The Occurrence of Non-Regulated Mycotoxins in Foods: A Systematic Review
Abstract
:1. Introduction
- To provide an update on occurrence data for non-regulated mycotoxins in foods with the highest dietary exposure (according to data retrieved from the European Food Safety Authority);
- To provide an overview of the current regulations and highlight any gaps in data related to these issues;
- Highlight the need for high-quality occurrence and metadata in risk assessments.
2. Results and Discussions
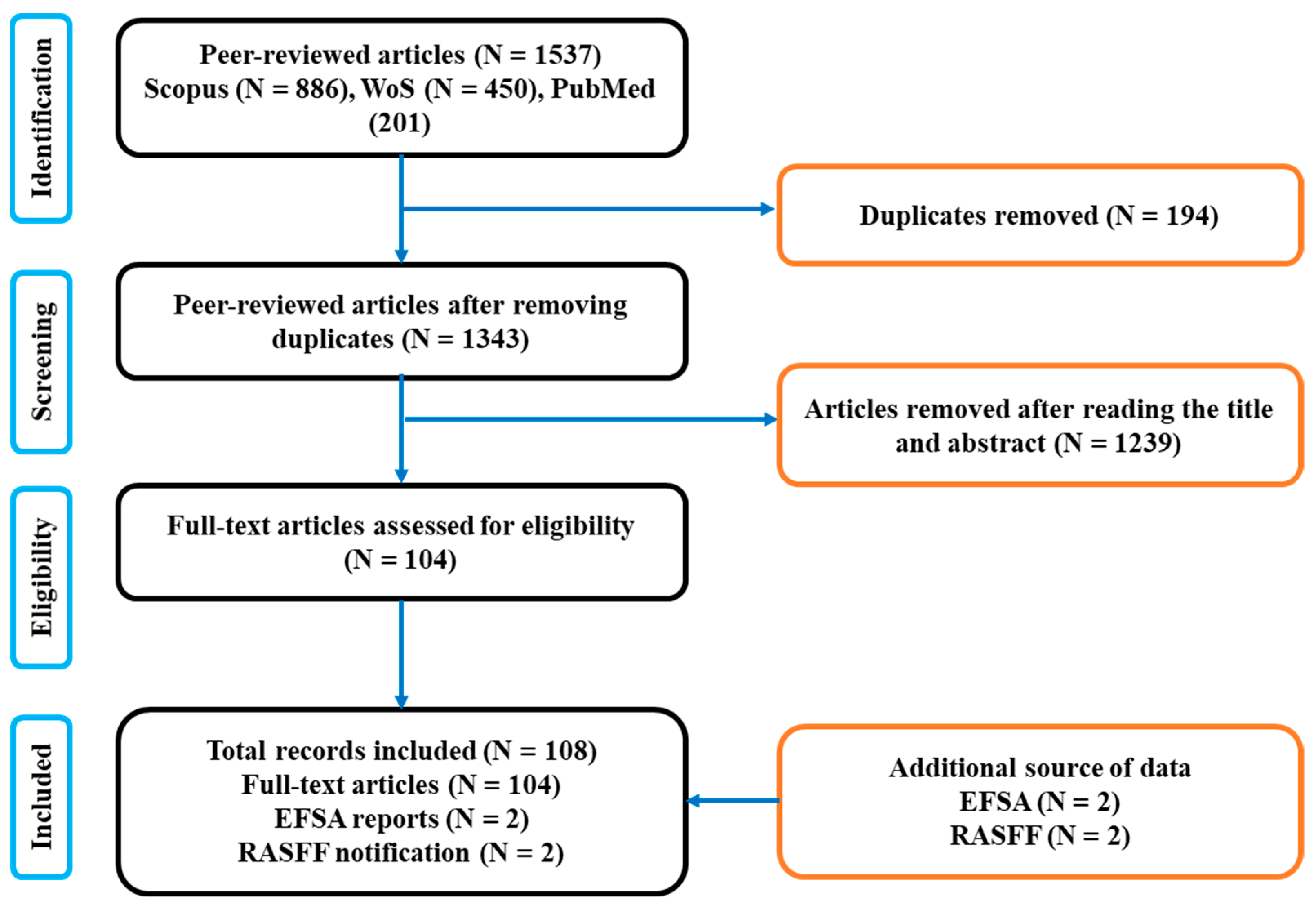
2.1. Systematic Review Process
2.2. Characteristics of the Articles Included in the Review
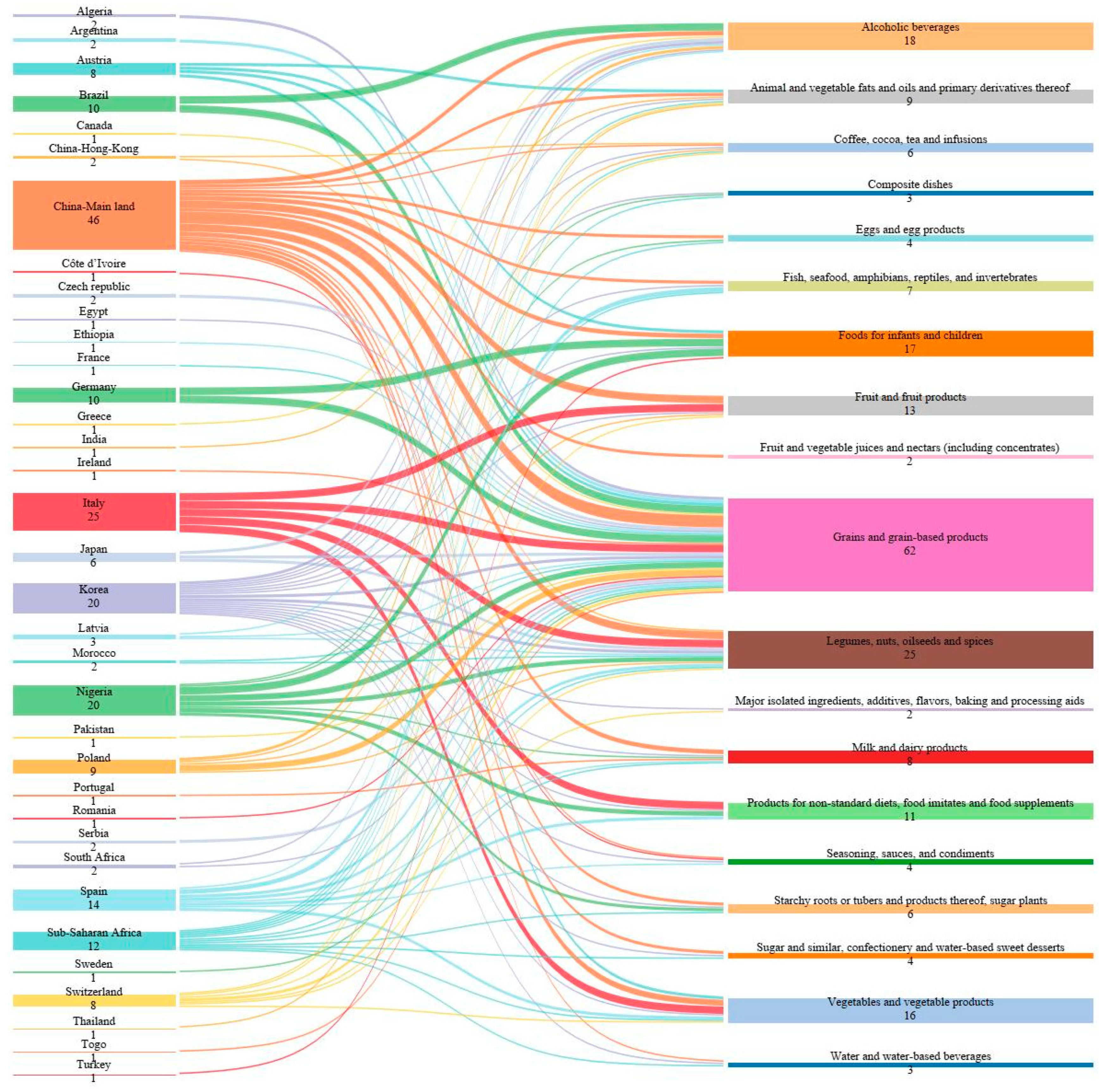
2.3. Worldwide Occurrence of Non-Regulated Mycotoxins in 2018–2022
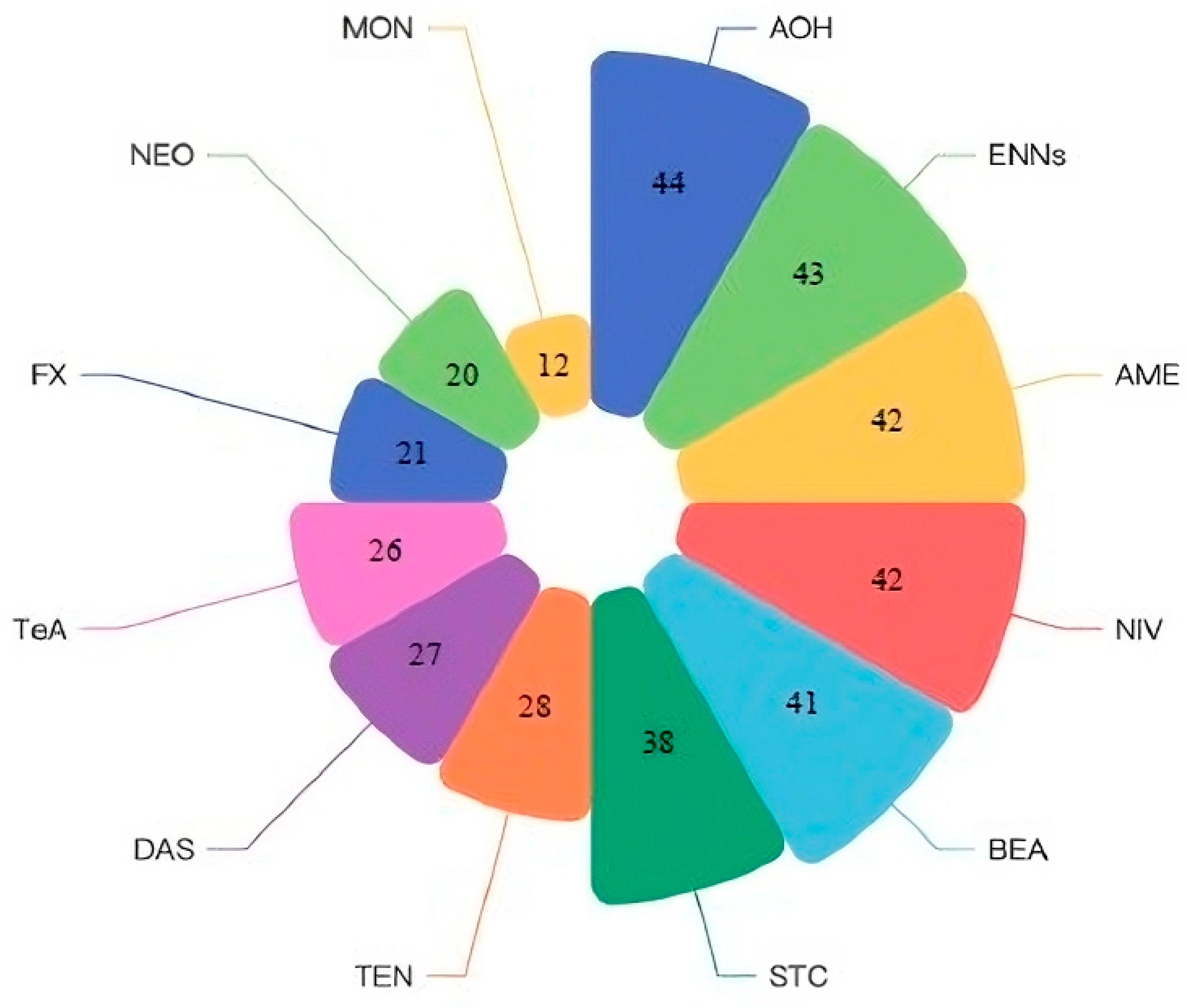
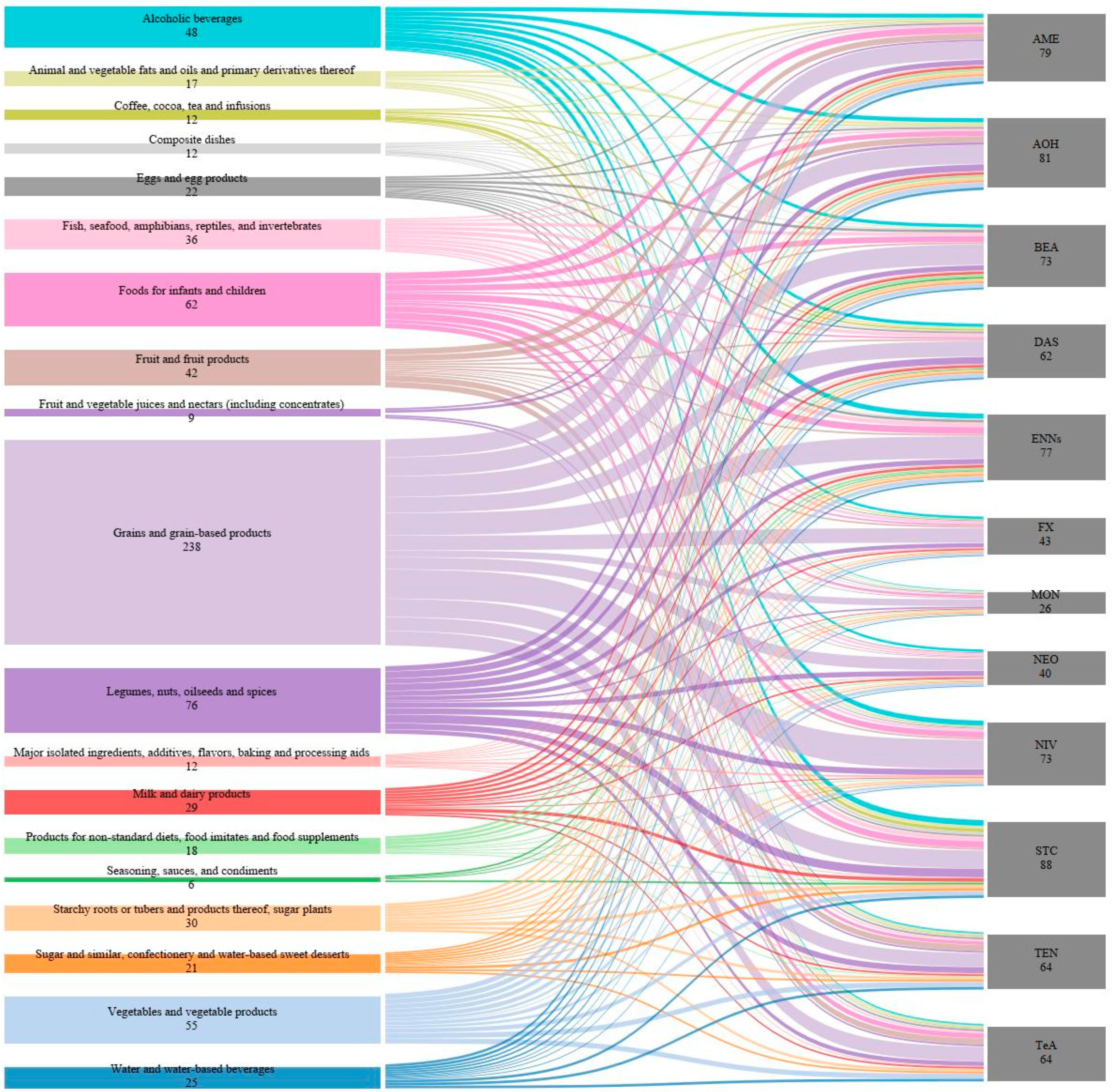
2.4. Worldwide Co-Occurrence of Non-Regulated Mycotoxins and Potential Health Effects
2.5. Data Quality in Occurrence Data
3. Study Limitations
4. Conclusions
5. Materials and Methods
5.1. Search Strategy
5.2. Inclusion and Exclusion Criteria of Data
5.3. Data Extraction
5.4. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sá, S.V.M.D.; Monteiro, C.; Fernandes, J.O.; Pinto, E.; Faria, M.A.; Cunha, S.C. Emerging Mycotoxins in Infant and Children Foods: A Review. Crit. Rev. Food Sci. Nutr. 2021, 63, 1707–1721. [Google Scholar] [CrossRef] [PubMed]
- Abia, W.A.; Warth, B.; Sulyok, M.; Krska, R.; Tchana, A.N.; Njobeh, P.B.; Dutton, M.F.; Moundipa, P.F. Determination of Multi-Mycotoxin Occurrence in Cereals, Nuts and Their Products in Cameroon by Liquid Chromatography Tandem Mass Spectrometry (LC-MS/MS). Food Control 2013, 31, 438–453. [Google Scholar] [CrossRef]
- López, P.; De Rijk, T.; Sprong, R.C.; Mengelers, M.J.B.; Castenmiller, J.J.M.; Alewijn, M. A Mycotoxin-Dedicated Total Diet Study in the Netherlands in 2013: Part II-Occurrence. World Mycotoxin J. 2016, 9, 89–108. [Google Scholar] [CrossRef]
- Oueslati, S.; Blesa, J.; Moltó, J.C.; Ghorbel, A.; Mañes, J. Presence of mycotoxins in sorghum and intake estimation in Tunisia. Food Addit. Contam. Part A 2014, 31, 307–318. [Google Scholar] [CrossRef] [PubMed]
- Arcella, D.; Eskola, M.; Gómez Ruiz, J.A. Dietary Exposure Assessment to Alternaria Toxins in the European Population. EFSA J. 2016, 14, e04654. [Google Scholar] [CrossRef]
- Knutsen, H.K.; Alexander, J.; Barregård, L.; Bignami, M.; Brüschweiler, B.; Ceccatelli, S.; Cottrill, B.; Dinovi, M.; Grasl-Kraupp, B.; Hogstrand, C.; et al. Risks to Human and Animal Health Related to the Presence of Moniliformin in Food and Feed. EFSA J. 2018, 16, e05082. [Google Scholar] [CrossRef]
- Schrenk, D.; Bignami, M.; Bodin, L.; Chipman, J.K.; del Mazo, J.; Grasl-Kraupp, B.; Hogstrand, C.; Hoogenboom, L.; Leblanc, J.C.; Nebbia, C.S.; et al. Risk Assessment of Aflatoxins in Food. EFSA J. 2020, 18, e06040. [Google Scholar] [CrossRef]
- Schrenk, D.; Bodin, L.; Chipman, J.K.; del Mazo, J.; Grasl-Kraupp, B.; Hogstrand, C.; Hoogenboom, L.; Leblanc, J.C.; Nebbia, C.S.; Nielsen, E.; et al. Risk Assessment of Ochratoxin A in Food. EFSA J. 2020, 18, e06113. [Google Scholar] [CrossRef]
- European Commission Regulation (EC) No 118/2006 of 19 December 2006 Setting Maximum Levels for Certain Contaminants in Foodstuffs. Off. J. Eur. Union 2006, 5–24.
- European Commission Regulation (EU). Recomendations on the Presence of T-2 and HT-2 Toxin in Cereals and Cereal Products. Off. J. Eur. Union 2013, 56, 12–15. [Google Scholar]
- European Commission Regulation (EU). COMMISSION RECOMMENDATION (EU) 2022/553 of 5 April 2022 on monitoring the presence of Alternaria toxins in food. Off. J. Eur. Union 2022. [Google Scholar]
- Vaclavikova, M.; Malachova, A.; Veprikova, Z.; Dzuman, Z.; Zachariasova, M.; Hajslova, J. ‘ Emerging ’ Mycotoxins in Cereals Processing Chains: Changes of Enniatins during Beer and Bread Making. Food Chem. 2013, 136, 750–757. [Google Scholar] [CrossRef] [PubMed]
- Bianchini, A.; Bullerman, L.B. Mycotoxins: Classification. Encycl. Food Microbiol. Second Ed. 2014, 2, 854–861. [Google Scholar] [CrossRef]
- EFSA CONTAM Panel (EFSA Panel on Contaminants in the Food Chain). Scientific Opinion on the risk for public and animal health relate to the presence of sterigmatocystin in food and feed. EFSA J. 2013, 11, 3254. [Google Scholar] [CrossRef]
- Battilani, P.; Toscano, P.; Van Der Fels-Klerx, H.J.; Moretti, A.; Camardo Leggieri, M.; Brera, C.; Rortais, A.; Goumperis, T.; Robinson, T. Aflatoxin B 1 Contamination in Maize in Europe Increases Due to Climate Change. Sci. Rep. 2016, 6, 24328. [Google Scholar] [CrossRef]
- Medina, Á.; González-Jartín, J.M.; Sainz, M.J. Impact of Global Warming on Mycotoxins. Curr. Opin. Food Sci. 2017, 18, 76–81. [Google Scholar] [CrossRef]
- Castañares, E.; Pavicich, M.A.; Dinolfo, M.I.; Moreyra, F.; Stenglein, S.A.; Patriarca, A. Natural Occurrence of Alternaria Mycotoxins in Malting Barley Grains in the Main Producing Region of Argentina. J. Sci. Food Agric. 2020, 100, 1004–1011. [Google Scholar] [CrossRef]
- Romero Bernal, Á.R.; Reynoso, C.M.; García Londoño, V.A.; Broggi, L.E.; Resnik, S.L. Alternaria Toxins in Argentinean Wheat, Bran, and Flour. Food Addit. Contam. Part B Surveill. 2019, 12, 24–30. [Google Scholar] [CrossRef]
- Yoshinari, T.; Takeda, N.; Watanabe, M.; Sugita-Konishi, Y. Development of an Analytical Method for Simultaneous Determination of the Modified Forms of 4,15-Diacetoxyscirpenol and Their Occurrence in Japanese Retail Food. Toxins 2018, 10, 178. [Google Scholar] [CrossRef]
- Orlando, B.; Grignon, G.; Vitry, C.; Kashefifard, K.; Valade, R. Fusarium Species and Enniatin Mycotoxins in Wheat, Durum Wheat, Triticale and Barley Harvested in France. Mycotoxin Res. 2019, 35, 369–380. [Google Scholar] [CrossRef]
- Lattanzio, V.M.T.; Verdini, E.; Sdogati, S.; Bibi, R.; Ciasca, B.; Pecorelli, I. Monitoring alternaria toxins in Italian food to support upcoming regulation. Food Addit. Contam. Part B 2021, 15, 42–51. [Google Scholar] [CrossRef]
- Sun, D.; Qiu, N.; Zhou, S.; Lyu, B.; Zhang, S.; Li, J.; Zhao, Y.; Wu, Y. Development of Sensitive and Reliable UPLC-MS/MS Methods for Food Analysis of Emerging Mycotoxins in China Total Diet Study. Toxins 2019, 11, 166. [Google Scholar] [CrossRef]
- Qiu, N.; Sun, D.; Zhou, S.; Li, J.; Zhao, Y.; Wu, Y. Rapid and Sensitive UHPLC-MS/MS Methods for Dietary Sample Analysis of 43 Mycotoxins in China Total Diet Study. J. Adv. Res. 2022, 39, 15–47. [Google Scholar] [CrossRef] [PubMed]
- Carballo, D.; Fernández-Franzón, M.; Ferrer, E.; Pallarés, N.; Berrada, H. Dietary Exposure to Mycotoxins through Alcoholic and Non-Alcoholic Beverages in Valencia, Spain. Toxins 2021, 13, 438. [Google Scholar] [CrossRef] [PubMed]
- Adekoya, I.; Obadina, A.; Adaku, C.C.; De Boevre, M.; Okoth, S.; De Saeger, S.; Njobeh, P. Mycobiota and Co-Occurrence of Mycotoxins in South African Maize-Based Opaque Beer. Int. J. Food Microbiol. 2018, 270, 22–30. [Google Scholar] [CrossRef]
- Rehagel, C.; Akineden, Ö.; Usleber, E. Microbiological and Mycotoxicological Analyses of Processed Cereal-Based Complementary Foods for Infants and Young Children from the German Market. J. Food Sci. 2022, 87, 1810–1822. [Google Scholar] [CrossRef]
- Ayeni, K.I.; Sulyok, M.; Krska, R.; Warth, B.; Ezekiel, C.N. Mycotoxins in Complementary Foods Consumed by Infants and Young Children within the First 18 Months of Life. Food Control 2023, 144, 109328. [Google Scholar] [CrossRef]
- Ezekiel, C.N.; Oyedele, O.A.; Kraak, B.; Ayeni, K.I.; Sulyok, M.; Houbraken, J.; Krska, R. Fungal Diversity and Mycotoxins in Low Moisture Content Ready-To-Eat Foods in Nigeria. Front. Microbiol. 2020, 11, 615. [Google Scholar] [CrossRef] [PubMed]
- Ezekiel, C.N.; Abia, W.A.; Braun, D.; Šarkanj, B.; Ayeni, K.I.; Oyedele, O.A.; Michael-Chikezie, E.C.; Ezekiel, V.C.; Mark, B.N.; Ahuchaogu, C.P.; et al. Mycotoxin Exposure Biomonitoring in Breastfed and Non-Exclusively Breastfed Nigerian Children. Environ. Int. 2022, 158, 106996. [Google Scholar] [CrossRef]
- Ojuri, O.T.; Ezekiel, C.N.; Sulyok, M.; Ezeokoli, O.T.; Oyedele, O.A.; Ayeni, K.I.; Eskola, M.K.; Šarkanj, B.; Hajšlová, J.; Adeleke, R.A.; et al. Assessing the Mycotoxicological Risk from Consumption of Complementary Foods by Infants and Young Children in Nigeria. Food Chem. Toxicol. 2018, 121, 37–50. [Google Scholar] [CrossRef]
- Rodríguez-Carrasco, Y.; Castaldo, L.; Gaspari, A.; Graziani, G.; Ritieni, A. Development of an UHPLC-Q-Orbitrap HRMS Method for Simultaneous Determination of Mycotoxins and Isoflavones in Soy-Based Burgers. LWT 2019, 99, 34–42. [Google Scholar] [CrossRef]
- Augustin Mihalache, O.; Carbonell-Rozas, L.; Cutroneo, S.; Dall’Asta, C. Multi-Mycotoxin Determination in Plant-Based Meat Alternatives and Exposure Assessment. Food Res. Int. 2023, 168, 112766. [Google Scholar] [CrossRef] [PubMed]
- Fraeyman, S.; Croubels, S.; Devreese, M.; Antonissen, G. Emerging Fusarium and Alternaria Mycotoxins: Occurrence, Toxicity and Toxicokinetics. Toxins 2017, 9, 228. [Google Scholar] [CrossRef]
- Fernández-Blanco, C.; Font, G.; Ruiz, M.J. Interaction Effects of Enniatin B, Deoxinivalenol and Alternariol in Caco-2 Cells. Toxicol. Lett. 2016, 241, 38–48. [Google Scholar] [CrossRef]
- Juan-García, A.; Carbone, S.; Ben-Mahmoud, M.; Sagratini, G.; Mañes, J. Beauvericin and Ochratoxin A Mycotoxins Individually and Combined in HepG2 Cells Alter Lipid Peroxidation, Levels of Reactive Oxygen Species and Glutathione. Food Chem. Toxicol. 2020, 139, 111247. [Google Scholar] [CrossRef]
- Springler, A.; Vrubel, G.J.; Mayer, E.; Schatzmayr, G.; Novak, B. Effect of Fusarium-Derived Metabolites on the Barrier Integrity of Differentiated Intestinal Porcine Epithelial Cells (IPEC-J2). Toxins 2016, 8, 345. [Google Scholar] [CrossRef]
- Mohammed, A.; Bekeko, Z.; Yusufe, M.; Sulyok, M.; Krska, R. Fungal Species and Multi-Mycotoxin Associated with Post-Harvest Sorghum (Sorghum bicolor (L.) Moench) Grain in Eastern Ethiopia. Toxins 2022, 14, 473. [Google Scholar] [CrossRef]
- EFSA CONTAM Panel (EFSA Panel on Contaminants in the Food Chain). Scientific Opinion on the risks to human and animal health related to the presence of beauvericin and enniatins in food and feed. EFSA J. 2014, 12, 3802. [Google Scholar] [CrossRef]
- Knutsen, H.K.; Barregård, L.; Bignami, M.; Brüschweiler, B.; Ceccatelli, S.; Cottrill, B.; Dinovi, M.; Edler, L.; Grasl-Kraupp, B.; Hogstrand, C.; et al. Appropriateness to Set a Group Health Based Guidance Value for Nivalenol and Its Modified Forms. EFSA J. 2017, 15, e04751. [Google Scholar] [CrossRef]
- Knutsen, H.K.; Alexander, J.; Barregård, L.; Bignami, M.; Brüschweiler, B.; Ceccatelli, S.; Cottrill, B.; Dinovi, M.; Grasl-Kraupp, B.; Hogstrand, C.; et al. Risk to Human and Animal Health Related to the Presence of 4,15-Diacetoxyscirpenol in Food and Feed. EFSA J. 2018, 16, e05367. [Google Scholar] [CrossRef]
- Rossi, F.; Gallo, A.; Bertuzzi, T. Emerging Mycotoxins in the Food Chain. Med. J. Nutr. Metab. 2020, 13, 7–27. [Google Scholar] [CrossRef]
- Ficheux, A.S.; Sibiril, Y.; Le Garrec, R.; Parent-Massin, D. Invitro Myelotoxicity Assessment of the Emerging Mycotoxins Beauvericin, Enniatin b and Moniliformin on Human Hematopoietic Progenitors. Toxicon 2012, 59, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Ficheux, A.S.; Sibiril, Y.; Parent-Massin, D. Co-Exposure of Fusarium Mycotoxins: In Vitro Myelotoxicity Assessment on Human Hematopoietic Progenitors. Toxicon 2012, 60, 1171–1179. [Google Scholar] [CrossRef] [PubMed]
- Bensassi, F.; Gallerne, C.; Sharaf El Dein, O.; Rabeh Hajlaoui, M.; Bacha, H.; Lemaire, C. Combined Effects of Alternariols Mixture on Human Colon Carcinoma Cells. Toxicol. Mech. Methods 2015, 25, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Kroemer, G.; Galluzzi, L.; Brenner, C. Mitochondrial Membrane Permeabilization in Cell Death. Physiol. Rev. 2007, 87, 99–163. [Google Scholar] [CrossRef] [PubMed]
- Zamzami, N.; Hirsch, T.; Dallaporta, B.; Petit, P.X.; Kroemer, G. Mitochondrial Implication in Accidental and Programmed Cell Death: Apoptosis and Necrosis. J. Bioenerg. Biomembr. 1997, 29, 185–193. [Google Scholar] [CrossRef]
- den Hollander, D.; Holvoet, C.; Demeyere, K.; De Zutter, N.; Audenaert, K.; Meyer, E.; Croubels, S. Cytotoxic Effects of Alternariol, Alternariol Monomethyl-Ether, and Tenuazonic Acid and Their Relevant Combined Mixtures on Human Enterocytes and Hepatocytes. Front. Microbiol. 2022, 13, 849243. [Google Scholar] [CrossRef] [PubMed]
- Alassane-Kpembi, I.; Kolf-Clauw, M.; Gauthier, T.; Abrami, R.; Abiola, F.A.; Oswald, I.P.; Puel, O. New Insights into Mycotoxin Mixtures: The Toxicity of Low Doses of Type B Trichothecenes on Intestinal Epithelial Cells Is Synergistic. Toxicol. Appl. Pharmacol. 2013, 272, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Meca, G.; Font, G.; Ruiz, M.J. Comparative Cytotoxicity Study of Enniatins A, A 1, A 2, B, B 1, B 4 and J 3 on Caco-2 Cells, Hep-G 2 and HT-29. Food Chem. Toxicol. 2011, 49, 2464–2469. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Fernández-Franzón, M.; Font, G.; Ruiz, M.J. Toxicity Evaluation of Individual and Mixed Enniatins Using an in Vitro Method with CHO-K1 Cells. Toxicol. Vitr. 2013, 27, 672–680. [Google Scholar] [CrossRef] [PubMed]
- Vejdovszky, K.; Warth, B.; Sulyok, M.; Marko, D. Non-Synergistic Cytotoxic Effects of Fusarium and Alternaria Toxin Combinations in Caco-2 Cells. Toxicol. Lett. 2016, 241, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Rothstein, H.R.; Sutton, A.J.; Borenstein, M. Publication Bias in Meta-Analysis: Prevention, Assessment and Adjustments; John Wiley & Sons, Ltd.: Chichester, UK, 2005. [Google Scholar]
- Mihalache, O.A.; Dall’Asta, C. Left-Censored Data and Where to Find Them: Current Implications in Mycotoxin-Related Risk Assessment, Legislative and Economic Impacts. Trends Food Sci. Technol. 2023, 136, 112–119. [Google Scholar] [CrossRef]
- More, S.J.; Hardy, A.; Bampidis, V.; Benford, D.; Hougaard Bennekou, S.; Bragard, C.; Boesten, J.; Halldorsson, T.I.; Hernández-Jerez, A.F.; Jeger, M.J.; et al. Guidance on Harmonised Methodologies for Human Health, Animal Health and Ecological Risk Assessment of Combined Exposure to Multiple Chemicals. EFSA J. 2019, 17, e05634. [Google Scholar] [CrossRef] [PubMed]
- More, S.J.; Bampidis, V.; Benford, D.; Bragard, C.; Hernandez-Jerez, A.; Bennekou, S.H.; Halldorsson, T.I.; Koutsoumanis, K.P.; Lambré, C.; Machera, K.; et al. Guidance Document on Scientific Criteria for Grouping Chemicals into Assessment Groups for Human Risk Assessment of Combined Exposure to Multiple Chemicals. EFSA J. 2021, 19, e07033. [Google Scholar] [CrossRef]
- Page, M.J.; Mckenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews Systematic Reviews and Meta-Analyses. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Rayyan—AI Powered Tool for Systematic Literature Reviews. Available online: https://www.rayyan.ai/ (accessed on 10 June 2023).

| Mean Contamination 1 and (or) Mean Range 2 μg/kg(L) | Alternaria | ENNs | BEA | MON | DAS | NIV | STC | NEO | FX | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AOH | AME | TeA | TEN | ENNA | ENNA1 | ENNB | ENNB1 | ||||||||
| Grains and grain-based products | 118.49 | 101.77 | 1178.59 | 2.5 | 6.78 | 12.32 | 118.41 | 37.78 | 47.53 | 231.58 | 7.49 | 70.7 | 2.07 | 0.93 | 80.68 |
| 1.22–36.08 | 0.73–38.79 | 2.62–306.06 | 0.54–10.72 | 0.81–176.92 | 0.51–574.68 | 0.31–2667.24 | 0.38–1449 | 0.39–1.86 | 53.35–4175.7 | 0.02–0.04 | 0.26–50 | 2.8–50 | |||
| Vegetables and vegetable products | 4.17 | 2.7 | 73.83 | 3.13 | 4.1 | 1.06 | 0.73 | 7.74 | ND | ND | ND | 0.09 | 0.52 | ND | |
| 2–65.3 | 5.10–548.36 | 1.09–1.97 | 0.16–0.38 | 15–57 | 0.09–0.17 | ||||||||||
| Starchy roots or tubers and products thereof | ND | 0.37 | 0.49 | 0.16 | ND | ND | ND | 0.23 | ND | ND | |||||
| 0.11–2.41 | 0.2–0.41 | 0.15–1.07 | 0.21–1.63 | 0.12–0.44 | 1.21–2.58 | 0.10–0.13 | |||||||||
| Legumes, nuts, oilseeds and spices | 121.47 | 13.98 | 1399.21 | 64.47 | 1.15 | 0.25 | 0.28 | 0.32 | 0.78 | 5.61 | 0.22 | 25.74 | 3.61 | 1.43 | ND |
| 4.70–93.27 | 5.50–29.67 | 27.79–4678.62 | 4.17–42.6 | 0.26–1.26 | 0.22–0.29 | 1.54–6.49 | 0.14–0.73 | 0.32–5.46 | 0.07–1.37 | ||||||
| Fruit and fruit products | 2.95 | 1.32 | 44.3 | 0.98 | 044 | 0.71 | ND | ND | ND | ND | ND | ND | |||
| 1.05–43.68 | 2–47.8 | 5.13–365.99 | 1.05–7.52 | ||||||||||||
| Fish, seafood, amphibians, reptiles and invertebrates | ND | 0.38 | 0.98 | 3.17 | 0.20 | ND | 3.71 | ND | 0.04 | 0.21 | ND | ||||
| 0.17–0.38 | 0.38–0.68 | 0.18–2.77 | 0.22–0.89 | 0.32–1.02 | |||||||||||
| Milk and dairy products | ND | 2.38 | ND | 1.16; liquid: 1.13 | 1.61; liquid: 0.77 | 1.73; liquid: 0.63 | 1.73; liquid: 0.76 | 3.84; liquid: 0.52 | ND | ND | ND | 1.9 | ND | ND | |
| 1.75–2.53 | 0.16–6.31 | ||||||||||||||
| Eggs and egg products | ND | 0.72 | 7.01 | 1.8 | ND | ND | ND | ND | ND | ||||||
| 0.72–1.31 | 0.26–27.73 | 0.29–1.55 | 0.27–0.51 | 0.11–2.18 | 0.14–0.58 | 0.78–6.7 | |||||||||
| Sugar and similar confectionery and water-based sweet desserts | ND | 0.12 | ND | 0.12 | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | 6.92 |
| Animal and vegetable fats and oils and primary derivatives thereof | 231.83 | 78.03 | 19.75 | 0.78 | - | - | - | - | 0.3 | ND | ND | - | 2.92 | - | - |
| 2–2.4 | 2–3.9 | ||||||||||||||
| Fruit and vegetable juices and nectars (including concentrates) | 1.71 | ND | 4.26 | ND | - | - | - | - | - | - | - | - | - | - | - |
| Water and water-based beverages | ND | 0.16 | 0.58 ng/L | 0.51 ng/L | ND | ND | ND | ND | ND | ND | |||||
| Alcoholic beverages | 23.29 | 21.03 | 0.16 | 13.8 | 0.62 | 2.24 | ND | ND | 4.62 | 18 | 14.82 | 167 | |||
| 2.26–17.62 | 0.15–0.67 | ||||||||||||||
| Coffee, cocoa, tea and infusions | 11.5 | - | - | - | - | - | - | ND | ND | 0.08 | - | ND | |||
| <3 | <1.3 | <1.3 | 0.13–4.48 | ||||||||||||
| Foods for infants and children | 1.26 | 0.58; breast milk: 3.98 ng/L | 22.7 | 0.96 | 2.13; breast milk: 0.5 ng/L | 0.53; breast milk: 0.9 ng/L | 1.83; breast milk: 4.38 ng/L | 0.93; breast milk: 0.56 ng/L | 1.85; breast milk: 3.88 ng/L | 64.24 | ND | 17.65 | 0.16; breast milk: 1.2 ng/L | - | ND |
| 0.4–2 | breast milk: 4–9 ng/L | breast milk: 11–19 ng/L | 16–19.5 | 0.1–0.2 | |||||||||||
| Products for non-standard diets, food imitates and food supplements | 92.6; liquid: 0.21 | 207.5 | ND | liquid: 21.3 | 323.8; liquid: 1.64 | 67.3; liquid: 1.53 | 138.4; liquid: 13.40 | 121.2; liquid: 8.22 | liquid: 1.4 | 11.2 | ND | ND | ND | - | |
| Composite dishes | 2.1 | ND | 114 | ND | ND | ND | ND | ND | 4.4 | 165 | ND | 16.4 | 0.43 | - | - |
| Seasoning, sauces and condiments | - | - | - | - | ND | ND | ND | 12.0 | 37.9 | - | ND | - | 0.08 | - | - |
| Major isolated ingredients, additives, flavors, baking and processing aids | ND | ND | ND | ND | ND | ND | ND | ND | ND | - | ND | ND | ND | ND | ND |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mihalache, O.A.; De Boevre, M.; Dellafiora, L.; De Saeger, S.; Moretti, A.; Pinson-Gadais, L.; Ponts, N.; Richard-Forget, F.; Susca, A.; Dall’Asta, C. The Occurrence of Non-Regulated Mycotoxins in Foods: A Systematic Review. Toxins 2023, 15, 583. https://doi.org/10.3390/toxins15090583
Mihalache OA, De Boevre M, Dellafiora L, De Saeger S, Moretti A, Pinson-Gadais L, Ponts N, Richard-Forget F, Susca A, Dall’Asta C. The Occurrence of Non-Regulated Mycotoxins in Foods: A Systematic Review. Toxins. 2023; 15(9):583. https://doi.org/10.3390/toxins15090583
Chicago/Turabian StyleMihalache, Octavian Augustin, Marthe De Boevre, Luca Dellafiora, Sarah De Saeger, Antonio Moretti, Laetitia Pinson-Gadais, Nadia Ponts, Florence Richard-Forget, Antonia Susca, and Chiara Dall’Asta. 2023. "The Occurrence of Non-Regulated Mycotoxins in Foods: A Systematic Review" Toxins 15, no. 9: 583. https://doi.org/10.3390/toxins15090583
APA StyleMihalache, O. A., De Boevre, M., Dellafiora, L., De Saeger, S., Moretti, A., Pinson-Gadais, L., Ponts, N., Richard-Forget, F., Susca, A., & Dall’Asta, C. (2023). The Occurrence of Non-Regulated Mycotoxins in Foods: A Systematic Review. Toxins, 15(9), 583. https://doi.org/10.3390/toxins15090583










