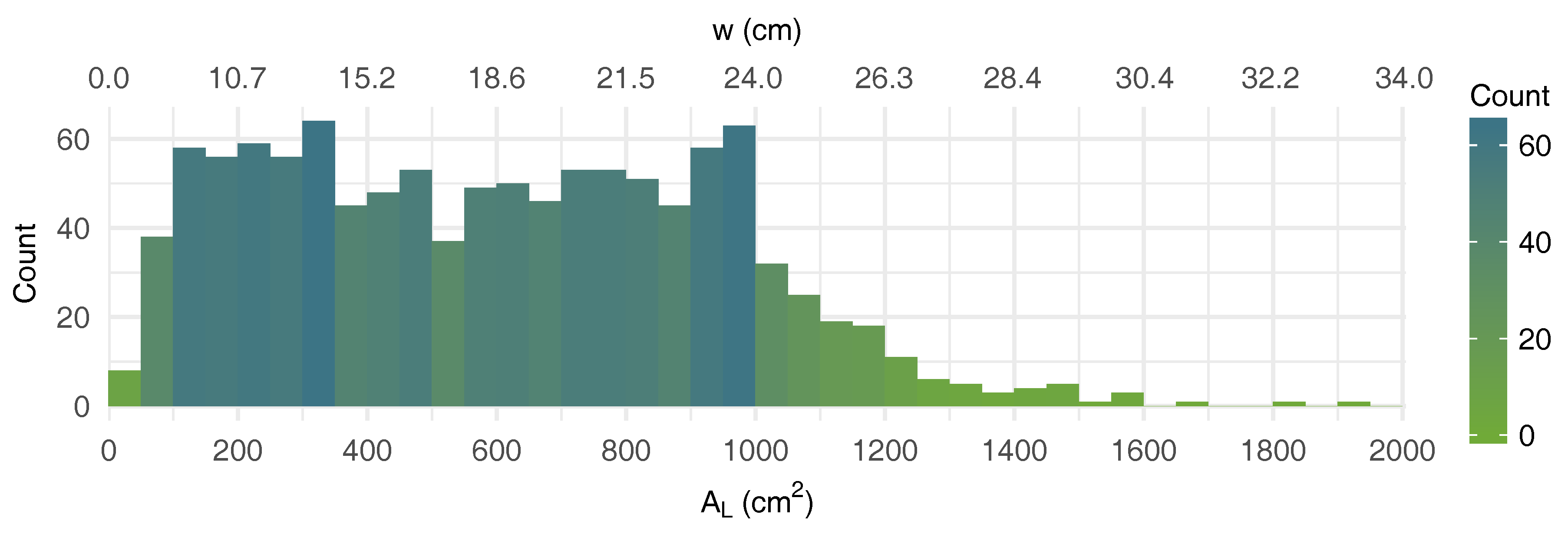
2.2. Experimental Data
2.2.1. Digitized Leaves Dataset
2.2.2. Plant Morphological Data
2.3. Morphometric Leaf Shape Analysis
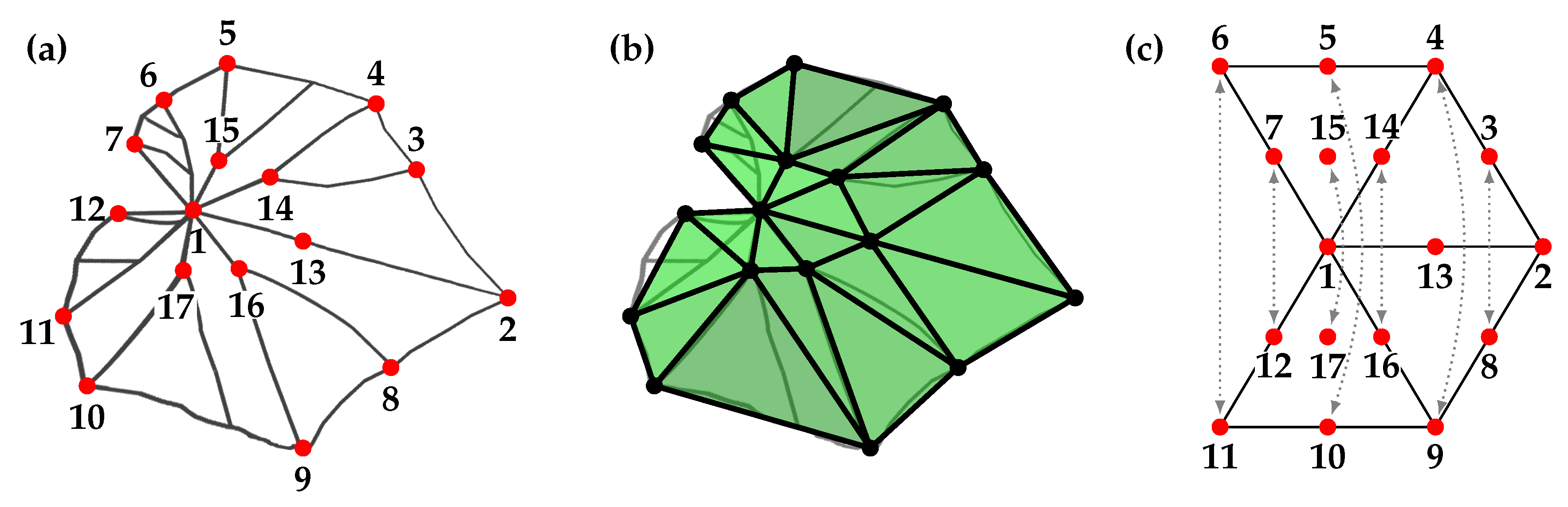
2.3.1. Preprocessing
2.3.2. Morphometric Measures
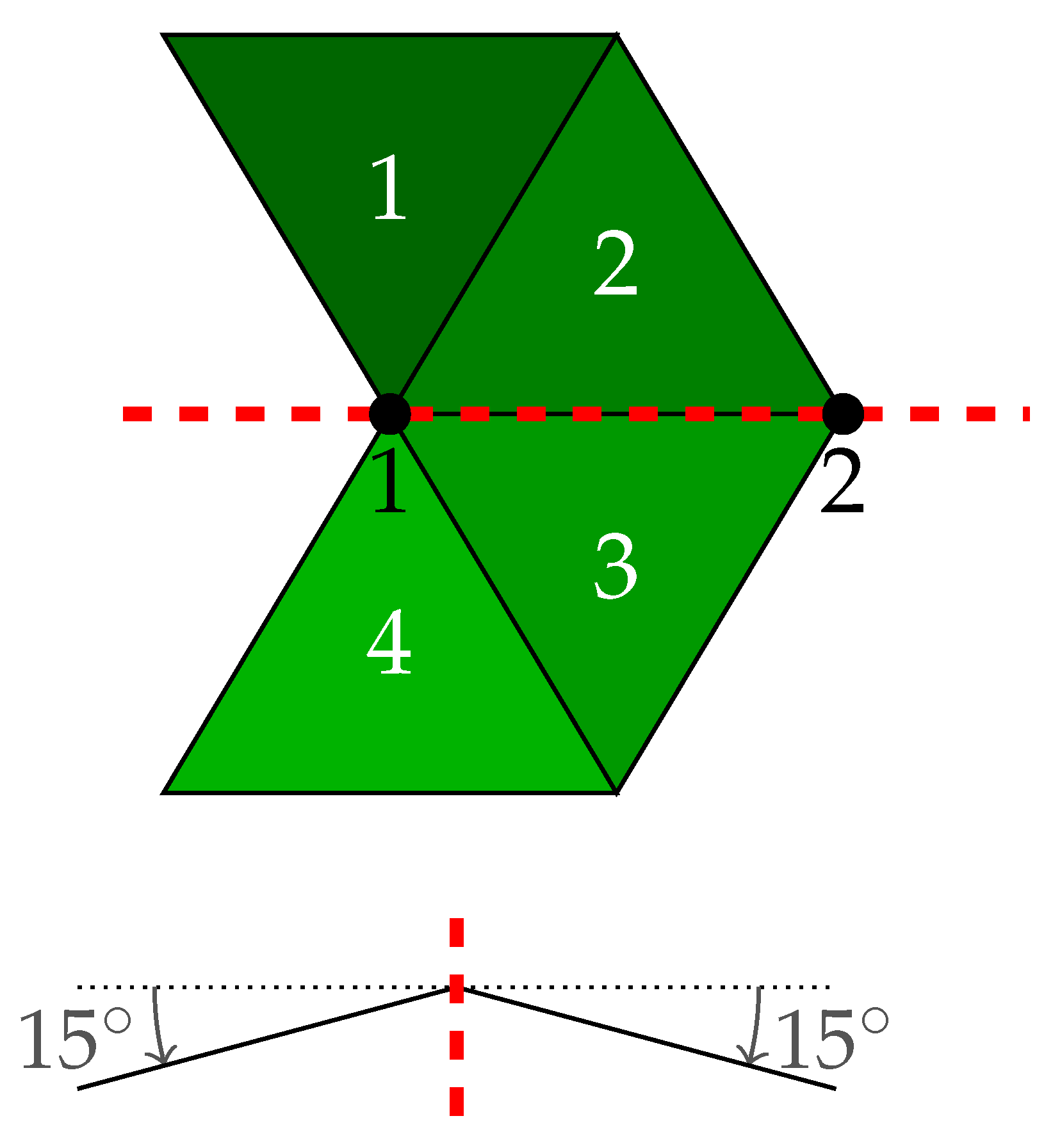
2.3.3. Symmetry Analysis
- calculate the left-right signed difference between the paired landmark shift for a single dimension (, or )
- conduct robust Bayesian estimation on the differences () to estimate posterior mean () and posterior standard deviation () as measures for directional and fluctuating asymmetry
- (a)
- if 95% highest probability density interval (HDI) of includes zero⇒ no directional asymmetry
- (b)
- if 95% highest probability density interval (HDI) of excludes zero⇒ directional asymmetry
- conduct robust Bayesian estimation on the pooled left-right data () to estimate overall posterior mean (absolute average displacement)
- if necessary: adjust average displacements using directional asymmetry information
2.4. Virtual Plant Simulations
3. Results and Discussion
3.1. Average Leaf Shape
3.2. Leaf Symmetry
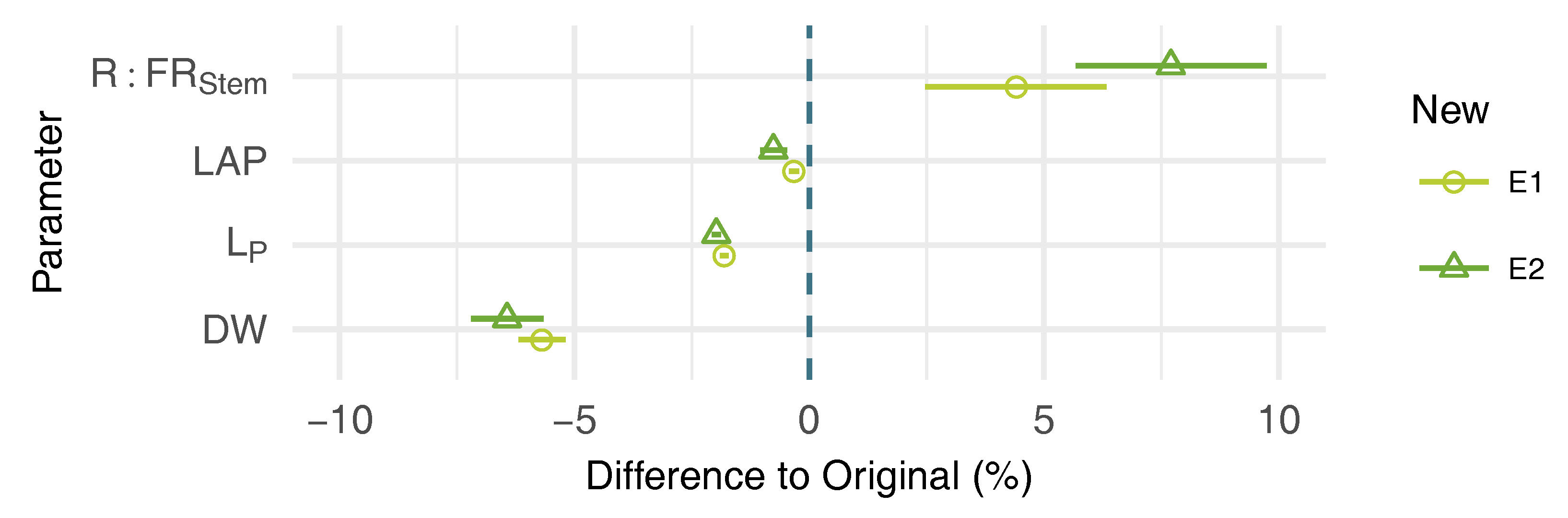
3.3. Effects of Leaf Shape on Virtual Plant Simulations
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| AL | leaf area (cm2) |
| DW | produced dry weight per plant and day (g) |
| internode elongation at day t (cm) | |
| temperature response function | |
| FIL | final internode length (cm) |
| FSPM | functional-structural plant model |
| GPA | Generalized Procrustes analysis (GPA) |
| HDI | 95% highest probability density interval |
| IL(t) | internode length at day t (cm) |
| plant length (cm) | |
| LAP | leaf area per plant (cm2) |
| L-System | Lindenmayer-System |
| leaf base | |
| leaf tip | |
| PAR | photosynthetically active radiation |
| R:FR | red-to-far-red ratio |
| daily steam red-to-far-red ratio from internodes weighted by their length (-) | |
| RMSD | root-mean-square deviation |
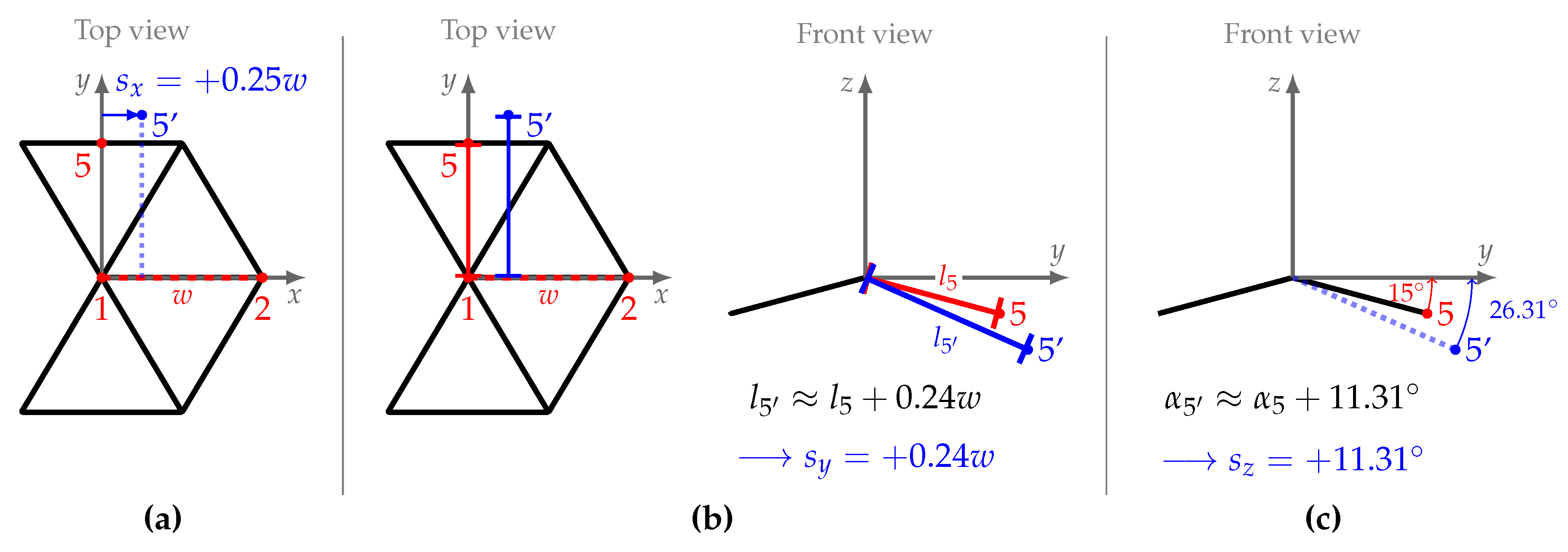
| point wise displacement along the x-axis | |
| point wise displacement in distance to the x-axis | |
| point wise displacement in roll angle around the x-axis | |
| , , | posterior mean of point wise displacement |
| , , | posterior standard deviation of point wise displacement |
| T | temperature (°C) |
| t | time (day) |
| w | equilateral triangle side (cm) |
References
- Vos, J.; Evers, J.B.; Buck-Sorlin, G.H.; Andrieu, B.; Chelle, M.; de Visser, P.H.B. Functional-structural plant modelling: A new versatile tool in crop science. J. Exp. Bot. 2010, 61, 2101–2115. [Google Scholar] [CrossRef] [PubMed]
- Sievänen, R.; Godin, C.; DeJong, T.M.; Nikinmaa, E. Functional-structural plant models: A growing paradigm for plant studies. Ann. Bot. 2014, 114, 599–603. [Google Scholar] [CrossRef] [PubMed]
- Vos, J.; Marcelis, L.; Evers, J. Functional-Structural plant modelling in crop production: Adding a dimension. In Functional-Structural Plant Modelling in Crop Production; Vos, J., Marcelis, L.F.M., Visser, P., Struik, P.C., Evers, J.B., Eds.; Springer: Dordrecht, The Netherlands, 2007; Volume 22, pp. 1–12. [Google Scholar]
- Prusinkiewicz, P.; Lindenmayer, A. The Algorithmic Beauty of Plants; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Kahlen, K.; Stützel, H. Modelling photo-modulated internode elongation in growing glasshouse cucumber canopies. New Phytol. 2011, 190, 697–708. [Google Scholar] [CrossRef] [PubMed]
- Kahlen, K.; Chen, T.W. Predicting Plant Performance Under Simultaneously Changing Environmental Conditions—The Interplay Between Temperature, Light, and Internode Growth. Front. Plant Sci. 2015, 6, 1130. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.W.; Nguyen, T.M.N.; Kahlen, K.; Stützel, H. High temperature and vapor pressure deficit aggravate architectural effects but ameliorate non-architectural effects of salinity on dry mass production of tomato. Front. Plant Sci. 2015, 6, 887. [Google Scholar] [CrossRef] [PubMed]
- Viaud, G.; Loudet, O.; Cournède, P.H. Leaf Segmentation and Tracking in Arabidopsis thaliana Combined to an Organ-Scale Plant Model for Genotypic Differentiation. Front. Plant Sci. 2017, 7, 2057. [Google Scholar] [CrossRef] [PubMed]
- Allen, M.T.; Prusinkiewicz, P.; DeJong, T.M. Using L-systems for modeling source–sink interactions, architecture and physiology of growing trees: The L-PEACH model. New Phytol. 2005, 166, 869–880. [Google Scholar] [CrossRef] [PubMed]
- Hanan, J.; Hearn, A. Linking physiological and architectural models of cotton. Agric. Syst. 2003, 75, 47–77. [Google Scholar] [CrossRef]
- Nikinmaa, E.; Sievänen, R.; Perttunen, J.; Hölttä, T. Simulated interaction between tree structure and xylem and phloem transport in 3D tree crowns using model LIGNUM. In Proceedings of the FSPM 2013 7th International Conference on Functional-Structural Plant Models, Saariselkä, Finland, 9–14 June 2013; Sievänen, R., Nikinmaa, E., Godin, C., Lintunen, A., Nygren, P.F.I., Eds.; [Google Scholar]
- Da Silva, D.; Han, L.; Faivre, R.; Costes, E. Influence of the variation of geometrical and topological traits on light interception efficiency of apple trees: Sensitivity analysis and metamodelling for ideotype definition. Ann. Bot. 2014, 114, 739–752. [Google Scholar] [CrossRef] [PubMed]
- Godin, C.; Sinoquet, H. Functional–structural plant modelling. New Phytol. 2005, 166, 705–708. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Li, J.; Cui, L.; Zhang, T.; Wu, Z.; Zhu, P.Y.; Meng, Y.J.; Zhang, K.J.; Yu, X.Q.; Lou, Q.F.; et al. New insights into the roles of cucumber TIR1 homologs and miR393 in regulating fruit/seed set development and leaf morphogenesis. BMC Plant Biol. 2017, 17, 130. [Google Scholar] [CrossRef] [PubMed]
- Kahlen, K.; Wiechers, D.; Stützel, H. Modelling leaf phototropism in a cucumber canopy. Funct. Plant Biol. 2008, 35, 876–884. [Google Scholar] [CrossRef]
- Kahlen, K. Towards functional-structural modelling of greenhouse cucumber. In Functional-Structural Plant Modelling in Crop Production; Vos, J., Marcelis, L.F.M., Visser, P., Struik, P.C., Evers, J.B., Eds.; Springer: Dordrecht, The Netherlands, 2007; Volume 22, pp. 209–217. [Google Scholar]
- Chambelland, J.C.; Dassot, M.; Adam, B.; Donès, N.; Balandier, P.; Marquier, A.; Saudreau, M.; Sonohat, G.; Sinoquet, H. A double-digitising method for building 3D virtual trees with non-planar leaves: Application to the morphology and light-capture properties of young beech trees (Fagus sylvatica). Funct. Plant Biol. 2008, 35, 1059–1069. [Google Scholar] [CrossRef]
- Chéné, Y.; Rousseau, D.; Lucidarme, P.; Bertheloot, J.; Caffier, V.; Morel, P.; Belin, É.; Chapeau-Blondeau, F. On the use of depth camera for 3D phenotyping of entire plants. Comput. Electron. Agric. 2012, 82, 122–127. [Google Scholar] [CrossRef]
- An, N.; Palmer, C.M.; Baker, R.L.; Markelz, R.C.; Ta, J.; Covington, M.F.; Maloof, J.N.; Welch, S.M.; Weinig, C. Plant high-throughput phenotyping using photogrammetry and imaging techniques to measure leaf length and rosette area. Comput. Electron. Agric. 2016, 127, 376–394. [Google Scholar] [CrossRef]
- Loch, B.I. Surface Fitting for the Modelling of Plant Leaves. Ph.D. Thesis, University of Queensland, Queensland, Australia, 2004. [Google Scholar]
- Zhang, L.; Weckler, P.; Wang, N.; Xiao, D.; Chai, X. Individual leaf identification from horticultural crop images based on the leaf skeleton. Comput. Electron. Agric. 2016, 127, 184–196. [Google Scholar] [CrossRef]
- Kahlen, K. 3D Architectural Modelling of Greenhouse Cucumber (Cucumis sativus L.) Using L-Systems; International Society for Horticultural Science (ISHS): Leuven, Belgium, 2006; pp. 51–58. [Google Scholar]
- Kahlen, K.; Stützel, H. Estimation of Geometric Attributes and Masses of Individual Cucumber Organs Using Three-dimensional Digitizing and Allometric Relationships. J. Am. Soc. Hort. Sci. 2007, 132, 439–446. [Google Scholar]
- Whitlock, M. The heritability of fluctuating asymmetry and the genetic control of developmental stability. Proc. R. Soc. Lond. Ser. B 1996, 263, 849–853. [Google Scholar] [CrossRef] [PubMed]
- DONGEN, S.V. Fluctuating asymmetry and developmental instability in evolutionary biology: Past, present and future. J. Evol. Biol. 2006, 19, 1727–1743. [Google Scholar] [CrossRef] [PubMed]
- Klingenberg, C.P. Analyzing Fluctuating Asymmetry with Geometric Morphometrics: Concepts, Methods, and Applications. Symmetry 2015, 7, 843–934. [Google Scholar] [CrossRef]
- Valen, L.V. A study of fluctuating asymmetry. Evolution 1962, 16, 125–142. [Google Scholar] [CrossRef]
- Klein, L.L.; Caito, M.; Chapnick, C.; Kitchen, C.; O’Hanlon, R.; Chitwood, D.H.; Miller, A.J. Digital Morphometrics of Two North American Grapevines (Vitis: Vitaceae) Quantifies Leaf Variation between Species, within Species, and among Individuals. Front. Plant Sci. 2017, 8, 373. [Google Scholar] [CrossRef] [PubMed]
- Chitwood, D.H.; Rundell, S.M.; Li, D.Y.; Woodford, Q.L.; Yu, T.T.; Lopez, J.R.; Greenblatt, D.; Kang, J.; Londo, J.P. Climate And Developmental Plasticity: Interannual Variability In Grapevine Leaf Morphology. Plant Physiol. 2016. Available online: http://www.plantphysiol.org/content/170/3/1480 (accessed on 9 July 2018).
- Chitwood, D.H.; Ranjan, A.; Martinez, C.C.; Headland, L.R.; Thiem, T.; Kumar, R.; Covington, M.F.; Hatcher, T.; Naylor, D.T.; Zimmerman, S.; et al. A Modern Ampelography: A Genetic Basis for Leaf Shape and Venation Patterning in Grape. Plant Physiol. 2014, 164, 259–272. [Google Scholar] [CrossRef] [PubMed]
- Martinez, C.C.; Chitwood, D.H.; Smith, R.S.; Sinha, N.R. Left–right leaf asymmetry in decussate and distichous phyllotactic systems. Philos. Trans. R. Soc. Lond. Ser. B 2016, 371. [Google Scholar] [CrossRef] [PubMed]
- Graham, J.H.; Whitesell, M.J., II; Hel-Or, H.; Nevo, E.; Raz, S. Fluctuating Asymmetry of Plant Leaves: Batch Processing with LAMINA and Continuous Symmetry Measures. Symmetry 2015, 7, 255–268. [Google Scholar] [CrossRef]
- Chen, T.W.; Henke, M.; de Visser, P.H.B.; Buck-Sorlin, G.; Wiechers, D.; Kahlen, K.; Stützel, H. What is the most prominent factor limiting photosynthesis in different layers of a greenhouse cucumber canopy? Ann. Bot. 2014, 114, 677–688. [Google Scholar] [CrossRef] [PubMed]
- Sarlikioti, V.; de Visser, P.H.B.; Buck-Sorlin, G.H.; Marcelis, L.F.M. How plant architecture affects light absorption and photosynthesis in tomato: Towards an ideotype for plant architecture using a functional-structural plant model. Ann. Bot. 2011, 108, 1065–1073. [Google Scholar] [CrossRef] [PubMed]
- Lewis, P. 3D canopy modelling as a tool in remote-sensing research. In Functional-Structural Plant Modelling in Crop Production; Vos, J., Marcelis, L.F.M., Visser, P., Struik, P.C., Evers, J.B., Eds.; Springer: Dordrecht, The Netherlands, 2007; Volume 22, pp. 219–229. [Google Scholar]
- Cieslak, M.; Lemieux, C.; Hanan, J.; Prusinkiewicz, P. Quasi-Monte Carlo simulation of the light environment of plants. Funct. Plant Biol. 2008, 35, 837–849. [Google Scholar] [CrossRef]
- Bookstein, F.L. Morphometric Tools for Landmark Data: Geometry and Biology; Cambridge University Press: Cambridge, UK, 1992. [Google Scholar]
- Walker, J.A. Ability of Geometric Morphometric Methods to Estimate a Known Covariance Matrix. Syst. Biol. 2000, 49, 686–696. [Google Scholar] [CrossRef] [PubMed]
- Zelditch, M.L.; Swiderski, D.L.; Sheets, H.D.; Fink, W.L. Geometric Morphometrics for Biologists; Academic Press: San Diego, CA, USA, 2004. [Google Scholar]
- Claes, P.; Daniels, K.; Walters, M.; Clement, J.; Vandermeulen, D.; Suetens, P. Dysmorphometrics: The modelling of morphological abnormalities. Theor. Biol. Med. Model. 2012, 9, 5. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Sheets, H.D.; Haney, R.A.; Mitchell, C.E. Morphometric Analysis of Ontogeny and Allometry of the Middle Ordovician Trilobite Triarthrus becki. Paleobiology 2002, 28, 364–377. [Google Scholar] [CrossRef]
- Webster, M.; Sheets, H.D.; Hughes, N.C. Allometric patterning in trilobite ontogeny: Testing for heterochrony in Nephrolenellus. In Beyond Heterochrony: The Evolution of Development; Zelditch, M.L., Ed.; Wiley-Liss: Hoboken, NJ, USA, 2001; pp. 105–144. [Google Scholar]
- Bookstein, F.L. Size and Shape Spaces for Landmark Data in Two Dimensions. Stat. Sci. 1986, 1, 181–222. [Google Scholar] [CrossRef]
- Mitteroecker, P.; Gunz, P. Advances in Geometric Morphometrics. Evol. Biol. 2009, 36, 235–247. [Google Scholar] [CrossRef]
- Kruschke, J.K. Bayesian estimation supersedes the t test. J. Exp. Psychol. Gen. 2013, 142, 573. [Google Scholar] [CrossRef] [PubMed]
- Kruschke, J.K.; Meredith, M. BEST: Bayesian Estimation Supersedes the t-Test, R Package Version 0.5.0; 2017. Available online: https://cran.r-project.org/package=BEST (accessed on 9 July 2018).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- Rakocevic, M.; Sinoquet, H.; Christophe, A.; Varlet-Grancher, C. Assessing the geometric structure of a white clover (Trifolium repens L.) canopy using3-D digitising. Ann. Bot. 2000, 86, 519–526. [Google Scholar] [CrossRef]
- Thanisawanyangkura, S.; Sinoquet, H.; Rivet, P.; Cretenet, M.; Jallas, E. Leaf orientation and sunlit leaf area distribution in cotton. Agric. For. Meteorol. 1997, 86, 1–15. [Google Scholar] [CrossRef]
- Kruschke, J.K.; Liddell, T.M. The Bayesian New Statistics: Hypothesis testing, estimation, meta-analysis, and power analysis from a Bayesian perspective. Psychon. Bull. Rev. 2017, 25, 178–206. [Google Scholar] [CrossRef] [PubMed]
- Merila, J.; Biorklund, M. Fluctuating Asymmetry and Measurement Error. Syst. Biol. 1995, 44, 97–101. [Google Scholar] [CrossRef]
- Schultheis, J.R.; Wehner, T.C.; Walters, S.A. Optimum planting density and harvest stage for little-leaf and normal-leaf cucumbers for once-over harvest. Can. J. Plant Sci. 1998, 78, 333–340. [Google Scholar] [CrossRef]



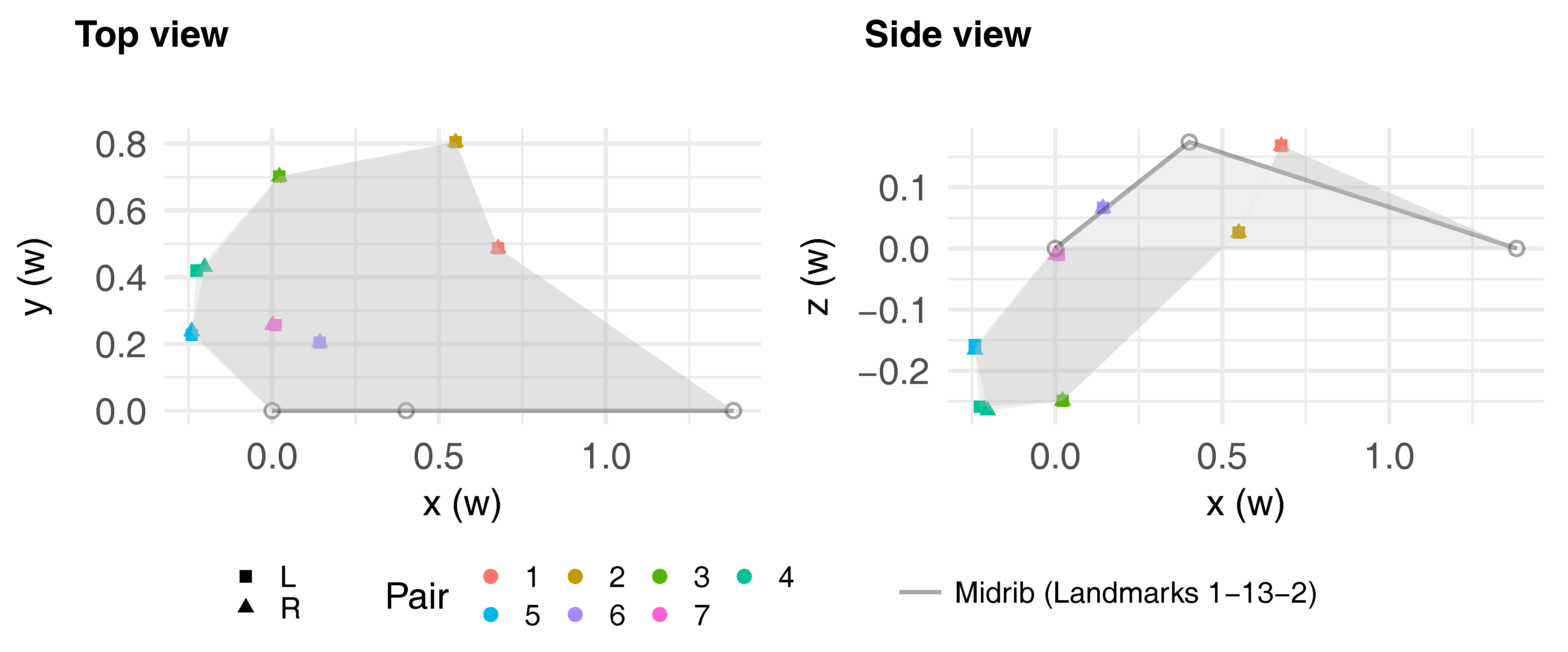
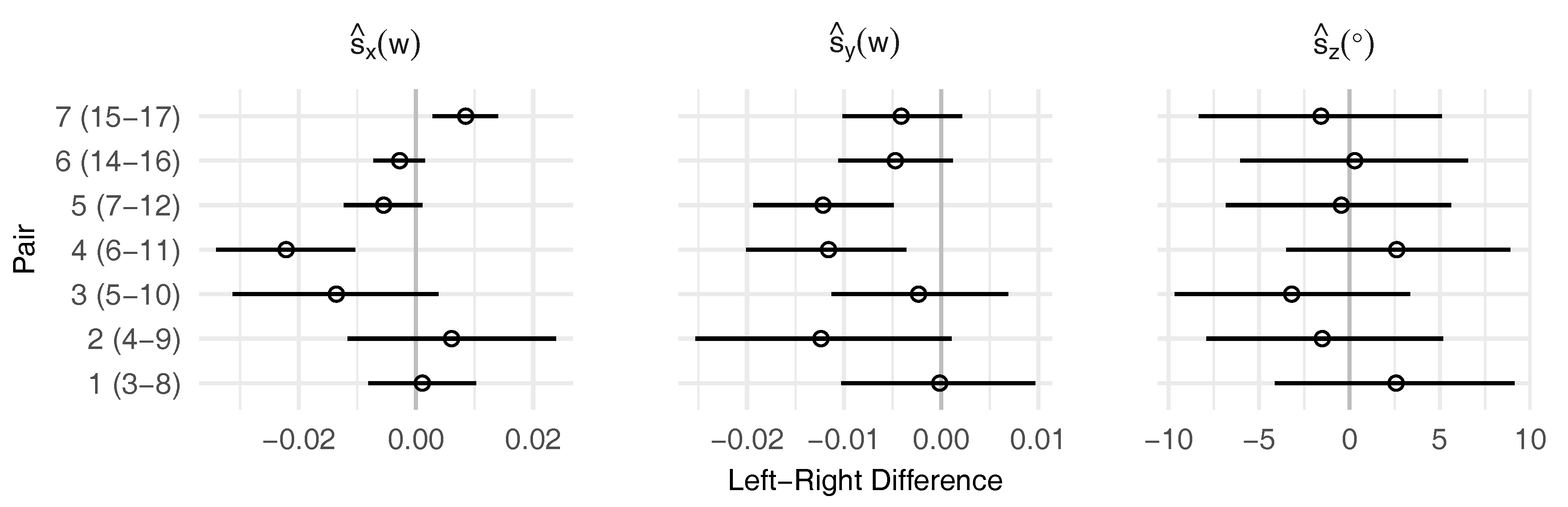
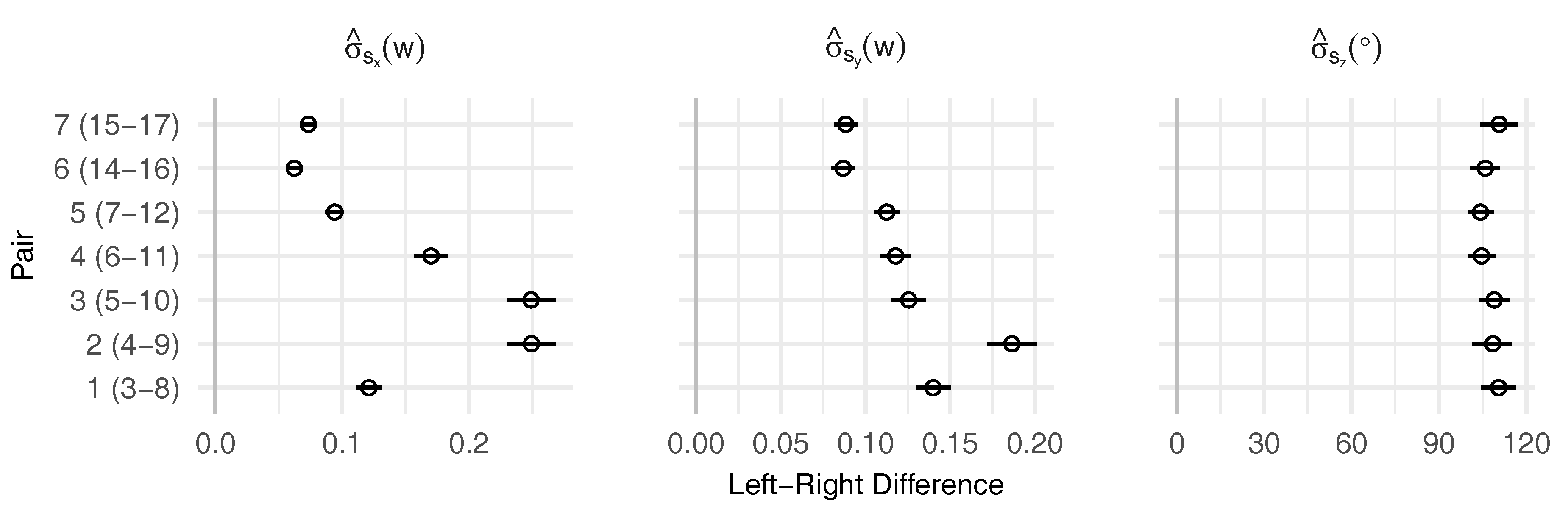


| Landmark | 3 | 8 | 4 | 9 | 5 | 10 | 6 | 11 | 7 | 12 | 14 | 16 | 15 | 17 | 2 | 13 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pair | 1 | 2 | 3 | 4 | 5 | 6 | 7 | |||||||||
| () | −0.12 | 0.01 | 0.02 | 0.29 | 0.31 | 0.02 | −0.12 | 0.009 | 0.001 | 0.29 | −0.12 | |||||
| () | 0.05 | −0.11 | −0.17 | −0.40 | −0.39 | −0.17 | −0.16 | −0.23 | −0.19 | – | 0.16 | |||||
| (°) | 34.03 | 16.86 | −4.52 | −16.63 | −19.85 | 33.00 | 12.65 | – | 0 | |||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schmidt, D.; Kahlen, K. Towards More Realistic Leaf Shapes in Functional-Structural Plant Models. Symmetry 2018, 10, 278. https://doi.org/10.3390/sym10070278
Schmidt D, Kahlen K. Towards More Realistic Leaf Shapes in Functional-Structural Plant Models. Symmetry. 2018; 10(7):278. https://doi.org/10.3390/sym10070278
Chicago/Turabian StyleSchmidt, Dominik, and Katrin Kahlen. 2018. "Towards More Realistic Leaf Shapes in Functional-Structural Plant Models" Symmetry 10, no. 7: 278. https://doi.org/10.3390/sym10070278
APA StyleSchmidt, D., & Kahlen, K. (2018). Towards More Realistic Leaf Shapes in Functional-Structural Plant Models. Symmetry, 10(7), 278. https://doi.org/10.3390/sym10070278





