Abstract
In this study, the feasibility of a new group of alkali-free high-entropy ABO3-δ perovskite cathodes, in which the B-site is occupied by a mixture of 3d and 4d/5d elements, is examined. Six different materials with a general formula of La(Cu0.2Ni0.2X10.2X20.2Y10.2)O3-δ (where X1, X2 = mixture of two Co, Ga, Fe, and Y1 = one of Nb/Ta): La(Cu0.2Ni0.2Co0.2Ga0.2Nb0.2)O3-δ, La(Cu0.2Ni0.2Co0.2Ga0.2Ta0.2)O3-δ, La(Cu0.2Ni0.2Fe0.2Ga0.2-Nb0.2)O3-δ, La(Cu0.2Ni0.2Fe0.2Ga0.2-Ta0.2)O3-δ, La(Cu0.2Ni0.2Co0.2Fe0.2Nb0.2)O3-δ, La(Cu0.2Ni0.2Co0.2Fe0.2Ta0.2)O3-δ were synthesized, with five of them possessing a single-phase, Pnma perovskite structure. While in the case of the basic properties, such as electrical conductivity or thermomechanical behavior, the studied oxides show a number of similarities, the differences between them become more apparent when low-temperature Oxygen Evolution Reaction (OER) and high-temperature Oxygen Reduction Reaction performance is evaluated. The overall best performance is achieved by La(Cu0.2Ni0.2Co0.2Fe0.2Nb0.2)O3-δ and La(Cu0.2Ni0.2Co0.2Fe0.2Ta0.2)O3-δ compositions, with the former offering slightly faster OER kinetics, and the latter exhibiting superior polarization resistance as an SOFC cathode. Overall, the materials exhibit a strong correlation between composition and properties, with potential for further development into non-equimolar compositions of superior performance.
1. Introduction
According to NOAA’s Global Monitoring Lab, the average atmospheric carbon dioxide concentration reached 422.8 ppm in 2024, a new record for recorded levels. Since 2000, its amount has increased by 15% (from ~368 ppm) [1]. One of the potential directions for decarbonizing the energy system is the development of various hydrogen technologies. In the case of transport, low-temperature proton-exchange membrane fuel cells (PEMFC) are commonly used, while in the case of stationary energy production in cogeneration systems—combined heat and power (CHP), high-temperature solid oxide fuel cells (SOFCs) are most often considered [2,3]. However, the potential of hydrogen technologies extends beyond just energy production, with the possibility of various fuel cells to work reversibly, as electrolysis cells, enabling conversion of electrical energy to chemical energy, and storage of the produced fuels [4]. This feature is of a high importance for renewable energy sources-based power grids. Regardless of whether high- or low-temperature H2 technologies are considered, in most of the systems several bottlenecks can be identified that affect the pace of their development. In the case of high-temperature technologies, such as solid-oxide fuel cells, the main limiting factor nowadays is the insufficient catalytic activity of the cathode materials, towards the oxygen reduction reaction (ORR) [2]. In contrast, for low-temperature technologies such as proton-exchange membrane electrolysis (PEM) or alkaline water electrolysis (AEM), the major challenges are associated with catalysis of the oxygen evolution reaction (OER), primarily due to the high cost of suitable catalysts and their insufficient long-term stability [5,6].
One of the groups of materials capable of meeting diverse requirements of various hydrogen technologies are perovskite-structured oxides. In terms of the SOFC applications, they have established themselves as by far the most widely used solution for ORR-active cathode materials, starting with the La1-xSrxMnO3-δ (LSM) utilized in the early, high-temperature generation of SOFC devices [2], through such compositions as La1-xSrxCo1-yFeyO3-δ (LSCF) [7] used in the intermediate-temperature SOFCs, up to the most recent, high-performance cathode materials for low-temperature SOFC devices, such as SrCo0.8Ta0.1Nb0.1O3 (SCNT) [8] or Ba0.5Sr0.5Co0.2Fe0.8O3 (BSCF) [9]. Similarly, perovskites have gathered a great deal of interest with regard to OERs, both at high [10] and low temperatures [11]. It is worth noting that, despite the obvious differences in the catalyzed processes, in many cases the same compositions display excellent ORR and OER performance [12], as the ORR/OER catalytic activity can be correlated with very similar features in electronic structure, such as suitable orbital filling [12], leading to cases where the same materials, e.g., BSCF, excel in both types of catalysis.
Despite all the development and unquestionable progress in the field of perovskite electrocatalysts, the state-of-the-art materials still suffer from multiple limitations. In the case of high-temperature applications, improvements in the ORR performance in most cases come at the expense of decreased stability, both in terms of chemical reactivity to volatile Cr species and CO2, originating from interconnects and atmosphere, respectively [13,14], and general instability of the structure, leading to its degradation over time [15]. Insufficient stability is also an issue for low-temperature OER, with problems such as dissolution and surface reconstruction affecting the potential of these materials in industrial-scale applications [16]. As a result, the past few years have seen a continuous search by scientists for novel, unconventional solutions.
One of the primary examples of new ideas originating from materials science is the recent development of high-entropy electrocatalysts. Originally introduced in metallic high-entropy alloys, the concept of near equimolar, multicomponent compounds was seamlessly transferred to other types of systems, including oxides [17]. Since then, high-entropy oxides (HEOs) have proved their immense potential with regard to a number of applications, including catalysis. In terms of SOFC applications, the first successful attempt to use the HEOs electrode was published in 2020 [18], with multiple other reports to follow [18,19,20,21,22,23]. Importantly, the available data justifies the statement that HEOs offer certain advantages over the conventional analogs, displaying excellent long-term stability [20], modest thermal expansion behavior [13,24], increased resistance to CO2- and Cr-poisoning [13,14,24], as well as opening new ways for designing effective cathode materials, such as alkali-free compositions [25]. Of interest, the notion of increased long-term stability seems to go beyond SOFC applications, as several high-performance HEO electrocatalysts of excellent stability have also been reported in the case of low-temperature OER catalysis [26]. Consequently, it can be stated that the utilization of high-entropy approach can bring a solution to a number of different issues affecting the development of hydrogen technologies.
One of the biggest strengths and simultaneously challenges associated with the high-entropy approach is the huge number of potential elemental combinations, with each potentially offering a new set of properties, resulting from the synergy between composing elements. In this work, our focus was to identify such new opportunities for the design of novel electrocatalysts to lay a solid foundation for future development. One of the particularly interesting groups of increasingly popular perovskite materials for catalysis are materials combining 3d, 4d, and/or 5d elements, simultaneously, which often leads to particularly beneficial results. Such examples include SrFe1-xMoxO3-δ perovskites [27], renowned for their exceptional stability under various pO2 conditions, and SrCo1-x-yTaxNbyO3-δ materials, which as of today are characterized by one of the best performances ever reported for SOFC cathodes [9]. For such systems, the high-entropy approach with its potential abundance of redox active elements, providing highly flexible mechanisms of charge compensation, appears to be particularly well suited.
In the current study, we explore the limits of the 3d/4d/5d combinations of B-site cations in perovskite materials by proposing a new group of La(Cu0.2Ni0.2X10.2X20.2Y10.2)O3-δ (where X1, X2 = mixture of two Co, Ga, Fe, and Y1 = one of Nb/Ta) high-entropy perovskites La(Cu0.2Ni0.2Co0.2Ga0.2Nb0.2)O3-δ, La(Cu0.2Ni0.2Co0.2Ga0.2Ta0.2)O3-δ, La(Cu0.2Ni0.2Fe0.2 Ga0.2Nb0.2)O3-δ, La(Cu0.2Ni0.2Fe0.2Ga0.2Ta0.2)O3-δ, La(Cu0.2Ni0.2Co0.2Fe0.2Nb0.2)O3-δ, La(Cu0.2 Ni0.2Co0.2Fe0.2Ta0.2)O3-δ. Not only does our proposed system break the most common trend of utilizing a mixture of almost exclusively 3d elements by including Nb and Ta, but it is also worth highlighting the presence of the lanthanum as the A-site cation in ABO3-δ perovskite. As the use of typically used alkali ions is often associated with several issues, such as susceptibility to various degradation mechanisms [13], replacement by less reactive lanthanides could bring certain benefits with respect to broadly understood stability of the materials studied. Furthermore, the presence of La3+ cations instead of typical Sr2+ ones is expected to affect the valence states of the 3d/4d/5d combination of the B-site cations in a previously unexplored way. The initial OER/ORR assessment of these novel materials is carried out, providing the baseline for further development.
2. Results and Discussion
2.1. Structural and Morphology Characteristics of the as Obtained Materials
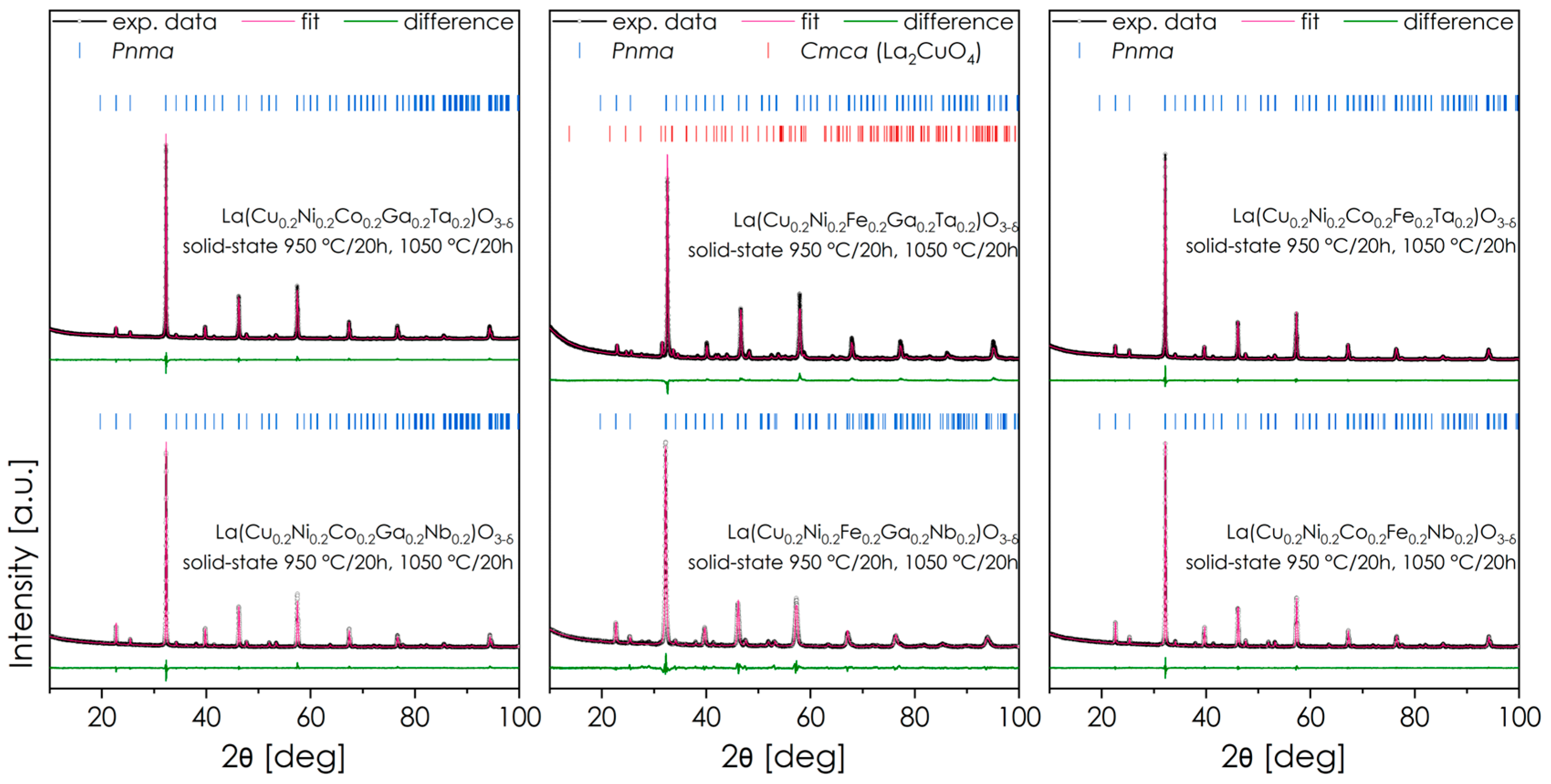
A series of materials was synthesized, in which the A position in the perovskite is occupied by La while the B position is occupied by an equimolar mixture of five elements: Cu and Ni, two elements selected from Co, Ga, and Fe, and one element selected from Ta or Nb. As a result, six base materials were obtained, in which three pairs can be distinguished: oxides with the same mixture of 3d elements and interchangeably Ta/Nb. The XRD diffractograms of the obtained powders after two cycles of sintering are presented in Figure 1, including Rietveld refinements. The residuals of Rietveld refinement procedures are summarized in Table 1. For each composition, an abbreviation used later in the text is provided, based on the elements used beside the common Cu/Ni core.
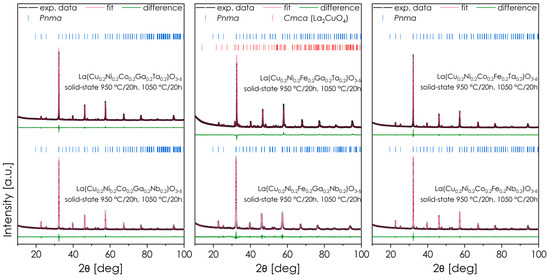
Figure 1.
XRD diffractograms of all obtained materials series La(Cu0.2Ni0.2X10.2X20.2Y10.2)O3-δ, sintered in air in two steps (at 950 °C for 20 h and 1050 °C for 20 h) and cooled down to RT with the rate of 2 °C min−1. Rietveld refinements are also included.

Table 1.
Structural data for the synthesized materials series La(Cu0.2Ni0.2X10.2X20.2Y10.2)O3-δ as obtained from the Rietveld refinements.
All of the considered powders crystallize predominantly in the Pnma space group (#62). Only for one material (FGT) were additional precipitates of the Ruddlesden-Popper (RP) Cmca (#64) phase detected. Interestingly, this issue was not observed for the analogous Nb-containing material, which may suggest a greater ability of isovalent Ta to stabilize the perovskite structure and prevent Cu and/or Ni from precipitating into the RP phase. The most likely reason for this behavior is that the lower electronegativity of Ta leads to more rigid B-O bonds, resulting in reduced structural flexibility, which in the case of multi-cation systems with a large variance of both valence states and ionic radii may be a limiting factor for successful incorporation of relatively large Cu2+ and Ni2+ ions. These differences are also reflected in the lattice parameters of FGT and FGN materials. Although the ionic radius of the elements Nb and Ta is the same (0.64 in 6-fold coordination), the normalized quasi-cubic parameter of the Ta-containing material is visibly smaller, which is in line with the more rigid lattice and part of the larger Cu2+ and Ni2+ ions not being incorporated into the structure.
The results of the SEM + EDS analysis conducted on the cross-sections of the pellets after two-step sintering are presented in Figure 2 and Figure 3. To further validate the consistency with the nominal compositions, the ICP-OES measurements were carried out for the selected materials, see Table S1, showing near-perfect agreement.

Figure 2.
Cross-section morphology with respective EDS mappings for selected obtained materials series La(Cu0.2Ni0.2X10.2X20.2Y10.2)O3-δ: (a) CGN, (b) CGT, (c) FGN.

Figure 3.
Cross-section morphology with respective EDS mappings for selected obtained materials series La(Cu0.2Ni0.2X10.2X20.2Y10.2)O3-δ: (a) FGT, (b) CFN, (c) CFT.
Only in one case, namely for the FGT composition (Figure 3a), does the SEM + EDS analysis lead to different conclusions from the XRD measurements—while this composition was expected to exhibit additional phase segregation (see also Figure S1), the EDS results indicate a single-phase structure. The reason for the observed discrepancy is probably the relatively low fraction of the secondary phase (about 8 wt.% according to XRD), which combined with the very fine-grain structure of the material may hinder the clear distinction between the precipitates and the main phase. In general, all materials examined by SEM exhibit characteristics of single-phase structure and exhibit a highly homogeneous distribution of elements. Nevertheless, based on the XRD results, the FGT composition was excluded from further characterization.
2.2. Functional Characterization of Obtained Materials
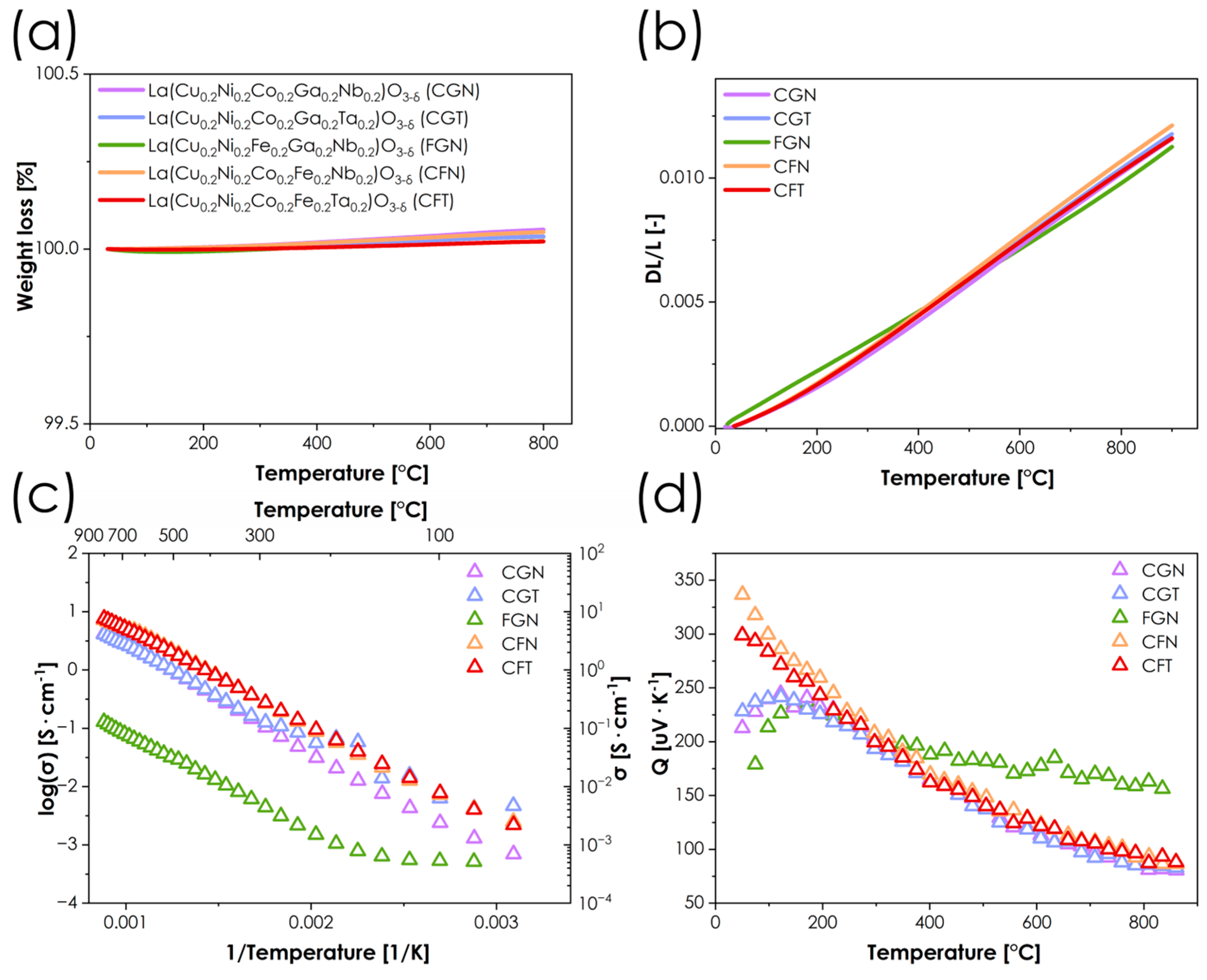
One of the most crucial aspects of using perovskite-structured materials as electrocatalysts, especially in SOFCs, is their oxygen non-stoichiometry. Unfortunately, in the case of the presented materials, determining the precise oxygen non-stoichiometry level is problematic for two main reasons. First, when using one of the most popular methods for determining oxygen non-stoichiometry in oxides, the iodometric titration, many assumptions have to be made, especially with regard to the lowest attainable oxidation states of cations undergoing reduction. While such an assumption is relatively straightforward for simpler systems and can be easily adjusted based on the results, in the case of an abundance of redox-active elements the situation becomes much more complex. The second problematic aspect is the presence of gallium in some of the materials studied. This element can partially/completely be reduced to Ga1+/Ga3+ under reducing conditions [28], making it impossible to reliably determine its final valence state during titration. Therefore, we focused on the temperature dependence of the oxygen content by determining mass loss by thermogravimetric analysis. The results of these measurements are shown in Figure 4a. Interestingly, all materials appear to have a nearly constant oxygen content from room temperature to 800 °C, rather than the decrease in oxygen content typically observed in perovskites at high temperature, due to lattice oxygen release. Such behavior, while atypical, especially for Co-containing compositions, is in line with previous reports concerning HEOs, indicating their much lower tendency toward oxygen loss. In principle, this can be beneficial for oxide materials, as the oxygen release is the main source of the deleterious chemical expansion behavior, which is a serious issue for most of the state-of-the-art cathode materials in SOFCs [29], leading to poor thermomechanical compatibility with other elements of the cell. On the other hand, the presence of oxygen vacancies is indispensable for achieving high values of ionic conductivity, which are crucial for effective ORR catalysis in SOFCs. For materials studied, assuming that all of the copper and nickel is likely in the +2 valence state, and Nb/Ta are +5 (see also XPS results in Figure S2), and that the remaining two elements, whether it is a Co/Fe, Co/Ga, or Fe/Ga combination, have an average oxidation state of +3.0 (which is likely), little to no oxygen non-stoichiometry would be expected. Therefore, in terms of future development, reducing the amount of Ta/Nb, e.g., to 10%, appears to be a logical next step.
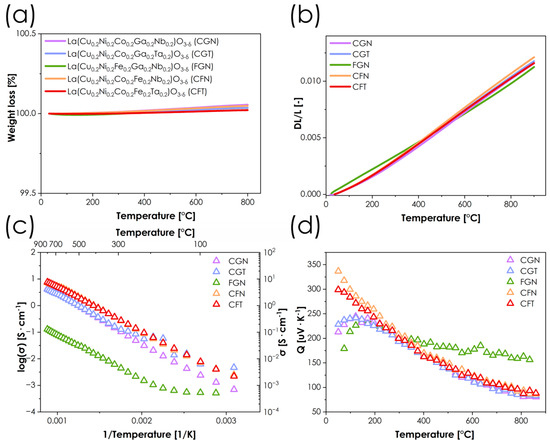
Figure 4.
(a) Dependence of oxygen non-stoichiometry as a function of temperature of obtained materials series; (b) thermal expansion behavior of materials series determined by dilatometry; (c) logarithmic conductivity values plot of materials series; (d) Seebeck coefficient dependence on temperature for the considered samples.
The thermal expansion properties of all the materials were investigated using the dilatometric method, with results presented in Figure 4b, while the obtained TEC values can be found in Table 2. The values obtained are relatively lower than those for typical perovskites [30], especially the ones with higher Co content. The correlation between cobalt content and thermomechanical properties is widely known for various perovskite systems, since cobalt Co3+ is known to undergo a transition from a low/medium-spin to high-spin configuration as a result of increasing temperature, leading to an increase in ionic radii and, consequently, an additional contribution to the total TEC values [31,32,33]. The reported herewith high-temperature TEC values (400–900 °C range) ranging from 14.4 to 15.2 × 10−6 K−1 for Co-containing materials can be considered very competitive compared to state-of-the-art SOFC cathode materials [2,30]. Such low values improve thermomechanical compatibility with typical electrolyte materials such as YSZ, GDC, and LSGM, for which the TEC values are within the range of 10–12.5 × 10−6 K−1 [34]. As expected, in the case of Co-free FGN material, the value is even lower. Comparing the low- and high-temperature TEC values, it can be noticed that the differences are low-to-moderate, and on average much lower than in the case of most of the state-of-the-art materials [30]. This is consistent with the TG results, Figure 4a, which clearly show that no high-temperature oxygen release takes place in any of the compositions studied and, therefore, the chemical expansion contribution can be expected to be negligible, with the Co spin transition likely being the main reason for the observed behavior.

Table 2.
Thermal expansion coefficients for the series based on the dilatometric studies.
The total electrical conductivity was determined using the 4-probe DC method combined with the simultaneous measurement of the Seebeck coefficient Q, with the results summarized in Figure 4c,d, as well as Table 3. In all cases, the highest conductivity values are observed at the highest temperature considered, which again is consistent with the lack of oxygen release (which can be seen in both TG and dilatometric measurements). One material, namely the Co-free FGN composition, clearly differs in behavior from the other oxides considered, with a maximum conductivity value as low as 0.1 S·cm−1 at 860 °C. However, it should be noted that such behavior is not surprising, as the total fraction of redox inactive elements (Nb and Ga) reaches in this case 40% of the available B-sites in the lattice. The rest of the materials are characterized by maximum total conductivity values of around 4 S·cm−1 and 7 S·cm−1 for Fe-free and Ga-free materials, respectively, which, although not particularly high, are not necessarily problematic from the point of view of catalytic applications [35]. Interestingly, in the case of CGN and CGT materials, in which the combined content of Ga and Nb/Ta also amounts to 40% of B-sites, the conductivity is still more than an order of magnitude higher than in FGN, which can be attributed to the fact that Co3+ is much easier to reduce than the Fe3+ ions with their stable d5 high-spin configuration. It is worth noting that switching between Nb and Ta in materials of otherwise identical elemental composition does not significantly affect the electrical behavior. From the perspective of conductivity mechanisms, the combination of Q values exceeding 100 μV·K−1, and energies of activation at the level of 0.3 eV (calculated based on the σT = σ0 exp(−Ea/kT) equation, Table 3), strongly suggests that the small polaron hopping mechanism is the dominant one [36,37,38]. The behavior of the Seebeck coefficient values (Figure 4d) shows that with the increase in temperature, the Q values decrease. This indicates that, despite nearly constant oxygen stoichiometry, the population of charge carriers involved in transport increases. This effect can be attributed to the temperature-dependent redistribution of charge carriers relative to the transport-active states, associated with the thermal population of localized and weakly delocalized 3d states. In materials with multicomponent B-sites, the transport-active density of states is expected to be energetically broadened because of the presence of multiple redox-active centers. As thermal broadening of the Fermi–Dirac distribution increases with temperature, an increasing fraction of these states contributes to charge transport, leading to a decrease in the Seebeck coefficient, even in the absence of oxygen vacancy formation and associated charge compensation mechanisms. This effect is the least noticeable in the FGN material, where the number of effective charge carriers is likely to be the lowest.

Table 3.
Activation energies (Ea), temperature ranges used for their determination (T), maximum conductivity value determined for series in temperature range 50–850 °C.
2.3. Low-Temperature OER Performance
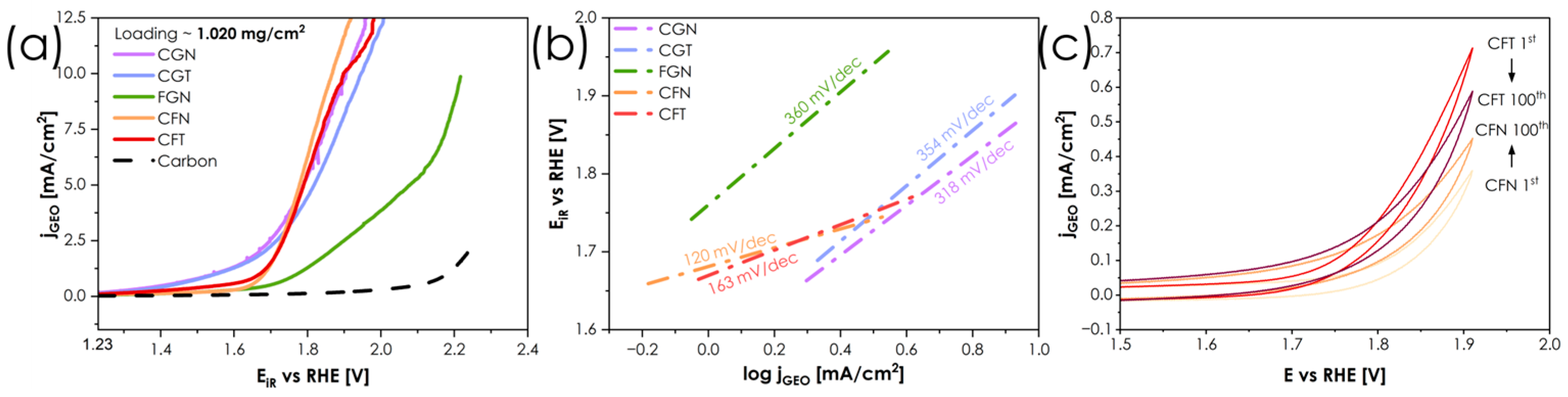
The OER cyclic voltamperometry curves obtained in 0.1 M KOH are presented in Figure 5.
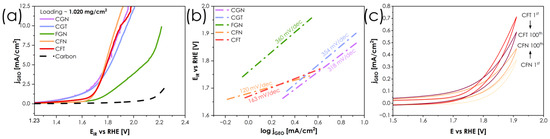
Figure 5.
(a) OER CV curves for the series obtained in 0.1 M KOH solution. (b) Tafel analysis based on polarization curves. (c) Stress test composed of 100 cyclic voltammetry scans up to 1.9 V vs. RHE.
Based on the OER activity measurements carried out for all single-phase compositions, the highest OER activity is achieved for the CFN composition, followed by CGN and CFT. For these three compounds, the geometric current densitiy jGEO of 10 mA·cm−2 is reached for the potential values equal to 1.868, 1.944 and 1.964 V, respectively, which at this stage of development of these materials, cannot be considered competitive with some of the best state-of-the-art materials which achieve similar current densities in the 1.5–1.6 V potential range [39,40]. It is not surprising that materials with a combination of Co and Fe exhibit the highest levels of performance, since the coupling of Fe3+/Fe4+ and Co2+/Co3+ redox pairs is well known from other, more conventional materials [41]. Tafel analysis shown in Figure 5b confirms that the presence of Fe3+/Fe4+and Co2+/Co3+ and synergistic effects of redox pairs in the structure facilitate the charge transfer capabilities of the catalysts. As a result, Tafel slopes of 120 mV/dec and 163 mV/dec were determined for CFN and CFT, respectively, which is comparable to several other perovskite electrocatalysts [42,43] and indicates that adsorption of *OH is the rate-determining step. On the other hand, CGN, which achieved similar overpotentials to CFN and CFT, is characterized by significantly slower OER kinetics, as evidenced by a Tafel slope of 318 mV/dec, additionally hindered by lower total conductivity. The apparently high activity of CGN inferred from polarization curves stems from either non-faradaic or parasitic currents related to changes in the structure of the catalysts themselves, e.g., creation of Me-OH or Me-OOH intermediate states on the surface, typical for oxide materials exhibiting conventional adsorbate evolution mechanism (AEM) [44,45]. As part of the benchmarking, accelerated stress test (AST) of the best performing catalysts—CFN and CFT—were carried out, Figure 5c. A distinct difference between 100th and 1st scans can be seen. CFN exhibits an apparent increase in activity from 0.35 mA/cm2 to 0.45 mA/cm2, while total current density drops from 0.7 mA/cm2 to 0.6 mA/cm2 for CFT. This effect, however, cannot be readily interpreted solely on the basis of catalyst composition, as the activity is also a function of ink/film quality. Nevertheless, when compared with other perovskite electrocatalysts [46], these values indicate high stability of the materials studied.
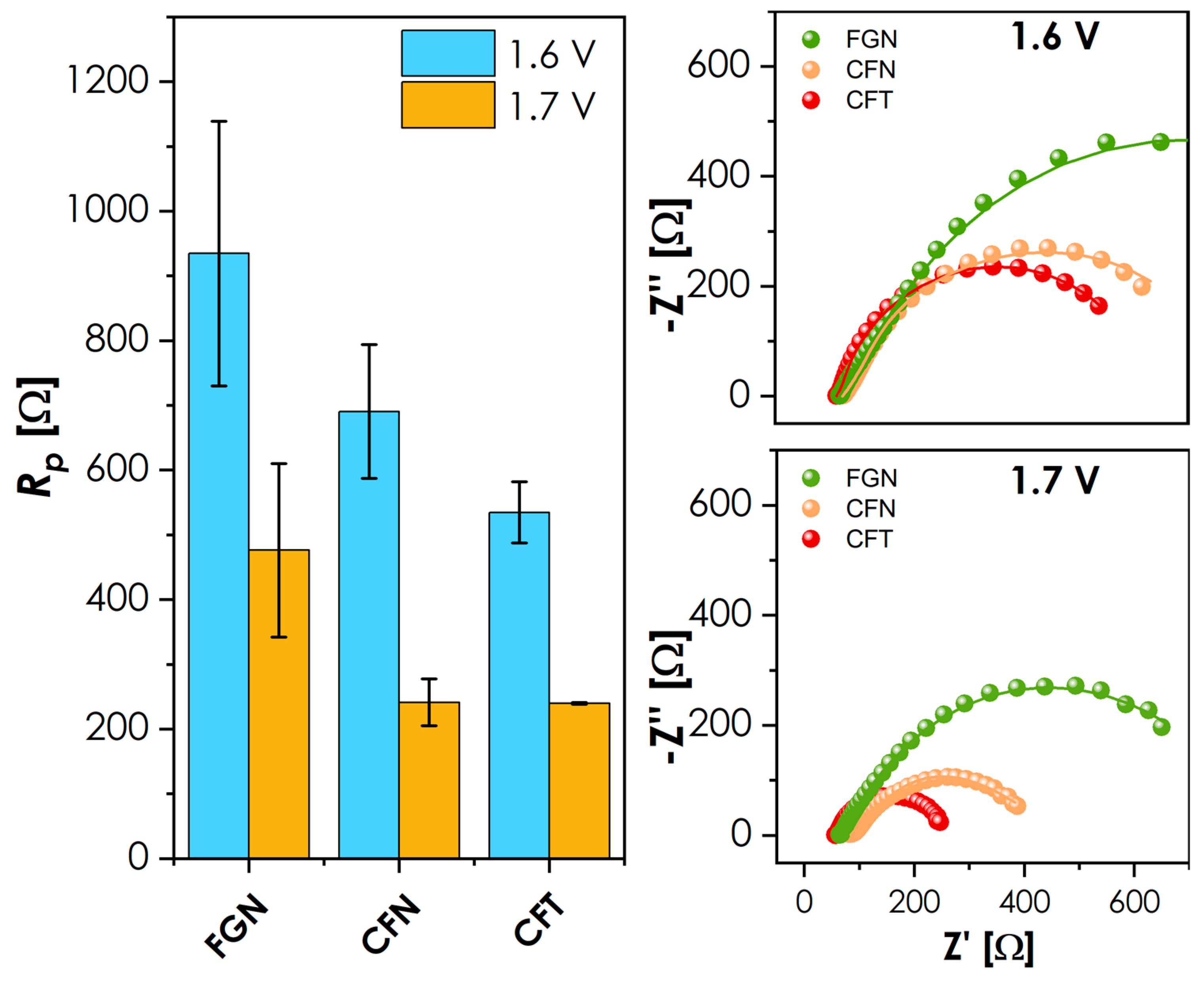
Furthermore, both CFT and CFN display relatively high total electrical conductivity, which may additionally facilitate charge transport during OER, although bulk conductivity alone is not sufficient to determine catalytic activity. At the other end of the spectrum is the FGN material, which clearly shows that the presence of Fe alone, combined with low conductivity, limits its performance. It should be clearly stated that, in the current bulk form, without any nanostructuring and at relatively high loadings, the materials studied are not directly competitive with some of the state-of-the-art materials. For comparison, conventional Sr-containing perovskite cathodes such as La0.6Sr0.4CoxFe1-xO3-δ are reported to reach comparable current densities at significantly lower loadings [41], highlighting that the present bulk materials exhibit only moderate intrinsic OER activity. Still, the results clearly indicate the presence of pronounced composition–activity relationships, which can be leveraged in future composition optimization and surface engineering strategies. Therefore, to provide further insight into the mechanisms of operation and to assess the relative performance of respective elemental combinations, three materials showing the most distinct behavior in the OER studies were selected for further EIS analysis: the best performing CFN and CFT, and the poorest performing FGN, see Figure 6.
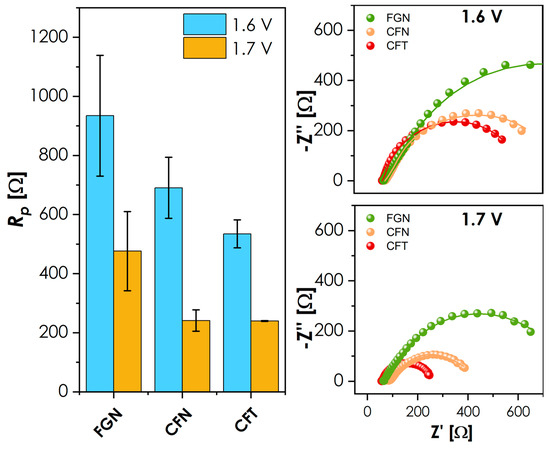
Figure 6.
Impedance spectra collected for measurement at low temperature obtained for selected materials. The comparison of the polarization resistance values is provided on the left-hand side.
The shape of the recorded EIS spectra with a single, slightly flattened semicircle and no visible Warburg-like behavior (Figure 6, right) suggests that the measured response is dominated by OER-related charge-transfer kinetics. At 1.6 V, CFT exhibits the lowest polarization resistance Rp, closely followed by CFN, with both significantly outperforming the FGN. The performance is consistent with the onset positions from Figure 5, highlighting the enhanced kinetics of the former compositions towards OER. At 1.7 V, the order of performance changes only slightly, with now almost identical Rp values for both CFT and CFN. When combined with the LSV results, CFN is characterized by more rapid kinetics than CFT, indicated by a steeper slope of the current response at potentials greater than 1.7 V. Overall, while the investigated materials require further optimization to reach competitive OER performance, the observed trends clearly demonstrate the critical role of the B-site composition in governing of OER kinetics in multicomponent La-based perovskites. On the basis of the results, the previously mentioned idea for further development of materials aimed at lowering the Nb/Ta content down to 10%, combined with the slightly increased content of Cu, Ni, and/or Co, should provide the system with much greater flexibility in terms of redox behavior, which is expected to translate into better OER performance.
2.4. High-Temperature ORR Performance as SOFC Cathode Materials
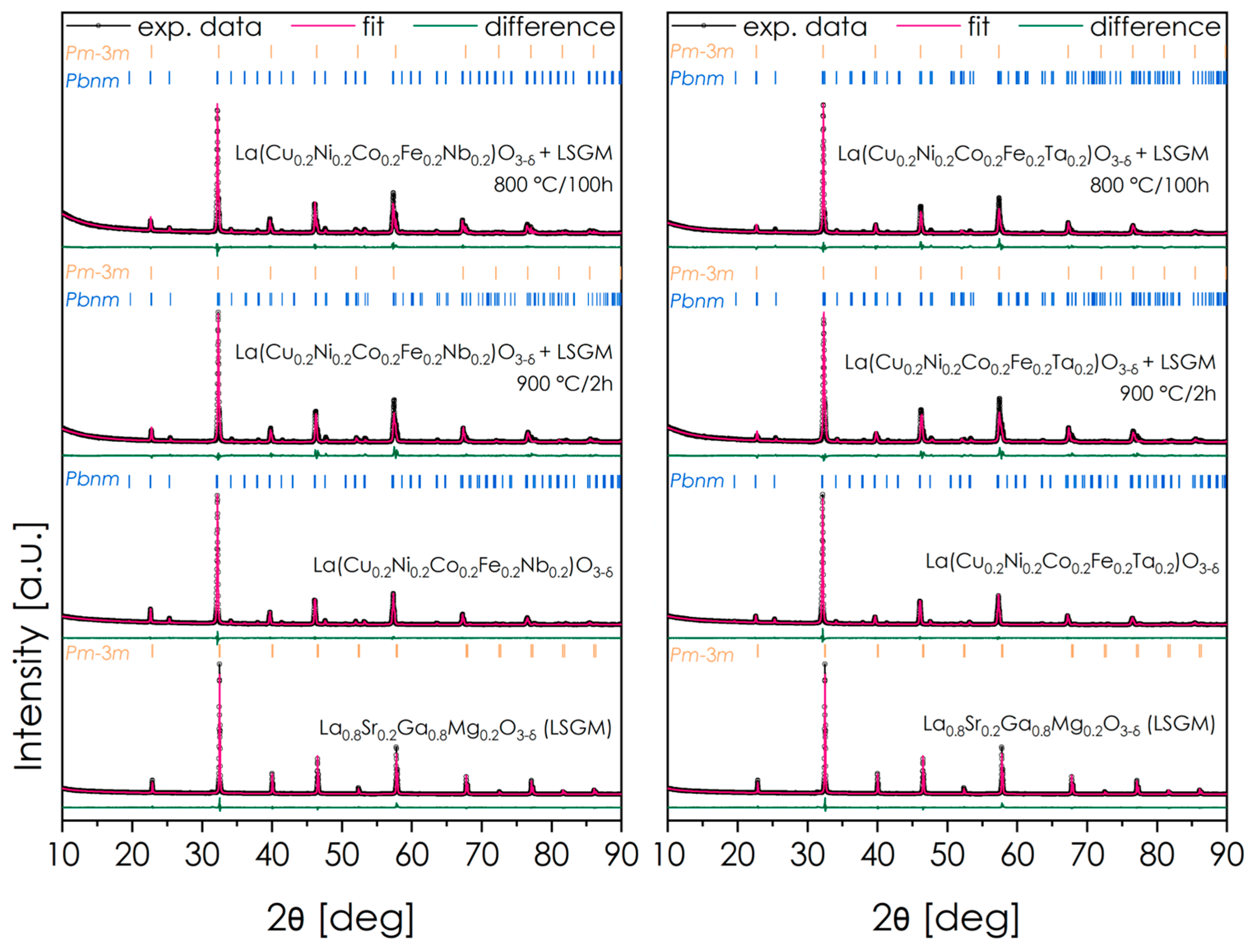
Two materials characterized by the highest values of total conductivity and relatively high OER activity—La(Cu0.2Ni0.2Co0.2Fe0.2Nb0.2)O3-δ and La(Cu0.2Ni0.2Co0.2Fe0.2Ta0.2)O3-δ—were investigated in the context of their potential usage in SOFC technology. In the first stage, the chemical stability toward the LSGM solid electrolyte used in low-temperature and intermediate-temperature SOFCs was evaluated, both under conditions corresponding to the electrode fabrication and long-term operation in SOFC. The diffractograms showing the results of the XRD analysis obtained for perovskite–electrolyte sintered mixture, and the corresponding results of the subsequent Rietveld analysis are collected in Figure 7 and Table 4. The patterns for pure LSGM and the respective electrode materials are included as references. Overall, both materials appear to be fully stable against the LSGM electrolyte.
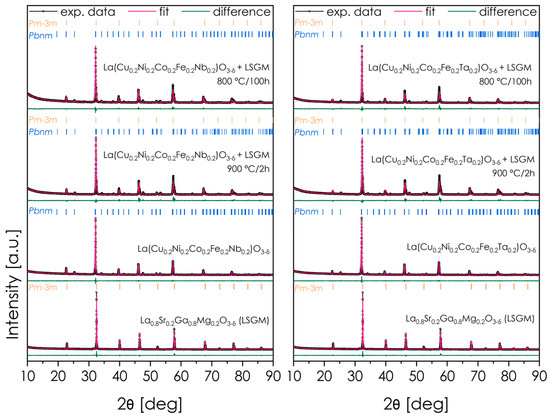
Figure 7.
XRD data concerning chemical stability measurements for the considered materials and electrolyte LSGM.

Table 4.
Results of the Rietveld refinements after chemical stability measurements for the considered materials and electrolyte LSGM.
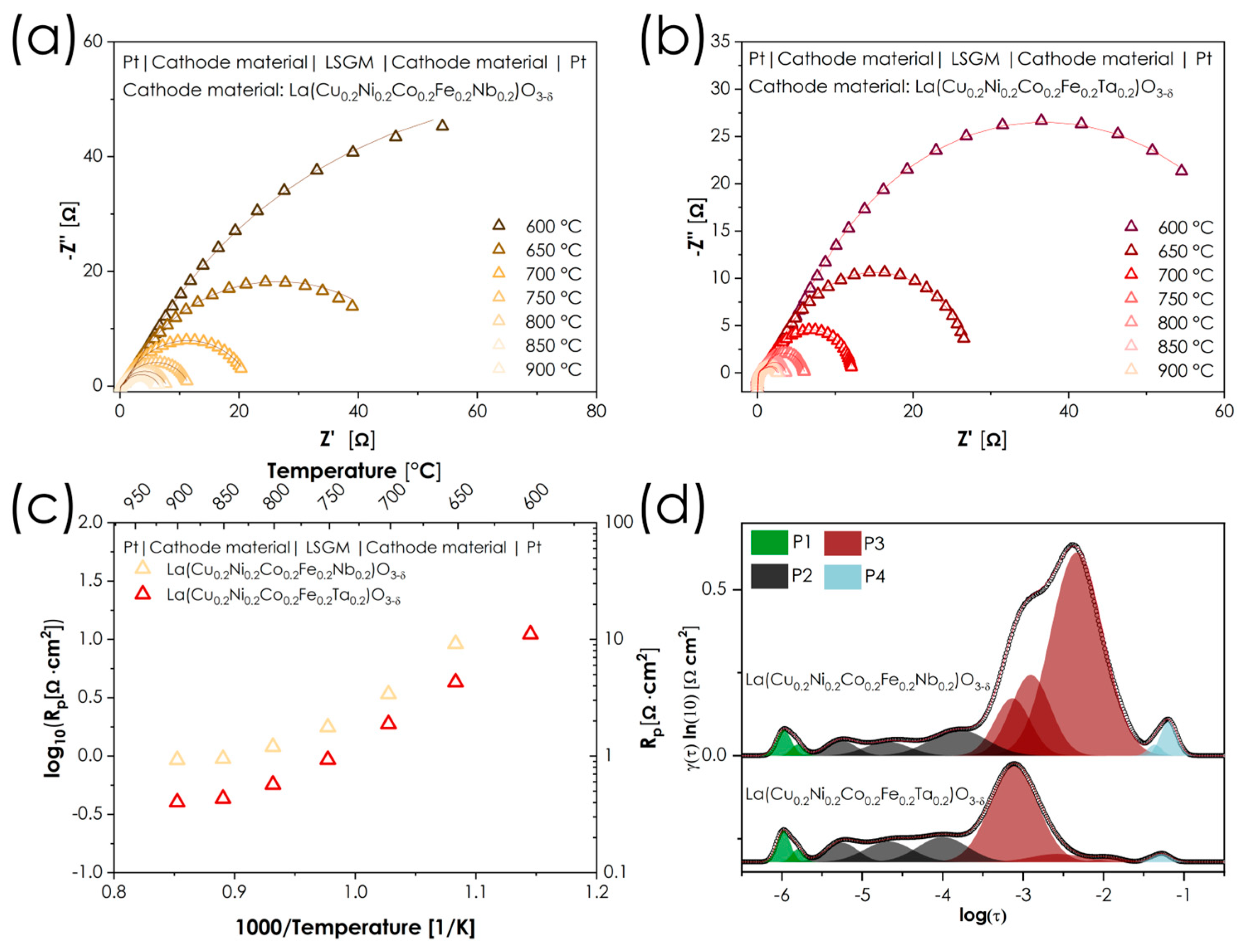
The selected CFN and CFT compositions were then used to prepare the symmetrical half-cells with LSGM electrolyte layer. The results of their assessment are presented in Figure 8. The values of determined cathodic polarization resistances in the temperature range 600–900 °C are summarized in Table 5.
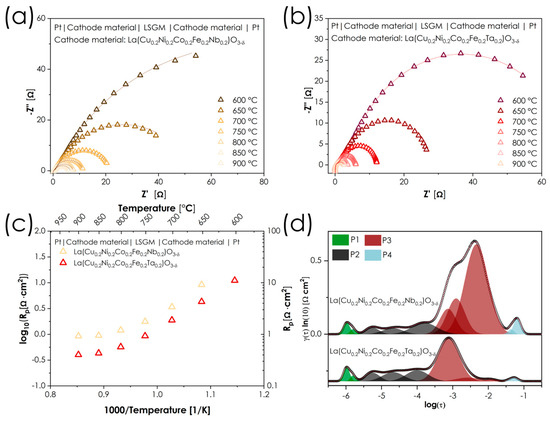
Figure 8.
(a,b) Impedance spectra collected for LSGM-based symmetrical cells; (c) summary of cathodic polarization resistances; (d) DRT results of the symmetrical cell with for both electrodes at 850 °C.

Table 5.
Total electrode polarization resistance Rp for the considered symmetric cells.
Compared to the recently published La(Co0.2Fe0.2Ga0.2Cu0.2Ni0.2)O3-δ composition, which has several similarities to materials studied, the performance of CFN and CFT is considerably lacking—while the former composition had a polarization resistance (Rp) at 800 °C equal to 0.089 Ω·cm2, the CFN and CFT exhibit values of 1.200 and 0.570 Ω·cm2, respectively [25]. It appears that replacing Ga with Ta or Nb significantly affects the catalytic activity of the material; considering that both CFN and CFT are characterized by a low (even negligible) concentration of oxygen vacancies, which additionally is temperature independent, it is likely that the introduction of a +5 cation suppresses charge transfer between the other transition-metal ions. In other words, introducing +5 cations into the lattice forces the remaining elements to adopt their lowest attainable oxidation states, leading to suppression of active redox pairs, such as Fe3+/Fe4+ or Co2+/Co3+/Co4+. Nevertheless, in this context, it is particularly interesting that, despite their overall similarities and identical valence states, replacing Nb with Ta resulted in a reduction in polarization resistance of more than 50%, regardless of temperature, which is an important clue for further development. To better understand the differences in the electrochemical mechanisms of these two cathode materials, a DRT (Distribution of Relaxation Times) analysis was performed based on the EIS measurements in air at 850 °C. The application of DRT enables the separation of individual processes that occur on the working electrode and reveals the contribution of each process to the total polarization resistance [47,48,49]. Based on the literature, four main groups of peaks can be proposed, although it should be noted that their fully unambiguous assignment would require pO2-dependent measurements: P1—the fastest processes associated with charge transfer across the electrolyte–electrode interface (theoretical slope of pO2 dependence m = 0), P2—combined oxygen-reduction processes (theoretical m = 0.25), P3—dissociation of the O2 molecule (theoretical m = 0.5), and P4—the slow oxygen adsorption process (theoretical m = 1). For the materials discussed, the P3 process is identified as the main source of differences between the two materials, with the prominent peak at log(τ) ≈ −2.5 visible for CFN and practically disappearing for CFT. The possible reason for this behavior can be attributed to the generally more covalent character of the Ta-O bonds, compared to the Nb-O ones, making it easier to transfer electron density from the oxide surface to the O2 molecule during the dissociation process. This step, while formally non-electrochemical, requires partial charge redistribution between the adsorbed O2 molecule and the oxide surface. The presence of Ta5+ facilitates such transient charge transfer by enhancing metal–oxygen covalency and charge delocalization, thereby lowering the activation barrier for O2 dissociation.
3. Materials and Methods
3.1. Synthesis Procedure
The presented series of high-entropy perovskite materials: La(Cu0.2Ni0.2Co0.2Ga0.2Nb0.2)O3-δ (abbreviated in this work as CGN), La(Cu0.2Ni0.2Co0.2Ga0.2Ta0.2)O3-δ (CGT), La(Cu0.2Ni0.2Fe0.2Ga0.2Nb0.2)O3-δ (FGN), La(Cu0.2Ni0.2Fe0.2Ga0.2Ta0.2)O3-δ (FGT), La(Cu0.2Ni0.2Co0.2Fe0.2Nb0.2)O3-δ (CFN), La(Cu0.2Ni0.2Co0.2Fe0.2Ta0.2)O3-δ (CFT) was synthesized using a standard solid-state method, dictated mainly by limited availability of Nb and Ta soluble salts, which could be used in combination with some of the more popular salts of 3d elements. As precursors to introduce considered cations, the following oxides were used: La2O3 Thermo Scientific, 99.99%), Co2O3 (Alfa Aesar, 99.7%), CuO (Alfa Aesar, 99.7%), Fe2O3 (Alfa Aesar, 99.9%), Ga2O3 (Alfa Aesar, 99.999%), Nb2O5 (Alfa Aesar, 99.9%), NiO (Alfa Aesar, 99.998%), Ta2O5 (Thermo Scientific, 99.99%). All precursor materials were weighed according to their stoichiometric ratios and milled in isopropanol using a ball mill (Fritsch Pulverisette 7 planetary mill with ZrO2 milling balls) at 600 RPM for 90 min. The resulting mixture was dried, pressed into 20 mm pellets, and sintered in air in a chamber furnace in two-step procedure: at 950 °C for 20 h in air, followed by cooling down to room temperature (RT) at a rate of 2 °C·min−1, and then at 1050 °C with the rest of the parameters analogical. Between the first and second sintering, the obtained powder was thoroughly ground in a mortar and pelletized again.
The resulting powders were used as base materials for XRD, SEM, TG, and HT-XRD measurements. To obtain dense pellets for dilatometric and electrical conductivity measurements, the respective powders were mixed with 2 wt.% of poly(vinyl butyral-co-vinyl alcohol-co-vinyl acetate) (PVB), then pelletized and re-sintered again at 1050 °C for 20 h in air in a chamber furnace, with cooling down to RT at a rate of 2 °C·min−1. For electrochemical studies, the base powders were additionally milled in isopropanol for 12 h at 600 RPM to achieve a finer particle morphology suitable for cathode paste preparation.
3.2. Characterization Methods
The structural properties of the obtained powders were investigated by using X-ray diffraction (XRD) measurements at room temperature (RT), carried out with the use of Panalytical Empyrean diffractometer with Cu Kα radiation, working in Bragg–Brentano geometry, within the 10–100 deg range. The obtained data were initially analyzed using X’Pert HighScore Plus 2.0 software and PDF-4+ ICDD database for phase analysis. GSAS-II v5.6.0 software was used for all Rietveld refinements [50]. Normalized cell volume V0 was calculated assuming that the equivalent cubic unit cell is 4 times smaller for orthorhombic structure. Then, the normalized quasi-cubic unit cell parameter a0 was calculated as the cube root of V0.
The scanning electron microscopy (SEM) method was used to provide morphological images of the pellet fracture after the second sintering cycle. Homogeneity and chemical composition were investigated by the energy dispersive X-ray spectroscopy method (EDS). ThermoFisher Scientific Phenom XL Desktop SEM with the ProSuite software package was used for all measurements, with accelerating voltage of 10 kV for morphology studies and 15 kV for the EDS analysis.
Chemical analysis of the material was performed in the Hydrogeochemical Laboratory of the Department of Hydrogeology and Engineering Geology, Faculty of Geology, Geophysics and Environmental Protection AGH University of Krakow. Approximately 0.05 g of each sample was digested in a microwave mineraliser UltraWAVE (Milestone Srl, Sorisole, Italy). HCl and HNO3 concentrated acids (3:1, v/v) were used in the digestion procedure. The process was carried out at a temperature of 230 °C and a maximum pressure of 75 bar. The digestion program used a power setting of 1500 W, a 15 min ramp to 230 °C, a 20 min hold at the target temperature, followed by cooling. Prior to analysis the digests were diluted with deionised water obtained from the HYDROLAB R20 system (Hydrolab Sp. z o.o.) to the final volume of 50 mL. The total concentration of elements was determined using the ICP-OES technique according to the 11 885 ISO standard. The iCAP PRO XP spectrometer from Thermo Fischer Scientific (Waltham, MA, USA) was used. Analytical lines 333.749 nm, 224.700 nm, 221.647 nm, 228.616 nm, and 238.204 nm were selected for the determination of La, Cu, Ni, Co and Fe. The limits of quantification for all elements ranged from 0.5 µg/L to 5.0 µg/L. Quality control was implemented during the whole analytical procedure. The temperature dependence of oxygen non-stoichiometry was studied using the thermogravimetric analysis performed with the TGA Q5000 apparatus. Each material was tested in two heating/cooling cycles, each time reaching a temperature of 800 °C with a rate of 2 °C·min−1. The measurements were conducted under an air atmosphere with a gas flow rate of 100.0 mL·min−1.
The thermal expansion coefficient in air atmosphere was determined using a Linseis L75 Platinum Series dilatometer over a temperature range from room temperature (RT) to 900 °C, with a heating/cooling rate of 5 °C·min−1.
The total electrical conductivity of the materials studied was evaluated using a 4-probe DC technique. Measurements were conducted during the cooling cycle from 850 °C to 25 °C, in increments of 25 °C, employing the Fine Instruments FRASB-1000 setup, composed of the TF1200 tube furnace, Keysight Multimeter 34465A, Keysight Function/Arbitrary Waveform Generator 33210A, control unit CFRASB-1000 with AM16/32B multiplexer, measuring probe SSC-15 and BR07 retain decade. The same system was used to determine the Seebeck coefficient values [51].
3.3. OER Performance
In order to evaluate the OER catalytic activity, the inks of active materials were prepared by mixing of 4 mg of the considered powder, 2 mg of Ketjenblack carbon and 50 µL of a 5% Nafion 115 solution. The total volume was adjusted to 1 mL using a H2O and isopropanol mixture (3:1). The resulting suspension was sonicated for 30 min in an ultrasonic bath and then deposited in amount of 4.5 µL of onto the polished GCE surface using drop-cast method per layer up to reaching total ink volume (22.5 µL). The oxygen evolution reaction activity was evaluated using a default three-electrode setup utilizing Pt wire as a counter electrode, Ag/AgCl as a reference electrode (3 M) and 0.1 M KOH electrolyte. All potentials in this section were converted to RHE (reversible hydrogen electrode) using formula (1):
where and . The electrolyte was constantly stirred during electrolysis, and the experiments were carried out using a Gamry Interface 1010E Potentiostat. The experimental sequence involved (1) twenty fast (200 mV·s−1) cyclic scans from 0.7 V to 1.0 V to obtain a reproducible surface state, (2) a slower (5 mV·s−1) subsequent linear anodic scan (LSV) from 0.7 V to 2.3 V, and (3) potentiostatic electrochemical impedance spectroscopy (EIS) measurements performed at 1.6 V and 1.7 V, using an AC perturbation amplitude of 10 mV (rms) and a frequency range from 105 Hz to 0.1 Hz. The fitting was done using ZView software, utilizing a Randles circuit with a CPE (constant phase element) component to account for non-ideal capacitive behavior of the electrochemical setup. All of the measurements were performed for three separate samples each. Tafel slopes were derived from linear sweep polarization curves using a linear fit. Potential window was set from 1.65 V to 1.95 V which corresponds to a current density span of 0.5 to 1 decade (measured in mA/cm2).
Accelerated stress test (AST) was performed for the best-performing materials—CFN and CFT. The test comprised two steps: (1) electrochemical cleaning—twenty fast scans (200 mV/s) from 0.7 V to 1.0 V, and (2) one hundred slower scans (5 mV/s) with the potential window ranging from 1 V to 1.9 V. Potentials higher than 1.9 V were not used due to the corrosion of the glassy carbon substrate, and 0.1 M KOH was used as an electrolyte. SEM imaging with EDS analysis was performed before and after this test to assess the morphology of the obtained electrodes.
3.4. SOFC Performance
The chemical stability with respect to commercial solid electrolytes was tested using La0.8Sr0.2Ga0.8Mg0.2O3-δ (LSGM) provided by fuelcellmaterials. Before mixing, the LSGM powder was pre-sintered at 1450 °C for 8 h. Subsequently, the electrolyte and selected perovskite oxide powders were mixed in a weight ratio of 1:1. The powders obtained were pressed into pellets and sintered under two conditions: (1) 900 °C for 2 h (with heating and cooling 2 °C·min−1), simulating cathode layer fabrication, and (2) 800 °C for 2 h (with heating and cooling 2 °C·min−1), corresponding to long-term symmetric cell testing. XRD measurements were used to analyze the resulting materials. LSGM powder was mixed with 1 wt.% of PVB and sintered as a pellet at 1450 °C for 8 h in air. The sinters were ground to a thickness of approx. 600 μm.
To evaluate the electrode polarization resistance, symmetrical cells (electrode|electrolyte|electrode) were fabricated using selected perovskite powders. The cathode pastes were prepared by mixing the powder with a commercial texanol-based binder (3D-nano) in a 1:0.4 weight ratio and adding starch (1:0.05 wt.%) as a pore-forming agent. The paste was screen-printed onto both sides of the electrolyte, forming ~30 μm thick layers with an area of ~0.28 cm2. Sintering was performed at 900 °C for 2 h in air, with 2 °C·min−1 heating and cooling rates. Platinum paste, sintered at 750 °C for 30 min, was used as the current collector.
Electrochemical impedance spectroscopy (EIS) measurements were performed using a NORECS Probostat™ setup combined with a Gamry Interface 5000E potentiostat/galvanostat. Data were analyzed using an equivalent circuit model (L–R0–Q1R1–Q2R2), where L represents the inductance of the wire, R0 the ohmic resistance (mainly from the electrolyte), and Q-R components reflect high-frequency (Q1R1) and low-frequency (Q2R2) electrode processes. The cathode polarization resistance (Rp) was calculated as (R1 + R2) × (cathode area)/2 [52].
To verify the material in long-term operation mode, we prepared symmetric cells with the same methodology as described above. EIS measurements were determined in 750 °C for 100 h using the same equipment.
To provide information on the mechanisms of the electrode processes, DRT analysis was performed for the selected materials. EIS measurements taken at 850 °C in air were considered. The subsequent DRT analysis was performed using the DRTtools Matlab toolbox [53]. The fitting was performed with the model including the contribution of inductance, and the regularization parameter λ was set at 10−4.
4. Conclusions
In this work, a new group of high-entropy perovskites with the general formula La(Cu,Ni,X1,X2,Y1)O3-δ (where X1, X2 are two of Co, Ga, or Fe, and Y1 is either Nb or Ta) was synthesized and investigated, demonstrating the possibility of obtaining alkali-free perovskites with mixed 3d/4d/5d B-site occupancy and establishing a framework for further development of these highly unconventional systems. The results show that the selection of the X1, X2, and Y1 ions has a profound impact on the usability and performance of the materials. In all cases, the materials with the Co/Fe combination appear to be the most promising, while the relative superiority between Nb and Ta changes depending on the intended application. The Nb-containing La(Cu0.2Ni0.2Co0.2Fe0.2Nb0.2)O3-δ appears to possess slightly more rapid OER kinetics compared with its Ta-containing La(Cu0.2Ni0.2Co0.2Fe0.2Ta0.2)O3-δ analog, while the opposite is true for the high-temperature ORR process in SOFC cathode applications. It appears that despite the close similarity between Ta and Nb, the differences in their electronegativity, and consequently the covalency of formed Nb/Ta-O bonds, may in some cases be a source of major effects. It must be noted that the proposed materials are proof-of-concept, showing the possibility of even synthesizing such compounds. In terms of their raw performance, in the current, equimolar form it is rather mediocre, with the presence of 20% (B-site) of pentavalent, redox-inactive elements drastically limiting the redox activity of 3d elements and suppressing the formation of oxygen vacancies, crucial from the perspective of high-temperature ORR. Surprisingly, CFN shows an increase in current density during 100 cycle AST test, suggesting improved electrochemical activity during prolonged operation. As a result, further studies will focus on optimizing the proposed systems by decreasing the relative amount of Ta and Nb, and, consequently, providing better flexibility for charge compensation and redox activity of other cations.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/catal16020192/s1, Table S1: ICP-OES analysis with molar ratios calculated relative to La performed for La(Cu0.2Ni0.2Co0.2Fe0.2Nb0.2)O3-δ and La(Cu0.2Ni0.2Co0.2Fe0.2Ta0.2)O3-δ; Figure S1: XPS results with fitting for material La(Cu0.2Ni0.2Fe0.2Ga0.2Nb0.2)O3-δ; Figure S2: Sintering optimization for La(Cu0.2Ni0.2Fe0.2Ga0.2Ta0.2)O3-δ composition.
Author Contributions
Conceptualization, J.D. and K.Ś.; methodology, K.Z., J.D., M.Z., M.K. and K.Ś.; software, M.Z.; validation, K.Z.; formal analysis, K.Z., J.D., M.Z., M.K. and K.Ś.; investigation, K.Z., M.Z., and M.K.; resources, J.D. and K.Ś.; data curation, K.Z., M.Z., and M.K.; writing—original draft preparation, K.Z. and J.D.; writing—review and editing, K.Z., J.D., M.Z., M.K. and K.Ś.; visualization, K.Z., and M.K.; supervision, J.D. and K.Ś.; project administration, K.Ś.; funding acquisition, K.Z., K.Ś. All authors have read and agreed to the published version of the manuscript.
Funding
Scientific work was funded by the state budget within the program of the Minister of Science and Higher Education in Poland under project Perły Nauki (PN/01/0127/2022), funding amount 188 100 PLN with total value of the project 188 100 PLN. This work was partially supported by the program “Excellence Initiative—Research University” for the AGH University of Krakow (IDUB AGH, No. 501.696.7996, Action 4, ID 9880).
Data Availability Statement
Data will be made available on request.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Global Monitoring Laboratory. Available online: https://gml.noaa.gov/ (accessed on 4 November 2025).
- Gao, Z.; Mogni, L.V.; Miller, E.C.; Railsback, J.G.; Barnett, S.A. A Perspective on Low-Temperature Solid Oxide Fuel Cells. Energy Environ. Sci. 2016, 9, 1602–1644. [Google Scholar] [CrossRef]
- Stambouli, A.B.; Traversa, E.; Stambouli, A. Solid Oxide Fuel Cells (SOFCs): A Review of an Environmentally Clean and Efficient Source of Energy. Renew. Sustain. Energy Rev. 2002, 6, 433–455. [Google Scholar] [CrossRef]
- Hauch, A.; Küngas, R.; Blennow, P.; Hansen, A.B.; Hansen, J.B.; Mathiesen, B.V.; Mogensen, M.B. Recent Advances in Solid Oxide Cell Technology for Electrolysis. Science 2020, 370, eaba6118. [Google Scholar] [CrossRef] [PubMed]
- Badgett, A.; Brauch, J.; Thatte, A.; Rubin, R.; Skangos, C.; Wang, X.; Ahluwalia, R.; Pivovar, B.; Ruth, M. Updated Manufactured Cost Analysis for Proton Exchange Membrane Water Electrolyzers; National Renewable Energy Laboratory (NREL): Golden, CO, USA, 2023. [Google Scholar]
- Du, N.; Roy, C.; Peach, R.; Turnbull, M.; Thiele, S.; Bock, C. Anion-Exchange Membrane Water Electrolyzers. Chem. Rev. 2022, 122, 11830–11895. [Google Scholar] [CrossRef] [PubMed]
- Kaur, P.; Singh, K. Review of Perovskite-Structure Related Cathode Materials for Solid Oxide Fuel Cells. Ceram. Int. 2020, 46, 5521–5535. [Google Scholar] [CrossRef]
- Li, M.; Zhao, M.; Li, F.; Zhou, W.; Peterson, V.K.; Xu, X.; Shao, Z.; Gentle, I.; Zhu, Z. A Niobium and Tantalum Co-Doped Perovskite Cathode for Solid Oxide Fuel Cells Operating Below 500 °C. Nat. Commun. 2017, 8, 13990. [Google Scholar] [CrossRef]
- Shao, Z.; Haile, S.M. A High-Performance Cathode for the next Generation of Solid-Oxide Fuel Cells. Nature 2004, 431, 170–173. [Google Scholar] [CrossRef]
- Laguna-Bercero, M.A. Recent Advances in High Temperature Electrolysis Using Solid Oxide Fuel Cells: A Review. J. Power Sources 2012, 203, 4–16. [Google Scholar] [CrossRef]
- Hong, W.T.; Risch, M.; Stoerzinger, K.A.; Grimaud, A.; Suntivich, J.; Shao-Horn, Y. Toward the Rational Design of Non-Precious Transition Metal Oxides for Oxygen Electrocatalysis. Energy Environ. Sci. 2015, 8, 1404–1427. [Google Scholar] [CrossRef]
- Hwang, J.; Rao, R.R.; Giordano, L.; Katayama, Y.; Yu, Y.; Shao-Horn, Y. Perovskites in Catalysis and Electrocatalysis. Science 2017, 358, 751–756. [Google Scholar] [CrossRef]
- Sreedhar, I.; Agarwal, B.; Goyal, P.; Agarwal, A. An Overview of Degradation in Solid Oxide Fuel Cells-Potential Clean Power Sources. J. Solid State Electrochem. 2020, 24, 1239–1270. [Google Scholar] [CrossRef]
- Harrison, C.M.; Slater, P.R.; Steinberger-Wilckens, R. A Review of Solid Oxide Fuel Cell Cathode Materials with Respect to Their Resistance to the Effects of Chromium Poisoning. Solid State Ion. 2020, 354, 115410. [Google Scholar] [CrossRef]
- Khan, M.S.; Xu, X.; Knibbe, R.; Zhu, Z. Air Electrodes and Related Degradation Mechanisms in Solid Oxide Electrolysis and Reversible Solid Oxide Cells. Renew. Sustain. Energy Rev. 2021, 143, 110918. [Google Scholar] [CrossRef]
- Chen, F.-Y.; Wu, Z.-Y.; Adler, Z.; Wang, H. Stability Challenges of Electrocatalytic Oxygen Evolution Reaction: From Mechanistic Understanding to Reactor Design. Joule 2021, 5, 1704–1731. [Google Scholar] [CrossRef]
- Rost, C.M.; Sachet, E.; Borman, T.; Moballegh, A.; Dickey, E.C.; Hou, D.; Jones, J.L.; Curtarolo, S.; Maria, J.-P. Entropy-Stabilized Oxides. Nat. Commun. 2015, 6, 8485. [Google Scholar] [CrossRef]
- Dąbrowa, J.; Olszewska, A.; Falkenstein, A.; Schwab, C.; Szymczak, M.; Zajusz, M.; Moździerz, M.; Mikuła, A.; Zielińska, K.; Berent, K.; et al. An Innovative Approach to Design SOFC Air Electrode Materials: High Entropy La1−xSrx(Co, Cr, Fe, Mn, Ni)O3−δ(x = 0, 0.1, 0.2, 0.3) Perovskites Synthesized by the Sol-Gel Method. J. Mater. Chem. A Mater. 2020, 8, 24455–24468. [Google Scholar] [CrossRef]
- Yang, Y.; Bao, H.; Ni, H.; Ou, X.; Wang, S.; Lin, B.; Feng, P.; Ling, Y. A Novel Facile Strategy to Suppress Sr Segregation for High-Entropy Stabilized La0·8Sr0·2MnO3-δ Cathode. J. Power Sources 2021, 482, 228959. [Google Scholar] [CrossRef]
- Han, X.; Yang, Y.; Fan, Y.; Ni, H.; Guo, Y.; Chen, Y.; Ou, X.; Ling, Y. New Approach to Enhance Sr-Free Cathode Performance by High-Entropy Multi-Component Transition Metal Coupling. Ceram. Int. 2021, 47, 17383–17390. [Google Scholar] [CrossRef]
- Shijie, Z.; Na, L.; Liping, S.; Qiang, L.; Lihua, H.; Hui, Z. A Novel High-Entropy Cathode with the A2BO4-Type Structure for Solid Oxide Fuel Cells. J. Alloys Compd. 2022, 895, 162548. [Google Scholar] [CrossRef]
- Li, Z.; Guan, B.; Xia, F.; Nie, J.; Li, W.; Ma, L.; Li, W.; Zhou, L.; Wang, Y.; Tian, H.; et al. High-Entropy Perovskite as a High-Performing Chromium-Tolerant Cathode for Solid Oxide Fuel Cells. ACS Appl. Mater. Interfaces 2022, 14, 24363–24373. [Google Scholar] [CrossRef]
- Dąbrowa, J.; Stępień, A.; Szymczak, M.; Zajusz, M.; Czaja, P.; Świerczek, K. High-Entropy Approach to Double Perovskite Cathode Materials for Solid Oxide Fuel Cells: Is Multicomponent Occupancy in (La, Pr, Nd, Sm, Gd)BaCo2O5+δ Affecting Physicochemical and Electrocatalytic Properties? Front. Energy Res. 2022, 10, 899308. [Google Scholar] [CrossRef]
- Yang, Z.; Guo, M.; Wang, N.; Ma, C.; Wang, J.; Han, M. A Short Review of Cathode Poisoning and Corrosion in Solid Oxide Fuel Cell. Int. J. Hydrogen Energy 2017, 42, 24948–24959. [Google Scholar] [CrossRef]
- Zielińska, K.; Dąbrowa, J.; Zajusz, M.; Szymczak, M.; Li, K.; Winiarz, P.; Gajewska, M.; Świerczek, K. Alkali Free Perovskites—Exploiting B-Site Synergies in Sr-Free, High-Entropy Cathodes for Intermediate-Temperature Solid-Oxide Fuel Cells. Chem. Eng. J. 2025, 504, 158974. [Google Scholar] [CrossRef]
- Zhang, J.; You, J.; Zhao, Y.; Liu, T. High-Entropy Oxides as Promising Electrocatalysts for Oxygen Evolution Reaction: A Review. J. Environ. Chem. Eng. 2025, 13, 116550. [Google Scholar] [CrossRef]
- Liu, Q.; Dong, X.; Xiao, G.; Zhao, F.; Chen, F. A Novel Electrode Material for Symmetrical SOFCs. Adv. Mater. 2010, 22, 5478–5482. [Google Scholar] [CrossRef]
- Chung, Y.; Lee, C.-W. Electrochemistry of Gallium. J. Electrochem. Sci. Technol. 2013, 4, 1–18. [Google Scholar] [CrossRef]
- He, S.; Jiang, S.P. Electrode/Electrolyte Interface and Interface Reactions of Solid Oxide Cells: Recent Development and Advances. Prog. Nat. Sci. Mater. Int. 2021, 31, 341–372. [Google Scholar] [CrossRef]
- Nikonov, A.V.; Kuterbekov, K.A.; Bekmyrza, K.Z.; Pavzderin, N.B. A Brief Review of Conductivity and Thermal Expansion of Perovskite-Related Oxides for SOFC Cathode. Eurasian J. Phys. Funct. Mater. 2018, 2, 274–292. [Google Scholar] [CrossRef]
- Petric, A. Evaluation of La–Sr–Co–Fe–O Perovskites for Solid Oxide Fuel Cells and Gas Separation Membranes. Solid State Ion. 2000, 135, 719–725. [Google Scholar] [CrossRef]
- Zhou, W.; Ran, R.; Shao, Z. Progress in Understanding and Development of Ba0.5Sr0.5Co0.8Fe0.2O3−δ-Based Cathodes for Intermediate-Temperature Solid-Oxide Fuel Cells: A Review. J. Power Sources 2009, 192, 231–246. [Google Scholar] [CrossRef]
- Kim, J.-H.; Manthiram, A. LnBaCo2O5+δ Oxides as Cathodes for Intermediate-Temperature Solid Oxide Fuel Cells. J. Electrochem. Soc. 2008, 155, B385. [Google Scholar] [CrossRef]
- Mahato, N.; Banerjee, A.; Gupta, A.; Omar, S.; Balani, K. Progress in Material Selection for Solid Oxide Fuel Cell Technology: A Review. Prog. Mater. Sci. 2015, 72, 141–337. [Google Scholar] [CrossRef]
- Duan, C.; Hook, D.; Chen, Y.; Tong, J.; O’Hayre, R. Zr and Y Co-Doped Perovskite as a Stable, High Performance Cathode for Solid Oxide Fuel Cells Operating below 500 °C. Energy Environ. Sci. 2017, 10, 176–182. [Google Scholar] [CrossRef]
- Zhou, X.-D.; Yang, J.B.; Thomsen, E.-C.; Cai, Q.; Scarfino, B.J.; Nie, Z.; Coffey, G.W.; James, W.J.; Yelon, W.B.; Anderson, H.U.; et al. Electrical, Thermoelectric, and Structural Properties of La(MxFe1−x)O3 (M=Mn, Ni, Cu). J. Electrochem. Soc. 2006, 153, J133. [Google Scholar] [CrossRef]
- Tai, L. Structure and Electrical Properties of La1−xSrxCo1−yFeyO3. Part 1. The System La0.8Sr0.2Co1−yFeyO3. Solid State Ion. 1995, 76, 259–271. [Google Scholar] [CrossRef]
- Anderson, H.U.; Zhou, X.-D.; Dogan, F. Defect Chemistry of Mixed Ionic/Electronic P-Type Oxides. In Mixed Ionic Electronic Conducting Perovskites for Advanced Energy Systems; Springer: Dordrecht, The Netherlands, 2004; pp. 303–312. [Google Scholar]
- Song, H.J.; Yoon, H.; Ju, B.; Kim, D. Highly Efficient Perovskite-Based Electrocatalysts for Water Oxidation in Acidic Environments: A Mini Review. Adv. Energy Mater. 2021, 11, 2002428. [Google Scholar] [CrossRef]
- Liu, D.; Zhou, P.; Bai, H.; Ai, H.; Du, X.; Chen, M.; Liu, D.; Ip, W.F.; Lo, K.H.; Kwok, C.T.; et al. Development of Perovskite Oxide-Based Electrocatalysts for Oxygen Evolution Reaction. Small 2021, 17, 2101605. [Google Scholar] [CrossRef]
- Wang, Z.; Tan, S.; Xiong, Y.; Wei, J. Effect of B Sites on the Catalytic Activities for Perovskite Oxides La.6Sr.4CoxFe1-XO3−δ as Metal-Air Batteries Catalysts. Prog. Nat. Sci. Mater. Int. 2018, 28, 399–407. [Google Scholar] [CrossRef]
- Sun, H.; Chen, G.; Sunarso, J.; Dai, J.; Zhou, W.; Shao, Z. Molybdenum and Niobium Codoped B-Site-Ordered Double Perovskite Catalyst for Efficient Oxygen Evolution Reaction. ACS Appl. Mater. Interfaces 2018, 10, 16939–16942. [Google Scholar] [CrossRef]
- Mahmoudi, E.; Fattahi, E.; Bakranova, D.; Delibas, N.; Asghari, E.; Coruh, A.; Niaei, A. Perovskite Engineering for Efficient Oxygen Evolution Reaction through Iron and Silver Doping. Sci. Rep. 2025, 15, 34426. [Google Scholar] [CrossRef]
- Li, K.; Tao, Z.; Ma, X.; Wu, J.; Wu, T.; Guo, C.; Qi, Y.; Yu, J.; Zheng, J.; Xue, J. The Application and Research Progress of D-Band Center Theory in the Field of Water Electrolysis. Int. J. Hydrogen Energy 2025, 132, 183–211. [Google Scholar] [CrossRef]
- Fabbri, E.; Schmidt, T.J. Oxygen Evolution Reaction—The Enigma in Water Electrolysis. ACS Catal. 2018, 8, 9765–9774. [Google Scholar] [CrossRef]
- Jones, M.; Lyu, C.; Trudgeon, D.; Corbin, J.; Nawrocka, I.; Zhang, Z.; Lykkegaard, M.B.; Guan, S.; Dodwell, T.; Li, X. Improving Electrocatalytic OER Activity of Perovskites through Insertion of Ternary B-Site Metals. Mater. Today Chem. 2024, 42, 102414. [Google Scholar] [CrossRef]
- Dierickx, S.; Weber, A.; Ivers-Tiffée, E. How the Distribution of Relaxation Times Enhances Complex Equivalent Circuit Models for Fuel Cells. Electrochim. Acta 2020, 355, 136764. [Google Scholar] [CrossRef]
- Xia, J.; Wang, C.; Wang, X.; Bi, L.; Zhang, Y. A Perspective on DRT Applications for the Analysis of Solid Oxide Cell Electrodes. Electrochim. Acta 2020, 349, 136328. [Google Scholar] [CrossRef]
- Zhang, Y.; Shen, L.; Wang, Y.; Du, Z.; Zhang, B.; Ciucci, F.; Zhao, H. Enhanced Oxygen Reduction Kinetics of IT-SOFC Cathode with PrBaCo2O5+δ/Gd0.1Ce1.9O2−δ Coherent Interface. J. Mater. Chem. A Mater. 2022, 10, 3495–3505. [Google Scholar] [CrossRef]
- Toby, B.H.; Von Dreele, R.B. GSAS-II: The Genesis of a Modern Open-Source All Purpose Crystallography Software Package. J. Appl. Crystallogr. 2013, 46, 544–549. [Google Scholar] [CrossRef]
- Stygar, M.; Dąbrowa, J.; Moździerz, M.; Zajusz, M.; Skubida, W.; Mroczka, K.; Berent, K.; Świerczek, K.; Danielewski, M. Formation and Properties of High Entropy Oxides in Co-Cr-Fe-Mg-Mn-Ni-O System: Novel (Cr, Fe, Mg, Mn, Ni)3O4 and (Co, Cr, Fe, Mg, Mn)3O4 High Entropy Spinels. J. Eur. Ceram. Soc. 2020, 40, 1644–1650. [Google Scholar] [CrossRef]
- Nielsen, J.; Hjelm, J. Impedance of SOFC Electrodes: A Review and a Comprehensive Case Study on the Impedance of LSM:YSZ Cathodes. Electrochim. Acta 2014, 115, 31–45. [Google Scholar] [CrossRef]
- Wan, T.H.; Saccoccio, M.; Chen, C.; Ciucci, F. Influence of the Discretization Methods on the Distribution of Relaxation Times Deconvolution: Implementing Radial Basis Functions with DRTtools. Electrochim. Acta 2015, 184, 483–499. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.