Abstract
The common alkaloid theophylline (Tph) is known to exist in five polymorphic forms. The structures of its 8-halo analogues were previously unreported. Here, we report three polymorphs for 8-chlorotheophylline (8-Cl-Tph) and an additional distinct one for 8-bromotheophylline (8-Br-Tph). While polymorphs for theophylline are dominated by NH---N inter-molecular hydrogen bonds, the halo compounds exclusively exhibit NH---O interactions. 8-Cl-Tph has two related structures with chains N(7)-H(7)---O(2); one is also the stable form for 8-Br-Tph. Polymorphs with a dimeric R22(10) ring structure using N(7)-H---O(6) H-bonds exist for all three compounds, though each with distinct 3D packing. DFT calculations indicate that the alkaloid ring nitrogen N(9) is a weaker base in 8-halo compounds, disfavoring NH---N interactions in their polymorphic forms.
1. Introduction
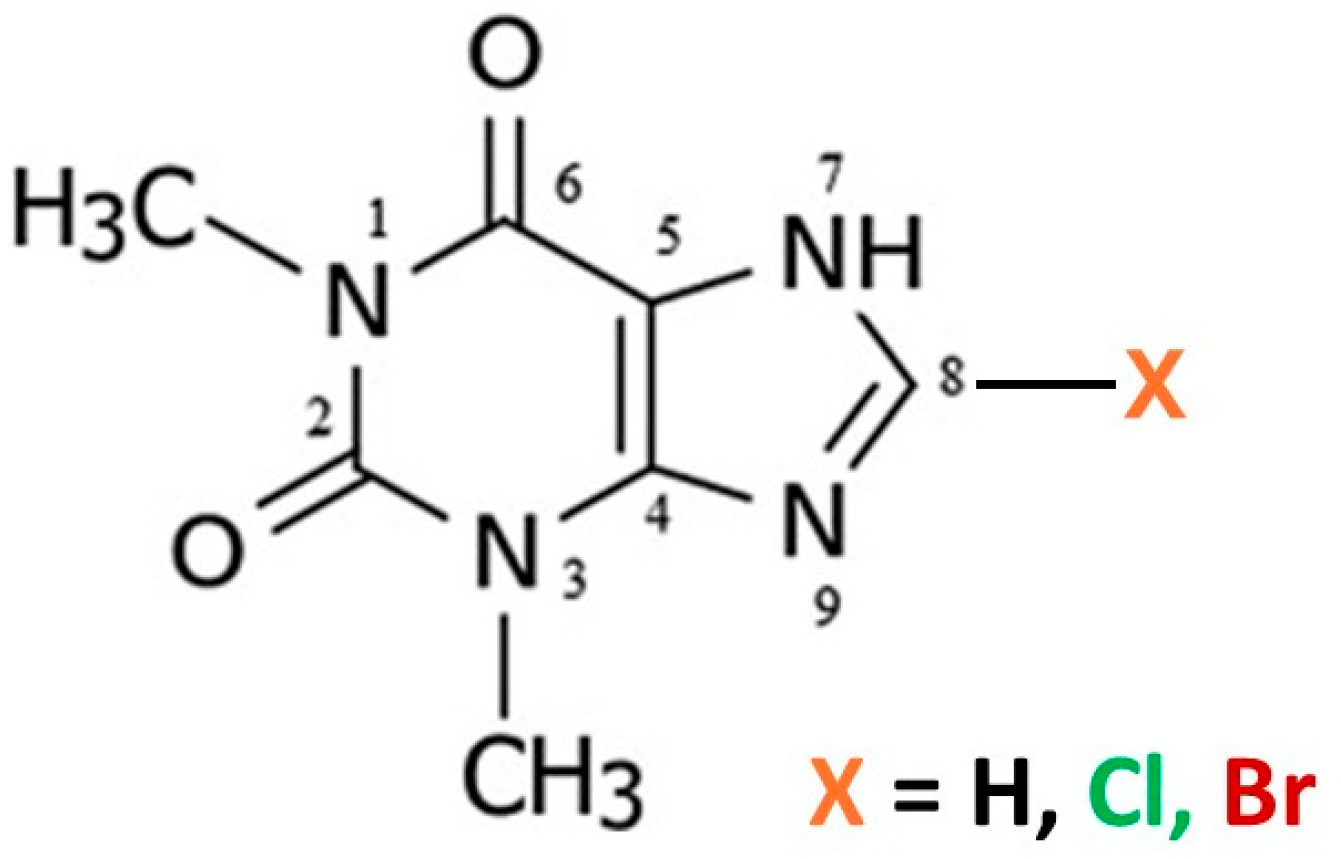
The crystal structures of many common xanthine alkaloids with important pharmacological properties have been extensively studied both experimentally and computationally [1,2,3,4,5]. In addition to caffeine (Caf), which is 1,3,7-trimethylxanthine, the related alkaloid theophylline (Tph), which is 1,3-dimethylxanthine (Figure 1), has also been the subject of considerable previous study.
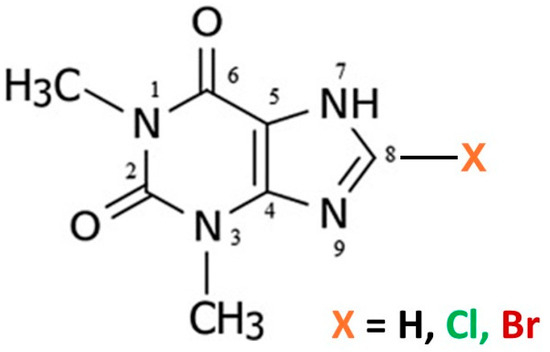
Figure 1.
Molecular scheme for theophylline alkaloids.
To date, five distinct polymorphic forms have been identified [6,7,8,9,10,11,12,13] along with a monohydrate, which is not readily suppressed under ambient crystallization conditions [9,12]. Their structural summary is given in Table 1. Form I [8,9] is the thermodynamically stable form above 232 °C, with the related Form II more stable at ambient temperature [6,9].

Table 1.
Summary of crystal data of theophylline polymorphs.
Form III is clearly metastable with a relatively poor packing efficiency, and its formation and structure were established by Madsen after desolvation of the common monohydrate form [10]. Form IV [7,8], which has the smallest specific molecular volume Vmol of the various polymorphs, has been regarded as the most thermodynamically favored form isolated to date [12]. Forms II and V and the monohydrate were obtained by us, and structures were redetermined at 100 K and are reported in Table 1 for completeness.
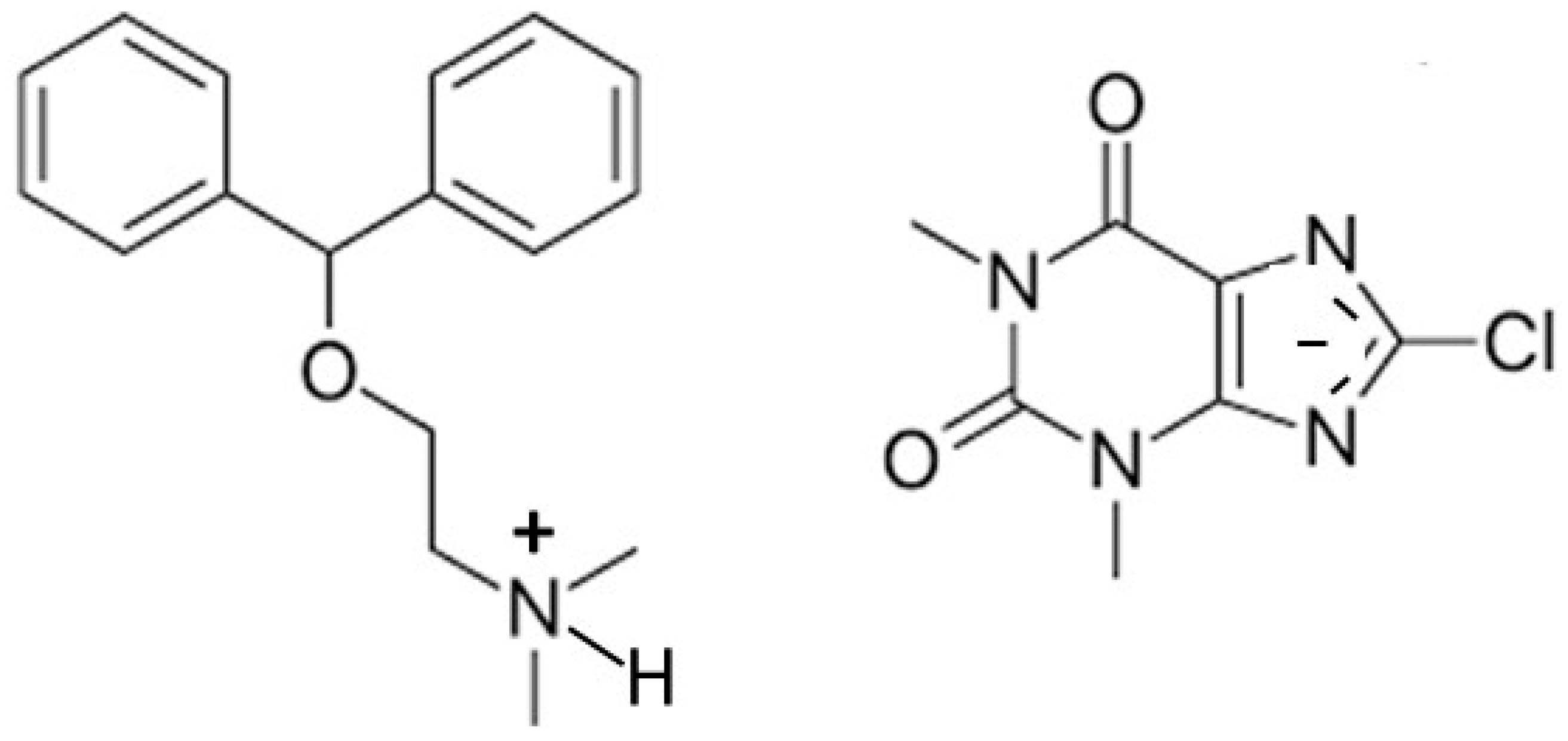
Two simple 8-halo-substituted theophyllines, 8-chlorotheophylline (8-Cl-Tph) and 8-bromotheophylline (8-Br-Tph), are commercially available, but neither had their crystal structures previously reported in the Cambridge Structural Database. This was rather surprising in the case of 8-Cl-Tph, since it is a well-known component of Dramamine (dimenhydrinate), in which it serves as the anion in a 1:1 salt with diphenhydramine and for which the crystal structure was determined (Figure 2) [14].
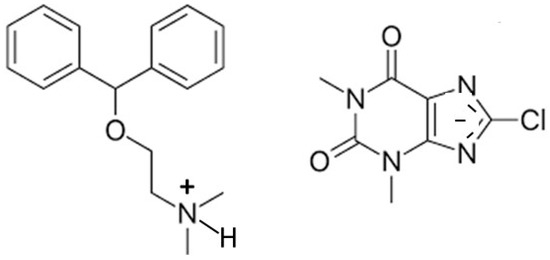
Figure 2.
Molecular scheme for dimenhydrinate salt containing 8-Cl-Tph anion.
The study of molecular analogues as potential templating seeds to provide access to new polymorphs has been explored by Price and co-workers [15,16]. In this system, we wished to discover whether the halo analogues would provide any new forms and if so whether these might induce a new polymorphic form of theophylline to be uncovered.
Through the use of differing crystallization conditions, we have uncovered three polymorphic forms of 8-Cl-Tph and two for 8-Br-Tph, all of which we report here. Only one pair of these structures is isostructural, and they are also distinct from the five previously reported Tph polymorphs [6,7,8,9,10,11,12,13]. Hence, a total of nine distinct packing arrangements have now been uncovered for these simple theophylline alkaloid molecules. This marked effect on polymorph type arising from a single halogen substitution is presented and rationalized here.
2. Materials and Methods
The theophylline alkaloids and solvents used were of reagent grade supplied by Meryer Chemical (Waters, Milford, MA, USA) with the following details: theophylline, 99% CAS 58-55-9, C7H8N4O2, Mw: 180.17; 8-chlorotheophylline >98%, CAS 85-18-7, C7H7ClN4O2, Mw: 214.61; 8-bromotheophylline 97%, CAS 10381-75-6, C7H7BrN4O2, Mw: 259.06. Attempts to form solid-solution phases of mixed 8-X-Tph (X = H, Br, Cl) were carried out by liquid-assisted grinding (LAG) [17] using a Tencan XQM-0.4A mini-planetary ball mill with zirconia vessels and media (Tencan, Changsha, China). A minimal amount of methanol (η factor = 0.2 mL/g) was used to accelerate the solid-state transformations, which took between 30 min and 2 hrs [18].
Crystal polymorphs were obtained by heating/cooling alcoholic or aprotic solutions of the compounds at varying temperatures (in some cases with flavonoid molecules present) or hydrates via solvent evaporation at room temperature. Attempts to form solid-solution single crystals of 8-X-Tph (X = Br, Cl) were made through common dissolution/reprecipitation from 50:50 mixtures from MeOH. The crystal size ranged from 80 to 250 microns.
Differential scanning calorimetry was conducted from ambient to 350 °C under a nitrogen atmosphere on a Universal V4.5A TA Instrument (Waters Corp., Milford, MA, USA). A heating rate of 10 °C per minute was employed using roughly 3 mg of sample.
2.1. X-Ray Crystallography
Powder X-ray diffraction data were obtained on crystalline powders at room temperature using Cu-Kα radiation by a PanAlytical X’Pert PRO diffractometer with a 1D X’celerator detector (Malvern Panalytical Ltd., Malvern, UK) or on a PanAlytical Aeris benchtop powder X-ray diffractometer and measured in the 2θ range 5° to 40° with a step size of 0.02°. Single crystal X-ray structure determinations of the various polymorphs and solid solutions of Tph, 8-Cl-Tph, and 8-Br-Tph were generally carried out at 100 K on a Rigaku Oxford Diffraction Supernova (Rigaku, Tokyo, Japan) operating with a micro-focus Cu-Kα source and Atlas detector. Some Br-containing phases have been reported using Mo-Kα, or from a Bruker Metal-jet with a Ga-Kα source (Billerica, MA, USA), where these gave superior results. The intensity data were processed using proprietary a CrysAlisPro or Apex5 software package, with multi-scan absorption corrections (Abspack, SADABS). Crystal structures were solved (charge-flipping algorithm) and refined using embedded SHELX programs [19,20] or internal options from within the Olex2 suite [21,22].
Structures were refined successfully, showing R-values typically between 2% and 4%. Bond length standard deviations were acceptably low (0.002–0.003 Å), and residual electron density was low except for absorption features associated with Br atoms, which were kept below 1.0e Å−3. No disorder of non-hydrogen atoms appeared present in these phases, except in the case of a 8-Cl/Br-Tph solid solution. Organic C-H hydrogens were placed geometrically with riding constraints and ADPs derived from the C atoms to which they were attached. All -CH and CH2 groups had H-Uiso fixed at 1.2 times the C atom. Methyls were idealized as freely rotating CH3 groups with H-Uiso fixed at 1.5 times that of the C atom. In some cases, these were split into two rotationally staggered orientations. Hydrogens on the heteroatoms NH and OH (theophylline hydrate) were refined as independent isotropic atoms.
2.2. Computational Methods
Density Functional Theory (DFT) calculations carried out on theophylline molecules (and derivatives) were performed using the Gaussian 09 program [23]. Molecular geometries were first optimized from starting coordinates from a polymorph of each molecule. The ωB97X-D functional [24] was chosen for the DFT calculations. The def2-TZVP basis set [25] (together with the built-in effective core potentials when present) was used to describe all the atoms in the calculations. The partial charges are estimated with two different schemes: (i) natural charges and the Wiberg bond index [26] in the natural atomic orbital basis are calculated using the Natural Bond Orbital (NBO) method [27] embedded in Gaussian 09, and (ii) the electrostatic potential-derived charges are calculated according to the Merz–Singh–Kollman [28,29] scheme in Gaussian 09. Full tables for crystallographic structures and computational results are listed in Supplementary Information.
3. Results and Discussion
3.1. Polymorphic Forms of Theophylline
In our own work, we have tried the crystallization of theophylline from ambient solvent evaporation methods, which has typically afforded the monohydrate or a mixture of phase types. The use of higher-temperature crystallizations is helpful in suppressing the formation of solvate and hydrated phases, and at 80 °C from ethanol–water, Form II is isolated. The use of even a higher temperature at 140 °C (solvothermal) from methanol–water affords a more recently isolated Form V [13]. Our structure redetermination of this indicates that this phase appears energetically competitive, with a specific molar volume of 195.5 Å3, which is essentially identical to our Form II. Only the 100 K structure from Form IV clearly gives a smaller specific molecular volume for Tph.
Descriptions of the theophylline polymorphic forms have been reported elsewhere, but it is instructive to consider here the three phase types we have isolated for a later comparison with the 8-halo analogues. The Form II anhydrous theophylline [6,9] is readily crystallized using a slightly higher temperature of crystallization, such as from hot aqueous ethanol. It consists of zigzag linear chains of a theophylline molecule screw related along the c-axis and connected via N(7)-H(7)---N(9) hydrogen bonds (N---N = 2.782(3) Å) (Figure 3, left).
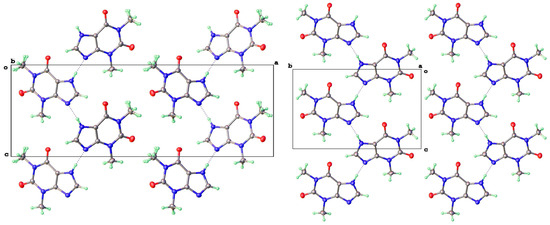
Figure 3.
Molecular packing, showing unit cell axes, for theophylline Forms II (left) and V (right) both viewed along a short b-axis.
Crystals of Form V Tph were formed using higher-temperature solvothermal crystal growth from methanol (140 °C). This is somewhat related to the use of DMF by Dvulgerov et al. in an attempted crystallization of a theophylline containing metal organic framework material [13]. The molecular packing diagram for Form V is also shown in Figure 3. The projections along the short b-axes look extremely similar for Forms II and V, and their packing densities are essentially identical. The hydrogen bond geometry in Form V is once again based on N(7)-H(7)---N(9), albeit with a slightly longer N---N separation of 2.820(3) Å.
The key difference between these two forms is illustrated in Figure 4, which shows that the hydrogen bond strands for Form II have a zigzag arrangement of molecular planes, while in Form V, the theophylline molecules in each strand lie in a coplanar manner.
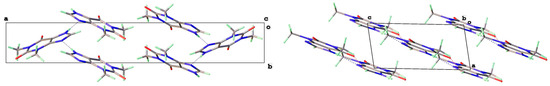
Figure 4.
Comparison of zig-zag and parallel hydrogen bonded strands for Forms II (left) and V (right) theophylline.
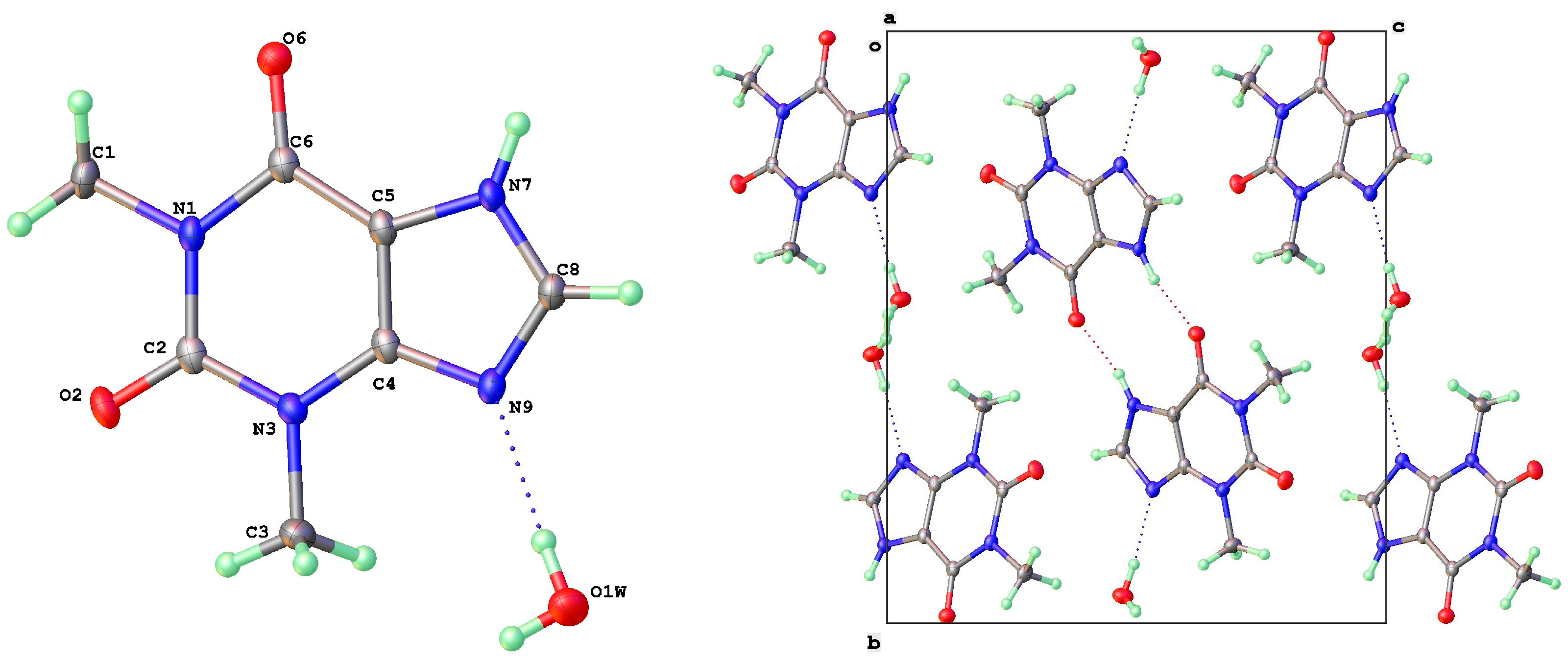
Theophylline monohydrate (Figure 5) is readily formed under ambient growth conditions if water is not carefully excluded from the system. The structure is monoclinic P21/n with 1-D water channels showing a statistical disorder of hydrogen atoms, as previously reported [9]. The water molecules hydrogen bond to themselves up and down the channel stack, as well as to the alkaloid base nitrogen N(9). N(7)-H(7) is the donor in a hydrogen bond to a neighbor O(6) keto oxygen (2.754 Å) such that the two theophyllines form a dimer R22(10) in an Etter graph set notation [30]. This dimer motif is anticipated as the most likely molecular aggregation in concentrated aqueous solutions of theophylline and has been examined computationally [31].
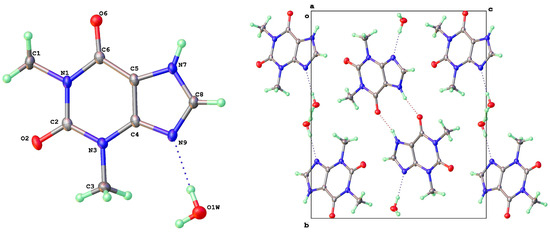
Figure 5.
Molecular structure (left) and packing (right) for theophylline. H2O (viewed along a-axis).
3.2. Polymorphic Forms of 8-Halotheophylline
The lack of reported polymorphic forms for the substituted alkaloids 8-chloro and 8-bromotheophylline prompted us to investigate their crystal structures. Given the rich diversity of theophylline structures discussed here, along with the common isolation of theophylline monohydrate, we anticipated that the halo substitutions would likely alter the existing polymorphic energies and potentially alter the stability ranking of these solid structures, or even give rise to new ones. A summary of crystal data from X-ray structure determinations for the 8-halotheophylline polymorphs isolated is given in Table 2 quickly reveals that there are no common structural phases to Tph itself. Further details of the structures for 8-Cl-Tph are also provided in Table 3.

Table 2.
Summary of crystal data of the 8-halotheophylline polymorphs at 100 K.

Table 3.
Structure determination summaries for three polymorphic forms of 8-Cl-Tph.
The material of 8-chlorotheophylline, as received, was a fine pale powder, but this gave a reasonable powder X-ray diffraction pattern with sharp peaks (vide infra Section 3.5) but with no resemblance to those from various theophylline polymorphs or its monohydrate. Thermal gravimetric analysis (TGA) indicated that 8-Cl-Tph was in an anhydrous form since there was no appreciable weight loss up to 150 °C and we have named this Form I. Recrystallization from aqueous methanol at 140 °C afforded acceptable single crystals of this Form I 8-Cl-Tph (Figure 6a). These crystals belong to the monoclinic system, whereas crystallization from various aprotic solvents gave triclinic Form II (Figure 6b). A third form 8-Cl-Tph Form III involving molecular dimers (Figure 6c) with a platy rhombus crystal habit was isolated from an unsuccessful attempt at the cocrystallization of 8-Cl-Tph with the flavonoid rutin, but has not yet been prepared in a phase-pure manner by a more rational methodology (Table 3).

Figure 6.
Crystal habits of 8-halotheophyllines: (a) 8-Cl-Tph Form I, (b) 8-Cl-Tph Form II, (c) 8-Cl-Tph Form III, (d) 8-Br-Tph Form I, (e) 8-Br-Tph Form IV, & (f) 8-Cl/8-Br-Tph solid solution Form I (Black lines are scale bars = 100 μm).
8-Bromotheophylline was also received as a pale tan powder, which gave a crude X-ray diffraction pattern, extremely similar to that of 8-Cl-Tph. Its recrystallization from hot methanol afforded smaller crystalline bars (Figure 6d). Despite the apparent difference in aspect ratios, these were later determined to be isostructural with Form I 8-Cl-Tph. In this case, the use of several aprotic organic solvents did not change the form of 8-Br-Tph produced, although a second polymorphic phase type was eventually isolated from an attempted cocrystallization of 8-Br-Tph with the flavonoid chrysin. This, which we have named Type IV, has a rectangular platy habit (Figure 6e).
Attempts at forming solid solution phases between different 8-X-Tph (X = H, Cl, Br) met with limited success, although unsurprisingly, the isostructurality of Form I 8-Cl-Tph and 8-Br-Tph afforded mixed solid solution crystals in this case (Figure 6f).
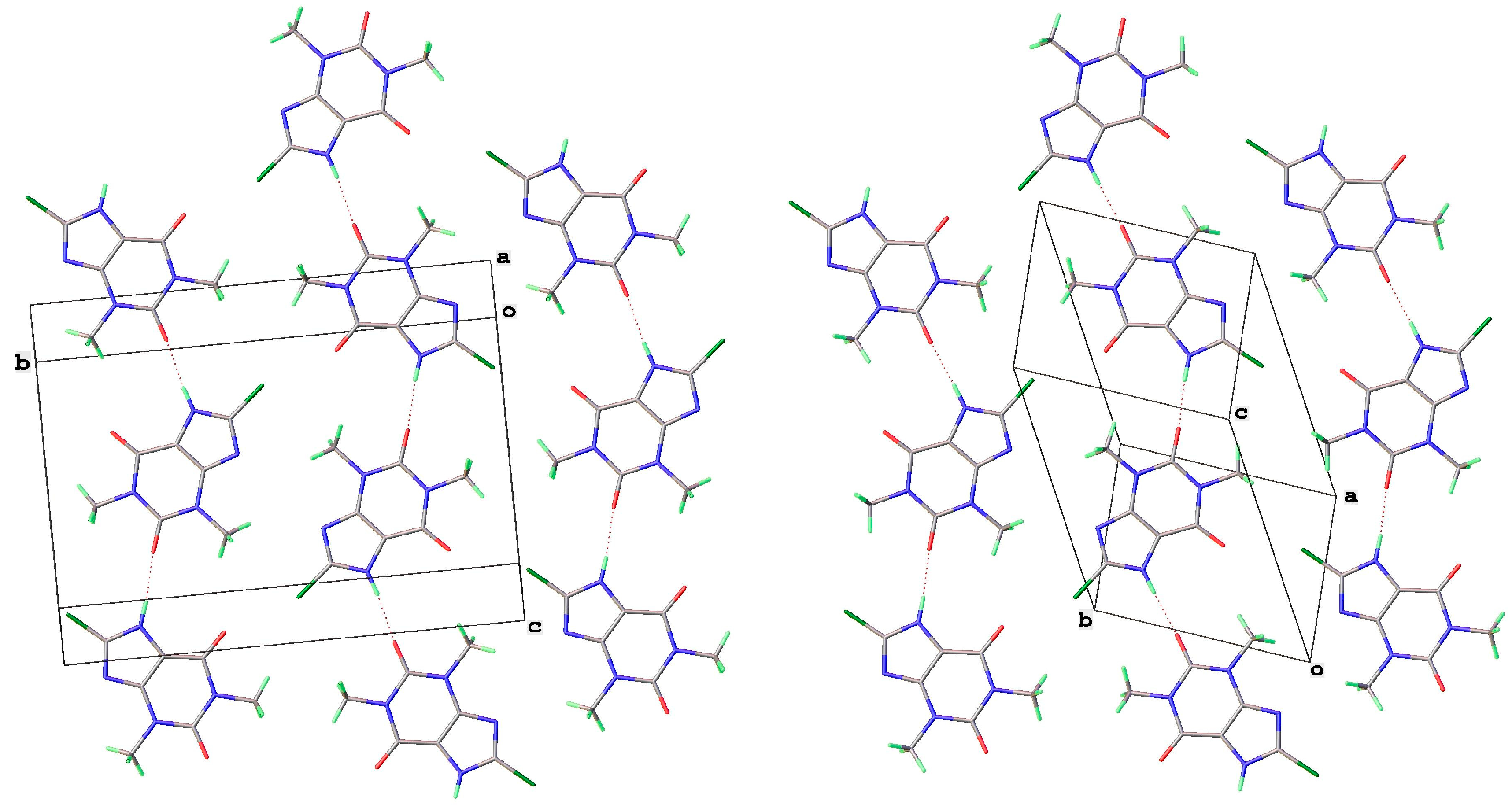
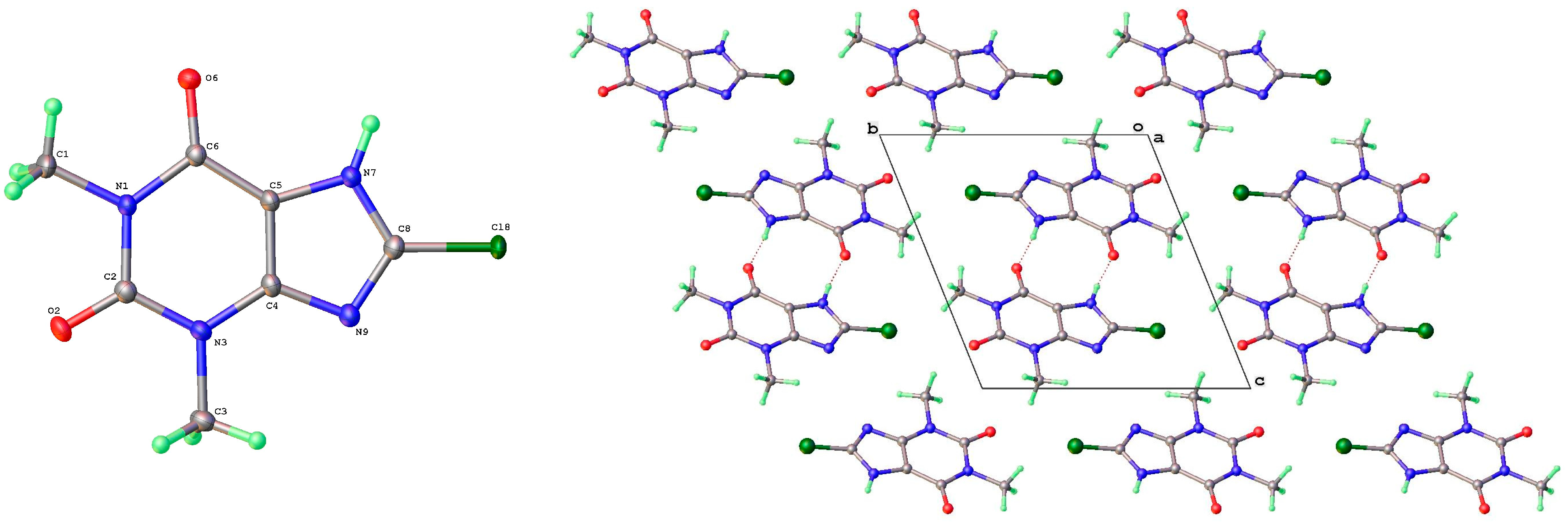
Both 8-Cl-Tph and 8-Br-Tph afford the common Form I (Figure 7, Figure 8 and Figure 9), which is isostructural in the two cases, with a slight cell expansion from 840 to 860 Å3, which is just around 5 Å3 per molecule. Unlike the polymorphs of theophylline, the protonated alkaloid nitrogen N(7)-H is now a hydrogen bond donor with keto O(2) of a neighboring molecule to form 1D H-bonded chains, which are distinct from the N(7)H---N(9) type found for most of the Tph polymorphs. Table 4 gives a list of key H-bond interactions in the 8-X-Tph polymorphs. Form II for 8-Cl-Tph that can be isolated by crystallization from various aprotic solvents is a structural variant of Form I. The asymmetric unit now includes two molecules, so the two forms differ considerably in configurational entropy [32]. In both Forms I and II of 8-X-Tph, essentially isometric 2D sheets of the 1D NH---O hydrogen-bonded strands are found in the two cases, as shown in Figure 8. However, a quite different stacking of these layers is found for the two forms, illustrated in Figure 9. The strongest interactions between adjacent layers are C-H---O in nature between methyl hydrogen atoms and keto oxygens. Since there are two chemically distinct CH3 and two keto groups per molecule, this gives a scope for several such arrangements of a very similar overall energy, as seems to be the case for Forms I and II.
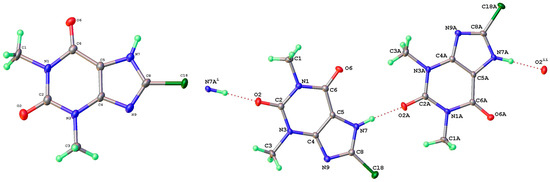
Figure 7.
Molecular structure and labelling scheme for 8-chlorotheophylline Forms I (left) and II (right).
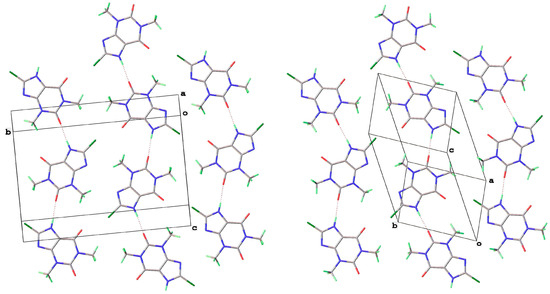
Figure 8.
Isostructurality of 2D layers in 8-Cl-Tph Forms I (left) and II (right) showing relationship to unit cells.
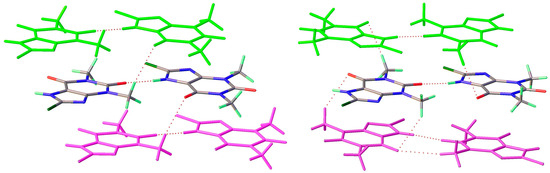
Figure 9.
Differentiation of 2D layer stacking in 8-Cl-Tph Forms I (left) and II (right).

Table 4.
Hydrogen bonds in 8-halotheophyllines.
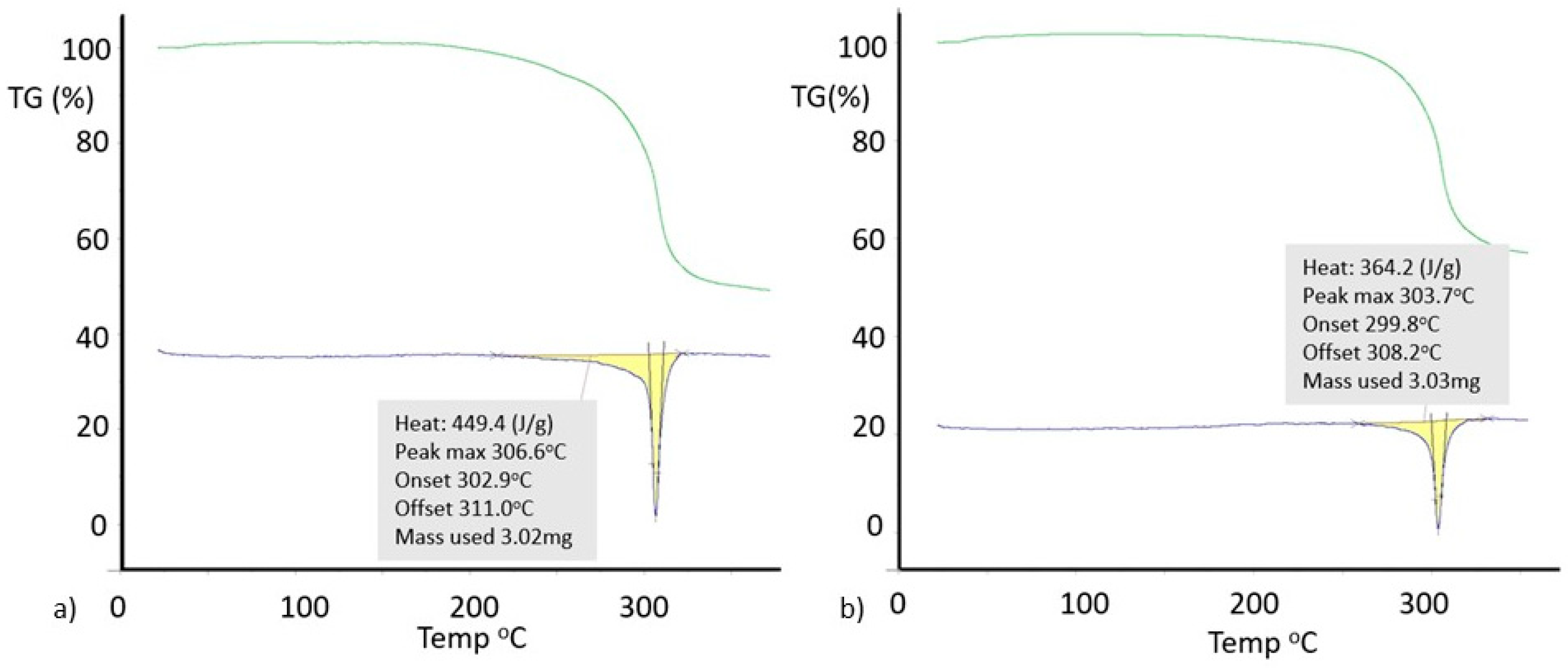
Differential scanning calorimetry (DSC) (Figure 10) gives a higher melting point (Tm = 306 °C versus 304 °C) and heat of fusion (HF = 449 Jg−1 versus 364 Jg−1) for Form I 8-Cl-Tph compared with Form II. Given the otherwise high similarity of the overall packing efficiency (Vmol are essentially equal) and geometric near equivalence of NH---O hydrogen bonds (Table 4) and the doubled asymmetric unit found for Form II, these two phases may have an enantiotropic stability relationship, with Form II becoming increasingly more stable relative to Form I at lower temperatures. This hypothesis has not yet been proven and needs further investigation.
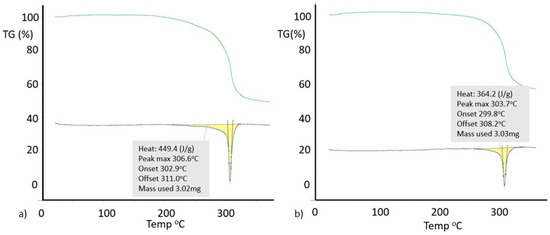
Figure 10.
Differential scanning calorimetry for 8-chlorotheophylline: (a) Form I; (b) Form II.
The related 8-Br-Tph (Table 5) also crystallizes with the monoclinic Form I structure; however, the second-chain Form II type has not yet been observed for 8-Br-Tph. An extensive search for polymorphic forms of this compound has not yet been conducted, so it may well exist. It should be noted that a slight expansion of the 2D sheet due to the Br incorporation would lead to slight energy changes in the CH3---keto O interactions between layers, such that Form II is disfavored in the 8-Br-Tph case.

Table 5.
Structure determination summaries for 8-Br-Tph and 8-Cl/Br-Tph (solid solution).
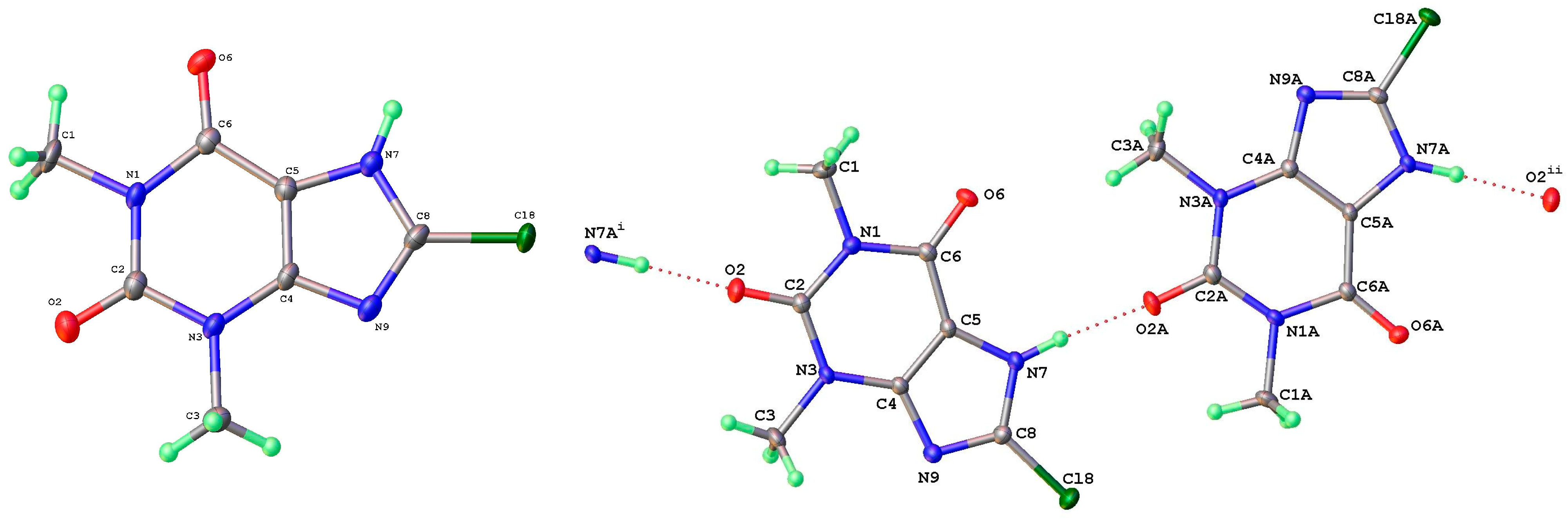
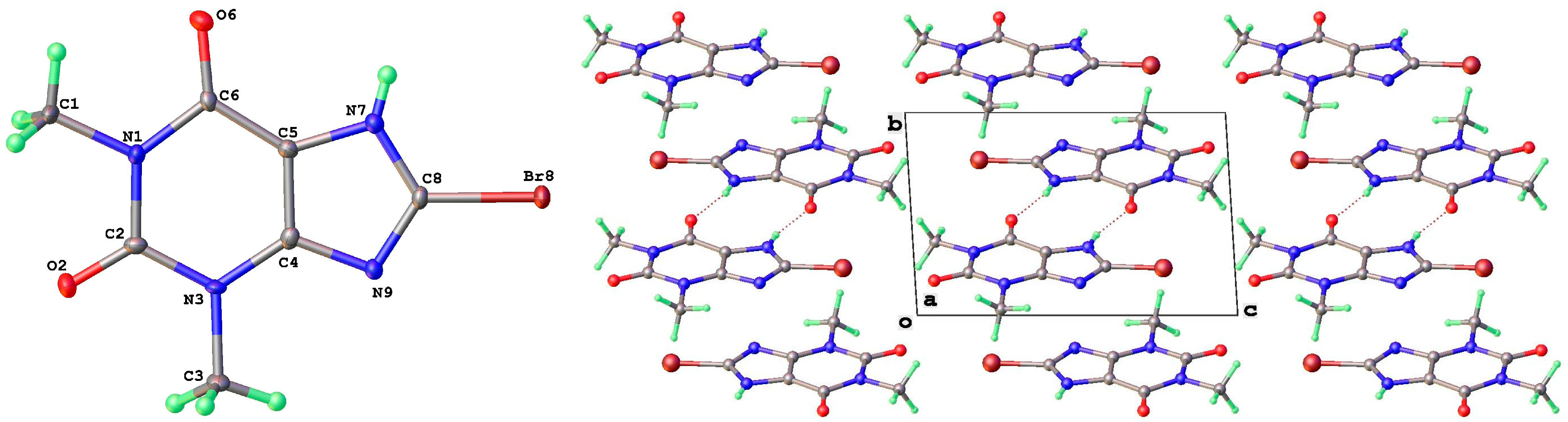
As mentioned above, a third polymorph of 8-Cl-Tph, Form III, was also isolated from a cocrystallization attempt with the flavonoid rutin. Like theophylline hydrate [9,12] and the meta-stable Form III of anhydrous Tph [10], this has a dimeric structure. This is formed by the pairing of Tph molecules via two N(7)-H(7)---O(6) hydrogen bonds, forming a R22(10) ring (Figure 11). Interestingly though, unlike the dimer polymorph for Tph, the specific volume in this 8-Cl-Tph polymorph is the smallest found for the three polymorphs. The opposite was true for Tph, which only gives Form III upon the desolvation of the open monohydrate phase.
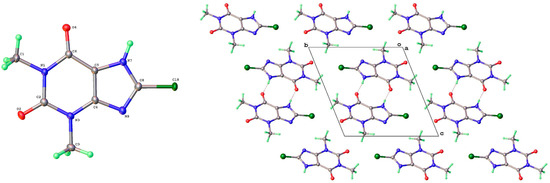
Figure 11.
Molecular structure (left) and packing (right) for 8-chlorotheophylline dimer Form III (viewed along the unit cell a-axis).
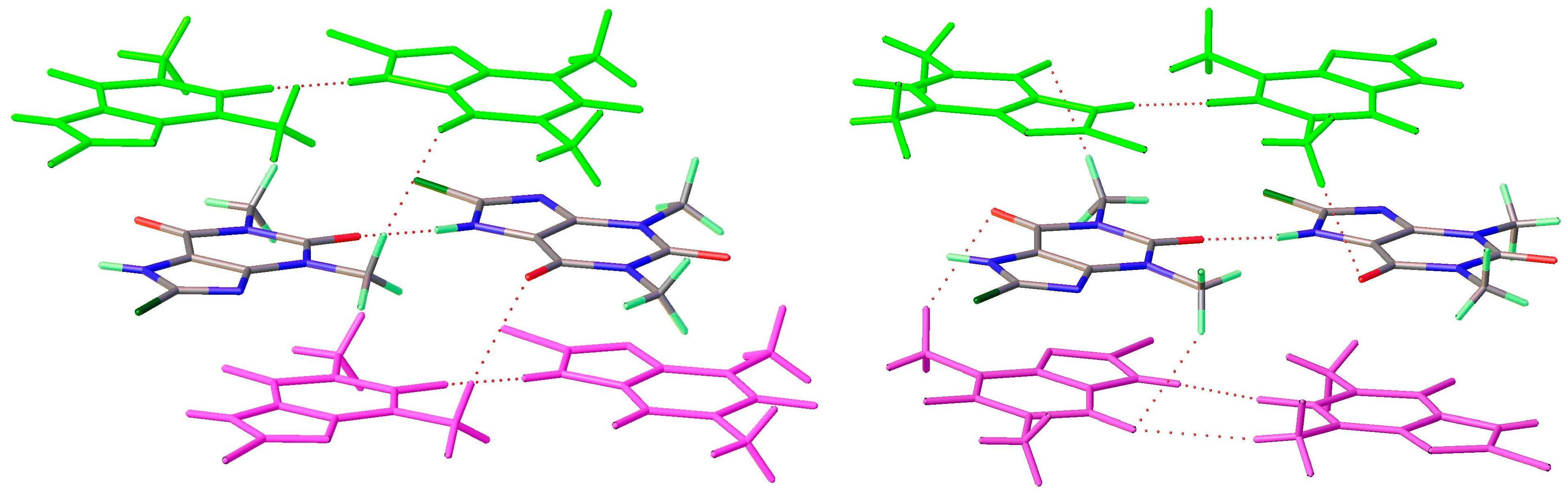
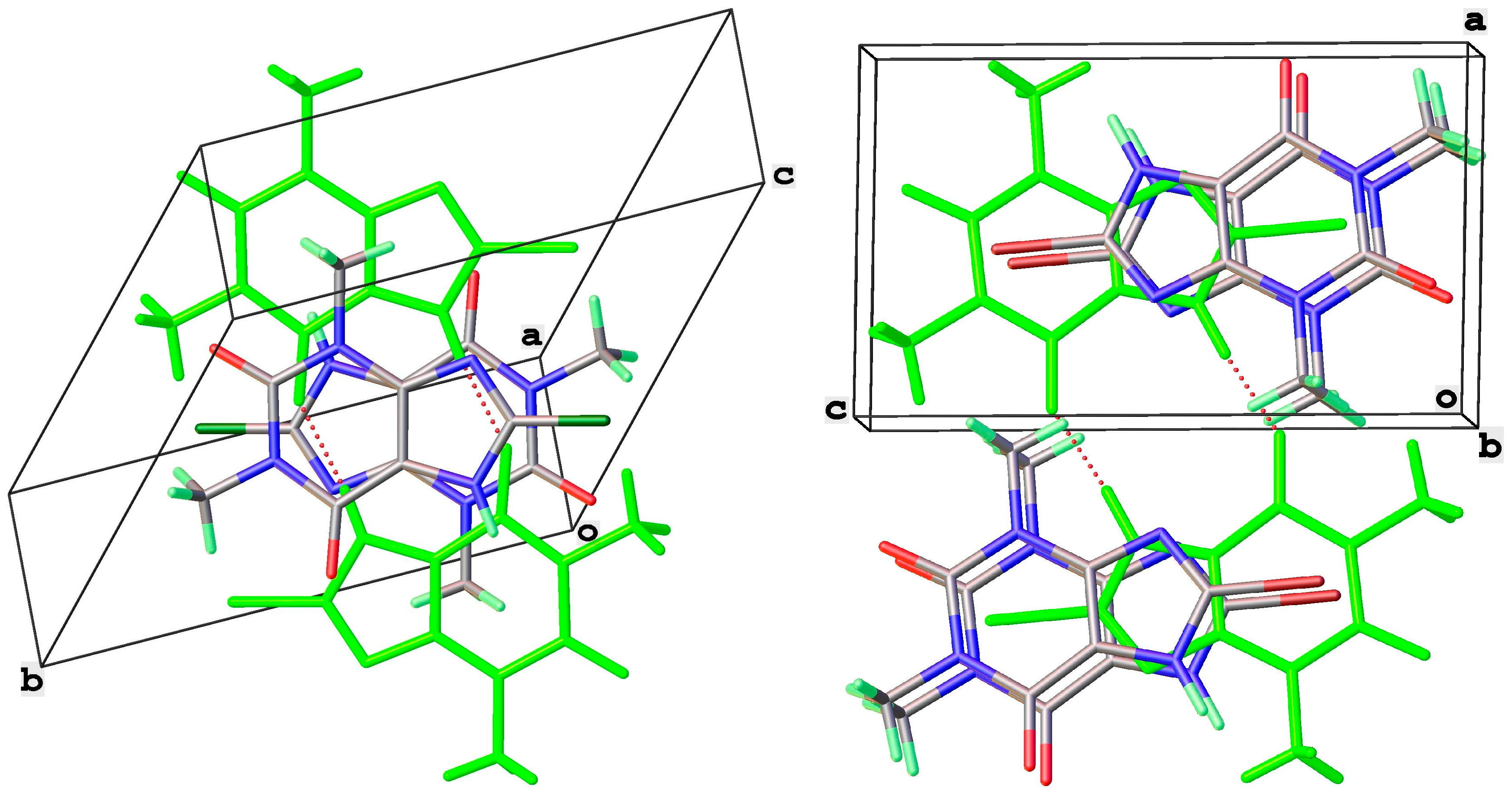
8-Br-Tph has also been isolated in a dimer polymorph (Table 5, Figure 12), this time from an attempted cocrystallization with chrysin (we will call this structural arrangement Form IV, since analog structures for 8-Br-Tph of the 8-Cl-Tph Forms II and III may yet be found). The 8-X-Tph Forms III and IV are similar in that the molecular dimers pack in coplanar layers that are metrically similar in the two cases, seen by the comparison of Figure 11 (right) and Figure 12 (right). Both forms are triclinic, but the unit cells are quite distinct with Form III for 8-Cl-Tph having cell dimensions of approximately 4.5 × 10.1 × 10.2 Å, whereas the 8-Br-Tph Form IV is 6.5 × 6.9 × 10.3 Å. Once again, the main difference is in the stacking of these 2D layers, which is quite different. This is shown in Figure 13 by illustrating the environments above and below a molecular dimer (shown in green). In the 8-Cl-case (Form III), the green dimer pair in a central layer has 8-Cl-Tph in an anti-parallel manner above/below the ring junction, an ABCD-type stacking, while in an 8-Br case (Form IV), a similar green dimer pair has molecules in register above and below that are rotated by 180° with respect to the central pair, creating a simpler ABAB-type layer stacking repeat.
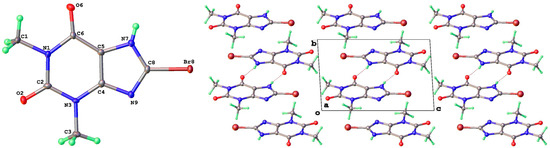
Figure 12.
Molecular structure (left) and packing (right) for 8-bromotheophylline dimer Form IV (viewed along the unit cell a-axis).
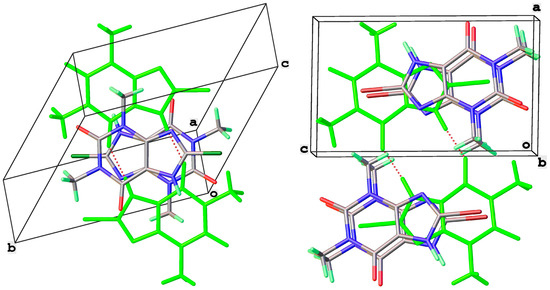
Figure 13.
Differential layer overlap for 8-Cl-Tph (Form III) (left) and 8-Br-Tph (Form IV) (right) dimer structures.
In both of the 8-Br-Tph polymorphs, weak Br---O halogen bond interactions can be seen, (Table 6) and it is notable that, in Form I, the related Cl---O separation is actually significantly longer, despite the overall 2D sheet contraction and the smaller atomic radius of chlorine.

Table 6.
Halogen–oxygen interactions in 8-halotheophylline polymorphs.
3.3. Computational Studies of 8-X-Theophyllines (X = H, Cl, Br)
Some rationalization of both this structural diversity and divergence between Tph and 8-X-Tph (X = Cl, Br) polymorphs has been sought from molecular electronic structure calculations using Density Functional Theory. The main point is that while theophylline polymorphs are dominated by N(7)-H(7)-N(9) hydrogen bonds, in the case of the 8-halo analogues, N(7)-H(7)---O hydrogen bonds are exclusively found. In general, these are to the keto oxygen O(2) leading to chain structures, which are thus distinct from Tph polymorph cases. The introduction of an 8-X substituent might be expected to modify the acidity/basicity of the nearby N(7)-H(7) and N(9) functionalities. In the well-known case of pyridine, compared with 2-chloro and 2-bromopyridines, the pKa of the respective pyridinium cations (or pKHB) are pyH = 5.17, 2-Cl-pyH = 0.72, and 2-Br-pyH = 0.90 [33]. This indicates that the halogen effect would render adjacent NH more acidic, but adjacent N less basic. This effect would be stronger for the chloro-substituent, but similar in both cases.
Density Functional Theory (DFT) calculations have been carried out on theophylline before by others [31,34,35]. This has allowed them to identify infrared vibrational modes and confirmed the expectation that, in concentrated aqueous solution, the dimer form will be a stable molecular aggregate [31]. Other calculations on the 8-Cl-Tph anion in dimenhydrinate have also been run previously to help confirm the salt, rather than the neutral cocrystal, formulation for this solid phase of Dramamine [14]. We undertook DFT calculations on Tph and 8-Cl-Tph and 8-Br-Tph to confirm the assumption that the halo-substituted molecule will have a substantial weakening of N(9) basicity, but less effect on keto oxygen O(2). The results in Table 7 provide an analysis of the partial charges on substituent atoms, calculated by both the Natural Bond Orbital (NBO) [27] and ESP [28,29] methods embedded in Gaussian09 software [23].

Table 7.
Atomic charges in theophylline molecules from DFT.
The NBO results show relatively small differences between the three molecules, and the ESP approach is better suited to for understanding likely electrostatic interactions between molecules, since it specifically models the electro-static potential of the molecular surface and how molecules would interact on that basis [36]. In this case, charges on O(2) oxygen and N(9) nitrogen can be seen to swap over so that, in Tph, N(9) is −0.616 and O(2) −0.532, helping to favor NH---N hydrogen bond formation, whereas in 8-Cl-Tph, N(9) is −0.516 and O(2) is −0.545, leading to the favorability of NH---O polymorphic forms. 8-Br-Tph is intermediate, but more similar to the chloro compound. The acidity of the NH functionality is not so clearly demonstrated in the charge on H(7) itself, or the computed bond distances, which are all 1.00 Å. The Wiberg bond order for N-H is 0.79 for Tph, slightly greater than in the 8-X-Tph cases (0.78). This difference is energetically significant and worth several kcal/mol−1.
3.4. Dimer Structures of 8-X-Theophyllines (X = H, Cl, Br)
The main difference between Tph and 8-X-Tph (X = Br, Cl) polymorphs involves the switch from N(7)H---N(9) to N(7)H---O(6) hydrogen bonding in the case of chain structures. However, each molecule also has an example of a dimer structure involving R22(10) rings and N(7)-H(7)---O(6) hydrogen bonds, but none of these are isostructural with each other. In the 8-Cl-Tph and 8-Br-Tph cases, the Br atoms lie above and below the alkaloid six-membered ring, whereas the Cl atoms interact with methyl groups lying outside the xanthine rings. This may reflect an electronic rather than steric preference. Subtle differences in a halogen environment exist in the 8-X-Tph polymorphs, and as mentioned earlier, shorter Br---O contacts than Cl---O contacts are found even in isostructural Form I (Table 6). We have observed that stronger halogen bond interactions in 11-azaartemisinin cocrystals with 4-halo-salicylic acids leads to the switching of energy preferences for polymorphic structures [37]. Different inter-planar C-H---X contacts where X = Cl, Br, O are also found and may influence the final 3D structural outcome. The dimer structure of Form III Tph is derived from the monohydrate and does not exhibit a perfect layer arrangement. As shown in the Tph hydrate structure in Figure 5 (right), dimers are stacked in registry with each other along the short a-axis, but such stacks have alternating tilts with respect to neighboring stacks. This structure orients C(1) methyl groups in proximity to each other [12], which is not the case for the halo analogues, so the dehydration of Tph does not lead to either the 8-Cl-Tph Form III or 8-Br-Tph Form IV arrangement.
3.5. Powder X-Ray Diffraction of 8-X-Tph Polymorphs
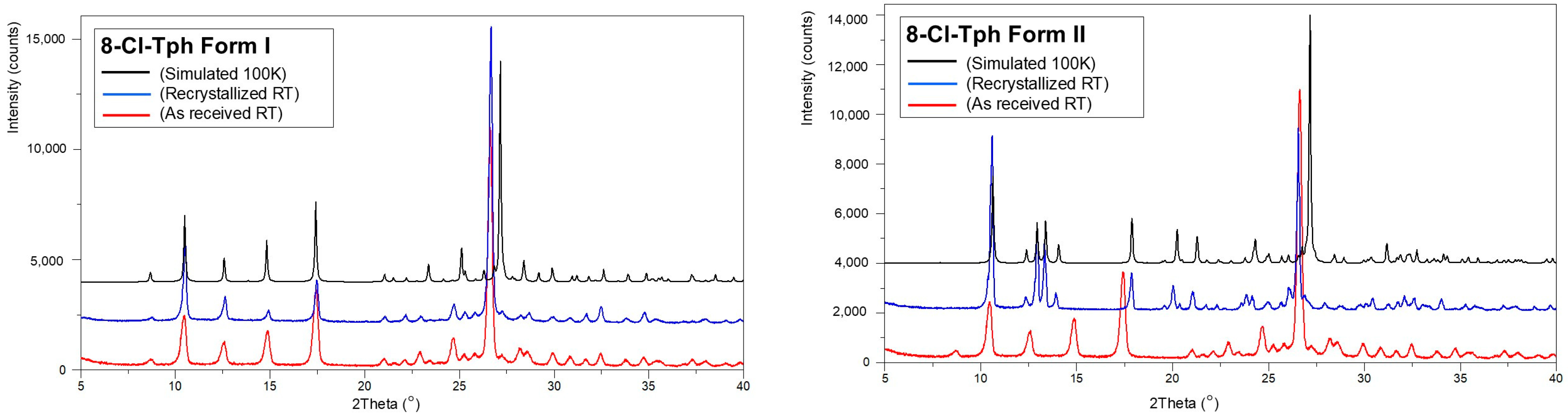
The isolation of phase pure polymorphs has not yet been achieved for all 8-X-Tph forms. In the case of 8-Cl-Tph, both Forms I and II can be prepared phase pure, and powder XRD patterns of phases isolated from high-temperature crystallization from ethanol and acetone fit those simulated from Forms I and Form II crystal structures measured at 100 K. The P-XRD of as-received 8-Cl-Tph also matches that of Form I (Figure 14).
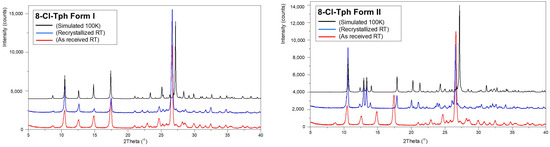
Figure 14.
Powder X-ray diffractograms for 8-Cl-Tph (left) Form I and (right) Form II.
3.6. Further Discussion of 8-X-Tph Polymorphs and Future Prospects
The difference and similarity of these different packing arrangements have also led us to study the possible solid-solution formation between these related molecules and the potential to template alternate polymorphic forms from analogue structures [15,16].
Unfortunately, Tph and the 8-halo analogues appear mutually insoluble, and LAG co-grinding [17,18] does not afford even limited solid solutions. In the case of 8-Cl-Tph and 8-Br-Tph, however, their electronic and steric similarities lead to the stable Form I being isostructural. Unsurprisingly then, solid solutions can be formed between them, which also adopt Form I, and a single crystal from a 1:1 co-dissolution in hot methanol and reprecipitation upon cooling gave a crystal structure indicating disorder at the 8-substituted site. Notably, this refined to occupancies of 72% 8-Cl and 28% 8-Br-Tph, indicating preferential incorporation of 8-Cl-Tph. The unit cell of this solid solution crystal is intermediate with V = 849 Å3 compared with 840 Å3 and 860 Å3 for pure 8-Cl-Tph and 8-Br-Tph structures.
Our initial purpose for the study of theophylline and 8-halotheophyllines was to examine their involvement in the formation of cocrystals with a variety of flavonoids. This was prompted by the recent report that cocrystals of caffeine with various flavonoids can be the basis for their separation based on differential solubility [38]. The measurement of the alkaloid crystal structures at 100 K was to provide benchmark-specific molecular volumes that might be used to assist in a comparison of coformer versus cocrystal volumes.
Unexpectedly, in some cases of ‘unsuccessful’ cocrystallization attempts, the alternate dimer polymorphs 8-X-Tph, namely, 8-Cl-Tph Form III and 8-Br-Tph Form IV, were isolated. The role of the flavonoid rutin in the case of 8-Cl-Tph and chrysin in the case of 8-Br-Tph is still being investigated and may have a subtle influence on the population of the molecular dimers in the solution or effects on the nucleation of competing polymorphic forms of the alkaloid. To date, no novel polymorphic form of theophylline itself was identified by us through such cocrystallization experiments with Tph. The switch of structure type from N(H)---N to N(H)---O hydrogen bonding and the DFT calculation of weaker basicity for 8-halo theophyllines hints that a weaker basicity of the alkaloid base N adversely affects the formation of cocrystals with the hydrogen bond donor flavonoids. Our initial survey supports this expectation, with the initial screening affording six cocrystals for Tph yet only two each for 8-Cl-Tph and 8-Br-Tph. The full results of these studies [39] will be reported elsewhere.
The computational studies carried out here were conducted on isolated ‘gas-phase’ molecules in order to check the inherent electronic modification of the molecule upon halo substitution. The polymorphic energies of the different structures have not yet been computed by applying boundary conditions, though a study of the nine polymorphic types for 8-X-Tph (X = H, Cl, Br) could be valuable. This might shed further light on the energetic ranking of the chain and dimer polymorphs for 8-X-Tph, and finally on whether a related more efficiently packed dimer polymorph of theophylline itself might be energetically competitive.
4. Conclusions
The crystal structures of several new polymorphic forms of the 8-halotheophyllines 8-Cl-Tph and 8-Br-Tph have been determined. Unlike the parent molecule theophylline, in which NH---N hydrogen bonds are typically favored, the structures are dominated by inter-molecular NH---O hydrogen bonding. This may be rationalized from the electronic perturbation of the halo substituent on the donor and acceptor properties of the proximal N(7)-H and N(9) groups. This renders N(7)H a slightly better donor, but crucially N(9) a weaker acceptor. DFT calculations support the idea that O(2) is more basic than N(9), leading to this switch. The most stable 8-Br-Tph polymorph appears isostructural with that of 8-Cl-Tph. In addition to chain arrangements, distinct dimer forms are found for all three molecules so that now a total of nine packing arrangements, Tph Forms I–V and 8-X-Tph (X = Br, Cl) Forms I–IV, have been found for these theophylline-type molecules.
Supplementary Materials
The following supporting information (full tables for crystallographic structures and computational results) can be downloaded at https://www.mdpi.com/article/10.3390/cryst15040340/s1.
Author Contributions
Conceptualization, I.D.W.; methodology, W.Y. and I.D.W.; computation, refinement, H.H.-Y.S., F.K.S., and L.W.-Y.W.; validation, C.Z. and L.W.-Y.W.; formal analysis, I.D.W. and F.K.S.; investigation, W.Y., C.Z., and F.K.S.; data curation, H.H.-Y.S. and F.K.S.; writing—original draft preparation, I.D.W.; writing—review and editing, all; supervision, I.D.W.; project administration, I.D.W.; funding acquisition, I.D.W. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Research Grants Council of Hong Kong (Grant No. 16306515) and equipment grant C6022-20E, the Hong Kong Branch of the Southern Marine Science and Engineering Guangdong Laboratory (Guangzhou) (Grant No. SMSEGL-20SC01-D), and the Hong Kong Branch of Chinese National Engineering Research Center for Tissue Restoration and Reconstruction, funded by the Innovation and Technology Commission (ITC-CNERC14SC01).
Data Availability Statement
Crystal structure determinations have been deposited with the Cambridge Crystallographic Data Centre (CCDC), deposition numbers 2420759 to 2420767.
Acknowledgments
Fanny L-Y. Shek and Carrie Law are thanked for their help with the DSC measurements.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Abbreviations
The following abbreviations are used in this manuscript:
| DFT | Density Functional Theory |
| DSC | differential scanning calorimetry |
| Tph | theophylline |
| LAG | liquid-assisted grinding |
| TGA | thermal gravimetric analysis |
| SS | solid solution |
References
- Carlucci, L.; Gavezzotti, A. Molecular Recognition and Crystal Energy Landscapes: An X-ray and Computational Study of Caffeine and Other Methylxanthines. Chem. Eur. J. 2004, 11, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Trask, A.V.; Motherwell, W.D.S.; Jones, W. Physical stability enhancement of theophylline via cocrystallization. Int. J. Pharm. 2006, 320, 114–123. [Google Scholar] [CrossRef]
- Sanphui, P.; Nangia, A. Salts and Co-crystals of Theobromine and their phase transformations in water. J. Chem. Sci. 2014, 126, 1249–1264. [Google Scholar] [CrossRef]
- Enright, G.D.; Terskikh, V.V.; Brouwer, D.H.; Ripmeester, J.A. The Structure of Two Anhydrous Polymorphs of Caffeine from Single-Crystal Diffraction and Ultrahigh-Field Solid-State 13C NMR Spectroscopy. Cryst. Growth Des. 2007, 7, 1406–1410. [Google Scholar] [CrossRef]
- Lehmann, C.W.; Stowasser, F. The crystal structure of anhydrous beta-caffeine as determined from X-ray powder-diffraction data. Chemistry 2007, 13, 2908–2911. [Google Scholar] [CrossRef] [PubMed]
- Ebisuzaki, Y.; Boyle, P.D.; Smith, J.A. Methylxanthines. I. Anhydrous Theophylline. Acta Crystallogr. Sect. C 1997, 53, 777–779. [Google Scholar] [CrossRef]
- Zhang, S.; Fischer, A. A monoclinic polymorph of theophylline. Acta Crystallogr. Sect. E 2011, 67, o3357. [Google Scholar] [CrossRef]
- Khamar, D.; Pritchard, R.G.; Bradshaw, I.J.; Hutcheon, G.A.; Seton, L. Polymorphs of anhydrous theophylline: Stable form IV consists of dimer pairs and metastable form I consists of hydrogen-bonded chains. Acta Crystallogr. Sect. C Cryst. Struct. Commun. 2011, 67, o496–o499. [Google Scholar] [CrossRef]
- Fucke, K.; McIntyre, G.J.; Wilkinson, C.; Henry, M.; Howard, J.A.K.; Steed, J.W. New insights into an Old Molecule: Interaction Energies of Theophylline Crystal Forms. Cryst. Growth Des. 2012, 12, 1395–1401. [Google Scholar] [CrossRef]
- Larsen, A.S.; Olsen, M.A.; Moustafa, H.; Larsen, F.H.; Sauer, S.P.A.; Rantanen, J.; Madsen, A.Ø. Determining short-lived solid forms during phase transformations using molecular dynamics. CrystEngComm 2019, 21, 4020–4024. [Google Scholar] [CrossRef]
- Seton, L.; Khamar, D.; Bradshaw, I.J.; Hutcheon, G.A. Solid State Forms of Theophylline: Presenting a New Anhydrous Polymorph. Cryst. Growth Des. 2010, 10, 3879–3886. [Google Scholar] [CrossRef]
- Sun, C.; Zhou, D.; Grant, D.J.W.; Young, V.G. Theophylline Monohydrate. Acta Crystallogr. Sect. E 2002, 58, o368–o370. [Google Scholar] [CrossRef]
- Dyulgerov, V.M.; Dimowa, L.T.; Kossev, K.; Nikolova, R.P.; Shivachev, B.L. Solvothermal synthesis of theophylline an N,N ′-(ethane-1,2-diyl)diformamide co-crystals from DMF decomposition and N-formylation trough catalytic effect of 3-carboxyphenylboronic acid and cadmium acetate. Bulgarian Chem. Commun. 2015, 47, 311–316. [Google Scholar]
- Putra, O.D.; Yoshida, T.; Umeda, D.; Higashi, K.; Uekusa, H.; Yonemochi, E. Crystal Structure Determination of Dimenhydrinate after More than 60 Years: Solving Salt–Cocrystal Ambiguity via Solid-State Characterizations and Solubility Study. Cryst. Growth Des. 2016, 16, 5223–5229. [Google Scholar] [CrossRef]
- Srirambhatla, V.K.; Guo, R.; Price, S.L.; Florence, A.J. Isomorphous template induced crystallisation: A robust method for the targeted crystallisation of computationally predicted metastable polymorphs. Chem. Commun. 2016, 52, 7384–7386. [Google Scholar] [CrossRef]
- Case, D.H.; Srirambhatla, V.K.; Guo, R.; Watson, R.E.; Price, L.S.; Polyzois, H.; Cockcroft, J.K.; Florence, A.J.; Tocher, D.A.; Price, S.L. Successful Computationally Directed Templating of Metastable Pharmaceutical Polymorphs. Cryst. Growth Des. 2018, 18, 5322–5331. [Google Scholar] [CrossRef]
- Trask, A.V.; Jones, W. Crystal Engineering of Organic Cocrystals by the Solid-State Grinding Approach. Top. Curr. Chem. 2005, 254, 41–70. [Google Scholar] [CrossRef]
- Trask, A.V.; van de Streek, J.; Motherwell, W.D.S.; Jones, W. Achieving Polymorphic and Stoichiometric Diversity in Cocrystal Formation: Importance of Solid-State Grinding, Powder X-ray Structure Determination, and Seeding. Cryst. Growth Des. 2005, 5, 2233–2241. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A 2008, 64, 112–122. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. Olex2: A complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Bourhis, L.J.; Dolomanov, O.V.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. The anatomy of a comprehensive constrained, restrained refinement program for the modern computing environment-Olex2 dissected. Acta Crystallogr. Sect. A 2015, 71, 59–75. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09 Revision E.01; Gaussian Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Chai, J.-D.; Head-Gordon, M. Long-range corrected hybrid density functionals with damped atom–atom dispersion corrections. Phys. Chem. Chem. Phys. 2008, 10, 6615–6620. [Google Scholar] [CrossRef] [PubMed]
- Weigend, F.; Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. 2005, 7, 3297–3305. [Google Scholar] [CrossRef] [PubMed]
- Wiberg, K.B. Application of the Pople-Santry-Segal CNDO method to the cyclopropylcarbinyl and cyclobutyl cation and to bicyclobutane. Tetrahedron 1968, 24, 1083–1096. [Google Scholar] [CrossRef]
- Glendening, E.D.; Landis, C.R.; Weinhold, F. Natural bond orbital methods. WIREs Comput. Mol. Sci. 2012, 2, 1–42. [Google Scholar] [CrossRef]
- Besler, B.H.; Merz, K.M.; Kollman, P.A. Atomic charges derived from semiempirical methods. J. Comp. Chem. 1990, 11, 431–439. [Google Scholar] [CrossRef]
- Singh, U.C.; Kollman, P.A. An approach to computing electrostatic charges for molecules. J. Comp. Chem. 1984, 5, 129–145. [Google Scholar] [CrossRef]
- Etter, M.C.; Macdonald, J.C.; Bernstein, J. Graph-set analysis of hydrogen bond patterns. Acta Crystallogr. Sect. B 1990, 46, 256–262. [Google Scholar] [CrossRef]
- Singh, V.B. Spectroscopic signatures and structural motifs in isolated and hydrated theophylline: A computational study. RSC Adv. 2015, 5, 11433–11444. [Google Scholar] [CrossRef]
- Krivovichev, S.V. Structural complexity and configurational entropy of crystals. Acta Crystallogr. Sect. B 2016, 72, 274–276. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.; Jencks, W.P.; Westheimer, F.H. Compilation of pKa Acidity Constants (ACS Organic Division). 2025. Available online: https://www.webqc.org/pkaconstants.php (accessed on 31 January 2025).
- Nolasco, M.M.; Amado, A.M.; Ribeiro-Claro, P.J.A. Computationally-Assisted Approach to the Vibrational Spectra of Molecular Crystals: Study of Hydrogen-Bonding and Pseudo-Polymorphism. ChemPhysChem 2006, 7, 2150–2161. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, B.A.; Du, J.J.; Lai, F.; Stanton, S.A.; Williams, P.A.; Groundwater, P.W.; Platts, J.A.; Overgaard, J.; Hibbs, D.E. An experimental and theoretical charge density study of theophylline and malonic acid cocrystallization. RSC Adv. 2022, 12, 15670–15684. [Google Scholar] [CrossRef]
- Lu, T.; Chen, F.-W. Comparison of Computational Methods for Atomic Charges. Acta Phys. Chim. Sin. 2012, 28, 1–18. [Google Scholar] [CrossRef]
- Li, K.; Roy, M.; Nisar, M.; Wong, L.W.-Y.; Sung, H.H.-Y.; Haynes, R.K.; Williams, I.D. Control of 11-Aza:4-X-SalA Cocrystal Polymorphs Using Heteroseeds That Switch On/Off Halogen Bonding. Crystals 2022, 12, 1368. [Google Scholar] [CrossRef]
- Xia, Y.; Wei, Y.; Chen, H.; Qian, S.; Zhang, J.; Gao, Y. Competitive cocrystallization and its application in the separation of flavonoids. IUCrJ 2021, 8, 195–207. [Google Scholar] [CrossRef] [PubMed]
- Ye, W. Isolation, Purification and Structural Studies of Natural Product Crystals and Cocrystals. Ph.D. Thesis, Hong Kong University of Science and Technology, Hong Kong, December 2024. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).