Endometrial Stromal Cells from Endometriosis Patients Reflect Lesion-Type-Specific Heterogeneity
Highlights
- Endometrial stromal cells from endometriotic lesions recapitulate the disease heterogeneity of endometriosis.
- Endometrial stromal cells show lesion-specific differences in migration, proliferation, contractility, and extracellular matrix.
- Endometrial stromal cells represent an excellent tool to study patho-mechanisms involved in endometriosis.
- Endometrial stromal cells represent a novel model to test therapeutic approaches that consider disease complexity and heterogeneity.
Abstract
1. Introduction
2. Materials and Methods
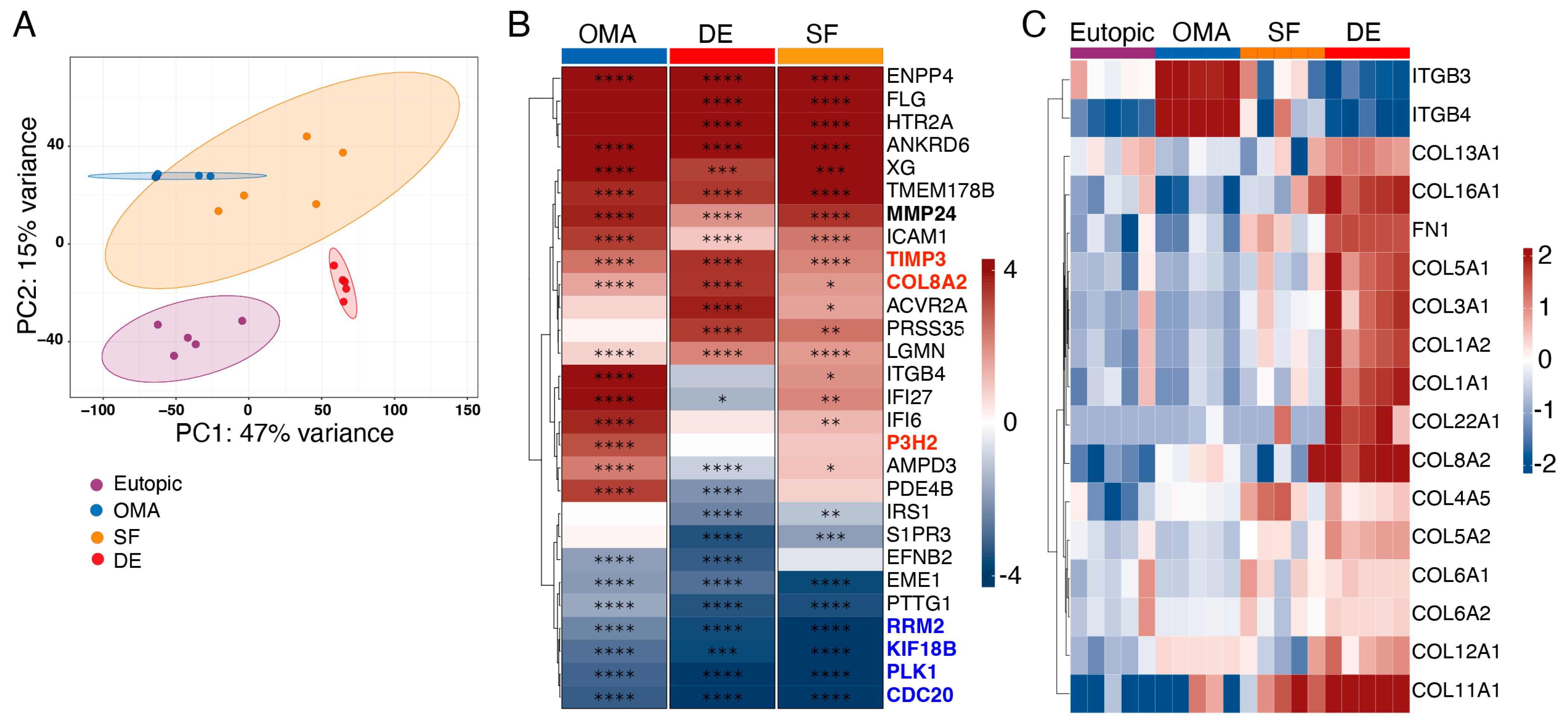
3. Results
3.1. Outgrowing of Cells from Endometriotic Lesions Yields Pure Fibroblast-like Stromal Cells
3.2. ESCs from Different Lesion Types Show Distinct Proliferation and Migration Phenotypes
3.3. Reduced mRNA Expression of Genes Involved in Cell Cycle and Proliferation
3.4. Altered Protein Expression in ESCs from EM Lesions
3.5. ESCs from Deep and Superficial Lesions Showed Increased Contractibility
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DE | Deep Endometriosis |
| EEC | Endometrial Epithelial Cell |
| EM | Endometriosis |
| EpCAM | Epithelial Cellular Adhesion Molecule |
| ER | Estrogen Receptor |
| ESC | Endometrial Stromal Cell |
| FGCZ | Functional Genomics Center Zürich |
| OMA | Endometrioma |
| PBS | Phosphate-Buffered Saline |
| SF | Superficial |
References
- Johnson, N.P.; Hummelshoj, L.; Adamson, G.D.; Keckstein, J.; Taylor, H.S.; Abrao, M.S.; Bush, D.; Kiesel, L.; Tamimi, R.; Sharpe-Timms, K.L.; et al. World Endometriosis Society Consensus on the Classification of Endometriosis. Hum. Reprod. 2017, 32, 315–324. [Google Scholar] [CrossRef]
- Richter, M.; Piwocka, O.; Musielak, M.; Piotrowski, I.; Suchorska, W.M.; Trzeciak, T. From Donor to the Lab: A Fascinating Journey of Primary Cell Lines. Front. Cell Dev. Biol. 2021, 9, 711381. [Google Scholar] [CrossRef] [PubMed]
- Sampson, J.A. Perforating Hemorragic (Chocolate) Cysts of the Ovary - Their Importance and Especially Their Relation to Pelvic Adenomas of Endometrial Type (“Adenomyoma” of the Uterus, Rectovaginal Septum, Sigmoid Etc.). Arch. Surg. 1921, 3, 245–323. [Google Scholar] [CrossRef]
- Golabek-Grenda, A.; Olejnik, A. In Vitro Modeling of Endometriosis and Endometriotic Microenvironment - Challenges and Recent Advances. Cell. Signal. 2022, 97, 110375. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.S.; Nair, H.B.; Lucidi, R.S.; Kirchner, A.J.; Schenken, R.S.; Tekmal, R.R.; Witz, C.A. Modeling the Early Endometriotic Lesion: Mesothelium-Endometrial Cell Co-Culture Increases Endometrial Invasion and Alters Mesothelial and Endometrial Gene Transcription. Fertil. Steril. 2008, 90, 1487–1495. [Google Scholar] [CrossRef] [PubMed]
- Donnez, J. Introduction: From Pathogenesis to Therapy, Deep Endometriosis Remains a Source of Controversy. Fertil. Steril. 2017, 108, 869–871. [Google Scholar] [CrossRef]
- Gordts, S.; Koninckx, P.; Brosens, I. Pathogenesis of Deep Endometriosis. Fertil. Steril. 2017, 108, 872–885.e1. [Google Scholar] [CrossRef]
- Andres, M.P.; Arcoverde, F.V.L.; Souza, C.C.C.; Fernandes, L.F.C.; Abrao, M.S.; Kho, R.M. Extrapelvic Endometriosis: A Systematic Review. J. Minim. Invasive Gynecol. 2020, 27, 373–389. [Google Scholar] [CrossRef]
- Imperiale, L.; Nisolle, M.; Noël, J.C.; Fastrez, M. Three Types of Endometriosis: Pathogenesis, Diagnosis and Treatment. State of the Art. J. Clin. Med. 2023, 12, 994. [Google Scholar] [CrossRef]
- Colgrave, E.M.; Keast, J.R.; Healey, M.; Rogers, P.A.; Girling, J.E.; Holdsworth-Carson, S.J. Extensive Heterogeneity in the Expression of Steroid Receptors in Superficial Peritoneal Endometriotic Lesions. Reprod. Biomed. Online 2023, 48, 103409. [Google Scholar] [CrossRef]
- Koninckx, P.R.; Ussia, A.; Adamyan, L.; Wattiez, A.; Gomel, V.; Martin, D.C. Pathogenesis of Endometriosis: The Genetic/Epigenetic Theory. Fertil. Steril. 2019, 111, 327–340. [Google Scholar] [CrossRef] [PubMed]
- Saha, R.; Pettersson, H.J.; Svedberg, P.; Olovsson, M.; Bergqvist, A.; Marions, L.; Tornvall, P.; Kuja-Halkola, R. Heritability of Endometriosis. Fertil. Steril. 2015, 104, 947–952. [Google Scholar] [CrossRef] [PubMed]
- Bulun, S.E.; Yildiz, S.; Adli, M.; Chakravarti, D.; Parker, J.B.; Milad, M.; Yang, L.; Chaudhari, A.; Tsai, S.; Wei, J.J.; et al. Endometriosis and Adenomyosis: Shared Pathophysiology. Fertil. Steril. 2023, 119, 746–750. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.C.; Wang, K.H.; Chuang, K.H.; Kao, A.P.; Kuo, T.C. Interleukin-33 Promotes Invasiveness of Human Ovarian Endometriotic Stromal Cells through the St2/Mapk/Mmp-9 Pathway Activated by 17β-Estradiol. Taiwan. J. Obstet. Gynecol. 2021, 60, 658–664. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, W.; Wang, L.; Zhang, Z.; Xiong, W.; Zhang, L.; Fu, T.; Li, X.; Chen, Y.; Liu, Y. Gli1 Is Increased in Ovarian Endometriosis and Regulates Migration, Invasion and Proliferation of Human Endometrial Stromal Cells in Endometriosis. Ann. Transl. Med. 2019, 7, 663. [Google Scholar] [CrossRef]
- Anupa, G.; Poorasamy, J.; Bhat, M.A.; Sharma, J.B.; Sengupta, J.; Ghosh, D. Endometrial Stromal Cell Inflammatory Phenotype during Severe Ovarian Endometriosis as a Cause of Endometriosis-Associated Infertility. Reprod. Biomed. Online 2020, 41, 623–639. [Google Scholar] [CrossRef]
- Delbandi, A.A.; Mahmoudi, M.; Shervin, A.; Heidari, S.; Kolahdouz-Mohammadi, R.; Zarnani, A.H. Evaluation of Apoptosis and Angiogenesis in Ectopic and Eutopic Stromal Cells of Patients with Endometriosis Compared to Non-Endometriotic Controls. BMC Women’s Health 2020, 20, 3. [Google Scholar] [CrossRef]
- Yang, H.L.; Zhou, W.J.; Chang, K.K.; Mei, J.; Huang, L.Q.; Wang, M.Y.; Meng, Y.; Ha, S.Y.; Li, D.J.; Li, M.Q. The Crosstalk between Endometrial Stromal Cells and Macrophages Impairs Cytotoxicity of Nk Cells in Endometriosis by Secreting Il-10 and Tgf-Beta. Reproduction 2017, 154, 815–825. [Google Scholar] [CrossRef]
- Taghizadeh, R.R.; Cetrulo, K.J.; Cetrulo, C.L. Collagenase Impacts the Quantity and Quality of Native Mesenchymal Stem/Stromal Cells Derived during Processing of Umbilical Cord Tissue. Cell Transplant. 2018, 27, 181–193. [Google Scholar] [CrossRef]
- Mattei, D.; Ivanov, A.; van Oostrum, M.; Pantelyushin, S.; Richetto, J.; Mueller, F.; Beffinger, M.; Schellhammer, L.; Vom Berg, J.; Wollscheid, B.; et al. Enzymatic Dissociation Induces Transcriptional and Proteotype Bias in Brain Cell Populations. Int. J. Mol. Sci. 2020, 21, 7944. [Google Scholar] [CrossRef]
- Pan, C.; Kumar, C.; Bohl, S.; Klingmueller, U.; Mann, M. Comparative Proteomic Phenotyping of Cell Lines and Primary Cells to Assess Preservation of Cell Type-Specific Functions. Mol. Cell. Proteom. 2009, 8, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Romano, A.; Xanthoulea, S.; Giacomini, E.; Delvoux, B.; Alleva, E.; Vigano, P. Endometriotic Cell Culture Contamination and Authenticity: A Source of Bias in in vitro Research? Hum. Reprod. 2020, 35, 364–376. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An Open-Source Platform for Biological-Image Analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Gasibat, Q.; Abdullah, B.; Samsudin, S.; Alexe, D.I.; Alexe, C.I.; Tohanean, D.I. Gender-Specific Patterns of Muscle Imbalance in Elite Badminton Players: A Comprehensive Exploration. Sports 2023, 11, 164. [Google Scholar] [CrossRef]
- Hughes, C.S.; Foehr, S.; Garfield, D.A.; Furlong, E.E.; Steinmetz, L.M.; Krijgsveld, J. Ultrasensitive Proteome Analysis Using Paramagnetic Bead Technology. Mol. Syst. Biol. 2014, 10, 757. [Google Scholar] [CrossRef]
- Leutert, M.; Rodriguez-Mias, R.A.; Fukuda, N.K.; Villen, J. R2-P2 Rapid-Robotic Phosphoproteomics Enables Multidimensional Cell Signaling Studies. Mol. Syst. Biol. 2019, 15, e9021. [Google Scholar] [CrossRef]
- Türker, C.; Akal, F.; Joho, D.; Panse, C.; Barkow-Oesterreicher, S.; Rehrauer, H.; Schlapbach, R. B-Fabric: The Swiss Army Knife for Life Sciences. In Proceedings of the 13th International Conference on Extending Database Technology, Lausanne Switzerland, 22–26 March 2010; Association for Computing Machinery: New York, NY, USA, 2010; pp. 717–720. [Google Scholar]
- Demichev, V.; Messner, C.B.; Vernardis, S.I.; Lilley, K.S.; Ralser, M. Dia-Nn: Neural Networks and Interference Correction Enable Deep Proteome Coverage in High Throughput. Nat Methods 2020, 17, 41–44. [Google Scholar] [CrossRef]
- Wolski, W.E.; Nanni, P.; Grossmann, J.; d’Errico, M.; Schlapbach, R.; Panse, C. Prolfqua: A Comprehensive R-Package for Proteomics Differential Expression Analysis. J. Proteome Res. 2023, 22, 1092–1104. [Google Scholar] [CrossRef]
- Huber, W.; von Heydebreck, A.; Sültmann, H.; Poustka, A.; Vingron, M. Variance Stabilization Applied to Microarray Data Calibration and to the Quantification of Differential Expression. Bioinformatics 2002, 18 (Suppl. S1), S96–S104. [Google Scholar] [CrossRef] [PubMed]
- Hatakeyama, M.; Opitz, L.; Russo, G.; Qi, W.; Schlapbach, R.; Rehrauer, H. Sushi: An Exquisite Recipe for Fully Documented, Reproducible and Reusable Ngs Data Analysis. BMC Bioinform. 2016, 17, 228. [Google Scholar] [CrossRef]
- Leary, P.; Rehrauer, H. exploreDE Interactive Shiny App. Zenodo 2024. [Google Scholar] [CrossRef]
- Bray, N.L.; Pimentel, H.; Melsted, P.; Pachter, L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 2016, 34, 525–527. [Google Scholar] [CrossRef]
- D’Urso, M.; Kurniawan, N.A. Mechanical and Physical Regulation of Fibroblast-Myofibroblast Transition: From Cellular Mechanoresponse to Tissue Pathology. Front. Bioeng. Biotechnol. 2020, 8, 609653. [Google Scholar] [CrossRef]
- Waise, S.; Parker, R.; Rose-Zerilli, M.J.J.; Layfield, D.M.; Wood, O.; West, J.; Ottensmeier, C.H.; Thomas, G.J.; Hanley, C.J. An Optimized Method to Isolate Human Fibroblasts from Tissue for ex vivo Analysis. Bio-Protocol 2019, 9, e3440. [Google Scholar] [CrossRef] [PubMed]
- Zlatska, A.V.; Rodnichenko, A.E.; Gubar, O.S.; Zubov, D.O.; Novikova, S.N.; Vasyliev, R.G. Endometrial Stromal Cells: Isolation, Expansion, Morphological and Functional Properties. Exp. Oncol. 2017, 39, 197–202. [Google Scholar] [CrossRef]
- Queckbörner, S.; Lundberg, E.S.; Gemzell-Danielsson, K.; Davies, L.C. Endometrial Stromal Cells Exhibit a Distinct Phenotypic and Immunomodulatory Profile. Stem Cell. Res. Ther. 2020, 11, 15. [Google Scholar] [CrossRef] [PubMed]
- Toki, T.; Shimizu, M.; Takagi, Y.; Ashida, T.; Konishi, I. Cd10 Is a Marker for Normal and Neoplastic Endometrial Stromal Cells. Int. J. Gynecol. Pathol. 2002, 21, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Park, J.E.; Lenter, M.C.; Zimmermann, R.N.; Garin-Chesa, P.; Old, L.J.; Rettig, W.J. Fibroblast Activation Protein, a Dual Specificity Serine Protease Expressed in Reactive Human Tumor Stromal Fibroblasts. J. Biol. Chem. 1999, 274, 36505–36512. [Google Scholar] [CrossRef]
- Tantengco, O.A.G.; Richardson, L.S.; Vink, J.; Kechichian, T.; Medina, P.M.B.; Pyles, R.B.; Menon, R. Progesterone Alters Human Cervical Epithelial and Stromal Cell Transition and Migration: Implications in Cervical Remodeling during Pregnancy and Parturition. Mol. Cell. Endocrinol. 2021, 529, 111276. [Google Scholar] [CrossRef]
- Alsharif, S.; Sharma, P.; Bursch, K.; Milliken, R.; Lam, V.; Fallatah, A.; Phan, T.; Collins, M.; Dohlman, P.; Tiufekchiev, S.; et al. Keratin 19 Maintains E-Cadherin Localization at the Cell Surface and Stabilizes Cell-Cell Adhesion of Mcf7 Cells. Cell Adh. Migr. 2021, 15, 1–17. [Google Scholar] [CrossRef]
- Conroy, K.P.; Kitto, L.J.; Henderson, N.C. Alphav Integrins: Key Regulators of Tissue Fibrosis. Cell Tissue Res. 2016, 365, 511–519. [Google Scholar] [CrossRef]
- Shinotsuka, N.; Denk, F. Fibroblasts: The Neglected Cell Type in Peripheral Sensitisation and Chronic Pain? A Review Based on a Systematic Search of the Literature. BMJ Open Sci. 2022, 6, e100235. [Google Scholar] [CrossRef]
- Vissers, G.; Giacomozzi, M.; Verdurmen, W.; Peek, R.; Nap, A. The Role of Fibrosis in Endometriosis: A Systematic Review. Hum. Reprod. Update 2024, 30, 706–750. [Google Scholar] [CrossRef]
- Malvezzi, H.; Marengo, E.B.; Podgaec, S.; Piccinato, C.A. Endometriosis: Current Challenges in Modeling a Multifactorial Disease of Unknown Etiology. J. Transl. Med. 2020, 18, 311. [Google Scholar] [CrossRef] [PubMed]
- Konrad, L.; Kortum, J.; Nabham, R.; Gronbach, J.; Dietze, R.; Oehmke, F.; Berkes, E.; Tinneberg, H.R. Composition of the Stroma in the Human Endometrium and Endometriosis. Reprod. Sci. 2018, 25, 1106–1115. [Google Scholar] [CrossRef]
- Koumas, L.; King, A.E.; Critchley, H.O.D.; Kelly, R.W.; Phipps, R.P. Fibroblast Heterogeneity: Existence of Functionally Distinct Thy 1+ and Thy 1− Human Female Reproductive Tract Fibroblasts. Am. J. Pathol. 2001, 159, 925–935. [Google Scholar] [CrossRef]
- Mareckova, M.; Garcia-Alonso, L.; Moullet, M.; Lorenzi, V.; Petryszak, R.; Sancho-Serra, C.; Oszlanczi, A.; Icoresi Mazzeo, C.; Wong, F.C.K.; Kelava, I.; et al. An Integrated Single-Cell Reference Atlas of the Human Endometrium. Nat. Genet. 2024, 56, 1925–1937. [Google Scholar] [CrossRef]
- Stejskalova, A.; Fincke, V.; Nowak, M.; Schmidt, Y.; Borrmann, K.; von Wahlde, M.K.; Schafer, S.D.; Kiesel, L.; Greve, B.; Gotte, M. Collagen I Triggers Directional Migration, Invasion and Matrix Remodeling of Stroma Cells in a 3d Spheroid Model of Endometriosis. Sci. Rep. 2021, 11, 4115. [Google Scholar] [CrossRef] [PubMed]
- Yuge, A.; Nasu, K.; Matsumoto, H.; Nishida, M.; Narahara, H. Collagen Gel Contractility Is Enhanced in Human Endometriotic Stromal Cells: A Possible Mechanism Underlying the Pathogenesis of Endometriosis-Associated Fibrosis. Hum. Reprod. 2007, 22, 938–944. [Google Scholar] [CrossRef] [PubMed]
- Young, V.J.; Brown, J.K.; Maybin, J.; Saunders, P.T.; Duncan, W.C.; Horne, A.W. Transforming Growth Factor-Beta Induced Warburg-Like Metabolic Reprogramming May Underpin the Development of Peritoneal Endometriosis. J. Clin. Endocrinol. Metab. 2014, 99, 3450–3459. [Google Scholar] [CrossRef]
- Kasvandik, S.; Samuel, K.; Peters, M.; Eimre, M.; Peet, N.; Roost, A.M.; Padrik, L.; Paju, K.; Peil, L.; Salumets, A. Deep Quantitative Proteomics Reveals Extensive Metabolic Reprogramming and Cancer-Like Changes of Ectopic Endometriotic Stromal Cells. J. Proteome Res. 2016, 15, 572–584. [Google Scholar] [CrossRef] [PubMed]
- Yotova, I.; Proestling, K.; Haslinger, I.; Witzmann-Stern, M.; Widmar, B.; Kuessel, L.; Husslein, H.; Wenzl, R.; Hudson, Q.J. Diras3 Regulates Autophagy in an Endometriosis Epithelial Cell Line. Reprod. Biomed. Online 2023, 47, 103251. [Google Scholar] [CrossRef] [PubMed]
- Tavaluc, R.T.; Hart, L.S.; Dicker, D.T.; El-Deiry, W.S. Effects of Low Confluency, Serum Starvation and Hypoxia on the Side Population of Cancer Cell Lines. Cell Cycle 2007, 6, 2554–2562. [Google Scholar] [CrossRef]
- Kobayashi, H.; Shigetomi, H.; Imanaka, S. Nonhormonal Therapy for Endometriosis Based on Energy Metabolism Regulation. Reprod. Fertil. 2021, 2, C42–C57. [Google Scholar] [CrossRef]
- Pandey, S. Metabolomics for the Identification of Biomarkers in Endometriosis. Arch. Gynecol. Obstet. 2024, 310, 2823–2827. [Google Scholar] [CrossRef]
- Hu, L.; Zhang, J.; Lu, Y.; Fu, B.; Hu, W. Estrogen Receptor Beta Promotes Endometriosis Progression by Upregulating Cd47 Expression in Ectopic Endometrial Stromal Cells. J. Reprod. Immunol. 2022, 151, 103513. [Google Scholar] [CrossRef]
- Monsivais, D.; Dyson, M.T.; Yin, P.; Coon, J.S.; Navarro, A.; Feng, G.; Malpani, S.S.; Ono, M.; Ercan, C.M.; Wei, J.J.; et al. Erbeta- and Prostaglandin E2-Regulated Pathways Integrate Cell Proliferation Via Ras-Like and Estrogen-Regulated Growth Inhibitor in Endometriosis. Mol. Endocrinol. 2014, 28, 1304–1315. [Google Scholar] [CrossRef]
- Perzelová, V.; Sabol, F.; Vasilenko, T.; Novotny, M.; Kovác, I.; Slezák, M.; Durkác, J.; Holly, M.; Pilátová, M.; Szabo, P.; et al. Pharmacological Activation of Estrogen Receptors-α and -β Differentially Modulates Keratinocyte Differentiation with Functional Impact on Wound Healing. Int. J. Mol. Med. 2016, 37, 21–28. [Google Scholar]
- Campbell, L.; Emmerson, E.; Davies, F.; Gilliver, S.C.; Krust, A.; Chambon, P.; Ashcroft, G.S.; Hardman, M.J. Estrogen Promotes Cutaneous Wound Healing Via Estrogen Receptor β Independent of Its Antiinflammatory Activities. J. Exp. Med. 2010, 207, 1825–1833. [Google Scholar] [CrossRef] [PubMed]
- Bian, X.; Liu, T.; Yang, M.; Gu, C.; He, G.; Zhou, M.; Tang, H.; Lu, K.; Lai, F.; Wang, F.; et al. The Absence of Oestrogen Receptor Beta Disturbs Collagen I Type Deposition during Achilles Tendon Healing by Regulating the Irf5-Ccl3 Axis. J. Cell. Mol. Med. 2020, 24, 9925–9935. [Google Scholar] [CrossRef] [PubMed]
- Flamini, M.I.; Sanchez, A.M.; Genazzani, A.R.; Simoncini, T. Estrogen Regulates Endometrial Cell Cytoskeletal Remodeling and Motility Via Focal Adhesion Kinase. Fertil. Steril. 2011, 95, 722–726. [Google Scholar] [CrossRef]
- Park, Y.; Sung, N.; Kim, E.; Jeong, J.; Sim, J.; Park, M.J.; Lydon, J.P.; Guan, X.; Han, S.J. Polychlorinated Biphenyls Alter Estrogen Receptor beta-mediated Epigenetic Regulation, Promoting Endometriosis. Endocrinology 2025, 166, bqaf146. [Google Scholar] [CrossRef]
- Ling, F.; Chen, Y.; Li, J.; Xu, M.; Song, G.; Tu, L.; Wang, H.; Li, S.; Zhu, L. Estrogen Receptor beta Activation Mitigates Colitis-associated Intestinal Fibrosis via Inhibition of TGF-beta/Smad and TLR4/MyD88/NF-kappaB Signaling Pathways. Inflamm. Bowel Dis. 2025, 31, 11–27. [Google Scholar] [CrossRef] [PubMed]
- Nie, J.; Yi, Y.; Liu, X.; Guo, S.W. Progressively Diminished Estrogen Signaling Concordant with Increased Fibrosis in Ectopic Endometriumm. Hum. Reprod. Open 2025, 2025, hoaf028. [Google Scholar] [PubMed]
- Hudelist, G.; Keckstein, J.; Czerwenka, K.; Lass, H.; Walter, I.; Auer, M.; Wieser, F.; Wenzl, R.; Kubista, E.; Singer, C.F. Estrogen Receptor Beta and Matrix Metalloproteinase 1 Are Coexpressed in Uterine Endometrium and Endometriotic Lesions of Patients with Endometriosis. Fertil. Steril. 2005, 84 (Suppl. S2), 1249–1256. [Google Scholar] [CrossRef] [PubMed]
- Cetera, G.E.; Merli, C.E.M.; Facchin, F.; Vigano, P.; Pesce, E.; Caprara, F.; Vercellini, P. Non-Response to First-Line Hormonal Treatment for Symptomatic Endometriosis: Overcoming Tunnel Vision. A Narrative Review. BMC Women’s Health 2023, 23, 347. [Google Scholar] [CrossRef]
- Almadani, Y.H.; Vorstenbosch, J.; Davison, P.G.; Murphy, A.M. Wound Healing: A Comprehensive Review. Semin. Plast. Surg. 2021, 35, 141–144. [Google Scholar] [CrossRef]
- Fiore, V.F.; Wong, S.S.; Tran, C.; Tan, C.; Xu, W.; Sulchek, T.; White, E.S.; Hagood, J.S.; Barker, T.H. Alphavbeta3 Integrin Drives Fibroblast Contraction and Strain Stiffening of Soft Provisional Matrix during Progressive Fibrosis. JCI Insight 2018, 3, e97597. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodriguez Gutierrez, D.; Spalinger, M.R.; Astourian, A.; Evrova, O.; Berclaz, L.; Hartmann, M.; Dedes, I.; Imesch, P.; Metzler, J.M.; Witzel, I.; et al. Endometrial Stromal Cells from Endometriosis Patients Reflect Lesion-Type-Specific Heterogeneity. Cells 2025, 14, 1891. https://doi.org/10.3390/cells14231891
Rodriguez Gutierrez D, Spalinger MR, Astourian A, Evrova O, Berclaz L, Hartmann M, Dedes I, Imesch P, Metzler JM, Witzel I, et al. Endometrial Stromal Cells from Endometriosis Patients Reflect Lesion-Type-Specific Heterogeneity. Cells. 2025; 14(23):1891. https://doi.org/10.3390/cells14231891
Chicago/Turabian StyleRodriguez Gutierrez, Daniel, Marianne R. Spalinger, Alina Astourian, Olivera Evrova, Lucie Berclaz, Monique Hartmann, Ioannis Dedes, Patrick Imesch, Julian M. Metzler, Isabelle Witzel, and et al. 2025. "Endometrial Stromal Cells from Endometriosis Patients Reflect Lesion-Type-Specific Heterogeneity" Cells 14, no. 23: 1891. https://doi.org/10.3390/cells14231891
APA StyleRodriguez Gutierrez, D., Spalinger, M. R., Astourian, A., Evrova, O., Berclaz, L., Hartmann, M., Dedes, I., Imesch, P., Metzler, J. M., Witzel, I., Shilaih, M., Vongrad, V., & Leeners, B. (2025). Endometrial Stromal Cells from Endometriosis Patients Reflect Lesion-Type-Specific Heterogeneity. Cells, 14(23), 1891. https://doi.org/10.3390/cells14231891





