New MEMS Tweezers for the Viscoelastic Characterization of Soft Materials at the Microscale
Abstract
:1. Introduction
2. The Adopted Microsystem
3. The Modeling Approach
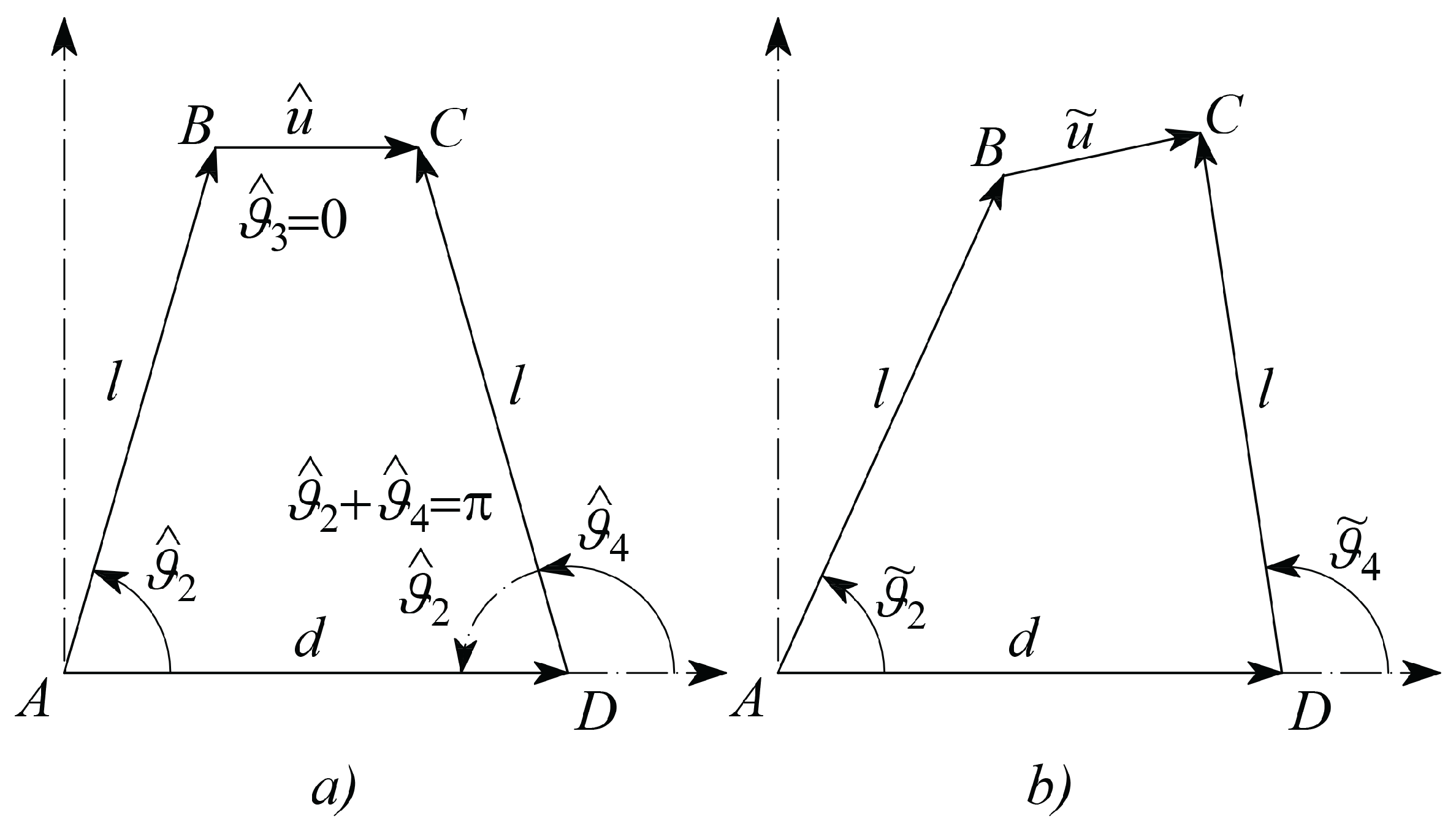
- hat refers to a constant parameter, such as, for example, those referring to the initial configuration, as shown in Figure 5a;
- tilde˜ refers to an actual parameter at the generic configuration, as represented in Figure 5b;
- superscript refers to the desired or target parameter;
- angles of bars are measured counterclockwise, starting from the positive abscissa;
- is the length of vector which is split in the initial length and the deformation ;
- , and refer to the variations of the angular positions of the link vectors , and , with respect to their initial position; in this way their actual absolute angular positions will be , , , respectively;
- (as Figure 5a shows that they are supplementary angles)
- , from geometry represented in Figure 5a;
- is the common length of the two vectors and ;
- is the length of the frame link ;
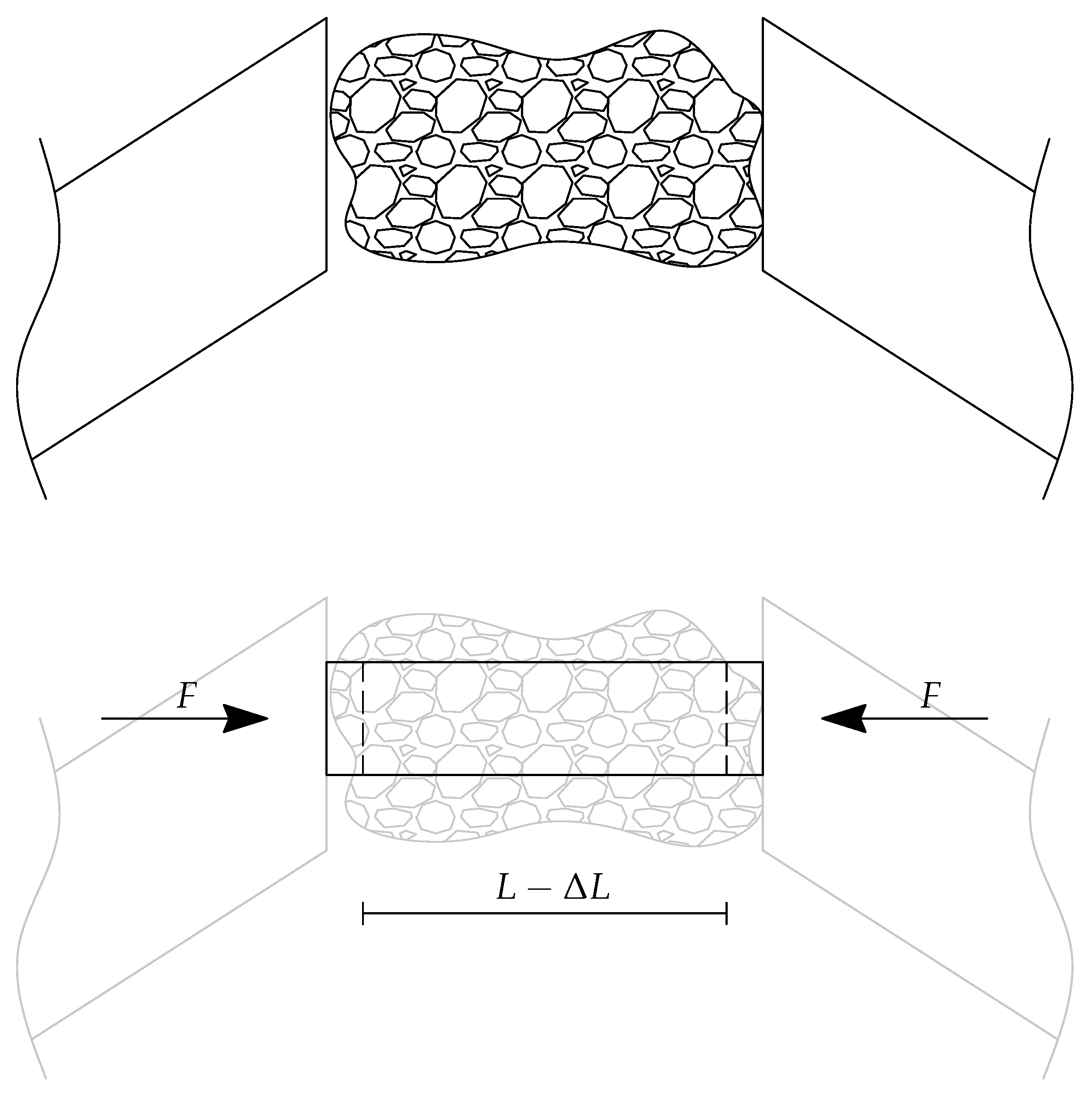
- is the stiffness coefficient of the tissue sample;
- and are the two jaws torsional stiffness, which are related to the CSFH curved beam material and geometry;
- , , and are the radius, width, thickness and beam subtended angle of the CSFH flexure curved beam;
- , and represent the viscous damping coefficients of the sample and of the two jaws;
- and represents the two jaws moments of inertia around and , with ;
- and are the tensions applied to the comb drives;
- , and are the overlap angle, gap and width of the comb drive fingers;
- device-handle gap (silicon oxide layer thickness);
- air viscosity at 25 °C;
- polar moment of area exposed to air viscous damping, calculated around the rotation points;
- equivalent radius employed to model the air viscous damping.
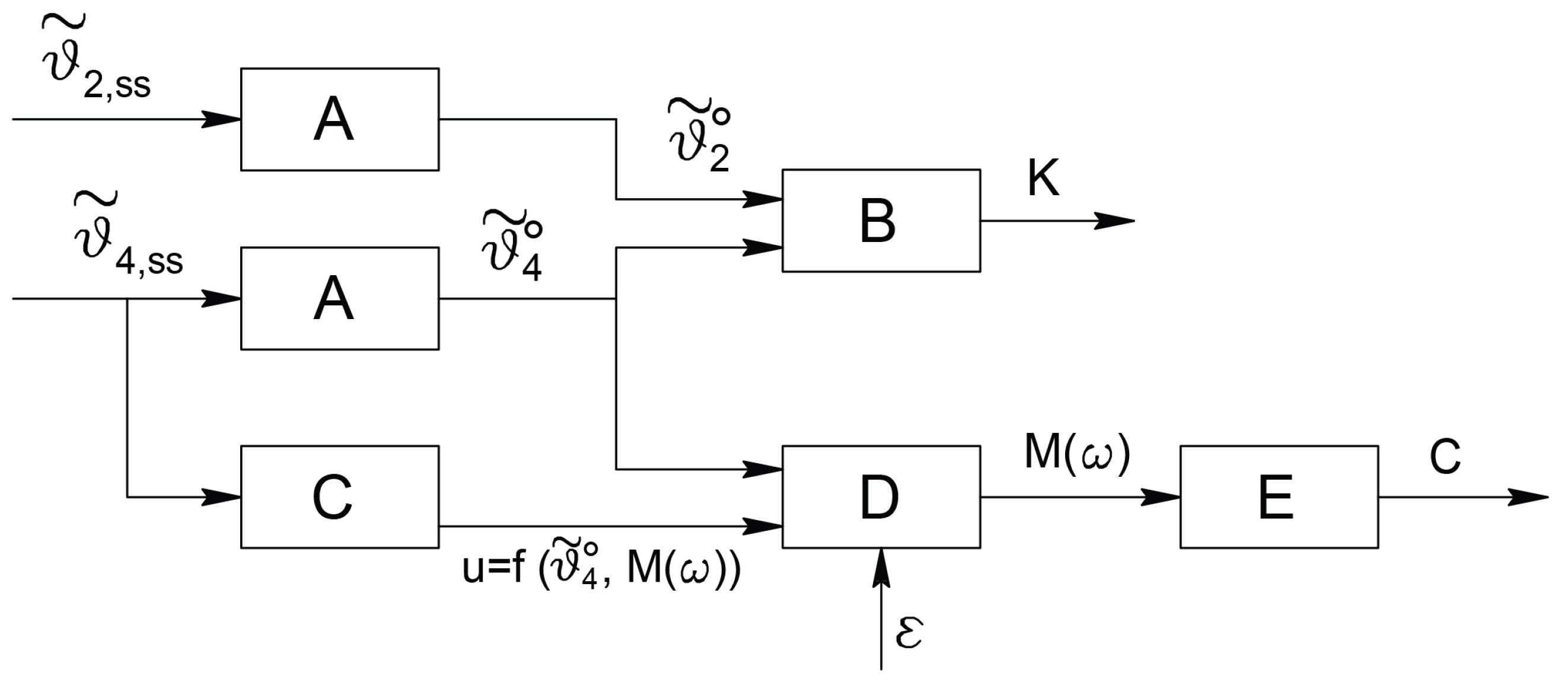
4. The Adopted Electromechanical Model
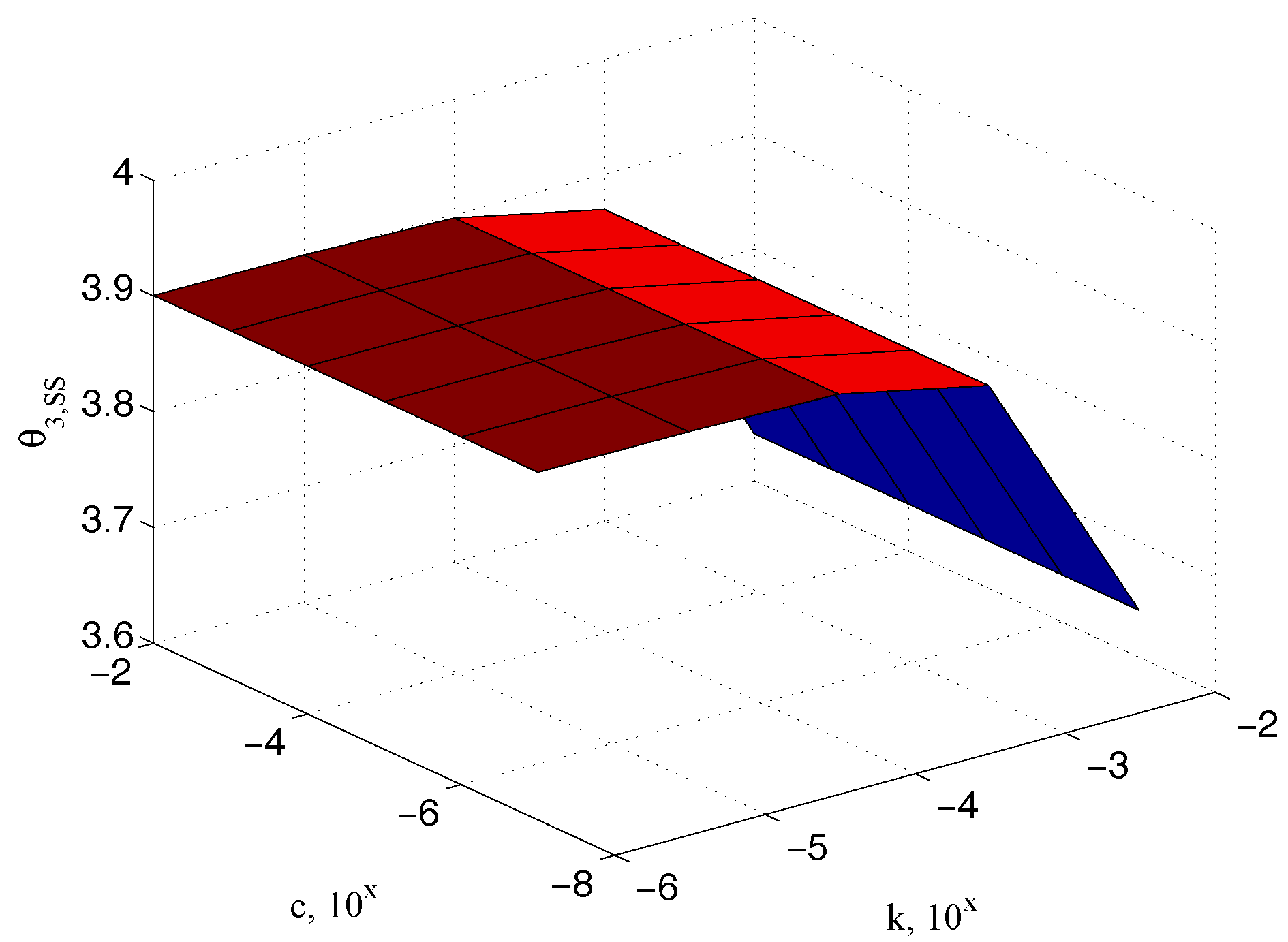
5. The Identification of the Sample Stiffness and the Damping Coefficients
5.1. Characterization of the Sample Stiffness
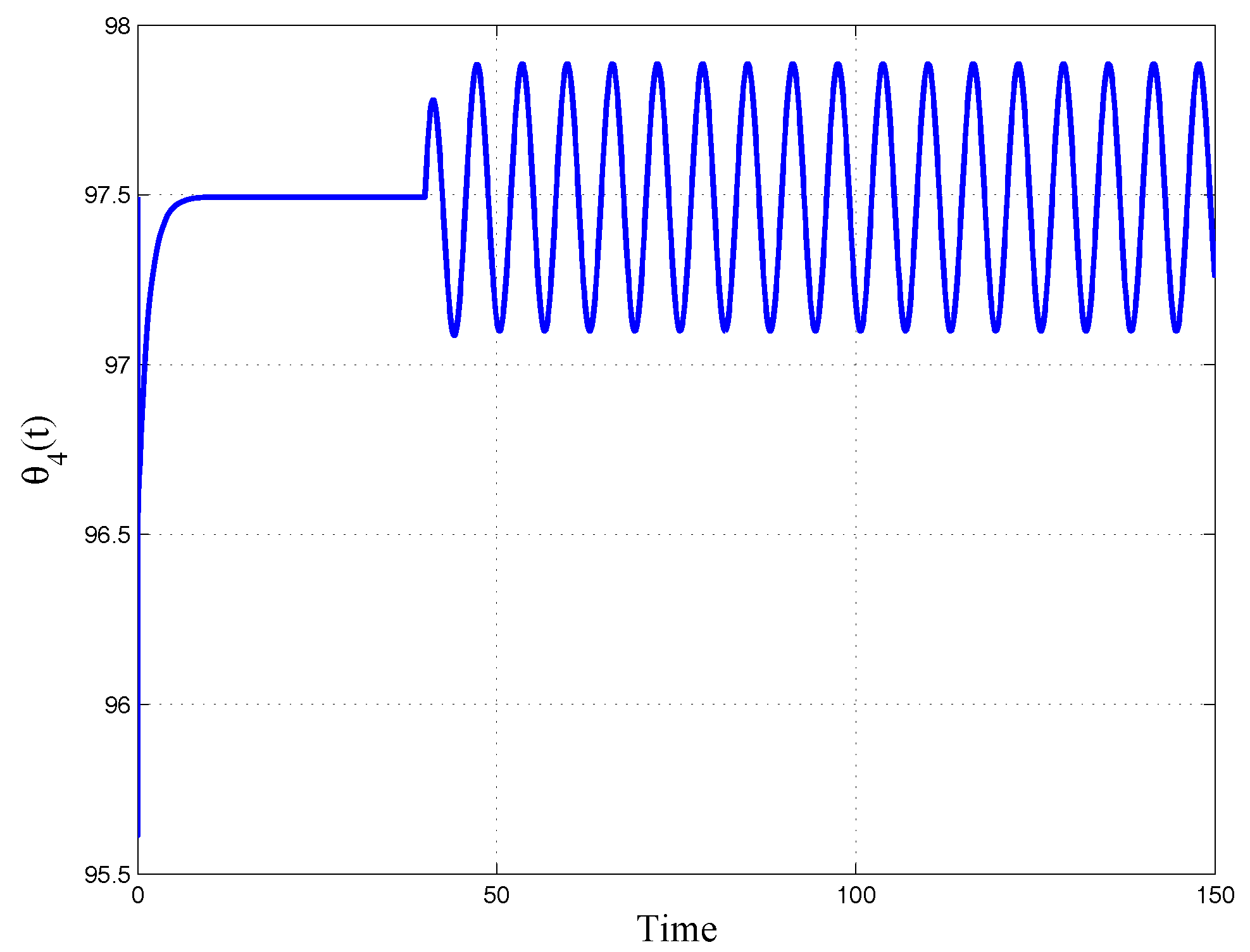
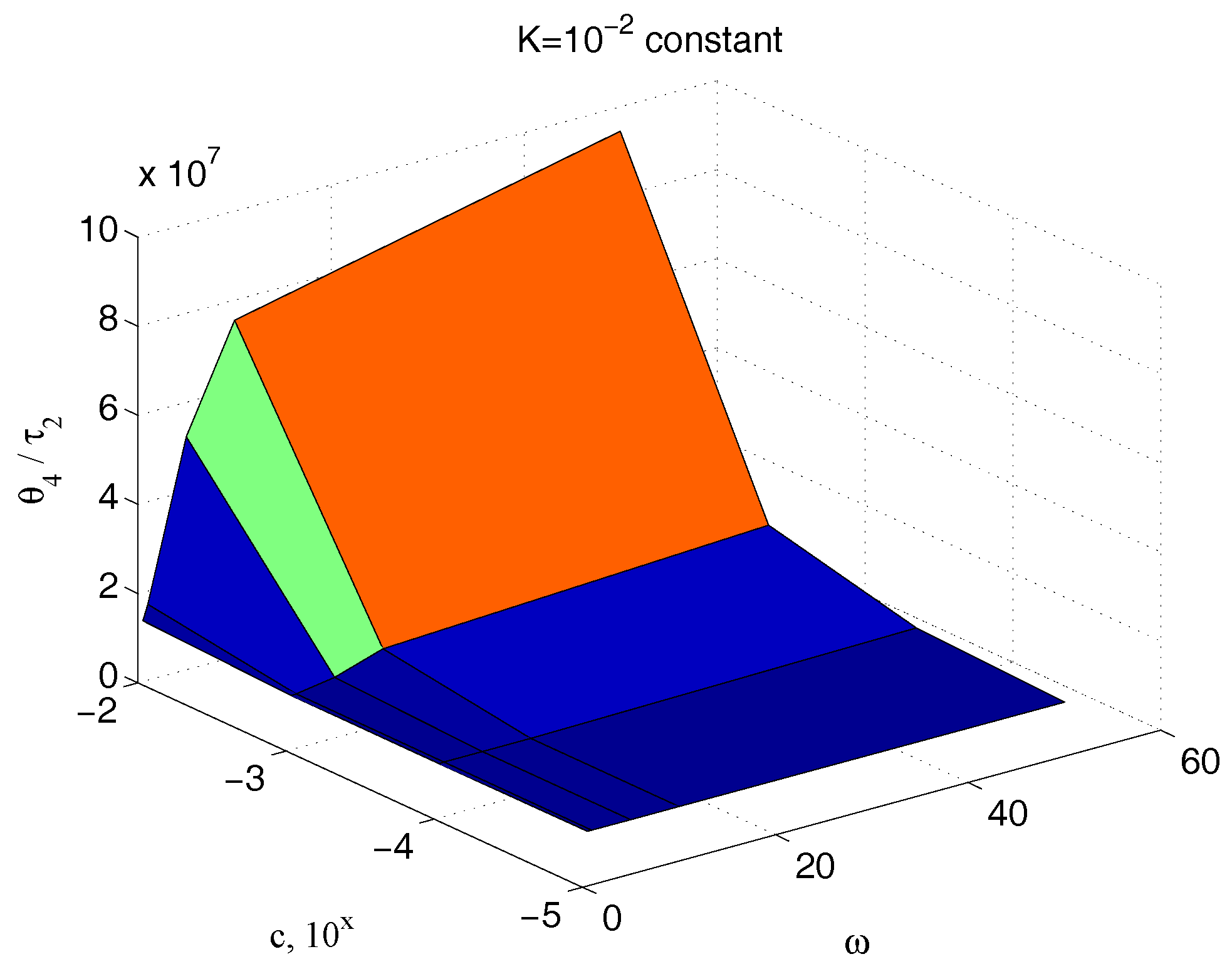
5.2. Characterization of Sample Characteristic Damping
5.3. The Adopted Operational Scheme
6. Conclusions
Author Contributions
Conflicts of Interest
References
- Morrison, B., III; Meaney, D.F.; McIntosh, T.K. Mechanical characterization of an in vitro device designed to quantitatively injure living brain tissue. Ann. Biomed. Eng. 1998, 26, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Edsberg, L.E.; Cutway, R.; Anain, S.; Natiella, J.R. Microstructural and mechanical characterization of human tissue at and adjacent to pressure ulcers. J. Rehabil. Res. Dev. 2000, 37, 463–471. [Google Scholar] [PubMed]
- Wakatsuki, T.; Kolodney, M.S.; Zahalak, G.I.; Elson, E.L. Cell mechanics studied by a reconstituted model tissue. Biophys. J. 2000, 79, 2353–2368. [Google Scholar] [CrossRef]
- Sacks, M.S.; Sun, W. Multiaxial mechanical behavior of biological materials. Annu. Rev. Biomed. Eng. 2003, 5, 251–284. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Fung, T.C.; Chian, K.S.; Chong, C.K. Viscoelasticity of esophageal tissue and application of a QLV model. J. Biomech. Eng. 2006, 128, 909–916. [Google Scholar] [CrossRef] [PubMed]
- Allen, K.D.; Athanasiou, K.A. Viscoelastic characterization of the porcine temporomandibular joint disc under unconfined compression. J. Biomech. 2006, 39, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Levesque, P.; Gauvin, R.; Larouche, D.; Auger, F.A.; Germain, L. A computer-controlled apparatus for the characterization of mechanical and viscoelastic properties of tissue-engineered vascular constructs. Cardiovasc. Eng. Technol. 2011, 2, 24–34. [Google Scholar] [CrossRef]
- Dagdeviren, C.; Shi, Y.; Joe, P.; Ghaffari, R.; Balooch, G.; Usgaonkar, K.; Gur, O.; Tran, P.L.; Crosby, J.R.; Meyer, M.; et al. Conformal piezoelectric systems for clinical and experimental characterization of soft tissue biomechanics. Nature Materials 2015, 14, 728–736. [Google Scholar] [CrossRef] [PubMed]
- Botta, F.; Marx, N.; Gentili, S.; Schwingshackl, C.; Di Mare, L.; Cerri, G.; Dini, D. Optimal placement of piezoelectric plates for active vibration control of gas turbine blades: experimental results. In SPIE Smart Structures and Materials + Nondestructive Evaluation and Health Monitoring; International Society for Optics and Photonics: San Diego, CA, USA, 2012; p. 83452H. [Google Scholar]
- Botta, F.; Dini, D.; Schwingshackl, C.; di Mare, L.; Cerri, G. Optimal placement of piezoelectric plates to control multimode vibrations of a beam. Adv. Acoust. Vib. 2013, 2013. [Google Scholar] [CrossRef]
- Botta, F.; Marx, N.; Schwingshackl, C.; Cerri, G.; Dini, D. A wireless vibration control technique for gas turbine blades using piezoelectric plates and contactless energy transfer. In Proceedings of the ASME Turbo Expo, San Antonio, TX, USA, 3–7 June 2013. [Google Scholar]
- Kiss, M.Z.; Varghese, T.; Hall, T.J. Viscoelastic characterization of in vitro canine tissue. Phys. Med. Biol. 2004, 49, 4207–4218. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Yao, A.; Zheng, E.E.; Lin, J.; Zheng, Y. Shear wave dispersion ultrasound vibrometry based on a different mechanical model for soft tissue characterization. J. Ultrasound Med. 2012, 31, 2001–2011. [Google Scholar] [CrossRef] [PubMed]
- Tavakoli, M.; Aziminejad, A.; Patel, R.; Moallem, M. Multi-sensory force/deformation cues for stiffness characterization in soft-tissue palpation. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society, New York, NY, USA, 31 August–3 September 2006; pp. 837–840. [Google Scholar]
- Boonvisut, P.; Çavuşoǧlu, M.C. Estimation of soft tissue mechanical parameters from robotic manipulation data. IEEE/ASME Trans. Mech. 2013, 18, 1602–1611. [Google Scholar] [CrossRef] [PubMed]
- Ebenstein, D.M.; Pruitt, L. Nanoindentation of biological materials. Nano Today 2006, 1, 26–33. [Google Scholar] [CrossRef]
- Cox, M.A.J.; Driessen, N.J.B.; Boerboom, R.A.; Bouten, C.V.C.; Baaijens, F.P.T. Mechanical characterization of anisotropic planar biological soft tissues using finite indentation: Experimental feasibility. J. Biomech. 2008, 41, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Saxena, T.; Gilbert, J.; Stelzner, D.; Hasenwinkel, J. Mechanical characterization of the injured spinal cord after lateral spinal hemisection injury in the rat. J. Neurotrauma 2012, 29, 1747–1757. [Google Scholar] [CrossRef] [PubMed]
- González-Cruz, R.D.; Fonseca, V.C.; Darling, E.M. Cellular mechanical properties reflect the differentiation potential of adipose-derived mesenchymal stem cells. Proc. Natl. Acad. Sci. USA 2012, 109, E1523–E1529. [Google Scholar] [CrossRef] [PubMed]
- Ficarella, E.; Lamberti, L.; Papi, M.; De Spirito, M.; Pappalettere, C. Viscohyperelastic calibration in mechanical characterization of soft matter. In Mechanics of Biological Systems and Materialsz; Springer International Publishing: Cham, Switzerland, 2017; Volume 6, pp. 33–37. [Google Scholar]
- Mazza, E.; Nava, A.; Hahnloser, D.; Jochum, W.; Bajka, M. The mechanical response of human liver and its relation to histology: An in vivo study. Med. Image Anal. 2007, 11, 663–672. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Sider, K.L.; Simmons, C.A. Measurement of layer-specific mechanical properties in multilayered biomaterials by micropipette aspiration. Acta Biomater. 2011, 7, 1220–1227. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.K. Mechanical characterization of biological tissues: Experimental methods based on mathematical modeling. Biomed. Eng. Lett. 2016, 6, 181–195. [Google Scholar] [CrossRef]
- Nava, A.; Mazza, E.; Kleinermann, F.; Avis, N.J.; McClure, J.; Bajka, M. Evaluation of the mechanical properties of human liver and kidney through aspiration experiments. Technol. Health Care 2004, 12, 269–280. [Google Scholar] [PubMed]
- Nava, A.; Mazza, E.; Furrer, M.; Villiger, P.; Reinhart, W.H. In vivo mechanical characterization of human liver. Med. Image Anal. 2008, 12, 203–216. [Google Scholar] [CrossRef] [PubMed]
- Boudou, T.; Ohayon, J.; Arntz, Y.; Finet, G.; Picart, C.; Tracqui, P. An extended modeling of the micropipette aspiration experiment for the characterization of the Young’s modulus and Poisson’s ratio of adherent thin biological samples: Numerical and experimental studies. J. Biomech. 2006, 39, 1677–1685. [Google Scholar] [CrossRef] [PubMed]
- Bosisio, M.R.; Talmant, M.; Skalli, W.; Laugier, P.; Mitton, D. Apparent Young’s modulus of human radius using inverse finite-element method. J. Biomech. 2007, 40, 2022–2028. [Google Scholar] [CrossRef] [PubMed]
- Valero, C.; Navarro, B.; Navajas, D.; García-Aznar, J.M. Finite element simulation for the mechanical characterization of soft biological materials by atomic force microscopy. J. Mech. Behav. Biomed. Mater. 2016, 62, 222–235. [Google Scholar] [CrossRef] [PubMed]
- Argento, G.; Simonet, M.; Oomens, C.W.J.; Baaijens, F.P.T. Multi-scale mechanical characterization of scaffolds for heart valve tissue engineering. J. Biomech. 2012, 45, 2893–2898. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, M.L.; McGarry, P.J.; Sniadecki, N.J. Review on cell mechanics: Experimental and modeling approaches. Appl. Mech. Rev. 2013, 65, 060801. [Google Scholar] [CrossRef]
- Ekpenyong, A.; Whyte, G.; Chalut, K.; Pagliara, S.; Lautenschläger, F.; Fiddler, C.; Paschke, S.; Keyser, U.; Chilvers, E.; Guck, J. Viscoelastic properties of differentiating blood cells are fate- and function-dependent. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Gossett, D.R.; Tse, H.T.K.; Lee, S.A.; Ying, Y.; Lindgren, A.G.; Yang, O.O.; Rao, J.; Clark, A.T.; Di Carlo, D. Hydrodynamic stretching of single cells for large population mechanical phenotyping. Proc. Natl. Acad. Sci. USA 2012, 109, 7630–7635. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, A.; Verstreken, C.; Fisher, C.; Keyser, U.; Pagliara, S.; Chalut, K. A microfluidic device for characterizing nuclear deformations. Lab Chip 2017, 17, 805–813. [Google Scholar] [CrossRef] [PubMed]
- Pajerowski, J.D.; Dahl, K.N.; Zhong, F.L.; Sammak, P.J.; Discher, D.E. Physical plasticity of the nucleus in stem cell differentiation. Proc. Natl. Acad. Sci. USA 2007, 104, 15619–15624. [Google Scholar] [CrossRef] [PubMed]
- Pagliara, S.; Franze, K.; McClain, C.; Wylde, G.; Fisher, C.; Franklin, R.; Kabla, A.; Keyser, U.; Chalut, K. Auxetic nuclei in embryonic stem cells exiting pluripotency. Nat. Mater. 2014, 13, 638–644. [Google Scholar] [CrossRef] [PubMed]
- Guck, J.; Schinkinger, S.; Lincoln, B.; Wottawah, F.; Ebert, S.; Romeyke, M.; Lenz, D.; Erickson, H.; Ananthakrishnan, R.; Mitchell, D.; et al. Optical deformability as an inherent cell marker for testing malignant transformation and metastatic competence. Biophys. J. 2005, 88, 3689–3698. [Google Scholar] [CrossRef] [PubMed]
- Otto, O.; Rosendahl, P.; Mietke, A.; Golfier, S.; Herold, C.; Klaue, D.; Girardo, S.; Pagliara, S.; Ekpenyong, A.; Jacobi, A.; et al. Real-time deformability cytometry: On-the-fly cell mechanical phenotyping. Nat. Methods 2015, 12, 199–202. [Google Scholar] [CrossRef] [PubMed]
- Guido, I.; Jaeger, M.; Duschl, C. Dielectrophoretic stretching of cells allows for characterization of their mechanical properties. Eur. Biophys. J. 2011, 40, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Kamble, H.; Vadivelu, R.; Barton, M.; Boriachek, K.; Munaz, A.; Park, S.; Shiddiky, M.; Nguyen, N.T. An electromagnetically actuated double-sided cell-stretching device for mechanobiology research. Micromachines 2017, 8, 256. [Google Scholar] [CrossRef]
- Verotti, M.; Crescenzi, R.; Balucani, M.; Belfiore, N.P. MEMS-based conjugate surfaces flexure hinge. J. Mech. Des. 2015, 137, 012301. [Google Scholar] [CrossRef]
- Belfiore, N.P.; Broggiato, G.B.; Verotti, M.; Balucani, M.; Crescenzi, R.; Bagolini, A.; Bellutti, P.; Boscardin, M. Simulation and construction of a MEMS CSFH based microgripper. Int. J. Mech. Control 2015, 16, 21–30. [Google Scholar]
- Balucani, M.; Belfiore, N.P.; Crescenzi, R.; Verotti, M. The development of a MEMS/NEMS-based 3 D.O.F. compliant micro robot. Int. J.Mech. Control 2011, 12, 3–10. [Google Scholar]
- Belfiore, N.P.; Pennestrì, E. An atlas of linkage-type robotic grippers. Mech. Mach. Theory 1997, 32, 811–833. [Google Scholar] [CrossRef]
- Dochshanov, A.; Verotti, M.; Belfiore, N.P. A comprehensive survey on microgrippers design: operational strategy. J. Mech. Des. 2017, 139, 070801. [Google Scholar] [CrossRef]
- Verotti, M.; Dochshanov, A.; Belfiore, N.P. A comprehensive survey on microgrippers design: mechanical structure. J. Mech. Des. 2017, 139, 060801. [Google Scholar] [CrossRef]
- Verotti, M.; Belfiore, N.P. Isotropic compliance in E(3): Feasibility and workspace mapping. J. Mech. Robot. 2016, 8, 061005. [Google Scholar] [CrossRef]
- Verotti, M.; Masarati, P.; Morandini, M.; Belfiore, N.P. Isotropic compliance in the Special Euclidean Group SE(3). Mech. Mach. Theory 2016, 98, 263–281. [Google Scholar] [CrossRef]
- Belfiore, N.P.; Simeone, P. Inverse kinetostatic analysis of compliant four-bar linkages. Mech. Mach. Theory 2013, 69, 350–372. [Google Scholar] [CrossRef]
- Belfiore, N.P.; Verotti, M.; Di Giamberardino, P.; Rudas, I. Active joint stiffness regulation to achieve isotropic compliance in the euclidean space. J. Mech. Robot. 2012, 4. [Google Scholar] [CrossRef]
- Verotti, M.; Dochshanov, A.; Belfiore, N.P. Compliance synthesis of CSFH MEMS-based microgrippers. J. Mech. Des. 2017, 139, 022301. [Google Scholar] [CrossRef]
- Cecchi, R.; Verotti, M.; Capata, R.; Dochshanov, A.; Broggiato, G.; Crescenzi, R.; Balucani, M.; Natali, S.; Razzano, G.; Lucchese, F.; et al. Development of micro-grippers for tissue and cell manipulation with direct morphological comparison. Micromachines 2015, 6, 1710–1728. [Google Scholar] [CrossRef]
- Bagolini, A.; Ronchin, S.; Bellutti, P.; Chiste, M.; Verotti, M.; Belfiore, N.P. Fabrication of novel MEMS microgrippers by deep reactive ion etching with metal hard mask. IEEE J. Microelectromechanical Syst. 2017, 26, 926–934. [Google Scholar] [CrossRef]
- Lim, C.; Zhou, E.; Quek, S. Mechanical models for living cells—A review. J. Biomech. 2006, 39, 195–216. [Google Scholar] [CrossRef] [PubMed]
- Verotti, M. Analysis of the center of rotation in primitive flexures: Uniform cantilever beams with constant curvature. Mech. Mach. Theory 2016, 97, 29–50. [Google Scholar] [CrossRef]
- Verotti, M. Effect of initial curvature in uniform flexures on position accuracy. Mech. Mach. Theory 2018, 119, 106–118. [Google Scholar] [CrossRef]
- Rao, S.S. Mechanical Vibrations; Addison-Wesley: Reading, MA, USA, 1993. [Google Scholar]
- Tu, C.C.; Fanchiang, K.; Liu, C.H. 1 × N rotary vertical micromirror for optical switching applications. Proc. SPIE 2005, 5719, 14–22. [Google Scholar]
- Hou, M.T.K.; Huang, J.Y.; Jiang, S.S.; Yeh, J.A. In-plane rotary comb-drive actuator for a variable optical attenuator. J. Micro/Nanolithography MEMS MOEMS 2008, 7. [Google Scholar] [CrossRef]

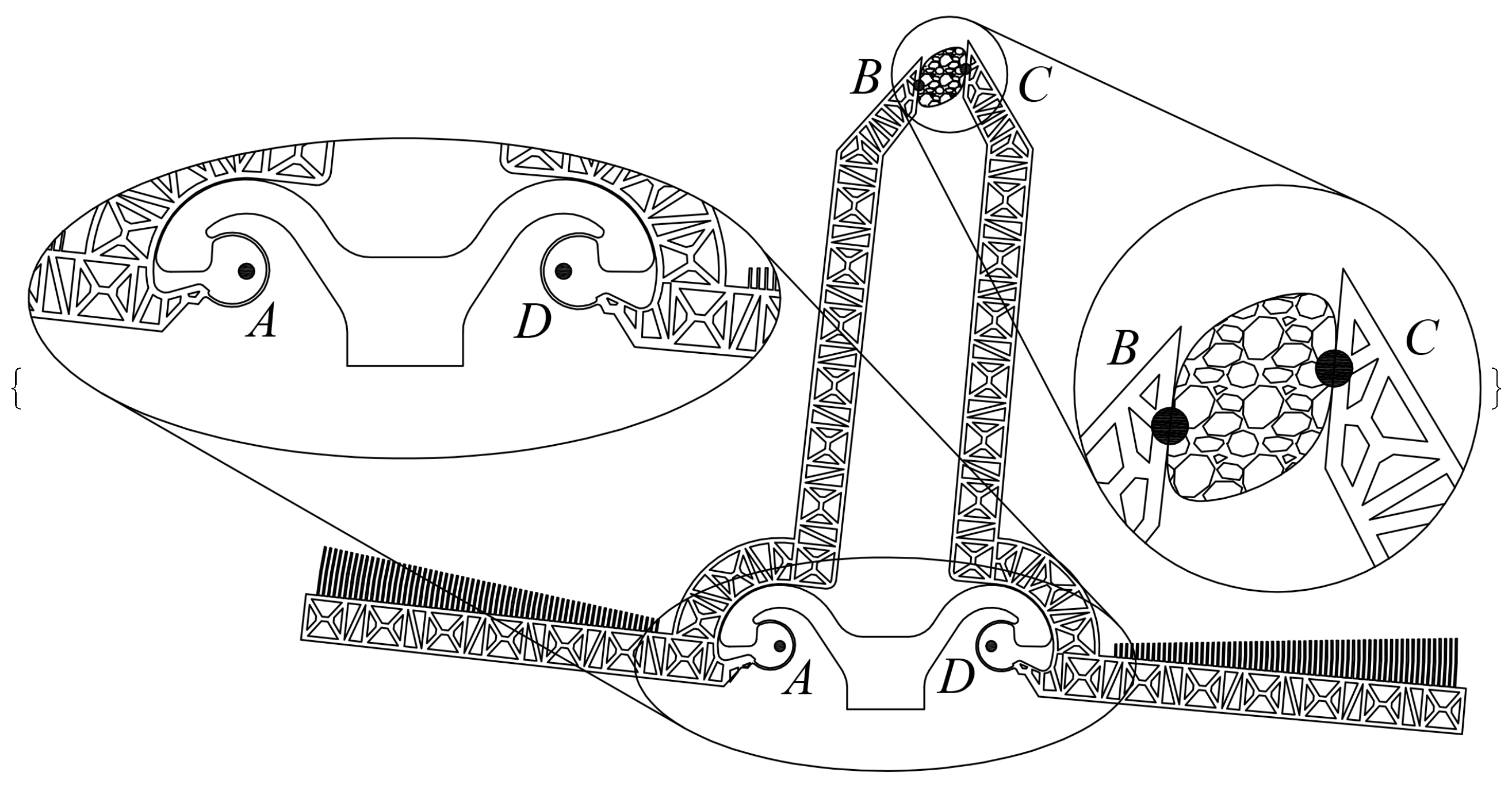
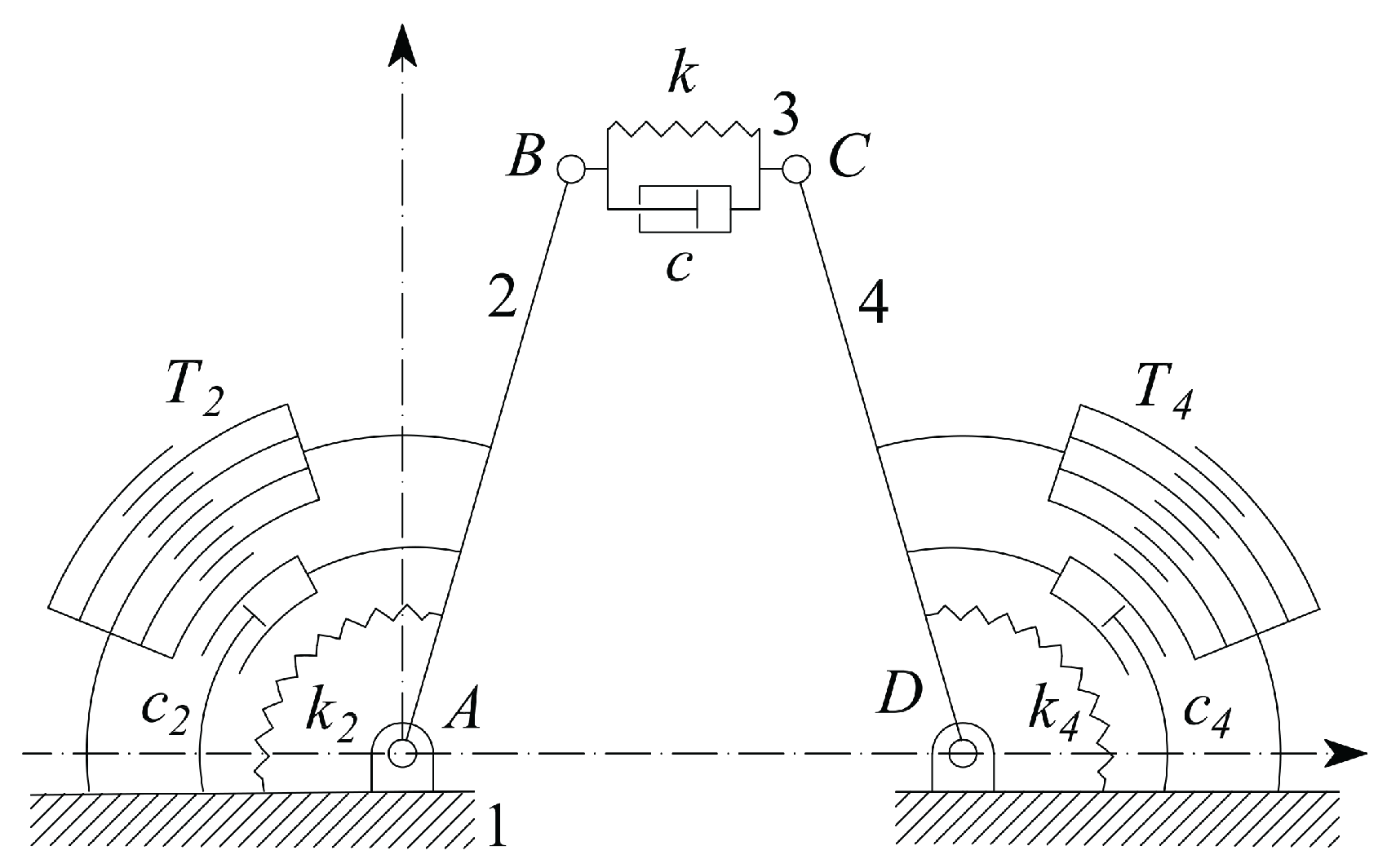
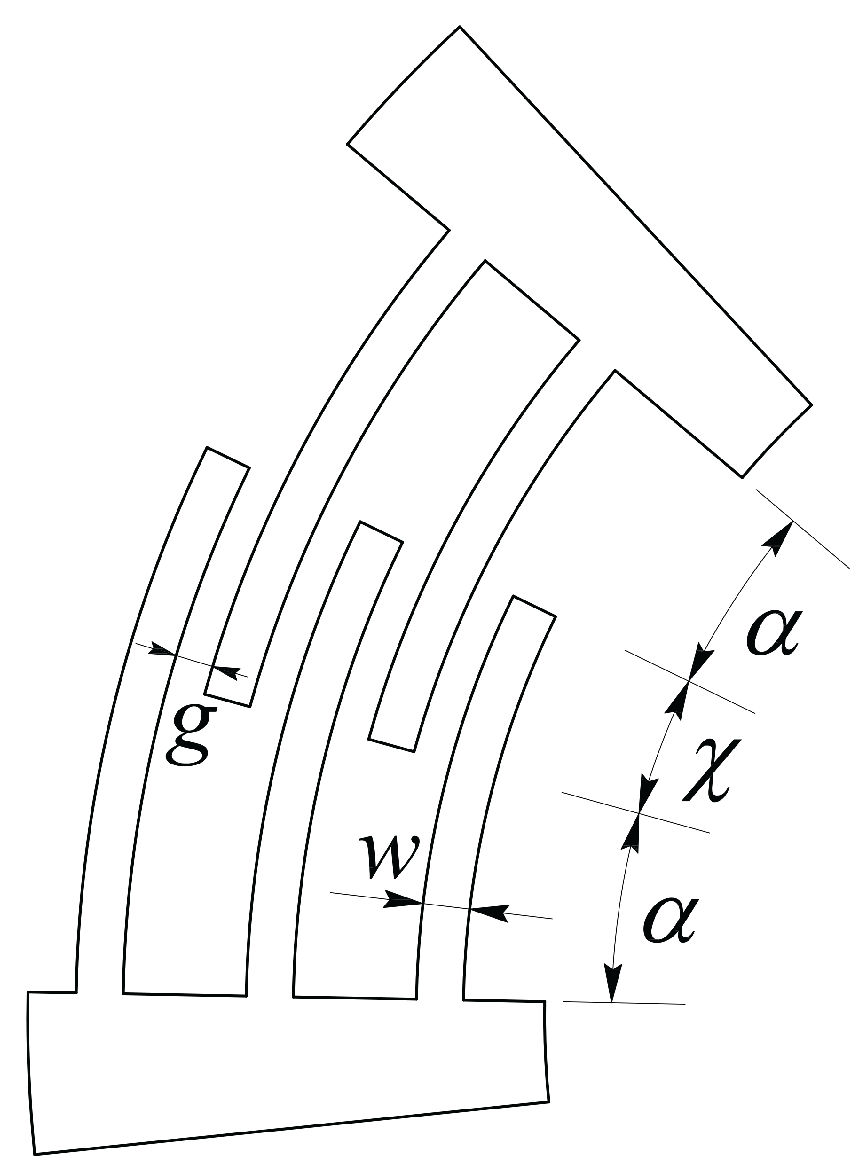
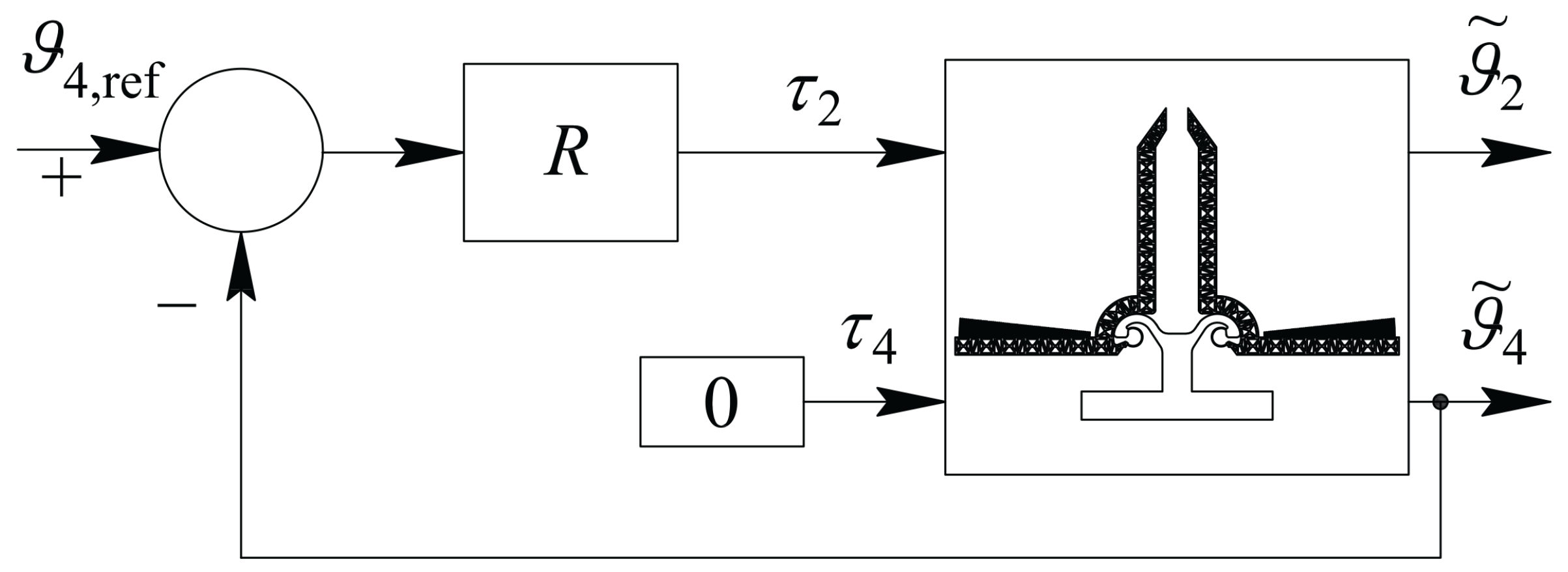
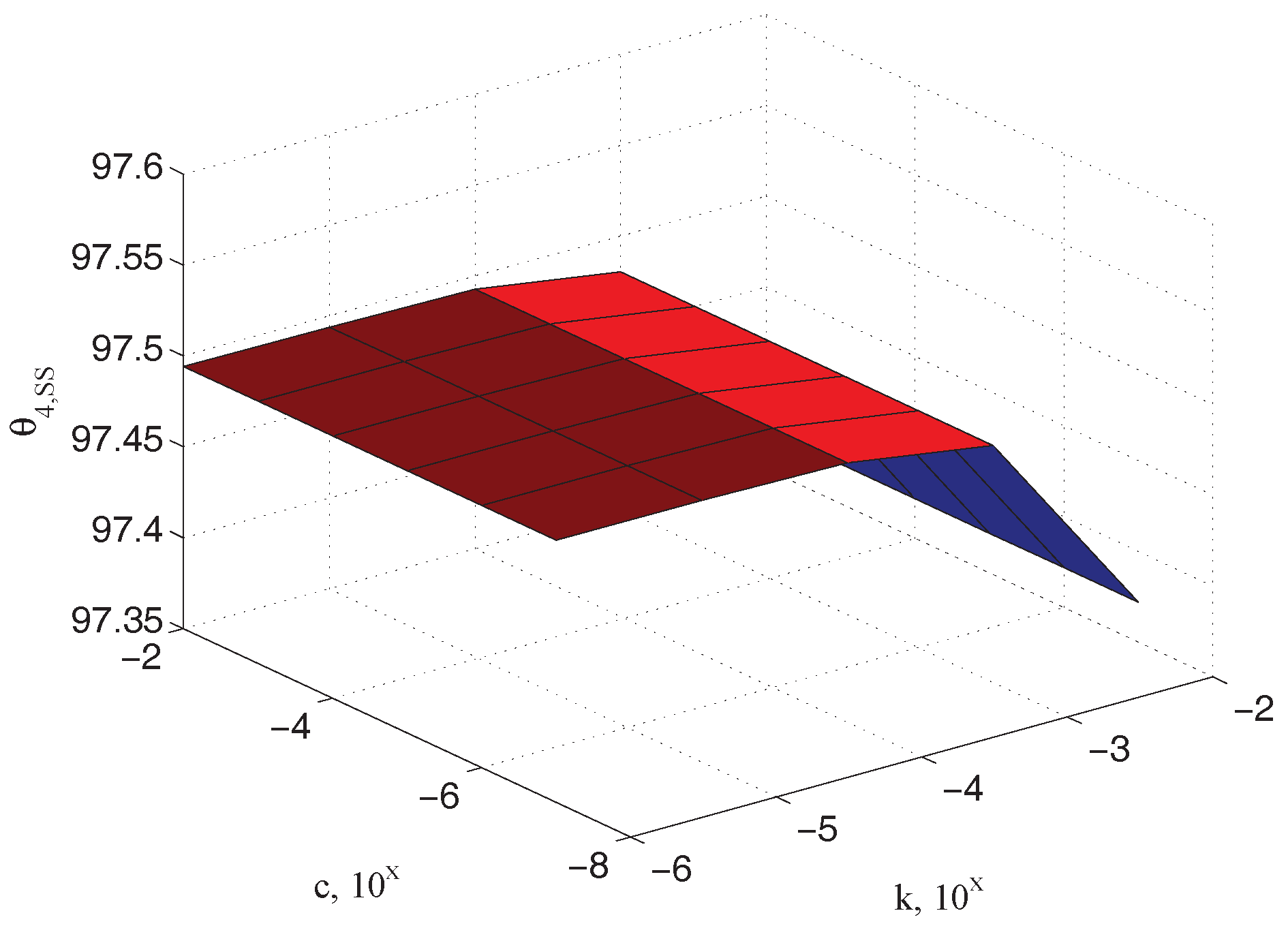
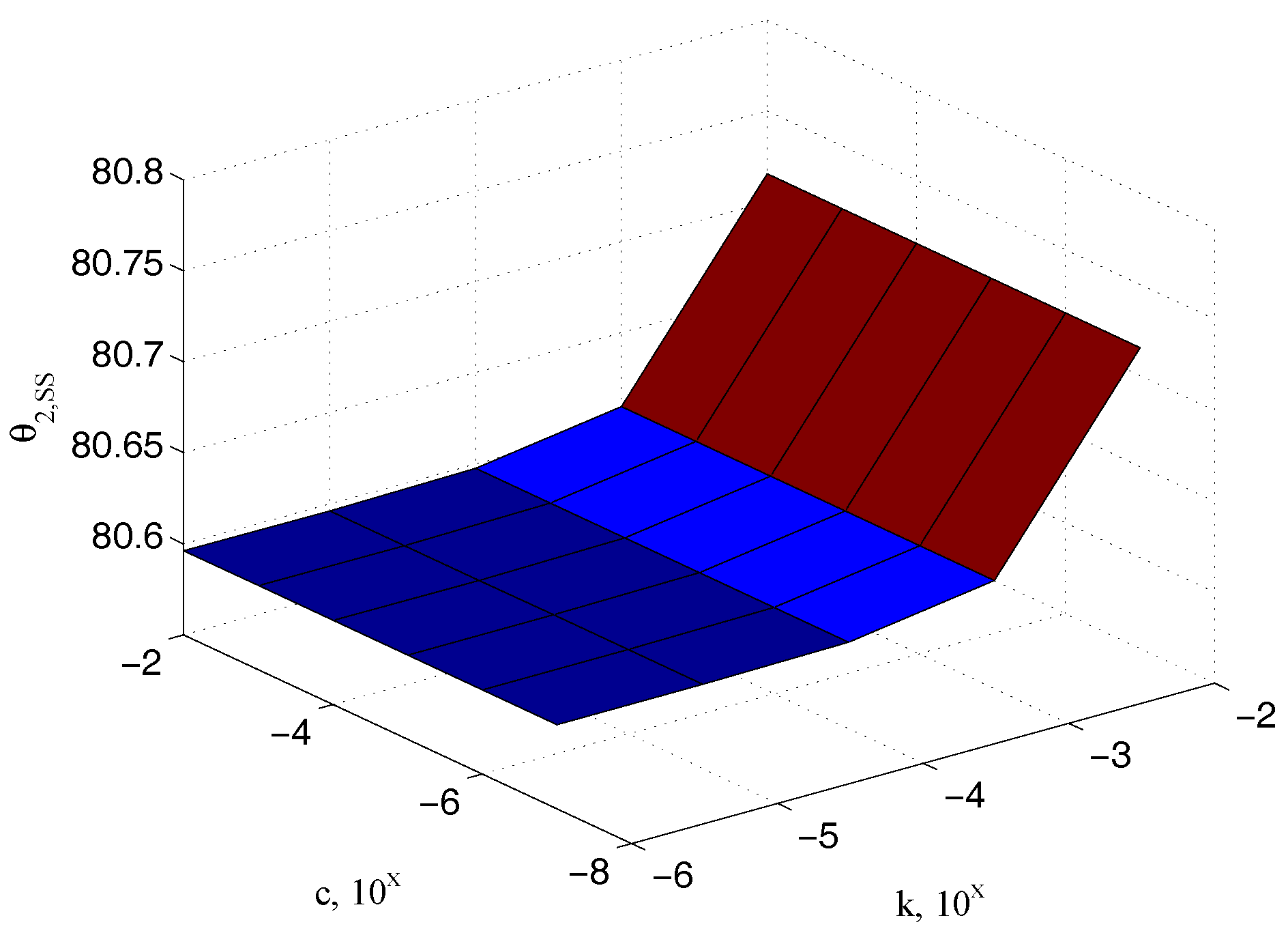
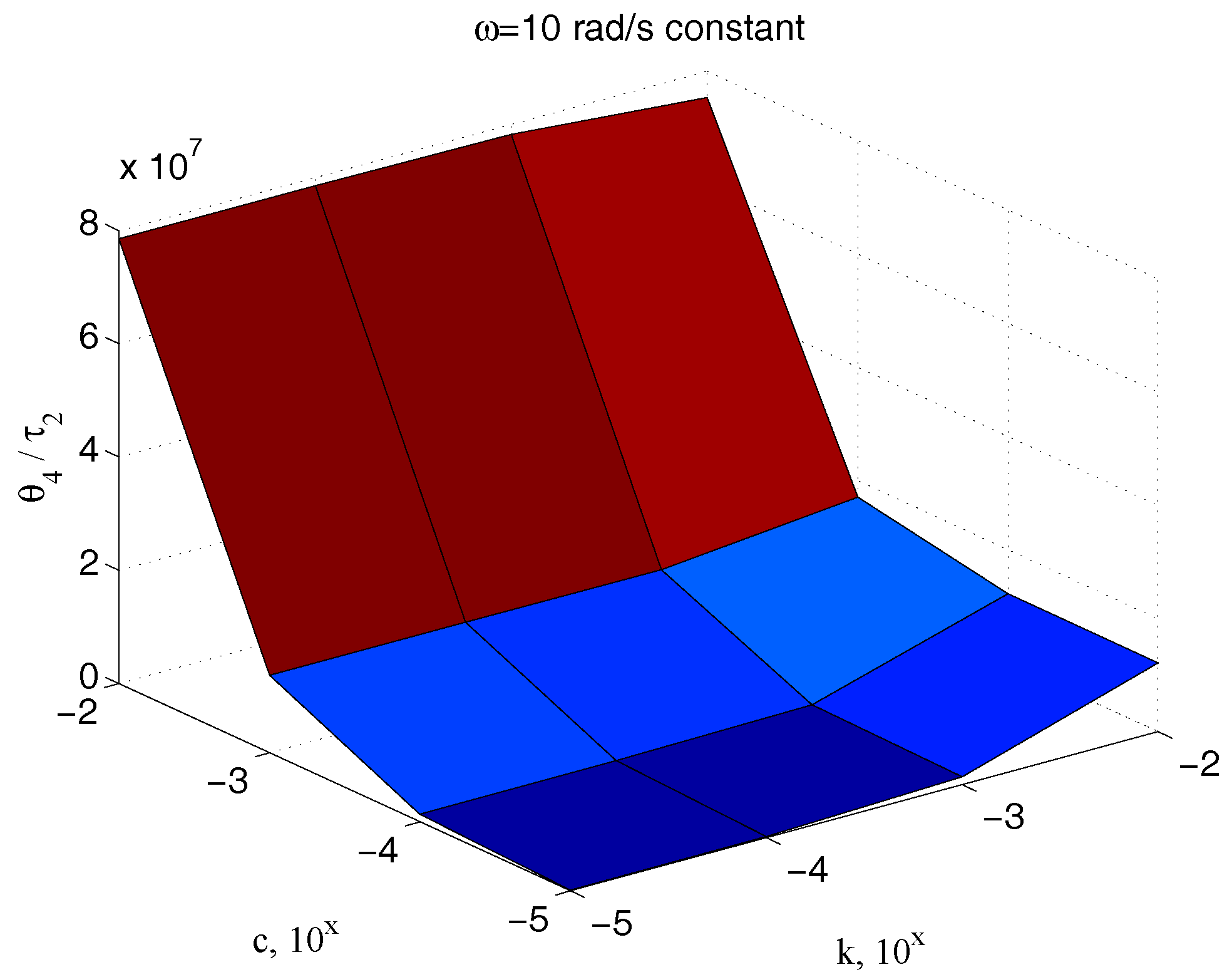
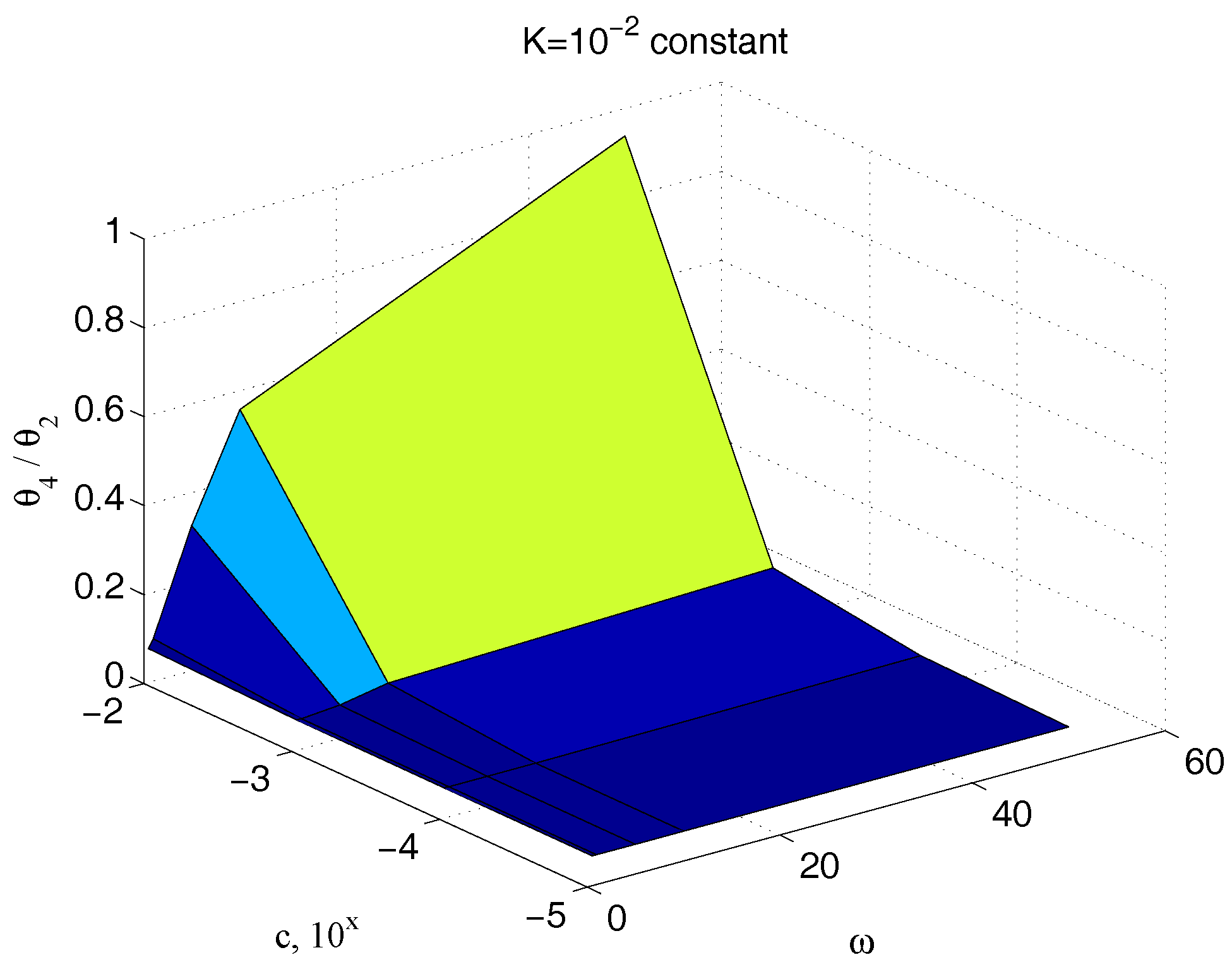

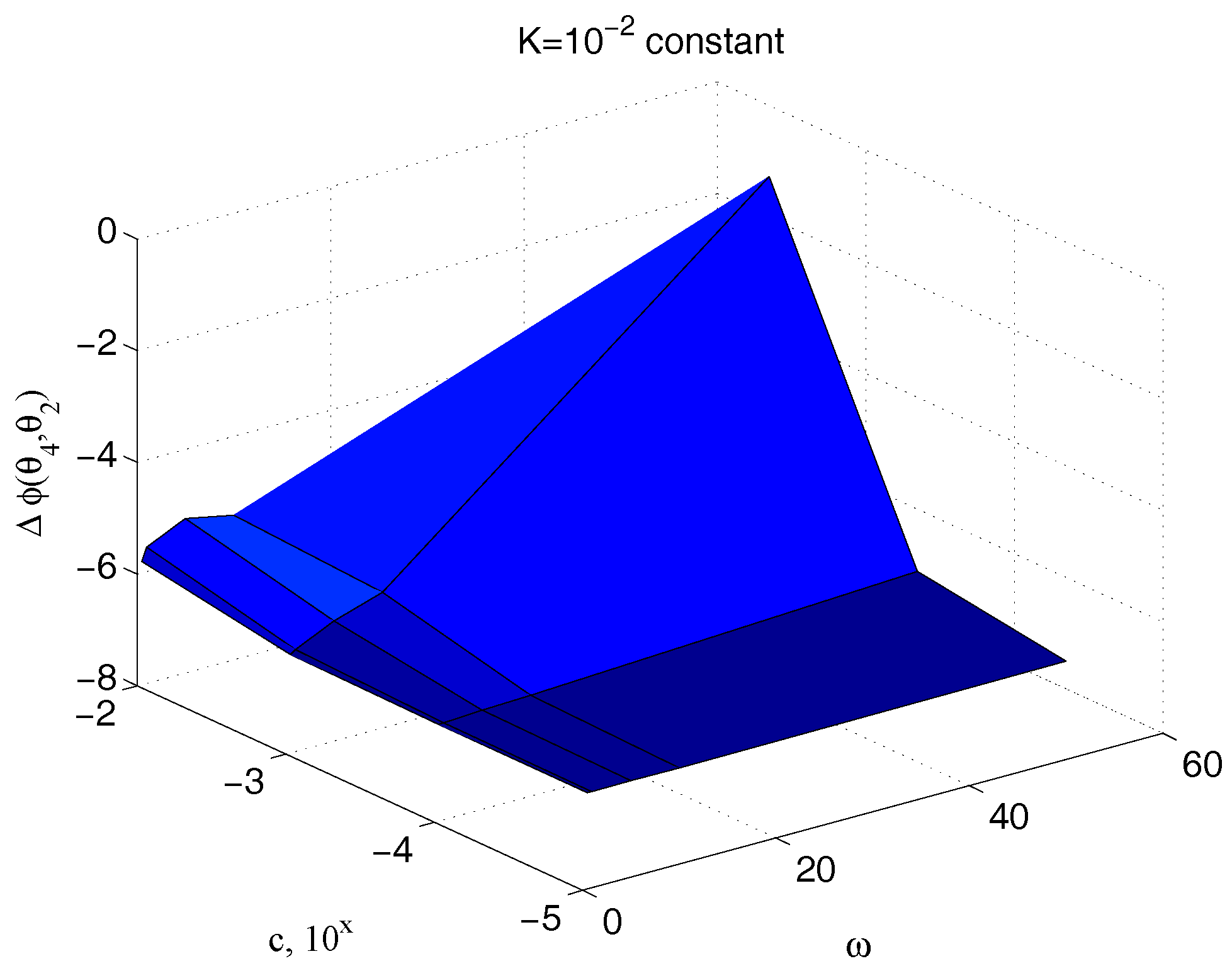
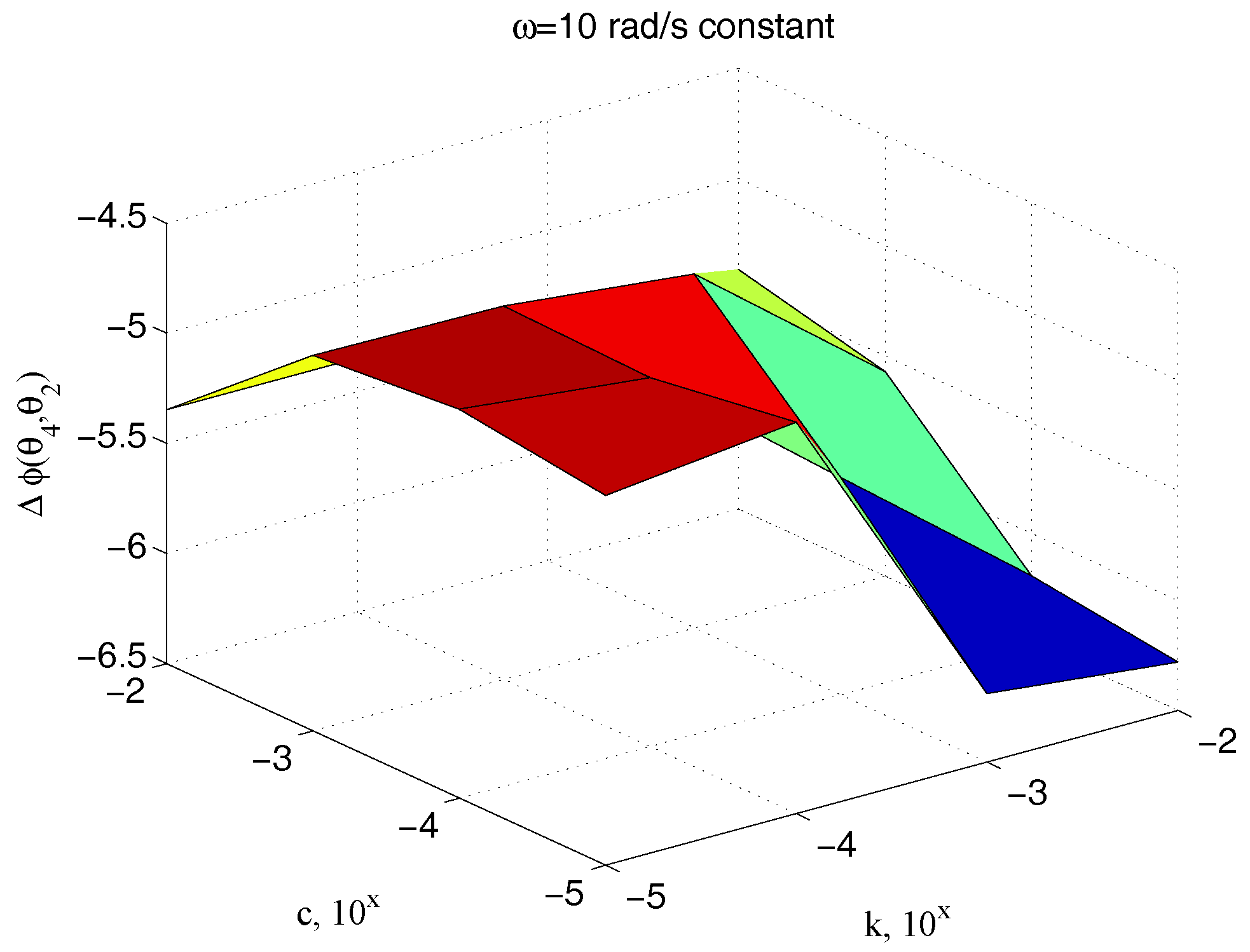
| Parameter | Value | Parameter | Value |
|---|---|---|---|
| 1.44 rad | m | ||
| 1.70 rad | m | ||
| 4.20 rad | m | ||
| rad | , | m | |
| m | , | kg | |
| m | , | kgm | |
| m | kg/ms | ||
| m | , | kgm/rad | |
| 2 m | , | kgm/srad |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Giamberardino, P.; Bagolini, A.; Bellutti, P.; Rudas, I.J.; Verotti, M.; Botta, F.; Belfiore, N.P. New MEMS Tweezers for the Viscoelastic Characterization of Soft Materials at the Microscale. Micromachines 2018, 9, 15. https://doi.org/10.3390/mi9010015
Di Giamberardino P, Bagolini A, Bellutti P, Rudas IJ, Verotti M, Botta F, Belfiore NP. New MEMS Tweezers for the Viscoelastic Characterization of Soft Materials at the Microscale. Micromachines. 2018; 9(1):15. https://doi.org/10.3390/mi9010015
Chicago/Turabian StyleDi Giamberardino, Paolo, Alvise Bagolini, Pierluigi Bellutti, Imre J. Rudas, Matteo Verotti, Fabio Botta, and Nicola P. Belfiore. 2018. "New MEMS Tweezers for the Viscoelastic Characterization of Soft Materials at the Microscale" Micromachines 9, no. 1: 15. https://doi.org/10.3390/mi9010015
APA StyleDi Giamberardino, P., Bagolini, A., Bellutti, P., Rudas, I. J., Verotti, M., Botta, F., & Belfiore, N. P. (2018). New MEMS Tweezers for the Viscoelastic Characterization of Soft Materials at the Microscale. Micromachines, 9(1), 15. https://doi.org/10.3390/mi9010015









