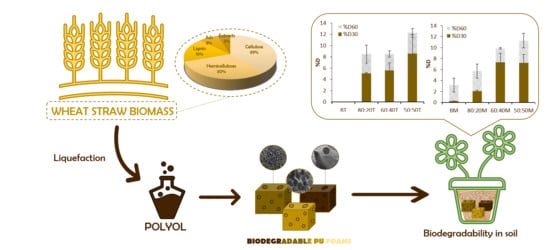
Bio-Degradable Polyurethane Foams Produced by Liquefied Polyol from Wheat Straw Biomass
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Liquefaction of wheat straw
2.3. Preparation of Polyurethane Foams
2.4. Characterization of Products
2.4.1. Liquefied Product
2.4.2. LWS-Formulated PU Foams
3. Results and Discussion
3.1. Chemical Composition of Wheat Straw
3.2. Wheat Straw Liquefaction and Product Properties
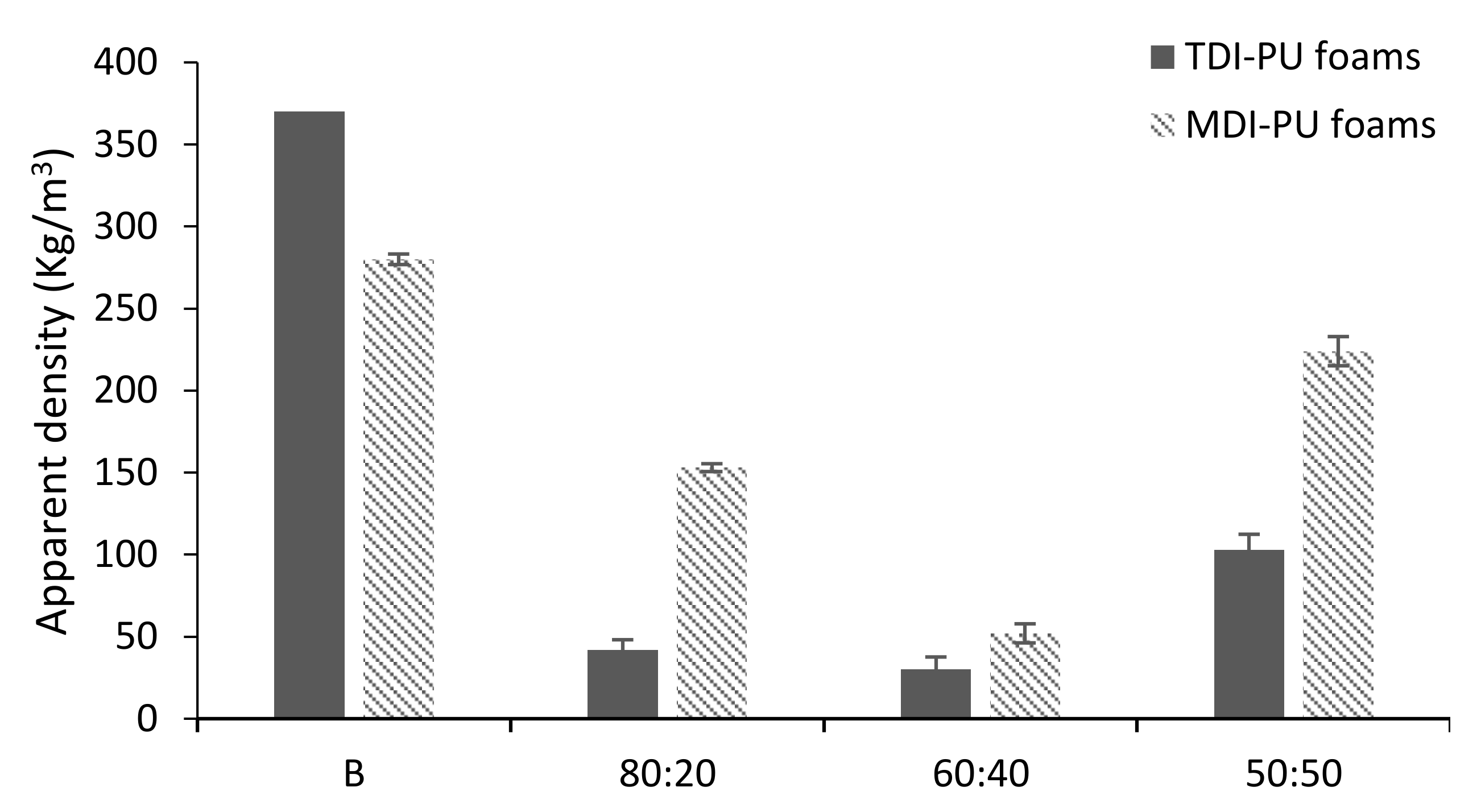
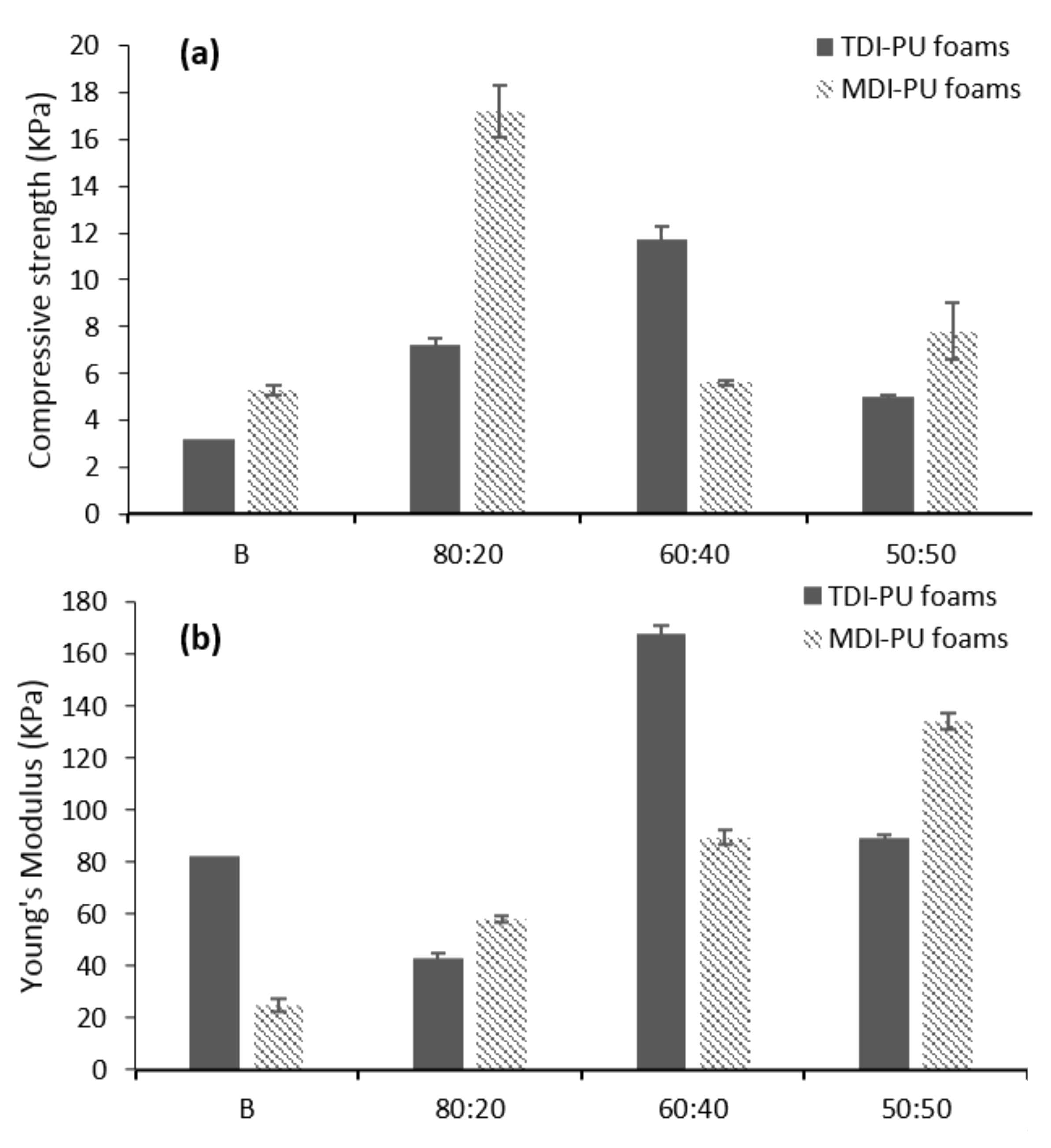
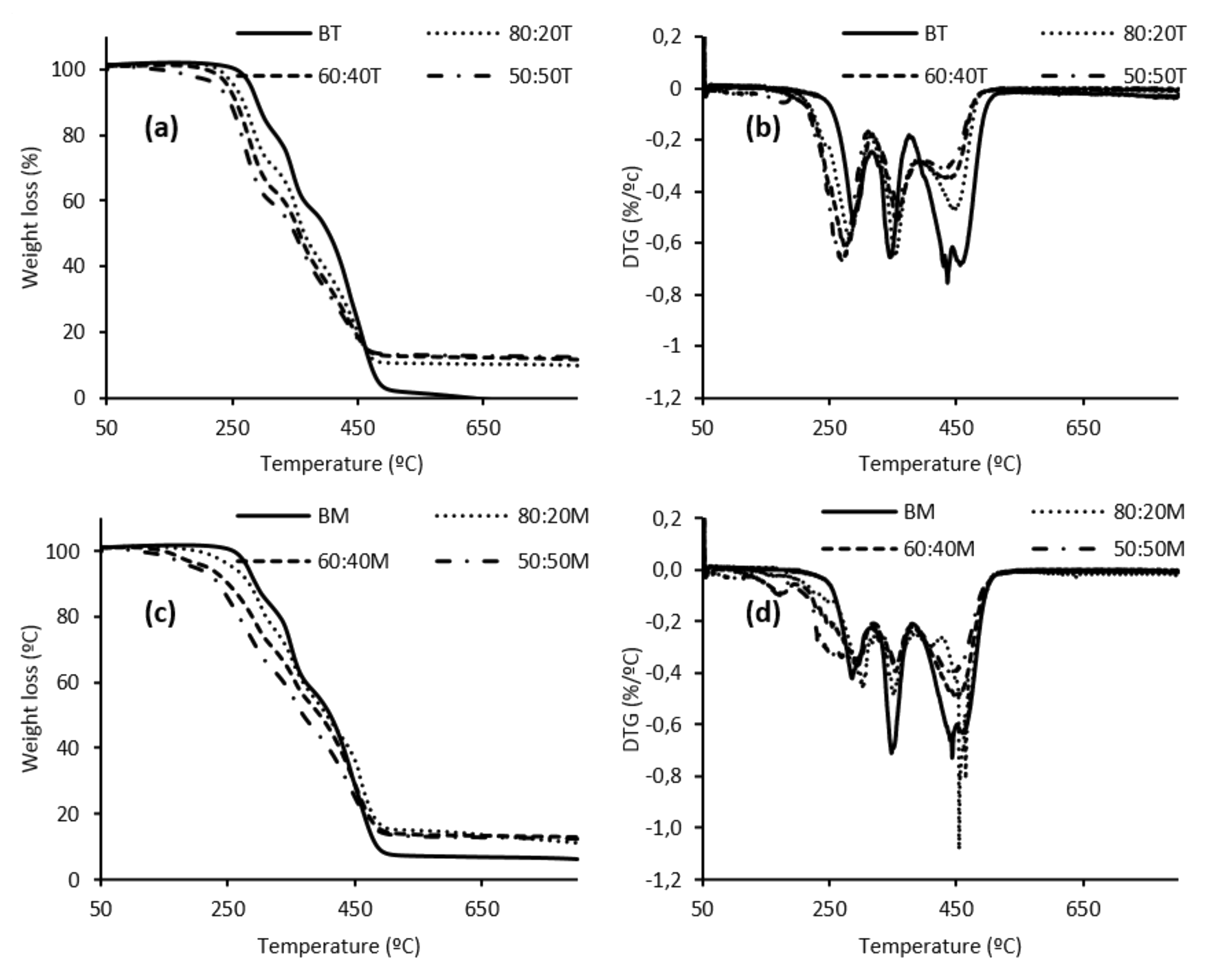
3.3. LWS-PU Foam Properties
3.4. LWS-PU Foam Application: Biodegradability in Soil Media
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Lozano, F.J.; Lozano, R. Assessing the potential sustainability benefits of agricultural residues: Biomass conversion to syngas for energy generation or to chemicals production. J. Clean. Prod. 2018, 172, 4162–4169. [Google Scholar] [CrossRef]
- Food and Agriculture Organization Corporate Statistical Database (FAOSTAT). New Food Balances. Available online: http://www.fao.org/faostat/en/#data/FBS (accessed on 2 December 2019).
- Saini, J.K.; Saini, R.; Tewari, L. Lignocellulosic agriculture wastes as biomass feedstocks for second-generation bioethanol production: Concepts and recent developments. 3 Biotech. 2015, 5, 337–353. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, E.; Tarrés, Q.; Delgado-Aguilar, M.; González, I.; Mutjé, P.; Rodríguez, A.; Espinosa, E.; Rodríguez, A. Suitability of wheat straw semichemical pulp for the fabrication of lignocellulosic nanofibres and their application to papermaking slurries. Cellulose 2015, 23, 837–852. [Google Scholar] [CrossRef]
- Tomás-Pejó, E.; Fermoso, J.; Herrador, E.; Hernando, H.; Jiménez-Sánchez, S.; Ballesteros, M.; González-Fernández, C.; Serrano, D. Valorization of steam-exploded wheat straw through a biorefinery approach: Bioethanol and bio-oil co-production. Fuel 2017, 199, 403–412. [Google Scholar] [CrossRef]
- Yang, S.; Bai, S.; Wang, Q. Sustainable packaging biocomposites from polylactic acid and wheat straw: Enhanced physical performance by solid state shear milling process. Compos. Sci. Technol. 2018, 158, 34–42. [Google Scholar] [CrossRef]
- Espinosa, E.; Bascón-Villegas, I.; Rosal, A.; Pérez-Rodríguez, F.; Chinga-Carrasco, G.; Rodríguez, A. PVA/(ligno)nanocellulose biocomposite films. Effect of residual lignin content on structural, mechanical, barrier and antioxidant properties. Int. J. Biol. Macromol. 2019, 141, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Robles, J.; Sánchez, R.; Díaz-Carrasco, P.; Espinosa, E.; García-Domínguez, M.; Rodríguez, A. Isolation and characterization of lignins from wheat straw: Application as binder in lithium batteries. Int. J. Biol. Macromol. 2017, 104, 909–918. [Google Scholar] [CrossRef]
- Ruiz, H.A.; Cerqueira, M.A.; Silva, H.D.; Rodríguez-Jasso, R.M.; Vicente, A.A.; Teixeira, J.A. Biorefinery valorization of autohydrolysis wheat straw hemicellulose to be applied in a polymer-blend film. Carbohydr. Polym. 2013, 92, 2154–2162. [Google Scholar] [CrossRef]
- Behrendt, F.; Neubauer, Y.; Oevermann, M.; Wilmes, B.; Zobel, N. Direct Liquefaction of Biomass. Chem. Eng. Technol. 2008, 31, 667–677. [Google Scholar] [CrossRef]
- Jiang, W.; Kumar, A.; Adamopoulos, S. Liquefaction of lignocellulosic materials and its applications in wood adhesives—A review. Ind. Crop. Prod. 2018, 124, 325–342. [Google Scholar] [CrossRef]
- Gómez-Jiménez-Aberasturi, O.; Ochoa-Gómez, J.R. New approaches to producing polyols from biomass. J. Chem. Technol. Biotechnol. 2017, 92, 705–711. [Google Scholar] [CrossRef]
- Tanaka, R.; Hirose, S.; Hatakeyama, H. Preparation and characterization of polyurethane foams using a palm oil-based polyol. Bioresour. Technol. 2008, 99, 3810–3816. [Google Scholar] [CrossRef] [PubMed]
- Bernardini, J.; Cinelli, P.; Anguillesi, I.; Coltelli, M.-B.; Lazzeri, A. Flexible polyurethane foams green production employing lignin or oxypropylated lignin. Eur. Polym. J. 2015, 64, 147–156. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, M.; Hu, L.; Zhou, Y. Synthesis of rigid polyurethane foams with castor oil-based flame retardant polyols. Ind. Crop. Prod. 2014, 52, 380–388. [Google Scholar] [CrossRef]
- Trevino, A.; Trumbo, D. Acetoacetylated castor oil in coatings applications. Prog. Org. Coatings 2002, 44, 49–54. [Google Scholar] [CrossRef]
- Technical Association of the Pulp and Paper Industry (TAPPI). TAPPI Standards: Regulation and Style Guidelines. Revised January 2018. Available online: http://www.tappi.org/content/pdf/standards/tm_guidelines_complete.pdf (accessed on 10 November 2020).
- Briones, R.; Serrano, L.; Labidi, J. Valorization of some lignocellulosic agro-industrial residues to obtain biopolyols. J. Chem. Technol. Biotechnol. 2011, 87, 244–249. [Google Scholar] [CrossRef]
- Briones, R.; Serrano, L.; Llano-Ponte, R.; Labidi, J. Polyols obtained from solvolysis liquefaction of biodiesel production solid residues. Chem. Eng. J. 2011, 175, 169–175. [Google Scholar] [CrossRef]
- Li, H.; Feng, S.; Yuan, Z.; Wei, Q.; Souzanchi, S. Highly efficient liquefaction of wheat straw for the production of bio-polyols and bio-based polyurethane foams. Ind. Crop. Prod. 2017, 109, 426–433. [Google Scholar] [CrossRef]
- Kim, D.-H.; Kwon, O.-J.; Yang, S.-R.; Park, J.; Chun, B.C. Structural, thermal, and mechanical properties of polyurethane foams prepared with starch as the main component of polyols. Fibers Polym. 2007, 8, 155–162. [Google Scholar] [CrossRef]
- Rincón, E.; Balu, A.M.; Luque, R.; Serrano, L. Insulating rigid polyurethane foams from laurel tree pruning based polyol. J. Appl. Polym. Sci. 2020, 138, 49789. [Google Scholar] [CrossRef]
- ASTM D4274-16, Standard Test Method for Testing Polyurethane Raw Materials: Determination of Hydroxyl Numbers of Polyols; ASTM International: West Conshohocken, PA, USA, 2016.
- ASTM D1622/D1622M-14, Standard Method For Apparent Density of Rigid Cellular Plastics; ASTM International: West Conshohocken, PA, USA, 2008.
- ASTM D1621-16, Standard Test Method for Compressive Properties of rigid Cellular Plastics; ASTM International: West Conshohocken, PA, USA, 2016.
- Huang, G.; Wang, P. Effects of preparation conditions on properties of rigid polyurethane foam composites based on liquefied bagasse and jute fibre. Polym. Test. 2017, 60, 266–273. [Google Scholar] [CrossRef]
- ASTM D5988-03, Standard Test Method for Determining Aerobic Biodegradation in Soil of Plastic Materials or Residual Plastic Materials After Composting; ASTM International: West Conshohocken, PA, USA, 2003.
- Kurańska, M.; Prociak, A. The influence of rapeseed oil-based polyols on the foaming process of rigid polyurethane foams. Ind. Crop. Prod. 2016, 89, 182–187. [Google Scholar] [CrossRef]
- Zlatanić, A.; Lava, C.; Zhang, W.; Petrović, Z.S. Effect of structure on properties of polyols and polyurethanes based on different vegetable oils. J. Polym. Sci. Part. B Polym. Phys. 2004, 42, 809–819. [Google Scholar] [CrossRef]
- Zhang, J.; Hori, N.; Takemura, A. Influence of NCO/OH ratio on preparation of four agricultural wastes liquefied polyols based polyurethane foams. Polym. Degrad. Stab. 2020, 179, 109256. [Google Scholar] [CrossRef]
- Kirpluks, M.; Kalnbunde, D.; Benes, H.; Cabulis, U. Natural oil based highly functional polyols as feedstock for rigid polyurethane foam thermal insulation. Ind. Crop. Prod. 2018, 122, 627–636. [Google Scholar] [CrossRef]
- Cinelli, P.; Anguillesi, I.; Lazzeri, A. Green synthesis of flexible polyurethane foams from liquefied lignin. Eur. Polym. J. 2013, 49, 1174–1184. [Google Scholar] [CrossRef]
- Wang, C.; Zheng, Y.; Xie, Y.; Qiao, K.; Sun, Y.; Yue, L. Synthesis of bio-castor oil polyurethane flexible foams and the influence of biotic component on their performance. J. Polym. Res. 2015, 22, 1–9. [Google Scholar] [CrossRef]
- Tu, Y.-C.; Suppes, G.J.; Hsieh, F.-H. Water-blown rigid and flexible polyurethane foams containing epoxidized soybean oil triglycerides. J. Appl. Polym. Sci. 2008, 109, 537–544. [Google Scholar] [CrossRef]
- Mosiewicki, M.; Dell’Arciprete, G.; Aranguren, M.; Marcovich, N. Polyurethane Foams Obtained from Castor Oil-based Polyol and Filled with Wood Flour. J. Compos. Mater. 2009, 43, 3057–3072. [Google Scholar] [CrossRef]
- Hejna, A.; Kirpluks, M.; Kosmela, P.; Cabulis, U.; Haponiuk, J.; Łukasz, P. The influence of crude glycerol and castor oil-based polyol on the structure and performance of rigid polyurethane-polyisocyanurate foams. Ind. Crop. Prod. 2017, 95, 113–125. [Google Scholar] [CrossRef]
- Wang, H.J.; Rong, M.Z.; Zhang, M.Q.; Hu, J.; Chen, H.W.; Czigány, T. Biodegradable Foam Plastics Based on Castor Oil. Biomacromolecules 2008, 9, 615–623. [Google Scholar] [CrossRef] [PubMed]
- Yeganeh, H.; Hojati-Talemi, P. Preparation and properties of novel biodegradable polyurethane networks based on castor oil and poly(ethylene glycol). Polym. Degrad. Stab. 2007, 92, 480–489. [Google Scholar] [CrossRef]




| Sample Name | Formulation (CO: LWS) | CO (g) | LWS (g) | TDI (g) | MDI (g) | RNCO/OH | Cream Time (s) | Free Rise Time (s) |
|---|---|---|---|---|---|---|---|---|
| BT | Blank | 20 | 0 | 11.5 | 0 | 0.554 | 42 | 72 |
| 80:20T | 80:20 | 16 | 4 | 11.5 | 0 | 0.647 | 50 | 98 |
| 60:40T | 60:40 | 12 | 8 | 11.5 | 0 | 0.779 | 111 | 173 |
| 50:50T | 50:50 | 10 | 10 | 11.5 | 0 | 0.867 | 102 | 148 |
| BM | Blank | 20 | 0 | 0 | 11.5 | 0.386 | 5.36 | 14.87 |
| 80:20M | 80:20 | 16 | 4 | 0 | 11.5 | 0.451 | 7.43 | 25.96 |
| 60:40M | 60:40 | 12 | 8 | 0 | 11.5 | 0.542 | 24.94 | 59.81 |
| 50:50M | 50:50 | 10 | 10 | 0 | 11.5 | 0.603 | 51 | 72 |
| Sample | Yield (%) | pH (25 °C) | IOH (mg KOH/g) | Acid number (mg KOH/g) | Viscosity (Pa·s) | Mw (g/mol) | Mn (g/mol) |
|---|---|---|---|---|---|---|---|
| LWS | 96.5 | 1.63 ± 0.02 | 604.1 ± 9.1 | 59.2 ± 0.86 | 0.6 ± 0.05 | 30,463 | 28,170 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serrano, L.; Rincón, E.; García, A.; Rodríguez, J.; Briones, R. Bio-Degradable Polyurethane Foams Produced by Liquefied Polyol from Wheat Straw Biomass. Polymers 2020, 12, 2646. https://doi.org/10.3390/polym12112646
Serrano L, Rincón E, García A, Rodríguez J, Briones R. Bio-Degradable Polyurethane Foams Produced by Liquefied Polyol from Wheat Straw Biomass. Polymers. 2020; 12(11):2646. https://doi.org/10.3390/polym12112646
Chicago/Turabian StyleSerrano, Luis, Esther Rincón, Araceli García, Jesús Rodríguez, and Rodrigo Briones. 2020. "Bio-Degradable Polyurethane Foams Produced by Liquefied Polyol from Wheat Straw Biomass" Polymers 12, no. 11: 2646. https://doi.org/10.3390/polym12112646
APA StyleSerrano, L., Rincón, E., García, A., Rodríguez, J., & Briones, R. (2020). Bio-Degradable Polyurethane Foams Produced by Liquefied Polyol from Wheat Straw Biomass. Polymers, 12(11), 2646. https://doi.org/10.3390/polym12112646