Abstract
This study develops a visible-light-driven heterogeneous Fenton-like system for the efficient degradation and detoxification of the persistent fluoroquinolone antibiotic gatifloxacin (GAT) in water. Three Fe-based metal–organic frameworks (MIL-53(Fe), MIL-88A(Fe), and MIL-101(Fe)) were synthesized and systematically evaluated as catalysts in a visible-light/H2O2 process. The three MOFs were systematically characterized, and a comparative analysis was conducted to elucidate how their structural differences influence catalytic performance. Among three MOFs, MIL-88A(Fe) exhibited superior photocatalytic activity, stability, recyclability, and low energy consumption in the visible-light-driven photo-Fenton process, which was attributed to its favorable structural and photo-induced redox properties. Under the optimal conditions (pH 7.2, H2O2 dosage of 1.2 mL·L−1, and catalyst loading of 0.1 g·L−1), 95.6% of GAT was degraded within 90 min. Radical scavenging experiments demonstrated that hydroxyl radicals (•OH) dominated the oxidation process. Based on intermediate identification, plausible degradation pathways were proposed, accompanied by a pronounced reduction in the ecological risks of transformation products. Furthermore, toxicity assays revealed that both the antibacterial activity and acute toxicity of the treated solutions were significantly alleviated. Overall, the Light/MIL-88A(Fe)/H2O2 system offers an effective and sustainable strategy for the removal and detoxification of fluoroquinolone antibiotics from aquatic environments.
1. Introduction
In recent years, the extensive use of antibiotics in healthcare, agriculture, and animal husbandry has led to their widespread occurrence in aquatic environments, and the phenomenon has raised serious ecological and public health concerns [1]. Gatifloxacin (GAT), a fourth-generation fluoroquinolone antibiotic, is widely used due to its broad-spectrum antibacterial activity. It has a detection rate of up to 93.8% in aquatic environments, and traditional wastewater treatment technologies have an elimination efficiency of less than 50.0% for it [2,3]. Existing antibiotic removal technologies include adsorption and electrocatalysis. However, these methods often have low efficiency or high energy consumption, and may cause secondary pollution [4,5]. Photocatalytic technology has become a research hotspot due to its environmental friendliness and high mineralization capability [6]. However, this technology also has significant limitations, such as electron-hole recombination, slow reaction rates under specific conditions, and high energy consumption due to ultraviolet light. Therefore, it is crucial to develop new photocatalytic materials that can effectively utilize visible light [7].
Among various photocatalytic materials, metal–organic frameworks (MOFs) have gained widespread attention due to their high porosity, wide pH adaptability, structural stability, and visible light responsiveness [8,9]. Fe-MOFs have advantages such as high specific surface area, lattice stability, and tunable structures. More importantly, under visible-light irradiation, they can simultaneously exhibit both photocatalytic and Fenton-like oxidative activities, making them a hot spot in the field of antibiotic degradation [10]. For example, Wang used a vacuum heating method to regulate the Fe2+/Fe3+ ratio in MIL-53(Fe) (optimal Fe2+/Fe3+ = 0.2725), which achieved a degradation rate of 95.0% for tetracycline (TC) under visible light after 100 min [11]. Juntao Tang designed MIL-100(Fe) with unsaturated iron sites, which completely degraded sulfamethoxazole (SMT) within 180 min at pH 4.0, with a mineralization efficiency of up to 52.4% [12]. Despite the demonstrated effectiveness of MIL-series Fe-MOFs toward various antibiotics, their application for gatifloxacin degradation remains scarcely explored. Moreover, a systematic comparison of the catalytic performance of different MIL materials (MIL-53(Fe), MIL-88A(Fe), and MIL-101(Fe)) within the same visible-light-driven Fenton-like system is still lacking. Previous studies have predominantly focused on degradation efficiency and kinetics, while the transformation pathways of GAT and the evolution of toxicity associated with degradation intermediates have received limited attention.
In this study, GAT was selected as a representative fluoroquinolone pollutant, and three Fe-MOFs (MIL-53(Fe), MIL-88A(Fe), and MIL-101(Fe)) were synthesized as heterogeneous catalysts to construct a visible-light-driven Fenton-like (Light/catalyst/H2O2) system. The degradation performance and stability of the three catalysts were systematically compared to identify the optimal material. Furthermore, the reaction mechanism was elucidated through electron Spin Resonance (ESR) analysis, while density functional theory (DFT) calculations were employed to predict vulnerable molecular sites of GAT. Degradation pathways were proposed based on liquid chromatography–mass spectrometry (LC–MS) analysis. In addition, the evolution of antibacterial activity and acute toxicity during the degradation process was evaluated using Escherichia coli, mixed bacterial cultures, and bioluminescent bacteria inhibition assays.
2. Results and Discussion
2.1. Characterization of Materials
MIL-88A(Fe) features a three-dimensional porous architecture in which fumaric acid ligands bridge trinuclear Fe(III) clusters, generating microporous channels and cage-like structures. In contrast, MIL-53(Fe) is constructed from trans-connected FeO6 octahedra linked by terephthalate ligands to form a three-dimensional framework, whereas MIL-101(Fe) is built from Fe3-oxo clusters coordinated with terephthalate linkers, giving rise to a distinct cluster-based three-dimensional porous network [9,12]. These structural differences are expected to influence mass transfer and interfacial reaction behavior during photocatalytic processes.
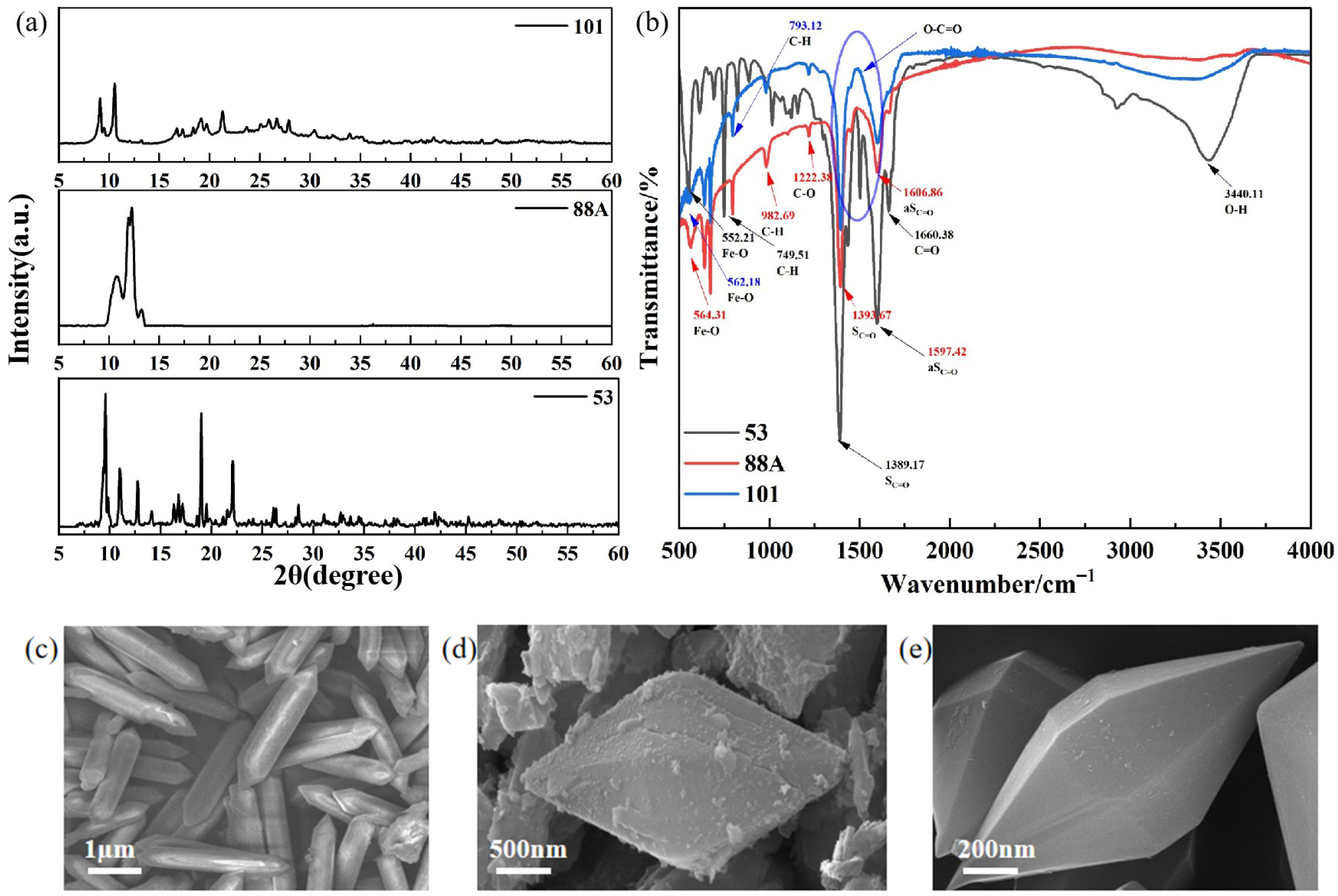
Scanning electron microscopy (SEM) images confirm the successful hydrothermal synthesis of the three MIL-series materials with well-defined and distinct morphologies (Figure 1c–e). MIL-88A(Fe) exhibits a uniform rod-like morphology with lengths of approximately 3–4 μm (Figure 1c). MIL-101(Fe) consists of well-faceted octahedral crystals (Figure 1d), whereas MIL-53(Fe) is composed of spindle-shaped particles (Figure 1e). All samples display intact crystal surfaces and well-developed morphologies, indicating good crystallinity and structural integrity, which are beneficial for subsequent photocatalytic reactions.
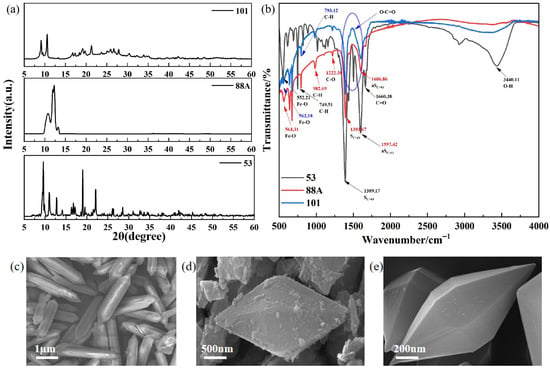
Figure 1.
(a) XRD patterns, (b) FTIR spectra, and (c–e) SEM images of the MIL-series catalysts (MIL-88A(Fe), MIL-101(Fe), and MIL-53(Fe)).
X-ray diffraction (XRD) was employed to identify the crystalline phases of the as-prepared catalysts (Figure 1a). For MIL-88A(Fe), distinct diffraction peaks were observed at 2θ = 11.3° and 11.8°, which correspond to the (101) and (002) crystal planes of MIL-88A(Fe), respectively, in good agreement with previously reported results [13]. The characteristic peaks of the prepared MIL-101(Fe) are consistent with the simulated MIL-101(Fe) and previous reports [14]. Meanwhile, the diffraction peaks at 2θ = 9.61°, 12.79°, 19.01°, and 22.09° are consistent with those reported in the CCDC database for MIL-53(Fe) (Nos. 690314–690316), confirming that the obtained sample corresponds to the MIL-53(Fe) crystalline phase [15].
To analyze the functional groups of the prepared materials, FT-IR spectroscopy was performed (Figure 1b), and the results are shown in Figure 1. For MIL-53, the broad peak at 3440 cm−1 is associated with the O-H stretching vibration of water molecules adsorbed on the surface [16]. In addition, the two absorption peaks at 1597 cm−1 and 1389 cm−1 correspond to the asymmetric νas(C=O) and symmetric νs(C=O) vibrations, respectively, indicating the presence of dicarboxylate organic linkers [17]. The absorption band at 1660 cm−1 corresponds to the stretching vibration of the C=O bond, whereas the peak at 749 cm−1 is attributed to the bending vibration of C–H on the benzene ring of the organic ligand [18]. Furthermore, the absorption band at 552 cm−1 is associated with Fe-O stretching vibrations, confirming the formation of Fe-O clusters between the central Fe metal and the carboxyl groups of the terephthalate ligands [16]. For MIL-88A(Fe), the characteristic peaks observed at 1397.86 cm−1 and 1606.86 cm−1 can be ascribed to the symmetric and asymmetric stretching vibrations of the hydroxyl groups in the fumarate ligands [19]. The peak at 1232.38 cm−1 is assigned to C–O stretching vibration [20], while those at 976.99 and 559.15 cm−1 correspond to C–H bending and Fe–O stretching vibrations, respectively [21]. In MIL-101, the band at 552 cm−1 also represents Fe–O vibrations. Peaks at 793 and 1001 cm−1 are related to C–H and C–O vibrations of the benzene ring, while those at 1389 and 1597 cm−1 correspond to the symmetric and asymmetric stretching of carboxylate groups. The band at 1703 cm−1 is attributed to C=O stretching [22].
The presence of Fe–O clusters is crucial for enhancing light absorption and catalytic activity. Sharp and intense Fe–O peaks indicate well-defined clusters with high crystallinity and structural order, whereas broad or weak peaks suggest structural defects. Stronger Fe–O signals are generally associated with more active sites [23]. Among the three materials, MIL-88A(Fe) exhibits the most intense Fe–O peak, suggesting superior catalytic potential. In contrast, MIL-53 shows a broad band at 3440 cm−1, attributed to O–H stretching of adsorbed water, indicating that water molecules may occupy pores and active sites, thereby hindering mass transfer and reducing its photocatalytic efficiency [24].
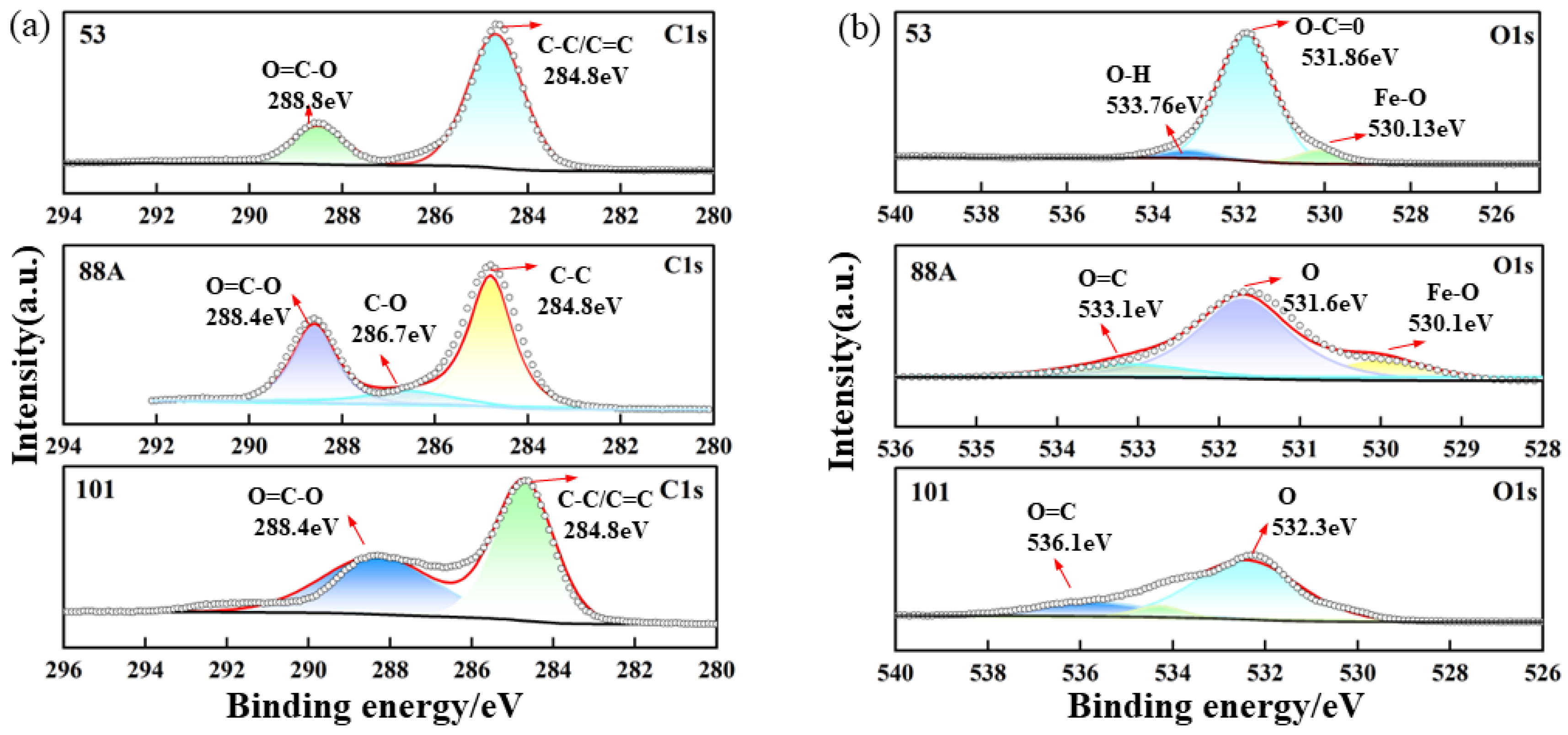
XPS was employed to investigate the surface elemental composition and chemical states of the three materials. As shown in Figure 2a–d, Fe, O, and C signals were detected in all samples, consistent with previous reports. In the high-resolution C 1s spectrum of MIL-88A(Fe), peaks at 284.8, 286.7, and 288.4 eV correspond to C–C, C–O, and O–C=O bonds, respectively [25]. For MIL-53, two peaks at 284.8 and 288.7 eV were observed, attributed to the aromatic ring (C–C/C=C) of terephthalic acid and the carboxylate carbon (O–C=O) [26]. MIL-101 exhibited similar features with peaks at 284.8 and 288.4 eV [27].
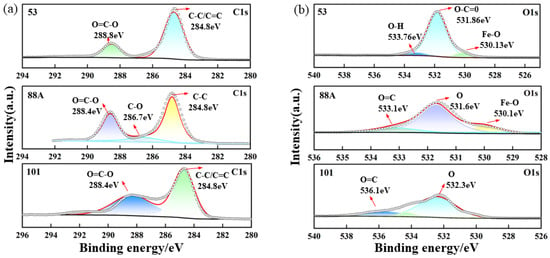
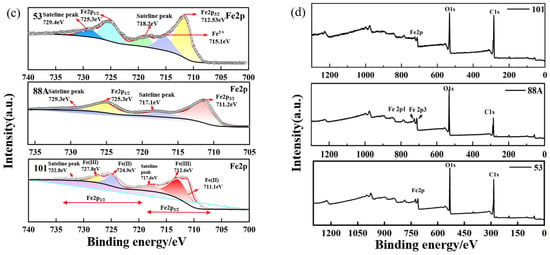
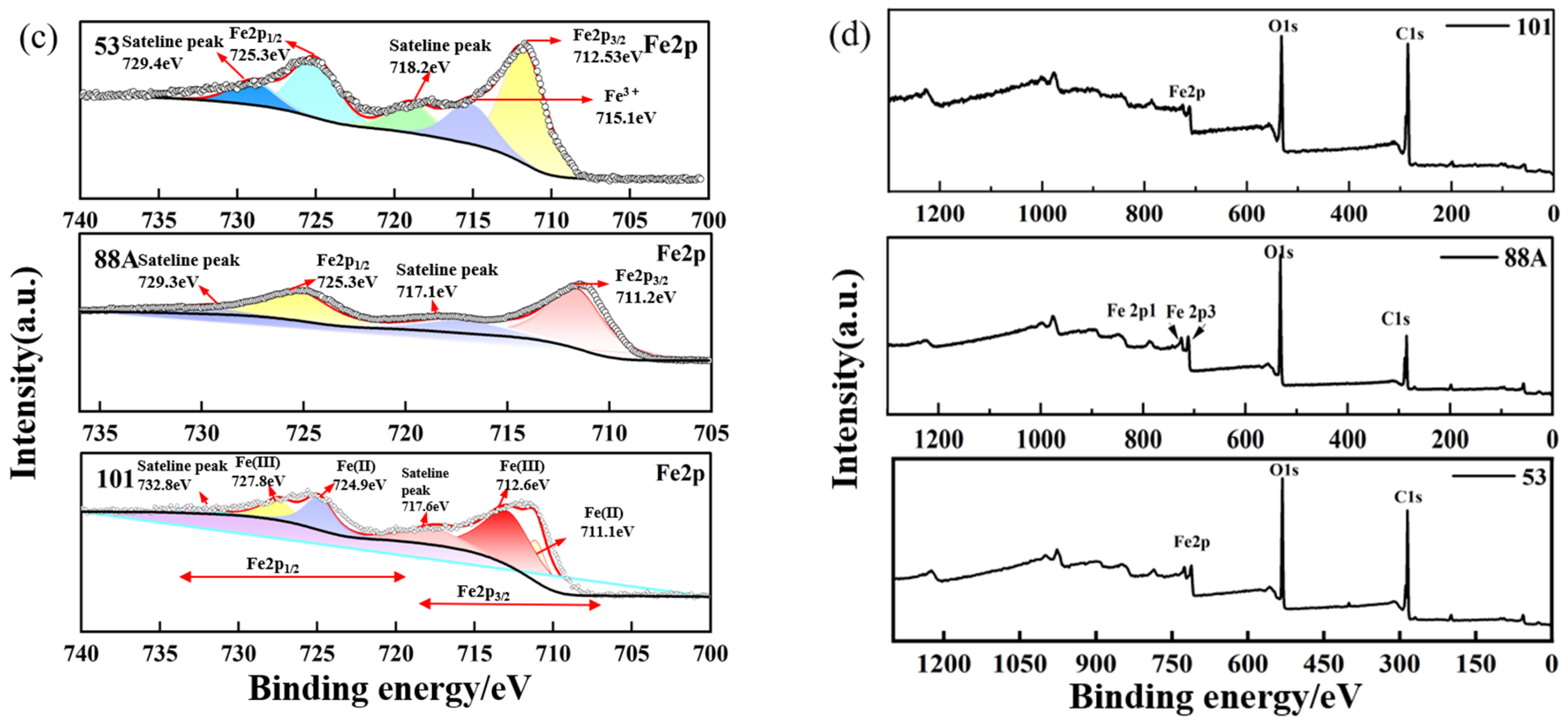
Figure 2.
High-resolution XPS spectra of the MIL series: (a) C 1s, (b) O 1s, (c) Fe 2p, and (d) survey spectrum.
The high-resolution O 1s spectrum of MIL-88A(Fe) showed peaks at 530.0, 531.7, and 533.1 eV, assigned to Fe–O, carboxylate oxygen, and O=C groups, respectively. In contrast, MIL-53 displayed peaks at 530.13, 531.86, and 533.76 eV, attributed to Fe–O bonds (Fe centers with organic linkers) and oxygen in O=C and O–H groups [28]. For MIL-101, two peaks at 532.3 and 536.1 eV were assigned to carboxylate and O=C oxygen species [27].
As shown in Figure 2c, the high-resolution Fe 2p spectrum of MIL-88A(Fe) revealed peaks at 711.2 eV (Fe 2p3/2) and 725.3 eV (Fe 2p1/2), suggesting the possible coexistence of Fe2+ and Fe3+. Satellite peaks at 717.1 and 729.9 eV, related to Fe3+, further support the presence of Fe3+ species. For MIL-53, the Fe 2p spectrum displayed peaks around 712.53 and 725.33 eV, corresponding to Fe 2p3/2 and Fe 2p1/2, respectively [28]. The high-resolution Fe 2p spectrum of MIL-101 exhibited characteristic peaks at binding energies of approximately 712.6 eV and 724.9 eV. Additionally, the peaks observed at 711.1 eV and 727.8 eV were assigned to the Fe 2p3/2 and Fe 2p1/2 states, respectively [27].
2.2. Comparison of Light Absorption Capacity
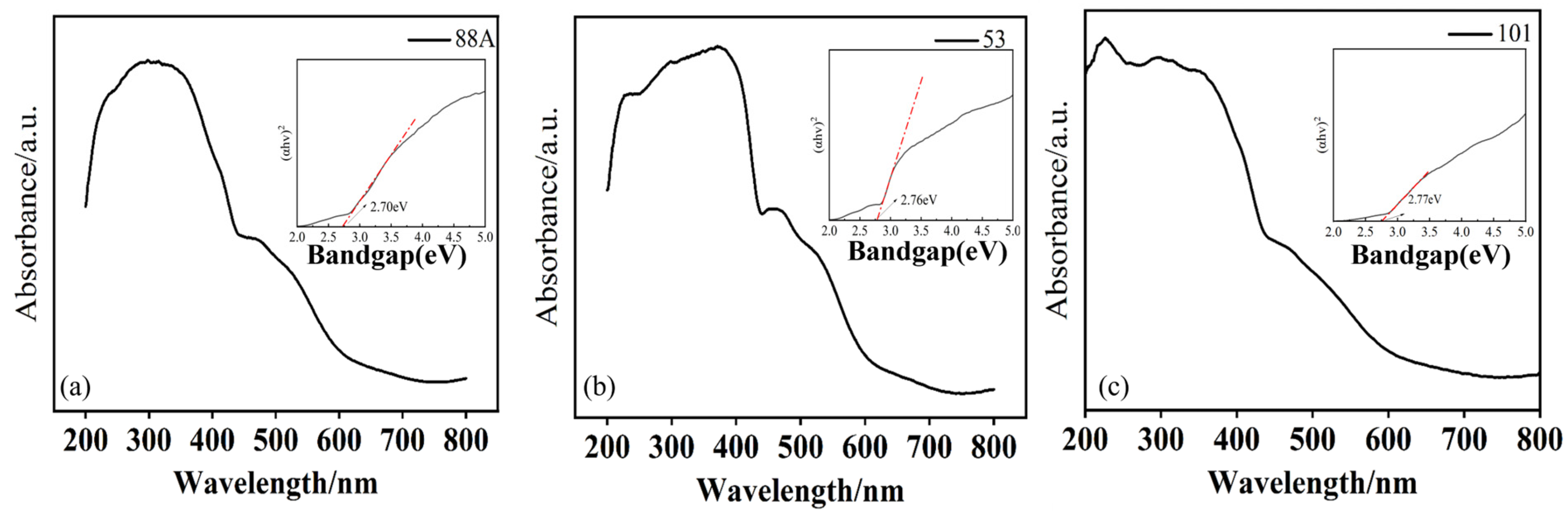
The optical properties of MIL-88A(Fe), MIL-53(Fe), and MIL-101(Fe) were characterized using ultraviolet–visible diffuse reflectance spectroscopy (UV–Vis DRS) (Figure 3a–c). As shown in the Figure 3, all three materials exhibited varying degrees of absorption in the 350–700 nm visible light range, indicating that they can all be excited by visible light. Among them, MIL-88A(Fe) showed the strongest absorption in the visible light region, suggesting it may possess the optimal photocatalytic activity. This visible light absorption primarily originates from the charge transfer transitions of the Fe–O clusters within the material [29].
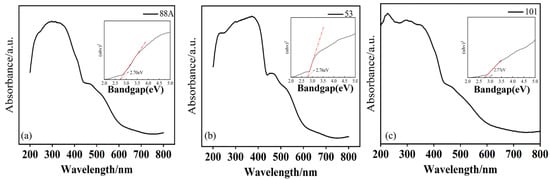
Figure 3.
(a–c) UV–Vis diffuse reflectance spectra of the MIL series (inset shows the band gap).
The band gap energy (Eg) of the materials was calculated using the Tauc equation: (αhν)1/n = A(hν − Eg), where α is the absorption coefficient, h is Planck’s constant, ν is the photon frequency, A is a constant, and n depends on the transition type (n = 1/2 for direct and n = 2 for indirect band gaps). Based on the DRS-derived Tauc plots (inset of Figure 3), the band gaps of MIL-88A(Fe), MIL-53(Fe), and MIL-101(Fe) were estimated to be 2.70 eV, 2.76 eV, and 2.77 eV, respectively.
2.3. Thermal Stability Investigation of MIL
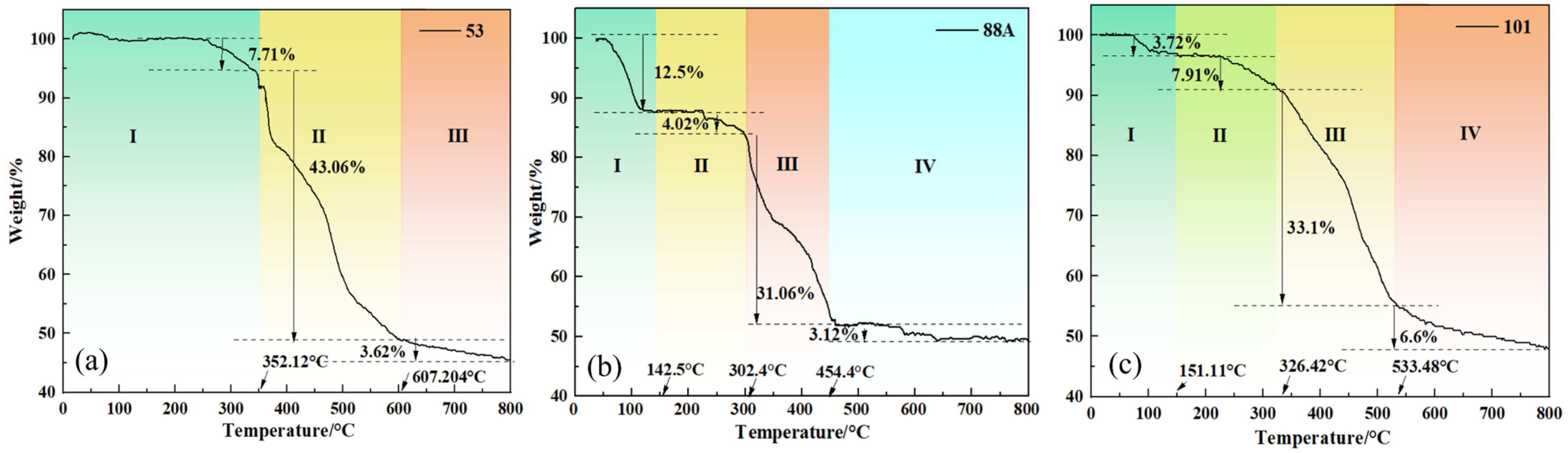
The thermal stability of the materials was studied using thermogravimetric analysis to examine the weight changes of MIL-88A(Fe), MIL-53(Fe), and MIL-101(Fe) with temperature. As shown in Figure 4a–c, all three materials exhibit high thermal stability. The weight loss curve of MIL-88A(Fe) is shown in Figure 4 and can be roughly divided into four stages: In Stage I, when the temperature increased to around 142.52 °C, the weight decreased by approximately 12.58%. This is likely due to the evaporation of water and residual ethanol in the material. In Stage II, when the temperature rises to around 302.21 °C, the weight loss is about 4.02%. This phenomenon is presumably attributed to the volatilization of residual organic solvents confined within the pores of the material. During Stage III, a pronounced mass loss is observed as the temperature increases to 454.4 °C, which can be ascribed to the decomposition of fumaric acid ligands accompanied by the progressive collapse of the framework structure [30]. In Stage IV, continued heating results in additional weight reduction, likely associated with carbonization processes occurring at elevated temperatures. The thermogravimetric behavior of MIL-53(Fe) is shown in Figure 4. In Stage I, when the temperature rises to about 259.30 °C and reaches 352.10 °C, the interaction between Fe3+ at the center of MIL-53 and water molecules is severed, leading to a weight loss of about 7.71%. In Stage II, when the temperature exceeds 352.10 °C, the weight loss rate sharply increases, mainly due to two processes: Firstly, at high temperatures, the organic ligand of terephthalic acid gradually detaches and undergoes carbonization. Secondly, as the temperature continues to rise, the framework structure of MIL-53 starts to collapse. MIL-101 exhibits similar thermogravimetric behavior, but its stability below 300 °C is superior to that of MIL-53.
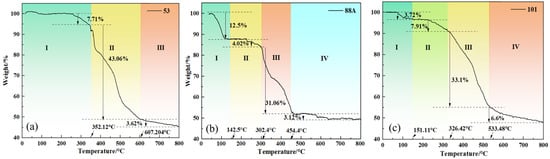
Figure 4.
(a–c) Thermogravimetric analysis (TGA) plots of different MIL series materials.
In summary, all three materials exhibit good thermal stability in the room to medium temperature range (<100 °C), which is crucial for their application in water treatment. It is only at higher temperatures (>300 °C) that significant ligand decomposition and framework collapse occur [30].
2.4. Comparison of Practical Degradation Performance
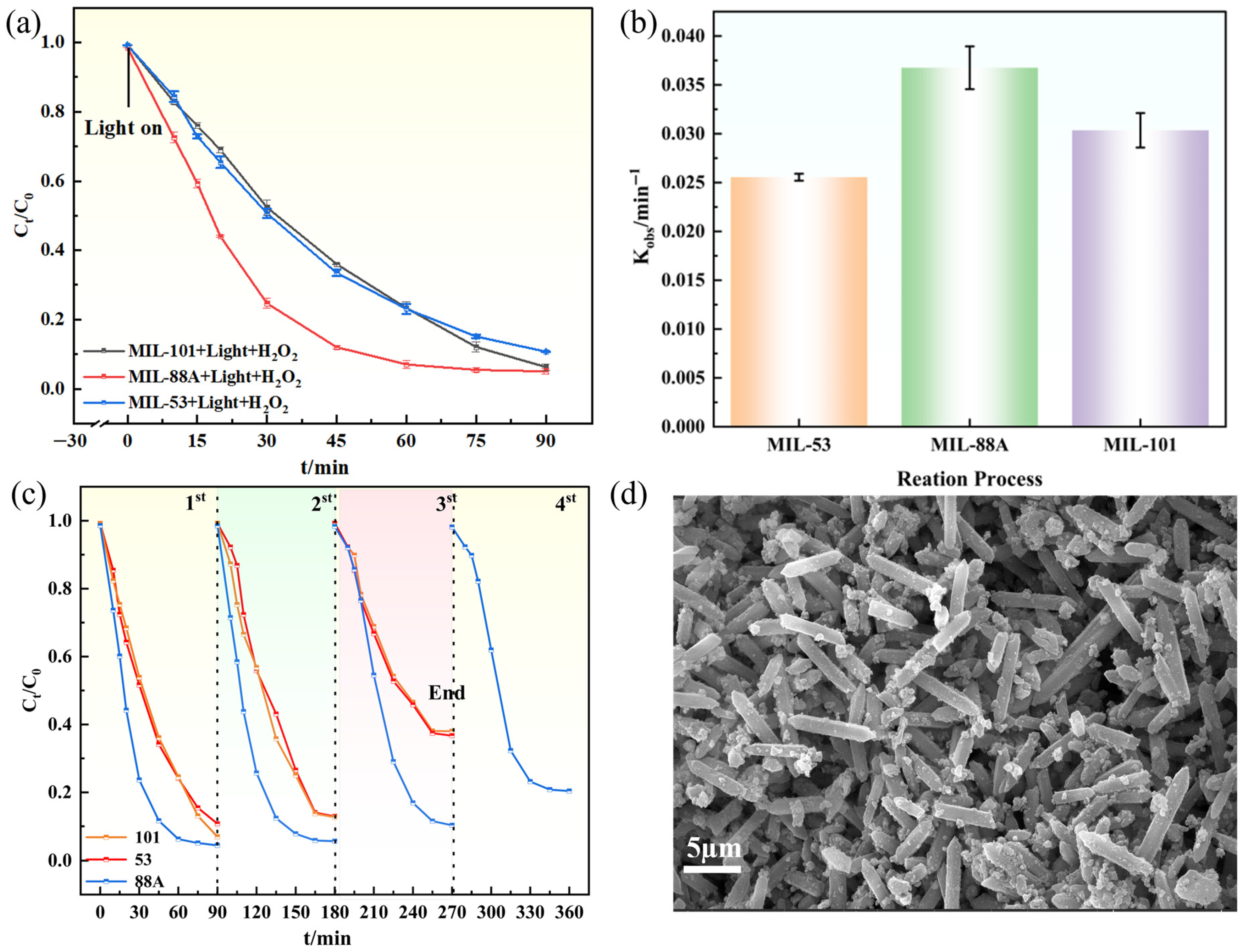
To evaluate the degradation performance of the three MIL catalysts toward GAT, experiments were carried out under optimized conditions (Figure 5a). Within 90 min of visible-light irradiation, the MIL-88A(Fe)/H2O2 system achieved the highest degradation efficiency (95.60%), slightly higher than MIL-101(Fe) (94.90%) and MIL-53(Fe) (90.10%). Kinetic analysis (Figure 5b) further confirmed the superior activity of MIL-88A(Fe), with the largest apparent rate constant (Kobs = 0.0385 ± 0.00219 min−1, R2 = 0.987). Table 1 provides an indicative comparison of the Light/MIL-88A(Fe)/H2O2 system with previously reported photocatalytic systems for GAT degradation. It should be noted that the experimental conditions vary among studies, including initial pollutant concentration, catalyst dosage, reaction time, light source, light intensity, and the presence or absence of H2O2. Therefore, the comparison is intended to demonstrate the relative competitiveness rather than to establish a strictly quantitative ranking.
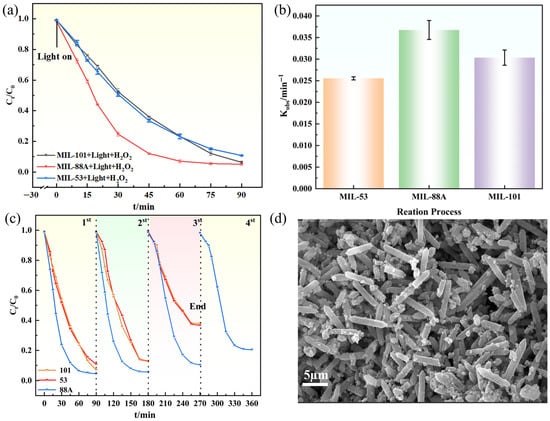
Figure 5.
(a) Degradation curves of the MIL series; (b) Degradation rate constants; (c) Cycling stability; (d) SEM of MIL-88A(Fe) after cycling.

Table 1.
Comparison of GAT degradation performance using different photocatalytic materials.
Notably, MIL-88A(Fe) achieves efficient GAT degradation under relatively mild synthesis conditions, moderate catalyst dosage, and near-neutral pH. In addition, the preparation of MIL-88A(Fe) requires lower temperature and shorter reaction time compared with many reported MOF-based systems, suggesting potential advantages in terms of energy input and sustainability.
Catalyst recyclability was subsequently examined (Figure 5c). After four consecutive cycles, MIL-88A(Fe) maintained 81% efficiency, showing only a slight 14.6% decline from its initial activity, whereas MIL-101(Fe) and MIL-53(Fe) dropped drastically to 62.10% and 63.40%, respectively. SEM images of MIL-88A(Fe) after cycling (Figure 5d) revealed well-preserved hexagonal rod-like morphology without structural collapse or aggregation, confirming its robust stability. In addition, MIL-88A(Fe) can be synthesized under milder conditions (85 °C, 2 h) compared with MIL-53(Fe) (120 °C, 10 h) and MIL-101(Fe) (110 °C, 12 h), resulting in lower energy demand and higher economic feasibility. To assess catalyst stability, Fe leaching during the reaction was quantified using the 1,10-phenanthroline method. The dissolved Fe concentration increased gradually with time and reached approximately 2.83 mg·L−1 at 90 min (typical reaction time), with Fe3+ as the dominant species. This corresponds to a small fraction of the total Fe content in the catalyst (7.9%), indicating limited metal dissolution under operational conditions. Although prolonged reaction (360 min) resulted in higher Fe concentration (5.63 mg·L−1), the retained catalytic activity and preserved structural features suggest that the framework remains largely intact and that the process is predominantly heterogeneous.
As summarized in Table 2, although MIL-101(Fe) exhibits a relatively higher Fe2+/Fe3+ ratio, MIL-88A(Fe) shows the highest Kobs and superior cycling stability. This indicates that the catalytic performance is not solely determined by the initial surface Fe2+ content. Instead, the combined effects of electronic structure (band gap), framework robustness, and charge transfer characteristics likely play more decisive roles in governing photocatalytic activity. Taken together, its outstanding activity, stability, and cost-effective synthesis highlight MIL-88A(Fe) as the optimal catalyst for further mechanistic and application studies.

Table 2.
Summary of key structural and catalytic parameters of MIL catalysts toward GAT degradation.
2.5. Photocatalytic Activation of H2O2 by MIL-88A(Fe) for GAT Degradation
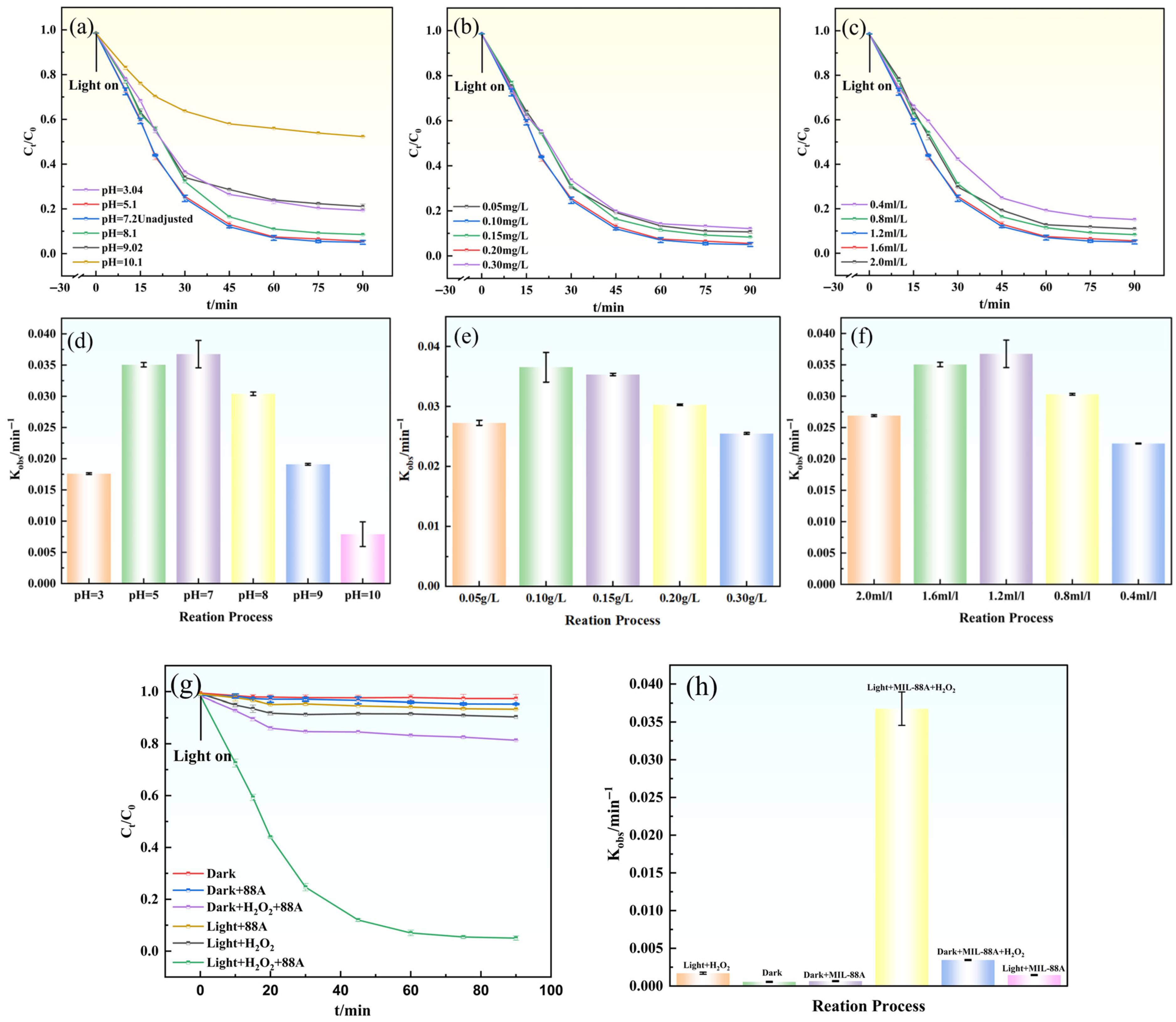
To elucidate the role of MIL-88A(Fe) in activating H2O2 for gatifloxacin (GAT) degradation under visible-light irradiation, a series of control experiments were conducted, as illustrated in Figure 6. Negligible GAT degradation was observed under visible-light irradiation alone, indicating that self-photolysis of GAT is insignificant. Similarly, the Light/H2O2 system exhibited very limited degradation efficiency (9.80% after 90 min), suggesting that H2O2 cannot be effectively activated by visible light in the absence of a catalyst.
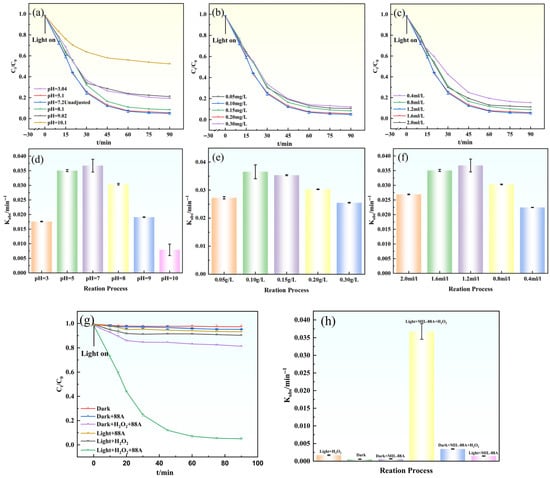
Figure 6.
(a–c,g) Effects of different conditions on GAT degradation; (d–f,h) degradation rate constants (Kobs) of GAT under different conditions.
In the dark MIL-88A(Fe)/H2O2 system, only a modest GAT removal (18.60% after 90 min) was achieved, implying that the heterogeneous Fenton-like reaction proceeds slowly without photoexcitation. The Light/MIL-88A(Fe) system also showed limited degradation performance, which can be attributed to the rapid recombination of photogenerated charge carriers, resulting in insufficient generation of reactive species.
In contrast, the Light/MIL-88A(Fe)/H2O2 system exhibited the highest degradation efficiency, reaching approximately 95.60% within 90 min. This pronounced enhancement indicates a strong synergistic effect among visible-light irradiation, MIL-88A(Fe), and H2O2, which facilitates efficient H2O2 activation and reactive oxygen species generation. Kinetic analysis further revealed that GAT degradation in all systems followed pseudo-first-order kinetics (Equation (8)). As shown in Figure 6h, the apparent rate constant (Kobs = 0.0385 ± 0.00216 min−1, R2 = 0.989) obtained for the Light/MIL-88A(Fe)/H2O2 system was significantly higher than those of the other control systems, further confirming the superior catalytic efficiency of this synergistic system.
Collectively, these results demonstrate that MIL-88A(Fe) plays a crucial role in promoting H2O2 activation under visible-light irradiation, thereby generating highly reactive oxidative species responsible for the rapid degradation of GAT.
2.5.1. Effect of Initial pH
In natural aquatic environments, pH values can fluctuate over a broad range, thereby exerting a substantial impact on the degradation efficiency of contaminants. To evaluate the applicability of the Light/MIL-88A/H2O2 system, the effect of initial pH (ranging from 3 to 10) on GAT degradation was investigated (Figure 6a,d). The results showed that within the pH range of 5–8, the degradation efficiency of GAT remained above 93.00% after 90 min, indicating that the system exhibits good adaptability under both mildly acidic and weakly alkaline conditions [35]. When the pH was increased to 10, the degradation efficiency dropped significantly to 52.30%. This decline is primarily due to the instability of H2O2 under strongly alkaline conditions, leading to its rapid decomposition into O2 and H2O; the precipitation of iron ions as hydroxides, which reduces the number of active sites; and the scavenging of •OH radicals by excess OH− to form the less reactive O− species. Considering that the pH of natural water bodies typically ranges from 6 to 8, and that the raw water used in this study had a pH of 7.2, all subsequent experiments were conducted at this native pH value.
2.5.2. Effect of H2O2 Concentration
As both an oxidizing agent and a radical precursor, the concentration of H2O2 plays a crucial role in regulating the generation of reactive radical species. As shown in Figure 6c,f, under fixed catalyst dosage and pH conditions, the effect of H2O2 concentration (0.4–2.0 mL·L−1, 30% solution) on GAT degradation was investigated. When the H2O2 concentration increased from 0.4 mL·L−1 to 1.2 mL·L−1, the GAT degradation efficiency within 60 min rose from 85.1% to 95.6%, with a corresponding increase in the Kobs (0.0385 ± 0.00219 min−1, R2 = 0.987) value. This enhancement is attributed to the sufficient supply of H2O2 ensuring continuous •OH generation. However, when the concentration was further increased to 2.0 mL·L−1, the degradation efficiency slightly decreased, which was likely due to excess H2O2 acting as a scavenger and consuming •OH (Equations (3) and (4)) [36]. Meanwhile, excessive H2O2 also leads to the ineffective generation of O2−. Considering both degradation efficiency and economic feasibility, 1.2 mL·L−1 was selected as the optimal H2O2 concentration.
2.5.3. Effect of MIL-88A Dosage
The catalyst dosage determines the number of active sites and the light absorption efficiency. As shown in Figure 6b,e the effect of MIL-88A(Fe) dosage (0.05–0.30 g·L−1) on GAT degradation was investigated. When the catalyst concentration increased from 0.05 g·L−1 to 0.10 g·L−1, the degradation efficiency at 90 min rose from 90.7% to 95.6%, and the corresponding Kobs (0.0385 ± 0.00219 min−1, R2 = 0.987) also increased (Figure 6e). This improvement is attributed to the greater number of active sites and light-harvesting units provided by the higher catalyst loading, which promotes the generation of reactive species. However, further increasing the catalyst concentration from 0.10 g·L−1 to 0.15 g·L−1 slightly reduced the degradation efficiency. This decline is due to the increased turbidity in the reactor, which decreases light transmittance and utilization efficiency, as well as particle agglomeration, which reduces the exposure of effective active sites. Therefore, 0.10 g·L−1 was selected as the optimal catalyst dosage.
2.5.4. Interference of Anions
Natural water bodies often contain various anions that may interfere with the degradation process. Therefore, the effects of representative anions (Cl−, SO42−, CO32−, and NO3−) on the photocatalytic degradation of GAT were investigated. In the presence of SO42− and Cl−, the GAT removal efficiency remained above 90%, indicating that these anions exert only a slight inhibitory effect on the photo-Fenton process. In contrast, the presence of CO32− and NO3− reduced the degradation efficiency from 95.3% to 40.3% and 62.4%, respectively. This pronounced inhibition may be attributed to the strong scavenging effect of CO32− and NO3− on hydroxyl radicals, as well as their reaction with •OH to form carbonate radicals (•CO32−) with lower oxidative capacity.
2.6. Degradation Mechanism of GAT
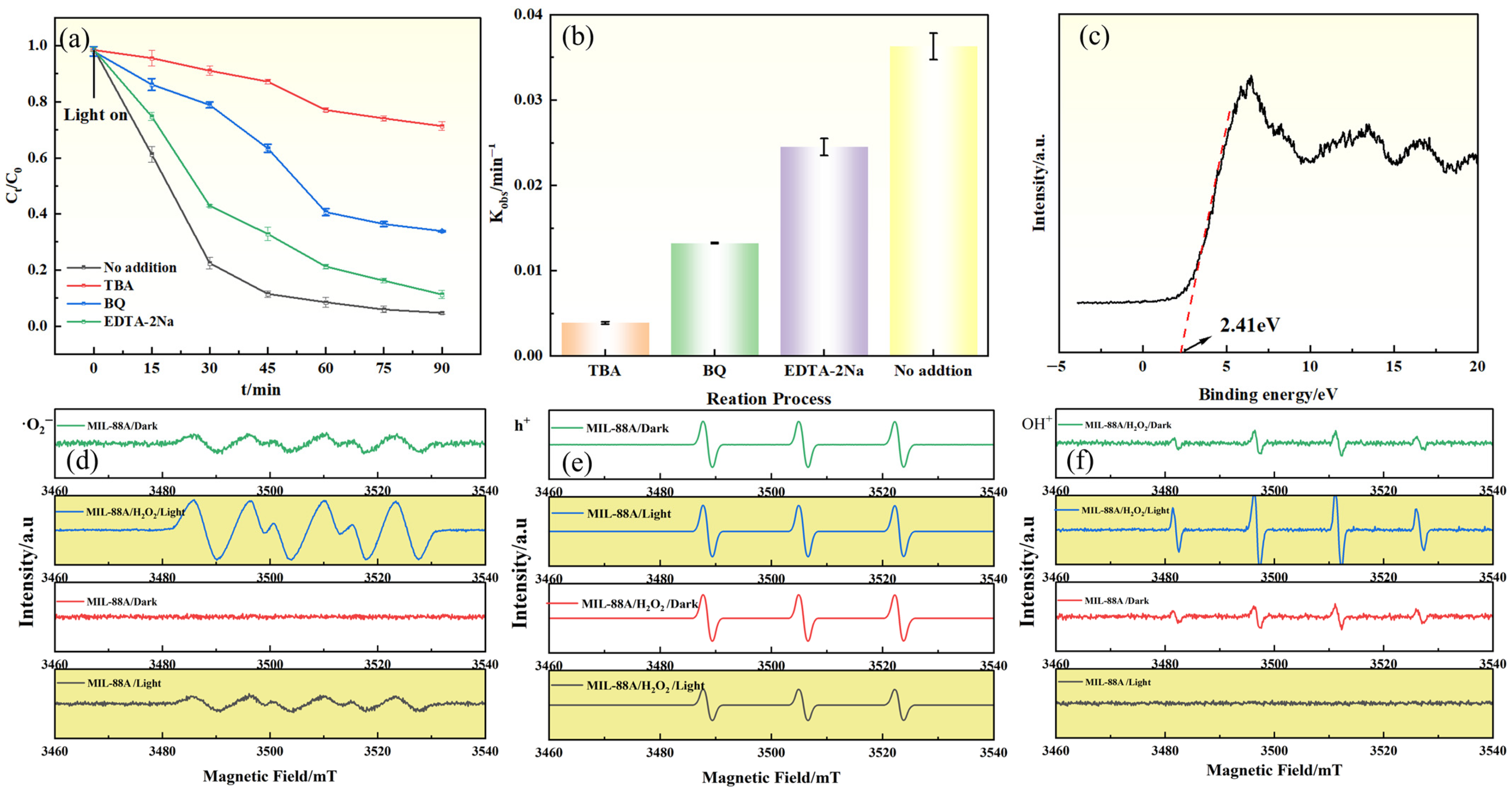
To elucidate the primary reactive species involved in GAT degradation by the Light/MIL-88A/H2O2 system, reactive species trapping experiments were conducted. Tert-butanol (TBA), disodium ethylenediaminetetraacetate (EDTA-2Na), and p-benzoquinone (BQ) were selected as radical scavengers to quench •OH, h+, and •O2−, respectively. As shown in Figure 7a,b, the addition of TBA markedly suppressed GAT degradation. The degradation efficiency decreased from 95.6% (No addition) to 29.7% (with TBA), corresponding to an inhibition of approximately 68.9%. This pronounced suppression indicates that •OH plays a dominant role in the Light/MIL-88A(Fe)/H2O2 system. Moreover, the addition of BQ and EDTA-2Na resulted in more moderate reductions in degradation efficiency (approximately 7.1–32.1% inhibition), suggesting that •O2− and h+ also participate in the degradation process but contribute to a lesser extent compared with •OH. Moreover, the ESR spectra of the reactive intermediates (Figure 7d–f) exhibited distinct signal features corresponding to •OH, •O2−, and h+, thereby further verifying the generation and involvement of these species in the Light/MIL-88A/H2O2 system.
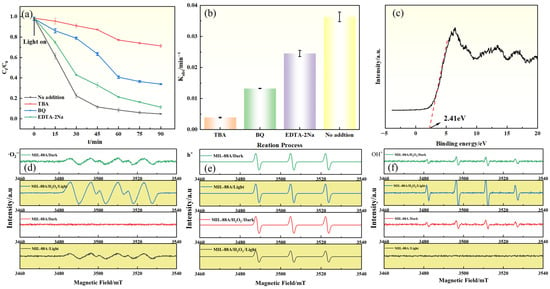
Figure 7.
(a) Effect of radical scavengers on GAT degradation; (b) Relationship between degradation rate constant (Kobs) and radical scavengers; (c) VB-XPS spectrum of MIL-88A(Fe); ESR spectra of (d) DMPO–•O2−, (e) TEMPO–h+, and (f) DMPO–•OH detected in different systems.
Based on the above experimental results and molecular orbital theory, a possible photocatalytic degradation mechanism is proposed under visible-light irradiation, electrons (e−) in MIL-88A(Fe) are excited from the HOMO to the LUMO, leaving holes (h+) in the HOMO (Equation (1)). The h+ can directly oxidize organic matter, or react with OH− to generate •OH (Equation (2)). The band positions of MIL-88A(Fe) were determined (Figure 7c), with its VB located at approximately 2.41 eV (vs. Fermi), via the equation EVB,NHE = ϕ + EVB,XPS − 4.44 [36,37] (φ represents the instrument work function of 4.2 eV). The EVB,NHE of MIL-88A(Fe) can be calculated as 2.17 Ev. Based on the relationship ECB = EVB − Eg, the conduction band potential (ECB) was calculated as −0.53 eV. Since this potential is more negative than the reduction potential of O2/•O2− (−0.33 eV vs. NHE), dissolved oxygen in the reaction system can capture photogenerated electrons to generate •O2− (Equation (3)), thereby contributing to GAT degradation. Nevertheless, MIL-88A(Fe) alone exhibits limited photocatalytic activity toward GAT under visible-light irradiation, which is likely due to the rapid recombination of photogenerated electron–hole pairs. The incorporation of an external electron acceptor, such as H2O2, can effectively inhibit this recombination process and consequently enhance. Meanwhile, H2O2, as an electron acceptor, can capture e− to generate •OH (Equation (4)), participating in GAT degradation. In addition, Fe3+ in the structure of MIL-88A(Fe) can accept electrons and be reduced to Fe2+ (Equation (5)), and under near-acidic conditions Fe2+ can react with H2O2 via the Fenton reaction to generate •OH (Equation (6)). In this process, surface-accessible Fe sites are primarily responsible for H2O2 activation, whereas Fe centers embedded within the framework may facilitate internal electron transfer. This cooperative effect between photoexcitation and the Fe2+/Fe3+ redox cycle accounts for the enhanced catalytic performance of the Light/MIL-88A(Fe)/H2O2 system.
2.7. Degradation Pathways of GAT
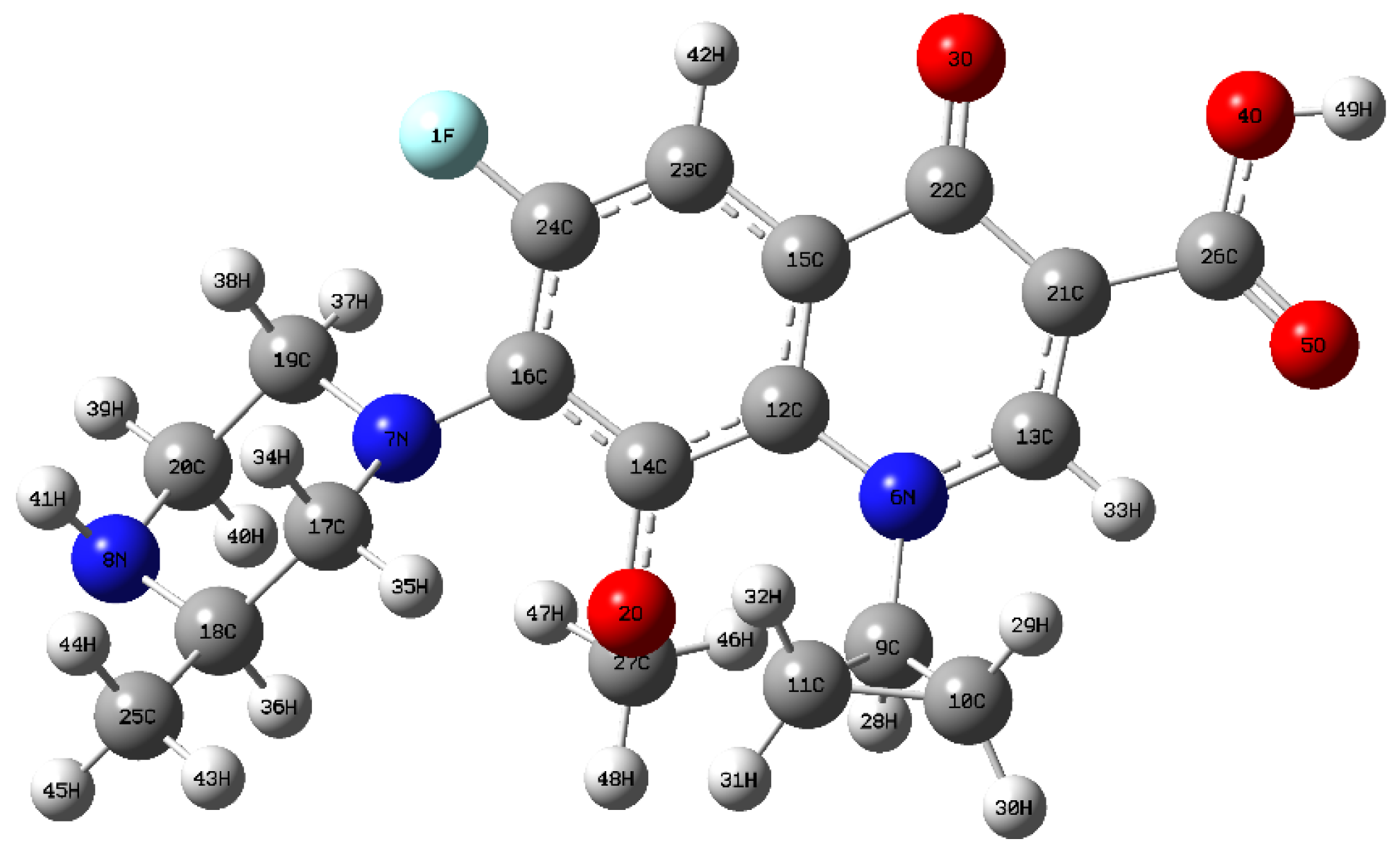
To identify the vulnerable sites of the GAT molecule, density functional theory (DFT) calculations based on Fukui function theory were performed. Geometry optimization and frequency calculations were carried out in the gas phase using Gaussian 09W at the B3LYP/6-311+G(d,p) level. Solvent effects were not explicitly included. The calculated Fukui indices and frontier molecular orbitals were used to qualitatively identify the most reactive sites of GAT rather than to provide quantitative energetic predictions. The nucleophilic Fukui index (f−) was computed to locate sites susceptible to electrophilic attack (e.g., •OH, h+). In addition, the spatial distributions of the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) were analyzed (Figure 8 and Figure 9). The HOMO regions are more likely to undergo electron loss (attacked by h+ or electrophilic radicals), whereas the LUMO regions are more likely to undergo electron gain (attacked by nucleophilic species). Furthermore, natural bond orbital (NBO) analysis was performed to investigate the atomic charge distribution. The results indicate that the 7N and 8N atoms on the piperazine ring exhibit both higher nucleophilic Fukui indices (f−) and higher HOMO density distributions (Figure 9), suggesting they are the most susceptible to electrophilic reactive species (•OH, h+). The 3O atom on the quinolone ring also shows relatively high reactivity.
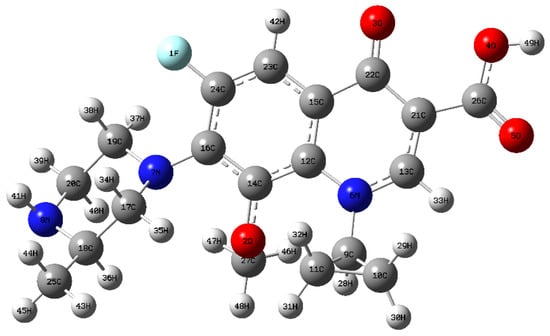
Figure 8.
Chemical structure of GAT.

Figure 9.
HOMO and LUMO of GAT.
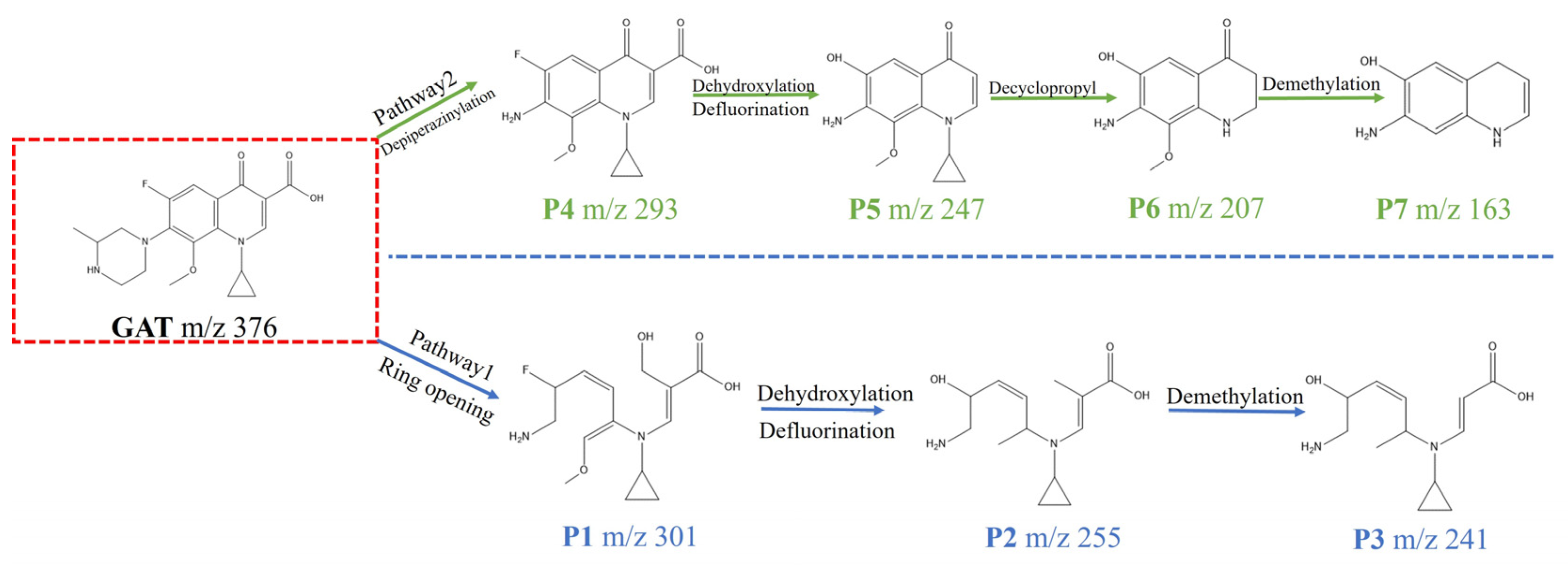
To experimentally verify the degradation pathways, liquid chromatography–mass spectrometry (LC–MS) was employed to identify the intermediate products formed during GAT degradation, and their structures were proposed based on mass spectral fragmentation patterns (Table S1 Supporting Information). By correlating the detected intermediates with the DFT-predicted reactive sites, two primary degradation pathways for GAT were proposed (Figure 10).
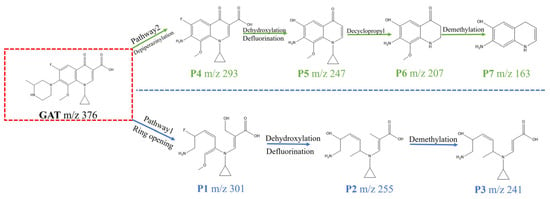
Figure 10.
Possible degradation pathways of GAT.
In Pathway I, electrophilic reactive species preferentially attack the piperazinyl and quinolone moieties of GAT (m/z = 376), resulting in bond cleavage and the formation of intermediate P1 (m/z = 301). P1 subsequently undergoes dehydroxylation, defluorination, and ether bond cleavage to generate P2 (m/z = 255), which is further demethylated to form P3 (m/z = 241). In Pathway II, the initial oxidative attack occurs predominantly on the piperazinyl group of GAT, yielding intermediate P4 (m/z = 293). Subsequent hydroxyl substitution of fluorine and decarboxylation lead to the formation of P5 (m/z = 247). P5 then undergoes cyclopropyl group removal to produce P6 (m/z = 209), followed by ether bond cleavage and decarbonylation to form P7 (m/z = 163). Ultimately, these intermediates are progressively transformed into smaller organic molecules and partially mineralized during the degradation process [8].
To provide a representative example of the structural assignment, the fragmentation behavior of intermediate P2 (m/z = 255) is discussed in detail. The parent compound GAT exhibits a protonated molecular ion at m/z = 376. After ring-opening and subsequent oxidative transformations, P2 was detected at m/z = 255, indicating a mass decrease of 121 Da relative to the parent molecule.
The mass difference can be rationalized by cleavage of the piperazine moiety and partial degradation of the quinolone skeleton, accompanied by dehydroxylation and defluorination. The loss of fluorine and hydroxyl group is consistent with oxidative defluorination and dehydration reactions commonly observed in photo-Fenton systems. The remaining fragment preserves the aromatic core structure, which is supported by the stability of the characteristic benzene-derived fragment ions observed in the MS spectrum.
Based on the observed m/z value, the proposed transformation pathway, and the typical fragmentation behavior of fluoroquinolone antibiotics reported in the literature, P2 was tentatively identified as the dehydroxylated and defluorinated ring-opening product. Detailed fragmentation assignments for all detected intermediates (P1–P7) are provided in Table S1 in the Supporting Information.
2.8. Toxicity Assessment
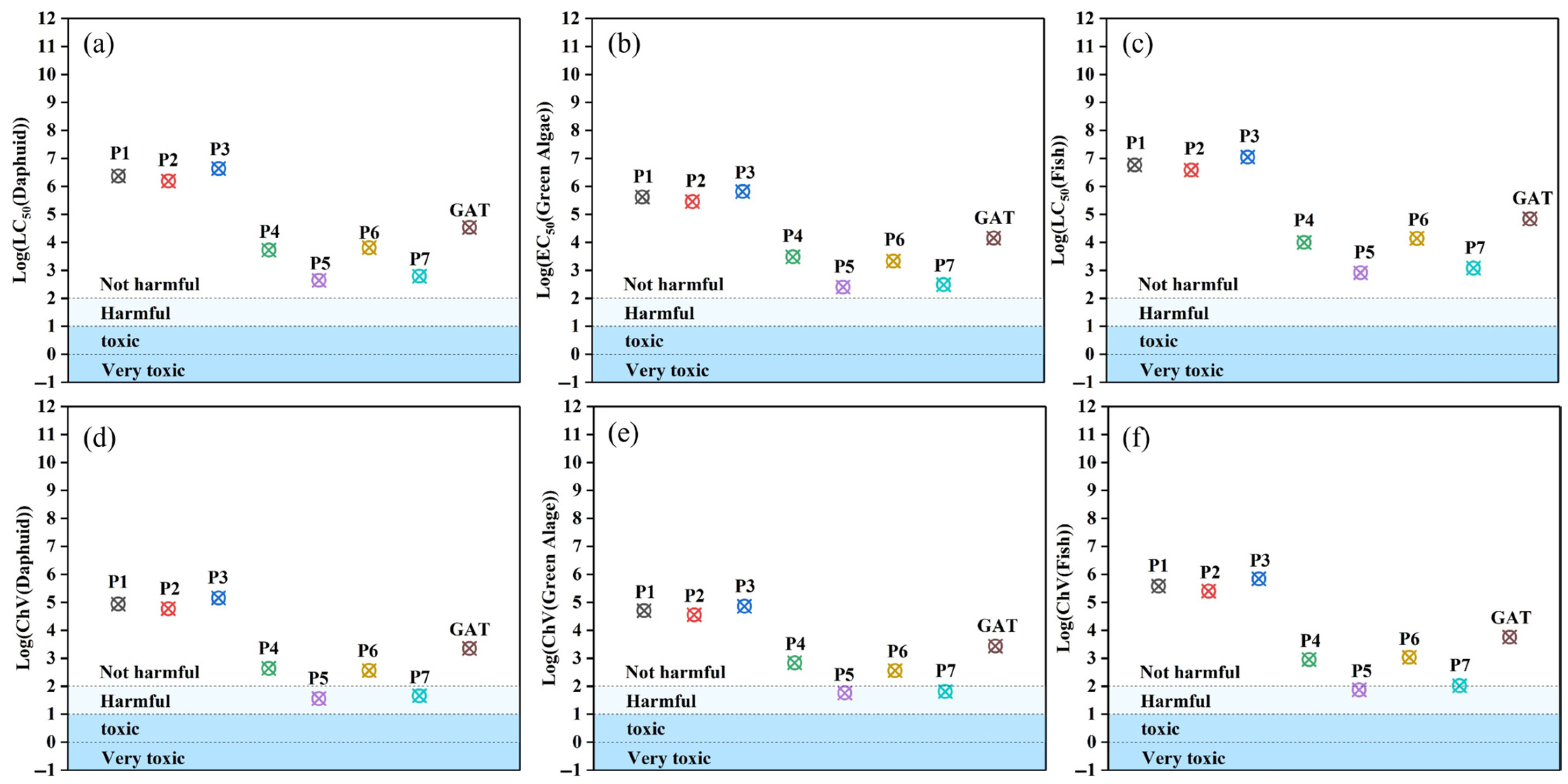
The ecological toxicity assessment of degradation products is crucial for evaluating the environmental safety of water treatment technologies [38]. The ECOSAR (Ecological Structure–Activity Relationship) software v2.2 (U.S. Environmental Protection Agency, Washington, DC, USA) was employed to predict the acute and chronic toxicity of the parent compound GAT and its major degradation intermediates (P1–P7) toward three aquatic organisms (fish, Daphnia, and green algae), based on its classification rules. Acute toxicity was expressed as the median lethal concentration or median effective concentration (LC/EC50), while chronic toxicity was expressed as the chronic value (ChV50). The prediction results are shown in Figure 11.
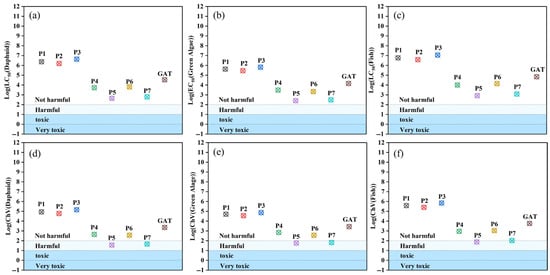
Figure 11.
LC50 of GAT and its intermediates for (a) Daphnia, (b) green algae, and (c) fish; (d–f) chronic toxicity toward the three aquatic species.
In general, the variation trends in toxicity of the intermediates relative to the parent compound were consistent across the three aquatic species. For simplicity, fish were selected as a representative example to illustrate the toxicity levels of the transformation products. As shown in Figure 11a–f, along the transformation from GAT to P1, P2, and P3, the LC50 values gradually increased, indicating a decrease in acute toxicity in pathway I. Similarly, according to the ChV50 values, the intermediates in pathway I also exhibited reduced chronic toxicity. In pathway II, one branch (GAT → P4 → P5 → P6 → P7) showed a slight increase in aquatic toxicity, although the overall toxicity level remained unchanged. However, the second branch demonstrated increased chronic toxicity, with P5 and P7 shifting from the “harmless” to the “harmful” category, suggesting a potential ecological risk. Therefore, photocatalytic degradation can be an effective approach for removing GAT and reducing its aquatic toxicity.
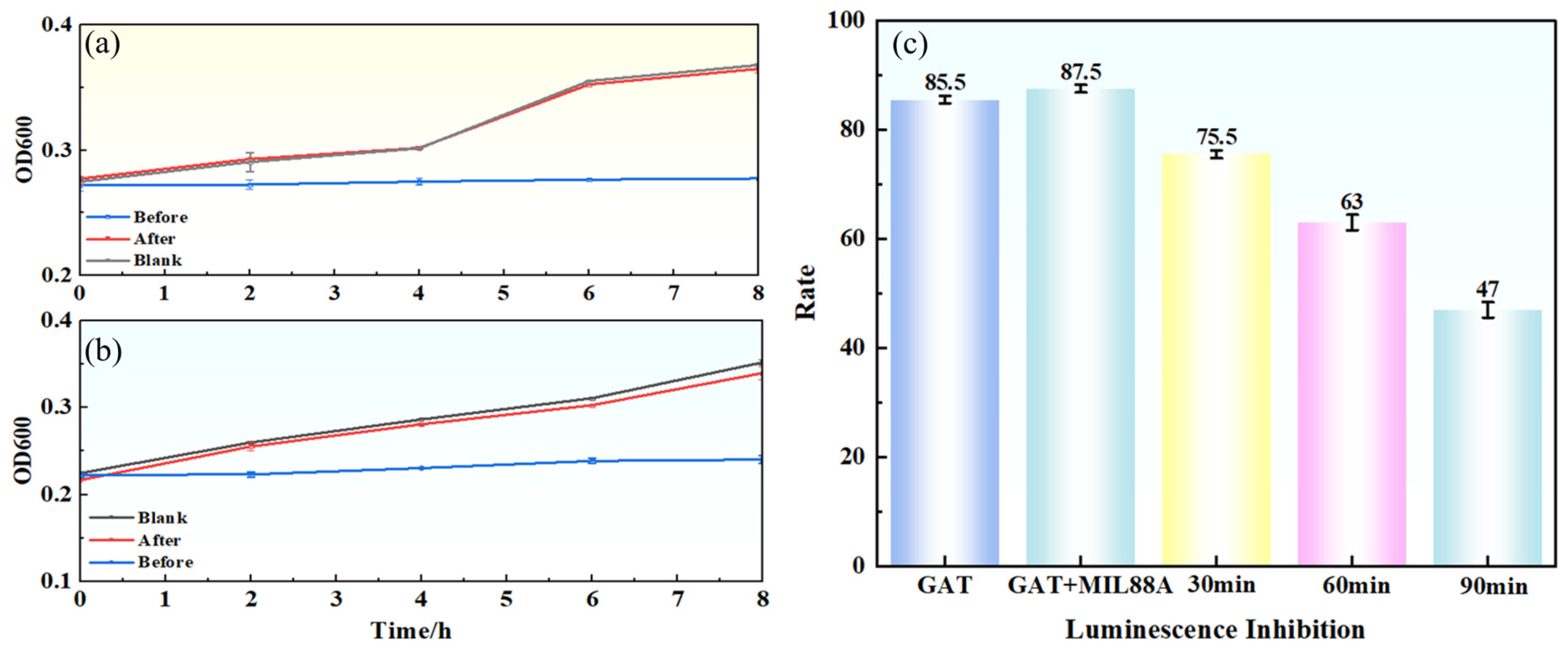
To further evaluate the changes in acute toxicity during the degradation process, a luminescent bacteria (Vibrio fischeri) inhibition assay was conducted to monitor the toxicity of samples at different time points (Figure 12c). The initial GAT solution (20 mg·L−1) exhibited high acute toxicity, with an inhibition rate of 87.5% (classified as “highly toxic”). As degradation proceeded, the solution toxicity decreased significantly. After 30 min and 60 min of degradation, the inhibition rates dropped to 75.5% and 63%, respectively. Upon complete degradation (90 min), the inhibition rate further decreased to 47% (classified as “moderately toxic”). Although the degraded solution still exhibited a certain degree of toxicity (likely due to residual intermediates), it was substantially lower than that of the original solution and met the acute toxicity discharge limit for industrial wastewater (IR < 50%) [39]. These results indicate that the Light/MIL-88A(Fe)/H2O2 system can effectively achieve both GAT degradation and simultaneous detoxification.
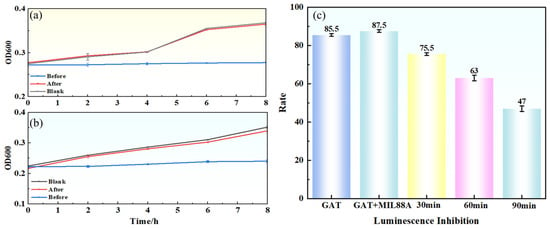
Figure 12.
Antibacterial activity and acute toxicity before and after degradation: (a) mixed bacteria, (b) E. coli, (c) luminescent bacteria inhibition rates.
To assess the impact of degradation on antibacterial activity, growth inhibition tests were performed using Escherichia coli and a mixed bacterial consortium (Figure 12a,b). The original GAT solution (20 mg·L−1) significantly inhibited the growth of both bacterial types (slow increase in OD600), indicating strong antibacterial activity. In contrast, after complete degradation (90 min), the bacterial growth curves showed no significant difference compared to the blank control group (ultrapure water or culture medium), demonstrating that the antibacterial activity of GAT was completely eliminated during degradation.
Collectively, the combined ECOSAR prediction and experimental toxicity evaluation provide a comprehensive assessment of the environmental implications of GAT degradation. Although the overall toxicity of the treated solution decreased significantly compared with the parent antibiotic, certain intermediates (notably P5 and P7) exhibited increased predicted chronic toxicity. Moreover, the solution after 90 min remained classified as “moderately toxic” in the luminescent bacteria assay, indicating that complete detoxification was not achieved under the present conditions.
Therefore, while the Light/MIL-88A(Fe)/H2O2 system effectively reduces GAT concentration and mitigates overall toxicity, it does not guarantee full environmental safety. Further optimization or integration with a subsequent biological treatment step may be required to achieve comprehensive detoxification.
3. Materials and Methods
3.1. Experimental Materials
Gatifloxacin (GAT, C19H22FN3O4 ≥ 98%), fumaric acid (C4H4O4, ≥99%), and terephthalic acid (C8H6O4, ≥99%) were purchased from Aladdin Biochemical Technology Co., Ltd. (Shanghai, China); hydrogen peroxide (H2O2, 30%), ethanol (C2H6O, ≥99%), and isopropanol (C3H8O, ≥99%) were purchased from China National Pharmaceutical Group Corporation (Sinopharm Chemical Reagent Co., Ltd., Shanghai, China); and ferric chloride hexahydrate (FeCl3·6H2O) was purchased from Macklin Biochemical Technology Co., Ltd. (Shanghai, China). All solutions were prepared using ultrapure water.
3.2. Catalyst Preparation
MIL-88A(Fe) was synthesized according to a previously reported procedure with minor modifications [40]. Specifically, 974.4 mg of fumaric acid and 2272.2 mg of FeCl3·6H2O were dissolved in 42 mL of ultrapure water. The solution was stirred for 2 h at 60 °C using a magnetic heating stirrer. It was then transferred to a Teflon-lined high-pressure reactor and heated at 85 °C for 2 h in a Teflon-lined stainless-steel autoclave. The product was collected by centrifugation and washed five times with anhydrous ethanol and ultrapure water, respectively. The resulting product was vacuum-dried at 100 °C in a vacuum oven to obtain MIL-88A(Fe) powder. MIL-101(Fe) and MIL-53(Fe) were synthesized following similar procedures reported in the literature [41,42], MIL-88A(Fe) was synthesized via a mild aqueous route at 85 °C for 2 h after pre-stirring at 60 °C, whereas MIL-101(Fe) and MIL-53(Fe) required solvothermal conditions in DMF at higher temperatures (110 °C for 12 h and 120 °C for 10 h, respectively). Therefore, compared with MIL-101(Fe) and MIL-53(Fe), the preparation of MIL-88A(Fe) was conducted under lower temperature, shorter reaction time, and in a water-based system. Detailed synthetic procedures are provided in Text S1 of the Supporting Information.
3.3. Characterization of Materials
The surface morphologies of MIL-53(Fe), MIL-88A(Fe), and MIL-101(Fe) were examined by scanning electron microscopy (SEM, SU5000, Hitachi, Tokyo, Japan). The crystal structures were identified using X-ray diffraction (XRD, Ultima IV, Rigaku, Akishima, Japan). The elemental compositions and valence states of the catalysts were analyzed by X-ray photoelectron spectroscopy (XPS, Escalab 250Xi, Thermo Fisher Scientific, Waltham, MA, USA). Fourier-transform infrared (FTIR) spectroscopy was conducted to characterize surface functional groups (Nexus 670, Thermo Nicolet, Waltham, MA, USA). The optical absorption properties and band gap energies were evaluated using UV–visible diffuse reflectance spectroscopy (UV–Vis DRS, UV-3600i, Shimadzu, Kyoto, Japan). Thermal stability was assessed by thermogravimetric analysis (TGA, TGA-1000C, Shanghai, China).
3.4. Photocatalytic Degradation Experiments
The degradation experiment was conducted in a photochemical reactor. A 200 mL solution of GAT (20 mg L−1) was prepared, and a predetermined amount of catalyst was added. The photocatalytic degradation performance of MIL-series materials toward GAT was evaluated under visible-light irradiation using a HEFAN 300 W xenon lamp system (Model: PE300W, total optical output power = 50 W,Hefan Instrument Co., Ltd., Shanghai, China) equipped with a 420 nm cutoff filter to eliminate ultraviolet light.
Prior to irradiation, the suspension was magnetically stirred under dark conditions for 30 min to establish adsorption–desorption equilibrium between the MIL-series materials and the GAT solution. A predetermined volume of H2O2 was added to the reaction solution at the beginning of illumination. During the illumination process, 4 mL of sample was taken every 15 min. The samples were immediately filtered through a 0.22 μm membrane to remove catalyst particles, and 20 μL of isopropanol was added to quench any residual •OH radicals and ensure that the measured concentration accurately reflects the state at the specific reaction time point. The concentration of GAT was determined using a spectrophotometer (UV7600, Shanghai Lingguang Technology Co., Ltd., Shanghai, China) at its maximum absorption wavelength of 294 nm. The GAT degradation rate was calculated according to the following formula [43]
where Ct and C0 represent the concentration (mg·L−1) of the GAT solution at a given reaction time and the initial concentration (mg·L−1) of the GAT solution, respectively; Kobs denotes the observed degradation rate constant; while t corresponds to the reaction time. All photocatalytic experiments were conducted in triplicate under identical conditions. The reported degradation efficiencies and kinetic parameters represent the mean values of three independent experiments. Error bars in the figures indicate standard deviations (SD). The apparent rate constants (Kobs) were obtained from linear fitting of ln(C0/Cₜ) versus time, and the corresponding coefficients of determination (R2) are provided to evaluate the goodness of fit.
3.5. Toxicity Assessment Experiments
The changes in antibacterial activity of the degradation solutions were evaluated using Escherichia coli and mixed bacterial growth inhibition assays. To eliminate the interference of residual hydrogen peroxide, manganese dioxide (MnO2) was added to the collected samples prior to biological tests. After centrifugation, the supernatants were collected and used as culture media for subsequent antibacterial experiments.
The experimental design consisted of three groups: (i) a blank control group containing bacteria cultured in fresh medium without GAT; (ii) a positive control group containing untreated GAT solution (20 mg·L−1) prior to degradation; (iii) an experimental group containing GAT-degraded water samples obtained at different reaction times. Corresponding parallel controls were prepared for each group. At designated degradation time points (2, 4, 6, 8, and 10 h), bacterial cells were collected, purified, and inoculated into sterile liquid media prepared from each group.
The cultures were incubated at 25 °C, and bacterial growth was monitored by measuring the optical density at 600 nm (OD600) using a microplate reader. Antibacterial activity was quantitatively evaluated by dynamic analysis of OD600 growth curves, based on the linear correlation between optical density and bacterial concentration within an appropriate range. The relative toxicity of the treated samples was assessed by comparing the growth rates (slopes) of bacterial growth curves between experimental and control groups [44].
Acute toxicity was evaluated using a bioluminescent bacteria inhibition assay in accordance with the ISO 11348 standard [45]. The following samples were selected for toxicity assessment: (i) a 20 mg L−1 GAT stock solution; (ii) a mixed solution containing 20 mg L−1 GAT and catalyst at a concentration identical to that used in the photocatalytic degradation experiments, collected after dark adsorption and prior to illumination; (iii) degradation solution obtained after 30 min of reaction; (iv) degradation solution obtained after 60 min of reaction; (v) degradation solution obtained after 90 min of reaction.
The acute toxicity of each sample was quantified by measuring the inhibition rate of luminescent bacteria, and the toxicity removal efficiency during the degradation process was evaluated based on ISO 11348 guidelines.
4. Conclusions
MIL-88A(Fe) exhibited markedly enhanced photocatalytic activity and recyclability toward GAT degradation compared to MIL-53 and MIL-101, retaining over 81% efficiency after four successive cycles. Under optimal conditions (pH 7.2, H2O2 = 1.2 mL·L−1, catalyst dosage = 0.1 g L−1), the Light/MIL-88A/H2O2 system achieved 95.6% GAT removal within 90 min and maintained robust stability upon reuse. Mechanistic investigations revealed that MIL-88A(Fe), upon photoexcitation, generated e−/h+ pairs, which subsequently activated H2O2 to yield •OH as the dominant reactive species, with h+ and •O2− also contributing to the degradation pathway. Ecotoxicological assessment, combining QSAR predictions and luminescent bacteria inhibition tests, confirmed that this system effectively transformed GAT into less toxic intermediates with mitigated antibacterial activity and reduced acute toxicity. These findings show that MIL-88A(Fe)-based photo-Fenton systems have great potential to efficiently remove fluoroquinolone antibiotics from aquatic environments.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/catal16030255/s1, Text S1: Preparation of catalysts; Table S1: DFT calculation result; Figure S1: Concentration of Fe2+ and Fe3+ in Solution.
Author Contributions
Y.Z.: Experiment, data analysis and writing the manuscript. Z.L.: Data analysis. S.J.: Formal analysis and funding acquisition. Y.X.: Supervision. C.G.: Supervision and funding acquisition. C.Y.: Supervision and funding acquisition. All authors have read and agreed to the published version of the manuscript.
Funding
The work was supported by Yunnan Higher Education Institutions’ Science and Technology Projects Serving Key Industries (FWCY-ZNT2024024).
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Xue, D.-M.; Zhu, G.-L.; Yan, X.-F.; Liu, M.-Q.; Krishna, R.; Zhang, H.-H.; Cai, R.-S. Amine-Functionalized Pillar-Layered Metal–Organic Frameworks for Multicomponent Natural Gas Purification with High-Methane Productivity and n-C4H10 Productivity. ACS Sustain. Chem. Eng. 2026, 14, 428–436. [Google Scholar] [CrossRef]
- Huang, F.; An, Z.; Moran, M.J.; Liu, F. Recognition of typical antibiotic residues in environmental media related to groundwater in China (2009−2019). J. Hazard. Mater. 2020, 399, 122813. [Google Scholar] [CrossRef] [PubMed]
- Gahrouei, A.E.; Vakili, S.; Zandifar, A.; Pourebrahimi, S. From wastewater to clean water: Recent advances on the removal of metronidazole. ciprofloxacin, and sulfamethoxazole antibiotics from water through adsorption and advanced oxidation processes (AOPs). Environ. Res. 2024, 252, 119029. [Google Scholar] [CrossRef] [PubMed]
- Xue, D.; Yan, X.; Liu, F.; Liu, M.; Zhang, H. Anthrathiophene-Based Covalent Organic Frameworks with Powerful π-Conjugation toward High-Efficiency CO2 Photoconversion. ACS Appl. Mater. Interfaces 2025, 17, 63467–63477. [Google Scholar] [CrossRef] [PubMed]
- Xue, D.-M.; Zhang, Y.-J.; Chen, J.-W. Molecular engineering in thizolo[5,4-d]thiazole-based donor-acceptor covalent organic framework induced high-efficient photosynthesis of H2O2. Chem. Eng. J. 2024, 502, 157874. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, J.; Zha, X.; Luo, Y.; Hu, Y.; Chen, G.; He, X. Interfacial chemical bond and oxygen vacancies modulated Mo2S3/BiOBr high-low junctions for enhanced photocatalysis gatifloxacin degradation. Appl. Surf. Sci. 2023, 641, 158548. [Google Scholar] [CrossRef]
- Qi, J.; Guo, L.; Yang, X.; Li, X.; Liu, W. Synergistic doped and loaded strategies to modify WO3 nanoflowers for efficient photocatalytic degradation of antibiotics through reactive oxygen species regulation. Sep. Purif. Technol. 2025, 361, 131557. [Google Scholar] [CrossRef]
- Yu, D.; Wu, M.; Hu, Q.; Wang, L.; Lv, C.; Zhang, L. Iron-based metal-organic frameworks as novel platforms for catalytic ozonation of organic pollutant: Efficiency and mechanism. J. Hazard. Mater. 2019, 367, 456–464. [Google Scholar] [CrossRef]
- Leng, X.; Dong, X.X.; Wang, W.P.; Sai, N.; Yang, C.; You, L.; Huang, H.; Yin, X.; Ni, J. Biocompatible Fe-based micropore metal-organic frameworks as sustained-release anticancer drug carriers. Molecules 2018, 23, 2490. [Google Scholar] [CrossRef]
- Ma, L.; Wang, T.; Wan, Y.; Zhang, G.; Li, X.; Jiang, M.; Zhang, L. Construction the soil-root-like heterostructure of Fe(III)-MOF/AgNWs composite membrane with photo-Fenton antifouling performance towards complex wastewater remediation. J. Environ. Chem. Eng. 2023, 11, 110125. [Google Scholar] [CrossRef]
- Guo, H.; Zhu, Y.; Wang, S.; Su, S.; Zhou, L.; Zhang, H. Boosting photocatalytic performance in mixed-valence MIL-53(Fe) by changing Fe2+/Fe3+ ratio. ACS Appl. Mater. Interfaces 2019, 11, 21280–21287. [Google Scholar] [CrossRef]
- Tang, J.; Wang, J. Metal–organic framework with coordinatively unsaturated sites as efficient Fenton-like catalyst for enhanced degradation of sulfamethazine. Environ. Sci. Technol. 2018, 52, 5367–5377. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, Y.; Li, X.; Xie, Y.; He, J.; Yu, J.; Song, Y. The MIL-88A-Derived Fe3O4-Carbon Hierarchical Nanocomposites for Electrochemical Sensing. Sci. Rep. 2015, 5, 14341. [Google Scholar] [CrossRef] [PubMed]
- Dao, X.-Y.; Guo, J.-H.; Zhang, X.-Y.; Wang, S.-Q.; Cheng, X.-M.; Sun, W.-Y. Structure-dependent iron-based metal–organic frameworks for selective CO2-to-CH4 photocatalytic reduction. J. Mater. Chem. A 2020, 8, 25850–25856. [Google Scholar] [CrossRef]
- Kachhadiya, D.D.; Murthy, Z.V.P. Highly efficient deep eutectic solvents coated MIL-53(Fe) embedded polyvinylidenefluoride MMMs for phenol separation using pervaporation. J. Mol. Liq. 2023, 390, 122981. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, W.; Zhang, X.; Ran, L.; Zhao, Q.; Zou, B.; Zhou, L.; Ye, Z. A novel design of self-assembled metal-organic frameworks MIL-53(Fe) modified resin as a catalyst for catalytic degradation of tetracycline. J. Clean. Prod. 2022, 348, 131385. [Google Scholar] [CrossRef]
- Zhao, M.; Li, Y.; Cui, S.; Chen, S.; Yang, J. Schottky junctions and localized surface plasmon resonance synergistic effect of Pt/MIL-53(Al. Cr. or Fe) for ketoprofen degradation: Pathways, DFT calculation, mechanism and structure-activity relationships. Appl. Surf. Sci. 2023, 607, 154980. [Google Scholar] [CrossRef]
- Sahoo, U.; Choudhury, S.; Pattnayak, S.; Aparajita, P.; Hota, G.; Ramakrishna, S. Strategic growth engineering of Ag self-doped Ag2CO3 on MIL-53 MOF: A novel p-n heterostructure facilitates serendipitous charge migration and remarkable multimodal photocatalytic activity. Mater. Today Commun. 2023, 35, 105842. [Google Scholar] [CrossRef]
- Gao, W.; Mu, B.; Zhu, Y.; Wang, A. Preparation of palygorskite/MIL-88A(Fe) composites for high-efficient removal of Congo red. Appl. Clay Sci. 2023, 242, 107003. [Google Scholar] [CrossRef]
- Wang, H.; Yu, S.; Gao, T.; Tan, X.; Meng, X.; Xiao, S. The efficient degradation of organic pollutants by Z-scheme MIL-88A@TiO2 heterojunction photo-Fenton catalyst: The synergistic effect of photocatalysis and Fenton catalysis. J. Alloys Compd. 2023, 960, 170688. [Google Scholar] [CrossRef]
- Yekkezare, H.; Tajik, H.; Mahmoodi, N.M. Green halogenation of aromatic compounds using environmentally friendly synthesized rod-like metal-organic framework (MIL-88A) catalyst. J. Mol. Struct. 2023, 1285, 135454. [Google Scholar] [CrossRef]
- Zhao, Q.; Yi, X.-H.; Wang, C.-C.; Wang, P.; Zheng, W. Photocatalytic Cr(VI) reduction over MIL-101(Fe)–NH2 immobilized on alumina substrate: From batch test to continuous operation. Chem. Eng. J. 2022, 429, 132497. [Google Scholar] [CrossRef]
- Liu, W.; Zhou, J.; Liu, D.; Liu, S.; Liu, X.; Xiao, S.; Feng, C.; Leng, C. Fe-MOF by ligand selective pyrolysis for Fenton-like process and photocatalysis: Accelerating effect of oxygen vacancy. J. Taiwan Inst. Chem. Eng. 2021, 127, 327–333. [Google Scholar] [CrossRef]
- She, Y.; Han, W.; Liu, H.; Hu, G.; Wang, H.; Wen, X.; Liu, L.; Feng, L.; Zhang, X.; Gong, J. Trace solvents-assisted mechanochemistry of waste poly(ethylene terephthalate) into MIL-53Al for efficient hydroelectricity generation. Chem. Eng. J. 2025, 515, 163895. [Google Scholar] [CrossRef]
- Wei, J.; Shen, W.; Liu, Y. Facile synthesis of SrWO4@MIL-88A(Fe) heterojunctions and their deep treatment of dye wastewater and municipal landfill leachate using photo-Fenton technology. J. Ind. Eng. Chem. 2023, 120, 103–120. [Google Scholar] [CrossRef]
- Liu, H.; Sun, H.; Fang, M.; Zhang, Y.; Kong, M.; Lv, Z.; Tan, X. Detection of Pb(II) in water via a NH2-MIL-53(Fe)@ITO electrochemical sensor. Mater. Chem. Phys. 2024, 314, 128833. [Google Scholar] [CrossRef]
- Li, X.; Guo, W.; Liu, Z.; Wang, R.; Liu, H. Quinone-modified NH2-MIL-101(Fe) composite as a+ redox mediator for improved degradation of bisphenol A. J. Hazard. Mater. 2017, 324, 665–672. [Google Scholar] [CrossRef]
- Mukherjee, D.; Das, P.; Kundu, S.; Kundu, L.M.; Mandal, B. Graphene quantum dots decorated MIL-100(Fe) composites for dye degradation. J. Photochem. Photobiol. A Chem. 2023, 442, 114776. [Google Scholar] [CrossRef]
- Xue, B.; Du, L.; Jin, J.; Meng, H.; Mi, J. In situ growth of MIL-88A into polyacrylate and its application in highly efficient photocatalytic degradation of organic pollutants in water. Appl. Surf. Sci. 2021, 564, 150404. [Google Scholar] [CrossRef]
- Hmoudah, M.; El-Qanni, A.; Tesser, R.; Esposito, R.; Petrone, A.; Jung, O.-S.; Salmi, T.; Russo, V.; Di Serio, M. Assessment of the robustness of MIL-88A in an aqueous solution: Experimental and DFT investigations. Mater. Sci. Eng. B 2023, 288, 116179. [Google Scholar] [CrossRef]
- Gan, W.; Guo, J.; Fu, X.; Jin, J.; Zhang, M.; Chen, R.; Ding, C.; Lu, Y.; Li, J.; Sun, Z. Introducing oxygen-doped g-C3N4 onto g-C3N4/TiO2 heterojunction for efficient catalytic gatifloxacin degradation and H2O2 production. Sep. Purif. Technol. 2023, 317, 123791. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, J.; Fang, T.; Tang, H.; Tang, H.; He, X. Oxygen vacancy-mediated BiVO4/Bi3O4Br S-scheme heterojunction for enhanced photocatalytic degradation of antibiotics. J. Colloid Interface Sci. 2025, 691, 137458. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Geng, J.; Li, H.; Bao, R.; Chen, H.; Wang, W.; Xia, J.; Wong, W.-Y. Exceedingly high photocatalytic activity of g-C3N4/Gd-N-TiO2 composite with nanoscale heterojunctions. Sol. Energy Mater. Sol. Cells 2017, 168, 91–99. [Google Scholar] [CrossRef]
- Yang, P.; Wu, C.; Zhang, X.; Sun, Q.; Hou, X.; Wang, T. Enhanced photocatalytic degradation of sulfathiazole via dual-ligand Zr-MOFs: A porphyrin and pyrene-based energy transfer. Sep. Purif. Technol. 2025, 354, 128727. [Google Scholar] [CrossRef]
- Wei, Y.; Lu, G.; Xie, D.; Sun, T.; Liu, Y.; Zhang, Y.; An, J.; Li, M.; Guo, H. Degradation of enrofloxacin in aqueous by DBD plasma and UV: Degradation performance, mechanism and toxicity assessment. Chem. Eng. J. 2022, 431, 133360. [Google Scholar] [CrossRef]
- Dias, F.F.; Oliveira, A.A.S.; Arcanjo, A.P.; Moura, F.C.C.; Pacheco, J.G.A. Residue-based iron catalyst for the degradation of textile dye via heterogeneous photo-Fenton. Appl. Catal. B Environ. 2016, 186, 136–142. [Google Scholar] [CrossRef]
- Li, X.; Kang, B.; Dong, F.; Zhang, Z.; Luo, X.; Han, L.; Huang, J.; Feng, Z.; Chen, Z.; Xu, J.; et al. Enhanced photocatalytic degradation and H2/H2O2 production performance of S-pCN/WO2.72 S-scheme heterojunction with appropriate surface oxygen vacancies. Nano Energy 2021, 81, 105671. [Google Scholar] [CrossRef]
- Daoud, F.; Pelzer, D.; Zuehlke, S.; Spiteller, M.; Kayser, O. Ozone pretreatment of process waste water generated in course of fluoroquinolone production. Chemosphere 2017, 185, 953–963. [Google Scholar] [CrossRef]
- Abbas, M.; Adil, M.; Ehtisham-ul-Haque, S.; Munir, B.; Yameen, M.; Ghaffar, A.; Shar, G.A.; Tahir, M.A. Vibrio fischeri bioluminescence inhibition assay for ecotoxicity assessment: A review. Sci. Total Environ. 2018, 626, 1295–1309. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, C.; Zheng, L.; Tang, M.; Ge, M. Activation of peroxydisulfate by MIL-88A(Fe) under visible light toward tetracycline degradation: Effect of synthesis temperature on catalytic performance. J. Solid State Chem. 2023, 323, 124051. [Google Scholar] [CrossRef]
- Fang, Z.; Liu, Y.; Qi, J.; Xu, Z.-F.; Qi, T.; Wang, L. Establishing a high-speed electron transfer channel via CuS/MIL-Fe heterojunction catalyst for photo-Fenton degradation of acetaminophen. Appl. Catal. B Environ. 2023, 320, 121979. [Google Scholar] [CrossRef]
- Gan, F.; Gao, C.; Ji, S.; Xing, Y.; Gong, T.; Tian, L.; Bai, Y.; Wu, Z. Efficient degradation of DDBAC in water by a heterogeneous Vis/H2O2 catalytic system based on MIL-53(Fe). J. Ind. Eng. Chem. 2025, 146, 366–377. [Google Scholar] [CrossRef]
- Wang, T.; Zhao, C.; Meng, L.; Li, Y.; Wang, D.; Wang, C.-C. Fe−O−P bond in MIL-88A(Fe)/BOHP heterojunctions as a highway for rapid electron transfer to enhance photo-Fenton abatement of enrofloxacin. Appl. Catal. B Environ. 2023, 334, 122832. [Google Scholar] [CrossRef]
- Domańska, M.; Hamal, K.; Jasionowski, B.; Łomotowski, J. Bacteriological contamination detection in water and wastewater samples using OD600. Pol. J. Environ. Stud. 2019, 28, 4503–4509. [Google Scholar] [CrossRef]
- ISO 11348-3:2007; Water Quality—Determination of the Inhibitory Effect of Water Samples on the Light Emission of Vibrio fischeri (Luminescent Bacteria Test)—Part 3: Method Using Freeze-Dried Bacteria. ISO: Geneva, Switzerland, 2007.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.