Metastatic Breast Cancer Recurrence after Bone Fractures
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design, Data Sources and Data Structure
2.2. Data Protection and Ethical Considerations
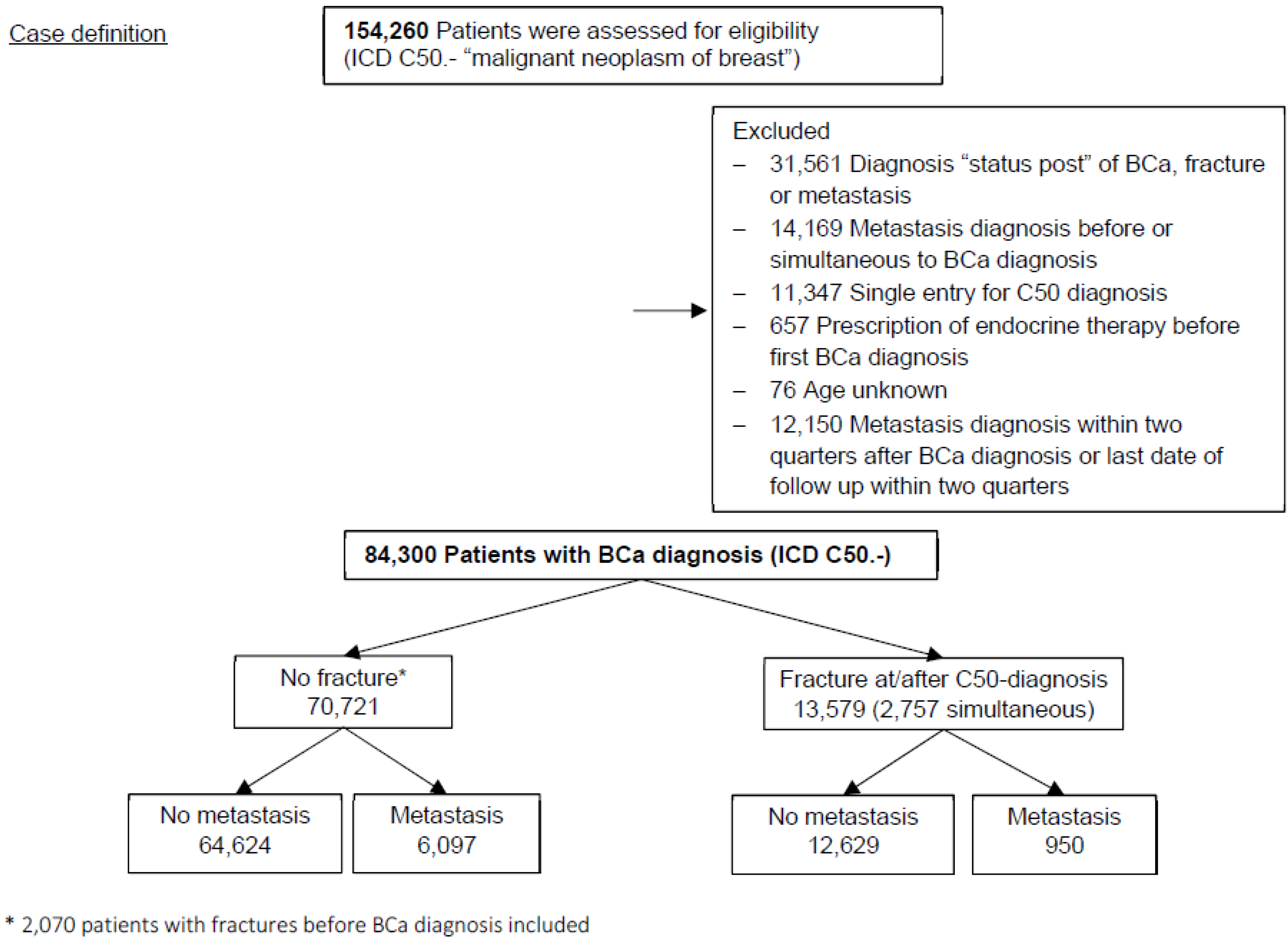
2.3. Study Population
2.4. Statistical Analysis
2.4.1. Exposure Variables
2.4.2. Outcomes
2.4.3. Other Covariates
2.4.4. Time-to-Event Regression
3. Results
3.1. Diagnosis of Bone Fractures and Risk of Metastasis
3.2. Sensitivity Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Cancer Report: Cancer Research for Cancer Prevention; Wild, C.P., Weiderpass, E., Stewart, B.W., Eds.; WHO: Geneva, Switzerland, 2020. [Google Scholar]
- Rueda, O.M.; Sammut, S.-J.; Seoane, J.A.; Chin, S.-F.; Caswell-Jin, J.L.; Callari, M.; Batra, R.; Pereira, B.; Bruna, A.; Ali, H.R.; et al. Dynamics of breast-cancer relapse reveal late-recurring ER-positive genomic subgroups. Nature 2019, 567, 399–404. [Google Scholar] [CrossRef]
- Pan, H.; Gray, R.; Braybrooke, J.; Davies, C.; Taylor, C.; McGale, P.; Peto, R.; Pritchard, K.I.; Bergh, J.; Dowsett, M.; et al. 20-Year Risks of Breast-Cancer Recurrence after Stopping Endocrine Therapy at 5 Years. N. Engl. J. Med. 2017, 377, 1836–1846. [Google Scholar] [CrossRef] [Green Version]
- Holleczek, B.; Stegmaier, C.; Radosa, J.C.; Solomayer, E.-F.; Brenner, H. Risk of loco-regional recurrence and distant metastases of patients with invasive breast cancer up to ten years after diagnosis—Results from a registry-based study from Germany. BMC Cancer 2019, 19, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turajlic, S.; Xu, H.; Litchfield, K.; Rowan, A.; Chambers, T.; Lopez, J.I.; Nicol, D.; O’brien, T.; Larkin, J.; Horswell, S.; et al. Tracking Cancer Evolution Reveals Constrained Routes to Metastases: TRACERx Renal. Cell 2018, 173, 581–594.e12. [Google Scholar] [CrossRef] [Green Version]
- DerHagopian, R.P.; Sugarbaker, E.V.; Ketcham, A. Inflammatory oncotaxis. JAMA 1978, 240, 374–375. [Google Scholar] [CrossRef]
- Ewing, J. The Modern Attitude Toward Traumatic Cancer. Bull. N. Y. Acad. Med. 1935, 11, 281–333. [Google Scholar]
- Jones, F.S.; Rous, P. On the cause of the localization of secondary tumors at points of injury. J. Exp. Med. 1914, 20, 404–412. [Google Scholar] [CrossRef] [Green Version]
- Smith, H.A.; Kang, Y. The metastasis-promoting roles of tumor-associated immune cells. J. Mol. Med. 2013, 91, 411–429. [Google Scholar] [CrossRef]
- Rybinski, B.; Franco-Barraza, J.; Cukierman, E. The wound healing, chronic fibrosis, and cancer progression triad. Physiol. Genom. 2014, 46, 223–244. [Google Scholar] [CrossRef]
- Abbas, S.; Ihle, P.; Köster, I.; Schubert, I. Estimation of Disease Incidence in Claims Data Dependent on the Length of Follow-Up: A Methodological Approach. Health Serv. Res. 2011, 47, 746–755. [Google Scholar] [CrossRef] [Green Version]
- Eastell, R.; Adams, J.; Clack, G.; Howell, A.; Cuzick, J.; Mackey, J.; Beckmann, M.W.; Coleman, R.E. Long-term effects of anastrozole on bone mineral density: 7-year results from the ATAC trial. Ann. Oncol. 2011, 22, 857–862. [Google Scholar] [CrossRef] [PubMed]
- Heeke, A.; Nunes, M.R.; Lynce, F. Bone-Modifying Agents in Early-Stage and Advanced Breast Cancer. Curr. Breast Cancer Rep. 2018, 10, 241–250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Braun, S.; Pantel, K.; Müller, P.; Janni, W.; Hepp, F.; Kentenich, C.R.; Gastroph, S.; Wischnik, A.; Dimpfl, T.; Kindermann, G.; et al. Cytokeratin-Positive Cells in the Bone Marrow and Survival of Patients with Stage I, II, or III Breast Cancer. N. Engl. J. Med. 2000, 342, 525–533. [Google Scholar] [CrossRef] [Green Version]
- Braun, S.; Schindlbeck, C.; Hepp, F.; Janni, W.; Kentenich, C.; Riethmüller, G.; Pantel, K. Occult Tumor Cells in Bone Marrow of Patients with Locoregionally Restricted Ovarian Cancer Predict Early Distant Metastatic Relapse. J. Clin. Oncol. 2001, 19, 368–375. [Google Scholar] [CrossRef]
- Braun, S.; Vogl, F.D.; Naume, B.; Janni, W.; Osborne, M.P.; Coombes, R.C.; Schlimok, G.; Diel, I.J.; Gerber, B.; Gebauer, G.; et al. A Pooled Analysis of Bone Marrow Micrometastasis in Breast Cancer. N. Engl. J. Med. 2005, 353, 793–802. [Google Scholar] [CrossRef]
- Werner, S.; Pantel, K. Tracing the Seeds in the Soil. Clin. Chem. 2017, 63, 1764–1765. [Google Scholar] [CrossRef]
- Riethdorf, S.; Müller, V.; Loibl, S.; Nekljudova, V.; Weber, K.; Huober, J.; Fehm, T.; Schrader, I.; Hilfrich, J.; Holms, F.; et al. Prognostic Impact of Circulating Tumor Cells for Breast Cancer Patients Treated in the Neoadjuvant “Geparquattro” Trial. Clin. Cancer Res. 2017, 23, 5384–5393. [Google Scholar] [CrossRef] [Green Version]
- Kang, Y.; Pantel, K. Tumor Cell Dissemination: Emerging Biological Insights from Animal Models and Cancer Patients. Cancer Cell 2013, 23, 573–581. [Google Scholar] [CrossRef] [Green Version]
- Pantel, K.; Hayes, D.F. Disseminated breast tumour cells: Biological and clinical meaning. Nat. Rev. Clin. Oncol. 2017, 15, 129–131. [Google Scholar] [CrossRef]
- Celià-Terrassa, T.; Kang, Y. Metastatic niche functions and therapeutic opportunities. Nat. Cell Biol. 2018, 20, 868–877. [Google Scholar] [CrossRef] [Green Version]
- Bahney, C.S.; Zondervan, R.L.; Allison, P.; Theologis, A.; Ashley, J.W.; Ahn, J.; Miclau, T.; Marcucio, R.S.; Hankenson, K.D. Cellular biology of fracture healing. J. Orthop. Res. 2019, 37, 35–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cassetta, L.; Fragkogianni, S.; Sims, A.H.; Swierczak, A.; Forrester, L.M.; Zhang, H.; Soong, D.Y.H.; Cotechini, T.; Anur, P.; Lin, E.Y.; et al. Human Tumor-Associated Macrophage and Monocyte Transcriptional Landscapes Reveal Cancer-Specific Reprogramming, Biomarkers, and Therapeutic Targets. Cancer Cell 2019, 35, 588–602.e10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohme, M.; Riethdorf, S.; Pantel, K. Circulating and disseminated tumour cells—Mechanisms of immune surveillance and escape. Nat. Rev. Clin. Oncol. 2017, 14, 155–167. [Google Scholar] [CrossRef] [PubMed]
- Sousa, S.; Määttä, J. The role of tumour-associated macrophages in bone metastasis. J. Bone Oncol. 2016, 5, 135–138. [Google Scholar] [CrossRef] [Green Version]
- Asiry, S.; Kim, G.; Filippou, P.S.; Sanchez, L.R.; Entenberg, D.; Marks, D.K.; Oktay, M.H.; Karagiannis, G.S. The Cancer Cell Dissemination Machinery as an Immunosuppressive Niche: A New Obstacle Towards the Era of Cancer Immunotherapy. Front. Immunol. 2021, 12, 1183. [Google Scholar] [CrossRef]
- Cuzick, J.; Otto, F.; Baron, J.A.; Brown, P.H.; Burn, J.; Greenwald, P.; Jankowski, J.; La Vecchia, C.; Meyskens, F.; Senn, H.J.; et al. Aspirin and non-steroidal anti-inflammatory drugs for cancer prevention: An international consensus statement. Lancet Oncol. 2009, 10, 501–507. [Google Scholar] [CrossRef] [Green Version]
- Loomans-Kropp, H.A.; Pinsky, P.; Umar, A. Evaluation of Aspirin Use with Cancer Incidence and Survival Among Older Adults in the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial. JAMA Netw. Open 2021, 4, e2032072. [Google Scholar] [CrossRef]
- Schäfer, I.; Kaduszkiewicz, H.; Nguyen, T.S.; Bussche, H.V.D.; Scherer, M.; Schön, G. Multimorbidity patterns and 5-year overall mortality: Results from a claims data–based observational study. J. Comorbidity 2018, 8, 2235042X18816588. [Google Scholar] [CrossRef] [Green Version]
- Hlatky, M.A.; Ray, R.M.; Burwen, D.R.; Margolis, K.L.; Johnson, K.C.; Kucharska-Newton, A.; Manson, J.E.; Robinson, J.G.; Safford, M.M.; Allison, M.; et al. Use of Medicare Data to Identify Coronary Heart Disease Outcomes in the Women’s Health Initiative. Circ. Cardiovasc. Qual. Outcomes 2014, 7, 157–162. [Google Scholar] [CrossRef] [Green Version]
- Sohn, S.; Kim, J.; Chung, C.K.; Lee, N.R.; Park, E.; Chang, U.-K.; Sohn, M.J.; Kim, S.H. A nationwide epidemiological study of newly diagnosed spine metastasis in the adult Korean population. Spine J. 2016, 16, 937–945. [Google Scholar] [CrossRef]
- Heller, G.; Günster, C.; Misselwitz, B.; Feller, A.; Schmidt, S. Jährliche Fallzahl pro Klinik und Überlebensrate sehr untergewichtiger Frühgeborener (VLBW) in Deutschland—Eine bundesweite Analyse mit Routinedaten. Z. Geburtshilfe Neonatol. 2007, 211, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Lowenstern, A.; Lippmann, S.J.; Brennan, J.M.; Wang, T.Y.; Curtis, L.H.; Feldman, T.; Glower, D.D.; Hammill, B.G.; Vemulapalli, S. Use of Medicare Claims to Identify Adverse Clinical Outcomes After Mitral Valve Repair. Circ. Cardiovasc. Interv. 2019, 12, e007451. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Chae, K.-H.; Chung, Y.-J.; Hwang, H.; Lee, M.; Kim, H.-K.; Cho, H.-H.; Kim, M.-R.; Jung, C.-Y.; Kim, S. The effect of the look-back period for estimating incidence using administrative data. BMC Health Serv. Res. 2020, 20, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Center for Cancer Registry Data in the Robert Koch Institute. Database Query with Estimates of the Incidence, Prevalence and Survival of Cancer in Germany Based on the Epidemiological State Cancer Registry Data; Center for Cancer Registry Data in the Robert Koch Institute: Berlin, Germany, 2021. [Google Scholar] [CrossRef]
- Cronin-Fenton, D.; Kjærsgaard, A.; Nørgaard, M.; Amelio, J.; Liede, A.; Hernandez, R.K.; Sørensen, H.T. Breast cancer recurrence, bone metastases, and visceral metastases in women with stage II and III breast cancer in Denmark. Breast Cancer Res. Treat. 2017, 167, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Wirtz, P.; Baumann, F.T. Physical Activity, Exercise and Breast Cancer - What Is the Evidence for Rehabilitation, Aftercare, and Survival A Review. Breast Care 2018, 13, 92–100. [Google Scholar] [CrossRef]
- Lee, J. A Meta-analysis of the Association Between Physical Activity and Breast Cancer Mortality. Cancer Nurs. 2019, 42, 271–285. [Google Scholar] [CrossRef]
| Total N = 84,300 | No Fracture at/after Diagnosis, N = 70,721 | Fracture at/after Diagnosis (ICD-10 S, T & M80), N = 13,579 | |
|---|---|---|---|
| Age at diagnosis (years), median (IQR) | 61 (52, 71) | 60 (52, 70) | 68 (58, 76) |
| Source of BCa diagnosis | |||
| Hospital care | 13,828 (16.4) | 12,527 (17.7) | 1301 (9.6) |
| Ambulant care | 70,472 (83.6) | 58,194 (82.3) | 12,278 (90.4) |
| Year of BCa diagnosis (col %; row %) b | |||
| 2015 § | 62,835 (74.5, 100) | 50,704 (71.7, 80.7) | 12,131 (89.3, 19.3) |
| 2016 | 8197 (9.7, 100) | 7406 (10.5, 90.4) | 791 (5.8, 9.7) |
| 2017 | 7082 (8.4, 100) | 6633 (9.4, 93.7) | 449 (3.3, 6.3) |
| 2018 | 6015 (7.1, 100) | 5810 (8.2, 96.6) | 205 (1.5, 3.4) |
| 2019 | 171 (0.2, 100) | 168 (0.2, 98.3) | 3 (0.02, 1.8) |
| Follow-up-time (years) (median (IQR)) | 4.25 (3.00, 4.25) | 4.25 (2.75, 4.25) | 4.25 (4.00, 4.25) |
| Time from diagnosis to fracture (years) (median (IQR)) | - | - | 1.50 (0.25, 2.50) |
| Metastasis * | |||
| No | 77,253 (91.6) | 64,624 (91.4) | 12,629 (93.0) |
| Yes | 7047 (8.4) | 6097 (8.6) | 950 (7.0) |
| Regional | 3226 (3.8) | 2865 (4.1) | 361 (2.7) |
| Distant (not bone) | 4903 (5.8) | 4230 (6.0) | 673 (5.0) |
| Distant bone | 2210 (2.6) | 1885 (2.7) | 325 (2.4) |
| No (2015 cases excluded) | 20,298 (94.6) | 18,908 (94.6) | 1390 (96.0) |
| Yes (2015 cases excluded) | 1167 (5.4) | 1109 (5.4) | 58 (4.0) |
| Regional | 675 (3.1) | 650 (3.3) | 25 (1.7) |
| Distant (not bone) | 632 (2.9) | 590 (3.0) | 42 (2.9) |
| Distant bone | 262 (1.2) | 239 (1.2) | 23 (1.6) |
| Second tumors at/after C50 | |||
| Yes | 14,606 (15.0) | 10,074 (14.2) | 2551 (18.8) |
| No | 71675 (85.0) | 60,647 (85.8) | 11,028 (81.2) |
| Other tumors prior C50 | |||
| Yes | 1996 (2.4) | 1815 (2.6) | 181 (1.3) |
| No | 82,304 (97.6) | 68,906 (97.4) | 13,398 (98.7) |
| Osteoporosis | |||
| Yes | 19,795 (23.5) | 13,429 (19.0) | 6366 (46.7) |
| No | 64,505 (76.5) | 57,292 (81.0) | 7213 (53.1) |
| Anti-estrogens | |||
| Yes | 24,268 (28.8) | 21,335 (30.2) | 2933 (21.6) |
| No | 60,032 (71.2) | 49,386 (69.8) | 10,646 (78.4) |
| Aromatase-inhibitors | |||
| Yes | 18,834 (22.3) | 15,615 (22.1) | 3219 (23.7) |
| No | 65,466 (77.7) | 55,106 (77.9) | 10,360 (76.3) |
| Bisphosphonates | |||
| Yes | 6678 (7.9) | 3986 (5.6) | 2692 (19.8) |
| No | 77,622 (92.1) | 66,735 (94.4) | 10,887 (80.2) |
| No. of entries per patient year without entries for fractures (median (IQR)) | 8.9 (5.4, 12.7) | 8.7 (5.4, 12.5) | 9.7 (6.1, 13.8) |
| Overall Metastasis | LN Metastasis | Distant Non-BM | Distant BM | |
|---|---|---|---|---|
| N (n events) | 84,300 (7047) | 84,300 (3226) | 84,300 (4903) | 84,300 (2210) |
| HR (95%-CI) | HR (95%-CI) | HR (95%-CI) | HR (95%-CI) | |
| Model 1 (unadjusted) | ||||
| Fractures at/after BCa diagnosis (Reference: none) | 1.14 (1.06, 1.23) | 1.03 (0.92, 1.14) | 1.20 (1.11, 1.30) | 1.21 (1.07, 1.36) |
| Model 2 (adjusted) | ||||
| Fractures at/after BCa diagnosis (Reference: none) | 1.12 (1.04, 1.20) | 1.08 (0.97, 1.21) | 1.16 (1.07, 1.26) | 1.18 (1.05, 1.34) |
| Second tumor after BCa | 1.91 (1.81, 2.03) | 2.11 (1.94, 2.29) | 2.27 (2.12, 2.42) | 2.07 (1.88, 2.28) |
| Other tumor before BCa | 1.49 (1.27, 1.75) | 1.29 (1.01, 1.64) | 1.60 (1.30, 1.96) | 1.66 (1.22, 2.26) |
| Osteoporosis | 1.00 (0.94, 1.07) | 0.96 (0.87, 1.05) | 0.95 (0.89, 1.02) | 1.08 (0.98, 1.20) |
| Anti-estrogens | 0.94 (0.89, 0.996) | 0.80 (0.73, 0.87) | 1.05 (0.99, 1.13) | 1.14 (1.04, 1.25) |
| Aromatase-inhibitors | 1.27 (1.20, 1.35) | 1.28 (1.18, 1.39) | 1.43 (1.45, 1.64) | 1.87 (1.70, 2.05) |
| Source of BCa diagnosis ambulant (Reference: hospital) | 0.90 (0.84, 0.97) | 0.96 (0.86, 1.06) | 0.90 (0.82, 0.99) | 0.98 (0.85, 1.12) |
| Age (years) | 0.999 (0.997, 1.001) | 0.99 (0.98, 0.99) | 1.00 (1.00, 1.01) | 0.996 (0.99, 1.00) |
| Exclusion of BCa diagnosis in 2015 N (n events) | 21,465 (1167) | 21,465 (675) | 21,465 (632) | 21,465 (262) |
| Model 3 (adjusted) | ||||
| Fractures at/after BCa diagnosis (reference: none) | 0.99 (0.76, 1.30) | 0.80 (0.54, 1.19) | 1.28 (0.94, 1.75) | 1.45 (0.94, 2.24) |
| Second tumor after BCa | 1.57 (1.30, 1.89) | 1.55 (1.22, 1.98) | 1.73 (1.37, 2.19) | 2.14 (1.54, 2.98) |
| Other tumor before BCa | 1.54 (1.26, 1.88) | 1.17 (0.89, 1.57) | 1.61 (1.24, 2.09) | 1.90 (1.28, 2.80) |
| Osteoporosis | 0.93 (0.78, 1.10) | 0.998 (0.79, 1.66) | 0.85 (0.67, 1.07) | 1.12 (0.81, 1.54) |
| Anti-estrogens | 0.49 (0.43, 0.57) | 0.36 (0.29, 0.44) | 0.54 (0.45, 0.66) | 0.61 (0.46, 0.81) |
| Aromatase inhibitors | 0.74 (0.63, 0.86) | 0.62 (0.50, 0.77) | 0.79 (0.64, 0.96) | 1.13 (0.84, 1.51) |
| Source of BCa diagnosis ambulant (Reference: hospital) | 0.93 (0.82, 1.05) | 1.01 (0.86, 1.20) | 0.85 (0.72, 1.10) | 0.95 (0.73, 1.23) |
| Age (years) | 0.994 (0.989, 0.999) | 0.99 (0.98, 0.99) | 1.00 (0.99, 1.01) | 0.99 (0.98, 1.01) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Obi, N.; Werner, S.; Thelen, F.; Becher, H.; Pantel, K. Metastatic Breast Cancer Recurrence after Bone Fractures. Cancers 2022, 14, 601. https://doi.org/10.3390/cancers14030601
Obi N, Werner S, Thelen F, Becher H, Pantel K. Metastatic Breast Cancer Recurrence after Bone Fractures. Cancers. 2022; 14(3):601. https://doi.org/10.3390/cancers14030601
Chicago/Turabian StyleObi, Nadia, Stefan Werner, Frank Thelen, Heiko Becher, and Klaus Pantel. 2022. "Metastatic Breast Cancer Recurrence after Bone Fractures" Cancers 14, no. 3: 601. https://doi.org/10.3390/cancers14030601
APA StyleObi, N., Werner, S., Thelen, F., Becher, H., & Pantel, K. (2022). Metastatic Breast Cancer Recurrence after Bone Fractures. Cancers, 14(3), 601. https://doi.org/10.3390/cancers14030601







