Genome-Wide Association Study Identifies Candidate Loci Associated with Opioid Analgesic Requirements in the Treatment of Cancer Pain
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.1.1. Patients Who Underwent Cancer Pain Treatment with Opioid Analgesics
2.1.2. Patient Characteristics and Clinical Data
2.2. Whole-Genome Genotyping, Quality Control, and Gene-Based and Gene-Set Analyses
2.2.1. Whole-Genome Genotyping and Quality Control
2.2.2. Gene-Based and Gene-Set Analyses
2.3. Statistical Analysis
2.4. Additional in Silico Analysis
2.4.1. Power Analysis
2.4.2. Linkage Disequilibrium Analysis
2.4.3. Reference of Databases
3. Results
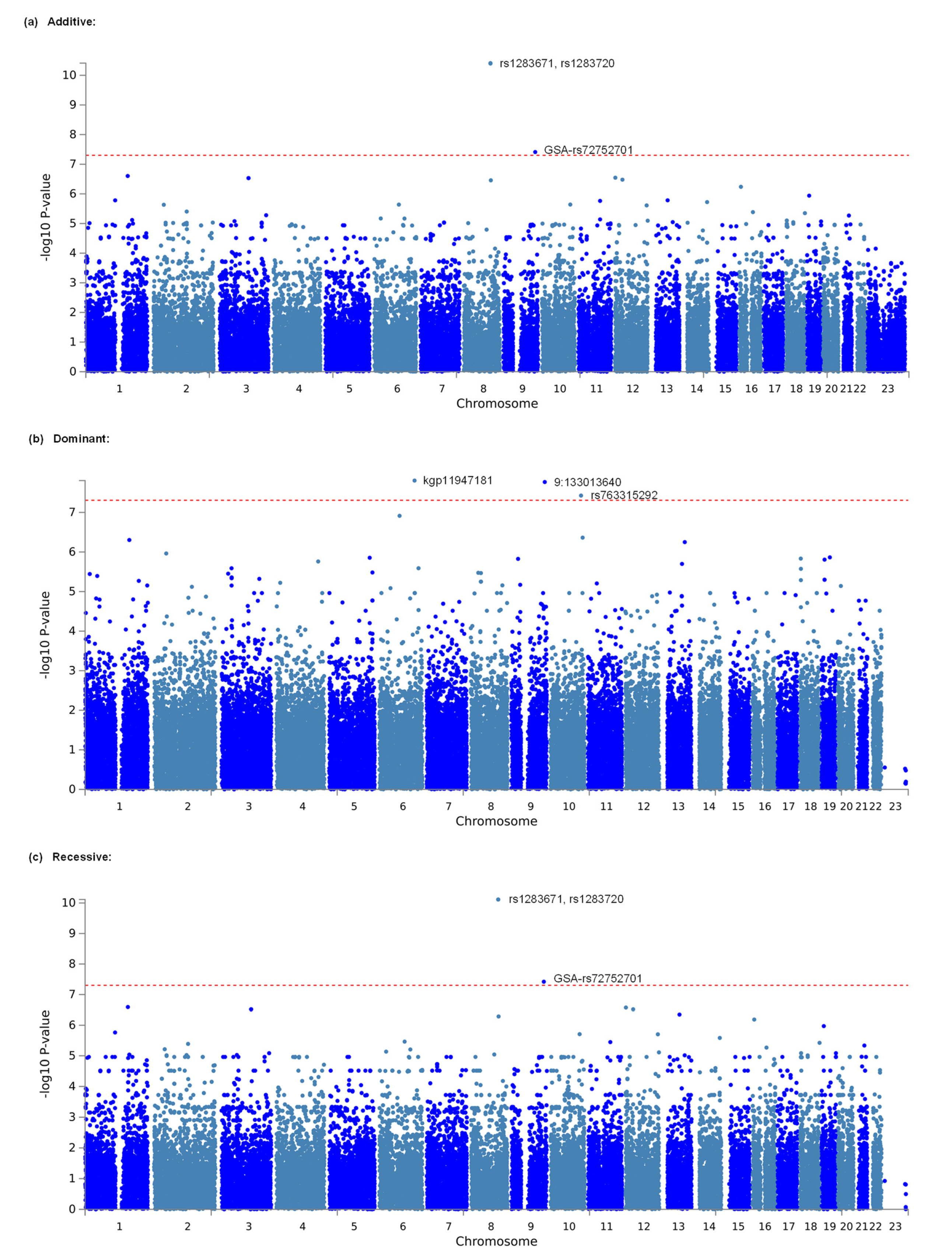
3.1. Identification of Genetic Polymorphisms Associated with Opioid Analgesic Requirements in the Treatment of Cancer Pain by GWAS
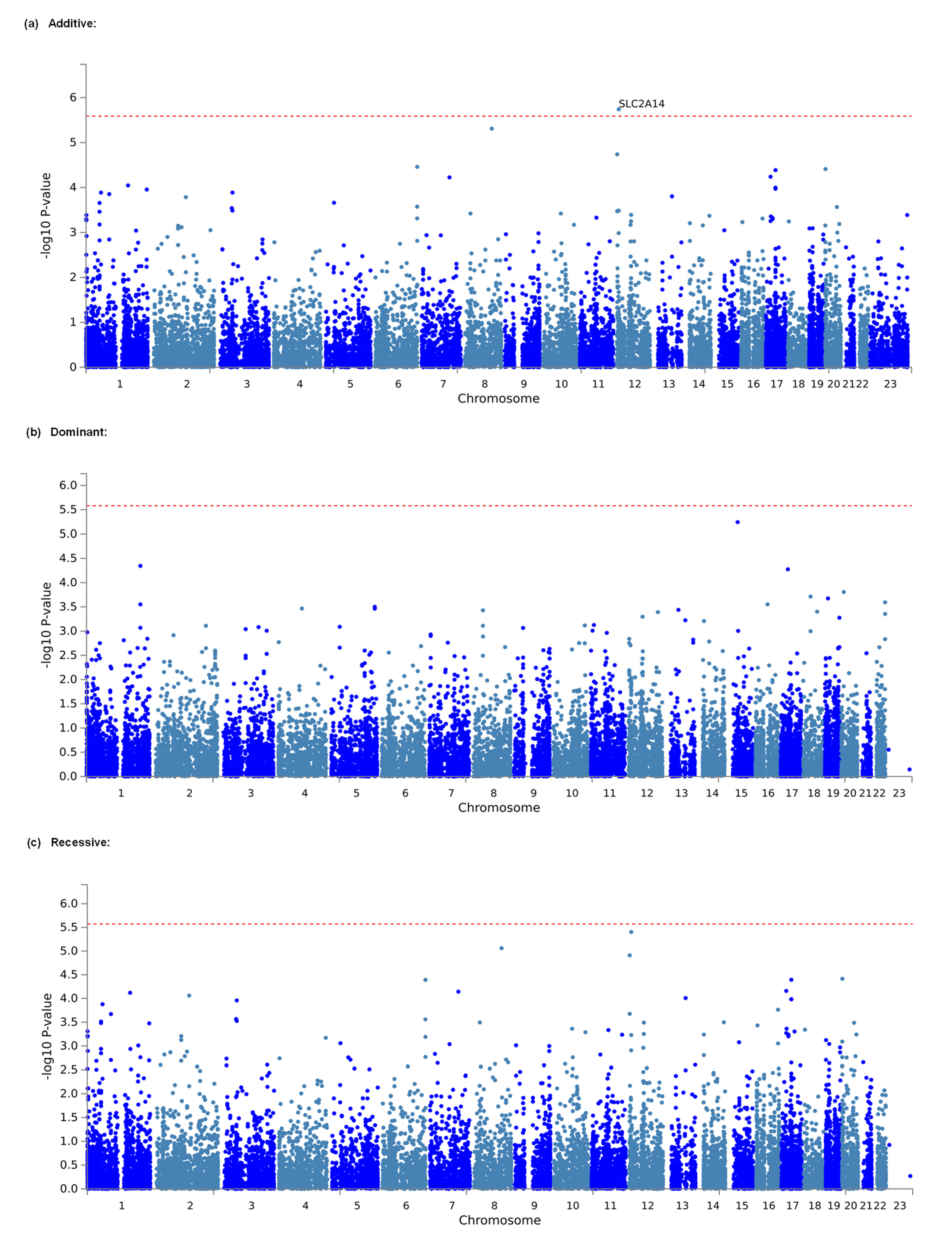
3.2. Identification of Genes and Gene Sets Associated with Opioid Analgesic Requirements in the Treatment of Cancer Pain by Gene-Based and Gene-Set Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Raja, S.N.; Carr, D.B.; Cohen, M.; Finnerup, N.B.; Flor, H.; Gibson, S.; Keefe, F.J.; Mogil, J.S.; Ringkamp, M.; Sluka, K.A.; et al. The revised International Association for the Study of Pain definition of pain: Concepts, challenges, and compromises. Pain 2020, 161, 1976–1982. [Google Scholar] [CrossRef]
- Van den Beuken-van Everdingen, M.H.; Hochstenbach, L.M.; Joosten, E.A.; Tjan-Heijnen, V.C.; Janssen, D.J. Update on prevalence of pain in patients with cancer: Systematic review and meta-analysis. J. Pain Symptom Manag. 2016, 51, 1070–1090. [Google Scholar] [CrossRef]
- WHO Guidelines for the Pharmacological and Radiotherapeutic Management of Cancer Pain in Adults and Adolescents. Available online: https://www.who.int/publications/i/item/9789241550390 (accessed on 9 August 2022).
- Ikeda, K.; Ide, S.; Han, W.; Hayashida, M.; Uhl, G.R.; Sora, I. How individual sensitivity to opiates can be predicted by gene analyses. Trends Pharmacol. Sci. 2005, 26, 311–317. [Google Scholar] [CrossRef]
- Nishizawa, D.; Ikeda, K. Genome-wide association studies and human opioid sensitivity. In Neuropathology of Drug Addictions and Substance Misuse Volume 1: Foundations of Understanding, Tobacco, Alcohol, Cannabinoids and Opioids; Preedy, V.R., Ed.; Academic Press: London, UK, 2016; pp. 909–921. [Google Scholar]
- Angst, M.S.; Phillips, N.G.; Drover, D.R.; Tingle, M.; Galinkin, J.L.; Christians, U.; Swan, G.E.; Lazzeroni, L.C.; Clark, J.D. Opioid pharmacogenomics using a twin study paradigm: Methods and procedures for determining familial aggregation and heritability. Twin Res. Hum. Genet. 2010, 13, 412–425. [Google Scholar] [CrossRef]
- Angst, M.S.; Phillips, N.G.; Drover, D.R.; Tingle, M.; Ray, A.; Swan, G.E.; Lazzeroni, L.C.; Clark, J.D. Pain sensitivity and opioid analgesia: A pharmacogenomic twin study. Pain 2012, 153, 1397–1409. [Google Scholar] [CrossRef]
- Branford, R.; Droney, J.; Ross, J.R. Opioid genetics: The key to personalized pain control? Clin. Genet. 2012, 82, 301–310. [Google Scholar] [CrossRef]
- Droney, J.; Riley, J.; Ross, J. Opioid genetics in the context of opioid switching. Curr. Opin. Supportive Palliat. Care 2012, 6, 10–16. [Google Scholar] [CrossRef]
- Vuilleumier, P.H.; Stamer, U.M.; Landau, R. Pharmacogenomic considerations in opioid analgesia. Pharm. Pers. Med. 2012, 5, 73–87. [Google Scholar] [CrossRef][Green Version]
- Yu, Z.; Wen, L.; Shen, X.; Zhang, H. Effects of the OPRM1 A118G polymorphism (rs1799971) on opioid analgesia in cancer pain: A systematic review and meta-analysis. Clin. J. Pain 2019, 35, 77–86. [Google Scholar] [CrossRef]
- Matic, M.; Jongen, J.L.; Elens, L.; de Wildt, S.N.; Tibboel, D.; Sillevis Smitt, P.A.; van Schaik, R.H. Advanced cancer pain: The search for genetic factors correlated with interindividual variability in opioid requirement. Pharmacogenomics 2017, 18, 1133–1142. [Google Scholar] [CrossRef]
- Yennurajalingam, S.; Astolfi, A.; Indio, V.; Beccaro, M.; Schipani, A.; Yu, R.; Shete, S.; Reyes-Gibby, C.; Lu, Z.; Williams, J.L.; et al. Genetic factors associated with pain severity, daily opioid dose requirement, and pain response among advanced cancer patients receiving supportive care. J. Pain Symptom Manag. 2021, 62, 785–795. [Google Scholar] [CrossRef]
- Yang, G.S.; Barnes, N.M.; Lyon, D.E.; Dorsey, S.G. Genetic variants associated with cancer pain and response to opioid analgesics: Implications for precision pain management. Semin. Oncol. Nurs. 2019, 35, 291–299. [Google Scholar] [CrossRef]
- Daly, A.K. Genome-wide association studies in pharmacogenomics. Nat. Rev. Genet. 2010, 11, 241–246. [Google Scholar] [CrossRef]
- Cook-Sather, S.D.; Li, J.; Goebel, T.K.; Sussman, E.M.; Rehman, M.A.; Hakonarson, H. TAOK3, a novel genome-wide association study locus associated with morphine requirement and postoperative pain in a retrospective pediatric day surgery population. Pain 2014, 155, 1773–1783. [Google Scholar] [CrossRef]
- Fukuda, K.; Hayashida, M.; Ide, S.; Saita, N.; Kokita, Y.; Kasai, S.; Nishizawa, D.; Ogai, Y.; Hasegawa, J.; Nagashima, M.; et al. Association between OPRM1 gene polymorphisms and fentanyl sensitivity in patients undergoing painful cosmetic surgery. Pain 2009, 147, 194–201. [Google Scholar] [CrossRef]
- Nishizawa, D.; Fukuda, K.; Kasai, S.; Hasegawa, J.; Aoki, Y.; Nishi, A.; Saita, N.; Koukita, Y.; Nagashima, M.; Katoh, R.; et al. Genome-wide association study identifies a potent locus associated with human opioid sensitivity. Mol. Psychiatry 2014, 19, 55–62. [Google Scholar] [CrossRef]
- Kurita, G.P.; Sjogren, P.; Ekholm, O.; Kaasa, S.; Loge, J.H.; Poviloniene, I.; Klepstad, P. Prevalence and predictors of cognitive dysfunction in opioid-treated patients with cancer: A multinational study. J. Clin. Oncol. 2011, 29, 1297–1303. [Google Scholar] [CrossRef]
- Galvan, A.; Skorpen, F.; Klepstad, P.; Knudsen, A.K.; Fladvad, T.; Falvella, F.S.; Pigni, A.; Brunelli, C.; Caraceni, A.; Kaasa, S.; et al. Multiple loci modulate opioid therapy response for cancer pain. Clin. Cancer Res. 2011, 17, 4581–4587. [Google Scholar] [CrossRef]
- Terui, T.; Koike, K.; Hirayama, Y.; Kusakabe, T.; Ono, K.; Mihara, H.; Kobayashi, K.; Takahashi, Y.; Nakajima, N.; Kato, J.; et al. Recent advances in palliative cancer care at a regional hospital in Japan. Am. J. Hosp. Palliat. Med. 2014, 31, 717–722. [Google Scholar] [CrossRef]
- Hirayama, Y.; Terui, T.; Kusakabe, T.; Koike, K.; Ono, K.; Kato, J.; Ishitani, K. A survey of patients who were referred to our palliative care division from other hospitals and appeared to have obvious indications for cancer chemotherapies. Am. J. Hosp. Palliat. Care 2014, 31, 804–807. [Google Scholar] [CrossRef]
- McPherson, M.L.; Walker, K.A.; Davis, M.P.; Bruera, E.; Reddy, A.; Paice, J.; Malotte, K.; Lockman, D.K.; Wellman, C.; Salpeter, S.; et al. Safe and appropriate use of methadone in hospice and palliative care: Expert consensus white paper. J. Pain Symptom Manag. 2019, 57, 635–645. [Google Scholar] [CrossRef] [PubMed]
- Quigley, C.; Wiffen, P. A systematic review of hydromorphone in acute and chronic pain. J. Pain Symptom Manag. 2003, 25, 169–178. [Google Scholar] [CrossRef]
- Cherny, N.I.; Fallon, M.T.; Kaasa, S.; Portenoy, R.K.; Currow, D.C. Oxford Textbook of Palliative Medicine, 6th ed.; Oxford University Press: Oxford, UK, 2021. [Google Scholar]
- De Leeuw, C.A.; Mooij, J.M.; Heskes, T.; Posthuma, D. MAGMA: Generalized gene-set analysis of GWAS data. PLoS Comput. Biol. 2015, 11, e1004219. [Google Scholar] [CrossRef]
- Watanabe, K.; Taskesen, E.; van Bochoven, A.; Posthuma, D. Functional mapping and annotation of genetic associations with FUMA. Nat. Commun. 2017, 8, 1826. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef] [PubMed]
- Purcell, S.; Neale, B.; Todd-Brown, K.; Thomas, L.; Ferreira, M.A.; Bender, D.; Maller, J.; Sklar, P.; de Bakker, P.I.; Daly, M.J.; et al. PLINK: A tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 2007, 81, 559–575. [Google Scholar] [CrossRef] [PubMed]
- Barrett, J.C.; Fry, B.; Maller, J.; Daly, M.J. Haploview: Analysis and visualization of LD and haplotype maps. Bioinformatics 2005, 21, 263–265. [Google Scholar] [CrossRef]
- De Bakker, P.I.; Ferreira, M.A.; Jia, X.; Neale, B.M.; Raychaudhuri, S.; Voight, B.F. Practical aspects of imputation-driven meta-analysis of genome-wide association studies. Hum. Mol. Genet. 2008, 17, R122–R128. [Google Scholar] [CrossRef]
- Barsh, G.S.; Copenhaver, G.P.; Gibson, G.; Williams, S.M. Guidelines for genome-wide association studies. PLoS Genet. 2012, 8, e1002812. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, revised ed.; Academic Press: New York, NY, USA, 1977. [Google Scholar]
- Gabriel, S.B.; Schaffner, S.F.; Nguyen, H.; Moore, J.M.; Roy, J.; Blumenstiel, B.; Higgins, J.; DeFelice, M.; Lochner, A.; Faggart, M.; et al. The structure of haplotype blocks in the human genome. Science 2002, 296, 2225–2229. [Google Scholar] [CrossRef] [PubMed]
- GTEx Consortium. The Genotype-Tissue Expression (GTEx) project. Nat. Genet. 2013, 45, 580–585. [Google Scholar] [CrossRef]
- Ward, L.D.; Kellis, M. HaploReg: A resource for exploring chromatin states, conservation, and regulatory motif alterations within sets of genetically linked variants. Nucleic. Acids Res. 2012, 40, D930–D934. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Taylor, J.A. SNPinfo: Integrating GWAS and candidate gene information into functional SNP selection for genetic association studies. Nucleic. Acids Res. 2009, 37, W600–W605. [Google Scholar] [CrossRef] [PubMed]
- Lotsch, J.; Klepstad, P.; Doehring, A.; Dale, O. A GTP cyclohydrolase 1 genetic variant delays cancer pain. Pain 2010, 148, 103–106. [Google Scholar] [CrossRef]
- Chen, J.; Yang, T.; Yu, H.; Sun, K.; Shi, Y.; Song, W.; Bai, Y.; Wang, X.; Lou, K.; Song, Y.; et al. A functional variant in the 3’-UTR of angiopoietin-1 might reduce stroke risk by interfering with the binding efficiency of microRNA 211. Hum. Mol. Genet. 2010, 19, 2524–2533. [Google Scholar] [CrossRef]
- Nathan, C. Points of control in inflammation. Nature 2002, 420, 846–852. [Google Scholar] [CrossRef]
- Elkind, M.S. Inflammation, atherosclerosis, and stroke. Neurologist 2006, 12, 140–148. [Google Scholar] [CrossRef]
- Parikh, S.M. Angiopoietins and Tie2 in vascular inflammation. Curr. Opin. Hematol. 2017, 24, 432–438. [Google Scholar] [CrossRef]
- Suri, C.; Jones, P.F.; Patan, S.; Bartunkova, S.; Maisonpierre, P.C.; Davis, S.; Sato, T.N.; Yancopoulos, G.D. Requisite role of angiopoietin-1, a ligand for the TIE2 receptor, during embryonic angiogenesis. Cell 1996, 87, 1171–1180. [Google Scholar] [CrossRef]
- Bafunno, V.; Firinu, D.; D’Apolito, M.; Cordisco, G.; Loffredo, S.; Leccese, A.; Bova, M.; Barca, M.P.; Santacroce, R.; Cicardi, M.; et al. Mutation of the angiopoietin-1 gene (ANGPT1) associates with a new type of hereditary angioedema. J. Allergy Clin. Immunol. 2018, 141, 1009–1017. [Google Scholar] [CrossRef] [PubMed]
- Converse, M.P.; Sobhanian, M.; Taber, D.J.; Houston, B.A.; Meadows, H.B.; Uber, W.E. Effect of angiotensin II inhibitors on gastrointestinal bleeding in patients with left ventricular assist devices. J. Am. Coll. Cardiol. 2019, 73, 1769–1778. [Google Scholar] [CrossRef] [PubMed]
- Poplawski, M.M.; Mastaitis, J.W.; Mobbs, C.V. Naloxone, but not valsartan, preserves responses to hypoglycemia after antecedent hypoglycemia: Role of metabolic reprogramming in counterregulatory failure. Diabetes 2011, 60, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Leu, J.; Cui, M.H.; Shamoon, H.; Gabriely, I. Hypoglycemia-associated autonomic failure is prevented by opioid receptor blockade. J. Clin. Endocrinol. Metab. 2009, 94, 3372–3380. [Google Scholar] [CrossRef] [PubMed]
- Caprio, S.; Gerety, G.; Tamborlane, W.V.; Jones, T.; Diamond, M.; Jacob, R.; Sherwin, R.S. Opiate blockade enhances hypoglycemic counterregulation in normal and insulin-dependent diabetic subjects. Am. J. Physiol. 1991, 260, E852–E858. [Google Scholar] [CrossRef] [PubMed]
- Pendyala, G.; Periyasamy, P.; Callen, S.; Fox, H.S.; Lisco, S.J.; Buch, S.J. Chronic SIV and morphine treatment increases heat shock protein 5 expression at the synapse. J. Neurovirol. 2015, 21, 592–598. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Freeze, H.H. GLUT14, a duplicon of GLUT3, is specifically expressed in testis as alternative splice forms. Genomics 2002, 80, 553–557. [Google Scholar] [CrossRef]
- Wang, W.; Yu, J.T.; Zhang, W.; Cui, W.Z.; Wu, Z.C.; Zhang, Q.; Tan, L. Genetic association of SLC2A14 polymorphism with Alzheimer’s disease in a Han Chinese population. J. Mol. Neurosci. 2012, 47, 481–484. [Google Scholar] [CrossRef]
- Amir Shaghaghi, M.; Zhouyao, H.; Tu, H.; El-Gabalawy, H.; Crow, G.H.; Levine, M.; Bernstein, C.N.; Eck, P. The SLC2A14 gene, encoding the novel glucose/dehydroascorbate transporter GLUT14, is associated with inflammatory bowel disease. Am. J. Clin. Nutr. 2017, 106, 1508–1513. [Google Scholar] [CrossRef]
- Edenberg, H.J.; Koller, D.L.; Xuei, X.; Wetherill, L.; McClintick, J.N.; Almasy, L.; Bierut, L.J.; Bucholz, K.K.; Goate, A.; Aliev, F.; et al. Genome-wide association study of alcohol dependence implicates a region on chromosome 11. Alcohol. Clin. Exp. Res. 2010, 34, 840–852. [Google Scholar] [CrossRef]
- Nutt, D.J. The role of the opioid system in alcohol dependence. J. Psychopharmacol. 2014, 28, 8–22. [Google Scholar] [CrossRef] [PubMed]
- Michalak, A.; Biala, G. Alcohol dependence: Neurobiology and treatment. Acta Pol. Pharm. 2016, 73, 3–12. [Google Scholar] [PubMed]
- Yoshida, K.; Nishizawa, D.; Ichinomiya, T.; Ichinohe, T.; Hayashida, M.; Fukuda, K.; Ikeda, K. Prediction formulas for individual opioid analgesic requirements based on genetic polymorphism analyses. PLoS ONE 2015, 10, e0116885. [Google Scholar] [CrossRef]
- Cheng, H.; Zeng, R.; Kong, L.; Ding, C.; He, Y.; Zhuang, W.; Sun, Y. Establishment of predicting equation for individual sufentanil dosage postoperatively based on gene polymorphisms. Pain Pract. 2022, 22, 39–46. [Google Scholar] [CrossRef] [PubMed]
| Demographic Data: | n | Minimum | Maximum | Mean | SD | Median | ||
|---|---|---|---|---|---|---|---|---|
| Gender | ||||||||
| male | 213 | |||||||
| female | 215 | |||||||
| Age [years] | 428 | 20 | 94 | 71.59 | 12.33 | 72.50 | ||
| Height [cm] | 423 | 136 | 181 | 157.61 | 8.56 | 158.00 | ||
| Weight [kg] | 421 | 30 | 110 | 50.56 | 11.07 | 50.00 | ||
| Diagnosis (primary disease): | n | Diagnosis (primary disease): | n | |||||
| lung cancer | 81 | breast cancer | 60 | |||||
| stomach cancer | 26 | pancreas cancer | 25 | |||||
| prostate cancer | 25 | colon cancer | 22 | |||||
| bladder cancer | 17 | rectal cancer | 13 | |||||
| hepatocellular cancer | 12 | esophageal cancer | 10 | |||||
| uterus cancer | 9 | ovary cancer | 8 | |||||
| others | 120 | |||||||
| Status and side effects: | absence | presence | ||||||
| somatic pain | 38 | 390 | ||||||
| visceral pain | 346 | 82 | ||||||
| neuropathic pain | 327 | 101 | ||||||
| nausea | 193 | 138 | ||||||
| constipation | 113 | 245 | ||||||
| Breakdown of opioids: | n | morphine | oxicodone | fentanyl | tapentadol | tramadol | methadone | hydromorphone |
| Number of administered patients | 426 | 97 | 209 | 107 | 10 | 31 | 5 | 41 |
| Administration of opioids: | n | Minimum | Maximum | Mean | SD | Median | ||
| Total dose of opioids [mg/kg] | 419 | 0.060241 | 32.407407 | 1.74 | 3.09 | 0.8163265 | ||
| (converted to oral morphine) | ||||||||
| Administration of other drugs: | absence | presence | ||||||
| NSAIDs | 137 | 291 | ||||||
| supplementary analgesics | 171 | 257 | ||||||
| Model | Rank | CHR | SNP | Position | p | Related Gene | Genotype (Patients) | Phenotype (Mean) | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A/A | A/B | B/B | A/A | A/B | B/B | |||||||
| Additive | 1 | 8 | rs1283671 | 107421459 | 3.876 × 10−11 * | ANGPT1 | 5 | 87 | 327 | 2.375 | 0.873 | 0.703 |
| Additive | 2 | 8 | rs1283720 | 107460100 | 4.007 × 10−11 * | ANGPT1 | 5 | 89 | 325 | 2.375 | 0.86 | 0.705 |
| Additive | 3 | 9 | GSA−rs72752701 | 125992274 | 3.879 × 10−8 * | 2 | 50 | 367 | 3.011 | 0.776 | 0.743 | |
| Additive | 4 | 1 | rs3917744 | 169608752 | 2.485 × 10−7 | SELP | 2 | 51 | 366 | 2.832 | 0.778 | 0.744 |
| Additive | 5 | 12 | rs11061996 | 1836333 | 2.846 × 10−7 | CACNA2D4,LRTM2 | 2 | 47 | 370 | 2.774 | 0.714 | 0.752 |
| Additive | 6 | 3 | rs61355450 | 117954317 | 0.000000294 | 2 | 36 | 381 | 2.869 | 0.786 | 0.744 | |
| Additive | 6 | 3 | rs6798512 | 118019819 | 0.000000294 | 2 | 36 | 381 | 2.869 | 0.786 | 0.744 | |
| Additive | 8 | 12 | GSA-rs117577524 | 30549638 | 3.316 × 10−7 | 2 | 27 | 390 | 2.869 | 0.794 | 0.744 | |
| Additive | 9 | 8 | rs4620259 | 108979439 | 0.000000349 | 5 | 74 | 340 | 2.175 | 0.828 | 0.722 | |
| Additive | 10 | 16 | rs74007038 | 6785856 | 5.776 × 10−7 | RBFOX1 | 5 | 79 | 335 | 2.076 | 0.781 | 0.733 |
| Additive | 11 | 19 | rs75017760 | 8940941 | 0.00000115 | MUC16 | 2 | 64 | 353 | 2.883 | 0.708 | 0.755 |
| Additive | 12 | 13 | rs9529111 | 66652839 | 0.000001648 | PCDH9 | 19 | 131 | 269 | 1.426 | 0.672 | 0.752 |
| Additive | 13 | 1 | 1:118813000 | 118270377 | 0.000001653 | 2 | 39 | 378 | 2.917 | 0.805 | 0.741 | |
| Additive | 14 | 11 | rs10501687 | 88799011 | 0.000001725 | GRM5 | 22 | 159 | 237 | 1.307 | 0.768 | 0.7 |
| Additive | 15 | 14 | kgp19644675 | 100077596 | 0.000001896 | EVL | 5 | 57 | 357 | 1.927 | 0.851 | 0.727 |
| Additive | 16 | 10 | rs10749151 | 114017465 | 0.000002292 | 7 | 105 | 307 | 1.833 | 0.704 | 0.752 | |
| Additive | 17 | 6 | rs17059990 | 99980179 | 0.0000023 | MCHR2 | 2 | 65 | 352 | 2.817 | 0.866 | 0.726 |
| Additive | 18 | 2 | 2:42166453 | 41939313 | 0.00000233 | C2orf91 | 35 | 162 | 220 | 1.168 | 0.74 | 0.702 |
| Additive | 19 | 12 | rs1554080 | 128183013 | 0.00000245 | 5 | 70 | 344 | 1.814 | 0.701 | 0.754 | |
| Additive | 20 | 2 | rs11692586 | 134159434 | 0.000003951 | 11 | 102 | 306 | 1.593 | 0.751 | 0.73 | |
| Additive | 21 | 16 | rs75384045 | 55308868 | 0.000004132 | 3 | 52 | 364 | 2.242 | 0.85 | 0.732 | |
| Additive | 22 | 18 | rs117231913 | 79691289 | 0.000004475 | CTDP1 | 3 | 55 | 361 | 2.421 | 0.693 | 0.754 |
| Additive | 23 | 3 | rs73184492 | 189405146 | 0.000005259 | 1 | 53 | 364 | 3.509 | 0.939 | 0.718 | |
| Additive | 24 | 21 | rs2834573 | 34668083 | 0.000005392 | 2 | 93 | 324 | 2.646 | 0.696 | 0.764 | |
| Additive | 25 | 6 | kgp17238235 | 27175279 | 0.000006765 | 4 | 85 | 330 | 1.919 | 0.795 | 0.734 | |
| Additive | 26 | 6 | kgp3351958 | 122754906 | 0.00000681 | 2 | 36 | 380 | 2.838 | 0.709 | 0.751 | |
| Additive | 27 | 11 | rs10831496 | 88824823 | 0.000007262 | GRM5 | 24 | 167 | 228 | 1.251 | 0.757 | 0.707 |
| Additive | 28 | 1 | rs75731751 | 187443485 | 0.000007626 | 1 | 50 | 368 | 3.509 | 0.944 | 0.725 | |
| Additive | 29 | 18 | rs989644 | 4231203 | 0.000007922 | DLGAP1 | 1 | 49 | 368 | 3.509 | 0.784 | 0.742 |
| Additive | 30 | 12 | 12:132932218 | 132355632 | 0.000008031 | 1 | 20 | 395 | 3.509 | 0.78 | 0.743 | |
| Model | Rank | CHR | SNP | Position | p | Related Gene | Genotype (Patients) | Phenotype (Mean) | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A/A | A/B | B/B | A/A | A/B | B/B | |||||||
| Dominant | 1 | 6 | kgp11947181 | 139582822 | 1.592 × 10−8 * | 0 | 2 | 417 | NA | 3.176 | 0.746 | |
| Dominant | 2 | 9 | 9:133013640 | 130251361 | 1.730 × 10−8 * | 0 | 2 | 417 | NA | 3.118 | 0.746 | |
| Dominant | 3 | 10 | rs763315292 | 119912126 | 3.807 × 10−8 * | SEC23IP | 0 | 3 | 416 | NA | 2.62 | 0.744 |
| Dominant | 4 | 6 | 6:80456944 | 79747227 | 1.223 × 10−7 | 0 | 4 | 415 | NA | 2.434 | 0.742 | |
| Dominant | 5 | 7 | 7:118263373 | 118623319 | 2.066 × 10−7 | 0 | 2 | 417 | NA | 2.953 | 0.747 | |
| Dominant | 6 | 10 | rs77717582 | 126393947 | 4.341 × 10−7 | 0 | 23 | 396 | NA | 1.411 | 0.72 | |
| Dominant | 7 | 1 | rs755431252 | 175365091 | 4.979 × 10−7 | TNR | 0 | 3 | 416 | NA | 2.677 | 0.744 |
| Dominant | 8 | 13 | 13:88770953 | 88118698 | 5.634 × 10−7 | 0 | 4 | 415 | NA | 2.109 | 0.745 | |
| Dominant | 9 | 2 | rs63751260 | 47403309 | 0.000001091 | MSH2 | 0 | 2 | 416 | NA | 2.883 | 0.749 |
| Dominant | 10 | 19 | rs146815072 | 32830639 | 0.000001368 | SLC7A9 | 0 | 4 | 415 | NA | 2.289 | 0.743 |
| Dominant | 11 | 5 | rs145778277 | 161311253 | 0.0000014 | GABRB2 | 0 | 13 | 406 | NA | 1.516 | 0.734 |
| Dominant | 12 | 18 | 18:3452222 | 3452225 | 0.000001471 | TGIF1 | 0 | 2 | 415 | NA | 2.859 | 0.75 |
| Dominant | 13 | 9 | GSA-rs10967750 | 27166161 | 0.000001495 | TEK | 0 | 5 | 414 | NA | 1.993 | 0.743 |
| Dominant | 14 | 19 | 19:12311461 | 12200646 | 0.000001562 | LOC100289333 | 1 | 9 | 408 | 2.471 | 1.537 | 0.738 |
| Dominant | 15 | 4 | JHU_4.166326986 | 165405835 | 0.000001738 | CPE | 3 | 54 | 362 | 0.942 | 1.079 | 0.708 |
| Dominant | 16 | 13 | 13:77841745 | 77267610 | 0.000002003 | MYCBP2 | 0 | 4 | 412 | NA | 2.031 | 0.747 |
| Dominant | 17 | 6 | rs192596782 | 155305908 | 0.000002575 | TFB1M | 0 | 3 | 413 | NA | 2.397 | 0.745 |
| Dominant | 18 | 3 | rs117341459 | 39630484 | 0.00000258 | 0 | 4 | 415 | NA | 2.144 | 0.744 | |
| Dominant | 19 | 18 | rs4632226 | 3989188 | 0.000002665 | DLGAP1,DLGAP1-AS4 | 1 | 27 | 390 | 0.179 | 1.287 | 0.722 |
| Dominant | 20 | 5 | kgp5717569 | 172042597 | 0.0000033 | STK10 | 1 | 31 | 386 | 2.125 | 1.136 | 0.725 |
| Dominant | 21 | 8 | GSA-rs147608943 | 30052419 | 0.000003361 | 0 | 5 | 414 | NA | 1.955 | 0.743 | |
| Dominant | 22 | 8 | 8:40320989 | 40463470 | 0.000003413 | 0 | 4 | 414 | NA | 2.163 | 0.739 | |
| Dominant | 23 | 3 | kgp2760268 | 26300600 | 0.000003538 | 0 | 3 | 416 | NA | 2.323 | 0.747 | |
| Dominant | 24 | 1 | kgp15630468 | 18045740 | 0.000003604 | 0 | 12 | 407 | NA | 1.479 | 0.737 | |
| Dominant | 25 | 1 | kgp15515856 | 47636211 | 0.000004042 | 0 | 8 | 411 | NA | 1.59 | 0.742 | |
| Dominant | 26 | 3 | 3:40166299 | 40124808 | 0.00000439 | MYRIP | 0 | 2 | 415 | NA | 2.646 | 0.75 |
| Dominant | 27 | 3 | 3:40027538 | 39986047 | 0.000004617 | MYRIP | 0 | 2 | 417 | NA | 2.646 | 0.749 |
| Dominant | 28 | 3 | 3:149817230 | 150099443 | 0.000004786 | 0 | 4 | 415 | NA | 2.075 | 0.745 | |
| Dominant | 29 | 19 | 19:12306462 | 12195647 | 0.000005029 | LOC100289333 | 1 | 14 | 404 | 2.471 | 1.345 | 0.733 |
| Dominant | 29 | 19 | 19:12352578 | 12241763 | 0.000005029 | 1 | 14 | 404 | 2.471 | 1.345 | 0.733 | |
| Model | Rank | CHR | SNP | Position | p | Related Gene | Genotype (Patients) | Phenotype (Mean) | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A/A | A/B | B/B | A/A | A/B | B/B | |||||||
| Recessive | 1 | 8 | rs1283671 | 107421459 | 7.698 × 10−11 * | ANGPT1 | 5 | 87 | 327 | 2.375 | 0.873 | 0.703 |
| Recessive | 1 | 8 | rs1283720 | 107460100 | 7.698 × 10−11 * | ANGPT1 | 5 | 89 | 325 | 2.375 | 0.86 | 0.705 |
| Recessive | 3 | 9 | GSA-rs72752701 | 125992274 | 3.814 × 10−8 * | 2 | 50 | 367 | 3.011 | 0.776 | 0.743 | |
| Recessive | 4 | 1 | rs3917744 | 169608752 | 2.568 × 10−7 | SELP | 2 | 51 | 366 | 2.832 | 0.778 | 0.744 |
| Recessive | 5 | 12 | rs11061996 | 1836333 | 2.672 × 10−7 | CACNA2D4,LRTM2 | 2 | 47 | 370 | 2.774 | 0.714 | 0.752 |
| Recessive | 6 | 3 | rs61355450 | 117954317 | 3.039 × 10−7 | 2 | 36 | 381 | 2.869 | 0.786 | 0.744 | |
| Recessive | 6 | 3 | rs6798512 | 118019819 | 3.039 × 10−7 | 2 | 36 | 381 | 2.869 | 0.786 | 0.744 | |
| Recessive | 6 | 12 | GSA-rs117577524 | 30549638 | 3.039 × 10−7 | 2 | 27 | 390 | 2.869 | 0.794 | 0.744 | |
| Recessive | 9 | 13 | rs9529111 | 66652839 | 4.469 × 10−7 | PCDH9 | 19 | 131 | 269 | 1.426 | 0.672 | 0.752 |
| Recessive | 10 | 8 | rs4620259 | 108979439 | 5.146 × 10−7 | 5 | 74 | 340 | 2.175 | 0.828 | 0.722 | |
| Recessive | 11 | 16 | rs74007038 | 6785856 | 6.499 × 10−7 | RBFOX1 | 5 | 79 | 335 | 2.076 | 0.781 | 0.733 |
| Recessive | 12 | 19 | rs75017760 | 8940941 | 0.000001066 | MUC16 | 2 | 64 | 353 | 2.883 | 0.708 | 0.755 |
| Recessive | 13 | 1 | 1:118813000 | 118270377 | 0.000001725 | 2 | 39 | 378 | 2.917 | 0.805 | 0.741 | |
| Recessive | 14 | 10 | rs10749151 | 114017465 | 0.000001958 | 7 | 105 | 307 | 1.833 | 0.704 | 0.752 | |
| Recessive | 15 | 12 | rs1554080 | 128183013 | 0.000001979 | 5 | 70 | 344 | 1.814 | 0.701 | 0.754 | |
| Recessive | 16 | 14 | kgp19644675 | 100077596 | 0.000002605 | EVL | 5 | 57 | 357 | 1.927 | 0.851 | 0.727 |
| Recessive | 17 | 6 | rs17059990 | 99980179 | 0.000003439 | MCHR2 | 2 | 65 | 352 | 2.817 | 0.866 | 0.726 |
| Recessive | 18 | 11 | rs10501687 | 88799011 | 0.000003562 | GRM5 | 22 | 159 | 237 | 1.307 | 0.768 | 0.7 |
| Recessive | 19 | 18 | rs117231913 | 79691289 | 0.000003767 | CTDP1 | 3 | 55 | 361 | 2.421 | 0.693 | 0.754 |
| Recessive | 20 | 2 | rs11692586 | 134159434 | 0.00000408 | 11 | 102 | 306 | 1.593 | 0.751 | 0.73 | |
| Recessive | 21 | 21 | rs2834573 | 34668083 | 0.000004617 | 2 | 93 | 324 | 2.646 | 0.696 | 0.764 | |
| Recessive | 22 | 16 | rs75384045 | 55308868 | 0.000005399 | 3 | 52 | 364 | 2.242 | 0.85 | 0.732 | |
| Recessive | 23 | 2 | 2:42166453 | 41939313 | 0.000006107 | C2orf91 | 35 | 162 | 220 | 1.168 | 0.74 | 0.702 |
| Recessive | 24 | 6 | kgp3351958 | 122754906 | 0.00000622 | 2 | 36 | 380 | 2.838 | 0.709 | 0.751 | |
| Recessive | 25 | 6 | kgp17238235 | 27175279 | 0.000007322 | 4 | 85 | 330 | 1.919 | 0.795 | 0.734 | |
| Recessive | 26 | 12 | 12:132932218 | 132355632 | 0.00000773 | 1 | 20 | 395 | 3.509 | 0.78 | 0.743 | |
| Recessive | 27 | 3 | rs73184492 | 189405146 | 0.000008219 | 1 | 53 | 364 | 3.509 | 0.939 | 0.718 | |
| Recessive | 27 | 18 | rs989644 | 4231203 | 0.000008219 | DLGAP1 | 1 | 49 | 368 | 3.509 | 0.784 | 0.742 |
| Recessive | 27 | 19 | rs112363595 | 56660873 | 0.000008219 | 1 | 18 | 399 | 3.509 | 0.707 | 0.748 | |
| Recessive | 30 | 8 | rs4734909 | 91861390 | 0.000009082 | 13 | 120 | 286 | 1.467 | 0.656 | 0.768 | |
| Model | Rank | CHR | Gene Start Position | Gene Stop Position | Gene | nSNPs | Z Statistic | p | P a |
|---|---|---|---|---|---|---|---|---|---|
| Additive | 1 | 12 | 7945108 | 8063744 | SLC2A14 | 30 | 4.6313 | 1.8167 × 10−6 | 0.035151328 * |
| Additive | 2 | 8 | 108241721 | 108530283 | ANGPT1 | 24 | 4.4216 | 4.8995 × 10−6 | 0.094800426 |
| Additive | 3 | 12 | 1909433 | 1965918 | LRTM2 | 19 | 4.1283 | 0.000018275 | 0.353602975 |
| Additive | 4 | 6 | 170131718 | 170201680 | ERMARD | 4 | 3.978 | 0.000034747 | 0.672319703 |
| Additive | 5 | 20 | 1141205 | 1186059 | TMEM74B | 8 | 3.9518 | 0.000038784 | 0.750431616 |
| Additive | 6 | 17 | 39113968 | 39163387 | KRT40 | 7 | 3.938 | 0.000041089 | 0.795031061 |
| Additive | 7 | 17 | 19102674 | 19145839 | AC106017.1 | 2 | 3.8561 | 0.000057607 | 1 |
| Additive | 8 | 7 | 112439202 | 112599971 | C7orf60 | 5 | 3.8481 | 0.000059519 | 1 |
| Additive | 9 | 1 | 169538087 | 169619431 | SELP | 21 | 3.7461 | 0.000089799 | 1 |
| Additive | 10 | 17 | 39094669 | 39143144 | KRT39 | 5 | 3.7175 | 0.00010059 | 1 |
| Additive | 11 | 17 | 39129686 | 39170385 | KRTAP3-3 | 6 | 3.7019 | 0.00010702 | 1 |
| Additive | 12 | 1 | 245113007 | 245310466 | EFCAB2 | 26 | 3.6928 | 0.00011092 | 1 |
| Additive | 13 | 3 | 50106341 | 50176454 | RBM5 | 4 | 3.6532 | 0.00012949 | 1 |
| Additive | 14 | 1 | 60338980 | 60412462 | CYP2J2 | 2 | 3.6525 | 0.00012986 | 1 |
| Additive | 15 | 1 | 93625476 | 93764287 | CCDC18 | 6 | 3.6329 | 0.00014014 | 1 |
| Additive | 16 | 13 | 75838808 | 76076250 | TBC1D4 | 42 | 3.6025 | 0.00015758 | 1 |
| Additive | 17 | 2 | 128828774 | 128973251 | UGGT1 | 6 | 3.5918 | 0.00016422 | 1 |
| Additive | 18 | 5 | 32669176 | 32811819 | NPR3 | 19 | 3.5168 | 0.00021839 | 1 |
| Additive | 19 | 1 | 54477347 | 54539177 | TMEM59 | 3 | 3.5143 | 0.00022045 | 1 |
| Additive | 20 | 6 | 169837307 | 170122159 | WDR27 | 15 | 3.4629 | 0.00026718 | 1 |
| Dominant | 1 | 15 | 41454923 | 41542941 | EXD1 | 5 | 4.3882 | 5.7145 × 10−6 | 0.108884083 |
| Dominant | 2 | 1 | 212983483 | 213040991 | C1orf227 | 5 | 3.9154 | 0.000045129 | 0.859887966 |
| Dominant | 3 | 17 | 28236218 | 28455470 | EFCAB5 | 10 | 3.876 | 0.000053086 | 1 |
| Dominant | 4 | 20 | 9498036 | 9839689 | PAK7 | 86 | 3.6046 | 0.00015634 | 1 |
| Dominant | 5 | 18 | 28936740 | 29014875 | DSG4 | 17 | 3.548 | 0.00019409 | 1 |
| Dominant | 6 | 19 | 12253879 | 12320064 | ZNF136 | 13 | 3.5248 | 0.00021191 | 1 |
| Dominant | 7 | 22 | 51041182 | 51086607 | ARSA | 19 | 3.4757 | 0.00025478 | 1 |
| Dominant | 8 | 16 | 47091614 | 47197908 | NETO2 | 6 | 3.4494 | 0.00028092 | 1 |
| Dominant | 9 | 1 | 213011597 | 213092705 | FLVCR1 | 9 | 3.4493 | 0.00028106 | 1 |
| Dominant | 10 | 5 | 171268553 | 171453877 | FBXW11 | 21 | 3.4178 | 0.00031566 | 1 |
| Dominant | 11 | 4 | 93198402 | 93245329 | RP11-9B6.1 | 3 | 3.3954 | 0.00034267 | 1 |
| Dominant | 12 | 5 | 171449077 | 171635390 | STK10 | 64 | 3.3934 | 0.00034512 | 1 |
| Dominant | 13 | 13 | 50466842 | 50530626 | SPRYD7 | 3 | 3.3782 | 0.00036482 | 1 |
| Dominant | 14 | 8 | 37680786 | 37727422 | BRF2 | 11 | 3.372 | 0.00037312 | 1 |
| Dominant | 15 | 18 | 54794293 | 54837531 | BOD1L2 | 15 | 3.3544 | 0.00039763 | 1 |
| Dominant | 16 | 12 | 116375711 | 116735143 | MED13L | 33 | 3.349 | 0.00040551 | 1 |
| Dominant | 17 | 22 | 51019114 | 51072409 | MAPK8IP2 | 25 | 3.3244 | 0.00044301 | 1 |
| Dominant | 18 | 12 | 55503465 | 55544586 | OR9K2 | 7 | 3.2881 | 0.00050434 | 1 |
| Dominant | 19 | 19 | 57301445 | 57372096 | PEG3 | 14 | 3.2739 | 0.00053034 | 1 |
| Dominant | 20 | 13 | 77598792 | 77921185 | MYCBP2 | 28 | 3.2397 | 0.00059826 | 1 |
| Recessive | 1 | 12 | 7945108 | 8063744 | SLC2A14 | 30 | 4.467 | 3.9655 × 10−6 | 0.073841576 |
| Recessive | 2 | 8 | 108241721 | 108530283 | ANGPT1 | 24 | 4.2961 | 8.6923 × 10−6 | 0.161859318 |
| Recessive | 3 | 12 | 1909433 | 1965918 | LRTM2 | 19 | 4.2181 | 0.000012317 | 0.229354857 |
| Recessive | 4 | 20 | 1141205 | 1186059 | TMEM74B | 8 | 3.9541 | 0.000038414 | 0.715307094 |
| Recessive | 5 | 17 | 39113968 | 39163387 | KRT40 | 7 | 3.9425 | 0.000040318 | 0.750761478 |
| Recessive | 6 | 6 | 170131718 | 170201680 | ERMARD | 4 | 3.9412 | 0.000040543 | 0.754951203 |
| Recessive | 7 | 17 | 19102674 | 19145839 | AC106017.1 | 2 | 3.8099 | 0.000069518 | 1 |
| Recessive | 8 | 7 | 112439202 | 112599971 | C7orf60 | 5 | 3.8012 | 0.000071998 | 1 |
| Recessive | 9 | 1 | 169538087 | 169619431 | SELP | 21 | 3.7878 | 0.000076002 | 1 |
| Recessive | 10 | 2 | 128828774 | 128973251 | UGGT1 | 6 | 3.7536 | 0.000087158 | 1 |
| Recessive | 11 | 13 | 75838808 | 76076250 | TBC1D4 | 42 | 3.7231 | 0.000098378 | 1 |
| Recessive | 12 | 17 | 39129686 | 39170385 | KRTAP3-3 | 6 | 3.7097 | 0.00010375 | 1 |
| Recessive | 13 | 17 | 39094669 | 39143144 | KRT39 | 5 | 3.7068 | 0.00010496 | 1 |
| Recessive | 14 | 3 | 50106341 | 50176454 | RBM5 | 4 | 3.6936 | 0.00011057 | 1 |
| Recessive | 15 | 1 | 60338980 | 60412462 | CYP2J2 | 2 | 3.6468 | 0.00013276 | 1 |
| Recessive | 16 | 16 | 85703690 | 85804735 | C16orf74 | 11 | 3.5785 | 0.00017279 | 1 |
| Recessive | 17 | 12 | 1881123 | 2048002 | CACNA2D4 | 41 | 3.5256 | 0.00021126 | 1 |
| Recessive | 18 | 1 | 93625476 | 93764287 | CCDC18 | 6 | 3.5227 | 0.00021358 | 1 |
| Recessive | 19 | 3 | 47249516 | 47344941 | KIF9 | 3 | 3.4581 | 0.00027197 | 1 |
| Recessive | 20 | 6 | 169837307 | 170122159 | WDR27 | 15 | 3.454 | 0.00027618 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nishizawa, D.; Terui, T.; Ishitani, K.; Kasai, S.; Hasegawa, J.; Nakayama, K.; Ebata, Y.; Ikeda, K. Genome-Wide Association Study Identifies Candidate Loci Associated with Opioid Analgesic Requirements in the Treatment of Cancer Pain. Cancers 2022, 14, 4692. https://doi.org/10.3390/cancers14194692
Nishizawa D, Terui T, Ishitani K, Kasai S, Hasegawa J, Nakayama K, Ebata Y, Ikeda K. Genome-Wide Association Study Identifies Candidate Loci Associated with Opioid Analgesic Requirements in the Treatment of Cancer Pain. Cancers. 2022; 14(19):4692. https://doi.org/10.3390/cancers14194692
Chicago/Turabian StyleNishizawa, Daisuke, Takeshi Terui, Kunihiko Ishitani, Shinya Kasai, Junko Hasegawa, Kyoko Nakayama, Yuko Ebata, and Kazutaka Ikeda. 2022. "Genome-Wide Association Study Identifies Candidate Loci Associated with Opioid Analgesic Requirements in the Treatment of Cancer Pain" Cancers 14, no. 19: 4692. https://doi.org/10.3390/cancers14194692
APA StyleNishizawa, D., Terui, T., Ishitani, K., Kasai, S., Hasegawa, J., Nakayama, K., Ebata, Y., & Ikeda, K. (2022). Genome-Wide Association Study Identifies Candidate Loci Associated with Opioid Analgesic Requirements in the Treatment of Cancer Pain. Cancers, 14(19), 4692. https://doi.org/10.3390/cancers14194692





