Electrochemical Removal of NOx on Ceria-Based Catalyst-Electrodes
Abstract
:1. Introduction
2. Results and Discussion
2.1. Catalyst-Electrode Characterizations
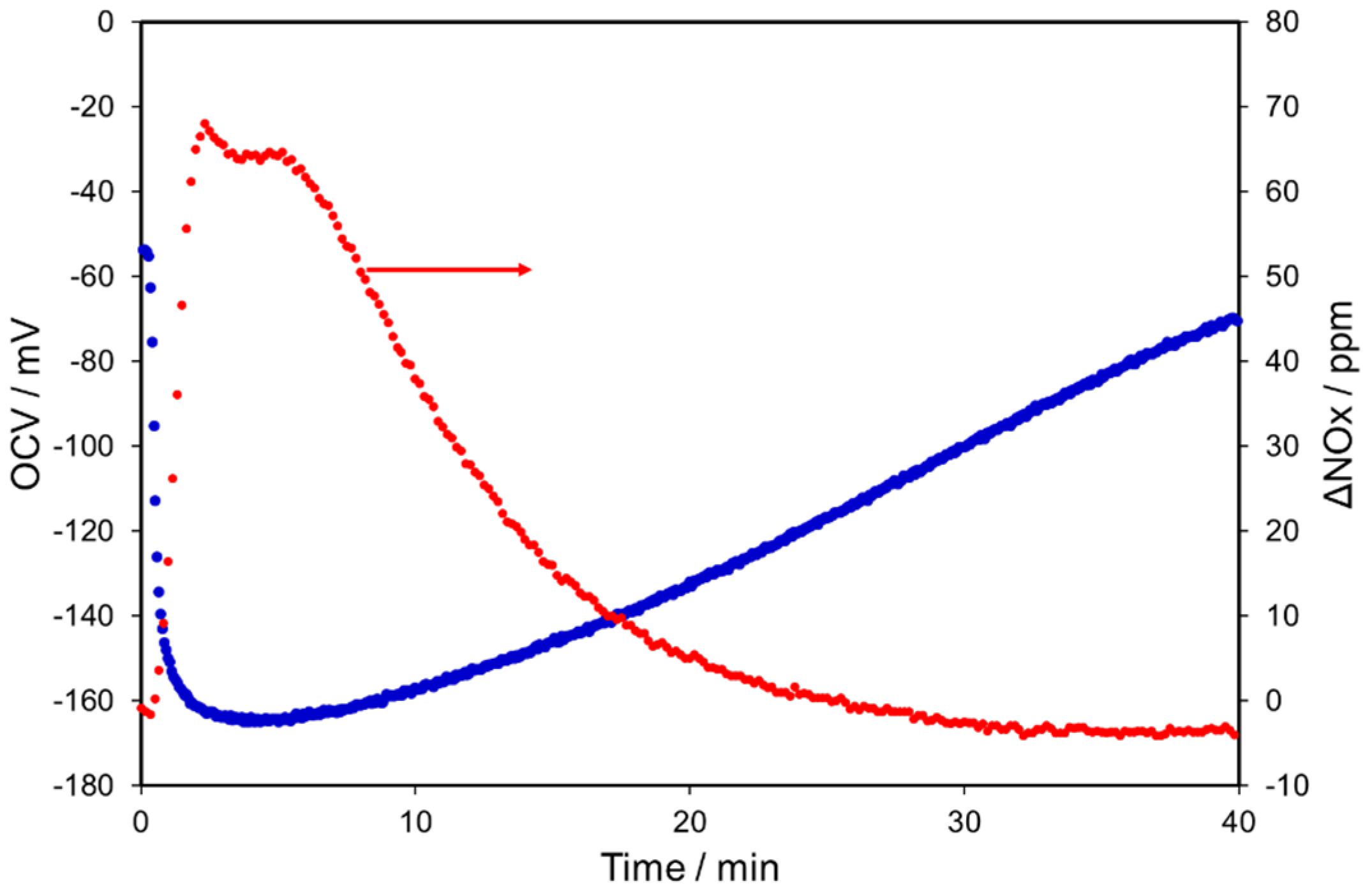
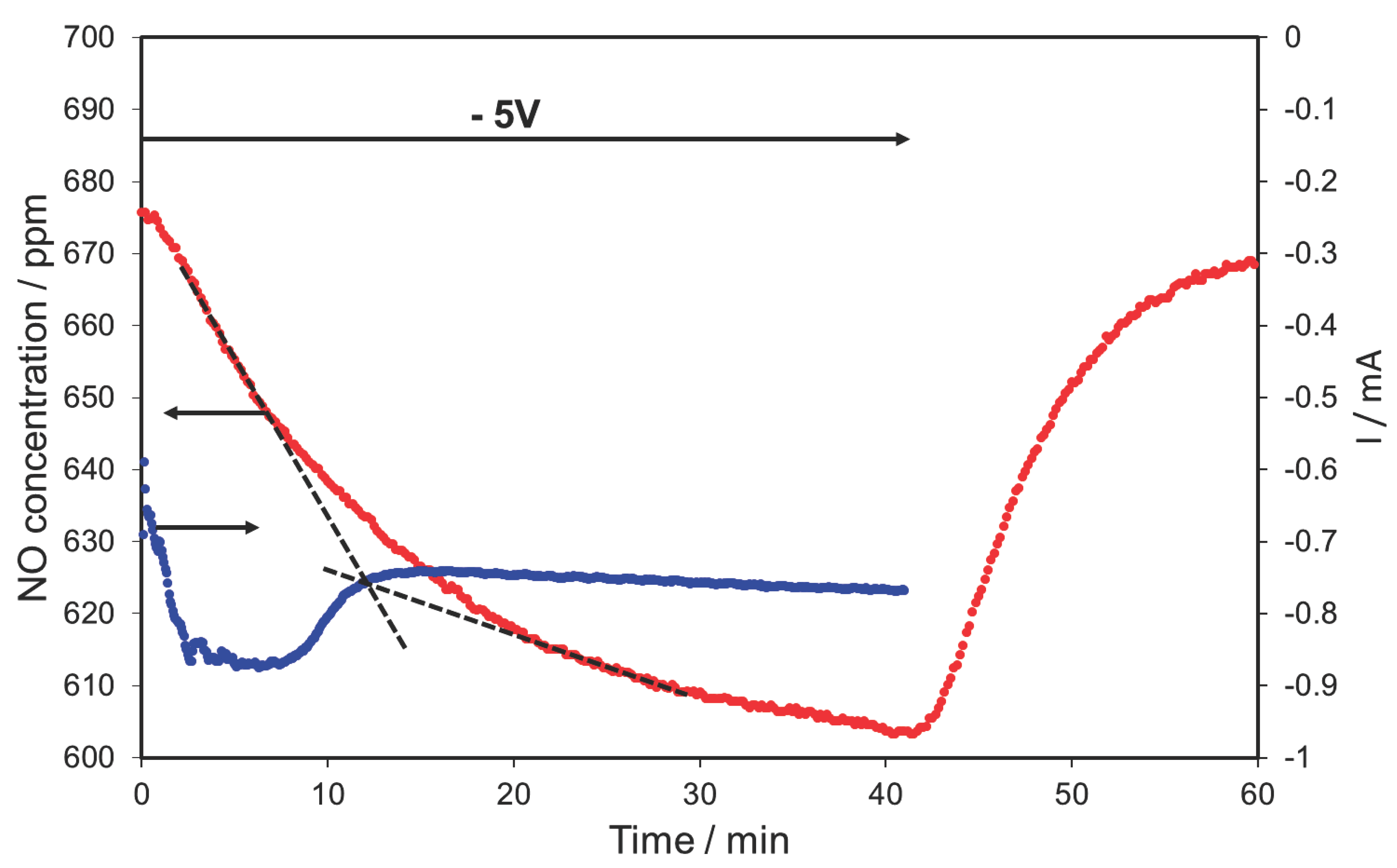
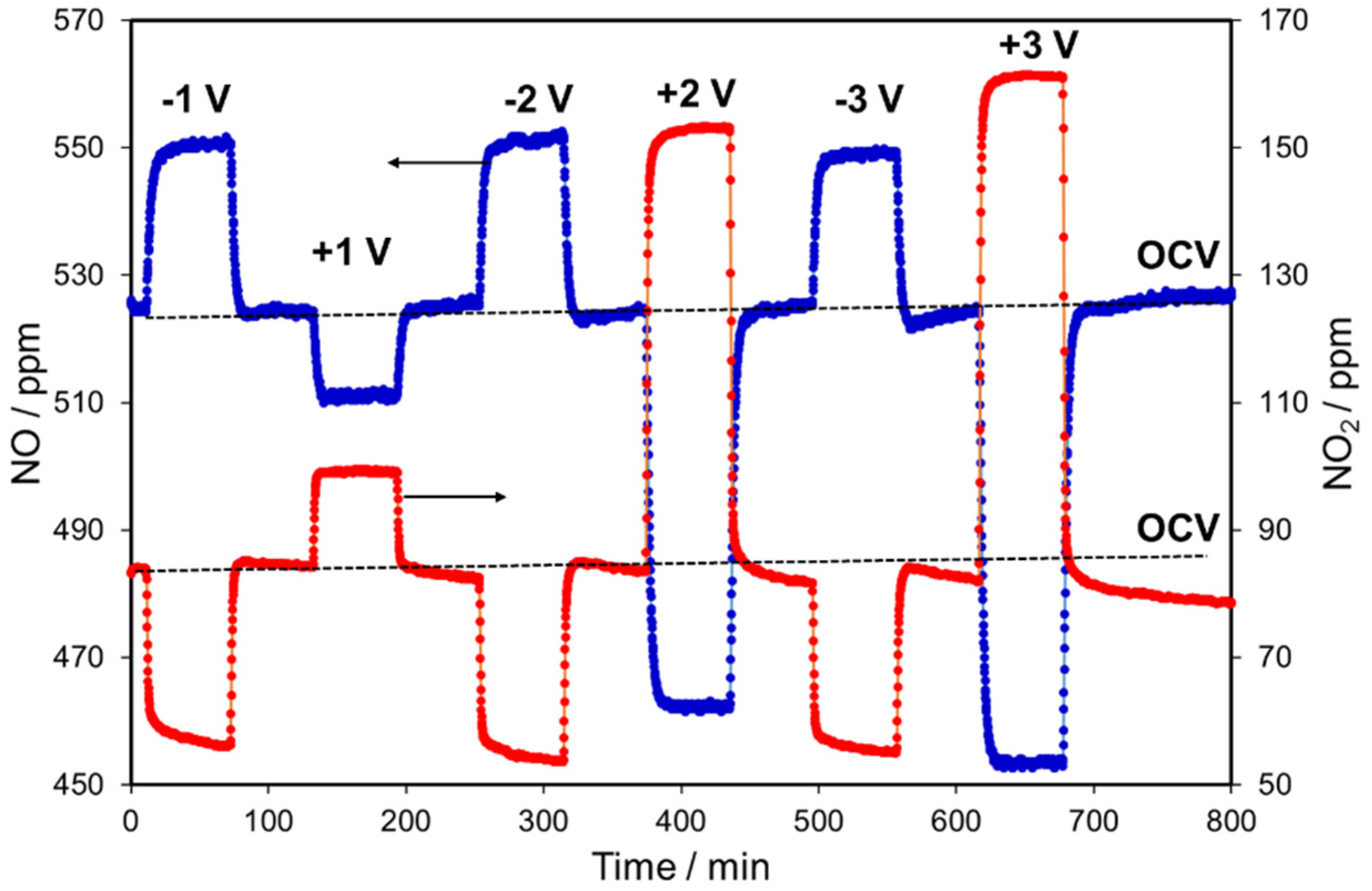
2.2. NOx Electrocatalytic Conversions without Oxygen in the Feed
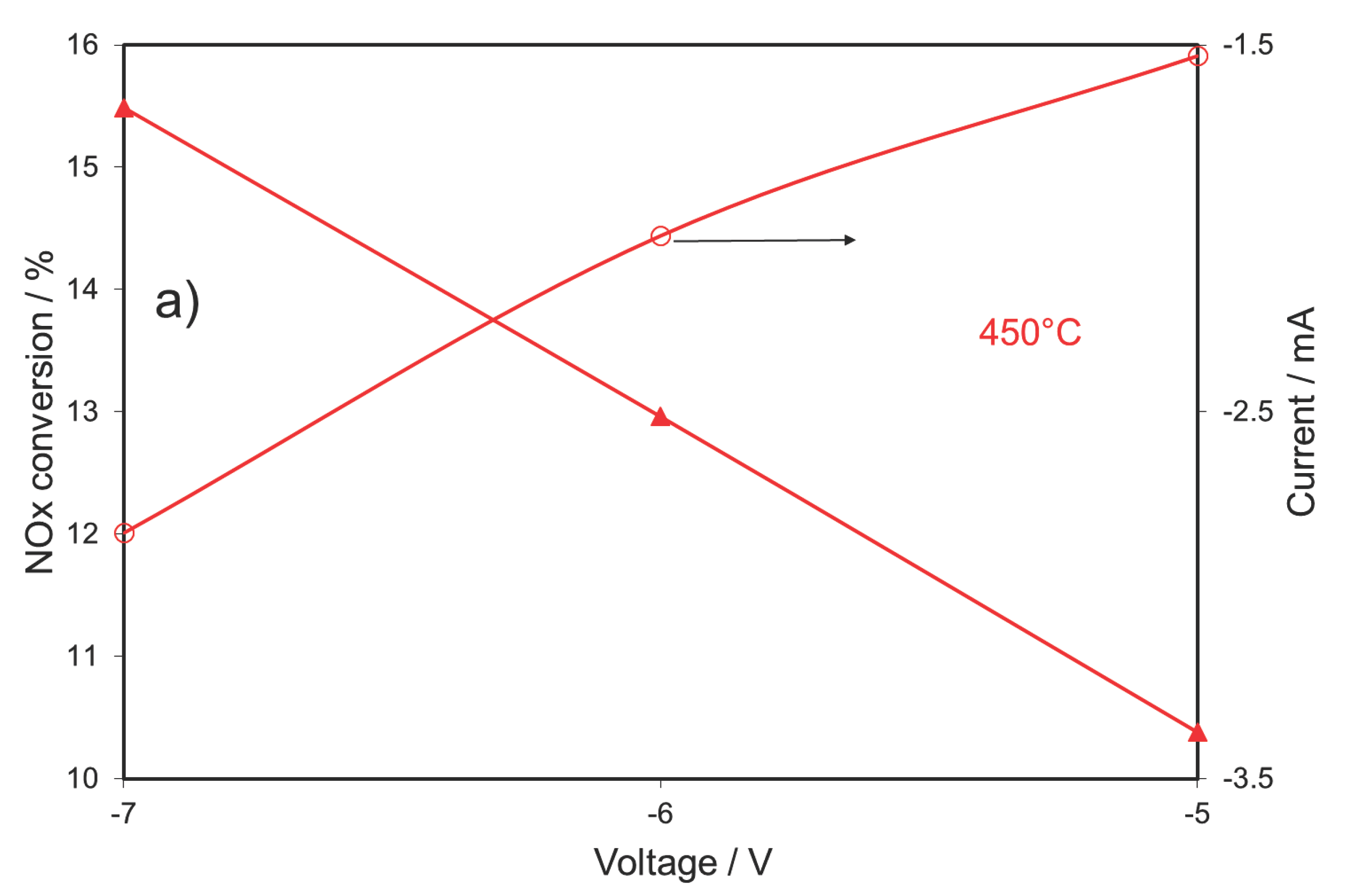
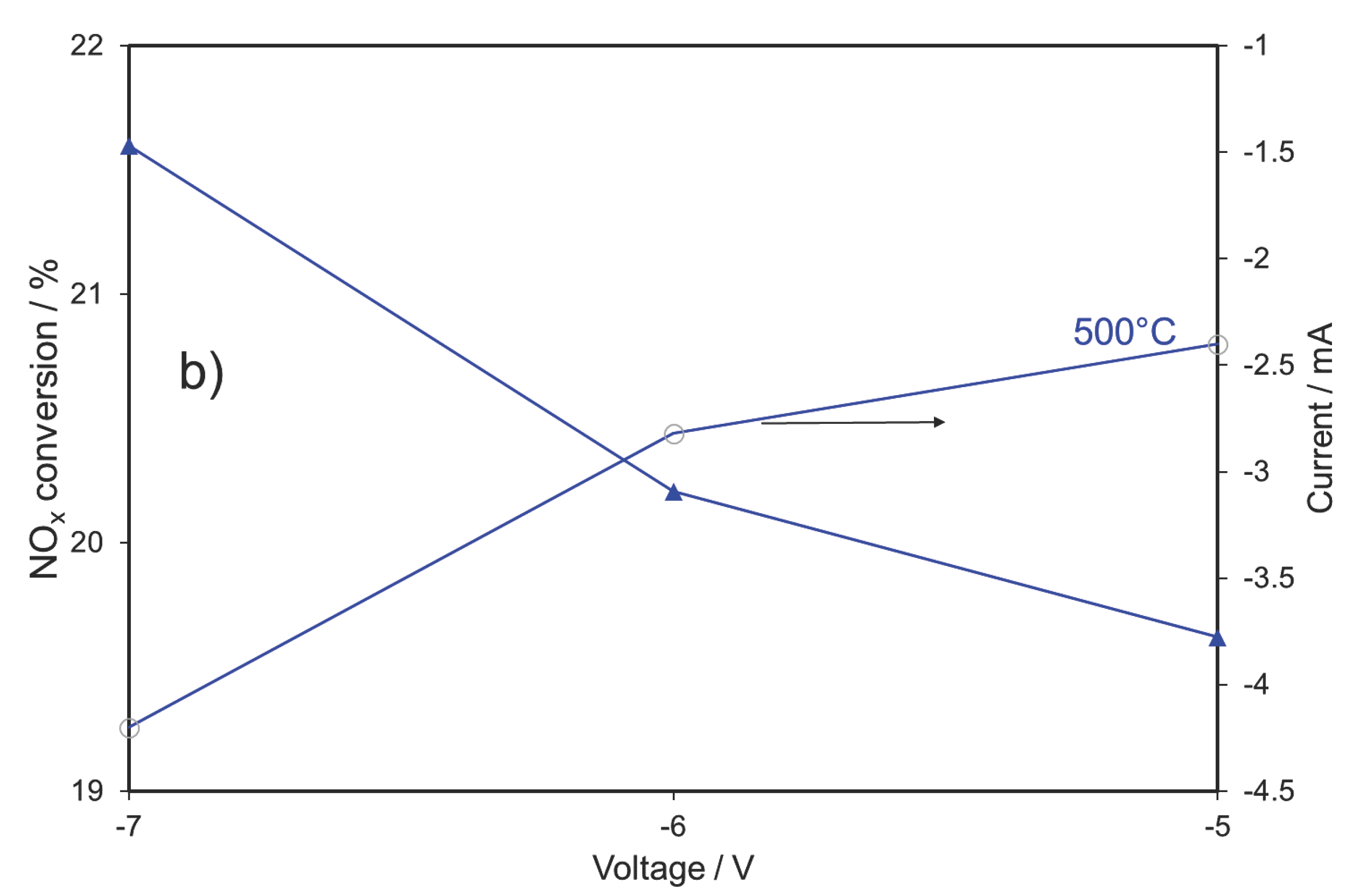
2.3. NOx Electrocatalytic Conversion in the Presence of 1% O2
3. Materials and Methods
3.1. Electrode Preparation
3.2. Measurements of the Electrocatalytic Performances
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Vestreng, V.; Ntziachristos, L.; Semb, A.; Reis, S.; Isaksen, I.S.A.; Tarrason, L. Evolution of NOx emissions in Europe with focus on road transport control measures. Atmos. Chem. Phys. 2011, 9, 1503–1520. [Google Scholar] [CrossRef]
- Granger, P.; Parvulescu, V.I. Catalytic NOx Abatement Systems for Mobile Sources: From Three-Way to Lean Burn after-Treatment Technologies. Chem. Rev. 2011, 111, 3155–3207. [Google Scholar] [CrossRef] [PubMed]
- Werchmeister, R.M.L.; Bentzen, J.J.; Andersen, K.B.; Hansen, K.K. Removal of NOx with Porous Cell Stacks with La0.85Sr0.15CoxMn1−xO3+δ-Ce0.9Gd0.1O1.95 Electrodes Infiltrated with BaO. J. Electrochem. Soc. 2014, 161, H663–H669. [Google Scholar] [CrossRef]
- Werchmeister, R.M.L.; Hansen, K.K. Electrochemical reduction of oxygen and nitric oxide at low temperature on Ce1−xPrxO2−δ cathodes. Electrochim. Acta 2013, 114, 474–477. [Google Scholar] [CrossRef]
- Huang, T.J.; Wu, C.Y.; Lin, Y.H. Electrochemical Enhancement of Nitric Oxide Removal from Simulated Lean-Burn Engine Exhaust via Solid Oxide Fuel Cells. Environ. Sci. Technol. 2011, 45, 5683–5688. [Google Scholar] [CrossRef] [PubMed]
- Pancharatnam, S.; Huggins, R.A.; Mason, D.M. Catalytic Decomposition of Nitric Oxide on Zirconia by Electrolytic Removal of Oxygen. J. Electrochem. Soc. 1975, 122, 869–875. [Google Scholar] [CrossRef]
- Gür, T.M.; Huggins, R.A. Decomposition of Nitric Oxide on Zirconia in a Solid State Electrochemical Cell. J. Electrochem. Soc. 1979, 126, 1067–1075. [Google Scholar] [CrossRef]
- Hansen, K.K. Electrochemical reduction of O2 and NO on Ni, Pt and Au. J. Appl. Electrochem. 2008, 38, 591–595. [Google Scholar] [CrossRef]
- Hamamoto, K.; Fujishiro, Y.; Awano, M. Low-Temperature NOx Decomposition Using an Electrochemical Reactor. J. Electrochem. Soc. 2008, 155, E109–E111. [Google Scholar] [CrossRef]
- Li, X.; Vernoux, P. A new NOx storage-reduction electrochemical catalyst. Appl. Catal. B 2005, 61, 267–273. [Google Scholar] [CrossRef]
- Shao, J.; Hansen, K.K. Enhancement of NOx removal performance for (La0.85Sr0.15)0.99MnO3/Ce0.9Gd0.1O1.95 electrochemical cells by NOx storage/reduction adsorption layers. Electrochim. Acta 2013, 90, 482–491. [Google Scholar] [CrossRef]
- Shao, J.; Hansen, K.K. Electrochemical NOx reduction on an LSM/CGO symmetric cell modified by NOx adsorbents. J. Mater. Chem. A 2013, 1, 7137–7146. [Google Scholar] [CrossRef]
- Friedberg, A.Z.; Hansen, K.K. Electrochemical reduction of NO with propene in the presence of oxygen on LSCoM/CGO porous cell stacks impregnated with BaO. J. Solid State Electrochem. 2015, 19, 1611–1620. [Google Scholar] [CrossRef]
- Traulsen, M.L.; Andersen, K.B.; Hansen, K.K. NOx conversion on LSM15-CGO10 cell stacks with BaO impregnation. J. Mater. Chem. 2012, 22, 11792–11800. [Google Scholar] [CrossRef]
- Leng, Y.J.; Chan, S.H.; Jiang, S.P.; Khor, K.A. Low-temperature SOFC with thin film GDC electrolyte prepared in situ by solid-state reaction. Solid State Ionics 2004, 170, 9–15. [Google Scholar] [CrossRef]
- Zha, S.W.; Moore, A.; Abernathy, H.; Liu, M.L. GDC-Based Low-Temperature SOFCs Powered by Hydrocarbon Fuels. Electrochem. Soc. 2004, 151, A1128–A1133. [Google Scholar] [CrossRef]
- Li, X.; Gaillard, F.; Vernoux, P. The Relationship of the Catalytic Activity and the Open-Circuit Potential of Pt Interfaced with YSZ. Ionics 2005, 11, 103–111. [Google Scholar] [CrossRef]
- Hadjar, A.; Hernández, W.Y.; Princivalle, A.; Tardivat, C.; Guizard, C.; Vernoux, P. Electrochemical activation of Pt–Ba/YSZ NOx TRAP catalyst under lean-burn conditions. Electrochem. Commun. 2011, 13, 924–927. [Google Scholar] [CrossRef]
- Sun, L.P.; Rieu, M.; Viricelle, J.P.; Pijolat, C.; Zhao, H. Fabrication and characterization of anodesupported single chamber solid oxide fuel cell based on La0.6Sr0.4Co0.2Fe0.8O3−δ-Ce0.9Gd0.1O1.95 composite cathode. Int. J. Hydrogen Energy 2014, 39, 1014–1022. [Google Scholar] [CrossRef]
- Kambolis, A.; Lizarraga, L.; Tsampas, M.N.; Burel, L.; Rieu, M.; Viricelle, J.P.; Vernoux, P. Electrochemical promotion of catalysis with highly dispersed Pt nanoparticles. Electrochem. Commun. 2012, 19, 5–8. [Google Scholar] [CrossRef]
- Vernoux, P.; Gaillard, F.; Bultel, L.; Siebert, E.; Primet, M. Electrochemical Promotion of Propane and Propene Oxidation on Pt/YSZ. J. Catal. 2002, 205, 412–421. [Google Scholar] [CrossRef]




© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Westermann, A.; Shi, Y.X.; Cai, N.S.; Rieu, M.; Viricelle, J.-P.; Vernoux, P. Electrochemical Removal of NOx on Ceria-Based Catalyst-Electrodes. Catalysts 2017, 7, 61. https://doi.org/10.3390/catal7020061
Wang X, Westermann A, Shi YX, Cai NS, Rieu M, Viricelle J-P, Vernoux P. Electrochemical Removal of NOx on Ceria-Based Catalyst-Electrodes. Catalysts. 2017; 7(2):61. https://doi.org/10.3390/catal7020061
Chicago/Turabian StyleWang, Xi, Alexandre Westermann, Yi Xiang Shi, Ning Sheng Cai, Mathilde Rieu, Jean-Paul Viricelle, and Philippe Vernoux. 2017. "Electrochemical Removal of NOx on Ceria-Based Catalyst-Electrodes" Catalysts 7, no. 2: 61. https://doi.org/10.3390/catal7020061
APA StyleWang, X., Westermann, A., Shi, Y. X., Cai, N. S., Rieu, M., Viricelle, J.-P., & Vernoux, P. (2017). Electrochemical Removal of NOx on Ceria-Based Catalyst-Electrodes. Catalysts, 7(2), 61. https://doi.org/10.3390/catal7020061






