T- and B-Cells in the Inner Invasive Margin of Hepatocellular Carcinoma after Resection Associate with Favorable Prognosis
Abstract
:Simple Summary
Abstract
1. Introduction
2. Results
2.1. Demographics of HCC Patients
2.2. Outcomes
2.3. Distribution of Immune Cells in Different Regions of Interest (ROIs)
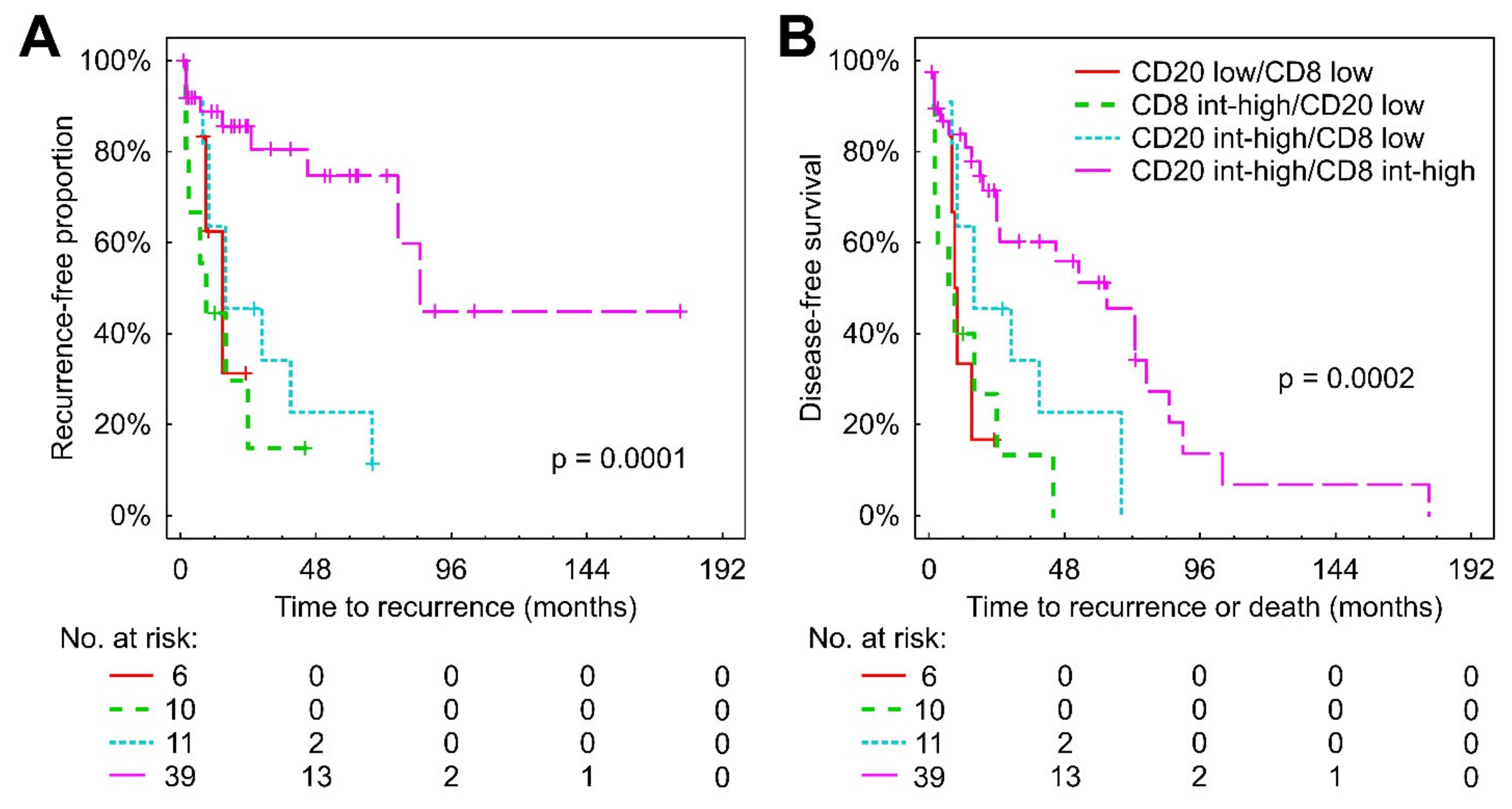
2.4. Prognostic Values of Immune Cells
2.5. Association between Subtypes of Immune Cells and Clinical and Pathology Variables
3. Discussion
3.1. Heterogeneity of the Tumor Invasive Margin
3.2. CD8+ Cells in TC and CD20+ Cells in the Inner Margin Confer the Best Prediction of TTR and DFS
3.3. CD8+ and CD20+ Cells in the Inner Margin Cooperate in Initiating Antitumor Immune Response
3.4. Greater T- and B-Cell Ratios between Inner and Outer Margin Associate with Longer TTR and DFS
3.5. Comparison to Relevant Literature
3.6. Limitations
4. Patients and Methods
4.1. Pathology and Immunohistology
4.2. Definitions of Regions of Interest
4.3. Stereological Analysis
4.4. Prognostic Factors and Outcomes
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Llovet, J.M.; Kelley, R.K.; Villanueva, A.; Singal, A.G.; Pikarsky, E.; Roayaie, S.; Lencioni, R.; Koike, K.; Zucman-Rossi, J.; Finn, R.S. Hepatocellular carcinoma. Nat. Rev. Dis. Primers 2021, 7, 6. [Google Scholar] [CrossRef] [PubMed]
- Roayaie, S.; Obeidat, K.; Sposito, C.; Mariani, L.; Bhoori, S.; Pellegrinelli, A.; Labow, D.; Llovet, J.M.; Schwartz, M.; Mazzaferro, V. Resection of hepatocellular cancer < /=2 cm: Results from two Western centers. Hepatology 2013, 57, 1426–1435. [Google Scholar] [CrossRef] [PubMed]
- Shinkawa, H.; Tanaka, S.; Takemura, S.; Amano, R.; Kimura, K.; Kinoshita, M.; Takahashi, K.; Matsuzaki, S.; Kubo, S. Nomograms predicting extra- and early intrahepatic recurrence after hepatic resection of hepatocellular carcinoma. Surgery 2021, 169, 922–928. [Google Scholar] [CrossRef] [PubMed]
- Centonze, L.; De Carlis, R.; Vella, I.; Carbonaro, L.; Incarbone, N.; Palmieri, L.; Sgrazzutti, C.; Ficarelli, A.; Valsecchi, M.G.; Dello Iacono, U.; et al. From LI-RADS Classification to HCC Pathology: A Retrospective Single-Institution Analysis of Clinico-Pathological Features Affecting Oncological Outcomes after Curative Surgery. Diagnostics 2022, 12, 160. [Google Scholar] [CrossRef]
- Najjar, M.; Agrawal, S.; Emond, J.C.; Halazun, K.J. Pretreatment neutrophil-lymphocyte ratio: Useful prognostic biomarker in hepatocellular carcinoma. J. Hepatocell. Carcinoma 2018, 5, 17–28. [Google Scholar] [CrossRef] [Green Version]
- Kamarajah, S.K.; Frankel, T.L.; Sonnenday, C.; Cho, C.S.; Nathan, H. Critical evaluation of the American Joint Commission on Cancer (AJCC) 8th edition staging system for patients with Hepatocellular Carcinoma (HCC): A Surveillance, Epidemiology, End Results (SEER) analysis. J. Surg. Oncol. 2018, 117, 644–650. [Google Scholar] [CrossRef]
- Zhou, L.; Rui, J.A.; Ye, D.X.; Wang, S.B.; Chen, S.G.; Qu, Q. Edmondson-Steiner grading increases the predictive efficiency of TNM staging for long-term survival of patients with hepatocellular carcinoma after curative resection. World J. Surg. 2008, 32, 1748–1756. [Google Scholar] [CrossRef] [PubMed]
- Galon, J.; Bruni, D. Tumor Immunology and Tumor Evolution: Intertwined Histories. Immunity 2020, 52, 55–81. [Google Scholar] [CrossRef]
- Bruni, D.; Angell, H.K.; Galon, J. The immune contexture and Immunoscore in cancer prognosis and therapeutic efficacy. Nat. Rev. Cancer 2020, 20, 662–680. [Google Scholar] [CrossRef] [PubMed]
- Pages, F.; Mlecnik, B.; Marliot, F.; Bindea, G.; Ou, F.S.; Bifulco, C.; Lugli, A.; Zlobec, I.; Rau, T.T.; Berger, M.D.; et al. International validation of the consensus Immunoscore for the classification of colon cancer: A prognostic and accuracy study. Lancet 2018, 391, 2128–2139. [Google Scholar] [CrossRef]
- Ding, W.; Xu, X.; Qian, Y.; Xue, W.; Wang, Y.; Du, J.; Jin, L.; Tan, Y. Prognostic value of tumor-infiltrating lymphocytes in hepatocellular carcinoma: A meta-analysis. Medicine (Baltimore) 2018, 97, e13301. [Google Scholar] [CrossRef]
- Hendry, S.; Salgado, R.; Gevaert, T.; Russell, P.A.; John, T.; Thapa, B.; Christie, M.; van de Vijver, K.; Estrada, M.V.; Gonzalez-Ericsson, P.I.; et al. Assessing Tumor-infiltrating Lymphocytes in Solid Tumors: A Practical Review for Pathologists and Proposal for a Standardized Method From the International Immunooncology Biomarkers Working Group: Part 1: Assessing the Host Immune Response, TILs in Invasive Breast Carcinoma and Ductal Carcinoma In Situ, Metastatic Tumor Deposits and Areas for Further Research. Adv. Anat. Pathol. 2017, 24, 235–251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, J.Y.; Gao, Q.; Wang, Z.C.; Zhou, J.; Wang, X.Y.; Min, Z.H.; Shi, Y.H.; Shi, G.M.; Ding, Z.B.; Ke, A.W.; et al. Margin-infiltrating CD20(+) B cells display an atypical memory phenotype and correlate with favorable prognosis in hepatocellular carcinoma. Clin. Cancer Res. 2013, 19, 5994–6005. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kolinko, Y.; Maleckova, A.; Kochova, P.; Grajciarova, M.; Blassova, T.; Kural, T.; Trailin, A.; Cervenkova, L.; Havrankova, J.; Vistejnova, L.; et al. Using virtual microscopy for the development of sampling strategies in quantitative histology and design-based stereology. Anat. Histol. Embryol. 2021. [Google Scholar] [CrossRef]
- Gabrielson, A.; Wu, Y.; Wang, H.; Jiang, J.; Kallakury, B.; Gatalica, Z.; Reddy, S.; Kleiner, D.; Fishbein, T.; Johnson, L.; et al. Intratumoral CD3 and CD8 T-cell Densities Associated with Relapse-Free Survival in HCC. Cancer Immunol. Res. 2016, 4, 419–430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, C.; Xu, J.; Song, J.; Liu, C.; Wang, J.; Weng, C.; Sun, H.; Wei, H.; Xiao, W.; Sun, R.; et al. The predictive value of centre tumour CD8(+) T cells in patients with hepatocellular carcinoma: Comparison with Immunoscore. Oncotarget 2015, 6, 35602–35615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schlageter, M.; Terracciano, L.M.; D'Angelo, S.; Sorrentino, P. Histopathology of hepatocellular carcinoma. World J. Gastroenterol. 2014, 20, 15955–15964. [Google Scholar] [CrossRef]
- Ishizaki, M.; Ashida, K.; Higashi, T.; Nakatsukasa, H.; Kaneyoshi, T.; Fujiwara, K.; Nouso, K.; Kobayashi, Y.; Uemura, M.; Nakamura, S.; et al. The formation of capsule and septum in human hepatocellular carcinoma. Virchows. Arch. 2001, 438, 574–580. [Google Scholar] [CrossRef] [PubMed]
- Lockwood, D.S.; Yeadon, T.M.; Clouston, A.D.; Crawford, D.G.; Fawcett, J.; Callaghan, S.A.; Gotley, D.C. Tumor progression in hepatocellular carcinoma: Relationship with tumor stroma and parenchymal disease. J. Gastroenterol. Hepatol. 2003, 18, 666–672. [Google Scholar] [CrossRef]
- Nielsen, K.; Rolff, H.C.; Eefsen, R.L.; Vainer, B. The morphological growth patterns of colorectal liver metastases are prognostic for overall survival. Mod. Pathol. 2014, 27, 1641–1648. [Google Scholar] [CrossRef] [Green Version]
- Galon, J.; Bruni, D. Approaches to treat immune hot, altered and cold tumours with combination immunotherapies. Nat. Rev. Drug Discov. 2019, 18, 197–218. [Google Scholar] [CrossRef]
- Joyce, J.A.; Fearon, D.T. T cell exclusion, immune privilege, and the tumor microenvironment. Science 2015, 348, 74–80. [Google Scholar] [CrossRef] [Green Version]
- Figenschau, S.L.; Fismen, S.; Fenton, K.A.; Fenton, C.; Mortensen, E.S. Tertiary lymphoid structures are associated with higher tumor grade in primary operable breast cancer patients. BMC Cancer 2015, 15, 101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martin-Sierra, C.; Martins, R.; Laranjeira, P.; Coucelo, M.; Abrantes, A.M.; Oliveira, R.C.; Tralhao, J.G.; Botelho, M.F.; Furtado, E.; Domingues, M.R.; et al. Functional and Phenotypic Characterization of Tumor-Infiltrating Leukocyte Subsets and Their Contribution to the Pathogenesis of Hepatocellular Carcinoma and Cholangiocarcinoma. Transl. Oncol. 2019, 12, 1468–1479. [Google Scholar] [CrossRef]
- Chen, D.S.; Mellman, I. Elements of cancer immunity and the cancer-immune set point. Nature 2017, 541, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Mlecnik, B.; Van den Eynde, M.; Bindea, G.; Church, S.E.; Vasaturo, A.; Fredriksen, T.; Lafontaine, L.; Haicheur, N.; Marliot, F.; Debetancourt, D.; et al. Comprehensive Intrametastatic Immune Quantification and Major Impact of Immunoscore on Survival. J. Natl. Cancer Inst. 2018, 110, 97–108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qin, M.; Wang, D.; Fang, Y.; Zheng, Z.; Liu, X.; Wu, F.; Wang, L.; Li, X.; Hui, B.; Ma, S.; et al. Current Perspectives on B Lymphocytes in the Immunobiology of Hepatocellular Carcinoma. Front. Oncol. 2021, 11, 647854. [Google Scholar] [CrossRef]
- Lund, F.E.; Randall, T.D. Effector and regulatory B cells: Modulators of CD4+ T cell immunity. Nat. Rev. Immunol. 2010, 10, 236–247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mariathasan, S.; Turley, S.J.; Nickles, D.; Castiglioni, A.; Yuen, K.; Wang, Y.; Kadel, E.E., III; Koeppen, H.; Astarita, J.L.; Cubas, R.; et al. TGFbeta attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature 2018, 554, 544–548. [Google Scholar] [CrossRef]
- Brunner, S.M.; Rubner, C.; Kesselring, R.; Martin, M.; Griesshammer, E.; Ruemmele, P.; Stempfl, T.; Teufel, A.; Schlitt, H.J.; Fichtner-Feigl, S. Tumor-infiltrating, interleukin-33-producing effector-memory CD8(+) T cells in resected hepatocellular carcinoma prolong patient survival. Hepatology 2015, 61, 1957–1967. [Google Scholar] [CrossRef] [PubMed]
- Giusca, S.E.; Wierzbicki, P.M.; Amalinei, C.; Caruntu, I.D.; Avadanei, E.R. Comparative analysis of CD4 and CD8 lymphocytes—Evidences for different distribution in primary and secondary liver tumors. Folia Histochem. Cytobiol. 2015, 53, 272–281. [Google Scholar] [CrossRef] [Green Version]
- Zheng, B.H.; Ma, J.Q.; Tian, L.Y.; Dong, L.Q.; Song, G.H.; Pan, J.M.; Liu, Y.M.; Yang, S.X.; Wang, X.Y.; Zhang, X.M.; et al. The distribution of immune cells within combined hepatocellular carcinoma and cholangiocarcinoma predicts clinical outcome. Clin. Transl. Med. 2020, 10, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Ramzan, M.; Sturm, N.; Decaens, T.; Bioulac-Sage, P.; Bancel, B.; Merle, P.; Tran Van Nhieu, J.; Slama, R.; Letoublon, C.; Zarski, J.P.; et al. Liver-infiltrating CD8(+) lymphocytes as prognostic factor for tumour recurrence in hepatitis C virus-related hepatocellular carcinoma. Liver Int. 2016, 36, 434–444. [Google Scholar] [CrossRef]
- Yao, W.; He, J.C.; Yang, Y.; Wang, J.M.; Qian, Y.W.; Yang, T.; Ji, L. The Prognostic Value of Tumor-infiltrating Lymphocytes in Hepatocellular Carcinoma: A Systematic Review and Meta-analysis. Sci. Rep. 2017, 7, 7525. [Google Scholar] [CrossRef]
- Martins-Filho, S.N.; Paiva, C.; Azevedo, R.S.; Alves, V.A.F. Histological Grading of Hepatocellular Carcinoma—A Systematic Review of Literature. Front. Med. (Lausanne) 2017, 4, 193. [Google Scholar] [CrossRef] [Green Version]
- Saxena, R. Practical Hepatic Pathology; Elsevier Saunders: Philadelphia, PA, USA, 2011; p. 675. [Google Scholar]
- Brunt, E.M.; Janney, C.G.; Di Bisceglie, A.M.; Neuschwander-Tetri, B.A.; Bacon, B.R. Nonalcoholic steatohepatitis: A proposal for grading and staging the histological lesions. Am. J. Gastroenterol. 1999, 94, 2467–2474. [Google Scholar] [CrossRef]
- Ishak, K.; Baptista, A.; Bianchi, L.; Callea, F.; De Groote, J.; Gudat, F.; Denk, H.; Desmet, V.; Korb, G.; MacSween, R.N.; et al. Histological grading and staging of chronic hepatitis. J. Hepatol. 1995, 22, 696–699. [Google Scholar] [CrossRef]
- Gundersen, H.J. Estimators of the number of objects per area unbiased by edge effects. Microsc. Acta 1978, 81, 107–117. [Google Scholar] [PubMed]
- Rothman, K.J.; Greenland, S. Introduction to regression models. In Modern Epidemiology, 2nd ed.; Winters, R., O’Connor, E., Eds.; Lippincott-Raven: Philadelphia, PA, USA, 1998; pp. 154–196. [Google Scholar]
- Kassambara, A.; Kosinski, M.; Biecek, P.; Fabian, S. Survminer: Drawing Survival Curves using ‘ggplot2’ 2021. Available online: https://rpkgs.datanovia.com/survminer/index.html (accessed on 25 November 2021).
- Therneau, T.M. A Package for Survival Analysis in R. R Package Version 3.2–13. 2021. Available online: https://CRAN.R-project.org/package=survival (accessed on 25 November 2021).



| QA | TTR | DFS | |||||
|---|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | p | ||
| Tumor Center | |||||||
| CD3,n = 67 p = 0.030 *, 0.014 † | int vs. low | 0.61 | 0.26–1.40 | 0.243 | 0.62 | 0.31–1.23 | 0.170 |
| high vs. low | 0.20 | 0.06–0.66 | 0.008 | 0.28 | 0.12–0.66 | 0.003 | |
| CD8,n = 67 p = 0.010 *, 0.004 † | int vs. low | 0.34 | 0.14–0.81 | 0.014 | 0.53 | 0.27–1.07 | 0.075 |
| high vs. low | 0.25 | 0.09–0.70 | 0.008 | 0.23 | 0.09–0.55 | 0.001 | |
| CD20,n = 67 p = 0.031 *, 0.020 † | int vs. low | 0.63 | 0.27–1.48 | 0.289 | 0.67 | 0.32–1.37 | 0.270 |
| high vs. low | 0.18 | 0.05–0.65 | 0.009 | 0.27 | 0.11–0.69 | 0.006 | |
| Inner invasive margin | |||||||
| CD3,n = 65 p = 0.014 *, 0.005 † | int vs. low | 0.77 | 0.33–1.79 | 0.545 | 0.77 | 0.39–1.55 | 0.472 |
| high vs. low | 0.14 | 0.04–0.54 | 0.004 | 0.24 | 0.09–0.59 | 0.002 | |
| CD8,n = 66 p = 0.029 *, 0.006 † | int vs. low | 0.52 | 0.23–1.19 | 0.120 | 0.77 | 0.40–1.51 | 0.455 |
| high vs. low | 0.23 | 0.08–0.69 | 0.009 | 0.22 | 0.09–0.57 | 0.002 | |
| CD20,n = 66 p = 0.002 *, <0.001 † | int vs. low | 0.38 | 0.16–0.90 | 0.026 | 0.36 | 0.18–0.74 | 0.005 |
| high vs. low | 0.09 | 0.02–0.36 | 0.001 | 0.13 | 0.05–0.35 | <0.001 | |
| Outer invasive margin | |||||||
| CD3,n = 65 p = 0.354 *, 0.280 † | int vs. low | 0.66 | 0.26–1.65 | 0.371 | 0.58 | 0.27–1.23 | 0.153 |
| high vs. low | 0.47 | 0.17–1.32 | 0.151 | 0.56 | 0.26–1.24 | 0.153 | |
| CD8,n = 66 p = 0.325 *, 0.061 † | int vs. low | 0.55 | 0.22–1.37 | 0.197 | 0.48 | 0.23–0.98 | 0.044 |
| high vs. low | 0.50 | 0.18–1.36 | 0.174 | 0.41 | 0.18–0.92 | 0.030 | |
| CD20,n = 66 p = 0.680 *, 0.475 † | int vs. low | 0.99 | 0.40–2.44 | 0.986 | 0.99 | 0.49–2.00 | 0.974 |
| high vs. low | 0.68 | 0.25–1.87 | 0.452 | 0.65 | 0.29–1.45 | 0.290 | |
| Peritumor liver | |||||||
| CD3, n = 64 | int vs. low | 0.53 | 0.20–1.38 | 0.193 | 0.74 | 0.34–1.63 | 0.452 |
| p = 0.418 *, 0.676 † | high vs. low | 0.78 | 0.30–2.04 | 0.608 | 0.95 | 0.42–2.14 | 0.902 |
| CD8, n = 65 | int vs. low | 0.82 | 0.33–2.04 | 0.663 | 1.09 | 0.52–2.26 | 0.828 |
| p = 0.753 *, 0.952 † | high vs. low | 1.15 | 0.43–3.04 | 0.780 | 1.14 | 0.50–2.62 | 0.757 |
| CD20, n = 65 | int vs. low | 0.24 | 0.09–0.64 | 0.004 | 0.43 | 0.20–0.92 | 0.030 |
| p= 0.014 *, 0.053 † | high vs. low | 0.72 | 0.30–1.71 | 0.452 | 0.86 | 0.40–1.84 | 0.699 |
| TTR | DFS | |||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | p | |
| CD3+ T cells | ||||||
| Inn M/out M > 0.361 †, n = 65 | 0.36 | 0.16–0.85 | 0.019 | 0.45 | 0.24–0.86 | 0.016 |
| TC/out M > 0.201, n = 65 | 0.41 | 0.18–0.92 | 0.031 | 0.56 | 0.31–1.02 | 0.057 |
| TC/M > 0.330, n = 65 | 0.53 | 0.24–1.17 | 0.117 | 0.64 | 0.35- 1.16 | 0.138 |
| TC/PT > 0.219, n = 64 | 0.43 | 0.19–1.00 | 0.050 | 0.61 | 0.33–1.13 | 0.116 |
| CD8+ T cells | ||||||
| Inn M/out M > 0.349, n = 66 | 0.53 | 0.24–1.17 | 0.119 | 0.45 | 0.24–0.85 | 0.013 |
| TC/out M > 0.166, n = 66 | 0.59 | 0.27–1.29 | 0.187 | 0.63 | 0.34–1.15 | 0.131 |
| TC/M > 0.218, n = 66 | 0.76 | 0.36–1.62 | 0.478 | 0.85 | 0.47–1.54 | 0.598 |
| TC/PT > 0.161, n = 65 | 0.41 | 0.18–0.93 | 0.032 | 0.42 | 0.22–0.81 | 0.010 |
| CD20+ B cells | ||||||
| Inn M/out M > 0.113, n = 66 | 0.28 | 0.12–0.65 | 0.003 | 0.29 | 0.15–0.57 | <0.001 |
| TC/out M > 0.051, n = 66 | 0.23 | 0.09–0.54 | 0.001 | 0.40 | 0.21–0.75 | 0.004 |
| TC/M > 0.078, n = 66 | 0.24 | 0.10–0.57 | 0.001 | 0.41 | 0.22–0.76 | 0.005 |
| TC/PT > 0.049, n = 65 | 0.27 | 0.12–0.65 | 0.003 | 0.49 | 0.27–0.90 | 0.022 |
| TTR | ||||
|---|---|---|---|---|
| HR | 95% CI | p | ||
| Density of nucleated profiles of TIL (QA) per ROI | ||||
| CD3 TC † p = 0.082 * | int vs. low | 0.78 | 0.32–1.96 | 0.601 |
| high vs. low | 0.26 | 0.07–0.89 | 0.032 | |
| CD8 TC p = 0.010 * | int vs. low | 0.34 | 0.14–0.81 | 0.014 |
| high vs. low | 0.25 | 0.09–0.70 | 0.008 | |
| CD20 TC † p = 0.017 * | int vs. low | 0.91 | 0.35–2.35 | 0.842 |
| high vs. low | 0.18 | 0.05–0.64 | 0.008 | |
| CD3 inn M † p = 0.014 * | int vs. low | 1.02 | 0.41–2.53 | 0.961 |
| high vs. low | 0.16 | 0.04–0.62 | 0.008 | |
| CD8 inn M † p = 0.065 * | int vs. low | 0.60 | 0.25–1.39 | 0.230 |
| high vs. low | 0.26 | 0.09–0.81 | 0.020 | |
| CD20 inn M † p = 0.004 * | int vs. low | 0.50 | 0.20–1.27 | 0.147 |
| high vs. low | 0.10 | 0.03–0.39 | 0.001 | |
| CD20 PT † p = 0.018 * | int vs. low | 0.24 | 0.09–0.66 | 0.005 |
| high vs. low | 0.70 | 0.29–1.67 | 0.420 | |
| Ratios between densities of nucleated profiles of TIL (QA) in two ROIs | ||||
| CD3 inn M/out M ǂ | above vs. under median (0.361) | 0.35 | 0.15–0.81 | 0.015 |
| CD3 TC/out M ǂ | above vs. under median (0.201) | 0.45 | 0.21–1.03 | 0.058 |
| CD3 TC/PT ǂ | above vs. under median (0.219) | 0.48 | 0.21–1.13 | 0.093 |
| CD8 TC/PT ǂ | above vs. under median (0.161) | 0.48 | 0.20–1.12 | 0.091 |
| CD20 inn M/out M ǂ | above vs. under median (0.113) | 0.26 | 0.11–0.62 | 0.002 |
| CD20 TC/out M ǂ | above vs. under median (0.051) | 0.21 | 0.09–0.51 | 0.001 |
| CD20 TC/PT ǂ | above vs. under median (0.049) | 0.28 | 0.12–0.66 | 0.004 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trailin, A.; Červenková, L.; Ambrozkiewicz, F.; Ali, E.; Kasi, P.; Pálek, R.; Hošek, P.; Třeška, V.; Daum, O.; Tonar, Z.; et al. T- and B-Cells in the Inner Invasive Margin of Hepatocellular Carcinoma after Resection Associate with Favorable Prognosis. Cancers 2022, 14, 604. https://doi.org/10.3390/cancers14030604
Trailin A, Červenková L, Ambrozkiewicz F, Ali E, Kasi P, Pálek R, Hošek P, Třeška V, Daum O, Tonar Z, et al. T- and B-Cells in the Inner Invasive Margin of Hepatocellular Carcinoma after Resection Associate with Favorable Prognosis. Cancers. 2022; 14(3):604. https://doi.org/10.3390/cancers14030604
Chicago/Turabian StyleTrailin, Andriy, Lenka Červenková, Filip Ambrozkiewicz, Esraa Ali, Phanindra Kasi, Richard Pálek, Petr Hošek, Vladislav Třeška, Ondrej Daum, Zbyněk Tonar, and et al. 2022. "T- and B-Cells in the Inner Invasive Margin of Hepatocellular Carcinoma after Resection Associate with Favorable Prognosis" Cancers 14, no. 3: 604. https://doi.org/10.3390/cancers14030604
APA StyleTrailin, A., Červenková, L., Ambrozkiewicz, F., Ali, E., Kasi, P., Pálek, R., Hošek, P., Třeška, V., Daum, O., Tonar, Z., Liška, V., & Hemminki, K. (2022). T- and B-Cells in the Inner Invasive Margin of Hepatocellular Carcinoma after Resection Associate with Favorable Prognosis. Cancers, 14(3), 604. https://doi.org/10.3390/cancers14030604







