Extracellular Vesicles from iPSC-Derived Glial Progenitor Cells Prevent Glutamate-Induced Excitotoxicity by Stabilising Calcium Oscillations and Mitochondrial Depolarisation
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolation of Glial Progenitor Cell-Derived Extracellular Vesicles (EV-GPCs)
2.2. Characteristics of the EV-GPCs
2.2.1. Nanoparticle Tracking Assay
2.2.2. Transmission Electron Microscopy
2.2.3. Western Blot Analysis
2.3. Preparation of Neuroglial Culture
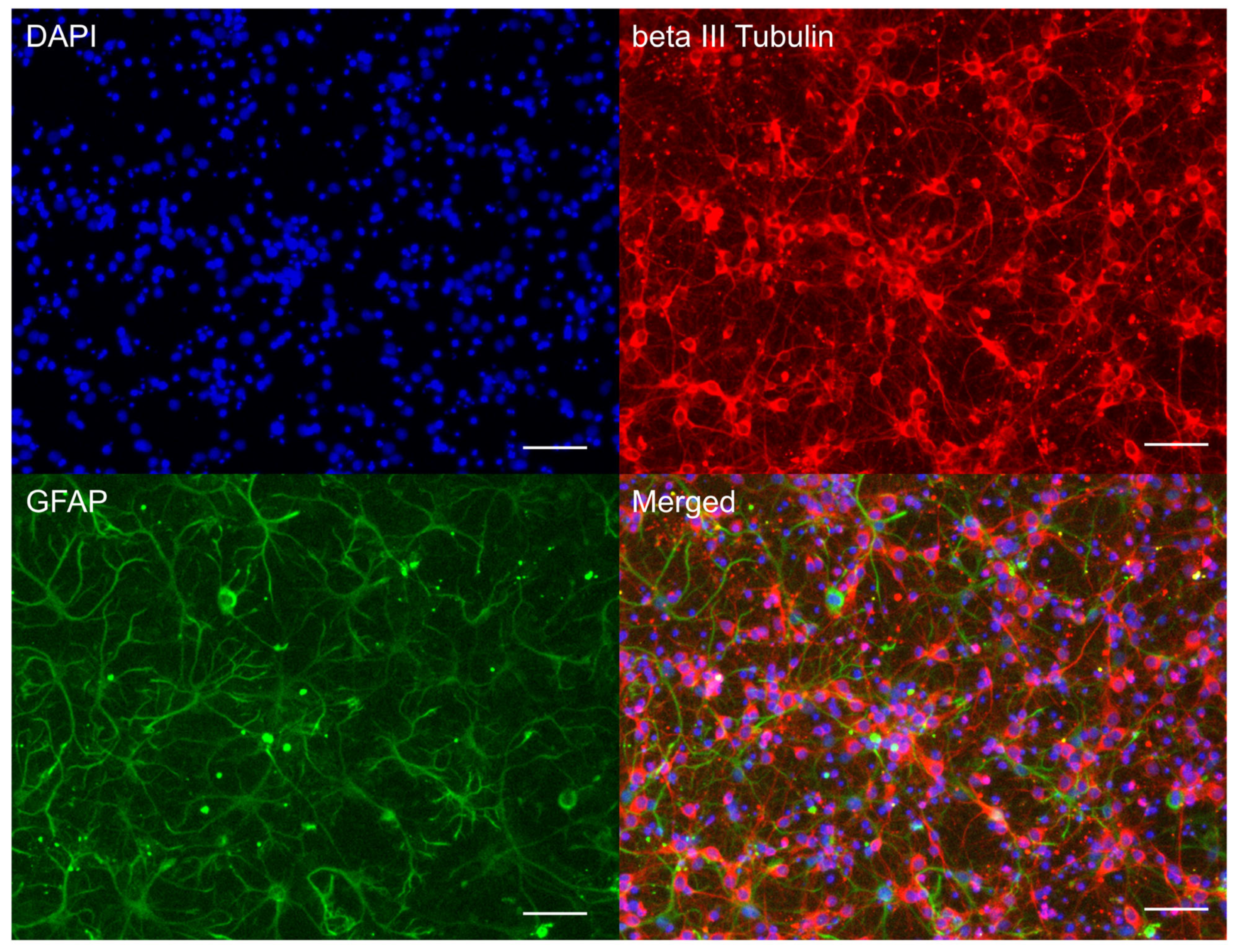
2.4. Immunohistochemistry of Neuroglial Culture
2.5. Induction of Glutamate Excitotoxicity In Vitro
2.6. Assessment of Cell Viability Using the MTT Test and Morphometric Evaluation of Neuronal Death
2.7. Proteomic Analysis of Extracellular Vesicles
2.8. Measurement of [Ca2+]i and Mitochondrial Potential (ΔΨm) in Cortical Neuroglial Cells
2.9. Transcriptomic Analysis (mRNA Sequencing)
2.10. Statistical Analysis
3. Results
3.1. Neuroglial Culture Characteristics
3.2. EV Characteristics
3.3. Proteomic Analysis of EVs
3.3.1. Belonging of Proteins to Intracellular Compartments
3.3.2. Participation of Cargo Proteins in Biological Processes
3.3.3. Molecular Functions of EV-GPC Cargo Proteins
3.3.4. Activated Signalling Pathways by Protein Cargo of EV-GPCs
3.4. Modelling Glutamate Excitotoxicity and the Neuroprotective Effect of Vesicles
3.5. Measurement of Intracellular Ca2+ Concentration ([Ca2+]i) and Mitochondrial Transmembrane Potential (ΔΨm)
3.6. The Role of the PI3K-Akt Pathway in Neuron Survival
3.7. The Effect of EV-GPCs on the Gene Expression Profile of Neuroglial Cultures. Comparison of Gene Expression in Intact Cells with Neuronal Cultures Pretreated by EV-GPCs (IC_vs._EV)
3.8. Assessment of Gene Expression in Neuroglial Cultures Under Glutamate-Induced Excitotoxicity
3.9. The Effect of EV-GPCs on the Gene Expression Profile of Neuroglial Cultures Under Glutamate Exitotoxicity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Di Maio, V. The glutamatergic synapse: A complex machinery for information processing. Cogn. Neurodyn. 2021, 15, 757–781. [Google Scholar] [CrossRef]
- Armada-Moreira, A.; Gomes, J.I.; Pina, C.C.; Savchak, O.K.; Gonçalves-Ribeiro, J.; Rei, N.; Pinto, S.; Morais, T.P.; Martins, R.S.; Ribeiro, F.F.; et al. Going the Extra (Synaptic) Mile: Excitotoxicity as the Road Toward Neurodegenerative Diseases. Front. Cell. Neurosci. 2020, 14, 90. [Google Scholar] [CrossRef] [PubMed]
- Ansari, U.; Alam, M.; Nadora, D.; Muttalib, Z.; Chen, V.; Taguinod, I.; FitzPatrick, M.; Wen, J.; Ansari, Z.; Lui, F. Assessing the efficacy of amyotrophic lateral sclerosis drugs in slowing disease progression: A literature review. AIMS Neurosci. 2024, 11, 166–177. [Google Scholar] [CrossRef] [PubMed]
- Tang, B.C.; Wang, Y.T.; Ren, J. Basic information about memantine and its treatment of Alzheimer’s disease and other clinical applications. Ibrain 2023, 9, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Bellingham, M.C. A Review of the Neural Mechanisms of Action and Clinical Efficiency of Riluzole in Treating Amyotrophic Lateral Sclerosis: What have we Learned in the Last Decade? CNS Neurosci. Ther. 2011, 17, 4–31. [Google Scholar] [CrossRef]
- Pope, C.; Karanth, S.; Liu, J. Pharmacology and toxicology of cholinesterase inhibitors: Uses and misuses of a common mechanism of action. Environ. Toxicol. Pharmacol. 2005, 19, 433–446. [Google Scholar] [CrossRef]
- Svendsen, S.P.; Svendsen, C.N. Cell therapy for neurological disorders. Nat. Med. 2024, 30, 2756–2770. [Google Scholar] [CrossRef]
- Liang, Y.; Ågren, L.; Lyczek, A.; Walczak, P.; Bulte, J.W. Neural progenitor cell survival in mouse brain can be improved by co-transplantation of helper cells expressing bFGF under doxycycline control. Exp. Neurol. 2013, 247, 73–79. [Google Scholar] [CrossRef]
- Nakaji-Hirabayashi, T.; Kato, K.; Iwata, H. In vivo study on the survival of neural stem cells transplanted into the rat brain with a collagen hydrogel that incorporates laminin-derived polypeptides. Bioconj. Chem. 2013, 24, 1798–1804. [Google Scholar] [CrossRef]
- Fujikawa, T.; Oh, S.-H.; Pi, L.; Hatch, H.M.; Shupe, T.; Petersen, B.E. Teratoma formation leads to failure of treatment for type I diabetes using embryonic stem cell-derived insulin-producing cells. Am. J. Pathol. 2005, 166, 1781–1791. [Google Scholar] [CrossRef]
- Richards, M.; Fong, C.-Y.; Chan, W.-K.; Wong, P.-C.; Bongso, A. Human feeders support prolonged undifferentiated growth of human inner cell masses and embryonic stem cells. Nat. Biotechnol. 2002, 20, 933–936. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kuroda, S.; Shichinohe, H.; Ikeda, J.; Seki, T.; Hida, K.; Tada, M.; Sawada, K.; Iwasaki, Y. Migration and differentiation of nuclear fluorescence-labeled bone marrow stromal cells after transplantation into cerebral infarct and spinal cord injury in mice. Neuropathology 2003, 23, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Saboori, M.; Riazi, A.; Taji, M.; Yadegarfar, G. Traumatic brain injury and stem cell treatments: A review of recent 10 years clinical trials. Clin. Neurol. Neurosurg. 2024, 239, 108219. [Google Scholar] [CrossRef] [PubMed]
- Baraniak, P.R.; McDevitt, T.C. Stem cell paracrine actions and tissue regeneration. Regen. Med. 2010, 5, 121–143. [Google Scholar] [CrossRef]
- Shimada, I.S.; Spees, J.L. Stem and progenitor cells for neurological repair: Minor issues, major hurdles, and exciting opportunities for paracrine-based therapeutics. J. Cell. Biochem. 2011, 112, 374–380. [Google Scholar] [CrossRef]
- Bogatcheva, N.V.; Coleman, M.E. Conditioned Medium of Mesenchymal Stromal Cells: A New Class of Therapeutics. Biochemistry 2019, 84, 1375–1389. [Google Scholar] [CrossRef]
- Drago, D.; Cossetti, C.; Iraci, N.; Gaude, E.; Musco, G.; Bachi, A.; Pluchino, S. The stem cell secretome and its role in brain repair. Biochimie 2013, 95, 2271–2285. [Google Scholar] [CrossRef]
- Xia, X.; Wang, Y.; Zheng, J.C. Extracellular vesicles, from the pathogenesis to the therapy of neurodegenerative diseases. Transl. Neurodegener. 2022, 11, 53. [Google Scholar] [CrossRef]
- Kumar, M.A.; Baba, S.K.; Sadida, H.Q.; Marzooqi, S.A.; Jerobin, J.; Altemani, F.H.; Algehainy, N.; Alanazi, M.A.; Abou-Samra, A.-B.; Kumar, R.; et al. Extracellular vesicles as tools and targets in therapy for diseases. Signal Transduct. Target. Ther. 2024, 9, 27. [Google Scholar] [CrossRef]
- Lee, H.-G.; Wheeler, M.A.; Quintana, F.J. Function and therapeutic value of astrocytes in neurological diseases. Nat. Rev. Drug Discov. 2022, 21, 339–358. [Google Scholar] [CrossRef]
- Salikhova, D.I.; Timofeeva, A.V.; Golovicheva, V.V.; Fatkhudinov, T.K.; Shevtsova, Y.A.; Soboleva, A.G.; Fedorov, I.S.; Goryunov, K.V.; Dyakonov, A.S.; Mokrousova, V.O.; et al. Extracellular vesicles of human glial cells exert neuroprotective effects via brain miRNA modulation in a rat model of traumatic brain injury. Sci. Rep. 2023, 13, 20388. [Google Scholar] [CrossRef]
- Salikhova, D.; Bukharova, T.; Cherkashova, E.; Namestnikova, D.; Leonov, G.; Nikitina, M.; Gubskiy, I.; Akopyan, G.; Elchaninov, A.; Midiber, K.; et al. Therapeutic Effects of hiPSC-Derived Glial and Neuronal Progenitor Cells-Conditioned Medium in Experimental Ischemic Stroke in Rats. Int. J. Mol. Sci. 2021, 22, 4694. [Google Scholar] [CrossRef] [PubMed]
- Bakaeva, Z.; Lizunova, N.; Tarzhanov, I.; Boyarkin, D.; Petrichuk, S.; Pinelis, V.; Fisenko, A.; Tuzikov, A.; Sharipov, R.; Surin, A. Lipopolysaccharide from E. coli Increases Glutamate-Induced Disturbances of Calcium Homeostasis, the Functional State of Mitochondria, and the Death of Cultured Cortical Neurons. Front. Mol. Neurosci. 2022, 14, 811171. [Google Scholar] [CrossRef] [PubMed]
- Andrews, S.; Biggins, L.; Inglesfield, S.; Carr, H.; Montgomery, J. FastQC: A Quality Control Tool for High Throughput Sequence Data. 2010. Available online: https://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (accessed on 1 October 2024).
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Patro, R.; Duggal, G.; Love, M.I.; Irizarry, R.A.; Kingsford, C. Salmon provides fast and bias-aware quantification of transcript expression. Nat. Methods 2017, 14, 417–419. [Google Scholar] [CrossRef]
- Soneson, C.; Love, M.I.; Robinson, M.D. Differential analyses for RNA-seq: Transcript-level estimates improve gene-level inferences. F1000Research 2015, 4, 1521. [Google Scholar] [CrossRef]
- Robinson, M.D.; McCarthy, D.J.; Smyth, G.K. edgeR: A Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 2009, 26, 139–140. [Google Scholar] [CrossRef]
- Kanehisa, M.; Goto, S. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef]
- Kanehisa, M. Toward understanding the origin and evolution of cellular organisms. Protein Sci. 2019, 28, 1947–1951. [Google Scholar] [CrossRef]
- Kanehisa, M.; Furumichi, M.; Sato, Y.; Matsuura, Y.; Ishiguro-Watanabe, M. KEGG: Biological systems database as a model of the real world. Nucleic Acids Res. 2025, 53, D672–D677. [Google Scholar] [CrossRef]
- Skjeldal, F.M.; Haugen, L.H.; Mateus, D.; Frei, D.M.; Rødseth, A.V.; Hu, X.; Bakke, O. De novo formation of early endosomes during Rab5-to-Rab7a transition. J. Cell Sci. 2021, 134, jcs254185. [Google Scholar] [CrossRef]
- Zhang, L.; Lu, S.-Y.; Guo, R.; Ma, J.-X.; Tang, L.-Y.; Wang, J.-J.; Shen, C.-L.; Lu, L.-M.; Liu, J.; Wang, Z.-G.; et al. STK10 knockout inhibits cell migration and promotes cell proliferation via modulating the activity of ERM and p38 MAPK in prostate cancer cells. Exp. Ther. Med. 2021, 22, 851. [Google Scholar] [CrossRef] [PubMed]
- Shah, J.A.; Berrington, W.R.; Vary, J.C.; Wells, R.D.; Peterson, G.J.; Kunwar, C.B.; Khadge, S.; Hagge, D.A.; Hawn, T.R. Genetic variation in toll-interacting protein is associated with leprosy susceptibility and cutaneous expression of interleukin 1 receptor antagonist. J. Infect. Dis. 2016, 213, 1189–1197. [Google Scholar] [CrossRef] [PubMed]
- Petranka, J.G.; E Fleenor, D.; Sykes, K.; E Kaufman, R.; Rosse, W.F. Structure of the CD59-encoding gene: Further evidence of a relationship to murine lymphocyte antigen Ly-6 protein. Proc. Natl. Acad. Sci. USA 1992, 89, 7876–7879. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, A.K.; Maji, S.; Bhoopathi, P.; Talukdar, S.; Mannangatti, P.; Guo, C.; Wang, X.-Y.; Cartagena, L.C.; Idowu, M.; Landry, J.W.; et al. Pharmacological inhibition of MDA-9/Syntenin blocks breast cancer metastasis through suppression of IL-1β. Proc. Natl. Acad. Sci. USA 2021, 118, e2103180118. [Google Scholar] [CrossRef]
- Fukumoto, I.; Kurozumi, A.; Goto, Y.; Matsushita, R.; Kato, M.; Nishikawa, R.; Sakamoto, S.; Ichikawa, T.; Seki, N. Abstract 1100: Targeting ITGA3/ITGB1 signaling by tumor-suppressive microRNA-223 inhibits cancer cell migration and invasion in prostate cancer. Cancer Res. 2016, 76, 1100. [Google Scholar] [CrossRef]
- Yang, Y.; Zhao, N.; Wang, R.; Zhan, Z.; Guo, S.; Song, H.; Wiemer, E.A.; Ben, J.; Ma, J. Macrophage MVP regulates fracture repair by promoting M2 polarization via JAK2-STAT6 pathway. Int. Immunopharmacol. 2023, 120, 110313. [Google Scholar] [CrossRef]
- Morozova, K.; Sidhar, S.; Zolla, V.; Clement, C.C.; Scharf, B.; Verzani, Z.; Diaz, A.; Larocca, J.N.; Hajjar, K.A.; Cuervo, A.M.; et al. Annexin A2 promotes phagophore assembly by enhancing Atg16L+ vesicle biogenesis and homotypic fusion. Nat. Commun. 2015, 6, 5856. [Google Scholar] [CrossRef]
- MacKenzie, E.L.; Ray, P.D.; Tsuji, Y. Role and regulation of ferritin H in rotenone-mediated mitochondrial oxidative stress. Free. Radic. Biol. Med. 2008, 44, 1762–1771. [Google Scholar] [CrossRef]
- Gao, D.; Lv, Y.; Hong, F.; Wu, D.; Wang, T.; Gao, G.; Lin, Z.; Yang, R.; Hu, J.; He, A.; et al. Peroxiredoxin 6 maintains mitochondrial homeostasis and promotes tumor progression through ROS/JNK/p38 MAPK signaling pathway in multiple myeloma. Sci. Rep. 2025, 15, 70. [Google Scholar] [CrossRef]
- Papadopoulou, D.; Mavrikaki, V.; Charalampous, F.; Tzaferis, C.; Samiotaki, M.; Papavasileiou, K.D.; Afantitis, A.; Karagianni, N.; Denis, M.C.; Sanchez, J.; et al. Discovery of the First-in-Class Inhibitors of Hypoxia Up-Regulated Protein 1 (HYOU1) Suppressing Pathogenic Fibroblast Activation. Angew. Chem. Int. Ed. Engl. 2024, 63, e202319157. [Google Scholar] [CrossRef]
- Masters, S.C.; Yang, H.; Datta, S.R.; Greenberg, M.E.; Fu, H. 14-3-3 inhibits Bad-induced cell death through interaction with serine-136. Mol. Pharmacol. 2001, 60, 1325–1331. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Buckley, S.M.; Cimmino, L.; Guillamot, M.; Strikoudis, A.; Cang, Y.; Goff, S.P.; Aifantis, I. The CUL4-DDB1 ubiquitin ligase complex controls adult and embryonic stem cell differentiation and homeostasis. eLife 2015, 4, e07539. [Google Scholar] [CrossRef] [PubMed]
- Bhat, T.A.; Chaudhary, A.K.; Kumar, S.; O’mAlley, J.; Inigo, J.R.; Kumar, R.; Yadav, N.; Chandra, D. Endoplasmic reticulum-mediated unfolded protein response and mitochondrial apoptosis in cancer. Biochim. Biophys. Acta (BBA)-Rev. Cancer 2017, 1867, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Cao, S.; Li, Y. Mechanistic study of heat shock protein 60-mediated apoptosis in DF-1 cells. Poult. Sci. 2024, 103, 103619. [Google Scholar] [CrossRef]
- Zhang, H.; Song, X.; Li, T.; Wang, J.; Xing, B.; Zhai, X.; Luo, J.; Hu, X.; Hou, X.; Wei, L. DDX1 from Cherry valley duck mediates signaling pathways and anti-NDRV activity. Veter.-Res. 2021, 52, 9. [Google Scholar] [CrossRef]
- Yan, T.; Lin, Z.; Jiang, J.; Lu, S.; Chen, M.; Que, H.; He, X.; Que, G.; Mao, J.; Xiao, J.; et al. MMP14 regulates cell migration and invasion through epithelial-mesenchymal transition in nasopharyngeal carcinoma. Am. J. Transl. Res. 2015, 7, 950–958. [Google Scholar]
- Sanchez, A.; Tripathy, D.; Yin, X.; Luo, J.; Martinez, J.; Grammas, P. Pigment epithelium-derived factor (PEDF) protects cortical neurons in vitro from oxidant injury by activation of extracellular signal-regulated kinase (ERK) 1/2 and induction of Bcl-2. Neurosci. Res. 2012, 72, 1–8. [Google Scholar] [CrossRef]
- Kim, T.H.; Sung, S.-E.; Yoo, J.C.; Park, J.-Y.; Yi, G.-S.; Heo, J.Y.; Lee, J.-R.; Kim, N.-S.; Lee, D.Y. Copine1 regulates neural stem cell functions during brain development. Biochem. Biophys. Res. Commun. 2018, 495, 168–173. [Google Scholar] [CrossRef]
- Namekata, K.; Tsuji, N.; Guo, X.; Nishijima, E.; Honda, S.; Kitamura, Y.; Yamasaki, A.; Kishida, M.; Takeyama, J.; Ishikawa, H.; et al. Neuroprotection and axon regeneration by novel low-molecular-weight compounds through the modification of DOCK3 conformation. Cell Death Discov. 2023, 9, 166. [Google Scholar] [CrossRef]
- Shirao, T.; Hanamura, K.; Koganezawa, N.; Ishizuka, Y.; Yamazaki, H.; Sekino, Y. The role of drebrin in neurons. J. Neurochem. 2017, 141, 819–834. [Google Scholar] [CrossRef] [PubMed]
- Kuramoto, T.; Kitada, K.; Inui, T.; Sasaki, Y.; Ito, K.; Hase, T.; Kawagachi, S.; Ogawa, Y.; Nakao, K.; Barsh, G.S.; et al. Attractin/mahogany/zitter plays a critical role in myelination of the central nervous system. Proc. Natl. Acad. Sci. USA 2001, 98, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Henderson, M.J.; Richie, C.T.; Airavaara, M.; Wang, Y.; Harvey, B.K. Mesencephalic astrocyte-derived neurotrophic factor (MANF) secretion and cell surface binding are modulated by KDEL receptors. J. Biol. Chem. 2013, 288, 4209–4225. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Sharma, P.; Dahiya, S.; Sharma, B. Plexins: Navigating through the neural regulation and brain pathology. Neurosci. Biobehav. Rev. 2025, 169, 105999. [Google Scholar] [CrossRef]
- Trochet, D.; Bitoun, M. A review of Dynamin 2 involvement in cancers highlights a promising therapeutic target. J. Exp. Clin. Cancer Res. 2021, 40, 238. [Google Scholar] [CrossRef]
- Murga, M.; Fernandez-Capetillo, O.; Tosato, G. Neuropilin-1 regulates attachment in human endothelial cells independently of vascular endothelial growth factor receptor-2. Blood 2005, 105, 1992–1999. [Google Scholar] [CrossRef]
- Sánchez-Elsner, T.; Botella, L.M.; Velasco, B.; Langa, C.; Bernabéu, C. Endoglin expression is regulated by transcriptional cooperation between the hypoxia and transforming growth factor-β pathways. J. Biol. Chem. 2002, 277, 43799–43808. [Google Scholar] [CrossRef]
- Zheng, Y.-S.; Liu, Y.-L.; Xu, Z.-G.; He, C.; Guo, Z.-Y. Is myeloid-derived growth factor a ligand of the sphingosine-1-phosphate receptor 2? Biochem. Biophys. Res. Commun. 2024, 706, 149766. [Google Scholar] [CrossRef]
- Farach-Carson, M.C.; Warren, C.R.; Harrington, D.A.; Carson, D.D. Border patrol: Insights into the unique role of perlecan/heparan sulfate proteoglycan 2 at cell and tissue borders. Matrix Biol. 2014, 34, 64–79. [Google Scholar] [CrossRef]
- Kang, S.W. Superoxide dismutase 2 gene and cancer risk: Evidence from an updated meta-analysis. Int. J. Clin. Exp. Med. 2015, 8, 14647–14655. [Google Scholar]
- Pei, J.; Pan, X.; Wei, G.; Hua, Y. Research progress of glutathione peroxidase family (GPX) in redoxidation. Front. Pharmacol. 2023, 14, 1147414. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, S.; Ding, D.; Yu, Z.; Sun, W.; Wang, Y. Up-regulation of Cdc37 contributes to schwann cell proliferation and migration after sciatic nerve crush. Neurochem. Res. 2018, 43, 1182–1190. [Google Scholar] [CrossRef]
- Giulino-Roth, L.; van Besien, H.J.; Dalton, T.; Totonchy, J.E.; Rodina, A.; Taldone, T.; Bolaender, A.; Erdjument-Bromage, H.; Sadek, J.; Chadburn, A. Inhibition of Hsp90 suppresses PI3K/AKT/mTOR signaling and has antitumor activity in Burkitt lym-phoma. Mol. Cancer Ther. 2017, 16, 1779–1790. [Google Scholar] [CrossRef] [PubMed]
- Bousoik, E.; Aliabadi, H.M. “Do We Know Jack” About JAK? A Closer Look at JAK/STAT Signaling Pathway. Front. Oncol. 2018, 8, 287. [Google Scholar] [CrossRef]
- Kamp, M.E.; Liu, Y.; Kortholt, A. Function and regulation of heterotrimeric g proteins during chemotaxis. Int. J. Mol. Sci. 2016, 17, 90. [Google Scholar] [CrossRef]
- Yuasa-Kawada, J.; Kinoshita-Kawada, M.; Tsuboi, Y.; Wu, J.Y. Neuronal guidance genes in health and diseases. Protein Cell 2023, 14, 238–261. [Google Scholar] [CrossRef]
- Andermatt, I.; Wilson, N.H.; Bergmann, T.; Mauti, O.; Gesemann, M.; Sockanathan, S.; Stoeckli, E.T. Semaphorin 6B acts as a receptor in post-crossing commissural axon guidance. Development 2014, 141, 3709–3720. [Google Scholar] [CrossRef]
- Xu, N.-J.; Henkemeyer, M. Ephrin reverse signaling in axon guidance and synaptogenesis. Semin. Cell Dev. Biol. 2012, 23, 58–64. [Google Scholar] [CrossRef]
- Long, H.; Sabatier, C.; Ma, L.; Plump, A.; Yuan, W.; Ornitz, D.M.; Tamada, A.; Murakami, F.; Goodman, C.S.; Tessier-Lavigne, M. Conserved roles for Slit and Robo proteins in midline commissural axon guidance. Neuron 2004, 42, 213–223. [Google Scholar] [CrossRef]
- Salikhova, D.I.; Shedenkova, M.O.; Sudina, A.K.; Belousova, E.V.; Krasilnikova, I.A.; Nekrasova, A.A.; Nefedova, Z.A.; Frolov, D.A.; Fatkhudinov, T.K.; Makarov, A.V.; et al. Neuroprotective and anti-inflammatory properties of proteins secreted by glial progenitor cells derived from human iPSCs. Front. Cell. Neurosci. 2024, 18, 1449063. [Google Scholar] [CrossRef]
- Zgodova, A.; Pavlova, S.; Nekrasova, A.; Boyarkin, D.; Pinelis, V.; Surin, A.; Bakaeva, Z. Isoliquiritigenin Protects Neuronal Cells against Glutamate Excitotoxicity. Membranes 2022, 12, 1052. [Google Scholar] [CrossRef]
- Reed, S.L.; Escayg, A. Extracellular vesicles in the treatment of neurological disorders. Neurobiol. Dis. 2021, 157, 105445. [Google Scholar] [CrossRef]
- Ginini, L.; Billan, S.; Fridman, E.; Gil, Z. Insight into Extracellular Vesicle-Cell Communication: From Cell Recognition to Intracellular Fate. Cells 2022, 11, 1375. [Google Scholar] [CrossRef] [PubMed]
- Bürger, S.; Meng, J.; Zwanzig, A.; Beck, M.; Pankonin, M.; Wiedemann, P.; Eichler, W.; Unterlauft, J.D. Pigment epithelium-derived factor (PEDF) receptors are involved in survival of retinal neurons. Int. J. Mol. Sci. 2021, 22, 369. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, S.; Gharagozloo, M.; Simard, C.; Gris, D. Astrocytes maintain glutamate homeostasis in the cns by controlling the balance between glutamate uptake and release. Cells 2019, 8, 184. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.-L.; Gong, X.-X.; Qin, Z.-H.; Wang, Y. Molecular mechanisms of excitotoxicity and their relevance to the pathogenesis of neurodegenerative diseases—An update. Acta Pharmacol. Sin. 2025, 46, 3129–3142. [Google Scholar] [CrossRef]
- Segal, M.; Korkotian, E. Endoplasmic reticulum calcium stores in dendritic spines. Front. Neuroanat. 2014, 8, 64. [Google Scholar] [CrossRef]
- Pivovarova, N.B.; Andrews, S.B. Calcium-dependent mitochondrial function and dysfunction in neurons: Minireview. FEBS J. 2010, 277, 3622–3636. [Google Scholar] [CrossRef]
- Spires, T.L.; Molnár, Z.; Kind, P.C.; Cordery, P.M.; Upton, A.L.; Blakemore, C.; Hannan, A.J. Activity-dependent regulation of synapse and dendritic spine morphology in developing barrel cortex requires phospholipase C-β1 signalling. Cereb. Cortex 2005, 15, 385–393. [Google Scholar] [CrossRef]
- Krishnan, A.; Areti, A.; Komirishetty, P.; Chandrasekhar, A.; Cheng, C.; Zochodne, D.W. Survival of compromised adult sensory neurons involves macrovesicular formation. Cell Death Discov. 2022, 8, 462. [Google Scholar] [CrossRef]
- Vaillant, A.R.; Zanassi, P.; Walsh, G.S.; Aumont, A.; Alonso, A.; Miller, F.D. Signaling mechanisms underlying reversible activity-dependent dendrite formation. Neuron 2002, 34, 985–998. [Google Scholar] [CrossRef]
- Nicosia, N.; Giovenzana, M.; Misztak, P.; Mingardi, J.; Musazzi, L. Glutamate-Mediated Excitotoxicity in the Pathogenesis and Treatment of Neurodevelopmental and Adult Mental Disorders. Int. J. Mol. Sci. 2024, 25, 6521. [Google Scholar] [CrossRef] [PubMed]
- Biswas, K.; Alexander, K.; Francis, M.M. Reactive Oxygen Species: Angels and Demons in the Life of a Neuron. NeuroSci 2022, 3, 130–145. [Google Scholar] [CrossRef] [PubMed]
- Phillips, M.B.; Povysheva, N.V.; Harnett-Scott, K.A.; Aizenman, E.; Johnson, J.W. State-specific inhibition of NMDA receptors by memantine depends on intracellular calcium and provides insights into NMDAR channel blocker tolerability. bioRxiv 2024. [Google Scholar] [CrossRef]
- Bonsergent, E.; Grisard, E.; Buchrieser, J.; Schwartz, O.; Théry, C.; Lavieu, G. Quantitative characterization of extracellular vesicle uptake and content delivery within mammalian cells. Nat. Commun. 2021, 12, 1864. [Google Scholar] [CrossRef]
- Povysheva, N.V.; Johnson, J.W. Effects of memantine on the excitation-inhibition balance in prefrontal cortex. Neurobiol. Dis. 2016, 96, 75–83. [Google Scholar] [CrossRef]
- Dong, H.; Yuede, C.M.; Coughlan, C.; Lewis, B.; Csernansky, J.G. Effects of memantine on neuronal structure and condi-tioned fear in the Tg2576 mouse model of Alzheimer’s disease. Neuropsychopharmacology 2008, 33, 3226–3236. [Google Scholar] [CrossRef]
- Read, D.E.; Gorman, A.M. Involvement of Akt in neurite outgrowth. Cell. Mol. Life Sci. 2009, 66, 2975–2984. [Google Scholar] [CrossRef]
- Sánchez-Alegría, K.; Flores-León, M.; Avila-Muñoz, E.; Rodríguez-Corona, N.; Arias, C. PI3K signaling in neurons: A central node for the control of multiple functions. Int. J. Mol. Sci. 2018, 19, 3725. [Google Scholar] [CrossRef]
- Neves, D.; Salazar, I.L.; Almeida, R.D.; Silva, R.M. Molecular mechanisms of ischemia and glutamate excitotoxicity. Life Sci. 2023, 328, 121814. [Google Scholar] [CrossRef]
- Wu, Q.J.; Tymianski, M. Targeting nmda receptors in stroke: New hope in neuroprotection Tim Bliss. Mol. Brain 2018, 11, 15. [Google Scholar] [CrossRef]
- Lai, T.W.; Zhang, S.; Wang, Y.T. Excitotoxicity and stroke: Identifying novel targets for neuroprotection. Prog. Neurobiol. 2014, 115, 157–188. [Google Scholar] [CrossRef] [PubMed]
- Martel, M.-A.; Ryan, T.J.; Bell, K.F.; Fowler, J.H.; McMahon, A.; Al-Mubarak, B.; Komiyama, N.H.; Horsburgh, K.; Kind, P.C.; Grant, S.G.; et al. The Subtype of GluN2 C-terminal Domain Determines the Response to Excitotoxic Insults. Neuron 2012, 74, 543–556. [Google Scholar] [CrossRef] [PubMed]
- Turovsky, E.A.; Golovicheva, V.V.; Varlamova, E.G.; Danilina, T.I.; Goryunov, K.V.; Shevtsova, Y.A.; Pevzner, I.B.; Zorova, L.D.; Babenko, V.A.; Evtushenko, E.A.; et al. Mesenchymal stromal cell-derived extracellular vesicles afford neuroprotection by modulating PI3K/AKT pathway and calcium oscillations. Int. J. Biol. Sci. 2022, 18, 5345–5368. [Google Scholar] [CrossRef] [PubMed]
- Bonafede, R.; Scambi, I.; Peroni, D.; Potrich, V.; Boschi, F.; Benati, D.; Bonetti, B.; Mariotti, R. Exosome derived from murine adipose-derived stromal cells: Neuroprotective effect on in vitro model of amyotrophic lateral sclerosis. Exp. Cell Res. 2016, 340, 150–158. [Google Scholar] [CrossRef]
- Rao, S.; Madhu, L.N.; Babu, R.S.; Nagarajan, A.; Narvekar, E.; Upadhya, R.; Shetty, A.K. Extracellular Vesicles from Human iPSC-derived NSCs Protect Human Neurons Against Aβ-42 Induced Neurodegeneration, Mitochondrial Dysfunction and Tau Phosphorylation. Alzheimer’s Dement. 2024, 20, e093150. [Google Scholar] [CrossRef]
- Xiong, W.-P.; Yao, W.-Q.; Wang, B.; Liu, K. BMSCs-exosomes containing GDF-15 alleviated SH-SY5Y cell injury model of Alzheimer’s disease via AKT/GSK-3β/β-catenin. Brain Res. Bull. 2021, 177, 92–102. [Google Scholar] [CrossRef]
- Xie, Y.; Chen, Y.; Zhu, Y.; Chen, X.; Lin, T.; Zhou, D. Adipose Mesenchymal Stem Cell-Derived Exosomes Enhance PC12 Cell Function through the Activation of the PI3K/AKT Pathway. Stem Cells Int. 2021, 2021, 2229477. [Google Scholar] [CrossRef]
- Wei, H.; Xu, Y.; Chen, Q.; Chen, H.; Zhu, X.; Li, Y. Mesenchymal stem cell-derived exosomal miR-223 regulates neuronal cell apoptosis. Cell Death Dis. 2020, 11, 290. [Google Scholar] [CrossRef]
- Yeung, A.W.K.; Choudhary, N.; Tewari, D.; El-Demerdash, A.; Horbańczuk, O.K.; Das, N.; Pirgozliev, V.; Lucarini, M.; Durazzo, A.; Souto, E.B.; et al. Quercetin: Total-scale literature landscape analysis of a valuable nutraceutical with numerous potential applications in the promotion of human and animal health a review. Anim. Sci. Pap. Rep. 2021, 39, 199–212. [Google Scholar]
- Deng, M.; Xiao, H.; Peng, H.; Yuan, H.; Xu, Y.; Zhang, G.; Tang, J.; Hu, Z. Preservation of neuronal functions by exosomes derived from different human neural cell types under ischemic conditions. Eur. J. Neurosci. 2018, 47, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Namini, M.S.; Beheshtizadeh, N.; Ebrahimi-Barough, S.; Ai, J. Human endometrial stem cell-derived small extracellular vesicles enhance neurite outgrowth and peripheral nerve regeneration through activating the PI3K/AKT signaling pathway. J. Transl. Med. 2025, 23, 6. [Google Scholar] [CrossRef] [PubMed]
- Saganich, M.J.; Machado, E.; Rudy, B. Differential expression of genes encoding subthreshold-operating voltage-gated K+ channels in brain. J. Neurosci. 2001, 21, 4609–4624. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Fan, G.; Shao, S. Role of TNFRSF12A in cell proliferation, apoptosis, and proinflammatory cytokine expression by regulating the MAPK and NF-κB pathways in thyroid cancer cells. Cytokine 2025, 186, 156841. [Google Scholar] [CrossRef]
- Ashutosh Chao, C.; Borgmann, K.; Brew, K.; Ghorpade, A. Tissue inhibitor of metalloproteinases-1 protects human neurons from staurosporine and HIV-1-induced apoptosis: Mechanisms and relevance to HIV-1-associated dementia. Cell Death Dis. 2012, 3, e332. [Google Scholar] [CrossRef]
- Gauthier-Kemper, A.; Alonso, M.S.; Sündermann, F.; Niewidok, B.; Fernandez, M.-P.; Bakota, L.; Heinisch, J.J.; Brandt, R. Annexins A2 and A6 interact with the extreme N terminus of tau and thereby contribute to tau’s axonal localization. J. Biol. Chem. 2018, 293, 8065–8076. [Google Scholar] [CrossRef]
- Mandal, P.; Belapurkar, V.; Nair, D.; Ramanan, N. Vinculin-mediated axon growth requires interaction with actin but not talin in mouse neocortical neurons. Cell. Mol. Life Sci. 2021, 78, 5807–5826. [Google Scholar] [CrossRef]
- Buttiglione, M.; Revest, J.-M.; Rougon, G.; Faivre-Sarrailh, C. F3 neuronal adhesion molecule controls outgrowth and fasciculation of cerebellar granule cell neurites: A cell-type-specific effect mediated by the Ig-like domains. Mol. Cell. Neurosci. 1996, 8, 53–69. [Google Scholar] [CrossRef]
- Istiaq, A.; Ohta, K. A review on Tsukushi: Mammalian development, disorders, and therapy. J. Cell Commun. Signal. 2022, 16, 505–513. [Google Scholar] [CrossRef]
- Gimenez-Cassina, A.; Lim, F.; Cerrato, T.; Palomo, G.M.; Diaz-Nido, J. Mitochondrial hexokinase II promotes neuronal survival and acts downstream of glycogen synthase kinase-3. J. Biol. Chem. 2009, 284, 3001–3011. [Google Scholar] [CrossRef]
- Nitti, M.; Piras, S.; Brondolo, L.; Marinari, U.M.; Pronzato, M.A.; Furfaro, A.L. Heme oxygenase 1 in the nervous system: Does it favor neuronal cell survival or induce neurodegeneration? Int. J. Mol. Sci. 2018, 19, 2260. [Google Scholar] [CrossRef]
- Dziennis, S.; Alkayed, N.J. Role of signal transducer and activator of transcription 3 in neuronal survival and regeneration. Rev. Neurobiol. 2008, 19, 341–362. [Google Scholar] [CrossRef]
- Yoshida, T.; Shiroshima, T.; Lee, S.-J.; Yasumura, M.; Uemura, T.; Chen, X.; Iwakura, Y.; Mishina, M. Interleukin-1 receptor accessory protein organizes neuronal synaptogenesis as a cell adhesion molecule. J. Neurosci. 2012, 32, 2588–2600. [Google Scholar] [CrossRef]
- Dzwonek, J.; Wilczyński, G.M. CD44: Molecular interactions, signaling and functions in the nervous system. Front. Cell. Neurosci. 2015, 9, 175. [Google Scholar] [CrossRef] [PubMed]
- Ji, E.; Zhang, Y.; Li, Z.; Wei, L.; Wu, Z.; Li, Y.; Yu, X.; Song, T.-J. The Chemokine CCL2 Promotes Excitatory Synaptic Transmission in Hippocampal Neurons via GluA1 Subunit Trafficking. Neurosci. Bull. 2024, 40, 1649–1666. [Google Scholar] [CrossRef] [PubMed]
- Lieberam, I.; Agalliu, D.; Nagasawa, T.; Ericson, J.; Jessell, T.M. A Cxcl12-Cxcr4 Chemokine signaling pathway defines the initial trajectory of mammalian motor axons. Neuron 2005, 47, 667–679. [Google Scholar] [CrossRef]
- Caminero, A.; Comabella, M.; Montalban, X. Role of tumour necrosis factor (TNF)-α and TNFRSF1A R92Q mutation in the pathogenesis of TNF receptor-associated periodic syndrome and multiple sclerosis. Clin. Exp. Immunol. 2011, 166, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Xu, X.; He, J.; Murray, A.; Sun, M.-A.; Wei, X.; Wang, X.; McCoig, E.; Xie, E.; Jiang, X.; et al. EGR1 recruits TET1 to shape the brain methylome during development and upon neuronal activity. Nat. Commun. 2019, 10, 3892. [Google Scholar] [CrossRef]
- Yang, N.J.; Isensee, J.; Neel, D.; Liu, S.M.; Zhang, H.X.B.; Belu, A.; Palan, S.; Kennedy-Curran, A.; Röderer, P.; Nitzsche, A.; et al. Anthrax Toxin as a Molecular Platform to Target Nociceptive Neurons and Modulate Pain. bioRxiv 2020. [Google Scholar] [CrossRef]
- Mohamedi, Y.; Fontanil, T.; Cobo, T.; Cal, S.; Obaya, A.J. New insights into adamts metalloproteases in the central nervous system. Biomolecules 2020, 10, 403. [Google Scholar] [CrossRef]
- Li, J.; Parker, B.; Martyn, C.; Natarajan, C.; Guo, J. The PMP22 gene and its related diseases. Mol. Neurobiol. 2013, 47, 673–698. [Google Scholar] [CrossRef] [PubMed]
- Camenisch, T.D.; Spicer, A.P.; Brehm-Gibson, T.; Biesterfeldt, J.; Augustine, M.L.; Calabro, A.; Kubalak, S.; Klewer, S.E.; McDonald, J.A. Disruption of hyaluronan synthase-2 abrogates normal cardiac morphogenesis and hyaluronan-mediated transformation of epithelium to mesenchyme. J. Clin. Investig. 2000, 106, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Joy, M.T.; Vrbova, G.; Dhoot, G.K.; Anderson, P.N. Sulf1 and Sulf2 expression in the nervous system and its role in limiting neurite outgrowth in vitro. Exp. Neurol. 2015, 263, 150–160. [Google Scholar] [CrossRef]
- Cao, Y.; Jin, H.-G.; Ma, H.-H.; Zhao, Z.-H. Comparative analysis on genome-wide DNA methylation in longissimus dorsi muscle between Small Tailed Han and DorperSmall Tailed Han crossbred sheep. Asian-Australas. J. Anim. Sci. 2017, 30, 1529–1539. [Google Scholar] [CrossRef]
- Leung, G.; Zhou, Y.; Ostrowski, P.; Mylvaganam, S.; Boroumand, P.; Mulder, D.J.; Guo, C.; Muise, A.M.; Freeman, S.A. ARPC1B binds WASP to control actin polymerization and curtail tonic signaling in B cells. J. Clin. Investig. 2021, 6, e149376. [Google Scholar] [CrossRef]
- Kim, H.-R.; Kwon, M.-S.; Lee, S.; Mun, Y.; Lee, K.-S.; Kim, C.-H.; Na, B.-R.; Kim, B.N.R.; Piragyte, I.; Lee, H.-S.; et al. TAGLN2 polymerizes G-actin in a low ionic state but blocks Arp2/3-nucleated actin branching in physiological conditions. Sci. Rep. 2018, 8, 5503. [Google Scholar] [CrossRef]
- Chen, Z.; Wang, C.; Li, M.; Cai, S.; Liu, X. SPRED3 regulates the NF-κB signaling pathway in thyroid cancer and promotes the proliferation. Sci. Rep. 2024, 14, 20506. [Google Scholar] [CrossRef]
- Pan, A.L.; Audrain, M.; Sakakibara, E.; Joshi, R.; Zhu, X.; Wang, Q.; Wang, M.; Beckmann, N.D.; Schadt, E.E.; Gandy, S.; et al. Dual-specificity protein phosphatase 6 (DUSP6) overexpression reduces amyloid load and improves memory deficits in male 5xFAD mice. Front. Aging Neurosci. 2024, 16, 1400447. [Google Scholar] [CrossRef]
- Huang, S.; Liu, F.; Niu, Q.; Li, Y.; Liu, C.; Zhang, L.; Ni, D.; Pu, X. GLIPR-2 Overexpression in HK-2 Cells Promotes Cell EMT and Migration through ERK1/2 Activation. PLoS ONE 2013, 8, e58574. [Google Scholar] [CrossRef]
- Satoh, J.-I.; Kino, Y.; Yanaizu, M.; Ishida, T.; Saito, Y. Microglia express GPNMB in the brains of Alzheimer’s disease and Nasu-Hakola disease. Intractable Rare Dis. Res. 2019, 8, 120–128. [Google Scholar] [CrossRef]
- Seijffers, R.; Zhang, J.; Matthews, J.C.; Chen, A.; Tamrazian, E.; Babaniyi, O.; Selig, M.; Hynynen, M.; Woolf, C.J.; Brown, R.H. ATF3 expression improves motor function in the ALS mouse model by promoting motor neuron survival and retaining muscle innervation. Proc. Natl. Acad. Sci. USA 2014, 111, 1622–1627. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, K.; Huang, Y.; Sun, M.; Tian, Q.; Zhang, S.; Qin, Y. TRIM25 Promotes TNF-α–Induced NF-κB Activation through Potentiating the K63-Linked Ubiquitination of TRAF2. J. Immunol. 2020, 204, 1499–1507. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Zhang, Y.; Cai, Z.; Jiang, M.; Li, B.; Chen, G.; Zeng, Y.; Liang, Y.; Wu, S.; Wang, Z.; et al. Increased expression of immediate early response gene 3 protein promotes aggressive progression and predicts poor prognosis in human bladder cancer. BMC Urol. 2018, 18, 82. [Google Scholar] [CrossRef]
- Srivastava, A.K.; Renusch, S.R.; Naiman, N.E.; Gu, S.; Sneh, A.; Arnold, W.D.; Sahenk, Z.; Kolb, S.J. Mutant HSPB1 overexpression in neurons is sufficient to cause age-related motor neuronopathy in mice. Neurobiol. Dis. 2012, 47, 163–173. [Google Scholar] [CrossRef]
- Jin, H.; Kanthasamy, A.; Anantharam, V.; Rana, A.; Kanthasamy, A.G. Transcriptional regulation of pro-apoptotic protein kinase Cδ: Implications for oxidative stress-induced neuronal cell death. J. Biol. Chem. 2011, 286, 19840–19859. [Google Scholar] [CrossRef]
- Léger, H.; Santana, E.; Leu, N.A.; Smith, E.T.; Beltran, W.A.; Aguirre, G.D.; Luca, F.C. Ndr kinases regulate retinal interneuron proliferation and homeostasis. Sci. Rep. 2018, 8, 12544. [Google Scholar] [CrossRef]
- Nakazawa, S.; Gotoh, N.; Matsumoto, H.; Murayama, C.; Suzuki, T.; Yamamoto, T. Expression of sorting Nexin 18 (SNX18) is dynamically regulated in developing Spinal Motor Neurons. J. Histochem. Cytochem. 2011, 59, 202–213. [Google Scholar] [CrossRef]
- Corgiat, E.B.; List, S.M.; Rounds, J.C.; Corbett, A.H.; Moberg, K.H. The RNA-binding protein Nab2 regulates the proteome of the developing Drosophila brain. J. Biol. Chem. 2021, 297, 100877. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, K.K. Glial fibrillary acidic protein: From intermediate filament assembly and gliosis to neurobiomarker. Trends Neurosci. 2015, 38, 364–374. [Google Scholar] [CrossRef]
- Cai, J.; Gao, L.; Wang, Y.; Li, Y.; Ye, Z.; Tong, S.; Yan, T.; Sun, Q.; Xu, Y.; Jiang, H.; et al. TMBIM1 promotes proliferation and attenuates apoptosis in glioblastoma cells by targeting the p38 MAPK signalling pathway. Transl. Oncol. 2022, 19, 101391. [Google Scholar] [CrossRef]
- Santoro, A.; Nicolin, V.; Florenzano, F.; Rosati, A.; Capunzo, M.; Nori, S.L. BAG3 is involved in neuronal differentiation and migration. Cell Tissue Res. 2017, 368, 249–258. [Google Scholar] [CrossRef][Green Version]
- Kerosuo, L.; Neppala, P.; Hsin, J.; Mohlin, S.; Vieceli, F.M.; Török, Z.; Laine, A.; Westermarck, J.; Bronner, M.E. Enhanced expression of MycN/CIP2A drives neural crest toward a neural stem cell-like fate: Implications for priming of neuroblastoma. Proc. Natl. Acad. Sci. USA 2018, 115, E7351–E7360. [Google Scholar] [CrossRef]
- Pantazopoulos, H.; Woo, T.-U.W.; Lim, M.P.; Lange, N.; Berretta, S. Extracellular matrix-glial abnormalities in the amygdala and entorhinal cortex of subjects diagnosed with schizophrenia. Arch. Gen. Psychiatry 2010, 67, 155–166. [Google Scholar] [CrossRef]
- Sampieri, L.; Funes Chabán, M.; Di Giusto, P.; Rozés-Salvador, V.; Alvarez, C. CREB3L2 Modulates Nerve Growth Fac-tor-Induced Cell Differentiation. Front. Mol. Neurosci. 2021, 14, 650338. [Google Scholar] [CrossRef]
- Shibasaki, K.; Murayama, N.; Ono, K.; Ishizaki, Y.; Tominaga, M. TRPV2 Enhances axon outgrowth through its activation by membrane stretch in developing sensory and motor neurons. J. Neurosci. 2010, 30, 4601–4612. [Google Scholar] [CrossRef] [PubMed]
- Franquinho, F.; Nogueira-Rodrigues, J.; Duarte, J.M.; Esteves, S.S.; Carter-Su, C.; Monaco, A.P.; Molnár, Z.; Velayos-Baeza, A.; Brites, P.; Sousa, M.M. The dyslexia-susceptibility protein kiaa0319 inhibits axon growth through smad2 signaling. Cereb. Cortex 2017, 27, 1732–1747. [Google Scholar] [CrossRef] [PubMed]
- Jósvay, K.; Winter, Z.; Katona, R.L.; Pecze, L.; Marton, A.; Buhala, A.; Szakonyi, G.; Oláh, Z.; Vizler, C. Besides neuro-imaging, the Thy1-YFP mouse could serve for visualizing experimental tumours, inflammation and wound-healing. Sci. Rep. 2014, 4, 6776. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.R.; Kim, D.H.; An, J.Y.; Kang, D.; Park, J.W.; Hwang, E.M.; Seo, E.J.; Jang, I.H.; Ha, C.M.; Lee, B.J. NELL2 function in axon development of hippocampal neurons. Mol. Cells 2020, 43, 581–589. [Google Scholar] [CrossRef]
- Wang, J.; Miao, Y.; Wicklein, R.; Sun, Z.; Wang, J.; Jude, K.M.; Fernandes, R.A.; Merrill, S.A.; Wernig, M.; Garcia, K.C.; et al. RTN4/NoGo-receptor binding to BAI adhesion-GPCRs regulates neuronal development. Cell 2021, 184, 5869–5885. [Google Scholar] [CrossRef]
- Sherchan, P.; Travis, Z.D.; Tang, J.; Zhang, J.H. The potential of Slit2 as a therapeutic target for central nervous system disorders. Expert Opin. Ther. Targets 2020, 24, 805–818. [Google Scholar] [CrossRef]
- Gatto, G.; Dudanova, I.; Suetterlin, P.; Davies, A.M.; Drescher, U.; Bixby, J.L.; Klein, R. Protein tyrosine phosphatase receptor type O inhibits trigeminal axon growth and branching by repressing TrkB and ret signaling. J. Neurosci. 2013, 33, 5399–5410. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Roesener, A.P.; Mendonca, P.R.; Mastick, G.S. Robo1 and Robo2 have distinct roles in pioneer longitudinal axon guidance. Dev. Biol. 2011, 358, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Orioli, D.; Klein, R. The eph receptor family: Axonal guidance by contact repulsion. Trends Genet. 1997, 13, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Corradini, I.; Verderio, C.; Sala, M.; Wilson, M.C.; Matteoli, M. SNAP-25 in neuropsychiatric disorders. Ann. N.Y. Acad. Sci. 2009, 1152, 93–99. [Google Scholar] [CrossRef]
- Ha, S.; Lee, D.; Cho, Y.S.; Chung, C.; Yoo, Y.-E.; Kim, J.; Lee, J.; Kim, W.; Kim, H.; Bae, Y.C.; et al. Cerebellar shank2 regulates excitatory synapse density, motor coordination, and specific repetitive and anxiety-like behaviors. J. Neurosci. 2016, 36, 12129–12143. [Google Scholar] [CrossRef]
- de Wit, J.; Ghosh, A. Control of neural circuit formation by leucine-rich repeat proteins. Trends Neurosci. 2014, 37, 539–550. [Google Scholar] [CrossRef]
- Bustos, F.J.; Ampuero, E.; Jury, N.; Aguilar, R.; Falahi, F.; Toledo, J.; Ahumada, J.; Lata, J.; Cubillos, P.; Henríquez, B.; et al. Epigenetic editing of the Dlg4/PSD95 gene improves cognition in aged and Alzheimer’s disease mice. Brain 2017, 140, 3252–3268. [Google Scholar] [CrossRef]
- Niftullayev, S.; Lamarche-Vane, N. Regulators of rho GTPases in the nervous system: Molecular implication in axon guidance and neurological disorders. Int. J. Mol. Sci. 2019, 20, 1497. [Google Scholar] [CrossRef]
- Chen, Y.; Fu, W.-Y.; Ip, J.P.K.; Ye, T.; Fu, A.K.Y.; Chao, M.V.; Ip, N.Y. Ankyrin repeat-rich membrane spanning protein (Kidins220) is required for neurotrophin and ephrin receptor-dependent dendrite development. J. Neurosci. 2012, 32, 8263–8269. [Google Scholar] [CrossRef]
- Shima, Y.; Kawaguchi, S.-Y.; Kosaka, K.; Nakayama, M.; Hoshino, M.; Nabeshima, Y.; Hirano, T.; Uemura, T. Opposing roles in neurite growth control by two seven-pass transmembrane cadherins. Nat. Neurosci. 2007, 10, 963–969. [Google Scholar] [CrossRef]
- Du, H.; Wang, Z.; Guo, R.; Yang, L.; Liu, G.; Zhang, Z.; Xu, Z.; Tian, Y.; Yang, Z.; Li, X.; et al. Transcription factors Bcl11a and Bcl11b are required for the production and differentiation of cortical projection neurons. Cereb. Cortex 2022, 32, 3611–3632. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, H.C.; O’lEary, A.; Freudenberg, F.; Fedele, G.; Wilkinson, R.; Markham, E.; van Eeden, F.; Reif, A.; Norton, W.H. Nitric oxide interacts with monoamine oxidase to modulate aggression and anxiety-like behaviour. Eur. Neuropsychopharmacol. 2020, 30, 30–43. [Google Scholar] [CrossRef] [PubMed]
- Pinna, A.; Serra, M.; Marongiu, J.; Morelli, M. Pharmacological interactions between adenosine A2A receptor antagonists and different neurotransmitter systems. Park. Relat. Disord. 2020, 80, S37–S44. [Google Scholar] [CrossRef]
- Tan, J.; Xiao, Y.; Kong, F.; Qian, J.; Zhu, A.; Yan, C. Structural insights into thyroid hormone transporter MCT8. Nat. Commun. 2025, 16, 2958. [Google Scholar] [CrossRef]
- Eriksen, J.; Li, F.; Edwards, R.H. The mechanism and regulation of vesicular glutamate transport: Coordination with the synaptic vesicle cycle. Biochim. Biophys. Acta-Biomembr. 2020, 1862, 183259. [Google Scholar] [CrossRef]
- Pizzagalli, M.D.; Bensimon, A.; Superti-Furga, G. A guide to plasma membrane solute carrier proteins. FEBS J. 2021, 288, 2784–2835. [Google Scholar] [CrossRef]
- Batten, S.R.; Matveeva, E.A.; Whiteheart, S.W.; Vanaman, T.C.; Gerhardt, G.A.; Slevin, J.T. Linking kindling to increased glutamate release in the dentate gyrus of the hippocampus through the STXBP5/tomosyn-1 gene. Brain Behav. 2017, 7, e00795. [Google Scholar] [CrossRef]
- Um, J.W.; Choii, G.; Park, D.; Kim, D.; Jeon, S.; Kang, H.; Mori, T.; Papadopoulos, T.; Yoo, T.; Lee, Y.; et al. IQ Motif and SEC7 domain-containing protein 3 (IQSEC3) interacts with gephyrin to promote inhibitory synapse formation. J. Biol. Chem. 2016, 291, 10119–10130. [Google Scholar] [CrossRef]
- Köster, J.-D.; Leggewie, B.; Blechner, C.; Brandt, N.; Fester, L.; Rune, G.; Schweizer, M.; Kindler, S.; Windhorst, S. Inositol-1,4,5-trisphosphate-3-kinase-A controls morphology of hippocampal dendritic spines. Cell. Signal. 2016, 28, 83–90. [Google Scholar] [CrossRef]
- Matt, L.; Kim, K.; Chowdhury, D.; Hell, J.W. Role of palmitoylation of postsynaptic proteins in promoting synaptic plasticity. Front. Mol. Neurosci. 2019, 12, 8. [Google Scholar] [CrossRef]
- Paul, M.S.; Michener, S.L.; Pan, H.; Chan, H.; Pfliger, J.M.; Rosenfeld, J.A.; Lerma, V.C.; Tran, A.; Longley, M.A.; Lewis, R.A.; et al. A syndromic neurodevelopmental disorder caused by rare variants in PPFIA3. Am. J. Hum. Genet. 2024, 111, 96–118. [Google Scholar] [CrossRef] [PubMed]
- Okerlund, N.D.; Kivimäe, S.; Tong, C.K.; Peng, I.-F.; Ullian, E.M.; Cheyette, B.N.R. Dact1 is a postsynaptic protein required for dendrite, spine, and excitatory synapse development in the mouse forebrain. J. Neurosci. 2010, 30, 4362–4368. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhang, Y.; Yu, T.; Li, N.; Wang, J.; Wang, J.; Ge, Y.; Yao, R. Psychomotor development and attention problems caused by a splicing variant of CNKSR2. BMC Med. Genom. 2020, 13, 182. [Google Scholar] [CrossRef]
- Kwon, S.E.; Chapman, E.R. Synaptophysin Regulates the Kinetics of Synaptic Vesicle Endocytosis in Central Neurons. Neuron 2011, 70, 847–854. [Google Scholar] [CrossRef]
- Barman, A.; Assmann, A.; Richter, S.; Soch, J.; Schã¼Tze, H.; Wã¼Stenberg, T.; Deibele, A.; Klein, M.; Richter, A.; Behnisch, G.; et al. Genetic variation of the RASGRF1 regulatory region affects human hippocampus-dependent memory. Front. Hum. Neurosci. 2014, 8, 260. [Google Scholar] [CrossRef]
- Ismail, V.; Zachariassen, L.G.; Godwin, A.; Sahakian, M.; Ellard, S.; Stals, K.L.; Baple, E.; Brown, K.T.; Foulds, N.; Wheway, G.; et al. Identification and functional evaluation of GRIA1 missense and truncation variants in individuals with ID: An emerging neurodevelopmental syndrome. Am. J. Hum. Genet. 2022, 109, 1217–1241. [Google Scholar] [CrossRef]
- Camp, C.R.; Yuan, H. GRIN2D/GluN2D NMDA receptor: Unique features and its contribution to pediatric developmental and epileptic encephalopathy. Eur. J. Paediatr. Neurol. 2020, 24, 89–99. [Google Scholar] [CrossRef]
- Rigter, P.M.; de Konink, C.; Dunn, M.J.; Onori, M.P.; Humberson, J.B.; Thomas, M.; Barnes, C.; Prada, C.E.; Weaver, K.N.; Ryan, T.D.; et al. Role of CAMK2D in neurodevelopment and associated conditions. Am. J. Hum. Genet. 2024, 111, 364–382. [Google Scholar] [CrossRef]
- Suthar, S.K.; Alam, M.M.; Lee, J.; Monga, J.; Joseph, A.; Lee, S.-Y. Bioinformatic Analyses of Canonical Pathways of TSPOAP1 and its Roles in Human Diseases. Front. Mol. Biosci. 2021, 8, 667947. [Google Scholar] [CrossRef]
- Wu, L.-Y.; Song, Y.-J.; Zhang, C.-L.; Liu, J. KV Channel-Interacting Proteins in the Neurological and Cardiovascular Systems: An Updated Review. Cells 2023, 12, 1894. [Google Scholar] [CrossRef]
- Geering, K. FXYD proteins: New regulators of Na-K-ATPase. Am. J. Physiol.-Physiol. 2006, 290, F241–F250. [Google Scholar] [CrossRef] [PubMed]
- Murata, K.; Kinoshita, T.; Ishikawa, T.; Kuroda, K.; Hoshi, M.; Fukazawa, Y. Region- and neuronal-subtype-specific expression of Na,K-ATPase alpha and beta subunit isoforms in the mouse brain. J. Comp. Neurol. 2020, 528, 2654–2678. [Google Scholar] [CrossRef] [PubMed]
- Strehler, E.E.; Thayer, S.A. Evidence for a role of plasma membrane calcium pumps in neurodegenerative disease: Recent developments. Neurosci. Lett. 2018, 663, 39–47. [Google Scholar] [CrossRef]
- Cusick, J.K.; Alcaide, J.; Shi, Y. The RELT Family of Proteins: An Increasing Awareness of Their Importance for Cancer, the Immune System, and Development. Biomedicines 2023, 11, 2695. [Google Scholar] [CrossRef]
- Ilic, K.; Auer, B.; Mlinac-Jerkovic, K.; Herrera-Molina, R. Neuronal signaling by thy-1 in nanodomains with specific ganglioside composition: Shall we open the door to a new complexity? Front. Cell Dev. Biol. 2019, 7, 27. [Google Scholar] [CrossRef]
- Huang, J.Y.; Wang, K.; Vermehren-Schmaedick, A.; Adelman, J.P.; Cohen, M.S. PARP6 is a Regulator of Hippocampal Dendritic Morphogenesis. Sci. Rep. 2016, 6, 18512. [Google Scholar] [CrossRef]
- Tsou, J.-H.; Yang, Y.-C.; Pao, P.-C.; Lin, H.-C.; Huang, N.-K.; Lin, S.-T.; Hsu, K.-S.; Yeh, C.-M.; Lee, K.-H.; Kuo, C.-J.; et al. Important Roles of Ring Finger Protein 112 in Embryonic Vascular Development and Brain Functions. Mol. Neurobiol. 2017, 54, 2286–2300. [Google Scholar] [CrossRef]
- Lin, T.-Y.; Wong, L.-C.; Hou, P.-S.; Wu, C.-K.; Cheng, H.-Y.; Zhao, H.-J.; Tung, C.-Y.; Lee, M.-H.; Lee, W.-T.; Tsai, J.-W. Functional defects in FOXG1 variants predict the severity of brain anomalies in FOXG1 syndrome. Mol. Psychiatry 2025, 30, 4824–4835. [Google Scholar] [CrossRef]
- Funa, K.; Sasahara, M. The roles of PDGF in development and during neurogenesis in the normal and diseased nervous system. J. Neuroimmune Pharmacol. 2014, 9, 168–181. [Google Scholar] [CrossRef]
- Brawley, C.M.; Uysal, S.; Kossiakoff, A.A.; Rock, R.S. Characterization of engineered actin binding proteins that control filament assembly and structure. PLoS ONE 2010, 5, e13960. [Google Scholar] [CrossRef]
- Stark, B.C.; Lanier, M.H.; Cooper, J.A. CARMIL family proteins as multidomain regulators of actin-based motility. Mol. Biol. Cell 2017, 28, 1713–1723. [Google Scholar] [CrossRef]
- Altas, B.; Romanowski, A.J.; Bunce, G.W.; Poulopoulos, A. Neuronal mTOR Outposts: Implications for Translation, Signaling, and Plasticity. Front. Cell. Neurosci. 2022, 16, 853634. [Google Scholar] [CrossRef]
- Chouinard, F.C.; Davis, L.; Gilbert, C.; Bourgoin, S.G. Functional Role of AGAP2/PIKE-A in Fcγ Receptor-Mediated Phag-ocytosis. Cells 2023, 12, 72. [Google Scholar] [CrossRef]
- Zhang, L.; Cao, Y.; Guo, X.; Wang, X.; Han, X.; Kanwore, K.; Hong, X.; Zhou, H.; Gao, D. Hypoxia-induced ROS aggravate tumor progression through HIF-1α-SERPINE1 signaling in glioblastoma. J. Zhejiang Univ. Sci. B 2023, 24, 32–49. [Google Scholar] [CrossRef]
- Chen, H.; Ma, D.; Yue, F.; Qi, Y.; Dou, M.; Cui, L.; Xing, Y. The Potential Role of Hypoxia-Inducible Factor-1 in the Progression and Therapy of Central Nervous System Diseases. Curr. Neuropharmacol. 2021, 20, 1651–1666. [Google Scholar] [CrossRef]
- Foxler, D.E.; Bridge, K.S.; Foster, J.G.; Grevitt, P.; Curry, S.; Shah, K.M.; Davidson, K.M.; Nagano, A.; Gadaleta, E.; Rhys, H.I.; et al. A HIF–LIMD 1 negative feedback mechanism mitigates the pro-tumorigenic effects of hypoxia. EMBO Mol. Med. 2018, 10, e8304. [Google Scholar] [CrossRef]
- Salma, J.; McDermott, J.C. Suppression of a MEF2-KLF6 survival pathway by PKA signaling promotes apoptosis in embryonic hippocampal neurons. J. Neurosci. 2012, 32, 2790–2803. [Google Scholar] [CrossRef] [PubMed]
- Bryan, L.; Kordula, T.; Spiegel, S.; Milstien, S. Regulation and functions of sphingosine kinases in the brain. Biochim. Biophys. Acta-Mol. Cell Biol. Lipids 2008, 1781, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Yue, W. VRK2, a Candidate Gene for Psychiatric and Neurological Disorders. Complex Psychiatry 2018, 4, 119–133. [Google Scholar] [CrossRef]
- Takata, S.; Sakata-Haga, H.; Shimada, H.; Tsukada, T.; Sakai, D.; Shoji, H.; Tomosugi, M.; Nakamura, Y.; Ishigaki, Y.; Iizuka, H.; et al. LIF–IGF Axis Contributes to the Proliferation of Neural Progenitor Cells in Developing Rat Cerebrum. Int. J. Mol. Sci. 2022, 23, 13199. [Google Scholar] [CrossRef]
- Joo, J.-Y.; Schaukowitch, K.; Farbiak, L.; Kilaru, G.; Kim, T.-K. Stimulus-specific combinatorial functionality of neuronal c-fos enhancers. Nat. Neurosci. 2015, 19, 75–83. [Google Scholar] [CrossRef]
- Lu, C.; Jiang, Y.; Xu, W.; Bao, X. Sestrin2: Multifaceted functions, molecular basis, and its implications in liver diseases. Cell Death Dis. 2023, 14, 160. [Google Scholar] [CrossRef]
- Baur, K.; Carrillo-García, C.; Şan, Ş.; von Hahn, M.; Strelau, J.; Hölzl-Wenig, G.; Mandl, C.; Ciccolini, F. Growth/differentiation factor 15 controls ependymal and stem cell number in the V-SVZ. Stem Cell Rep. 2024, 19, 351–365. [Google Scholar] [CrossRef]
- Li, L.; Mo, H.; Zhang, J.; Zhou, Y.; Peng, X.; Luo, X. The role of heat shock protein 90B1 in patients with polycystic ovary syndrome. PLoS ONE 2016, 11, e0152837. [Google Scholar] [CrossRef]
- Moon, B.-S.; Yoon, J.-Y.; Kim, M.-Y.; Lee, S.-H.; Choi, T.; Choi, K.-Y. Bone morphogenetic protein 4 stimulates neuronal differentiation of neuronal stem cells through the ERK pathway. Exp. Mol. Med. 2009, 41, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.-Y.; Zukin, R.S. REST, a master transcriptional regulator in neurodegenerative disease. Curr. Opin. Neurobiol. 2018, 48, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Peiris, M.; Hockley, J.R.; E Reed, D.; Smith, E.S.J.; Bulmer, D.C.; Blackshaw, L.A. Peripheral Kv7 channels regulate visceral sensory function in mouse and human colon. Mol. Pain 2017, 13, 1744806917709371. [Google Scholar] [CrossRef]
- Zhong, L.; Fang, S.; Wang, A.-Q.; Zhang, Z.-H.; Wang, T.; Huang, W.; Zhou, H.-X.; Zhang, H.; Yin, Z.-S. Identification of the Fosl1/AMPK/autophagy axis involved in apoptotic and inflammatory effects following spinal cord injury. Int. Immunopharmacol. 2022, 103, 108492. [Google Scholar] [CrossRef]
- Singh, K.; Han, C.; Fleming, J.L.; Becker, A.P.; McElroy, J.; Cui, T.; Johnson, B.; Kumar, A.; Sebastian, E.; Showalter, C.A.; et al. TRIB1 confers therapeutic resistance in GBM cells by activating the ERK and Akt pathways. Sci. Rep. 2023, 13, 12424. [Google Scholar] [CrossRef]
- Navarro, E.; Esteras, N. Multitarget Effects of Nrf2 Signalling in the Brain: Common and Specific Functions in Different Cell Types. Antioxidants 2024, 13, 1502. [Google Scholar] [CrossRef]
- Kim, S.; Lee, W.; Jo, H.; Sonn, S.-K.; Jeong, S.-J.; Seo, S.; Suh, J.; Jin, J.; Kweon, H.Y.; Kim, T.K.; et al. The antioxidant enzyme Peroxiredoxin-1 controls stroke-associated microglia against acute ischemic stroke. Redox Biol. 2022, 54, 102347. [Google Scholar] [CrossRef]
- Biermanns, M.; Gärtner, J. Genomic organization and characterization of human PEX2 encoding a 35-kDa peroxisomal membrane protein. Biochem. Biophys. Res. Commun. 2000, 273, 985–990. [Google Scholar] [CrossRef]
- Winship, A.; Sorby, K.; Correia, J.; Rainczuk, A.; Yap, J.; Dimitriadis, E. Interleukin-11 up-regulates endoplasmic reticulum stress induced target, PDIA4 in human first trimester placenta and in vivo in mice. Placenta 2017, 53, 92–100. [Google Scholar] [CrossRef]
- Petrova, K.; Oyadomari, S.; Hendershot, L.M.; Ron, D. Regulated association of misfolded endoplasmic reticulum lumenal proteins with P58/DNAJc3. EMBO J. 2008, 27, 2862–2872. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, L.P.d.S.; da Silva, R.A.; Borges, P.P.; Xavier, L.F.; Scharf, P.; Sandri, S.; Oliani, S.M.; Farsky, S.H.; Gil, C.D. Annexin A1 in neurological disorders: Neuroprotection and glial modulation. Pharmacol. Ther. 2025, 267, 108809. [Google Scholar] [CrossRef] [PubMed]
- Kollárovič, G.; E Topping, C.; Shaw, E.P.; Chambers, A.L. The human HELLS chromatin remodelling protein promotes end resection to facilitate homologous recombination and contributes to DSB repair within heterochromatin. Nucleic Acids Res. 2020, 48, 1872–1885. [Google Scholar] [CrossRef]
- Jin, H.; Suh, D.-S.; Kim, T.-H.; Yeom, J.-H.; Lee, K.; Bae, J. IER3 is a crucial mediator of TAp73β-induced apoptosis in cervical cancer and confers etoposide sensitivity. Sci. Rep. 2015, 5, 8367. [Google Scholar] [CrossRef]
- Raoul, C.; Estévez, A.G.; Nishimune, H.; Cleveland, D.W.; deLapeyrière, O.; Henderson, C.E.; Haase, G.; Pettmann, B. Motoneuron death triggered by a specific pathway downstream of fas: Potentiation by ALS-linked SOD1 mutations. Neuron 2002, 35, 1067–1083. [Google Scholar] [CrossRef]
- Chen, W.; Wang, H.; Tao, S.; Zheng, Y.; Wu, W.; Lian, F.; Jaramillo, M.; Fang, D.; Zhang, D.D. Tumor protein translationally controlled 1 is a p53 target gene that promotes cell survival. Cell Cycle 2013, 12, 2321–2328. [Google Scholar] [CrossRef]
- Lozinski, B.M.; Ta, K.; Dong, Y. Emerging role of galectin 3 in neuroinflammation and neurodegeneration. Neural Regen. Res. 2024, 19, 2004–2009. [Google Scholar] [CrossRef]
- Hausott, B.; Klimaschewski, L. Sprouty2—A Novel Therapeutic Target in the Nervous System? Mol. Neurobiol. 2019, 56, 3897–3903. [Google Scholar] [CrossRef]
- Cheng, Z.; Zou, X.; Jin, Y.; Gao, S.; Lv, J.; Li, B.; Cui, R. The role of KLF4 in Alzheimer’s disease. Front. Cell. Neurosci. 2018, 12, 325. [Google Scholar] [CrossRef]
- Hao, Q.; Liu, Y.; Liu, Y.; Shi, L.; Chen, Y.; Yang, L.; Jiang, Z.; Liu, Y.; Wang, C.; Wang, S.; et al. Cysteine- and glycine-rich protein 1 predicts prognosis and therapy response in patients with acute myeloid leukemia. Clin. Exp. Med. 2024, 24, 57. [Google Scholar] [CrossRef]
- Jiang, D.; de Vries, J.C.; Muschhammer, J.; Schatz, S.; Ye, H.; Hein, T.; Fidan, M.; Romanov, V.S.; Rinkevich, Y.; Scharffetter-Kochanek, K. Local and transient inhibition of p21 expression ameliorates age-related delayed wound healing. Wound Repair Regen. 2020, 28, 49–60. [Google Scholar] [CrossRef]
- Gambarotta, G.; El Soury, M. Soluble neuregulin-1 (NRG1): A factor promoting peripheral nerve regeneration by affecting Schwann cell activity immediately after injury. Neural Regen. Res. 2019, 14, 1374–1375. [Google Scholar] [CrossRef]
- Srivastava, S.; Ramdass, B.; Nagarajan, S.; Rehman, M.; Mukherjee, G.; Krishna, S. Notch1 regulates the functional contribution of RhoC to cervical carcinoma progression. Br. J. Cancer 2010, 102, 196–205. [Google Scholar] [CrossRef]
- Yuan, J.; Yu, J. Human tissue factor pathway inhibitor-2 suppresses the wound-healing activities of human Tenon’s capsule fibroblasts in vitro. Mol. Vis. 2009, 15, 2306–2312. [Google Scholar]
- Farooq, M.; Khan, A.W.; Kim, M.S.; Choi, S. The role of fibroblast growth factor (FGF) signaling in tissue repair and regeneration. Cells 2021, 10, 3242. [Google Scholar] [CrossRef] [PubMed]
- Takouda, J.; Katada, S.; Imamura, T.; Sanosaka, T.; Nakashima, K. SoxE group transcription factor Sox8 promotes astrocytic differentiation of neural stem/precursor cells downstream of Nfia. Pharmacol. Res. Perspect. 2021, 9, e00749. [Google Scholar] [CrossRef]
- Kim, J.; Lo, L.; Dormand, E.; Anderson, D.J. SOX10 maintains multipotency and inhibits neuronal differentiation of neural crest stem cells. Neuron 2003, 38, 17–31. [Google Scholar] [CrossRef]
- Warner, L.E.; Mancias, P.; Butler, I.J.; McDonald, C.M.; Keppen, L.; Koob, K.G.; Lupski, J.R. Mutations in the early growth response 2 (EGR2) gene are associated with hereditary myelinopathies. Nat. Genet. 1998, 18, 382–384. [Google Scholar] [CrossRef]
- Greene, L.A.; Lee, H.Y.; Angelastro, J.M. The transcription factor ATF5: Role in neurodevelopment and neural tumors. J. Neurochem. 2009, 108, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Briscoe, J.; Sussel, L.; Serup, P.; Hartigan-O’Connor, D.; Jessell, T.M.; Rubenstein, J.L.R.; Ericson, J. Homeobox gene Nkx2.2 and specification of neuronal identity by graded Sonic hedgehog signalling. Nature 1999, 398, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Tu, M.; Zhu, P.; Hu, S.; Wang, W.; Su, Z.; Guan, J.; Sun, C.; Zheng, W. Notch1 signaling activation contributes to adult hippocampal neurogenesis following traumatic brain injury. Med. Sci. Monit. 2017, 23, 5480–5487. [Google Scholar] [CrossRef]
- Escobedo, N.; Contreras, O.; Muñoz, R.; Farías, M.; Carrasco, H.; Hill, C.; Tran, U.; Pryor, S.E.; Wessely, O.; Copp, A.J.; et al. Syndecan 4 interacts genetically with Vangl2 to regulate neural tube closure and planar cell polarity. Development 2013, 140, 3008–3017. [Google Scholar] [CrossRef]
- Wu, Y.; Peng, H.; Cui, M.; Whitney, N.P.; Huang, Y.; Zheng, J.C. CXCL12 increases human neural progenitor cell proliferation through Akt-1/FOXO3a signaling pathway. J. Neurochem. 2009, 109, 1157–1167. [Google Scholar] [CrossRef]
- Chi, S.; Cui, Y.; Wang, H.; Jiang, J.; Zhang, T.; Sun, S.; Zhou, Z.; Zhong, Y.; Xiao, B. Astrocytic Piezo1-mediated mechanotransduction determines adult neurogenesis and cognitive functions. Neuron 2022, 110, 2984–2999.e8. [Google Scholar] [CrossRef]
- Tamura, R.E.; de Vasconcellos, J.F.; Sarkar, D.; Libermann, T.A.; Fisher, P.B.; Zerbini, L.F. GADD45 Proteins: Central Players in Tumorigenesis. Curr. Mol. Med. 2012, 12, 634–651. [Google Scholar] [CrossRef]
- Liu, C.; Chen, L.; Cong, Y.; Cheng, L.; Shuai, Y.; Lv, F.; Chen, K.; Song, Y.; Xing, Y. Protein phosphatase 1 regulatory subunit 15 A promotes translation initiation and induces G2M phase arrest during cuproptosis in cancers. Cell Death Dis. 2024, 15, 149. [Google Scholar] [CrossRef]
- Grubbs, E.G.; Williams, M.D.; Scheet, P.; Vattathil, S.; Perrier, N.D.; Lee, J.E.; Gagel, R.F.; Hai, T.; Feng, L.; Cabanillas, M.E.; et al. Role of CDKN2C copy number in sporadic medullary thyroid carcinoma. Thyroid® 2016, 26, 1553–1562. [Google Scholar] [CrossRef]
- Rogers, C.D.; Phillips, J.L.; Bronner, M.E. Elk3 is essential for the progression from progenitor to definitive neural crest cell. Dev. Biol. 2013, 374, 255–263. [Google Scholar] [CrossRef]
- Rudenok, M.M.; Shadrina, M.I.; Filatova, E.V.; Rybolovlev, I.N.; Nesterov, M.S.; Abaimov, D.A.; Ageldinov, R.A.; Kolacheva, A.A.; Ugrumov, M.V.; Slominsky, P.A.; et al. Expression Analysis of Genes Involved in Transport Processes in Mice with MPTP-Induced Model of Parkinson’s Disease. Life 2022, 12, 751. [Google Scholar] [CrossRef]
- Ould-Yahoui, A.; Tremblay, E.; Sbai, O.; Ferhat, L.; Bernard, A.; Charrat, E.; Gueye, Y.; Lim, N.H.; Brew, K.; Risso, J.-J.; et al. A new role for TIMP-1 in modulating neurite outgrowth and morphology of cortical neurons. PLoS ONE 2009, 4, e8289. [Google Scholar] [CrossRef]
- Huang, T.; Fu, G.; Gao, J.; Zhang, Y.; Cai, W.; Wu, S.; Jia, S.; Xia, S.; Bachmann, T.; Bekker, A.; et al. Fgr contributes to hemorrhage-induced thalamic pain by activating NF-κB/ERK1/2 pathways. J. Clin. Investig. 2020, 5, e139987. [Google Scholar] [CrossRef]
- Islam, M.I.; Sultana, S.; Padmanabhan, N.; Rashid, M.-U.; Siddiqui, T.J.; Coombs, K.M.; Vitiello, P.F.; Karimi-Abdolrezaee, S.; Eftekharpour, E. Thioredoxin-1 protein interactions in neuronal survival and neurodegeneration. Biochim. Biophys. Acta-Mol. Basis Dis. 2025, 1871, 167548. [Google Scholar] [CrossRef]
- Mohammadzadeh, P.; Amberg, G.C. AXL/Gas6 signaling mechanisms in the hypothalamic-pituitary-gonadal axis. Front. Endocrinol. 2023, 14, 1212104. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.-L.; Li, Z.-Y.; Song, J.; Liu, J.-M.; Miao, C.-Y. Metrnl: A secreted protein with new emerging functions. Acta Pharmacol. Sin. 2016, 37, 571–579. [Google Scholar] [CrossRef]
- Fischer-Huchzermeyer, S.; Dombrowski, A.; Hagel, C.; Mautner, V.F.; Schittenhelm, J.; Harder, A. The Cellular Retinoic Acid Binding Protein 2 Promotes Survival of Malignant Peripheral Nerve Sheath Tumor Cells. Am. J. Pathol. 2017, 187, 1623–1632. [Google Scholar] [CrossRef]
- Schaeffer, J.; Tannahill, D.; Cioni, J.-M.; Rowlands, D.; Keynes, R. Identification of the extracellular matrix protein Fibulin-2 as a regulator of spinal nerve organization. Dev. Biol. 2018, 442, 101–114. [Google Scholar] [CrossRef]
- Nonaka, M.; Fukuda, M. Galectin-1 for Neuroprotection? Immunity 2012, 37, 187–189. [Google Scholar] [CrossRef]
- Chen, M.X.; Oh, Y.-S.; Kim, Y. S100A10 and its binding partners in depression and antidepressant actions. Front. Mol. Neurosci. 2022, 15, 953066. [Google Scholar] [CrossRef]
- Iwanicka, J.; Balcerzyk-Matić, A.; Iwanicki, T.; Mizia-Stec, K.; Bańka, P.; Filipecki, A.; Gawron, K.; Jarosz, A.; Nowak, T.; Krauze, J.; et al. The Association of ADAMTS7 Gene Polymorphisms with the Risk of Coronary Artery Disease Occurrence and Cardiovascular Survival in the Polish Population: A Case-Control and a Prospective Cohort Study. Int. J. Mol. Sci. 2024, 25, 2274. [Google Scholar] [CrossRef] [PubMed]
- Pagnamenta, A.T.; Kaiyrzhanov, R.; Zou, Y.; Da’As, S.I.; Maroofian, R.; Donkervoort, S.; Dominik, N.; Lauffer, M.; Ferla, M.P.; Orioli, A.; et al. An ancestral 10-bp repeat expansion in VWA1 causes recessive hereditary motor neuropathy. Brain 2021, 144, 584–600. [Google Scholar] [CrossRef]
- Konietzny, A.; Bär, J.; Mikhaylova, M. Dendritic actin cytoskeleton: Structure, functions, and regulations. Front. Cell. Neurosci. 2017, 11, 147. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.C.; Andrews, P.C.; Asselbergs, F.W.; Frost, H.R.; Williams, S.M.; Harris, B.T.; Read, C.; Askland, K.D.; Moore, J.H. Gene ontology analysis of pairwise genetic associations in two genome-wide studies of sporadic ALS. BioData Min. 2012, 5, 9. [Google Scholar] [CrossRef]
- Notter, T.; Schalbetter, S.M.; Clifton, N.E.; Mattei, D.; Richetto, J.; Thomas, K.; Meyer, U.; Hall, J. Neuronal activity increases translocator protein (TSPO) levels. Mol. Psychiatry 2021, 26, 2025–2037. [Google Scholar] [CrossRef]
- Silbereis, J.C.; Nobuta, H.; Tsai, H.-H.; Heine, V.M.; McKinsey, G.L.; Meijer, D.H.; Howard, M.A.; Petryniak, M.A.; Potter, G.B.; Alberta, J.A.; et al. Olig1 Function Is Required to Repress Dlx1/2 and Interneuron Production in Mammalian Brain. Neuron 2014, 81, 574–587. [Google Scholar] [CrossRef]
- José, N.G.d.S.; Massa, F.; Halbgebauer, S.; Oeckl, P.; Steinacker, P.; Otto, M. Neuronal pentraxins as biomarkers of synaptic activity: From physiological functions to pathological changes in neurodegeneration. J. Neural Transm. 2022, 129, 207–230. [Google Scholar] [CrossRef]
- Uzor, N.-E.; Scheihing, D.M.; Kim, G.S.; Moruno-Manchon, J.F.; Zhu, L.; Reynolds, C.R.; Stephenson, J.M.; Holmes, A.; McCullough, L.D.; Tsvetkov, A.S. Aging lowers PEX5 levels in cortical neurons in male and female mouse brains. Mol. Cell. Neurosci. 2020, 107, 103536. [Google Scholar] [CrossRef]
- Chapleau, A.; Boucher, R.-M.; Pastinen, T.; Thiffault, I.; Gould, P.V.; Bernard, G. Neuropathological characterization of the cavitating leukoencephalopathy caused by COA8 cytochrome c oxidase deficiency: A case report. Front. Cell. Neurosci. 2023, 17, 1216487. [Google Scholar] [CrossRef]
- Mucha, M.; Skrzypiec, A.E.; Schiavon, E.; Attwood, B.K.; Kucerova, E.; Pawlak, R. Lipocalin-2 controls neuronal excitability and anxiety by regulating dendritic spine formation and maturation. Proc. Natl. Acad. Sci. USA 2011, 108, 18436–18441. [Google Scholar] [CrossRef] [PubMed]
- Kolobynina, K.G.; Solovyova, V.V.; Levay, K.; Rizvanov, A.A.; Slepak, V.Z. Emerging roles of the single EF-hand Ca2+ sensor tescalcin in the regulation of gene expression, cell growth and differentiation. J. Cell Sci. 2016, 129, 3533–3540. [Google Scholar] [CrossRef] [PubMed]
- Nixon, B.; Bromfield, E.G.; Dun, M.D.; Redgrove, K.; McLaughlin, E.; Aitken, R.J. The role of the molecular chaperone heat shock protein A2 (HSPA2) in regulating human sperm-egg recognition. Asian J. Androl. 2015, 17, 568–573. [Google Scholar] [CrossRef]
- Wang, J.; Owji, A.P.; Kittredge, A.; Clark, Z.; Zhang, Y.; Yang, T. GAD65 tunes the functions of Best1 as a GABA receptor and a neurotransmitter conducting channel. Nat. Commun. 2024, 15, 8051. [Google Scholar] [CrossRef]
- Gotliv, I.L. FXYD5: Na+/K+-ATPase regulator in health and disease. Front. Cell Dev. Biol. 2016, 4, 26. [Google Scholar] [CrossRef]
- Shen, J.; Shi, D.; Suzuki, T.; Xia, Z.; Zhang, H.; Araki, K.; Wakana, S.; Takeda, N.; Yamamura, K.-I.; Jin, S.; et al. Severe ocular phenotypes in Rbp4-deficient mice in the C57BL/6 genetic background. Mod. Pathol. 2016, 96, 680–691. [Google Scholar] [CrossRef]
- Reichmann, F.; Holzer, P. Neuropeptide Y: A stressful review. Neuropeptides 2016, 55, 99–109. [Google Scholar] [CrossRef]
- Zhu, Z.; Bolt, E.; Newmaster, K.; Osei-Bonsu, W.; Cohen, S.; Cuddapah, V.A.; Gupta, S.; Paudel, S.; Samanta, D.; Dang, L.T.; et al. SCN1B Genetic Variants: A Review of the Spectrum of Clinical Phenotypes and a Report of Early Myoclonic Encephalopathy. Children 2022, 9, 1507. [Google Scholar] [CrossRef]
- Pang, W.; Yi, X.; Li, L.; Liu, L.; Xiang, W.; Xiao, L. Untangle the Multi-Facet Functions of Auts2 as an Entry Point to Understand Neurodevelopmental Disorders. Front. Psychiatry 2021, 12, 580433. [Google Scholar] [CrossRef]
- Wagner, M.; Lévy, J.; Jung-Klawitter, S.; Bakhtiari, S.; Monteiro, F.; Maroofian, R.; Bierhals, T.; Hempel, M.; Elmaleh-Bergès, M.; Kitajima, J.P.; et al. Loss of TNR causes a nonprogressive neurodevelopmental disorder with spasticity and transient opisthotonus. Anesthesia Analg. 2020, 22, 1061–1068. [Google Scholar] [CrossRef]
- Ageta-Ishihara, N.; Kinoshita, M. Developmental and postdevelopmental roles of septins in the brain. Neurosci. Res. 2021, 170, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Cukier, H.N.; Duarte, C.L.; Laverde-Paz, M.J.; Simon, S.A.; Van Booven, D.J.; Miyares, A.T.; Whitehead, P.L.; Hamilton-Nelson, K.L.; Adams, L.D.; Carney, R.M.; et al. An Alzheimer’s disease risk variant in TTC3 modifies the actin cytoskeleton organization and the PI3K-Akt signaling pathway in iPSC-derived forebrain neurons. Neurobiol. Aging 2023, 131, 182–195. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, S.D.-S.; Moreau, M.M.; Hien, Y.E.; Garcia, M.; Aubailly, N.; Henderson, D.J.; Studer, V.; Sans, N.; Thoumine, O.; Montcouquiol, M. Vangl2 acts at the interface between actin and N-cadherin to modulate mammalian neuronal outgrowth. eLife 2020, 9, e51822. [Google Scholar] [CrossRef] [PubMed]
- Rolando, C.; Erni, A.; Grison, A.; Beattie, R.; Engler, A.; Gokhale, P.J.; Milo, M.; Wegleiter, T.; Jessberger, S.; Taylor, V. Multipotency of Adult Hippocampal NSCs In Vivo Is Restricted by Drosha/NFIB. Cell Stem Cell 2016, 19, 653–662. [Google Scholar] [CrossRef]
- Ruiz-Gabarre, D.; Carnero-Espejo, A.; Ávila, J.; García-Escudero, V. What’s in a Gene? The Outstanding Diversity of MAPT. Cells 2022, 11, 840. [Google Scholar] [CrossRef]
- Li, W.; Cheng, T.; Dong, X.; Chen, H.; Yang, L.; Qiu, Z.; Zhou, W. KIF5C deficiency causes abnormal cortical neuronal migration, dendritic branching, and spine morphology in mice. Pediatr. Res. 2022, 92, 995–1002. [Google Scholar] [CrossRef]
- Sakabe, I.; Hu, R.; Jin, L.; Clarke, R.; Kasid, U.N. TMEM33: A new stress-inducible endoplasmic reticulum transmembrane protein and modulator of the unfolded protein response signaling. Breast Cancer Res. Treat. 2015, 153, 285–297. [Google Scholar] [CrossRef]
- Martínez, J.C.; Randolph, L.K.; Iascone, D.M.; Pernice, H.F.; Polleux, F.; Hengst, U. Pum2 Shapes the Transcriptome in Developing Axons through Retention of Target mRNAs in the Cell Body. Neuron 2019, 104, 931–946.e5. [Google Scholar] [CrossRef]
- Desole, C.; Gallo, S.; Vitacolonna, A.; Montarolo, F.; Bertolotto, A.; Vivien, D.; Comoglio, P.; Crepaldi, T. HGF and MET: From Brain Development to Neurological Disorders. Front. Cell Dev. Biol. 2021, 9, 683609. [Google Scholar] [CrossRef]
- Łuczyńska, K.; Zhang, Z.; Pietras, T.; Zhang, Y.; Taniguchi, H. NFE2L1/Nrf1 serves as a potential therapeutical target for neurodegenerative diseases. Redox Biol. 2024, 69, 103003. [Google Scholar] [CrossRef]
- Higelin, J.; Catanese, A.; Semelink-Sedlacek, L.L.; Oeztuerk, S.; Lutz, A.-K.; Bausinger, J.; Barbi, G.; Speit, G.; Andersen, P.M.; Ludolph, A.C.; et al. NEK1 loss-of-function mutation induces DNA damage accumulation in ALS patient-derived motoneurons. Stem Cell Res. 2018, 30, 150–162. [Google Scholar] [CrossRef] [PubMed]
- Santo, E.E.; Paik, J. FOXO in Neural Cells and Diseases of the Nervous System. In Current Topics in Developmental Biology; Elsevier BV: Amsterdam, The Netherlands, 2018; Volume 127, pp. 105–118. [Google Scholar]
- Klopf, E.; Schmidt, H.A.; Clauder-Münster, S.; Steinmetz, L.M.; Schüller, C. INO80 represses osmostress induced gene expression by resetting promoter proximal nucleosomes. Nucleic Acids Res. 2017, 45, 3752–3766. [Google Scholar] [CrossRef] [PubMed]
- Yue, W.; Zhang, K.; Jiang, M.; Long, W.; Cui, J.; Li, Y.; Zhang, Y.; Li, A.; Fang, Y. Deubiquitination of SARM1 by USP13 regulates SARM1 activation and axon degeneration. Life Med. 2023, 2, lnad040. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhao, Y. Progress on the roles of MEF2C in neuropsychiatric diseases. Mol. Brain 2022, 15, 8. [Google Scholar] [CrossRef]
- Renkilaraj, M.R.L.M.; Baudouin, L.; Wells, C.M.; Doulazmi, M.; Wehrlé, R.; Cannaya, V.; Bachelin, C.; Barnier, J.-V.; Jia, Z.; Oumesmar, B.N.; et al. The intellectual disability protein PAK3 regulates oligodendrocyte precursor cell differentiation. Neurobiol. Dis. 2017, 98, 137–148. [Google Scholar] [CrossRef]
- Flores, C.E.; Méndez, P. Shaping inhibition: Activity dependent structural plasticity of GABAergic synapses. Front. Cell. Neurosci. 2014, 8, 327. [Google Scholar] [CrossRef]
- Hussain, N.K.; Hsin, H.; Huganir, R.L.; Sheng, M. MINK and TNIK differentially act on Rap2-mediated signal transduction to regulate neuronal structure and AMPA receptor function. J. Neurosci. 2010, 30, 14786–14794. [Google Scholar] [CrossRef]
- Aabdien, A.; Sichlinger, L.; Borgel, Z.; Jones, M.R.; Waston, I.A.; Gatford, N.J.F.; Raval, P.; Tanangonan, L.; Powell, T.R.; Duarte, R.R.R.; et al. Schizophrenia risk proteins ZNF804A and NT5C2 interact in cortical neurons. Eur. J. Neurosci. 2024, 59, 2102–2117. [Google Scholar] [CrossRef]
- Guo, H.; Bettella, E.; Marcogliese, P.C.; Zhao, R.; Andrews, J.C.; Nowakowski, T.J.; Gillentine, M.A.; Hoekzema, K.; Wang, T.; Wu, H.; et al. Disruptive mutations in TANC2 define a neurodevelopmental syndrome associated with psychiatric disorders. Nat. Commun. 2019, 10, 4679. [Google Scholar] [CrossRef]
- Assali, A.; Chenaux, G.; Cho, J.Y.; Berto, S.; Ehrlich, N.A.; Cowan, C.W. EphB1 controls long-range cortical axon guidance through a cell non-autonomous role in GABAergic cells. Development 2024, 151, dev201439. [Google Scholar] [CrossRef]
- Nagy, G.N.; Zhao, X.-F.; Karlsson, R.; Wang, K.; Duman, R.; Harlos, K.; El Omari, K.; Wagner, A.; Clausen, H.; Miller, R.L.; et al. Structure and function of Semaphorin-5A glycosaminoglycan interactions. Nat. Commun. 2024, 15, 2723. [Google Scholar] [CrossRef]
- Le, V.-H.; Orniacki, C.; Murcia-Belmonte, V.; Denti, L.; Schütz, D.; Stumm, R.; Ruhrberg, C.; Erskine, L. CXCL12 promotes the crossing of retinal ganglion cell axons at the optic chiasm. Development 2024, 151, dev202446. [Google Scholar] [CrossRef]
- Chang, C.; Banerjee, S.L.; Park, S.S.; Zhang, X.L.; Cotnoir-White, D.; Opperman, K.J.; Desbois, M.; Grill, B.; Kania, A. Ubiquitin ligase and signalling hub MYCBP2 is required for efficient EPHB2 tyrosine kinase receptor function. eLife 2024, 12, RP89176. [Google Scholar] [CrossRef]
- Koga, M.; Ishiguro, H.; Yazaki, S.; Horiuchi, Y.; Arai, M.; Niizato, K.; Iritani, S.; Itokawa, M.; Inada, T.; Iwata, N.; et al. Involvement of SMARCA2/BRM in the SWI/SNF chromatin-remodeling complex in schizophrenia. Hum. Mol. Genet. 2009, 18, 2483–2494. [Google Scholar] [CrossRef] [PubMed]
- El Hayek, L.; Tuncay, I.O.; Nijem, N.; Russell, J.; Ludwig, S.; Kaur, K.; Li, X.; Anderton, P.; Tang, M.; Gerard, A.; et al. Kdm5a mutations identified in autism spectrum disorder using forward genetics. eLife 2020, 9, e56883. [Google Scholar] [CrossRef] [PubMed]
- Condylis, C.; Ghanbari, A.; Manjrekar, N.; Bistrong, K.; Yao, S.; Yao, Z.; Nguyen, T.N.; Zeng, H.; Tasic, B.; Chen, J.L. Dense functional and molecular readout of a circuit hub in sensory cortex. Science 2022, 375, eabl5981. [Google Scholar] [CrossRef]
- Sollazzo, R.; Puma, D.D.L.; Aceto, G.; Paciello, F.; Colussi, C.; Vita, M.G.; Giuffrè, G.M.; Pastore, F.; Casamassa, A.; Rosati, J.; et al. Structural and functional alterations of neurons derived from sporadic Alzheimer’s disease hiPSCs are associated with downregulation of the LIMK1-cofilin axis. Alzheimer’s Res. Ther. 2024, 16, 267. [Google Scholar] [CrossRef]
- Baumgärtel, K.; Green, A.; Hornberger, D.; Lapira, J.; Rex, C.; Wheeler, D.G.; Peters, M. PDE4D regulates Spine Plasticity and Memory in the Retrosplenial Cortex. Sci. Rep. 2018, 8, 3895. [Google Scholar] [CrossRef]
- Nishioka, M.; Shimada, T.; Bundo, M.; Ukai, W.; Hashimoto, E.; Saito, T.; Kano, Y.; Sasaki, T.; Kasai, K.; Kato, T.; et al. Neuronal cell-type specific DNA methylation patterns of the Cacna1c gene. Int. J. Dev. Neurosci. 2013, 31, 89–95. [Google Scholar] [CrossRef]
- Muresan, V.; Abramson, T.; Lyass, A.; Winter, D.; Porro, E.; Hong, F.; Chamberlin, N.L.; Schnapp, B.J. KIF3C and KIF3A form a novel neuronal heteromeric kinesin that associates with membrane vesicles. Mol. Biol. Cell 1998, 9, 637–652. [Google Scholar] [CrossRef]
- Kanai, Y.; Okada, Y.; Tanaka, Y.; Harada, A.; Terada, S.; Hirokawa, N. KIF5C, a novel neuronal kinesin enriched in motor neurons. J. Neurosci. 2000, 20, 6374–6384. [Google Scholar] [CrossRef]












Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shedenkova, M.; Gurianova, A.; Krasilnikova, I.; Sudina, A.; Karpulevich, E.; Maksimov, Y.; Samburova, M.; Guguchkin, E.; Nefedova, Z.; Babenko, V.; et al. Extracellular Vesicles from iPSC-Derived Glial Progenitor Cells Prevent Glutamate-Induced Excitotoxicity by Stabilising Calcium Oscillations and Mitochondrial Depolarisation. Cells 2025, 14, 1915. https://doi.org/10.3390/cells14231915
Shedenkova M, Gurianova A, Krasilnikova I, Sudina A, Karpulevich E, Maksimov Y, Samburova M, Guguchkin E, Nefedova Z, Babenko V, et al. Extracellular Vesicles from iPSC-Derived Glial Progenitor Cells Prevent Glutamate-Induced Excitotoxicity by Stabilising Calcium Oscillations and Mitochondrial Depolarisation. Cells. 2025; 14(23):1915. https://doi.org/10.3390/cells14231915
Chicago/Turabian StyleShedenkova, Margarita, Anastasiia Gurianova, Irina Krasilnikova, Anastasia Sudina, Evgeny Karpulevich, Yaroslav Maksimov, Marina Samburova, Egor Guguchkin, Zlata Nefedova, Valentina Babenko, and et al. 2025. "Extracellular Vesicles from iPSC-Derived Glial Progenitor Cells Prevent Glutamate-Induced Excitotoxicity by Stabilising Calcium Oscillations and Mitochondrial Depolarisation" Cells 14, no. 23: 1915. https://doi.org/10.3390/cells14231915
APA StyleShedenkova, M., Gurianova, A., Krasilnikova, I., Sudina, A., Karpulevich, E., Maksimov, Y., Samburova, M., Guguchkin, E., Nefedova, Z., Babenko, V., Frolov, D., Savostyanov, K., Fatkhudinov, T., Goldshtein, D., Bakaeva, Z., & Salikhova, D. (2025). Extracellular Vesicles from iPSC-Derived Glial Progenitor Cells Prevent Glutamate-Induced Excitotoxicity by Stabilising Calcium Oscillations and Mitochondrial Depolarisation. Cells, 14(23), 1915. https://doi.org/10.3390/cells14231915









