Cetuximab for Immunotherapy-Refractory/Ineligible Cutaneous Squamous Cell Carcinoma
Abstract
Simple Summary
Abstract
1. Introduction
2. Patients and Methods
3. Results
3.1. Patients
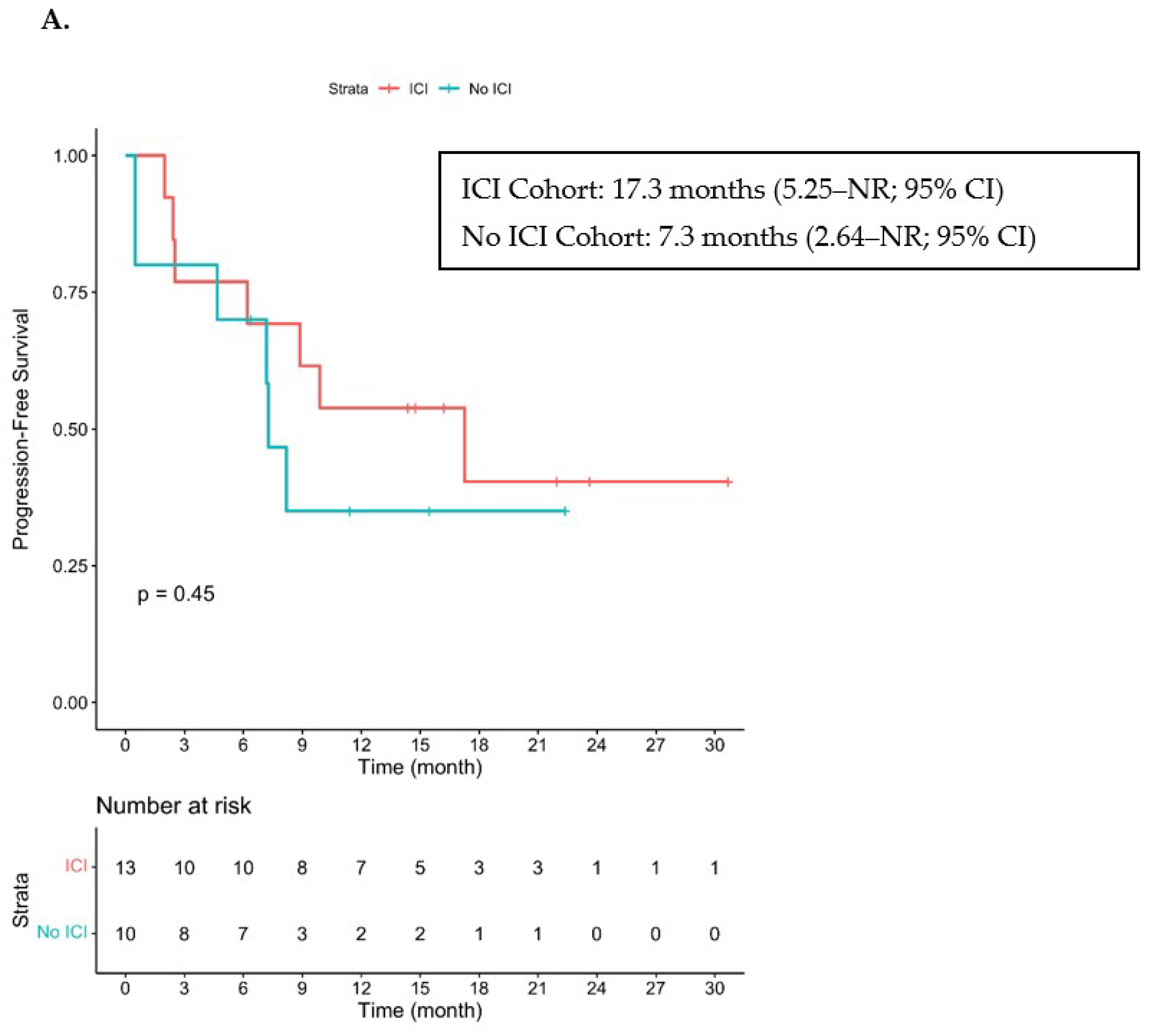
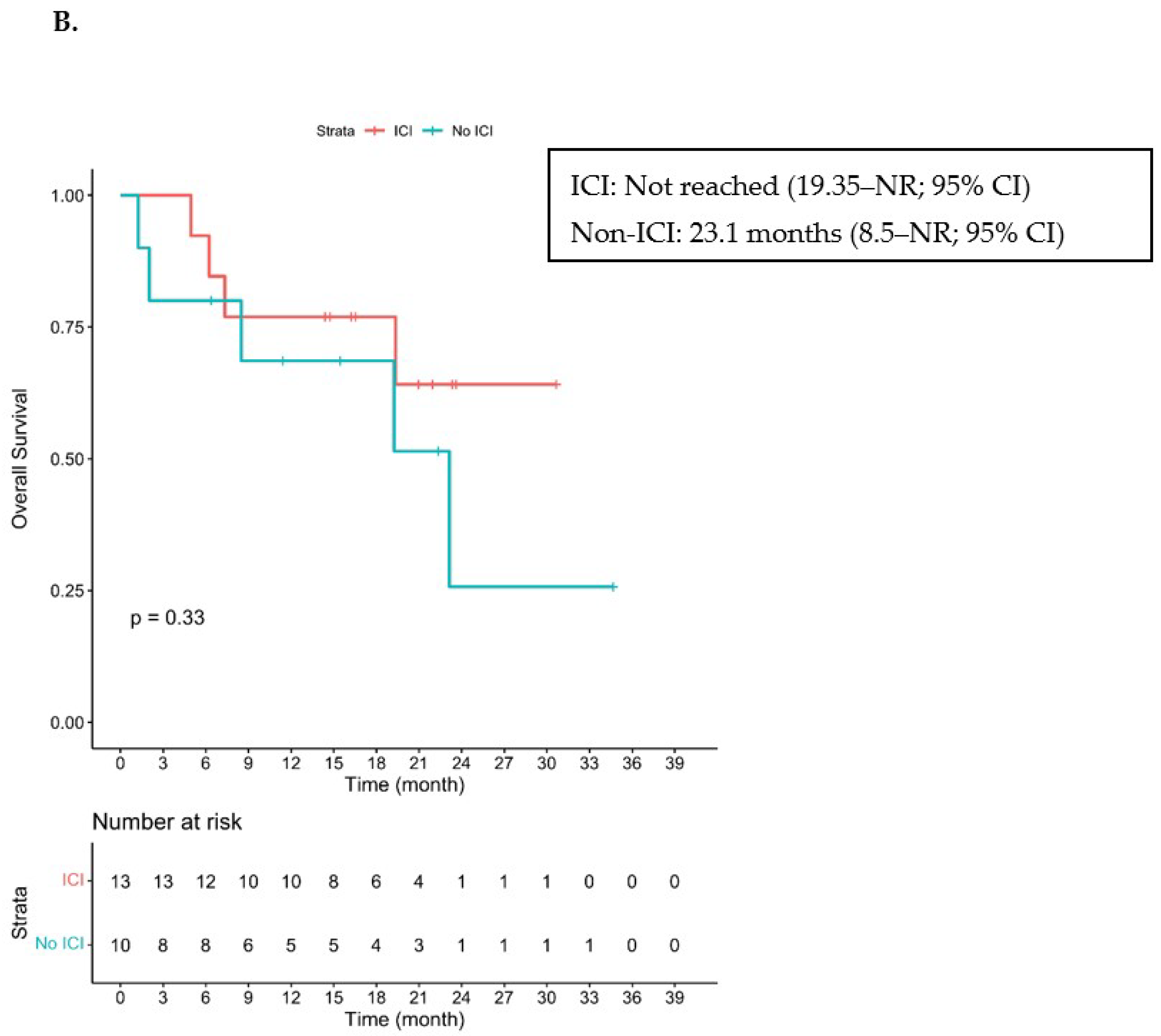
3.2. Efficacy
3.3. Toxicities
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rogers, H.W.; Weinstock, M.A.; Feldman, S.R.; Coldiron, B.M. Incidence Estimate of Nonmelanoma Skin Cancer (Keratinocyte carcinomas) in the U.S. Population, 2012. JAMA Dermatol. 2015, 151, 1081–1086. [Google Scholar] [CrossRef] [PubMed]
- Burton, K.A.; Ashack, K.A.; Khachemoune, A. Cutaneous Squamous Cell Carcinoma: A Review of High-Risk and Metastatic Disease. Am. J. Clin. Dermatol. 2016, 17, 491–508. [Google Scholar] [CrossRef]
- Karia, P.S.; Han, J.; Schmults, C.D. Cutaneous squamous cell carcinoma: Estimated incidence of disease, nodal metastasis, and deaths from disease in the United States, 2012. J. Am. Acad. Dermatol. 2013, 68, 957–966. [Google Scholar] [CrossRef] [PubMed]
- Schmults, C.D.; Karia, P.S.; Carter, J.B.; Han, J.; Qureshi, A.A. Factors predictive of recurrence and death from cutaneous squamous cell carcinoma: A 10-year, single-institution cohort study. JAMA Dermatol. 2013, 149, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Brantsch, K.D.; Meisner, C.; Schonfisch, B.; Trilling, B.; Wehner-Caroli, J.; Rocken, M.; Breuninger, H. Analysis of risk factors determining prognosis of cutaneous squamous-cell carcinoma: A prospective study. Lancet Oncol. 2008, 9, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Guthrie, T.H., Jr.; Porubsky, E.S.; Luxenberg, M.N.; Shah, K.J.; Wurtz, K.L.; Watson, P.R. Cisplatin-based chemotherapy in advanced basal and squamous cell carcinomas of the skin: Results in 28 patients including 13 patients receiving multimodality therapy. J. Clin. Oncol. 1990, 8, 342–346. [Google Scholar] [CrossRef]
- Ikegawa, S.; Saida, T.; Obayashi, H.; Sasaki, A.; Esumi, H.; Ikeda, S.; Kiyohara, Y.; Hayasaka, K.; Ishihara, K. Cisplatin combination chemotherapy in squamous cell carcinoma and adenoid cystic carcinoma of the skin. J. Dermatol. 1989, 16, 227–230. [Google Scholar] [CrossRef]
- Khansur, T.; Kennedy, A. Cisplatin and 5-fluorouracil for advanced locoregional and metastatic squamous cell carcinoma of the skin. Cancer 1991, 67, 2030–2032. [Google Scholar] [CrossRef]
- Sadek, H.; Azli, N.; Wendling, J.L.; Cvitkovic, E.; Rahal, M.; Mamelle, G.; Guillaume, J.C.; Armand, J.P.; Avril, M.F. Treatment of advanced squamous cell carcinoma of the skin with cisplatin, 5-fluorouracil, and bleomycin. Cancer 1990, 66, 1692–1696. [Google Scholar] [CrossRef]
- Shin, D.M.; Glisson, B.S.; Khuri, F.R.; Clifford, J.L.; Clayman, G.; Benner, S.E.; Forastiere, A.A.; Ginsberg, L.; Liu, D.; Lee, J.J.; et al. Phase II and biologic study of interferon alfa, retinoic acid, and cisplatin in advanced squamous skin cancer. J. Clin. Oncol. 2002, 20, 364–370. [Google Scholar] [CrossRef]
- Maubec, E.; Duvillard, P.; Velasco, V.; Crickx, B.; Avril, M.F. Immunohistochemical analysis of EGFR and HER-2 in patients with metastatic squamous cell carcinoma of the skin. Anticancer Res. 2005, 25, 1205–1210. [Google Scholar]
- Bauman, J.E.; Eaton, K.D.; Martins, R.G. Treatment of recurrent squamous cell carcinoma of the skin with cetuximab. Arch. Derm. 2007, 143, 889–892. [Google Scholar] [CrossRef] [PubMed]
- Maubec, E.; Petrow, P.; Scheer-Senyarich, I.; Duvillard, P.; Lacroix, L.; Gelly, J.; Certain, A.; Duval, X.; Crickx, B.; Buffard, V.; et al. Phase II study of cetuximab as first-line single-drug therapy in patients with unresectable squamous cell carcinoma of the skin. J. Clin. Oncol. 2011, 29, 3419–3426. [Google Scholar] [CrossRef] [PubMed]
- Montaudie, H.; Viotti, J.; Combemale, P.; Dutriaux, C.; Dupin, N.; Robert, C.; Mortier, L.; Kaphan, R.; Duval-Modeste, A.B.; Dalle, S.; et al. Cetuximab is efficient and safe in patients with advanced cutaneous squamous cell carcinoma: A retrospective, multicentre study. Oncotarget 2020, 11, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Migden, M.R.; Rischin, D.; Schmults, C.D.; Guminski, A.; Hauschild, A.; Lewis, K.D.; Chung, C.H.; Hernandez-Aya, L.; Lim, A.M.; Chang, A.L.S.; et al. PD-1 Blockade with Cemiplimab in Advanced Cutaneous Squamous-Cell Carcinoma. N. Engl. J. Med. 2018, 379, 341–351. [Google Scholar] [CrossRef]
- Migden, M.R.; Khushalani, N.I.; Chang, A.L.S.; Lewis, K.D.; Schmults, C.D.; Hernandez-Aya, L.; Meier, F.; Schadendorf, D.; Guminski, A.; Hauschild, A.; et al. Cemiplimab in locally advanced cutaneous squamous cell carcinoma: Results from an open-label, phase 2, single-arm trial. Lancet Oncol. 2020, 21, 294–305. [Google Scholar] [CrossRef]
- Lowe, M.; Yushak, M.L.; Olson, B.; Mokhtari, A.; Harutyunyan, A.; Delman, K.; Parker, D.; Evans, E.; Fisher, T.; Lesinski, G.; et al. 792MO Neoadjuvant pepinemab in combination with nivolumab and/or ipilimumab in resectable stage III melanoma. Ann Oncol. 2022, 33, S906. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration; Center for Drug Evaluation and Research. FDA Approves Cemiplimab-Rwlc for Metastatic or Locally Advanced Cutaneous Squamous Cell Carcinoma. 28 September 2018. Available online: https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-cemiplimab-rwlc-metastatic-or-locally-advanced-cutaneous-squamous-cell-carcinoma (accessed on 15 June 2021).
- Hughes, B.G.M.; Munoz-Couselo, E.; Mortier, L.; Bratland, A.; Gutzmer, R.; Roshdy, O.; Gonzalez Mendoza, R.; Schachter, J.; Arance, A.; Grange, F.; et al. Pembrolizumab for locally advanced and recurrent/metastatic cutaneous squamous cell carcinoma (KEYNOTE-629 study): An open-label, nonrandomized, multicenter, phase II trial. Ann. Oncol. 2021, 32, 1276–1285. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration; Center for Drug Evaluation and Research. FDA Approves Pembrolizumab for Cutaneous Squamous Cell Carcinoma. 24 June 2020. Available online: https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-pembrolizumab-cutaneous-squamous-cell-carcinoma (accessed on 15 June 2021).
- Garcia-Sancha, N.; Corchado-Cobos, R.; Bellido-Hernandez, L.; Roman-Curto, C.; Cardenoso-Alvarez, E.; Perez-Losada, J.; Orfao, A.; Canueto, J. Overcoming Resistance to Immunotherapy in Advanced Cutaneous Squamous Cell Carcinoma. Cancers 2021, 13, 5134. [Google Scholar] [CrossRef]
- Hanna, G.J.; Ruiz, E.S.; LeBoeuf, N.R.; Thakuria, M.; Schmults, C.D.; Decaprio, J.A.; Silk, A.W. Real-world outcomes treating patients with advanced cutaneous squamous cell carcinoma with immune checkpoint inhibitors (CPI). Br. J. Cancer 2020, 123, 1535–1542. [Google Scholar] [CrossRef]
- Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0. Available online: https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcae_v5_quick_reference_5x7.pdf (accessed on 15 May 2021).
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef] [PubMed]
- Mo, J.; Bohne, A.S.; Gaudy Marqueste, C.; Della Marta, N.; Festino, L.; Chen, C.; Lo, S.N.; Park, J.J.; Ascierto, P.A.; Ladwa, R.; et al. 838P Patterns of response/progressive disease (PD) and management following PD with anti-PD-1 (PD1) in patients (pts) with advanced cutaneous squamous cell carcinoma (cSCC). Ann. Oncol. 2022, 33, S932–S933. [Google Scholar] [CrossRef]
- (FDA), F.a.D.A. Full Prescribing Information. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/125084s273lbl.pdf (accessed on 6 February 2023).
- Cowey, C.L.; Robert, N.J.; Espirito, J.L.; Davies, K.; Frytak, J.; Lowy, I.; Fury, M.G. Clinical outcomes among unresectable, locally advanced and metastatic cutaneous squamous cell carcinoma patients treated with systemic therapy. Cancer Med. 2020, 9, 7381–7387. [Google Scholar] [CrossRef] [PubMed]
- Bossi, P.; Alberti, A.; Bergamini, C.; Resteghini, C.; Locati, L.D.; Alfieri, S.; Cavalieri, S.; Colombo, E.; Gurizzan, C.; Lorini, L.; et al. Immunotherapy followed by cetuximab in locally advanced/metastatic (LA/M) cutaneous squamous cell carcinomas (cSCC): The I-TACKLE trial. J. Clin. Oncol. 2022, 40, 9520. [Google Scholar] [CrossRef]
- He, X.; Xu, C. Immune checkpoint signaling and cancer immunotherapy. Cell Res. 2020, 30, 660–669. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.; Wei, T.; Meng, H.; Luo, P.; Zhang, J. Role of the dynamic tumor microenvironment in controversies regarding immune checkpoint inhibitors for the treatment of non-small cell lung cancer (NSCLC) with EGFR mutations. Mol. Cancer 2019, 18, 139. [Google Scholar] [CrossRef]
- Atkins, M.B.; Lee, S.J.; Chmielowski, B.; Tarhini, A.A.; Cohen, G.I.; Truong, T.G.; Moon, H.H.; Davar, D.; O’Rourke, M.; Stephenson, J.J.; et al. Combination Dabrafenib and Trametinib Versus Combination Nivolumab and Ipilimumab for Patients With Advanced BRAF-Mutant Melanoma: The DREAMseq Trial-ECOG-ACRIN EA6134. J. Clin. Oncol. 2023, 41, 186–197. [Google Scholar] [CrossRef] [PubMed]

| All Patients (N = 23) | Cohorts A and B (N = 13) | Cohort A (N = 11) | Cohort B (N = 2) | Cohort C (N = 10) | |
|---|---|---|---|---|---|
| Age | 68 {49–89} | 72 {54–89} | 72 {54–89} | 71.5 {65–77} | 67.5 {49–75} |
| Gender | |||||
| Males | 15 (65.2%) | 11 (84.6%) | 10 (90.9%) | 1 (50%) | 4 (40%) |
| Females | 8 (34.8%) | 2 (15.4%) | 1 (9.1%) | 1 (50%) | 6 (60%) |
| Race | |||||
| White | 23 (100%) | 13 (100%) | 11 (100%) | 2 (100%) | 10 (100%) |
| Comorbidities on Systemic Immunosuppression or Lymphodepleting Therapy | 9 (39.1%) | 1 (7.7%) | - | 1 (50%) | 8 (80%) |
| Secondary Cancer | 1 (4.3%) | 1 (7.7%)—CLL | - | 1 (50%)—CLL | - |
| Autoimmunity | 3 (13%) | - | - | - | 3 (30%) |
| Kidney Transplant | 3 (13%) | - | - | - | 3 (30%) |
| Heart Transplant | 1 (4.3%) | - | - | - | 1 (10%) |
| Liver Transplant | 1 (4.3%) | - | - | - | 1 (10%) |
| Primary Site | |||||
| Scalp | 2 (8.7%) | 2 (15.4%) | 2 (18.2%) | - | - |
| Face | 10 (43.5%) | 3 (23.1%) | 3 (27.3%) | - | 7 (70%) |
| Trunk | 3 (13%) | 2 (15.4%) | 1 (9.1%) | 1 (50%) | 1 (10%) |
| Upper Extremity | 3 (13%) | 3 (23.1%) | 2 (18.2%) | 1 (50%) | - |
| Lower Extremity | 5 (21.7%) | 3 (23.1%) | 3 (27.3%) | - | 2 (20%) |
| Disease Stage at Time of Therapy | |||||
| Locally Advanced | 9 (39.1%) | 3 (23.1%) | 3 (27.3%) | - | 6 (60%) |
| Metastatic | 14 (60.9%) | 10 (76.9%) | 8 (72.3%) | 2 (100%) | 4 (40%) |
| Line Cetuximab Use | |||||
| First | 9 (39.1%) | - | - | - | 9 (90%) |
| Second or More | 14 (60.9%) | 13 (100%) | 11 (100%) | 2 (100%) | 1 (10%) |
| Concurrent Radiation | 9 (39.1%) | 6 (46.2%) | 6 (54.5%) | - | 3 (30%) |
| Cohort | Patient | Location of Radiation | Dosing and Fractions | Radiation Goal | Measurable Distant Disease | Response to Cetuximab | Progression after Cetuximab |
|---|---|---|---|---|---|---|---|
| A (n = 6/11, 54.5%) | 1 | Primary Tumor | 66 Gy, 33 fractions | Definitive | Yes: Distal Lymph Nodes | PR | No |
| 2 | Regional Lymph Node | 30 Gy, 10 fractions | Palliative | Yes: Distal Lymph Nodes | PR | No | |
| 3 | Regional Lymph Node | 54 Gy, 18 fractions | Palliative | Yes: Distal Lymph Nodes and Bone | SD | Yes | |
| 4 | Primary Tumor + Regional Lymph Node | 70 Gy, 35 fractions | Definitive | No | SD | No | |
| 5 | Metastatic Cutaneous Lesion | 40 Gy, 10 fractions | Palliative | Yes: Distal Lymph Nodes | PR | Yes | |
| 6 | Primary Tumor | 66 Gy, 33 fractions | Definitive | Yes: Pulmonary Metastasis | PR | No | |
| C (n = 3/10, 30%) | 1 | Primary Tumor | 70 Gy, 35 fractions | Definitive | No | PR | No |
| 2 | Regional Lymph Nodes | 30 Gy, 15 fractions | Palliative | No | PR | Yes | |
| 3 | Regional Lymph Nodes | 70 Gy, 35 fractions | Definitive | No | PR | Yes |
| Associated Condition (n = 10) | Immunosuppressive Regimens | Concurrent Radiation | Best Response | Progression after Cetuximab |
|---|---|---|---|---|
| Kidney Transplantation (n = 3) | Cyclosporine + prednisone (n = 1) | No | CR | No * |
| Tacrolimus (n = 1) | No | PD | N/A | |
| Tacrolimus + mycophenolate (n = 1) | No | PD | N/A | |
| Liver Transplantation (n = 1) | Tacrolimus + mycophenolate | Yes | PR | No |
| Heart Transplantation (n = 1) | Tacrolimus + prednisone | Yes | PR | Yes |
| Multiple Sclerosis (n = 1) | Fingolimod | No | PR | No * |
| Wegener’s Granulomatosis (n = 1) | Prednisone | No | PR | Yes |
| Severe Lichen Planus (n = 1) | Beclomethasone + triamcinolone | No | PR | No * |
| Sjögren’s Syndrome (n = 1) | None (n = 1) | No | PR | No ** |
| None (n = 1) | None (n = 1) | Yes | PR | Yes *** |
| Response | All Patients (N = 23) | Cohorts A/B (N = 13) | Cohort A (N = 11) | Cohort B (N = 2) | Cohort C (N = 10) |
|---|---|---|---|---|---|
| ORR | 65.2% (15) | 53.9% (7) | 63.6% (7) | 0% (0) | 80% (8) |
| DCR | 78.2% (18) | 77% (10) | 90.9% (10) | 0% (0) | 80% (8) |
| CR | 8.7% (2) | 7.7% (1) | 9.1% (1) | - | 10% (1) |
| PR | 56.5% (13) | 46.2% (6) | 54.5% (6) * | - | 70% (7) **** |
| SD | 13% (3) | 23.1% (3) | 27.3% (3) ** | - | - |
| PD | 21.7% (5) | 23.1% (3) | 9.1% (1) | 100% (2) *** | 20% (2) |
| Toxicities | All Patients (N = 23) | Cohort A (N = 11) | Cohort B (N = 2) | Cohort C (N = 10) |
|---|---|---|---|---|
| All | 22 (95.7%) | 11 (100%) | 2 (100%) | 9 (90%) |
| Rash | 6 (26.1%) | 4 (36.3%) | 1 (50%) | 1 (10%) |
| Grade 1 | 1 (4.3%) | 1 (9.1%) | 1 (50%) | 0 (0%) |
| Grade 2 | 5 (21.7%) | 4 (36.3%) | 0 (0%) | 1 (10%) ** |
| Hypomagnesemia | 4 (17.4%) | 1 (9.1%) | 0 (0%) | 3 (30%) |
| Grade 1 | 3 (13%) | 1 (9.1%) | 0 (0%) | 2 (20%) |
| Grade 2 | 1 (4.3%) | 0 (0%) | 0 (0%) | 1 (10%) ** |
| Hypomagnesemia with Rash | 10 (43.5%) | 5 (45.5%) | 1 (50%) | 4 (40%) |
| Grade 1 | 3 (13%) | 2 (18.2%) | 0 (0%) | 1 (10%) |
| Grade 2 | 7 (30.4%) | 3 (27.3%) | 1 (50%) | 3 (30%) **, Δ |
| Other (all Grade 5) | 1 (4.3%) * | 0 (0%) | 0 (0%) | 1 (10%) *, ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marin-Acevedo, J.A.; Withycombe, B.M.; Kim, Y.; Brohl, A.S.; Eroglu, Z.; Markowitz, J.; Tarhini, A.A.; Tsai, K.Y.; Khushalani, N.I. Cetuximab for Immunotherapy-Refractory/Ineligible Cutaneous Squamous Cell Carcinoma. Cancers 2023, 15, 3180. https://doi.org/10.3390/cancers15123180
Marin-Acevedo JA, Withycombe BM, Kim Y, Brohl AS, Eroglu Z, Markowitz J, Tarhini AA, Tsai KY, Khushalani NI. Cetuximab for Immunotherapy-Refractory/Ineligible Cutaneous Squamous Cell Carcinoma. Cancers. 2023; 15(12):3180. https://doi.org/10.3390/cancers15123180
Chicago/Turabian StyleMarin-Acevedo, Julian A., Bethany M. Withycombe, Youngchul Kim, Andrew S. Brohl, Zeynep Eroglu, Joseph Markowitz, Ahmad A. Tarhini, Kenneth Y. Tsai, and Nikhil I. Khushalani. 2023. "Cetuximab for Immunotherapy-Refractory/Ineligible Cutaneous Squamous Cell Carcinoma" Cancers 15, no. 12: 3180. https://doi.org/10.3390/cancers15123180
APA StyleMarin-Acevedo, J. A., Withycombe, B. M., Kim, Y., Brohl, A. S., Eroglu, Z., Markowitz, J., Tarhini, A. A., Tsai, K. Y., & Khushalani, N. I. (2023). Cetuximab for Immunotherapy-Refractory/Ineligible Cutaneous Squamous Cell Carcinoma. Cancers, 15(12), 3180. https://doi.org/10.3390/cancers15123180








