Aquatic Biota Is Not Exempt from Coronavirus Infections: An Overview
Abstract
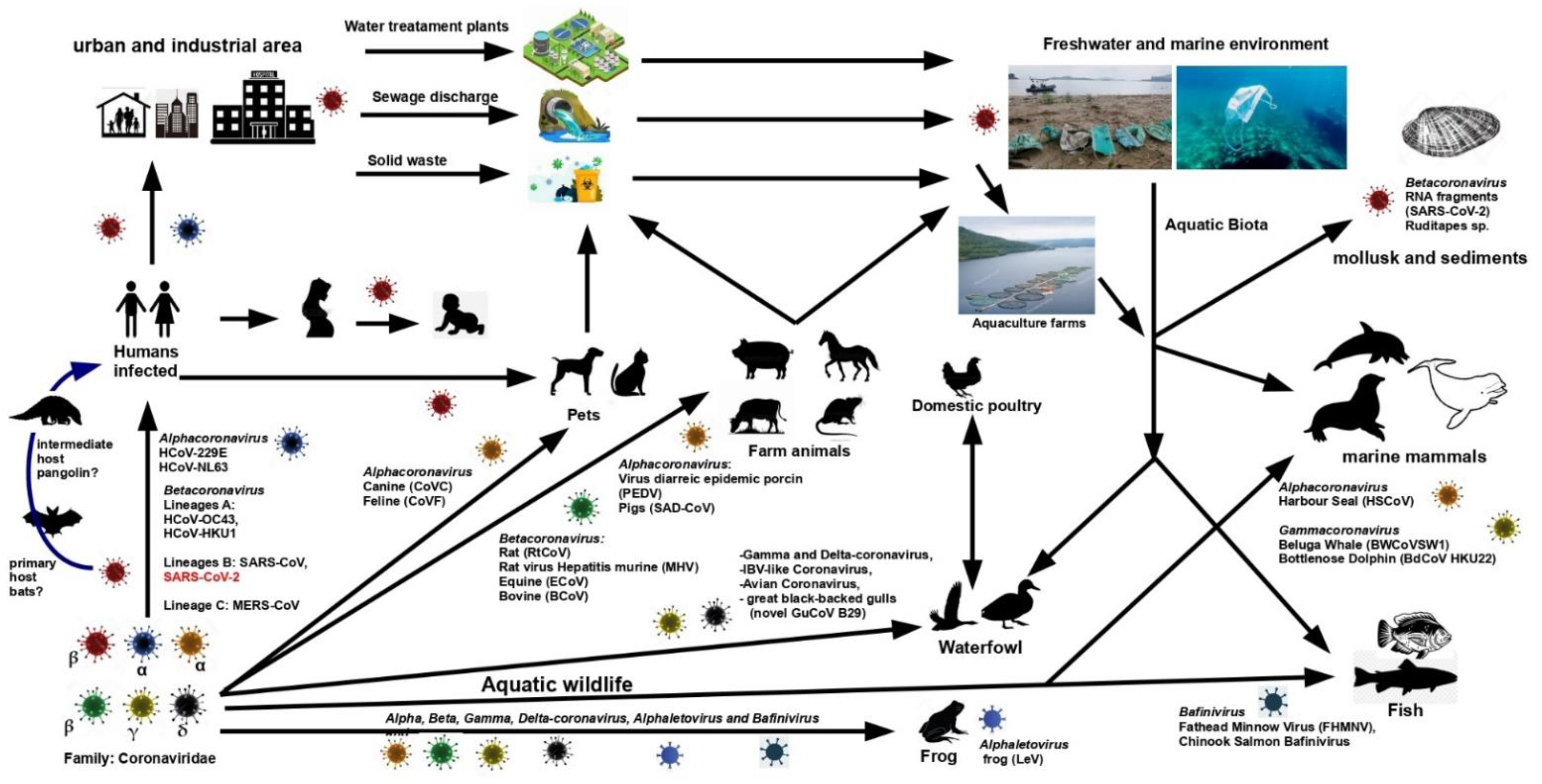
1. Introduction
2. Coronavirus and Aquatic Environments
2.1. Coronavirus in Natural Waters
2.2. CoVs and Aquatic Organisms
2.2.1. Fish
2.2.2. Waterfowl
2.2.3. Amphibians
2.2.4. Marine Mammals
2.2.5. Crustaceans and Mollusks
3. Some Viral Infections to Humans from Aquatic Organisms
4. Further Considerations about CoVs and Aquatic Biota
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Burki, T. Outbreak of coronavirus disease 2019. Lancet Infect. Dis. 2020, 20, 292–293. [Google Scholar] [CrossRef]
- Costagliola, A.; Liguori, G.; D’angelo, D.; Costa, C.; Ciani, F.; Giordano, A. Do animals play a role in the transmission of severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2)? a commentary. Animals 2021, 11, 16. [Google Scholar] [CrossRef]
- Haleem, A.; Javaid, M.; Vaishya, R.; Deshmukh, S.G. Areas of academic research with the impact of COVID-19. Am. J. Emerg. Med. 2020, 38, 1524–1526. [Google Scholar] [CrossRef] [PubMed]
- Qu, G.; Li, X.; Hu, L.; Jiang, G. An Imperative Need for Research on the Role of Environmental Factors in Transmission of Novel Coronavirus (COVID-19). Environ. Sci. Technol. 2020, 54, 3730–3732. [Google Scholar] [CrossRef]
- Bosch, A.; Pintó, R.M.; Le Guyader, F.S. Viral contaminants of molluscan shellfish: Detection and characterisation. In Shellfish Safety and Quality; Shumway, S.E., Rodrick, G.E., Eds.; Academic Press: London, UK, 2009; pp. 83–107. [Google Scholar] [CrossRef]
- Bouseettine, R.; Hassou, N.; Bessi, H.; Ennaji, M.M. Waterborne Transmission of Enteric Viruses and Their Impact on Public Health; Elsevier Inc.: London, UK, 2019; ISBN 9780128194003. [Google Scholar]
- de Groot, R.; Baker, S.; Baric, R.; Enjuanes, L.; Gorbalenya, A.; Holmes, K.; Perlman, S.; Poon, L.; Rottier, P.; Talbot, P.; et al. Part II—The Positive Sense Single Stranded RNA Viruses Family Coronaviridae. In Virus Taxonomy: Ninth Report of the International Committee on Taxonomy of Viruses; King, A.M.Q., Adams, M.J., Carstens, E.B., Leftkowitz, E.J., Eds.; Academic Press: London, UK, 2012; pp. 806–828. ISBN 9780123846846. [Google Scholar]
- Avendaño-López, C. Aportaciones de las ciencias biomédicas en el estado de alarma motivado por la pandemia del virus COV-2. An. la Real Académia Nac. Farm. 2020, 86, 9–17. [Google Scholar]
- Malik, Y.S.; Sircar, S.; Bhat, S.; Sharun, K.; Dhama, K.; Dadar, M.; Tiwari, R.; Chaicumpa, W. Emerging novel coronavirus (2019-nCoV)—current scenario, evolutionary perspective based on genome analysis and recent developments. Vet. Q. 2020, 40, 68–76. [Google Scholar] [CrossRef]
- Woo, P.C.Y.; Lau, S.K.P.; Lam, C.S.F.; Tsang, A.K.L.; Hui, S.-W.; Fan, R.Y.Y.; Martelli, P.; Yuen, K.-Y. Discovery of a Novel Bottlenose Dolphin Coronavirus Reveals a Distinct Species of Marine Mammal Coronavirus in Gammacoronavirus. J. Virol. 2014, 88, 1318–1331. [Google Scholar] [CrossRef] [PubMed]
- Kasmi, Y.; Khataby, K.; Souiri, A.; Ennaji, M.M. Coronaviridae: 100,000 years of emergence and reemergence. In Emerging and Reemerging Viral Pathogens: Volume 1: Fundamental and Basic Virology Aspects of Human, Animal and Plant Pathogens; Ennaji, M.M., Ed.; Academic Press: London, UK, 2019; pp. 127–149. ISBN 9780128194003. [Google Scholar]
- Bukhari, K.; Mulley, G.; Gulyaeva, A.A.; Zhao, L.; Shu, G.; Jiang, J.; Neuman, B.W. Description and initial characterization of metatranscriptomic nidovirus-like genomes from the proposed new family Abyssoviridae, and from a sister group to the Coronavirinae, the proposed genus Alphaletovirus. Virology 2018, 524, 160–171. [Google Scholar] [CrossRef] [PubMed]
- Granzow, H.; Weiland, F.; Fichtner, D.; Schütze, H.; Karger, A.; Mundt, E.; Dresenkamp, B.; Martin, P.; Mettenleiter, T.C. Identification and ultrastructural characterization of a novel virus from fish. J. Gen. Virol. 2001, 82, 2849–2859. [Google Scholar] [CrossRef] [PubMed]
- Leong, J.C. Fish Viruses. In Encyclopedia of Virology; Mahy, B.W.J., Van Regenmortel, M.H.V., Eds.; Academic Press: London, UK, 2008; pp. 227–234. ISBN 9780123744104. [Google Scholar]
- Alexyuk, M.S.; Turmagambetova, A.S.; Alexyuk, P.G.; Bogoyavlenskiy, A.P.; Berezin, V.E. Comparative study of viromes from freshwater samples of the Ile-Balkhash region of Kazakhstan captured through metagenomic analysis. VirusDisease 2017, 28, 18–25. [Google Scholar] [CrossRef]
- Aguiar-Oliveira, M.d.L.; Campos, A.; Matos, A.R.; Rigotto, C.; Sotero-Martins, A.; Teixeira, P.F.P.; Siqueira, M.M. Wastewater-based epidemiology (Wbe) and viral detection in polluted surface water: A valuable tool for covid-19 surveillance—A brief review. Int. J. Environ. Res. Public Health 2020, 17, 9251. [Google Scholar] [CrossRef] [PubMed]
- Roa, V.C.; Melnick, J.L. Environmental Virology; Van Nostrand Reinhold Co. Ltd.: Berkshire, UK, 1986; ISBN 9788578110796. [Google Scholar]
- Melnick, J.L. Etiologic Agents and Their Potential for Causing Waterborne Virus Diseases. Monogr. Virol. 1984, 15, 1–16. [Google Scholar]
- Petrović, T.; D’Agostino, M. Viral Contamination of Food. In Antimicrobial Food Packaging; Barros-Velázquez, J., Ed.; Academic Press: Amsterdam, The Netherlands, 2016; pp. 65–79. ISBN 9780128007235. [Google Scholar]
- Farthing, M.J.G. Viruses and the Gut; Farthing, M.J.G., Ed.; Smith Kline & French Ltd.: Walwyn Garden City, Hertfordshire, UK, 1989; ISBN 0948271086. [Google Scholar]
- Lesté-Lasserre, C. Coronavirus found in Paris sewage points to early warning system. Sci. News. 2020, 1. Available online: https://www.sciencemag.org/news/2020/04/coronavirus-found-paris-sewage-points-early-warning-system (accessed on 8 May 2020). [CrossRef]
- Macnaughton, M.R. Occurrence and frequency of coronavirus infections in humans as determined by enzyme-linked immunosorbent assay. Infect. Immun. 1982, 38, 419–423. [Google Scholar] [CrossRef]
- Feichtmayer, J.; Deng, L.; Griebler, C. Antagonistic microbial interactions: Contributions and potential applications for controlling pathogens in the aquatic systems. Front. Microbiol. 2017, 8, 1–14. [Google Scholar] [CrossRef]
- Wang, X.W.; Li, J.S.; Guo, T.K.; Zhen, B.; Kong, Q.X.; Yi, B.; Li, Z.; Song, N.; Jin, M.; Xiao, W.J.; et al. Concentration and detection of SARS coronavirus in sewage from Xiao Tang Shan Hospital and the 309th Hospital. J. Virol. Methods 2005, 128, 156–161. [Google Scholar] [CrossRef]
- Casanova, L.; Rutala, W.A.; Weber, D.J.; Sobsey, M.D. Survival of surrogate coronaviruses in water. Water Res. 2009, 43, 1893–1898. [Google Scholar] [CrossRef]
- Wang, X.W.; Li, J.S.; Jin, M.; Zhen, B.; Kong, Q.X.; Song, N.; Xiao, W.J.; Yin, J.; Wei, W.; Wang, G.J.; et al. Study on the resistance of severe acute respiratory syndrome-associated coronavirus. J. Virol. Methods 2005, 126, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Prussin, A.J.; Schwake, D.O.; Lin, K.; Gallagher, D.L.; Buttling, L.; Marr, L.C. Survival of the enveloped virus Phi6 in droplets as a function of relative humidity, absolute humidity, and temperature. Appl. Environ. Microbiol. 2018, 84, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hasija, M.; Li, L.; Rahman, N.; Ausar, S.F. Forced degradation studies: An essential tool for the formulation development of vaccines. Vaccine Dev. Ther. 2013, 3, 11–33. [Google Scholar] [CrossRef][Green Version]
- Sharma, S.; Gray, D.K.; Read, J.S.; O’Reilly, C.M.; Schneider, P.; Qudrat, A.; Gries, C.; Stefanoff, S.; Hampton, S.E.; Hook, S.; et al. A global database of lake surface temperatures collected by in situ and satellite methods from 1985–2009. Sci. Data 2015, 2, 1–19. [Google Scholar] [CrossRef]
- Padisák, J.; Reynolds, C.S. Shallow lakes: The absolute, the relative, the functional and the pragmatic. Hydrobiologia 2003, 506–509, 1–11. [Google Scholar] [CrossRef]
- Heldal, M.; Bratbak, G. Production and decay of viruses in aquatic environments. Mar. Ecol. Prog. Ser. 1991, 72, 205–212. [Google Scholar] [CrossRef]
- Wommack, K.E.; Hill, R.T.; Muller, T.A.; Colwell, R.R. Effects of sunlight on bacteriophage viability and structure. Appl. Environ. Microbiol. 1996, 62, 1336–1341. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, S.W.; Weinbauer, M.G.; Suttle, C.A.; Jeffrey, W.H. The role of sunlight in the removal and repair of viruses in the sea. Limnol. Oceanogr. 1998, 43, 586–592. [Google Scholar] [CrossRef]
- Darnell, M.E.R.; Subbarao, K.; Feinstone, S.M.; Taylor, D.R. Inactivation of the coronavirus that induces severe acute respiratory syndrome, SARS-CoV. J. Virol. Methods 2004, 121, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Pratelli, A. Canine coronavirus inactivation with physical and chemical agents. Vet. J. 2008, 177, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Murray, A.G.; Jackson, G.A. Viral dynamics: A model of the effects of size, shape, motion and abundance of single-celled planktonic organisms and other particles. Mar. Ecol. Prog. Ser. 1992, 89, 103–116. [Google Scholar] [CrossRef]
- Noble, R.T.; Fuhrman, J.A. Virus decay and its causes in coastal waters. Appl. Environ. Microbiol. 1997, 63, 77–83. [Google Scholar] [CrossRef]
- Wartecki, A.; Rzymski, P. On the coronaviruses and their associations with the aquatic environment and wastewater. Water 2020, 12, 1598. [Google Scholar] [CrossRef]
- Seyer, A.; Şanlıdağ, T. The fate of SARS-CoV-2 in the marine environments: Are marine environments safe of COVID-19? Erciyes Med. J. 2021, 10–11. [Google Scholar] [CrossRef]
- Bosch, A.; Abad, F.X.; Pintó, R.M. Human pathogenic viruses in the marine environment. In Oceans and Health: Pathogens in the Marine Environment; Belkin, C., Ed.; Springer: New York, NY, USA, 2005; pp. 109–131. ISBN 9780387237091. [Google Scholar]
- Jeong, H.W.; Kim, S.; Kim, H.; Kim, Y.; Kim, J.H.; Cho, J.Y.; Kim, S.; Kang, H.; Kim, S.; Park, S.; et al. Viable SARS-CoV-2 in various specimens from COVID-19 patients. Clin. Microbiol. Infect. 2020, 26, 1520–1524. [Google Scholar] [CrossRef]
- Sobsey, M.D.; Meschke, J.S. Virus Survival in the Environment with Special Attention to Survival in Sewage Droplets and Other Environmental Media of Fecal or Respiratory Origin; World Health Organization: Geneva, Switzerland, 2003; p. 70. [Google Scholar]
- Geller, C.; Varbanov, M.; Duval, R.E. Human coronaviruses: Insights into environmental resistance and its influence on the development of new antiseptic strategies. Viruses 2012, 4, 3044–3068. [Google Scholar] [CrossRef]
- Saraswat, R.; Kouthanker, M.; Kurtarkar, S.; Nigam, R.; Linshy, V.N. Effect of salinity induced pH changes on benthic foraminifera: A laboratory culture experiment. Biogeosciences Discuss. 2011, 8, 8423–8450. [Google Scholar] [CrossRef]
- NOAA. Why Is the Ocean Salty? Available online: https://oceanservice.noaa.gov/facts/whysalty.html (accessed on 3 June 2020).
- NOAA. Ocean Acidification. Available online: https://www.noaa.gov/education/resource-collections/ocean-coasts/ocean-acidification.html (accessed on 3 June 2020).
- Natarajan, P.; Miller, A. Recreational Infections. In Infectious Diseases; Cohen, J., Powerly, W.G., Opal, S.M., Eds.; Elsevier Ltd.: London, UK, 2017; pp. 643–646. [Google Scholar]
- Shapiro, D.S. Infections Acquired from Animals Other Than Pets. In Infectious Diseases; Cohen, J., Powderly, W.G., Opal, S.M., Eds.; Elsevier Ltd.: London, UK, 2017; pp. 663–669. [Google Scholar]
- Polo, D.; Lois, M.; Fernández-Núñez, M.T.; Romalde, J.L. Detection of SARS-CoV-2 RNA in bivalve mollusks and marine sediments. Sci. Total Environ. 2021, 786, 147534. [Google Scholar] [CrossRef] [PubMed]
- Sano, T.; Yamaki, T.; Fukuda, H. A novel carp coronavirus: Characterization and pathogenicity. In Proceedings of the Fishery Health Conference, Vancouver, BC, Canada, 19–21 July 1988; p. 160. [Google Scholar]
- Schütze, H. Coronaviruses in Aquatic Organisms. In Aquaculture Virology; Kibenge, F., Godoy, M., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2016; pp. 327–335. ISBN 9780128017548. [Google Scholar]
- Miao, W. Aquaculture Production and Trade Trends: Carp, Tilapia and Shrimp Weimin Miao; FAO RAP: Rome, Italy, 2015. [Google Scholar]
- Karnai, L.; Szucs, I. Outlooks and Perspectives of the Common Carp Production. Ann. Polish Assoc. Agric. Agribus. Econ. 2018, 64–72. [Google Scholar] [CrossRef]
- Batts, W.N.; Goodwin, A.E.; Winton, J.R. Genetic analysis of a novel nidovirus from fathead minnows. J. Gen. Virol. 2012, 93, 1247–1252. [Google Scholar] [CrossRef]
- Iwanowicz, L.R.; Goodwin, A.E. A new bacilliform fathead minnow rhabdovirus that produces syncytia in tissue culture. Arch. Virol. 2002, 147, 899–915. [Google Scholar] [CrossRef]
- Lord, S.D.; Raymond, M.J.; Krell, P.J.; Kropinski, A.M.; Stevenson, R.M.W. Novel chinook salmon bafinivirus isolation from ontario fish health monitoring. In Proceedings of the Proceedings of the Seventh International Symposium on Aquatic Animal Health; Portland, OR, USA, 31 August–4 September 2014, p. 242.
- Ahne, W. Viral infections of aquatic animals with special reference to Asian aquaculture. Annu. Rev. Fish Dis. 1994, 4, 375–388. [Google Scholar] [CrossRef]
- Miyazaki, T.; Okamoto, H.; Kageyama, T.; Kobayashi, T. Viremia-associated ana-aki-byo, a new viral disease in color carp Cyprinus carpio in Japan. Dis. Aquat. Organ. 2000, 39, 183–192. [Google Scholar] [CrossRef]
- Bossart, G.D.; Schwartz, J.C. Acute Necrotizing Enteritis Associated with Suspected Coronavirus Infection in Three Harbor seals (Phoca vitulina). J. Zoo Wildl. Med. 1990, 21, 84–87. [Google Scholar]
- Nollens, H.H.; Wellehan, J.F.X.; Archer, L.; Lowenstine, L.J.; Gulland, F.M.D. Detection of a respiratory coronavirus from tissues archived during a pneumonia epizootic in free-ranging pacific harbor seals Phoca vitulina richardsii. Dis. Aquat. Organ. 2010, 90, 113–120. [Google Scholar] [CrossRef]
- Mihindukulasuriya, K.A.; Wu, G.; St. Leger, J.; Nordhausen, R.W.; Wang, D. Identification of a Novel Coronavirus from a Beluga Whale by Using a Panviral Microarray. J. Virol. 2008, 82, 5084–5088. [Google Scholar] [CrossRef]
- Youdao, N. Can freshwater fish transmit novel coronavirus? Chinese W. 2020, 19–20. Available online: https://covid-19.chinadaily.com.cn/a/202003/20/WS5e73f268a310128217280910.html (accessed on 6 August 2021).
- Casanova, L.M.; Jeon, S.; Rutala, W.A.; Weber, D.J.; Sobsey, M.D. Effects of air temperature and relative humidity on coronavirus survival on surfaces. Appl. Environ. Microbiol. 2010, 76, 2712–2717. [Google Scholar] [CrossRef] [PubMed]
- Araujo, M.B.; Naimi, B. Spread of SARS-CoV-2 Coronavirus likely to be constrained by climate. medRxiv 2020, 1–15. [Google Scholar] [CrossRef]
- Eckert, R.; Randall, D.; Augustine, G. Fisología Animal: Mecanismos y Adaptaciones, 3rd ed.; McGraw-Hill-Interamericana: Madrid, España, 1990. [Google Scholar]
- Peteri, A. Cyprinus carpio (Linnaeus, 1758). In Cultured Aquatic Species Fact Sheets; Crespi, V., New, M., Eds.; FAO: Rome, Italy, 2009; pp. 1–15. [Google Scholar]
- Melero, M.; Rodríguez-Prieto, V.; Rubio-García, A.; García-Párraga, D.; Sánchez-Vizcaíno, J.M. Thermal reference points as an index for monitoring body temperature in marine mammals. BMC Res. Notes 2015, 8, 411. [Google Scholar] [CrossRef] [PubMed]
- Nuñez-Nogueira, G. Coronavirus en aves acuáticas. Kuxulkab' 2020, 26, 51–59. [Google Scholar] [CrossRef]
- Chu, D.K.W.; Leung, C.Y.H.; Gilbert, M.; Joyner, P.H.; Ng, E.M.; Tse, T.M.; Guan, Y.; Peiris, J.S.M.; Poon, L.L.M. Avian Coronavirus in Wild Aquatic Birds. J. Virol. 2011, 85, 12815–12820. [Google Scholar] [CrossRef]
- Shi, Z.; Hu, Z. A review of studies on animal reservoirs of the SARS coronavirus. Virus Res. 2008, 133, 74–87. [Google Scholar] [CrossRef]
- Barbosa, C.M.; Durigon, E.L.; Thomazelli, L.M.; Ometto, T.; Marcatti, R.; Nardi, M.S.; de Aguiar, D.M.; Pinho, J.B.; Petry, M.V.; Neto, I.S.; et al. Divergent coronaviruses detected in wild birds in Brazil, including a central park in São Paulo. Braz. J. Microbiol. 2019, 50, 547–556. [Google Scholar] [CrossRef] [PubMed]
- Hepojoki, S.; Lindh, E.; Vapalahti, O.; Huovilainen, A. Prevalence and genetic diversity of coronaviruses in wild birds, Finland. Infect. Ecol. Epidemiol. 2017, 7, 1408360. [Google Scholar] [CrossRef] [PubMed]
- de Sales Lima, F.E.; Gil, P.; Pedrono, M.; Minet, C.; Kwiatek, O.; Campos, F.S.; Spilki, F.R.; Roehe, P.M.; Franco, A.C.; Maminiaina, O.F.; et al. Diverse gammacoronaviruses detected in wild birds from Madagascar. Eur. J. Wildl. Res. 2015, 61, 635–639. [Google Scholar] [CrossRef]
- Uhart, M.; Vanstreels, R.E.T.; Gallo, L.; Cook, R.A.; Karesh, W.B. Serological survey for select infectious agents in wild magellanic penguins (Spheniscus magellanicus) in Argentina, 1994–2008. J. Wildl. Dis. 2020, 56, 66–81. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.M.; Karesh, W.B.; Majluf, P.; Paredes, R.; Reul, A.H.; Stetter, M.; Braselton, W.E.; Puche, H.; Cook, R.A.; Kristine, M.; et al. Health Evaluation of Free-Ranging Humboldt Penguins (Spheniscus humboldti) in Peru. Avian Dis. 2008, 52, 130–135. [Google Scholar] [CrossRef]
- Karesh, W.B.; Uhart, M.M.; Frere, E.; Gandini, P.; Braselton, E.; Puche, H.; Cook, R.A. Health evaluation of free-ranging rockhopper penguins (Eudyptes chrysocomes) in Argentina. J. Zoo Wildl. Med. 1999, 30, 25–31. [Google Scholar] [PubMed]
- FAO. Food Chain Crisis Early Warning Bulletin; FAO: Rome, Italy, 2016. [Google Scholar]
- Kydyrmanov, A.I.; Karamendin, K.O. Viruses of Marine Mammals and Metagenomic Monitoring of Infectious Diseases. Bull. Natl. Acad. Sci. Repub. Kazakhstan 2019, 4, 147–153. [Google Scholar] [CrossRef]
- Bosch, A.; Pintó, R.M.; Guix, S. Foodborne viruses. Curr. Opin. Food Sci. 2016, 8, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Chantanachookin, C.; Boonyaratpalin, S.; Kasornchandra, J.; Direkbusarakom, S.; Ekpanithanpong, U.; Supamataya, K.; Sriurairatana, S.; Flegel, T.W. Histology and ultrastructure reveal a new granulosis-like virus in Penaeus monodon affected by yellow-head disease. Dis. Aquat. Org. 1993, 17, 145–157. [Google Scholar] [CrossRef]
- De la Rosa-Velez, J.; Cedano-Thomas, Y.; Cid-Becerra, J.; Mendez-Payan, J.C.; Vega-Perez, C.; Zambrano-Garcia, J.; Bonami, J.R. Presumptive detection of yellow head virus by reverse transcriptasepolymerase chain reaction and dot-blot hybridization in Litopenaeus vannamei and L. stylirostris cultured on the Northwest coast of Mexico. J. Fish Dis. 2006, 29, 717–726. [Google Scholar] [CrossRef]
- Walker, P.J.; Cowley, J.A.; Spann, K.M.; Hodgson, R.A.J.; Hall, M.R.; Withychumnarnkul, B. Yellow head complex viruses: Transmission cycles and topographical distribution in the Asia-Pacific region. In Proceedings of the the New Wave: Proceedings of the Special Session of Sustainable Shrimp Culture, World Aquaculture 2001, Baton Rouge, LA, USA, January 2001; Browdy, C.L., Jory, D.J., Eds.; World Aquaculture Society: Baton Rouge, LA, USA, 2001; pp. 292–302. [Google Scholar]
- Walker, P.J.; Winton, J.R. Emerging viral diseases of fish and shrimp. Vet. Res. 2010, 41, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Booth, M. Climate Change and the Neglected Tropical Diseases, 1st ed.; Rollinson, D., Stothard, R., Eds.; Elsevier Ltd.: London, UK, 2018; Volume 100, ISBN 9780128151693. [Google Scholar]
- Al-taee, S.K.; Al-jumaa, Z.M.; Ali, F.F. Coronavirus and COVID-19 disease in aquatic animals´aspects. Vet. Pract. 2020, 21, 107–112. [Google Scholar]
- Chen, Y.; Liu, Q.; Guo, D. Emerging coronaviruses: Genome structure, replication, and pathogenesis. J. Med. Virol. 2020, 92, 418–423. [Google Scholar] [CrossRef] [PubMed]
- Gheblawi, M.; Wang, K.; Viveiros, A.; Nguyen, Q.; Zhong, J.C.; Turner, A.J.; Raizada, M.K.; Grant, M.B.; Oudit, G.Y. Angiotensin-Converting Enzyme 2: SARS-CoV-2 Receptor and Regulator of the Renin-Angiotensin System: Celebrating the 20th Anniversary of the Discovery of ACE2. Circ. Res. 2020, 126, 1456–1474. [Google Scholar] [CrossRef] [PubMed]
- Fichtner, D.; Bergmann, S.M.; Dauber, M.; Enzmann, P.J.; Granzow, H.; Schmidt-Posthaus, H.; Et, A. Dituation of Fish Epidemics in Germany and Selected Case Reports from the National Reference Laboratory, in XII. Gemeninschaftstagung der Deutschen, der Österreichischen und der Schweizer Sektion der European Association of Fish Pathologists (EAFP); European Association of Fish Pathologists: Jena, Germany, 2009. [Google Scholar]
- Damas, J.; Hughes, G.M.; Keough, K.C.; Painter, C.A.; Persky, N.S.; Corbo, M.; Hiller, M.; Koepfli, K.P.; Pfenning, A.R.; Zhao, H.; et al. Broad host range of SARS-CoV-2 predicted by comparative and structural analysis of ACE2 in vertebrates. Proc. Natl. Acad. Sci. USA 2020, 117, 22311–22322. [Google Scholar] [CrossRef]
- Mordecai, G.J.; Hewson, I. Coronaviruses in the Sea. Front. Microbiol. 2020, 11, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Boxman, I.L.A.; Tilburg, J.J.H.C.; te Loeke, N.A.J.M.; Vennema, H.; Jonker, K.; de Boer, E.; Koopmans, M. Detection of noroviruses in shellfish in the Netherlands. Int. J. Food Microbiol. 2006, 108, 391–396. [Google Scholar] [CrossRef]
- Boxman, I.L.A. Human enteric viruses occurrence in shellfish from european markets. Food Environ. Virol. 2010, 2, 156–166. [Google Scholar] [CrossRef]
- FAO/WHO. Viruses in Food: Scientific Advice to Support Risk Management Activities; FAO: Rome, Italy, 2008; Volume 13. [Google Scholar]
- EFSA. Scientific Opinion on an update on the present knowledge on the occurrence and control of foodborne viruses. EFSA J. 2011, 9, 1–96. [Google Scholar] [CrossRef]
- Tumpey, T.M.; Suarez, D.L.; Perkins, L.E.L.; Senne, D.A.; Lee, J.; Lee, Y.J.; Mo, I.P.; Sung, H.W.; Swayne, D.E. Evaluation of a High-Pathogenicity H5N1 Avian Influenza A Virus Isolated from Duck Meat. Avian Dis. 2003, 47, 951–955. [Google Scholar] [CrossRef]
- WHO. Novel Coronavirus (2019-nCoV) Advice for the Public; WHO: Geneva, Switzerland, 2020. [Google Scholar]
- Zaporozhets, T.S.; Besednova, N.N. Biologically active compounds from marine organisms in the strategies for combating coronaviruses. AIMS Microbiol. 2020, 6, 470–494. [Google Scholar] [CrossRef]
- Abad Martinez, M.J.; Bedoya Del Olmo, L.M.; Bermejo Benito, P. Natural marine antiviral products. Stud. Nat. Prod. Chem. 2008, 35, 101–134. [Google Scholar] [CrossRef]
- Koehn, F.E.; Gunasekera, S.P.; Niel, D.N.; Cross, S.S. Halitunal, an unusual disterpene aldehyde from the marine alga Halmeda tuna. Tetrahedron Lett. 1991, 32, 169–172. [Google Scholar] [CrossRef]
- Donia, M.; Hamann, M.T. Marine natural products and their potential applications as anti-infective agents. Lancet Infect. Dis. 2003, 3, 338–348. [Google Scholar] [CrossRef]
- Nakao, Y.; Masuda, A.; Matsunaga, S.; Fusetani, N. Pseudotheonamides, serine protease inhibitors from the marine sponge Theonella swinhoei. J. Am. Chem. Soc. 1999, 121, 2425–2431. [Google Scholar] [CrossRef]
- Gómez-Archila, L.G.; Rugeles, M.T.; Zapata, W. Actividad antiviral de compuestos aislados de esponjas marinas. Rev. Biol. Mar. Oceanogr. 2014, 49, 401–412. [Google Scholar] [CrossRef]
- De Lira, S.P.; Seleghim, M.H.R.; Williams, D.E.; Marion, F.; Hamill, P.; Jean, F.; Andersen, R.J.; Hajdu, E.; Berlinck, R.G.S. A SARS-coronovirus 3CL protease inhibitor isolated from the marine sponge Axinella cf. corrugata: Structure elucidation and synthesis. J. Braz. Chem. Soc. 2007, 18, 440–443. [Google Scholar] [CrossRef]
- Gentile, D.; Patamia, V.; Scala, A.; Sciortino, M.T.; Piperno, A.; Rescifina, A. Putative inhibitors of SARS-CoV-2 main protease from a library of marine natural products: A virtual screening and molecular modeling study. Mar. Drugs 2020, 18, 225. [Google Scholar] [CrossRef] [PubMed]
- Abdelrheem, D.A.; Ahmed, S.A.; Abd El-Mageed, H.R.; Mohamed, H.S.; Rahman, A.A.; Elsayed, K.N.M.; Ahmed, S.A. The inhibitory effect of some natural bioactive compounds against SARS-CoV-2 main protease: Insights from molecular docking analysis and molecular dynamic simulation. J. Environ. Sci. Health—Part A Toxic/Hazardous Subst. Environ. Eng. 2020, 55, 1373–1386. [Google Scholar] [CrossRef]
- Mishchenko, N.P.; Krylova, N.V.; Iunikhina, O.V.; Vasileva, E.A.; Likhatskaya, G.N.; Pislyagin, E.A.; Tarbeeva, D.V.; Dmitrenok, P.S.; Fedoreyev, S.A. Antiviral potential of sea urchin aminated spinochromes against herpes simplex virus type 1. Mar. Drugs 2020, 18, 550. [Google Scholar] [CrossRef] [PubMed]
- Fedoreyev, S.A.; Krylova, N.V.; Mishchenko, N.P.; Vasileva, E.A.; Pislyagin, E.A.; Iunikhina, O.V.; Lavrov, V.F.; Svitich, O.A.; Ebralidze, L.K.; Leonova, G.N. Antiviral and antioxidant properties of echinochrome A. Mar. Drugs 2018, 16, 509. [Google Scholar] [CrossRef]
- Soleimani, S.; Yousefzadi, M.; moein, S.; Rezadoost, H.; Bioki, N.A. Identification and antioxidant of polyhydroxylated naphthoquinone pigments from sea urchin pigments of Echinometra mathaei. Med. Chem. Res. 2016, 25, 1476–1483. [Google Scholar] [CrossRef]
- Khan, M.T.; Ali, A.; Wang, Q.; Irfan, M.; Khan, A.; Zeb, M.T.; Zhang, Y.J.; Chinnasamy, S.; Wei, D.Q. Marine natural compounds as potents inhibitors against the main protease of SARS-CoV-2—A molecular dynamic study. J. Biomol. Struct. Dyn. 2021, 39, 3627–3637. [Google Scholar] [CrossRef] [PubMed]
- Vijayaraj, R.; Altaff, K.; Rosita, A.S.; Ramadevi, S.; Revathy, J. Bioactive compounds from marine resources against novel corona virus (2019-nCoV): In silico study for corona viral drug. Nat. Prod. Res. 2020, 1–5. [Google Scholar] [CrossRef]
- Rubilar, T.; Cardozo, D. Los erizos de mar y su potencial terapéutico para tratar Covid-19. Atek 2020, 1479, 375–384. [Google Scholar]
- Barbieri Elena, S.; Tamara, R.; Ayelén, G.; Marisa, A.; Seiler, E.N.; Mercedes, V.P.; Agustín, G.; Chaar, F.; Pia, F.J.; Lucas, S. Sea urchin pigments as potential therapeutic agents against the spike protein of SARS-CoV-2 based on in silico analysis. ChemRxiv 2020, 1–10. [Google Scholar] [CrossRef]
- Rubilar Panasiuk, C.T.; Barbieri, E.S.; Gázquez, A.; Avaro, M.; Vera Piombo, M.; Gittardi Calderón, A.A.; Seiler, E.N.; Fernandez, J.P.; Sepúlveda, L.R.; Chaar, F. In silico analysis of sea urchin pigments as potential therapeutic agents against SARS-CoV-2: Main protease (Mpro) as a target. ChemRxiv 2020, 1–14. [Google Scholar] [CrossRef]
- Ponce Rey, L.D.R.; del Barrio Alonso, G.D.C.; Spengler Salabarría, I.; Resik Aguirre, S.; Roque Quintero, A. Evaluación de la actividad antiviral del alga parda Sargassum fluitans frente a Echovirus 9. Rev. Cubana Med. Trop. 2018, 70, 1–12. [Google Scholar]
- Torres, B. El sargazo. La Cienc. y el Hombre 2019, XXXII, 1. [Google Scholar]
| Group | Genus | CoV Type | Host | Health Effects | Year | Reference | |
|---|---|---|---|---|---|---|---|
| Order Suborder Family | Nidoviales Cornidovirinea Coronaviridae | Coronavirus (?) * | Carp CoV | Common carp (Japan) | Erythema, necrosis (abdomen and liver) Dermal ulcerations, necrotic lesion. Found in spleen and hematopoietic | 1988 | [51,57] |
| Coronavirus (?) * | Carp Viremia-Associated Ana-Aki-Byo | Common carp (Japan) | 1997–1998 | [57,58] | |||
| Subfamily | Orthocoronavirinae | Alphacoronavirus | HSCoV | Harbor seals (Aquatic Park, FL, USA) | Acute enteritis, pulmonary edema | 1987 | [59] |
| HSCoV | Pacific Harbor seals (CA, USA) | Pulmonary congestion, consolidation, and hemorrhage, pneumonia | 2000 | [60] | |||
| Betacoronavirus | Not reported | ||||||
| Gammacoronavirus | BWCoVSW1 | Beluga whale (Aquatic Park, CA, USA) | Hepatic necrosis and pulmonary disease | 2008 | [7,61] | ||
| BdCoV HKU22 | Bottlenose Dolphin (Aquatic Park, Hong Kong) | Found in feces | ? | [10] | |||
| Deltacoronavirus | Not reported | ||||||
| Torivirinae | Torovirus (?) * | CIVH 33/86 | Grass carp (Hungary) | Not known | 1986 | [51] | |
| Bafinivirus | WBV DF24/00 | White bream fish (Saxony-Anhalt, Germany) | Not known | 2000 | [13,51] | ||
| FHMNV | Fathead minnow fish (AR, USA) | Eyes and skin hemorrhage, tissue lesions (spleen, liver, and kidney) | 1997 | [54,55] | |||
| Chinook Salmon Bfinivirus | Chinook salmon (ON, Canada) | Not known | 2014 | [51,56] | |||
| Letovirinae | Alphaletovirus | Microhyla letovirus MLev | Ornamental pygmy frog | Not reported | 2018 | [12] |
| Order | Genus | Species | Type of Bird | Type of CoV | References |
|---|---|---|---|---|---|
| Anseriformes | Anas | domestica | Duck | Gamma (SARS-CoV) | [70] |
| platyhynchos | Spot-billed duck | Gamma (SARS-CoV) | [70] | ||
| americana | American wigeon | Delta (JQ065048.1) | [71] | ||
| crecca | Common teal | Gamma (J0109, J0212, J0126, J0559, J0579, J1393); Delta (J1420) | [69,72,73] | ||
| clypeata | Northern shoveler | Gamma (K547, K554, K561, K589, J0554, J0807, J1300,J0901, J1491); Delta (J0590) | [69,73] | ||
| penelope | Eurasian wigeon | Gamma (K596, J0588, J1561) | [69] | ||
| acuta | Northern pintail | Gamma (J1375, J1393, J1404,, J1407, J1435, J1616, J1451,, PBA-10, PBA-15, PBA-16,, PBA-25, PBA-37, PBA-124) | [69,72,73] | ||
| erythrorhyncha | Red-billed duck | Gamma, (KM093874, KM093875,, KM093876, KM093877) | [73] | ||
| hottentota | Hottentot teal | Gamma (KM093880) | [71] | ||
| Clangula | hyemalis | Long-tailed duck | Gamma (Fin14395) | [72] | |
| Cygnus | cygnus | Whooper swan | Gamma (Fin4983) | [72] | |
| Dendrocygna | javanica | Lesser whistling | Gamma (KH08-0852) | [69,71] | |
| viduata | White-faced whistling duck | Gamma (KM093872, KM093873, KM093878) | [73] | ||
| Aythya | fuligula | Tufted duck | Gamma (J1482) | [69] | |
| Anser | caerulescens | Snow goose | Gamma (WIR-159) | [69,71] | |
| anser | Greylag goose | Gamma (SARS) | [70] | ||
| cygnoides | Swan goose | Gamma (DPV_16), Delta (DPV_5, DPV_10) | [71] | ||
| Branta | bernicola | Brent goose | Gamma (KR-69, KR-70, KR88) | [69,71,73] |
| Order | Genus | Species | Type of Bird | Type of CoV | References |
|---|---|---|---|---|---|
| Pelecaniformes | Ardeola | bacchus | Pond heron | Delta (KH08-1475, KH08-1474) | [69] |
| Ardea | cinerea | Gray heron | Delta (K581, K513) | [69] | |
| Bubulcus | ibis | Heron | Gamma (KM093897) | [71,73] | |
| Platalea | minor | Black-faced spoonbill | Delta (J0569) | [69] | |
| Phalacrocorax | carbo | Great cormorant | Delta (J0982, J1517) | [69] | |
| Gruiformes | Rallus | madagascariensis | Madagascar rail | Gamma (KM093896) | [73] |
| Porphyrula | alleni | Allen’s gallinule | Gamma (KM093890, KM093891, KM093892 KM093893, KM093894) | [71,73] | |
| Gallinula | chloropus | Common moorhen | Gamma (KM093881, KM093885, KM093887). Delta (JQ065049.1) | [71,73] | |
| Charadriiformes | Charadrius | pecuarius | Kittlitz´s plover | Gamma (KM093879, KM093883, KM093884) | [71,73] |
| Gallinago | macrodactyla | Madagascan snipe | Gamma (KM093888, KM093889, KM093895) | [73] | |
| Calidris | mauri | Western sandpiper | Gamma (KR-28) | [71] | |
| ptilocnemis | Rock sandpiper | Gamma (CIR-66187, CIR-665821, CIR-665828) | [69,71,73] | ||
| alba | Sanderling | Gamma (PNLP100) | [71] | ||
| fuscicollis | White-rumped sandpiper | Gamma (PNLP159) | [71] | ||
| Larus | sp | Gull | Delta (JX548304) | [71] | |
| argentatus | Herring gull | Gamma (Fin9211, Fin10877, Fin10879, Fin12822, Fin13125) | [72] | ||
| hyperboreus | Glaucous gull | Gamma (PBA-173) | [69] | ||
| fuscus | Lesser Black-back gull | Gamma (Fin10059) | [72] | ||
| glauscescens | Glaucous-winged gull | Gamma (CIR-66002, GU396682) | [69] | ||
| Chroicocepphalus | ridibundus | Black-headed gull | Gamma (CIR-66187, GU396679, GU396680, GU396683, KX588674, Fin10083) | [69,72] | |
| Rostratula | benghalensis | Greater Painted-snipe | Gamma (KM093883) | [71] | |
| Rynchops | niger | Black skimmer | Delta (PNLP115) | [71] | |
| Sphenisciformes | Spheniscus | magellanicus | Magellanic penguin | Avian CoV M41, C46, A99, JMK | [74] |
| humboldti | Humboldt penguin | Avian CoV M41, C46, A99 | [75] | ||
| Eudyptes | chrysocome | Southern rockhopper | Avian CoV C46, A99, JMK | [76] |
| Substances (Compound) | Species | Source | Reference |
|---|---|---|---|
| Peptides (Pseudoteonamides C y D) | Theonella swinhoei | Marine sponge | [101] |
| Nucleosides (micalamide A) | Mycale sp, | Marine sponge | [100] |
| Nucleosides (Spongotimidine and Ara-A9 | Cryptotethya crypta | Marine sponge | [102] |
| Esculetin ethyl ester | Axinella corrugata | Marine sponge | [103] |
| Alkaloid (Dragmacidine F) | Halicortex sp. | Marine sponge | [102] |
| Alkaloid (4-methylaptamine) | Aaptos aaptos | Marine sponge | [102] |
| Terpenic aldehydes (halitunal) | Halimeda sp. | Tuna seaweed | [99] |
| Florotannins (Dieckol,, 6,6’-Bieckol, 8,8’-Bieckol) | Ecklonia cava | Brown seaweed | [104] |
| Florotannins (Heptafuhalol A, Phlorethopentafuhalol A, Pseudopentafuhalol B, Pseudopentafuhalol C, Hydroxypentafuhalol A | Sargassum sp. Sargassum spinuligerum | Brown seaweed | [104] |
| Fatty acids (oleic acid) | Ceramium virgatum | Red algae | |
| Ulva intestinalis | Green algae | [105] | |
| Fucus sp. | Brown seaweed | ||
| Phytosterols (Saryngosterols) | Acanthophora spicifera | Red algae | |
| Cladophora fascicularis | Green algae | [105] | |
| Sargassum muticum | Brown seaweed | ||
| Phytosterols (β-sitosterol) | Euchema cottonii | Red algae | |
| Ulva fasciata | Green algae | [105] | |
| Sargassum glaucescens | Brown seaweed | ||
| Glycoglycerolipids | Exophyllum wentii | Red algae | |
| Sargassum horneri, Phormidium sp. | Brown seaweed, Cyanobacteria | [105] | |
| Kjellmanianone | Sargassum naozhouense | Brown seaweed | [105] |
| Loliolide | Sargassum naozhouense | Brown seaweed | [105] |
| Caulerpin | Caulerpa racemosa | Green algae | |
| Chondria armata | Red algae | [105] | |
| Sargassum platycarpum | Brown seaweed | ||
| Polyhydroxy naphthoquinones natural pigments (Echinochromo A, Echinaminas A and B, beta-carotene, Astaxanthin and Fucoxanthin) | Scaphechinus mirabilis Strongylocentrotus nudus S. pallidus S. polyacanthus Echinarachnius parma Evechinus chloroticus Echinometra mathaei Arbacia dufresnii | Sea urchin (gonads, spines and carapace) | [106,107,108] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Núñez-Nogueira, G.; Valentino-Álvarez, J.A.; Granados-Berber, A.A.; Ramírez-Ayala, E.; Zepeda-González, F.A.; Tintos-Gómez, A. Aquatic Biota Is Not Exempt from Coronavirus Infections: An Overview. Water 2021, 13, 2215. https://doi.org/10.3390/w13162215
Núñez-Nogueira G, Valentino-Álvarez JA, Granados-Berber AA, Ramírez-Ayala E, Zepeda-González FA, Tintos-Gómez A. Aquatic Biota Is Not Exempt from Coronavirus Infections: An Overview. Water. 2021; 13(16):2215. https://doi.org/10.3390/w13162215
Chicago/Turabian StyleNúñez-Nogueira, Gabriel, Jesús Alberto Valentino-Álvarez, Andrés Arturo Granados-Berber, Eduardo Ramírez-Ayala, Francisco Alberto Zepeda-González, and Adrián Tintos-Gómez. 2021. "Aquatic Biota Is Not Exempt from Coronavirus Infections: An Overview" Water 13, no. 16: 2215. https://doi.org/10.3390/w13162215
APA StyleNúñez-Nogueira, G., Valentino-Álvarez, J. A., Granados-Berber, A. A., Ramírez-Ayala, E., Zepeda-González, F. A., & Tintos-Gómez, A. (2021). Aquatic Biota Is Not Exempt from Coronavirus Infections: An Overview. Water, 13(16), 2215. https://doi.org/10.3390/w13162215






