Abstract
The development of materials showing hydrogen sorption reactions close to room temperature and ambient pressure will promote the use of hydrogen as energy carrier for mobile and stationary large-scale applications. In the present study, in order to reduce the thermodynamic stability of MgH2, Ni has been added to form Mg2NiH4, which has been mixed with various borohydrides to further tune hydrogen release reactions. De-hydrogenation/re-hydrogenation properties of Mg2NiH4-LiBH4-M(BH4)x (M = Na, K, Mg, Ca) systems have been investigated. Mixtures of borohydrides have been selected to form eutectics, which provide a liquid phase at low temperatures, from 110 °C up to 216 °C. The presence of a liquid borohydride phase decreases the temperature of hydrogen release of Mg2NiH4 but only slight differences have been detected by changing the borohydrides in the eutectic mixture.
1. Introduction
To overcome the increasing worldwide demand of energy, the use of hydrogen as sustainable and efficient energy carrier plays an essential role. In this frame, the development of materials able to store hydrogen close to room temperature and ambient pressure will enable the use of hydrogen in mobile and stationary large-scale applications. Low cost and light weight hydrides are promising materials for solid-state hydrogen storage, owing to their high gravimetric and volumetric hydrogen capacity [1]. The theoretical hydrogen content of LiBH4 is 18.5% wt, i.e., the highest among metal borohydrides, while it is equal to 10.7% wt for NaBH4, 7.5% wt for KBH4, 14.9% wt for Mg(BH4)2 and 11.6% wt for Ca(BH4)2. Observed values are usually lower, depending on the experimental conditions.
Usually light metal hydrides absorb and release hydrogen through sluggish kinetics at relatively high temperatures. In order to reduce their thermodynamic stabilities, transition metals have been added to form complex metal hydrides [2], such as in the case of MgH2 mixed with Ni to form Mg2NiH4, with a theoretical hydrogen content equal to 3.6% wt. This complex metal hydride, upon heating, shows a polymorphic transition from a monoclinic to a cubic structure at 244 °C, together with the release of one hydrogen to form Mg2NiH3 [3,4]. The main dehydrogenation reaction occurs at 305 °C under 5%H2/95%N2 flow or 1 bar H2 [3,5]. The thermodynamic properties of the Mg-Ni-H ternary system were assessed by Zeng et al. [6], where the values of temperature of decomposition as a function of hydrogen pressure are reported. Upon cycling at low hydrogen pressure, Mg2Ni can absorb hydrogen (at Tpeak = 247 °C, at 3 bar H2) to form the partial hydrogenated hexagonal solid solution Mg2NiH0.3 [7].
Borohydrides are a class of inorganic ionic materials suitable for hydrogen storage in the solid state [8]. The most promising ones contain a light alkali or alkali-earth metal cation ionically bonded to the complex borohydride anion (BH4−) [8]. They attracted the attention of the latest research thanks to their extremely rich chemistry and tuneable properties in relation to their structures and interactions in mixtures. Among borohydrides, several bimetallic compounds are present. Moreover, selected mixtures of borohydrides form eutectic melts at a relatively low temperatures [9]. This feature can be used to easily infiltrate them in nanometric scaffold to improve the hydrogen absorption/desorption kinetics and cyclability [10]. Recently, a systematic study of hydrogen release in pure and eutectic mixtures of borohydrides was reported by Paskevicius et al. [9].
The full understanding of thermodynamic and kinetic properties of hydrogen sorption in borohydride-based systems will allow obtaining new suitable materials for effective hydrogen storage. In the literature, many tailoring routes have been proposed to improve thermodynamic and kinetic properties of both metal hydrides and complex hydrides. The so called Reactive Hydride Composites (RHC), for example, allow mixtures of metal hydrides and borohydrides to release hydrogen in a reversible manner under moderate temperature and hydrogen pressure conditions [11].
The thermodynamic properties and phase diagrams of the LiBH4-NaBH4-KBH4 system were recently assessed [12]. In this system, two eutectic compositions are present. 0.725LiBH4-0.275KBH4 (LiK) and 0.70LiBH4-0.30NaBH4 (LiNa) melt at an onset temperature of 105 °C and 216 °C, respectively. Even if the LiK eutectic shows a stable liquid above 105 °C, it does not decompose below 400 °C [13]. The decomposition of the 0.68LiBH4-0.32NaBH4 mixture was investigated in the literature and it shows a release above 400 °C [14], lowered to 250 °C when nanoconfined [15]. 0.55LiBH4-0.45Mg(BH4)2 eutectic composition (LiMg) melts at 180 °C and releases hydrogen during melting and above 250 °C [16,17,18], its decomposition is lowered to 150 °C upon nanoconfinement, thus suppressing the evolution of diborane and the formation of closoboranes [19,20]. 0.68LiBH4-0.32Ca(BH4)2 eutectic composition (LiCa) melts at 200 °C and decomposes at 350 °C [21], lowered down to 200 °C upon confinement [22,23,24,25].
Recently various studies have explored the hydrogen release properties of mixtures of complex metal hydrides and borohydrides, such in the case of Mg2FeH6-M(BH4)x systems (M = Li, Na, K, Mg, Ca) [26]. Mg2NiH4-M(BH4)x systems (M = Li, Na, Ca) have been studied as well, showing an improvement of the hydrogen release properties and cyclability [27,28,29,30,31]. The decomposition reactions can form boride species (e.g., MgNi2.5B2), which reversibly play as B-donors to form back the borohydride during the re-hydrogenation process. In the Mg2NiH4-LiBH4 system, hydrogen release under argon flow starts at roughly 300 °C and it can be re-hydrogenated at 100 bar and 350 °C in about 4 h [27]. The hydrogen release in the Mg2NiH4-NaBH4 system has an onset temperature close to 250 °C under vacuum and it occurs in three different steps [29]. The Mg2NiH4-Ca(BH4)2 system releases hydrogen above 300 °C [30]. There is no report in the literature on the Mg2NiH4-KBH4 system, since it is expected to release hydrogen at temperatures that are considered not suitable for the employment of this material in real applications. The Mg2NiH4-Mg(BH4)2 system has not been studied yet.
As reported above, mixtures of borohydrides have shown the formation of eutectic melts, which allow the release of hydrogen from the liquid state at low temperatures [13,32]. In the present study, we apply the RHC approach to eutectic mixtures of borohydrides, aiming to further improve hydrogen sorption properties of the mixed borohydrides. In order to compare our results with the literature, we investigated the as prepared Mg2NiH4 and the RHC mixture Mg2NiH4-LiBH4. Then Mg2NiH4 was mixed with all the binary eutectic compositions of LiBH4 and M(BH4)x (M = Na, K, Mg, Ca). The thermal behaviour and decomposition temperature were analysed by High-Pressure Differential Scanning Calorimetry (HP-DSC). Ball milled mixtures (BM) and samples after thermal cycling where analysed by powder X-ray diffraction (PXD) to identify crystalline decomposition products. This study will allow an understanding of the behaviour of a RHC mixtures with a liquid phase and its role in the thermodynamic destabilization of hydrogen sorption reactions.
2. Materials and Methods
Mg2NiH4 was synthesized by ball milling (BM) MgH2 and Ni in a molar ratio 2:1. Approximately 5 g of powder were milled in a planetary Fritsch Pulverisette 6 mill for 10 h at 350 r.p.m. in a custom-made stainless steel (SS) 250 mL vial, under 10 bar of hydrogen and a ball-to-powder mass ratio of 5:1, using 15 mm SS balls. After milling, the powder was transferred in a Parr Reactor and heated under 10 bar of hydrogen up to 370 °C for 10 h.
Lithium borohydride (LiBH4, purity >99% from Rockwood Lithium), sodium borohydride (NaBH4, purity 99.99% from Sigma-Aldrich), potassium borohydride (KBH4, purity 99% from Sigma-Aldrich), magnesium borohydride (Mg(BH4)2, purity >95% from KatChem) and calcium borohydride (Ca(BH4)2, purity >95% from KatChem) were mixed by BM in the eutectic molar composition. Approximately 1 g of sample was milled under nitrogen atmosphere in 80 mL SS vials and with SS balls (o.d. 5 mm), with a balls-to-powder mass ratio of 30:1. The samples were milled at 350 r.p.m. for 5 min for 12 times, with a 2 min pause in between each run. Using the same BM settings, the eutectic borohydride mixtures and Mg2NiH4 were ball milled together in the correct molar ratio to obtain a complete conversion to MgNi2.5B2 after decomposition of the borohydrides. All preparations and manipulations of the samples were performed in an argon or nitrogen-filled glove box with a circulation purifier, p(O2, H2O) < 1 ppm. An overview of milled samples and corresponding compositions is reported in Table 1. The investigated mixtures will be named by the main metals that are present (e.g., the LiBH4 and KBH4 in eutectic composition will be named LiK and its mixture with Mg2NiH4 will be named MgNiLiK).

Table 1.
Investigated samples and their molar composition.
To analyse the phase mixtures and decomposition products, powder X-ray diffraction (PXD) measurements were performed at room temperature. A Panalytical X-pert (Almelo, The Netherlands) (Cu Kα = 1.54059 Å, Kβ = 1.54446 Å; Kα/Kβ = 0.375) in capillary transmission set-up (Debye-Scherer geometry) was used. Patterns were collected from 10° to 70° 2θ range, step size 0.016, time step 60 s. Samples were mounted in the glove box in 0.5 mm glass capillaries and sealed with plastiline, then moved out of the glove box and sealed with flame.
To analyse hydrogen release and thermal behaviour of the samples, approximately 5 mg of powder were loaded into aluminium crucibles with a lid, where a hole was made to let the gas flow out. A high-pressure 204 Netsch DSC (HP-DSC, NETZSCH GmbH, Selb, Germany), inside a nitrogen-filled glove box, was used to analyse all the samples. Heating and cooling were performed from room temperature (RT) up to 440 °C at 5 °C/min under a static pressure of 2.2 bar of H2 loaded at RT, that rose up to 2.8 bar at 440 °C. To clarify reversibility and phase transformation various cycling were performed.
3. Results
Figure 1a, Figure 2a, Figure 3a, Figure 4a, Figure 5a and Figure 6a, report PXD patterns of the as-prepared samples, which can be compared with the patterns collected after thermal cycling. Figure 1b, Figure 2b, Figure 3b, Figure 4b, Figure 5b and Figure 6b show the DSC traces upon cycling.
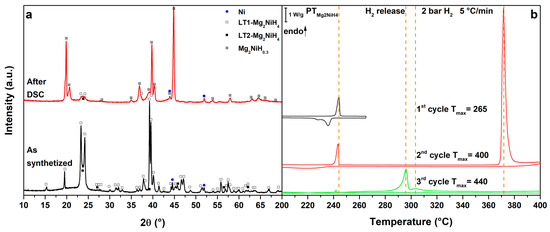
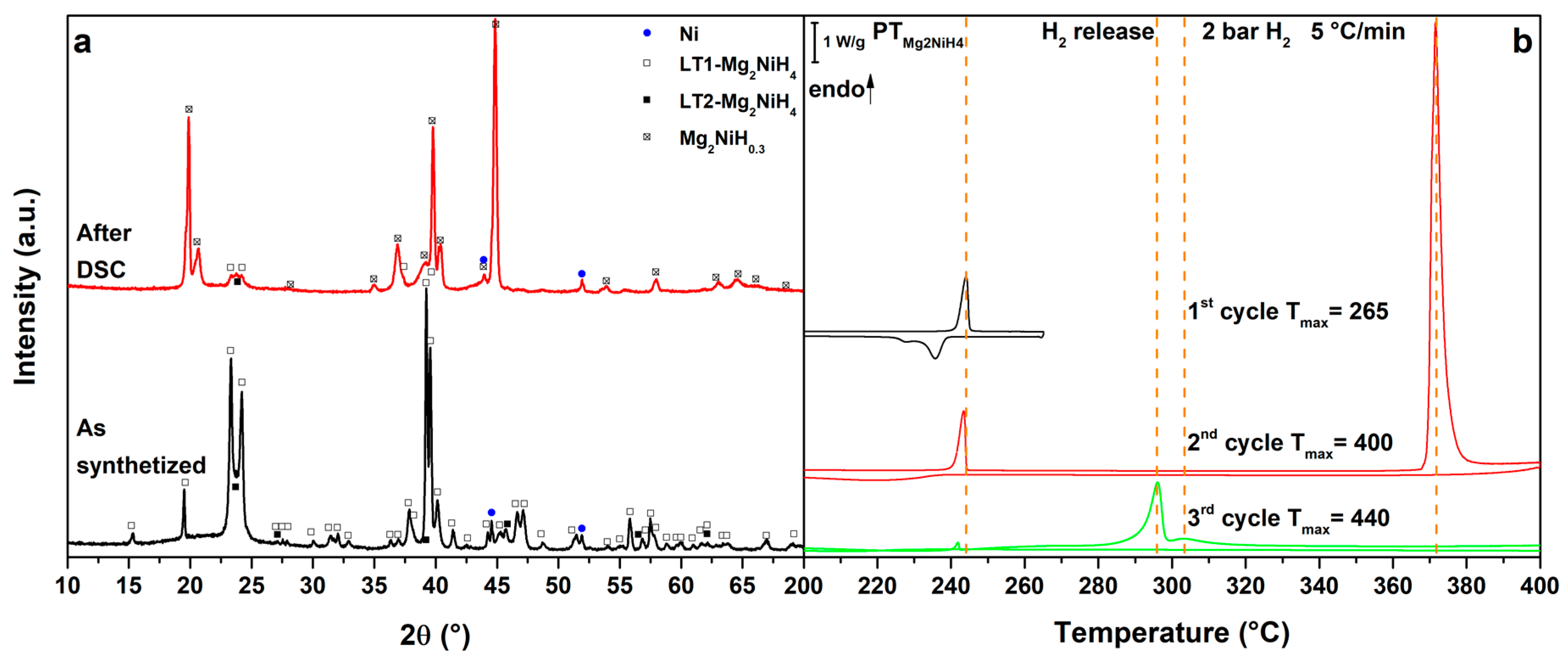
Figure 1.
(a) PXD of the as synthetized Mg2NiH4 (black, down) and after cycling in HP-DSC (red, up); (b) HP-DSC cycling of Mg2NiH4 at 5 °C/min under a static pressure of 2.2 bar of H2.
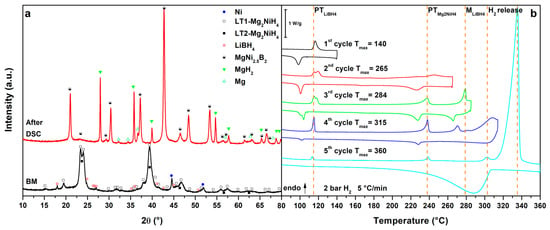
Figure 2.
(a) PXD of the ball milled MgNiLi system (black, down) and after cycling in HP-DSC (red, up); (b) HP-DSC cycling of MgNiLi at 5 °C/min under a static pressure of 2.2 bar of H2.
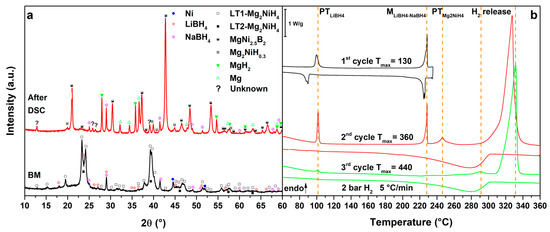
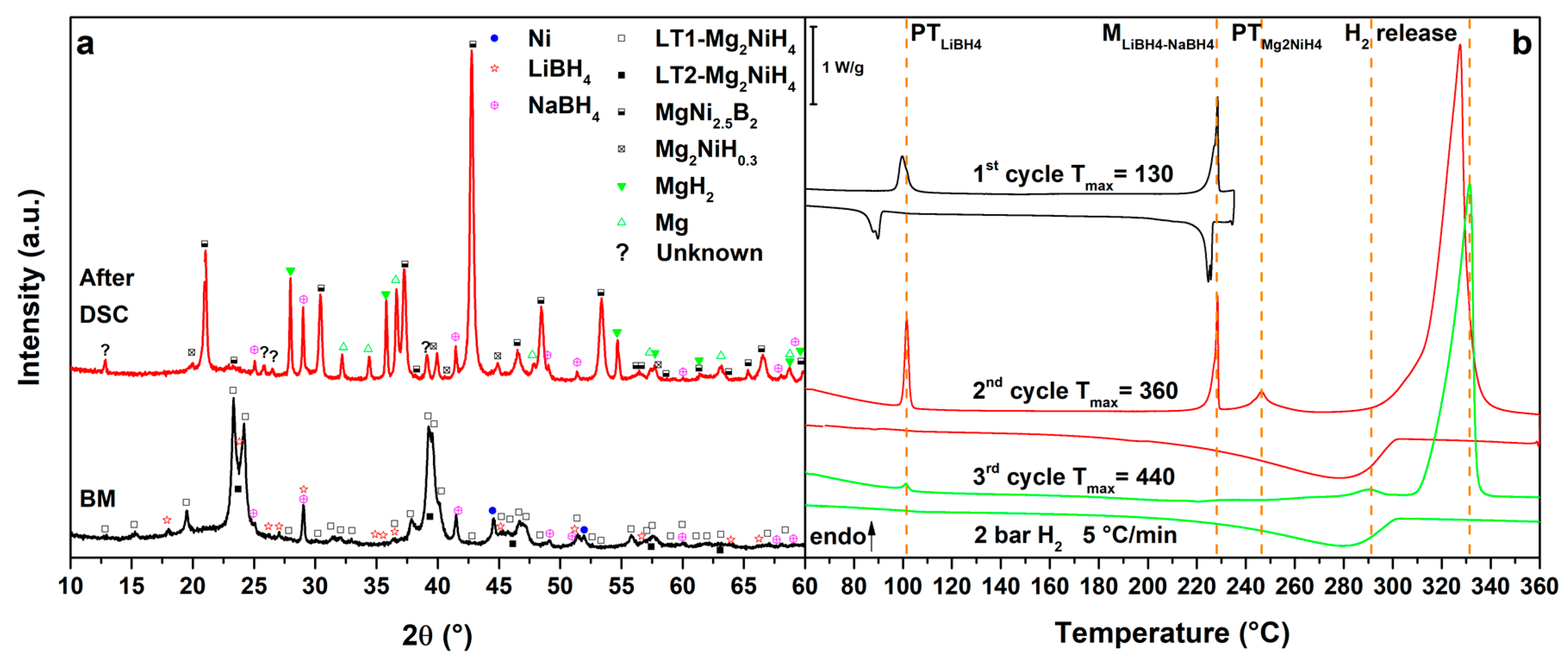
Figure 3.
(a) PXD of the ball milled MgNiLiNa (black, down) and after cycling in HP-DSC (red, up); (b) HP-DSC cycling of MgNiLiNa at 5 °C/min under a static pressure of 2.2 bar of H2.
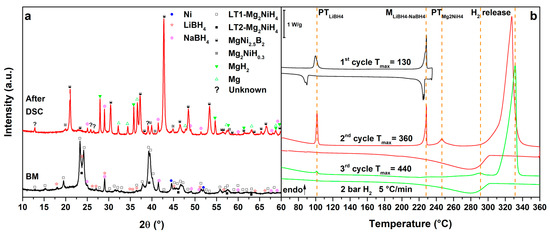
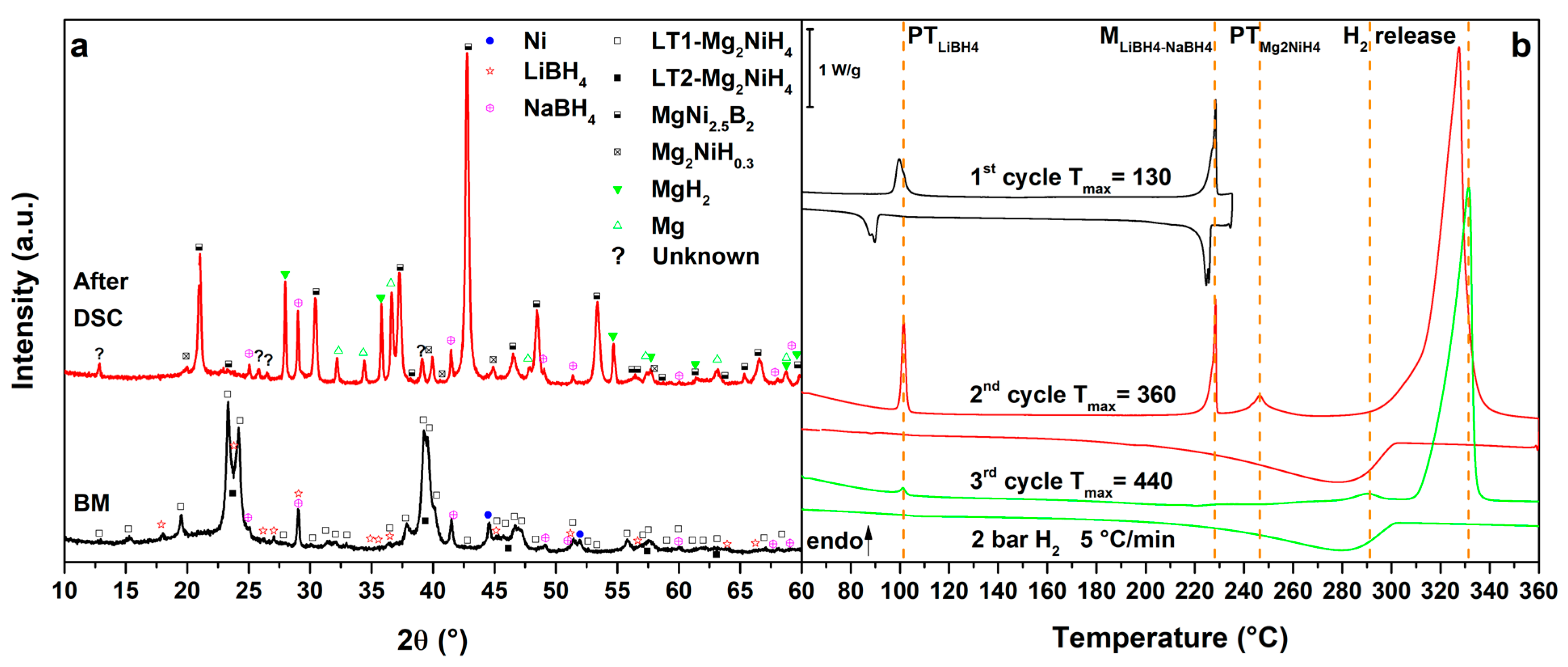
Figure 4.
(a) PXD of the ball milled MgNiLiNa (black, down) and after cycling in HP-DSC (red, up); (b) HP-DSC cycling of MgNiLiNa at 5 °C/min under a static pressure of 2.2 bar of H2.
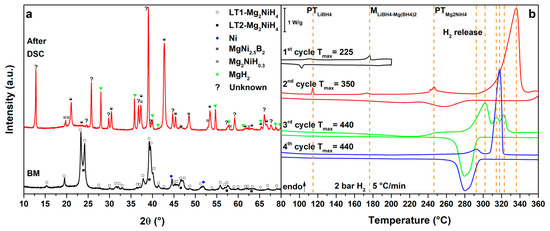
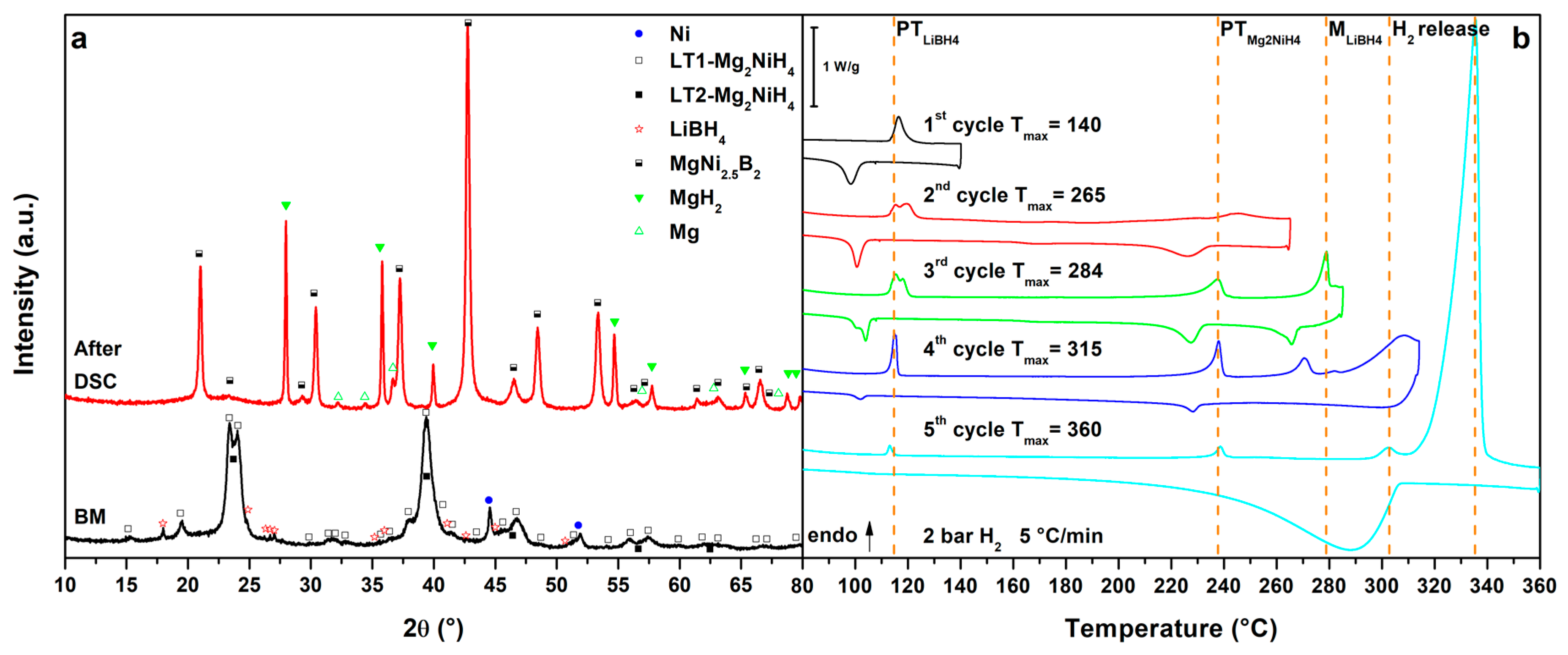
Figure 5.
(a) PXD of the ball milled MgNiLiMg (black, down) and after cycling in HP-DSC (red, up); (b) HP-DSC cycling of MgNiLiMg at 5 °C/min under a static pressure of 2.2 bar of H2.
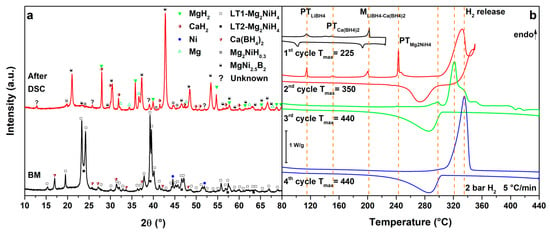
Figure 6.
(a) PXD of the ball milled MgNiLiCa (black, down) and after cycling in HP-DSC (red, up); (b) HP-DSC cycling of MgNiLiCa at 5 °C/min under a static pressure of 2.2 bar of H2.
3.1. Mg2NiH4
As a consequence of the synthesis procedure, the diffraction pattern of Mg2NiH4 (Figure 1a, as synthetized) presents broad diffraction peaks of the monoclinic phase stable at room temperature, LT1. The orange-reddish colour of the powder indicates the presence of a microtwinned modification of the room-temperature phase, called LT2 [33], as confirmed by PXD pattern. As previously reported for a similar synthesis route [34], even if a molar ratio 2:1 of starting MgH2 and Ni have been used to obtain the complex metal hydride, traces of unreacted Ni are present in the sample.
In the DSC trace of pure Mg2NiH4 on heating (Figure 1b, 1st cycle), at Tpeak = 244 °C, the monoclinic-to-cubic polymorphic phase transition can be observed. This transition is reversible, as clearly shown by the exothermic peak upon cooling at Tpeak = 234 °C, due to the cubic high temperature phase forming LT1 and LT2 [3,4]. In the 2nd cycle (Figure 1b, 2nd cycle), the polymorphic transition is observed again and upon further heating an endothermic peak related to hydrogen release can be detected. The complex metal hydride decompose to Mg2Ni [34], under a hydrogen backpressure of 2.7 bar, at Tpeak = 372 °C. Upon cooling, in the 2nd cycle, a broad exothermic peak of hydrogenation to form Mg2NiH0.3 can be observed below 235 °C [7]. Upon further cycling (Figure 1b, 3rd cycle), the sample releases hydrogen starting from 280 °C (Tpeak = 296 °C), from Mg2NiH0.3.
Mg2NiH0.3, together with traces of Mg2NiH4, is observed in PXD after cooling (Figure 1a, after DSC).
3.2. RHC Mixtures
In almost all the diffraction patterns of Mg2NiH4 mixed with borohydrides, the composing phases are visible (Figure 2a, Figure 3a, Figure 4a, Figure 5a and Figure 6a, BM). However, in the MgNiLiK system (Figure 3a, BM), the milling causes the formation of the LiK(BH4)2 bimetallic phase, in agreement with the literature [32]. In the MgNiLiMg system (Figure 5a, BM), LiBH4 and Mg(BH4)2 are hardly visible after milling.
3.3. Mg2NiH4–LiBH4
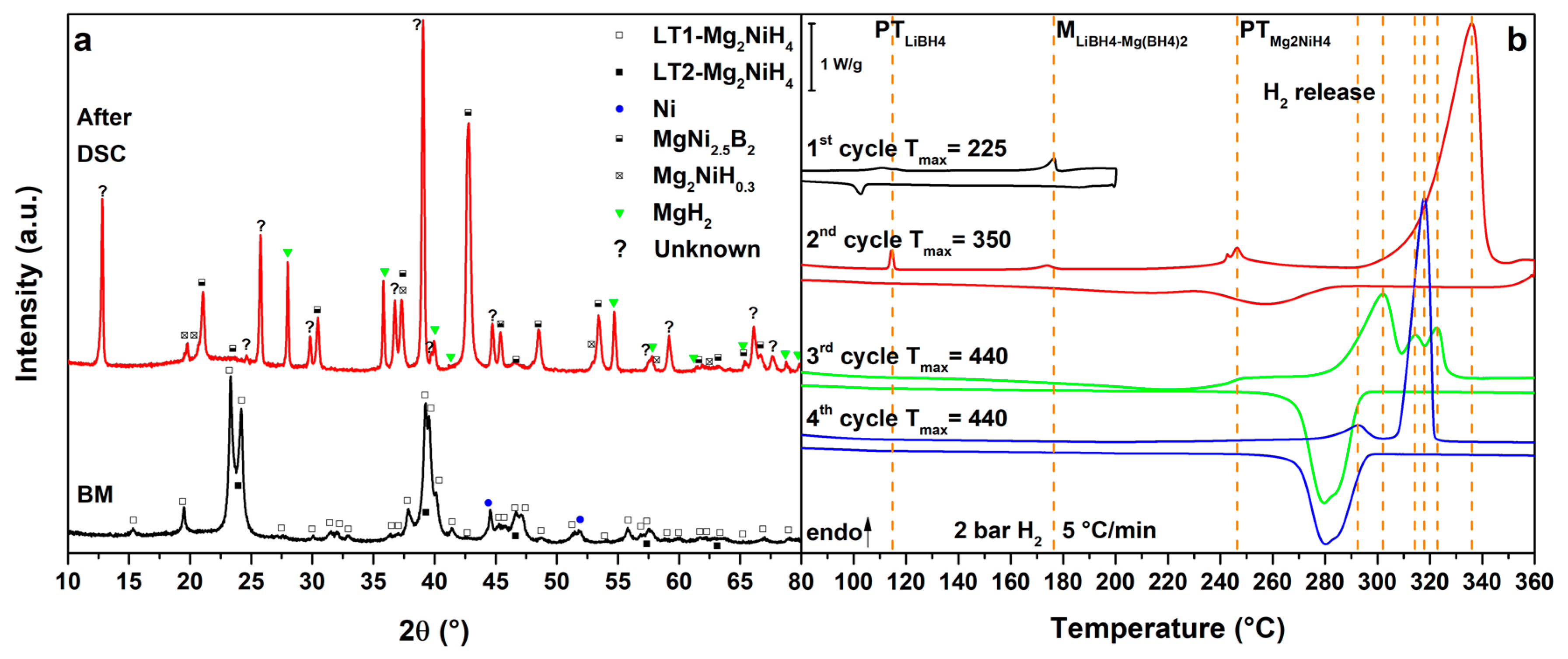
Figure 2b shows the DSC signals for the RHC mixture MgNiLi upon cycling. In the 1st cycle (Figure 2b, 1st cycle), the polymorphic transition of LiBH4 is identified Tpeak = 117 °C, which is reversible upon cooling. In the 2nd cycle (Figure 2b, 2nd cycle), after the polymorphic transition of LiBH4, also the polymorphic phase transition of Mg2NiH4 is observed as a broad peak around 245 °C. Both DSC signals can be observed also on cooling. Furthermore, in the 3rd cycle (Figure 2b, 3rd cycle), after the polymorphic transition peaks, the melting of LiBH4 can be detected at Tpeak = 279 °C. All transitions are reversible and well visible during the cooling process and temperature values are in good agreement with the literature [3,4,35]. In the 4th cycle (Figure 2b, 4th cycle), after the polymorphic transition peaks and LiBH4 melting, at Tpeak = 308 °C a broad endothermic peak due to hydrogen release from LiBH4 can be observed under 2.7 bar H2. In this case, on cooling, only small peaks from the polymorphic transitions can be observed, suggesting the occurrence of a reaction. In fact, in the last cycle (Figure 2b, 5th cycle) only small DSC peaks of Mg2NiH4 polymorphic transition, LiBH4 polymorphic transition and melting can be observed on heating, evidencing the presence of a small quantity of parent hydrides in the sample. At higher temperatures, at Tpeak = 335 °C, the main hydrogen release from the RHC mixture can be observed, with LiBH4 and Mg2NiH4 forming MgNi2.5B2 and Mg. Upon cooling below 307 °C, Mg absorbs hydrogen to form MgH2.
The PXD pattern after the DSC analysis (Figure 2a, after DSC) confirms the total decomposition of LiBH4 and Mg2NiH4. In fact, MgNi2.5B2, MgH2 and a small quantity of unreacted Mg are observed but no Li-containing phases are evidenced, likely because of the light scattering factor. LiBH4 cannot be reformed because of the low hydrogen pressure applied. The obtained results are in good agreement with the literature [27] and confirms the decrease of the decomposition temperature of both Mg2NiH4 and LiBH4 when mixed together into a RHC.
3.4. Mg2NiH4–LiBH4–KBH4 Eutectic Composition
Figure 3 reports the results obtained for the MgNiLiK system. Upon heating, in the 1st cycle (Figure 3b, 1st cycle), the melting of the LiK eutectic can be observed at Tpeak = 110 °C, which is reversible upon cooling [12]. In the 2nd cycle (Figure 3b, 2nd cycle), after the eutectic melting, the polymorphic transition of Mg2NiH4 is observed as a broad peak at Tpeak = 246 °C. Above 290 °C (Tpeak = 325 °C) the main hydrogen release from the RHC mixture can be observed under 2.7 bar H2. The mixture has decomposed into MgNi2.5B2, Mg2Ni and Mg. In fact, a broad peak of hydrogenation can be detected, upon cooling, below 300 °C to form Mg2NiH0.3 and MgH2. In the 3rd cycle (Figure 3b, 3rd cycle), no transitions from LiK eutectic and Mg2NiH4 are observed, endorsing their decomposition. Hydrogen release is observed in a broad peak around 292 °C, related to the hydrogen release from Mg2NiH0.3 and above 310 °C, related to decomposition of MgH2. In this case, the hydrogen release temperature of the RHC mixture is lower of about 10 °C compared to the MgNiLi system. Upon cooling, the Mg hydrogenation peak is more defined in the 3rd cycle because of a possible activation upon previous thermal cycling.
Mg2NiH0.3, MgH2 and MgNi2.5B2 are present in the PXD pattern after DSC (Figure 3a, after DSC), together with an unknown phase. The XRD peak positions of this unknown phase correspond to those observed after decomposition of the Mg2NiH4-Ca(BH4)2 system [30]. The presence of KBH4 in the mixture suggests that not all the borohydride present in the mixture has been decomposed upon thermal cycling. Therefore, from the liquid eutectic mixture of borohydrides, only LiBH4 actively reacts with Mg2NiH4, whereas the most stable KBH4 remains unaffected.
3.5. Mg2NiH4–LiBH4–NaBH4 Eutectic Composition
Figure 4 presents the results obtained for the MgNiLiNa system. Upon heating in the 1st cycle (Figure 4b, 1st cycle), we observe the polymorphic transition of LiBH4 at Tpeak = 98 °C, stabilized for the presence of NaBH4 [36] and the melting of the LiNa eutectic at Tpeak = 228 °C. Both phase transformations are reversible and slightly undercooled on cooling. In the 2nd cycle (Figure 4b, 2nd cycle), after the previous phase transformations, also the polymorphic transition of Mg2NiH4 is observed at Tpeak = 246 °C, followed by the main hydrogen release from the RHC mixture at Tpeak = 327 °C under 2.7 bar H2. The RHC mixture decomposes similarly to the MgNiLiK system and, on cooling, Mg2Ni and Mg are hydrogenated to form Mg2NiH0.3 and MgH2 below 302 °C. In the 3rd cycle (Figure 4b, 3rd cycle), previous transitions from LiNa and Mg2NiH4 are hardly visible, confirming their decomposition. In fact, the hydrogen release from Mg2NiH0.3 and MgH2 are observed at Tpeak = 290 °C and 331 °C, respectively. No improvement in the hydrogenation of Mg can be seen on cooling in the 3th cycle. In comparison to the starting components, the hydrogen release temperature of the RHC mixture has not been lowered by the substitution of KBH4 with NaBH4.
The decomposition products of the MgNiLiNa system are similar to those of MgNiLiK, as evidenced by the PXD analysis (Figure 4a, after DSC). Even in this case, the most stable NaBH4 borohydride is still present after thermal cycling.
3.6. Mg2NiH4–LiBH4–Mg(BH4)2 Eutectic Composition
Results obtained for the MgNiLiMg system are shown in Figure 5. In the 1st cycle (Figure 5b, 1st cycle), the polymorphic transition of LiBH4 is observed as a broad peak at Tpeak = 111 °C, followed by the melting of the eutectic at Tpeak = 176 °C. Upon cooling the crystallization of the eutectic is not clearly detected, possibly because some Mg(BH4)2 has already reacted, enriching the LiBH4 fraction in the composition of the borohydride mixture [16,17,18]. In fact, the DSC peak due to the LiBH4 phase transformation appears more intense on cooling with respect to that observed on heating. Consequently, in the 2nd cycle (Figure 5b, 2nd cycle), the polymorphic transition of LiBH4 is more defined and the LiMg eutectic melting is hardly detected as a broad peak. At higher temperatures, the Mg2NiH4 polymorphic transition is detected at Tpeak = 246 °C, together with a small peak of hydrogen release from the liquid borohydride, as previously observed in the literature [20]. Furthermore, the main hydrogen release from the RHC mixture starts above 290 °C under 2.7 bar H2 (Tpeak = 336 °C). In this temperature range, the decomposition of Mg(BH4)2 is also expected [9]. On the other hands, after the decomposition of the mixture, MgNi2.5B2 and Mg are expected to be formed, so that the decomposition of Mg(BH4)2 to Mg, B and H2 is limited. Upon cooling in the 2nd cycle, a broad hydrogenation peak is observed at Tpeak = 260 °C related to the formation of MgH2. Actually, in the 3rd cycle (Figure 5b, 3rd cycle), no peaks related to LiBH4 or Mg2NiH4 phase transformations are observed but several peaks related to hydrogen release can be observed above 231 °C, in a complex multi-steps reaction (Tpeak = 302 °C, 315 °C and 323 °C). These peaks are related to the full decomposition of residual Mg(BH4)2 [9], together with hydrogen release from Mg2NiH0.3 and MgH2. Hydrogenation of Mg2Ni and Mg is clearly observed upon cooling below 300 °C. Finally, in the 4th cycle (Figure 5b, 4th cycle), only hydrogen release peaks from Mg2NiH0.3 (Tpeak = 293 °C) and MgH2 (Tpeak = 317 °C) are observed and, during cooling, a peak related to hydrogenation can be detected again below 297 °C.
The decomposition products detected in the PXD patter after thermal cycling (Figure 5a, after DSC) are always the same, as observed for the MgNiLiK and MgNiLiNa systems. However, in this case, no traces of residual borohydrides are observed, suggesting the full reaction of Mg(BH4)2. It is worth to note that the intensity of the diffraction peaks of the unknown phase are higher with respect to the previous systems. The high content of Mg and B in this system suggests that the unknown phase could be related to the Mg-Ni-B system.
3.7. Mg2NiH4–LiBH4–Ca(BH4)2 Eutectic Composition
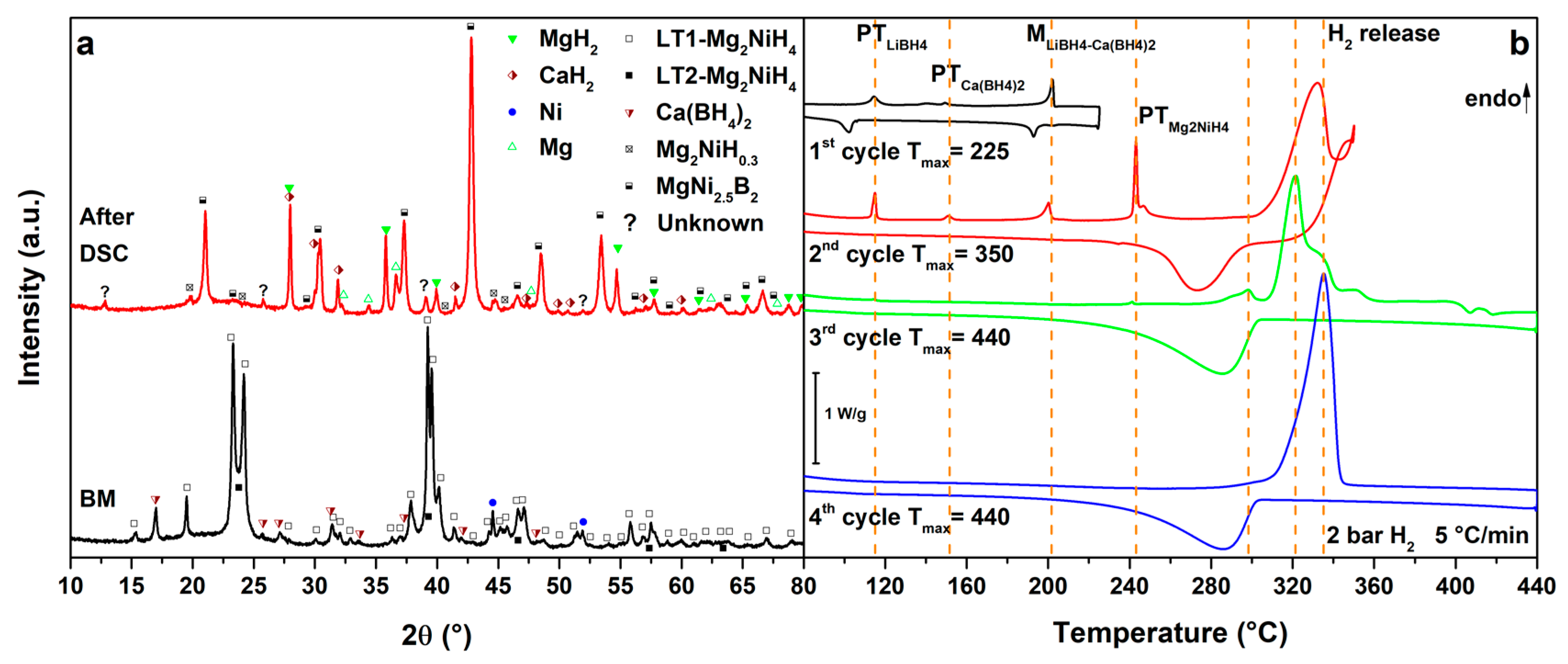
Finally, the results obtained for the MgNiLiCa system are reported in Figure 6. The DSC trace of the 1st cycle (Figure 6b, 1st cycle) presents the polymorphic transition of LiBH4 at Tpeak = 115 °C, the polymorphic transition of Ca(BH4)2 at Tpeak = 150 °C and the melting of the LiCa eutectic at Tpeak = 202 °C. Only the polymorphic transition of Ca(BH4)2 cannot be observed by DSC on cooling, because of sluggish kinetics [21]. In the 2nd cycle (Figure 6b, 2nd cycle), all the previous transitions of the LiCa system are observed. At higher temperatures, the polymorphic transition of Mg2NiH4 is detected at Tpeak = 243 °C, together with a small peak of hydrogen release from the liquid borohydride, as previously observed in the MgNiLiMg system and in the literature [37]. Furthermore, a DSC endothermic peak due to hydrogen release starts above 300 °C (Tpeak = 332 °C), under 2.7 bar H2, so that heating has been stopped at 350 °C, in order to separate hydrogen release reactions. The DSC exothermic DSC peaks due to hydrogenation reactions of Mg2Ni and Mg can be observed on cooling below 304 °C. In the 3rd cycle (Figure 6b, 3rd cycle), no peaks from the LiCa system are observed, suggesting the occurrence of reactions between Mg2NiH4 and the borohydride mixture in the previous cycle. A DSC peak at Tpeak = 298 °C is clearly related to the hydrogen release from Mg2NiH0.3, followed by the decomposition of MgH2 and, at higher temperatures, by the decomposition of Ca(BH4)2 to CaH2 up to 440 °C. In fact, in the 4th cycle (Figure 6b, 4th cycle), the decomposition of the Mg2NiH0.3 is hardly observed but the decomposition of MgH2 is clearly occurring at Tpeak = 335 °C. During cooling, a broad peak of hydrogenation can be detected below 304 °C both in the 3rd and 4th cycle.
CaH2 is observed, in addition to the previously observed decomposition products, in the PXD pattern after cycling (Figure 6a, after DSC).
4. Discussion
The results obtained in this work for the Mg2NiH4 and the Mg2NiH4–LiBH4 (MgNiLi) system are in good agreement with previous studies reported in the literature [3,7,27]. Mg2NiH4 releases hydrogen under 2.7 bar of H2 at relatively high temperature (Tpeak = 372 °C) and it is partially hydrogenated to Mg2NiH0.3 on cooling. However, this temperature can be lowered by the addition of LiBH4. In fact, in the RHC mixture MgNiLi, the release of hydrogen, under a backpressure of 2.7 bar, starts above 330 °C. Compared with the pristine compounds, their mixture leads to a release of hydrogen at lower temperature for both LiBH4 and Mg2NiH4 to form MgNi2.5B2, Mg2Ni and Mg.
Mg2NiH4 was then mixed with all the binary eutectic compositions of LiBH4 and M(BH4)x (M = Na, K, Mg, Ca). In all systems, the decomposition of both LiBH4 and Mg2NiH4 is evidenced by a broad DSC peak, localized around Tpeak = 320–330 °C. Upon cooling, in the present conditions, the decomposed samples, in all RHC studied mixtures, can be re-hydrogenated to form Mg2NiH0.3 and MgH2. In further thermal cycles, two decomposition peaks are observed, related to Mg2NiH0.3 and MgH2 hydrogen release, respectively. Table 2 summarizes the peak temperatures of transitions and reactions for all investigated systems.

Table 2.
Peak temperatures of transitions and reactions for all investigated systems (H: heating, C: cooling, PT: polymorphic transition, M: melting, Cr: crystallization).
All the systems investigated in this work present the decomposition of borohydrides from the liquid state, which appears to play a role on the temperature of decomposition of the RHC mixtures. When only LiBH4 is present, the decomposition peak is at a slightly higher temperature with respect to RHC mixtures containing eutectic borohydrides mixtures, though it does not change significantly in all different systems. When a highly stable borohydride (i.e., NaBH4 and KBH4) is present in the eutectic mixture, only LiBH4 plays and active role in the mixture and the RHC mixture does not decompose completely, so that the stable borohydride is still present in the decomposition products after thermal cycling. On the other hand, if an alkali-earth borohydride (i.e., Mg(BH4)2 and Ca(BH4)2) is mixed with LiBH4 to form a eutectic, the liquid phase fully decomposes, namely at lower temperatures compared to the pure eutectic mixture alone but complex multi-step reactions are taking place.
In all systems, when the borohydrides and the Mg2NiH4 decompose, they always react to form MgNi2.5B2 and Mg. The presence of an unknown decomposition product has also been observed in all RHC mixtures with eutectic borohydride mixtures. The low backpressure applied in this case is not high enough to form back the starting borohydrides, though it is able to hydrogenate the decomposed Mg2Ni and Mg to form Mg2NiH0.3 and MgH2.
Decomposition temperatures in investigated systems have not been lowered significantly compared to previous studies of Mg2NiH4-MBH4 mixtures. In fact, the hydrogen release temperature is still far from ambient conditions. A full rehydrogenation has not been obtained because of the limited H2 pressure applied in the experimental conditions. As for previous studied mixtures [27,28,29,30,31], a hydrogen pressure of 100 bar is expected to fully rehydrogenate the systems. The nanoconfinement of the studied mixtures into a nanoporous scaffold to obtain nanostructured materials [13,15,19,20,22,23,24,25,31,32] might be explored to improve the thermodynamics and kinetics of hydrogen sorption reactions and cyclability.
5. Conclusions
In this study RHC systems obtained by mixing Mg2NiH4 and eutectic mixtures of borohydrides have been investigated, to explore possible improvements in hydrogen release properties of those systems.
In the investigated RHC mixtures, when LiBH4 is in eutectic mixture with borohydrides that contain stable single charged metal cation, a slight improvement in the decomposition temperature of the RHC mixture is observed but a leftover of the more stable borohydrides is detect after cycling.
The presence of double charged metal cation in the eutectic mixture causes a full decomposition of the borohydrides in complex multi-steps reactions. The presence of a stable liquid at low temperature promotes the decrease of the hydrogen release temperature, if compared to that of pure Mg2NiH4.
Acknowledgments
European Marie Curie Actions under ECOSTORE grant agreement No. 607040 is acknowledged for supporting this work.
Author Contributions
E.M.D. and M.B. conceived and designed the experiments; E.M.D. and S.V. performed the experiments and analysed the data; C.P. and M.D. contributed with materials and fruitful discussion of the results, E.M.D. and M.B. wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Callini, E.; Atakli, Z.Ö.K.; Hauback, B.C.; Orimo, S.; Jensen, C.; Dornheim, M.; Grant, D.; Cho, Y.W.; Chen, P.; Hjörvarsson, B.; et al. Complex and liquid hydrides for energy storage. Appl. Phys. A 2016, 122, 353. [Google Scholar] [CrossRef]
- Dornheim, M. Thermodynamics of metal hydrides: Tailoring reaction enthalpies of hydrogen storage materials. In Thermodynamics—Interaction Studies—Solids, Liquids and Gases; InTech: Rijeka, Croatia, 2011; pp. 891–918. [Google Scholar]
- Martínez-Coronado, R.; Retuerto, M.; Torres, B.; Martínez-Lope, M.J.; Fernández-Díaz, M.T.; Alonso, J.A. High-pressure synthesis, crystal structure and cyclability of the Mg2NiH4 hydride. Int. J. Hydrogen Energy 2013, 38, 5738–5745. [Google Scholar] [CrossRef]
- Noréus, D. Properties of formal low-valence transition metal—hydrogen complexes in Mg2NiH4 and Na2PdH2. Z. Phys. Chem. 1989, 163, 575–578. [Google Scholar] [CrossRef]
- Čermák, J.; Král, L.; David, B. Hydrogen diffusion in Mg2NiH4 intermetallic compound. Intermetallics 2008, 16, 508–517. [Google Scholar] [CrossRef]
- Zeng, K.; Klassen, T.; Oelerich, W.; Bormann, R. Thermodynamic analysis of the hydriding process of Mg-Ni alloys. J. Alloys Compd. 1999, 283, 213–224. [Google Scholar] [CrossRef]
- Révész, Á.; Gajdics, M.; Schafler, E.; Calizzi, M.; Pasquini, L. Dehydrogenation-hydrogenation characteristics of nanocrystalline Mg2Ni powders compacted by high-pressure torsion. J. Alloys Compd. 2017, 702, 84–91. [Google Scholar] [CrossRef]
- Paskevicius, M.; Jepsen, L.H.; Schouwink, P.; Černý, R.; Ravnsbæk, D.B.; Filinchuk, Y.; Dornheim, M.; Besenbacher, F.; Jensen, T.R. Metal borohydrides and derivatives—Synthesis, structure and properties. Chem. Soc. Rev. 2017, 46, 1565–1634. [Google Scholar] [CrossRef] [PubMed]
- Paskevicius, M.; Ley, M.B.; Sheppard, D.A.; Jensen, T.R.; Buckley, C.E. Eutectic melting in metal borohydrides. Phys. Chem. Chem. Phys. 2013, 15, 19774. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, T.K.; Besenbacher, F.; Jensen, T.R. Nanoconfined hydrides for energy storage. Nanoscale 2011, 3, 2086. [Google Scholar] [CrossRef] [PubMed]
- Rude, L.H.; Nielsen, T.K.; Ravnsbæk, D.B.; Bösenberg, U.; Ley, M.B.; Richter, B.; Arnbjerg, L.M.; Dornheim, M.; Filinchuk, Y.; Besenbacher, F.; et al. Tailoring properties of borohydrides for hydrogen storage: A review. Phys. Status Solidi (a) 2011, 208, 1754–1773. [Google Scholar] [CrossRef]
- Dematteis, E.M.; Pinatel, E.R.; Corno, M.; Jensen, T.R.; Baricco, M. Phase diagrams of the LiBH4–NaBH4–KBH4 system. Phys. Chem. Chem. Phys. 2017, 19, 25071–25079. [Google Scholar] [CrossRef] [PubMed]
- Roedern, E.; Hansen, B.R.S.; Ley, M.B.; Jensen, T.R. Effect of Eutectic Melting, Reactive Hydride Composites and Nanoconfinement on Decomposition and Reversibility of LiBH4–KBH4. J. Phys. Chem. C 2015, 119, 25818–25825. [Google Scholar] [CrossRef]
- Liu, Y.; Reed, D.; Paterakis, C.; Contreras Vasquez, L.; Baricco, M.; Book, D. Study of the decomposition of a 0.62LiBH4–0.38NaBH4 mixture. Int. J. Hydrogen Energy 2017, 42, 22480–22488. [Google Scholar] [CrossRef]
- Javadian, P.; Sheppard, D.A.; Buckley, C.E.; Jensen, T.R. Hydrogen storage properties of nanoconfined LiBH4–NaBH4. Int. J. Hydrogen Energy 2015, 40, 14916–14924. [Google Scholar] [CrossRef]
- Liu, X.; Peaslee, D.; Sheehan, T.P.; Majzoub, E.H. Decomposition behaviour of eutectic LiBH4–Mg(BH4)2 and its confinement effects in ordered nanoporous carbon. J. Phys. Chem. C 2014, 118, 27265–27271. [Google Scholar] [CrossRef]
- Fang, Z.-Z.; Kang, X.-D.; Wang, P.; Li, H.-W.; Orimo, S.-I. Unexpected dehydrogenation behaviour of LiBH4/Mg(BH4)2 mixture associated with the in situ formation of dual-cation borohydride. J. Alloys Compd. 2010, 491, L1–L4. [Google Scholar] [CrossRef]
- Bardají, E.G.; Zhao-Karger, Z.; Boucharat, N.; Nale, A.; van Setten, M.J.; Lohstroh, W.; Röhm, E.; Catti, M.; Fichtner, M. LiBH4—Mg(BH4)2: A physical mixture of metal borohydrides as hydrogen storage material. J. Phys. Chem. C 2011, 115, 6095–6101. [Google Scholar] [CrossRef]
- Zhao-Karger, Z.; Witter, R.; Bardaji, E.G.; Wang, D.; Cossement, D.; Fichtner, M. Altered reaction pathways of eutectic LiBH4-Mg(BH4)2 by nanoconfinement. J. Mater. Chem. A 2013, 1, 3379. [Google Scholar] [CrossRef]
- Javadian, P.; Jensen, T.R. Enhanced hydrogen reversibility of nanoconfined LiBH4–Mg(BH4)2. Int. J. Hydrogen Energy 2014, 39, 9871–9876. [Google Scholar] [CrossRef]
- Lee, J.Y.; Ravnsbæk, D.B.; Lee, Y.S.; Kim, Y.; Cerenius, Y.; Shim, J.; Jensen, T.R.; Hur, N.H.; Cho, Y.W. Decomposition reactions and reversibility of the LiBH4-Ca(BH4)2 composite. J. Phys. Chem. C 2009, 113, 15080–15086. [Google Scholar] [CrossRef]
- Lee, Y.-S.; Filinchuk, Y.; Lee, H.S.; Suh, J.-Y.; Kim, J.W.; Yu, J.-S.; Cho, Y.W. On the Formation and the Structure of the First Bimetallic Borohydride Borate, LiCa 3 (BH 4)(BO 3) 2. J. Phys. Chem. C 2011, 115, 10298–10304. [Google Scholar] [CrossRef]
- Ampoumogli, A.; Charalambopoulou, G.; Javadian, P.; Richter, B.; Jensen, T.R.; Steriotis, T. Hydrogen desorption and cycling properties of composites based on mesoporous carbons and a LiBH4–Ca(BH4)2 eutectic mixture. J. Alloys Compd. 2015, 645, S480–S484. [Google Scholar] [CrossRef]
- Javadian, P.; Sheppard, D.A.; Buckley, C.E.; Jensen, T.R. Hydrogen storage properties of nanoconfined LiBH4-Ca(BH4)2. Int. J. Hydrogen Energy 2015, 11, 96–103. [Google Scholar] [CrossRef]
- Lee, H.S.; Hwang, S.-J.; Kim, H.K.; Lee, Y.-S.; Park, J.; Yu, J.-S.; Cho, Y.W. In Situ NMR Study on the Interaction between LiBH4–Ca(BH4)2 and Mesoporous Scaffolds. J. Phys. Chem. Lett. 2012, 3, 2922–2927. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, A.-L.; Li, G.; Matsuo, M.; Orimo, S.; Deledda, S.; Sørby, M.H.; Hauback, B.C.; Pistidda, C.; Klassen, T.; Dornheim, M. Simultaneous desorption behaviour of M borohydrides and Mg2FeH6 reactive hydride composites (M = Mg, then Li, Na, K, Ca). Appl. Phys. Lett. 2015, 107, 073905. [Google Scholar] [CrossRef]
- Vajo, J.J.; Li, W.; Liu, P. Thermodynamic and kinetic destabilization in LiBH4/Mg2NiH4: Promise for borohydride-based hydrogen storage. Chem. Commun. 2010, 46, 6687–6689. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Vajo, J.J.; Cumberland, R.W.; Liu, P.; Hwang, S.-J.; Kim, C.; Bowman, R.C. Hydrogenation of magnesium nickel boride for reversible hydrogen storage. J. Phys. Chem. Lett. 2010, 1, 69–72. [Google Scholar] [CrossRef]
- Afonso, G.; Bonakdarpour, A.; Wilkinson, D.P. Hydrogen storage properties of the destabilized 4NaBH4/5Mg2NiH4 composite system. J. Phys. Chem. C 2013, 117, 21105–21111. [Google Scholar] [CrossRef]
- Bergemann, N.; Pistidda, C.; Milanese, C.; Emmler, T.; Karimi, F.; Chaudhary, A.-L.; Chierotti, M.R.; Klassen, T.; Dornheim, M. Ca(BH4)2–Mg2NiH4: On the pathway to a Ca(BH4)2 system with a reversible hydrogen cycle. Chem. Commun. 2016, 52, 4836–4839. [Google Scholar] [CrossRef] [PubMed]
- Javadian, P.; Zlotea, C.; Ghimbeu, C.M.; Latroche, M.; Jensen, T.R. Hydrogen storage properties of nanoconfined LiBH4–Mg2NiH4 Reactive Hydride Composites. J. Phys. Chem. C 2015, 119, 5819–5826. [Google Scholar] [CrossRef]
- Ley, M.B.; Roedern, E.; Jensen, T.R. Eutectic melting of LiBH4–KBH4. Phys. Chem. Chem. Phys. 2014, 16, 24194–24199. [Google Scholar] [CrossRef] [PubMed]
- Blomqvist, H.; Noréus, D. Mechanically reversible conductor–insulator transition in Mg2NiH4. J. Appl. Phys. 2002, 91, 5141–5148. [Google Scholar] [CrossRef]
- Polanski, M.; Nielsen, T.K.; Kunce, I.; Norek, M.; Płociński, T.; Jaroszewicz, L.R.; Gundlach, C.; Jensen, T.R.; Bystrzycki, J. Mg2NiH4 synthesis and decomposition reactions. Int. J. Hydrogen Energy 2013, 38, 4003–4010. [Google Scholar] [CrossRef]
- El Kharbachi, A.; Pinatel, E.R.; Nuta, I.; Baricco, M. A thermodynamic assessment of LiBH4. Calphad 2012, 39, 80–90. [Google Scholar] [CrossRef]
- Dematteis, E.M.; Roedern, E.; Pinatel, E.R.; Corno, M.; Jensen, T.R.; Baricco, M. A thermodynamic investigation of the LiBH4–NaBH4 system. RSC Adv. 2016, 6, 60101–60108. [Google Scholar] [CrossRef]
- Javadian, P.; GharibDoust, S.P.; Li, H.-W.; Sheppard, D.A.; Buckley, C.E.; Jensen, T.R. Reversibility of LiBH4 facilitated by the LiBH4–Ca(BH4)2 eutectic. J. Phys. Chem. C 2017, 121, 18439–18449. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).