Abstract
Immobilized microorganism technology offers a promising approach for remediating heavy metal-contaminated soils. This study developed a novel bio-mineral composite (B-AM) by coupling acid-modified maifanite (AM) with Bacillus mucilaginosus to enhance lead (Pb) immobilization. Comparative experiments demonstrated that B-AM outperformed conventional amendments, including oyster shell, pristine maifanite, AM and B. mucilaginosus in Pb immobilization. The B-AM treatment optimized soil pH, improved soil fertility with increases in available potassium (1.06-fold) and available phosphorus (1.28-fold). Additionally, B-AM transformed Pb into more stable fractions, reducing labile Pb fractions by 52.52% while increasing the residual fraction by 88.36%. These improvements resulted in an 83.24% reduction in Pb accumulation and a 63.95% increase in the fresh root weight of radish. Mechanistic insights revealed that the enhanced remediation performance stems from both the individual contributions of AM (adsorption capacity) and B. mucilaginosus (biosorption and biomineralization) and their synergistic interaction. Specifically, AM acts as a carrier and pH buffer, promoting microbial proliferation and reducing Pb remobilization from cell lysis. The resulting sustained microbial activity further leads to the formation of stable Pb minerals. Collectively, our results establish a theoretical and practical basis for using B-AM to remediate Pb-contaminated soils.
1. Introduction
Lead (Pb), a highly toxic and persistent heavy metal with strong bioaccumulation potential, is widespread in the environment [1]. Soil Pb contamination poses an escalating global threat due to its concealed nature, nonbiodegradability, and tendency to accumulate in terrestrial ecosystems. Recent studies indicate that approximately 16% of global farmland exhibits excessive concentrations of toxic metals, with Pb among the predominant contaminants [2]. A World Health Organization report estimated that long-term Pb exposure caused approximately 0.9 million deaths in 2019, with 21.7 million disability-adjusted life years [3]. Pb can enter the human body via inhalation, ingestion, or dermal absorption. Once absorbed, it is not readily eliminated through normal metabolic processes, leading to progressive accumulation in tissues [1]. When physiological thresholds are exceeded, Pb can cause severe damage to the hematopoietic, renal, and reproductive systems [4]. Consequently, effective remediation of Pb-contaminated soil is imperative to mitigate these health risks and ensure food safety.
For farmland soils with low to moderate levels of contamination, immobilization remediation is a widely adopted technique. This approach reduces the activity and bioavailability of heavy metals by applying immobilizing agents that convert metals into insoluble, low-mobility, and low-toxicity forms. Commonly used immobilization agents include burnt lime, oyster shell (OS), and maifanite (M). Burnt lime primarily immobilizes soil Pb through precipitation, while OS and M achieve immobilization via precipitation, chemisorption, or formation of crystalline compounds [5,6,7]. However, long-term soil acidification can remobilize previously immobilized Pb, reducing treatment effectiveness. Furthermore, repeated annual applications increase costs and may lead to soil compaction. Therefore, achieving efficient and stable remediation of Pb-contaminated soils remains an urgent challenge. Conventional remediation methods, particularly physicochemical approaches, are often constrained by high energy consumption, substantial costs, and insufficient long-term stability [8,9]. Integrating green technologies such as microbial-assisted immobilization into existing approaches may offer a sustainable solution and merits further investigation.
Microbial remediation has garnered increasing attention due to its efficiency, environmental compatibility, and cost-effectiveness. Certain microorganisms can alter the fractionation of heavy metals and reduce their toxicity through adaptive mechanisms, such as biotransformation under metal stress [10]. Bacillus mucilaginosus exhibits notable tolerance to heavy metals and demonstrates adaptive responses under stress conditions. For example, Shen et al. [11] observed enhanced urease activity in this species under cadmium (Cd) stress, enabling bioavailable Cd2+ mineralization and removal from aqueous systems. Bybin et al. [12] also reported its arsenic tolerance and the formation of highly structured, honeycomb-like colonies that facilitate heavy metal immobilization. However, research on the capacity of B. mucilaginosus for Pb immobilization remains limited, and existing studies have predominantly focused on aqueous systems, resulting in a significant knowledge gap regarding its efficacy in complex soil matrices. Moreover, B. mucilaginosus improves soil nutrient availability and fertility. Li et al. [13] demonstrated that its co-application with organic amendments significantly increases soil carbon, nitrogen, and phosphorus levels. Similarly, Sattar et al. [14] documented its plant-growth-promoting traits, including phytohormone production, nitrogen fixation, phosphate solubilization, root development, and antibiotic synthesis. These multifunctional properties position B. mucilaginosus as promising candidates for commercial soil remediation conditioners, offering a viable eco-friendly approach for sustainable agricultural production.
In recent years, immobilized microbial technology has gained increasing application in remediation efforts. This approach physically or chemically confines free microorganisms within carrier materials, providing benefits such as high biomass retention, prolonged microbial activity, and enhanced operational stability [15]. It effectively addresses key limitations of free-living microorganisms in contaminated soils, including low survival rates, poor establishment of dominant populations, and the risk of heavy metal re-release after cell death. Maifanite has been shown to enhance microbially driven biogeochemical processes, and previous research highlights its potential as an effective immobilization carrier [16]. Notably, acid modification increases the specific surface area of materials, which not only enhances their adsorption capacity but also makes them more suitable as microbial carriers [17]. Consequently, the coupling of B. mucilaginosus with acid-modified maifanite (AM) offers a promising strategy for remediating heavy metal-contaminated soils, capitalizing on the superior adsorption properties of the mineral and the effective bioimmobilization promoted by the bacteria. However, the potential of coupling B. mucilaginosus and AM for Pb contamination remediation remains relatively unexplored, particularly the underlying interaction mechanisms responsible for long-term Pb stabilization.
This study first systematically evaluated the performance of AM and B. mucilaginosus in adsorbing and removing Pb from aqueous solution, with material and microbial characterization conducted. Based on these findings, AM was employed as a carrier for immobilizing B. mucilaginosus, leading to the development of a novel coupled agent (B-AM). The remediation performance of the B-AM was systematically evaluated in Pb-contaminated soil through pot experiments, with comparative assessment against traditional and individual treatments, including OS, pristine M, AM, and free B. mucilaginosus (B). Treatment efficacy was comprehensively evaluated through measurements of soil pH, key fertility parameters, Pb bioavailability, Pb accumulation in indicator crops and plant weight. Furthermore, the remediation mechanisms of the B-AM were thoroughly investigated. This research seeks to establish a scientific basis for innovative strategies to remediate Pb-contaminated soils, thereby safeguarding environmental health and ensuring food security.
2. Materials and Methods
2.1. Soil and Materials
2.1.1. Soil Collection and Characterization
Soil samples were collected from a government-designated contaminated paddy field (26°57′58″ N, 117°44′19″ E) near a chemical plant in Dagan Town, Nanping City, Fujian Province. Topsoil samples (0–20 cm depth) were air-dried, homogenized, and sieved (60-mesh). Soil pH was determined using the CaCl2 extraction method, and organic matter was quantified using the hydration heat-K2Cr2O7 colorimetric method. Ammonium nitrogen was analyzed by flow injection analysis. Available potassium was extracted with NH4OAc and quantified by flame photometry. Available phosphorus was extracted with NaHCO3 and determined by the molybdenum blue colorimetric method. Total Pb content was assessed via microwave-assisted acid digestion, which revealed a concentration of 145.2 mg·kg−1, indicating a moderate level of lead contamination. The other physicochemical properties of the soil are summarized in Table S1.
2.1.2. Sources and Preparation of Immobilization Agent
Pristine OS and M were provided by The Second Geological Exploration Institute of China Geological Survey Bureau (Fuzhou, China). AM samples were prepared by treating 50 g of pristine M with HCl solutions at varying concentrations (0.5, 1.0, 1.5, and 2.0 mol·L−1) under continuous stirring for 2 h. After acid treatment, each sample was rinsed repeatedly with distilled water until neutral pH was achieved and filtered. All obtained materials, including pristine OS, M and AM samples, were ground and sieved through a 100-mesh sieve before further use.
B. mucilaginosus (SHBCC D12018) was obtained from the Shanghai Bioresource Collection Center (Shanghai, China). The strain was activated and pre-cultured in a nitrogen-free medium with the following composition (per liter): glucose, 10 g; NaCl, 2 g; MgSO4·7H2O, 2 g; KH2PO4, 2 g; CaCO3, 5 g; pH 7.2. Large-scale fermentation was conducted using a nitrogen-containing medium containing (per liter): sucrose, 10 g; yeast extract, 1 g; NaCl, 1 g; MgSO4, 0.2 g; K2HPO4, 0.5 g; pH 7.2. All media were sterilized by autoclaving at 121 °C for 20 min.
The composite immobilizing agent (B-AM) was prepared by immobilizing B. mucilaginosus onto AM. The bacterial strain was pre-cultured in a nitrogen-free liquid medium at 30 °C and 150 r·min−1 for 48 h. The seed culture was inoculated at 5% (v/v) into a nitrogen-containing liquid medium and fermented under the same conditions for 72 h. The resulting bacterial culture was mixed with sterilized AM at a 1:2 (v/w) ratio under stirring for 20 min. The mixture was subsequently spray-dried with the following parameters: feed rate 8 mL·min−1, nozzle duration 10 s, fan frequency 30 Hz, inlet temperature 130 °C, and outlet temperature 80 °C. In addition, B. mucilaginosus enumeration was performed using the standard plate count method to confirm the B. mucilaginosus loading in the B-AM composite.
2.2. Pb Adsorption and Removal Experiments in Aqueous Solution
Batch experiments were conducted to evaluate the Pb2+ removal capabilities of the pristine maifanite (M), acid-modified maifanite (AM), and B. mucilaginosus separately. For the M and AM, 0.5 g of each material was added to 50 mL of Pb2+ solution and agitated at 150 rpm. For Pb removal by B. mucilaginosus, 1.0 g of wet biomass was introduced into 20 mL of Pb2+ solution under identical shaking conditions. The effects of pH (2.0, 3.0, 4.0, 5.0, 5.5), initial Pb2+ concentration (50, 100, 200, 300, 400, 500 mg·L−1), reaction time (10, 30, 60, 90, 120, 150, 180 min), and temperature (10, 20, 25, 30, 35, 40 °C) on Pb2+ removal were investigated. Unless specifically varied, the default conditions were maintained at pH 5.5, Pb2+ concentration of 300 mg·L−1, temperature of 30 °C, and reaction time of 120 min. After each experiment, the mixture was centrifuged and the supernatant analyzed to determine residual Pb2+ concentration and removal efficiency.
2.3. Characterization of Materials
The morphological characteristics of pristine M, AM, B. mucilaginosus, post-Pb-removal B. mucilaginosus, and the B-AM were examined. Material characterization was performed using the following techniques: surface morphology was observed via scanning electron microscopy (SEM; Verios G4, Thermo Fisher Scientific, Waltham, MA, USA), with elemental composition simultaneously determined by energy-dispersive X-ray spectroscopy (EDS; System SIX, Thermo Fisher Scientific, USA); Brunauer–Emmett–Teller (BET) surface area, pore volume and average pore width were analyzed by Automated Gas Sorption Analyzer (ASAP 2020 HD88, Micromeritics, Norcross, GA, USA); crystalline structures were analyzed by X-ray diffraction (XRD; MiniFlex 600, Rigaku, Tokyo, Japan); surface functional groups were identified using Fourier-transform infrared spectroscopy (FT-IR; Nicolet iS10, Thermo Fisher Scientific, USA).
2.4. Pot Experiment for Soil Remediation
2.4.1. Experimental Design
Pot experiments were conducted to evaluate the effectiveness of various immobilizing agents in remediating Pb-contaminated soil. Cherry radish (Raphanus sativus L. var. radiculus pers) was selected as the indicator crop due to its short growth cycle (60–80 days from sowing to harvest) and high sensitivity to heavy metal stress, making it an ideal species for efficiently monitoring soil contamination [18]. Plastic pots (10 cm × 10 cm × 10 cm) were each filled with 1 kg of contaminated soil. Six treatments were applied: untreated soil (CK), OS, M, AM, B, and B-AM. Each treatment was applied at a rate of 30 g·kg−1, with three replicates per treatment. Cherry radish seedlings, pre-germinated in darkness, were transplanted into the pots. The plants were grown under a 12 h/12 h light/dark cycle with artificial lighting and irrigated every 48 h to maintain soil moisture at 60% of the water-holding capacity. After 90 days of growth, the plants were harvested for analysis.
2.4.2. Analysis of Soil Remediation Indicators
To evaluate the overall effect of the immobilization treatments on soil quality, changes in soil pH and fertility parameters before and after remediation were compared. Then, soil Pb fractionation was determined using a modified Tessier sequential extraction method. The Tessier method categorizes soil-bound Pb into five operationally defined fractions, in order of decreasing mobility and bioavailability: exchangeable (T1), carbonate-bound (T2), Fe/Mn oxide-bound (T3), organic matter-bound (T4), and residual (T5). The effectiveness and stability of Pb immobilization were evaluated by comparing the distribution of these fractions between treated soils and the CK.
The bioavailable Pb fraction was extracted using the diethylenetriaminepentaacetic acid method [15]. Specifically, 2.0 g of sieved soil (100-mesh) was placed in a centrifuge tube, mixed with 10 mL of Diethylenetriaminepentaacetic acid extractant, and shaken at 180 rpm and 25 °C for 2 h. The mixture was then centrifuged at 5000 rpm for 10 min. The Pb concentration in the supernatant was measured to quantify the readily bioavailable fraction, which primarily includes exchangeable and carbonate-bound Pb. These forms are highly mobile and pose significant risks due to their potential for plant uptake and entry into the food chain. The immobilization efficiency was calculated using Equation (1):
where E is the immobilization efficiency (%), w1 is the bioavailable Pb concentration in the control soil (mg·kg−1), and w2 is the bioavailable Pb concentration in the treated soil (mg·kg−1).
Since the root is the primary edible part of radish and serves as the main accumulation site for Pb [19], our assessment of Pb enrichment and associated health risks specifically targeted root samples, which were processed using a dry ashing protocol. Porcelain crucibles were pre-treated by soaking in 20% HNO3 overnight, rinsed thoroughly with deionized water, and oven-dried. Subsequently, 2.0 g of finely ground plant tissue was weighed into each crucible. Samples were carbonized on a hotplate until smoke emission ceased, then ashed in a muffle furnace at 500 °C for 6 h. After cooling, the ash was dissolved in 1 mL of concentrated HNO3 and quantitatively transferred to a 10 mL volumetric flask. The crucible was rinsed three times with 2 mL deionized water, and the rinsates were combined into the flask. The solution was diluted to volume, mixed thoroughly, and analyzed for Pb concentration. Following the Pb concentration analysis, the fresh weight of the radish roots was measured to assess the improvement in soil productivity and plant growth resulting from the remediation treatments.
2.5. Data Analysis
All statistical analyses were performed using SPSS 23.0 (IBM, Armonk, NY, USA). Data were expressed as mean ± standard deviation of three replicates. One-way analysis of variance followed by Duncan’s multiple range test was used to determine significant differences between treatments at p < 0.05. Data visualizations were generated using Origin 2018 (OriginLab, Northampton, MA, USA).
3. Results and Discussion
3.1. Pb Adsorption and Characterization of Acid-Modified Maifanite
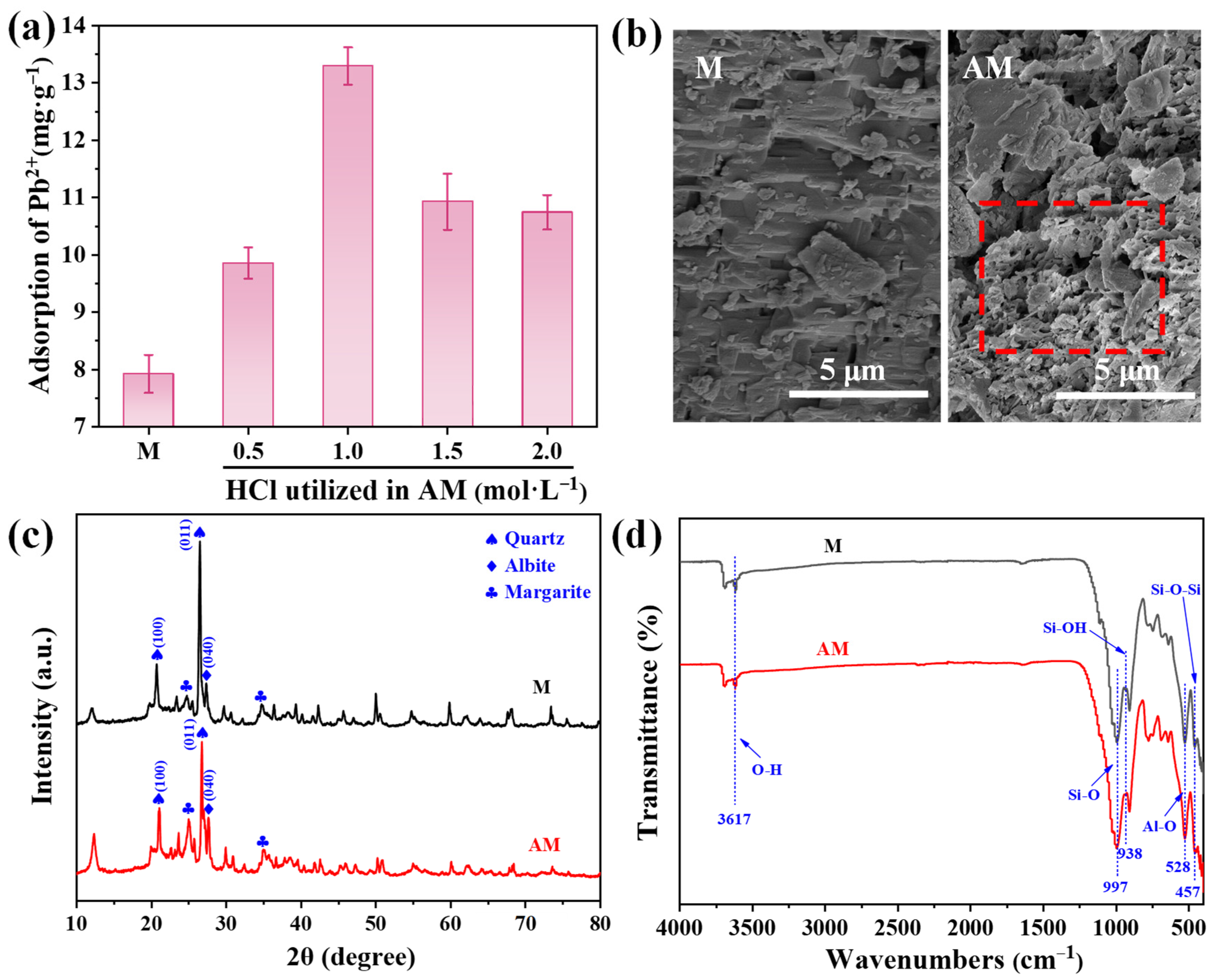
The Pb2+ adsorption performance of pristine M and AM treated with different HCl concentrations is shown in Figure 1a. All AM samples exhibited enhanced Pb2+ adsorption compared to pristine M. The sample modified with 1.0 mol·L−1 HCl demonstrated the highest adsorption capacity (13.29 mg·g−1), representing a 1.68-fold increase over the unmodified material. Given its superior performance, the optimally modified AM (treated with 1.0 mol·L−1 HCl) and pristine M were selected for further characterization to analyze their key physicochemical properties.
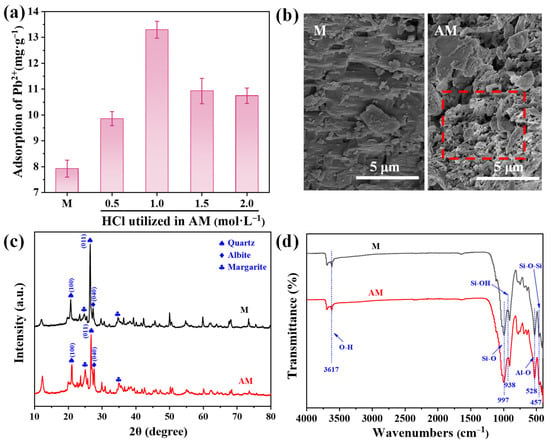
Figure 1.
(a) Pb2+ adsorption capacity of pristine maifanite (M) and acid-modified maifanite (AM) prepared at different HCl concentrations; (b) SEM images, (c) XRD patterns, and (d) FT-IR spectra of M and AM.
SEM analysis revealed distinct differences in surface morphology between the M and AM (Figure 1b). Pristine M exhibited a secondary dissolution pore structure, characterized by laminated and sponge-like channels. However, these channels appear partially obstructed by surface impurities. In contrast, AM displayed noticeably rougher particle surfaces and more open porous structures, which facilitate contaminant adsorption. BET analysis further showed that AM possessed a larger specific surface area (13.19 m2·g−1) and pore volume (0.04 cm3·g−1) than pristine M (Table S2), providing more active sites for Pb2+ adsorption.
XRD patterns indicated that acid modification did not alter the fundamental mineral composition of maifanite (Figure 1c). The diffractogram of pristine M exhibits characteristic diffraction peaks at 2θ = 20.8°and 26.5°, which correspond to the (100) and (011) planes of quartz (SiO2) [20]. The peak at 2θ = 27.3° is attributed to the (040) plane of albite (Na2O·Al2O3·6SiO2) [21], while those at 2θ = 24.8° and 34.8° are assigned to the (002) and (130) planes of margarite (CaAl2(Si2Al2)O10(OH)2), respectively [22]. Although no new peaks appeared after acid treatment, the reduction in peak intensities indicated effective removal of surface impurities and pore blockages [23], thereby enhancing the availability of adsorption sites.
FT-IR spectroscopy confirmed the preservation of key functional groups after acid modification (Figure 1d). Both samples exhibited similar spectra, including O–H stretching vibration at 3617 cm−1, Si–O stretching at 997 cm−1 [24], Si–OH stretching at 938 cm−1 [25], Al–O vibrations at 528 cm−1 [26], Si–O–Si stretching (in-plane bending vibrations) at 457 cm−1 [25], and complex vibrational modes between 400–500 cm−1 associated with Si–O and Al–O structures [27]. The consistency in FT-IR spectra indicates that acid modification did not alter the fundamental mineralogy of maifanite but primarily changed its morphology, leading to improved Pb2+ adsorption capacity.
3.2. Efficiency and Characterization of Pb Removal by B. mucilaginosus
3.2.1. Pb Removal Efficiency by B. mucilaginosus
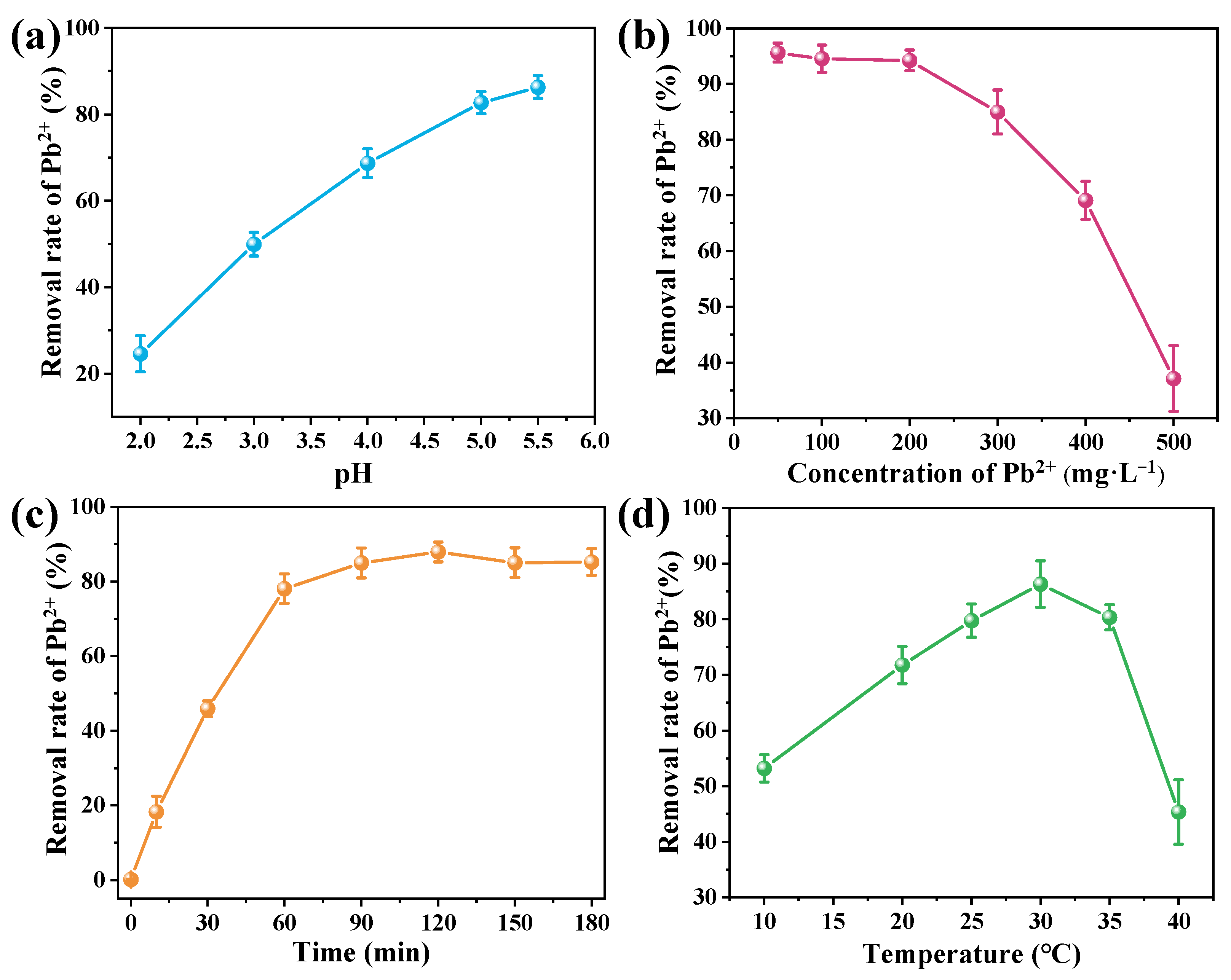
Microbial Pb removal is a complex process influenced by multiple environmental factors. To assess the potential of B. mucilaginosus for mitigating Pb contamination, its Pb removal efficiency was systematically evaluated under varying conditions of pH, initial Pb2+ concentration, reaction time, and temperature (Figure 2). The effect of pH was investigated within the range of 2.0–5.5 to prevent Pb(OH)2 precipitation and ensure removal was primarily microbial-mediated. Removal efficiency increased progressively with pH, reaching an optimum of 86.26% at pH 5.5. This enhancement is mainly due to reduced proton competition for binding sites and increased deprotonation of functional groups such as carboxyl, phosphoryl, and amino residues, which strengthens electrostatic attraction and complexation with Pb2+ ions [28]. The initial Pb2+ concentration significantly affected removal performance, which exceeded 90% at concentrations between 50–200 mg·L−1. Beyond this range, performance declined due to saturation of binding sites on bacterial cells. Reaction time experiments revealed rapid removal within the first 60 min, followed by a gradual approach to equilibrium at 120 min, with a maximum efficiency of 87.92%. This profile suggests initial rapid surface binding followed by slower biomineralization and intracellular uptake. Temperature exerted a substantial influence on removal efficiency, which increased with temperature up to an optimum of 30 °C (removal efficiency 86.33%), beyond which performance declined due to thermal stress on microbial activity and protein integrity. These results demonstrate the effective removal of Pb2+ from aqueous solutions by B. mucilaginosus. Its high removal efficiency and environmental resilience indicate strong potential for remediating Pb-contaminated soils, providing a scientific basis for developing practical bioremediation strategies.
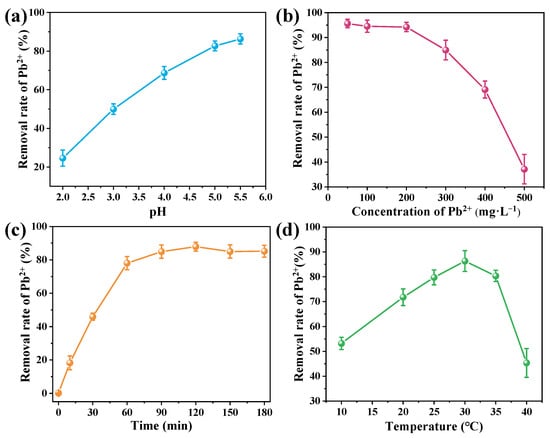
Figure 2.
Effects of (a) pH, (b) initial Pb2+ concentration, (c) reaction time, and (d) temperature on Pb2+ removal by B. mucilaginosus.
3.2.2. Characterization of B. mucilaginosus After Pb Removal
To elucidate the morphological and compositional changes of B. mucilaginosus following Pb2+ removal, a series of characterization analyses were conducted. SEM analysis was conducted to visualize morphological changes and elemental distribution in B. mucilaginosus following Pb exposure. As shown in Figure 3, untreated cells exhibited smooth and intact rod-like morphologies. In contrast, Pb-treated cells displayed notably rough and wrinkled surfaces covered with dense crystalline ellipsoidal particles. Elemental mapping confirmed the presence of C (32.37%), O (32.06%), N (14.35%), P (6.60%), Cl (3.31%), and Pb (12.91%) (Figure S1). The C, N, O, and P signals originated from organic cell matrices and EPS, while Cl likely originates from extracellular chloride sources. The significant accumulation of lead (12.91%) demonstrates the definitive immobilization of Pb by B. mucilaginosus.

Figure 3.
SEM images of B. mucilaginosus (a) before and (b) after Pb2+ removal.
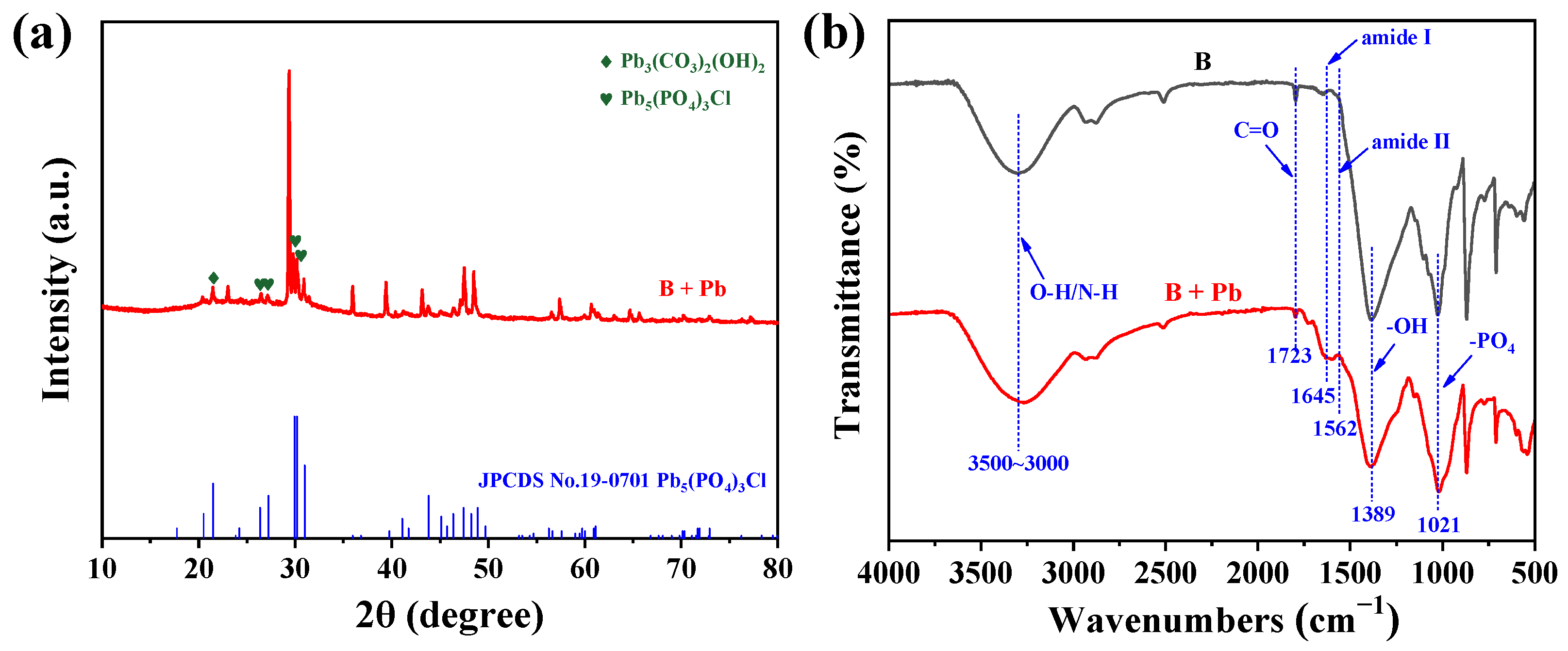
XRD analysis was performed to identify the crystalline phases of the Pb-containing particles. As shown in Figure 4a, diffraction peaks emerged after treatment, including a signal at 2θ = 21.4°, corresponding to Pb3(CO3)2(OH)2 [29], and peaks at 26.5°, 27.2°, 30.2° and 31.0° consistent with pyromorphite [Pb5(PO4)3Cl] (JCPDS 19-0701) [30]. The formation of Pb5(PO4)3Cl is likely attributed to the reaction between Pb2+ and phosphate groups derived from the bacterial EPS [30]. These results confirm the formation of thermodynamically stable pyromorphite. Such phases are known to remain highly stable across a wide pH range (3–9) and are considered one of the most persistent forms of Pb in soil environments [31].
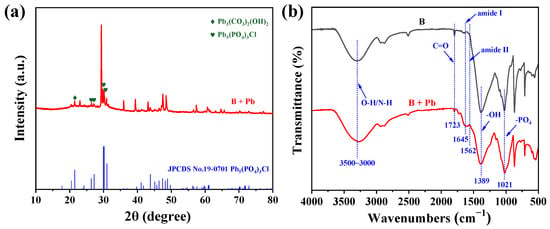
Figure 4.
(a) XRD patterns and (b) FT-IR spectra of B. mucilaginous before (B) and after Pb2+ removal (B + Pb).
To gain insight into the molecular-level interactions between functional groups and Pb, FT-IR spectroscopy was employed (Figure 4b). For untreated B. mucilaginosus, a broad absorption band from 3500 to 3000 cm−1 was observed, corresponding to O–H and N–H stretching vibrations [32,33]. Other characteristic peaks were identified at 1723 cm−1 (aldehyde C=O), and 1021 cm−1 (–PO4). These functional groups are abundantly present in the EPS (e.g., carboxyl, phosphate, amino, and hydroxyl groups) secreted by the bacterial cells and play a critical role in metal binding [34]. After Pb adsorption, several spectral shifts were noted: the O–H stretching band shifts from 3300 to 3270 cm−1, the aldehyde C=O signal shifts from 1723 to 1732 cm−1. Notably, the broadening of the –PO4 band at 1021 cm−1 further indicates complexation between Pb and phosphate groups hydrogen bonding and other associations [35]. Furthermore, the increased intensity of the amide I (1645 cm−1) and II (1562 cm−1) bands suggest that EPS proteins play a more substantial role in the biomineralization process. These FT-IR results collectively elucidate the critical role played by the functional groups of B. mucilaginosus, particularly those within its EPS, in the immobilization and adsorption of Pb.
3.3. Coupling of B. mucilaginosus and Acid-Modified Maifanite
The composite interface formed between AM and B. mucilaginosus was characterized using SEM. The increased surface roughness and porosity of AM resulting from acid modification facilitated microbial adhesion. As shown in Figure 5, extensive bacterial colonization was observed, with numerous cells tightly adhered to the AM surface and effectively anchored within its porous structure. These morphological findings confirm the successful preparation of the B-AM composite, which provides the structural basis for its remediation function. The plate counting method was used to quantitatively assess bacterial survival rates before and after spray drying. As shown in Table 1, the survival rate of free B. mucilaginosus after spray drying was only 24.15 ± 1.07%. In contrast, the survival rate of B. mucilaginosus immobilized on AM was significantly higher (56.18 ± 1.72%). This demonstrates that the AM provides a protective effect during spray drying, likely due to its porous structure, which offers physical shelter and mitigates the direct exposure of bacterial cells to harsh conditions.

Figure 5.
SEM images of the composite formed by coupling B. mucilaginosus with acid-modified Maifanite.

Table 1.
The survival rates of B. mucilaginosus after spray drying.
The developed B-AM composite system shows promising potential for the remediation of Pb-contaminated soils. In this system, AM rapidly adsorbs Pb2+ ions via its high adsorption capacity, while the B. mucilaginosus promotes the biomineralization of Pb into stable mineral phases. This synergistic mechanism is expected to substantially improve the efficiency of Pb immobilization. Although aqueous adsorption studies are commonly used for preliminary evaluation of adsorbents, this research is specifically aimed at developing a deployable soil remediation technology. Given the complexity of soil environments, which includes factors such as organic matter, competing ions, and pH fluctuations, our assessment focused directly on the behavior of B-AM in Pb-contaminated soil. Accordingly, pot experiments were conducted to evaluate the impact of B-AM on soil properties, Pb speciation transformation, and plant Pb accumulation, offering a comprehensive assessment of its practical remediation efficacy.
3.4. Remediation Performance of the Immobilizing Agent
3.4.1. Improvement of Soil Properties
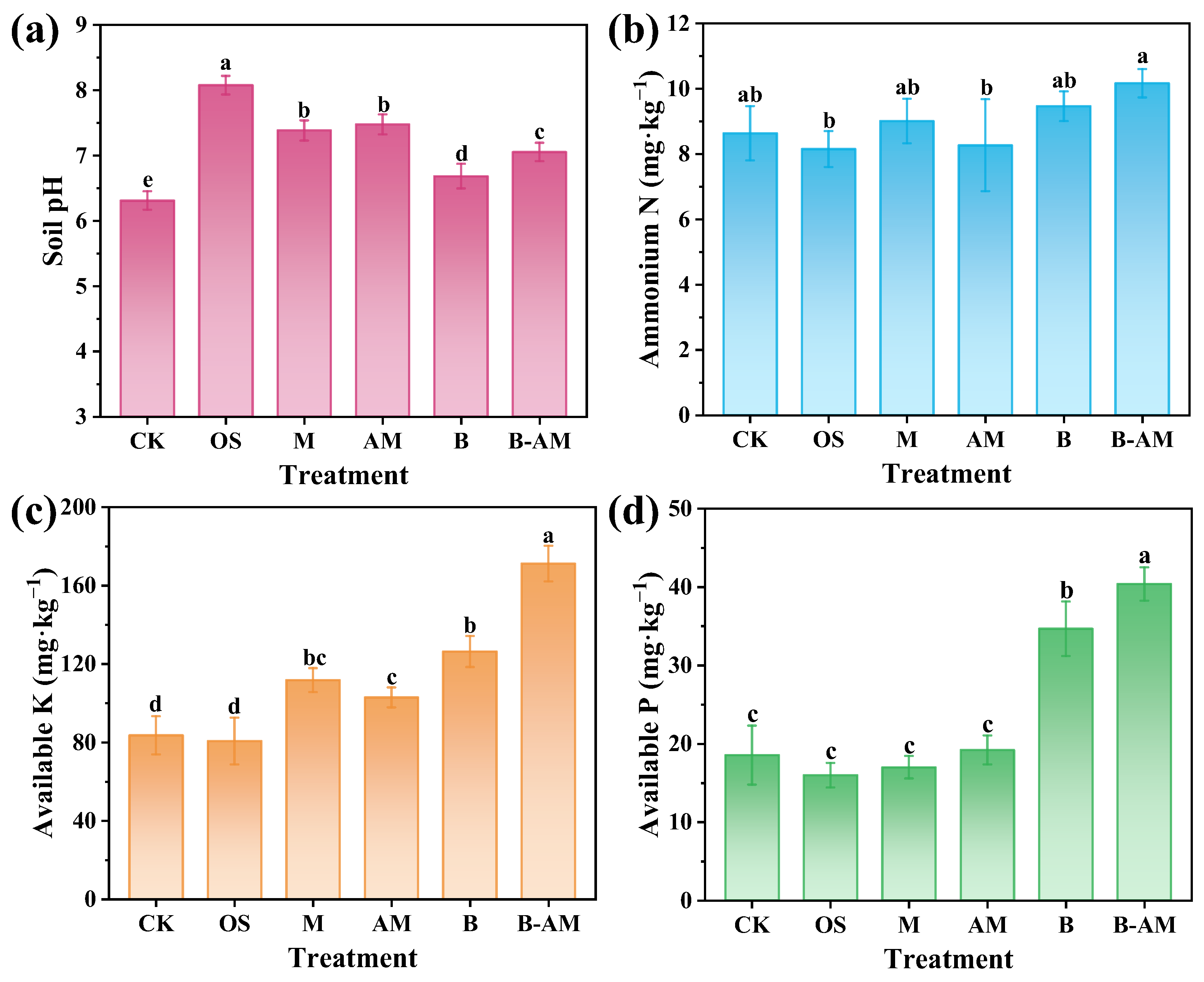
Changes in key soil properties, including pH and fertility-related parameters, were systematically evaluated after the application of different amendments. As shown in Figure 6a, all treatments significantly increased soil pH (p < 0.05). This increase is critical for remediation, as a higher pH promotes metal immobilization through enhanced precipitation. OS exhibited the greatest increase, raising pH to 8.1, primarily due to its CaCO3 content, which effectively neutralizes soil acidity. It should be noted that this pH level approaches the upper threshold of the optimal range for many crops [36,37]. Similarly, both M and AM also induced a moderate soil pH increase to approximately 7.4, attributable to the proton consumption capacity of their Al2O3 content. The treatment of B induced a slight pH increase to 6.7, which can be explained by the opposing effects of two metabolic processes: ammonium ions released from amino acid decomposition by B. mucilaginosus tend to increase pH, while concurrently secreted bio-organic acids counteract this alkalinization effect [38]. In the B-AM treatment, the AM improved soil pH buffering capacity due to its inherent alkaline mineral constituents, maintaining a stable pH of approximately 7.1. The resulting neutral pH not only facilitates Pb immobilization via enhanced adsorption and precipitation but also lies within the range suitable for most crops.
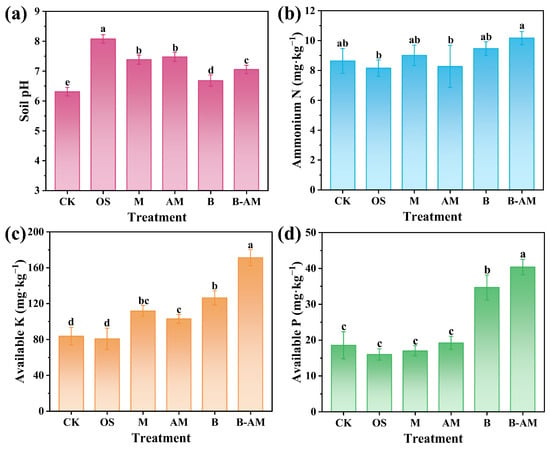
Figure 6.
Soil properties across different treatment groups: (a) pH, (b) ammonium nitrogen, (c) available K, and (d) available P. (CK: untreated soil; OS: oyster shell; M: pristine maifanite; AM: acid-modified maifanite; B: B. mucilaginosus; B-AM: coupling of B. mucilaginosus and acid-modified maifanite). Different letters indicate significant differences among treatments, with p < 0.05 level.
Soil NH4+–N is a key indicator of soil nitrogen availability. As shown in Figure 6b, although slightly higher NH4+–N content was observed in the B and B-AM treatments compared to the control, these differences were not statistically significant (p > 0.05).These subtle elevations may be attributed to the mineralization of organic nitrogen, mediated by B. mucilaginosus, which releases NH4+–N [39]. The lack of significant differences is likely explained by concurrent NH4+ consumption processes, including microbial assimilation, nitrification, and volatilization [40]. Notably, the B-AM treatment resulted in higher NH4+–N levels compared to the B treatment alone, suggesting that AM improved microbial activity and facilitated sustained organic nitrogen transformation.
The changes in available K content following the application of different amendments are presented in Figure 6c. Significant differences were observed among treatments, with the overall trend following the order: B-AM > B > M > AM > OS. No significant change was observed in the OS treatment (p > 0.05), whereas M and AM significantly increased available K (p < 0.05). This phenomenon occurs because maifanite, as a silicate mineral, undergoes gradual weathering and dissolution, a process that liberates K+ ions from its crystalline lattice structure [41]. The B treatment showed enhanced potassium solubilization, consistent with the known behavior of B. mucilaginosus as a potassium-solubilizing microorganism, which converts insoluble K to plant-available forms via organic acid secretion [42]. The B-AM treatment achieved the highest available K concentration, reaching a level 2.06-folds that of the CK. This significant improvement resulted from the synergistic interaction between mineral and microbial.
Soil phosphorus exists predominantly in recalcitrant forms, including organic types such as phytic acid and inorganic types such as apatite and secondary phosphate minerals. Only a small fraction is directly available to plants as orthophosphate [43]. Figure 6d shows no significant changes in available P across the M, AM, and OS treatments (p > 0.05). In contrast, the B and B-AM treatments significantly increased soil available P (p < 0.05), achieving 1.87-fold and 2.28-fold enhancements compared to CK, respectively. This improvement is attributed to the phosphate-solubilizing activity of B. mucilaginosus, consistent with its known role as a phosphate-solubilizing microorganism [44]. B. mucilaginosus can secrete organic acids to dissolve inorganic phosphorus minerals through acidification and chelation, converting insoluble forms to plant-available orthophosphates [45]. They can also produce phosphatases that hydrolyze organic phosphorus compounds [46], a process that releases soluble phosphate and mobilizes recalcitrant phosphorus pools. Notably, the B-AM treatment outperformed the B treatment alone, likely due to the AM providing improved microbial survival and sustained activity, which enhances both organic acid secretion and phosphatase production over time.
3.4.2. Reduction in Pb Bioavailability
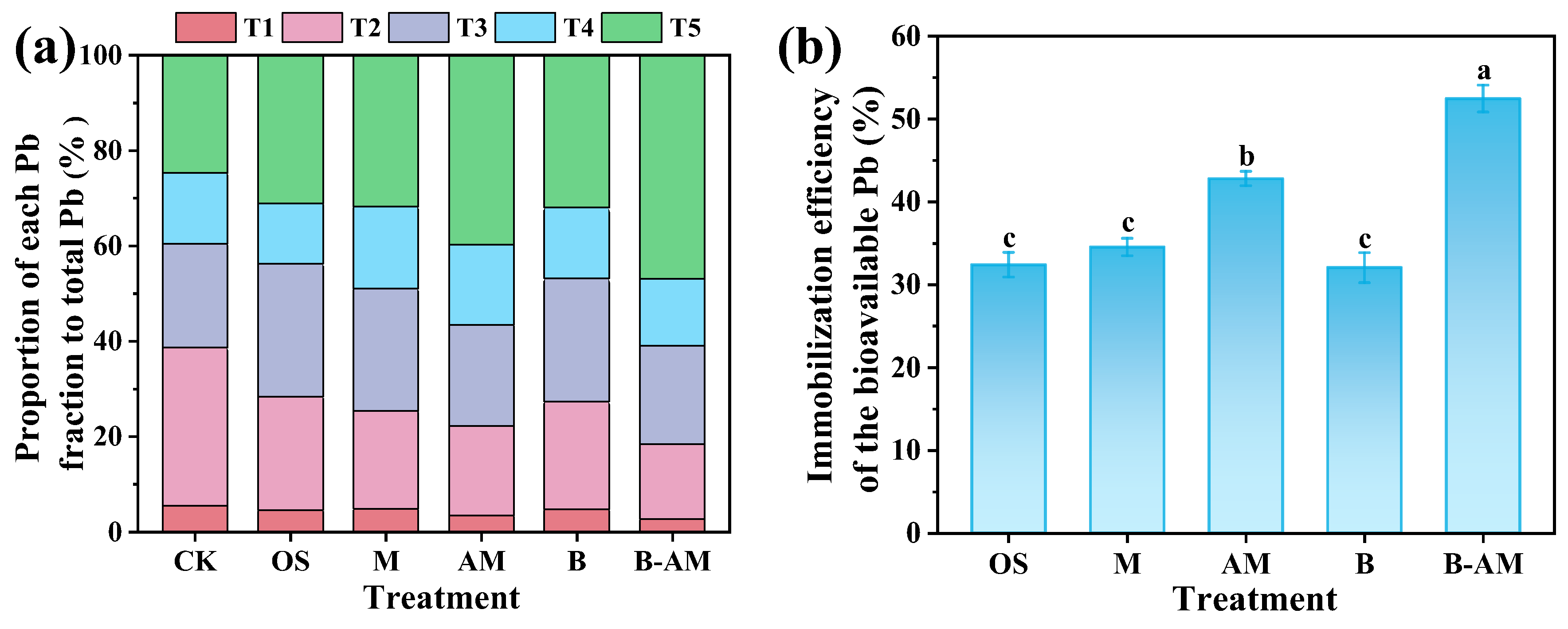
The application of immobilization agents markedly altered the fractionation patterns of Pb in the soil. As illustrated in Figure 7a, distinct fractionation profiles were observed across the treatment groups. In the CK, Pb was predominantly present in the T2 fraction (33.87%), whereas T1 (5.27%) accounted for the smallest proportion. The remaining Pb was distributed as follows: 20.52% in the T3, 15.17% in the T4, and 25.17% in the T5. Notably, both T1 and T2 are characterized by high mobility and activity, collectively referred to as the labile fraction of soil Pb, which has been positively correlated with oral bioaccessibility [47]. Collectively, these labile fractions (T1 + T2) constituted 39.14% of the total Pb, highlighting the need for effective remediation strategies to mitigate environmental mobility and associated health risks. After immobilization remediation, all amendments significantly (p < 0.05) reduced the proportion of the labile Pb fractions (T1 + T2) by 26.71–52.52%, while simultaneously increasing the T5 by 26.10–88.36%. In contrast, the T3 and T4 fractions remained largely unchanged. Among all treatments, B-AM exhibited the most pronounced transformation efficacy, achieving a 53.41% reduction in the T2 and an 88.36% increase in the T5. The superior performance arises from synergy between AM and B. mucilaginosus: AM enhances bacterial viability and function, leading to intensified biomineralization, while the resulting biogenic minerals, combined with AM’s own adsorption capabilities, collectively convert labile Pb into more stable forms.
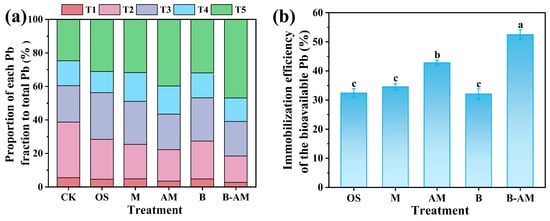
Figure 7.
(a) Changes in Pb fractionation and (b) immobilization efficiency of bioavailable Pb across different treatment groups. (CK: untreated soil; OS: oyster shell; M: pristine maifanite; AM: acid-modified maifanite; B: B. mucilaginosus; B-AM: coupling of B. mucilaginosus and acid-modified maifanite). Different letters indicate significant differences among treatments, with p < 0.05 level.
Bioavailable Pb, defined as the readily mobile and plant-accessible fraction, is a critical indicator of ecological and health risks. As shown in Figure 7b, the immobilization efficiency for bioavailable Pb across treatments followed the order: B-AM (52.46%) > AM (42.08%) > M (34.54%) > OS (32.41%) > B (32.06%). Statistical analysis indicates that the differences among OS, M, and B were not significant (p > 0.05), while B-AM significantly outperformed all other treatments (p < 0.05). The superior performance of B-AM over abiotic amendments (OS, AM, M) stems from the synergistic between the mineral carrier and the bacteria. The lower efficiency in the B treatment is likely due to the lack of a protective carrier, which compromises microbial viability and leads to Pb remobilization upon cell death. In contrast, the immobilized carrier in B-AM not only provides physical protection for the bacteria, sustaining their metabolic activity and enabling continuous biomineralization, but also contributes through its own high adsorption capacity to direct Pb immobilization. This integrated approach combines physical, chemical, and biological mechanisms, significantly improving Pb stabilization in complex soil environments.
3.4.3. Pb Accumulation and Plant Growth
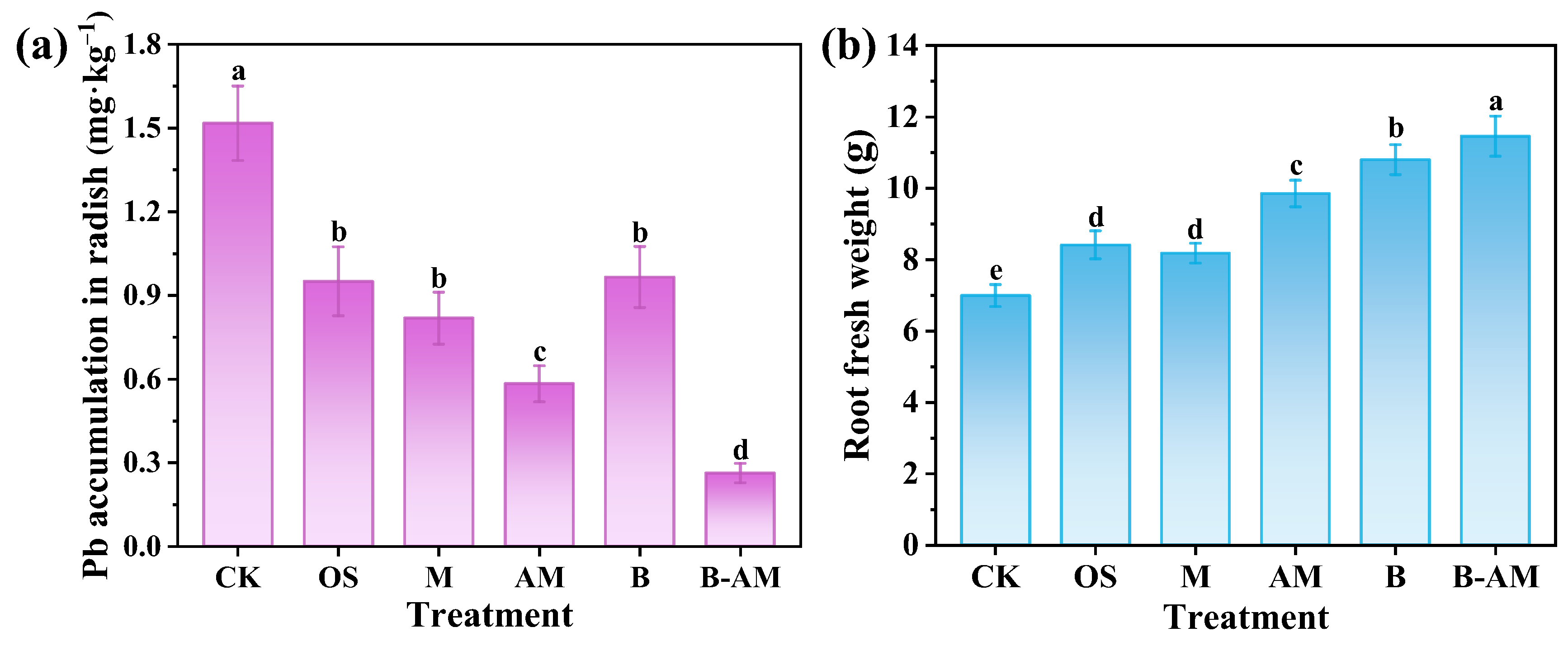
Assessing Pb accumulation in edible crops and corresponding plant growth responses is crucial for evaluating the practical effectiveness of immobilization remediation and its implications for both crop productivity and food safety. In the CK, the Pb concentration in the radish roots reached 1.52 mg·kg−1 (Figure 8a). All remediation treatments significantly reduced Pb accumulation, with concentrations ranging from 0.55 to 1.25 mg·kg−1. The B-AM treatment was the most effective, achieving a reduction of 83.24% (p < 0.05) compared to the CK. This result demonstrates the exceptional capability of the B-AM amendment to inhibit Pb translocation from soil to the crop, thereby significantly enhancing food safety in contaminated agricultural soils.
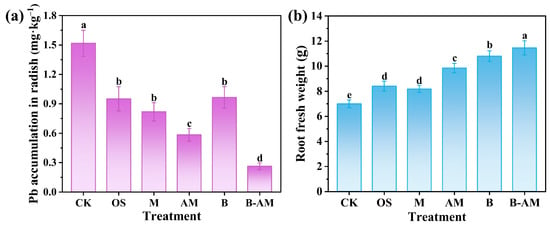
Figure 8.
(a) Pb accumulation and (b) fresh weight of radish roots under different treatments. (CK: untreated soil; OS: oyster shell; M: pristine maifanite; AM: acid-modified maifanite; B: B. mucilaginosus; B-AM: coupling of B. mucilaginosus and acid-modified maifanite). Different letters indicate significant differences among treatments, with p < 0.05 level.
The translocation of heavy metals from soil to plants involves complex physicochemical and biological processes. The immobilizing agents, including OS, M, and AM, contribute to Pb stabilization primarily through adsorption and by increasing soil pH. These processes reduce Pb mobility and bioavailability, thereby limiting plant uptake. In contrast, B. mucilaginosus enhances Pb immobilization via both biosorption and biomineralization, ultimately facilitating the formation of highly stable pyromorphite (Pb5(PO4)3Cl), which is highly persistent and stable [48]. Through synergistic action, AM and B. mucilaginosus collectively convert labile Pb fractions into inert forms, significantly decreasing soil Pb bioavailability. As a result, the B-AM treatment effectively minimizes Pb transfer into the edible tissues of cherry radish, demonstrating its superior remediation performance.
Plant growth responses further confirmed the benefits of the amendments. The fresh weight of radish roots differed significantly across treatments (Figure 8b). In the CK group, plant growth was severely inhibited by Pb toxicity, resulting in the lowest fresh weight of only 6.99 g. In contrast, all amendments significantly increased biomass compared to the CK. Notably, the B-AM treatment resulted in the highest fresh weight of 11.46 g, representing a 63.95% increase compared to the CK. This improvement can be attributed to the combined effects of reduced Pb phytotoxicity, optimized soil pH, and improved nutrient availability resulting from the synergistic interaction between AM and B. mucilaginosus.
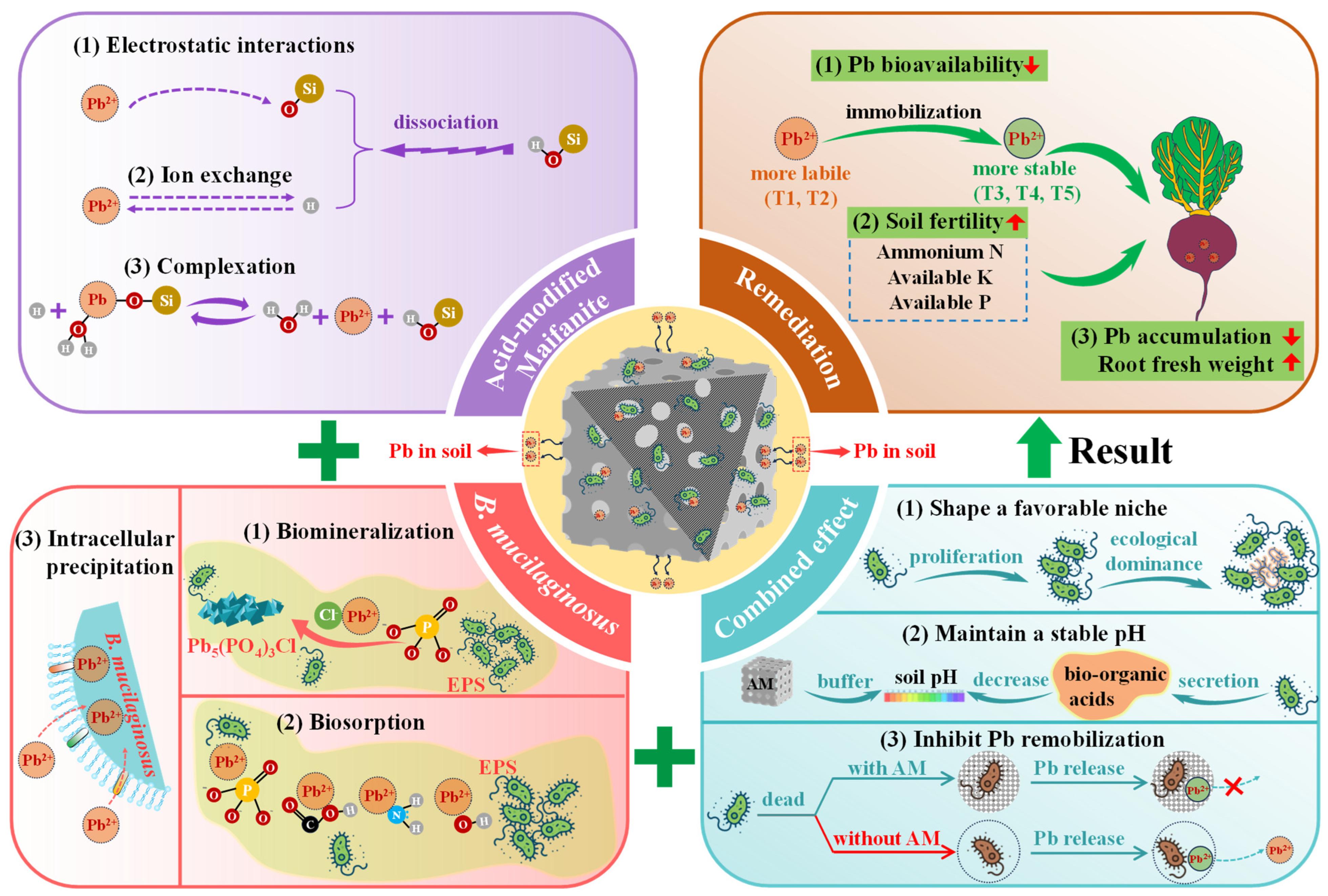
3.5. Proposed Remediation Mechanisms for Pb-Contaminated Soil
Based on a comprehensive analysis, Figure 9 summarizes the proposed Pb immobilization mechanism of the coupled immobilizing agent B-AM, which arises from the individual and synergistic contributions of its components. As confirmed by FT-IR analysis, the presence of abundant Si–OH groups on AM serves as the primary active site for Pb immobilization. Specifically, AM contributes to Pb immobilization through the following mechanisms: (1) Dissociation of Si–OH groups produce Si–O− and H+, allowing Pb2+ to replace exchangeable H+ at negatively charged sites [49]. (2) Under neutral to alkaline conditions, the dissociation of Si–OH groups generate a negatively charged surface (Si–O−), which facilitates the electrostatic attraction and accumulation of cationic Pb2+ ions [49]. (3) The Si–OH groups on the AM surface contribute to Pb immobilization through complexation. Specifically, Pb2+ can interact with Si–OH groups to form stable surface complexes such as Si–O–PbOH2+, as represented by Equation (2) [50].
Si–OH + Pb2+ + H2O ↔ Si–O–PbOH2+ + H+
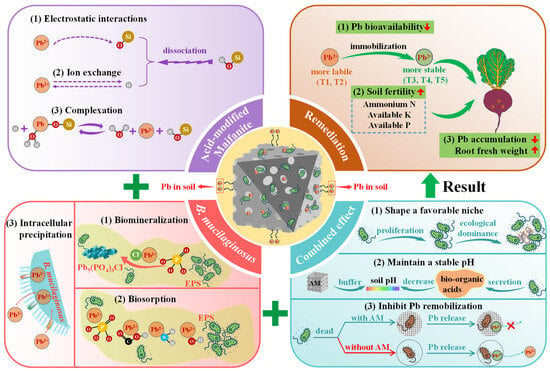
Figure 9.
Remediation mechanism of the B-AM complex for Pb-contaminated soil.
Meanwhile, B. mucilaginosus contributes to the bioimmobilization of Pb through the following mechanisms: (1) It secretes EPS rich in functional groups (e.g., carboxyl, phosphate, amino, and hydroxyl groups), which facilitate Pb biosorption through ion exchange, chelation, and surface coordination [35]. (2) Phosphate groups in EPS can react with Pb2+ and environmental Cl− to form stable minerals such as pyromorphite (Pb5(PO4)3Cl), as shown in Equation (3) [29]. (3) Pb2+ binds to cytoplasmic components, such as metallothionein, or reacts with intracellular phosphates and sulfides, leading to the formation of insoluble deposits that accumulate within the cell [51]. However, this intracellular precipitation mechanism generally plays a relatively minor role in the overall Pb adsorption process.
5Pb2+ + 3PO43− + Cl− → Pb5(PO4)3Cl
Critically, the synergy between AM and B. mucilaginosus substantially enhances the overall immobilization performance of Pb: (1) AM offers an ideal microenvironment for bacterial colonization and biofilm development, promoting microbial proliferation and ecological dominance. (2) It also serves as a pH buffer, counteracting acidification from microbial organic acids and maintaining a stable pH favorable for both microbial activity and chemical immobilization. (3) The coupled system immobilizes bacterial cells, thereby minimizing the risk of Pb remobilization caused by cell lysis and enhancing long-term remediation stability.
Together, these interactions between AM and B. mucilaginosus establish an integrated mechanism combining adsorption, biosorption, and biomineralization, resulting in highly effective and durable Pb immobilization. A key feature of this synergy is the rapid adsorption and concentration of Pb2+ by AM, which creates a high-concentration microenvironment around the bacteria that enhances microbial biomineralization; the subsequent fixation of Pb disrupts the adsorption equilibrium, enabling continuous adsorption and establishing an efficient adsorption–bioimmobilization cycle. The conversion of labile Pb into geochemically inert forms significantly reduces its bioavailability in soil, thereby decreasing Pb accumulation in crops. Furthermore, the favorable microenvironment provided by AM promotes the metabolic activity and growth of B. mucilaginosus, enhancing its ability to solubilize and mobilize essential nutrients such as nitrogen, phosphorus, and potassium [52]. This synergy not only sustains microbial vitality but also improves soil fertility and plant nutrient availability, thereby increasing the fresh root weight of radish plants, highlighting the dual role of the B-AM system in heavy metal immobilization and soil health improvement.
3.6. Feasibility and Considerations for Field Application
B-AM offers distinct advantages that may justify its application in specific scenarios. Unlike simple amendments that primarily function as adsorbents or pH adjusters, B-AM simultaneously improves soil fertility (increasing available P and K) and promotes the transformation of Pb into stable residual fractions. More importantly, this enhanced immobilization effectively reduces Pb bioavailability in soil, as evidenced by the reduction in Pb accumulation in radish roots, thereby mitigating potential health risks associated with food chain transfer. This multifunctionality could reduce the need for multiple amendments or repeated applications.
While the B-AM composite demonstrates promising Pb immobilization performance in this laboratory-scale study, several practical considerations must be acknowledged for its potential field application. The production process involving acid modification, microbial cultivation, and spray-drying inevitably incurs higher material costs compared to conventional amendments such as OS or raw maifanite. It is important to note that this study represents an early-stage exploration focused on establishing proof-of-concept and mechanistic understanding. For practical deployment, future research should address cost optimization strategies.
4. Conclusions
This study demonstrates that AM exhibits superior adsorption of Pb from aqueous solutions compared to the pristine M, owing to its enhanced surface roughness and specific surface area. Simultaneously, B. mucilaginosus proves highly effective in Pb removal, with spectroscopic and microscopic analyses confirming that its immobilization mechanism involves biosorption and the formation of stable mineral precipitates, such as pyromorphite (Pb5(PO4)3Cl). Subsequently, a novel coupled agent, B-AM, was developed and demonstrated superior performance in remediating Pb-contaminated soil compared to individual treatments such as OS, M, AM, and B. The B-AM agent not only adjusted soil pH to a range optimal for microbial activity and plant growth but also improved soil fertility by increasing available potassium, and available phosphorus. Furthermore, it significantly reduced Pb bioavailability and accumulation in indicator crops, while simultaneously increasing the fresh weight of radish roots. There was enhanced performance from the individual contributions of AM and B. mucilaginosus, as well as their synergistic interaction: AM serves as both a carrier and a pH buffer, enhancing microbial proliferation and reducing the risk of Pb remobilization from lysed cells. Furthermore, this sustained microbial activity facilitates the formation of stable Pb minerals. Therefore, the B-AM system proves to be a promising strategy for simultaneously immobilizing Pb, improving soil properties, and reducing ecological risks.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/agronomy16060641/s1, Figure S1: EDS spectra of B. mucilaginosus after Pb2+ removal; Table S1: Basic physicochemical properties of soil sample; Table S2: Surface textural properties of maifanite (M) and acid-modified maifanite (AM).
Author Contributions
X.C.: Conceptualization, Funding acquisition, Investigation, Project administration, Supervision, Validation, Writing—review and editing. M.Z.: Data curation, Formal analysis, Investigation, Methodology, Validation, Writing—original draft. Z.L.: Conceptualization, Resources, Supervision. H.F.: Formal analysis, Investigation. Y.C.: Formal analysis, Funding acquisition, Investigation. Z.C.: Data curation, Formal analysis, Investigation, Methodology, Writing—original draft. Q.G.: Formal analysis, Investigation. J.Z.: Project administration, Resources. X.Z.: Project administration, Resources. K.X.: Resources, Supervision. C.Z.: Conceptualization, Funding acquisition, Supervision, Writing—review and editing. J.N.: Conceptualization, Supervision, Writing—review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Key Research and Development Program of China (No. 2024YFD1701800); the Scientific and Technological Innovation Project of China Metallurgical Geology Bureau (No. CMGBKY202301); the National College Student Innovation Training Program (No. 202510386029); the Fujian College Association Instrumental Analysis Center of Fuzhou University Testing Fund of Precious Apparatus (Nos. 2026T040 and 2026T034).
Data Availability Statement
The original contributions presented in this study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding authors.
Conflicts of Interest
Authors Jianyu Zhang and Xuchuan Zhang are employed by the Jiangsu Longchang Chemical Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Oh, S.J.; Irshad, M.K.; Kang, M.W.; Roh, H.-S.; Jeon, Y.; Lee, S.S. In-Situ Physical and Chemical Remediation of Cd and Pb Contaminated Mine Soils Cultivated with Chinese Cabbage: A Three-Year Field Study. J. Hazard. Mater. 2023, 459, 132091. [Google Scholar] [CrossRef]
- Hou, D.; Jia, X.; Wang, L.; McGrath, S.P.; Zhu, Y.-G.; Hu, Q.; Zhao, F.-J.; Bank, M.S.; O’Connor, D.; Nriagu, J. Global Soil Pollution by Toxic Metals Threatens Agriculture and Human Health. Science 2025, 388, 316–321. [Google Scholar] [CrossRef]
- WHO. Guideline for Clinical Management of Exposure to Lead: Executive Summary, 1st ed.; World Health Organization: Geneva, Switzerland, 2021; ISBN 978-92-4-003688-8. [Google Scholar]
- Qu, J.; Yu, R.; Wang, M.; Wang, N.; Bi, F.; Gao, H.; Qin, C.; Wang, Y.; Zhang, Y. Tailoring Sponge-like Nitrogen-Enriched Biochar: A Green and Sustainable Remediation Technology for Pb and Cu Contaminated Soil. Chem. Eng. J. 2025, 522, 167603. [Google Scholar] [CrossRef]
- Hamid, Y.; Tang, L.; Hussain, B.; Usman, M.; Gurajala, H.K.; Rashid, M.S.; He, Z.; Yang, X. Efficiency of Lime, Biochar, Fe Containing Biochar and Composite Amendments for Cd and Pb Immobilization in a Co-Contaminated Alluvial Soil. Environ. Pollut. 2020, 257, 113609. [Google Scholar] [CrossRef]
- Hossain, M.F.; Islam, M.S.; Kashem, M.A.; Osman, K.T.; Zhou, Y. Lead Immobilization in Soil Using New Hydroxyapatite-like Compounds Derived from Oyster Shell and Its Uptake by Plant. Chemosphere 2021, 279, 130570. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.; Zhang, Q.; Wu, Z.; Guo, T.; Huang, X.; Chen, L.; Zhan, Y.; Zhao, S. Rice Varieties, Soil Conditioners, and Foliar Blockers for Remedying Cd-Pb Contamination on Paddy Fields. Fujian J. Agric. Sci. 2025, 39, 1306–1314. [Google Scholar] [CrossRef]
- Li, D.; Qi, Z.; Guo, J.; Wang, T.; Li, X.; Hou, N. Study on the Screening of High-Efficiency Salt and Alkali-Tolerant Microbial Agents and Their Roles and Mechanisms in Enhancing Saline-Alkaline Soil Remediation. J. Clean. Prod. 2025, 519, 145992. [Google Scholar] [CrossRef]
- Zhao, C.; Yao, J.; Knudsen, T.Š.; Hu, W.; Cao, Y. Combined Modified Montmorillonite and Microbial Consortium Enhanced the Remediation Effect of As and Cd-Contaminated Soil in a Smelting Area. J. Clean. Prod. 2025, 501, 145329. [Google Scholar] [CrossRef]
- Yin, K.; Wang, Q.; Lv, M.; Chen, L. Microorganism Remediation Strategies towards Heavy Metals. Chem. Eng. J. 2019, 360, 1553–1563. [Google Scholar] [CrossRef]
- Shen, Y.; Yu, C.; Yu, X. Bioremediation of Cadmium Ions by Bacillus mucilaginosus and Its Removal Mechanism. Water Air Soil Pollut. 2022, 233, 132. [Google Scholar] [CrossRef]
- Bybin, V.A.; Belogolova, G.A.; Markova, Y.A.; Sokolova, M.G.; Sidorov, A.V.; Gordeeva, O.N.; Poletaeva, V.I. Influence of Heavy Metals and Arsenic on Survival and Biofilm Formation of Some Saprotrophic Soil Microorganisms. Water Air Soil Pollut. 2021, 232, 343. [Google Scholar] [CrossRef]
- Li, Z.; Li, J.; Li, T.; Zhang, Q.; Gao, C.; Lu, J.; Jin, D.; Xu, M. Effects of Functional Microbial Agents on the Microbial Community and Fertility of Reclaimed Soil in a Coal Mining Area. Environ. Technol. Innov. 2024, 36, 103891. [Google Scholar] [CrossRef]
- Sattar, A.; Naveed, M.; Ali, M.; Zahir, Z.A.; Nadeem, S.M.; Yaseen, M.; Meena, V.S.; Farooq, M.; Singh, R.; Rahman, M.; et al. Perspectives of Potassium Solubilizing Microbes in Sustainable Food Production System: A Review. Appl. Soil Ecol. 2019, 133, 146–159. [Google Scholar] [CrossRef]
- Zhu, G.; Wang, X.; Chen, R.; Wang, Y.; Zhao, X.; Wu, P.; Tu, Q. Remediation of Lead-Zinc-Cadmium Co-Contaminated Soil Using Distiller’s Grain Biochar Immobilized with Lactobacillus plantarum. Chem. Eng. J. 2025, 522, 167304. [Google Scholar] [CrossRef]
- Chen, Z.; Chen, L.; Cheng, Y.; Zhang, B.; Zhao, S.; Chen, J.; Chen, Y.; Chen, X.; Lin, R. Thermal Protection of Encapsulated Bacillus mucilaginosus. Fujian J. Agric. Sci. 2024, 38, 360–366. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, Y.; Han, F.; Yan, P.; Liu, B.; Zhou, Q.; Min, F.; He, F.; Wu, Z. Investigation on the Adsorption of Phosphorus in All Fractions from Sediment by Modified Maifanite. Sci. Rep. 2018, 8, 15619. [Google Scholar] [CrossRef]
- Wang, X.; Liu, Y.; Zhao, W.; Wang, P.; Zhao, S.; Zhao, X.; Wang, D. The Disturbance of the Antioxidant System Results in Internal Blue Discoloration of Postharvest Cherry Radish (Raphanus sativus L. var. radculus pers) Roots. Foods 2023, 12, 677. [Google Scholar] [CrossRef]
- Xiao, J.; Wen, Y.; Kang, W.; Yu, F.; Liu, C.; Peng, Z.; Xu, D. The Transcription Factor Basic Pentacysteine 5, RsBPC5, Enhances Lead Stress Tolerance in Raphanus sativus. Plants 2025, 14, 2362. [Google Scholar] [CrossRef]
- Yang, H.; Zhang, Y.; Xia, S. Study on the Co-Effect of Maifanite-Based Photocatalyst and Humic Acid in the Photodegradation of Organic Pollutant. Environ. Sci. Pollut. Res. 2021, 28, 15731–15742. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, S.; Wang, J.; Dong, J.; Cheng, B.; Xu, L.; Shan, A. A Novel Adsorbent Albite Modified with Cetylpyridinium Chloride for Efficient Removal of Zearalenone. Toxins 2019, 11, 674. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Feng, D.; Bai, C.; Sun, S.; Zhang, Y.; Zhao, Y.; Li, Y.; Zhang, F.; Chang, G.; Qin, Y. Thermal Synergistic Treatment of Municipal Solid Waste Incineration (MSWI) Fly Ash and Fluxing Agent in Specific Situation: Melting Characteristics, Leaching Characteristics of Heavy Metals. Fuel Process. Technol. 2022, 233, 107311. [Google Scholar] [CrossRef]
- Biswas, B.; Sarkar, B.; Rusmin, R.; Naidu, R. Mild Acid and Alkali Treated Clay Minerals Enhance Bioremediation of Polycyclic Aromatic Hydrocarbons in Long-Term Contaminated Soil: A 14C-Tracer Study. Environ. Pollut. 2017, 223, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Simonsen, M.E.; Sønderby, C.; Li, Z.; Søgaard, E.G. XPS and FT-IR Investigation of Silicate Polymers. J. Mater. Sci. 2009, 44, 2079–2088. [Google Scholar] [CrossRef]
- Baig, M.M.; Zulfiqar, S.; Yousuf, M.A.; Shakir, I.; Aboud, M.F.A.; Warsi, M.F. DyxMnFe2−xO4 Nanoparticles Decorated over Mesoporous Silica for Environmental Remediation Applications. J. Hazard. Mater. 2021, 402, 123526. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lei, H.; Jiang, P.; Liu, L.; Cui, K.; Cao, W. Synthesis and Optical Properties of Intense Blue Colors Oxides Based on Mn5+ in Tetrahedral Sites in Ba7Al2−xMnxO10+y. Ceram. Int. 2021, 47, 686–691. [Google Scholar] [CrossRef]
- Qin, J.; Liu, W.; Wu, H.; Wang, J.; Xue, Q.; Zhao, H.; Zuo, H. A Comprehensive Investigation on the Viscosity and Structure of CaO-SiO2-Al2O3-MgO-BaO Slag with Different Al2O3/SiO2 Ratios. J. Mol. Liq. 2022, 365, 120060. [Google Scholar] [CrossRef]
- Fang, R.; Lu, C.; Zhong, Y.; Xiao, Z.; Liang, C.; Huang, H.; Gan, Y.; Zhang, J.; Pan, G.; Xia, X.; et al. Puffed Rice Carbon with Coupled Sulfur and Metal Iron for High-Efficiency Mercury Removal in Aqueous Solution. Environ. Sci. Technol. 2020, 54, 2539–2547. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhao, X.; Jiang, Y.; Ding, C.; Liu, J.; Zhu, C. Synergistic Remediation of Lead Pollution by Biochar Combined with Phosphate Solubilizing Bacteria. Sci. Total Environ. 2023, 861, 160649. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, H.; Nie, Q.; Ding, Y.; Lei, Z.; Zhang, Z.; Shimizu, K.; Yuan, T. Pb(II) Bioremediation Using Fresh Algal-Bacterial Aerobic Granular Sludge and Its Underlying Mechanisms Highlighting the Role of Extracellular Polymeric Substances. J. Hazard. Mater. 2023, 444, 130452. [Google Scholar] [CrossRef]
- Plunkett, S.A.; Wijayawardena, M.A.A.; Naidu, R.; Siemering, G.S.; Tomaszewski, E.J.; Ginder-Vogel, M.; Soldat, D.J. Use of Routine Soil Tests to Estimate Pb Bioaccessibility. Environ. Sci. Technol. 2018, 52, 12556–12562. [Google Scholar] [CrossRef]
- Tran, L.T.; Dang, H.T.M.; Tran, H.V.; Hoang, G.T.L.; Huynh, C.D. MIL-88B(Fe)-NH2: An Amine-Functionalized Metal–Organic Framework for Application in a Sensitive Electrochemical Sensor for Cd2+, Pb2+, and Cu2+ Ion Detection. RSC Adv. 2023, 13, 21861–21872. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, A.; Hua, T.; Chen, X.; Zhu, M.; Guo, Z.; Song, Y.; Yang, G.; Li, S.; Feng, J.; et al. Revealing the Interaction Forms between Hg(II) and Group Types (–Cl, –CN, –NH2, –OH, –COOH) in Functionalized Poly(Pyrrole Methane)s for Efficient Mercury Removal. Environ. Pollut. 2024, 351, 124049. [Google Scholar] [CrossRef]
- Kuang, X.; Hu, Y.; Zhu, S.; Du, L.; Li, C.; Xin, J.; Peng, L. Contribution and Composition of Extracellular Polymeric Substances in Cadmium Removal by Two Types of Biological Crusts. J. Environ. Manag. 2025, 392, 126857. [Google Scholar] [CrossRef] [PubMed]
- Mahto, K.U.; Vandana; Priyadarshanee, M.; Samantaray, D.P.; Das, S. Bacterial Biofilm and Extracellular Polymeric Substances in the Treatment of Environmental Pollutants: Beyond the Protective Role in Survivability. J. Clean. Prod. 2022, 379, 134759. [Google Scholar] [CrossRef]
- Börjesson, G.; Kirchmann, H. Interactive Long-Term Effects of Liming and P Application on Clay Soil: Crop Yield Increases up to pH 7.5(Aq). Plant Soil 2022, 473, 407–421. [Google Scholar] [CrossRef]
- Holland, J.E.; White, P.J.; Glendining, M.J.; Goulding, K.W.T.; McGrath, S.P. Yield Responses of Arable Crops to Liming—An Evaluation of Relationships between Yields and Soil pH from a Long-Term Liming Experiment. Eur. J. Agron. 2019, 105, 176–188. [Google Scholar] [CrossRef] [PubMed]
- Park, S.A.; Bhatia, S.K.; Park, H.A.; Kim, S.Y.; Sudheer, P.D.V.N.; Yang, Y.-H.; Choi, K.-Y. Bacillus Subtilis as a Robust Host for Biochemical Production Utilizing Biomass. Crit. Rev. Biotechnol. 2021, 41, 827–848. [Google Scholar] [CrossRef]
- Chen, M.; Huang, Y.; Wang, C.; Gao, H. The Conversion of Organic Nitrogen by Functional Bacteria Determines the End-Result of Ammonia in Compost. Bioresour. Technol. 2020, 299, 122599. [Google Scholar] [CrossRef]
- Chen, J.; Hu, G.; Liu, J.; Poulain, A.J.; Pu, Q.; Huang, R.; Meng, B.; Feng, X. The Divergent Effects of Nitrate and Ammonium Application on Mercury Methylation, Demethylation, and Reduction in Flooded Paddy Slurries. J. Hazard. Mater. 2023, 460, 132457. [Google Scholar] [CrossRef] [PubMed]
- Basak, B.B.; Sarkar, B.; Maity, A.; Chari, M.S.; Banerjee, A.; Biswas, D.R. Low-Grade Silicate Minerals as Value-Added Natural Potash Fertilizer in Deeply Weathered Tropical Soil. Geoderma 2023, 433, 116433. [Google Scholar] [CrossRef]
- Vincze, É.-B.; Becze, A.; Laslo, É.; Mara, G. Beneficial Soil Microbiomes and Their Potential Role in Plant Growth and Soil Fertility. Agriculture 2024, 14, 152. [Google Scholar] [CrossRef]
- Wang, S.; Song, M.; Wang, C.; Dou, X.; Wang, X.; Li, X. Mechanisms Underlying Soil Microbial Regulation of Available Phosphorus in a Temperate Forest Exposed to Long-Term Nitrogen Addition. Sci. Total Environ. 2023, 904, 166403. [Google Scholar] [CrossRef]
- Li, M.; Teng, Z.; Zhu, J.; Song, M. Research Advances in Heavy Metal Contaminated Soil Remediation by Phosphate Solubilizing Microorganisms. Acta Ecol. Sin. 2018, 38, 3393–3402. [Google Scholar] [CrossRef][Green Version]
- Matilla, M.A.; Krell, T. Plant Growth Promotion and Biocontrol Mediated by Plant-Associated Bacteria. In Plant Microbiome: Stress Response; Egamberdieva, D., Ahmad, P., Eds.; Springer: Singapore, 2018; pp. 45–80. [Google Scholar]
- Alori, E.T.; Glick, B.R.; Babalola, O.O. Microbial Phosphorus Solubilization and Its Potential for Use in Sustainable Agriculture. Front. Microbiol. 2017, 8, 971. [Google Scholar] [CrossRef]
- Chen, X.; Huang, Z.; Chen, Y.; Yao, C.; Zhang, J.; Diao, G.; Yuan, C.; Xu, K.; Liu, X. In Vitro Test-Based Study on Health Risks of Arsenic in Typical Soils of China and Their Influencing Factors. Acta Pedol. Sin. 2020, 59, 172–182. [Google Scholar] [CrossRef]
- Debela, F.; Arocena, J.M.; Thring, R.W.; Whitcombe, T. Organic Acid-Induced Release of Lead from Pyromorphite and Its Relevance to Reclamation of Pb-Contaminated Soils. Chemosphere 2010, 80, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Awan, S.A.; Rizwan, M.; Ali, S.; Hassan, M.J.; Brestic, M.; Zhang, X.; Huang, L. Effects of Silicon on Heavy Metal Uptake at the Soil-Plant Interphase: A Review. Ecotoxicol. Environ. Saf. 2021, 222, 112510. [Google Scholar] [CrossRef]
- Bradl, H.B. Adsorption of Heavy Metal Ions on Soils and Soils Constituents. J. Colloid Interface Sci. 2004, 277, 1–18. [Google Scholar] [CrossRef]
- Liu, X.; Ju, Y.; Mandzhieva, S.; Pinskii, D.; Minkina, T.; Rajput, V.D.; Roane, T.; Huang, S.; Li, Y.; Ma, L.Q.; et al. Sporadic Pb Accumulation by Plants: Influence of Soil Biogeochemistry, Microbial Community and Physiological Mechanisms. J. Hazard. Mater. 2023, 444, 130391. [Google Scholar] [CrossRef]
- Saravanan, A.; Swaminaathan, P.; Kumar, P.S.; Yaashikaa, P.R.; Kamalesh, R.; Rangasamy, G. A Comprehensive Review on Immobilized Microbes—Biochar and Their Environmental Remediation: Mechanism, Challenges and Future Perspectives. Environ. Res. 2023, 236, 116723. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.