Abstract
Electrocatalytic hydrogenation (ECH) represents an environmentally friendly pathway for converting 5-hydroxymethylfurfural (HMF) into the high-value chemical 2,5-bis(hydroxymethyl)furan (BHMF). However, its selectivity and Faradaic efficiency are often constrained by competitive hydrogen evolution at the cathode and insufficient supply of active hydrogen at the surface. To address this challenge, this study developed an Ir-decorated copper oxide nanowire catalyst (denoted as CuIr) featuring a hydrogen-rich adsorption (Hads) surface. The incorporation of Ir significantly enhances the catalyst’s water dissociation capacity, creating abundant Hads sources that selectively accelerate HMF hydrogenation while suppressing side reactions. Under a mild applied potential of −0.45 V vs. RHE and a current density of approximately −20 mA cm−2, the optimal CuIr40 catalyst achieved near-complete conversion of HMF (99%), a BHMF yield of 99%, and a high Faradaic efficiency of 97% within 120 min of electrolysis. Mechanistic studies reveal that this catalytic leap stems from the synergistic functional interaction between Cu and Ir sites in substrate activation and hydrogen supply. This work presents a novel strategy for designing efficient electrocatalysts for biomass hydrogenation by regulating surface Hads concentration.
1. Introduction
With the escalating environmental and resource challenges associated with fossil fuel consumption, the conversion of renewable biomass into high-value-added chemicals has emerged as a critical pathway for achieving sustainable development [1,2,3,4]. The catalytic conversion of biomass not only offers a forward-looking solution for producing high-value chemicals but also helps alleviate the depletion of finite fossil fuel resources [5,6,7,8]. Among numerous biomass derivatives, furan-based aldehydes—particularly 5-hydroxymethylfurfural (HMF)—serve as pivotal platform molecules bridging biomass resources and the chemical industry due to their unique chemical structure [9,10,11]. HMF can be synthesized from biomass carbohydrates and demonstrates exceptional application potential among numerous platform compounds [12,13]. Its hydrogenation product, 2,5-bis(hydroxymethyl)furan (BHMF), serves as a crucial precursor for synthesizing bio-based polymers, fuels, and pharmaceutical intermediates, offering broad application prospects [14,15,16,17,18]. Currently, the industrial conversion of HMF to BHMF typically requires high-temperature and high-pressure conditions, relying on H2 as the hydrogen source [19]. Consequently, developing a catalytic system capable of efficiently converting HMF under mild conditions holds significant importance [5,20,21].
In recent years, electrocatalytic hydrogenation (ECH) technology has garnered significant attention as a green and highly efficient alternative solution [22]. Due to its ability to achieve highly efficient selective reactions under mild conditions, coupled with the advantages of energy-tunable and green sustainable properties, it is regarded as a highly promising catalytic technology [23]. In ECH systems, selective regulation of reaction products can be achieved by adjusting parameters such as current density and voltage [24]. Copper-based, nickel-based, and other non-precious metal catalysts have been widely utilized in selective hydrogenation reactions, allowing for the targeted synthesis of desired products through the modulation of metal electronic structures, surface acid–base properties, and support synergistic effects [25,26]. Among numerous electrode materials, copper(Cu)-based catalysts demonstrate application potential in the ECH of biomass-derived aldehydes due to their low cost and moderate adsorption strength towards carbonyl groups [27,28,29]. The rich crystal surface structure provides a structural design basis for regulating reaction pathways [30].
In this context, the use of Cu-based catalysts for the hydrogenation of HMF to BHMF has emerged as a research focus. It has been demonstrated that Cu can effectively adsorb and activate the C=O bond in HMF molecules, which is crucial for its hydrogenation conversion [31]. For example, Yang et al. systematically reviewed studies on the electrocatalytic hydrogenation of furanic aldehydes, detailing reaction parameters and mechanisms involving HMF [31]. Elsayed et al. achieved efficient catalytic reduction of HMF to BHMF under hydrogen-free conditions using a copper–iron oxide bimetallic nanocatalyst [32]. To further enhance performance, researchers often combine Cu with other metals to construct bimetallic catalysts. For instance, Liu et al. prepared CuM (M = Pt, Pd, Au, Ag) bimetallic catalysts via a simple replacement method, and these catalysts exhibited enhanced activity in the electrocatalytic conversion of furanic aldehydes [33]. Studies have also indicated that combining Cu with noble metals (e.g., Pt, Pd) or non-noble metals can effectively modulate the electronic structure of Cu, strengthen its adsorption and activation toward HMF, and simultaneously improve the catalyst’s ability to generate adsorbed hydrogen (Hads), thereby enhancing the selectivity and reaction kinetics for BHMF [33,34]. However, single Cu-based catalysts frequently encounter challenges of low Faraday efficiency (FE) and BHMF selectivity in the hydrogenation of HMF. This is primarily attributable to competition from the hydrogen evolution reaction (HER) and insufficient supply of active Hads on the catalyst surface, leading to side reactions [27,35]. Research indicates that the generation and utilization efficiency of Hads is one of the key factors determining the selectivity of furan–aldehyde compounds toward ECH [36,37]. Its formation depends on the characteristics of the electrode surface and involves direct interactions between the reactants and the electrode surface [38].
To overcome the aforementioned limitations, constructing bimetallic catalysts to harness intermetallic synergistic effects represents an effective strategy for enhancing the performance of copper-based catalysts [39,40]. By combining two metals, not only can their respective advantages be leveraged, but the overall catalytic performance, selectivity, and stability can also be optimized through adjustments to the electronic structure [41]. Common precious metals such as platinum (Pt) and palladium (Pd) are widely used due to their high reactivity [42], but they come with a high cost. In contrast, iridium (Ir) is regarded as an ideal component for promoting Hads generation due to its exceptional water dissociation capability and moderate adsorption strength towards hydrogen species [43,44]. Introducing Ir into the Cu matrix is expected to significantly enhance the Hads enrichment capacity on the catalyst surface by constructing bimetallic active sites [45]. Accordingly, this study designed and prepared a novel nanowire catalyst featuring iridium species decorated on a copper oxide substrate. Through an electrolytically induced displacement method, Ir was etched onto the surface of copper nanowires, successfully constructing this composite architecture. Electron spin resonance (ESR) analysis reveals that this catalyst possesses highly active Hads, effectively promoting water dissociation and establishing a Hads-rich interfacial environment. Kinetic analyses and spectroscopic characterizations demonstrate that Ir species catalyze the formation of interfacial Hads, thereby enhancing HMF hydrogenation selectivity and effectively suppressing undesirable dimerization side reactions. This research offers novel insights for the rational design of interfacial aquatic environments and provides a viable approach for the efficient catalytic conversion of biomass-derived molecules.
2. Results and Discussion
2.1. Characterization of the Physicochemical Properties of Catalysts
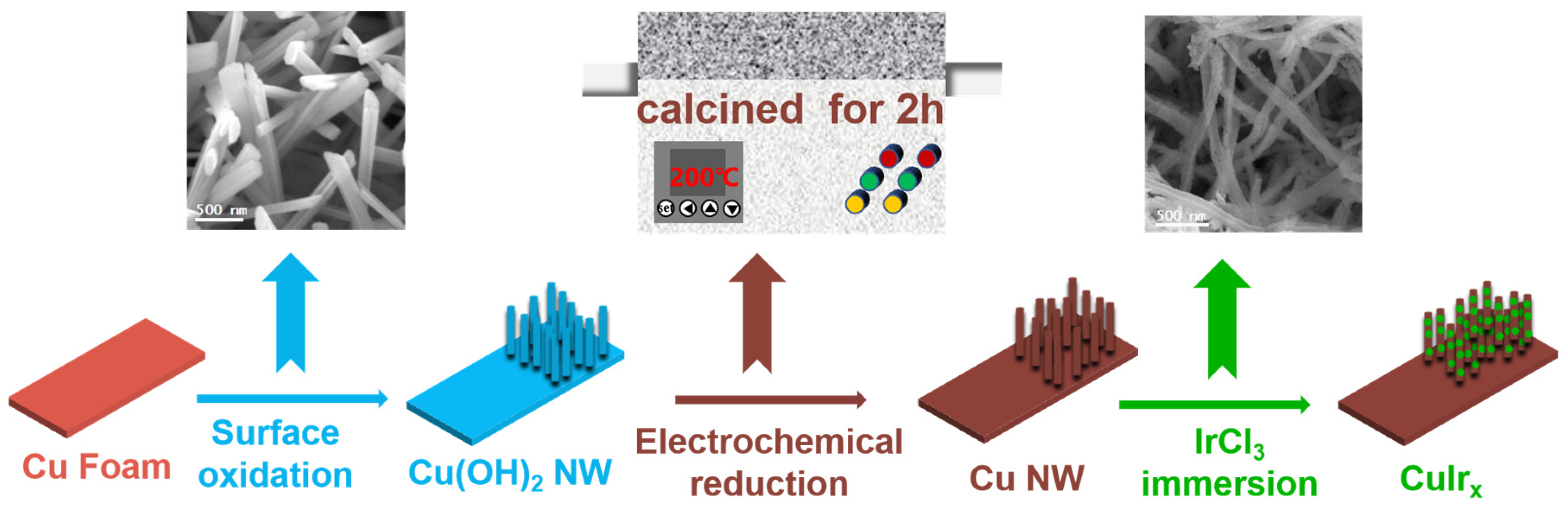
The synthesis process for the Ir-decorated copper oxide nanowire catalyst (denoted as CuIrx) is exhibited in Figure 1. Employing the galvanic replacement method, copper nanowires were immersed in an IrCl3 solution for surface etching and deposition, successfully yielding a CuIr bimetallic nanowire catalyst loaded with iridium species. The iridium content of the samples was quantified by ICP, as shown in Table S1.
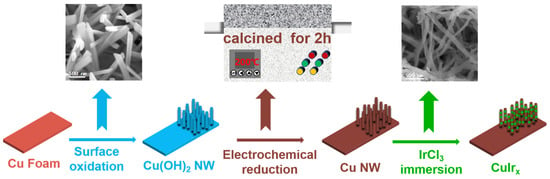
Figure 1.
The schematic illustration for CuIrx preparation.
As shown in Figure S2, the SEM image of the Cu nanowire clearly demonstrates its nanowire structure. With increasing electrostatic displacement time, the CuIr10, CuIr20, CuIr30, CuIr40, and CuIr50 samples retained their distinct nanowire structure without exhibiting any signs of damage or deformation. Figure 2 presents a comparative analysis of CuIr40 and Cu nanowires. Figure 2a–d shows SEM and TEM images of Cu nanowires before and after galvanic replacement, respectively. It can be observed that the surface of the etched nanowires becomes rougher, which will enhance contact between the catalyst surface and HMF. Figure 2e–h presents the EDX elemental distribution maps for CuIr40, revealing uniform dispersion of Ir across the surface. This confirms successful galvanic replacement, wherein Ir has etched away portions of the Cu atoms. Furthermore, Figure 2i,j displays the SAED patterns for CuIr40 and Cu NW. The results indicate no new diffraction rings emerged prior to or following the etching process. Further HRTEM characterization (Figure 2k,l) revealed, through measurement of lattice spacings, that the (111), (200) and (220) crystal planes of Cu exhibit spacings of 0.2325 nm, 0.1834 nm and 0.1243 nm, respectively, whereas the Ir (111) crystal plane spacing is 0.2325 nm. (200), and (220) crystal planes were determined to be 0.2325 nm, 0.1834 nm, and 0.1243 nm, respectively, whilst the Ir (111) plane measured 0.2467 nm. Based on the distribution of surface lattice planes, this indicates that the Ir etching did not remain confined to the surface but penetrated into the nanowire structure.

Figure 2.
The SEM images of samples Cu NW and CuIr40 (a,b), TEM images of Cu NW and CuIr40 (c,d), EDX images of CuIr40 (e–h), SAED images of CuIr40 and Cu NW (i,j), and HRTEM images of Cu NW and CuIr40 (k,l).
To investigate the influence of varying metal deposition amounts on the specific surface area of the catalyst, N2 adsorption–desorption experiments were performed. The results are presented in Figure 3, with corresponding data summarized in Table 1. The presence of hysteresis loops in the isotherms (Figure 3) confirm the mesoporous nature of all catalysts. Compared to the pristine Cu NW, the introduction of Ir generally resulted in an increase in specific surface area and a shift in the average pore size toward a smaller range. This structural evolution toward more defined mesoporosity likely enhances the substrate adsorption capacity. The observed non-linear variation in surface area with etching time is attributable to the complex galvanic replacement process, which can lead to non-uniform etching and deposition. The increased specific surface area provides a greater number of active sites and improves metal dispersion, strengthening the interaction between Ir and the nanowire support. This contributes to enhanced catalyst stability and water dissociation capability, thereby facilitating the supply of Hads for the protonation step in the hydrogenation reaction.
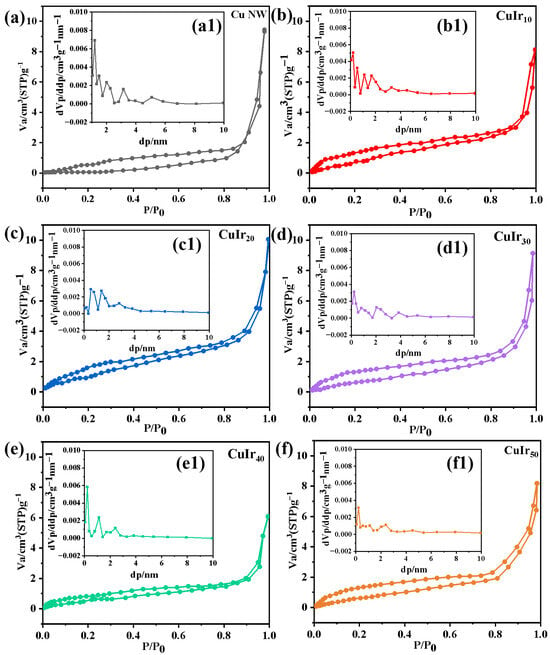
Figure 3.
The N2 adsorption-desorption isotherms and pore size distribution of samples Cu NW (a,a1), CuIr10 (b,b1), CuIr20 (c,c1), CuIr30 (d,d1), CuIr40 (e,e1) and CuIr50 (f,f1).

Table 1.
The SBET value and average pore diameter of catalysts.
As shown in Figure 4, X-ray diffraction (XRD) was used to analyze the crystal structures of Cu NW and CuIr catalysts. Three characteristic diffraction peaks were observed at 43.7°, 50.9°, and 74.3°, corresponding to the Cu (111), Cu (200), and Cu (220) planes, respectively. These characteristic peaks confirm the presence of a well-defined Cu crystalline structure in the samples. No distinct peaks corresponding to Ir were detected, and no significant peak shifts in the Cu diffraction pattern were observed after Ir etching, which could be attributed to the low Ir content [46].
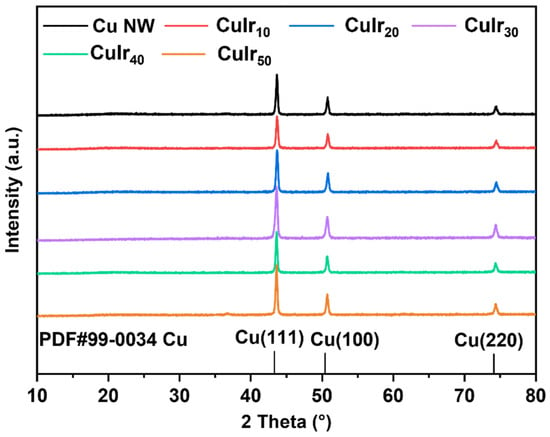
Figure 4.
XRD pattern of prepared catalysts.
X-ray photoelectron spectroscopy (XPS) was employed to analyze the electronic structure changes in surface elements (Ir, Cu, and O) in the catalysts, as shown in Figure 5. The full XPS spectra of CuIr40 and Cu NW are displayed in Figure 5a. Figure 5b shows the Ir element peaks in the CuIr40 catalyst, indicating the presence of Ir primarily in the metallic state with a small portion in the oxidized state. A detailed analysis of the O 1s region for CuIr40 and Cu NW is shown in Figure 5c. The results reveal two primary oxidation states of oxygen species on the surface, corresponding to Cu-O and Cu-O-Cu. The former arises from slight oxidation of the electrode surface and adsorption of oxygen atoms, while the latter likely results from the partial conversion of Cu2O during the calcination process [47]. Figure 5d illustrates the change in the valence state of Cu on the surfaces of CuIr40 and Cu NW. Both catalysts exhibit strong peaks associated with Cu0/Cu+ species, with only minor satellite peaks of Cu2+, indicating that most of the surface copper species have been reduced to lower oxidation states or metallic copper [48]. Notably, a positive shift of 0.43 eV in the Cu peak was observed on the surface of CuIr40, which was absent in Cu NW, suggesting the formation of a Cu-Ir bimetallic interaction at the catalyst surface.

Figure 5.
The XPS spectra of CuIr40 and Cu NW (a) high-resolution spectra of Ir 4f (b), O 1s (c), and Cu 2p (d).
2.2. Catalytic Performance
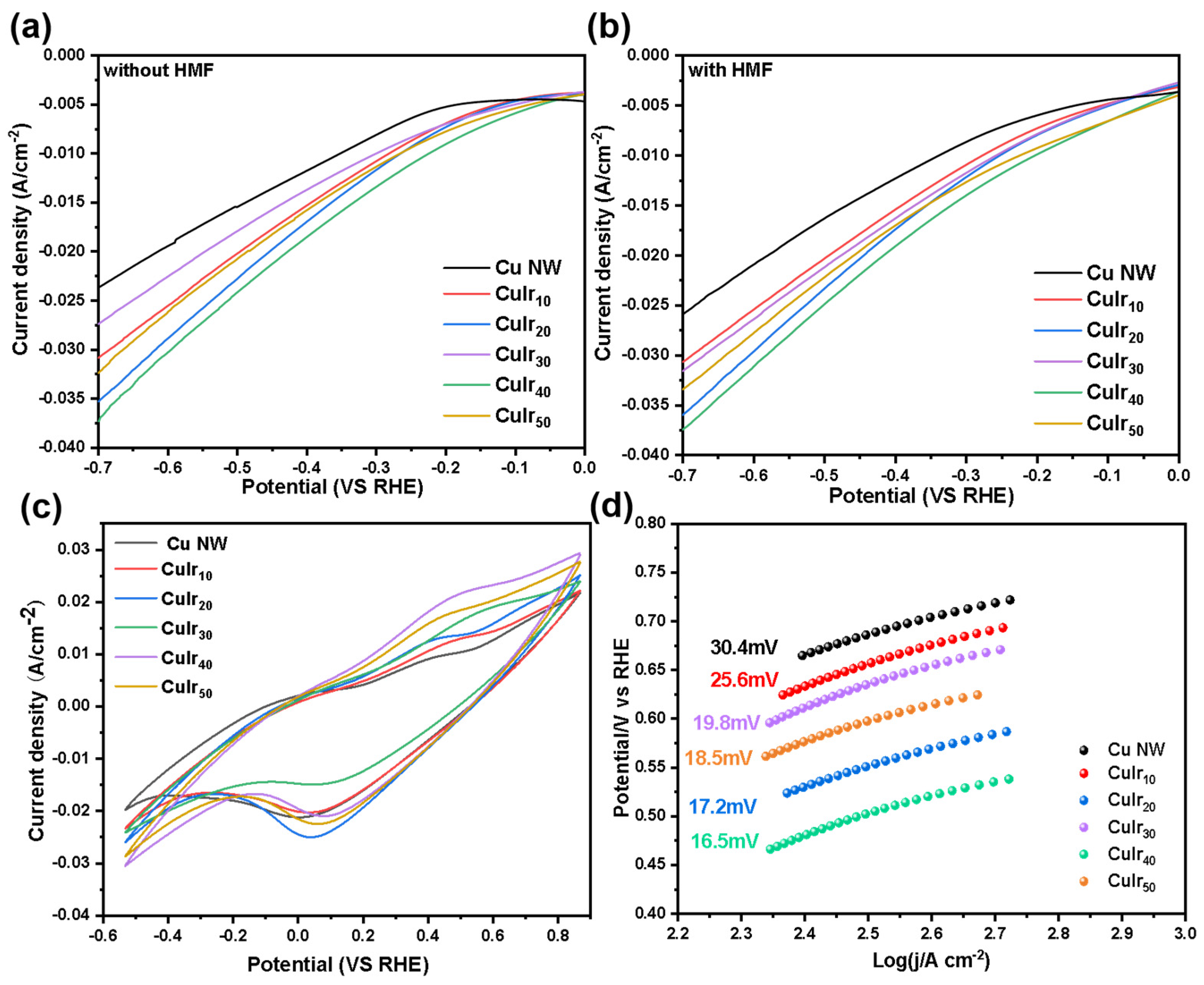
As shown in Figure 6, to evaluate the electrocatalytic performance of the prepared catalysts, linear sweep voltammetry (LSV), cyclic voltammetry (CV), and Tafel slope measurements were carried out for CuIrx nanowire catalysts with different immersion durations. Figure 6a,b show that the CuIr40 catalyst exhibited the highest current density over a wide potential range both before and after the addition of HMF substrate, significantly outperforming the other samples. Therefore, CuIr40 was selected as the optimal catalyst for further investigation. Figure 6c presents the CV curves recorded in 0.1 M KOH. All catalysts displayed pronounced peaks around 0.4–0.45 V, which can be attributed to the desorption of Hads species, indicating that Hads desorption occurred at these potentials. Notably, the desorption peak observed on CuIr40 was significantly more intense, implying a higher generation of Hads on its surface. This observation was further confirmed by ESR analysis, which highlighted the crucial role of Ir in enhancing Hads formation. Figure 6d presents the Tafel plots of the different catalysts. In line with the LSV results, CuIr40 exhibited the lowest Tafel slope, indicating superior reaction kinetics and the lowest overpotential barrier. This also suggests that the proton–electron coupling process on its surface is well-regulated, and the overall hydrogenation reaction is primarily governed by the adsorption and desorption behavior of Hads.

Figure 6.
LSV curves of different catalysts measured in 0.1 M KOH without (a) and with (b) 20 mM HMF, CV curves of different catalysts (c), and Tafel plots of different catalysts (d).
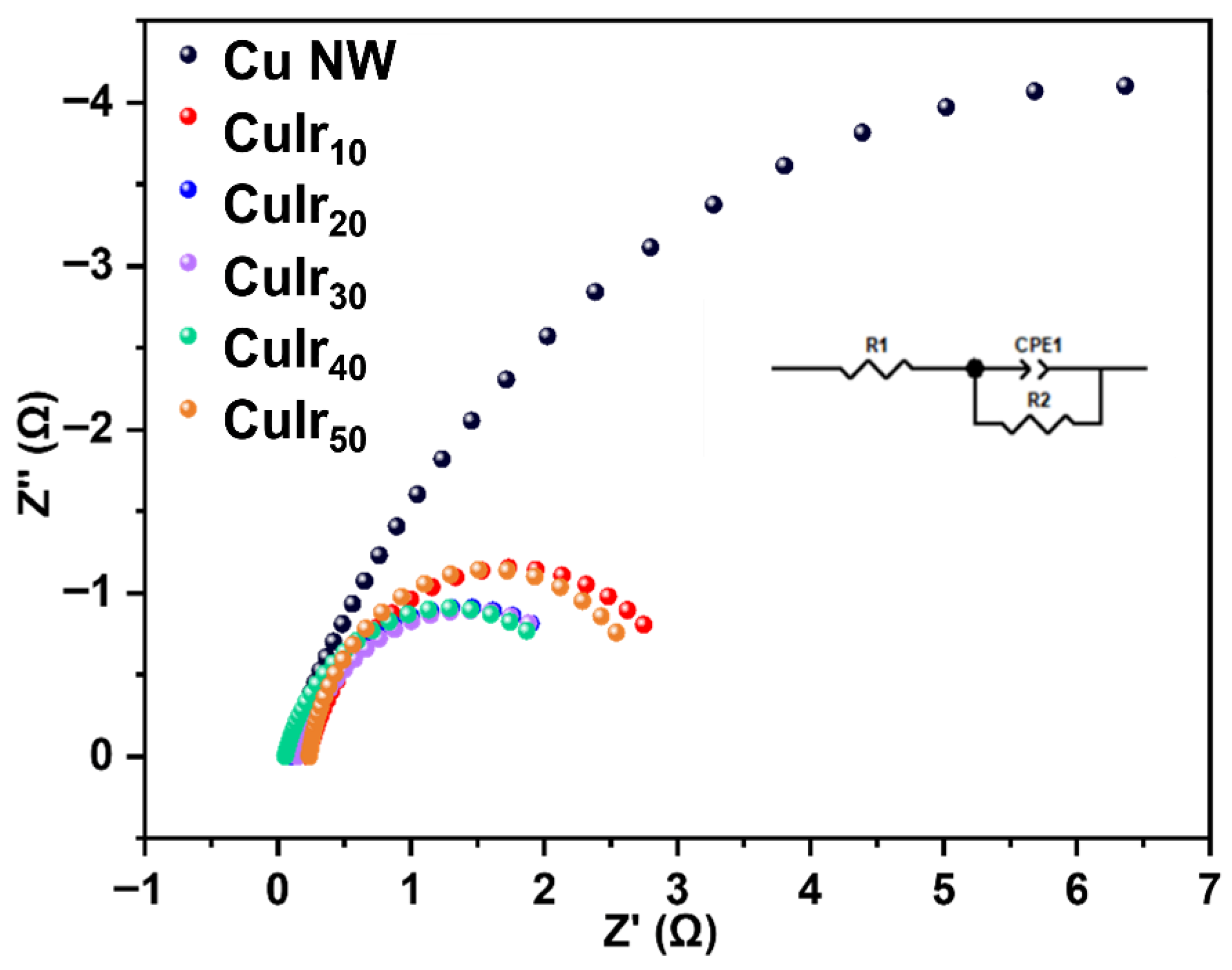
As shown in Figure 7, to further investigate the charge transfer capability of the catalysts during the electrocatalytic process, electrochemical impedance spectroscopy (EIS) was conducted on different catalyst samples. The results reveal that the pristine Cu NW sample, without any metal doping, exhibits relatively high charge transfer resistance. In contrast, upon the incorporation of Ir, the CuIrx series catalysts show significantly reduced impedance values, indicating improved electronic conductivity. This suggests that the introduction of Ir effectively modifies the surface electronic structure of the catalyst, enhances charge migration efficiency between active sites, and lowers the energy barrier for interfacial charge transfer. Meanwhile, the proton/intermediate diffusion efficiency on the catalyst surface is also improved, indicating that the CuIrx catalysts possess higher intrinsic activity and faster reaction kinetics in the ECH of HMF. These findings further confirm the significant role of bimetallic synergy in enhancing catalytic performance. The observed non-monotonic variation in charge transfer resistance (Rct) with increasing Ir content can be attributed to the evolution of surface electronic structure and active site distribution. Initially, the introduction of Ir enhances the overall conductivity and facilitates charge transfer, leading to a decrease in Rct. However, beyond an optimal loading (CuIr40), further Ir incorporation may slightly disrupt the optimal electronic coupling between Cu and Ir sites, as inferred from the positive shift in the Cu 2p XPS peak (Figure 5d). This could modestly increase the interfacial charge transfer barrier. Consequently, the minimum Rct attained for CuIr40 corresponds to the most favorable electronic environment for efficient charge transfer, which aligns well with its highest activity and selectivity in HMF hydrogenation. This optimal charge transfer property of CuIr40 is consistent with its superior catalytic activity and selectivity observed in Figure 6 and Figure 8. This structure–activity relationship underscores the importance of optimizing bimetallic composition to achieve synergistic effects in electrocatalytic performance.
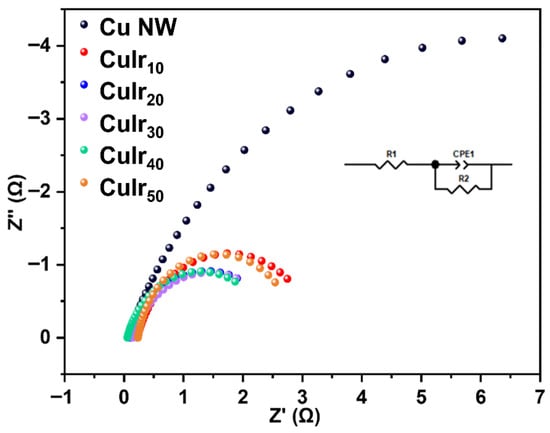
Figure 7.
EIS spectra of different catalysts.
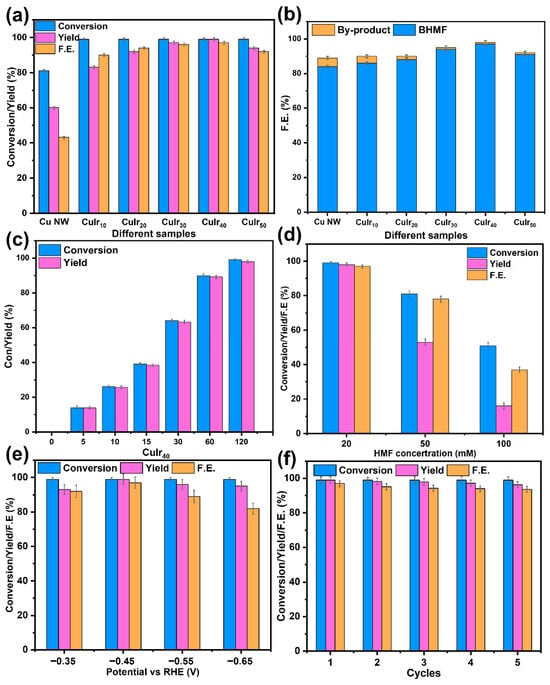
Figure 8.
Catalytic performance of different samples (a,b), reaction kinetics of the best sample (c), performance at different substrate concentrations (d), performance at different voltages (e) and regeneration performance (f).
To investigate the effect of Ir introduction on the hydrogenation performance of HMF, electrochemical measurements and product analyses were conducted on samples subjected to different etching times. As shown in Figure 8a,b, which present the hydrogenation capabilities of different catalysts, the obtained results are consistent with the data from electrochemical tests. The introduction of Ir significantly enhances the selectivity toward BHMF and improves the FE. This demonstrates that increasing the generation of Hads indeed exerts a positive influence on the hydrogenation reaction. Compared with the Cu NW catalyst, the CuIrx series not only improved the conversion of HMF but also enhanced the yield of BHMF. The FE for BHMF formation exhibited a volcano-shaped trend across the series, initially increasing and then declining with higher Ir content, with CuIr40 showing the maximum value. These results indicate that CuIr40 is the optimal catalyst. Figure 8c illustrates the hydrogenation kinetics of CuIr40. Both yield and conversion increase synchronously during the reaction, but a fast-to-slow trend is observed over time due to the gradual depletion of HMF substrate and the reduced adsorption efficiency. After 120 min of reaction, a conversion rate and product yield of 99% were achieved, with almost no detectable byproducts. Figure 8d presents the hydrogenation performance of CuIr40 under different substrate concentrations. When the substrate concentration was increased to 2.5 and 5 times the original level, both the FE and selectivity decreased, which may be attributed to insufficient Hads generation to match the protonation pathway. This result also indicates that the optimal concentration of HMF is 20 mM. Figure 8e shows the hydrogenation performance of CuIr40 at different applied voltages. As the voltage increased, the conversion rate remained relatively stable, but both FE and selectivity declined. This phenomenon could result from the occurrence of HER and excessive hydrogenation involving 4 Hads under high Hads conditions. Thus, −0.45 V (vs. RHE) is identified as the optimal applied potential. Figure 8f presents the regeneration performance of CuIr40. After five reaction cycles, the catalyst maintained its performance without significant degradation, indicating excellent reusability.
2.3. Mechanistic Insights into the HMF-to-BHMF Hydrogenation Pathway over CuIrx Catalysts
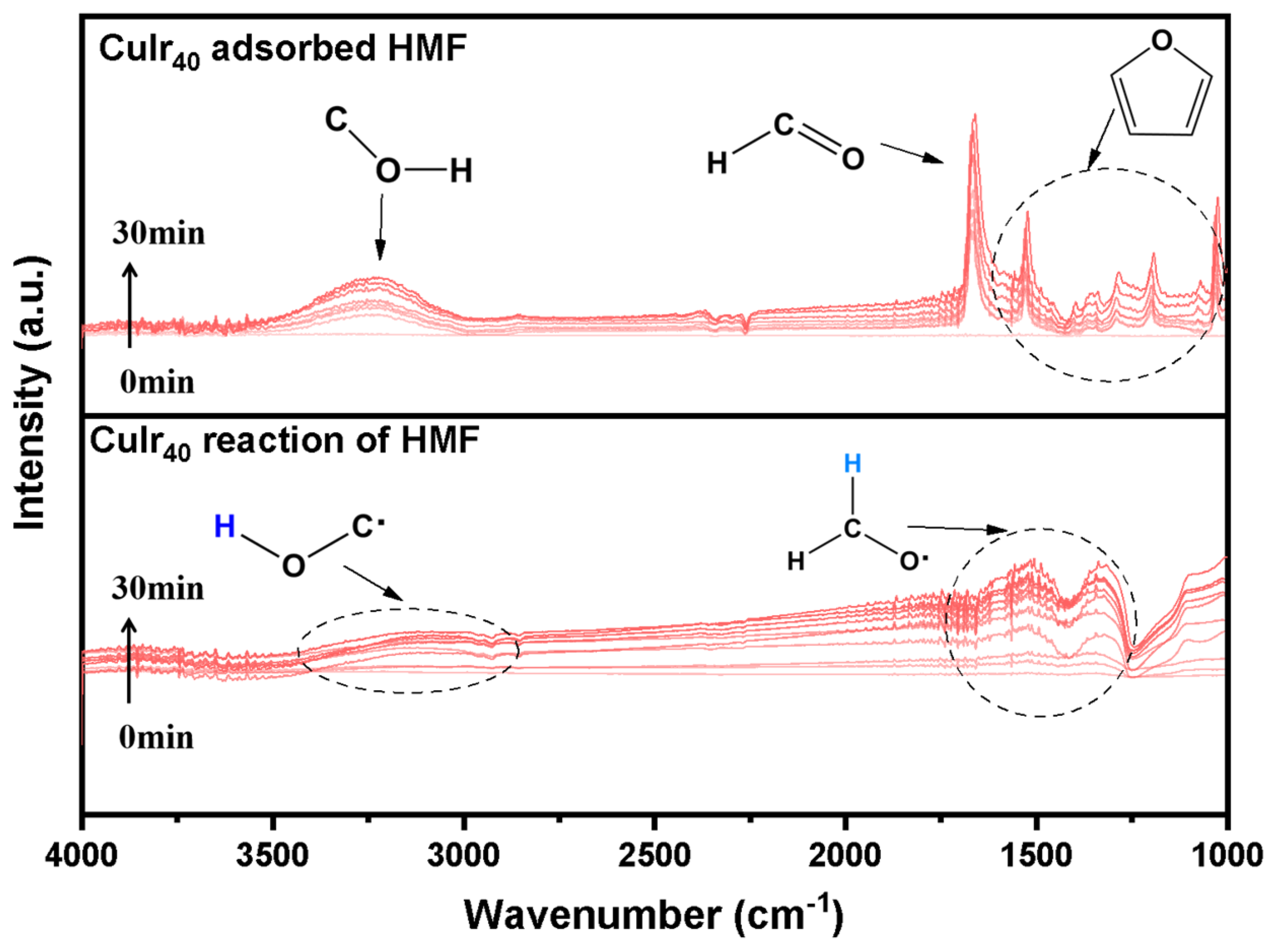
As shown in Figure 9, the adsorption and reaction process of HMF on the surface of the CuIr40 electrode was analyzed by electrochemical in situ infrared (ECIR). During the initial adsorption stage, a distinct C=O stretching vibration peak appeared at 1650 cm−1 in the spectrum, and its intensity gradually increased with prolonged reaction time, indicating a stable and continuous adsorption behavior of the aldehyde group from HMF on the catalyst surface. Concurrently, a broad peak associated with the hydroxymethyl (-CH2OH) group was observed around 3300 cm−1, suggesting that the hydroxymethyl moiety of the HMF molecule also effectively interacted with the catalytic surface. Furthermore, characteristic vibration peaks of the furan ring skeleton were clearly visible, further demonstrating the favorable overall adsorption capability of the CuIr40 catalyst for HMF. This multi-site adsorption behavior facilitates the stable formation of reaction intermediates and promotes subsequent reaction steps [49]. As the electrocatalytic reaction progressed, new infrared bands associated with radical intermediates began to emerge and showed increasing intensity over time. This indicates that the aldehyde group underwent electron transfer on the electrode surface, leading to the formation of radicals such as C·-OH and CH-O·. The generation of these radical intermediates requires the involvement of adsorbed hydrogen species, implying that the CuIr40 surface provides a sufficient quantity and diversity of Hads species to support the stepwise hydrogenation of the aldehyde group. Considering that Ir sites on the catalyst surface possess high water dissociation activity, it is inferred that a Hads-rich environment is formed under the synergistic effect of the Cu-Ir bimetallic system. This environment significantly accelerates the conversion of radical intermediates into the target product BHMF. This finding not only confirms the excellent HMF adsorption and catalytic properties of CuIr40 but also reveals the evolution pathway of key intermediates and highlights the mechanistic role of Hads in the electrocatalytic hydrogenation process.

Figure 9.
ECIR spectra of HMF adsorption and reaction on CuIr40.
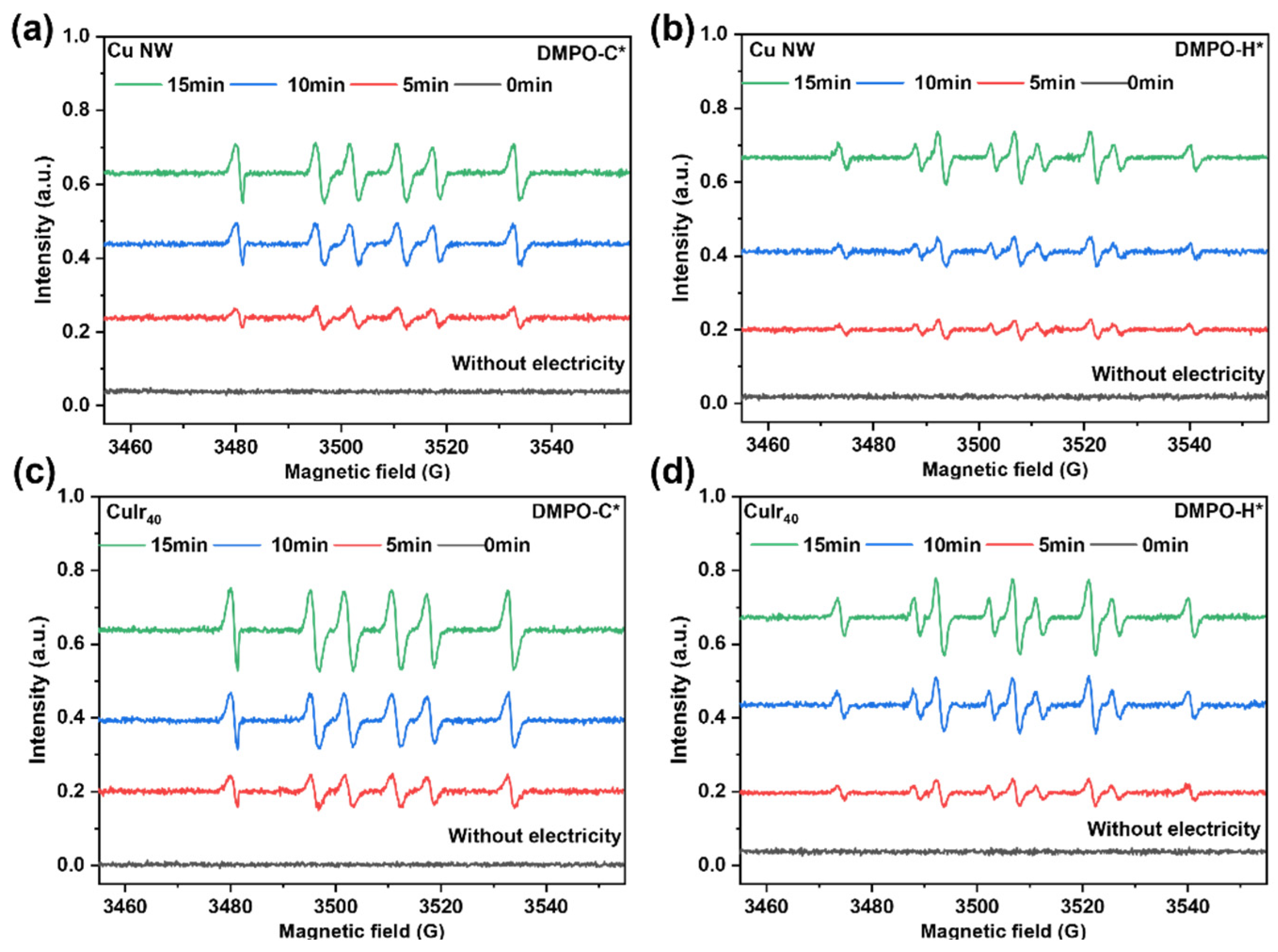
The generation of radical species was analyzed by ESR spectroscopy, as presented in Figure 10. The introduction of Ir notably enhanced the formation of both DMPO-H* and DMPO-C* adducts (DMPO: 5,5-dimethyl-1-pyrroline N-oxide, a spin-trapping agent). Quantitative integration of the ESR spectra revealed that the signal intensities corresponding to DMPO-H* and DMPO-C* increased by factors of 1.51 and 2.39, respectively, for CuIr40 relative to Cu NW (Supplementary Figure S5). This differential enhancement indicates that Ir not only promotes the formation of active hydrogen species (H*) but also, more markedly, facilitates the generation of carbon-centered radicals (C*) derived from HMF activation. The pronounced increase in DMPO-C* signals suggests that Ir effectively assists in the cleavage and electron transfer of the carbonyl (C=O) group in HMF. The concurrent rise in both radical types supports a synergistic mechanism in which the abundant H* species—supplied through Ir-enhanced water dissociation—efficiently couple with the readily formed C* intermediates on the catalyst surface. This cooperative radical environment directly accelerates the hydrogenation kinetics and improves the overall catalytic activity. Furthermore, the Hads-rich surface effectively suppresses undesirable dimerization side reactions. Together, these results demonstrate that Ir significantly boosts catalytic performance by co-promoting the formation and coupling of key radical species essential for the selective hydrogenation pathway [50].
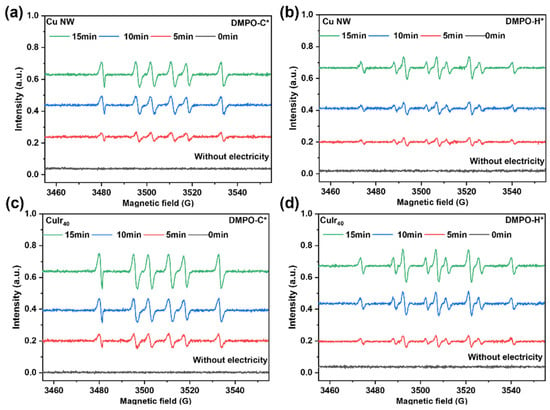
Figure 10.
ESR spectra of DMPO-trapped radical adducts for different catalysts. DMPO-C* adduct for Cu NW (a), DMPO-H* adduct for Cu NW (b), DMPO-C* adduct for CuIr40 (c) and DMPO-H* adduct for CuIr40 (d).
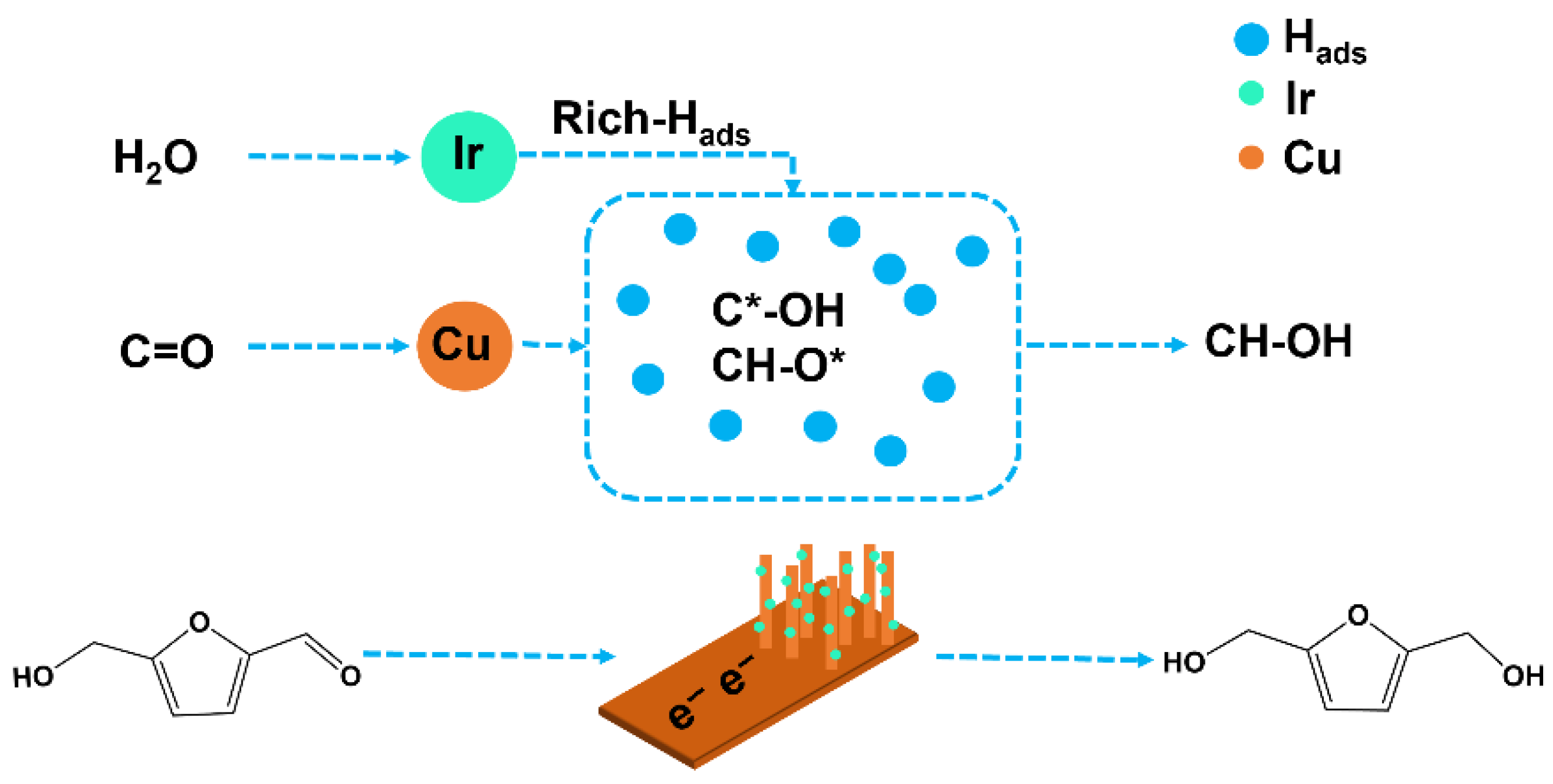
In summary, as shown in Figure 11, the electrocatalytic hydrogenation mechanism of HMF involves electron transfer from the carbonyl group, the formation of a radical intermediate, and subsequent stepwise hydrogenation. In the CuIr bimetallic catalyst system, Cu primarily facilitates the adsorption and activation of HMF molecules, particularly through directional adsorption at the C=O carbonyl group and initial electron transfer. The introduction of Ir significantly alters the catalyst’s surface reaction environment, exhibiting notably higher activity in water molecule dissociation behavior. Ir exhibits excellent water dissociation capability, effectively decomposing water molecules into protons and hydroxyl groups. The generated H+ ions then bind electrons at the electrode surface to form Hads. This process significantly increases the concentration of Hads on the catalyst surface, thereby creating a Hads-rich environment. This enriched Hads environment exerts a significant influence on the hydrogenation process: First, it enables rapid coupling reactions with free radical intermediates adsorbed on the catalyst surface (e.g., C·-OH, CH-O·), promoting the conversion of intermediates into the target product BHMF. Second, the high Hads concentration helps lower the reaction energy barrier and increase the reaction rate, thereby achieving higher Faraday efficiency and yield. Additionally, a certain degree of electronic interaction or alloying behavior may exist between Cu and Ir, thereby regulating the electronic structure of Cu and further optimizing its adsorption behavior and reaction selectivity. This synergistic effect plays a crucial role in promoting the directional adsorption of HMF molecules, stabilizing intermediate structures, and enhancing overall catalytic performance. Therefore, the introduction of Ir not only provides active sites for water dissociation but also plays a central role in promoting HMF-to-BHMF conversion by establishing a Hads-rich catalytic environment.
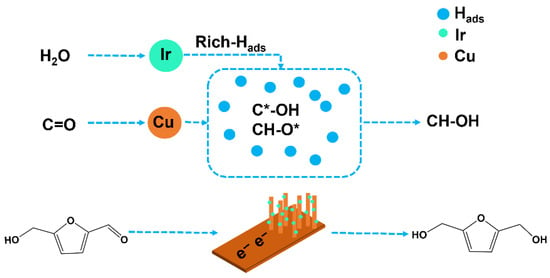
Figure 11.
Mechanism diagram of electrocatalytic hydrogenation of HMF to form BHMF in Hads-rich environment.
3. Experimental Section
3.1. Reagents and Materials
Copper foam (CF, thickness 1.0 mm) was purchased from Jiangsu Province. Ammonium persulfate (AR), sodium sulfate (AR), sodium hydroxide (AR), ammonium sulfate (AR), potassium hydroxide (AR), hydrochloric acid (AR), ethanol (AR) and iridium (III) chloride tetrahydrate (IrCl3·4H2O, AR) were obtained from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). Acetonitrile (HPLC grade) and methanol (HPLC grade) were supplied by Shanghai Aladdin Biochemical Technology Co., Ltd. (Shanghai, China). 5-Hydroxymethylfurfural (HMF, AR) and 2,5-bis(hydroxymethyl)furan (BHMF, AR) were purchased from Shanghai Macklin Biochemical Co., Ltd. (Shanghai, China). Deionized water was produced by a Milli-Q water purification system. All chemicals were used as received without further purification.
3.2. Synthesis of Catalyst
The pretreated clean foam copper (1 cm × 2 cm) was immersed into an 80 mL aqueous solution containing 6.4 g NaOH and 2.0 g (NH4)2S2O8, then reacted at room temperature for 30 min. The resulting sample was washed with water and dried with nitrogen gas to obtain the Cu(OH)2 nanowire precursor. The precursor was heated at 5 °C/min to 200 °C under air atmosphere and calcined for 2 h to convert it into CuO nanowires. Using the CuO nanowires as the working electrode, constant-current reduction was performed at a current density of −40 mA cm−2 in 0.5 M Na2SO4 electrolyte for 30 min, yielding Cu nanowires (Cu NW). Then, the prepared Cu NW were immersed in a 0.3 mM IrCl3·4H2O solution for different durations (10 min, 20 min, 30 min, 40 min, and 50 min) to perform galvanic replacement. This process yielded Ir-decorated copper oxide nanowires with varying Ir contents. The obtained samples were designated as CuIr10, CuIr20, CuIr30, CuIr40, and CuIr50, respectively, where the “CuIr” notation refers to this Ir-decorated copper oxide structure. After the galvanic replacement reaction, the samples were thoroughly washed with deionized water to remove residual solution and then dried under a nitrogen flow.
3.3. Electrochemical Testing and Product Analysis
Electrochemical testing was conducted in a 40 mL H-type electrolytic cell equipped with a cation-exchange membrane. In the experiment, the prepared catalyst, electrolytic platinum electrode, and mercury oxide electrode served as the working electrode, counter electrode, and reference electrode, respectively. The working electrode had a geometric surface area of 1 cm2. The electrolyte consisted of a 0.1 M KOH solution containing 20 mM HMF (pH = 13). All potentials were converted to reversible hydrogen electrode (RHE) potentials. Constant-potential electrolysis was performed using the chronoamperometric method. The reaction solution was filtered through a 0.22 μm filter head and analyzed quantitatively for HMF and BHMF concentrations using high-performance liquid chromatography (HPLC). In addition, The Faradaic efficiency of HMF was calculated using Equation (1). The HMF yield (%) was calculated by Equation (2), and the HMF conversion (%) was calculated using Equation (3).
Here, n represents the molar quantity of HMF after the reaction (mol), F denotes Faraday’s constant (96,485 C mol−1), Q indicates the number of electrons transferred per product molecule, I is the current density (A cm−2), and t is the reaction time measured via the electrochemical workstation (s).
Here, nHMF,0 denotes the molar quantity of HMF prior to the reaction (mol), nHMF represents the molar quantity of HMF after the reaction (mol), and nBHMF indicates the molar quantity of BHMF measured via HPLC after the reaction (mol).
3.4. Cycling Tests
Recyclability tests were performed to evaluate the stability of the catalyst. Under identical electrochemical conditions, the catalyst with the best performance was subjected to repeated HMF hydrogenation cycles. After each reaction, the catalyst was thoroughly washed with deionized water and anhydrous ethanol, then dried under a nitrogen stream. The treated catalyst was reused in the next cycle, and a total of four cycles were carried out. The composition of the reaction solution was analyzed using HPLC.
4. Conclusions
In this study, a series of Ir-decorated copper oxide nanowire electrocatalysts were successfully synthesized for the electrocatalytic hydrogenation of HMF to BHMF. By enhancing the water dissociation capability and constructing a Hads-rich environment, the catalysts exhibited outstanding catalytic performance. Through the rational control of the number of water dissociation active sites, the optimized CuIr catalyst achieved a remarkable HMF conversion of 99% and BHMF yield of 99% at a current density of −20 mA cm−2, significantly outperforming the pristine Cu nanowire catalyst. Comprehensive characterizations revealed that Cu plays a key role in the adsorption and activation of the carbonyl (C=O) group, serving as the main active site for HMF binding. Meanwhile, the incorporation of Ir introduces highly active sites for water dissociation, markedly enhancing the generation of Hads on the catalyst surface. The synergistic effect between Cu and Ir effectively promotes the protonation pathway of the hydrogenation reaction, facilitating the coupling between HMF-derived intermediates and Hads, while suppressing the formation of undesired dimers. In addition, the nanowire morphology strengthens the interaction between HMF molecules and the catalyst surface, contributing to the high catalytic efficiency. The catalyst also demonstrated excellent stability in repeated cycling experiments, with no significant loss in activity. These findings highlight the crucial role of optimized hydrogen activation in the hydrogenation of furanic aldehydes, offering new insights into the design of electrocatalysts with enriched Hads environments. This work provides valuable guidance for the development of high-performance Cu-based electrocatalysts for the efficient hydrogenation of furfural derivatives into furan alcohols.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/catal16020116/s1, Figure S1. The standard curves of HMF (a) and BHMF (b). Figure S2. SEM images of Cu NW (a), CuIr10 (b), CuIr20 (c), CuIr30 (d), CuIr40 (e), and CuIr50 (f). Figure S3. XRD patterns before and after cycles. Figure S4. Magnified view of the low-pressure region of N2 adsorption isotherms. Figure S5. ESR spectra of DMPO-C* (a) and DMPO-H* (b) adducts generated over different catalysts after 15 min of reaction. Figure S6. Integral fitting of ESR spectra of DMPO-C* (a) and DMPO-H* (b) for Cu NW and CuIr40. Figure S7. The hydrogenation path of BHMF prepared by HMF. Table S1. The Ir content on the catalyst surface.
Author Contributions
Methodology, H.L.; Software, C.C. and Y.L.; Validation, C.Y. and Y.L.; Formal analysis, C.Y.; Investigation, C.C. and H.L.; Data curation, H.L.; Writing—original draft, C.C.; Writing—review & editing, Y.C.; Supervision, Y.C. and Y.Z.; Project administration, Y.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financially supported by the Collaborative Innovation Center for Water Treatment Technology and Materials, the Special Fund of Henan Key Laboratory of Water Pollution Control and Rehabilitation Technology (CJSZ2024010).
Data Availability Statement
The data presented in this study are available upon request from the corresponding authors.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Sen, S.; Ganguly, S. Opportunities, barriers and issues with renewable energy development—A discussion. Renew. Sustain. Energy Rev. 2017, 69, 1170–1181. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, Y.; Guan, W.; Cao, Y.; Wang, F.; Zhang, Y. Mechanistic Studies into the Selective Production of 2,5-furandicarboxylic Acid from 2,5-bis(hydroxymethyl)furan Using Au-Pd Bimetallic Catalyst Supported on Nitrated Carbon Material. Catalysts 2023, 13, 435. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, Y.; Xia, Q.; Chen, Y.; Kong, L.; Yan, X.; Guan, W.; Pan, J. Ambient temperature catalyzed air-oxidation of 5-hydroxymethylfurfural via ternary metal and oxygen vacancies. Green Energy Environ. 2025, 10, 1568–1582. [Google Scholar] [CrossRef]
- Zhou, M.; Fakayode, O.A.; Ren, M.; Li, H.; Liang, J.; Zhou, C. Green and sustainable extraction of lignin by deep eutectic solvent, its antioxidant activity, and applications in the food industry. Crit. Rev. Food Sci. Nutr. 2023, 64, 7201–7219. [Google Scholar] [CrossRef]
- Suliman, M.A.; Basheer, C.; Farooq, W. Cobalt Boride/g-C3N4 Nanosheets-Assisted Electrocatalytic Oxidation of 5-Hydroxymethylfurfural into 2,5-Furandicarboxylic Acid. Catalysts 2021, 11, 1241. [Google Scholar] [CrossRef]
- Cai, J.; Xi, S.; Zhang, C.; Li, X.; Helal, M.H.; El-Bahy, Z.M.; Ibrahim, M.M.; Zhu, H.; Singh, M.V.; Wasnik, P.; et al. Overview of biomass valorization: Case study of nanocarbons, biofuels and their derivatives. J. Agric. Food Res. 2023, 14, 100714. [Google Scholar] [CrossRef]
- Wang, T.; Zhang, J.; Xie, W.; Tang, Y.; Guo, D.; Ni, Y. Catalytic Transfer Hydrogenation of Biobased HMF to 2,5-Bis-(Hydroxymethyl)Furan over Ru/Co3O4. Catalysts 2017, 7, 92. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, C.; Yan, X.; Mao, Y.; Cao, Y.; Liu, Y.; Guan, W.; Chen, Y. High-efficient microwave-assisted conversion of fructose to 5-hydroxymethylfurfural over rice straw-derived sulfonated porous carbonaceous catalyst. Fuel 2024, 373, 132348. [Google Scholar] [CrossRef]
- Slak, J.; Pomeroy, B.; Kostyniuk, A.; Grilc, M.; Likozar, B. A review of bio-refining process intensification in catalytic conversion reactions, separations and purifications of hydroxymethylfurfural (HMF) and furfural. Chem. Eng. J. 2022, 429, 132325. [Google Scholar] [CrossRef]
- Long, J.; Zhao, W.; Xu, Y.; Li, H.; Yang, S. Carbonate-Catalyzed Room-Temperature Selective Reduction of Biomass-Derived 5-Hydroxymethylfurfural into 2,5-Bis(hydroxymethyl)furan. Catalysts 2018, 8, 633. [Google Scholar] [CrossRef]
- Zhang, Y.; Cao, Y.; Yan, C.; Liu, W.; Chen, Y.; Guan, W.; Wang, F.; Liu, Y.; Huo, P. Rationally designed Au-ZrOx interaction for boosting 5-hydroxymethylfurfural oxidation. Chem. Eng. J. 2023, 459, 141644. [Google Scholar] [CrossRef]
- Gong, C.; Meng, X.; Jin, C.; Yang, M.; Liu, J.; Sheng, K.; Pu, Y.; Ragauskas, A.; Ji, G.; Zhang, X. Green synthesis of cellulose formate and its efficient conversion into 5-hydroxymethylfurfural. Ind. Crops Prod. 2023, 192, 115985. [Google Scholar] [CrossRef]
- Li, M.; Jiang, H.; Zhang, L.; Yu, X.; Liu, H.; Yagoub, A.E.A.; Zhou, C. Synthesis of 5-HMF from an ultrasound-ionic liquid pretreated sugarcane bagasse by using a microwave-solid acid/ionic liquid system. Ind. Crops Prod. 2020, 149, 112361. [Google Scholar] [CrossRef]
- Zhang, Z.-G.; Shen, X.; Jiang, S.-K.; Lin, J.-C.; Yi, Y.; Ji, X.-J. Biocatalytic Hydrogenation of Biomass-Derived Furan Aldehydes to Alcohols. J. Agric. Food Chem. 2025, 73, 2266–2278. [Google Scholar] [CrossRef]
- Abdul Rani, M.A.A.B.; Karim, N.A.; Kamarudin, S.K. Recent reaction systems for the synthesis of 5-hydroxymethylfurfural (HMF) from carbohydrates with process development analysis: A review. Int. J. Energy Res. 2022, 46, 18996–19050. [Google Scholar] [CrossRef]
- Wang, Y.; Fan, L.; Xiao, L.; Wang, L.; Shen, D.; Long, Y. Role of reaction adsorption on the production of 5-hydroxymethylfurfural from fructose under microwave hydrothermal process. Fuel 2023, 340, 127530. [Google Scholar] [CrossRef]
- Phan, H.B.; Luong, C.M.; Nguyen, L.P.; Bui, B.T.; Nguyen, H.T.; Mai, B.V.; Mai, T.V.T.; Huynh, L.K.; Tran, P.H. Eco-Friendly Synthesis of 5-Hydroxymethylfurfural and Its Applications as a Starting Material to Synthesize Valuable Heterocyclic Compounds. ACS Sustain. Chem. Eng. 2022, 10, 8673–8684. [Google Scholar] [CrossRef]
- Jung, S.; Kim, K.S.; Park, G.H.; Cha, H.G.; Jeong, H.; Kang, M.J. Strategies on utilizing biomass derived 5-hydroxymethylfufural by catalytic reactions: Pathways and mechanisms. Mater. Today Sustain. 2025, 29, 101058. [Google Scholar] [CrossRef]
- Gallezot, P. Conversion of biomass to selected chemical products. Chem. Soc. Rev. 2012, 41, 1538–1558. [Google Scholar] [CrossRef]
- Aswin, P.; Kothari, A.C.; Neethu, P.P.; Bal, R.; Venkatesha, N.J.; Hsu, H.-L.; Ganesh, V.; Sakthivel, A. Hydrogenation of 5-Hydroxymethyl Furfural (HMF) Using Noble Metal-Free Copper-Molybdenum-Based Catalyst. Catal. Lett. 2024, 154, 4906–4917. [Google Scholar] [CrossRef]
- Xu, C.; Paone, E.; Rodríguez-Padrón, D.; Luque, R.; Mauriello, F. Recent catalytic routes for the preparation and the upgrading of biomass derived furfural and 5-hydroxymethylfurfural. Chem. Soc. Rev. 2020, 49, 4273–4306. [Google Scholar] [CrossRef]
- Wang, T.; Huang, Z.; Liu, T.; Tao, L.; Tian, J.; Gu, K.; Wei, X.; Zhou, P.; Gan, L.; Du, S.; et al. Transforming Electrocatalytic Biomass Upgrading and Hydrogen Production from Electricity Input to Electricity Output. Angew. Chem. Int. Ed. 2022, 61, e202115636. [Google Scholar] [CrossRef]
- Tang, C.; Zheng, Y.; Jaroniec, M.; Qiao, S.Z. Electrocatalytic Refinery for Sustainable Production of Fuels and Chemicals. Angew. Chem. Int. Ed. 2021, 60, 19572–19590. [Google Scholar] [CrossRef]
- Zhou, L.; Li, Y.; Lu, Y.; Wang, S.; Zou, Y. pH-Induced selective electrocatalytic hydrogenation of furfural on Cu electrodes. Chin. J. Catal. 2022, 43, 3142–3153. [Google Scholar] [CrossRef]
- Yu, Z.; Lu, X.; Wang, X.; Xiong, J.; Li, X.; Zhang, R.; Ji, N. Metal-Catalyzed Hydrogenation of Biomass-Derived Furfural: Particle Size Effects and Regulation Strategies. ChemSusChem 2020, 13, 5185–5198. [Google Scholar] [CrossRef] [PubMed]
- Ohyama, J.; Hayashi, Y.; Ueda, K.; Yamamoto, Y.; Arai, S.; Satsuma, A. Effect of FeOx Modification of Al2O3 on Its Supported Au Catalyst for Hydrogenation of 5-Hydroxymethylfurfural. J. Phys. Chem. C 2016, 120, 15129–15136. [Google Scholar] [CrossRef]
- Murthy, P.S.; Liang, W.; Jiang, Y.; Huang, J. Cu-Based Nanocatalysts for CO2 Hydrogenation to Methanol. Energy Fuels 2021, 35, 8558–8584. [Google Scholar] [CrossRef]
- Chang, F.; Xiao, M.; Miao, R.; Liu, Y.; Ren, M.; Jia, Z.; Han, D.; Yuan, Y.; Bai, Z.; Yang, L. Copper-Based Catalysts for Electrochemical Carbon Dioxide Reduction to Multicarbon Products. Electrochem. Energy Rev. 2022, 5, 4. [Google Scholar] [CrossRef]
- Zhang, Q.; Tackett, B.M.; Wu, Q.; Chen, J.G. Trends in Hydrogen Evolution Activity of Metal-Modified Molybdenum Carbides in Alkaline and Acid Electrolytes. ChemElectroChem 2016, 3, 1686–1693. [Google Scholar] [CrossRef]
- Xie, Q.; Larrazábal, G.O.; Ma, M.; Chorkendorff, I.; Seger, B.; Luo, J. Copper-indium hydroxides derived electrocatalysts with tunable compositions for electrochemical CO2 reduction. J. Energy Chem. 2021, 63, 278–284. [Google Scholar] [CrossRef]
- Yang, M.; Yuan, Z.; Peng, R.; Wang, S.; Zou, Y. Recent Progress on Electrocatalytic Valorization of Biomass-Derived Organics. Energy Environ. Mater. 2022, 5, 1117–1138. [Google Scholar] [CrossRef]
- Elsayed, I.; Jackson, M.A.; Hassan, E.B. Hydrogen-Free Catalytic Reduction of Biomass-Derived 5-Hydroxymethylfurfural into 2,5-Bis(hydroxymethyl)furan Using Copper–Iron Oxides Bimetallic Nanocatalyst. ACS Sustain. Chem. Eng. 2020, 8, 1774–1785. [Google Scholar] [CrossRef]
- Liu, H.; Yu, J.; Chen, Y.; Lee, J.; Huang, W.; Li, W. Cu-Based Bimetallic Catalysts for Electrocatalytic Oxidative Dehydrogenation of Furfural with Practical Rates. ACS Appl. Mater. Interfaces 2023, 15, 37477–37485. [Google Scholar] [CrossRef]
- Sun, Y.; Miao, J.; Fan, X.; Zhang, K.; Zhang, T. Recent Progress in Electrochemical Conversion from Biomass Derivatives into High-Value-Added Chemicals. Small Struct. 2024, 5, 2300576. [Google Scholar] [CrossRef]
- Viar, N.; Agirre, I.; Gandarias, I. Process design, kinetics, and techno-economic assessment of an integrated liquid phase furfural hydrogenation process. Chem. Eng. J. 2024, 480, 147873. [Google Scholar] [CrossRef]
- Zhang, W.; Qi, Y.; Zhao, Y.; Ge, W.; Dong, L.; Shen, J.; Jiang, H.; Li, C. Rh-dispersed Cu nanowire catalyst for boosting electrocatalytic hydrogenation of 5-hydroxymethylfurfural. Sci. Bull. 2023, 68, 2190–2199. [Google Scholar] [CrossRef]
- Bender, M.T.; Yuan, X.; Goetz, M.K.; Choi, K.-S. Electrochemical Hydrogenation, Hydrogenolysis, and Dehydrogenation for Reductive and Oxidative Biomass Upgrading Using 5-Hydroxymethylfurfural as a Model System. ACS Catal. 2022, 12, 12349–12368. [Google Scholar] [CrossRef]
- Chadderdon, X.H.; Chadderdon, D.J.; Matthiesen, J.E.; Qiu, Y.; Carraher, J.M.; Tessonnier, J.-P.; Li, W. Mechanisms of Furfural Reduction on Metal Electrodes: Distinguishing Pathways for Selective Hydrogenation of Bioderived Oxygenates. J. Am. Chem. Soc. 2017, 139, 14120–14128. [Google Scholar] [CrossRef] [PubMed]
- Moyo, P.S.; Mehlana, G.; Matsinha, L.C.; Makhubela, B.C.E. Copper-Based Metal–Organic Framework: Synthesis, Characterization and Evaluation for the Hydrogenation of Furfural to Furfuryl Alcohol. J. Inorg. Organomet. Polym. Mater. 2024, 35, 2257–2273. [Google Scholar] [CrossRef]
- Li, R.; Liu, W.; Liu, H.; Niu, H.; Zhou, Q.; Liang, C.; Chen, X. Dual Active Sites of Ni and FeNi3 Constructed from Layered Double Hydroxides for One-Pot Hydrogenation of Furfural to Tetrahydrofurfuryl Alcohol. Ind. Eng. Chem. Res. 2023, 62, 21631–21643. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, Y.; Gu, B.; Tang, Q.; Cao, Q.-E.; Fang, W. Regulating the Interaction within Pd-Cu Dual Metal Sites for Selective Hydrogenation of Furfural Using Ambient H2 Pressure. ACS Sustain. Chem. Eng. 2023, 11, 12798–12808. [Google Scholar] [CrossRef]
- Ratrey, G.; Solanki, B.S.; Kamble, S.P.; Rode, C.V. Highly Efficient Chemoselective Hydrogenation of 5-HMF to BHMF over Reusable Bimetallic Pd-Ir/C Catalyst. ChemistrySelect 2022, 7, e202200456. [Google Scholar] [CrossRef]
- Hu, J.; Liu, Y.; Zhou, Y.; Zhao, H.; Xu, Z.; Li, H. Recent advances in rhenium-based nanostructures for enhanced electrocatalysis. Appl. Catal. A Gen. 2023, 663, 119304. [Google Scholar] [CrossRef]
- Yu, P.; Wang, F.; Shifa, T.A.; Zhan, X.; Lou, X.; Xia, F.; He, J. Earth abundant materials beyond transition metal dichalcogenides: A focus on electrocatalyzing hydrogen evolution reaction. Nano Energy 2019, 58, 244–276. [Google Scholar] [CrossRef]
- Luo, J.; Liang, C. Rhenium in Heterogeneous Catalysis: A Rising Star for Hydrogenation Reactions. ACS Catal. 2024, 14, 7032–7049. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, B.; Wei, Z.; Zhou, W.; Wang, D.; Tian, J.; Wang, T.; Zhao, S.; Liu, J.; Tao, L.; et al. Coupling Glucose-Assisted Cu(I)/Cu(II) Redox with Electrochemical Hydrogen Production. Adv. Mater. 2021, 33, 2104791. [Google Scholar] [CrossRef]
- Zhu, B.; Yang, J.; Wang, Q.; Yu, X.; Fan, S.; Xie, W.; Zhang, J.; Chen, C. Corrosion-induced CoCu microwire arrays for efficient electroreduction of 5-hydroxymethylfurfural. Chem Catal. 2025, 5, 101259. [Google Scholar] [CrossRef]
- Sanghez de Luna, G.; Ho, P.H.; Lolli, A.; Ospitali, F.; Albonetti, S.; Fornasari, G.; Benito, P. Ag Electrodeposited on Cu Open-Cell Foams for the Selective Electroreduction of 5-Hydroxymethylfurfural. ChemElectroChem 2020, 7, 1238–1247. [Google Scholar] [CrossRef]
- Wang, L.; Jin, H.; Liu, Z.; Yang, S.; He, G.; Liu, H.; Meng, X.; Xu, C. Investigation on the preparation of 5-Hydroxymethylfurfural through fructose dehydration using in-line FTIR and in-situ 13C NMR. J. Catal. 2024, 432, 115450. [Google Scholar] [CrossRef]
- Gao, Y.; Tang, C.; Zheng, Y. Manipulating adsorbed hydrogen for enhanced HMF electrocatalytic hydrogenation. J. Energy Chem. 2025, 105, 439–445. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.