Seasonal and Spatial Variation of Mo Isotope Compositions in Headwater Stream of Xijiang River Draining the Carbonate Terrain, Southwest China
Abstract
1. Introduction
2. Materials and Methods
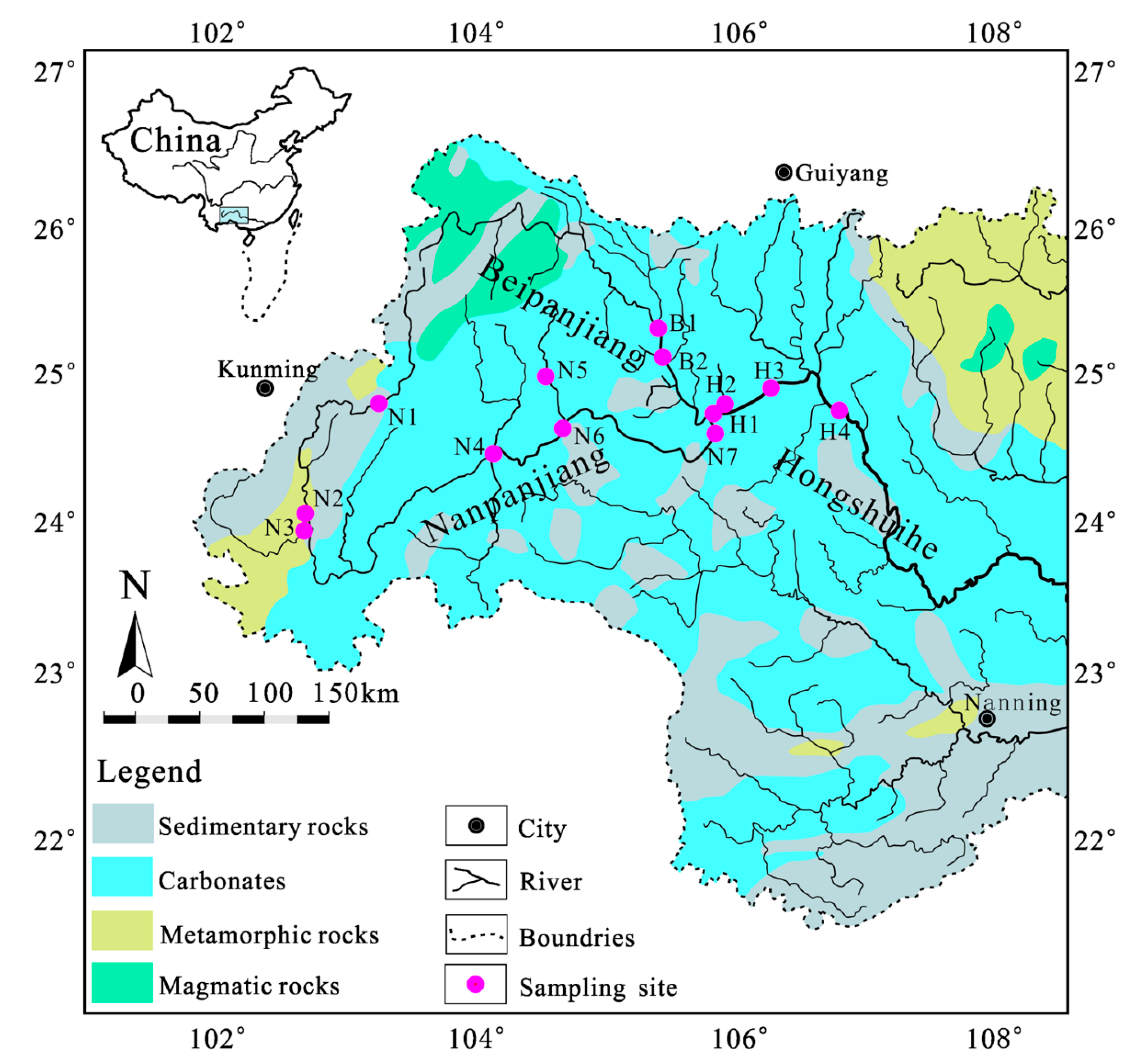
2.1. Study Area
2.2. Sampling and Chemical Analysis
2.3. Analytical Technique for Mo Isotope Composition
3. Results
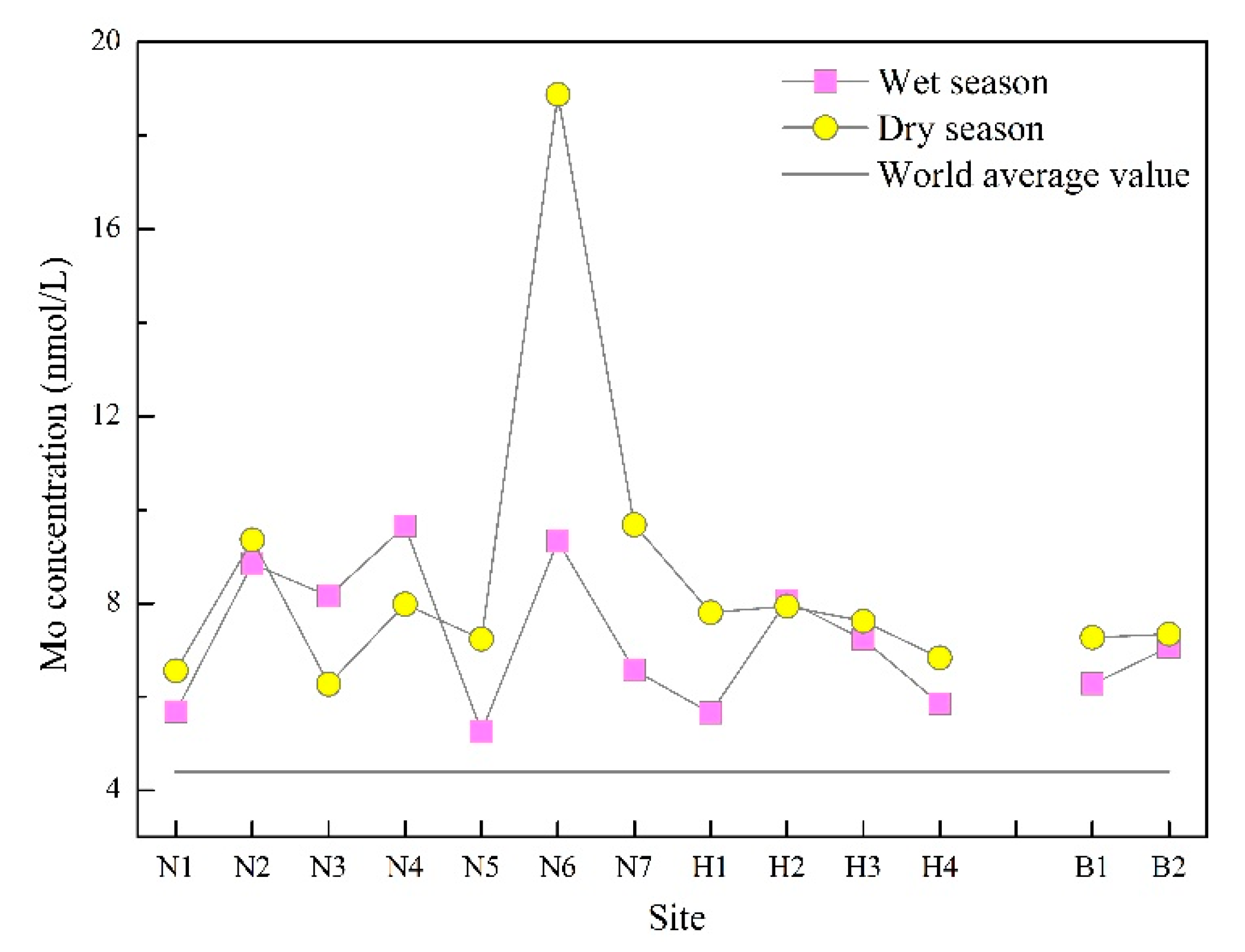
3.1. Seasonal and Spatial Variations in Mo Concentration
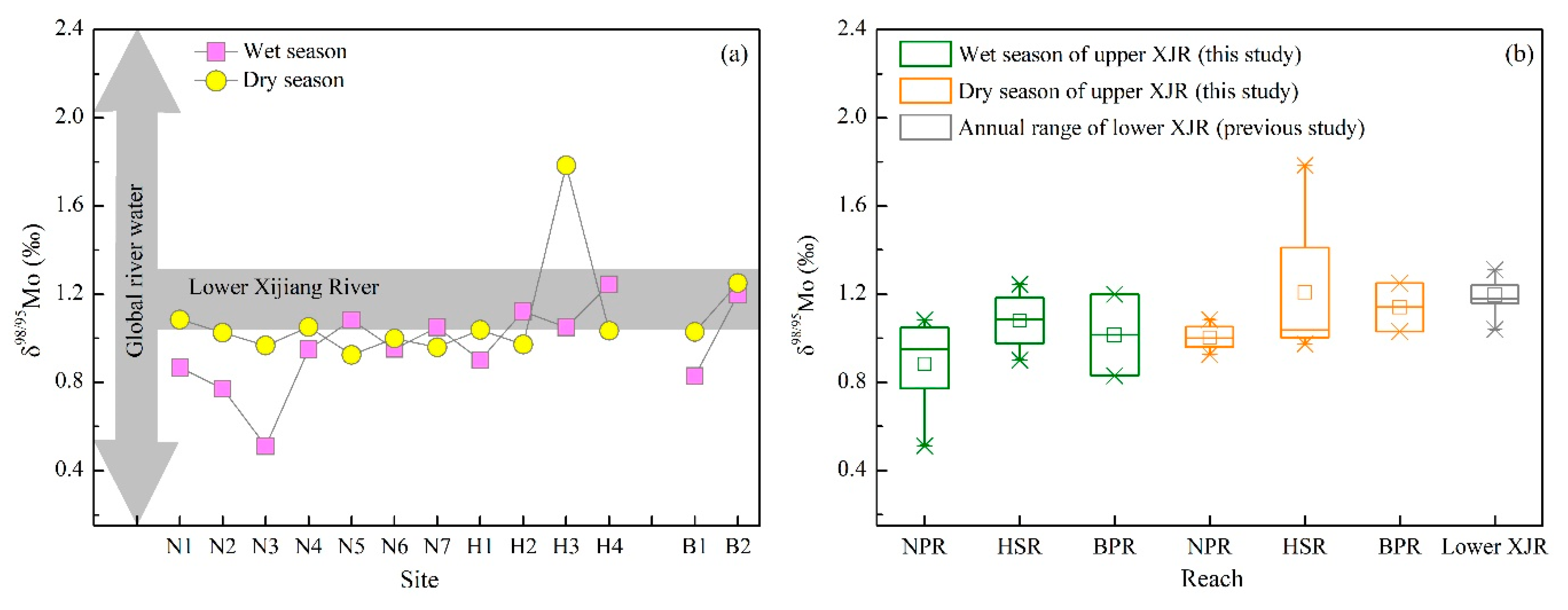
3.2. Mo Isotopic Composition
4. Discussion
4.1. Source of Dissolved Mo in the River Water
4.1.1. Atmospheric and Anthropogenic Inputs
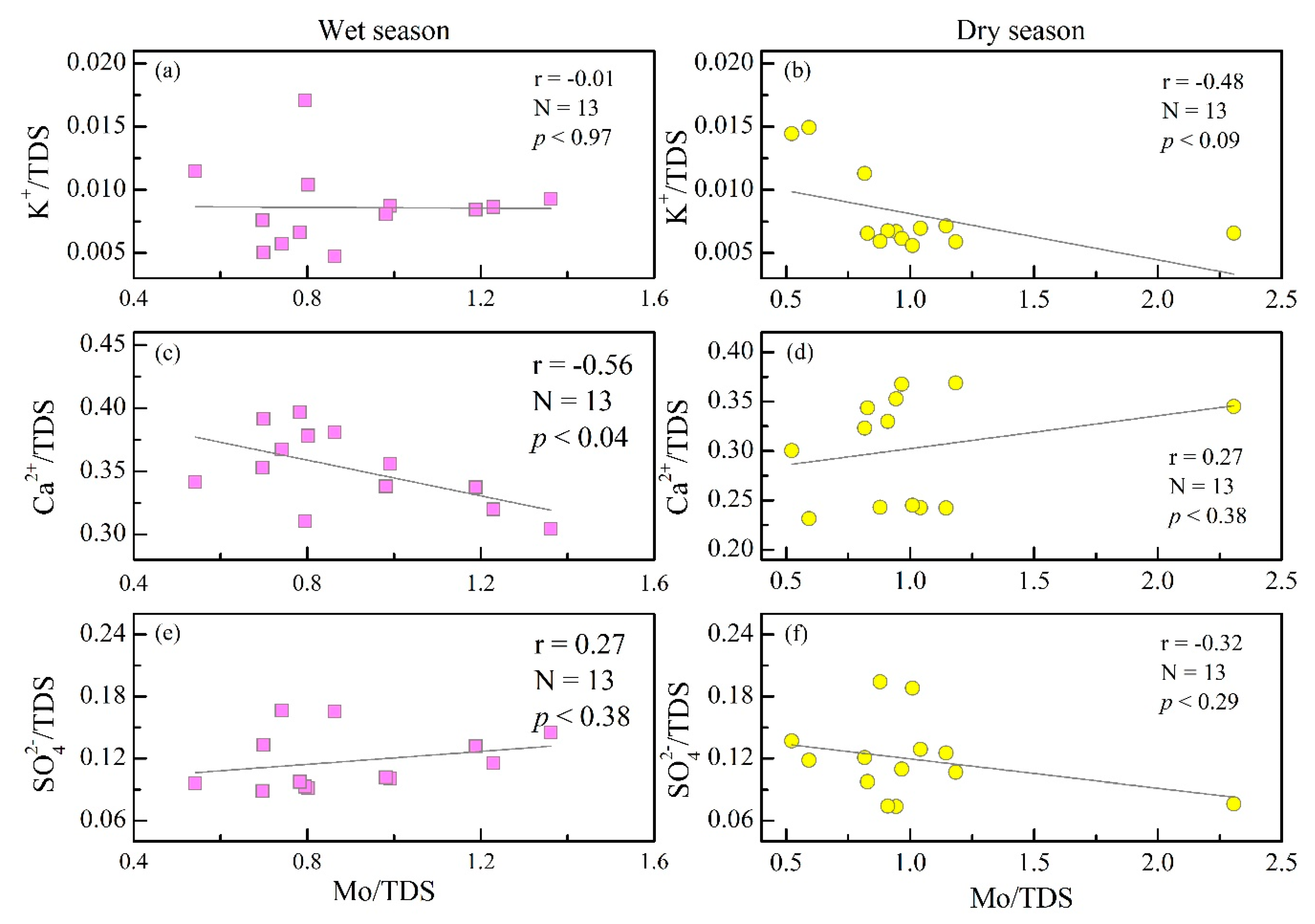
4.1.2. Weathering of Rock
4.2. Mo Isotope Fractionation during Weathering and Riverine Transportation
4.3. Consequences of River in Carbonate Terrain for the Global Mo Isotope Budget
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barbieri, M. Isotopes in Hydrology and Hydrogeology. Water 2019, 11, 291. [Google Scholar]
- Han, G.; Lv, P.; Tang, Y.; Song, Z. Spatial and temporal variation of H and O isotopic compositions of the Xijiang River system, Southwest China. Isot. Environ. Health Stud. 2018, 54, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Han, G.; Liu, M.; Li, X.; Liu, J.; Zhang, Q. Spatial and Seasonal Variation of O and H Isotopes in the Jiulong River, Southeast China. Water 2018, 10, 1677. [Google Scholar] [CrossRef]
- Wang, W.; Li, S.-L.; Zhong, J.; Li, C.; Yi, Y.; Chen, S.; Ren, Y. Understanding transport and transformation of dissolved inorganic carbon (DIC) in the reservoir system using δ13CDIC and water chemistry. J. Hydrol. 2019, 574, 193–201. [Google Scholar] [CrossRef]
- Wang, X.; Qiao, W.; Chen, J.; Liu, X.; Yang, F. Understanding the Burial and Migration Characteristics of Deep Geothermal Water Using Hydrogen, Oxygen and Inorganic Carbon Isotopes. Water 2018, 10, 7. [Google Scholar] [CrossRef]
- Boschetti, T.; Awaleh, M.O.; Barbieri, M. Waters from the Djiboutian Afar: A Review of Strontium Isotopic Composition and a Comparison with Ethiopian Waters and Red Sea Brines. Water 2018, 10, 1700. [Google Scholar] [CrossRef]
- Han, G.; Tang, Y.; Xu, Z. Fluvial geochemistry of rivers draining karst terrain in Southwest China. J. Asian Earth Sci. 2010, 38, 65–75. [Google Scholar] [CrossRef]
- Zhang, X.; Xu, Z.; Liu, W.; Moon, S.; Zhao, T.; Zhou, X.; Zhang, J.; Wu, Y.; Jiang, H.; Zhou, L. Hydro-Geochemical and Sr Isotope Characteristics of the Yalong River Basin, Eastern Tibetan Plateau: Implications for Chemical Weathering and Controlling Factors. Geochem. Geophys. Geosyst. 2019, 20, 1221–1239. [Google Scholar] [CrossRef]
- Wu, W.; Sun, M.; Ji, X.; Qu, S. Sr isotopic characteristics and fractionation during weathering of a small granitic watershed system in the Jiuhua Mountains (eastern China). J. Hydrol. 2019, 568, 135–146. [Google Scholar] [CrossRef]
- Li, C.; Li, S.-L.; Yue, F.-J.; Liu, J.; Zhong, J.; Yan, Z.-F.; Zhang, R.-C.; Wang, Z.-J.; Xu, S. Identification of sources and transformations of nitrate in the Xijiang River using nitrate isotopes and Bayesian model. Sci. Total Environ. 2019, 646, 801–810. [Google Scholar] [CrossRef]
- Yue, F.-J.; Liu, C.-Q.; Li, S.-L.; Zhao, Z.-Q.; Liu, X.-L.; Ding, H.; Liu, B.-J.; Zhong, J. Analysis of δ 15 N and δ 18 O to identify nitrate sources and transformations in Songhua River, Northeast China. J. Hydrol. 2014, 519, 329–339. [Google Scholar] [CrossRef]
- Peters, M.; Guo, Q.; Strauss, H.; Wei, R.; Li, S.; Yue, F. Contamination patterns in river water from rural Beijing: A hydrochemical and multiple stable isotope study. Sci. Total Environ. 2019, 654, 226–236. [Google Scholar] [CrossRef]
- Jiménez-Madrid, A.; Castaño, S.; Vadillo, I.; Martinez, C.; Carrasco, F.; Soler, A. Applications of Hydro-Chemical and Isotopic Tools to Improve Definitions of Groundwater Catchment Zones in a Karstic Aquifer: A Case Study. Water 2017, 9, 595. [Google Scholar] [CrossRef]
- Moore, L.J.; Machlan, L.A.; Shields, W.R.; Garner, E.L. Internal normalization techniques for high accuracy isotope dilution analyses. Application to molybdenum and nickel in standard reference materials. Anal. Chem. 1974, 46, 1082–1089. [Google Scholar] [CrossRef]
- Wang, Z.; Ma, J.; Li, J.; Wei, G.; Chen, X.; Deng, W.; Xie, L.; Lu, W.; Zou, L. Chemical weathering controls on variations in the molybdenum isotopic composition of river water: Evidence from large rivers in China. Chem. Geol. 2015, 410, 201–212. [Google Scholar] [CrossRef]
- Pearce, C.R.; Burton, K.W.; von Strandmann, P.A.E.P.; James, R.H.; Gíslason, S.R. Molybdenum isotope behaviour accompanying weathering and riverine transport in a basaltic terrain. Earth Planet. Sci. Lett. 2010, 295, 104–114. [Google Scholar] [CrossRef]
- Wille, M.; Kramers, J.D.; Nägler, T.F.; Beukes, N.J.; Schröder, S.; Meisel, T.; Lacassie, J.P.; Voegelin, A.R. Evidence for a gradual rise of oxygen between 2.6 and 2.5Ga from Mo isotopes and Re-PGE signatures in shales. Geochim. Cosmochim. Acta 2007, 71, 2417–2435. [Google Scholar] [CrossRef]
- Wille, M.; Naegler, T.F.; Lehmann, B.; Schroeder, S.; Kramers, J.D. Hydrogen sulphide release to surface waters at the Precambrian/Cambrian boundary. Nature 2008, 453, 767–769. [Google Scholar] [CrossRef] [PubMed]
- Archer, C.; Vance, D. The isotopic signature of the global riverine molybdenum flux and anoxia in the ancient oceans. Nat. Geosci. 2008, 1, 597–600. [Google Scholar] [CrossRef]
- Wang, Z.; Ma, J.; Li, J.; Wei, G.; Zeng, T.; Li, L.; Zhang, L.; Deng, W.; Xie, L.; Liu, Z. Fe (hydro) oxide controls Mo isotope fractionation during the weathering of granite. Geochim. Cosmochim. Acta 2018, 226, 1–17. [Google Scholar] [CrossRef]
- Neubert, N.; Heri, A.R.; Voegelin, A.R.; Nägler, T.F.; Schlunegger, F.; Villa, I.M. The molybdenum isotopic composition in river water: Constraints from small catchments. Earth Planet. Sci. Lett. 2011, 304, 180–190. [Google Scholar] [CrossRef]
- Hammond, C.R. The Elements. In CRC Handbook of Chemistry and Physics; Lide, D.R., Ed.; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Xu, Z.; Liu, C.Q. Chemical weathering in the upper reaches of Xijiang River draining the Yunnan–Guizhou Plateau, Southwest China. Chem. Geol. 2007, 239, 83–95. [Google Scholar] [CrossRef]
- Li, S.-L.; Calmels, D.; Han, G.; Gaillardet, J.; Liu, C.-Q. Sulfuric acid as an agent of carbonate weathering constrained by δ13CDIC: Examples from Southwest China. Earth Planet. Sci. Lett. 2008, 270, 189–199. [Google Scholar] [CrossRef]
- Han, G.; Liu, C.-Q. Water geochemistry controlled by carbonate dissolution: A study of the river waters draining karst-dominated terrain, Guizhou Province, China. Chem. Geol. 2004, 204, 1–21. [Google Scholar] [CrossRef]
- Wu, Q.; Han, G.; Li, F.; Tang, Y. Characteristic and source analysis of major ions in Nanpanjiang and Beipanjiang at the upper Pearl River during the wet season. Environ. Chem. 2015, 34, 1289–1296. (In Chinese) [Google Scholar]
- Ma, K.; Wu, Q.; Han, G.; Dong, A. Hydrochemical characteristics and sources of Nanpanjiang and Beipanjiang river basins during dry seasons. Carsol. Sin. 2018, 37, 192–202. (In Chinese) [Google Scholar]
- Liu, J.; Li, S.-L.; Chen, J.-B.; Zhong, J.; Yue, F.-J.; Lang, Y.; Ding, H. Temporal transport of major and trace elements in the upper reaches of the Xijiang River, SW China. Environ. Earth Sci. 2017, 76, 299. [Google Scholar] [CrossRef]
- Zeng, J.; Han, G.; Wu, Q.; Tang, Y. Geochemical characteristics of dissolved heavy metals in Zhujiang River, Southwest China: Spatial-temporal distribution, source, export flux estimation and a water quality assessment. PeerJ 2019, 7, e6578. [Google Scholar] [CrossRef]
- Li, J.; Liang, X.-R.; Zhong, L.-F.; Wang, X.-C.; Ren, Z.-Y.; Sun, S.-L.; Zhang, Z.-F.; Xu, J.-F. Measurement of the Isotopic Composition of Molybdenum in Geological Samples by MC-ICP-MS using a Novel Chromatographic Extraction Technique. Geostand. Geoanal. Res. 2014, 38, 345–354. [Google Scholar] [CrossRef]
- Gaillardet, J.; Viers, J.; Dupré, B. Trace Elements in River Waters. In Treatise on Geochemistry, 2nd ed.; Holland, H.D., Turekian, K.K., Eds.; Elsevier: Oxford, UK, 2014; pp. 195–235. [Google Scholar]
- Li, S.; Zhang, Q. Spatial characterization of dissolved trace elements and heavy metals in the upper Han River (China) using multivariate statistical techniques. J. Hazard. Mater. 2010, 176, 579–588. [Google Scholar] [CrossRef]
- Rahaman, W.; Goswami, V.; Singh, S.K.; Rai, V.K. Molybdenum isotopes in two Indian estuaries: Mixing characteristics and input to oceans. Geochim. Cosmochim. Acta 2014, 141, 407–422. [Google Scholar] [CrossRef]
- Voegelin, A.R.; Nägler, T.F.; Pettke, T.; Neubert, N.; Steinmann, M.; Pourret, O.; Villa, I.M. The impact of igneous bedrock weathering on the Mo isotopic composition of stream waters: Natural samples and laboratory experiments. Geochim. Cosmochim. Acta 2012, 86, 150–165. [Google Scholar] [CrossRef]
- Siebert, C.; Nägler, T.F.; von Blanckenburg, F.; Kramers, J.D. Molybdenum isotope records as a potential new proxy for paleoceanography. Earth Planet. Sci. Lett. 2003, 211, 159–171. [Google Scholar] [CrossRef]
- An, H. Remote sensing survey and analysis of current land utilization in the drainage area of Nanpanjiang and Beipanjiang rivers in Guizhou province. Guizhou Geol. 1996, 13, 344–349. (In Chinese) [Google Scholar]
- Tang, Y.; Han, G.L.; Li, F.S.; Wu, Q.X. Natural and anthropogenic sources of atmospheric dust at a remote forest area in Guizhou karst region, southwest China. Geochem.-Explor. Environ. Anal. 2016, 16, 159–163. [Google Scholar] [CrossRef]
- Tang, Y.; Han, G. Seasonal Variation and Quality Assessment of the Major and Trace Elements of Atmospheric Dust in a Typical Karst City, Southwest China. Int. J. Environ. Res. Public Health 2019, 16, 325. [Google Scholar] [CrossRef] [PubMed]
- Han, G.; Wu, Q.; Tang, Y. Acid rain and alkalization in southwestern China: Chemical and strontium isotope evidence in rainwater from Guiyang. J. Atmos. Chem. 2011, 68, 139–155. [Google Scholar] [CrossRef]
- Han, G.; Tang, Y.; Wu, Q.; Tan, Q. Chemical and strontium isotope characterization of rainwater in karst virgin forest, Southwest China. Atmos. Environ. 2010, 44, 174–181. [Google Scholar] [CrossRef]
- Nie, X.; Wang, Y.; Li, Y.; Sun, L.; Li, T.; Yang, M.; Yang, X.; Wang, W. Characteristics and impacts of trace elements in atmospheric deposition at a high-elevation site, southern China. Environ. Sci. Pollut. Res. 2017, 24, 22839–22851. [Google Scholar] [CrossRef]
- Huang, S.; Tu, J.; Liu, H.; Hua, M.; Liao, Q.; Feng, J.; Weng, Z.; Huang, G. Multivariate analysis of trace element concentrations in atmospheric deposition in the Yangtze River Delta, East China. Atmos. Environ. 2009, 43, 5781–5790. [Google Scholar] [CrossRef]
- Zheng, X.; Guo, X.; Zhao, W.; Shu, T.; Xin, Y.; Yan, X.; Xiong, Q.; Chen, F.; Lv, M. Spatial variation and provenance of atmospheric trace elemental deposition in Beijing. Atmos. Pollut. Res. 2016, 7, 260–267. [Google Scholar] [CrossRef]
- Lyu, Y.; Zhang, K.; Chai, F.; Cheng, T.; Yang, Q.; Zheng, Z.; Li, X. Atmospheric size-resolved trace elements in a city affected by non-ferrous metal smelting: Indications of respiratory deposition and health risk. Environ. Pollut. 2017, 224, 559–571. [Google Scholar] [CrossRef]
- Lee, P.-K.; Choi, B.-Y.; Kang, M.-J. Assessment of mobility and bio-availability of heavy metals in dry depositions of Asian dust and implications for environmental risk. Chemosphere 2015, 119, 1411–1421. [Google Scholar] [CrossRef]
- Liu, J.; Li, S.; Zhong, J.; Zhu, X.; Guo, Q.; Lang, Y.; Han, X. Sulfate sources constrained by sulfur and oxygen isotopic compositions in the upper reaches of the Xijiang River, China. Acta Geochim. 2017, 36, 611–618. [Google Scholar] [CrossRef]
- Gaillardet, J.; Dupré, B.; Louvat, P.; Allègre, C.J. Global silicate weathering and CO2 consumption rates deduced from the chemistry of large rivers. Chem. Geol. 1999, 159, 3–30. [Google Scholar] [CrossRef]
- Lü, P.; Han, G.; Wu, Q. Chemical characteristics of rainwater in karst rural areas, Guizhou Province, Southwest China. Acta Geochim. 2017, 36, 572–576. [Google Scholar] [CrossRef]
- Wu, Q.; Han, G.; Tao, F.; Tang, Y. Chemical composition of rainwater in a karstic agricultural area, Southwest China: The impact of urbanization. Atmos. Res. 2012, 111, 71–78. [Google Scholar] [CrossRef]
- Xu, L.; Lehmann, B.; Mao, J. Seawater contribution to polymetallic Ni–Mo–PGE–Au mineralization in Early Cambrian black shales of South China: Evidence from Mo isotope, PGE, trace element and REE geochemistry. Ore Geol. Rev. 2013, 52, 66–84. [Google Scholar] [CrossRef]
- Voegelin, A.R.; Nägler, T.F.; Samankassou, E.; Villa, I.M. Molybdenum isotopic composition of modern and Carboniferous carbonates. Chem. Geol. 2009, 265, 488–498. [Google Scholar] [CrossRef]
- Goldberg, T.; Archer, C.; Vance, D.; Poulton, S.W. Mo isotope fractionation during adsorption to Fe (oxyhydr)oxides. Geochim. Cosmochim. Acta 2009, 73, 6502–6516. [Google Scholar] [CrossRef]
- Siebert, C.; Pett-Ridge, J.C.; Opfergelt, S.; Guicharnaud, R.A.; Halliday, A.N.; Burton, K.W. Molybdenum isotope fractionation in soils: Influence of redox conditions, organic matter and atmospheric inputs. Geochim. Cosmochim. Acta 2015, 162, 1–24. [Google Scholar] [CrossRef]
- Dai, Q.; Liu, Z.; Shao, H.; Yang, Z. Karst bare slope soil erosion and soil quality: A simulation case study. Solid Earth 2015, 6, 985–995. [Google Scholar] [CrossRef]
- Dai, Q.H.; Peng, X.D.; Wang, P.J.; Li, C.L.; Shao, H.B. Surface erosion and underground leakage of yellow soil on slopes in karst regions of southwest China. Land Degrad. Dev. 2018, 29, 2438–2448. [Google Scholar] [CrossRef]
- An, Y.; Hou, Y.; Wu, Q.; Qing, L.; Li, L. Chemical weathering and CO2 consumption of a high-erosion-rate karstic river: A case study of the Sanchahe River, southwest China. Chin. J. Geochem. 2015, 34, 601–609. [Google Scholar] [CrossRef]
- Zeng, J.; Han, G.; Wu, Q.; Tang, Y. Heavy Metals in Suspended Particulate Matter of Zhujiang River, Southwest China: Contents, Sources and Health Risks. Int. J. Environ. Res. Public Health 2019, 94, e94145. [Google Scholar]
- Zou, J. Geochemical characteristics and organic carbon sources within the upper reaches of the Xi River, southwest China during high flow. J. Earth Syst. Sci. 2017, 126, 6. [Google Scholar] [CrossRef][Green Version]
- McManus, J.; Berelson, W.M.; Severmann, S.; Poulson, R.L.; Hammond, D.E.; Klinkhammer, G.P.; Holm, C. Molybdenum and uranium geochemistry in continental margin sediments: Paleoproxy potential. Geochim. Cosmochim. Acta 2006, 70, 4643–4662. [Google Scholar] [CrossRef]

| Sample | pH a | T a | EC a | DO a | Mo a | δ98/95Mo | Na+ | K+ | Mg2+ | Ca2+ | HCO3− | Cl− | NO3− | SO42− |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| °C | μS/cm | mg/L | nmol/L | ‰ | mmol/L b | |||||||||
| Wet season | ||||||||||||||
| N1 | 7.6 | 21.3 | 249 | 4.9 | 5.7 | 0.87 | 0.45 | 0.12 | 0.63 | 1.79 | 3.29 | 0.48 | 0.29 | 0.50 |
| N2 | 7.8 | 22.7 | 267 | 6.9 | 8.8 | 0.77 | 0.23 | 0.11 | 0.68 | 2.09 | 3.27 | 0.44 | 0.45 | 0.50 |
| N3 | 8.1 | 23.3 | 259 | 7.1 | 8.2 | 0.51 | 0.56 | 0.18 | 0.79 | 1.59 | 2.84 | 0.56 | 0.41 | 0.48 |
| N4 | 7.9 | 23.9 | 225 | 6.8 | 9.7 | 0.95 | 0.26 | 0.08 | 0.58 | 1.73 | 2.95 | 0.27 | 0.56 | 0.49 |
| N5 | 8.1 | 20.3 | 181 | 8.1 | 5.3 | 1.08 | 0.13 | 0.04 | 0.39 | 1.47 | 2.15 | 0.16 | 0.31 | 0.50 |
| N6 | 8.1 | 27.1 | 163 | 7.4 | 9.3 | 0.95 | 0.29 | 0.06 | 0.54 | 1.04 | 1.97 | 0.17 | 0.21 | 0.50 |
| N7 | 8.4 | 30.6 | 158 | 12.3 | 6.6 | 1.05 | 0.22 | 0.05 | 0.47 | 1.13 | 2.21 | 0.17 | 0.16 | 0.34 |
| H1 | 8.0 | 31.9 | 193 | 8.4 | 5.7 | 0.90 | 0.34 | 0.06 | 0.46 | 1.43 | 2.74 | 0.41 | 0.06 | 0.36 |
| H2 | 8.4 | 31.7 | 162 | 12.4 | 8.1 | 1.12 | 0.26 | 0.06 | 0.51 | 1.05 | 2.05 | 0.18 | 0.14 | 0.38 |
| H3 | 8.3 | 33.3 | 155 | 10.0 | 7.2 | 1.05 | 0.24 | 0.05 | 0.43 | 1.03 | 1.84 | 0.14 | 0.10 | 0.40 |
| H4 | 7.7 | 22.7 | 182 | 6.6 | 5.9 | 1.25 | 0.17 | 0.05 | 0.36 | 1.48 | 2.60 | 0.11 | 0.12 | 0.36 |
| B1 | 8.3 | 27.0 | 206 | 9.0 | 6.3 | 0.83 | 0.23 | 0.05 | 0.46 | 1.55 | 2.33 | 0.14 | 0.28 | 0.70 |
| B2 | 8.0 | 21.8 | 201 | 8.5 | 7.1 | 1.20 | 0.25 | 0.04 | 0.37 | 1.56 | 2.33 | 0.11 | 0.25 | 0.68 |
| Dry season | ||||||||||||||
| N1 | 8.2 | 11.4 | 602 | 9.0 | 6.6 | 1.09 | 0.92 | 0.18 | 0.67 | 1.88 | 3.36 | 0.73 | 0.52 | 0.86 |
| N2 | 8.3 | 13.4 | 530 | 8.5 | 9.4 | 1.03 | 0.44 | 0.13 | 0.78 | 1.85 | 3.41 | 0.39 | 0.44 | 0.69 |
| N3 | 8.3 | 13.6 | 588 | 8.8 | 6.3 | 0.97 | 0.66 | 0.16 | 0.81 | 1.23 | 3.70 | 0.56 | 0.19 | 0.63 |
| N4 | 8.3 | 15.3 | 396 | 7.9 | 8.0 | 1.05 | 0.22 | 0.06 | 0.51 | 1.49 | 3.17 | 0.17 | 0.21 | 0.31 |
| N5 | 8.3 | 15.4 | 401 | 7.8 | 7.2 | 0.93 | 0.21 | 0.05 | 0.48 | 1.31 | 3.15 | 0.16 | 0.20 | 0.30 |
| N6 | 8.3 | 15.7 | 398 | 7.1 | 18.9 | 1.00 | 0.21 | 0.05 | 0.50 | 1.41 | 3.12 | 0.16 | 0.20 | 0.31 |
| N7 | 8.1 | 17.8 | 384 | 7.9 | 9.7 | 0.96 | 0.23 | 0.05 | 0.46 | 1.51 | 2.74 | 0.13 | 0.22 | 0.44 |
| H1 | 8.0 | 17.5 | 383 | 7.7 | 7.8 | 1.04 | 0.22 | 0.05 | 0.46 | 0.83 | 2.76 | 0.14 | 0.23 | 0.43 |
| H2 | 8.1 | 18.0 | 381 | 8.0 | 7.9 | 0.97 | 0.23 | 0.05 | 0.47 | 1.51 | 2.73 | 0.14 | 0.21 | 0.45 |
| H3 | 8.3 | 10.6 | 465 | 9.2 | 7.6 | 1.78 | 0.21 | 0.05 | 0.60 | 1.05 | 3.11 | 0.17 | 0.12 | 0.84 |
| H4 | 7.8 | 19.1 | 370 | 7.7 | 6.8 | 1.03 | 0.22 | 0.05 | 0.42 | 0.80 | 2.70 | 0.12 | 0.20 | 0.42 |
| B1 | 8.0 | 10.2 | 414 | 6.6 | 7.3 | 1.03 | 0.21 | 0.06 | 0.60 | 1.51 | 3.15 | 0.14 | 0.16 | 0.43 |
| B2 | 8.2 | 16.2 | 409 | 9.0 | 7.3 | 1.25 | 0.32 | 0.04 | 0.45 | 0.89 | 2.56 | 0.12 | 0.18 | 0.68 |
| Item | DO | FeSPM a | MnSPM a | DOC b | |
|---|---|---|---|---|---|
| δ98/95Mo | Pearson correlation | 0.31 | −0.69 | −0.72 | −0.66 |
| significance (two-tailed) | 0.12 | 0.31 | 0.28 | 0.05 | |
| N | 26 | 4 | 4 | 9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeng, J.; Han, G.; Zhu, J.-M. Seasonal and Spatial Variation of Mo Isotope Compositions in Headwater Stream of Xijiang River Draining the Carbonate Terrain, Southwest China. Water 2019, 11, 1076. https://doi.org/10.3390/w11051076
Zeng J, Han G, Zhu J-M. Seasonal and Spatial Variation of Mo Isotope Compositions in Headwater Stream of Xijiang River Draining the Carbonate Terrain, Southwest China. Water. 2019; 11(5):1076. https://doi.org/10.3390/w11051076
Chicago/Turabian StyleZeng, Jie, Guilin Han, and Jian-Ming Zhu. 2019. "Seasonal and Spatial Variation of Mo Isotope Compositions in Headwater Stream of Xijiang River Draining the Carbonate Terrain, Southwest China" Water 11, no. 5: 1076. https://doi.org/10.3390/w11051076
APA StyleZeng, J., Han, G., & Zhu, J.-M. (2019). Seasonal and Spatial Variation of Mo Isotope Compositions in Headwater Stream of Xijiang River Draining the Carbonate Terrain, Southwest China. Water, 11(5), 1076. https://doi.org/10.3390/w11051076







