Comparing Local and Systemic Control between Partial- and Whole-Breast Radiotherapy in Low-Risk Breast Cancer—A Meta-Analysis of Randomized Trials
Simple Summary
Abstract
1. Introduction
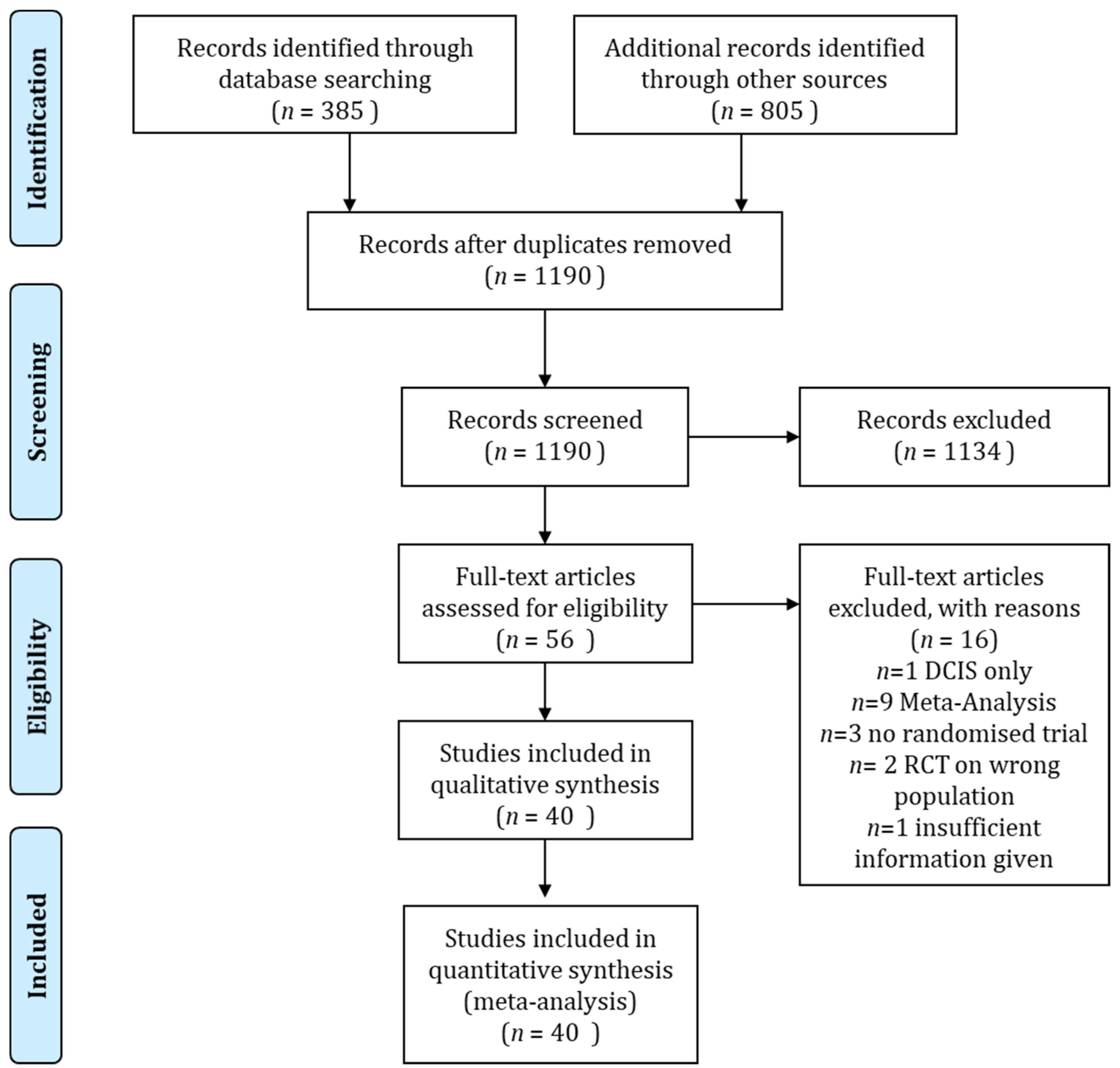
2. Material and Methods
3. Results
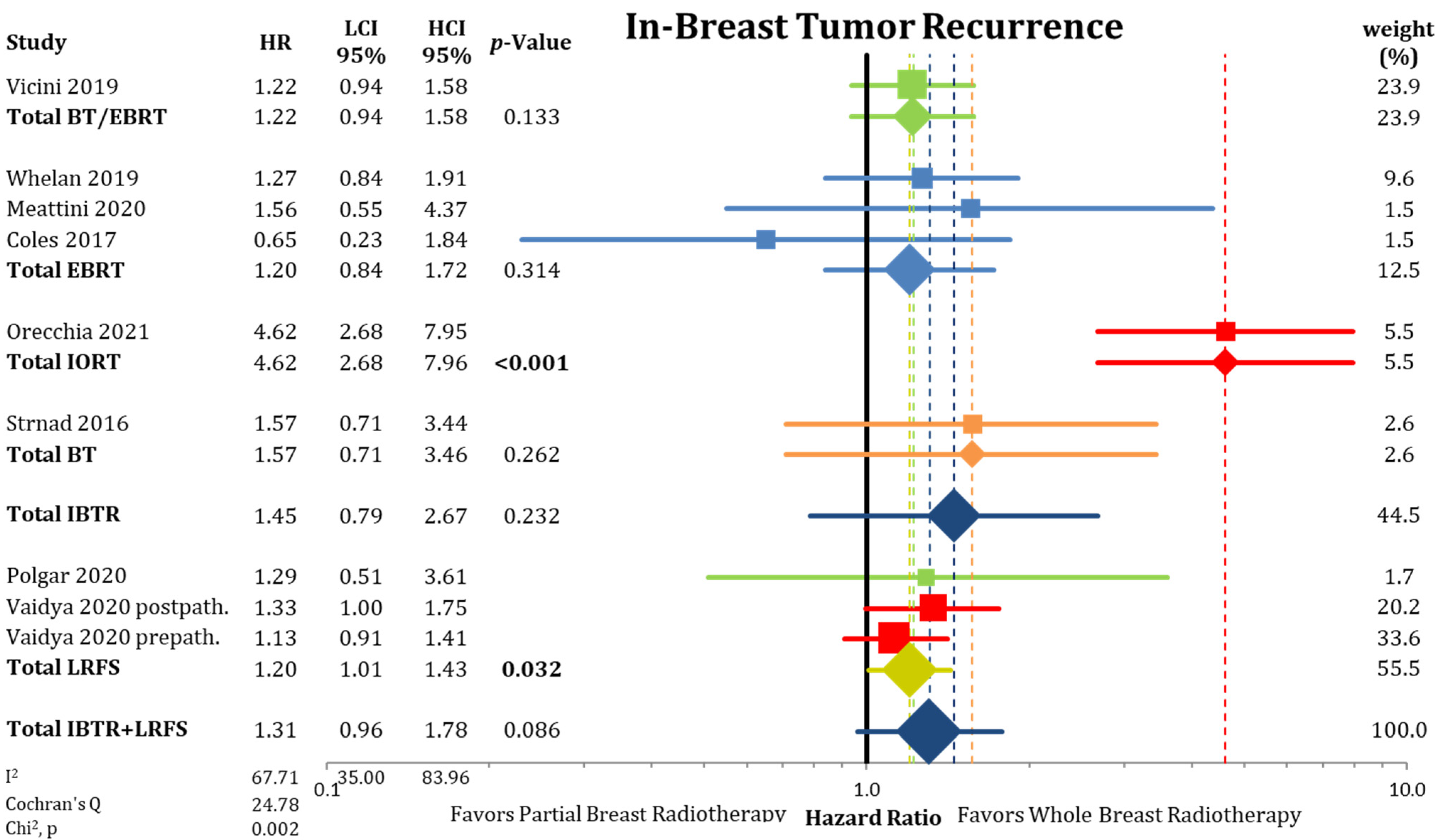
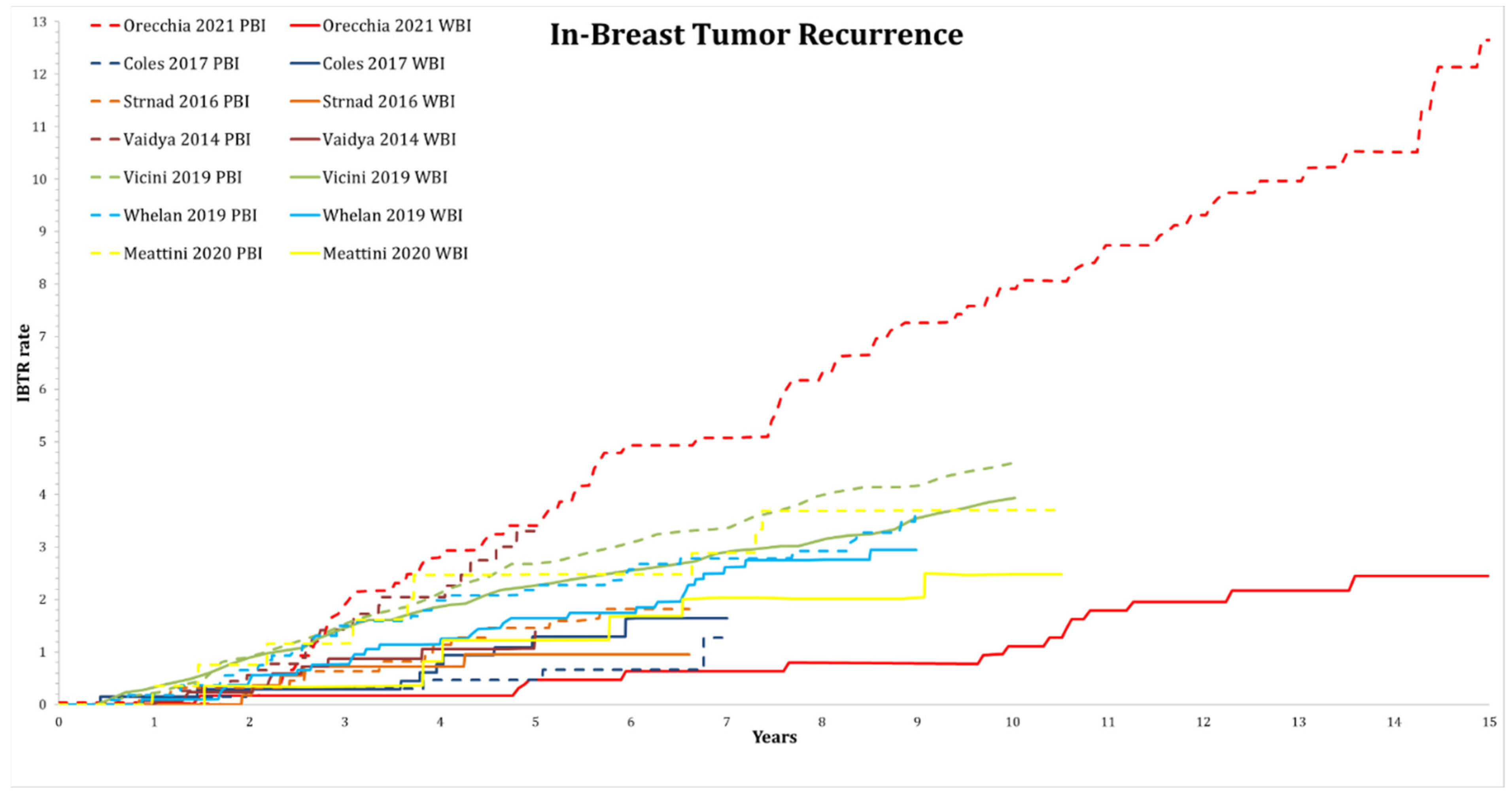
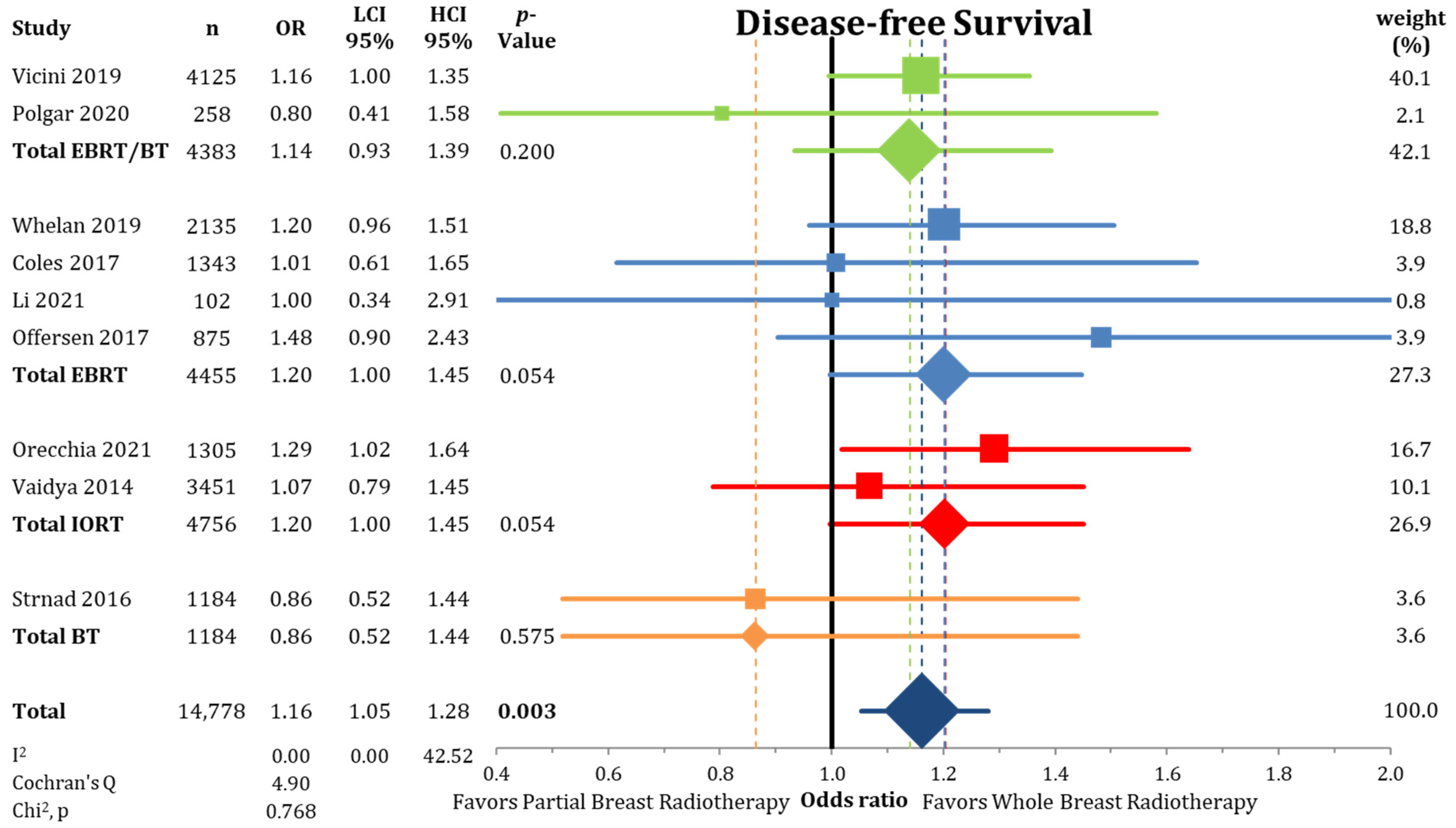
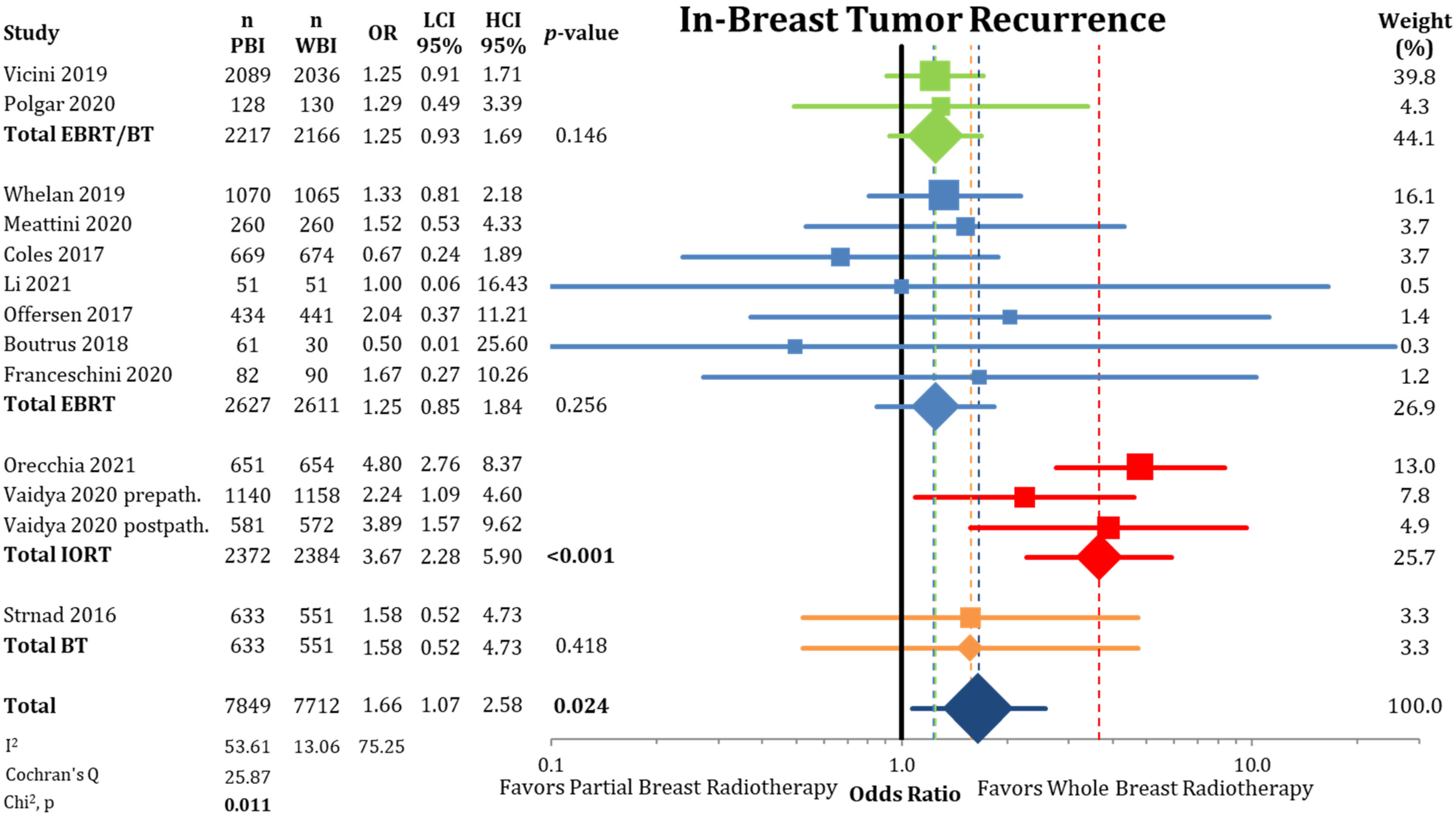
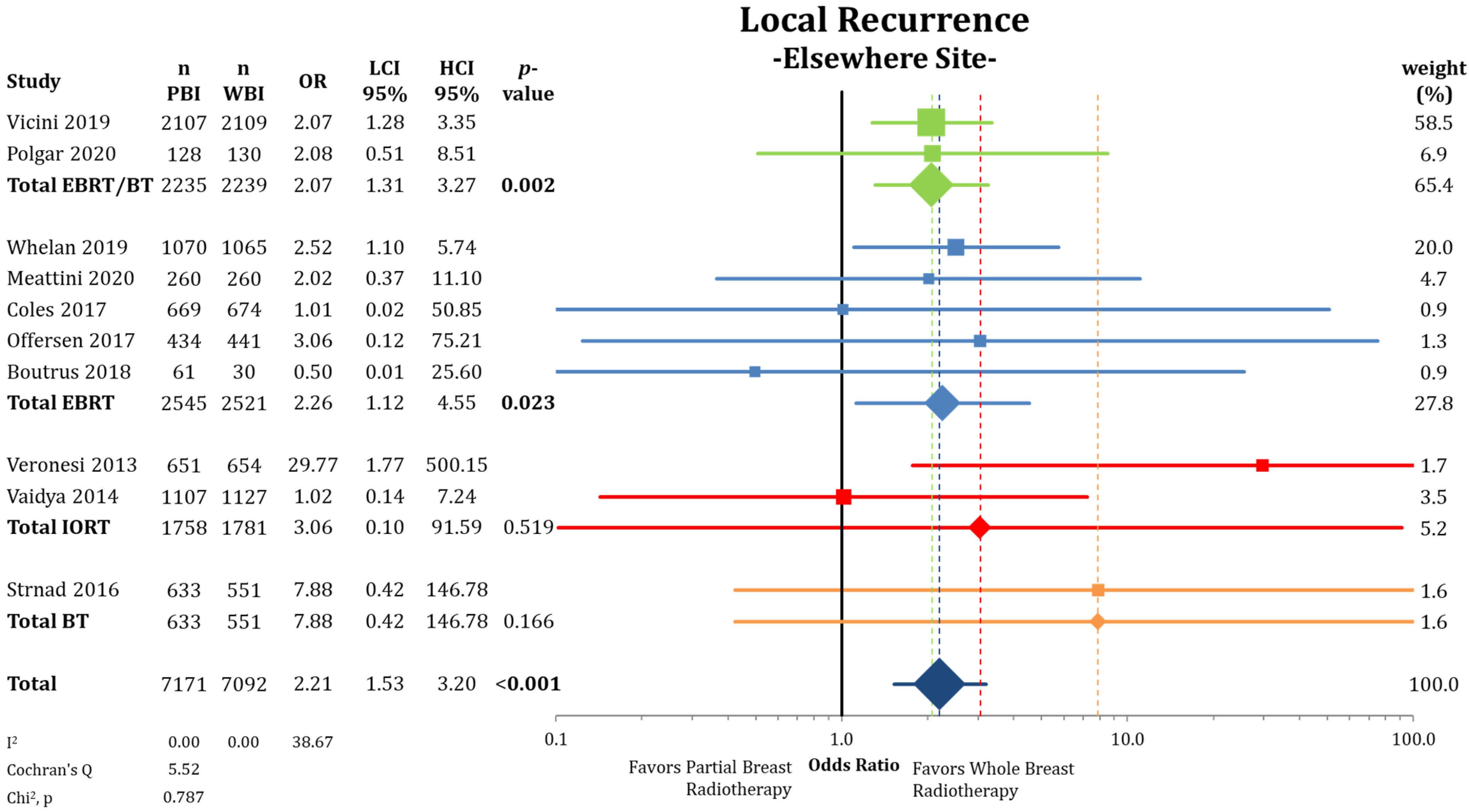
3.1. Local Control
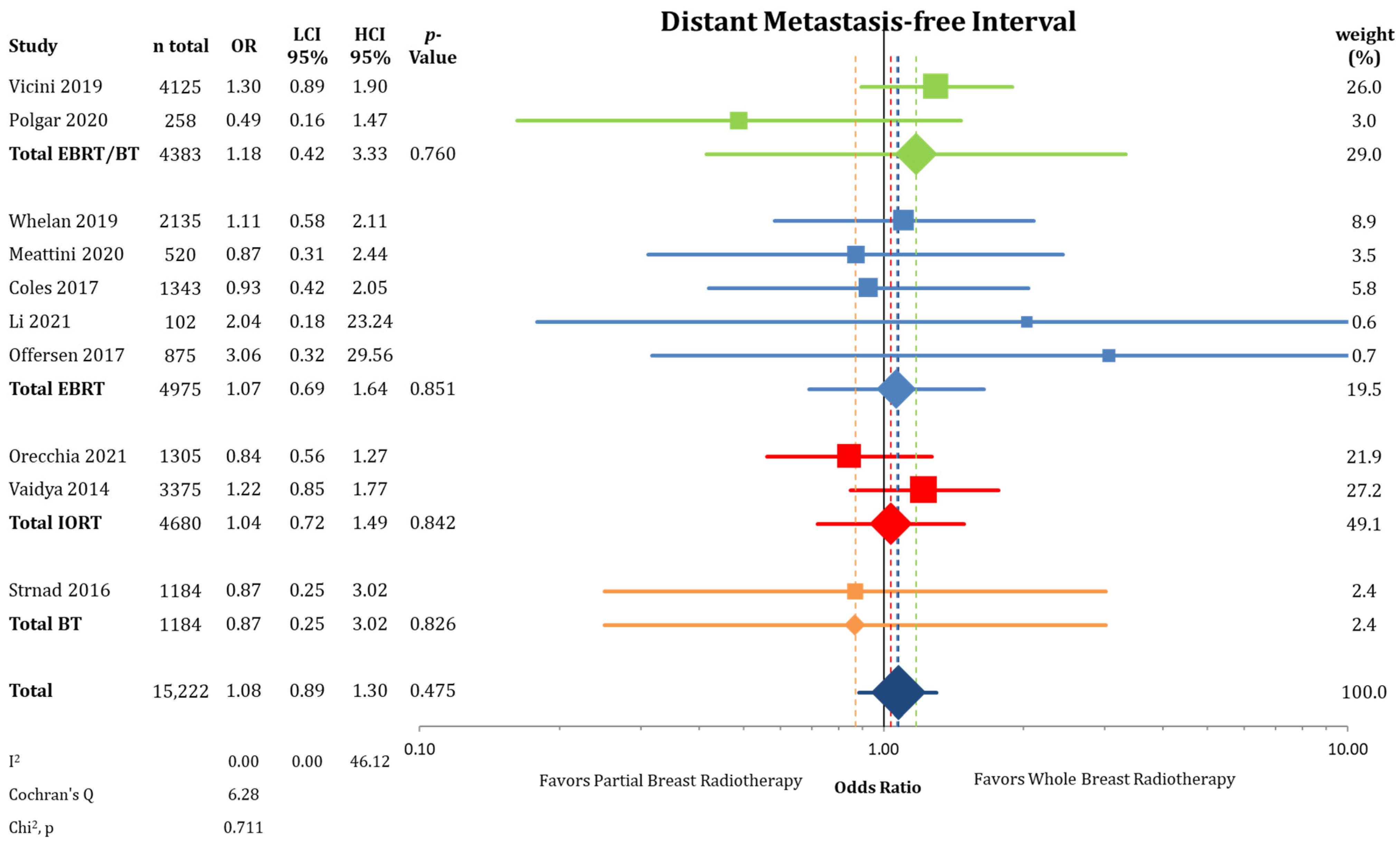
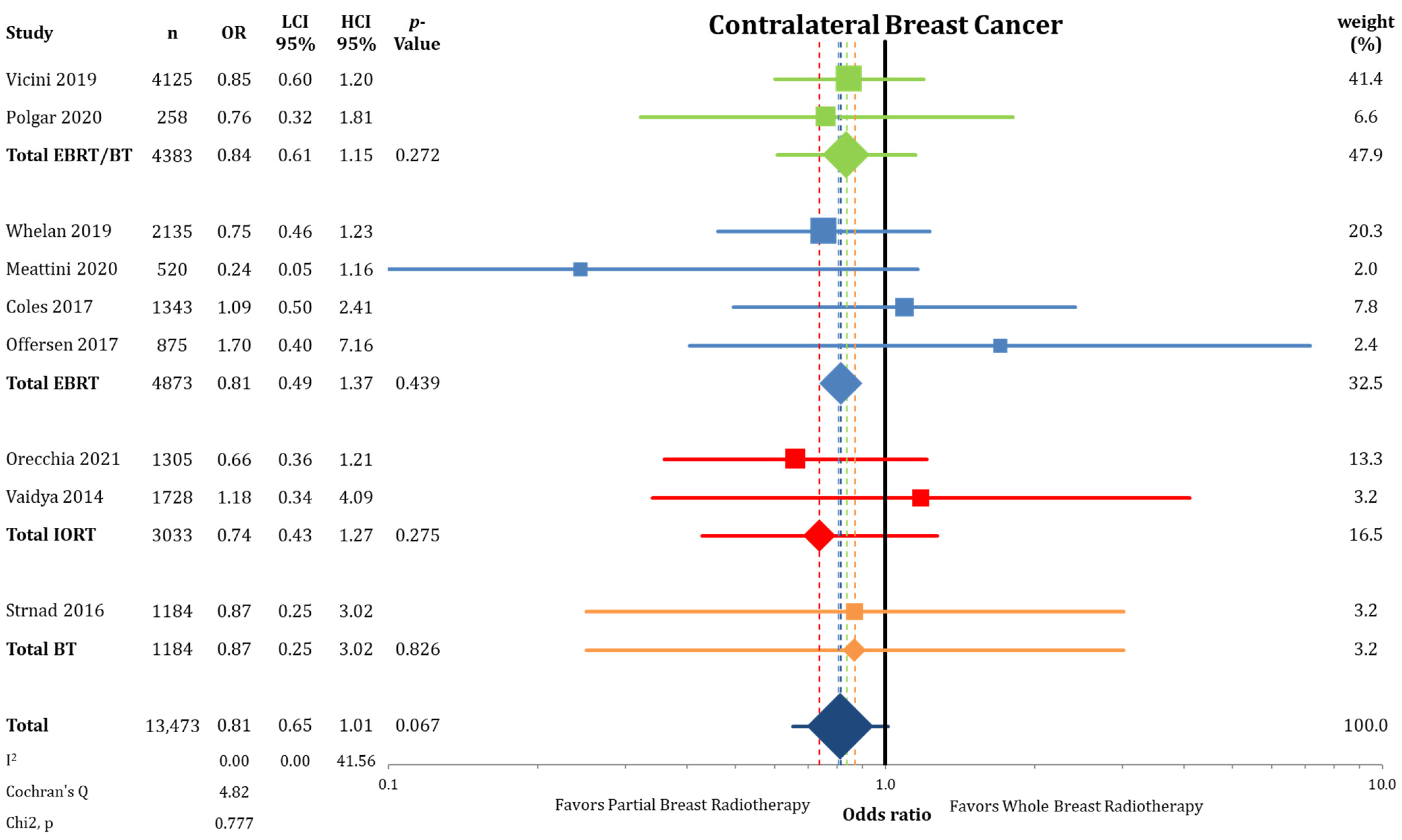
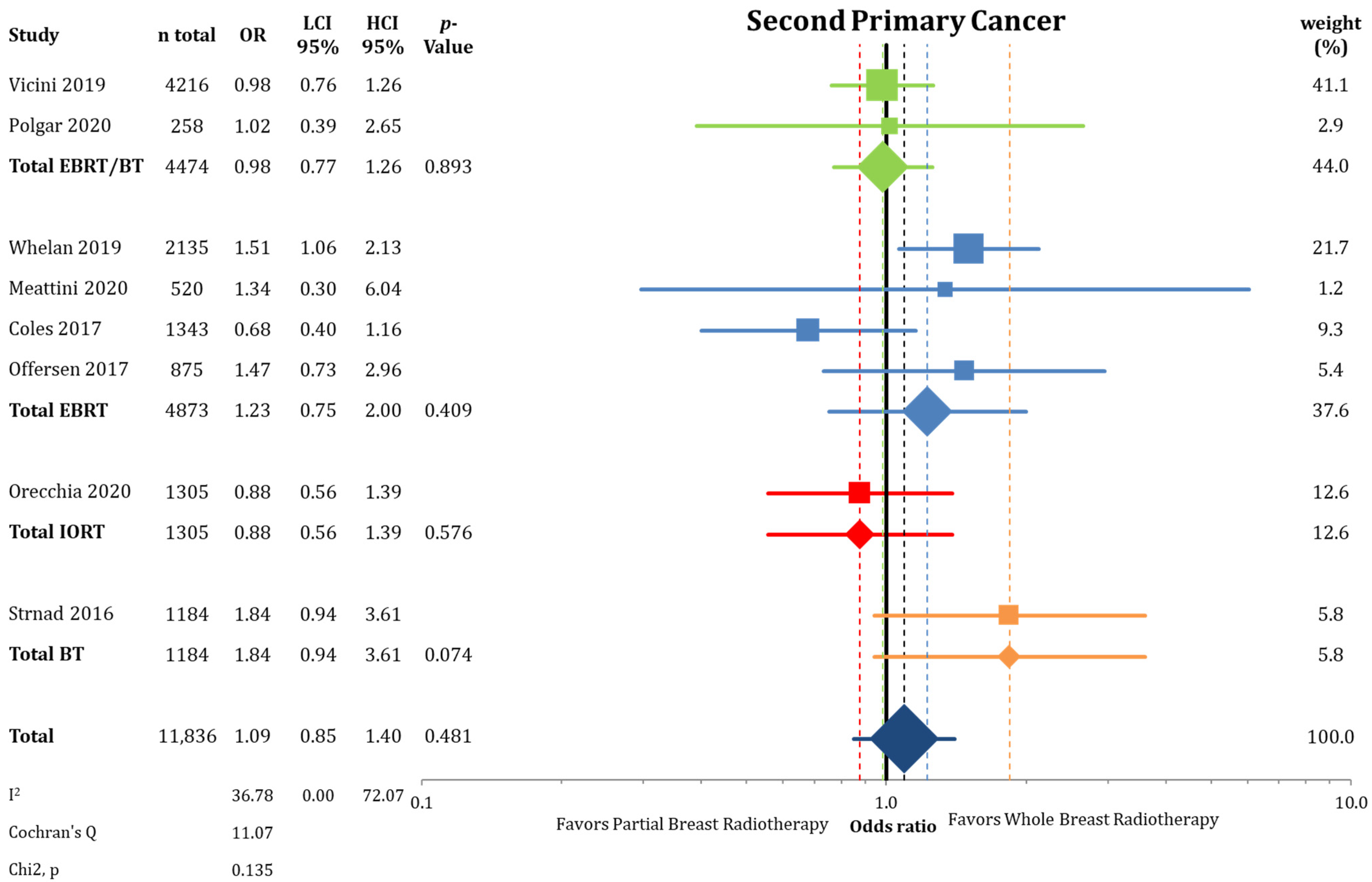
3.2. Other Endpoints
4. Discussion
4.1. Main Results
4.2. Techniques
4.3. Subgroups
4.4. Limitations of Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 3DCRT | 3D conventional radiation therapy |
| BCS | breast-conserving surgery |
| BID | twice daily |
| BT | brachytherapy |
| CTx | chemotherapy |
| DCIS | ductal carcinoma in situ |
| DMFI | distant metastasis-free interval |
| e- | electrons |
| EBRT | external beam radiotherapy |
| EP | endpoint |
| ER | estrogen receptor |
| ET | endocrine therapy |
| FU | follow-up |
| IBC | invasive breast cancer |
| IDC | invasive ductal cancer |
| IBTR | in-breast tumor recurrence |
| IMRT | intensity-modulated radiation therapy |
| IORT | intraoperative radiotherapy |
| ivhet | inverse variance heterogeneity mode |
| HR+ | hormone receptor positive |
| LR | local recurrence |
| LREB | local recurrence elsewhere in the ipsilateral breast |
| LRFS | local recurrence-free survival |
| LRPS | local recurrence at the primary site |
| Med. | median |
| n | number |
| N+ | nodal positive |
| Noninf | non-inferiority |
| n.r. | not reported |
| PBI | partial-breast irradiation |
| Pop | population |
| QD | once daily |
| RT | radiotherapy |
| q.o.d. | every other day |
| SIB | simultaneous integrated boost |
| Stat. | statistical |
| Strat. | stratification |
| WBI | whole-breast irradiation |
| X | photons |
| y | years |
Appendix A
References
- Fisher, B.; Anderson, S.; Bryant, J.; Margolese, R.G.; Deutsch, M.; Fisher, E.R.; Jeong, J.; Wolmark, N. Twenty-Year Follow-up of a Randomized Trial Comparing Total Mastectomy, Lumpectomy, and Lumpectomy plus Irradiation for the Treatment of Invasive Breast Cancer. N. Engl. J. Med. 2002, 347, 1233–1241. [Google Scholar] [CrossRef]
- Veronesi, U.; Marubini, E.; Mariani, L.; Galimberti, V.; Luini, A.; Salvadori, B.; Zucali, R. Radiotherapy after breast-conserving surgery in small breast carcinoma: Long-term results of a randomized trial. Ann. Oncol. 2001, 12, 997–1003. [Google Scholar] [CrossRef]
- Van Dongen, J.A.; Voogd, A.C.; Fentiman, I.S.; Legrand, C.; Sylvester, R.J.; Tong, D.; Van Der Schueren, E.; Helle, P.A.; Van Zijl, K.; Bartelink, H. Long-Term Results of a Randomized Trial Comparing Breast-Conserving Therapy with Mastectomy: European Organization for Research and Treatment of Cancer 10801 Trial. J. Natl. Cancer Inst. 2000, 92, 1143–1150. [Google Scholar] [CrossRef]
- Blichert-Toft, M.; Nielsen, M.; Düring, M.; Møller, S.; Rank, F.; Overgaard, M.; Mouridsen, H.T. Long-term results of breast conserving surgery vs. mastectomy for early stage invasive breast cancer: 20-year follow-up of the Danish randomized DBCG-82TM protocol. Acta Oncol. 2008, 47, 672–681. [Google Scholar] [CrossRef]
- Lichter, A.S.; Lippman, M.E.; Danforth, D.N.; D’Angelo, T.; Steinberg, S.M.; Demoss, E.; Macdonald, H.D.; Reichert, C.M.; Merino, M.; Swain, S.M. Mastectomy versus breast-conserving therapy in the treatment of stage I and II carcinoma of the breast: A randomized trial at the National Cancer Institute. J. Clin. Oncol. 1992, 10, 976–983. [Google Scholar] [CrossRef] [PubMed]
- Darby, S.C.; McGale, P.; Correa, C.R.; Taylor, C.A.; Arriagada, R.; Clarke, M.; Cutter, D.; Davies, C.; Ewertz, M.; Godwin, J.; et al. Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: Meta-analysis of individual patient data for 10 801 women in 17 randomised trials. Lancet 2011, 378, 1707–1716. [Google Scholar] [CrossRef] [PubMed]
- Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: Patient-level meta-analysis of randomised trials. Lancet 2011, 378, 771–784. [Google Scholar] [CrossRef]
- Kunkler, I.H.; Williams, L.J.; Jack, W.J.L.; Cameron, D.A.; Dixon, J.M. Breast-conserving surgery with or without irradiation in women aged 65 years or older with early breast cancer (PRIME II): A randomised controlled trial. Lancet Oncol. 2015, 16, 266–273. [Google Scholar] [CrossRef]
- Williams, L.; Kunkler, I.; King, C.; Jack, W.; Van Der Pol, M. A randomised controlled trial of post-operative radiotherapy following breast-conserving surgery in a minimum-risk population. Quality of life at 5 years in the PRIME trial. Clin. Gov. Int. J. 2011, 16. [Google Scholar] [CrossRef]
- Fyles, A.W.; McCready, D.R.; Manchul, L.A.; Trudeau, M.E.; Merante, P.; Pintilie, M.; Weir, L.M.; Olivotto, I.A. Tamoxifen with or without Breast Irradiation in Women 50 Years of Age or Older with Early Breast Cancer. N. Engl. J. Med. 2004, 351, 963–970. [Google Scholar] [CrossRef]
- Hughes, K.S.; Schnaper, L.A.; Bellon, J.R.; Cirrincione, C.T.; Berry, D.A.; McCormick, B.; Muss, H.B.; Smith, B.L.; Hudis, C.A.; Winer, E.P.; et al. Lumpectomy Plus Tamoxifen with or Without Irradiation in Women Age 70 Years or Older With Early Breast Cancer: Long-Term Follow-Up of CALGB 9343. J. Clin. Oncol. 2013, 31, 2382–2387. [Google Scholar] [CrossRef] [PubMed]
- Hughes, K.S.; Schnaper, L.A.; Berry, D.; Cirrincione, C.; McCormick, B.; Shank, B.; Wheeler, J.; Champion, L.A.; Smith, T.J.; Smith, B.L. Lumpectomy plus tamoxifen with or without irradiation in women 70 years of age or older with early breast cancer. N. Eng. J. Med. 2004, 351, 971–977. [Google Scholar] [CrossRef]
- Fisher, B.; Bryant, J.; Dignam, J.J.; Wickerham, D.L.; Mamounas, E.P.; Fisher, E.R.; Margolese, R.G.; Nesbitt, L.; Paik, S.; Pisansky, T.M.; et al. Tamoxifen, Radiation Therapy, or Both for Prevention of Ipsilateral Breast Tumor Recurrence after Lumpectomy in Women with Invasive Breast Cancers of One Centimeter or Less. J. Clin. Oncol. 2002, 20, 4141–4149. [Google Scholar] [CrossRef] [PubMed]
- Winzer, K.-J.; Sauerbrei, W.; Braun, M.; Liersch, T.; Dunst, J.; Guski, H.; Schumacher, M. Radiation therapy and tamoxifen after breast-conserving surgery: Updated results of a 2 × 2 randomised clinical trial in patients with low risk of recurrence. Eur. J. Cancer 2010, 46, 95–101. [Google Scholar] [CrossRef]
- Blamey, R.; Bates, T.; Chetty, U.; Duffy, S.; Ellis, I.; George, D.; Mallon, E.; Mitchell, M.; Monypenny, I.; Morgan, D.; et al. Radiotherapy or tamoxifen after conserving surgery for breast cancers of excellent prognosis: British Association of Surgical Oncology (BASO) II trial. Eur. J. Cancer 2013, 49, 2294–2302. [Google Scholar] [CrossRef]
- Matuschek, C.; Bölke, E.; Haussmann, J.; Mohrmann, S.; Nestle-Krämling, C.; Gerber, P.A.; Corradini, S.; Orth, K.; Kammers, K.; Budach, W. The benefit of adjuvant radiotherapy after breast conserving surgery in older patients with low risk breast cancer—A meta-analysis of randomized trials. Radiat. Oncol. 2017, 12, 60. [Google Scholar] [CrossRef]
- Chesney, T.R.; Yin, J.X.; Rajaee, N.; Tricco, A.C.; Fyles, A.W.; Acuna, S.A.; Scheer, A.S. Tamoxifen with radiotherapy compared with Tamoxifen alone in elderly women with early-stage breast cancer treated with breast conserving surgery: A systematic review and meta-analysis. Radiother. Oncol. 2017, 123, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.M.; Wilkinson, R.H.; Miceli, P.N.; Macdonald, W.D. Breast Cancer Experiences with Conservation Therapy. Am. J. Clin. Oncol. 1987, 10, 461–468. [Google Scholar] [CrossRef]
- The Uppsala-Orebro Breast Cancer Study Group Sector Resection with or without Postoperative Radiotherapy for Stage I Breast Cancer: A Randomized Trial. J. Natl. Cancer Inst. 1990, 82, 277–282. [CrossRef]
- Goldstein, N.S.; Kestin, L.; Vicini, F.; Goldstein, N.S.; Kestin, L. Factors Associated with Ipsilateral Breast Failure and Distant Metastases in Patients with Invasive Breast Carcinoma Treated with Breast-Conserving Therapy: A Clinicopathologic Study of 607 Neoplasms From 583 Patients. Am. J. Clin. Pathol. 2003, 120, 500–527. [Google Scholar] [CrossRef]
- Faverly, D.; Holland, R.; Burgers, L. An original stereomicroscopic analysis of the mammary glandular tree. Virchows Archiv 1992, 421, 115–119. [Google Scholar] [CrossRef]
- Haffty, B.G.; Carter, D.; Flynn, S.D.; Fischer, D.B.; Brash, D.E.; Simons, J.; Ziegler, A.; Fischer, J.J. Local recurrence versus new primary: Clinical analysis of 82 breast relapses and potential applications for genetic fingerprinting. Int. J. Radiat. Oncol. 1993, 27, 575–583. [Google Scholar] [CrossRef]
- Imamura, H.; Haga, S.; Shimizu, T.; Watanabe, O.; Kinoshita, J.; Nagumo, H.; Kajiwara, T.; Aiba, M. Relationship between the morphological and biological characteristics of intraductal components accompanying invasive ductal breast carcinoma and patient age. Breast Cancer Res. Treat. 2000, 62, 177–184. [Google Scholar] [CrossRef]
- Ohtake, T.; Abe, R.; Kimijima, I.; Fukushima, T.; Tsuchiya, A.; Hoshi, K.; Wakasa, H. Intraductal extension of primary invasive breast carcinoma treated by breast conservative surgery. Computer graphic three-dimensional reconstruction of the mammary duct-lobular systems. Cancer 1995, 76, 32–45. [Google Scholar] [CrossRef]
- Fisher, E.R.; Sass, R.; Fisher, B.; Gregorio, R.; Brown, R.; Wickerham, L. Pathologic findings from the National Surgical Adjuvant Breast Project (protocol 6). II. Relation of local breast recurrence to multicentricity. Cancer 1986, 57, 1717–1724. [Google Scholar] [CrossRef]
- Liljegren, G.; Holmberg, L.; Bergh, J.; Lindgren, A.; Tabár, L.; Nordgren, H.; Adami, H. 10-Year Results After Sector Resection with or without Postoperative Radiotherapy for Stage I Breast Cancer: A Randomized Trial. J. Clin. Oncol. 1999, 17, 2326. [Google Scholar] [CrossRef]
- Clark, R.M.; Whelan, T.; Levine, M.; Roberts, R.; Willan, A.; McCulloch, P.; Lipa, M.; Wilkinson, R.H.; Mahoney, L.J. Randomized Clinical Trial of Breast Irradiation Following Lumpectomy and Axillary Dissection for Node-Negative Breast Cancer: An Update. J. Natl. Cancer Inst. 1996, 88, 1659–1664. [Google Scholar] [CrossRef]
- Malmström, P.; Holmberg, L.; Anderson, H.; Mattsson, J.; Jönsson, P.-E.; Tennvall-Nittby, L.; Balldin, G.; Lovén, L.; Svensson, J.-H.; Ingvar, C.; et al. Breast conservation surgery, with and without radiotherapy, in women with lymph node-negative breast cancer: A randomised clinical trial in a population with access to public mammography screening. Eur. J. Cancer 2003, 39, 1690–1697. [Google Scholar] [CrossRef]
- Holland, R.; Veling, S.H.J.; Mravunac, M.; Hendriks, J.H.C.L. Histologic multifocality of tis, T1–2 breast carcinomas implications for clinical trials of breast-conserving surgery. Cancer 1985, 56, 979–990. [Google Scholar] [CrossRef]
- Magee, B.; Swindell, R.; Harris, M.; Banerjee, S. Prognostic factors for breast recurrence after conservative breast surgery and radiotherapy: Results from a randomised trial. Radiother. Oncol. 1996, 39, 223–227. [Google Scholar] [CrossRef]
- Ribeiro, G.; Magee, B.; Swindell, R.; Harris, M.; Banerjee, S. The christie hospital breast conservation trial: An update at 8 years from inception. Clin. Oncol. 1993, 5, 278–283. [Google Scholar] [CrossRef]
- Haussmann, J.; Budach, W.; Corradini, S.; Krug, D.; Tamaskovics, B.; Bölke, E.; Djiepmo-Njanang, F.-J.; Simiantonakis, I.; Kammers, K.; Matuschek, C. No Difference in Overall Survival and Non-Breast Cancer Deaths after Partial Breast Radiotherapy Compared to Whole Breast Radiotherapy—A Meta-Analysis of Randomized Trials. Cancers 2020, 12, 2309. [Google Scholar] [CrossRef] [PubMed]
- Parmar, M.K.; Torri, V.; Stewart, L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat. Med. 1998, 17, 2815–2834. [Google Scholar] [CrossRef]
- Tierney, J.F.; Stewart, L.A.; Ghersi, D.; Burdett, S.; Sydes, M.R. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials 2007, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Doi, S.A.; Barendregt, J.J.; Khan, S.; Thalib, L.; Williams, G. Advances in the meta-analysis of heterogeneous clinical trials I: The inverse variance heterogeneity model. Contemp. Clin. Trials 2015, 45, 130–138. [Google Scholar] [CrossRef]
- Friedrich, J.O.; Adhikari, N.K.J.; Beyene, J. Inclusion of zero total event trials in meta-analyses maintains analytic consistency and incorporates all available data. BMC Med. Res. Methodol. 2007, 7, 5–6. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Vaidya, J.S.; Bulsara, M.; Saunders, C.; Flyger, H.; Tobias, J.S.; Corica, T.; Massarut, S.; Wenz, F.; Pigorsch, S.; Alvarado, M.; et al. Effect of Delayed Targeted Intraoperative Radiotherapy vs Whole-Breast Radiotherapy on Local Recurrence and Survival: Long-term Results From the TARGIT-A Randomized Clinical Trial in Early Breast Cancer. JAMA Oncol. 2020, 6, e200249. [Google Scholar] [CrossRef]
- Vaidya, J.S.; Wenz, F.; Bulsara, M.; Tobias, J.S.; Joseph, D.J.; Keshtgar, M.; Flyger, H.L.; Massarut, S.; Alvarado, M.; Saunders, C.; et al. Risk-adapted targeted intraoperative radiotherapy versus whole-breast radiotherapy for breast cancer: 5-year results for local control and overall survival from the TARGIT—A randomised trial. Lancet 2014, 383, 603–613. [Google Scholar] [CrossRef]
- Vaidya, J.S.; Wenz, F.; Bulsara, M.; Tobias, J.S.; Joseph, D.J.; Saunders, C.; Brew-Graves, C.; Potyka, I.; Morris, S.; Vaidya, H.J.; et al. An international randomised controlled trial to compare TARGeted Intraoperative radioTherapy (TARGIT) with conventional postoperative radiotherapy after breast-conserving surgery for women with early-stage breast cancer (the TARGIT-A trial). Health Technol. Assess. 2016, 20, 1–188. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, J.S.; Bulsara, M.; Baum, M.; Wenz, F.; Massarut, S.; Pigorsch, S.; Alvarado, M.; Douek, M.; Saunders, C.; Flyger, H.L.; et al. Long term survival and local control outcomes from single dose targeted intraoperative radiotherapy during lumpectomy (TARGIT-IORT) for early breast cancer: TARGIT—A randomised clinical trial. BMJ 2020, 370, m2836. [Google Scholar] [CrossRef]
- Polgár, C.; Major, T.; Takácsi-Nagy, Z.; Fodor, J. Breast-Conserving Surgery Followed by Partial or Whole Breast Irradiation: Twenty-Year Results of a Phase 3 Clinical Study. Int. J. Radiat. Oncol. Biol. Phys. 2021, 109, 998–1006. [Google Scholar] [CrossRef] [PubMed]
- Coles, C.E.; Griffin, C.L.; Kirby, A.M.; Titley, J.; Agrawal, R.K.; Alhasso, A.; Bhattacharya, I.; Brunt, A.M.; Ciurlionis, L.; Chan, C.; et al. Partial-breast radiotherapy after breast conservation surgery for patients with early breast cancer (UK IMPORT LOW trial): 5-year results from a multicentre, randomised, controlled, phase 3, non-inferiority trial. Lancet 2017, 390, 1048–1060. [Google Scholar] [CrossRef]
- Offersen, B.; Nielsen, H.; Thomsen, M.; Jacobsen, E.; Nielsen, M.; Stenbygaard, L.; Pedersen, A.; Krause, M.; Jensen, M.; Overgaard, J. SP-0315: Partial breast radiotherapy after breast conservation for breast cancer: Early results from the randomised DBCG PBI trial. Radiother. Oncol. 2017, 123, S163–S164. [Google Scholar] [CrossRef]
- Boutrus, R.; El Hossieny, H.; El Sherif, S.; Amin, A.; Bayomy, M.; Hashim, T.; Farahat, A.; El-Sebaie, M. Two-Year Results of Once Daily Accelerated Partial Breast Irradiation: A Randomized Controlled Study. Int. J. Radiat. Oncol. 2018, 102, S81. [Google Scholar] [CrossRef]
- Meattini, I.; Marrazzo, L.; Saieva, C.; Desideri, I.; Scotti, V.; Simontacchi, G.; Bonomo, P.; Greto, D.; Mangoni, M.; Scoccianti, S.; et al. Accelerated Partial-Breast Irradiation Compared with Whole-Breast Irradiation for Early Breast Cancer: Long-Term Results of the Randomized Phase III APBI-IMRT-Florence Trial. J. Clin. Oncol. 2020, 38, 4175–4183. [Google Scholar] [CrossRef]
- Franceschini, D.; Loi, M.; Chiola, I.; Arculeo, S.; Marzo, M.; Fernandes, B.; Masci, G.; Torrisi, R.; Tinterri, C.; Testori, A.; et al. Preliminary Results of a Randomized Study on Postmenopausal Women with Early Stage Breast Cancer: Adjuvant Hypofractionated Whole Breast Irradiation Versus Accelerated Partial Breast Irradiation (HYPAB Trial). Clin. Breast Cancer 2020. [Google Scholar] [CrossRef]
- Vicini, F.A.; Cecchini, R.S.; White, J.R.; Arthur, D.W.; Julian, T.B.; Rabinovitch, R.A.; Kuske, R.R.; Ganz, P.A.; Parda, D.S.; Scheier, M.F.; et al. Long-term primary results of accelerated partial breast irradiation after breast-conserving surgery for early-stage breast cancer: A randomised, phase 3, equivalence trial. Lancet 2019, 394, 2155–2164. [Google Scholar] [CrossRef]
- Whelan, T.J.; Julian, J.A.; Berrang, T.S.; Kim, D.-H.; Germain, I.; Nichol, A.M.; Akra, M.; Lavertu, S.; Germain, F.; Fyles, A.; et al. External beam accelerated partial breast irradiation versus whole breast irradiation after breast conserving surgery in women with ductal carcinoma in situ and node-negative breast cancer (RAPID): A randomised controlled trial. Lancet 2019, 394, 2165–2172. [Google Scholar] [CrossRef]
- Rodríguez, N.; Sanz, X.; Dengra, J.; Foro, P.; Membrive, I.; Reig, A.; Quera, J.; Fernández-Velilla, E.; Pera, Ó.; Lio, J.; et al. Five-Year Outcomes, Cosmesis, and Toxicity With 3-Dimensional Conformal External Beam Radiation Therapy to Deliver Accelerated Partial Breast Irradiation. Int. J. Radiat. Oncol. 2013, 87, 1051–1057. [Google Scholar] [CrossRef]
- Vaidya, J.S.; Joseph, D.J.; Tobias, J.S.; Bulsara, M.; Wenz, F.; Saunders, C.; Alvarado, M.; Flyger, H.L.; Massarut, S.; Eiermann, W.; et al. Targeted intraoperative radiotherapy versus whole breast radiotherapy for breast cancer (TARGIT-A trial): An international, prospective, randomised, non-inferiority phase 3 trial. Lancet 2010, 376, 91–102. [Google Scholar] [CrossRef]
- Vicini, F.; Cecchini, R.; White, J.; Julian, T.; Arthur, D.; Rabinovitch, R.; Kuske, R.; Parda, D.; Ganz, P.; Scheier, M.; et al. Abstract GS4-04: Primary results of NSABP B-39/RTOG 0413 (NRG Oncology): A randomized phase III study of conventional whole breast irradiation (WBI) versus partial breast irradiation (PBI) for women with stage 0, I, or II breast cancer. General Session Abstracts 2019, 79, GS4-04. [Google Scholar] [CrossRef]
- White, J.; Winter, K.; Cecchini, R.; Vicini, F.; Arthur, D.; Kuske, R.; Rabinovitch, R.; Sehkon, A.; Khan, A.; Chmura, S.; et al. Cosmetic Outcome from Post Lumpectomy Whole Breast Irradiation (WBI) Versus Partial Breast Irradiation (PBI) on the NRG Oncology/NSABP B39-RTOG 0413 Phase III Clinical Trial. Int. J. Radiat. Oncol. 2019, 105, S3–S4. [Google Scholar] [CrossRef]
- Olivotto, I.A.; Whelan, T.J.; Parpia, S.; Kim, D.-H.; Berrang, T.; Truong, P.T.; Kong, I.; Cochrane, B.; Nichol, A.; Roy, I.; et al. Interim Cosmetic and Toxicity Results From RAPID: A Randomized Trial of Accelerated Partial Breast Irradiation Using Three-Dimensional Conformal External Beam Radiation Therapy. J. Clin. Oncol. 2013, 31, 4038–4045. [Google Scholar] [CrossRef] [PubMed]
- Peterson, D.; Truong, P.T.; Parpia, S.; Olivotto, I.A.; Berrang, T.; Kim, D.-H.; Kong, I.; Germain, I.; Nichol, A.; Akra, M.; et al. Predictors of Adverse Cosmetic Outcome in the RAPID Trial: An Exploratory Analysis. Int. J. Radiat. Oncol. 2015, 91, 968–976. [Google Scholar] [CrossRef]
- Whelan, T.; Julian, J.; Levine, M.; Berrang, T.; Kim, D.-H.; Gu, C.; Germain, I.; Nichol, A.; Akra, M.; Lavertu, S.; et al. Abstract GS4-03: RAPID: A randomized trial of accelerated partial breast irradiation using 3-dimensional conformal radiotherapy (3D-CRT). Gen. Sess. Abstr. 2019, 79, GS4-03. [Google Scholar] [CrossRef]
- Livi, L.; Buonamici, F.B.; Simontacchi, G.; Scotti, V.; Fambrini, M.; Compagnucci, A.; Paiar, F.; Scoccianti, S.; Pallotta, S.; Detti, B.; et al. Accelerated Partial Breast Irradiation with IMRT: New Technical Approach and Interim Analysis of Acute Toxicity in a Phase III Randomized Clinical Trial. Int. J. Radiat. Oncol. 2010, 77, 509–515. [Google Scholar] [CrossRef]
- Livi, L.; Meattini, I.; Marrazzo, L.; Simontacchi, G.; Pallotta, S.; Saieva, C.; Paiar, F.; Scotti, V.; Cardillo, C.D.L.; Bastiani, P.; et al. Accelerated partial breast irradiation using intensity-modulated radiotherapy versus whole breast irradiation: 5-year survival analysis of a phase 3 randomised controlled trial. Eur. J. Cancer 2015, 51, 451–463. [Google Scholar] [CrossRef]
- Meattini, I.; Saieva, C.; Miccinesi, G.; Desideri, I.; Francolini, G.; Scotti, V.; Marrazzo, L.; Pallotta, S.; Meacci, F.; Muntoni, C.; et al. Accelerated partial breast irradiation using intensity modulated radiotherapy versus whole breast irradiation: Health-related quality of life final analysis from the Florence phase 3 trial. Eur. J. Cancer 2017, 76, 17–26. [Google Scholar] [CrossRef]
- Meattini, I.; Saieva, C.; Lucidi, S.; Russo, M.L.; Scotti, V.; Desideri, I.; Marrazzo, L.; Simontacchi, G.; Mangoni, M.; Becherini, C.; et al. Abstract GS4-06: Accelerated partial breast or whole breast irradiation after breast conservation surgery for patients with early breast cancer: 10-year follow up results of the APBI IMRT Florence randomized phase 3 trial. Gen. Sess. Abstr. 2020, 80, GS4-06. [Google Scholar] [CrossRef]
- Orecchia, R.; Veronesi, U.; Maisonneuve, P.; Galimberti, V.E.; Lazzari, R.; Veronesi, P.; Jereczek-Fossa, B.A.; Cattani, F.; Sangalli, C.; Luini, A.; et al. Intraoperative irradiation for early breast cancer (ELIOT): Long-term recurrence and survival outcomes from a single-centre, randomised, phase 3 equivalence trial. Lancet Oncol. 2021, 22, 597–608. [Google Scholar] [CrossRef]
- Veronesi, U.; Orecchia, R.; Maisonneuve, P.; Viale, G.; Rotmensz, N.; Sangalli, C.; Luini, A.; Veronesi, P.; Galimberti, V.; Zurrida, S.; et al. Intraoperative radiotherapy versus external radiotherapy for early breast cancer (ELIOT): A randomised controlled equivalence trial. Lancet Oncol. 2013, 14, 1269–1277. [Google Scholar] [CrossRef]
- Andersen, K.G.; Gärtner, R.; Kroman, N.; Flyger, H.; Kehlet, H. Persistent pain after targeted intraoperative radiotherapy (TARGIT) or external breast radiotherapy for breast cancer: A randomized trial. Breast 2012, 21, 46–49. [Google Scholar] [CrossRef]
- Sperk, E.; Welzel, G.; Keller, A.; Kraus-Tiefenbacher, U.; Gerhardt, A.; Sütterlin, M.; Wenz, F. Late radiation toxicity after intraoperative radiotherapy (IORT) for breast cancer: Results from the randomized phase III trial TARGIT A. Breast Cancer Res. Treat. 2012, 135, 253–260. [Google Scholar] [CrossRef]
- Welzel, G.; Boch, A.; Sperk, E.; Hofmann, F.; Kraus-Tiefenbacher, U.; Gerhardt, A.; Suetterlin, M.; Wenz, F. Radiation-related quality of life parameters after targeted intraoperative radiotherapy versus whole breast radiotherapy in patients with breast cancer: Results from the randomized phase III trial TARGIT-A. Radiat. Oncol. 2013, 8, 9. [Google Scholar] [CrossRef]
- Keshtgar, M.R.S.; Williams, N.R.; Bulsara, M.; Saunders, C.; Flyger, H.; Cardoso, J.S.; Corica, T.; Bentzon, N.; Michalopoulos, N.V.; Joseph, D.J. Objective assessment of cosmetic outcome after targeted intraoperative radiotherapy in breast cancer: Results from a randomised controlled trial. Breast Cancer Res. Treat. 2013, 140, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Corica, T.; Nowak, A.K.; Saunders, C.M.; Bulsara, M.; Taylor, M.; Vaidya, J.S.; Baum, M.; Joseph, D.J. Cosmesis and Breast-Related Quality of Life Outcomes After Intraoperative Radiation Therapy for Early Breast Cancer: A Substudy of the TARGIT-A Trial. Int. J. Radiat. Oncol. 2016, 96, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Corica, T.; Nowak, A.K.; Saunders, C.M.; Bulsara, M.K.; Taylor, M.; Williams, N.R.; Keshtgar, M.; Joseph, D.J.; Vaidya, J.S. Cosmetic outcome as rated by patients, doctors, nurses and BCCT.core software assessed over 5 years in a subset of patients in the TARGIT-A Trial. Radiat. Oncol. 2018, 13, 68. [Google Scholar] [CrossRef] [PubMed]
- Strnad, V.; Ott, O.J.; Hildebrandt, G.; Kauer-Dorner, D.; Knauerhase, H.; Major, T.; Lyczek, J.; Guinot, J.L.; Dunst, J.; Miguelez, C.G.; et al. 5-year results of accelerated partial breast irradiation using sole interstitial multicatheter brachytherapy versus whole-breast irradiation with boost after breast-conserving surgery for low-risk invasive and in-situ carcinoma of the female breast: A randomised, phase 3, non-inferiority trial. Lancet 2016, 387, 229–238. [Google Scholar] [CrossRef]
- Polgár, C.; Ott, O.J.; Hildebrandt, G.; Kauer-Dorner, D.; Knauerhase, H.; Major, T.; Lyczek, J.; Guinot, J.L.; Dunst, J.; Miguelez, C.G.; et al. Late side-effects and cosmetic results of accelerated partial breast irradiation with interstitial brachytherapy versus whole-breast irradiation after breast-conserving surgery for low-risk invasive and in-situ carcinoma of the female breast: 5-year results of a randomised, controlled, phase 3 trial. Lancet Oncol. 2017, 18, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Schäfer, R.; Strnad, V.; Polgár, C.; Uter, W.; Hildebrandt, G.; Ott, O.J.; Kauer-Dorner, D.; Knauerhase, H.; Major, T.; Lyczek, J.; et al. Quality-of-life results for accelerated partial breast irradiation with interstitial brachytherapy versus whole-breast irradiation in early breast cancer after breast-conserving surgery (GEC-ESTRO): 5-year results of a randomised, phase 3 trial. Lancet Oncol. 2018, 19, 834–844. [Google Scholar] [CrossRef]
- Bhattacharya, I.; Haviland, J.S.; Kirby, A.M.; Kirwan, C.C.; Hopwood, P.; Yarnold, J.R.; Bliss, J.; Coles, C.E.; IMPORT Trialists. Patient-Reported Outcomes Over 5 Years After Whole- or Partial-Breast Radiotherapy: Longitudinal Analysis of the IMPORT LOW (CRUK/06/003) Phase III Randomized Controlled Trial. J. Clin. Oncol. 2019, 37, 305–317. [Google Scholar] [CrossRef]
- Bhattacharya, I.S.; Haviland, J.S.; Hopwood, P.; Coles, C.E.; Yarnold, J.R.; Bliss, J.M.; Kirby, A.M.; Trialists, I. Can patient-reported outcomes be used instead of clinician-reported outcomes and photographs as primary endpoints of late normal tissue effects in breast radiotherapy trials? Results from the IMPORT LOW trial. Radiother. Oncol. 2019, 134, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, I.; Haviland, J.S.; Perotti, C.; Eaton, D.; Gulliford, S.; Harris, E.; Coles, C.E.; Kirwan, C.C.; Bliss, J.M.; Kirby, A.M.; et al. Is breast seroma after tumour resection associated with patient-reported breast appearance change following radiotherapy? Results from the IMPORT HIGH (CRUK/06/003) trial. Radiother. Oncol. 2019, 136, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Polgár, C.; Major, T.; Fodor, J.; Németh, G.; Orosz, Z.; Sulyok, Z.; Udvarhelyi, N.; Somogyi, A.; Takácsi-Nagy, Z.; Lövey, K.; et al. High-dose-rate brachytherapy alone versus whole breast radiotherapy with or without tumor bed boost after breast-conserving surgery: Seven-year results of a comparative study. Int. J. Radiat. Oncol. 2004, 60, 1173–1181. [Google Scholar] [CrossRef]
- Polgár, C.; Fodor, J.; Major, T.; Németh, G.; Lövey, K.; Orosz, Z.; Sulyok, Z.; Takácsi-Nagy, Z.; Kásler, M. Breast-Conserving Treatment with Partial or Whole Breast Irradiation for Low-Risk Invasive Breast Carcinoma—5-Year Results of a Randomized Trial. Int. J. Radiat. Oncol. 2007, 69, 694–702. [Google Scholar] [CrossRef]
- Lövey, K.; Fodor, J.; Major, T.; Szabó, É.; Orosz, Z.; Sulyok, Z.; Jánváry, L.; Fröhlich, G.; Kásler, M.; Polgár, C. Fat Necrosis after Partial-Breast Irradiation with Brachytherapy or Electron Irradiation Versus Standard Whole-Breast Radiotherapy—4-Year Results of a Randomized Trial. Int. J. Radiat. Oncol. 2007, 69, 724–731. [Google Scholar] [CrossRef]
- Polgár, C.; Fodor, J.; Major, T.; Sulyok, Z.; Kásler, M. Breast-conserving therapy with partial or whole breast irradiation: Ten-year results of the Budapest randomized trial. Radiother. Oncol. 2013, 108, 197–202. [Google Scholar] [CrossRef]
- Polgár, C.; Major, T.; Sulyok, Z.; Takácsi-Nagy, Z.; Fodor, J. Long-Term Toxicity and Cosmetic Results of Partial Versus Whole Breast Irradiation: 10-Year Results of a Phase III APBI Trial. Int. J. Radiat. Oncol. 2014, 90, S133–S134. [Google Scholar] [CrossRef]
- Li, X.; Sanz, J.; Foro, P.; Martínez, A.; Zhao, M.; Reig, A.; Liu, F.; Huang, Y.; Membrive, I.; Algara, M.; et al. Long-term results of a randomized partial irradiation trial compared to whole breast irradiation in the early stage and low-risk breast cancer patients after conservative surgery. Clin. Transl. Oncol. 2021. [Google Scholar] [CrossRef]
- Gujral, D.M.; Sumo, G.; Owen, J.R.; Ashton, A.; Bliss, J.M.; Haviland, J.; Yarnold, J.R. Ipsilateral Breast Tumor Relapse: Local Recurrence Versus New Primary Tumor and the Effect of Whole-Breast Radiotherapy on the Rate of New Primaries. Int. J. Radiat. Oncol. 2011, 79, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Mannino, M.; Yarnold, J. Accelerated partial breast irradiation trials: Diversity in rationale and design. Radiother. Oncol. 2009, 91, 16–22. [Google Scholar] [CrossRef]
- Jagsi, R.; Chadha, M.; Moni, J.; Ballman, K.; Laurie, F.; Buchholz, T.A.; Giuliano, A.; Haffty, B.G. Radiation Field Design in the ACOSOG Z0011 (Alliance) Trial. J. Clin. Oncol. 2014, 32, 3600–3606. [Google Scholar] [CrossRef]
- Giuliano, A.E.; Ballman, K.V.; McCall, L.; Beitsch, P.D.; Brennan, M.B.; Kelemen, P.R.; Ollila, D.W.; Hansen, N.M.; Whitworth, P.W.; Blumencranz, P.W.; et al. Effect of Axillary Dissection vs No Axillary Dissection on 10-Year Overall Survival Among Women with Invasive Breast Cancer and Sentinel Node Metastasis. JAMA 2017, 318, 918–926. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, G.; Stachs, A.; Gerber, B.; Potenberg, J.; Krug, D.; Wolter, K.; Kühn, T.; Zierhut, D.; Sedlmayer, F.; Kaiser, J.; et al. Central Review of Radiation Therapy Planning Among Patients with Breast-Conserving Surgery: Results from a Quality Assurance Process Integrated into the INSEMA Trial. Int. J. Radiat. Oncol. 2020, 107, 683–693. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.; Correa, C.; Duane, F.K.; Aznar, M.C.; Anderson, S.J.; Bergh, J.; Dodwell, D.; Ewertz, M.; Gray, R.; Jagsi, R.; et al. Estimating the Risks of Breast Cancer Radiotherapy: Evidence from Modern Radiation Doses to the Lungs and Heart and From Previous Randomized Trials. J. Clin. Oncol. 2017, 35, 1641–1649. [Google Scholar] [CrossRef]
- Ramin, C.; Withrow, D.; Lynn, B.D.; Gierach, G.; De González, A.B. Contralateral breast cancer risk according to first breast cancer characteristics among United States women from 1992 to 2015. J. Clin. Oncol. 2019, 37, 1549. [Google Scholar] [CrossRef]
- Cheung, K.J.; Davidson, N.E. Double Trouble: Contralateral Breast Cancer Risk Management in the Modern Era. J. Natl. Cancer Inst. 2019, 111, 641–643. [Google Scholar] [CrossRef]
- Kirby, A.M. Updated ASTRO guidelines on accelerated partial breast irradiation (APBI): To whom can we offer APBI outside a clinical trial? Br. J. Radiol. 2018, 91, 20170565. [Google Scholar] [CrossRef]
- Shah, C.; Badiyan, S.; Ben Wilkinson, J.; Vicini, F.; Beitsch, P.; Keisch, M.; Arthur, D.; Lyden, M. Treatment Efficacy with Accelerated Partial Breast Irradiation (APBI): Final Analysis of the American Society of Breast Surgeons MammoSite® Breast Brachytherapy Registry Trial. Ann. Surg. Oncol. 2013, 20, 3279–3285. [Google Scholar] [CrossRef]
- Strnad, V.; Major, T.; Polgar, C.; Lotter, M.; Guinot, J.-L.; Gutierrez-Miguelez, C.; Galalae, R.; Van Limbergen, E.; Guix, B.; Niehoff, P.; et al. ESTRO-ACROP guideline: Interstitial multi-catheter breast brachytherapy as Accelerated Partial Breast Irradiation alone or as boostx—GEC-ESTRO Breast Cancer Working Group practical recommendations. Radiother. Oncol. 2018, 128, 411–420. [Google Scholar] [CrossRef]
- Correa, C.; Harris, E.E.; Leonardi, M.C.; Smith, B.D.; Taghian, A.G.; Thompson, A.M.; White, J.; Harris, J.R. Accelerated Partial Breast Irradiation: Executive summary for the update of an ASTRO Evidence-Based Consensus Statement. Pract. Radiat. Oncol. 2017, 7, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Shah, C.; Vicini, F.; Shaitelman, S.F.; Hepel, J.; Keisch, M.; Arthur, D.; Khan, A.J.; Kuske, R.; Patel, R.; Wazer, D.E. The American Brachytherapy Society consensus statement for accelerated partial-breast irradiation. Brachytherapy 2018, 17, 154–170. [Google Scholar] [CrossRef] [PubMed]
- Ciervide, R.; Dhage, S.; Guth, A.; Shapiro, R.L.; Axelrod, D.; Roses, D.F.; Formenti, S.C. Five Year Outcome of 145 Patients with Ductal Carcinoma In Situ (DCIS) After Accelerated Breast Radiotherapy. Int. J. Radiat. Oncol. 2012, 83, e159–e164. [Google Scholar] [CrossRef]
- Shah, C.; McGee, M.; Ben Wilkinson, J.; Berry, S.; Grills, I.; Wallace, M.; Mitchell, C.; Vicini, F. Clinical Outcomes Using Accelerated Partial Breast Irradiation in Patients with Ductal Carcinoma In Situ. Clin. Breast Cancer 2012, 12, 259–263. [Google Scholar] [CrossRef]
- Vicini, F.; Shah, C.; Ben Wilkinson, J.; Keisch, M.; Beitsch, P.; Lyden, M. Should Ductal Carcinoma-in-situ (DCIS) Be Removed from the ASTRO Consensus Panel Cautionary Group for Off-protocol Use of Accelerated Partial Breast Irradiation (APBI)? A Pooled Analysis of Outcomes for 300 Patients with DCIS Treated with APBI. Ann. Surg. Oncol. 2013, 20, 1275–1281. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Schneider, F.; Ma, L.; Wenz, F.; Herskind, C. Relative Biologic Effectiveness (RBE) of 50 kV X-rays Measured in a Phantom for Intraoperative Tumor-Bed Irradiation. Int. J. Radiat. Oncol. 2013, 85, 1127–1133. [Google Scholar] [CrossRef] [PubMed]
- Watson, P.G.F.; Bekerat, H.; Papaconstadopoulos, P.; Davis, S.; Seuntjens, J. An investigation into the INTRABEAM miniature x-ray source dosimetry using ionization chamber and radiochromic film measurements. Med Phys. 2018, 45, 4274–4286. [Google Scholar] [CrossRef]
- Kunkler, I.H.; Williams, L.J.; Jack, W.; Cameron, D.A.; Dixon, M. Abstract GS2-03: Prime 2 randomised trial (postoperative radiotherapy in minimum-risk elderly): Wide local excision and adjuvant hormonal therapy +/- whole breast irradiation in women =/> 65 years with early invasive breast cancer: 10 year results. Gen. Ses. Abstr. 2021, 81, GS2-03. [Google Scholar] [CrossRef]
- Haviland, J.S.; Owen, J.R.; Dewar, J.A.; Agrawal, R.K.; Barrett, J.; Barrett-Lee, P.; Dobbs, H.J.; Hopwood, P.; Lawton, P.A.; Magee, B.J.; et al. The UK Standardisation of Breast Radiotherapy (START) trials of radiotherapy hypofractionation for treatment of early breast cancer: 10-year follow-up results of two randomised controlled trials. Lancet Oncol. 2013, 14, 1086–1094. [Google Scholar] [CrossRef]
- Brunt, A.M.; Haviland, J.S.; Wheatley, D.A.; Sydenham, M.A.; Alhasso, A.; Bloomfield, D.J.; Chan, C.; Churn, M.; Cleator, S.; Coles, C.E.; et al. Hypofractionated breast radiotherapy for 1 week versus 3 weeks (FAST-Forward): 5-year efficacy and late normal tissue effects results from a multicentre, non-inferiority, randomised, phase 3 trial. Lancet 2020, 395, 1613–1626. [Google Scholar] [CrossRef]
- Bartelink, H.; Maingon, P.; Poortmans, P.; Weltens, C.; Fourquet, A.; Jager, J.; Schinagl, D.; Oei, B.; Rodenhuis, C.; Horiot, J.-C.; et al. Whole-breast irradiation with or without a boost for patients treated with breast-conserving surgery for early breast cancer: 20-year follow-up of a randomised phase 3 trial. Lancet Oncol. 2015, 16, 47–56. [Google Scholar] [CrossRef]
- Romestaing, P.; Lehingue, Y.; Carrie, C.; Coquard, R.; Montbarbon, X.; Ardiet, J.M.; Mamelle, N.; Gérard, J.P. Role of a 10-Gy boost in the conservative treatment of early breast cancer: Results of a randomized clinical trial in Lyon, France. J. Clin. Oncol. 1997, 15, 963–968. [Google Scholar] [CrossRef]
- Killander, F.; Karlsson, P.; Anderson, H.; Mattsson, J.; Holmberg, E.; Lundstedt, D.; Malmström, P. No breast cancer subgroup can be spared postoperative radiotherapy after breast-conserving surgery. Fifteen-year results from the Swedish Breast Cancer Group randomised trial, SweBCG 91 RT. Eur. J. Cancer 2016, 67, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Polgár, C.; Strnad, V.; Kovács, G. Partial-Breast Irradiation or Whole-Breast Radiotherapy for Early Breast Cancer: A Meta-Analysis of Randomized Trials. Strahlenther. Onkol. 2010, 186, 113–114. [Google Scholar] [CrossRef] [PubMed]
- Valachis, A.; Mauri, D.; Polyzos, N.P.; Mavroudis, D.; Georgoulias, V.; Casazza, G. Partial Breast Irradiation or Whole Breast Radiotherapy for Early Breast Cancer: A Meta-Analysis of Randomized Controlled Trials. Breast J. 2010, 16, 245–251. [Google Scholar] [CrossRef]
- Vaidya, J.S.; Bulsara, M.; Wenz, F.; Coombs, N.; Singer, J.; Ebbs, S.; Massarut, S.; Saunders, C.; Douek, M.; Williams, N.R.; et al. Reduced Mortality with Partial-Breast Irradiation for Early Breast Cancer: A Meta-Analysis of Randomized Trials. Int. J. Radiat. Oncol. 2016, 96, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Hickey, B.; Lehman, M.; Francis, D.P.; See, A.M. Partial breast irradiation for early breast cancer. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef]
- Marta, G.N.; Macedo, C.R.; Carvalho, H.D.A.; Hanna, S.A.; Da Silva, J.L.F.; Riera, R. Accelerated partial irradiation for breast cancer: Systematic review and meta-analysis of 8653 women in eight randomized trials. Radiother. Oncol. 2015, 114, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.; Cheng, J.; Ding, X.; Li, B.; Zhang, J.; Li, H.; Huang, W.; Zhou, T.; Sun, H. Efficacy and Safety of Accelerated Partial Breast Irradiation after Breast-conserving Surgery: A Meta-analysis of Published Comparative Studies. Breast J. 2013, 20, 116–124. [Google Scholar] [CrossRef]
- Ye, X.-P.; Bao, S.; Guo, L.-Y.; Wang, C.-H.; Ma, Y.-P.; Wei, Z.; Chun-Hua, W.; Yan-Fang, Z.; Zhi, F.; Gao, Y.; et al. Accelerated Partial Breast Irradiation for Breast Cancer: A Meta-Analysis. Transl. Oncol. 2013, 6, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Korzets, Y.; Fyles, A.; Shepshelovich, D.; Amir, E.; Goldvaser, H. Toxicity and clinical outcomes of partial breast irradiation compared to whole breast irradiation for early-stage breast cancer: A systematic review and meta-analysis. Breast Cancer Res. Treat. 2019, 175, 531–545. [Google Scholar] [CrossRef] [PubMed]
- Viani, G.A.; Arruda, C.V.; Faustino, A.C.; De Fendi, L.I. Partial-breast irradiation versus whole-breast radiotherapy for early breast cancer: A systematic review and update meta-analysis. Brachytherapy 2020, 19, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Shah, C.; Jia, X.; Hobbs, B.P.; Tendulkar, R.D.; Sittenfeld, S.M.C.; Al-Hilli, Z.; Arthur, D.W.; Keisch, M.E.; Khan, A.J.; Shaitelman, S.F.; et al. Outcomes with Partial Breast Irradiation vs. Whole Breast Irradiation: A Meta-Analysis. Ann. Surg. Oncol. 2021. [Google Scholar] [CrossRef] [PubMed]




| Study | Synonym | Additional Publications | Years of Trial | FU | NTotal | Med. Age | Stat. Setting | Prim. EP | Population | Stratification Factors | PBI Technique | PBI Dose | WBI Dose | G3 | DCIS | N+ | HR+ | Her2+ | CTx | ET | Boost |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vicini 2019 [48] | NSABP B-39 | Vicini et al. 2019 [52] White et al. 2019 [53] | 2005–2013 | 10.2 | 4216 | 54 | Equiv. | IBTR | IBC or DCIS; T < 3 cm, ≤N1; R0; >18 y | Stage, menopausal, ER, CTx | 3DCRT, single- and multicath. BT | 34/3.4; 38.5/3.85 10x in 5–8 d | 50/2; 50.4/1.8; opt. Boost | 26% | 24% | 10% | 81% | n.r. | 29% | n.r. | 80% |
| Whelan 2019 [49] | RAPID | Olivotto et al. 2013 [54] Peterson et al. 2015 [55] Whelan et al. 2019 [56] | 02/2006–07/2011 | 8.6 | 2135 | 61 | noninf. | IBTR | IBC or DCIS; T < 3 cm; R0; N0; >40 y; unifocal | Age >< 50; histology, T >< 1.5 cm; ER, center | 3DCRT IMRT | 38.5/3.85 BID in 5–8 d | 50/2; 42.5/2.66+ opt. Boost | 16% | 18% | 0% | 84% | 6% | 13% | 55% | 21% |
| Meattini 2020 [46] | Florence | Livi et al. 2010 [57] Livi et al. 2015 [58] Meattini et al. 2017 [59] Meattini et al. 2020 [60] | 03/2005–06/2013 | 10.7 | 520 | n.r. | Equiv. | IBTR | IBC or DCIS; T < 2.5 cm; >40 y; BCS+ | None | IMRT | 30/6 q.o.d | 50/2+ opt. Boost 10/2 | 11.4% | 11% | 10% | 96% | 4% | 7% | 62% | n.r. |
| Orecchia 2021 [61] | ELIOT | Veronesi et al. 2013 [62] | 11/2000–12/2007 | 12.4 | 1305 | n.r. | Equiv. | IBTR | IBC; T < 2.5 cm; cN0, R0; 48–75 y; unifocal | T < 1 cm, T 1–1.4 cm, T > 1.5 cm | IORT e- | 21/21 | 50/2+ opt. Boost 10/2 | 20.9% | 0% | 27% | 91% | 3% | 22% | 89% | n.r. |
| Vaidya 2020 [41] | TARGIT-A prepathology | Vaidya et al. 2010 [51] Andersen et al. 2012 [63] Sperk et al. 2012 [64] Welzel et al. 2013 [65] Keshtgar et al. 2013 [66] Vaidya 2014 [39] Corica et al. 2016 [67] Corica et al. 2018 [68] | 03/2000–06/2012 | 8.6 | 2298 | Mean 63 | noninf. | LRFS | IDC; T < 2.5 cm; R0; >45 y; unifocal | Center, timing | IORT x | 20/20 | n.r. | 20% | 0% | ~21% | 90% | 15% | 21% | 81% | 38% |
| Vaidya 2020 [38] | TARGIT-A postpathology | 03/2000–06/2012 | 9 | 1153 | Mean 63 | noninf. | LRFS | IDC; T < 2.5 cm; R0; >45 y; unifocal | Center, timing | IORT x | 20/20 | n.r. | 6% | 3% | 5% | 98% | 6% | 4% | 87% | n.r. | |
| Strnad 2016 [69] | GEC Estro | Polgar et al. 2017 [70] Schäfer et al. 2018 [71] | 04/2004–07/2009 | 6.6 | 1328 | 62 | noninf. | IBTR | IBC or DCIS; T < 3 cm; R0; N0; >40 y; BCS+ | Center, menopausal, stage | multicath. BT | 32/4; 30.3/4.3 or PDR | 50/2; 50.4/1.8; Boost opt. | 8.3% | 5% | 6% | 95% | n.r. | 11% | 90% | 98% |
| Coles 2017 [43] | Import low | Bhattacharya et al. 2019 [72] Bhattacharya et al. 2019 [73] Bhattacharya et al. 2019 [74] | 05/2007–10/2010 | 6 | 1343 | 62 | noninf. | IBTR | IDC; T < 3 cm; >50 y; pN0–1 | Centre | 3DCRT | 40/2.67 QD | 40/2.67 | 9.7% | 0% | 3% | 95% | 4% | 5% | 80% | n.r. |
| Polgar 2020 [42] | Budapest | Polgar et al. 2004 [75] Polgar et al. 2007 [76] Lövey et al. 2007 [77] Polgar et al. 2013 [78] Polgar et al. 2014 [79] | 1998–2004 | 17 | 258 | Mean 59 | noninf. | LR | IBC; T < 2 cm; N0; R0; G1–2; unifocal | None | multicath. BT 3DCRT e- | BT: 36.4/5.2 BID; e-:50/2 QD | 50/2+ opt. 16/2 | 0.0% | 0% | 0% | 88% | n.r. | 3% | 99% | 0.8% |
| Li 2021 [80] | Barcelona | Rodriguez et al. 2013 [50] | 2007–2013 | 10.3 | 102 | Mean 68 | noninf. | IBTR | IBC; T < 3 cm; R0; N0; >60 y; unifocal; G 1–2 | n.r. | 3DCRT | 37.5/3.75 BID | 48/2+ opt. Boost 10/2 or 20/2 | 0.0% | 0% | 0% | 98% | 1% | 3% | 99% | n.r. |
| Offersen 2017 [44] | DBCG PBI | 2009–2016 | 3 | 882 | 66 | noninf. | Breast induration | IBC, T1, R0, >60 y, G1–2, HER2−, pN0 | Center, ET | 3DCRT | 40/2.67 QD | 40/2.67 | <1.0% | 0% | 0% | 100% | 0% | n.r. | 80% | n.r. | |
| Boutrus 2018 [45] | Cairo | n.r. | 2 | 91 | 50 | n.r. | IBTR | IBC; T < 3 cm; R0; N0; >40 y; unifocal; G1–3 | n.r. | 3DCRT | 38.5/3.85 QD 38.5/3.85 BID | 50/2+ opt: Boost | n.r. | 0% | 0% | 80% | n.r. | 19% | n.r. | n.r. | |
| Franceschini 2020 [47] | HYPAB | 01/2015–01/2018 | 3 | 172 | 64 | Cosmesis | T1–2, postmeno, cN0, BCS, ER +, unicentric, R0 > 5 mm, | n.r. | VMAT | 30/6 q.o.d | 40.5/2.7 SIB to 48/3.2 | 3% | 0% | n.r. | 100% | n.r. | n.r. | 97% | 100% |
| Five-Year in-Breast Tumor Recurrence | |||||||
|---|---|---|---|---|---|---|---|
| Study | Rate [%] PBI | Rate [%] WBI | OR | LCI 95% | HCI 95% | Weight (%) | p-Value |
| NSABP B-39 | 2.68 | 2.26 | 1.19 | 0.80 | 1.77 | 42.5 | |
| Budapest | 3.91 | 3.08 | 1.28 | 0.34 | 4.88 | 3.7 | |
| IBTR 5y EBTR/BT | 2.76 | 2.32 | 1.20 | 0.82 | 1.75 | 46.2 | 0.349 |
| RAPID | 2.35 | 1.67 | 1.42 | 0.75 | 2.70 | 15.9 | |
| IMPORT LOW | 0.53 | 1.04 | 0.51 | 0.13 | 1.97 | 3.6 | |
| Florence | 2.31 | 1.15 | 2.02 | 0.50 | 8.18 | 3.4 | |
| IBTR 5y EBTR | 1.70 | 1.41 | 1.17 | 0.44 | 3.15 | 22.9 | 0.749 |
| ELIOT | 4.26 | 0.52 | 8.53 | 2.56 | 28.50 | 4.6 | |
| TARGIT prepath | 2.11 | 0.95 | 2.24 | 1.09 | 4.60 | 12.8 | |
| TARGIT postpath | 3.96 | 1.05 | 3.89 | 1.57 | 9.62 | 8.1 | |
| IBTR 5y IORT | 3.07 | 0.90 | 3.39 | 1.64 | 7.00 | 25.4 | 0.001 |
| GEC ESTRO | 1.57 | 1.02 | 1.55 | 0.52 | 4.65 | 5.5 | |
| IBTR 5y BT | 1.57 | 1.02 | 1.55 | 0.52 | 4.65 | 5.5 | 0.436 |
| Total 5y IBTR | 2.47 | 1.46 | 1.61 | 0.97 | 2.66 | 100.0 | 0.066 |
| I2 | 53.45 | 1.14 | 78.08 | ||||
| Cochran’s Q | 17.19 | ||||||
| Chi2, p | 0.028 | ||||||
| Outcome (Median Follow-Up Range) | Anticipated Absolute Effects | Relative Effect (95% CI) | No. of Participants (Studies) | |||||
|---|---|---|---|---|---|---|---|---|
| Risk with WBI per 100 | Risk with PBI (95% CI) per 100 | |||||||
| In-breast tumor recurrence (2–17 years) | 2.05 | 3.40 | 2.19 | 5.27 | 1.66 | 1.07 | 2.58 | 15,561 (13 RCTs) |
| In-breast tumor recurrence at primary site (2–17 years) | 1.34 | 1.36 | 0.87 | 2.13 | 1.01 | 0.65 | 1.59 | 14,161 (10 RCTs) |
| In-breast tumor recurrence at elsewhere site (2–17 years) | 0.53 | 1.17 | 0.81 | 1.69 | 2.21 | 1.53 | 3.20 | 14,161 (10 RCTs) |
| Regional recurrence (3–17 years) | 0.33 | 0.58 | 0.35 | 0.95 | 1.75 | 1.07 | 2.88 | 15,485 (11 RCTs) |
| Distant metastasis-free interval (3–17 years) | 2.60 | 2.80 | 2.31 | 3.39 | 1.08 | 0.89 | 1.30 | 15,222 (10 RCTs) |
| Disease-free survival (3–17 years) | 11.71 | 13.60 | 12.34 | 15.00 | 1.16 | 1.05 | 1.28 | 14,778 (9 RCTs) |
| Contralateral breast cancer (3–12.4 years) | 2.58 | 2.10 | 1.68 | 2.62 | 0.81 | 0.65 | 1.01 | 13,473 (9 RCTs) |
| Second primary cancer (3–12.4 years) | 5.04 | 5.51 | 4.30 | 7.06 | 1.09 | 0.85 | 1.40 | 11,745 (8 RCTs) |
| Study | HR | LCI 95% | HCI 95% | Weight (%) | p-Value | Study | HR | LCI 95% | HCI 95% | Weight (%) | p-Value |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Age < 50 years | N0 | ||||||||||
| Whelan 2019 [49] | 0.78 | 0.29 | 2.11 | 72.6 | Vicini 2019 [48] | 1.31 | 0.85 | 2.00 | 40.1 | ||
| Livi 2015 [58] | 1.52 | 0.96 | 24.23 | 27.4 | Whelan 2019 [49] | 1.27 | 0.84 | 1.91 | 43.6 | ||
| Total | 0.94 | 0.40 | 2.18 | 100.0 | 0.879 | Livi 2015 [58] | 1.08 | 0.15 | 7.70 | 1.9 | |
| Age > 50 years | Orecchia 2021 [61] | 5.47 | 2.68 | 11.19 | 14.4 | ||||||
| Whelan 2019 [49] | 1.44 | 0.91 | 2.11 | 84.2 | Total | 1.58 | 0.76 | 3.28 | 100 | 0.219 | |
| Livi 2015 [58] | 1.07 | 0.07 | 17.08 | 2.0 | N1 | ||||||
| Coles 2017 [43] | 0.65 | 0.23 | 1.84 | 13.8 | Vicini 2019 [48] | 1.91 | 0.57 | 6.34 | 32.9 | ||
| Total | 1.28 | 0.87 | 1.89 | 100.0 | 0.206 | Orecchia 2021 [61] | 3.41 | 1.47 | 7.92 | 67.1 | |
| Age > 70 years | Total | 2.82 | 1.41 | 5.62 | 100.0 | 0.003 | |||||
| Livi 2015 [58] | 1.07 | 0.07 | 17.08 | 23.1 | Interaction N0/N1 | 0.260 | |||||
| Orecchia 2021 [61] | 1.86 | 0.41 | 8.33 | 76.9 | Grade 1–2 | ||||||
| Total | 1.64 | 0.44 | 6.13 | 100.0 | 0.464 | Whelan 2019 [49] | 1.10 | 0.60 | 2.01 | 54.2 | |
| Interaction Age | 0.732 | Orecchia 2021 [61] | 4.50 | 2.33 | 8.68 | 45.8 | |||||
| DCIS | Total | 2.10 | 0.53 | 8.37 | 100.0 | 0.294 | |||||
| Vicini 2019 [48] | 1.01 | 0.61 | 1.68 | 69.7 | Grade 3 | ||||||
| Whelan 2019 [49] | 1.81 | 0.84 | 3.91 | 30.3 | Whelan 2019 [49] | 1.06 | 0.44 | 2.55 | 42.4 | ||
| Total | 1.21 | 0.69 | 2.11 | 100.0 | 0.513 | Livi 2015 [58] | 1.43 | 0.09 | 22.92 | 4.3 | |
| Invasive Cancer | Orecchia 2021 [61] | 2.18 | 1.00 | 4.79 | 53.3 | ||||||
| Vicini 2019 [48] | 1.37 | 0.91 | 2.05 | 24.3 | Total | 1.58 | 0.89 | 2.79 | 100.0 | 0.118 | |
| Whelan 2019 [49] | 1.12 | 0.69 | 1.84 | 16.4 | Interaction G1–2/G3 | 0.709 | |||||
| Orecchia 2021 [61] | 4.62 | 2.68 | 7.95 | 13.4 | Estrogen Receptor Positive | ||||||
| Vaidya 2014 [39] | 2.13 | 1.01 | 4.49 | 7.1 | Vicini 2019 [48] | 1.32 | 0.91 | 1.92 | 50.5 | ||
| Coles 2017 [43] | 0.65 | 0.23 | 1.84 | 3.7 | Whelan 2019 [49] | 1.19 | 0.69 | 2.07 | 23.3 | ||
| Polgar 2013 [78] | 1.09 | 0.88 | 1.72 | 35.2 | Livi 2015 [58] | 1.79 | 0.80 | 10.69 | 4.2 | ||
| Total | 1.44 | 0.85 | 2.44 | 100.0 | 0.171 | Orecchia 2021 [61] | 4.21 | 2.39 | 7.42 | 21.9 | |
| Interaction DCIS/Invasive | 0.644 | Total | 1.68 | 0.84 | 3.35 | 100.0 | 0.139 | ||||
| T1a/b | Estrogen Receptor Negative | ||||||||||
| Vicini 2019 [48] | 0.58 | 0.27 | 1.22 | 68.2 | Vicini 2019 [48] | 0.98 | 0.54 | 1.77 | 72.6 | ||
| Orecchia 2021 [61] | 4.01 | 1.33 | 12.1 | 31.8 | Whelan 2019 [49] | 1.01 | 0.34 | 3.04 | 21.3 | ||
| Total | 1.07 | 0.15 | 7.82 | 100.0 | 0.945 | Orecchia 2021 [61] | 9.25 | 1.19 | 71.70 | 6.1 | |
| T1c | Total | 1.13 | 0.40 | 3.16 | 100.0 | 0.815 | |||||
| Vicini 2019 [48] | 2.66 | 1.24 | 5.68 | 48.2 | Interaction ER + /ER- | 0.529 | |||||
| Livi 2015 [58] | 1.32 | 0.08 | 21.23 | 3.6 | Her2 Negative | ||||||
| Orecchia 2021 [61] | 4.91 | 2.30 | 10.51 | 48.3 | Orecchia 2021 [61] | 4.35 | 2.47 | 7.64 | 92.3 | ||
| Total | 2.53 | 1.22 | 5.28 | 100.0 | 0.013 | Livi 2015 [58] | 1.13 | 0.16 | 8.02 | 7.7 | |
| T2 | Total | 3.92 | 1.15 | 13.40 | 100.0 | 0.029 | |||||
| Vicini 2019 [48] | 1.34 | 0.52 | 3.46 | 57.9 | ASTRO Risk Group Criteria—Suitable | ||||||
| Orecchia 2021 [61] | 4.80 | 1.58 | 14.58 | 42.1 | Vicini 2019 [48] | 1.12 | 0.46 | 2.76 | 32.0 | ||
| Total | 2.29 | 0.65 | 8.05 | 100.0 | 0.195 | Whelan 2019 [49] | 1.06 | 0.57 | 1.95 | 68.0 | |
| Interaction T1ab/T1c/T2 | 0.729 | Total | 1.08 | 0.65 | 1.79 | 100.0 | 0.769 | ||||
| T < 1.5 cm | ASTRO Risk Group Criteria—Not Suitable | ||||||||||
| Whelan 2019 [49] | 1.02 | 0.59 | 1.75 | 63.7 | Vicini 2019 [48] | 1.26 | 0.77 | 2.08 | 56.6 | ||
| Orecchia 2021 [61] | 4.92 | 2.39 | 10.12 | 36.3 | Whelan 2019 [49] | 1.46 | 0.83 | 2.58 | 43.4 | ||
| Total | 1.80 | 0.37 | 8.85 | 100.0 | 0.467 | Total | 1.34 | 0.92 | 1.95 | 100.0 | 0.122 |
| T > 1.5 cm | Interaction ASTRO Risk Group | 0.495 | |||||||||
| Whelan 2019 [49] | 2.01 | 1.03 | 3.93 | 60.8 | |||||||
| Orecchia 2021 [61] | 4.31 | 1.87 | 9.92 | 39.2 | |||||||
| Total | 2.71 | 1.28 | 5.72 | 100.0 | 0.009 | ||||||
| Interaction T-size 1.5 cm | 0.650 | ||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haussmann, J.; Budach, W.; Strnad, V.; Corradini, S.; Krug, D.; Schmidt, L.; Tamaskovics, B.; Bölke, E.; Simiantonakis, I.; Kammers, K.; et al. Comparing Local and Systemic Control between Partial- and Whole-Breast Radiotherapy in Low-Risk Breast Cancer—A Meta-Analysis of Randomized Trials. Cancers 2021, 13, 2967. https://doi.org/10.3390/cancers13122967
Haussmann J, Budach W, Strnad V, Corradini S, Krug D, Schmidt L, Tamaskovics B, Bölke E, Simiantonakis I, Kammers K, et al. Comparing Local and Systemic Control between Partial- and Whole-Breast Radiotherapy in Low-Risk Breast Cancer—A Meta-Analysis of Randomized Trials. Cancers. 2021; 13(12):2967. https://doi.org/10.3390/cancers13122967
Chicago/Turabian StyleHaussmann, Jan, Wilfried Budach, Vratislav Strnad, Stefanie Corradini, David Krug, Livia Schmidt, Balint Tamaskovics, Edwin Bölke, Ioannis Simiantonakis, Kai Kammers, and et al. 2021. "Comparing Local and Systemic Control between Partial- and Whole-Breast Radiotherapy in Low-Risk Breast Cancer—A Meta-Analysis of Randomized Trials" Cancers 13, no. 12: 2967. https://doi.org/10.3390/cancers13122967
APA StyleHaussmann, J., Budach, W., Strnad, V., Corradini, S., Krug, D., Schmidt, L., Tamaskovics, B., Bölke, E., Simiantonakis, I., Kammers, K., & Matuschek, C. (2021). Comparing Local and Systemic Control between Partial- and Whole-Breast Radiotherapy in Low-Risk Breast Cancer—A Meta-Analysis of Randomized Trials. Cancers, 13(12), 2967. https://doi.org/10.3390/cancers13122967








