Removal of Ciprofloxacin with Aluminum-Pillared Kaolin Sodium Alginate Beads (CA-Al-KABs): Kinetics, Isotherms, and BBD Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Fabrication of CA-Al-KABs
2.1.1. Preparation of Na-Kaolin
2.1.2. Preparation of Al-Kaolin
2.1.3. Preparation of CA-Al-KABs
2.2. Characterizations
2.3. Adsorption Experiment
3. Results and Discussion
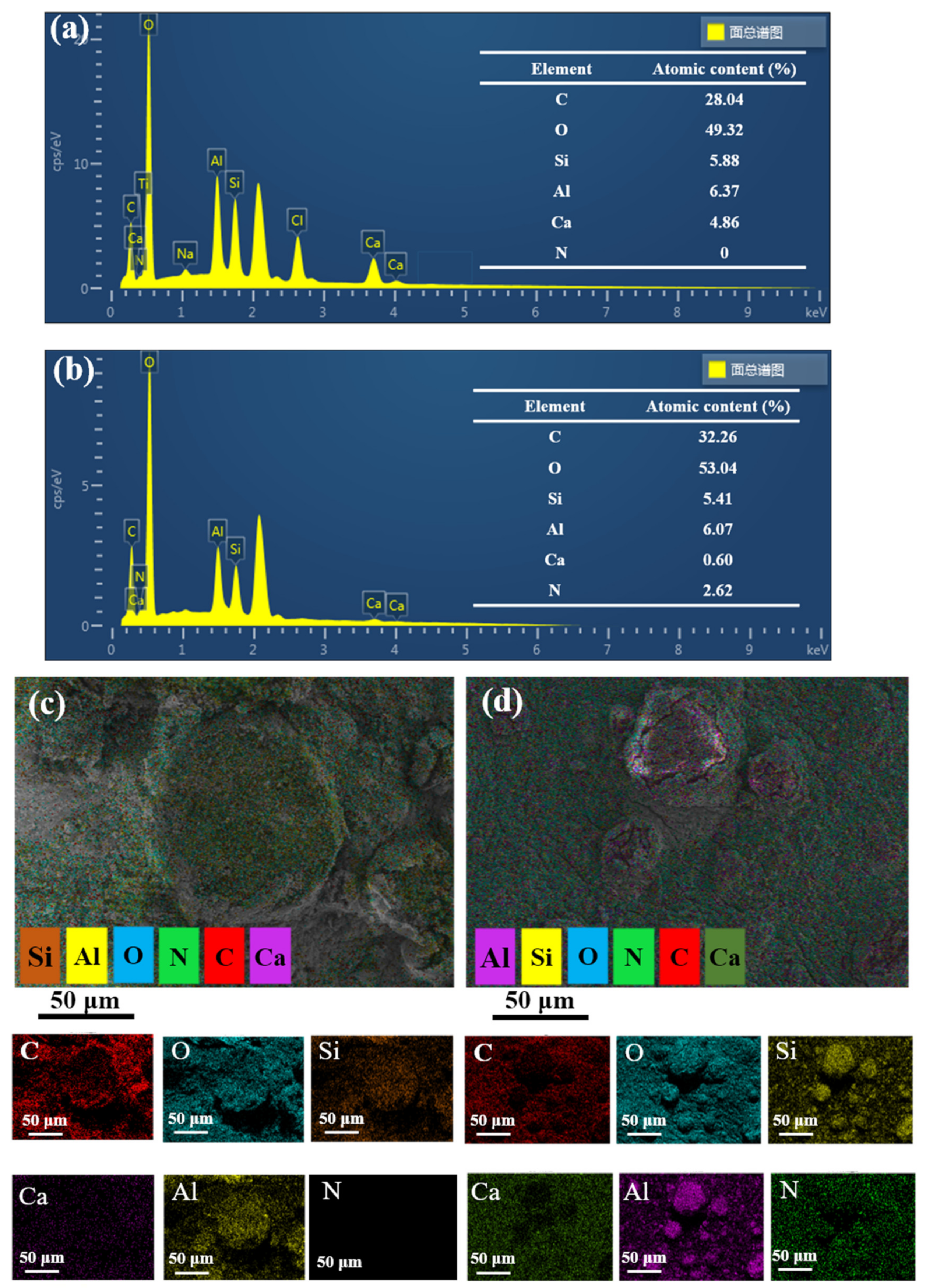
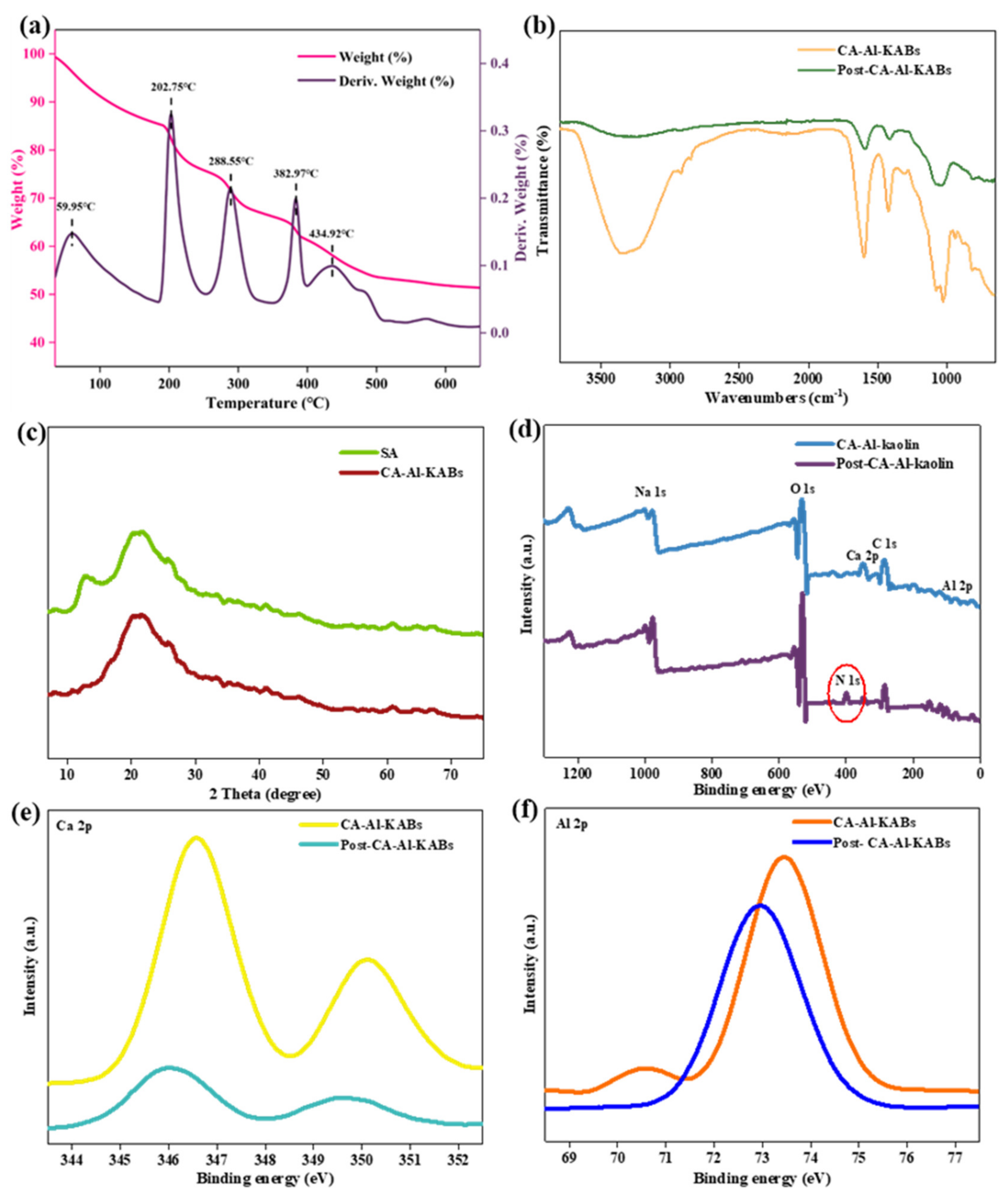
3.1. Characteristics
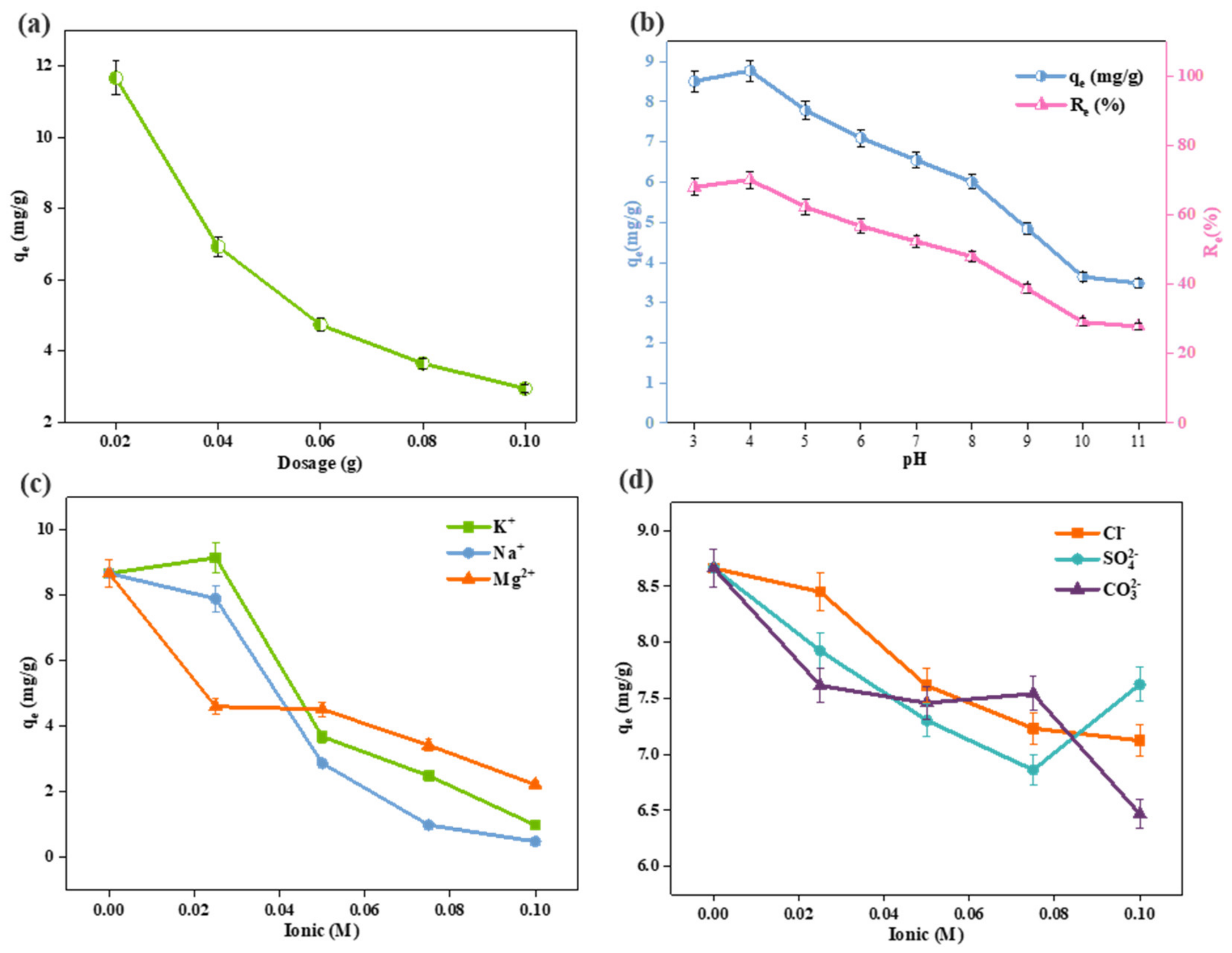
3.2. Effects of Dosage on Adsorption Capacity
3.3. Effects of pH on Adsorption Capacity
3.4. Effects of Iron Strength’s Effect on Adsorption Capacity
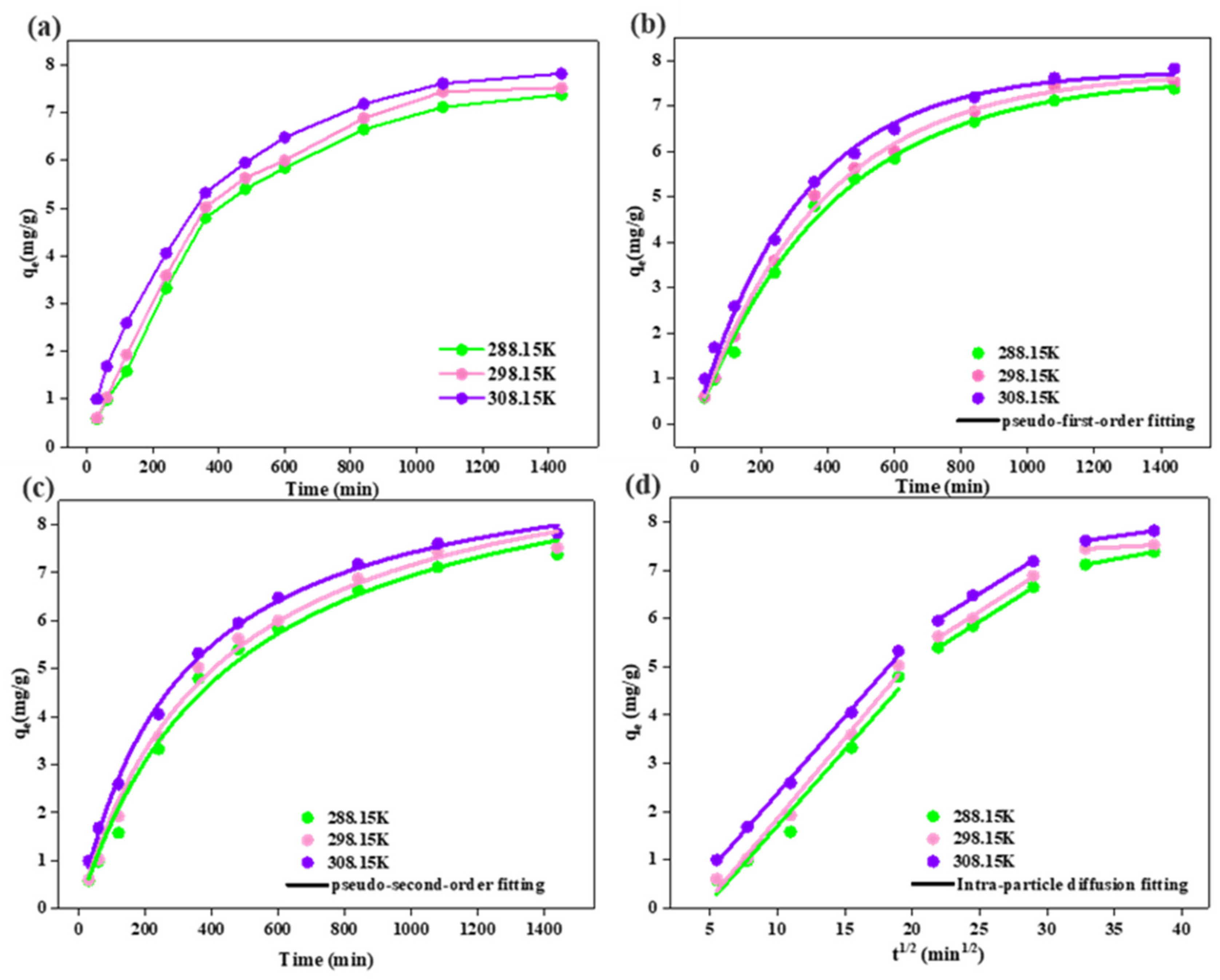
3.5. Adsorption Kinetics
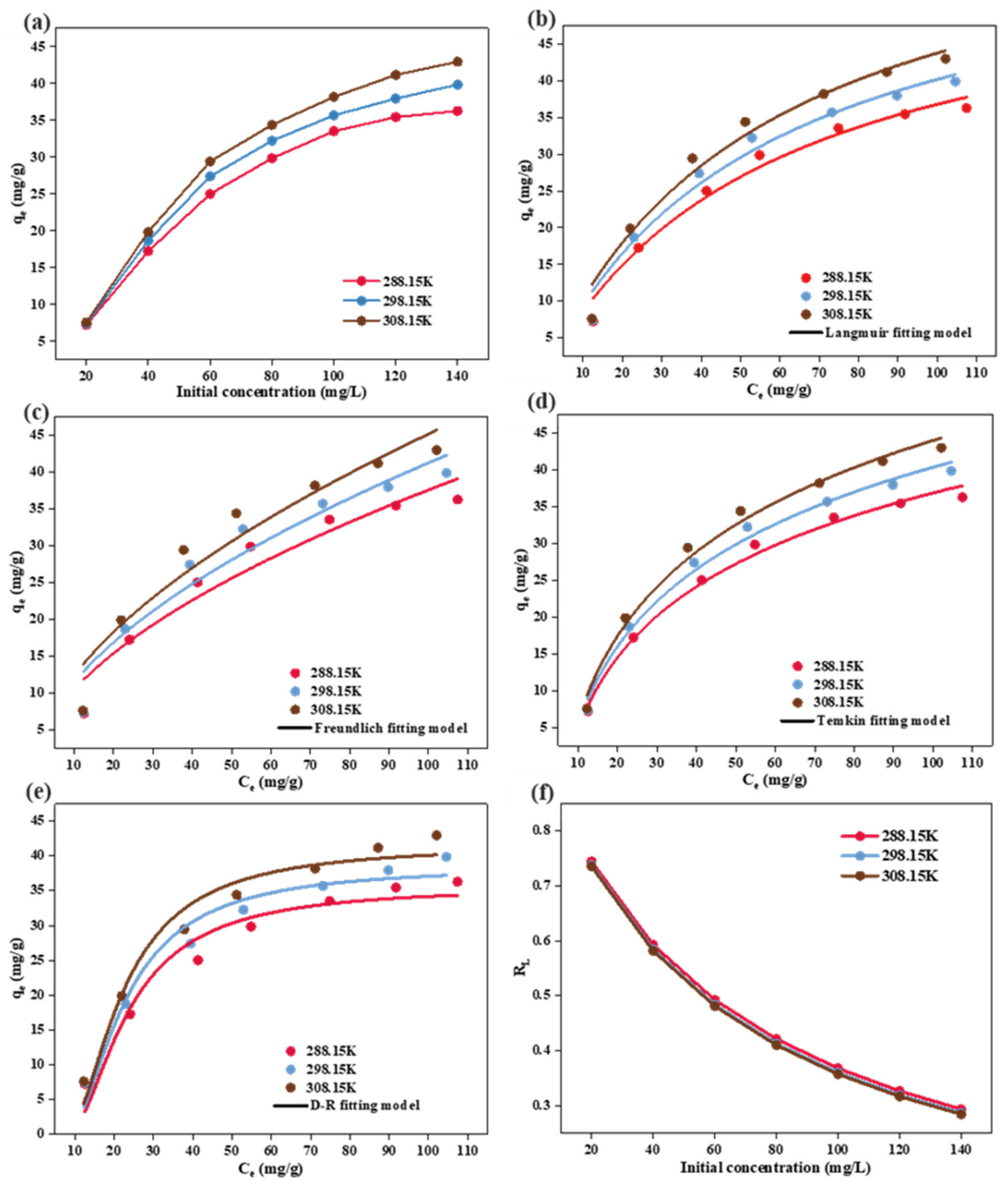
3.6. Adsorption Isotherms
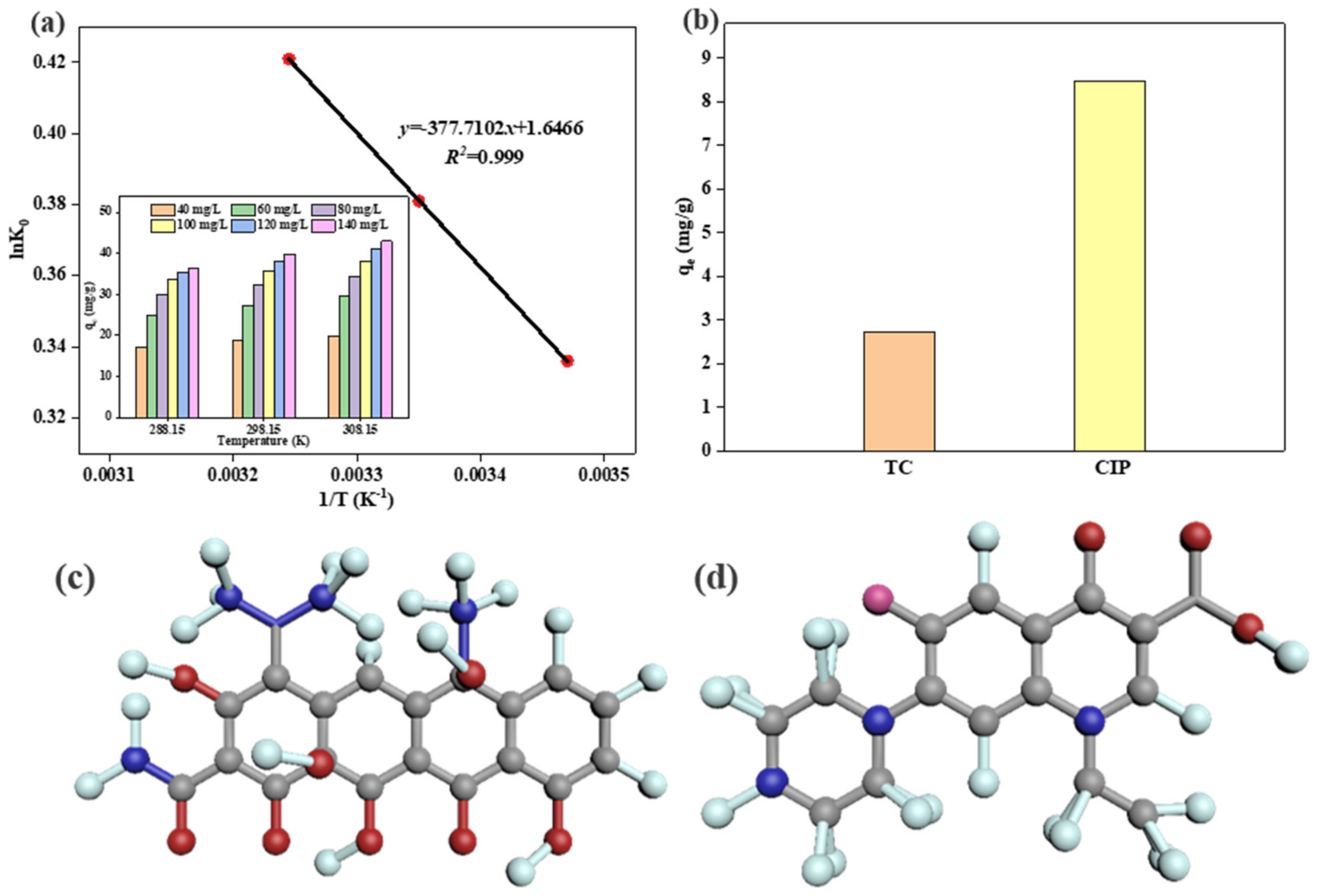
3.7. Adsorption Thermodynamics
3.8. Adsorption Capacity of Different Antibiotics
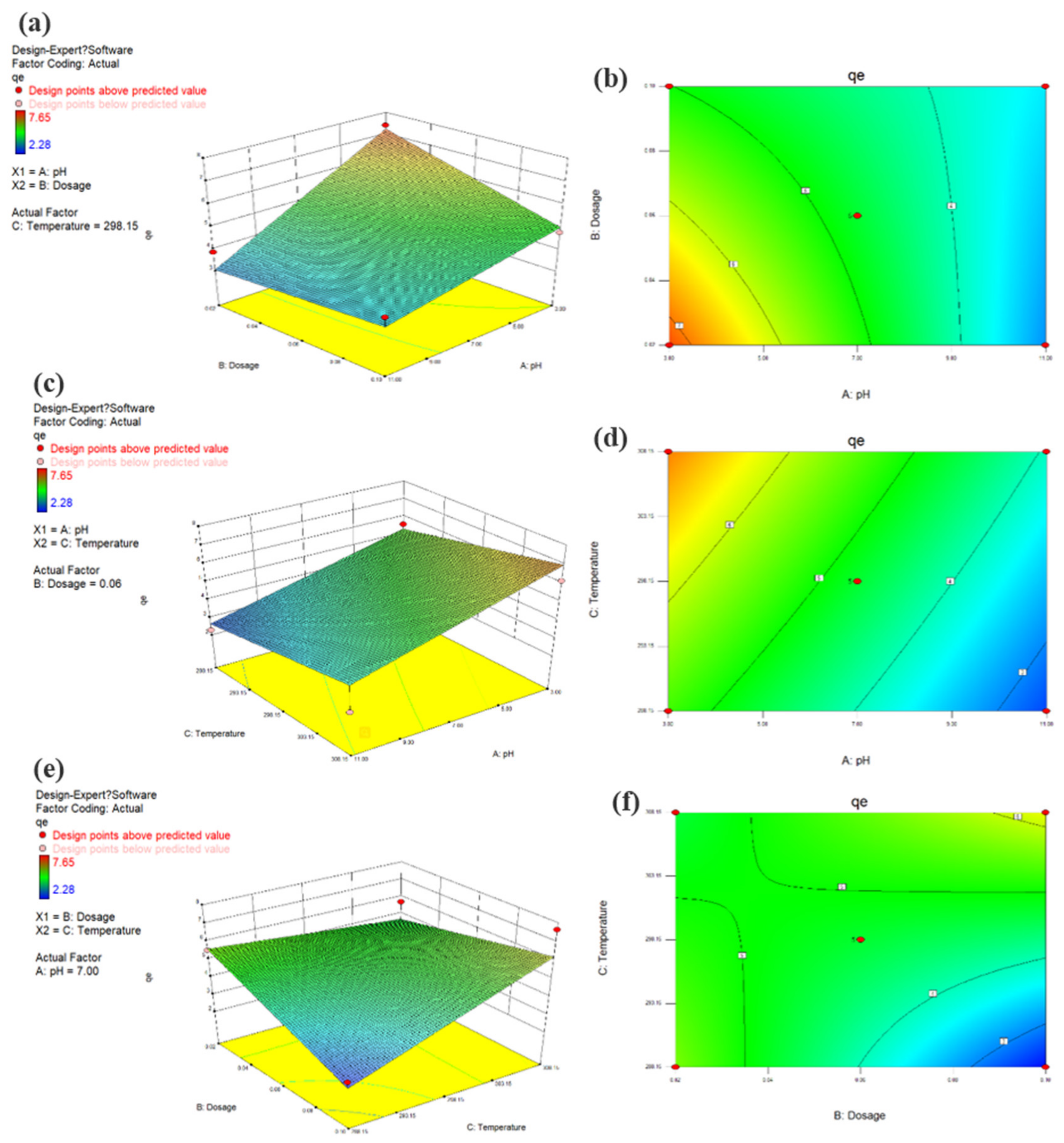
3.9. Adsorption Modeling by RSM
3.9.1. Modeling Set Up
3.9.2. Regression Model Equation Establishment
3.9.3. ANOVA
3.9.4. Effect of Model Arguments and Their Interaction
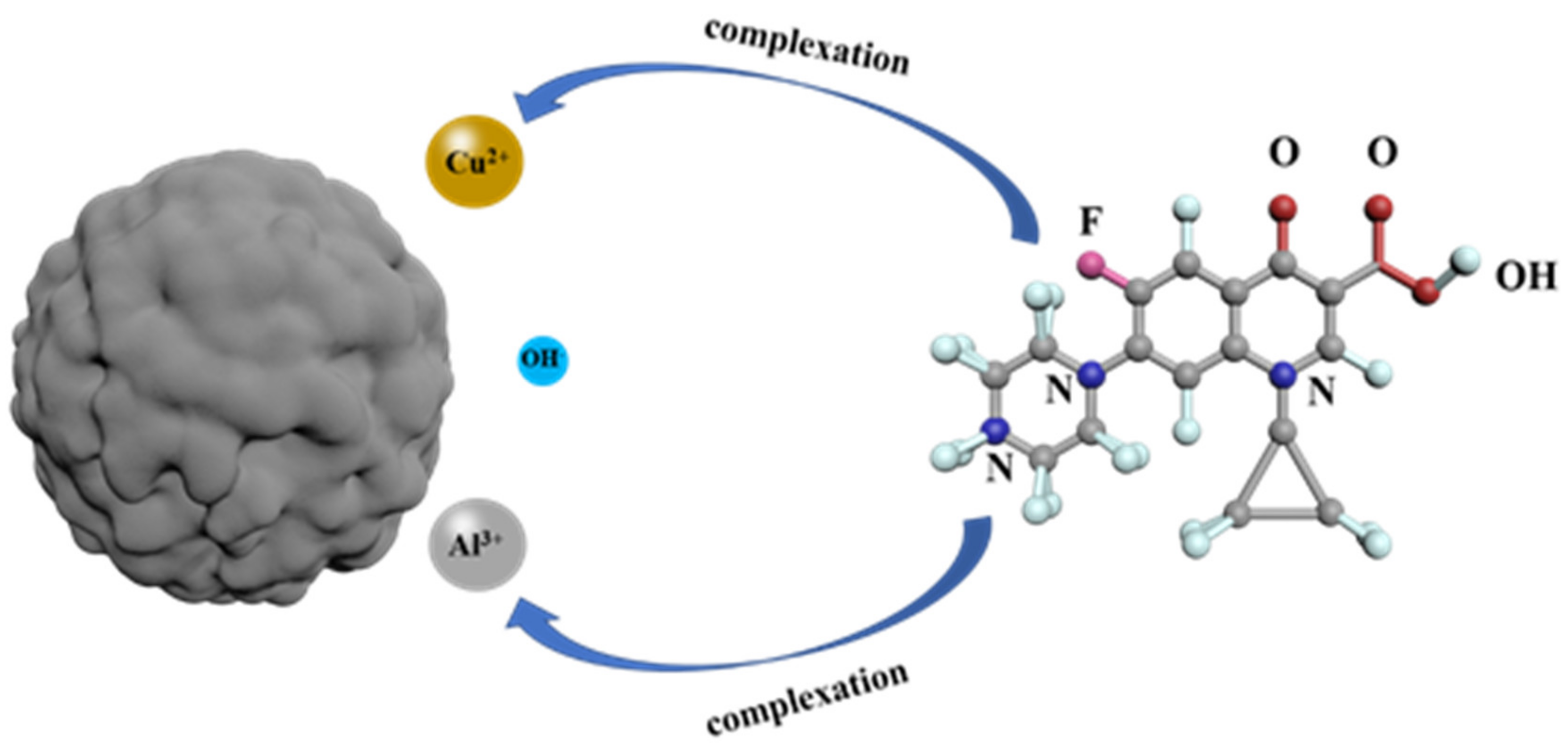
3.10. Adsorbing Mechanism
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yue, Y.; Shen, C.; Ge, Y. Biochar accelerates the removal of tetracyclines and their intermediates by altering soil properties. J. Hazard. Mater. 2019, 380, 120821. [Google Scholar] [CrossRef]
- Antón-Herrero, R.; García-Delgado, C.; Alonso-Izquierdo, M.; García-Rodríguez, G.; Cuevas, J.; Eymar, E. Comparative adsorption of tetracyclines on biochars and stevensite: Looking for the most effective adsorbent. Appl. Clay Sci. 2018, 160, 162–172. [Google Scholar] [CrossRef]
- He, K.; Niu, C.-G.; Chen, A.; Huang, Z.; Peng, M.; Huang, T.; Chen, G. Graphene hybridized polydopamine-kaolin composite as effective adsorbent for methylene blue removal. Compos. Part B Eng. 2019, 161, 141–149. [Google Scholar] [CrossRef]
- Mustapha, S.; Ndamitso, M.; Abdulkareem, A.; Tijani, J.; Mohammed, A.; Shuaib, D. Potential of using kaolin as a natural adsorbent for the removal of pollutants from tannery wastewater. Heliyon 2019, 5, 02923. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Lingfa, P. Sodium bentonite and kaolin clays: Comparative study on their FT-IR, XRF, and XRD. Mater. Today Proc. 2020, 22, 737–742. [Google Scholar] [CrossRef]
- Abdulsalam, I.O.; Adegoke, H.I.; Adekola, F.A. Batch Sorption of Ciprofloxacin on Kaolinitic Clay and nHematite Composite: Equilibrium and Thermodynamics Studies. Moroc. J. Chem. 2016, 4, 384. [Google Scholar]
- Ma, W.; Dai, J.; Dai, X.; Yan, Y. Preparation and Characterization of Chitosan/Kaolin/Fe3O4 Magnetic Microspheres and Their Application for the Removal of Ciprofloxacin. Adsorpt. Sci. Technol. 2014, 32, 775–790. [Google Scholar] [CrossRef]
- Ibrahim, H.; Jamil, T.; Hegazy, E. Application of zeolite prepared from Egyptian kaolin for the removal of heavy metals: II. Isotherm models. J. Hazard. Mater. 2010, 182, 842–847. [Google Scholar] [CrossRef]
- Zhou, K.; Wu, B.; Dai, X.; Chai, X. Development of polymeric iron/zirconium-pillared clinoptilolite for simultaneous removal of multiple inorganic contaminants from wastewater. Chem. Eng. J. 2018, 347, 819–827. [Google Scholar] [CrossRef]
- Mnasri-Ghnimi, S.; Frini-Srasra, N. Removal of heavy metals from aqueous solutions by adsorption using single and mixed pillared clays. Appl. Clay Sci. 2019, 179, 105151. [Google Scholar] [CrossRef]
- Belhouchat, N.; Zaghouane-Boudiaf, H.; Viseras, C. Removal of anionic and cationic dyes from aqueous solution with activated organo-bentonite/sodium alginate encapsulated beads. Appl. Clay Sci. 2017, 135, 9–15. [Google Scholar] [CrossRef]
- Chen, P.-P.; Zhang, H.-P.; Ding, J.; Lin, X.-Y.; Lu, X.; Liu, C.; Tang, Y. Carboxylmethyl konjac glucomannan conjugated polydopamine composites for Pb(II) removal. Carbohydr. Polym. 2017, 162, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Filho, E.C.D.S.; Lima, L.C.B.; Silva, F.C.; Sousa, K.S.; Fonseca, M.G.; Santana, S.A. Immobilization of ethylene sulfide in aminated cellulose for removal of the divalent cations. Carbohydr. Polym. 2013, 92, 1203–1210. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Kang, H.; Zhang, W.; Zhang, S.; Li, J. Improvement of interfacial interactions using natural polyphenol-inspired tannic acid-coated nanoclay enhancement of soy protein isolate biofilms. Appl. Surf. Sci. 2017, 401, 271–282. [Google Scholar] [CrossRef]
- Dambies, L.; Guimon, C.; Yiacoumi, S.; Guibal, E. Characterization of metal ion interactions with chitosan by X-ray photoelectron spectroscopy. Colloids Surfaces A Physicochem. Eng. Asp. 2001, 177, 203–214. [Google Scholar] [CrossRef]
- Wu, D.; Zhao, J.; Zhang, L.; Wu, Q.; Yang, Y. Lanthanum adsorption using iron oxide loaded calcium alginate beads. Hydrometallurgy 2010, 101, 76–83. [Google Scholar] [CrossRef]
- Lim, S.-F.; Zheng, Y.-M.; Zou, S.-W.; Chen, J.P. Characterization of Copper Adsorption onto an Alginate Encapsulated Magnetic Sorbent by a Combined FT-IR, XPS, and Mathematical Modeling Study. Environ. Sci. Technol. 2008, 42, 2551–2556. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, S.; Zhong, L.; Wu, G. Preparation of high-stable silver nanoparticle dispersion by using sodium alginate as a stabilizer under gamma radiation. Radiat. Phys. Chem. 2009, 78, 251–255. [Google Scholar] [CrossRef]
- Deng, Z.; Wang, F.; Zhou, B.; Li, J.; Li, B.; Liang, H. Immobilization of pectinases into calcium alginate microspheres for fruit juice application. Food Hydrocoll. 2019, 89, 691–699. [Google Scholar] [CrossRef]
- Turel, I.; Bukovec, N.; Farkas, E. Complex formation between some metals and a quinolone family member (ciprofloxacin). Polyhedron 1996, 15, 269–275. [Google Scholar] [CrossRef]
- Chen, H.; Ma, L.Q.; Gao, F.; Gu, C. Effects of Cu and Ca cations and Fe/Al coating on ciprofloxacin sorption onto sand media. J. Hazard. Mater. 2013, 252, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Lin, X.; He, Y.; Luo, X. Phenolic hydroxyl derived copper alginate microspheres as superior adsorbent for effective adsorption of tetracycline. Int. J. Boil. Macromol. 2019, 136, 445–459. [Google Scholar] [CrossRef] [PubMed]
- Gu, C.; Karthikeyan, K.G. Sorption of the Antimicrobial Ciprofloxacin To Aluminum and Iron Hydrous Oxides. Environ. Sci. Technol. 2005, 39, 9166–9173. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-J.; Li, Z.; Jiang, W.-T.; Jean, J.-S.; Liu, C.-C. Cation exchange interaction between antibiotic ciprofloxacin and montmorillonite. J. Hazard. Mater. 2010, 183, 309–314. [Google Scholar] [CrossRef] [PubMed]
- El-Shafey, E.-S.I.; Al-Lawati, H.A.; Al-Sumri, A.S. Ciprofloxacin adsorption from aqueous solution onto chemically prepared carbon from date palm leaflets. J. Environ. Sci. 2012, 24, 1579–1586. [Google Scholar] [CrossRef]
- Patel, S.; Bajpai, A.; Bajpai, J.; Saini, R.K.; Acharya, S. Facile preparation of iron loaded calcium alginate nanocarriers and study of controlled release of iron. J. Environ. Chem. Eng. 2017, 5, 5337–5346. [Google Scholar] [CrossRef]
- Lapointe, M.; Barbeau, B. Understanding the roles and characterizing the intrinsic properties of synthetic vs. natural polymers to improve clarification through interparticle Bridging: A review. Sep. Purif. Technol. 2020, 231, 115893. [Google Scholar] [CrossRef]

| Type | Parameter | T(K) | ||
|---|---|---|---|---|
| 288.15 | 298.15 | 308.15 | ||
| Pseudo-first-order kinetics | qe,cal (mg/g) | 7.644 | 7.772 | 7.779 |
| k1 (min−1) | 2.46 × 10−3 | 2.63 × 10−3 | 3.18 × 10−3 | |
| R2 | 0.995 | 0.996 | 0.994 | |
| Pseudo-second-order kinetics | qe,cal (mg/g) | 10.113 | 10.124 | 9.686 |
| k2 (g·(mg·min)−1) | 2.16 × 10−4 | 2.38×10−4 | 3.37 × 10−4 | |
| R2 | 0.989 | 0.991 | 0.997 | |
| Intra-particle diffusion model | k1 (mg·(g·min1/2)−1) | 0.315 | 0.332 | 0.318 |
| R2 | 0.962 | 0.983 | 0.997 | |
| k2 (mg·(g·min1/2)−1) | 0.177 | 0.179 | 0.172 | |
| R2 | 0.999 | 0.988 | 0.989 | |
| k3 (mg·(g·min1/2)−1) | 0.051 | 0.016 | 0.040 | |
| R2 | 1 | 1 | 1 | |
| Type | Parameter | T (K) | ||
|---|---|---|---|---|
| 288.15 | 298.15 | 308.15 | ||
| Langmuir | qm (mg·g−1) | 58.124 | 62.712 | 68.36 |
| KL (L·mg−1) | 0.0172 | 0.0178 | 0.0180 | |
| R2 | 0.973 | 0.967 | 0.963 | |
| Freundlich | KF (mg·g−1) (L·mg−1)1/n | 2.916 | 3.193 | 3.387 |
| 1/n | 0.555 | 0.555 | 0.562 | |
| R2 | 0.923 | 0.917 | 0.913 | |
| Temkin | KT (J·mol−1) | 13.88 | 15.128 | 16.509 |
| f (L·mg−1) | 0.142 | 0.144 | 0.144 | |
| R2 | 0.990 | 0.989 | 0.987 | |
| Dubinin-Radushkevich | qm (mg·g−1) | 35.575 | 38.487 | 41.564 |
| (mol2·J−2) | 7.1094n−5 | 6.1774n−5 | 5.5834n−5 | |
| R2 | 0.940 | 0.954 | 0.959 | |
| ΔH0 (kJ·mol−1) | ΔS0 (J·(mol·K)−1) | ΔG0 ( kJ·mol−1) | ||
| 288.15 K | 298.15 K | 308.15 K | ||
| 45.43 | 0.198 | −0.805 | −0.944 | −1.079 |
| Standard Orders | Independent Values(X) | Responses (Y) | |||||
|---|---|---|---|---|---|---|---|
| Real Values | Coded Values | ||||||
| pH | Temperature (K) | Dosage (g) | pH | Temperature (K) | Dosage (g) | qe (mg/g) | |
| 1 | 7 | 288.15 | 0.1 | 0 | −1 | +1 | 2.67 |
| 2 | 7 | 298.15 | 0.06 | 0 | 0 | 0 | 4.42 |
| 3 | 11 | 308.15 | 0.06 | +1 | +1 | 0 | 2.47 |
| 4 | 7 | 308.15 | 0.02 | 0 | +1 | −1 | 5.71 |
| 5 | 3 | 288.15 | 0.06 | −1 | −1 | 0 | 5.57 |
| 6 | 7 | 298.15 | 0.06 | 0 | 0 | 0 | 4.42 |
| 7 | 7 | 298.15 | 0.06 | 0 | 0 | 0 | 4.42 |
| 8 | 11 | 288.15 | 0.06 | +1 | −1 | 0 | 2.28 |
| 9 | 11 | 298.15 | 0.02 | +1 | 0 | −1 | 3.88 |
| 10 | 7 | 308.15 | 0.10 | 0 | +1 | +1 | 7.65 |
| 11 | 3 | 298.15 | 0.10 | −1 | 0 | +1 | 4.78 |
| 12 | 3 | 298.15 | 0.02 | −1 | 0 | −1 | 7.42 |
| 13 | 7 | 288.15 | 0.02 | 0 | −1 | −1 | 5.54 |
| 14 | 3 | 308.15 | 0.06 | −1 | 1 | 0 | 6.14 |
| 15 | 11 | 298.15 | 0.10 | +1 | 0 | +1 | 3.95 |
| 16 | 7 | 298.15 | 0.06 | 0 | 0 | 0 | 4.42 |
| 17 | 7 | 298.15 | 0.06 | 0 | 0 | 0 | 4.42 |
| Source | Sum of Squares | Degree of Freedom | Mean Square | F−Value | P−Value |
|---|---|---|---|---|---|
| Model | 29.60 | 6 | 4.93 | 6.46 | 0.0052 |
| A | 16.05 | 1 | 16.05 | 21.01 | 0.0010 |
| B | 1.53 | 1 | 1.53 | 2.01 | 0.1872 |
| C | 4.37 | 1 | 4.37 | 5.72 | 0.0379 |
| AB | 1.84 | 1 | 1.84 | 2.40 | 0.1521 |
| AC | 0.036 | 1 | 0.036 | 0.047 | 0.8323 |
| BC | 5.78 | 1 | 5.78 | 7.57 | 0.0204 |
| Residual | 7.64 | 10 | 0.76 | ||
| Lack of Fit | 7.64 | 6 | 1.27 | ||
| Pure Error | 0.00 | 4 | 0.00 | ||
| Cor Total | 37.24 | 16 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, Y.; Pan, C.; Zheng, X.; Liu, S.; Hu, F.; Xu, L.; Xu, G.; Peng, X. Removal of Ciprofloxacin with Aluminum-Pillared Kaolin Sodium Alginate Beads (CA-Al-KABs): Kinetics, Isotherms, and BBD Model. Water 2020, 12, 905. https://doi.org/10.3390/w12030905
Hu Y, Pan C, Zheng X, Liu S, Hu F, Xu L, Xu G, Peng X. Removal of Ciprofloxacin with Aluminum-Pillared Kaolin Sodium Alginate Beads (CA-Al-KABs): Kinetics, Isotherms, and BBD Model. Water. 2020; 12(3):905. https://doi.org/10.3390/w12030905
Chicago/Turabian StyleHu, Yuying, Cheng Pan, Xiaohuan Zheng, Susu Liu, Fengping Hu, Li Xu, Gaoping Xu, and Xiaoming Peng. 2020. "Removal of Ciprofloxacin with Aluminum-Pillared Kaolin Sodium Alginate Beads (CA-Al-KABs): Kinetics, Isotherms, and BBD Model" Water 12, no. 3: 905. https://doi.org/10.3390/w12030905
APA StyleHu, Y., Pan, C., Zheng, X., Liu, S., Hu, F., Xu, L., Xu, G., & Peng, X. (2020). Removal of Ciprofloxacin with Aluminum-Pillared Kaolin Sodium Alginate Beads (CA-Al-KABs): Kinetics, Isotherms, and BBD Model. Water, 12(3), 905. https://doi.org/10.3390/w12030905