Human Stem Cell Use in Androgenetic Alopecia: A Systematic Review
Abstract
1. Introduction
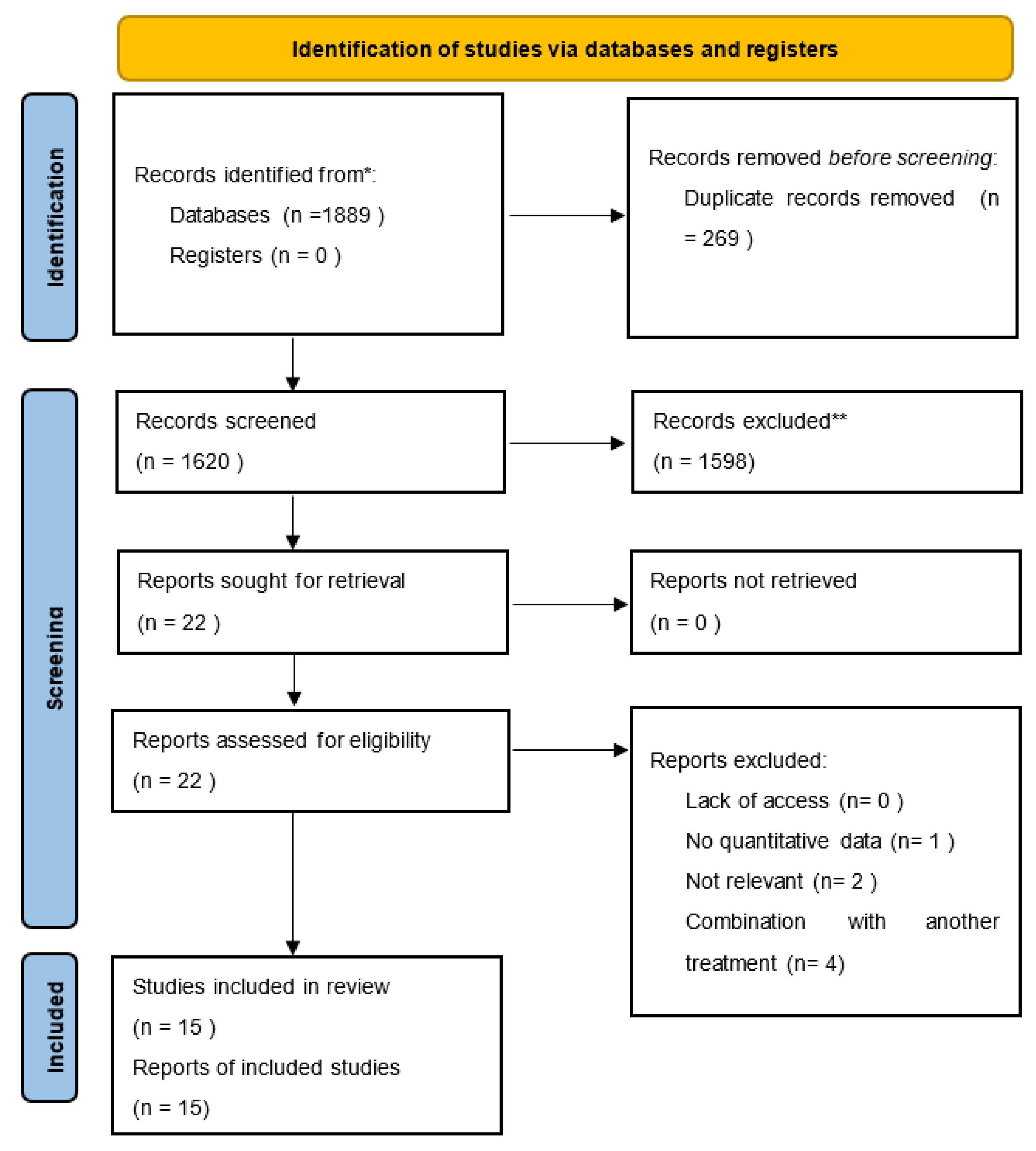
2. Materials and Methods
3. Results
3.1. Studies Comparing the Use of Stem Cells with a Placebo
3.2. Studies Analyzing the Efficacy of Stem Cell Treatment Alone
3.3. Studies Comparing Stem Cells and/or Other Treatments
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Blumeyer, A.; Tosti, A.; Messenger, A.; Reygagne, P.; Del Marmol, V.; Spuls, P.I.; Trakatelli, M.; Finner, A.; Kiesewetter, F.; Trüeb, R.; et al. Evidence-based (S3) guideline for the treatment of androgenetic alopecia in women and in men. JDDG J. Dtsch. Dermatol. Ges. 2011, 9, S1–S57. [Google Scholar] [CrossRef] [PubMed]
- Rushton, D.H.; Westgate, G.E.; Van Neste, D.J. Following historical “tracks” of hair follicle miniaturisation in patterned hair loss: Are elastin bodies the forgotten aetiology? Exp. Dermatol. 2022, 31, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Chan, L.; Cook, D.K. Female pattern hair loss. Aust. J. Gen. Pract. 2018, 47, 459–464. [Google Scholar] [CrossRef]
- Alfonso, M.; Richter-Appelt, H.; Tosti, A.; Viera, M.S.; García, M. The psychosocial impact of hair loss among men: A multinational European study. Curr. Med. Res. Opin. 2005, 21, 1829–1836. [Google Scholar] [CrossRef] [PubMed]
- Cash, T.F. The psychosocial consequences of androgenetic alopecia: A review of the research literature: Psychosocial consequences of androgenetic alopecia. Br. J. Dermatol. 1999, 141, 398–405. [Google Scholar] [CrossRef]
- Choi, G.-S.; Sim, W.-Y.; Kang, H.; Huh, C.H.; Lee, Y.W.; Shantakumar, S.; Ho, Y.-F.; Oh, E.-J.; Duh, M.S.; Cheng, W.Y.; et al. Long-Term Effectiveness and Safety of Dutasteride versus Finasteride in Patients with Male Androgenic Alopecia in South Korea: A Multicentre Chart Review Study. Ann. Dermatol. 2022, 34, 349. [Google Scholar] [CrossRef] [PubMed]
- Adil, A.; Godwin, M. The effectiveness of treatments for androgenetic alopecia: A systematic review and meta-analysis. J. Am. Acad. Dermatol. 2017, 77, 136–141.e5. [Google Scholar] [CrossRef]
- Gupta, A.K.; Bamimore, M.A.; Foley, K.A. Efficacy of non-surgical treatments for androgenetic alopecia in men and women: A systematic review with network meta-analyses, and an assessment of evidence quality. J. Dermatol. Treat. 2022, 33, 62–72. [Google Scholar] [CrossRef]
- Egger, A.; Tomic-Canic, M.; Tosti, A. Advances in Stem Cell-Based Therapy for Hair Loss. CellR4—Repair Replace. Regen. Reprogramming 2020, 8, e2894. [Google Scholar]
- Anudeep, T.C.; Jeyaraman, M.; Muthu, S.; Rajendran, R.L.; Gangadaran, P.; Mishra, P.C.; Sharma, S.; Jha, S.K.; Ahn, B.-C. Advancing Regenerative Cellular Therapies in Non-Scarring Alopecia. Pharmaceutics 2022, 14, 612. [Google Scholar] [CrossRef]
- Ji, S.; Zhu, Z.; Sun, X.; Fu, X. Functional hair follicle regeneration: An updated review. Signal Transduct. Target. Ther. 2021, 6, 66. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Liu, X.-M.; Liu, Z.-N.; Wang, Y.; Han, X.; Lian, A.-B.; Mu, Y.; Jin, M.-H.; Liu, J.-Y. Human hair follicle-derived mesenchymal stem cells: Isolation, expansion, and differentiation. World J. Stem Cells 2020, 12, 462–470. [Google Scholar] [CrossRef]
- Kiani, M.T.; Higgins, C.A.; Almquist, B.D. The Hair Follicle: An Underutilized Source of Cells and Materials for Regenerative Medicine. ACS Biomater. Sci. Eng. 2018, 4, 1193–1207. [Google Scholar] [CrossRef]
- Owczarczyk-Saczonek, A.; Krajewska-Włodarczyk, M.; Kruszewska, A.; Banasiak, Ł.; Placek, W.; Maksymowicz, W.; Wojtkiewicz, J. Therapeutic Potential of Stem Cells in Follicle Regeneration. Stem Cells Int. 2018, 2018, 1049641. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, P.; Youssef, K.K.; Abbasalizadeh, S.; Baharvand, H.; Aghdami, N. Human Hair Reconstruction: Close, But Yet So Far. Stem Cells Dev. 2016, 25, 1767–1779. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-L.; Huang, W.-Y.; Wang EH, C.; Tai, K.-Y.; Lin, S.-J. Functional complexity of hair follicle stem cell niche and therapeutic targeting of niche dysfunction for hair regeneration. J. Biomed. Sci. 2020, 27, 43. [Google Scholar] [CrossRef]
- Mysore, V.; Alexander, S.; Nepal, S.; Venkataram, A. Regenerative Medicine Treatments for Androgenetic Alopecia. Indian J. Plast. Surg. Off. Publ. Assoc. Plast. Surg. India 2021, 54, 514–520. [Google Scholar] [CrossRef]
- Alexander, R. Understanding Adipose-Derived Stromal Vascular Fraction (SVF) Cell Biology In Reconstructive and Regenerative Applications on the Basis of Mononucleated Cell Components 1. J. Prolotherapy 2013, 10, 15–29. [Google Scholar] [CrossRef]
- Perez-Meza, D.; Ziering, C.; Sforza, M.; Krishnan, G.; Ball, E.; Daniels, E. Hair follicle growth by stromal vascular fraction-enhanced adipose transplantation in baldness. Stem Cells Cloning Adv. Appl. 2017, 10, 1–10. [Google Scholar] [CrossRef]
- Anderi, R.; Makdissy, N.; Azar, A.; Rizk, F.; Hamade, A. Cellular therapy with human autologous adipose-derived adult cells of stromal vascular fraction for alopecia areata. Stem Cell Res. Ther. 2018, 9, 141. [Google Scholar] [CrossRef]
- Dong, Y.; Sui, L.; Yamaguchi, F.; Kamitori, K.; Hirata, Y.; Hossain, A.; Noguchi, C.; Katagi, A.; Nishio, M.; Suzuki, A.; et al. The expression of pten in the development of mouse cochlear lateral wall. Neuroscience 2014, 258, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Yoo, B.Y.; Shin, Y.H.; Yoon, H.H.; Kim, Y.J.; Seo, Y.K.; Song, K.Y.; Park, J.K. Evaluation of Human Umbilical Cord-derived Mesenchymal Stem Cells on In Vivo Hair Inducing Activity. Tissue Eng. Regen. Med. 2009, 6, 15–22. [Google Scholar]
- Richardson, S.M.; Kalamegam, G.; Pushparaj, P.N.; Matta, C.; Memic, A.; Khademhosseini, A.; Mobasheri, R.; Poletti, F.L.; Hoyland, J.A.; Mobasheri, A. Mesenchymal stem cells in regenerative medicine: Focus on articular cartilage and intervertebral disc regeneration. Methods 2016, 99, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Tsuboi, R.; Niiyama, S.; Irisawa, R.; Harada, K.; Nakazawa, Y.; Kishimoto, J. Autologous cell–based therapy for male and female pattern hair loss using dermal sheath cup cells: A randomized placebo-controlled double-blinded dose-finding clinical study. J. Am. Acad. Dermatol. 2020, 83, 109–116. [Google Scholar] [CrossRef]
- Gentile, P.; Scioli, M.G.; Bielli, A.; Orlandi, A.; Cervelli, V. Stem cells from human hair follicles: First mechanical isolation for immediate autologous clinical use in androgenetic alopecia and hair loss. Stem Cell Investig. 2017, 4, 58. [Google Scholar] [CrossRef]
- El-Khalawany, M.; Rageh MA, A.; Elnokrashy, I.; Ibrahim, S.M.A. Efficacy of autologous stromal vascular fraction injection in the treatment of androgenic alopecia. Arch. Dermatol. Res. 2022, 1–8. [Google Scholar] [CrossRef]
- Gentile, P.; Scioli, M.G.; Bielli, A.; De Angelis, B.; De Sio, C.; De Fazio, D.; Ceccarelli, G.; Trivisonno, A.; Orlandi, A.; Cervelli, V.; et al. Platelet-Rich Plasma and Micrografts Enriched with Autologous Human Follicle Mesenchymal Stem Cells Improve Hair Re-Growth in Androgenetic Alopecia. Biomolecular Pathway Analysis and Clinical Evaluation. Biomedicines 2019, 7, 27. [Google Scholar] [CrossRef]
- Shin, H.; Ryu, H.H.; Kwon, O.; Park, B.-S.; Jo, S.J. Clinical use of conditioned media of adipose tissue-derived stem cells in female pattern hair loss: A retrospective case series study. Int. J. Dermatol. 2015, 54, 730–735. [Google Scholar] [CrossRef]
- Kim, S.J.; Kim, M.J.; Lee, Y.J.; Lee, J.C.; Kim, J.H.; Kim, D.H.; Do, Y.H.; Choi, J.W.; Chung, S.I.; Do, B.-R. Innovative method of alopecia treatment by autologous adipose-derived SVF. Stem Cell Res. Ther. 2021, 12, 486. [Google Scholar] [CrossRef]
- Fukuoka, H.; Suga, H. Hair Regeneration Treatment Using Adipose-Derived Stem Cell Conditioned Medium: Follow-up With Trichograms. Eplasty 2015, 15, e10. [Google Scholar]
- Gentile, P.; Scioli, M.G.; Cervelli, V.; Orlandi, A.; Garcovich, S. Autologous Micrografts from Scalp Tissue: Trichoscopic and Long-Term Clinical Evaluation in Male and Female Androgenetic Alopecia. Biomed Res. Int. 2020, 1–10. [Google Scholar] [CrossRef]
- Lee, Y.I.; Kim, J.; Kim, J.; Park, S.; Lee, J.H. The Effect of Conditioned Media From Human Adipocyte-Derived Mesenchymal Stem Cells on Androgenetic Alopecia After Nonablative Fractional Laser Treatment. Dermatol. Surg. Off. Publ. Am. Soc. Dermatol. Surg. Al 2020, 46, 1698–1704. [Google Scholar] [CrossRef] [PubMed]
- Tak, Y.J.; Lee, S.Y.; Cho, A.R.; Kim, Y.S. A randomized, double-blind, vehicle-controlled clinical study of hair regeneration using adipose-derived stem cell constituent extract in androgenetic alopecia. Stem Cells Transl. Med. 2020, 9, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Zari, S. Short-Term Efficacy of Autologous Cellular Micrografts in Male and Female Androgenetic Alopecia: A Retrospective Cohort Study. Clin. Cosmet. Investig. Dermatol. 2021, 14, 1725–1736. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, R.G.; Rosell, J.M.C.; Ceccarelli, G.; De Sio, C.; De Angelis, G.C.; Pinto, H.; Astarita, C.; Graziano, A. Progenitor-cell-enriched micrografts as a novel option for the management of androgenetic alopecia. J. Cell. Physiol. 2020, 235, 4587–4593. [Google Scholar] [CrossRef]
- Elmaadawi, I.H.; Mohamed, B.M.; Ibrahim, Z.A.; Abdou, S.M.; El Attar, Y.A.; Youssef, A.; Shamloula, M.M.; Taha, A.; Metwally, H.G.; El Afandy, M.M.; et al. Stem cell therapy as a novel therapeutic intervention for resistant cases of alopecia areata and androgenetic alopecia. J. Dermatol. Treat. 2018, 29, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Narita, K.; Fukuoka, H.; Sekiyama, T.; Suga, H.; Harii, K. Sequential Scalp Assessment in Hair Regeneration Therapy Using an Adipose-Derived Stem Cell-Conditioned Medium. Dermatol. Surg. Off. Publ. Am. Soc. Dermatol. Surg. Al 2020, 46, 819–825. [Google Scholar] [CrossRef]
- Halilovic, E. Trichoscopic Findings in Androgenetic Alopecia. Med. Arch. 2021, 75, 109. [Google Scholar] [CrossRef]
- Morgun, E.I.; Vorotelyak, E.A. Epidermal Stem Cells in Hair Follicle Cycling and Skin Regeneration: A View from the Perspective of Inflammation. Front. Cell Dev. Biol. 2020, 8, 581697. [Google Scholar] [CrossRef]
| Patients = women and men with androgenic alopecia | Descriptions: female pattern hair loss, male pattern hair loss, female androgenic alopecia, male androgenic alopecia, alopecia in women, alopecia in men. |
| Interventions = treatment with stem cells | Descriptions: ADSC, HFSC, BMSC. |
| Comparison | Descriptions: placebo, different treatments. |
| Outcomes | Objective measurement of hair density. |
| Author, Year | Design Country | N, Age (Year) F (%) | Duration and Type of Treatment | Progression of the Disease (Scale) | Assessment | Hair Density |
|---|---|---|---|---|---|---|
| Tsuboi et al. [24], 2020 | RCT Japan | 65 33–64 23 | HFSCs 1 session n/r injections | N-H 3-6 S 3-6 | P, hair density, hair diameter, trichogram | IMPROVEMENT: total hair density increased vs. placebo (p < 0.025), an increase was greater in people ≥51 years of age |
| Gentile et al. [24], 2017 | NRCT Italy | 11 38–61 0 | HFSCs 1 session subcutaneous injections | N-H 3-5 | P | IMPROVEMENT: hair density increased by 29% ± 5% for a treated area and by less than 1% for the placebo area |
| Elmadaavi et al. [25], 2018 | RCT Egypt | 20 10–50 (26 ± 8) 60 | BMSCs and HFSCs 1 session intradermal injections | n/r | P, dermosopic examination, digital dermoscopic, and histological examination | IMPROVEMENT: increase in hair density of 52 ± 28 in subjects receiving BMMC and 42 ± 27 in subjects receiving FSC with a non-statistically significant difference |
| Zari et al. [26], 2021 | single arm, non-controlled Saudi Arabia | 140 18–65 (mean age 32.1) 80,7 | HFSCs 1 session subcutaneous injections | S 2–4, N-H 2–4 | P, trichoscopy (TrichoScan®) | IMPROVEMENT: hair density increases by +4.5 to 7.12 hair/cm |
| Narita et al. [27], 2020 | NRCT Japan | 40 23–74 47,5 | ADSCs-CM 6 sessions intradermal injections | L 1-2, N-H 2–6 | P, trichoscopy (TrichoScan®) | IMPROVEMENT: increased hair density from T0 to T6 (p < 0.001) |
| Ruiz et al. [28], 2019 | single arm, non-controlled Spain | 100 n/r n/r | HFSCs 1 session n/r injections | n/r | P, trichoscopy (TrichoScan®) | IMPROVEMENT: increased hair density of 30% ± 3.0% |
| Gentile et al. [29], 2019 | ROCA Italy | 21 25–72 28,2 | HFSCs 2 sessions subcutaneous injections | L 1-2, N-H 2-5 | P | IMPROVEMENT: increased hair density of 30 ± 5.0% for the treatment group and less than a 1% increase for the placebo |
| Kim et al. [29], 2021 | single arm, non-controlled Republic of Korea | 9 43–64 (53 ± 1.22) men: 51.5 ± 3.43; women 55,5 | ADSCs 1 session subcutaneous injections | L 1-3, N–H 4-5 | P | IMPROVEMENT: hair density increased compared to the non-treated side (p = 0.01 and p = 0.009 per each); density increased in the treated site by 48.11% as compared to the non-treated site density of 35.48% |
| Fukuoka et al. [30], 2015 | NRCT Japan | 32 20–70 40,2 | ADSCs-CM 6 sessions intradermal injections | n/r | P, trichoscopy (TrichoScope) | IMPROVEMENT: the mean increase in hair density was 29 ± 4.1 in male patients and 15.6 ± 4.2 in female patients |
| El-Khalawany et al. [26], 2022 | single arm, non-controlled Egypt | 30 21–45 (30.1 ± 6.3) 53,3 | ADSCs 1 session intradermal injections | L 1-3, N-H 1–6 | P, trichoscopy | IMPROVEMENT: hair count/cm2 showed a high statistically significant increase from 130.87 ± 14/cm2 before the study to 151.93 ± 22.36/cm2 at the 6-month follow-up visit with a 16.09% improvement |
| Shin et al. [28], 2015 | ROCS Republic of Korea | 27 22–69 (41.9 ± 13.4) 100 | ADSCs-CM 12 sessions topical wit micro-niddle roller | L 1 | medical records and phototrichographic images were analyzed | IMPROVEMENT: hair density increased from 105.4 to 122.7 hairs/cm2 over the 12 weeks of treatment (p < 0.001), representing an increase of 16.4%. |
| Gentile et al. [31], 2020 | RCT Italy | 27 n/r 37 | HFSCs 3 sessions subcutaneous injections | L 1-2, N-H 2-5 | P, phototrichograms | IMPROVEMENT: an increase of hair count and hair density, respectively, of 18.0 hairs per 0.65 cm2 and 23.3 hairs per cm2 compared with the baseline, while the control area displayed a mean decrease of 1.1 hairs per 0.65 cm2 and 0.7 hairs per cm2 (control vs. treatment: p < 0.0001) |
| Perez Meza et al. [19], 2017 | ROCS Spain | 6 18–55 11,1 | ADSCs 1 session subcutaneous injections | L 1-3, N–H 2-6 | P, trichoscopy (FotooFinder®) | IMPROVEMENT: the mean increase of hair density was 31 hairs/cm2 of (represents a 23% relative percentage increase) |
| Lee et al. [32], 2020 | RCT Republic of Korea | 30 20–61 (46.6) 50 | ADSCs-CM topical 12 weeks, 1 session weekly | n/r | P, phototrichogram | IMPROVEMENT: the hair density of the placebo group was 89.3 ± 3.79/cm2 and that of the ADSC-CM group was 102.1 ± 4.09/cm2, showing a significant difference (p < 0.05) |
| Tak et al. [33], 2020 | RCT Republic of Korea | 38 45.3 23,7 | ADSCs-CM topical 16 weeks twice daily | n/r | P, phototrichogram | IMPROVEMENT: in the treatment group the mean hair density and thickness increased by 28.1% and 14.2% by 16 weeks, which were 3.95 and 2.25 times those in control group using the vehicle placebo |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krefft-Trzciniecka, K.; Piętowska, Z.; Nowicka, D.; Szepietowski, J.C. Human Stem Cell Use in Androgenetic Alopecia: A Systematic Review. Cells 2023, 12, 951. https://doi.org/10.3390/cells12060951
Krefft-Trzciniecka K, Piętowska Z, Nowicka D, Szepietowski JC. Human Stem Cell Use in Androgenetic Alopecia: A Systematic Review. Cells. 2023; 12(6):951. https://doi.org/10.3390/cells12060951
Chicago/Turabian StyleKrefft-Trzciniecka, Katarzyna, Zuzanna Piętowska, Danuta Nowicka, and Jacek C. Szepietowski. 2023. "Human Stem Cell Use in Androgenetic Alopecia: A Systematic Review" Cells 12, no. 6: 951. https://doi.org/10.3390/cells12060951
APA StyleKrefft-Trzciniecka, K., Piętowska, Z., Nowicka, D., & Szepietowski, J. C. (2023). Human Stem Cell Use in Androgenetic Alopecia: A Systematic Review. Cells, 12(6), 951. https://doi.org/10.3390/cells12060951







