Interaction of Pregnancy-Specific Glycoprotein 1 With Integrin α5β1 Is a Modulator of Extravillous Trophoblast Functions
Abstract
1. Introduction
2. Materials and Methods
2.1. Protein Production and Purification
2.2. Cell Lines
2.3. Placental Tissue Collection and Primary Cells Isolation
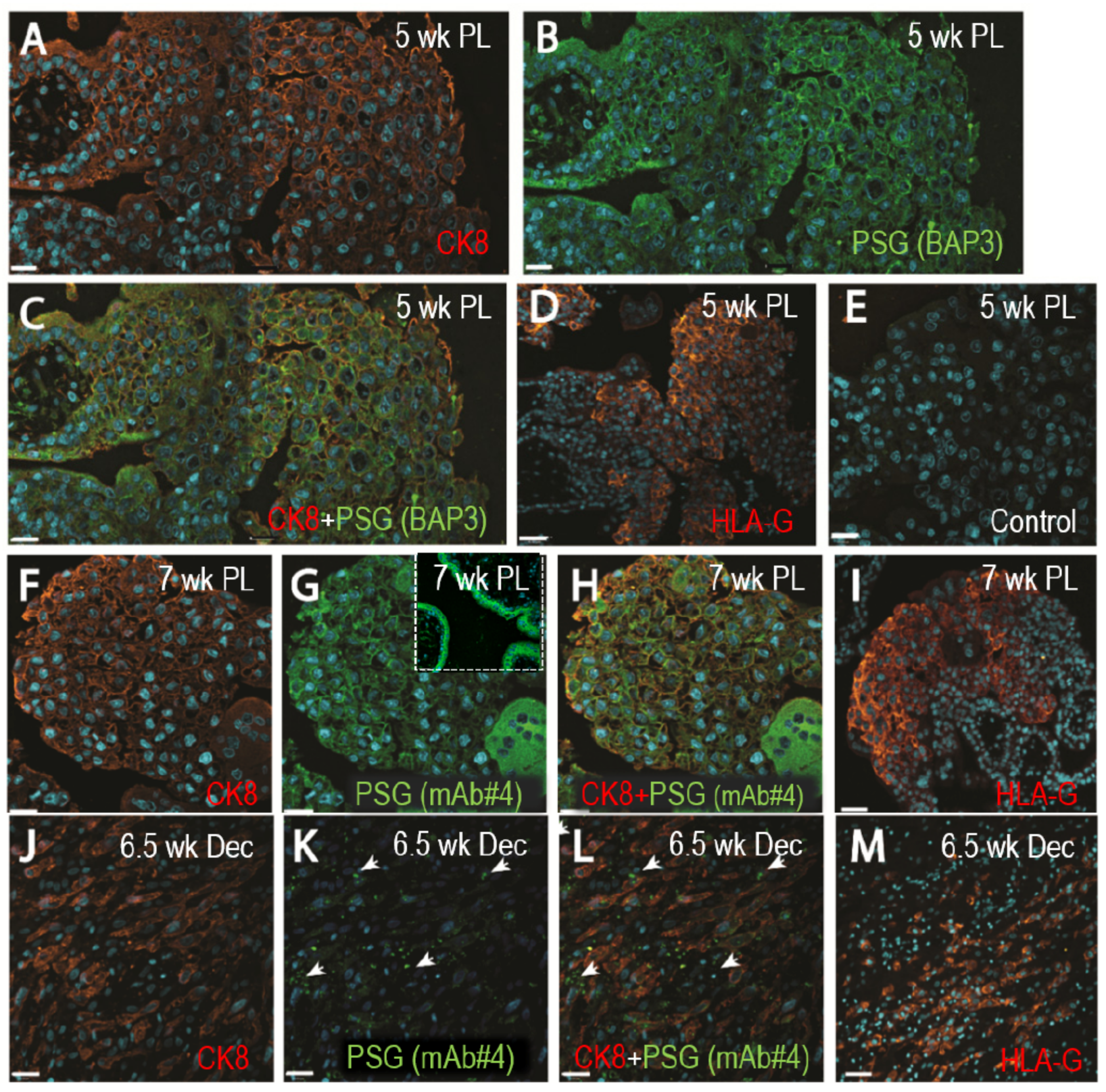
2.4. Fluorescence Immunohistochemistry for Analysis of PSGs Expression and Localization with Integrin α5 in First-Trimester Placentas
2.5. Immunoblot Analysis of PSG1 Expression in Second-Trimester Trophoblasts
2.6. Cell Adhesion
2.7. Integrin Binding ELISA
2.8. FAK Phosphorylation Analysis by Western Blot
2.9. Focal Adhesion Structure Analysis by Fluorescence Microscopy
2.10. PSG1-ECM Binding Assay
2.11. Wound Healing Assay
2.12. Tracking of Single Cell Migration
2.13. HIF1α Stabilization Analysis by Western Blot
2.14. Flow Cytometry
2.15. Invasion Assays
2.16. Determination of PSG1 Concentration in Serum of Pregnant Women
3. Results
3.1. Expression of PSGs in EVTs
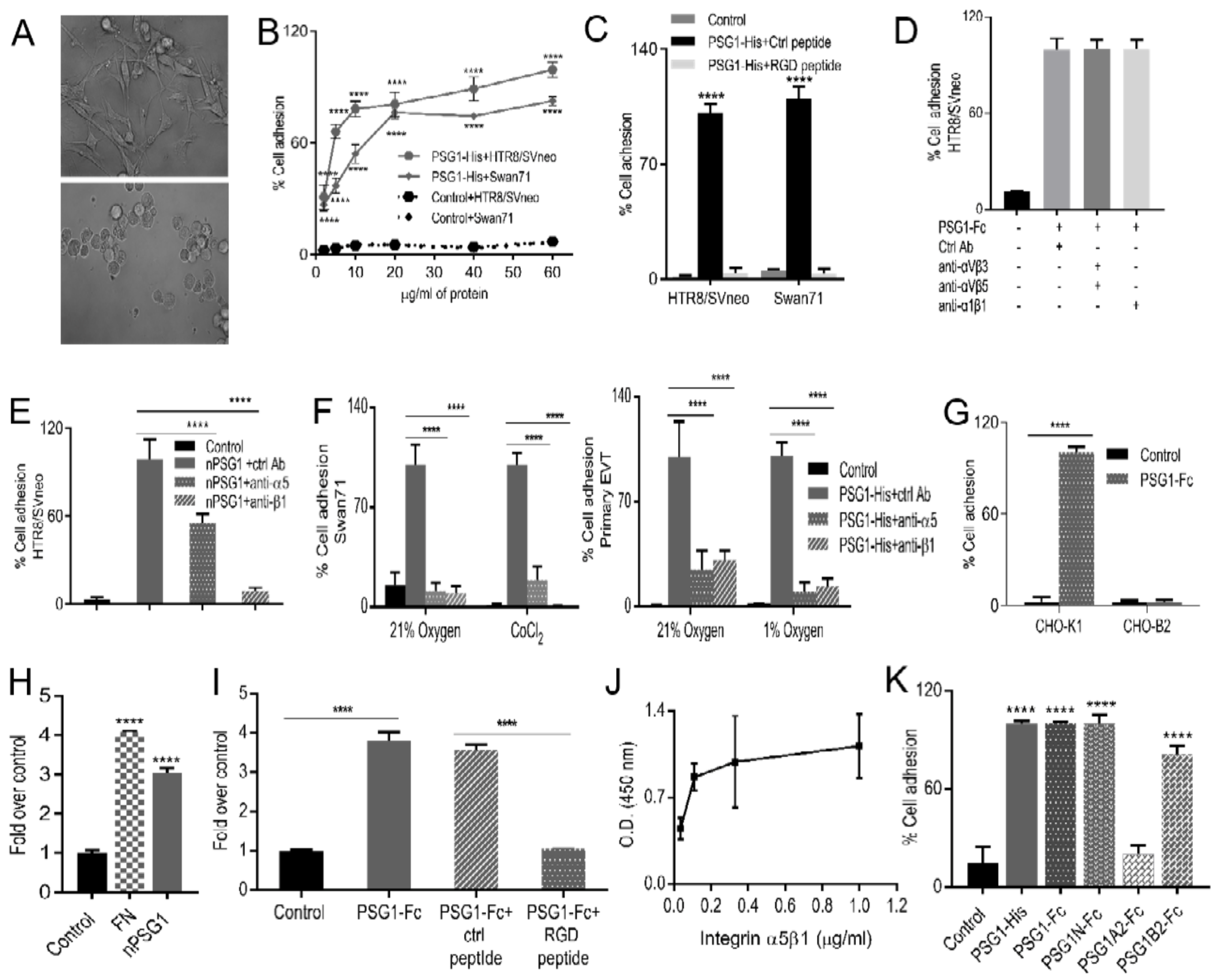
3.2. Immobilized PSG1 Induces Adhesion of EVTs by Binding to Integrin α5β1
3.3. More Than One Domain of PSG1 Mediates EVT Adhesion
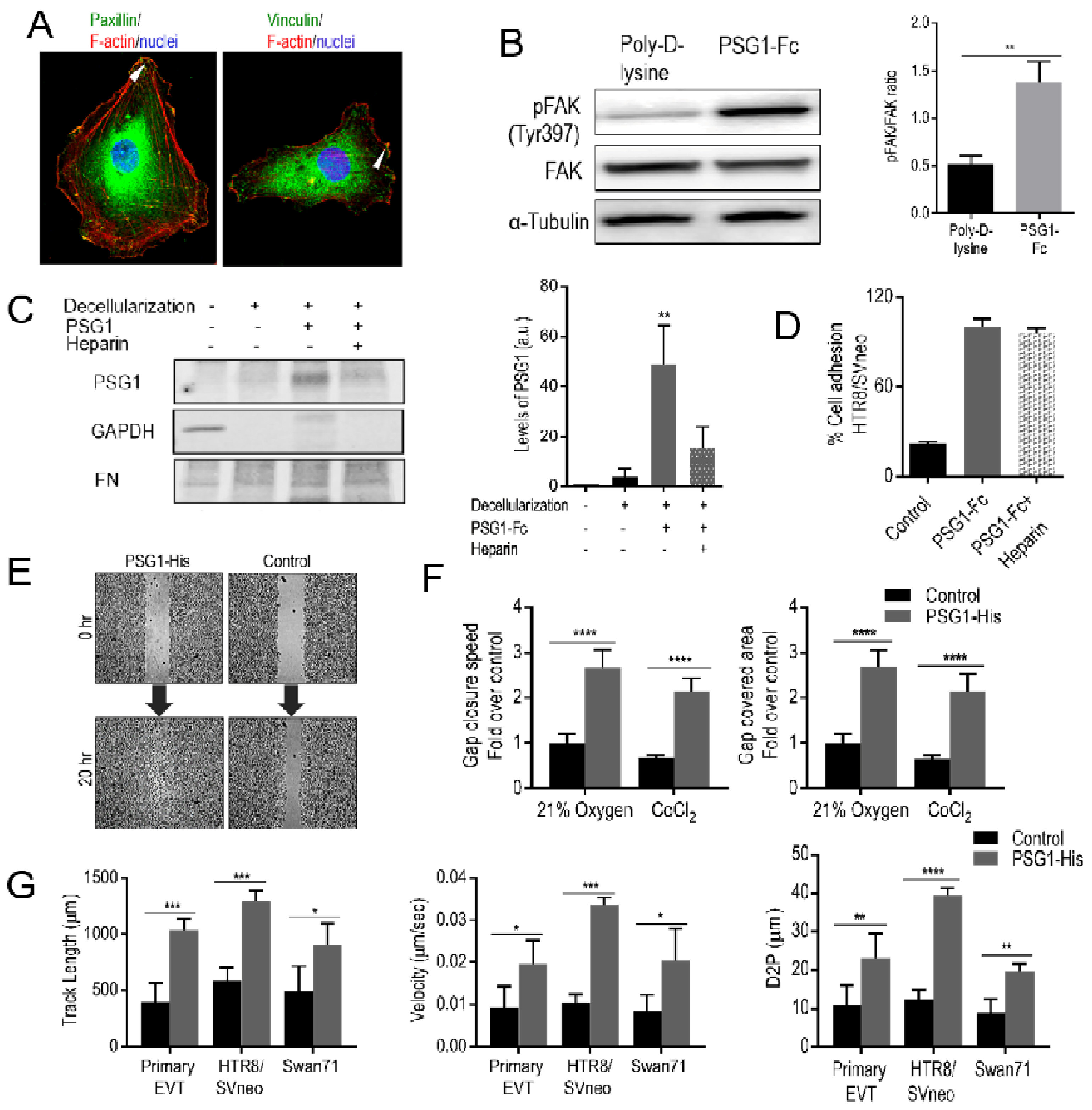
3.4. PSG1 Induces Focal Adhesion (FA) Structures and Focal Adhesion Kinase (FAK) Phosphorylation
3.5. PSG1 Binds to HSPGs in the ECM and can Concurrently Bind to Integrin α5β1 on the Cell Surface
3.6. PSG1 Increases EVT Migration
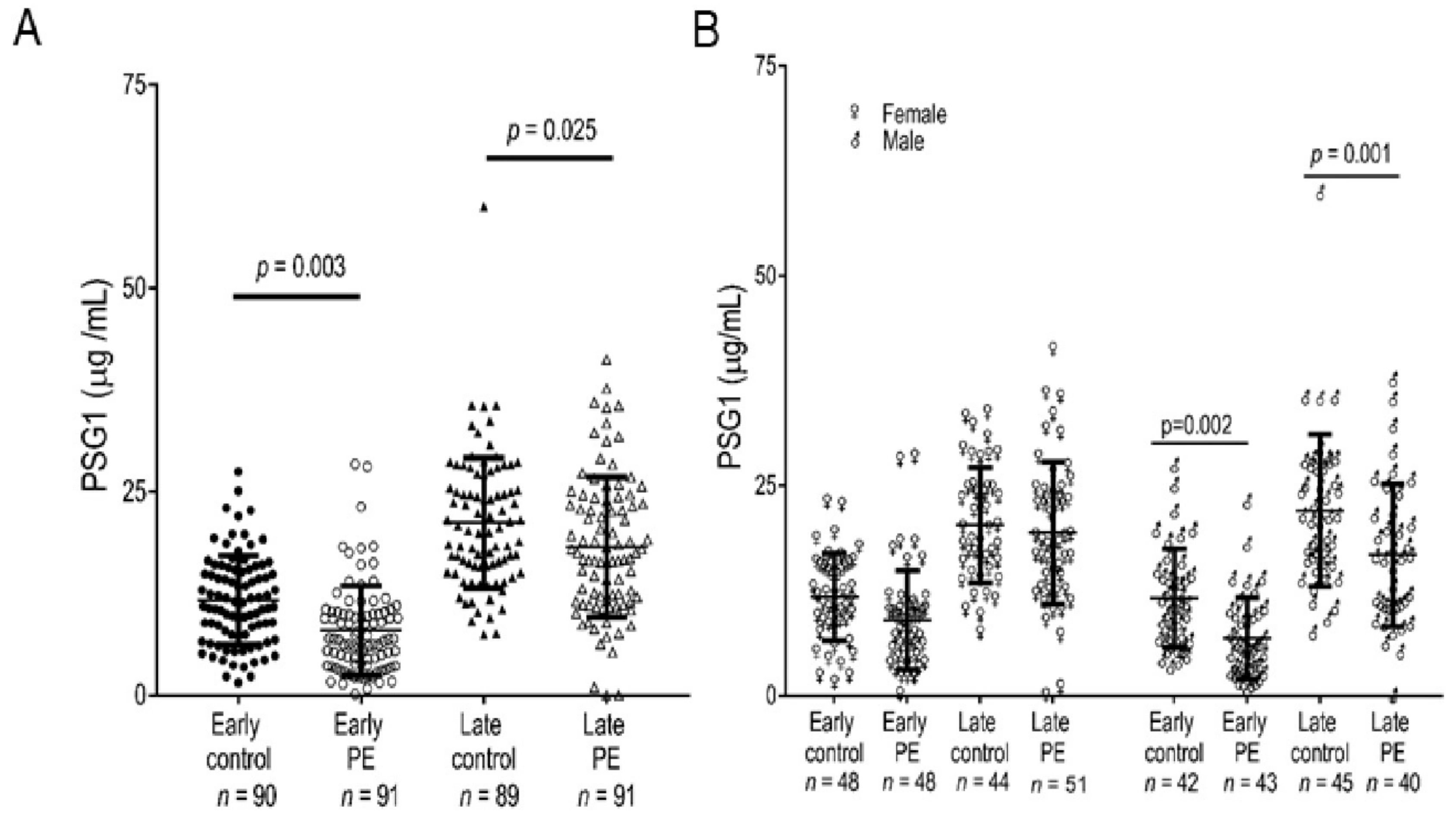
3.7. The PSG1 Concentration Is Significantly Reduced in Pregnant Women Diagnosed with Early-Onset and Late-Onset PE
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A

References
- Zhou, G.Q.; Hammarstrom, S. Pregnancy-specific glycoprotein (PSG) in baboon (Papio hamadryas): Family size, domain structure, and prediction of a functional region in primate PSGs. Biol. Reprod. 2001, 64, 90–99. [Google Scholar] [CrossRef][Green Version]
- Kammerer, R.; Mansfeld, M.; Hanske, J.; Missbach, S.; He, X.; Kollner, B.; Mouchantat, S.; Zimmermann, W. Recent expansion and adaptive evolution of the carcinoembryonic antigen family in bats of the Yangochiroptera subgroup. BMC Genom. 2017, 18, 717. [Google Scholar] [CrossRef]
- Sorensen, S. Pregnancy-“specific” beta 1-glycoprotein (SP1): Purification, characterization, quantification and clinical application in malignancies (a review). Tumour Biol. 1984, 5, 275–302. [Google Scholar]
- Bohn, H. Detection and characterization of pregnancy proteins in the human placenta and their quantitative immunochemical determination in sera from pregnant women. Arch. Gynakol. 1971, 210, 440–457. [Google Scholar] [CrossRef]
- Kammerer, R.; Zimmermann, W. Coevolution of activating and inhibitory receptors within mammalian carcinoembryonic antigen families. BMC Biol. 2010, 8, 12. [Google Scholar] [CrossRef]
- Moore, T.; Dveksler, G.S. Pregnancy-specific glycoproteins: Complex gene families regulating maternal-fetal interactions. Int. J. Dev. Biol. 2014, 58, 273–280. [Google Scholar] [CrossRef]
- McLellan, A.S.; Fischer, B.; Dveksler, G.; Hori, T.; Wynne, F.; Ball, M.; Okumura, K.; Moore, T.; Zimmermann, W. Structure and evolution of the mouse pregnancy-specific glycoprotein (Psg) gene locus. BMC Genom. 2005, 6, 4. [Google Scholar] [CrossRef]
- Teglund, S.; Olsen, A.; Khan, W.N.; Frangsmyr, L.; Hammarstrom, S. The pregnancy-specific glycoprotein (PSG) gene cluster on human chromosome 19: Fine structure of the 11 PSG genes and identification of 6 new genes forming a third subgroup within the carcinoembryonic antigen (CEA) family. Genomics 1994, 23, 669–684. [Google Scholar] [CrossRef]
- Shanley, D.K.; Kiely, P.A.; Golla, K.; Allen, S.; Martin, K.; O’Riordan, R.T.; Ball, M.; Aplin, J.D.; Singer, B.B.; Caplice, N.; et al. Pregnancy-specific glycoproteins bind integrin alphaIIbbeta3 and inhibit the platelet-fibrinogen interaction. PLoS ONE 2013, 8, e57491. [Google Scholar] [CrossRef]
- Wurz, H.; Geiger, W.; Kunzig, H.J.; Jabs-Lehmann, A.; Bohn, H.; Luben, G. Radioimmunoassay of SP1 (pregnancy-specific beta1-glycoprotein) in maternal blood and in amniotic fluid normal and pathologic pregnancies. J. Perinat. Med. 1981, 9, 67–78. [Google Scholar] [CrossRef][Green Version]
- McLellan, A.S.; Zimmermann, W.; Moore, T. Conservation of pregnancy-specific glycoprotein (PSG) N domains following independent expansions of the gene families in rodents and primates. BMC Evol. Biol. 2005, 5, 39. [Google Scholar] [CrossRef]
- Jones, K.; Ballesteros, A.; Mentink-Kane, M.; Warren, J.; Rattila, S.; Malech, H.; Kang, E.; Dveksler, G. PSG9 stimulates increase in FoxP3+ regulatory T-Cells through the TGF-beta1 pathway. PLoS ONE 2016, 11, e0158050. [Google Scholar] [CrossRef]
- Warren, J.; Im, M.; Ballesteros, A.; Ha, C.; Moore, T.; Lambert, F.; Lucas, S.; Hinz, B.; Dveksler, G. Activation of latent transforming growth factor-beta1, a conserved function for pregnancy-specific beta 1-glycoproteins. Mol. Hum. Reprod. 2018, 24, 602–612. [Google Scholar] [CrossRef]
- Snyder, S.K.; Wessner, D.H.; Wessells, J.L.; Waterhouse, R.M.; Wahl, L.M.; Zimmermann, W.; Dveksler, G.S. Pregnancy-Specific glycoproteins function as immunomodulators by inducing secretion of IL-10, IL-6 and TGF-beta1 by human monocytes. Am. J. Reprod. Immunol. 2001, 45, 205–216. [Google Scholar] [CrossRef]
- Motran, C.C.; Diaz, F.L.; Montes, C.L.; Bocco, J.L.; Gruppi, A. In vivo expression of recombinant pregnancy-specific glycoprotein 1a induces alternative activation of monocytes and enhances Th2-type immune response. Eur. J. Immunol. 2003, 33, 3007–3016. [Google Scholar] [CrossRef]
- Blois, S.M.; Sulkowski, G.; Tirado-Gonzalez, I.; Warren, J.; Freitag, N.; Klapp, B.F.; Rifkin, D.; Fuss, I.; Strober, W.; Dveksler, G.S. Pregnancy-specific glycoprotein 1 (PSG1) activates TGF-beta and prevents dextran sodium sulfate (DSS)-induced colitis in mice. Mucosal Immunol. 2014, 7, 348–358. [Google Scholar] [CrossRef]
- Lisboa, F.A.; Warren, J.; Sulkowski, G.; Aparicio, M.; David, G.; Zudaire, E.; Dveksler, G.S. Pregnancy-specific glycoprotein 1 induces endothelial tubulogenesis through interaction with cell surface proteoglycans. J. Biol. Chem. 2011, 286, 7577–7586. [Google Scholar] [CrossRef]
- Ha, C.T.; Waterhouse, R.; Warren, J.; Zimmermann, W.; Dveksler, G.S. N-Glycosylation is required for binding of murine pregnancy-specific glycoproteins 17 and 19 to the receptor CD9. Am. J. Reprod. Immunol. 2008, 59, 251–258. [Google Scholar] [CrossRef]
- Houston, A.; Williams, J.M.; Rovis, T.L.; Shanley, D.K.; O’Riordan, R.T.; Kiely, P.A.; Ball, M.; Barry, O.P.; Kelly, J.; Fanning, A.; et al. Pregnancy-Specific glycoprotein expression in normal gastrointestinal tract and in tumors detected with novel monoclonal antibodies. MAbs 2016, 8, 491–500. [Google Scholar] [CrossRef]
- Shupert, W.L.; Chan, W.Y. Pregnancy specific beta 1-glycoprotein in human intestine. Mol. Cell Biochem. 1993, 120, 159–170. [Google Scholar] [CrossRef]
- Lee, J.N.; Salem, H.T.; Al-Ani, A.T.; Chard, T.; Huang, S.C.; Ouyang, P.C.; Wei, P.Y.; Seppala, M. Circulating concentrations of specific placental proteins (human chorionic gonadotropin, pregnancy-specific beta-1 glycoprotein, and placental protein 5) in untreated gestational trophoblastic tumors. Am. J. Obstet. Gynecol. 1981, 139, 702–704. [Google Scholar] [CrossRef]
- Campo, E.; Algaba, F.; Palacin, A.; Germa, R.; Sole-Balcells, F.J.; Cardesa, A. Placental proteins in high-grade urothelial neoplasms. An immunohistochemical study of human chorionic gonadotropin, human placental lactogen, and pregnancy-specific beta-1-glycoprotein. Cancer 1989, 63, 2497–2504. [Google Scholar] [CrossRef]
- Salahshor, S.; Goncalves, J.; Chetty, R.; Gallinger, S.; Woodgett, J.R. Differential gene expression profile reveals deregulation of pregnancy specific beta1 glycoprotein 9 early during colorectal carcinogenesis. BMC Cancer 2005, 5, 66. [Google Scholar] [CrossRef]
- Kawano, K.; Ebisawa, M.; Hase, K.; Fukuda, S.; Hijikata, A.; Kawano, S.; Date, Y.; Tsuneda, S.; Itoh, K.; Ohno, H. Psg18 is specifically expressed in follicle-associated epithelium. Cell Struct. Funct. 2007, 32, 115–126. [Google Scholar] [CrossRef]
- Maltepe, E.; Fisher, S.J. Placenta: The forgotten organ. Annu. Rev. Cell Dev. Biol. 2015, 31, 523–552. [Google Scholar] [CrossRef]
- Chang, C.W.; Wakeland, A.K.; Parast, M.M. Trophoblast lineage specification, differentiation and their regulation by oxygen tension. J. Endocrinol. 2018, 236, R43–R56. [Google Scholar] [CrossRef]
- Tarrade, A.; Lai Kuen, R.; Malassiné, A.; Tricottet, V.; Blain, P.; Vidaud, M.; Evain-Brion, D. Characterization of human villous and extravillous trophoblasts isolated from first trimester placenta. Lab. Investig. 2001, 81. [Google Scholar] [CrossRef]
- Knofler, M.; Haider, S.; Saleh, L.; Pollheimer, J.; Gamage, T.; James, J. Human placenta and trophoblast development: Key molecular mechanisms and model systems. Cell Mol. Life Sci. 2019. [Google Scholar] [CrossRef]
- Pollheimer, J.; Vondra, S.; Baltayeva, J.; Beristain, A.G.; Knöfler, M. Regulation of placental extravillous trophoblasts by the maternal uterine environment. Front Immunol. 2018, 9. [Google Scholar] [CrossRef]
- Pujal, J.M.; Roura, S.; Munoz-Marmol, A.M.; Mate, J.L.; Bayes-Genis, A. Fetal-maternal interface: A chronicle of allogeneic coexistence. Chimerism 2012, 3, 18–20. [Google Scholar] [CrossRef][Green Version]
- Gleeson, L.M.; Chakraborty, C.; McKinnon, T.; Lala, P.K. Insulin-Like growth factor-binding protein 1 stimulates human trophoblast migration by signaling through alpha 5 beta 1 integrin via mitogen-activated protein Kinase pathway. J. Clin. Endocrinol. Metab. 2001, 86, 2484–2493. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhou, G.Q.; Baranov, V.; Zimmermann, W.; Grunert, F.; Erhard, B.; Mincheva-Nilsson, L.; Hammarstrom, S.; Thompson, J. Highly specific monoclonal antibody demonstrates that pregnancy-specific glycoprotein (PSG) is limited to syncytiotrophoblast in human early and term placenta. Placenta 1997, 18, 491–501. [Google Scholar] [CrossRef]
- Gormley, M.; Ona, K.; Kapidzic, M.; Garrido-Gomez, T.; Zdravkovic, T.; Fisher, S.J. Preeclampsia: Novel insights from global RNA profiling of trophoblast subpopulations. Am. J. Obstet. Gynecol. 2017, 217. [Google Scholar] [CrossRef] [PubMed]
- Okae, H.; Toh, H.; Sato, T.; Hiura, H.; Takahashi, S.; Shirane, K.; Kabayama, Y.; Suyama, M.; Sasaki, H.; Arima, T. Derivation of human trophoblast stem cells. Cell Stem Cell 2018, 22, 50–63. [Google Scholar] [CrossRef] [PubMed]
- Blois, S.; Tirado-Gonzalez, I.; Wu, J.; Barrientos, G.; Johnson, B.; Warren, J.; Freitag, N.; Klapp, B.; Irmak, S.; Ergun, S.; et al. Early expression of pregnancy-specific glycoprotein 22 (PSG22) by trophoblast cells modulates angiogenesis in mice. Biol. Reprod. 2012, 86, 1–9. [Google Scholar] [CrossRef]
- Sulkowski, G.N.; Warren, J.; Ha, C.T.; Dveksler, G.S. Characterization of receptors for murine pregnancy specific glycoproteins 17 and 23. Placenta 2011, 32, 603–610. [Google Scholar] [CrossRef]
- Damsky, C.H.; Librach, C.; Lim, K.H.; Fitzgerald, M.L.; McMaster, M.T.; Janatpour, M.; Zhou, Y.; Logan, S.K.; Fisher, S.J. Integrin switching regulates normal trophoblast invasion. Development 1994, 120, 3657–3666. [Google Scholar]
- Korhonen, M.; Ylanne, J.; Laitinen, L.; Cooper, H.M.; Quaranta, V.; Virtanen, I. Distribution of the alpha 1-alpha 6 integrin subunits in human developing and term placenta. Lab. Investig. 1991, 65, 347–356. [Google Scholar]
- Aplin, J.D. Expression of integrin alpha 6 beta 4 in human trophoblast and its loss from extravillous cells. Placenta 1993, 14, 203–215. [Google Scholar] [CrossRef]
- Silva, J.F.; Serakides, R. Intrauterine trophoblast migration: A comparative view of humans and rodents. Cell Adh. Migr. 2016, 10, 88–110. [Google Scholar] [CrossRef]
- Bronson, R.A.; Fusi, F.M. Integrins and human reproduction. Mol. Hum. Reprod. 1996, 2, 153–168. [Google Scholar] [CrossRef] [PubMed]
- Bilban, M.; Haslinger, P.; Prast, J.; Klinglmuller, F.; Woelfel, T.; Haider, S.; Sachs, A.; Otterbein, L.E.; Desoye, G.; Hiden, U.; et al. Identification of novel trophoblast invasion-related genes: Heme oxygenase-1 controls motility via peroxisome proliferator-activated receptor gamma. Endocrinology 2009, 150, 1000–1013. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Damsky, C.H.; Chiu, K.; Roberts, J.M.; Fisher, S.J. Preeclampsia is associated with abnormal expression of adhesion molecules by invasive cytotrophoblasts. J. Clin. Investig. 1993, 91, 950–960. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros, A.; Mentink-Kane, M.M.; Warren, J.; Kaplan, G.G.; Dveksler, G.S. Induction and activation of latent transforming growth factor-beta1 are carried out by two distinct domains of pregnancy-specific glycoprotein 1 (PSG1). J. Biol. Chem. 2015, 290, 4422–4431. [Google Scholar] [CrossRef]
- Cohen, M.; Ribaux, P.; Epiney, M.; Irion, O. Role of prostate apoptosis response 4 in translocation of GRP78 from the endoplasmic reticulum to the cell surface of trophoblastic cells. PLoS ONE 2013, 8, e80231. [Google Scholar] [CrossRef]
- Fisher, S.J.; Cui, T.Y.; Zhang, L.; Hartman, L.; Grahl, K.; Zhang, G.Y.; Tarpey, J.; Damsky, C.H. Adhesive and degradative properties of human placental cytotrophoblast cells in vitro. J. Cell Biol. 1989, 109, 891–902. [Google Scholar] [CrossRef]
- Zhou, Y.; Fisher, S.J.; Janatpour, M.; Genbacev, O.; Dejana, E.; Wheelock, M.; Damsky, C.H. Human cytotrophoblasts adopt a vascular phenotype as they differentiate. A strategy for successful endovascular invasion? J. Clin. Investig. 1997, 99, 2139–2151. [Google Scholar] [CrossRef]
- Akiyama, S.K.; Yamada, S.S.; Chen, W.T.; Yamada, K.M. Analysis of fibronectin receptor function with monoclonal antibodies: Roles in cell adhesion, migration, matrix assembly, and cytoskeletal organization. J. Cell Biol. 1989, 109, 863–875. [Google Scholar] [CrossRef]
- Yamada, K.M.; Kennedy, D.W.; Yamada, S.S.; Gralnick, H.; Chen, W.T.; Akiyama, S.K. Monoclonal antibody and synthetic peptide inhibitors of human tumor cell migration. Cancer Res. 1990, 50, 4485–4496. [Google Scholar]
- ACOG Committee on Obstetric Practice. ACOG practice bulletin. Diagnosis and management of preeclampsia and eclampsia. Obstet. Gynecol. 2002, 99, 159–167. [Google Scholar]
- Von Dadelszen, P.; Magee, L.A.; Roberts, J.M. Subclassification of preeclampsia. Hypertens. Pregnancy 2003, 22, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Harris, L.K.; Jones, C.J.; Aplin, J.D. Adhesion molecules in human trophoblast—A review. II. extravillous trophoblast. Placenta 2009, 30, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Jauniaux, E.; Watson, A.L.; Hempstock, J.; Bao, Y.P.; Skepper, J.N.; Burton, G.J. Onset of maternal arterial blood flow and placental oxidative stress. A possible factor in human early pregnancy failure. Am. J. Pathol. 2000, 157, 2111–2122. [Google Scholar] [CrossRef]
- Rodesch, F.; Simon, P.; Donner, C.; Jauniaux, E. Oxygen measurements in endometrial and trophoblastic tissues during early pregnancy. Obstet. Gynecol. 1992, 80, 283–285. [Google Scholar] [PubMed]
- Yamanaka-Tatematsu, M.; Nakashima, A.; Fujita, N.; Shima, T.; Yoshimori, T.; Saito, S. Autophagy induced by HIF1alpha overexpression supports trophoblast invasion by supplying cellular energy. PLoS ONE 2013, 8, e76605. [Google Scholar] [CrossRef] [PubMed]
- Arimoto-Ishida, E.; Sakata, M.; Sawada, K.; Nakayama, M.; Nishimoto, F.; Mabuchi, S.; Takeda, T.; Yamamoto, T.; Isobe, A.; Okamoto, Y.; et al. Up-Regulation of alpha5-integrin by E-cadherin loss in hypoxia and its key role in the migration of extravillous trophoblast cells during early implantation. Endocrinology 2009, 150, 4306–4315. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wu, C.; Bauer, J.S.; Juliano, R.L.; McDonald, J.A. The alpha 5 beta 1 integrin fibronectin receptor, but not the alpha 5 cytoplasmic domain, functions in an early and essential step in fibronectin matrix assembly. J. Biol. Chem. 1993, 268, 21883–21888. [Google Scholar]
- Orecchia, A.; Mettouchi, A.; Uva, P.; Simon, G.C.; Arcelli, D.; Avitabile, S.; Ragone, G.; Meneguzzi, G.; Pfenninger, K.H.; Zambruno, G.; et al. Endothelial cell adhesion to soluble vascular endothelial growth factor receptor-1 triggers a cell dynamic and angiogenic phenotype. FASEB J. 2014, 28, 692–704. [Google Scholar] [CrossRef]
- Chen, C.P.; Liu, S.H.; Lee, M.Y.; Chen, Y.Y. Heparan sulfate proteoglycans in the basement membranes of the human placenta and decidua. Placenta 2008, 29, 309–316. [Google Scholar] [CrossRef]
- Sarrazin, S.; Lamanna, W.C.; Esko, J.D. Heparan sulfate proteoglycans. Cold Spring Harb. Perspect. Biol. 2011, 3. [Google Scholar] [CrossRef]
- Irving, J.A.; Lala, P.K. Functional role of cell surface integrins on human trophoblast cell migration: Regulation by TGF-beta, IGF-II, and IGFBP-1. Exp. Cell Res. 1995, 217, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Kabir-Salmani, M. The role of 51-integrin in the IGF-I-induced migration of extravillous trophoblast cells during the process of implantation. Mol. Hum. Reprod. 2004, 10, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Dunk, C.E.; Pappas, J.J.; Lye, P.; Kibschull, M.; Javam, M.; Bloise, E.; Lye, S.J.; Szyf, M.; Matthews, S.G. P-Glycoprotein (P-gp)/ABCB1 plays a functional role in extravillous trophoblast (EVT) invasion and is decreased in the pre-eclamptic placenta. J. Cell Mol. Med. 2018, 22, 5378–5393. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Damsky, C.H.; Fisher, S.J. Preeclampsia is associated with failure of human cytotrophoblasts to mimic a vascular adhesion phenotype. One cause of defective endovascular invasion in this syndrome? J. Clin. Investig. 1997, 99, 2152–2164. [Google Scholar] [CrossRef]
- Aplin, J.D.; Beristain, A.; DaSilva-Arnold, S.; Dunk, C.; Duzyj, C.; Golos, T.G.; Kemmerling, U.; Knofler, M.; Mitchell, M.D.; Olson, D.M.; et al. IFPA meeting 2016 workshop report III: Decidua-trophoblast interactions; trophoblast implantation and invasion; immunology at the maternal-fetal interface; placental inflammation. Placenta 2017. [Google Scholar] [CrossRef]
- Khong, T.Y.; De Wolf, F.; Robertson, W.B.; Brosens, I. Inadequate maternal vascular response to placentation in pregnancies complicated by pre-eclampsia and by small-for-gestational age infants. Br. J. Obstet. Gynaecol. 1986, 93, 1049–1059. [Google Scholar] [CrossRef]
- Kramer, N.; Walzl, A.; Unger, C.; Rosner, M.; Krupitza, G.; Hengstschlager, M.; Dolznig, H. In vitro cell migration and invasion assays. Mutat. Res. 2013, 752, 10–24. [Google Scholar] [CrossRef]
- Pihl, K.; Larsen, T.; Laursen, I.; Krebs, L.; Christiansen, M. First trimester maternal serum pregnancy-specific beta-1-glycoprotein (SP1) as a marker of adverse pregnancy outcome. Prenat. Diagn. 2009, 29, 1256–1261. [Google Scholar] [CrossRef]
- Camolotto, S.; Racca, A.; Rena, V.; Nores, R.; Patrito, L.C.; Genti-Raimondi, S.; Panzetta-Dutari, G.M. Expression and transcriptional regulation of individual pregnancy-specific glycoprotein genes in differentiating trophoblast cells. Placenta 2010, 31, 312–319. [Google Scholar] [CrossRef]
- Bamberger, A.M.; Sudahl, S.; Loning, T.; Wagener, C.; Bamberger, C.M.; Drakakis, P.; Coutifaris, C.; Makrigiannakis, A. The adhesion molecule CEACAM1 (CD66a, C-CAM, BGP) is specifically expressed by the extravillous intermediate trophoblast. Am. J. Pathol. 2000, 156, 1165–1170. [Google Scholar] [CrossRef]
- Shao, L.; Jacobs, A.R.; Johnson, V.V.; Mayer, L. Activation of CD8+ regulatory T cells by human placental trophoblasts. J. Immunol. 2005, 174, 7539–7547. [Google Scholar] [CrossRef] [PubMed]
- Kromer, B.; Finkenzeller, D.; Wessels, J.; Dveksler, G.; Thompson, J.; Zimmermann, W. Coordinate expression of splice variants of the murine pregnancy-specific glycoprotein (PSG) gene family during placental development. Eur. J. Biochem. 1996, 242, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Wynne, F.; Ball, M.; McLellan, A.S.; Dockery, P.; Zimmermann, W.; Moore, T. Mouse pregnancy-specific glycoproteins: Tissue-specific expression and evidence of association with maternal vasculature. Reproduction 2006, 131, 721–732. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Carlson, T.R.; Feng, Y.; Maisonpierre, P.C.; Mrksich, M.; Morla, A.O. Direct cell adhesion to the angiopoietins mediated by integrins. J. Biol. Chem. 2001, 276, 26516–26525. [Google Scholar] [CrossRef]
- Wang, Z.; Collighan, R.J.; Gross, S.R.; Danen, E.H.; Orend, G.; Telci, D.; Griffin, M. RGD-independent cell adhesion via a tissue transglutaminase-fibronectin matrix promotes fibronectin fibril deposition and requires syndecan-4/2 alpha5beta1 integrin co-signaling. J. Biol. Chem. 2010, 285, 40212–40229. [Google Scholar] [CrossRef]
- Pankov, R.; Yamada, K.M. Fibronectin at a glance. J. Cell Sci. 2002, 115, 3861–3863. [Google Scholar] [CrossRef]
- Huppertz, B.; Gauster, M.; Orendi, K.; Konig, J.; Moser, G. Oxygen as modulator of trophoblast invasion. J. Anat. 2009, 215, 14–20. [Google Scholar] [CrossRef]
- Jauniaux, E.; Watson, A.; Burton, G. Evaluation of respiratory gases and acid-base gradients in human fetal fluids and uteroplacental tissue between 7 and 16 weeks’ gestation. Am. J. Obstet. Gynecol. 2001, 184, 998–1003. [Google Scholar] [CrossRef]
- Rainero, E.; Norman, J.C. Endosomal integrin signals for survival. Nat. Cell Biol. 2015, 17, 1373–1375. [Google Scholar] [CrossRef]
- MacPhee, D.J.; Mostachfi, H.; Han, R.; Lye, S.J.; Post, M.; Caniggia, I. Focal adhesion kinase is a key mediator of human trophoblast development. Lab. Invest. 2001, 81, 1469–1483. [Google Scholar] [CrossRef]
- Zeng, B.X.; Fujiwara, H.; Sato, Y.; Nishioka, Y.; Yamada, S.; Yoshioka, S.; Ueda, M.; Higuchi, T.; Fujii, S. Integrin alpha5 is involved in fibronectin-induced human extravillous trophoblast invasion. J. Reprod. Immunol. 2007, 73, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bersinger, N.A.; Odegard, R.A. Second- and third-trimester serum levels of placental proteins in preeclampsia and small-for-gestational age pregnancies. Acta Obstet. Gynecol. Scand. 2004, 83, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Amsalem, H.; Gaiger, A.; Mizrahi, S.; Yagel, S.; Rachmilewitz, J. Characterization of a lymphocyte subset displaying a unique regulatory activity in human decidua. Int. Immunol. 2008, 20, 1147–1154. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Salem, H.T.; Lee, J.N.; Seppala, M.; Vaara, L.; Aula, P.; Al-Ani, A.T.; Chard, T. Measurement of placental protein 5, placental lactogen and pregnancy-specific beta 1 glycoprotein in mid-trimester as a predictor of outcome of pregnancy. Br. J. Obstet. Gynaecol. 1981, 88, 371–374. [Google Scholar] [CrossRef] [PubMed]
- Towler, C.M.; Horne, C.H.; Jandial, V.; Campbell, D.M.; MacGillivray, I. Plasma levels of pregnancy-specific beta 1-glycoprotein in complicated pregnancies. Br. J. Obstet. Gynaecol. 1977, 84, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Irwin, D.E.; Savitz, D.A.; Hertz-Picciotto, I.; St Andre, K.A. The risk of pregnancy-induced hypertension: Black and white differences in a military population. Am. J. Public Health 1994, 84, 1508–1510. [Google Scholar] [CrossRef]
- Deepthi, G.; Chaithri, P.K.; Latha, P.; Usha Rani, V.; Rahman, P.F.; Jahan, P. TGFB1 functional gene polymorphisms (C-509T and T869C) in the maternal susceptibility to pre-eclampsia in South Indian Women. Scand. J. Immunol. 2015, 82, 390–397. [Google Scholar] [CrossRef]
- Tanaka, M.; Jaamaa, G.; Kaiser, M.; Hills, E.; Soim, A.; Zhu, M.; Shcherbatykh, I.Y.; Samelson, R.; Bell, E.; Zdeb, M.; et al. Racial disparity in hypertensive disorders of pregnancy in New York State: A 10-year longitudinal population-based study. Am. J. Public Health 2007, 97, 163–170. [Google Scholar] [CrossRef]
- Lisonkova, S.; Joseph, K.S. Incidence of preeclampsia: Risk factors and outcomes associated with early- versus late-onset disease. Am. J. Obstet. Gynecol. 2013, 209. [Google Scholar] [CrossRef]
- Blankley, R.T.; Fisher, C.; Westwood, M.; North, R.; Baker, P.N.; Walker, M.J.; Williamson, A.; Whetton, A.D.; Lin, W.; McCowan, L.; et al. A label-free SRM workflow identifies a subset of pregnancy specific glycoproteins as potential predictive markers of early-onset pre-eclampsia. Mol. Cell Proteom. 2013. [Google Scholar] [CrossRef]
- Sulem, P.; Helgason, H.; Oddson, A.; Stefansson, H.; Gudjonsson, S.A.; Zink, F.; Hjartarson, E.; Sigurdsson, G.T.; Jonasdottir, A.; Jonasdottir, A.; et al. Identification of a large set of rare complete human knockouts. Nat. Genet. 2015, 47, 448–452. [Google Scholar] [CrossRef] [PubMed]
- Redman, C.W.; Staff, A.C. Preeclampsia, biomarkers, syncytiotrophoblast stress, and placental capacity. Am. J. Obstet. Gynecol. 2015, 213. [Google Scholar] [CrossRef] [PubMed]
- Hutter, S.; Knabl, J.; Andergassen, U.; Mayr, D.; Hofmann, S.; Kuhn, C.; Mahner, S.; Arck, P.; Jeschke, U. Fetal gender specific expression of tandem-repeat galectins in placental tissue from normally progressed human pregnancies and intrauterine growth restriction (IUGR). Placenta 2015, 36, 1352–1361. [Google Scholar] [CrossRef] [PubMed]
- Blois, S.M.; Dveksler, G.; Vasta, G.R.; Freitag, N.; Blanchard, V.; Barrientos, G. Pregnancy galectinology: Insights into a complex network of glycan binding proteins. Front. Immunol. 2019, 10, 1166. [Google Scholar] [CrossRef] [PubMed]
- Di Renzo, G.C.; Rosati, A.; Sarti, R.D.; Cruciani, L.; Cutuli, A.M. Does fetal sex affect pregnancy outcome? Gend. Med. 2007, 4, 19–30. [Google Scholar] [CrossRef]
- Thagard, A.S.; Slack, J.L.; Estrada, S.M.; Kazanjian, A.A.; Chan, S.; Burd, I.; Napolitano, P.G.; Ieronimakis, N. Long-term impact of intrauterine neuroinflammation and treatment with magnesium sulphate and betamethasone: Sex-specific differences in a preterm labor murine model. Sci. Rep. 2017, 7, 17883. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rattila, S.; Dunk, C.E.; Im, M.; Grichenko, O.; Zhou, Y.; Cohen, M.; Yanez-Mo, M.; Blois, S.M.; Yamada, K.M.; Erez, O.; et al. Interaction of Pregnancy-Specific Glycoprotein 1 With Integrin α5β1 Is a Modulator of Extravillous Trophoblast Functions. Cells 2019, 8, 1369. https://doi.org/10.3390/cells8111369
Rattila S, Dunk CE, Im M, Grichenko O, Zhou Y, Cohen M, Yanez-Mo M, Blois SM, Yamada KM, Erez O, et al. Interaction of Pregnancy-Specific Glycoprotein 1 With Integrin α5β1 Is a Modulator of Extravillous Trophoblast Functions. Cells. 2019; 8(11):1369. https://doi.org/10.3390/cells8111369
Chicago/Turabian StyleRattila, Shemona, Caroline E. Dunk, Michelle Im, Olga Grichenko, Yan Zhou, Marie Cohen, Maria Yanez-Mo, Sandra M. Blois, Kenneth M. Yamada, Offer Erez, and et al. 2019. "Interaction of Pregnancy-Specific Glycoprotein 1 With Integrin α5β1 Is a Modulator of Extravillous Trophoblast Functions" Cells 8, no. 11: 1369. https://doi.org/10.3390/cells8111369
APA StyleRattila, S., Dunk, C. E., Im, M., Grichenko, O., Zhou, Y., Cohen, M., Yanez-Mo, M., Blois, S. M., Yamada, K. M., Erez, O., Gomez-Lopez, N., Lye, S. J., Hinz, B., Romero, R., & Dveksler, G. (2019). Interaction of Pregnancy-Specific Glycoprotein 1 With Integrin α5β1 Is a Modulator of Extravillous Trophoblast Functions. Cells, 8(11), 1369. https://doi.org/10.3390/cells8111369





