Crystal Chemistry and Structural Complexity of the Uranyl Molybdate Minerals and Synthetic Compounds
Abstract
:1. Introduction
2. Materials and Methods
2.1. Structural Data
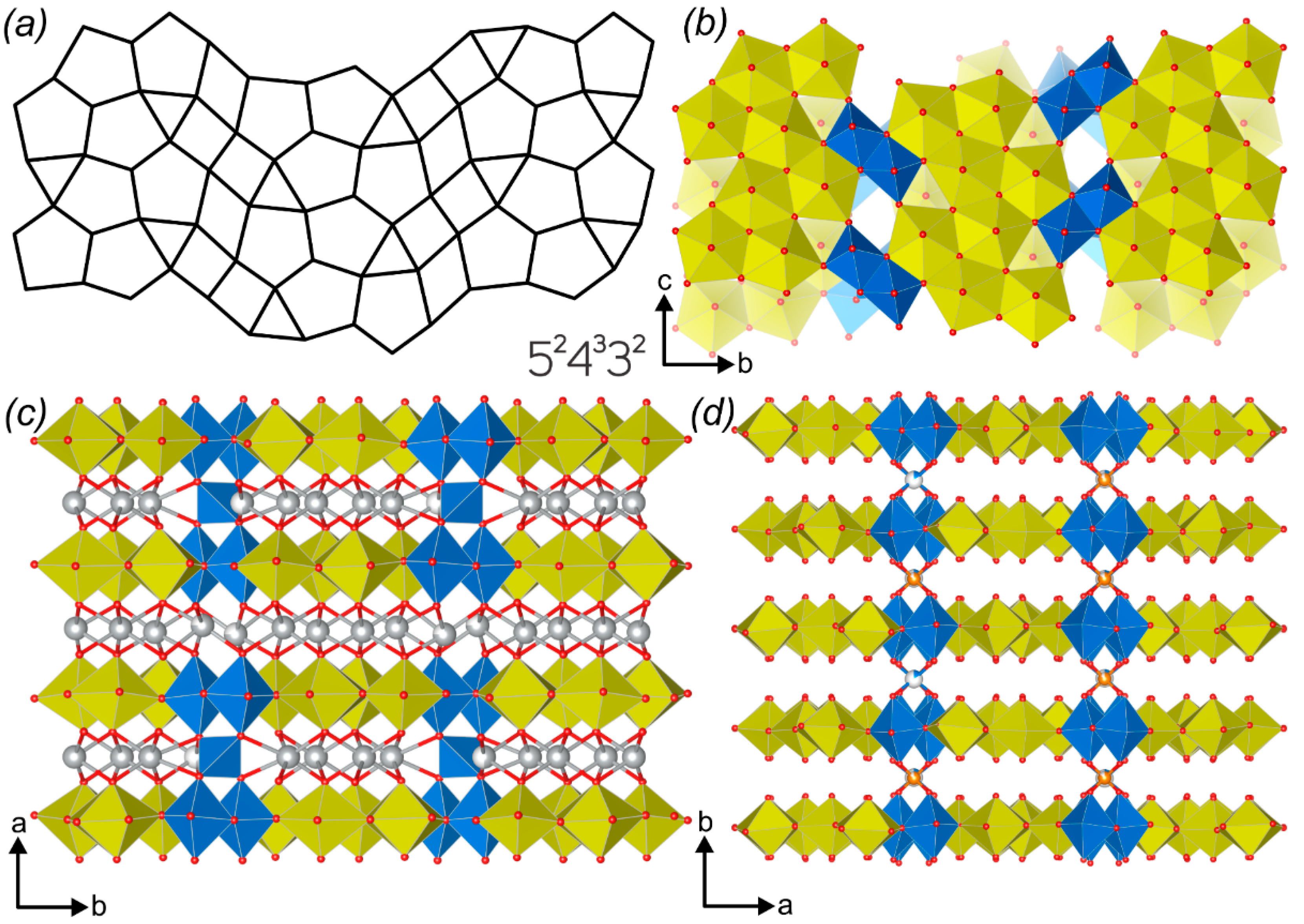
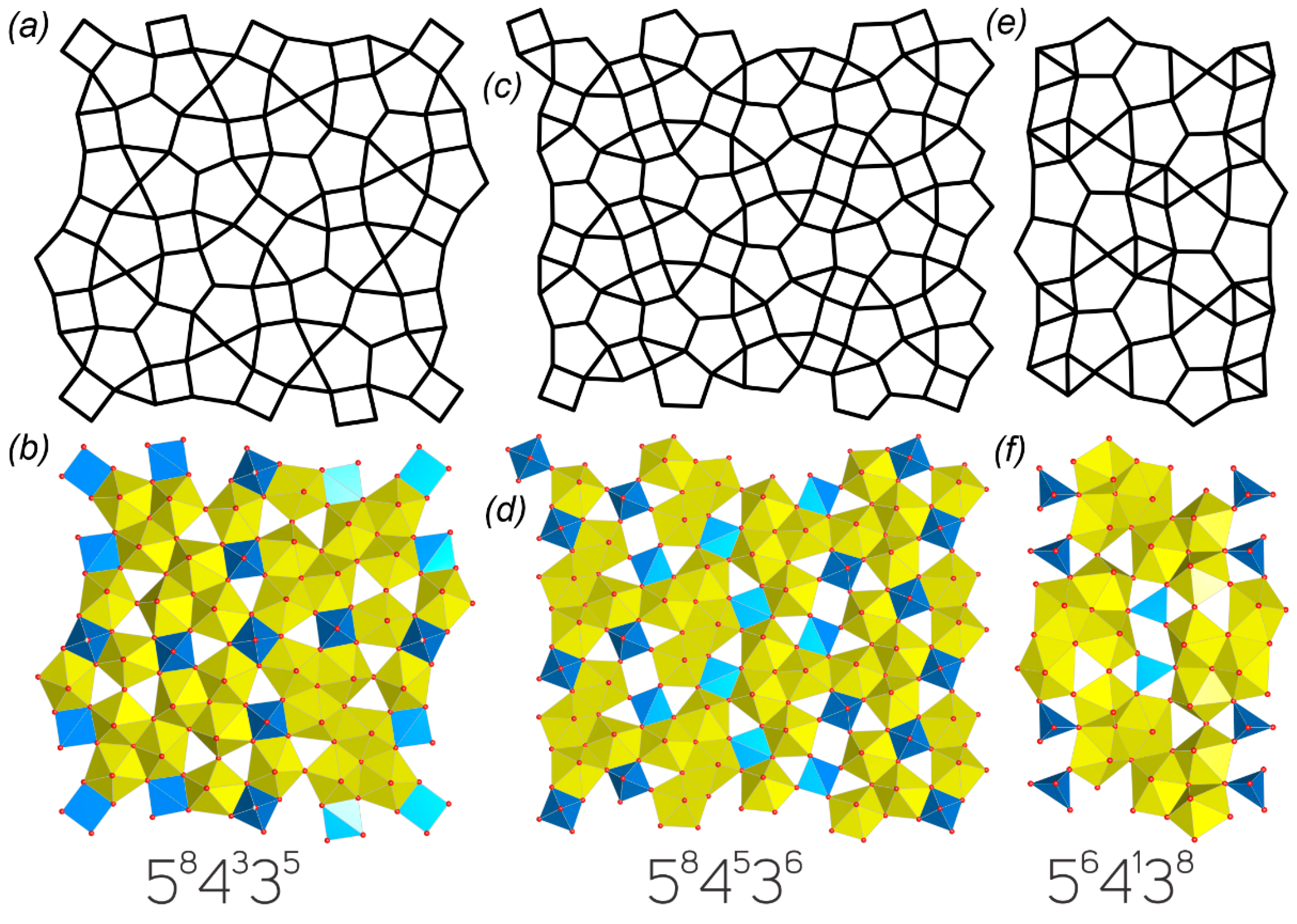
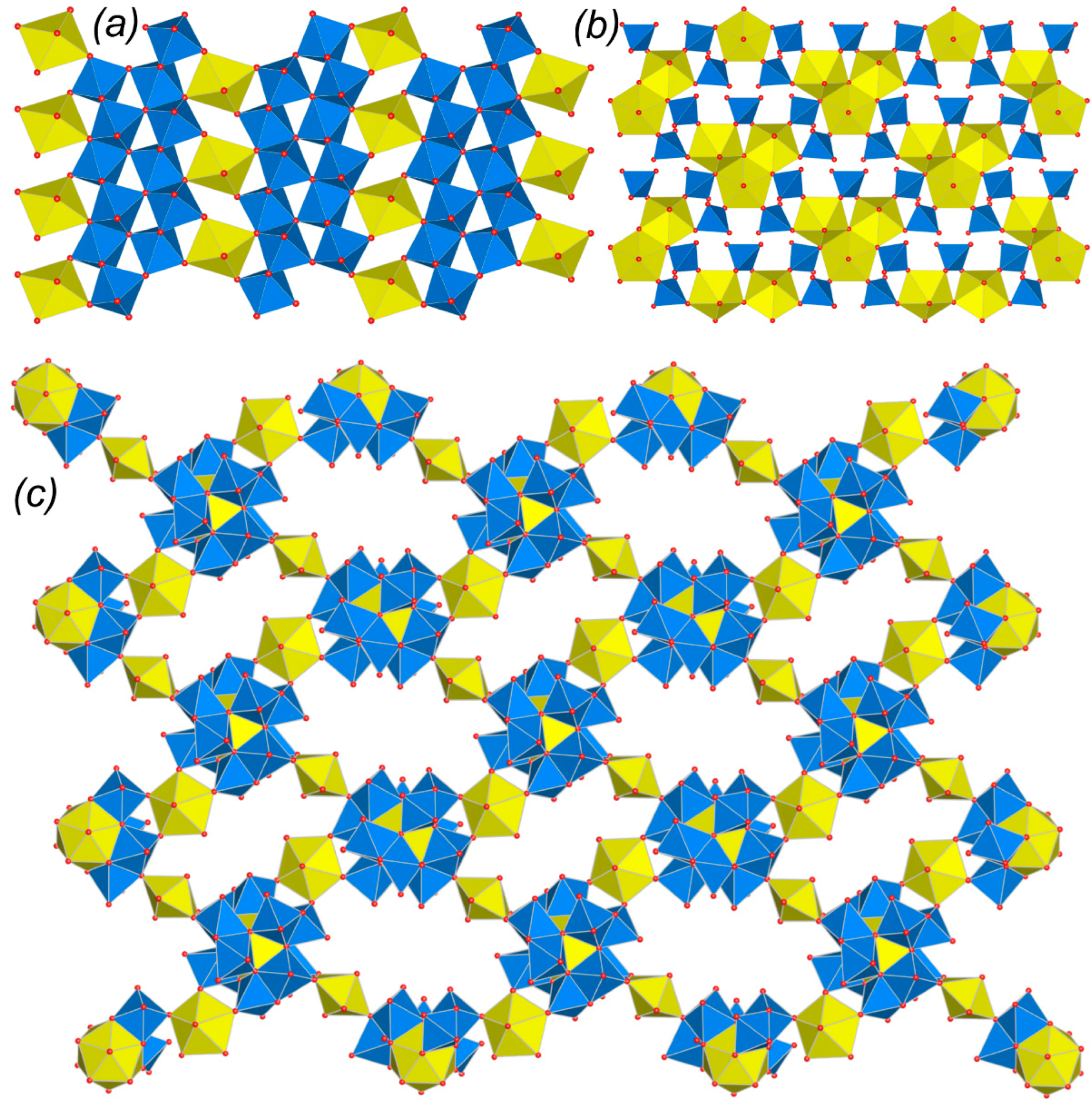
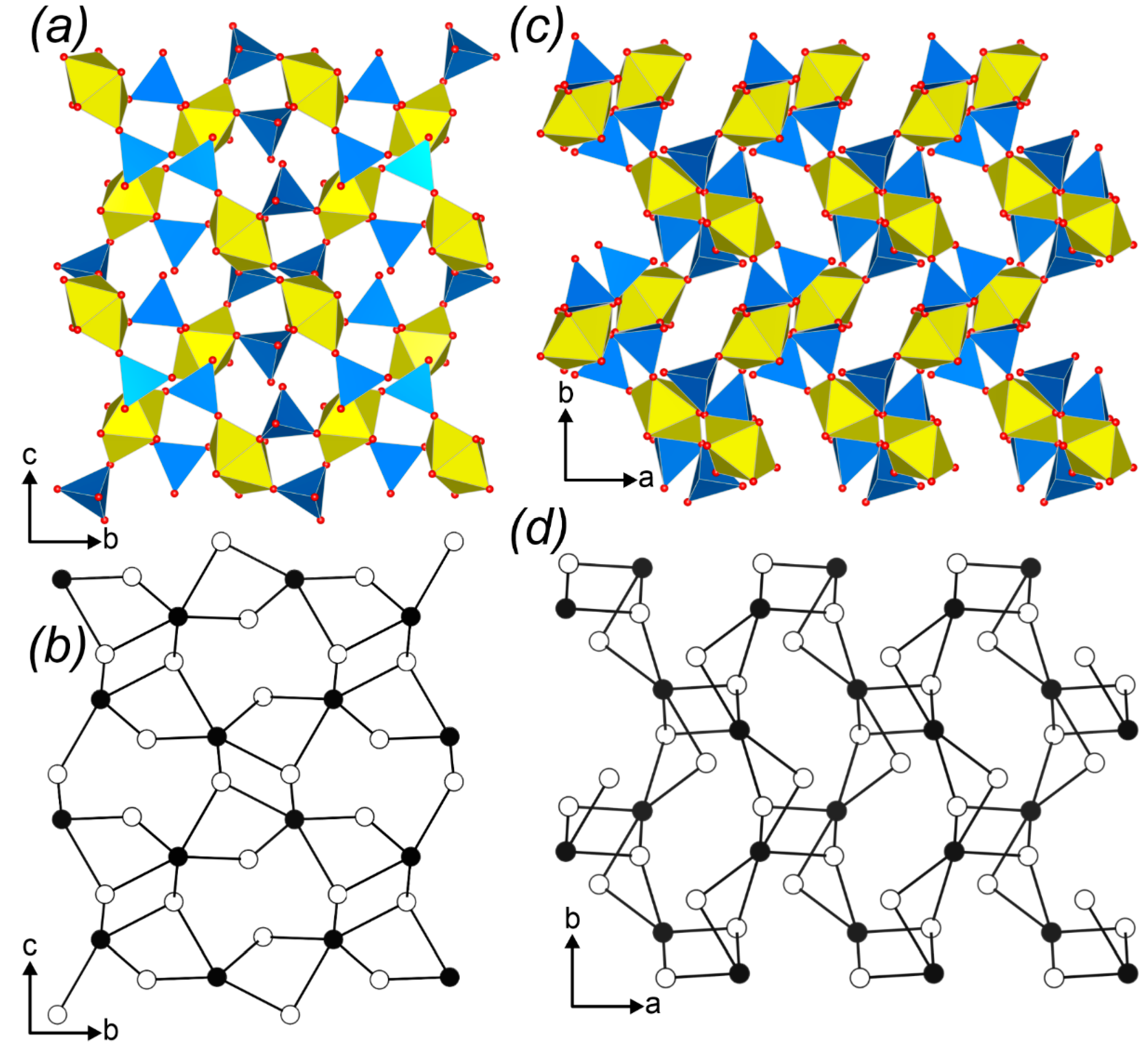
2.2. Graphical Representation and Anion Topologies
2.3. Complexity Calculations
| No. | Chemical Formula | Mineral Name | Sp. Gr. | а, Å/ α, ° | b, Å/ β, ° | с, Å/ γ, ° | Structural Complexity Parameters, Bits per Atom/Bits per Unit Cell | Ref. | |
|---|---|---|---|---|---|---|---|---|---|
| Infinite Chains | U-Bearing Unit | Entire Structure | |||||||
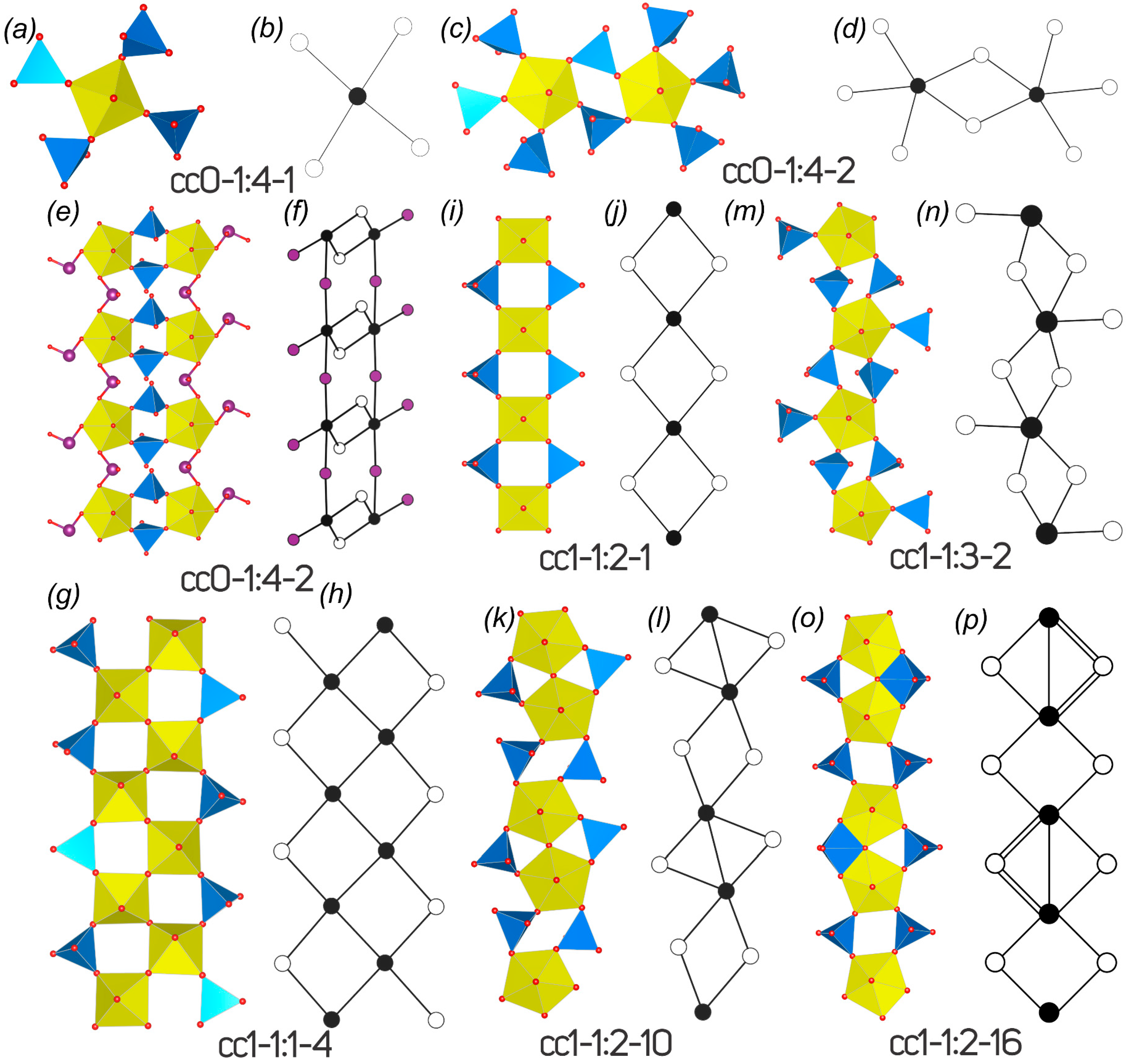
| cc1–1:2–1 | |||||||||
| 1 | Cu4(UO2)(Mo2O8)(OH)6 | Deloryite | C2/m | 19.940(10)/90 | 6.116(2)/104.18(5) | 5.520(3)/90 | 2.470/32.106 | 3.586/125.525 | [31,32,33] |
| Layers | |||||||||
| Sheets of clusters of uranyl polyhedra | |||||||||
| Uranophane topology, 514131 | |||||||||
| 2 | [(UO2)(MoO4)(H2O)](H2O) | Umohoite | P-1 | 6.3748(4)/82.64(1) | 7.5287(5)/85.95(1) | 14.628(1)/89.91(1) | 4.000/128.000 | 4.585/220.078 | [34,35,36,37,38] |
| 2a | (UO2)[(MoO4)(H2O)](H2O)1.45 | Umohoite | P-1 | 14.69(3)/90.07 | 7.535(4)/85.9 | 6.372(3)/97.1 | 4.000/128.000 | 4.700/244.423 | [39] |
| 2b | [(UO2)(MoO4)(H2O)](H2O) | Umohoite 14A | P21/c | 6.32/90 | 7.5/94 | 57.8/90 | 5.358/878.639 | 5.512/992.534 | [40] |
| Iriginite topology, 514331 | |||||||||
| 3 | [(UO2)(Mo2O7)(H2O)](H2O) | Iriginite | Pbcm | 6.705(1)/90 | 12.731(2)/90 | 11.524(2)/90 | 2.918/140.078 | 3.741/344.168 | [41,42,43,44] |
| Sheets formed by chains of uranyl polyhedra | |||||||||
| 534332 | |||||||||
| 4 | Ba0.5[(UO2)3O8Mo2(OH)3](H2O)∼3 | Baumoite | Monoclinic * | 9.8337(3)/90 | 15.0436(5)/108.978(3) | 14.2055(6)/90 | [45] | ||
| Sheets of uranyl polyhedra | |||||||||
| 564138 | |||||||||
| 5 | Ca(UO2)6(MoO4OH)2O2(OH)49H2O | Tengchongite | C2221 | 13.0866(8)/90 | 17.6794(12)/90 | 15.6800(9)/90 | 4.170/200.235 | 5.387/894.257 | [46,47] |
| Undefined structures | |||||||||
| 6 | H4U4+(UO2)3(MoO4)7(H2O)18 | Moluranite | [42,48] | ||||||
| 7 | (UO2)(Mo6+)5O16(H2O)5 | Mourite | C2, Cm or C2/m | 24.426(6)/90 | 7.185(1)/102.10(1) | 9.895(1)/90 | [49,50] | ||
| 8 | Ca[(UO2)3(MoO4)2(OH)4](H2O)~5.0 | Calcurmolite | Monoclinic | 16.30(3)/90 | 25.49(5)/90.07 | 19.50(6)/90 | [51,52,53,54] | ||
| 9 | MgU4+2(MoO4)2(OH)6·2H2O (?) | Cousinite | [55,56] | ||||||
| 10 | U4+(MoO4)2 | Sedovite | Orthorombic | 3.36(6)/90 | 11.08(3)/90 | 6.42(5)/90 | [57] | ||
| No. | Chemical Formula | Sp. Gr. | а, Å/ α, ° | b, Å/ β, ° | с, Å/ γ, ° | Syn. * | Structural Complexity Parameters, Bits per Atom/Bits per Unit Cell | Ref. | ||
| FINITE CLUSTERS | ||||||||||
| cc0–1:4–1 | ||||||||||
| 11 | Cs6[(UO2)(MoO4)4] | P-1 | 11.613(3)/102.713(6) | 12.545(3)/95.281(6) | 14.466(3)/106.182(6) | SS[850] | 5.123/353.488 | 5.454/474.536 | [58] | |
| 12 | Rb6[(UO2)(MoO4)4] | C2/c | 17.312(1)/90 | 11.5285(8)/127.634(1) | 13.916(1)/90 | SS[700] | 3.567/164.084 | 3.961/229.763 | [59] | |
| cc0–1:4–2 | ||||||||||
| 13 | Na6[(UO2)(MoO4)4] | P-1 | 7.0958(8)/73.692(2) | 9.566(1)/86.621(2) | 13.415(2)/82.940(2) | SS[850] | 4.524/208.084 | 4.858/281.763 | [7] | |
| 14 | Na3Tl3[(UO2)(MoO4)4] | Pbcn | 20.5823(14)/90 | 7.4391(5)/90 | 26.2514(17)/90 | HTwA[120] | 4.524/832.335 | 4.858/1127.052 | [60] | |
| INFINITE CHAINS | ||||||||||
| cc1–1:1–4 | ||||||||||
| 15 | Cs2[(UO2)O(MoO4)] | Pca21 | 12.018(2)/90 | 12.438(2)/90 | 17.917(3)/90 | SS[870] | 4.755/513.528 | 5.044/665.860 | [61] | |
| cc1–1:2–1 | ||||||||||
| 16 | Cu4[(UO2)(MoO4)2](OH)6 | B2/m | 19.8392(11)/90 | 5.5108(3)/90 | 6.1009(4)/104.477(4) | HT[280] | 2.470/32.106 | 3.617/104.881 | [31] | |
| 17 | Li2[(UO2)(MoO4)2] | P-1 | 5.3455(4)/108.267(2) | 5.8297(4)/100.566(2) | 8.2652(6)/104.121(2) | SS[650] | 2.777/36.106 | 2.974/44.603 | [62] | |
| cc1–1:2–10 | ||||||||||
| 18 | Na6[(UO2)O(MoO4)4] | P-1 | 7.637(2)/72.329(5) | 8.164(2)/79.364(5) | 8.746(2)/65.795(4) | SS[850] | 3.792/102.382 | 4.075/134.465 | [7] | |
| 19 | K6[(UO2)2O(MoO4)4] | P-1 | 7.8282(8)/83.893(2) | 7.8298(8)/73.131(2) | 10.302(1)/80.338(2) | SS[850] | 3.792/102.382 | 4.075/134.465 | [7] | |
| 20 | Rb6[(UO2)2O(MoO4)4] | P-1 | 10.1567(5)/76.921(1) | 10.1816(5)/76.553(1) | 13.1129(6)/65.243(1) | SS[700] | 4.755/256.764 | 5.044/332.930 | [58] | |
| cc1–1:2–16 | ||||||||||
| 21 | Cs6[(UO2)2(MoO4)3(MoO5)] | P-1 | 10.4275(14)/70.717(2) | 15.075(2)/80.382(2) | 17.806(2)/86.386(2) | SS[950] | 5.755/621.528 | 6.044/797.860 | [63] | |
| cc1–1:3–2 | ||||||||||
| 22 | Na3Tl5[(UO2)(MoO4)3]2(H2O)3 | P212121 | 10.7662(6)/90 | 11.9621(6)/90 | 12.8995(7)/90 | HTwA[180] | 4.248/322.842 | 4.700/488.846 | [59] | |
| 23 | Na12.9Tl3.1[(UO2)(MoO4)3]4(H2O)6.6 | P2/c | 19.7942(11)/90 | 7.1913(4)/97.828(1) | 22.8835(13)/90 | HTwA[120] | 5.248/797.685 | 5.716/1177.419 | [59] | |
| cc1–1:3–3 | ||||||||||
| 24 | K2[(UO2)(MoO4)(IO3)2] | P21/c | 11.3717(6)/90 | 7.2903(4)/108.167(1) | 15.7122(8)/90 | HT[180] | 3.459/152.215 | 4.170/300.235 | [64] | |
| LAYERS | ||||||||||
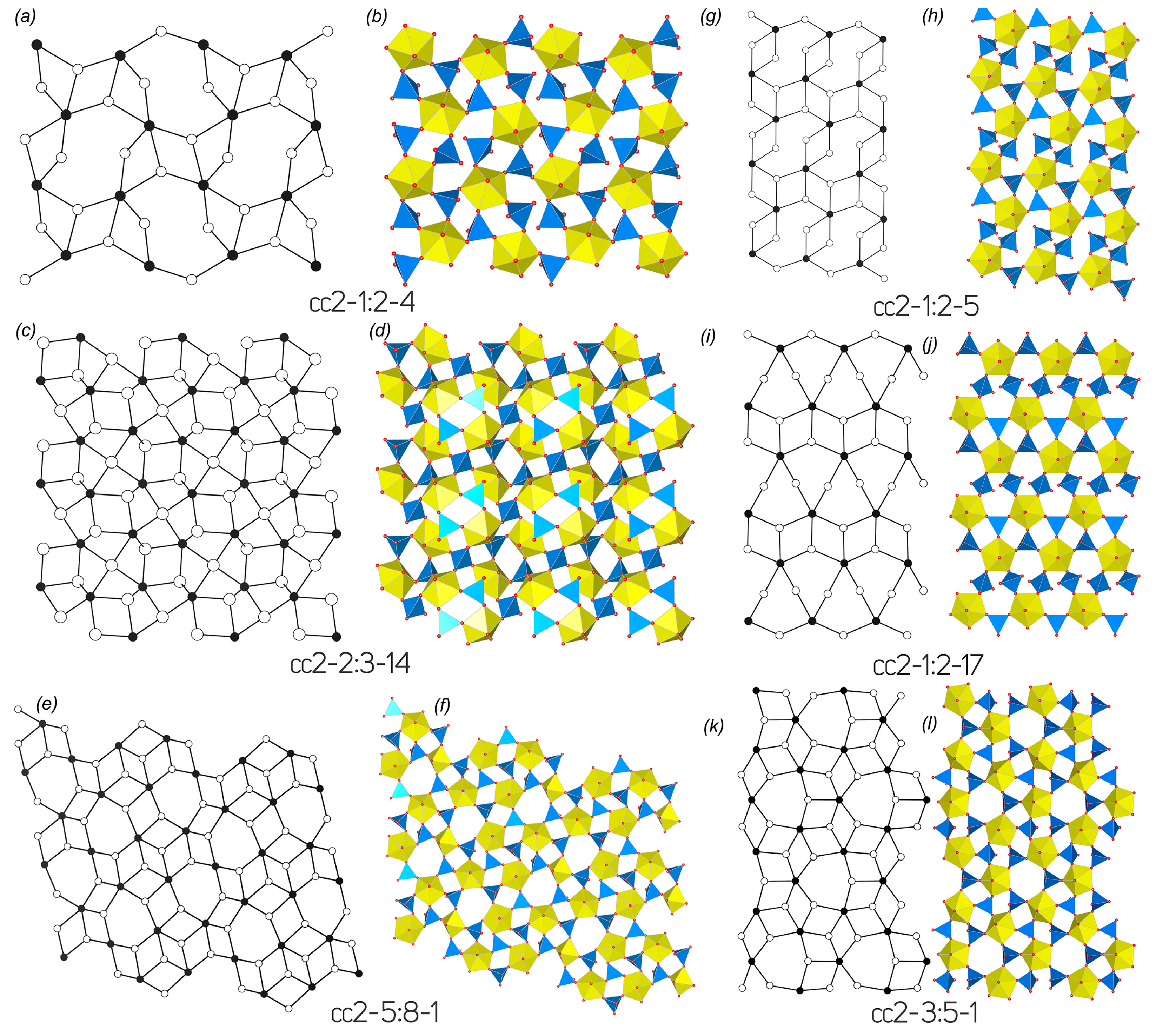
| Vertex-sharing interpolyhedral linkage | ||||||||||
| cc2–1:2–4 | ||||||||||
| 25 | Cs2[(UO2)(MoO4)2] | Pbca | 11.762(2)/90 | 14.081(2)/90 | 14.323(2)/90 | SS[600] | 3.700/384.846 | 3.907/468.827 | [65] | |
| 26 | Cs2[(UO2)(MoO4)2](H2O) | P21/c | 8.2222(9)/90 | 11.0993(10)/95.155(8) | 13.9992(13)/90 | HT[120] | 3.700/192.423 | 4.322/345.754 | [65] | |
| 27 | Cs2(UO2)(MoO4)2(H2O) | P21/a | 14.031(1)/90 | 8.272(2)/90 | 11.067(2)/95.63 | HTwA | 3.700/192.423 | 4.322/345.754 | [66] | |
| 28 | K2[(UO2)(MoO4)2] | P21/c | 12.269(5)/90 | 13.468(5)/95.08(3) | 12.857(6)/90 | HTwA | 4.700/488.846 | 4.907/588.827 | [67] | |
| 29 | K2[(UO2)(MoO4)2](H2O) | P21/c | 7.893(2)/90 | 10.907(2)/98.70(3) | 13.558(3)/90 | HT[150] | 3.700/192.423 | 4.17/300.235 | [68] | |
| 30 | Rb2[(UO2)(MoO4)2] | P21/c | 12.302(1)/90 | 13.638(1)/94.975(1) | 13.508(1)/90 | SS[700] | 4.700/488.846 | 4.907/588.827 | [58] | |
| 31 | (C2H10N2)[(UO2)(MoO4)2] | P-1 | 8.4004(4)/86.112(1) | 11.2600(5)/86.434(1) | 13.1239(6)/76.544(1) | HT[180] | 4.700/244.423 | 5.755/621.528 | [69] | |
| 32 | Rb2[(UO2)(MoO4)2](H2O) | P21/c | 7.967(3)/90 | 10.956(4)/96.69(3) | 13.679(5)/90 | Aq/HT[120–180] | 3.700/192.423 | 4.17/300.235 | [70] | |
| 33 | Tl2[(UO2)(MoO4)2] | Pca21 | 10.977(3)/90 | 14.004(3)/90 | 14.041(3)/90 | SS[600] | 4.700/488.846 | 4.907/588.827 | [71] | |
| 34 | (C5H14N2)[(UO2)(MoO4)2](H2O) | Pbca | 12.697(1)/90 | 13.247(1)/90 | 17.793(1)/90 | HT[180] | 3.700/384.846 | 5.129/1436.199 | [72] | |
| 35 | Na2[(UO2)(MoO4)2](H2O)4 | P21/n | 8.9023(5)/90 | 11.5149(6)/107.743(1) | 13.8151(7)/90 | HT[120] | 3.700/192.423 | 4.755/513.528 | [59] | |
| cc2–1:2–5 | ||||||||||
| 36 | (C4H12N2)[(UO2)(MoO4)2] | P-1 | 7.096(1)/97.008(3) | 8.388(1)/96.454(2) | 11.634(1)/110.456(3) | HT[180] | 3.700/96.211 | 4.954/307.160 | [72] | |
| cc2–1:2–17 | ||||||||||
| 37 | Na2[(UO2)(MoO4)2] | P212121 | 7.2298(5)/90 | 11.3240(8)/90 | 12.0134(8)/90 | SS[850] | 3.700/192.423 | 3.907/234.413 | [68] | |
| cc2–2:3–14 | ||||||||||
| 38 | Beta-Cs2[(UO2)2(MoO4)3] | P42/n | 10.1367(8)/90 | 10.1367(8)/90 | 16.2831(17)/90 | SS[850] | 3.488/292.955 | 3.697/340.168 | [73] | |
| cc2–3:5–1 | ||||||||||
| 39 | [C3H9NH+]4[(UO2)3(MoO4)5] | Cc | 16.768(6)/90 | 20.553(8)/108.195(7) | 11.897(4)/90 | HT[220] | 5.087/345.947 | 6.492/1168.534 | [74] | |
| 40 | (NH3(CH2)3NH3)(H3O)2[(UO2)3(MoO4)5] | Pbnm | 10.465(1)/90 | 16.395(1)/90 | 20.241(1)/90 | HT[180] | 4.264/579.895 | 4.898/1038.319 | [72] | |
| cc2–5:8–1 | ||||||||||
| 41 | (C6H14N2)3[(UO2)5(MoO4)8](H2O)4 | P-1 | 11.8557(9)/96.734(2) | 11.8702(9)/91.107(2) | 12.6746(9)/110.193(2) | HT[180] | 4.800/263.975 | 6.041/791.378 | [69] | |
| Edge-sharing interpolyhedral linkage | ||||||||||
| Layers with clusters of uranyl polyhedra | ||||||||||
| Iriginite topology | ||||||||||
| 42 | [Ca(UO2)(Mo2O7)2] | P-1 | 13.239(5)/90.00(4) | 6.651(2)/90.38(4) | 8.236(3)/120.16(3) | SQT[600] | 4.392/184.477 | 4.459/196.215 | [75] | |
| 43 | [(UO2)(Mo2O7)(H2O)2] | C2/c | 35.071(6)/90 | 6.717(1)/90.069(6) | 11.513(2)/90 | HT[230] | 4.301/326.842 | 4.684/468.386 | [76] | |
| Layers of miscellaneous topologies | ||||||||||
| 44 | Ag6[(UO2)3O(MoO4)5] | C2/c | 16.4508(14)/90 | 11.3236(14)/100.014(8) | 12.7418(13)/90 | SS[650] | 4.215/295.05 | 4.431/363.319 | [77] | |
| 45 | K2[UO2(Mo2O7)2] | P21/c | 9.0775(5)/90 | 4.9444(2)/106.912(6) | 15.2017(10)/- | SS[900]/HP | 3.440/144.477 | 3.567/164.084 | [5] | |
| 46 | (NH4)4((UO2)2(H2O)3 UMo12O42)(H2O)18 | P-1 | 11.429(2)/84.58(2) | 14.359(3)/87.96(2) | 16.491(3)/87.38(2) | Aq | 6.016/770.000 | 6.625/1298.483 | [78] | |
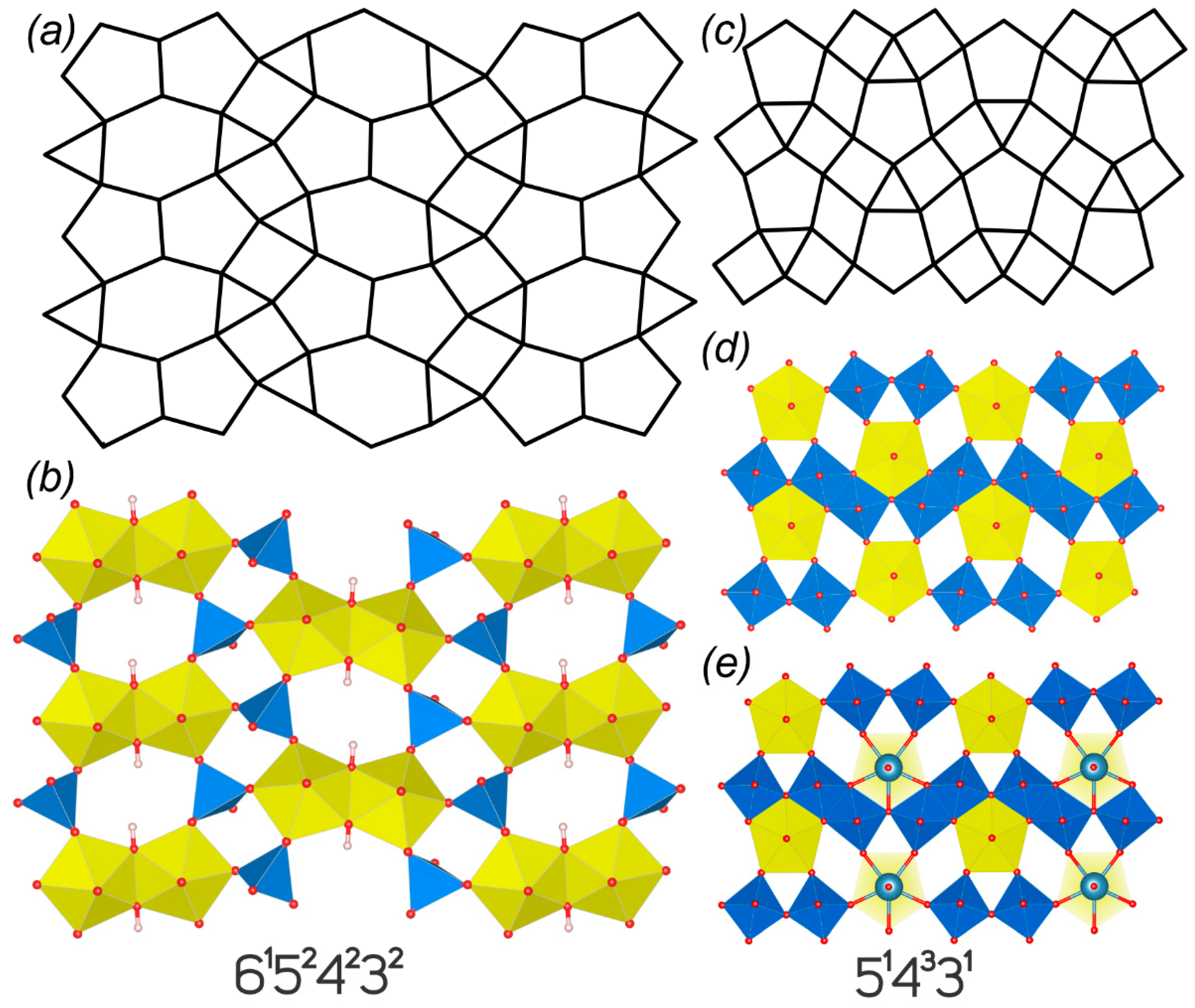
| Phosphuranylite topology, 61524232 | ||||||||||
| 47 | C(NH2)3(UO2)(OH)(MoO4) | P21/c | 15.411(1)/90 | 7.086(1)/113.125(2) | 18.108(1)/90 | HT[180] | 4.322/345.754 | 5.248/797.685 | [72] | |
| Layers with chains of uranyl polyhedra | ||||||||||
| 48 | Cs4[(UO2)3O(MoO4)2(MoO5)] | P1 | 7.510(2)/79.279(5) | 7.897(2)/81.269(5) | 9.774(2)/87.251(5) | SS[850] | 4.700/122.211 | 4.907/147.207 | [58] | |
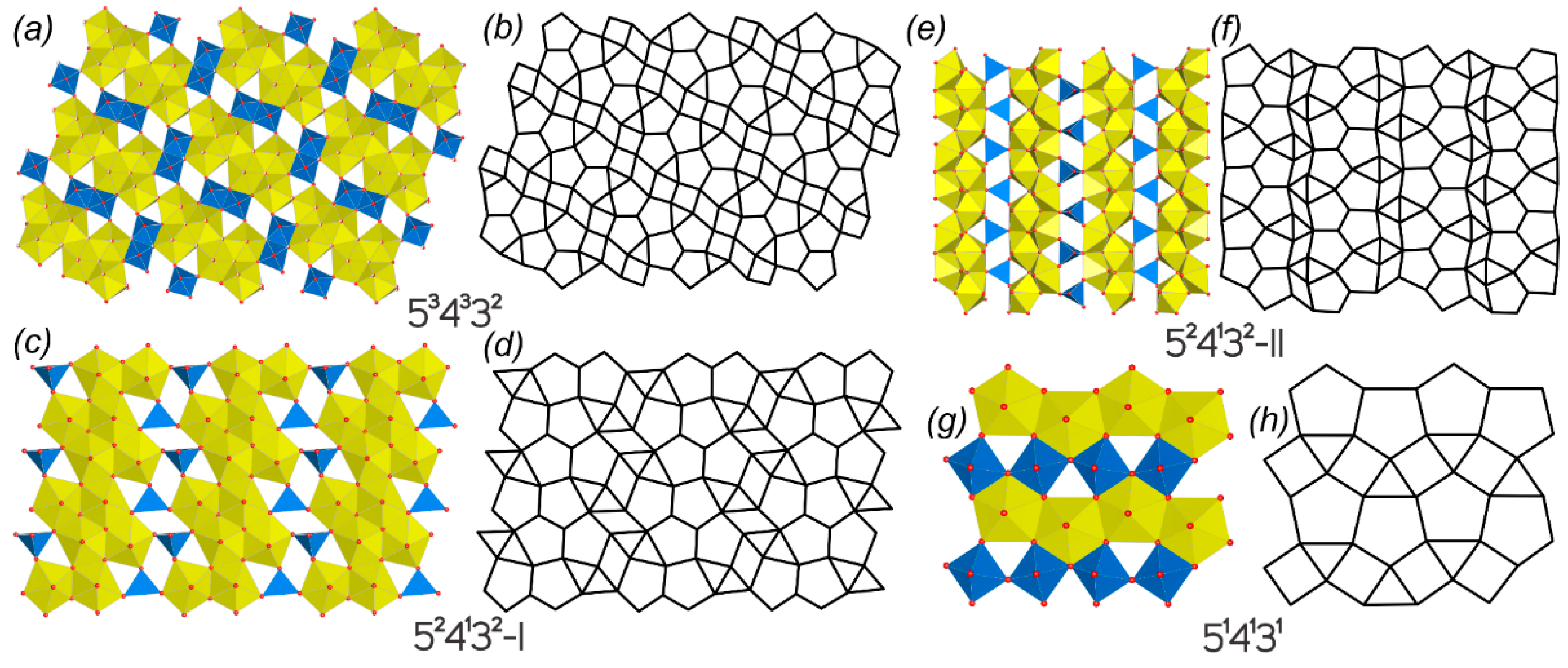
| 534332-I | ||||||||||
| 49 | CsNa3[(UO2)4O4(Mo2O8)] | P-1 | 6.4655(13)/84.325(10) | 6.9057(10)/77.906(9) | 11.381(2)/80.230(9) | SS[950] | 3.700/96.211 | 3.974/119.207 | [79] | |
| 524332 | ||||||||||
| 50 | Ag10[(UO2)8O8(Mo5O20)] | C2/c | 24.672(2)/90 | 23.401(2)/94.985(2) | 6.7932(4)/90 | SS[650] | 4.850/552.949 | 5.176/703.895 | [80] | |
| 51 | K2Na8(UO2)8(Mo4O24)[(S,Mo)O4] | C2/c | 24.282(4)/90 | 12.1170(18)/106.33(1) | 13.6174(17)/90 | SS[700] | 4.892/567.526 | 5.117/695.895 | [81] | |
| 52 | Cs2Na8[(UO2)8O8(Mo5O20)] | Ibam | 6.8460(2)/90 | 23.3855(7)/90 | 12.3373(3)/- | SS[950] | 3.479/201.763 | 3.793/257.947 | [79] | |
| 534332-II | ||||||||||
| 53 | Rb2[(UO2)2(MoO4)O2] | P21/c | 8.542(1)/90 | 15.360(2)/104.279(3) | 8.436(1)/90 | SS[1000] | 3.700/192.423 | 3.907/234.413 | [82] | |
| 54 | K2(UO2)2(MoO4)O2 | P21/c | 8.2498(9)/90 | 15.337(2)/104.748(5) | 8.3514(9)/90 | Flx[950] | 3.700/192.423 | 3.907/234.413 | [83] | |
| Layers with 2D linkage of uranyl polyhedra | ||||||||||
| 584536 | ||||||||||
| 55 | K8[(UO2)8(MoO5)3O6] | P4/n | 23.488(3)/90 | 23.488(3)/90 | 6.7857(11)/90 | Flx[950] | 4.682/908.383 | 4.891/1105.360 | [83] | |
| 584335 | ||||||||||
| 56 | Tl2[(UO2)2O(MoO5)] | P21/n | 8.2527(3)/90 | 28.5081(12)/104.122(1) | 9.1555(4)/90 | SS[650] | 4.700/488.846 | 4.907/588.827 | [84] | |
| FRAMEWORKS | ||||||||||
| Vertex-sharing interpolyhedral linkage | ||||||||||
| 57 | Ca[(UO2)6(MoO4)7(H2O)2](H2O)7.6 | C2221 | 11.3691(9)/90 | 20.0311(5)/90 | 23.8333(18)/90 | HT[230] | 4.800/527.95 | 5.800/1275.899 | [85] | |
| 57a | Ca((UO2)6(MoO4)7(H2O)2)(H2O)15 ** | C2221 | 11.3691(9)/90 | 20.0311(15)/90 | 23.8333(18)/90 | HT[230] | 4.800/527.95 | 6.016/1540 | [85] | |
| 58 | α-Cs2[(UO2)2(MoO4)3] | Pna21 | 20.4302(15)/90 | 8.5552(7)/90 | 9.8549(7)/90 | HT[180] | 4.492/368.955 | 4.524/416.168 | [73] | |
| 59 | Rb2[(UO2)2(MoO4)3] | Pna21 | 20.214(1)/90 | 8.3744(4)/90 | 9.7464(5)/90 | SS[700] | 4.492/368.955 | 4.524/416.168 | [59] | |
| 60 | Tl2[(UO2)2(MoO4)3] | Pna21 | 20.1296(9)/90 | 8.2811(4)/90 | 9.7045(4)/90 | SS[600] | 4.492/368.955 | 4.524/416.168 | [86] | |
| 61 | α-[(UO2)(MoO4)(H2O)2] | P21/c | 13.612(5)/90 | 11.005(4)/113.05(3) | 10.854(3)/90 | HT[190] | 4.170/300.235 | 4.755/513.528 | [87] | |
| 62 | Sr[(UO2)6(MoO4)7(H2O)15] | C2221 | 11.166(5)/90 | 20.281(10)/90 | 24.061(12)/90 | HT | 4.800/527.95 | 5.311/828.523 | [88] | |
| 63 | Mg[(UO2)6(MoO4)7](H2O)18 | C2221 | 11.313(5)/90 | 20.163(10)/90 | 23.877(11)/90 | HT | 4.800/527.95 | 5.416/909.909 | [88] | |
| 64 | (NH4)2[(UO2)6(MoO4)7(H2O)2] | Pbcm | 13.970(1)/90 | 10.747(1)/90 | 25.607(2)/90 | HT[180] | 4.945/1087.899 | 5.196/1371.720 | [89] | |
| 65 | Rb2[(UO2)6(MoO4)7(H2O)2] | Pbcm | 13.961(2)/90 | 10.752(2)/90 | 25.579(4)/90 | HT[230] | 4.945/1087.899 | 5.192/1349.816 | [59] | |
| 66 | Cs2[(UO2)6(MoO4)7(H2O)2] | Pbcm | 13.990(2)/90 | 10.808(1)/90 | 25.671(3)/90 | HT[230] | 4.945/1087.899 | 5.192/1349.816 | [89] | |
| 67 | Ag2[(UO2)6(MoO4)7(H2O)2](H2O)2 | Pbcm | 14.1309(9)/90 | 10.6595(7)/90 | 25.8281(16)/90 | HT[220] | 4.945/1087.899 | 5.264/1431.790 | [90] | |
| 68 | K2.98[(UO2)6(OH)2(MoO4)6(MoO3OH)] | Pbcm | 13.9807(12)/90 | 10.7427(9)/90 | 25.517(2)/90 | SS[900]/HP | 4.945/1087.899 | 5.152/1298.275 | [5] | |
| 69 | Na2UO2(MoO4)2(H2O) | P212121 | 8.6005(16)/90 | 10.749(2)/90 | 11.086(2)/90 | HT[200] | 3.700/192.423 | 4.170/200.235 | [91] | |
| 70 | Ba[(UO2)3(MoO4)4(H2O)4] | Pbca | 17.797(8)/90 | 11.975(6)/90 | 23.33(1)/90 | HT | 4.907/1177.654 | 5.285/1649.045 | [92] | |
| 71 | Mg[(UO2)3(MoO4)4(H2O)8] | Cmc21 | 17.105(8)/90 | 13.786(6)/90 | 10.908(5)/90 | HT[240] | 4.115/255.160 | 4.996/579.526 | [93] | |
| 72 | Zn[(UO2)3(MoO4)4(H2O)8] | Cmc21 | 17.056(8)/90 | 13.786(6)/90 | 10.919(5)/90 | HT[240] | 4.115/255.160 | 4.752/456.156 | [93] | |
| 73 | (NH4)4[(UO2)5(MoO4)7](H2O)5 | P61 | 11.4067(5)/90 | 11.4067(5)/90 | 70.659(5)/120 | HT[180] | 5.644/1693.157 | 6.066/2438.568 | [94] | |
| 74 | [(C2H5)2NH2]2[(UO2)4(MoO4)5(H2O)](H2O) | P6522 | 11.3612(13)/90 | 11.3612(13)/90 | 52.698(8)/120 | HT[220] | 4.524/1248.503 | 4.807/1615.271 | [95] | |
| 75 | [(UO2)(MoO4)] | P21/c | 7.202(3)/90 | 5.484(2)/104.54(2) | 13.599(2)/90 | 3.000/96.000 | 3.000/96.000 | [96] | ||
| Edge-sharing interpolyhedral linkage | ||||||||||
| 76 | K2[(UO2)2(Mo(VI)4Mo(IV)(OH)2)O16] | P-1 | 5.0468(9)/88.456(10) | 6.8982(7)/69.815(15) | 7.2414(12)/69.493(12) | SS[900]/HP | 3.125/50.000 | 2.974/44.603 | [5] | |
| 77 | Li4[(UO2)10O10(Mo2O8)] | P21/c | 7.9426(4)/90 | 19.9895(9)/90.575(2) | 10.0796(5)/90 | SS[870] | 4.684/468.386 | 4.792/517.528 | [97] | |
| 78 | K5[(UO2)10MoO5O11OH]H2O | P1 | 8.0728(5)/111.118(6) | 11.0224(7)/102.845(5) | 11.4744(6)/104.506(6) | SS[900]/HP | 5.585/268.078 | 5.728/303.580 | [5] | |
3. Results and Discussion
3.1. Uranyl Molybdate Minerals
3.2. Synthetic Uranyl Molybdates
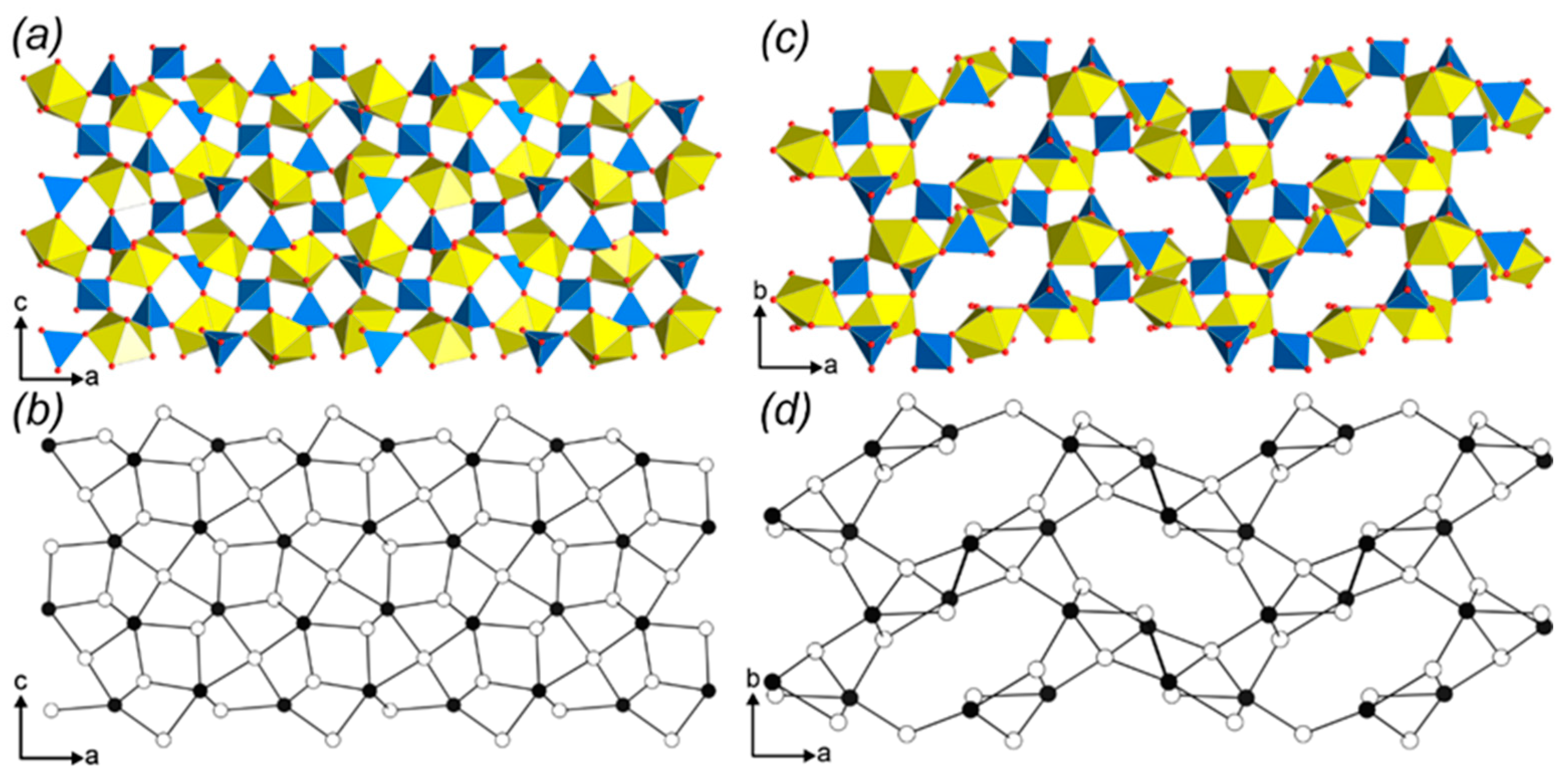
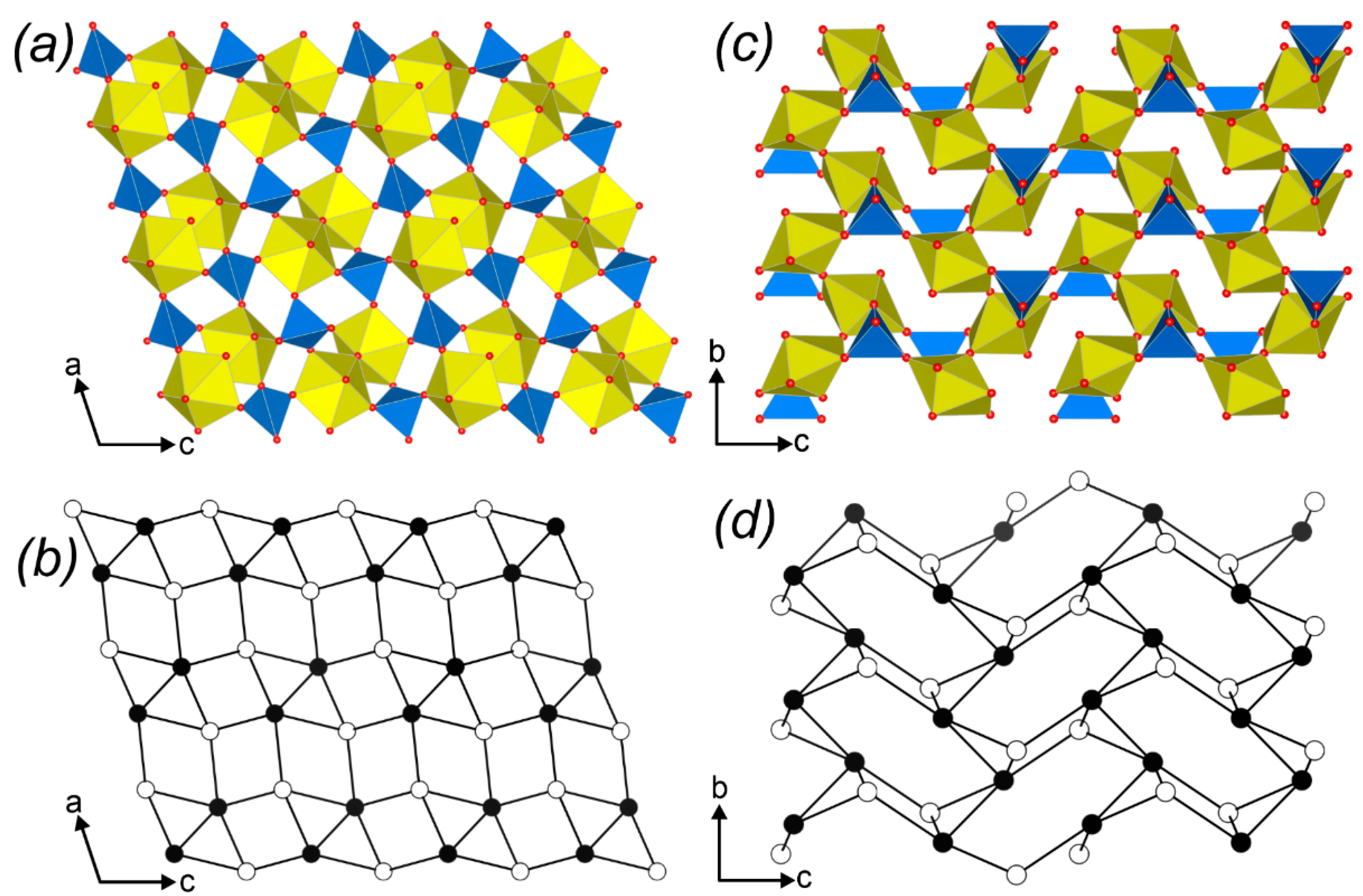
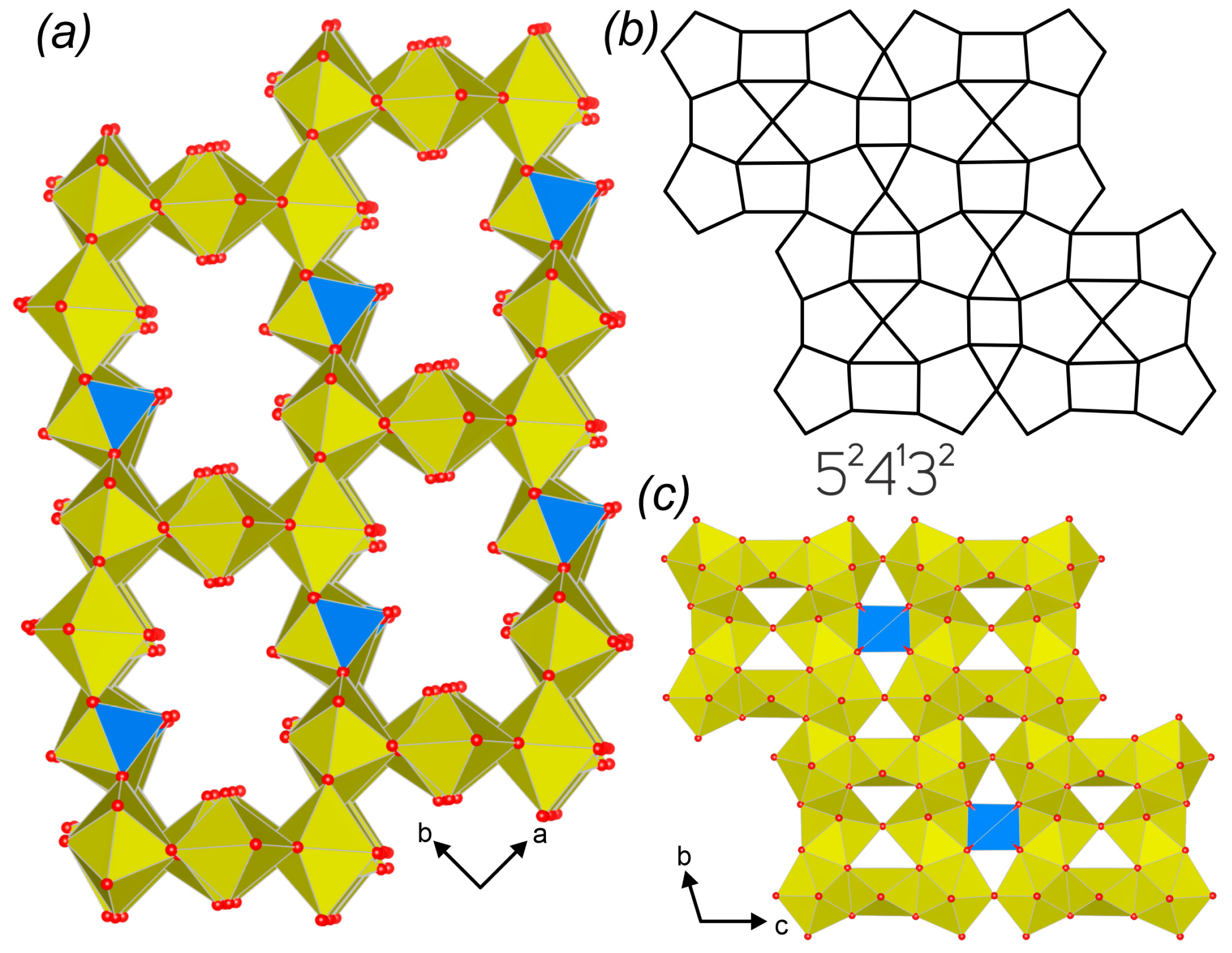
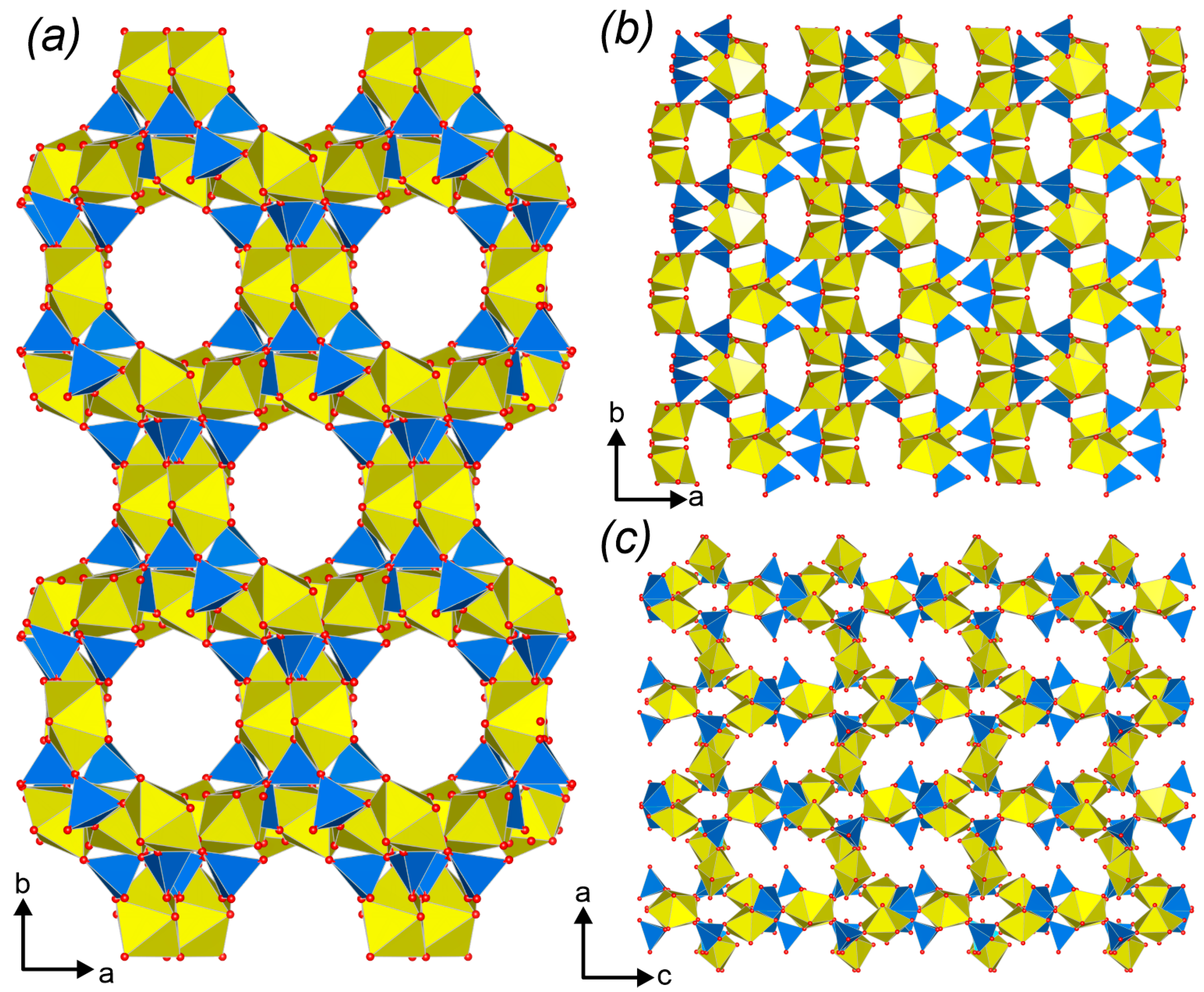
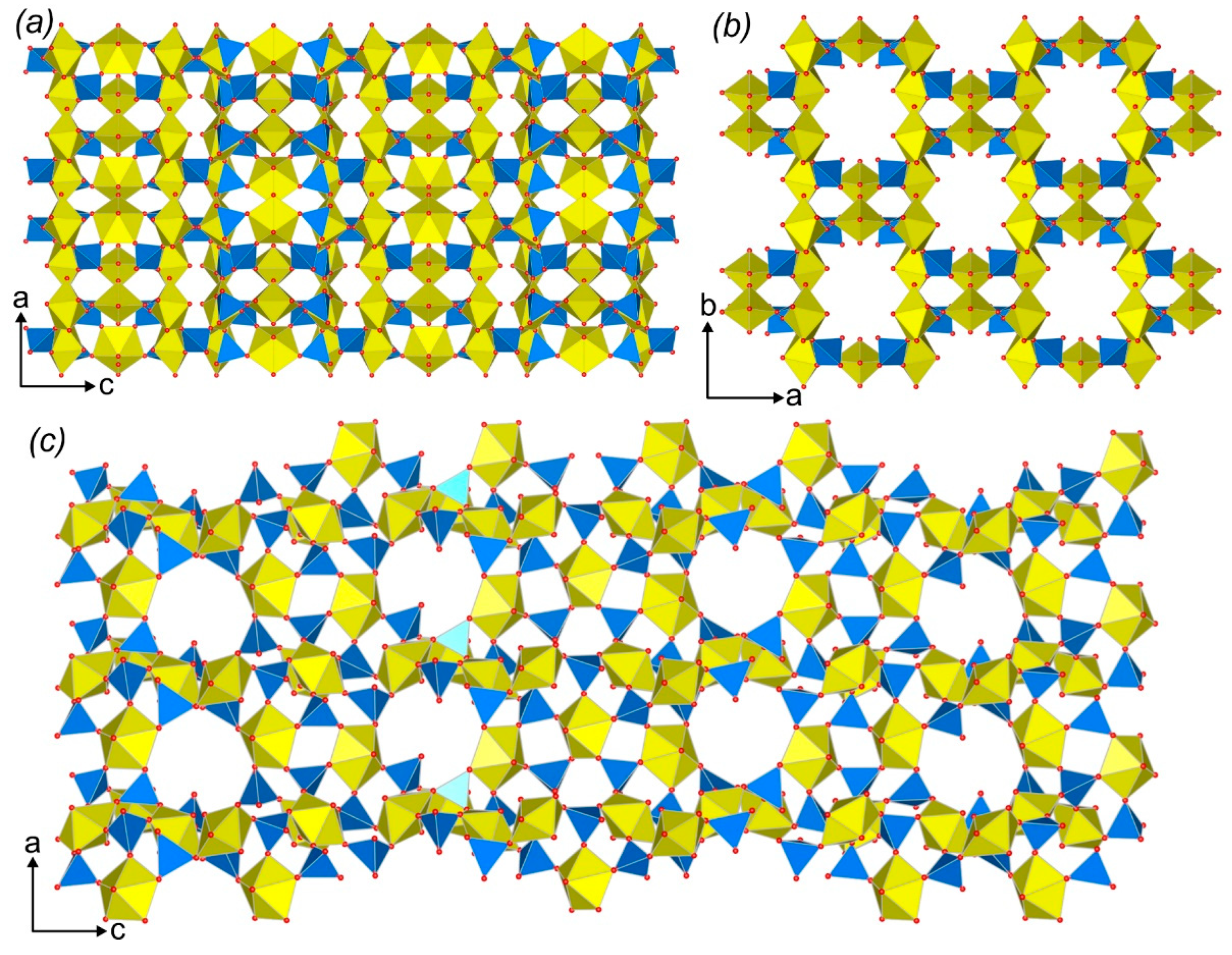
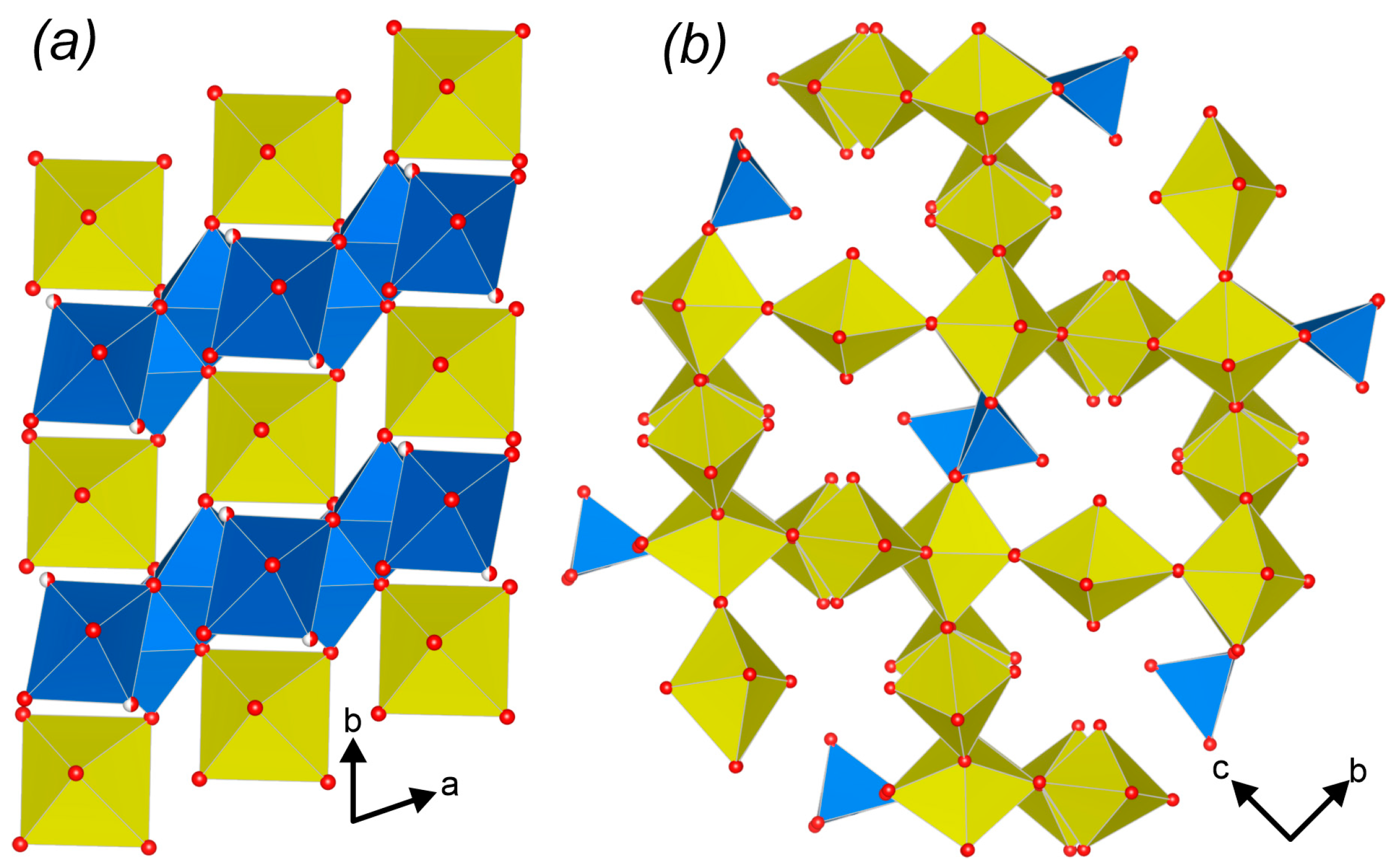
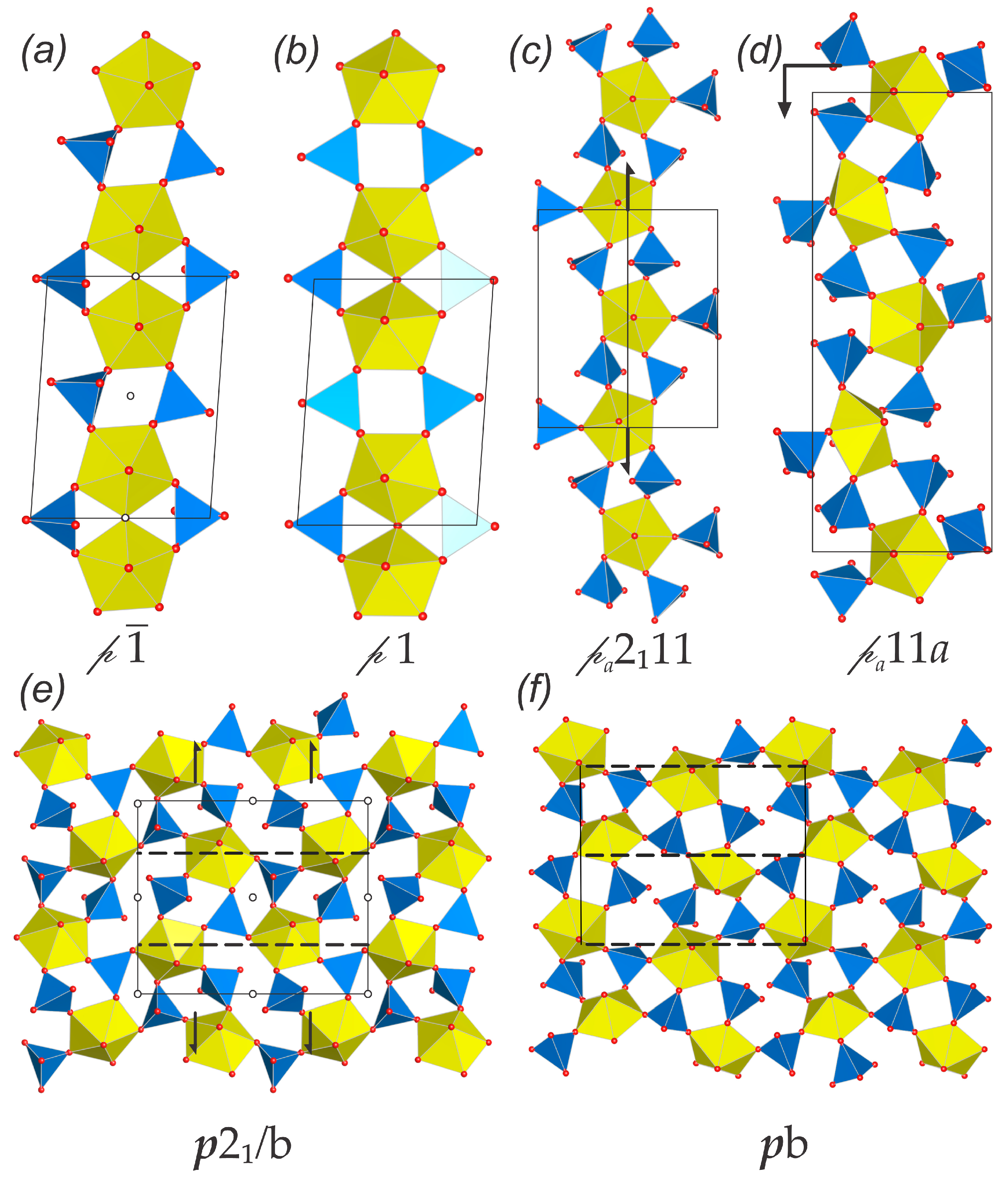
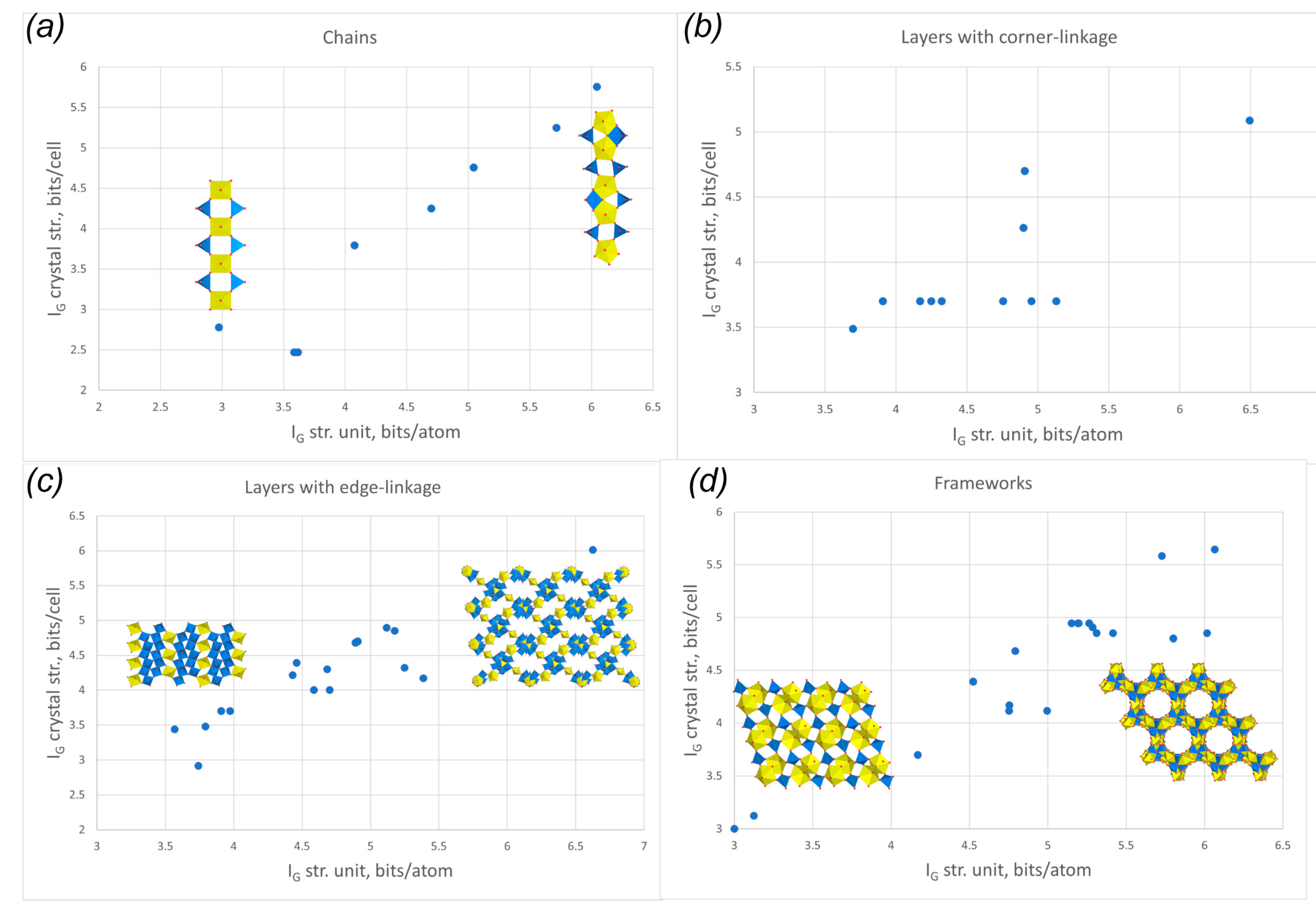
3.3. Topological Analysis
3.4. Crystal Structures vs. Synthesis Conditions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Basiev, T.T.; Sobol, A.A.; Voronko, Y.K.; Zverev, P.G. Spontaneous Raman Spectroscopy of Tungstate and Molybdate Crystals for Raman Lasers. Opt. Mater. 2000, 15, 205–216. [Google Scholar] [CrossRef]
- Dorzhieva, S.G.; Sofich, D.O.; Bazarov, B.G.; Shendrik, R.Y.; Bazarova, J.G. Optical Properties of Molybdates Containing a Combination of Rare-Earth Elements. Inorg. Mater. 2021, 57, 54–59. [Google Scholar] [CrossRef]
- Malavasi, L.; Fisher, C.A.J.; Islam, M.S. Oxide-ion and proton conducting electrolyte materials for clean energy applications: Structural and mechanistic features. Chem. Soc. Rev. 2010, 39, 4370–4387. [Google Scholar] [CrossRef] [PubMed]
- Lind, C.; VanDerveer, D.G.; Wilkinson, A.P.; Chen, J.H.; Vaughan, M.T.; Weidner, D.J. New high-pressure form of the negative thermal expansion materials zirconium molybdate and hafnium molybdate. Chem. Mater. 2001, 13, 487–490. [Google Scholar] [CrossRef]
- Murphy, G.L.; Kegler, P.; Klinkenberg, M.; Wang, S.; Alekseev, E.V. Extreme condition high temperature and high pressure studies of the K–U–Mo–O system. Dalton Trans. 2020, 49, 15843–15853. [Google Scholar] [CrossRef] [PubMed]
- Finch, R.J.; Murakami, T. Systematics and Paragenesis of Uranium Minerals. In Uranium: Mineralogy, Geochemistry and the Environment; Burns, P.C., Finch, R.J., Eds.; Reviews in Mineralogy; Mineralogical Society of America: Chantilly, VA, USA, 1999; Volume 38, pp. 91–180. [Google Scholar]
- Krivovichev, S.V.; Burns, P.C. Crystal chemistry of uranyl molybdates. III. New structural themes in Na6[(UO2)2O(MoO4)4], Na6[(UO2)(MoO4)4] and K6[(UO2)2O(MoO4)4]. Can. Mineral. 2001, 39, 197–206. [Google Scholar] [CrossRef]
- Buck, E.C.; Wronkiewicz, D.J.; Finn, P.A.; Bates, J.K. A new uranyl oxide hydrate phase derived from spent fuel alteration. J. Nucl. Mater. 1997, 249, 70–76. [Google Scholar] [CrossRef]
- Buck, E.C.; Wronkiewicz, D.J. Uranium Mineralogy and the Geologic Disposal of Spent Nuclear Fuel. Rev. Mineral. Geochem. 1999, 38, 474–497. [Google Scholar]
- Lafuente, B.; Downs, R.T.; Yang, H.; Stone, N. The power of databases: The RRUFF project. In Highlights in Mineralogical Crystallography; Armbruster, T., Danisi, R.M., Eds.; De Gruyter: Berlin, Germany, 2015; pp. 1–30. [Google Scholar]
- Burns, P.C.; Miller, M.L.; Ewing, R.C. U6+ minerals and inorganic phases: A comparison and hierarchy of structures. Can. Mineral. 1996, 34, 845–880. [Google Scholar]
- Lussier, A.J.; Lopez, R.A.K.; Burns, P.C. A revised and expanded structure hierarchy of natural and synthetic hexavalent uranium compounds. Can. Mineral. 2016, 54, 177–283. [Google Scholar] [CrossRef]
- Hawthorne, F.C. Graphical enumeration of polyhedral clusters. Acta Crystallogr. A 1983, 39, 724–736. [Google Scholar] [CrossRef]
- Krivovichev, S.V. Combinatorial topology of salts of inorganic oxoacids: Zero-, one- and two-dimensional units with corner-sharing between coordination polyhedra. Crystallogr. Rev. 2004, 10, 185–232. [Google Scholar] [CrossRef]
- Krivovichev, S.V. Structural Crystallography of Inorganic Oxysalts; Oxford University Press: Oxford, UK, 2008; p. 303. [Google Scholar]
- Krivovichev, S.V. Topological complexity of crystal structures: Quantitative approach. Acta Crystallogr. A 2012, 68, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Krivovichev, S.V. Structural complexity of minerals: Information storage and processing in the mineral world. Mineral. Mag. 2013, 77, 275–326. [Google Scholar] [CrossRef]
- Krivovichev, S.V. Which inorganic structures are the most complex? Angew. Chem. Int. Ed. 2014, 53, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Krivovichev, S.V. Structural complexity of minerals and mineral parageneses: Information and its evolution in the mineral world. In Highlights in Mineralogical Crystallography; Danisi, R., Armbruster, T., Eds.; Walter de Gruyter: Berlin, Germany, 2015; pp. 31–73. [Google Scholar]
- Krivovichev, S.V. Structural complexity and configurational entropy of crystalline solids. Acta Crystallogr. B 2016, 72, 274–276. [Google Scholar] [CrossRef] [PubMed]
- Gurzhiy, V.V.; Tyumentseva, O.S.; Izatulina, A.R.; Krivovichev, S.V.; Tananaev, I.G. Chemically Induced Polytypic Phase Transitions in the Mg[(UO2)(TO4)2(H2O)](H2O)4 (T = S, Se) System. Inorg. Chem. 2019, 58, 14760–14768. [Google Scholar] [CrossRef] [PubMed]
- Gurzhiy, V.V.; Tyumentseva, O.S.; Belova, E.V.; Krivovichev, S.V. Chemically induced symmetry breaking in the crystal structure of guanidinium uranyl sulfate. Mendeleev Commun. 2019, 29, 408–410. [Google Scholar] [CrossRef]
- Tyumentseva, O.S.; Kornyakov, I.V.; Britvin, S.N.; Zolotarev, A.A.; Gurzhiy, V.V. Crystallographic Insights into Uranyl Sulfate Minerals Formation: Synthesis and Crystal Structures of Three Novel Cesium Uranyl Sulfates. Crystals 2019, 9, 660. [Google Scholar] [CrossRef]
- Kornyakov, I.V.; Kalashnikova, S.A.; Gurzhiy, V.V.; Britvin, S.N.; Belova, E.V.; Krivovichev, S.V. Synthesis, characterization and morphotropic transitions in a family of M[(UO2)(CH3COO)3](H2O)n (M = Na, K, Rb, Cs; n = 0–1.0) compounds. Z. Kristallogr. 2020, 235, 95–103. [Google Scholar] [CrossRef]
- Kornyakov, I.V.; Tyumentseva, O.S.; Krivovichev, S.V.; Gurzhiy, V.V. Dimensional evolution in hydrated K+-bearing uranyl sulfates: From 2D-sheets to 3D frameworks. Cryst. Eng. Comm. 2020, 22, 4621–4629. [Google Scholar] [CrossRef]
- Gurzhiy, V.V.; Plasil, J. Structural complexity of natural uranyl sulfates. Acta Crystallogr. B 2019, 75, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Gurzhiy, V.V.; Kuporev, I.V.; Kovrugin, V.M.; Murashko, M.N.; Kasatkin, A.V.; Plášil, J. Crystal chemistry and structural complexity of natural and synthetic uranyl selenites. Crystals 2019, 9, 639. [Google Scholar] [CrossRef]
- Krivovichev, V.G.; Krivovichev, S.V.; Charykova, M.V. Selenium Minerals: Structural and Chemical Diversity and Complexity. Minerals 2019, 9, 455. [Google Scholar] [CrossRef]
- Gurzhiy, V.V.; Kalashnikova, S.A.; Kuporev, I.V.; Plášil, J. Crystal Chemistry and Structural Complexity of the Uranyl Carbonate Minerals and Synthetic Compounds. Crystals 2021, 11, 704. [Google Scholar] [CrossRef]
- Blatov, V.A.; Shevchenko, A.P.; Proserpio, D.M. Applied topological analysis of crystal structures with the program package ToposPro. Cryst. Growth. Des. 2014, 14, 3576–3586. [Google Scholar] [CrossRef]
- Sarp, H.; Chiappero, P.J. Deloryite, Cu4(UO2)(MoO4)2(OH)6, a new mineral from the Cap Garonne mine near Le Pradet, Var, France. Neues Jahrb. Mineral. Monatshefte 1992, 2, 58–64. [Google Scholar]
- Tali, R.; Tabachenko, V.V.; Kovba, L.M. Crystal structure of Cu4UO2(MoO4)2(OH)6. Russ. J. Inorg. Chem. 1993, 38, 1350–1352. [Google Scholar]
- Pushcharovsky, D.Y.; Rastsvetaeva, R.K.; Sarp, H. Crystal structure of deloryite, Cu4(UO2)[Mo2O8](OH)6. J. Alloys Compd. 1996, 239, 23–26. [Google Scholar] [CrossRef]
- Brophy, G.P.; Kerr, P.F. Hydrous Uranium Molybdate in Maryvale Ore; Annual Report for June 30, 1952 to April 1, 1953 RME-3046; U.S. Atomic Energy Commission: Oak Ridge, Tennessee, 1953; pp. 45–51. [Google Scholar]
- Coleman, R.G.; Appleman, D.E. Umohoite from the Lucky MC mine, Wyoming. Am. Mineral. 1957, 42, 657–660. [Google Scholar]
- Skvortsova, K.V.; Kopchenova, E.V.; Silant’eva, N.I.; Sidorenko, G.A.; Dara, A.D. On the conditions of umohoite formation on U.S.S.R. uranium-molybdenum deposits. Geol. Ore Depos. 1961, 5, 53–63. [Google Scholar]
- Krivovichev, S.V.; Burns, P.C. Crystal chemistry of uranyl molybdates. I. The structure and formula of umohoite. Can. Mineral. 2000, 38, 717–726. [Google Scholar] [CrossRef]
- Sidorenko, G.A.; Rastsvetaeva, R.K.; Chukanov, N.V.; Chistyakova, N.I.; Barinova, A.V. Umohoite: New data on its mineralogy and crystal-chemistry. Zap. Vseross. Mineral. Obs. 2003, 132, 73–80. [Google Scholar]
- Rastsvetaeva, R.K.; Barinova, A.V.; Sidorenko, G.A.; Pushcharovskii, D.Y. Crystal structure of triclinic umohoite [UMoO6(H2O)](H2O). Dokl. Akad. Nauk 2000, 373, 202–205. [Google Scholar]
- Makarov, E.S.; Anikina, L.I. Crystal structure of umohoite (UMoO6(H2O)2)·2H2O. Geochemistry 1963, 1, 14–21. [Google Scholar]
- Soboleva, M.V.; Pudovkina, I.A. Iriginite. In Uranium Minerals Handbook; Gosgeoltekhizdat: Moscow, Russia, 1957; pp. 249–251. (In Russian) [Google Scholar]
- Epshtein, G.Y. On the molybdates of uranium—Moluranite and iriginite. Zap. Vsesoyuznogo Mineral. Obs. 1959, 88, 564–570. (In Russian) [Google Scholar]
- Serezhkin, V.N.; Chuvaev, V.F.; Kovba, L.M.; Trunov, V.K. The structure of synthetic iriginite. Dokl. Akad. Nauk SSSR 1973, 210, 873–876. (In Russian) [Google Scholar]
- Krivovichev, S.V.; Burns, P.C. The crystal chemistry of uranyl molybdates. II. The crystal structure of iriginite. Can. Mineral. 2000, 38, 847–851. [Google Scholar] [CrossRef]
- Elliott, P.; Plášil, J.; Petříček, V.; Čejka, J.; Bindi, L. Twinning and incomensurate modulation in baumoite, Ba0.5[(UO2)3O8Mo2(OH)3](H2O)~3, the first natural Ba uranyl molybdate. Mineral. Mag. 2019, 83, 507–514. [Google Scholar] [CrossRef]
- Chen, Z.; Luo, K.; Tan, F.; Zhang, Y.; Gu, X. Tengchongite, a new mineral of hydrated calcium uranyl molybdate. Kexue Tongbao 1986, 31, 396–401. [Google Scholar]
- Li, T.; Fan, G.; Ge, X.; Wang, T.; Yu, A.; Deng, L. Crystal structure of tengchongite with a revised chemical formula Ca(UO2)6(MoO4OH)2O2(OH)4·9H2O. Can. Mineral. 2022, 60, 533–542. [Google Scholar] [CrossRef]
- Soboleva, M.V.; Pudovkina, I.A. Moluranite. In Uranium Minerals Handbook; Gosgeoltekhizdat: Moscow, Russia, 1957; pp. 252–253. [Google Scholar]
- Kopchenova, E.V.; Skvortsova, K.V.; Silantieva, N.I.; Sidorenko, G.A.; Mikhailova, L.V. Mourite, a new supergene uranium-molybdenum mineral. Zap. Vsesoyuznogo Mineral. Obs. 1962, 91, 67–71. [Google Scholar]
- Smith, M.L.; Marinenko, J.; Weeks, A.D. Comparison of mourite from Karnes County, Texas with mourite from the U.S.S.R. Am. Mineral. 1971, 56, 163–173. [Google Scholar]
- Rudnitskaya, L.S. Calcium molybdate of uranium. Yad. Goryuchee I Reakt. Met. 1959, 3, 160–161. [Google Scholar]
- Fedorov, O.V. The second finding of the calcium molybdate of uranium in the USSR. Zap. Vsesoyuznogo Mineral. Obs. 1963, 92, 464–465. [Google Scholar]
- Sidorenko, G.A.; Chistyakova, N.I.; Chukanov, N.V.; Naumova, I.S.; Rassulov, V.A. Carcurmolite: New data on chemical composition and constitution the mineral. New Data Miner. 2005, 40, 29–36. [Google Scholar]
- Frost, R.L.; Čejka, J.; Dickfos, M.J. Raman and infrared spectroscopic study of the molybdate-containing uranyl mineral calcurmolite. J. Raman Spectrosc. 2008, 39, 779–785. [Google Scholar] [CrossRef]
- Vaes, J.F. Cousiniet, een nieuw uraanmineraal. Geol. Mijnb. 1958, 20, 449. [Google Scholar]
- Deliens, M. Une association de molybdates d’uranium de Shinkolobwe (Région du Shaba, République du Zaire). Ann. Soc. Géol. Belg. 1975, 98, 155–160. [Google Scholar]
- Skvortsova, K.V.; Sidorenko, G.A. Sedovite, a new supergene mineral of uranium and molybdenum. Zap. Vsesoyuznogo Mineral. Obs. 1965, 94, 548–554. [Google Scholar]
- Krivovichev, S.V.; Burns, P.C. Crystal chemistry of uranyl molybdates. VI. New uranyl molybdate units in the structures of Cs4[(UO2)3O(MoO4)2(MoO5)] and Cs6[(UO2)(MoO4)4]. Can. Mineral. 2002, 40, 201–209. [Google Scholar] [CrossRef]
- Krivovichev, S.V.; Burns, P.C. Crystal Chemistry of Rubidium Uranyl Molybdates: Crystal Structures of Rb6[(UO2)(MoO4)4], Rb6[(UO2)2O(MoO4)4], Rb2[(UO2)(MoO4)2], Rb2[(UO2)2(MoO4)3] and Rb2[(UO2)6(MoO4)7(H2O)2. J. Solid State Chem. 2002, 168, 245–258. [Google Scholar] [CrossRef]
- Krivovichev, S.V.; Burns, P.C. Crystal chemistry of uranyl molybdates. VIII. Crystal structures of Na3Tl3[(UO2)(MoO4)4], Na13–xTl3+x[(UO2)(MoO4)3]4(H2O)6+x (x = 0.1), Na3Tl5[(UO2)(MoO4)3]2(H2O)3 and Na2[(UO2)(MoO4)2](H2O)4. Can. Mineral. 2003, 41, 707–719. [Google Scholar] [CrossRef]
- Alekseev, E.V.; Krivovichev, S.V.; Armbruster, T.; Depmeier, W.; Suleimanov, E.V.; Chuprunov, E.V.; Golubeva, A.V. Dimensional reduction in alkali metal uranyl molybdates: Synthesis and structure of Cs2[(UO2)O(MoO4)]. Z. Anorg. Allg. Chem. 2007, 633, 1979–1984. [Google Scholar] [CrossRef]
- Krivovichev, S.V.; Burns, P.C. Synthesis and crystal structure of Li2[(UO2)(MoO4)2], a uranyl molybdate with chains of corner-sharing uranyl square bipyramids and MoO4 tetrahedra. Solid State Sci. 2003, 5, 481–485. [Google Scholar] [CrossRef]
- Yagoubi, S.; Obbade, S.; Saad, S.; Abraham, F. From ∞1[(UO2)2O(MoO4)4]6− to ∞1[(UO2)2(MoO4)3(MoO5)]6− infinite chains in A6U2Mo4O21 (A = Na, K, Rb, Cs) compounds: Synthesis and crystal structure of Cs6[(UO2)2(MoO4)3(MoO5)]. J. Solid State Chem. 2011, 184, 971–981. [Google Scholar] [CrossRef]
- Sykora, R.E.; McDaniel, S.M.; Wells, D.M.; Albrecht-Schmitt, T.E. Mixed-Metal Uranium(VI) Iodates: Hydrothermal Syntheses, Structures, and Reactivity of Rb[UO2(CrO4)(IO3)(H2O)], A2[UO2(CrO4)(IO3)2] (A = K, Rb, Cs), and K2[UO2(MoO4)(IO3)2. Inorg. Chem. 2002, 41, 5126–5132. [Google Scholar] [CrossRef] [PubMed]
- Krivovichev, S.V.; Burns, P.C. Crystal chemistry of uranyl molybdates. XI. Crystal structures of Cs2[(UO2)(MoO4)2] and Cs2[(UO2)(MoO4)2](H2O). Can. Mineral. 2005, 43, 713–720. [Google Scholar] [CrossRef]
- Rastsvetaeva, R.K.; Barinova, A.V.; Fedoseev, A.M.; Budantseva, N.A. Synthesis and crystal structure of a new hydrated cesium uranyl molybdate Cs2UO2(MoO4)2(H2O). Dokl. Akad. Nauk 1999, 365, 68–71. (In Russian) [Google Scholar]
- Sadikov, G.G.; Krasovskaya, T.I.; Polyakov, Y.A.; Nikolaev, V.P. Structure and spectral analysis of potassium uranyl dimolybdate. Izv. Akad. Nauk SSSR Neorg. Mater. 1988, 24, 109–115. (In Russian) [Google Scholar]
- Krivovichev, S.V.; Finch, R.J.; Burns, P.C. Crystal chemistry of uranyl molybdates. V. Topologically distinct uranyl dimolybdate sheets in the structures of Na2[(UO2)(MoO4)2] and K2[(UO2)(MoO4)2](H2O). Can. Mineral. 2002, 40, 193–200. [Google Scholar] [CrossRef]
- Krivovichev, S.V.; Burns, P.C. Combinatorial topology of uranyl molybdate sheets: Syntheses and crystal structures of (C6H14N2)3[(UO2)5(MoO4)8](H2O)4 and (C2H10N2)[(UO2)(MoO4)2]. J. Solid State Chem. 2003, 170, 106–117. [Google Scholar] [CrossRef]
- Khrustalev, V.N.; Andreev, G.B.; Antipin, M.Y.; Fedoseev, A.M.; Budantseva, N.A.; Shirokova, I.B. Synthesis and crystal structure of rubidium uranyl dimolybdate Rb2UO2(MoO4)2(H2O). Zhurnal Neorg. Khimii 2000, 45, 1996–1998. (In Russian) [Google Scholar]
- Krivovichev, S.V.; Locock, A.J.; Burns, P.C. Lone electron pair stereoactivity, cation arrangements and distortion of heteropolyhedral sheets in the structures of Tl2[(UO2)(AO4)2] (A = Cr, Mo). Z. Kristallogr. 2005, 220, 10–18. [Google Scholar] [CrossRef]
- Shiv Halasyamani, P.; Francis, R.J.; Walker, S.M.; O’Hare, D. New Layered Uranium(VI) Molybdates: Syntheses and Structures of (NH3(CH2)3NH3)(H3O)2(UO2)3(MoO4)5, C(NH2)3(UO2)(OH)(MoO4), (C4H12N2)(UO2)(MoO4)2, and (C5H14N2)(UO2)(MoO4)2(H2O). Inorg. Chem. 1999, 38, 271–279. [Google Scholar] [CrossRef]
- Krivovichev, S.V.; Cahill, C.L.; Burns, P.C. Syntheses and Crystal Structures of Two Topologically Related Modifications of Cs2[(UO2)2(MoO4)3]. Inorg. Chem. 2002, 41, 34–39. [Google Scholar] [CrossRef]
- Nazarchuk, E.V.; Charkin, D.; Siidra, O.; Kalmykov, S. Organically Templated Layered Uranyl Molybdate [C3H9NH+]4[(UO2)3(MoO4)5] Structurally Based on Mineral-Related Modular Units. Minerals 2020, 10, 659. [Google Scholar] [CrossRef]
- Lee, M.R.; Jaulmes, S. Nouvelle serie d’oxydes derives de la structure de α-U3U8: MiiUMo4O16. J. Solid State Chem. 1987, 67, 364–368. [Google Scholar] [CrossRef]
- Krivovichev, S.V.; Burns, P.C. Crystal chemistry of uranyl molybdates. VII. An iriginite-type sheet of polyhedra in the structure of [(UO2)Mo2O7(H2O)2]. Can. Mineral. 2002, 40, 1571–1577. [Google Scholar] [CrossRef]
- Krivovichev, S.V.; Burns, P.C. Synthesis and Structure of Ag6[(UO2)3O(MoO4)5]: A Novel Sheet of Triuranyl Clusters and MoO4 Tetrahedra. Inorg. Chem. 2002, 41, 4108–4110. [Google Scholar] [CrossRef]
- Kotvanova, M.K.; Molchanov, V.N.; Torchenkova, E.A.; Spitsyn, V.I. The crystal structure of (NH4)4[(UO2)2(H2O)3UMo12O42) (H2O)18. Zhurnal Neorg. Khimii 1984, 29, 1790–1797. (In Russian) [Google Scholar]
- Nazarchuk, E.V.; Siidra, O.I.; Krivovichev, S.V.; Malcherek, T.; Depmeier, W. First Mixed Alkaline Uranyl Molybdates: Synthesis and Crystal Structures of CsNa3[(UO2)4O4(Mo2O8)] and Cs2Na8[(UO2)8O8(Mo5O20)]. Z. Anorg. Allg. Chem. 2009, 635, 1231–1235. [Google Scholar] [CrossRef]
- Krivovichev, S.V.; Burns, P.C. Crystal chemistry of uranyl molybdates. X. The crystal structure of Ag10[(UO2)8O8(Mo5O20)]. Can. Mineral. 2003, 41, 1455–1462. [Google Scholar] [CrossRef]
- Krivovichev, S.V. K2Na8(UO2)8Mo4O24[(S,Mo)O4], the first uranium molybdosulfate: Synthesis, crystal structure, and comparison to related compounds. J. Geosci. 2014, 59, 115–121. [Google Scholar] [CrossRef]
- Alekseev, E.V.; Suleimanov, E.V.; Chuprunov, E.V.; Golubev, A.V.; Fukin, G.K.; Marychev, M.O. Synthesis and Crystal Structure of a New Representative of the Rb2U2MoO10 Uranomolybdate Series. Russ. J. Inorg. Chem. 2007, 52, 1446–1449. [Google Scholar] [CrossRef]
- Obbade, S.; Yagoubi, S.; Dion, C.; Saadi, M.; Abraham, F. Synthesis, crystal structure and electrical characterization of two new potassium uranyl molybdates K2(UO2)2(MoO4)O2 and K8(UO2)8(MoO5)3O6. J. Solid State Chem. 2003, 174, 19–31. [Google Scholar] [CrossRef]
- Krivovichev, S.V.; Burns, P.C. Crystal chemistry of uranyl molybdates. IX. A novel uranyl molybdate sheet in the structure of Tl2[(UO2)2O(MoO5)]. Can. Mineral. 2003, 41, 1225–1231. [Google Scholar] [CrossRef]
- Nazarchuk, E.V.; Krivovichev, S.V.; Burns, P.C. Crystal structure and phase transformations of Ca[(UO2)6(MoO4)7 (H2O)2](H2O)n (n~7.6). Zap. Ross. Mineral. 2005, 134, 110–117. (In Russian) [Google Scholar]
- Nazarchuk, E.V.; Krivovichev, S.V.; Burns, P.C. Crystal structure of Tl2[(UO2)2(MoO4)3] and crystal chemistry of the compounds M2[(UO2)2(MoO4)3] (M = Tl, Rb, Cs). Radiokhimiya 2005, 47, 408–411. [Google Scholar] [CrossRef]
- Serezhkin, V.N.; Efremov, V.A.; Trunov, V.K. The crystal structure of alpha-UO2MoO4(H2O)2. Kristallografiya 1980, 25, 861–865. (In Russian) [Google Scholar]
- Tabachenko, V.V.; Kovba, L.M.; Serezhkin, V.N. Crystal structures of Mg(UO2)6(MoO4)7(H2O)18 and Sr(UO2)6(MoO4)7(H2O)15. Koord. Khimiya 1984, 10, 558–562. (In Russian) [Google Scholar]
- Krivovichev, S.V.; Burns, P.C. Crystal chemistry of uranyl molybdates. IV. The structures of M2[(UO2)6(MoO4)7(H2O)2] (M = Cs, NH4). Can. Mineral. 2001, 39, 207–214. [Google Scholar] [CrossRef]
- Nazarchuk, E.V.; Siidra, O.I.; Krivovichev, S.V. Synthesis and Crystal Structure of Ag2[(UO2)6(MoO4)7(H2O)2](H2O)2. Radiochemistry 2016, 58, 1–5. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y.; Dai, X.; Liu, W.; Yin, X.; Chen, L.; Zhai, F.; Diwu, J.; Zhang, C.; Zhou, R.; et al. Inorganic X-ray scintillators based on a previously unnoticed but intrinsically advantageous metal center. Inorg. Chem. 2019, 58, 2807–2812. [Google Scholar] [CrossRef] [PubMed]
- Tabachenko, V.V.; Balashov, V.L.; Kovba, L.M.; Serezhkin, V.N. Crystal Structure of Barium Uranyl Molybdate Ba(UO2)3(MoO4)4(H2O)4. Koord. Khimiya 1984, 10, 854–857. (In Russian) [Google Scholar]
- Tabachenko, V.V.; Kovba, L.M.; Serezhkin, V.N. The crystal structure of molybdatouranylates of magnesium and zinc of composition M(UO2)3(MoO4)4(H2O)8 (M = Mg, Zn). Koord. Khimiya 1983, 9, 1568–1571. (In Russian) [Google Scholar]
- Krivovichev, S.V.; Cahill, C.L.; Burns, P.C. A novel open framework uranyl molybdate: Synthesis and structure of (NH4)4((UO2)5(MoO4)7)(H2O)5. Inorg. Chem. 2003, 42, 2459–2464. [Google Scholar] [CrossRef]
- Krivovichev, S.V.; Cahill, C.L.; Nazarchuk, E.V.; Burns, P.C.; Armbruster, T.; Depmeier, W. Chiral open-framework uranyl molybdates. 1. Topological diversity: Synthesis and crystal structure of [(C2H5)2NH2]2[(UO2)4(MoO4)5(H2O)](H2O). Microporous Mesoporous Mater. 2005, 78, 209–215. [Google Scholar] [CrossRef]
- Serezhkin, V.N.; Trunov, V.K.; Makarevich, L.G. The refined crystal structure of uranyl molybdate. Kristallografiya 1980, 25, 858–860. [Google Scholar]
- Alekseev, E.V.; Krivovichev, S.V.; Malcherek, T.; Depmeier, W. One-dimensional array of two- and three-center cation–cation bonds in the structure of Li4[(UO2)10O10(Mo2O8)]. Inorg. Chem. 2007, 46, 8442–8444. [Google Scholar] [CrossRef]
- Smith, D.K. Uranium mineralogy. In Uranium Geochemistry, Mineralogy, Geology, Exploration and Resources; De Vivo, B., Ippolito, F., Capaldi, G., Simpson, P.R., Eds.; Institute of Mining and Metallurgy: London, UK, 1984; pp. 43–88. [Google Scholar]
- Gaines, R.V.; Skinner, H.C.W.; Foord, E.E.; Mason, B.; Rosenzweig, A. Dana’s New Mineralogy, 8th ed.; John Wiley & Sons: New York, NY, USA, 1997. [Google Scholar]
- Obbade, S.; Dion, C.; Bekaert, E.; Yagoubi, S.; Saadi, M.; Abraham, F. Synthesis and crystal structure of new uranyl tungstates M2(UO2)(W2O8) (M=Na, K), M2(UO2)2(WO5)O (M=K, Rb), and Na10(UO2)8(W5O20)O8. J. Solid State Chem. 2003, 172, 305–318. [Google Scholar] [CrossRef]
- Durova, E.V.; Kuporev, I.V.; Gurzhiy, V.V. Organically Templated Uranyl Sulfates and Selenates: Structural Complexity and Crystal Chemical Restrictions for Isotypic Compounds Formation. Int. J. Mol. Sci. 2023, 24, 13020. [Google Scholar] [CrossRef] [PubMed]
- Kovrugin, V.M.; Gurzhiy, V.V.; Krivovichev, S.V. Structural topology and dimensional reduction in uranyl oxysalts: Eight novel phases in the methylamine–(UO2)(NO3)2–H2SeO4–H2O system. Struct. Chem. 2012, 23, 2003–2017. [Google Scholar] [CrossRef]
- Gurzhiy, V.V.; Kornyakov, I.V.; Szymanowski, J.E.S.; Felton, D.; Tyumentseva, O.S.; Krzhizhanovskaya, M.G.; Krivovichev, S.V.; Burns, P.C. Chemically-induced structural variations of a family of Cs2[(AnO2)2(TO4)3] (An = U, Np; T = S, Se, Cr, Mo) compounds: Thermal behavior, calorimetry studies and spectroscopy characterization of Cs uranyl sulfate and selenate. J. Solid State Chem. 2020, 282, 121077–121084. [Google Scholar] [CrossRef]
- Krivovichev, S.V. Ladders of information: What contributes to the structural complexity in inorganic crystals. Z. Kristallogr. 2018, 233, 155–161. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuporev, I.V.; Kalashnikova, S.A.; Gurzhiy, V.V. Crystal Chemistry and Structural Complexity of the Uranyl Molybdate Minerals and Synthetic Compounds. Crystals 2024, 14, 15. https://doi.org/10.3390/cryst14010015
Kuporev IV, Kalashnikova SA, Gurzhiy VV. Crystal Chemistry and Structural Complexity of the Uranyl Molybdate Minerals and Synthetic Compounds. Crystals. 2024; 14(1):15. https://doi.org/10.3390/cryst14010015
Chicago/Turabian StyleKuporev, Ivan V., Sophia A. Kalashnikova, and Vladislav V. Gurzhiy. 2024. "Crystal Chemistry and Structural Complexity of the Uranyl Molybdate Minerals and Synthetic Compounds" Crystals 14, no. 1: 15. https://doi.org/10.3390/cryst14010015
APA StyleKuporev, I. V., Kalashnikova, S. A., & Gurzhiy, V. V. (2024). Crystal Chemistry and Structural Complexity of the Uranyl Molybdate Minerals and Synthetic Compounds. Crystals, 14(1), 15. https://doi.org/10.3390/cryst14010015







