Abstract
C2-symmetric diimine generated in situ from (R,R)-HPEN and 8-formylquinoline was readily transformed to novel chiral diimine pre-III bearing 1(S),2(S)-bis(quinolin-8-yl) ethane and salicylic aldehyde fragments via stereoselective diaza-Cope rearrangement. The structure and absolute configuration of the product was established by the X-ray diffraction study of the corresponding Rh(III) complex. In the presence of pre-III, practically important enantioselective reactions of 4-hydroxycoumarin with α,β-enones afforded anticoagulant (S)-warfarin and its derivatives in aqueous THF in high yield (88–92%) with enantioselectivity 85–93% ee.
1. Introduction
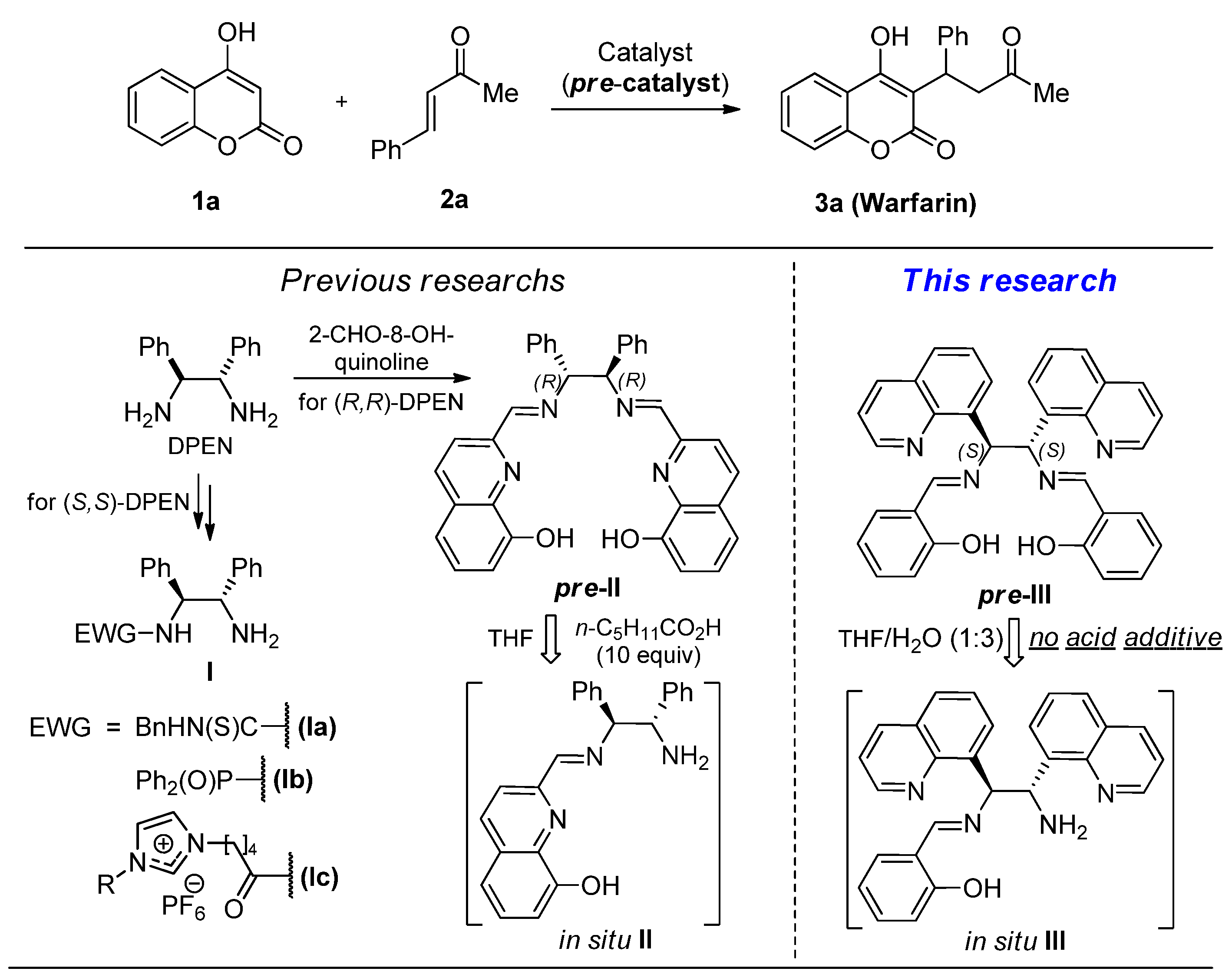
Asymmetric reactions of β-dicarbonyls 1 with α,β-enones 2 are widely used for stereoselective synthesis of biologically active compounds [1,2,3]. In particular, warfarin (3a), an efficient chiral anticoagulant [4], can be readily prepared by this research strategy [5]. The (S)-enantiomer of this medication is 4–5 times more active than the (R)-enantiomer [6]. Enantioselective warfarin synthesis requires the use of enzymes [7], chiral metal complexes [8], or organocatalysts [9] as sources of chirality. Enzymatic catalysis commonly results in the formation of less active (R)-warfarin enantiomer with a low yield [10]. Metal complexes are inconvenient in handling due to their instability towards moisture and air [11]. Organocatalysts are usually more practical. Moreover, they do not contaminate pharmaceutical products by toxic heavy metals [12,13]. Among efficient organocatalysts for enantioselective warfarin synthesis are chiral primary amines Ia–c, which can be prepared from available enantiomerically pure C2-symmetric 1,2-diphenylethane diamines (DPEN) [14,15,16,17,18] (Figure 1). However, selective functionalization of just one of the two amino groups in DPEN requires auxiliary protection/deprotection steps, which complicate the synthesis of compounds Ia–c.
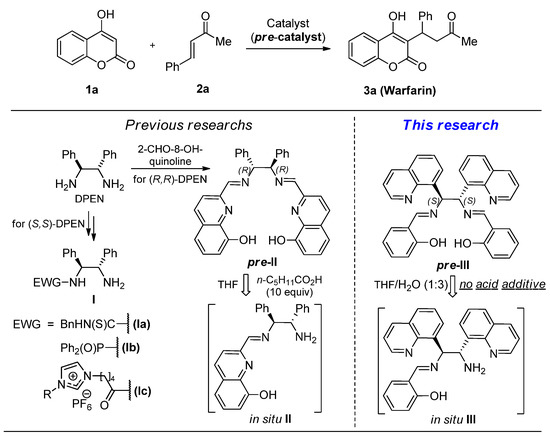
Figure 1.
Research strategy for asymmetric synthesis of warfarin.
Cheng with co-workers nicely contributed to this challenge [18]. They generated primary amine II from diimine precursor pre-II (10 mol.%) under the action of n-hexanoic acid (10 equiv.) in the presence of starting substrates 1 and 2. As a result, (R)-warfarin was formed over the one-pot procedure in 96% yield with 91% ee. Yet, the expensiveness of 2-formyl-8-hydroxyquinoline (>EUR 100 per gram) used for the preparation of diimine pre-II, and the tedious separation of product 3 from n-hexanoic acid are major shortcomings of this approach.
We hypothesized that the asymmetric synthesis of warfarin and its analogues in the presence of pre-catalyst pre-III, an isomer of compound pre-II, generated in situ from more available starting compounds, would be superior in terms of practicality. Furthermore, we expected that the substitution of n-hexanoic acid with water, an easily removable natural hydrolyzing agent, would make the one-pot procedure more convenient and environmentally friendly.
2. Results and Discussion
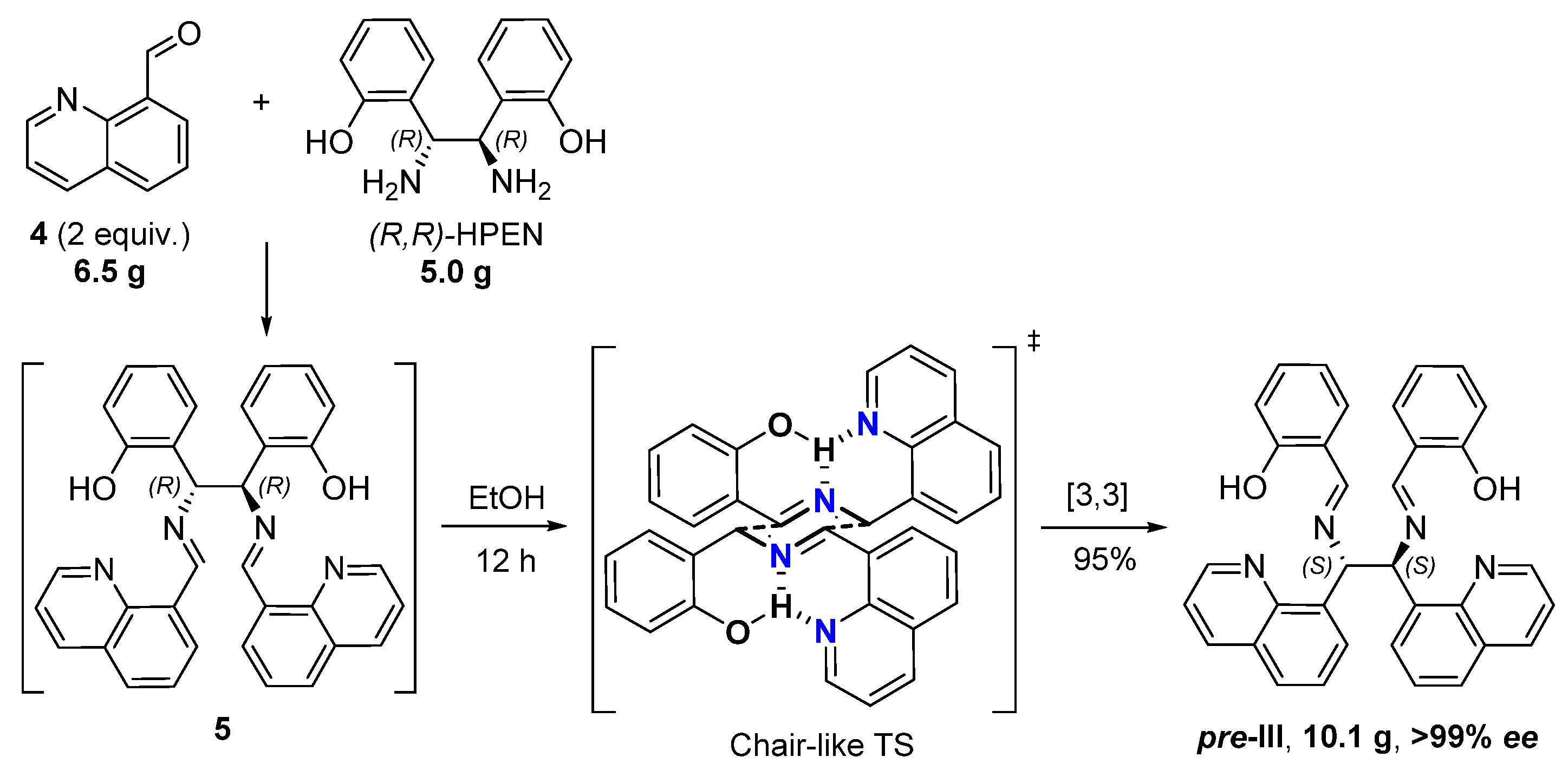
We synthesized novel C2-symmetric diimine pre-III by the asymmetric reaction of 8-quinoline carbaldehyde (4) (2 equiv.) with commercially available (1R,2R)-bis(ortho-hydroxyphenyl)ethane diamine ((R,R)-HPEN) accompanied by stereoselective diaza-Cope rearrangement [19,20,21] of the initially generated diimine 5 (Figure 2). The domino reaction was carried out in EtOH at ambient temperature over 12 h. Diastereo- and enantiomerically pure product pre-III was precipitated from the reaction mixture (yield 95%) and did not require further purification. The excellent stereoselectivity may be attributed to the formation of chair-like six-membered transition state (TS) with favorable pseudo-equatorial orientation of substituents stabilized by hydrogen bonds [22]. The reaction is readily scalable and allows practical asymmetric synthesis of more than 10 g of pre-III over a single experimental operation.
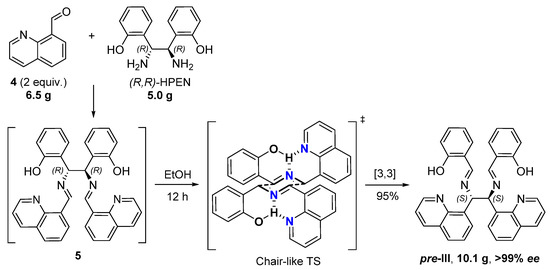
Figure 2.
Stereoselective synthesis of pre-III from 8-quinolinecarbaldehyde (4) and (R,R)-HPEN via the diaza-Cope rearrangement.
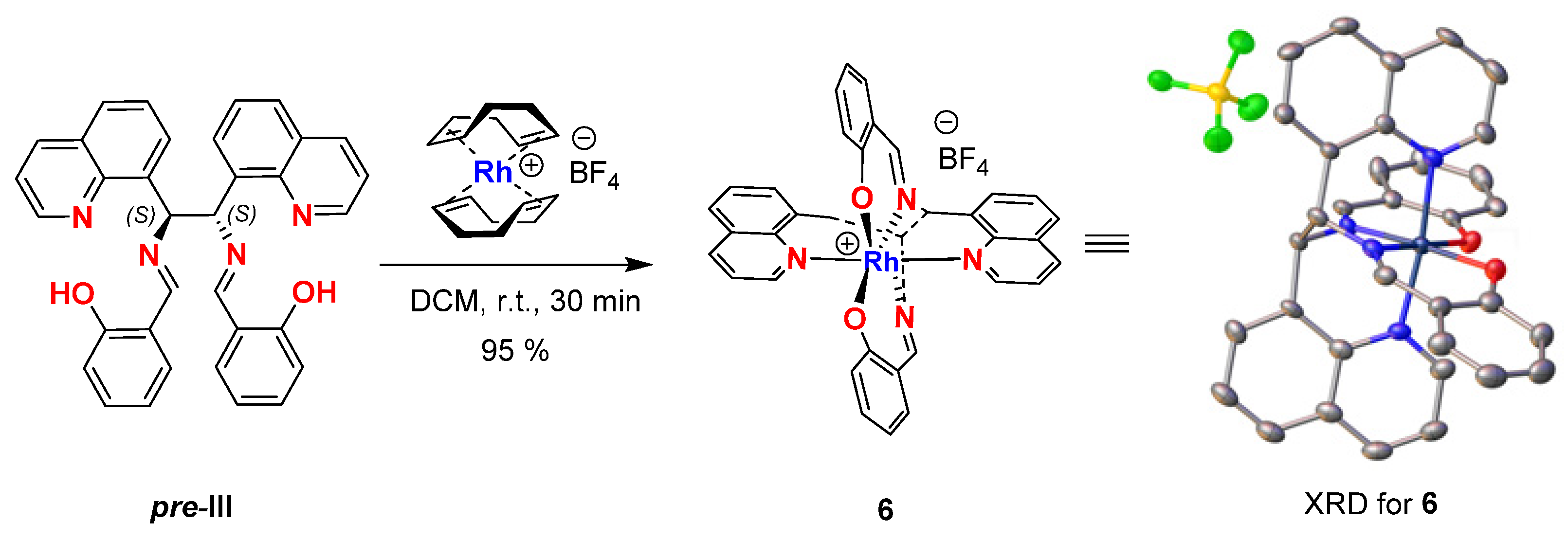
To determine the structure and absolute configuration of pre-III, we synthesized complex 6 of this ligand with Rh[(COD)2]BF4 by slow evaporation of CHCl3 from the binary solution of both components in this solvent (Figure 3). According to the X-ray diffraction (XRD) study, the obtained crystals of 6 had an octahedral configuration with a Rh atom coordinated with four nitrogen and two oxygen atoms at the apexes of the octahedron. The XRD data proved the (S)-configuration of both stereogenic centers in 6 (for details see SI), the assignment being highly reliable and taking into account the presence of Rh heavy metal in the complex [23].
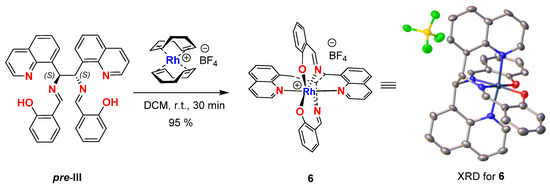
Figure 3.
The synthesis and crystal structure obtained from the XRD analysis of Rh(III) complex 6.
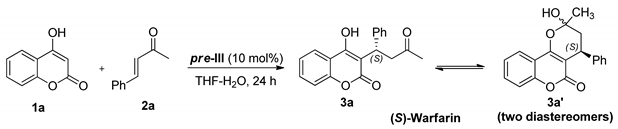
Then we examined the asymmetric Michael reaction of 4-hydroxycoumarin (1a) with benzylideneacetone (2a), in the presence of pre-III (10 mol%). The reaction was tested in different solvents at ambient temperature for 24 h. The reactants were unchanged in protic solvents (THF, CH2Cl2, toluene) (Table 1, entry 1). However, in the presence of n-hexanoic acid (10 equiv.), the desirable (S)-warfarin (3a) was formed with a 76% yield with 87% ee (entry 2), comparable with results attained in the presence of pre-II [18]. We succeeded in significantly improving the reaction outcome by carrying it out in aqueous THF even without an acidic additive (entries 3–5). The best yield (88%) and enantiomeric purity of product 3a (86% ee) were attained at 3:1 THF/H2O ratio (entry 3). The warfarin yield was nearly the same (85%) in pure water, but the enantioselectivity was noticeably lower in this case (74% ee) (entry 6).

Table 1.
Optimization of the reaction conditions a.
High-resolution mass spectrometry (HRMS) data testify to the presence (see Supplementary Materials) in aqueous reaction mixtures of amino-imide III (m/z 419.1872), a product of partial hydrolysis of diimide pre-III, which obviously acts as a true catalyst in the asymmetric Michael reaction (see Figure 1). A favorable impact of water on various asymmetric organocatalytic transformations has been reported [24,25,26,27,28]. However, the role of water in these reactions mainly consisted of the inhibition of side processes, which push the organocatalyst out of the catalytic cycle [29,30] or in the creation of heterogeneous conditions favorable for catalytic transformations on a boundary between organic and aqueous phases (“on water” reactions) [31,32,33]. To our knowledge, this research provided the first example of the generation of an active organocatalyst from its latent precursor in situ under the action of water.
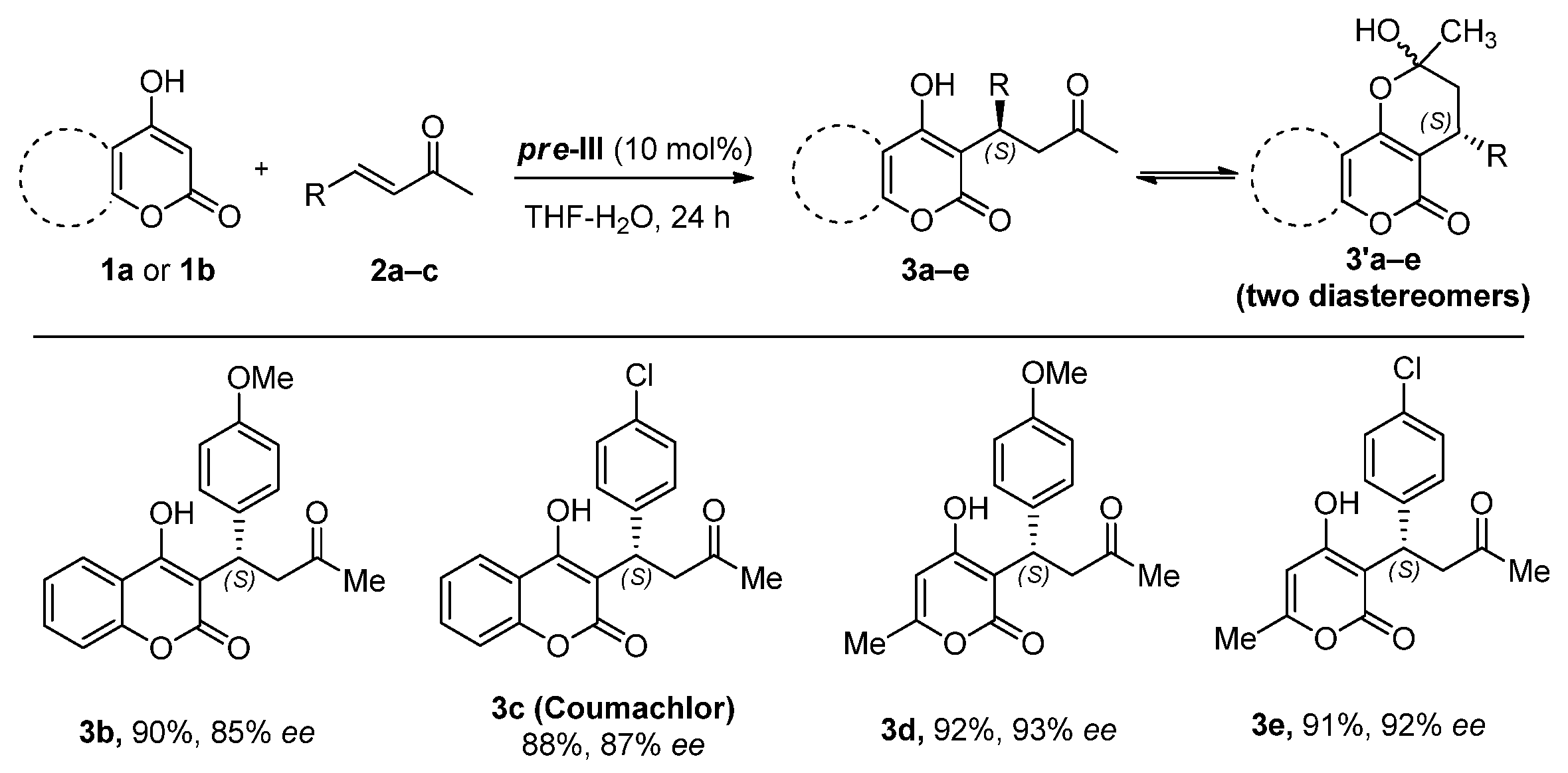
Benzylideneacetone derivatives 2b and 2c bearing donor (OMe) or acceptor (Cl) groups in the aromatic ring reacted with 4-hydroxycoumarin (1a) under optimal conditions (Figure 4). 6-Methyl-4-hydroxypyranone (1b), a mononuclear analogue of 1a, was also identified as a suitable nucleophilic component. In all cases, corresponding adducts 3a–e were formed as dynamic equilibrium mixtures of linear and cyclic tautomers in nearly quantitative yields with enantioselectivity up to 93% ee. The absolute S configuration was assigned to products 3b–e by analogy with warfarin 3a.
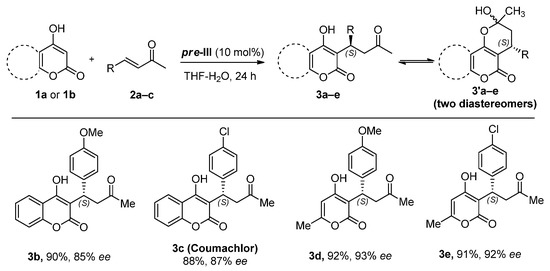
Figure 4.
The reaction scope.
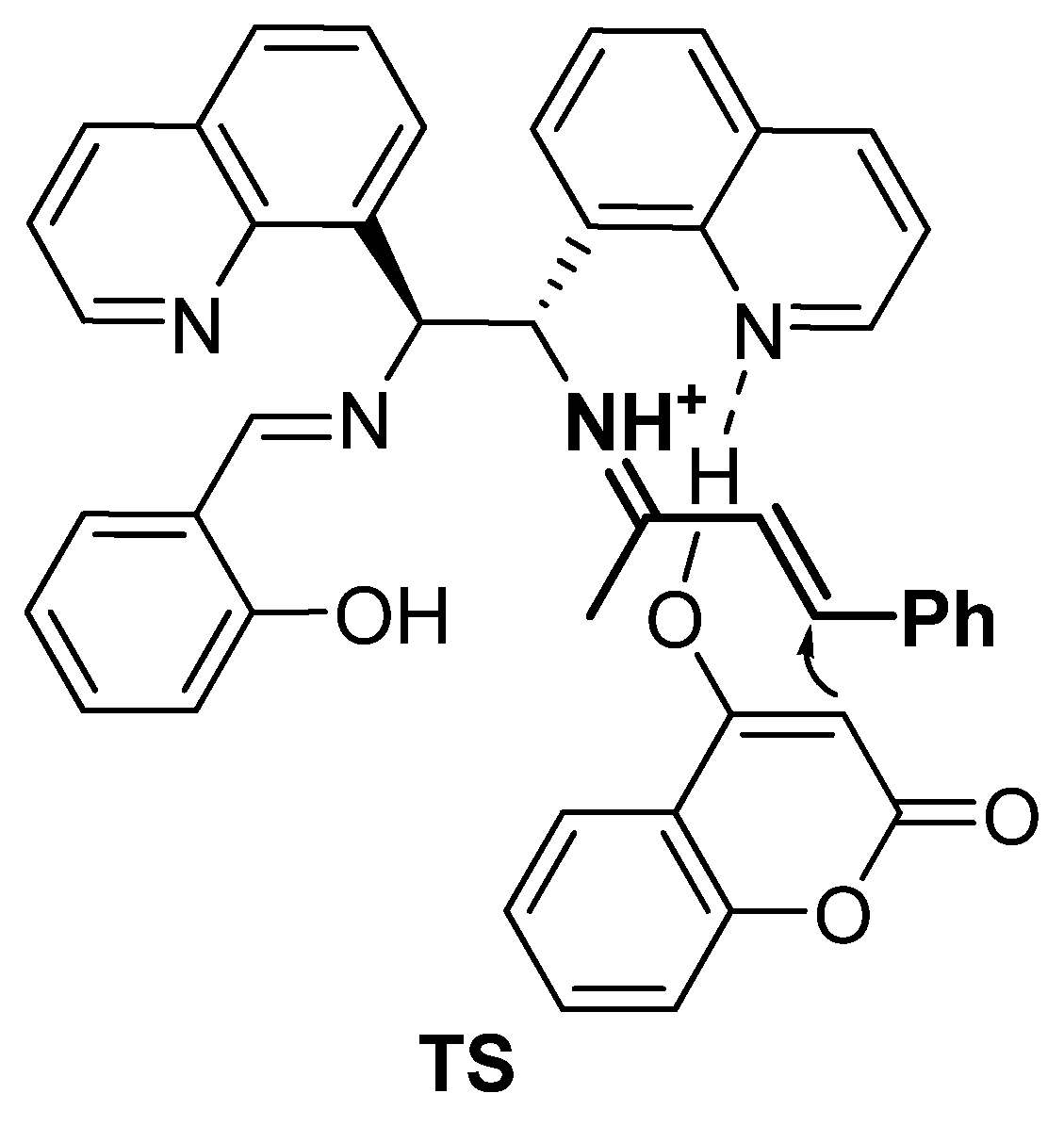
Most likely, the asymmetric Michael reaction includes classical activation of α,β-enone 2 via the formation of an iminium ion with a primary amine group of catalyst III, generated in situ (Figure 5). The subsequent addition of 4-hydroxycoumarin to the iminium ion from the reface through a plausible transition state, TS, affords (S)-warfarin.
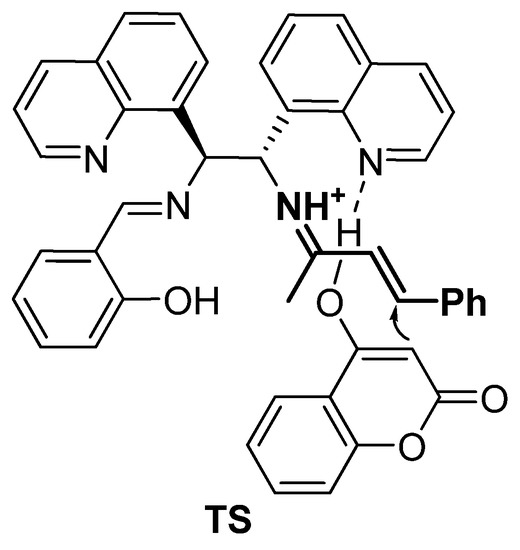
Figure 5.
Plausible transition state (TS).
3. Materials and Methods
The 1H and 13C NMR spectra were recorded on a 300 MHz spectrometer (Avance-300, Bruker, Billerica, MA, USA). The high-resolution mass spectra (HRMSAgilent Technologies, Santa Clara, CA, USA) were measured using electrospray ionization (ESI) and a time-of-flight (TOF) mass analyzer. The measurements were taken in the positive ion mode (interface capillary voltage 4500 V) in the mass range from m/z = 50 Da to m/z = 3000 Da; external and internal calibrations were conducted with the electrospray calibrant solution. Optical rotations were measured on a polarimeter JASCO P-2000 (JASCO corporation, Tokyo, Japan) and calibrated with a pure solvent as a blank. HPLC analyses were performed on an HPLC system (Akvilon, Moscow, Russia) equipped with chiral stationary phase column (AD-H, AS-H, OJ-H, OD-H), detection at 220 or 254 nm. Starting compounds 1 and 2 are commercially available. Reagents and solvents were purified by standard methods. Solvents were removed from reaction mixtures by means of vacuum (10 Torr, 40 L min−1).
3.1. Synthesis of pre-III
8-Quiniolinecarboxaldehyde (6.50 g, 41.0 mmol) was added in one portion to a solution of (R,R)-HPEN (5.00 g, 20.5 mmol) in abs. EtOH (50 mL) and the resulting mixture was stirred at ambient temperature for 12 h. The yellow precipitate was filtered, washed with cold EtOH (2 × 25 mL), and dried in vacuo (10 Torr), to afford pre-III as yellow powder, yield 10.1 g (95%), m.p. = 197–200 °C. 1H NMR (300 MHz, CD2Cl2) δ 13.81 (s, 2H), 8.96 (s, 2H), 8.52 (s, 2H), 8.00–7.98 (m, 4H), 7.52–7.50 (m, 2H), 7.17–7.10 (m, 6H), 6.95 (m, 2H), 6.90 (s, 2H), 6.88 (s, 2H), 6.79–6.74 (t, 2H) ppm. 13C NMR (75 MHz, CD2Cl2,) δ 166.4, 161.3, 149.5, 145.8, 137.8, 136.1, 132.2, 131.6, 129.1, 128.1, 127.2, 126.0, 121.0, 118.9, 118.4, 116.7, 70.4 ppm. HPLC data: >99% ee (Chiralpak AD-H, n-hexane: i-PrOH = 85:15, 220 nm; flow rate: 1.0 mL/min; tminor = 12.1 min, tmajor = 14.9 min). HRMS (ESI): m/z [M + H]+ calcd for [C34H27N4O2]+: 523.2129, found 523.2124.
3.2. Synthesis of Rh(III) Complex 6
[Rh(COD)2]BF4 (1.51 g, 3.83 mmol) was added to a solution of pre-III (2.00 g, 3.83 mmol) in dry CH2Cl2 (10 mL) and the resulting mixture was stirred for 3 h. The resulting light orange precipitate was filtered, washed with cold CH2Cl2 (2 × 5 mL), and dried in vacuo (10 Torr) to afford Rh(III) complex 6 as light orange solid, yield 2.59 g (95%), m.p. > 200 °C (decomp.). 1H NMR (300 MHz, CD2Cl2,) δ 9.27–9.25 (d, J = 4.9 Hz, 2H), 8.57–8.54 (d, J = 7.1 Hz, 2H), 8.42–8.39 (d, J = 8.3 Hz, 2H), 8.04–8.01 (d, J = 8.1 Hz, 2H), 7.95 (s, 2H), 7.91–7.86 (t, J = 7.7 Hz, 2H), 7.56–7.52 (dd, J = 7.9, 5.3 Hz, 2H), 7.34–7.25 (m, 4H), 6.90–6.87 (d, J = 7.8 Hz, 2H), 6.44–6.39 (t, J = 7.0 Hz, 2H), 6.00 (s, 2H) ppm. 13C NMR (75 MHz, CD2Cl2) δ 167.0, 163.0, 153.6, 143.4, 141.2, 135.9, 135.1, 134.0, 132.4, 131.3, 130.6, 128.6, 128.1, 122.6, 121.8, 118.6, 118.5, 115.4, 78.7 ppm. HRMS (ESI): m/z [M + H]+ calcd for C34H25N4O2RhBF4 712.9236, found 712.9239.
3.3. Gram-Scale Synthesis of (S)-Warfarin 3a
Pre-III (1.61 g, 3.09 mmol) was added to a solution of 1a (5.00 g, 30.9 mmol) and 2a (4.96 g, 34 mmol) in the H2O-THF solvent system (1:3, v/v, 15 mL). The reaction mixture was stirred at ambient temperature for 24 h. The solvents were evaporated reduced pressure (10 Torr), water (15 mL) was added to the residue, thus obtained precipitate was filtered and dried in vacuo (10 Torr) to afford 3a (warfarin) as colorless solid, yield 8.40 g (88%), m.p. = 156–159 °C. 1H NMR (300 MHz, CDCl3) δ 9.67 (s, 0.16H), 7.96–7.74 (m, 1.42H), 7.59–7.39 (m, 1.67H), 7.39–7.13 (m, 8.47H), 4.77–4.74 (d, J = 10.1 Hz, 0.16H), 4.30–4.13 (m, 1.23H), 3.90–3.78 (m, 0.36H), 3.37–3.30 (d, 0.19H), 2.53–2.35 (m, 1.50H), 2.29 (s, 0.32H) 2.07–1.95 (m, 0.74), 1.69–1.67 (m, 3H) ppm. [α]D25 = −21.7 (c 1, CH2Cl2). HPLC data: 86% ee (Chiralpak AD-H, 70:30 (n-hexane: i-PrOH), 254 nm, flow rate 1.00 mL/min, tminor = 4.0 min, tmajor = 7.8 min). All characterization data in accordance with literature [34].
3.4. Synthesis of Compounds 3b–e
General procedure:Pre-III (0.05 mmol, 26 mg) was added to a solution of 1a–b (0.50 mmol) and 2b–c (0.55 mmol) in the water-THF solvent system (1:3, v/v, 500 μL). The reaction mixture was stirred at ambient temperature for 24 h. The solvents were evaporated in vacuo (10 Torr), water (2 mL) was added to the residue, thus obtained precipitate was filtered and dried in vacuo (10 Torr) to afford analytically pure adducts 3b–e.
3.4.1. (S)-4-hydroxy-3-(1-(4-methoxyphenyl)-3-oxobutyl)-2H-chromen-2-one (3b)
Colorless solid, yield 152 mg (90%), m.p. = 165–167 °C. 1H NMR (300 MHz, CDCl3) δ 7.92–7.89 (d, J = 7.6 Hz, 0.61H), 7.83–7.81 (d, J = 6.2 Hz, 0.58H), 7.59–7.47 (m, 1.78H), 7.36–7.11 (m, 5.53H), 6.95–6.92 (d, J = 8.7 Hz, 0.41H), 6.88–6.83 (t, J = 7.2 Hz, 2.65H), 6.65–6.59 (d, J = 16.3 Hz, 0.18H), 4.25–4.15 (m, 1.17H), 3.86 (s, 0.51H), 3.78 (s, 4.06H), 2.55–2.34 (m, 2.25H), 2.04–1.97 (m, 0.66H), 1.71–1.68 (m, 3H) ppm. HPLC data: 85% ee (Chiralpak AS-H, 70:30 (n-hexane: i-PrOH), 254 nm, flow rate 1.00 mL/min, tminor = 13.8 min, tmajor = 17.1 min). All characterization data in accordance with literature [35].
3.4.2. (S)-3-(1-(4-chlorophenyl)-3-oxobutyl)-4-hydroxy-2H-chromen-2-one (3c) (Coumachlor)
Colorless solid, yield 151 mg (88%), m.p. = 175–176 °C. 1H NMR (300 MHz, CDCl3) δ 9.72 (0.18H), 7.90–7.81 (m, 1H), 7.59–7.44 (m, 1.29H), 7.37–7.14 (m, 6.64), 4.68 (d, J = 8.4 Hz, 0.16H), 4.38 (s, 0.41H), 4.16 (m, 1.10H), 3.83 (m, 0.55H), 3.32–3.26 (m, 0.20H), 2.48–2.35 (m, 1.28H), 2.28 (s, 0.31H), 2.05–1.89 (m, 1.25H), 1.72 (s, 1.43H), 1.69 (s, 0.98H) ppm. HPLC data: 87% ee (Chiralpak AS-H, 85:15 (n-hexane: i-PrOH), 254 nm, flow rate 1.00 mL/min, tmajor = 21.8 min, tminor = 26.0 min,). All characterization data in accordance with literature [36].
3.4.3. (S)-4-hydroxy-3-(1-(4-methoxyphenyl)-3-oxobutyl)-6-methyl-2H-pyran-2-one (3d)
Yellowish oil, yield 139 mg (92%). 1H NMR (300 MHz, CDCl3) δ 9.42 (s, 0.22H), 7.34–7.28 (m, 0.64H), 7.16–7.08 (m, 1.53H), 6.86–6.80 (m, 1.96H), 5.88 (s, 0.,33H), 5.79 (s, 0.36H), 5.68 (s, 0.24H), 4.66–4.62 (q, J = 8.8, 5.4 Hz, 0.25H), 4.08–4.05 (m, 0.35H), 4.01–3.95 (m, 0.42H), 3.77 (s, 3H), 3.68–3.59 (m, 0.38H), 3.24–3.22 (d, J = 5.5 Hz, 0.14H), 3.18–3.17 (d, J = 5.1 Hz, 0.11H), 2.44–2.43 (d, J = 3.2 Hz, 0.13H), 2.39–2.27 (m, 2.15H), 2.20–2.19 (m, 1.84H), 2.07 (s, 1.02H), 1.91–1.83 (m, 0.49H), 1.55 (s, 2.14H) ppm. HPLC data: 93% ee (Chiralpak OJ-H, 90:10 (n-hexane: i-PrOH), 254 nm, flow rate 1.00 mL/min, tminor = 25.1 min, tmajor = 32.9 min). All characterization data in accordance with literature [37].
3.4.4. (S)-3-(1-(4-chlorophenyl)-3-oxobutyl)-4-hydroxy-6-methyl-2H-pyran-2-one (3e)
Yellowish oil, yield 139 mg (91%). 1H NMR (300 MHz, CDCl3) δ 9.59 (s, 0.35H), 7.35–7.32 (d, J = 8.9 Hz, 1.07H), 7.28–7.23 (m, 3.1H), 7.15–7.09 (t, J = 12.1 Hz, 2.24H), 5.87 (s, 0.41H), 5.81 (s, 0.61H), 5.66 (s, 0.38H), 4.68–4.64 (q, J = 2.1, 8.0 Hz, 0.38H), 4.02–3.96 (m, 1.09H), 3.88 (s, 0.49H), 3.64–3.55 (m, 0.46H), 3.38 (s, 0.34H), 3.26–3.24 (d, J = 5.4Hz, 0.23H), 3.20–3.18 (d, J = 5.7 Hz, 0.17H), 2.35–2.08 (m, 5.98H), 2.08 (s, 0.96H), 1.85–1.76 (t, J = 12.0 Hz, 0.81H); 1.57–1.55 (m, 3H). HPLC data: 92% ee (Chiralpak OD-H, 85:15 (n-hexane: i-PrOH), 254 nm, flow rate 1.00 mL/min, tminor = 13.6 min, tmajor = 16.2 min). All characterization data in accordance with literature [37].
3.5. X-ray Crystallography
X-ray diffraction data were collected at 100 K on a four-circle Rigaku Synergy S diffractometer (Tokyo, Japan) equipped with a HyPix600HE area-detector (kappa geometry, shutterless ω-scan technique), using graphite monochromatized CuKα radiation. The intensity data were integrated and corrected for absorption and decay by the CrysAlisPro v42 program. The structure was solved by direct methods using SHELXT2 and refined on F2 using SHELXL-20183 in the OLEX2-1.5 program. All non-hydrogen atoms were refined with individual anisotropic displacement parameters. All hydrogen atoms were placed in ideal calculated positions and refined as riding atoms with relative isotropic displacement parameters.
General Procedure for Crystal Growing
To the clear solution of 6 (250 mg, 0.35 mmol) in MeOH (2 mL), the heptane (1 mL) was added dropwise. The obtained solution was allowed to warm to 50 °C, and then to freeze to −5 °C. Then the obtained orange crystals were collected and dried under vacuum (10 torr) for 1 h.
4. Conclusions
In summary, a novel C2-symmetric diamine, a latent form of an efficient organocatalyst for the asymmetric synthesis of anticoagulant warfarin and its analogues, was prepared from commercially available reagents via diaza-Cope rearrangement. The pre-catalyst structure and absolute configuration of the stereogenic centers were established by the X-ray diffraction study of its Rh(III) complex. A simple and environment friendly one-pot catalyst generation/catalytic transformation procedure based on performing the pre-III-induced asymmetric reaction of 4-hydroxycoumarin or 4-hydroxypyranone with α,β-enones in aqueous tetrahydrofuran medium was developed.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/catal13010136/s1. Figure S1. HRMS data for reaction mixture; Figure S2. 1H, 13C NMR pictures for all compounds; Figure S3. HPLC Data; Figure S4. X-Ray data for Rh (III)-complex 6; Table S1. Crystal data and structure refinement for 6.
Author Contributions
Investigation, I.D.N.; resources, O.Y.K.; data curation, R.A.K.; supervision, A.S.K. and S.G.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
Not applicable.
Conflicts of Interest
There are no conflicts to declare.
References
- Han, B.; He, X.H.; Liu, Y.Q.; He, G.; Peng, C.; Li, J.L. Asymmetric organocatalysis: An enabling technology for medicinal chemistry. Chem. Soc. Rev. 2021, 50, 1522–1586. [Google Scholar] [CrossRef] [PubMed]
- Mondal, A.; Bhowmick, S.; Ghosh, A.; Chanda, T.; Bhowmick, K.C. Advances on asymmetric organocatalytic 1, 4-conjugate addition reactions in aqueous and semi-aqueous media. Tetrahedron Asymmetry 2017, 28, 849–875. [Google Scholar] [CrossRef]
- Chopade, M.U.; Chopade, A.U.; Momin, A.A. Asymmetric michael adducts as key building blocks for synthesis of bioactive molecules. MOJ Biorg. Org. Chem. 2017, 1, 148–151. [Google Scholar] [CrossRef]
- Hirsh, J.; Fuster, V.; Ansell, J.; Halperin, J.L. American Heart Association/American College of Cardiology foundation guide to warfarin therapy. J. Am. Coll. Cardiol. 2003, 41, 1633–1652. [Google Scholar] [CrossRef] [PubMed]
- Marcia de Figueiredo, R.; Christmann, M. Organocatalytic Synthesis of Drugs and Bioactive Natural Products. EurJOC 2007, 16, 2575–2600. [Google Scholar] [CrossRef]
- Park, B.K. Warfarin: Metabolism and mode of action. Biochem. Pharmacol. 1988, 37, 19–27. [Google Scholar] [CrossRef]
- Sano, K.; Saito, S.; Hirose, Y.; Kohari, Y.; Nakano, H.; Seki, C.; Tokiwa, M.; Takeshita, M.; Uwai, K. Development of a novel method for warfarin synthesis via lipase-catalyzed steroselective Michael reaction. Heterocycles 2013, 87, 1269–1278. [Google Scholar]
- Yang, H.M.; Gao, Y.H.; Li, L.; Jiang, Z.Y.; Lai, G.Q.; Xia, C.G.; Xu, L.W. Iron-catalyzed Michael reactions revisited: A synthetically useful process for the preparation of tri-carbonyl compounds and chiral warfarin. Tetrahedron Lett. 2010, 51, 3836–3839. [Google Scholar] [CrossRef]
- Dong, Z.; Wang, L.; Chen, X.; Liu, X.; Lin, L.; Feng, X. Organocatalytic enantioselective Michael addition of 4-hydroxycoumarin to α,β-unsaturated ketones: A simple synthesis of warfarin. EurJOC 2009, 2009, 5192–5197. [Google Scholar] [CrossRef]
- Xie, B.H.; Guan, Z.; He, Y.H. Promiscuous enzyme-catalyzed Michael addition: Synthesis of warfarin and derivatives. J. Chem. Technol. Biotechnol. 2012, 87, 1709–1714. [Google Scholar] [CrossRef]
- Tsuchiya, Y.; Hamashima, Y.; Sodeoka, M. A new entry to Pd− H chemistry: Catalytic asymmetric conjugate reduction of enones with EtOH and a highly enantioselective synthesis of warfarin. Org. Lett. 2006, 8, 4851–4854. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, V.D.G.; Cardoso, M.F.D.C.; Forezi, L.D.S.M. Organocatalysis: A brief overview on its evolution and applications. Catalysts 2018, 8, 605. [Google Scholar] [CrossRef]
- Krištofíková, D.; Modrocká, V.; Mečiarová, M.; Šebesta, R. Green asymmetric organocatalysis. ChemSusChem 2020, 13, 2828–2858. [Google Scholar] [CrossRef] [PubMed]
- Mei, R.Q.; Xu, X.Y.; Li, Y.C.; Fu, J.Y.; Huang, Q.C.; Wang, L.X. Highly effective and enantioselective Michael addition of 4-hydroxycoumarin to α, β-unsaturated ketones promoted by simple chiral primary amine thiourea bifunctional catalysts. Tetrahedron Lett. 2011, 52, 1566–1568. [Google Scholar] [CrossRef]
- Dong, J.; Du, D.M. Highly enantioselective synthesis of Warfarin and its analogs catalysed by primary amine–phosphinamide bifunctional catalysts. Org. Biomol. Chem. 2012, 10, 8125–8131. [Google Scholar] [CrossRef]
- Kucherenko, A.S.; Siyutkin, D.E.; Nigmatov, A.G.; Chizhov, A.O.; Zlotin, S.G. Chiral Primary Amine Tagged to Ionic Group as Reusable Organocatalyst for Asymmetric Michael Reactions of C-Nucleophiles with α, β-Unsaturated Ketones. Adv. Synth. Catal. 2012, 354, 3078. [Google Scholar] [CrossRef]
- Kucherenko, A.S.; Lisnyak, V.G.; Chizhov, A.O.; Zlotin, S.G. Primary Amine Attached to an N-(Carboxyalkyl) imidazolium Cation: A Recyclable Organocatalyst for the Asymmetric Michael Reaction. Eur. J. Org. Chem. 2014, 18, 3808–3813. [Google Scholar] [CrossRef]
- Zhu, X.; Lin, A.; Shi, Y.; Guo, J.; Zhu, C.; Cheng, Y. Enantioselective synthesis of polycyclic coumarin derivatives catalyzed by an in situ formed primary amine-imine catalyst. Org. Lett. 2011, 13, 4382–4385. [Google Scholar] [CrossRef]
- Kim, H.; Nguyen, Y.; Lough, A.J.; Chin, J. Stereospecific Diaza-Cope Rearrangement Driven by Steric Strain. Angew. Chem. 2008, 120, 8806–8809. [Google Scholar] [CrossRef]
- Zhu, Q.; Huang, H.; Shi, D.; Shen, Z.; Xia, C. An Efficient Synthesis of Chiral Diamines with Rigid Backbones: Application in Enantioselective Michael Addition of Malonates to Nitroalkenes. Org. Lett. 2009, 11, 4536–4539. [Google Scholar] [CrossRef]
- So, S.M.; Mui, L.; Kim, H.; Chin, J. Understanding the Interplay of Weak Forces in [3,3]-Sigmatropic Rearrangement for Stereospecific Synthesis of Diamines. Acc. Chem. Res. 2012, 45, 1345–1355. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Kim, H.; Kim, H.; Chin, J. Synthesis of Enantiopure Mixed Alkyl-Aryl Vicinal Diamines by the Diaza-Cope Rearrangement: A Synthesis of (+)-CP-99,994. J. Org. Chem. 2017, 82, 12050–12058. [Google Scholar] [CrossRef] [PubMed]
- Saito, F.; Schreiner, P.R. Determination of the absolute configurations of chiral alkanes–an analysis of the available tools. Eur. J. Org. Chem. 2020, 40, 6328–6339. [Google Scholar] [CrossRef]
- Jimeno, C. Water in asymmetric organocatalytic systems: A global perspective. Org. Biomol. Chem. 2016, 14, 6147–6164. [Google Scholar] [CrossRef] [PubMed]
- Mlynarski, J.; Baś, S. Catalytic asymmetric aldol reactions in aqueous media—A 5 year update. Chem. Soc. Rev. 2014, 43, 577–587. [Google Scholar] [CrossRef]
- Bhowmick, S.; Mondal, A.; Ghosh, A.; Bhowmick, K.C. Water: The most versatile and nature’s friendly media in asymmetric organocatalyzed direct aldol reactions. Tetrahedron Asymmetry 2015, 26, 1215–1244. [Google Scholar] [CrossRef]
- Bhowmick, S.; Bhowmick, K.C. Catalytic asymmetric carbon–carbon bond-forming reactions in aqueous media. Tetrahedron Asymmetry 2011, 22, 1945–1979. [Google Scholar] [CrossRef]
- Raj, M.; Singh, V.K. Organocatalytic reactions in water. Chem. Commun. 2009, 44, 6687–6703. [Google Scholar] [CrossRef]
- Blackmond, D.G.; Armstrong, A.; Coombe, V.; Wells, A. Water in organocatalytic processes: Debunking the myths. Angew. Chem. Int. Ed. 2007, 46, 3798–3800. [Google Scholar] [CrossRef]
- Nyberg, A.I.; Usano, A.; Pihko, P.M. Proline-catalyzed ketone-aldehyde aldol reactions are accelerated by water. Synlett 2004, 11, 1891–1896. [Google Scholar] [CrossRef]
- Guillena, G.; Alonso, D.; Baeza, A.; Chinchilla, R.; Flores-Ferrándiz, J.; Gómez-Martínez, M.; Trillo, P. Pursuing chemical efficiency by using supported organocatalysts for asymmetric reactions under aqueous conditions. Curr. Organocatal. 2015, 2, 102–123. [Google Scholar] [CrossRef]
- Mase, N.; Barbas III, C.F. In water, on water, and by water: Mimicking nature’s aldolases with organocatalysis and water. Org. Biomol. Chem. 2010, 8, 4043–4050. [Google Scholar] [CrossRef] [PubMed]
- Gruttadauria, M.; Giacalone, F.; Noto, R. Water in stereoselective organocatalytic reactions. Adv. Synth. Catal. 2009, 351, 33–57. [Google Scholar] [CrossRef]
- Kucherenko, A.S.; Kostenko, A.A.; Zhdankina, G.M.; Kuznetsova, O.Y.; Zlotin, S.G. Green asymmetric synthesis of Warfarin and Coumachlor in pure water catalyzed by quinoline-derived 1,2-diamines. Green Chem. 2018, 20, 754–759. [Google Scholar] [CrossRef]
- Xie, J.W.; Yue, L.; Chen, W.; Du, W.; Zhu, J.; Deng, J.G.; Chen, Y.C. Highly enantioselective Michael addition of cyclic 1, 3-dicarbonyl compounds to α, β-unsaturated ketones. Org. Lett. 2007, 9, 413–415. [Google Scholar] [CrossRef] [PubMed]
- Shi, T.; Guo, Z.; Yu, H.; Xie, J.; Zhong, Y.; Zhu, W. Atom-economic synthesis of optically active Warfarin anticoagulant over a chiral MOF organocatalyst. Adv. Synth. Catal. 2013, 355, 2538–2543. [Google Scholar] [CrossRef]
- Kochetkov, S.V.; Kucherenko, A.S.; Zlotin, S.G. Asymmetric synthesis of warfarin and its analogs catalyzed by C2-symmetric squaramide-based primary diamines. Org. Biomol. Chem. 2018, 16, 6423–6429. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).