UV-Initiated Crosslinking Reaction Mechanism and Electrical Breakdown Performance of Crosslinked Polyethylene
Abstract
1. Introduction
2. Theoretical Methodology and Experiments
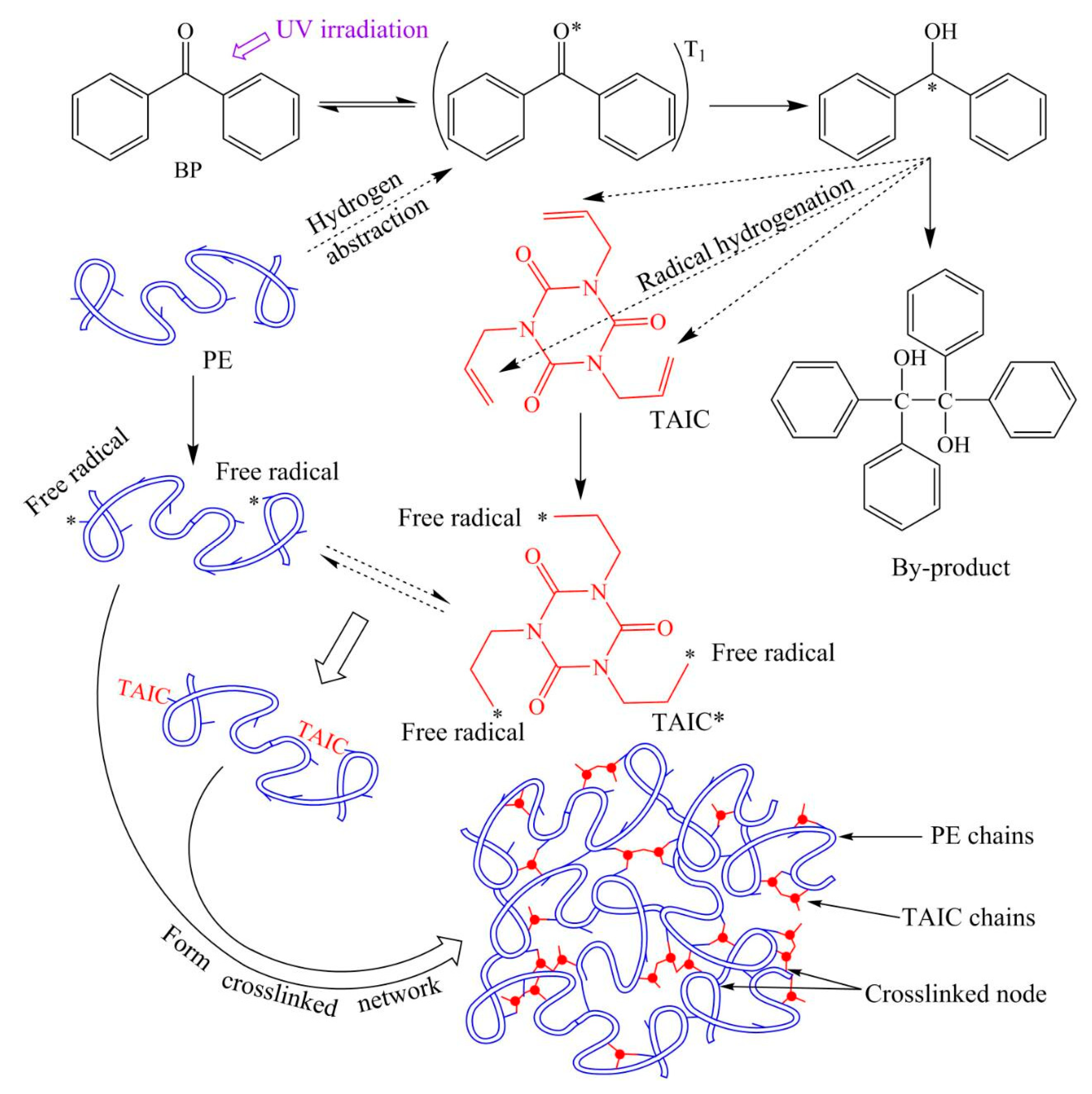
2.1. Polyethylene Photon-Crosslinking Reaction Mechanism
2.2. Molecular Model and Calculation Schemes
2.3. Material Preparation and Test Methods
3. Results and Discussion
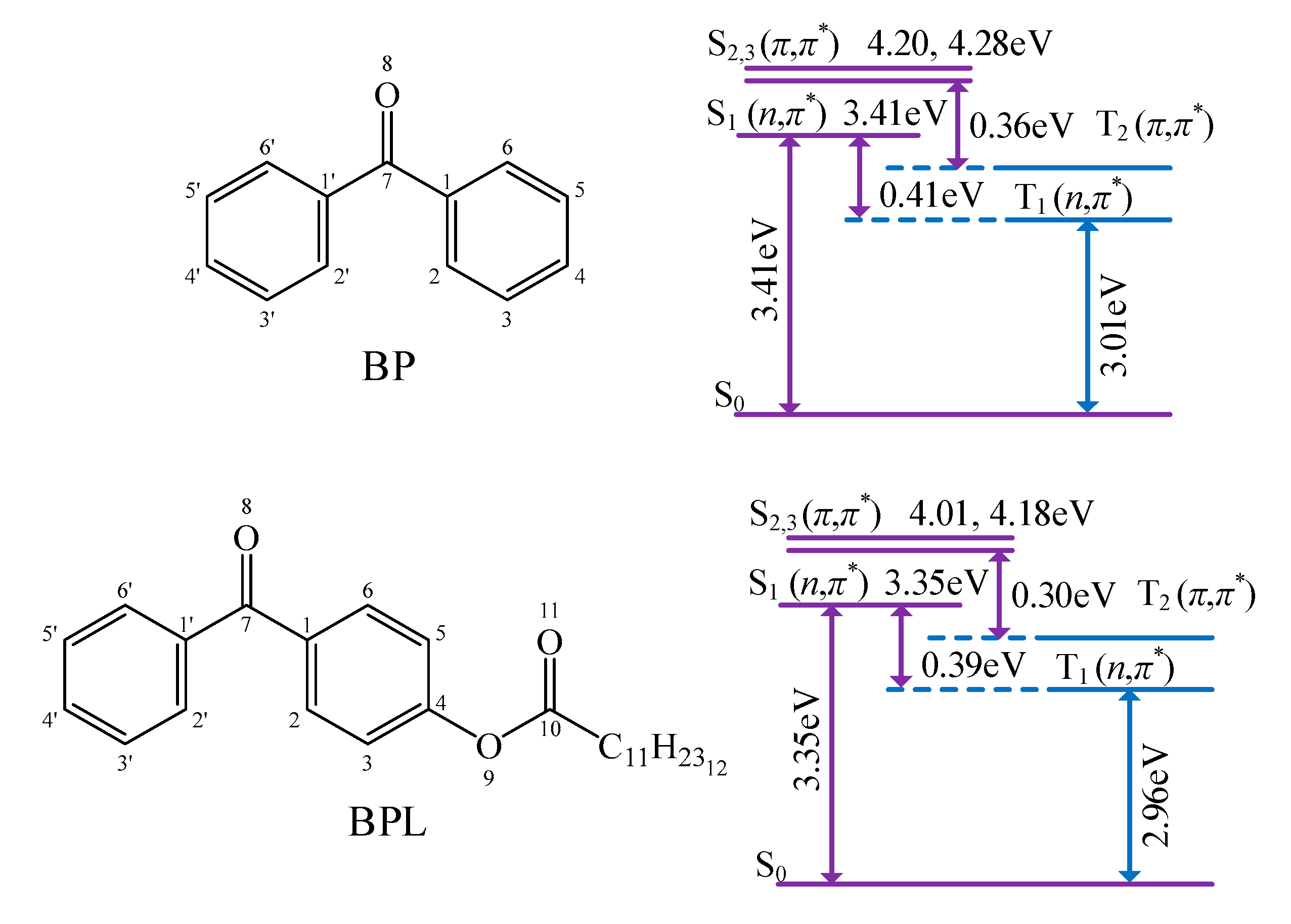
3.1. Photon-Initiated Free Radicals
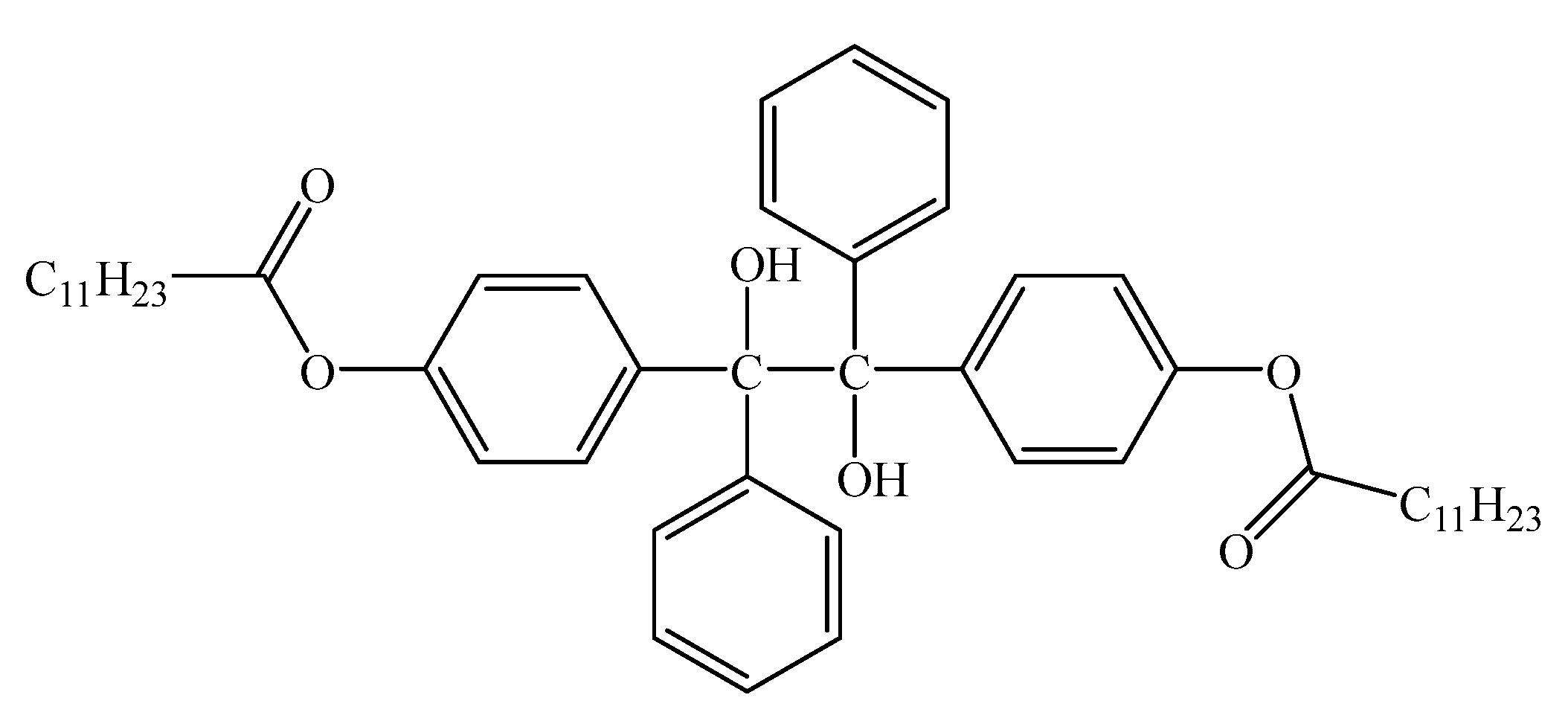
3.2. Crosslinking Reactions of Radical Polyethylene Molecules
3.3. Energetic Electronic Properties of Molecules in Crosslinking Reactions
3.4. Infrared Spectrum Characterization
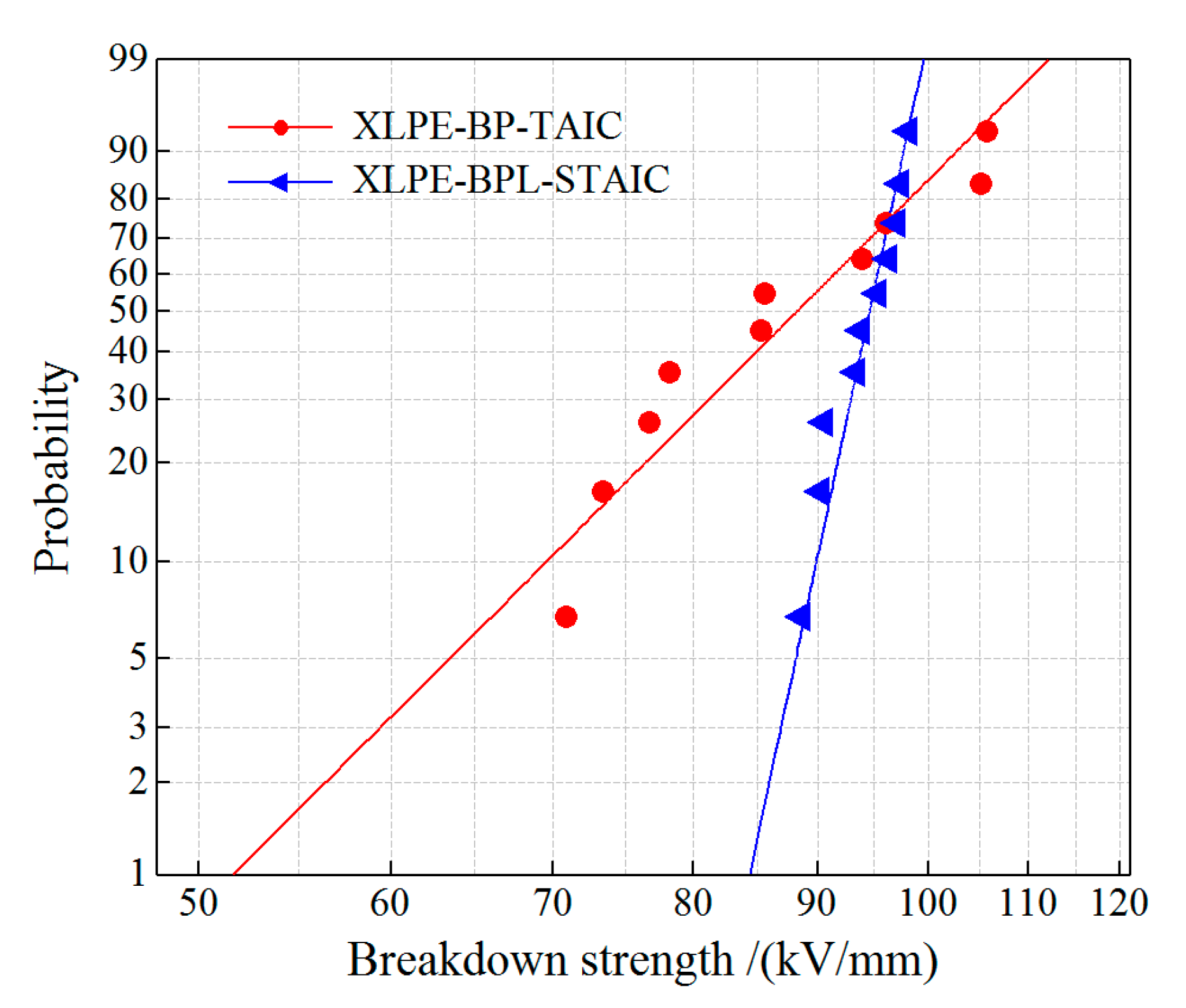
3.5. Dielectric Breakdown Strength
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Orton, H. History of underground power cables. IEEE Electr. Insul. Mag. 2013, 29, 52–57. [Google Scholar] [CrossRef]
- Takaoka, M.; Ono, I.; Kaji, I. Development of 275 kV XLPE cable system and prospect of 500 kV XLPE cable. IEEE Power Eng. Rev. 1983, 9, 3254–3263. [Google Scholar]
- Qu, B.J. Recent developments in photo-initiated crosslinking of polyethylene and its industrial applications. Chin. J. Polym. Sci. 2001, 19, 189–207. [Google Scholar]
- Deng, J.P.; Wang, L.F.; Liu, L.Y.; Yang, W.T. Developments and new applications of UV-induced surface graft polymerizations. Prog. Polym. Sci. 2009, 34, 156–193. [Google Scholar] [CrossRef]
- Qu, B.J.; Shi, W.F.; Rånby, B. Photocrosslinking of LDPE and its application for wires and cables. J. Photopolym. Sci. Technol. 1989, 2, 269–276. [Google Scholar] [CrossRef]
- Oster, G.; Oster, G.K.; Moroson, H. Ultraviolet induced crosslinking and grafting of solid high polymers. J. Polym. Sci. 1959, 34, 671–684. [Google Scholar] [CrossRef]
- Qu, B.J.; Rånby, B. Photocross-linking of low-density polyethylene. I. Kinetics and reaction parameters. J. Appl. Polym. Sci. 1993, 48, 701–709. [Google Scholar] [CrossRef]
- Qu, B.J.; Rånby, B. Photocross-linking of low-density polyethylene. II. Structure and morphology. J. Appl. Polym. Sci. 1993, 48, 711–719. [Google Scholar] [CrossRef]
- Fu, Y.W.; Wang, X.; Wu, Q.H.; Zhao, H. Study of crosslinked and electrical characteristics for cableinsulating material of new UV XLPE. Trans. Chin. Electrotechnol. Soc. 2018, 33, 178–186. [Google Scholar]
- Wu, Q.H.; Qu, B.J. Photoinitiating characteristics of benzophenone derivatives as new initiators in the photocrosslinking of polyethylene. Polym. Eng. Sci. 2001, 41, 1220–1226. [Google Scholar] [CrossRef]
- Yang, Q.; Rånby, B. Photoinitiated crosslinking of low-density polyethylene. IV: Continuous extrusion application. Polym. Eng. Sci. 1992, 32, 831–835. [Google Scholar]
- Ye, L.; Wu, Q.H.; Qu, B.J. Photocrosslinking and related properties of intumescent flame-retardant LLDPE/EVA/IFR blends. Polym. Adv. Technol. 2012, 23, 858–865. [Google Scholar] [CrossRef]
- Chen, Y.L.; Rånby, B. Photocrosslinking of polyethylene. I. Photoinitiators, crosslinking agent, and reaction kinetics. J. Polym. Sci. A 1989, 27, 4051–4075. [Google Scholar] [CrossRef]
- Qu, B.J.; Xu, Y.H.; Shi, W.F.; Rånby, B. Photoinitiated crosslinking of low-density polyethylene. 6. Spin-trapping ESR studies on radical intermediates. Macromolecules 1992, 25, 5215–5219. [Google Scholar] [CrossRef]
- Qu, B.J.; Xu, Y.H.; Shi, W.F. Mechanism of photoinitiated crosslinking of low density polyethylene I. ND Spin trapping ESR Study. Chin. J. Magn. Reson. 1990, 7, 359–365. [Google Scholar]
- Slagle, I.R.; Bernhardt, J.R.; Gutman, D.; Hanning-Lee, M.A.; Pilling, M.J. Kinetics of the reaction between oxygen atoms and propargyl radicals. J. Phys. Chem. 1990, 94, 3652–3656. [Google Scholar] [CrossRef]
- Henning, S.K.; Boye, W.M. Fundamentals of curing elastomers with peroxides and coagents II: Understanding the relationship between coagent and elastomer. Rubber World 2009, 240, 31–39. [Google Scholar]
- Zlata, H.M.; Jasenka, J.; Leonard, M. The mechanism of triallylcyanurate as a coagent in EPDM peroxide vulcanization. Polym. Eng. Sci. 1998, 38, 689–692. [Google Scholar]
- Zamotaev, P.; Shibirin, E.; Nogellova, Z. Photocrosslinking of polypropylene: The effect of different photo-initiators and coagents. Polym. Degrad. Stab. 1995, 47, 93–107. [Google Scholar] [CrossRef]
- Loan, L.D. Peroxide crosslinking of ethylene-propylene rubber. J. Polym. Sci. Part A Gen. Pap. 1964, 2, 3053–3066. [Google Scholar] [CrossRef]
- Zhang, H.; Shang, Y.; Li, M.X.; Zhao, H.; Wang, X.; Han, B.Z. Theoretical study on the radical reaction mechanism in the cross-linking process of polyethylene. RSC Adv. 2015, 5, 90343–90353. [Google Scholar] [CrossRef]
- Zhang, H.; Shang, Y.; Zhao, H.; Wang, X.; Han, B.Z.; Li, Z.S. Theoretical study on the reaction of maleic anhydride in the UV radiation cross-linking process of polyethylene. Polymer 2017, 133, 232–239. [Google Scholar] [CrossRef]
- Zhao, H.; Chen, J.Q.; Zhang, H.; Shang, Y.; Wang, X.; Han, B.Z.; Li, Z.S. Theoretical study on the reaction of triallyl isocyanurate in the UV radiation cross-linking of polyethylene. RSC Adv. 2017, 7, 37095–37104. [Google Scholar] [CrossRef]
- Cima, L.G.; Lopina, S.T. Network structures of radiation-crosslinked star polymer gels. Macromolecules 1995, 28, 6787–6794. [Google Scholar] [CrossRef]
- Amkreutz, M.; Hoffmann, M.; Wilke, Y.; Zilke, A.; Beck, E. Relating mechanical properties of UV-cured coatings to the molecular network: A new approach to predict crosslinking of coatings. Radtech Rep. 2012, 3, 18–26. [Google Scholar]
- Li, Y.S.; Hua, X.; Dai, Y.P.; Liu, Z.P. Study on molecular structure change and micro-mechanism of electrical aging of XLPE dielectric materials under external electric fields. J. Atom. Mol. Phys. 2019, 36, 413–420. [Google Scholar]
- Matsumoto, A.; Ogawa, S.; Matsuda, T.; Ueda, A.; Aota, H.; Fujii, T.; Toridome, H. Further discussion on correlation between brittleness and inhomogeneous network structure of cross-linked resins originating in specific polymerization behavior of triallyl isocyanurate. Macromolecules 2008, 41, 7938–7945. [Google Scholar] [CrossRef]
- Yang, W.; Rånby, B. Photoinitiation performance of some ketones in the LDPE–acrylic acid surface photografting system. Eur. Polym. J. 1999, 35, 1557–1568. [Google Scholar] [CrossRef]
- Köhler, C.; Seifert, G.; Frauenheim, T. Density functional based calculations for Fen (n ≤ 32). Chem. Phys. 2005, 309, 23–31. [Google Scholar] [CrossRef]
- Andzelm, J.; King-smith, R.D.; Fitzgerald, G. Geometry optimization of solids using delocalized internal coordinates. Chem. Phys. Lett. 2001, 335, 321–326. [Google Scholar] [CrossRef]
- Baker, J.; Kessi, A.; Delley, B. The generation and use of delocalized internal coordinates in geometry optimization. J. Chem. Phys. 1996, 105, 192–196. [Google Scholar] [CrossRef]
- Peverati, R.; Truhlar, D.G. M11-L: A local density functional that provides improved accuracy for electronic structure calculations in chemistry and physics. J. Phys. Chem. Lett. 2012, 3, 117–124. [Google Scholar] [CrossRef]
- Kresse, G.; Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 1996, 54, 11169–11186. [Google Scholar] [CrossRef] [PubMed]
- Henkelman, G.; Jonsson, H. Improved tangent estimate in the nudged elastic band method for finding energy paths and saddle points. J. Chem. Phys. 2000, 113, 9978–9985. [Google Scholar] [CrossRef]
- Sipaut, C.S.; Dayou, J. In situ FTIR analysis in determining possible chemical reactions for peroxide crosslinked LDPE in the presence of triallylcyanurate. Funct. Compos. Struct. 2019, 1, 025003. [Google Scholar] [CrossRef]
- Barboiu, V.; Avadanei, M.I. Chemical reactions of benzophenone photoirradiated in 1,2-polybutadiene. J. Photochem. Photobiol. A Chem. 2011, 222, 170–179. [Google Scholar] [CrossRef]
- Aloïse, S.; Ruckebusch, C.; Blanchet, L.; Réhault, J.; Buntinx, G.; Huvenne, J.P. The Benzophenone S1(n,π*)→T1(n,π*) states intersystem crossing reinvestigated by ultrafast absorption spectroscopy and multivariatecurve resolution. J. Phys. Chem. A 2008, 112, 224–231. [Google Scholar] [CrossRef]
- Spighi, G.; Gaveau, M.A.; Mestdagh, J.M.; Poisson, L.; Soep, B. Gas phase dynamics of triplet formation in benzophenone. Phys. Chem. Chem. Phys. 2014, 16, 9610–9618. [Google Scholar] [CrossRef]
- Zhang, H.; Shang, Y.; Wang, X.; Zhao, H.; Han, B.Z.; Li, Z.S. Mechanisms on electrical breakdown strength increment of polyethylene by aromatic carbonyl compounds addition: A theoretical study. J. Mol. Model. 2013, 19, 5429–5438. [Google Scholar] [CrossRef]

| Reactant Molecules | Characteristic Groups | Absorption Peaks/cm−1 | |
|---|---|---|---|
| BPL | ketone carbonyl –C=O | 1658 | |
| ester carbonyl O–C=O | 1766 | ||
| ester C–O–C | 1203 | ||
| STAIC | allyl double bond =CH2/C=C | 929/1598 | |
| phenylcarbamoyl N–C=O | 1700 | ||
| ester carbonyl O–C=O | 1764 | ||
| ester –C–O–C– | 1206 | ||
| LDPE | alkyl –CH2- | stretching | 2898 |
| bending | 1460 | ||
| alkyl –C4H9 | 720 | ||
| Photon Initiator | States | Mulliken Charges/e | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C1/C1′ | C2/C2′ | C3/C3′ | C4/C4′ | C5/C5′ | C6/C6′ | C7 | O8 | O9 | C10 | O11 | ||
| BP | S0 | 0.04 | −0.14 | −0.09 | −0.08 | −0.08 | −0.09 | 0.31 | −0.39 | |||
| S1 | 0.05 | −0.15 | −0.09 | −0.08 | −0.08 | −0.09 | 0.33 | −0.41 | ||||
| T1 | 0.06 | −0.15 | −0.08 | −0.09 | −0.07 | −0.09 | 0.39 | −0.50 | ||||
| BPL | S0 | 0.03/0.04 | −0.14/−0.14 | −0.13/−0.09 | 0.34/−0.08 | −0.14/−0.08 | −0.09/−0.09 | 0.33 | −0.40 | −0.41 | 0.51 | −0.37 |
| S1 | 0.04/0.06 | −0.12/−0.12 | −0.13/−0.08 | 0.32/−0.11 | −0.12/−0.08 | −0.10/−0.12 | 0.34 | −0.41 | −0.39 | 0.51 | −0.38 | |
| T1 | 0.05/0.06 | −0.13/−0.13 | −0.13/−0.08 | 0.31/−0.10 | −0.12/−0.09 | −0.11/−0.13 | 0.42 | −0.51 | −0.40 | 0.50 | −0.38 | |
| Schematic Molecular Reactions | LR | Rb/f | LP | fi | Eb | ΔG0298 |
|---|---|---|---|---|---|---|
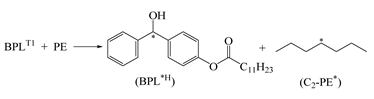 | 1.098 | 0.881 | 0.958 | 1247 | 0.14 | −11.833 |
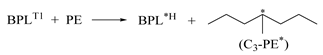 | 1.104 | 0.870 | 0.958 | 1235 | 0.25 | −10.475 |
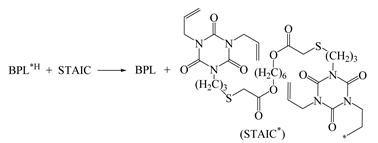 | 0.958 | 0.847 | 1.102 | 1306 | 0.37 | −7.649 |
| BPL*H + TAIC → BPL + TAIC* | 0.958 | 0.915 | 1.092 | 1327 | 0.46 | −5.182 |
| Schematic Molecular Reactions | LR/Å | LP/Å | ΔG0298/(kcal/mol) |
|---|---|---|---|
| 2 C2-PE* → PE-PE | 1.524 | −85.655 | |
| C2-PE* + TAIC* → PE-TAIC | 1.481 | 1.511 | −101.096 |
| 2 TAIC* → -[TAIC]2- | 1.481 | 1.497 | −122.999 |
| C2-PE* + STAIC* → PE-STAIC | 1.476 | 1.517 | −107.480 |
| 2 STAIC* → -[STAIC]2- | 1.476 | 1.504 | −131.105 |
| Molecules | M11-L | B3LYP | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Eg | IP(a) | IP(v) | EA(a) | EA(v) | Eg | IP(a) | IP(v) | EA(a) | EA(v) | |
| BP | 3.795 | 8.695 | 8.766 | 0.790 | 0.546 | 3.808 | 8.912 | 8.661 | 0.819 | 0.560 |
| BPL | 3.783 | 8.561 | 8.594 | 0.816 | 0.675 | 3.964 | 8.668 | 8.886 | 0.795 | 0.642 |
| TAIC | 5.890 | 8.776 | 8.828 | −0.461 | −0.519 | 6.206 | 8.970 | 8.632 | −0.438 | −0.545 |
| STAIC | 4.366 | 7.767 | 7.823 | 0.458 | 0.372 | 4.635 | 8.216 | 8.165 | 0.426 | 0.358 |
| PBB | 4.390 | 7.976 | 8.065 | −0.073 | −0.228 | 4.447 | 8.349 | 8.398 | −0.075 | −0.216 |
| PBBL | 4.291 | 7.855 | 7.977 | 1.104 | 1.022 | 4.583 | 8.359 | 7.869 | 1.074 | 0.969 |
| PE | 8.232 | 7.644 | 7.701 | −0.874 | −0.913 | 8.229 | 7.476 | 7.992 | −0.919 | 0.873 |
| AP | 3.223 | 8.757 | 8.873 | 0.214 | 0.025 | 3.415 | 8.685 | 9.277 | 0.229 | 0.024 |
| Experimental Samples | Shape Parameter | Characteristic DBS/(kV/mm) |
|---|---|---|
| XLPE-BP-TAIC | 7.91 | 87.6 |
| XLPE-BPL-STAIC | 37.14 | 95.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, Y.-W.; Sun, W.-F.; Wang, X. UV-Initiated Crosslinking Reaction Mechanism and Electrical Breakdown Performance of Crosslinked Polyethylene. Polymers 2020, 12, 420. https://doi.org/10.3390/polym12020420
Fu Y-W, Sun W-F, Wang X. UV-Initiated Crosslinking Reaction Mechanism and Electrical Breakdown Performance of Crosslinked Polyethylene. Polymers. 2020; 12(2):420. https://doi.org/10.3390/polym12020420
Chicago/Turabian StyleFu, Yu-Wei, Wei-Feng Sun, and Xuan Wang. 2020. "UV-Initiated Crosslinking Reaction Mechanism and Electrical Breakdown Performance of Crosslinked Polyethylene" Polymers 12, no. 2: 420. https://doi.org/10.3390/polym12020420
APA StyleFu, Y.-W., Sun, W.-F., & Wang, X. (2020). UV-Initiated Crosslinking Reaction Mechanism and Electrical Breakdown Performance of Crosslinked Polyethylene. Polymers, 12(2), 420. https://doi.org/10.3390/polym12020420







