Brain-Derived Extracellular Vesicle microRNA Signatures Associated with In Utero and Postnatal Oxycodone Exposure
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Oxycodone Treatment
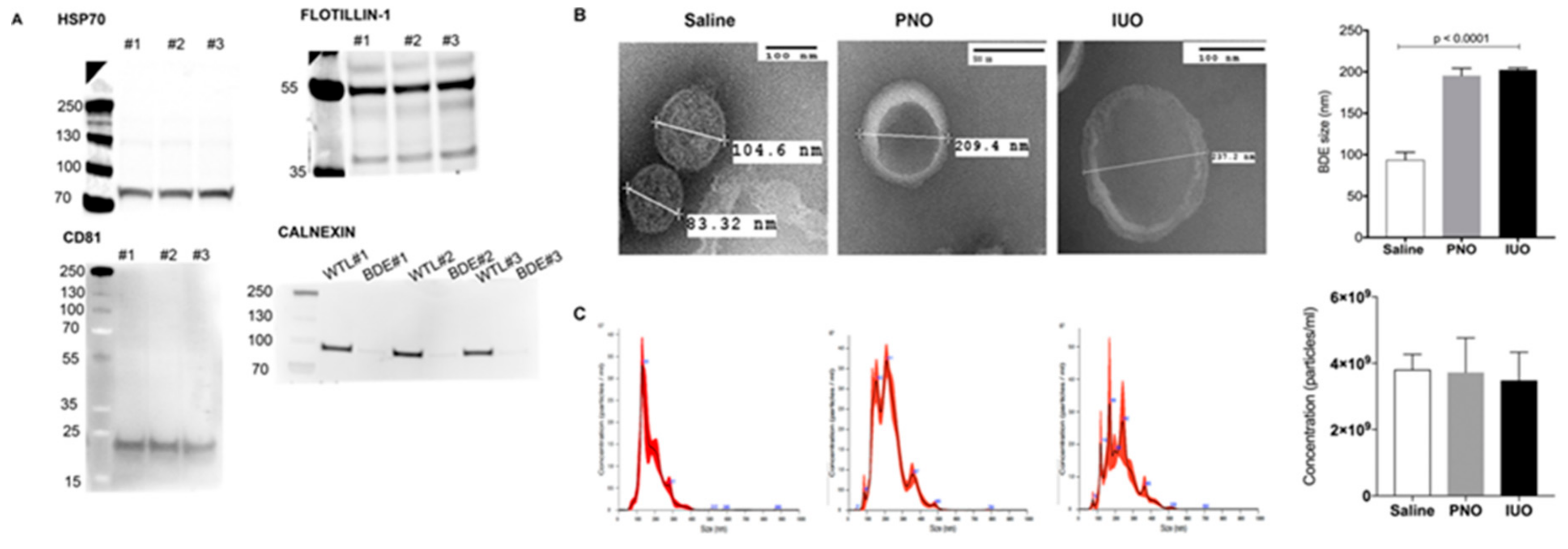
2.3. Brain-Derived Extracellular Vesicle Isolation
2.4. RNA Isolation and Sequencing
2.5. Western Blot
2.6. Transmission Electron Microscopy (TEM)
2.7. Bioinformatics
2.8. Primary Rat Neuronal Cultures and BDEs Treatment
2.9. Immunostaining of Dendritic Spines
2.10. Confocal Imaging and Image J Analysis
2.11. Crude Synaptosome Isolation
2.12. Statistical Analyses
2.13. Bioinformatics for 2D Hierarchical Clustering
3. Results
3.1. Purity and Characterization of Isolated BDEs
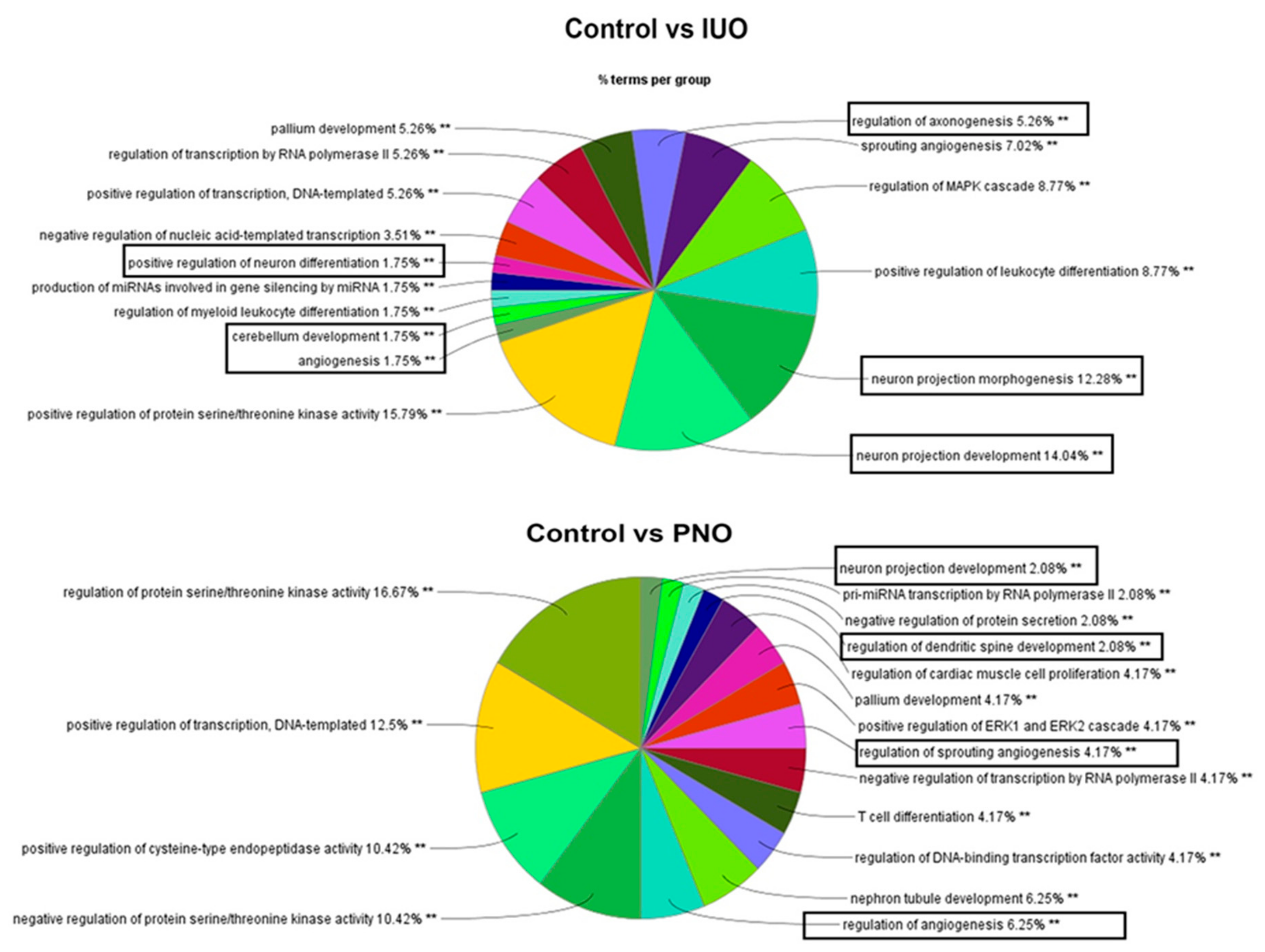
3.2. RNA Seq and Bioinformatics Analysis
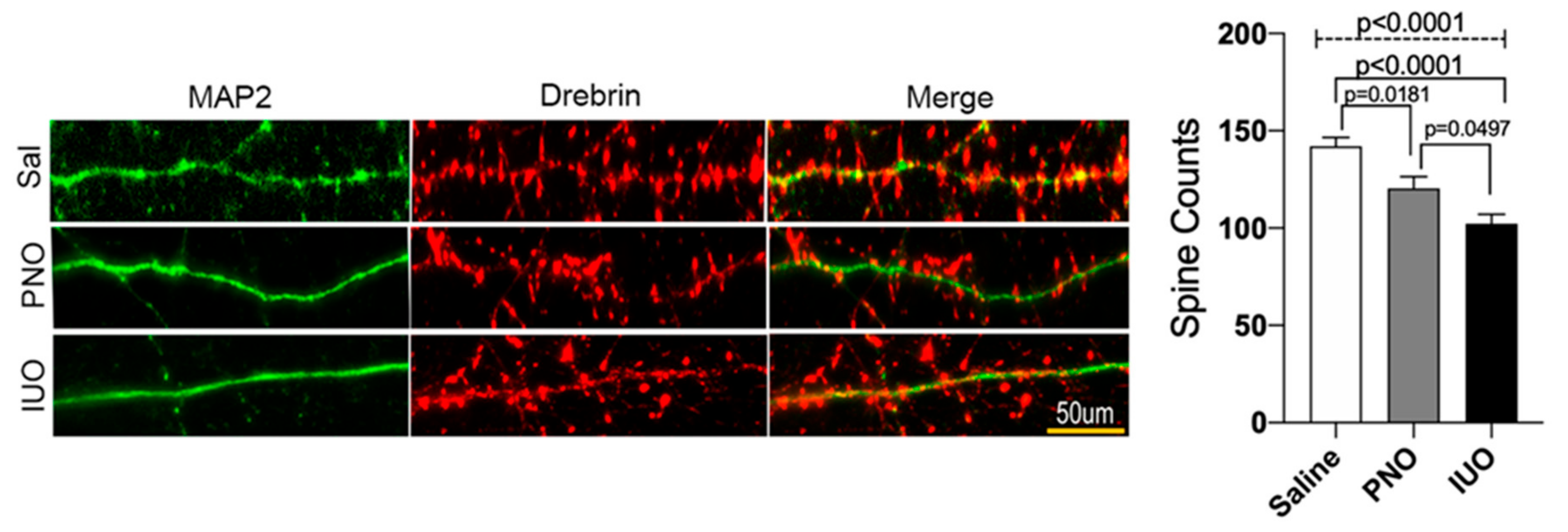
3.3. BDEs from IUO Offspring Enhance Synaptodendritic Damage
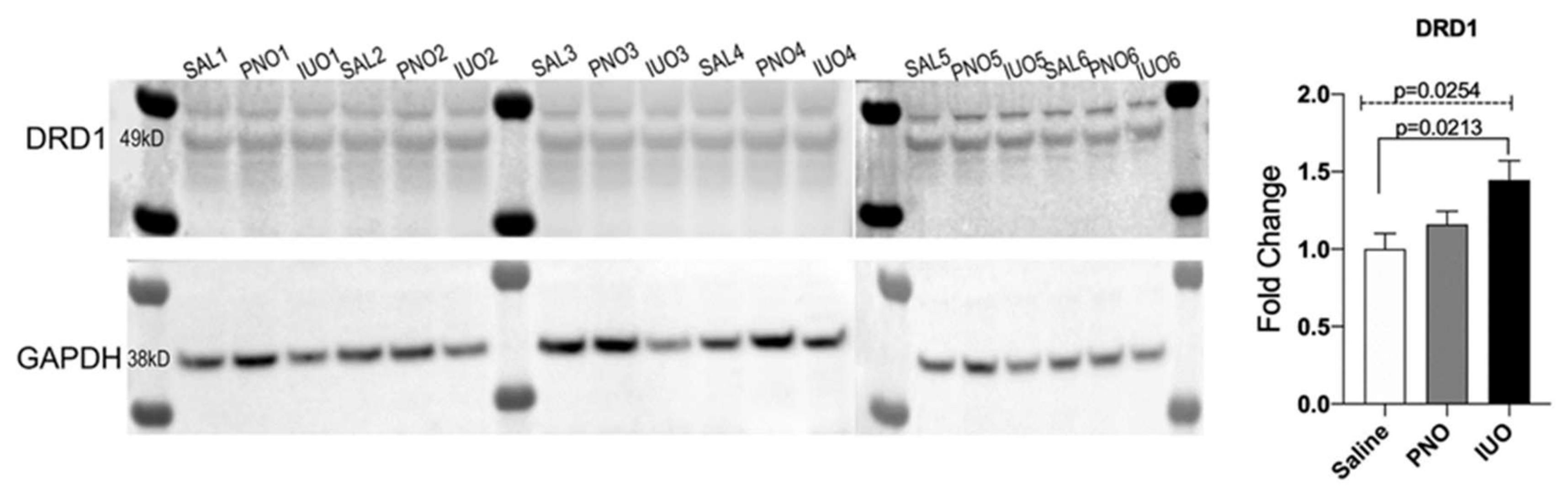
3.4. DRD1 Receptor Is Upregulated in Synaptosomal Fractions in the IUO Group
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Compton, W.M.; Volkow, N.D. Major increases in opioid analgesic abuse in the United States: concerns and strategies. Drug Alcohol Depend. 2006, 81, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Zosel, A.; Bartelson, B.B.; Bailey, E.; Lowenstein, S.; Dart, R. Characterization of adolescent prescription drug abuse and misuse using the Researched Abuse Diversion and Addiction-related Surveillance (RADARS((R))) System. J. Am. Acad. Child Adolesc. Psychiatry 2013, 52, 196–204.e192. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.S. Oxycodone/Naloxone Prolonged Release: A Review in Severe Chronic Pain. Clin. Drug Investig. 2017, 37, 1191–1201. [Google Scholar] [CrossRef] [PubMed]
- Chaves, C.; Remiao, F.; Cisternino, S.; Decleves, X. Opioids and the Blood-Brain Barrier: A Dynamic Interaction with Consequences on Drug Disposition in Brain. Curr. Neuropharmacol. 2017, 15, 1156–1173. [Google Scholar] [CrossRef]
- Okura, T.; Higuchi, K.; Deguchi, Y. The blood-brain barrier transport mechanism controlling analgesic effects of opioid drugs in CNS. Yakugaku Zasshi 2015, 135, 697–702. [Google Scholar] [CrossRef]
- Wasser, C.R.; Kavalali, E.T. Leaky synapses: regulation of spontaneous neurotransmission in central synapses. Neuroscience 2009, 158, 177–188. [Google Scholar] [CrossRef]
- Mittelbrunn, M.; Gutierrez-Vazquez, C.; Villarroya-Beltri, C.; Gonzalez, S.; Sanchez-Cabo, F.; Gonzalez, M.A.; Bernad, A.; Sanchez-Madrid, F. Unidirectional transfer of microRNA-loaded exosomes from T cells to antigen-presenting cells. Nat. Commun. 2011, 2, 282. [Google Scholar] [CrossRef]
- Fabbri, M.; Paone, A.; Calore, F.; Galli, R.; Gaudio, E.; Santhanam, R.; Lovat, F.; Fadda, P.; Mao, C.; Nuovo, G.J.; et al. MicroRNAs bind to Toll-like receptors to induce prometastatic inflammatory response. Proc. Natl. Acad. Sci. USA 2012, 109, E2110–E2116. [Google Scholar] [CrossRef]
- Montecalvo, A.; Larregina, A.T.; Shufesky, W.J.; Stolz, D.B.; Sullivan, M.L.; Karlsson, J.M.; Baty, C.J.; Gibson, G.A.; Erdos, G.; Wang, Z.; et al. Mechanism of transfer of functional microRNAs between mouse dendritic cells via exosomes. Blood 2012, 119, 756–766. [Google Scholar] [CrossRef]
- Thery, C.; Ostrowski, M.; Segura, E. Membrane vesicles as conveyors of immune responses. Nat. Rev. Immunol. 2009, 9, 581–593. [Google Scholar] [CrossRef]
- Izadpanah, M.; Seddigh, A.; Ebrahimi Barough, S.; Fazeli, S.A.S.; Ai, J. Potential of Extracellular Vesicles in Neurodegenerative Diseases: Diagnostic and Therapeutic Indications. J. Mol. Neurosci. 2018. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.P.; Franklin, L.M.; Johnson, G.S.; Schrott, L.M. Prenatal oxycodone exposure impairs spatial learning and/or memory in rats. Behav. Brain Res. 2010, 212, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Harrison, E.B.; Hochfelder, C.G.; Lamberty, B.G.; Meays, B.M.; Morsey, B.M.; Kelso, M.L.; Fox, H.S.; Yelamanchili, S.V. Traumatic brain injury increases levels of miR-21 in extracellular vesicles: Implications for neuroinflammation. FEBS Open Bio 2016, 6, 835–846. [Google Scholar] [CrossRef] [PubMed]
- Yelamanchili, S.V.; Lamberty, B.G.; Rennard, D.A.; Morsey, B.M.; Hochfelder, C.G.; Meays, B.M.; Levy, E.; Fox, H.S. MiR-21 in Extracellular Vesicles Leads to Neurotoxicity via TLR7 Signaling in SIV Neurological Disease. PLoS Pathog. 2015, 11, e1005032. [Google Scholar] [CrossRef]
- Pendyala, G.; Buescher, J.L.; Fox, H.S. Methamphetamine and inflammatory cytokines increase neuronal Na+/K+-ATPase isoform 3: relevance for HIV associated neurocognitive disorders. PLoS ONE 2012, 7, e37604. [Google Scholar] [CrossRef] [PubMed]
- Pendyala, G.; Ninemire, C.; Fox, H.S. Protective role for the disulfide isomerase PDIA3 in methamphetamine neurotoxicity. PLoS ONE 2012, 7, e38909. [Google Scholar] [CrossRef]
- Whittemore, E.R.; Korotzer, A.R.; Etebari, A.; Cotman, C.W. Carbachol increases intracellular free calcium in cultured rat microglia. Brain Res. 1993, 621, 59–64. [Google Scholar] [CrossRef]
- Agarwal, V.; Bell, G.W.; Nam, J.W.; Bartel, D.P. Predicting effective microRNA target sites in mammalian mRNAs. Elife 2015, 4. [Google Scholar] [CrossRef]
- Bindea, G.; Mlecnik, B.; Hackl, H.; Charoentong, P.; Tosolini, M.; Kirilovsky, A.; Fridman, W.H.; Pages, F.; Trajanoski, Z.; Galon, J. ClueGO: A Cytoscape plug-in to decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics 2009, 25, 1091–1093. [Google Scholar] [CrossRef]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef]
- Thery, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhu, X.; Bai, M.; Zhang, L.; Xue, L.; Yi, J. Maternal deprivation enhances behavioral vulnerability to stress associated with miR-504 expression in nucleus accumbens of rats. PLoS ONE 2013, 8, e69934. [Google Scholar] [CrossRef] [PubMed]
- Nanovskaya, T.; Deshmukh, S.; Brooks, M.; Ahmed, M.S. Transplacental transfer and metabolism of buprenorphine. J. Pharmacol. Exp. Ther. 2002, 300, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Nanovskaya, T.N.; Nekhayeva, I.A.; Hankins, G.D.; Ahmed, M.S. Transfer of methadone across the dually perfused preterm human placental lobule. Am. J. Obstet. Gynecol. 2008, 198, e121–e124. [Google Scholar] [CrossRef] [PubMed]
- Gerdin, E.; Rane, A.; Lindberg, B. Transplacental transfer of morphine in man. J. Perinat. Med. 1990, 18, 305–312. [Google Scholar] [CrossRef]
- Niklasson, B.; Arnelo, C.; Ohman, S.G.; Segerdahl, M.; Blanck, A. Oral oxycodone for pain after caesarean section: A randomized comparison with nurse-administered IV morphine in a pragmatic study. Scand. J. Pain 2015, 7, 17–24. [Google Scholar] [CrossRef]
- Nie, J.J.; Sun, S.; Huang, S.Q. Effect of oxycodone patient-controlled intravenous analgesia after cesarean section: A randomized controlled study. J. Pain Res. 2017, 10, 2649–2655. [Google Scholar] [CrossRef]
- Lewis, T.; Erfe, B.L.; Ezell, T.; Gauda, E. Pharmacoepidemiology of opiate use in the neonatal ICU: Increasing cumulative doses and iatrogenic opiate withdrawal. J. Opioid Manag. 2015, 11, 305–312. [Google Scholar] [CrossRef]
- Nair, J.; Lakshminrusimha, S. Update on PPHN: Mechanisms and treatment. Semin. Perinatol. 2014, 38, 78–91. [Google Scholar] [CrossRef]
- Lakshminrusimha, S.; Keszler, M. Persistent Pulmonary Hypertension of the Newborn. Neoreviews 2015, 16, e680–e692. [Google Scholar] [CrossRef]
- Atkinson, H.C.; Begg, E.J.; Darlow, B.A. Drugs in human milk. Clinical pharmacokinetic considerations. Clin. Pharmacokinet. 1988, 14, 217–240. [Google Scholar] [CrossRef] [PubMed]
- Seaton, S.; Reeves, M.; McLean, S. Oxycodone as a component of multimodal analgesia for lactating mothers after Caesarean section: Relationships between maternal plasma, breast milk and neonatal plasma levels. Aust. N. Z. J. Obstet. Gynaecol. 2007, 47, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Olkkola, K.T.; Hamunen, K.; Seppälä, T.; Maunuksela, E.L. Pharmacokinetics and ventilatory effects of intravenous oxycodone in postoperative children. Br. J. Clin. Pharmacol. 1994, 38, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Pokela, M.L.; Anttila, E.; Seppälä, T.; Olkkola, K.T. Marked variation in oxycodone pharmacokinetics in infants. Paediatr. Anaesth. 2005, 15, 560–565. [Google Scholar] [CrossRef] [PubMed]
- Kokki, H.; Rasanen, I.; Reinikainen, M.; Suhonen, P.; Vanamo, K.; Ojanperä, I. Pharmacokinetics of oxycodone after intravenous, buccal, intramuscular and gastric administration in children. Clin. Pharmacokinet. 2004, 43, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Kokki, M.; Heikkinen, M.; Välitalo, P.; Hautajärvi, H.; Hokkanen, J.; Pitkänen, H.; Sankilampi, U.; Ranta, V.P.; Kokki, H. Maturation of oxycodone pharmacokinetics in neonates and infants: Oxycodone and its metabolites in plasma and urine. Br. J. Clin. Pharmacol. 2017, 83, 791–800. [Google Scholar] [CrossRef] [PubMed]
- Lam, J.; Kelly, L.; Ciszkowski, C.; Landsmeer, M.L.; Nauta, M.; Carleton, B.C.; Hayden, M.R.; Madadi, P.; Koren, G. Central nervous system depression of neonates breastfed by mothers receiving oxycodone for postpartum analgesia. J. Pediatr. 2012, 160, 33–37. [Google Scholar] [CrossRef]
- Devarapalli, M.; Leonard, M.; Briyal, S.; Stefanov, G.; Puppala, B.L.; Schweig, L.; Gulati, A. Prenatal Oxycodone Exposure Alters CNS Endothelin Receptor Expression in Neonatal Rats. Drug Res. (Stuttg.) 2016, 66, 246–250. [Google Scholar] [CrossRef]
- Fan, R.; Schrott, L.M.; Arnold, T.; Snelling, S.; Rao, M.; Graham, D.; Cornelius, A.; Korneeva, N.L. Chronic oxycodone induces axonal degeneration in rat brain. BMC Neurosci. 2018, 19. [Google Scholar] [CrossRef]
- Sithisarn, T.; Legan, S.J.; Westgate, P.M.; Wilson, M.; Wellmann, K.; Bada, H.S.; Barron, S. The Effects of Perinatal Oxycodone Exposure on Behavioral Outcome in a Rodent Model. Front. Pediatr. 2017, 5. [Google Scholar] [CrossRef]
- Fan, R.; Schrott, L.M.; Snelling, S.; Ndi, J.; Arnold, T.; Korneeva, N.L. Chronic oxycodone induces integrated stress response in rat brain. BMC Neurosci. 2015, 16. [Google Scholar] [CrossRef] [PubMed]
- Yuferov, V.; Zhang, Y.; Liang, Y.; Zhao, C.; Randesi, M.; Kreek, M.J. Oxycodone Self-Administration Induces Alterations in Expression of Integrin, Semaphorin and Ephrin Genes in the Mouse Striatum. Front. Psychiatry 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liang, Y.; Levran, O.; Randesi, M.; Yuferov, V.; Zhao, C.; Kreek, M.J. Alterations of expression of inflammation/immune-related genes in the dorsal and ventral striatum of adult C57BL/6J mice following chronic oxycodone self-administration: A RNA sequencing study. Psychopharmacology 2017, 234, 2259–2275. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Mayer-Blackwell, B.; Schlussman, S.D.; Randesi, M.; Butelman, E.R.; Ho, A.; Ott, J.; Kreek, M.J. Extended access oxycodone self-administration and neurotransmitter receptor gene expression in the dorsal striatum of adult C57BL/6 J mice. Psychopharmacology 2014, 231, 1277–1287. [Google Scholar] [CrossRef]
- Zhang, Y.; Brownstein, A.J.; Buonora, M.; Niikura, K.; Ho, A.; Correa da Rosa, J.; Kreek, M.J.; Ott, J. Self administration of oxycodone alters synaptic plasticity gene expression in the hippocampus differentially in male adolescent and adult mice. Neuroscience 2015, 285, 34–46. [Google Scholar] [CrossRef]
- Randesi, M.; Zhou, Y.; Mazid, S.; Odell, S.C.; Gray, J.D.; Correa da Rosa, J.; McEwen, B.S.; Milner, T.A.; Kreek, M.J. Sex differences after chronic stress in the expression of opioid-, stress- and neuroplasticity-related genes in the rat hippocampus. Neurobiol. Stress 2018, 8, 33–41. [Google Scholar] [CrossRef]
- Bali, P.; Kenny, P.J. MicroRNAs and Drug Addiction. Front. Genet. 2013, 4, 43. [Google Scholar] [CrossRef]
- Im, H.I.; Kenny, P.J. MicroRNAs in neuronal function and dysfunction. Trends Neurosci. 2012, 35, 325–334. [Google Scholar] [CrossRef]
- Kenny, P.J. Epigenetics, microRNA, and addiction. Dialogues Clin. Neurosci. 2014, 16, 335–344. [Google Scholar]
- Valadi, H.; Ekstrom, K.; Bossios, A.; Sjostrand, M.; Lee, J.J.; Lotvall, J.O. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 2007, 9, 654–659. [Google Scholar] [CrossRef]
- Chaudhuri, A.D.; Dastgheyb, R.M.; Yoo, S.W.; Trout, A.; Talbot, C.C., Jr.; Hao, H.; Witwer, K.W.; Haughey, N.J. TNFalpha and IL-1beta modify the miRNA cargo of astrocyte shed extracellular vesicles to regulate neurotrophic signaling in neurons. Cell Death Dis. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Cesselli, D.; Parisse, P.; Aleksova, A.; Veneziano, C.; Cervellin, C.; Zanello, A.; Beltrami, A.P. Extracellular Vesicles: How Drug and Pathology Interfere With Their Biogenesis and Function. Front. Physiol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Buran, I.; Etem, E.O.; Tektemur, A.; Elyas, H. Treatment with TREK1 and TRPC3/6 ion channel inhibitors upregulates microRNA expression in a mouse model of chronic mild stress. Neurosci. Lett. 2017, 656, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Gecz, J.; Baker, E.; Donnelly, A.; Ming, J.E.; McDonald-McGinn, D.M.; Spinner, N.B.; Zackai, E.H.; Sutherland, G.R.; Mulley, J.C. Fibroblast growth factor homologous factor 2 (FHF2): Gene structure, expression and mapping to the Borjeson-Forssman-Lehmann syndrome region in Xq26 delineated by a duplication breakpoint in a BFLS-like patient. Hum. Genet. 1999, 104, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.Y.; Pang, K.; Kim, J.Y.; Ryu, J.R.; Kang, H.; Liu, Z.; Kim, W.K.; Sun, W.; Kim, H.; Han, K. Post-transcriptional regulation of SHANK3 expression by microRNAs related to multiple neuropsychiatric disorders. Mol. Brain 2015, 8. [Google Scholar] [CrossRef]
- Chen, C.L.; Liu, H.; Guan, X. Changes in microRNA expression profile in hippocampus during the acquisition and extinction of cocaine-induced conditioned place preference in rats. J. Biomed. Sci. 2013, 20. [Google Scholar] [CrossRef]
- Jimenez, K.M.; Pereira-Morales, A.J.; Forero, D.A. A Functional Polymorphism in the DRD1 Gene, That Modulates Its Regulation by miR-504, Is Associated with Depressive Symptoms. Psychiatry Investig. 2018, 15, 402–406. [Google Scholar] [CrossRef]
- Huang, W.; Li, M.D. Differential allelic expression of dopamine D1 receptor gene (DRD1) is modulated by microRNA miR-504. Biol. Psychiatry 2009, 65, 702–705. [Google Scholar] [CrossRef]
- Hersch, S.M.; Ciliax, B.J.; Gutekunst, C.A.; Rees, H.D.; Heilman, C.J.; Yung, K.K.; Bolam, J.P.; Ince, E.; Yi, H.; Levey, A.I. Electron microscopic analysis of D1 and D2 dopamine receptor proteins in the dorsal striatum and their synaptic relationships with motor corticostriatal afferents. J. Neurosci. 1995, 15, 5222–5237. [Google Scholar] [CrossRef]
- Alvarez, V.A.; Sabatini, B.L. Anatomical and physiological plasticity of dendritic spines. Annu. Rev. Neurosci. 2007, 30, 79–97. [Google Scholar] [CrossRef]
- Sun, T.; Li, T.; Davies, H.; Li, W.; Yang, J.; Li, S.; Ling, S. Altered Morphologies and Functions of the Olfactory Bulb and Hippocampus Induced by miR-30c. Front. Neurosci. 2016, 10. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Li, W.; Ling, S. miR-30c and semaphorin 3A determine adult neurogenesis by regulating proliferation and differentiation of stem cells in the subventricular zones of mouse. Cell Prolif. 2016, 49, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Alsharafi, W.; Xiao, B. Dynamic Expression of MicroRNAs (183, 135a, 125b, 128, 30c and 27a) in the Rat Pilocarpine Model and Temporal Lobe Epilepsy Patients. CNS Neurol. Disord. Drug Targets 2015, 14, 1096–1102. [Google Scholar] [CrossRef] [PubMed]
- Gu, Q.H.; Yu, D.; Hu, Z.; Liu, X.; Yang, Y.; Luo, Y.; Zhu, J.; Li, Z. miR-26a and miR-384-5p are required for LTP maintenance and spine enlargement. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Ignacio, C.; Mooney, S.M.; Middleton, F.A. Effects of Acute Prenatal Exposure to Ethanol on microRNA Expression are Ameliorated by Social Enrichment. Front. Pediatr. 2014, 2. [Google Scholar] [CrossRef] [PubMed]
- Korpi, E.R.; Den Hollander, B.; Farooq, U.; Vashchinkina, E.; Rajkumar, R.; Nutt, D.J.; Hyytia, P.; Dawe, G.S. Mechanisms of Action and Persistent Neuroplasticity by Drugs of Abuse. Pharmacol. Rev. 2015, 67, 872–1004. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, A.; Dixit, A.B.; Paul, D.; Tripathi, M.; Sarkar, C.; Chandra, P.S.; Banerjee, J. Comparative analysis of cytokine/chemokine regulatory networks in patients with hippocampal sclerosis (HS) and focal cortical dysplasia (FCD). Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Cook, J.L.; Green, C.R.; De la Ronde, S.; Dell, C.A.; Graves, L.; Morgan, L.; Ordean, A.; Ruiter, J.; Steeves, M.; Wong, S. Screening and Management of Substance Use in Pregnancy: A Review. J. Obstet. Gynaecol. Can. 2017, 39, 897–905. [Google Scholar] [CrossRef] [PubMed]
- Horiguchi, H.; Kobune, M.; Kikuchi, S.; Yoshida, M.; Murata, M.; Murase, K.; Iyama, S.; Takada, K.; Sato, T.; Ono, K.; et al. Extracellular vesicle miR-7977 is involved in hematopoietic dysfunction of mesenchymal stromal cells via poly(rC) binding protein 1 reduction in myeloid neoplasms. Haematologica 2016, 101, 437–447. [Google Scholar] [CrossRef] [PubMed]
- Godlewski, J.; Nowicki, M.O.; Bronisz, A.; Nuovo, G.; Palatini, J.; De Lay, M.; Van Brocklyn, J.; Ostrowski, M.C.; Chiocca, E.A.; Lawler, S.E. MicroRNA-451 regulates LKB1/AMPK signaling and allows adaptation to metabolic stress in glioma cells. Mol. Cell 2010, 37, 620–632. [Google Scholar] [CrossRef]
- Tantawy, M.; Elzayat, M.G.; Yehia, D.; Taha, H. Identification of microRNA signature in different pediatric brain tumors. Genet. Mol. Biol. 2018, 41, 27–34. [Google Scholar] [CrossRef]
- Prins, S.A.; Przybycien-Szymanska, M.M.; Rao, Y.S.; Pak, T.R. Long-term effects of peripubertal binge EtOH exposure on hippocampal microRNA expression in the rat. PLoS ONE 2014, 9, e83166. [Google Scholar] [CrossRef]
- Wu, Y.C.; Chen, C.H.; Mercer, A.; Sokol, N.S. Let-7-complex microRNAs regulate the temporal identity of Drosophila mushroom body neurons via chinmo. Dev. Cell 2012, 23, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Bacaj, T.; Yang, X.; Pang, Z.P.; Sudhof, T.C. Lipid-anchored SNAREs lacking transmembrane regions fully support membrane fusion during neurotransmitter release. Neuron 2013, 80, 470–483. [Google Scholar] [CrossRef] [PubMed]
- Caygill, E.E.; Johnston, L.A. Temporal regulation of metamorphic processes in Drosophila by the let-7 and miR-125 heterochronic microRNAs. Curr. Biol. 2008, 18, 943–950. [Google Scholar] [CrossRef] [PubMed]
- Edbauer, D.; Neilson, J.R.; Foster, K.A.; Wang, C.F.; Seeburg, D.P.; Batterton, M.N.; Tada, T.; Dolan, B.M.; Sharp, P.A.; Sheng, M. Regulation of synaptic structure and function by FMRP-associated microRNAs miR-125b and miR-132. Neuron 2010, 65, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Lv, J.; Zeng, Y.; Qian, Y.; Dong, J.; Zhang, Z.; Zhang, J. MicroRNA let-7c-5p improves neurological outcomes in a murine model of traumatic brain injury by suppressing neuroinflammation and regulating microglial activation. Brain Res. 2018, 1685, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Yang, Q.; Wei, X.; Liu, Y.; Ma, D.; Li, J.; Wan, Y.; Luo, Y. The role of miR-190a-5p contributes to diabetic neuropathic pain via targeting SLC17A6. J. Pain Res. 2017, 10, 2395–2403. [Google Scholar] [CrossRef] [PubMed]
- Boulland, J.L.; Qureshi, T.; Seal, R.P.; Rafiki, A.; Gundersen, V.; Bergersen, L.H.; Fremeau, R.T., Jr.; Edwards, R.H.; Storm-Mathisen, J.; Chaudhry, F.A. Expression of the vesicular glutamate transporters during development indicates the widespread corelease of multiple neurotransmitters. J. Comp. Neurol. 2004, 480, 264–280. [Google Scholar] [CrossRef]
- Fremeau, R.T., Jr.; Kam, K.; Qureshi, T.; Johnson, J.; Copenhagen, D.R.; Storm-Mathisen, J.; Chaudhry, F.A.; Nicoll, R.A.; Edwards, R.H. Vesicular glutamate transporters 1 and 2 target to functionally distinct synaptic release sites. Science 2004, 304, 1815–1819. [Google Scholar] [CrossRef]
- Herzog, E.; Takamori, S.; Jahn, R.; Brose, N.; Wojcik, S.M. Synaptic and vesicular co-localization of the glutamate transporters VGLUT1 and VGLUT2 in the mouse hippocampus. J. Neurochem. 2006, 99, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Watakabe, A.; Hioki, H.; Fujiyama, F.; Tanaka, Y.; Yamamori, T.; Kaneko, T. Transiently increased colocalization of vesicular glutamate transporters 1 and 2 at single axon terminals during postnatal development of mouse neocortex: A quantitative analysis with correlation coefficient. Eur. J. Neurosci. 2007, 26, 3054–3067. [Google Scholar] [CrossRef] [PubMed]
- Shan, L.; Ma, D.; Zhang, C.; Xiong, W.; Zhang, Y. miRNAs may regulate GABAergic transmission associated genes in aged rats with anesthetics-induced recognition and working memory dysfunction. Brain Res. 2017, 1670, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wu, Y.; Huang, Q.; Zou, D.; Qin, W.; Chen, Z. Grouping Pentylenetetrazol-Induced Epileptic Rats According to Memory Impairment and MicroRNA Expression Profiles in the Hippocampus. PLoS ONE 2015, 10, e0126123. [Google Scholar] [CrossRef] [PubMed]
- Augustin, R.; Endres, K.; Reinhardt, S.; Kuhn, P.H.; Lichtenthaler, S.F.; Hansen, J.; Wurst, W.; Trumbach, D. Computational identification and experimental validation of microRNAs binding to the Alzheimer-related gene ADAM10. BMC Med. Genet. 2012, 13, 35. [Google Scholar] [CrossRef]
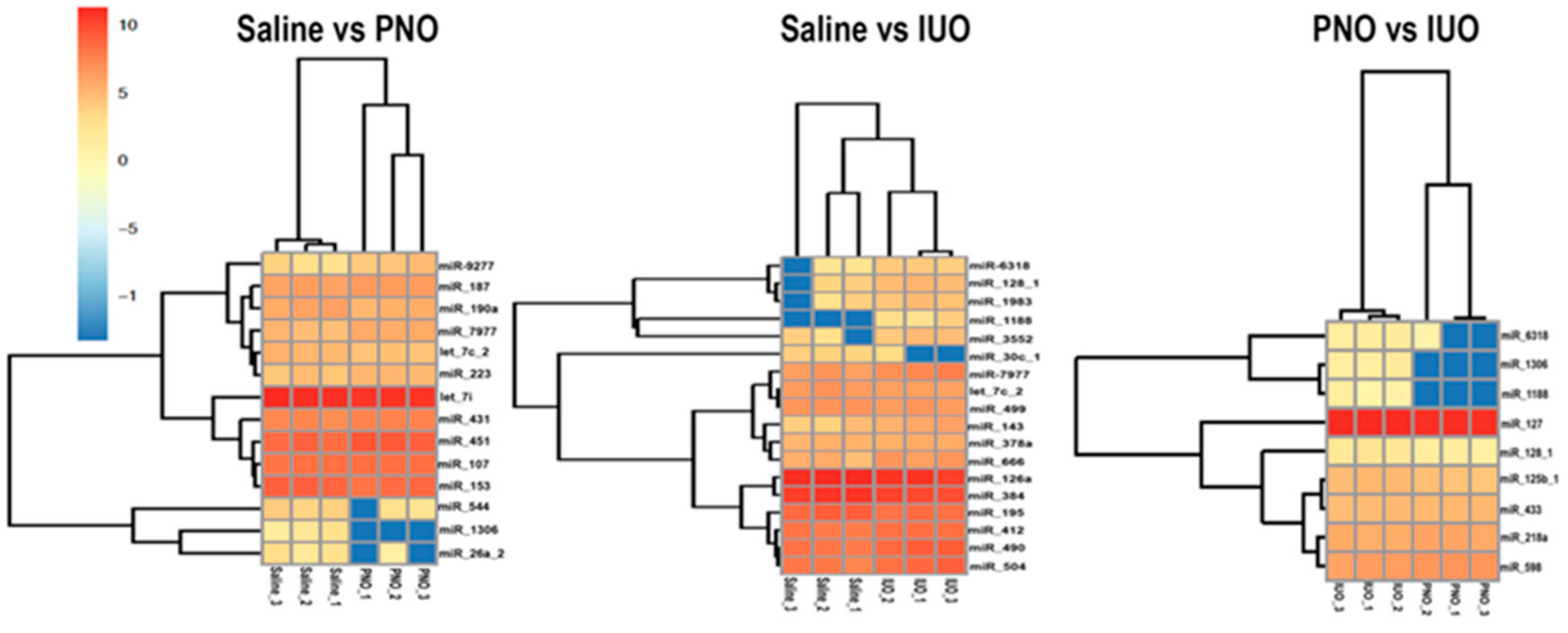
| PNO vs. Control | ||||
| miRNA | Seed sequence | up/down | Fold Change (PNO/Control) | p-value (student’s T-test) |
| rno-miR-1306-5p | CCACCTCCCCTGCAAACGTCCA | down | -inf | 0.018 |
| mmu-miR-26a-2-3p_1ss4GA | CCTATTCTTGATTACTTGTTTC | down | 0.09 | 0.021 |
| rno-miR-544-3p_R+1 | ATTCTGCATTTTTAGCAAGCTT | down | 0.29 | 0.018 |
| rno-let-7c-2-3p | CTATACAATCTACTGTCTTTCC | down | 0.64 | 0.015 |
| rno-miR-190a-5p_R+1 | TGATATGTTTGATATATTAGGTT | down | 0.66 | 0.048 |
| rno-miR-451-5p | AAACCGTTACCATTACTGAGTT | up | 1.59 | 0.035 |
| hsa-miR-7977_1ss6AG | TTCCCGGCCAACGCACCA | up | 1.88 | 0.006 |
| efu-mir-9277-p3_1ss9AT | CAGGTTCGTATCCTGCCGACT | up | 2.64 | 0.023 |
| IUO vs. Control | ||||
| miRNA | Seed sequence | up/down | Fold Change (IUO/Control) | p-value (student’s T-test) |
| rno-miR-30c-1-3p | CTGGGAGAGGGTTGTTTACTCC | down | 0.23 | 0.039 |
| rno-miR-384-5p | TGTAAACAATTCCTAGGCAATGT | down | 0.63 | 0.013 |
| rno-miR-195-5p_R+1 | TAGCAGCACAGAAATATTGGCA | down | 0.64 | 0.047 |
| rno-miR-504_L-1R+1 | GACCCTGGTCTGCACTCTGTCT | up | 1.84 | 0.039 |
| rno-miR-490-3p_R+1 | CAACCTGGAGGACTCCATGCTGT | up | 1.93 | 0.016 |
| hsa-miR-7977_1ss6AG | TTCCCGGCCAACGCACCA | up | 1.98 | 0.044 |
| rno-miR-666-5p_R+4 | AGCGGGCACGGCTGTGAGAGCCCC | up | 2.23 | 0.014 |
| rno-miR-143-5p_R+1 | GGTGCAGTGCTGCATCTCTGGT | up | 2.76 | 0.024 |
| rno-miR-128-1-5p_R+2 | CGGGGCCGTAGCACTGTCTGAGA | up | 3.19 | 0.019 |
| mmu-mir-1983-p5_1ss1GA | AAAAGCATGCTCCAGTGGCGC | up | 3.23 | 0.037 |
| rno-miR-3552 | AGGCTGCAGGCCCACTTCCCT | up | 3.32 | 0.037 |
| rno-miR-6318_R+1 | CTGCCTGGCGCAGGGCCTGTAGT | up | 4.60 | 0.003 |
| rno-miR-1188-3p_L+2R+5 | TCCGAGGCTCCCCACCACACCCTG | up | inf | 0.012 |
| IUO vs. PNO | ||||
| miRNA | Seed sequence | up/down | Fold Change (IUO/PNO) | p-value (student’s T-test) |
| rno-miR-128-1-5p_R+2 | CGGGGCCGTAGCACTGTCTGAGA | up | 2.16 | 0.005 |
| rno-miR-6318_R+1 | CTGCCTGGCGCAGGGCCTGTAGT | up | 6.29 | 0.007 |
| rno-miR-1306-5p | CCACCTCCCCTGCAAACGTCCA | up | inf | 0.001 |
| rno-miR-1188-3p_L+2R+5 | TCCGAGGCTCCCCACCACACCCTG | up | inf | 0.012 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shahjin, F.; Guda, R.S.; Schaal, V.L.; Odegaard, K.; Clark, A.; Gowen, A.; Xiao, P.; Lisco, S.J.; Pendyala, G.; Yelamanchili, S.V. Brain-Derived Extracellular Vesicle microRNA Signatures Associated with In Utero and Postnatal Oxycodone Exposure. Cells 2020, 9, 21. https://doi.org/10.3390/cells9010021
Shahjin F, Guda RS, Schaal VL, Odegaard K, Clark A, Gowen A, Xiao P, Lisco SJ, Pendyala G, Yelamanchili SV. Brain-Derived Extracellular Vesicle microRNA Signatures Associated with In Utero and Postnatal Oxycodone Exposure. Cells. 2020; 9(1):21. https://doi.org/10.3390/cells9010021
Chicago/Turabian StyleShahjin, Farah, Rahul S. Guda, Victoria L. Schaal, Katherine Odegaard, Alexander Clark, Austin Gowen, Peng Xiao, Steven J. Lisco, Gurudutt Pendyala, and Sowmya V. Yelamanchili. 2020. "Brain-Derived Extracellular Vesicle microRNA Signatures Associated with In Utero and Postnatal Oxycodone Exposure" Cells 9, no. 1: 21. https://doi.org/10.3390/cells9010021
APA StyleShahjin, F., Guda, R. S., Schaal, V. L., Odegaard, K., Clark, A., Gowen, A., Xiao, P., Lisco, S. J., Pendyala, G., & Yelamanchili, S. V. (2020). Brain-Derived Extracellular Vesicle microRNA Signatures Associated with In Utero and Postnatal Oxycodone Exposure. Cells, 9(1), 21. https://doi.org/10.3390/cells9010021







