The authors wish to make the following changes to their paper [1]. In the original publication, there was a mistake uploading the figures, and Figure 1, Figure 2 and Figure 3 as published do not correspond to the updated versions after peer review. In particular, Figure 1 needed to be corrected regarding the transcriptional status of the X chromosomes during the preimplantation stages. Figure 1, Figure 2 and Figure 3 should be changed to:
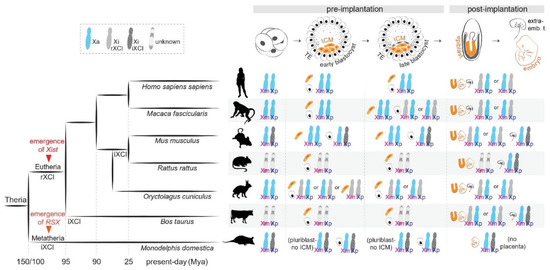
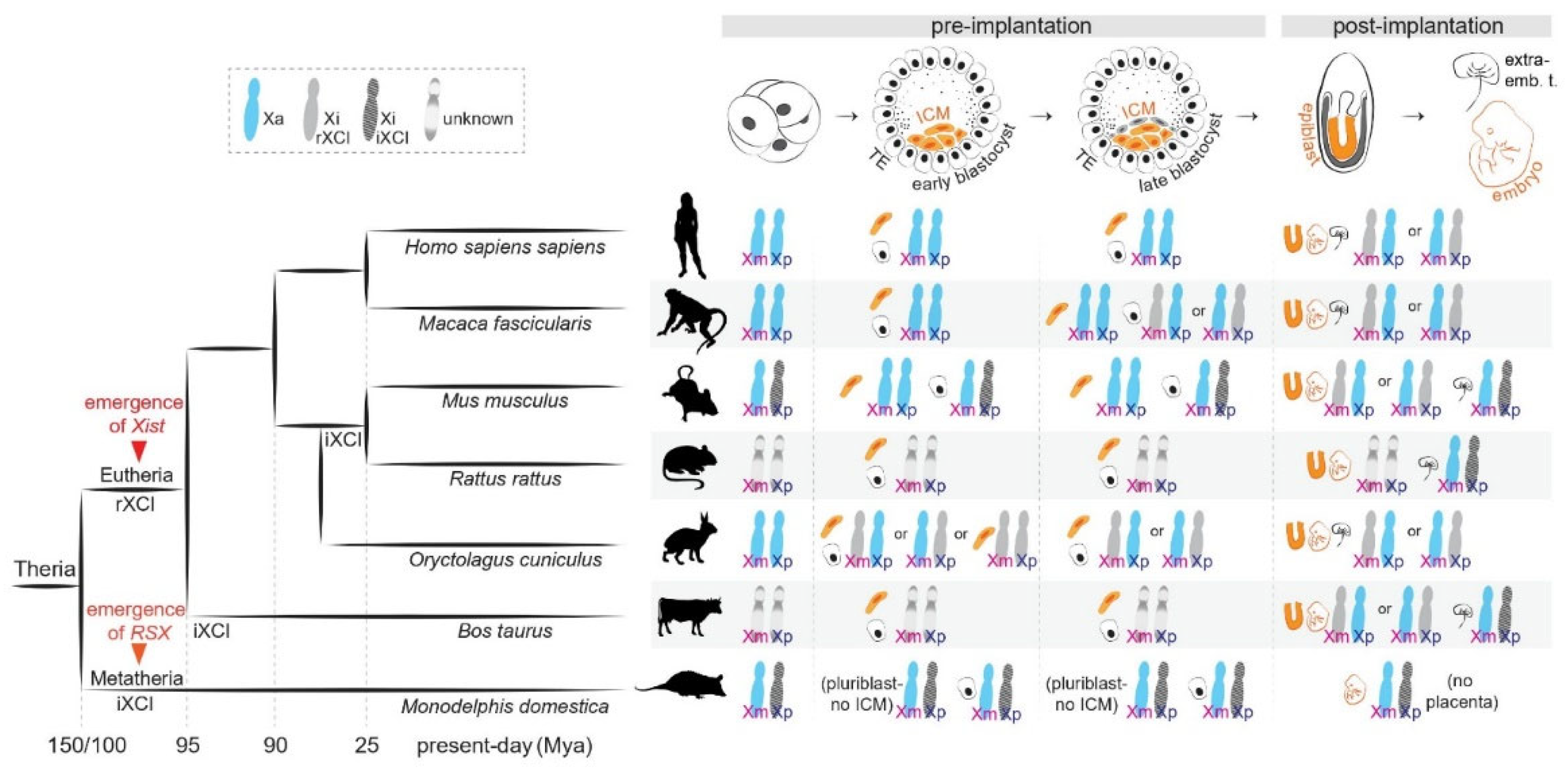
Figure 1.
X-chromosome inactivation across species. (Left): Phylogenetic tree indicating the evolution of random and imprinted XCI and the emergence of long non-coding RNAs Xist and RSX in Theria. (Right): X-chromosome inactivation dynamics across development in representative species.
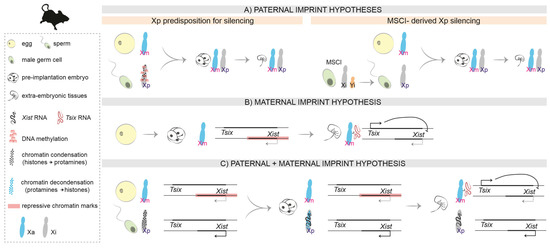
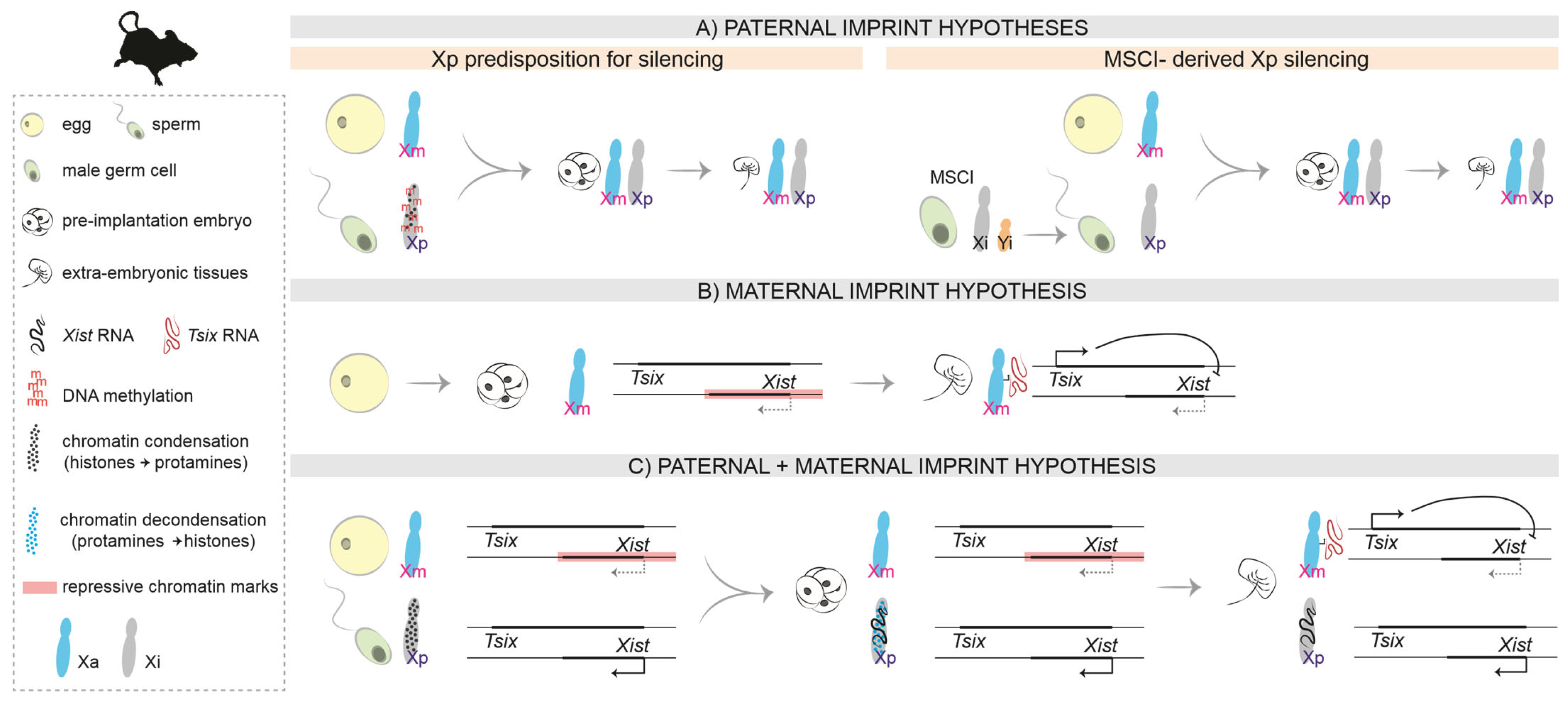
Figure 2.
Hypotheses on the molecular nature of the imprint in mice. (A). Paternal imprint: The Xp inherits a predisposition for silencing from its life cycle in the male. (B). Maternal imprint: In the preimplantation embryo, repressive chromatin marks on the Xm (including the Xist promoter region) prevent Xist expression on the Xm. In the extra-embryonic tissues of the post-implantation embryo, Tsix expression prevents Xist upregulation in cis. (C). Paternal and maternal imprint: A combination of both hypotheses, also considering the different chromatin condensation states of the Xp in the sperm and in the paternal pronucleus after fertilization.
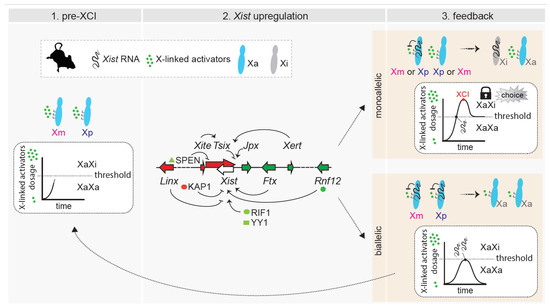
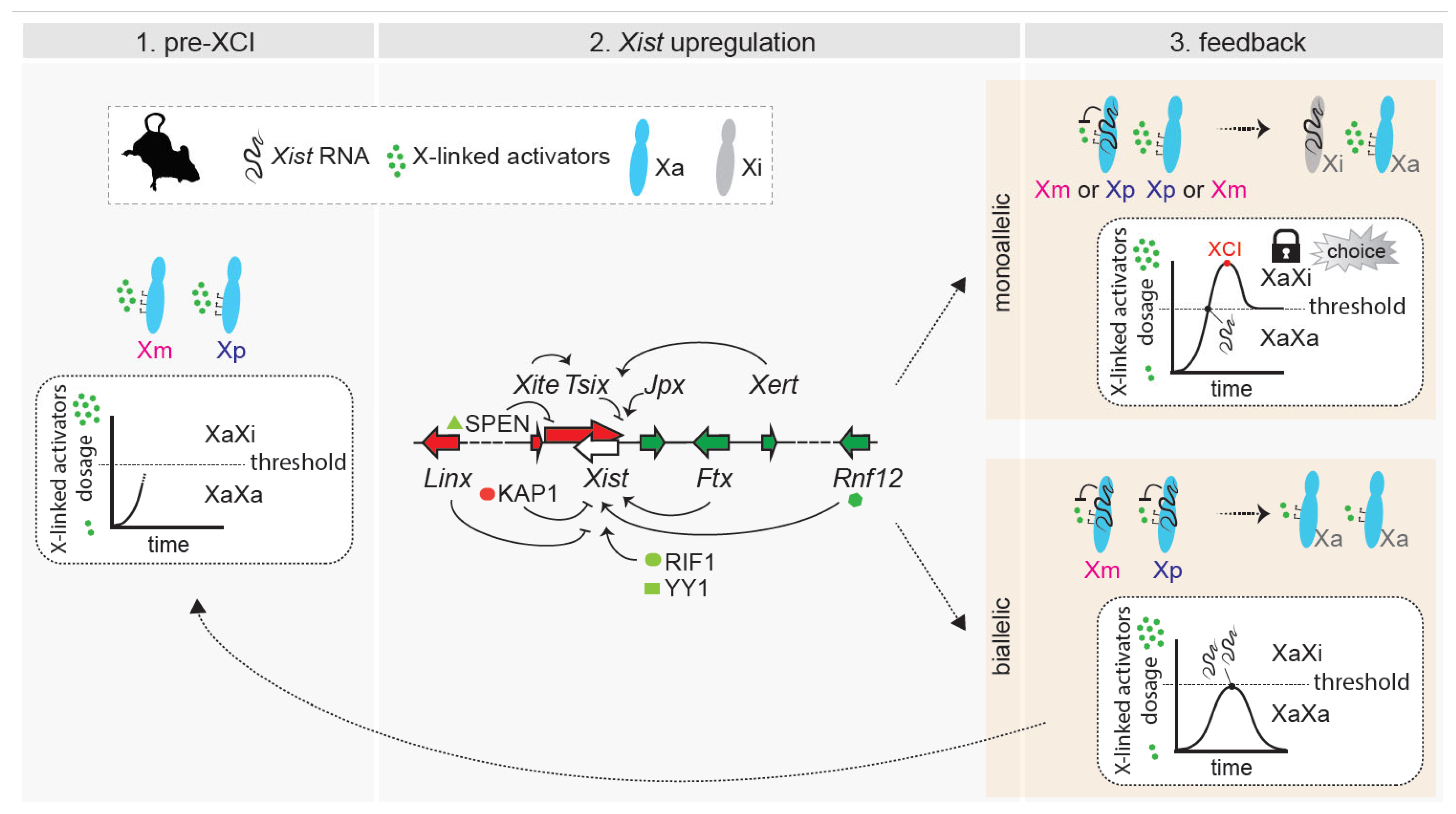
Figure 3.
Dynamic model of choice during random XCI. (Left): Pre-XCI status. Both X chromosomes are active and transcribe X-linked genes. The dose of X-linked activators increases towards the threshold necessary for productive Xist upregulation. (Middle): Biallelic X-chromosome transcription allows the cell to reach the threshold for Xist activation. X-linked and autosomal cis and trans positive and negative regulators influence the initiation of Xist upregulation, which can occur on a single X chromosome or on both of them. Only factors and loci discussed in the text have been included in the figure. (Right (top)): In cells that have upregulated Xist monoallelically, X-wide cis-silencing triggered by Xist RNA causes a drop in the level of activators, preventing the second chromosome from upregulating Xist. The choice is locked in. Monoallelic Xist expression (and cis-silencing) has to be sustained through enough dosage of activators and/or feedback mechanisms. (Right (bottom)): In cells that have upregulated Xist biallelically, excess Xist expression triggers rapid downregulation of X-linked activators on both X chromosomes, and this drop in levels below the threshold causes Xist expression to switch off. Both X chromosomes remain active, and the process has to start again.
The authors apologize for any inconvenience caused and state that the scientific conclusions are unaffected. This correction was approved by the academic editor. The original publication has also been updated.
Conflicts of Interest
The authors declare no conflict of interest.
Reference
- Furlan, G.; Galupa, R. Mechanisms of Choice in X-Chromosome Inactivation. Cells 2022, 11, 535. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).