Build-To-Specification Vanillin and Phloroglucinol Derived Biobased Epoxy-Amine Vitrimers
Abstract
1. Introduction
2. Materials and Methods
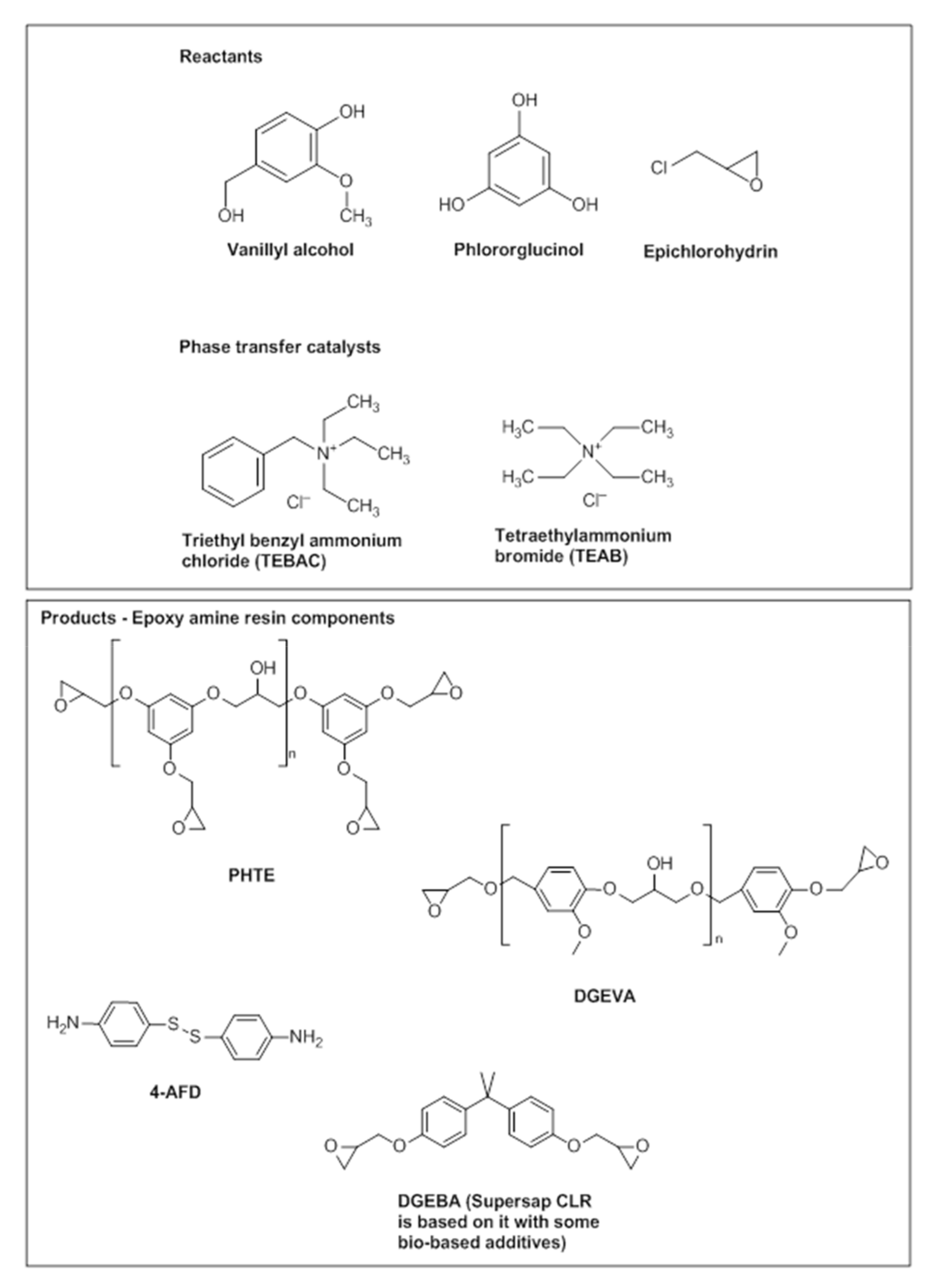
2.1. Materials
2.2. Methods
2.3. Synthesis of Biobased Epoxy Resins and Vitrimers
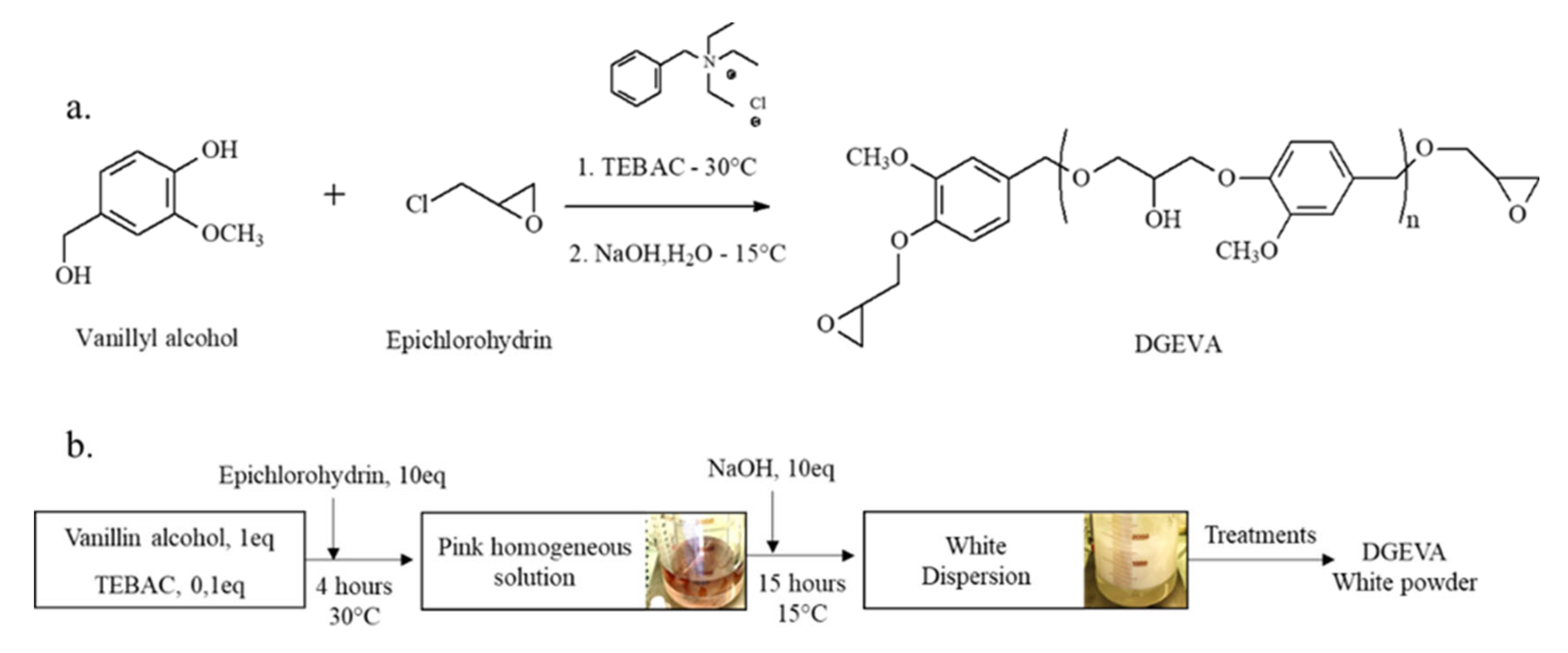
2.3.1. Synthesis of DGEVA
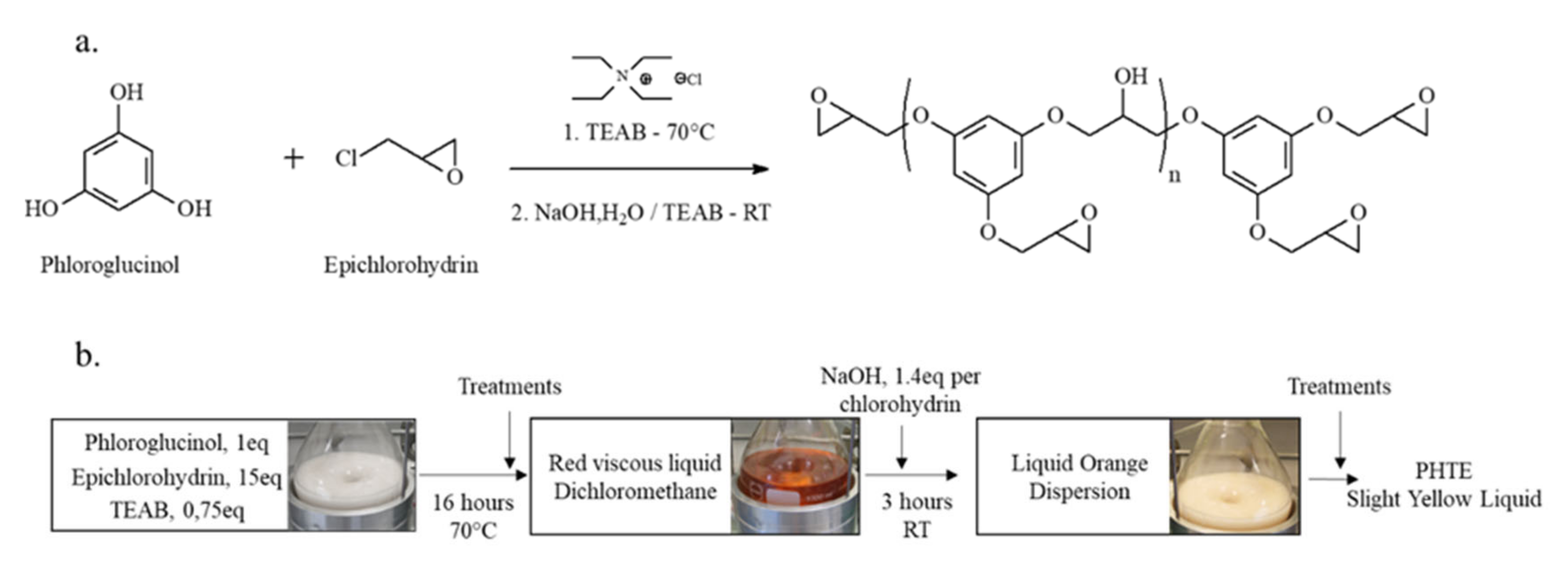
2.3.2. Synthesis of PHTE
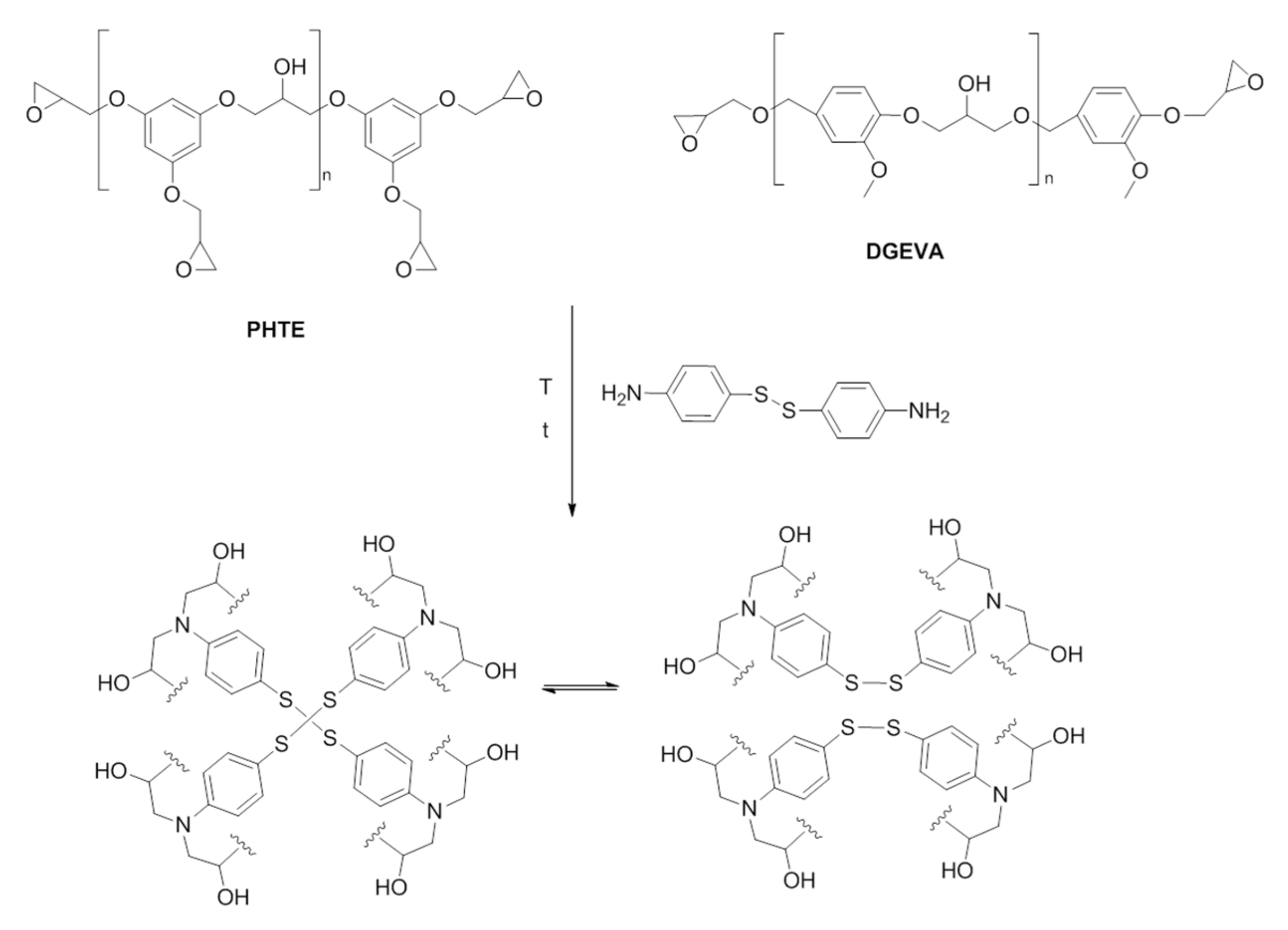
2.3.3. Synthesis of Epoxy-Vitrimers
3. Results and Discussion
3.1. Thermal and Mechanical Characterization of the Pristine Material
3.1.1. Thermal Properties
3.1.2. Mechanical Properties
3.1.3. Crosslink Density
3.1.4. Dynamic Properties
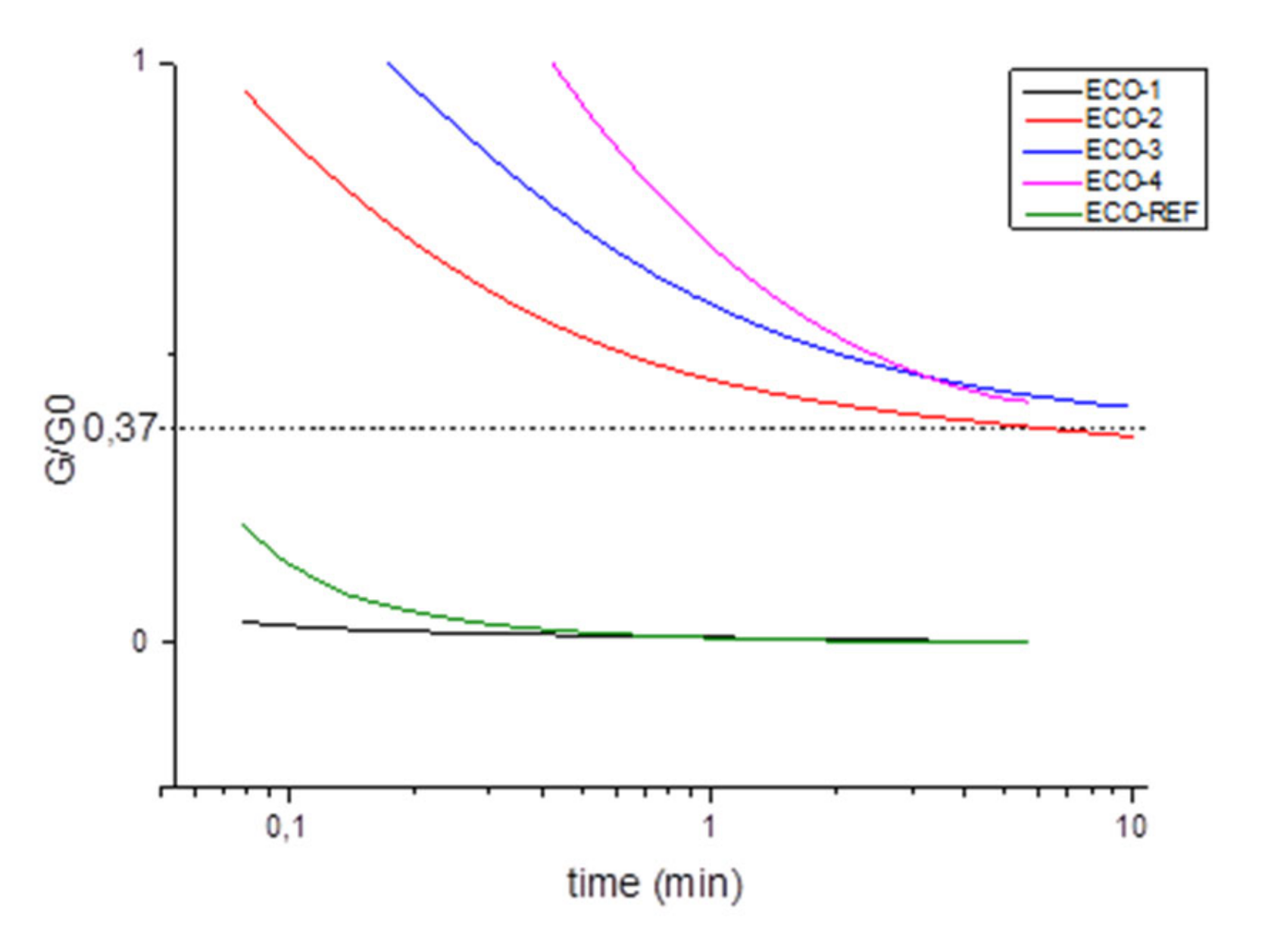
- Reprocessability
- Repairability
- Reciclability
3.2. Thermal and Mechanical Characterization of the Recycled Material
Thermomechanical Properties Recovery
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Vozzola, E.; Overcash, M.; Twomey, J.; Griffing, E.; Asmatulu, E. Thermoset composite recycling—Driving forces, development, and evolution of new opportunities. J. Compos. Mater. 2017, 52, 1033–1043. [Google Scholar]
- Denissen, W.; Winne, J.M.; Du Prez, F.E. Vitrimers: Permanent organic networks with glass-like fluidity. Chem. Sci. 2016, 7, 30–38. [Google Scholar] [PubMed]
- Addicoat, M.A.; Tsotsalas, M. Covalently linked organic networks. Front. Mater. 2015, 2, 1–5. [Google Scholar] [CrossRef]
- Alabiso, W. The Impact of Vitrimers on the Industry of the Future: Chemistry, Properties and Sustainable Forward-Looking Applications. Polymers 2020, 12, 1660. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, M. Implantation of Recyclability and Healability into Cross-Linked Commercial Polymers by Applying the Vitrimer Concept. Polymers 2020, 12, 1322. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Ahmed, N.; Rabnawaz, M. Covalent Adaptable Network and Self-Healing Materials: Current Trends and Future Prospects in Sustainability. Polymers 2020, 12, 2027. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Feng, Y.; He, H.; Yang, Z. Preparation and Characteristics of Biocomposites Based on Steam Exploded Sisal Fiber Modified with Amphiphatic Epoxidized Soybean Oil Resin. Materials 2018, 11, 1731. [Google Scholar] [CrossRef]
- Garcia-garcia, D.; Id, R.B. Manufacturing and Characterization of Composite Fibreboards with Posidonia oceanica Wastes with an Environmentally-Friendly Binder from Epoxy Resin. Materials 2018, 11, 35. [Google Scholar] [CrossRef] [PubMed]
- Auvergne, R.; Caillol, S.; David, G.; Boutevin, B.; Pascault, J.P. Biobased thermosetting epoxy: Present and future. Chem. Rev. 2014, 114, 1082–1115. [Google Scholar] [CrossRef]
- Fache, M.; Boutevin, B.; Caillol, S. Vanillin, a key-intermediate of biobased polymers. Eur. Polym. J. 2015, 68, 488–502. [Google Scholar] [CrossRef]
- Wang, S.; Ma, S.; Xu, C.; Liu, Y.; Dai, J.; Wang, Z.; Liu, X.; Chen, J.; Shen, X.; Wei, J. Vanillin-Derived High-Performance Flame Retardant Epoxy Resins: Facile Synthesis and Properties. Macromolecules 2017, 50, 1892–1901. [Google Scholar] [CrossRef]
- Shibata, M.; Ohkita, T. Fully biobased epoxy resin systems composed of a vanillin-derived epoxy resin and renewable phenolic hardeners. Eur. Polym. J. 2017, 92, 165–173. [Google Scholar] [CrossRef]
- Fache, M.; Darroman, E.; Besse, V.; Auvergne, R.; Caillol, S.; Boutevin, B. Vanillin, a promising biobased building-block for monomer synthesis. Green Chem. 2014, 16, 1987–1998. [Google Scholar] [CrossRef]
- Nikafshar, S.; Zabihi, O.; Hamidi, S.; Moradi, Y.; Barzegar, S.; Ahmadi, M.; Naebe, M. A renewable bio-based epoxy resin with improved mechanical performance that can compete with DGEBA. RSC Adv. 2017, 7, 8694–8701. [Google Scholar] [CrossRef]
- Frost, J.W. Biosynthesis of Philoroglucinol and Preparation of 1,3-dihydroxybenzene Therefrom. U.S. Patent 8,329,445B2, 27 April 2006. [Google Scholar]
- Noè, C.; Malburet, S.; Bouvet-Marchand, A.; Graillot, A.; Loubat, C.; Sangermano, M. Cationic photopolymerization of bio-renewable epoxidized monomers. Prog. Org. Coat. 2019, 133, 131–138. [Google Scholar] [CrossRef]
- Zou, W.; Dong, J.; Luo, Y.; Zhao, Q.; Xie, T. Dynamic Covalent Polymer Networks: From Old Chemistry to Modern Day Innovations. Adv. Mater. 2017, 29, 1606100. [Google Scholar] [CrossRef] [PubMed]
- Ciarella, S.; Wouter, G. Ellenbroek. Swap-Driven Self-Adhesion and Healing of Vitrimers. Coatings 2019, 9, 114. [Google Scholar] [CrossRef]
- De Luzuriaga, A.R.; Martin, R.; Markaide, N.; Rekondo, A.; Cabañero, G.; Rodríguez, J.; Odriozola, I. Epoxy resin with exchangeable disulfide crosslinks to obtain reprocessable, repairable and recyclable fiber-reinforced thermoset composites. Mater. Horiz. 2016, 3, 241–247. [Google Scholar] [CrossRef]
- De Luzuriaga, A.R.; Martin, R.; Markaide, N.; Rekondo, A.; Cabañero, G.; Rodríguez, J.; Odriozola, I. Correction: Epoxy resin with exchangeable disulfide crosslinks to obtain reprocessable, repairable and recyclable fiber-reinforced thermoset composites. Mater. Horiz. 2020, 7, 2460–2461. [Google Scholar] [CrossRef]
- Flory, P.J.; Rehner, J. Statistical mechanics of cross-linked polymer networks I. Rubberlike elasticity. J. Chem. Phys. 1943, 11, 512–520. [Google Scholar] [CrossRef]





| Sample Name | DGEVA/PHTE Ratio (wt) | DGEVA | PHTE | SUPERSAP CLR | 4-AFD | Curing Cycle |
|---|---|---|---|---|---|---|
| ECO-1 | 100/0 | 30 g, 0.22 epoxy eq. | - | - | 16.14 g, 0.26 amino eq. | 1 h 150 °C |
| ECO-2 | 60/40 | 18 g, 0.13 epoxy eq. | 12 g, 0.08 epoxy eq. | - | 15.52 g, 0.25 amino eq. | 1 h 150 °C |
| ECO-3 | 40/60 | 12 g, 0.09 epoxy eq. | 18 g, 0.12 epoxy eq. | - | 15.52 g, 0.25 amino eq. | 1 h 160 °C |
| ECO-4 | 0/100 | - | 30 g, 0.20 epoxy eq. | - | 14.90 g, 0.24 amino eq. | 1 h 150 °C + 30′ 180 °C |
| ECO-REF | - | - | - | 30 g, 0.16 epoxy eq. | 11.80 g, 0.19 amino eq | 1 h 150 °C |
| Ref. | TgDSC (°C) | TgDMA (°C) | Td5% (°C) | Stress a (MPa) | Strain a (%) | E’ (30 °C) (GPa) | E’ (at Tg + 30 °C) (MPa) | νXL b·103 (mol·cm−3) |
|---|---|---|---|---|---|---|---|---|
| ECO-1 | 105 | 103 | 259 | 86 ± 1 | 7 ± 1 | 6 | 17 | 1.7 |
| ECO-2 | 135 | 137 | 251 | 105 ± 2 | 7 ± 0.6 | 9 | 28 | 2.6 |
| ECO-3 | 157 | 160 | 257 | 94 ± 4 | 5 ± 0.4 | 7 | 41 | 3.6 |
| ECO-4 | 194 | 197 | 239 | 92 ± 6 | 5 ± 0.3 | 11 | 83 | 6.7 |
| ECO-REF | 107 | 108 | 252 | 66 ± 10 | 4 ± 1 | 7 | 16 | 1.6 |
| Ref. | Relaxation Time (s) | |
|---|---|---|
| Tg + 20 °C | Tg + 50 °C | |
| ECO-1 | It does not relax (at 120 °C) | 100 s (at 150 °C) |
| ECO-2 | It does not relax (at 150 °C) | 163 s (at 180 °C) |
| ECO-3 | 215 s (at 170 °C) | 35 s (at 200 °C) |
| ECO-4 | 359 s (at 210 °C–too high T) | Too high T (240 °C) |
| ECO-REF | It does not relax (at 120 °C) | 212 s (at 150 °C) |
| Ref. | Initial TgDSC (°C) | TgDSC after 20 min at 200 °C (°C) | TgDSC after 20 min at 230 °C (°C) |
|---|---|---|---|
| ECO-1 | 105 | 97 | 95 |
| ECO-2 | 134 | 131 | 129 |
| ECO-3 | 156 | 152 | 137 |
| ECO-4 | 194 | 177 | 162 |
| ECO-REF | 107 | 102 | 92 |
| Ref. | Initial TgDSC (°C) | Recycled Tg DSC (°C) | Initial TgDMA (°C) | Recycled TgDMA (°C) | Initial Td5% (°C) | Recycled Td5% (°C) |
|---|---|---|---|---|---|---|
| ECO-1 | 105 | 106 | 103 | 103 | 259 | 255 |
| ECO-2 | 135 | 149 | 137 | 146 | 251 | 255 |
| ECO-3 | 157 | 165 | 160 | 169 | 257 | 255 |
| ECO-REF | 107 | 104 | 108 | 107 | 252 | 249 |
| Ref. | Initial E’ (30 °C) (GPa) | Recycled E’ (30 °C) (GPa) | Initial E’ (Tg + 30 °C) (MPa) | Recycled E’ (Tg + 30 °C) (MPa) | Initial νXL a·103 (mol·cm−3) | Recycled νXL a·103 (mol·cm−3) |
|---|---|---|---|---|---|---|
| ECO-1 | 6 | 4 | 17 | 20 | 1.7 | 2.0 |
| ECO-2 | 9 | 10 | 28 | 26 | 2.6 | 2.3 |
| ECO-3 | 7 | 4 | 41 | 54 | 3.6 | 4.6 |
| ECO-REF | 7 | 3 | 16 | 16 | 1.6 | 1.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Genua, A.; Montes, S.; Azcune, I.; Rekondo, A.; Malburet, S.; Daydé-Cazals, B.; Graillot, A. Build-To-Specification Vanillin and Phloroglucinol Derived Biobased Epoxy-Amine Vitrimers. Polymers 2020, 12, 2645. https://doi.org/10.3390/polym12112645
Genua A, Montes S, Azcune I, Rekondo A, Malburet S, Daydé-Cazals B, Graillot A. Build-To-Specification Vanillin and Phloroglucinol Derived Biobased Epoxy-Amine Vitrimers. Polymers. 2020; 12(11):2645. https://doi.org/10.3390/polym12112645
Chicago/Turabian StyleGenua, Aratz, Sarah Montes, Itxaso Azcune, Alaitz Rekondo, Samuel Malburet, Bénédicte Daydé-Cazals, and Alain Graillot. 2020. "Build-To-Specification Vanillin and Phloroglucinol Derived Biobased Epoxy-Amine Vitrimers" Polymers 12, no. 11: 2645. https://doi.org/10.3390/polym12112645
APA StyleGenua, A., Montes, S., Azcune, I., Rekondo, A., Malburet, S., Daydé-Cazals, B., & Graillot, A. (2020). Build-To-Specification Vanillin and Phloroglucinol Derived Biobased Epoxy-Amine Vitrimers. Polymers, 12(11), 2645. https://doi.org/10.3390/polym12112645








