Abstract
We report the facile synthesis of hierarchical spiked cobalt selenide (Co0.85Se) microcrystals grown on nickel foam (NF) via a hydrothermal method followed by selenization. Derived from cobalt hydroxyl fluoride (Co(OH)F) microcrystals, the resulting Co0.85Se structures exhibit a robust architecture with well-defined spikes that offer abundant active sites and promote efficient charge transfer, thereby enhancing their electrocatalytic bifunctional activity toward the oxygen evolution reaction (OER) and urea oxidation reaction (UOR). The Co0.85Se/NF electrode delivers low overpotentials of 357 mV for OER and 236 mV for UOR at 100 mA cm−2. Furthermore, it exhibits a small Tafel slope (34.3 mV dec−1) and excellent durability for 24 h at 100 mA cm−2 during UOR. This simple and cost-effective strategy highlights the potential of hierarchical spiked Co0.85Se microcrystals as highly efficient electrocatalysts for urea-assisted OER and related sustainable energy conversion applications.
1. Introduction
The growing depletion of fossil fuels and the resulting environmental challenges have intensified the global pursuit of sustainable and clean energy technologies. Among various strategies, electrochemical water splitting is considered one of the most promising routes for hydrogen production [1,2,3]. However, its efficiency is significantly limited by the sluggish kinetics of the oxygen evolution reaction (OER) occurring at the anode [4,5]. A practical strategy to mitigate this issue involves replacing OER with the urea oxidation reaction (UOR), which proceeds at a much lower thermodynamic potential (0.37 V vs. 1.23 V for OER) [6,7,8,9]. Moreover, UOR not only reduces the overall energy input for hydrogen generation but also enables the electrochemical remediation of urea-rich wastewater. Therefore, the development of efficient, stable, and low-cost electrocatalysts for UOR is of great importance for both energy and environmental applications [10].
To date, a wide range of transition metal-based materials, particularly Ni-, Co-, and Fe-containing oxides, hydroxides, phosphides, and sulfides, have been explored as UOR electrocatalysts [11,12,13,14,15,16]. Among them, Ni-based catalysts dominate due to their ability to form high-valence Ni3+/Ni4+ species that act as the active centers during urea oxidation [17,18,19]. Recent studies have shown that Ni-based materials, such as Ni(OH)2/NiOOH, Ni-based alloys, and Ni chalcogenides, can achieve overpotentials typically in the range of 200–350 mV at current densities of 10–100 mA cm−2, with Tafel slopes of 40–80 mV dec−1, owing to the in situ formation of catalytically active NiOOH species under alkaline UOR conditions [17,18,19]. Despite their promising activity, Ni-based catalysts often suffer from limited intrinsic electrical conductivity and structural degradation at high current densities, motivating the exploration of alternative transition-metal-based systems. In this context, cobalt-based catalysts have attracted increasing attention because of their similar redox behavior and flexible electronic configuration, which can facilitate the adsorption and dehydrogenation of urea molecules [20,21,22].
In recent years, cobalt selenides have emerged as promising candidates for diverse electrochemical reactions, including OER, the hydrogen evolution reaction (HER), and overall water splitting [23,24,25]. Their high electrical conductivity, adjustable electronic structure, and stable surface chemistry enable fast charge transfer and excellent catalytic durability. Despite these advantages, studies on the application of cobalt selenides toward UOR remain limited. For example, Xu et al. prepared yolk–shell CoSe2 for efficient urea electrolysis, achieving notable UOR performance [26]. Zhao et al. synthesized self-supported CoSe2 nanorods that delivered an overpotential of 260 mV and a Tafel slope of 94.83 mV dec−1 [27]. Similarly, Lu et al. reported a CoSe2 hollow cube/CoSe2 nanosheet heterostructure that achieved 100 mA cm−2 at a low potential of 1.48 V (250 mV), saving 270 mV compared to OER [28]. Among cobalt selenide phases, non-stoichiometric Co0.85Se is particularly attractive due to its intrinsic cobalt vacancies, which modulate the electronic structure, enhance electrical conductivity, and increase the density of unsaturated Co active sites compared with stoichiometric phases such as CoSe2. These characteristics are highly beneficial for the UOR, where fast charge transfer and effective adsorption of urea-derived intermediates are required. In addition, Co0.85Se readily undergoes surface reconstruction under alkaline anodic conditions to form catalytically active CoOOH species while preserving a conductive selenide framework. The defect-rich lattice also facilitates the formation of hierarchical architectures with high electrochemically active surface area, making Co0.85Se a promising electrocatalyst for efficient and durable UOR.
Given these findings, a systematic investigation of cobalt selenide as a potential UOR electrocatalyst is essential to elucidate its intrinsic activity, stability, and surface reconstruction behavior under operating conditions. In this work, we synthesized spiked Co0.85Se microcrystals directly grown on nickel foam (NF) via a facile hydrothermal-selenization approach and evaluated their bifunctional electrocatalytic performance toward OER and UOR in alkaline media. The precursor cobalt hydroxyl fluoride (Co(OH)F) served as a structural template to generate the spiked Co0.85Se architecture. The resulting Co0.85Se/NF electrode exhibited outstanding catalytic activity, requiring only 357 mV and 236 mV to achieve 100 mA cm−2 for OER and UOR, respectively, along with excellent durability over 24 h and a small Tafel slope (34.3 mV dec−1). This study provides an effective strategy for constructing robust Co-based selenide architectures as efficient and durable electrocatalysts for urea-assisted water electrolysis and other sustainable energy applications.
2. Results and Discussion
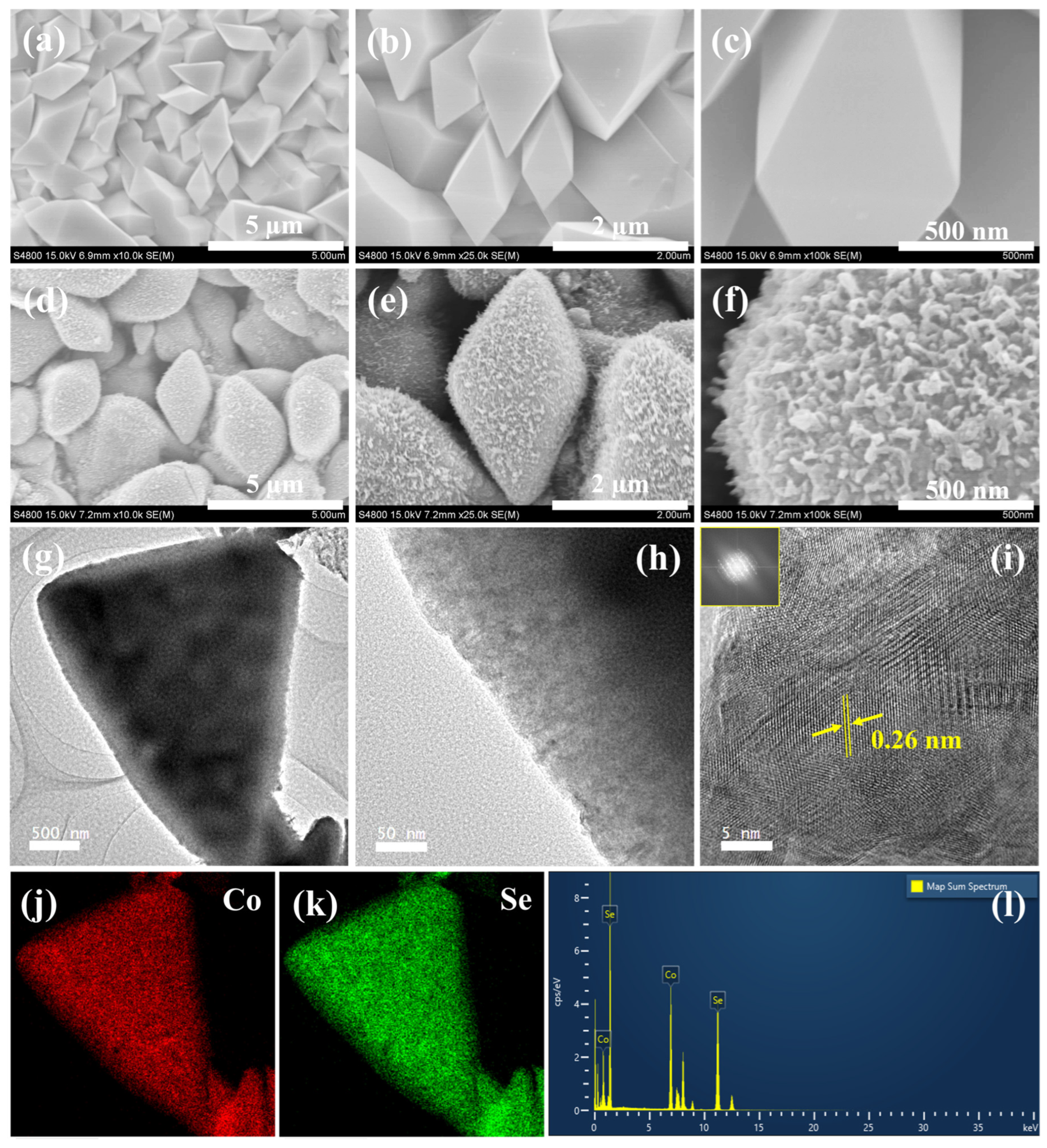
The morphology of the Co(OH)F and Co0.85Se electrodes was characterized by field-emission scanning electron microscopy (FESEM). As shown in Figure 1a–c, the Co(OH)F precursor consists of densely packed microcrystals with well-defined polyhedral shapes, primarily exhibiting smooth surfaces and sharp edges. The individual particles are about 1 µm in size and display compact, faceted geometries, indicating good crystallinity and mechanical robustness. Such a stable polyhedral framework provides a suitable template for the subsequent selenization process. The corresponding energy-dispersive X-ray spectroscopy (EDS) spectrum (Figure S1a) confirms the presence of Co, O, and F elements in the Co(OH)F precursor.
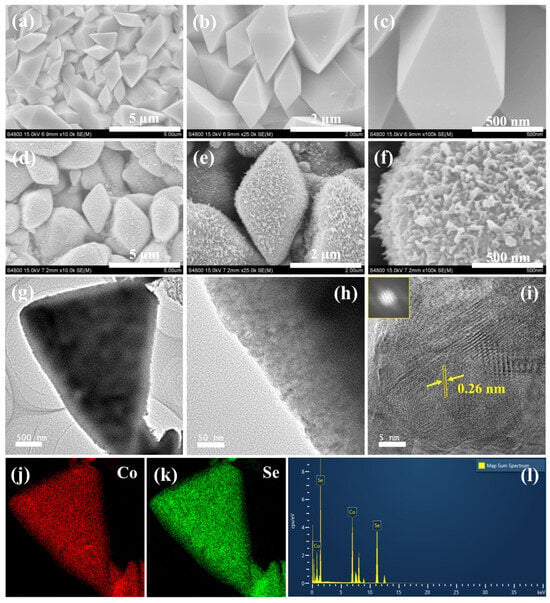
Figure 1.
(a–c) FESEM images of Co(OH)F microcrystals. (d–f) FESEM images of Co0.85Se microcrystals. (g–i) TEM and HRTEM images of Co0.85Se microcrystals. (j,k) Elemental mapping of Co and Se. (l) Corresponding EDX spectrum.
After selenization, the Co(OH)F precursor is converted into Co0.85Se microcrystals with a distinct hierarchical surface structure (Figure 1d–f). The overall polyhedral morphology is retained; however, the surfaces become densely decorated with fine, spike-like protrusions, resulting in a roughened texture. This transformation significantly enhances the surface area and exposes numerous electroactive sites, which are beneficial for facilitating the urea-assisted OER. Owing to the non-uniform distribution and varying dimensions of the spike-like surface features, precise quantification of individual protrusions is challenging. As observed from FESEM analysis (Figure 1d), the polyhedral Co0.85Se microcrystals exhibit typical overall sizes in the range of approximately 1–3 μm, confirming their micrometer-scale hierarchical architecture. The EDS spectrum of Co0.85Se (Figure S1b) confirms the presence of Co and Se, verifying successful selenization.
Field-emission transmission electron microscopy (FETEM) images (Figure 1g,h) further corroborate the FESEM observations, showing a clear contrast between the dense core and the roughened surface layer composed of nanoscale features. The high-resolution TEM image (Figure 1i) reveals distinct lattice fringes with an interplanar spacing of 0.26 nm, corresponding to the (101) plane of Co0.85Se, confirming its crystalline nature [29,30]. The inset SAED pattern displays sharp diffraction spots, consistent with a well-defined crystal structure. Elemental mapping (Figure 1j,k) demonstrates the uniform distribution of Co and Se throughout the microcrystal, while the corresponding EDS spectrum (Figure 1l) shows Co and Se peaks, confirming the high purity of the synthesized Co0.85Se material, making it a promising electrocatalyst for urea-assisted OER.
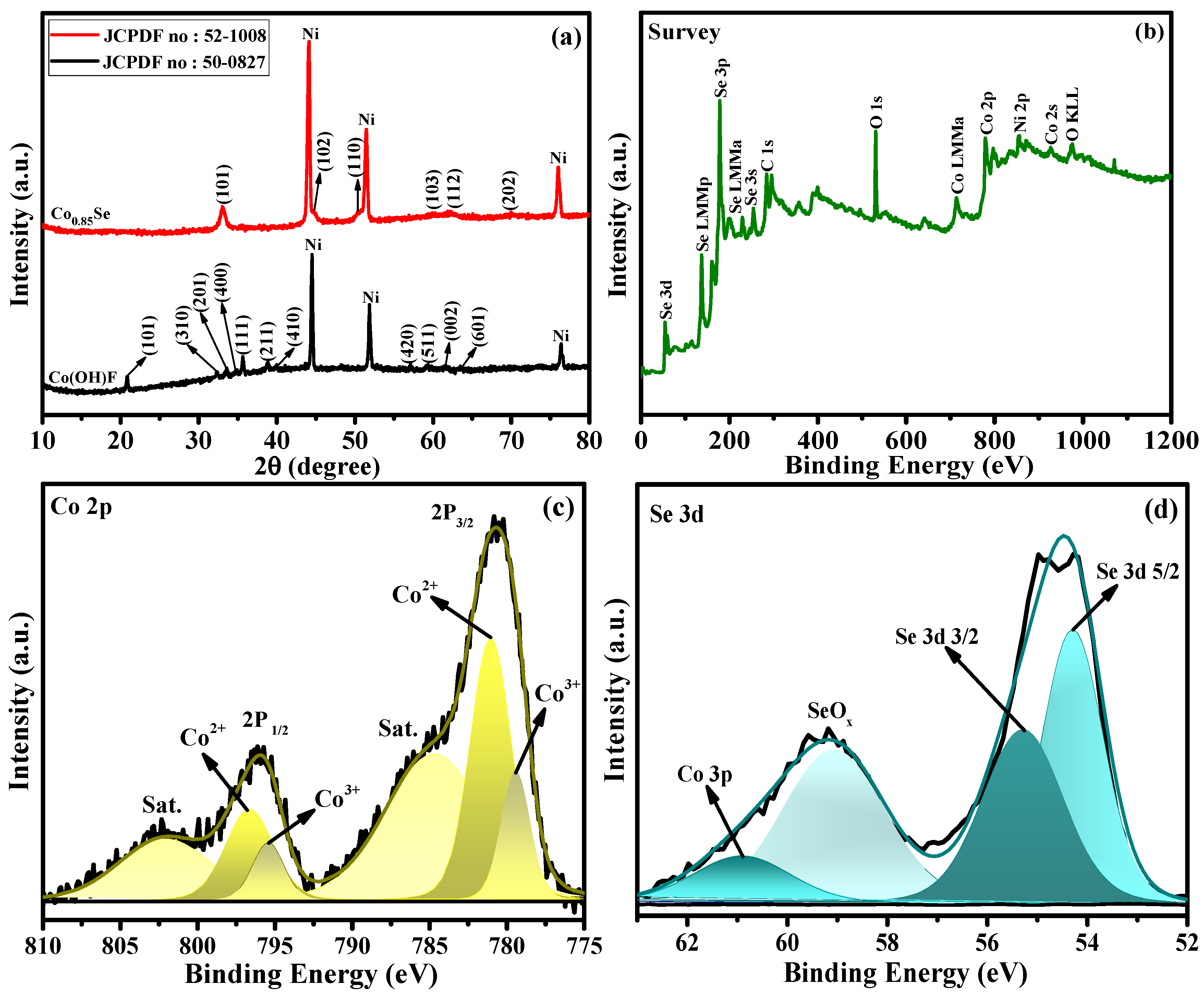
Figure 2a shows the XRD patterns of the synthesized samples, confirming the formation of orthorhombic Co(OH)F (JCPDS No. 50-0827) [31,32] and its successful conversion into hexagonal Co0.85Se (JCPDS No. 52-1008) after the selenization treatment. The diffraction peaks located at 33.6° (101), 45.0° (102), 51.1° (110), 60.5° (103), 62.5° (112), and 70.2° (202) match well with the standard Co0.85Se phase [33,34], confirming its high crystallinity and phase purity without any detectable secondary phases.
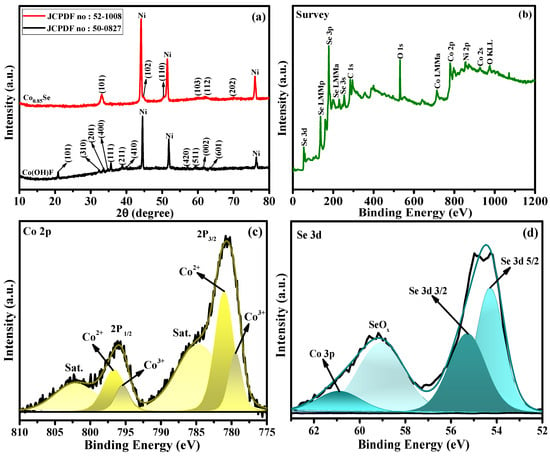
Figure 2.
(a) XRD patterns of Co0.85Se/NF and Co(OH)F/NF electrocatalysts. (b) XPS survey spectrum of Co0.85Se. (c) High-resolution Co 2p spectrum. (d) High-resolution Se 3d spectrum.
The surface chemical states of the Co0.85Se microcrystals were further examined using X-ray photoelectron spectroscopy (XPS). The survey scan (Figure 2b) reveals the presence of Co, Se, O, and C elements. The strong oxygen signals arise from Co–O, Se–O, and Co–O–Se bonding, while the weak carbon signals are related to atmospheric adventitious carbon. The high-resolution Co 2p spectrum (Figure 2c) displays two prominent peaks at 780.6 eV (Co 2p3/2) and 796.1 eV (Co 2p1/2), accompanied by well-defined satellite peaks at 785.1 eV and 802.2 eV [29,33,35]. These spectral features are characteristic of Co2+ and Co3+ species, confirming that cobalt exists in the +2 and +3 oxidation states. The Se 3d spectrum (Figure 2d) exhibits two main peaks centered at 54.3 eV (Se 3d5/2) and 55.5 eV (Se 3d3/2), corresponding to Se2− species in metal selenides. The higher binding energy peak at 58.6 eV originates from oxidized Se–O species, indicating surface oxidation due to air exposure [36,37]. In addition to the Co 2p doublet, a subtle contribution from the Co 3p level is also observed in the lower binding energy region (around ~60 eV) [35]. Although the Co 3p signal is weak and often overlapped with Se–O related features, its presence supports the identification of cobalt in the selenide framework and further validates the Co–Se bonding environment. The coexistence of Se2− and Se–O components suggest a mixed surface environment that may facilitate electron transfer processes relevant to UOR activity.
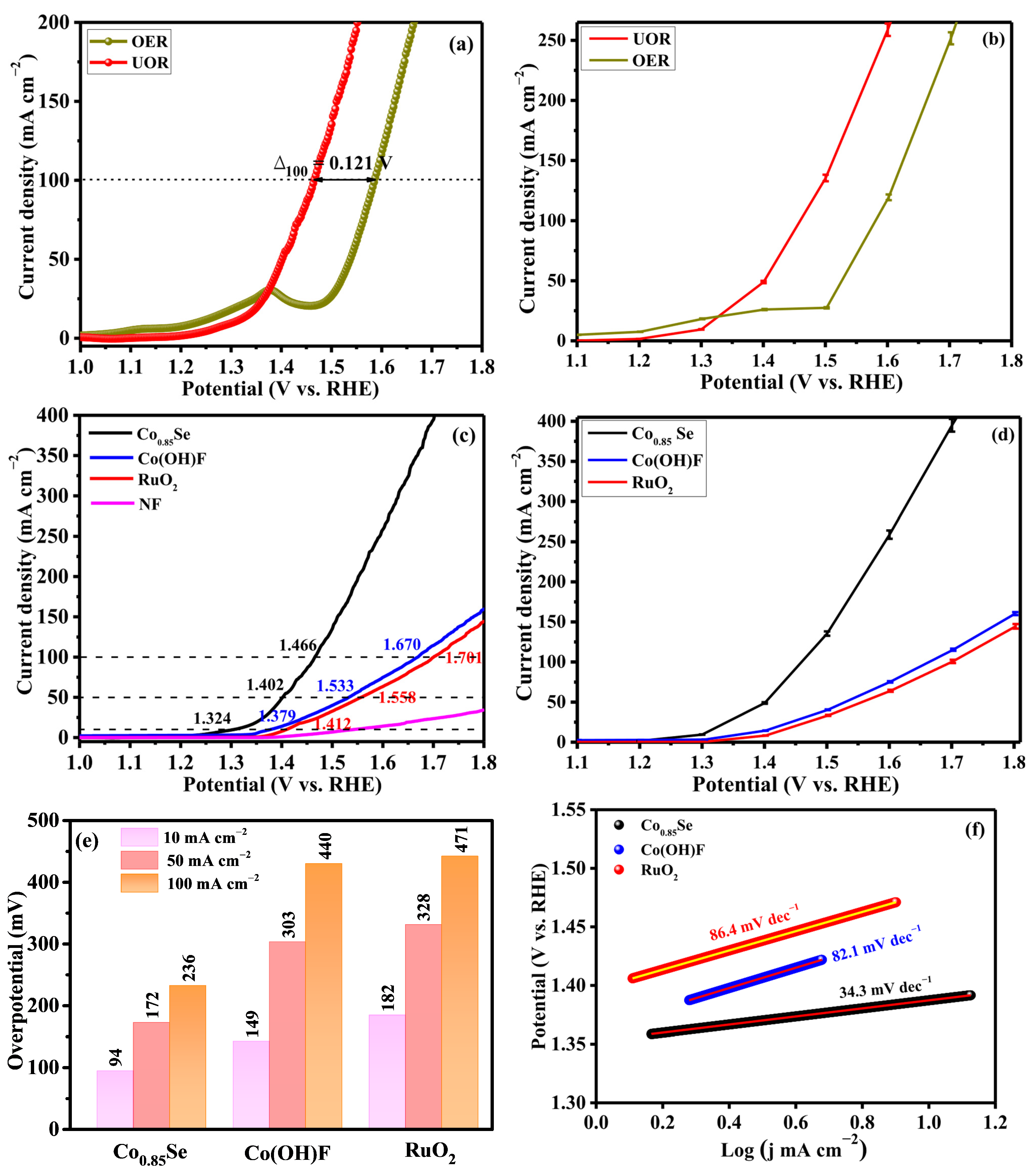
The electrocatalytic behavior of Co0.85Se toward both the OER and UOR was systematically evaluated in alkaline electrolyte (1.0 M KOH with and without 0.33 M urea). Linear sweep voltammetry (LSV) curves recorded at a scan rate of 5 mV s−1 (Figure 3a) demonstrate a pronounced advantage for UOR compared with OER. For the OER, the small current feature observed around 1.35 V is attributed to the surface oxidation of cobalt species under alkaline conditions. In urea-containing electrolyte, this oxidation-related feature is not observed because the urea oxidation reaction is kinetically favored and dominates the anodic process at lower potentials, thereby suppressing or masking the surface oxidation current. To achieve a benchmark current density of 100 mA cm−2, Co0.85Se requires only 1.466 V (vs. RHE) for UOR, whereas 1.587 V is needed for OER, corresponding to a 121 mV reduction in overpotential. This substantial potential saving highlights both the lower thermodynamic barrier of urea oxidation and the superior catalytic activity of Co0.85Se under operating conditions. Consequently, Co0.85Se emerges as a promising anode material for energy-efficient electrolysis coupled with urea remediation.
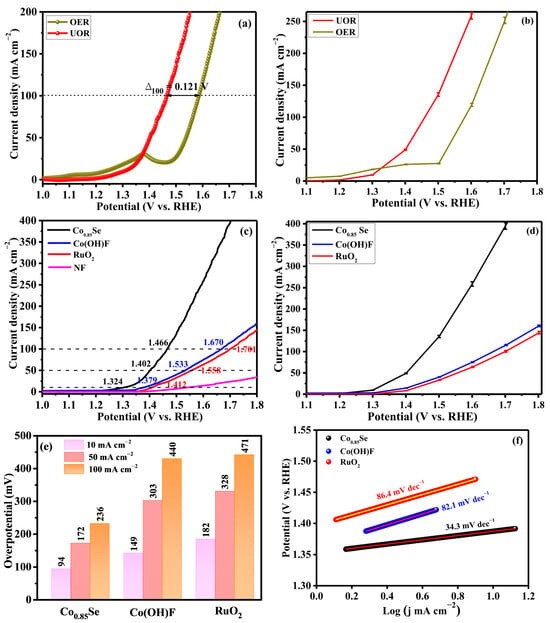
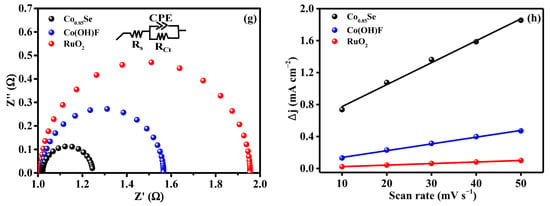
Figure 3.
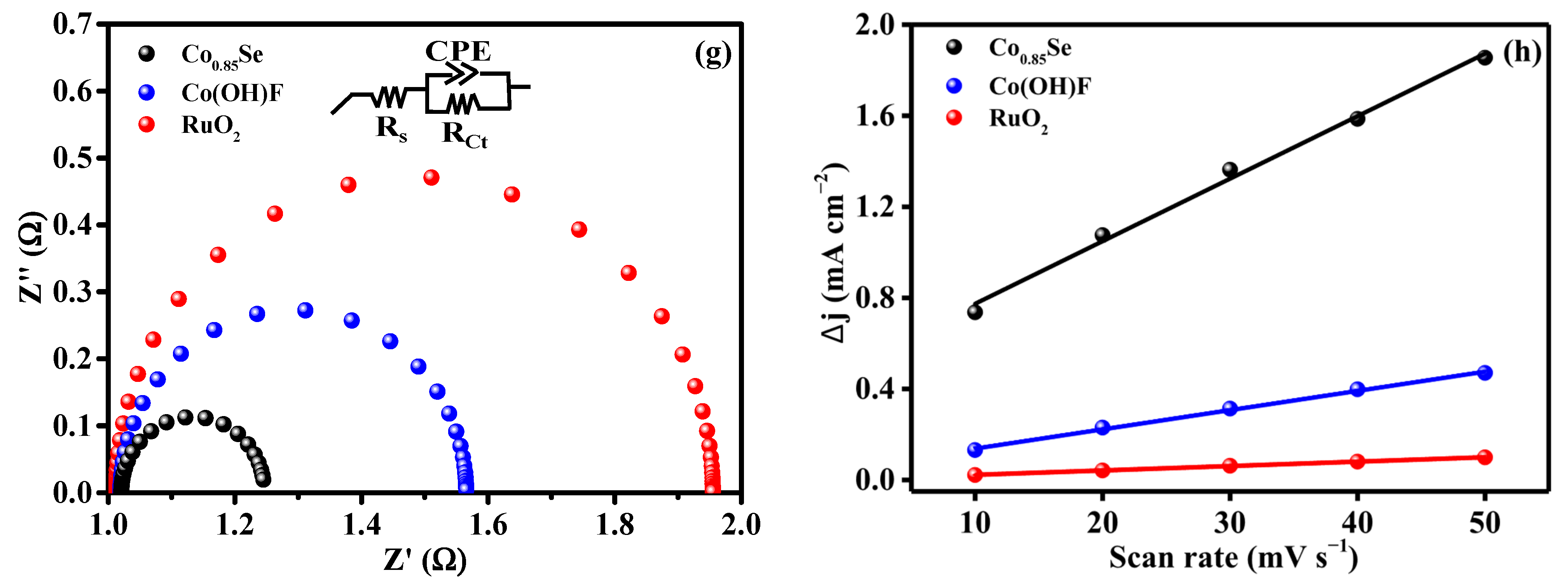
(a) LSV curves of Co0.85Se/NF in 1 M KOH and 1 M KOH + 0.33 M urea. (b) Error-bar polarization curves for OER and UOR in alkaline electrolyte (potential vs. RHE). (c) Comparison of UOR polarization curves for Co0.85Se, Co(OH)F, RuO2, and NF electrocatalysts. (d) Error-bar polarization curves of Co0.85Se, Co(OH)F, and RuO2 under identical conditions. (e) Overpotentials at 10, 50 and 100 mA cm−2 for different catalysts. (f) Tafel plots. (g) Nyquist plots obtained from EIS. (h) Double-layer capacitance (Cdl) extracted from CV curves.
Figure 3b presents statistical error-bar analysis comparing the anodic responses of OER and UOR, obtained from three independent measurements, and the error bars correspond to the standard deviation (±SD). For OER, the current density increases gradually with applied potential and rises sharply beyond ~1.50 V vs. RHE, indicating the onset of oxygen evolution. In contrast, UOR shows a markedly higher current response over the entire potential range, with an earlier and steeper increase. The small error bars for both reactions indicate excellent experimental reproducibility, while slightly larger deviations at higher potentials arise from intensified gas evolution and transient mass-transport effects. The superior UOR activity is attributed to the lower theoretical oxidation potential of urea (0.37 V vs. RHE) compared to water (1.23 V vs. RHE), enabling lower operating voltage and higher current output.
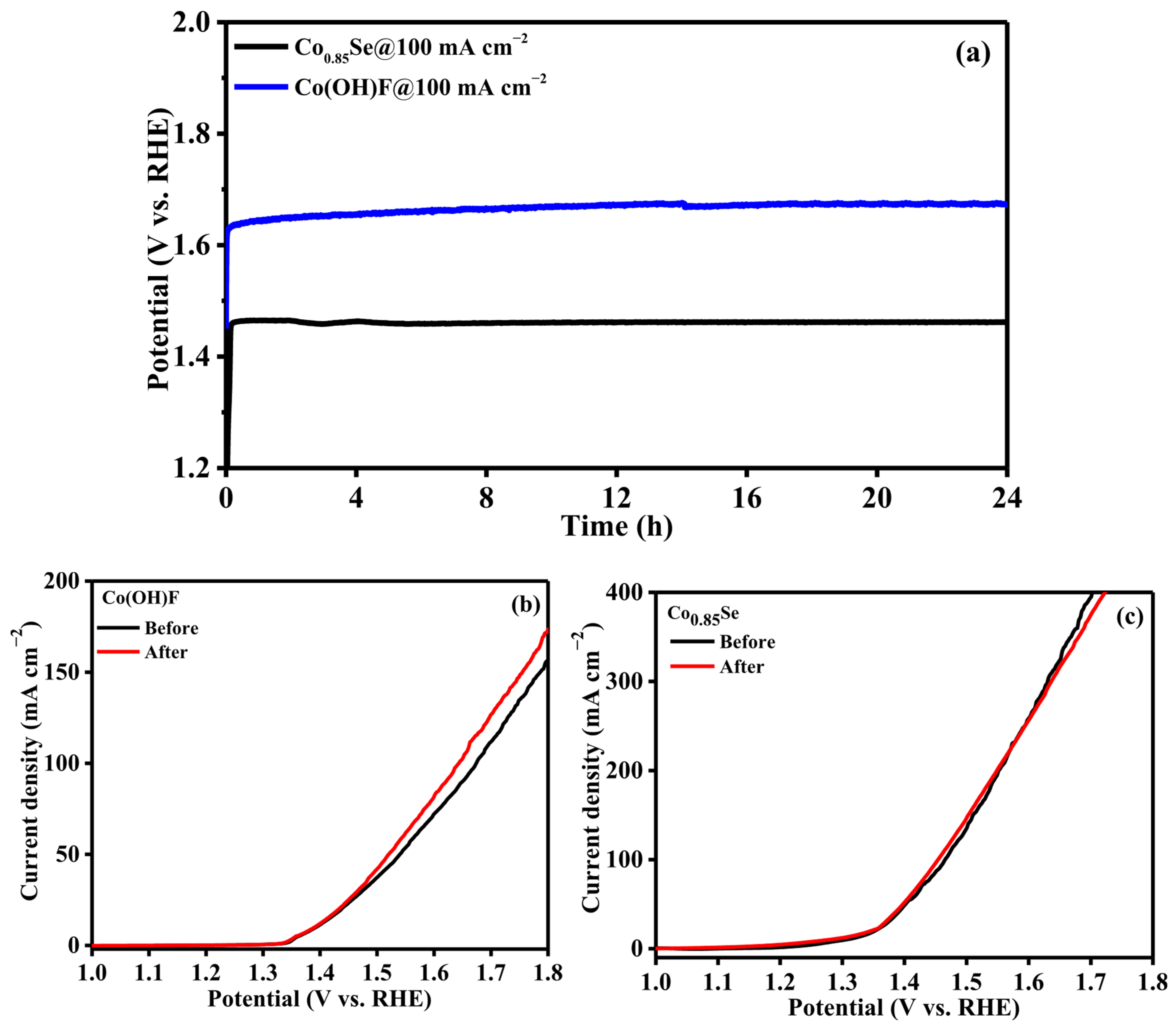
The catalytic performance of Co0.85Se was further compared with its precursor Co(OH)F and the benchmark RuO2 catalyst (Figure 3c). At a current density of 10 mA cm−2, Co0.85Se exhibits an overpotential of only 94 mV, which is significantly lower than that of Co(OH)F (149 mV) and RuO2 (182 mV). The lower onset potential and higher current density over the entire potential range indicate that Co0.85Se possesses favorable reaction thermodynamics and enhanced charge-transfer kinetics. The improved catalytic activity can be attributed to selenium incorporation, which modulates the electronic environment of cobalt and optimizes the adsorption energies of reaction intermediates. To evaluate the intrinsic contribution of the substrate, LSV curve of bare NF was performed under identical UOR conditions. As shown in Figure 3c, bare NF displays negligible urea oxidation activity compared with Co0.85Se/NF, confirming that NF serves mainly as a conductive current collector and structural support, while the catalytic performance is dominated by the Co0.85Se active phase. Figure 3d presents statistical error bars obtained from three independent measurements for Co0.85Se, Co(OH)F, and RuO2. The current density values represent mean responses, while the error bars indicate the standard deviation (±SD). Co0.85Se exhibits a steady increase in current density with applied potential, followed by a pronounced rise beyond ~1.50 V vs. RHE, reflecting fast anodic reaction kinetics. Co(OH)F delivers higher current densities than RuO2 across the entire potential range; for example, at 1.55 V vs. RHE, Co(OH)F reaches 58 mA cm−2 compared to 46 mA cm−2 for RuO2. The narrow error bars confirm excellent reproducibility and electrochemical stability, while slightly larger deviations at higher potential arise from intensified gas evolution and transient mass-transport effects.
Furthermore, Co0.85Se requires the lowest potential to reach current densities of 50 and 100 mA cm−2 for UOR compared with Co(OH)F and RuO2 (Figure 3e). The performance of Co0.85Se is also comparable to or even superior to that of many reported Co-based catalysts, such as yolk-shell CoSe2 (160 mV at 50 mA cm−2) [26], CoSe2/Co nanorods (260 mV at 10 mA cm−2) [27], CoSe2 hollow cube/CoSe2 nanosheets (250 mV at 100 mA cm−2) [28], and Co/CoSe2@CNx (140 mV at 10 mA cm−2) [38] (Table 1). To visually highlight the superior UOR performance, a bar chart comparing the overpotential at 10 mA cm−2 (η10) of Co0.85Se with representative catalysts from the literature is presented in Figure S2.

Table 1.
Comparison of UOR performance for Co0.85Se microcrystals with other selenide-based materials [39,40,41,42,43,44,45].
Further insight into the intrinsic reaction kinetics was obtained from the Tafel analysis. Co0.85Se exhibits a Tafel slope of 34.3 mV dec−1, which is significantly smaller than those of Co(OH)F (82.1 mV dec−1) and RuO2 (86.4 mV dec−1) (Figure 3f). The reduced slope indicates faster charge-transfer kinetics and a more favorable reaction pathway. A Tafel slope in the range of 30–50 mV dec−1 suggests a mechanism involving a rapid pre-equilibrium step followed by a rate-determining electron-transfer or chemical step. Accordingly, Co0.85Se sustains high current densities with smaller incremental energy input, a desirable characteristic for practical electrolyzer operation. The Co0.85Se catalyst exhibits an exceptionally low Tafel slope of 34.3 mV dec−1 for the urea oxidation reaction, indicating a distinct kinetic regime dominated by a fast electrochemical step rather than interfacial charge-transfer limitations. In alkaline media, UOR proceeds through urea adsorption, sequential dehydrogenation of –NH2 groups, C–N bond cleavage, and oxidation of carbon-containing intermediates to carbonate species. The low Tafel slope suggests that urea adsorption and *CO oxidation are not rate-determining on Co0.85Se. Instead, the kinetics are consistent with a rate-determining dehydrogenation step of adsorbed urea-derived intermediates (e.g., *NH2 → *NH), which involves a relatively low activation barrier. The incorporation of Se plays a critical role in modulating the electronic structure of Co active sites. Owing to its larger atomic radius and lower electronegativity, Se induces electron redistribution around Co centers and shifts the Co d-band center closer to the Fermi level, thereby optimizing the adsorption energies of key nitrogen-containing intermediates (*NH2 and *NH). This electronic tuning facilitates urea dehydrogenation, reduces the energy barrier of the rate-determining step, and results in accelerated UOR kinetics with a markedly reduced Tafel slope. These results indicate that the enhanced catalytic performance arises from intrinsic electronic structure modulation rather than solely from improved charge-transfer properties.
Electrochemical impedance spectroscopy (EIS) measurements (Figure 3g) further corroborate the kinetic advantages of Co0.85Se. The EIS spectra were recorded at 1.330 V (vs. RHE), and the impedance data were fitted using the equivalent circuit model shown in the inset. The Nyquist plots reveal a much smaller semicircle diameter for Co0.85Se than for Co(OH)F and RuO2, indicating a lower charge-transfer resistance (Rct). The Rct value of Co0.85Se (22 Ω) is significantly lower than that of Co(OH)F (58 Ω) and RuO2 (65 Ω). The reduced Rct demonstrates improved electrical conductivity and faster interfacial electron exchange between the catalyst surface and reactive intermediates. Such behavior is consistent with the metallic or semi-metallic nature of the Co–Se bonding network, which facilitates more efficient charge transport compared with hydroxide or oxyfluoride phases, thus supporting the superior LSV and Tafel results.
The electrochemical double-layer capacitance (Cdl) was estimated from non-Faradaic current–scan rate relationships (Figure 3h). Cyclic voltammetry (CV) measurements were conducted at different scan rates, and the corresponding CV curves are shown in Figure S3. Co0.85Se exhibits a substantially larger Cdl (44.1 mF cm−2) than Co(OH)F (2.44 mF cm−2) and RuO2 (13.9 mF cm−2), indicating a significantly increased ECSA. This enhancement is attributed to the surface roughening and structural reconstruction induced during the selenization process, which expose more catalytically active sites. The synergy between the enlarged active surface area and the improved intrinsic site activity arising from Se incorporation collectively accounts for the superior UOR performance of Co0.85Se.
Long-term stability is a key requirement for the practical application of UOR electrocatalysts. The durability of the Co0.85Se electrode was evaluated by chronopotentiometry at a constant current density of 100 mA cm−2 for 24 h. As shown in Figure 4a, the potential of Co0.85Se remains nearly unchanged throughout the entire test period, demonstrating excellent operational stability. Moreover, the LSV curve recorded after the stability test closely overlaps with the initial one (Figure 4c), confirming the remarkable electrochemical durability of Co0.85Se under continuous anodic operation. For comparison, the stability of the Co(OH)F electrode was also tested at 100 mA cm−2 for 24 h under identical conditions (Figure 4a). Unlike Co0.85Se, the polarization curve of Co(OH)F exhibited a noticeable shift after the long-term test (Figure 4b), indicating structural or compositional changes that lead to performance degradation. This comparison further highlights the superior durability of Co0.85Se as a UOR electrocatalyst. This outstanding stability can be attributed to two main factors: (i) the strong Co–Se bonding that suppresses catalyst dissolution and lattice distortion, and (ii) the in situ formation of a thin, stable CoOOH surface layer during anodic polarization, which preserves catalytic activity and protects the underlying structure from degradation. These synergistic effects ensure both the chemical and structural robustness of Co0.85Se under harsh electrolysis conditions.
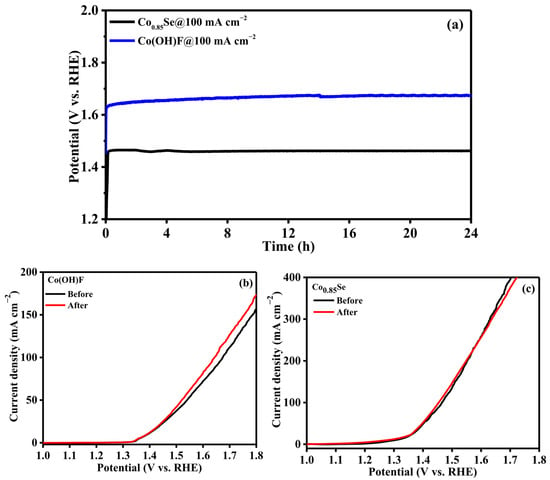
Figure 4.
(a) Chronopotentiometry stability test of Co0.85Se/NF and Co(OH)F/NF at 100 mA cm−2 for 24 h. (b,c) LSV curves recorded before and after CP.
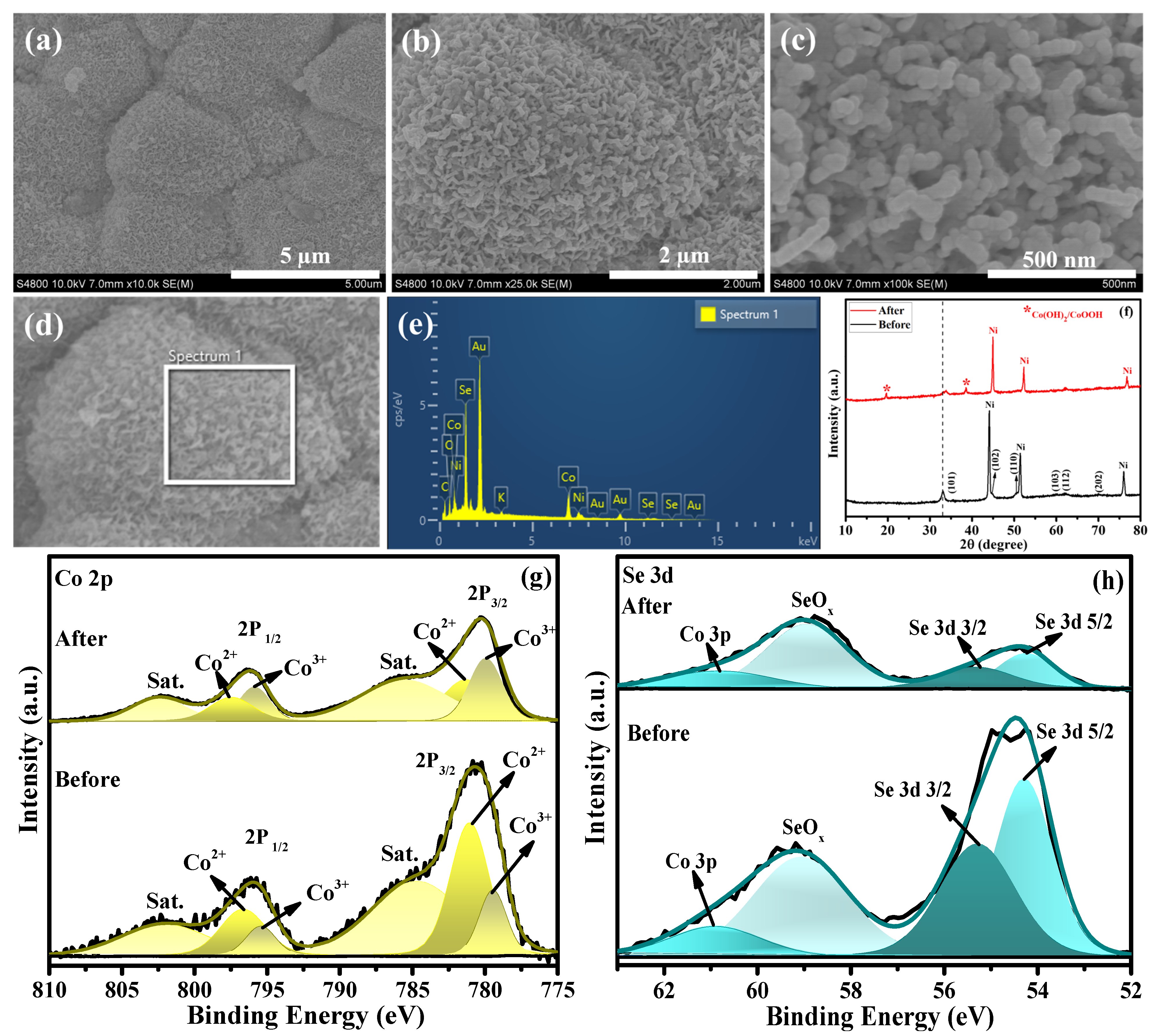
The surface morphology of the Co0.85Se/NF electrode before and after the long-term UOR stability test was examined by FESEM, as shown in Figure S4 and Figure 5. Before electrolysis (Figure S4a,b), the Co0.85Se electrode exhibits densely packed polyhedral microcrystals with roughened surfaces covered by fine nanoscale protrusions. This hierarchical surface texture, originating from the selenization of the Co(OH)F precursor, provides abundant active sites and efficient electrolyte accessibility. After 24 h of UOR operation at 100 mA cm−2, the overall polyhedral morphology remains largely preserved (Figure S4c,d), indicating excellent structural robustness. However, the microcrystals display slightly coarsened surfaces with partial aggregation of nanoscale features (Figure 5a–c), reflecting mild surface reconstruction during prolonged electrolysis. The corresponding EDX spectrum (Figure 5e) confirms the presence of Co, Se, Ni, O, and minor C and K elements. The increased oxygen signal after the stability test suggests the formation of a surface CoOOH layer while the detectable Se signal indicates retention of the underlying Co–Se framework. These observations collectively demonstrate that Co0.85Se maintains its microcrystalline architecture and chemical integrity, accounting for its excellent long-term stability and durability during urea oxidation.
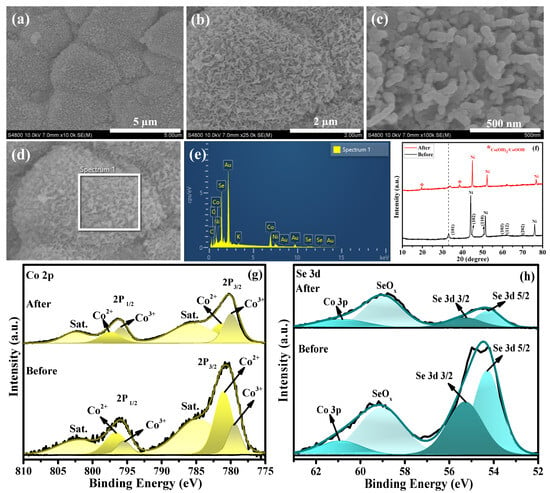
Figure 5.
(a–d) FESEM images of Co0.85Se/NF electrocatalyst after the CP stability test. (e) Corresponding EDX spectrum. (f) XRD patterns of Co0.85Se/NF electrocatalysts before and after the UOR. (g) Deconvoluted Co 2p spectra before and after UOR. (h) Deconvoluted Se 3d spectra before and after UOR.
The crystalline phase of the Co0.85Se/NF electrocatalyst after the UOR stability test was examined by XRD (Figure 5f). After the UOR stability test, the XRD pattern of Co0.85Se/NF largely preserves the characteristic diffraction peaks of the Co0.85Se phase, confirming the structural stability of the bulk selenide framework. Notably, two additional sharp peaks appear at approximately 19.5° and 38.5°, along with slight distortion of the main diffraction peak near 33°. These features are attributed to the formation of surface cobalt hydroxide/oxyhydroxide (Co(OH)2/CoOOH) species induced by anodic oxidation under alkaline UOR conditions. Such surface reconstruction, while retaining a conductive selenide core, is commonly observed for cobalt chalcogenide electrocatalysts and is considered beneficial for sustained catalytic activity.
The excellent catalytic activity of Co0.85Se toward UOR can be understood from the XPS analysis. The XPS spectra recorded before and after UOR clearly reveal the electron-transfer processes occurring in Co0.85Se. After UOR, the Co 2p spectrum (Figure 5g) displays two main peaks at binding energies of 780.3 and 795.2 eV ± 0.1 eV, corresponding to the Co 2p3/2 and Co 2p1/2 spin–orbit components. These peaks can be deconvoluted into sub-peaks at 779.9 and 796.2 eV, which represent Co3+ species and confirm the effective oxidation of Co2+ to Co3+ compared with the pre-UOR state. In addition, the peaks appearing at 781.8 and 796.9 eV ± 0.5 eV are assigned to Co2+, indicating the coexistence of mixed oxidation states. For Se 3d, the spectra obtained before and after UOR (Figure 5h) show characteristic peaks at 54.2 and 56.2 eV, corresponding to Se 3d5/2 and Se 3d3/2, along with a typical SeOx feature at 58.89 eV. The preservation of the main Se2− peaks, along with the observable oxide peak, indicates partial surface oxidation with an overlap of Co 3p further confirms the formation Co–Se framework.
Post-stability XPS analysis (Figure 5g,h) reveals a pronounced increase in the Co3+ component after prolonged urea oxidation, indicating surface reconstruction of Co0.85Se under anodic polarization. During electrochemical urea oxidation in alkaline media, the catalyst surface undergoes in situ oxidation under anodic bias, generating high-valent metal oxyhydroxide surface phases (Co3+ in CoOOH). These high-valent species are widely recognized as electrocatalytically active centers for both OER and UOR due to their ability to facilitate multi-electron and proton transfer processes, as confirmed by the XPS analysis [46]. Mechanistically, the formation of Co3+ occurs through the electro-oxidation of lower-valent cobalt (Co2+) under applied positive potentials:
Co(OH)2 + OH− → CoOOH + H2O + e−
Surface reconstruction leads to the formation of an oxyhydroxide layer on Co0.85Se. Once Co3+/CoOOH species are present on the surface, the adsorption and activation of urea (NH2–CO–NH2) can be rationalized in the context of electrochemical–chemical steps. Urea molecules coordinate more strongly to high-valent Co3+ active sites than to metallic or low-valent cobalt species because Co3+ exhibits a higher affinity for polar moieties, such as C=O and N–H donor/acceptor groups [47]. This enhanced adsorption orients the urea molecule such that the carbonyl carbon and adjacent NH2 groups are favorably positioned for facile deprotonation and C–N bond activation, which is often the rate-determining step in UOR [8].
Following adsorption on Co3+ sites, Co3+ acts as an electron acceptor, while the urea molecule undergoes dehydrogenation through the loss of protons from its N–H groups and electrons to the external circuit. The high-valent Co3+ species facilitate this process by enabling efficient electron transfer and stabilizing partially dehydrogenated intermediates (e.g., *CONH2 and *NH species). Similar behavior has been reported for NiOOH systems, in which Ni3+–O species interact with adsorbed urea and facilitate its oxidation to CO2 and N2, while Ni3+ is transiently reduced to Ni2+ during the reaction cycle [48].
In addition, the presence of Se2− in the cobalt selenide lattice plays a synergistic role in stabilizing high-valence cobalt species and modulating the electronic structure of the catalyst. Selenium, having a lower electronegativity than oxygen, donates electron density to cobalt, thereby facilitating the reversible Co2+/Co3+ redox transition under anodic polarization. This electronic interaction lowers the energy barrier for Co3+ formation and enhances charge-transfer kinetics, which is consistent with the low Tafel slope observed for the Co0.85Se catalyst [49]. Furthermore, partial surface oxidation of Co–Se bonds during UOR likely generates Co–Se–O interfacial sites, which have been proposed as highly active motifs for urea adsorption and oxidation.
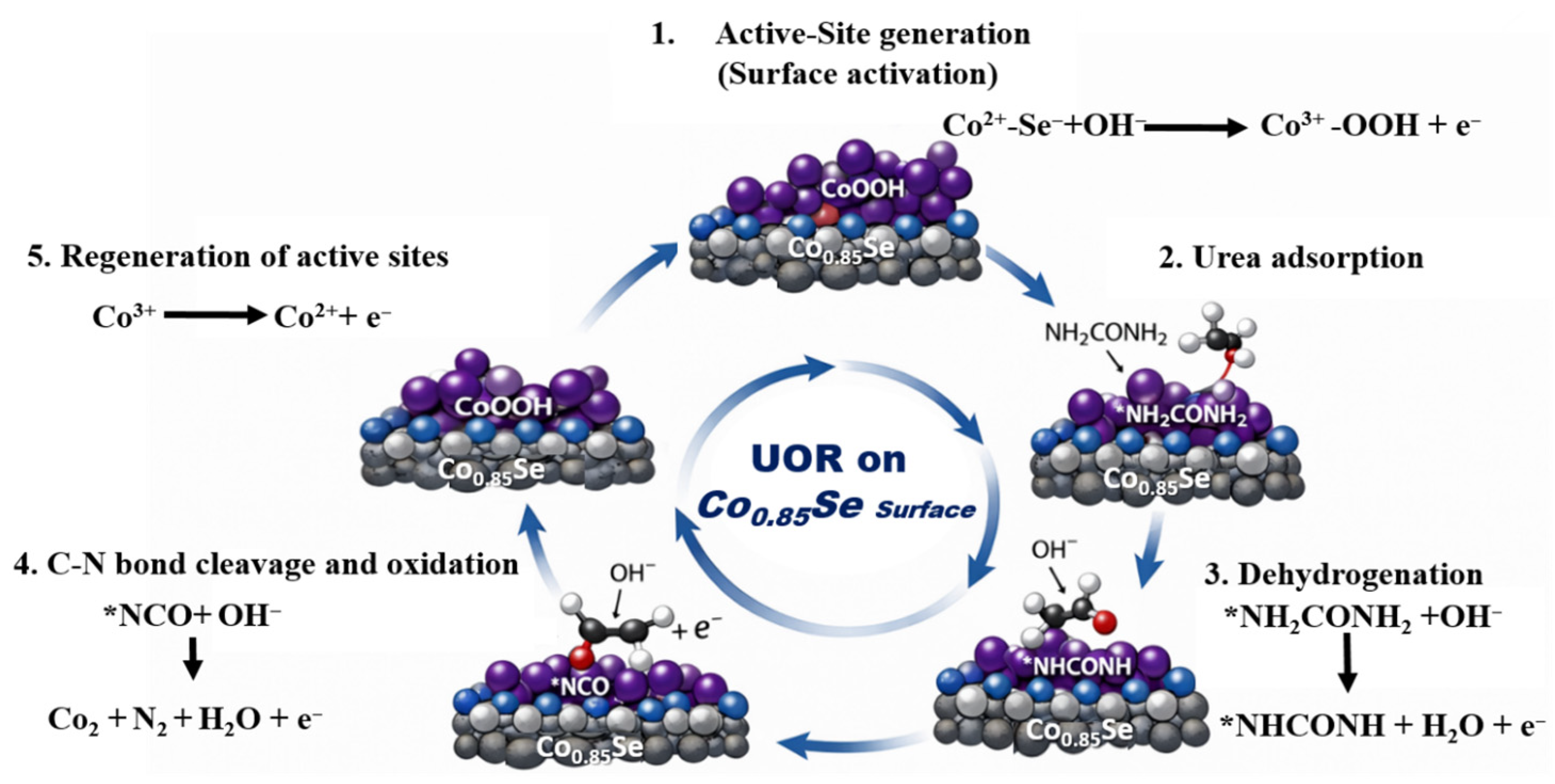
Based on these observations, a plausible UOR mechanism on Co0.85Se is proposed, as illustrated in Figure 6: (i) electrochemical oxidation of surface Co2+ to Co3+ to generate active sites; (ii) urea adsorption on Co3+ centers; (iii) Co3+-mediated dehydrogenation and C–N bond cleavage assisted by OH−; and (iv) reduction of Co3+ back to Co2+, completing the redox cycle [11]. The increased Co3+ content observed in the post-stability XPS spectra therefore provides direct spectroscopic evidence that high-valence cobalt species are not only stable under UOR conditions but also play a decisive role in sustaining high catalytic activity.
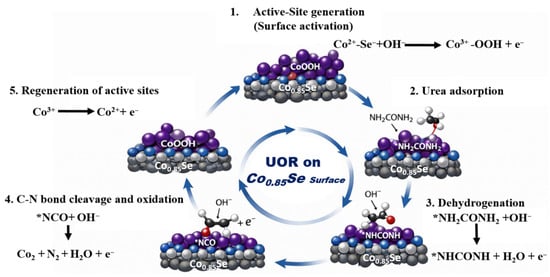
Figure 6.
Schematic illustration of the UOR mechanism on the Co0.85Se surface.
Overall, the effective redox transitions of Co and Se reveal the electron-transfer processes involved during UOR. Co is predominantly oxidized from Co2+ to Co3+, whereas Se 3d accepts electron density to maintain strong Co–Se2− bonding. This synergistic electronic interaction enhances electrocatalytic activity by facilitating charge transfer and reducing the UOR overpotential.
3. Experimental Section
3.1. Materials
Cobalt nitrate hexahydrate (Co(NO3)2·6H2O), ammonium fluoride (NH4F), sodium selenite (Na2SeO3), hydrazine hydrate (N2H4·H2O), urea, and potassium hydroxide (KOH) were purchased from Sigma-Aldrich. The RuO2 benchmark catalyst used in this study was a commercially available material (Ruthenium (IV) oxide, 99.9% trace metals basis, Alfa Aesar). Ethanol (C2H6O) and deionized water (DI H2O) were used as solvents, and NF substrates were obtained from MTI Corporation. All chemicals were of analytical grade and used without further purification.
3.2. Synthesis of Co(OH)F/NF Precursor
For the growth of the Co(OH)F nanostructure, 50 mM Co(NO3)2·6H2O and 150 mM NH4F were dissolved in 20 mL of DI H2O under magnetic stirring for 10 min. The homogeneous solution was transferred to a 100 mL Teflon-lined stainless-steel autoclave containing a pre-cleaned NF substrate (10 mm × 50 mm) fully immersed in the reaction solution. The hydrothermal reaction was conducted at 120 °C for 12 h. After cooling to room temperature, the obtained Co(OH)F/NF precursor was thoroughly rinsed with DI H2O and ethanol and then dried at 70 °C overnight.
3.3. Synthesis of Co0.85Se/NF
The conversion of Co(OH)F to Co0.85Se was achieved through a low-temperature selenization process. In a typical synthesis, 0.1 g of Na2SeO3 and 1 mL of hydrazine hydrate were dissolved in 20 mL DI H2O under stirring for 10 min. The prepared Co(OH)F/NF precursor was then immersed into the solution and maintained at 120 °C for 6 h in a Teflon-lined autoclave. During this process, Se2− species generated in situ from Na2SeO3 and hydrazine gradually replaced the F− ions, leading to the formation of spiked Co0.85Se structures on the NF substrate. The resulting black product was collected, washed repeatedly with DI H2O and ethanol, and dried at 70 °C for 12 h to obtain the final Co0.85Se/NF electrode.
The morphology of Co0.85Se is closely related to the synthesis conditions of the Co(OH)F precursor. Our previous study demonstrated that an optimized cobalt precursor concentration yields polyhedral Co(OH)F microcrystals with superior electrochemical properties [32]. Using this established condition, Co(OH)F templates were obtained in the present work and subsequently converted into hierarchical spiked Co0.85Se during selenization, leading to enhanced UOR activity.
In this process, hydrazine hydrate serves as a reducing agent, converting SeO32− ions from Na2SeO3 into reactive Se2− species under hydrothermal conditions. The in situ-generated Se2− ions subsequently replace F−/OH− species in the Co(OH)F precursor through an anion-exchange reaction, leading to the formation of hierarchical spiked Co0.85Se microcrystals on the NF substrate.
Detailed characterization parameters and electrochemical testing procedures are provided in the Supplementary Information.
4. Conclusions
In summary, hierarchical spiked polyhedral Co0.85Se microcrystals were successfully synthesized directly on NF via a facile hydrothermal method followed by selenization. The resulting Co0.85Se/NF electrode exhibits outstanding UOR activity, delivering low overpotentials of 94 mV at 10 mA cm−2 and 236 mV at 100 mA cm−2, along with a small Tafel slope of 34.3 mV dec−1 and excellent operational stability over 24 h in alkaline media. The superior UOR performance is attributed to the defect-rich Co0.85Se phase, hierarchical spike-like morphology, and efficient electron transport enabled by the conductive NF substrate. From a practical perspective, the synthesis approach is amenable to scale-up, as it employs low-temperature, solution-based processing with commercially available precursors. The direct growth of Co0.85Se on NF enables binder-free electrode fabrication and straightforward integration into urea electrolysis systems. Together with its high activity at industrially relevant current densities and excellent durability, the Co0.85Se/NF electrode shows strong potential for large-scale urea-assisted hydrogen production and wastewater remediation.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/catal16030232/s1, Experimental parts such as, materials characterization and electrochemical measurements. Figure S1: (a,b) EDX spectra of Co(OH)F and Co0.85Se microcrystals, respectively; Figure S2: Comparison of η10 values for Co0.85Se/NF and representative cobalt-selenide UOR electrocatalysts in alkaline media; Figure S3: Typical CV curves recorded at different scan rates in the non-Faradaic region for (a) Co0.85Se, (b) Co(OH)F, and (c) RuO2 electrocatalysts; Figure S4: (a,b) FESEM images of as-prepared Co0.85Se electrocatalyst. (c,d) FESEM images of Co0.85Se electrocatalyst after CP stability study.
Author Contributions
Conceptualization, J.A.R.; Methodology, J.A.R.; Validation, J.A.R., S.M.N.J., S.-H.K. and K.-S.A.; Investigation, J.A.R., S.M.N.J. and K.-S.A.; Data curation, J.A.R. and S.M.N.J.; Writing—original draft, J.A.R.; Writing—review & editing, J.A.R., S.M.N.J. and K.-S.A.; Visualization, J.A.R., S.M.N.J., S.-H.K. and K.-S.A.; Supervision, K.-S.A.; Project administration, K.-S.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the Regional Innovation System & Education (RISE) program through the Gyeongbuk RISE Center, funded by the Ministry of Education (MOE) and Gyeongsangbuk-do, Republic of Korea (2025-RISE-B0080529002339).
Data Availability Statement
The data used is already included in the manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Aslam, S.; Rani, S.; Lal, K.; Fatima, M.; Hardwick, T.; Shirinfar, B.; Ahmed, N. Electrochemical hydrogen production: Sustainable hydrogen economy. Green Chem. 2023, 25, 9543–9573. [Google Scholar] [CrossRef]
- Qian, Q.; Zhu, Y.; Ahmad, N.; Feng, Y.; Zhang, H.; Cheng, M.; Liu, H.; Xiao, C.; Zhang, G.; Xie, Y. Recent advancements in electrochemical hydrogen production via hybrid water splitting. Adv. Mater. 2024, 36, 2306108. [Google Scholar] [CrossRef]
- Deng, R.; Zhang, B.; Zhang, Q. Electrochemical water splitting for scale hydrogen production: From the laboratory to industrial applications. ChemCatChem 2024, 16, e202301165. [Google Scholar] [CrossRef]
- Gong, S.; Zhang, T.; Meng, J.; Sun, W.; Tian, Y. Advances in the mechanism investigation for the oxygen evolution reaction: Fundamental theory and monitoring techniques. Mater. Chem. Front. 2024, 8, 603–626. [Google Scholar] [CrossRef]
- Song, J.; Wei, C.; Huang, Z.-F.; Liu, C.; Zeng, L.; Wang, X.; Xu, Z.J. A review on fundamentals for designing oxygen evolution electrocatalysts. Chem. Soc. Rev. 2020, 49, 2196–2214. [Google Scholar] [CrossRef]
- Wang, H.; Zheng, X.; Fang, L.; Lu, S. Urea electrooxidation in alkaline environment: Fundamentals and applications. ChemElectroChem 2023, 10, e202300138. [Google Scholar] [CrossRef]
- Ge, W.; Lin, L.; Wang, S.-Q.; Wang, Y.; Ma, X.; An, Q.; Zhao, L. Electrocatalytic urea oxidation: Advances in mechanistic insights, nanocatalyst design, and applications. J. Mater. Chem. A 2023, 11, 15100. [Google Scholar] [CrossRef]
- Zhang, C.; Chen, S.; Guo, L.; Li, Z.; Yan, C.; Lv, C. Recent advances in urea electrocatalysis: Applications, materials and mechanisms. Chin. J. Chem. 2024, 42, 3441–3468. [Google Scholar] [CrossRef]
- Gautam, J.; Lee, S.-Y.; Park, S.-J. Cutting-edge optimization strategies and in situ characterization techniques for urea oxidation reaction catalysts: A comprehensive review. Adv. Energy Mater. 2025, 15, 2406047. [Google Scholar] [CrossRef]
- Zhu, B.; Liang, Z.; Zou, R. Designing advanced catalysts for energy conversion based on urea oxidation reaction. Small 2020, 16, 1906133. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Ruan, X.; Ganesan, M.; Wu, J.; Ravi, S.K.; Cui, X. Transition metal-based catalysts for urea oxidation reaction (UOR): Catalyst design strategies, applications, and future perspectives. Adv. Funct. Mater. 2024, 34, 2313309. [Google Scholar] [CrossRef]
- Ge, J.; Liu, Z.; Guan, M.; Kuang, J.; Xiao, Y.; Yang, Y.; Tsang, C.H.; Lu, X.; Yang, C. Investigation of the electrocatalytic mechanisms of urea oxidation reaction on the surface of transition metal oxides. J. Colloid. Interface Sci. 2022, 620, 442–453. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Zheng, X.; Fang, L.; Yin, F.; Liu, H. Rational engineering design of nickel hydroxides for urea oxidation reaction: A mini-review. Electrochem. Commun. 2023, 157, 107599. [Google Scholar] [CrossRef]
- Kumar, S.; Bhanuse, G.B.; Fu, Y.-P. Phosphide-based electrocatalysts for urea electrolysis: Recent trends and progress. ChemPhysChem 2024, 25, e202300924. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, N.; Du, X.; Han, X.; Zhang, X. Transition metal atoms M (M = Mn, Fe, Cu, Zn) doped nickel-cobalt sulfides on the Ni foam for efficient oxygen evolution reaction and urea oxidation reaction. J. Alloys Compd. 2022, 893, 162269. [Google Scholar] [CrossRef]
- Desalegn, B.Z.; Hern, K.; Seo, J.G. Synergistically interfaced bifunctional transition metal selenides for high-rate hydrogen production via urea electrolysis. ChemCatChem 2022, 14, e202100969. [Google Scholar] [CrossRef]
- Li, Q.; Wang, J.; Shi, Y.; Li, H.; Yang, H.; Xiang, K.; You, W.; Liang, J. Urea oxidation catalysts: A review on non-metallic enhancements in nickel-based electrocatalysts. Mater. Horiz. 2025, 12, 9952–9965. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Hao, L.; Hou, Y.; Zhang, J.; Yang, J.-H. Summary and application of Ni-based catalysts for electrocatalytic urea oxidation. Int. J. Hydrogen Energy 2024, 51, 966–981. [Google Scholar] [CrossRef]
- Ma, Y.; Ma, C.; Wang, Y.; Wang, K. Advanced nickel-based catalysts for urea oxidation reaction: Challenges and developments. Catalysts 2022, 12, 337. [Google Scholar] [CrossRef]
- Fang, M.; Xu, W.-B.; Han, S.; Cao, P.; Xu, W.; Zhu, D.; Lu, Y.; Liu, W. Enhanced urea oxidization electrocatalysis on spinel cobalt oxide nanowires via on-site electrochemical defect engineering. Mater. Chem. Front. 2021, 5, 3717–3724. [Google Scholar] [CrossRef]
- Zheng, S.; Qin, H.; Cao, X.; Wang, T.; Lu, W.; Jiao, L. Electron modulation of cobalt carbonate hydroxide by Mo doping for urea-assisted hydrogen production. J. Energy Chem. 2022, 70, 258–265. [Google Scholar] [CrossRef]
- Ding, M.; Zhang, S.; Zhou, H.; Xu, H.; Lin, T.; Tao, H.; Ma, Y.; Li, Y. Enhanced electrochemical oxidation of urea contaminant and simultaneous H2 generation over highly active Co-based array catalysts. Sep. Purif. Technol. 2025, 355, 129438. [Google Scholar] [CrossRef]
- Truong, H.B.; Tran, N.T.; Do, H.H. Recent advances, challenges, and perspectives of CoSe2-based electrocatalysts for oxygen evolution reaction. Int. J. Hydrogen Energy 2025, 162, 150765. [Google Scholar] [CrossRef]
- Zhong, H.; Roldan, C.A.C.; Zhao, Y.; Zhang, S.; Feng, Y.; Vante, N.A. Recent advances of cobalt-based electrocatalysts for oxygen electrode reactions and hydrogen evolution reaction. Catalysts 2018, 8, 559. [Google Scholar] [CrossRef]
- Zhao, Y.; Jin, B.; Zheng, Y.; Jin, H.; Jiao, Y.; Qiao, S.-Z. Charge state manipulation of cobalt selenide catalyst for overall seawater electrolysis. Adv. Energy Mater. 2018, 8, 1801926. [Google Scholar] [CrossRef]
- Xu, H.; Ye, K.; Yin, J.; Zhu, K.; Yan, J.; Wang, G.; Cao, D. In situ growth of ZIF67 at the edge of nanosheet transformed into yolk-shell CoSe2 for high efficiency urea electrolysis. J. Power Sources 2021, 491, 229592. [Google Scholar] [CrossRef]
- Zhao, K.; Chen, X.; Liu, H.; Wang, J.; Zhang, J. Self-supported CoSe2 nanorods for efficient oxygen evolution and urea oxidation. ACS Appl. Nano Mater. 2024, 7, 6927–6934. [Google Scholar] [CrossRef]
- Lu, B.; Li, Z.; Yin, J.; Zhu, K.; Ye, K. The CoSe2 hollow cube/CoSe2 nanosheet interface catalyst for efficient electrolysis of urea–assisted hydrogen production at industrial–grade currents. Appl. Catal. B Environ. 2024, 350, 123940. [Google Scholar] [CrossRef]
- Rajesh, J.A.; Kwon, S.-J.; Manikandan, R.; Kang, S.-H.; Ahn, K.-S. Design of Co0.85Se microsphere-like architectures for high-performance hybrid supercapacitors. Crystals 2025, 15, 217. [Google Scholar] [CrossRef]
- Zhong, Q.-S.; Xia, W.-Y.; Liu, B.-C.; Xu, C.-W.; Li, N. Co0.85Se on three-dimensional hierarchical porous graphene-like carbon for highly effective oxygen evolution reaction. Int. J. Hydrogen Energy 2019, 44, 10182–10189. [Google Scholar] [CrossRef]
- Alam, S.; Sahu, T.K.; Qureshi, M. One-dimensional Co(OH)F as a noble metal-free redox mediator and hole extractor for boosted photoelectrochemical water oxidation in worm-like bismuth vanadate. ACS Sustain. Chem. Eng. 2021, 9, 5155–5165. [Google Scholar] [CrossRef]
- Rajesh, J.A.; Park, J.-Y.; Kang, S.-H.; Ahn, K.-S. Effect of molar concentration on the crystallite structures and electrochemical properties of cobalt fluoride hydroxide for hybrid supercapacitors. Electrochim. Acta 2022, 414, 140203. [Google Scholar] [CrossRef]
- Rajesh, J.A.; Kim, J.-Y.; Kang, S.-H.; Ahn, K.-S. Facile synthesis of microsphere-like Co0.85Se structures on nickel foam for a highly efficient hydrogen evolution reaction. Micromachines 2023, 14, 1905. [Google Scholar] [CrossRef]
- Hussain, N.; Wu, F.; Xu, L.; Qian, Y. Co0.85Se hollow spheres constructed of ultrathin 2D mesoporous nanosheets as a novel bifunctional-electrode for supercapacitor and water splitting. Nano Res. 2019, 12, 2941–2946. [Google Scholar] [CrossRef]
- Zheng, H.; Xu, H.-S.; Hu, J.; Liu, H.; Wei, L.; Wu, S.; Li, J.; Huang, Y.; Tang, K. Electrochemical performance of CoSe2 with mixed phases decorated with N-doped rGO in potassium ion-batteries. RSC Adv. 2022, 12, 21374–21384. [Google Scholar] [CrossRef]
- Pan, Z.; Zhan, Y.; Shen, P.K. Mo-doped Co3S4/CoSe2 nanosheet heterostructures for efficient water splitting in a wide pH range. ACS Appl. Nano Mater. 2025, 8, 17174–17183. [Google Scholar] [CrossRef]
- Peng, Q.; Zhuang, X.; Wei, L.; Shi, L.; Isimjan, T.T.; Hou, R.; Yang, X. Niobium-incorporated CoSe2 nanothorns with electronic structural alterations for efficient alkaline oxygen evolution reaction at high current density. ChemSusChem 2022, 15, e202200827. [Google Scholar] [CrossRef]
- Li, H.; Liu, Y.; Huang, L.; Xin, J.; Zhang, T.; Liu, P.; Chen, L.; Guo, W.; Gu, T.; Wang, G. 2D metal–organic framework derived Co/CoSe2 heterojunctions with interfacial electron redistribution as bifunctional electrocatalysts for urea-assisted rechargeable Zn–air batteries. J. Mater. Chem. A 2023, 11, 5179–5187. [Google Scholar] [CrossRef]
- Boakye, F.O.; Sendeku, M.G.; Kumar, A.; Ajmal, S.; Owusu, K.A.; Ibrahim, K.B.; Tabish, M.; Zaman, F.uz.; Mushtaq, M.A.; Alotaibi, K.M.; et al. Engineering active sites on binary metal selenide heterointerface catalyst to boost urea electrooxidation. Appl. Catal. B Environ. 2024, 352, 124013. [Google Scholar] [CrossRef]
- Wang, R.; Cao, J.; Zhang, R.; Sun, Z.; Wang, F.; Wu, H. Self-assembly of Ni2P/CoSe2 heterostructure as bifunctional electrocatalysts for urea-water electrolysis. Int. J. Hydrogen Energy 2022, 47, 29593–29603. [Google Scholar] [CrossRef]
- Yuan, S.; Wu, Y.; Huang, L.; Zhang, Z.; Chen, W.; Wang, Y. Engineering Ni0.85Se/CoSe2 heterojunction for enhanced bifunctional catalysis in urea-assisted hydrogen production. J. Colloid Interface Sci. 2025, 683, 981–994. [Google Scholar] [CrossRef] [PubMed]
- Meshesha, M.M.; Chanda, D.; Jang, S.G.; Yang, B.L. Enhancing cobalt-based bimetallic selenide performance for urea and water electrolysis through interface engineering. Chem. Eng. J. 2023, 474, 145708. [Google Scholar] [CrossRef]
- Li, B.; Yang, Y.; Zhang, M.; Wu, H.; Ding, Y. Novel nickel-doped CoMnxSe2 Chalcogenides for boosting hydrogen evolution and urea oxidation. Int. J. Hydrogen Energy 2024, 51, 1208–1215. [Google Scholar] [CrossRef]
- Wang, H.; Du, X.; Zhang, X. Transition metal-based selenide materials derived from ZIF-67 as efficient urea and seawater splitting electrocatalyst. Renew. Energy 2025, 244, 122663. [Google Scholar] [CrossRef]
- Cheng, Y.; Zhu, L.; Gong, Y. V-doped transition metal selenides derived from NiFe Prussian blue analogues for efficient oxygen evolution reaction and urea oxidation reaction. Int. J. Hydrogen Energy 2024, 92, 1049–1058. [Google Scholar] [CrossRef]
- Alotaibi, N.; Hammud, H.H.; Otaibi, N.A.; Prakasam, T. Electrocatalytic properties of 3D hierarchical graphitic carbon−cobalt nanoparticles for urea oxidation. ACS Omega 2020, 5, 26038–26048. [Google Scholar] [CrossRef]
- Li, S.; Peng, S.; Huang, L.; Cui, X.; Al-Enizi, A.M.; Zheng, G. Carbon-coated Co3+-rich cobalt selenide derived from ZIF-67 for efficient electrochemical water oxidation. ACS Appl. Mater. Interfaces 2016, 8, 20534–20539. [Google Scholar] [CrossRef]
- Zhou, T.; Jagadeesan, S.N.; Zhang, L.; Deskins, N.A.; Teng, X. Enhanced urea oxidation electrocatalytic activity by synergistic cobalt and nickel mixed oxides. J. Phys. Chem. Lett. 2024, 15, 81–89. [Google Scholar] [CrossRef]
- Shi, J.; Wei, T.; Yan, X.; Sun, K.; Du, L.; Qian, K.; Shi, M.; Shang, J.; Guo, H.; Sultana, F.; et al. Constructing efficient-active-centers selenide heterointerface for enhanced urea oxidation-driven hydrogen evolution. Int. J. Hydrogen Energy 2025, 127, 576–585. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.