Sustainable Polyculture of Hybrid Yellow Catfish (Pelteobagrus fulvidraco ♀ × P. vachelli ♂) and Chinese Olive Mussel (Solenaia oleivora) Enhances Water Quality and Modulates Bacterial Community Assembly in Pond Ecosystems
Abstract
1. Introduction
2. Materials and Methods
2.1. Pond Culture and Experimental Design
2.2. Sample Collection
2.3. DNA Extraction, PCR Amplification, and 16S rRNA Sequencing
2.4. Bacterial Community Profiling and Analysis
2.5. Bacterial Community-Water Quality Parameter Relationships
3. Results
3.1. The Influence of Polyculture Mode on the Weight Gain Rate
3.2. Water Quality Dynamics
3.3. Overview of 16S rRNA Sequencing Analysis
3.4. Bacterial Community Shifts
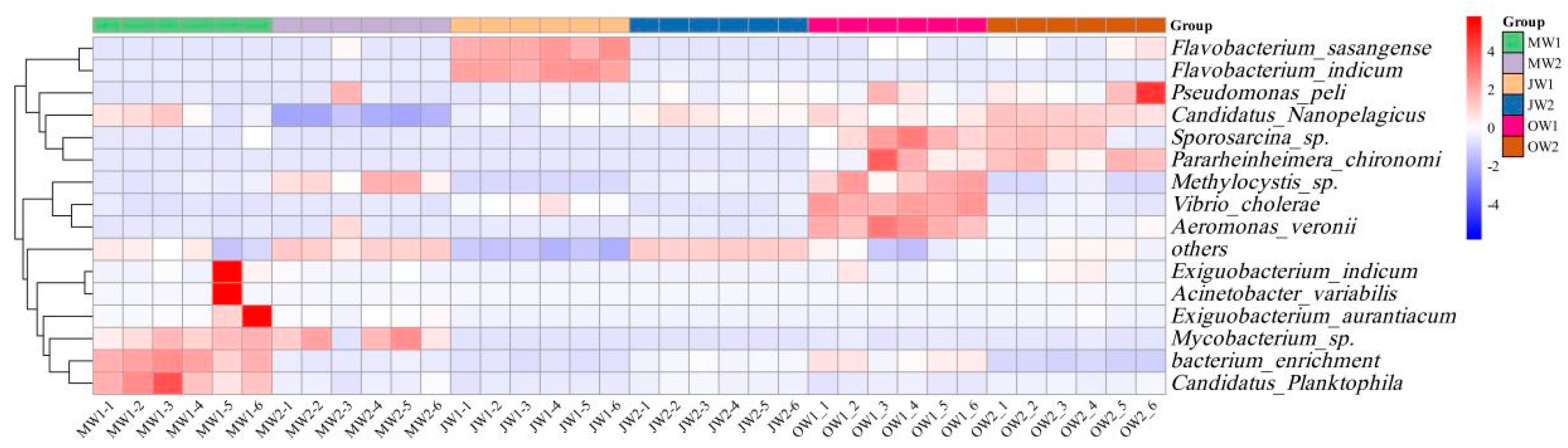
3.5. Taxonomic Composition of the Microbial Community
3.6. Correlations Between Water Quality Indicators and Dominant Bacteria
4. Discussion
4.1. Water Quality Dynamics in Polyculture Systems
4.2. Microbial Community Restructuring
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahmed, N.; Thompson, S. The blue dimensions of aquaculture: A global synthesis. Sci. Total Environ. 2019, 652, 851–861. [Google Scholar] [CrossRef] [PubMed]
- Ruben, M.O.; Akinsanola, A.B.; Okon, M.E.; Shitu, T.; Jagunna, I.I. Emerging challenges in aquaculture: Current perspectives and human health implications. Vet. World 2025, 18, 15–28. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Shin, S.K.; Do, Y.H.; Yarish, C.; Kim, J.K. Application of open water integrated multi-trophic aquaculture to intensive monoculture: A review of the current status and challenges in Korea. Aquaculture 2018, 497, 174–183. [Google Scholar] [CrossRef]
- Ahmed, N.; Thompson, S.; Turchini, G.M. Organic aquaculture productivity, environmental sustainability, and food security: Insights from organic agriculture. Food Secur. 2020, 12, 1253–1267. [Google Scholar] [CrossRef]
- Ghosh, A.K.; Hasanuzzaman, A.F.M.; Islam, S.S.; Sarower, M.G.; Mistry, S.K.; Arafat, S.T.; Huq, K.A. Integrated multi-trophic aquaculture (IMTA): Enhancing growth, production, immunological responses, and environmental management in aquaculture. Aquac. Int. 2025, 33, 336. [Google Scholar] [CrossRef]
- Thomas, M.; Pasquet, A.; Aubin, J.; Nahon, S.; Lecocq, T. When more is more: Taking advantage of species diversity to move towards sustainable aquaculture. Biol. Rev. 2021, 96, 767–784. [Google Scholar] [CrossRef]
- Batır, E.; Aydın, İ.; Theodorou, J.A.; Rakaj, A. Mytilus galloprovincialis’s role in Integrated Multi-Trophic Aquaculture (IMTA): A comprehensive review. J. World Aquac. Soc. 2025, 56, e70013. [Google Scholar] [CrossRef]
- Zheng, X.; Zhang, D.; Qin, J.; Wang, Y. The effect of C/N ratio on bacterial community and water quality in a mussel-fish integrated system. Aquac. Res. 2018, 49, 1699–1708. [Google Scholar] [CrossRef]
- Zhou, J.; Qiu, H.; Chen, Y.; Ma, X.; Yu, G.; Hong, Y.; Hu, B. Fish-mussel-algae-bacteria model remedied eutrophication pollution: Application in Dongxiang district reservoir. Environ. Pollut. 2024, 342, 123011. [Google Scholar] [CrossRef]
- Ji, Z.; Lu, X.; Xue, M.; Fan, Y.; Tian, J.; Dong, L.; Zhu, C.; Wen, H.; Jiang, M. The probiotic effects of host-associated Bacillus velezensis in diets for hybrid yellow catfish (Pelteobagrus fulvidraco ♀ × Pelteobagrus vachelli ♂). Anim. Nutr. 2023, 15, 114–125. [Google Scholar] [CrossRef]
- Zhang, E.; Yang, H.; Tu, X.; Gu, Z. First Investigation of Trichodinid Species (Ciliophora: Trichodinidae) on Farmed Hybrid Yellow Catfish (Tachysurus fulvidraco × Tachysurus vachelli) in China. J. Fish Dis. 2025, 48, e14100. [Google Scholar] [CrossRef] [PubMed]
- Amirkolaie, A.K. Reduction in the environmental impact of waste discharged by fish farms through feed and feeding. Rev. Aquac. 2011, 3, 19–26. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, G.-R.; Guo, S.-S.; Guo, X.-Z.; Wei, K.-J.; Ge, T.-M. Isolation and characterization of fifteen polymorphic microsatellite loci in the threatened freshwater mussel Solenaia oleivora (Bivalvia: Unionidae). Biochem. Syst. Ecol. 2013, 47, 104–107. [Google Scholar] [CrossRef]
- Chen, P.; Li, D.; Chen, X.; Zhang, G.; Yang, S. Molecular identification and phylogenetic analysis of the mitogenome of Solenaia oleivora MG. Mitochondrial DNA Part B 2020, 5, 2796–2798. [Google Scholar] [CrossRef] [PubMed]
- Yavuzcan Yildiz, H.; Robaina, L.; Pirhonen, J.; Mente, E.; Domínguez, D.; Parisi, G. Fish Welfare in Aquaponic Systems: Its Relation to Water Quality with an Emphasis on Feed and Faeces—A Review. Water 2017, 9, 13. [Google Scholar] [CrossRef]
- Nguyen, T.T.T.; Foysal, M.J.; Fotedar, R.; Gupta, S.K.; Siddik, M.A.B.; Tay, C.-Y. The Effect of Two Dietary Protein Sources on Water Quality and the Aquatic Microbial Communities in Marron (Cherax cainii) Culture. Microb. Ecol. 2021, 82, 299–308. [Google Scholar] [CrossRef]
- Wan Mahari, W.A.; Waiho, K.; Fazhan, H.; Azwar, E.; Shu-Chien, A.C.; Hersi, M.A.; Kasan, N.A.; Foo, S.S.; Wong, K.Y.; Draman, A.S.; et al. Emerging paradigms in sustainable shellfish aquaculture: Microalgae and biofloc technologies for wastewater treatment. Aquaculture 2024, 587, 740835. [Google Scholar] [CrossRef]
- Lacoste, É.; McKindsey, C.W.; Archambault, P. Biodiversity–Ecosystem Functioning (BEF) approach to further understanding aquaculture–environment interactions with application to bivalve culture and benthic ecosystems. Rev. Aquac. 2020, 12, 2027–2041. [Google Scholar] [CrossRef]
- Zheng, X.; Tang, J.; Zhang, C.; Qin, J.; Wang, Y. Bacterial composition, abundance and diversity in fish polyculture and mussel–fish integrated cultured ponds in China. Aquac. Res. 2017, 48, 3950–3961. [Google Scholar] [CrossRef]
- Hu, B.; Zhou, J.; Dong, J.; Yang, H.; Yu, G.; Hong, Y. Association of algae diversity and Hyriopsis schlegelii growth in mixed fish-mussel aquaculture. Algal Res. 2022, 65, 102736. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Segata, N.; Izard, J.; Waldron, L.; Gevers, D.; Miropolsky, L.; Garrett, W.S.; Huttenhower, C. Metagenomic biomarker discovery and explanation. Genome Biol. 2011, 12, R60. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zhou, Y.; Wen, H.; Ma, X.; Xu, D. Integrated analysis of physiological, transcriptome, and metabolome analyses of the gills in Solenaia oleivora under ammonia exposure. Ecotoxicol. Environ. Saf. 2024, 271, 115949. [Google Scholar] [CrossRef]
- Stefani, F.; Schiavon, A.; Tirozzi, P.; Gomarasca, S.; Marziali, L. Functional response of fish communities in a multistressed freshwater world. Sci. Total Environ. 2020, 740, 139902. [Google Scholar] [CrossRef]
- Huang, Y.; Yang, H.; Li, K.; Meng, Q.; Wang, S.; Wang, Y.; Zhu, P.; Niu, Q.; Yan, H.; Li, X.; et al. Red mud conserved compost nitrogen by enhancing nitrogen fixation and inhibiting denitrification revealed via metagenomic analysis. Bioresour. Technol. 2022, 346, 126654. [Google Scholar] [CrossRef]
- Yuan, H.; Zhang, R.; Lu, Q.; Huang, Q.; Chen, J. Response of bacterioplankton communities and their phosphorus metabolic functions to algal extinction and growth in a eutrophic plateau lake. Environ. Technol. Innov. 2025, 38, 104108. [Google Scholar] [CrossRef]
- Korlević, M.; Šupraha, L.; Ljubešić, Z.; Henderiks, J.; Ciglenečki, I.; Dautović, J.; Orlić, S. Bacterial diversity across a highly stratified ecosystem: A salt-wedge Mediterranean estuary. Syst. Appl. Microbiol. 2016, 39, 398–408. [Google Scholar] [CrossRef]
- Brindefalk, B.; Ettema, T.J.G.; Viklund, J.; Thollesson, M.; Andersson, S.G.E. A Phylometagenomic Exploration of Oceanic Alphaproteobacteria Reveals Mitochondrial Relatives Unrelated to the SAR11 Clade. PLoS ONE 2011, 6, e24457. [Google Scholar] [CrossRef]
- Zhao, Y.-X.; Liu, X.-G.; Lu, M.; Zhou, R.-F.; Sun, Z.-Y.; Xiao, S.-W. Characteristics of Bacterial Community in Pelteobagrus fulvidraco Integrated Multi-Trophic Aquaculture System. Water 2022, 14, 3192. [Google Scholar] [CrossRef]
- Ruprecht, J.E.; Birrer, S.C.; Dafforn, K.A.; Mitrovic, S.M.; Crane, S.L.; Johnston, E.L.; Wemheuer, F.; Navarro, A.; Harrison, A.J.; Turner, I.L.; et al. Wastewater effluents cause microbial community shifts and change trophic status. Water Res. 2021, 200, 117206. [Google Scholar] [CrossRef]
- Liu, J.; Fu, B.; Yang, H.; Zhao, M.; He, B.; Zhang, X.-H. Phylogenetic shifts of bacterioplankton community composition along the Pearl Estuary: The potential impact of hypoxia and nutrients. Front. Microbiol. 2015, 6, 64. [Google Scholar] [CrossRef]
- Dahle, S.W.; Bakke, I.; Birkeland, M.; Nordøy, K.; Dalum, A.S.; Attramadal, K.J.K. Production of lumpfish (Cyclopterus lumpus L.) in RAS with distinct water treatments: Effects on fish survival, growth, gill health and microbial communities in rearing water and biofilm. Aquaculture 2020, 522, 735097. [Google Scholar] [CrossRef]
- Wang, M.; Feng, W.; Wang, Y.; Li, B.; Wang, J.; Zhu, X.; Zhang, L. Water quality, plankton composition, and growth performance of juvenile yellow catfish (Pelteobagrus fulvidraco) in mono- and polyculture systems. Aquaculture 2022, 552, 738017. [Google Scholar] [CrossRef]






| Culture Modes | Initial Body Weight (g) | Harvest Body Weight (g) | Weight Gain Rate (%) |
|---|---|---|---|
| Monoculture | 24.99 ± 1.37 | 181.54 ± 10.47 | 626.45 |
| Polyculture | 25.25 ± 1.39 | 201.86 ± 13.03 * | 699.45 |
| Index | Culture Mode | May | July | October |
|---|---|---|---|---|
| T (°C) | Monoculture | 25.16 ± 0.06 a | 35.93 ± 0.15 a | 22.2 ± 0.07 a |
| Polyculture | 25.77 ± 0.12 a | 36.23 ± 0.25 a | 21.7 ± 0.13 a | |
| pH | Monoculture | 7.87 ± 0.07 a | 8.52 ± 0.06 a | 8.49 ± 0.05 a |
| Polyculture | 7.88 ± 0.28 a | 8.59 ± 0.04 a | 8.51 ± 0.09 a | |
| DO (mg/L) | Monoculture | 8.81 ± 0.28 a | 11.96 ± 0.29 a | 6.57 ± 0.12 a |
| Polyculture | 8.69 ± 1.14 a | 11.67 ± 0.22 a | 6.08 ± 0.09 a | |
| TN (mg/L) | Monoculture | 2.33 ± 0.21 a | 2.74 ± 0.91 a | 3.44 ± 0.13 a |
| Polyculture | 2.94 ± 0.43 a | 1.95 ± 0.37 b | 1.89 ± 0.05 b | |
| DTN (mg/L) | Monoculture | 2.13 ± 0.19 a | 2.46 ± 0.69 a | 2.41 ± 0.008 a |
| Polyculture | 2.43 ± 0.45 a | 1.52 ± 0.28 b | 1.64 ± 0.006 b | |
| NH3-N (mg/L) | Monoculture | 0.14 ± 0.09 b | 1.88 ± 0.68 a | 1.16 ± 0.10 a |
| Polyculture | 0.33 ± 0.06 a | 1.25 ± 0.32 a | 0.29 ± 0.05 b | |
| NO2−-N(mg/L) | Monoculture | 0.12 ± 0.01 b | 0.007 ± 0.003 a | 0.006 ± 0.001 |
| Polyculture | 0.20 ± 0.03 a | 0.003 ± 0.002 a | ND | |
| TP (mg/L) | Monoculture | 0.37 ± 0.02 a | 1.45 ± 0.04 a | 0.44 ± 0.005 a |
| Polyculture | 0.29 ± 0.02 b | 0.73 ± 0.01 b | 0.32 ± 0.008 b | |
| DTP (mg/L) | Monoculture | 0.28 ± 0.02 a | 1.39 ± 0.01 a | 0.09 ± 0.006 a |
| Polyculture | 0.16 ± 0.01 b | 0.71 ± 0.00 b | 0.06 ± 0.004 a | |
| PO43−-P (mg/L) | Monoculture | 0.30 ± 0.02 a | 1.31 ± 0.03 a | 0.42 ± 0.006 a |
| Polyculture | 0.16 ± 0.03 b | 0.69 ± 0.02 b | 0.26 ± 0.007 b | |
| CODMn (mg/L) | Monoculture | 8.78 ± 0.12 a | 8.43 ± 0.06 a | 9.66 ± 0.08 a |
| Polyculture | 8.65 ± 0.13 a | 8.16 ± 0.15 b | 9.65 ± 0.11 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Zhou, H.; Hu, F.; Hu, Y.; Liu, A.; Zhang, Y.; Duan, G. Sustainable Polyculture of Hybrid Yellow Catfish (Pelteobagrus fulvidraco ♀ × P. vachelli ♂) and Chinese Olive Mussel (Solenaia oleivora) Enhances Water Quality and Modulates Bacterial Community Assembly in Pond Ecosystems. Water 2025, 17, 3208. https://doi.org/10.3390/w17223208
Wang H, Zhou H, Hu F, Hu Y, Liu A, Zhang Y, Duan G. Sustainable Polyculture of Hybrid Yellow Catfish (Pelteobagrus fulvidraco ♀ × P. vachelli ♂) and Chinese Olive Mussel (Solenaia oleivora) Enhances Water Quality and Modulates Bacterial Community Assembly in Pond Ecosystems. Water. 2025; 17(22):3208. https://doi.org/10.3390/w17223208
Chicago/Turabian StyleWang, Huan, Huaxing Zhou, Feiyu Hu, Yuting Hu, Amei Liu, Ye Zhang, and Guoqing Duan. 2025. "Sustainable Polyculture of Hybrid Yellow Catfish (Pelteobagrus fulvidraco ♀ × P. vachelli ♂) and Chinese Olive Mussel (Solenaia oleivora) Enhances Water Quality and Modulates Bacterial Community Assembly in Pond Ecosystems" Water 17, no. 22: 3208. https://doi.org/10.3390/w17223208
APA StyleWang, H., Zhou, H., Hu, F., Hu, Y., Liu, A., Zhang, Y., & Duan, G. (2025). Sustainable Polyculture of Hybrid Yellow Catfish (Pelteobagrus fulvidraco ♀ × P. vachelli ♂) and Chinese Olive Mussel (Solenaia oleivora) Enhances Water Quality and Modulates Bacterial Community Assembly in Pond Ecosystems. Water, 17(22), 3208. https://doi.org/10.3390/w17223208






