Paralytic Shellfish Toxins Occurrence in Non-Traditional Invertebrate Vectors from North Atlantic Waters (Azores, Madeira, and Morocco)
Abstract
1. Introduction
2. Results and Discussion
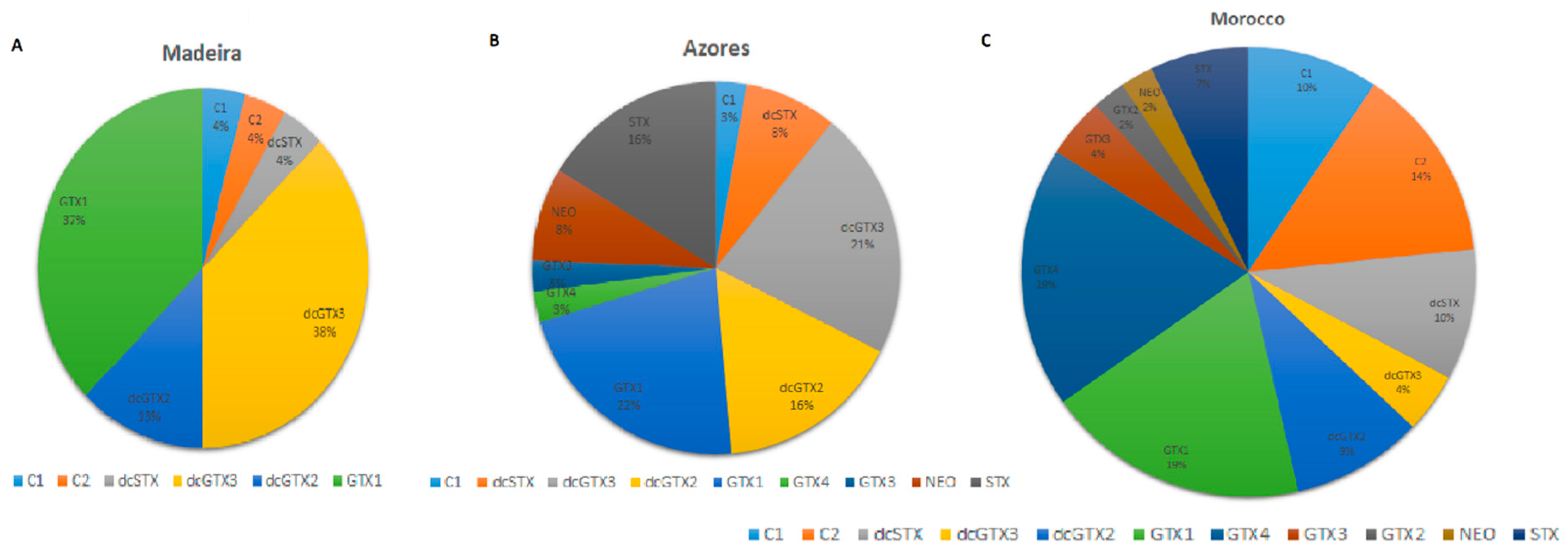
2.1. Madeira Island (Madeira Archipelago)
2.2. São Miguel Island (Azores Archipelago)
2.3. Moroccan Coast
2.4. Statistical Analysis
2.5. General Discussion
3. Conclusions
4. Materials and Methods
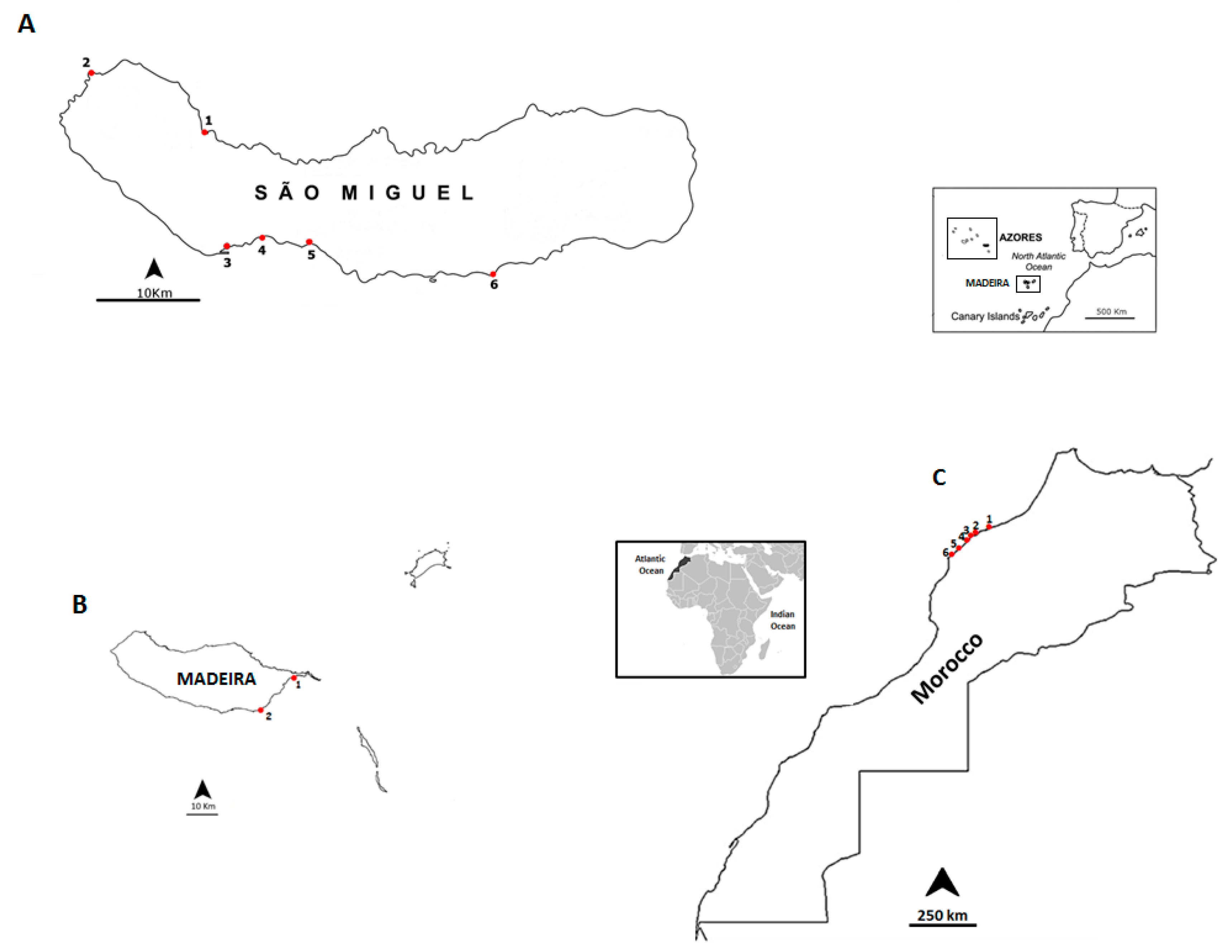
4.1. Sampling Sites and Selected Species
4.2. PSTs Extraction and Analysis Method
4.2.1. Chemicals and Solutions
4.2.2. Apparatus
4.2.3. Extraction Protocol
4.2.4. HPLC Analysis
4.2.5. Post-Column Derivatization
4.2.6. Toxin Identification and Quantification
4.3. Statistical Analyses
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wiese, M.; D’Agostino, P.; Mihali, T.; Moffitt, M.; Neilan, B.A. Neurotoxic alkaloids: Saxitoxin and its analogues. Mar. Drugs 2010, 8, 2185–2211. [Google Scholar] [CrossRef] [PubMed]
- Negri, A.P.; Stirling, D.J.; Quilliam, M.; Blackburn, S.; Bolch, C.; Burton, I.; Eaglesham, G.; Thomas, K.; Walter, J.; Willis, R. Three novel hydroxybenzoate saxitoxin analogues isolated from the dinoflagellate Gymnodinium catenatum. Chem. Res. Toxicol. 2003, 16, 1029–1033. [Google Scholar] [CrossRef] [PubMed]
- Vale, P. Complex profiles of hydrophobic paralytic shellfish poisoning compounds in Gymnodinium catenatum identified by liquid chromatography with fluorescence detection and mass spectrometry. J. Chromatogr. A 2008, 1195, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Shumway, S.E. A review of the effects of algal blooms on shellfish and aquaculture. J. World Aquacul. Soc. 1990, 21, 65–104. [Google Scholar] [CrossRef]
- Shumway, S.E. Phycotoxin-related shellfish poisoning: Bivalve mollusks are not the only vectors. Rev. Fish. Sci. 1995, 3, 1–31. [Google Scholar] [CrossRef]
- Kellmann, R.; Ploux, O.; Neilan, B.A. Neurotoxic alkalids from cyanobacteria. In Natural Products-Phytochemistry, Botany and Metabolism of Alkaloids, Phenolics and Terpenes; Ramawat, K., Merillon, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 39–83. [Google Scholar]
- Bricelj, M.V.; Shumway, S.E. Paralytic shellfish toxins in bivalve moluscs: Ocurrence, transfer kinetics and biotransformation. Rev. Fish. Sci. 1998, 6, 315–383. [Google Scholar] [CrossRef]
- Deeds, J.R.; Landsberg, J.H.; Etheridge, S.M.; Pitcher, G.C.; Longan, S.W. Non-traditional vectors for paralytic shellfish poisoning. Mar. Drugs 2008, 6, 308–348. [Google Scholar] [CrossRef] [PubMed]
- García, C.; Pérez, F.; Contreras, C.; Figueroa, D.; Barriga, A.; López-Rivera, A.; Araneda, O.F.; Contreras, H.R. Saxitoxins and okadaic acid group: Accumulation and distribution in invertebrate marine vectors from Southern Chile. Food Addit. Contam. Part. A 2015, 32, 984–1002. [Google Scholar] [CrossRef] [PubMed]
- Zamorano, R.; MarãN, M.; Cabrera, F.; Contreras, C.; Barriga, A.; Lagos, N.; García, C. Determination of the variability of both hydrophilic and lipophilic toxins in endemic wild bivalves and carnivorous gastropods from the Southerm part of Chile. Food Addit. Contam. Part. A 2013, 30, 1660–1677. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.; Barreiro, A.; Rodriguez, P.; Otero, P.; Azevedo, J.; Alfonso, A.; Botana, L.M.; Vasconcelos, V. New Invertebrate Vectors for PST, Spirolides and Okadaic Acid in the North Atlantic. Mar. Drugs 2013, 11, 1936–1960. [Google Scholar] [CrossRef] [PubMed]
- Costa, P.R.; Botelho, M.J.; Lefebvre, K.A. Characterization of paralytic shellfish toxins in seawater and sardines (Sardina pilchardus) during blooms of Gymnodinium catenatum. Hydrobiologia 2010, 655, 89–97. [Google Scholar] [CrossRef]
- Cembella, A.D.; Quilliam, M.A.; Lewis, N.I.; Bauder, A.G.; Dell’Aversano, C.; Thomasa, K.; Jellet, J.; Cusack, R.R. The toxigenic marine dinoflagellate Alexandrium tamarense as the probable cause of mortality of caged salmon in Nova Scotia. Harmful Algae 2002, 1, 313–325. [Google Scholar] [CrossRef]
- Sephton, D.H.; Haya, K.; Martin, J.L.; LeGresley, M.M.; Page, F.H. Paralytic shellfish toxins in zooplankton, mussels, lobsters and caged Atlantic salmon, Salmo salar, during a bloom of Alexandrium fundyense off Gran Manan Island, in the Bay of Fundy. Harmful Algae 2007, 6, 745–758. [Google Scholar] [CrossRef]
- White, A.W. Paralytic shellfish toxins and finfish. In Seafood Toxins; Ragelis, E.P., Ed.; ACS Symposium Series 262; ACS: Washington, DC, USA, 1984; pp. 171–180. [Google Scholar]
- Haya, K.; Martin, J.L.; Waiwood, B.A.; Burridge, L.E.; Hungerford, J.M.; Zitko, V. Identification of paralytic shellfish toxins in mackerel from southwest Bay of Fundy, Canada. In Toxic Marine Phytoplankton; Graneli, E., Sundstrom, B., Edler, L., Anderson, D.M., Eds.; Elsevier Press: New York, NY, USA, 1990; pp. 350–355. [Google Scholar]
- Castonguay, M.; Levasseur, M.; Beaulieu, J.L.; Gregoire, F.; Michaud, S.; Bonneau, E.; Bates, S.S. Accumulation of PSP toxins in Atlantic mackerel: Seasonal and ontogenetic variations. J. Fish. Biol. 1997, 50, 1203–1213. [Google Scholar] [CrossRef]
- Shumway, S.E.; Barter, J.; Sherman-Caswell, S. Auditing the impact of toxic algal blooms on oysters. Environ. Audit. 1990, 2, 41–56. [Google Scholar]
- EU Council. Regulation (EC) No 853/2004 of the European Parliament and of the Council of 29 April 2004 laying down specific hygiene rules for food of animal origin. Off. J. Eur. Union 2004, L139, 55–255. [Google Scholar]
- AOAC. Paralytic shellfish poison. Method 958.08. In Official Methods of Analysis os AOAC International, 17th ed.; Horwitz, W., Ed.; The Association of Official Analytical Chemists International: Gaithersbury, MD, USA, 2000. [Google Scholar]
- Horwitz, W. Protocol for the design, conduct and interpretation of method-performance studies. Pure Appl. Chem. 1995, 67, 331–343. [Google Scholar] [CrossRef]
- AOAC. Official Method 2005.06. Paralytic Shellfish Poisoning Toxins in Shellfish. Prechomatographic Oxidation and Liquid Chromatography with Fluorescence Detection. First Action 2005. In Official Methods of Analysis of AOAC International; Horwitz, W., Ed.; AOAC Internatioanl: Gaithersburg, MD, USA, 2005. [Google Scholar]
- AOAC. Official Method 2011.02. Determination of Paralytic Shellfish Poisoning Toxins in mussels, clams, oysters and scallops. Post-column oxidation method (PCOX). First action 2011. In Official Methods of Analysis; AOAC Internatioanl: Gaithersburg, MD, USA, 2011. [Google Scholar]
- EU Council. Regulation (EC) No 2074/2005 of 5 December 2005, laying down implementing measures for certain products unde Regulation (EC) No 853/2004 of the European Parliament and of the Council and for the organisation of official controls under Regulation (EC) No 854/2004 of the European Parliament and of the Council and Regulation (EC) No 882/2004 of the European Parliament and of the Council, derogating from the Regulation (EC) No 852/2004 of the European Parliament and of the Council and amending Regulations (EC) No 853/2004 and (EC) No 854/2004. Off. J. Eur. Union 2005, 338, 1–54. [Google Scholar]
- Rey, V.; Botana, A.M.; Botana, L.M. Quantification of PSP toxins in toxic shellfish matrices using post-column oxidation liquid chromatography and pre-column oxidation liquid chromatography methods suggest post-column oxidation liquid chromatography as a good monitoring method of choice. Toxicon 2017, 129, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Costa, P.R.; Costa, S.T.; Braga, A.C.; Rodrigues, S.M.; Vale, P. Relevance and challenges in monitoring marine biotoxins in non-bivalve vectors. Food Control. 2017, 76, 24–33. [Google Scholar] [CrossRef]
- FAO. Fishery and aquaculture statistics. In Global Capture Production 1950–2013 (FishstatJ); FAO Fisheries and Aquaculture Department: Rome, Italy, 2015. [Google Scholar]
- Rey, V.; Alfonso, A.; Botana, L.M.; Botana, A.M. Influence of Different Shellfish Matrices on the Separation of PSP Toxins Using a Postcolumn Oxidation Liquid Chromatography Method. Toxins 2015, 7, 1324–1340. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.; Rey, V.; Botana, A.; Vasconcelos, V.; Botana, L.M. Determination of Gonyautoxin-4 in Echinoderms and Gastropod Matrices by Conversion to Neosaxitoxin Using 2-Mercaptoethanol and Post-Column Oxidation Liquid Chromatography with Fluorescence Detection. Toxins 2016, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.; Auðunsson, G.A.; Benford, D.; Cockburn, A.; Cravedi, J.P.; Dogliotti, E.; Di Domenico, A.; Fernández-Cruz, M.L.; Fink-Gremmels, J.; Fürst, P.; et al. Scientific Opinion of the Panel on Contaminants in the Food Chain on a request from the European Commission on Marine Biotoxins in Shellfish-Saxitoxin Group. Efsa J. 2009, 1019, 1–76. [Google Scholar]
- Regulation (EC) No 854/2004 of the European Parliament and of the Council of 29 April 2004 Laying Down Specific Rules for the Organisation of Official Controls on Products of Animal Origin Intended for Human Consumption. OJ L 139. pp. 206–320. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32007R1246 (accessed on 18 June 2015).
- Silva, M.; Azevedo, J.; Rodriguez, P.; Alfonso, A.; Botana, L.M.; Vasconcelos, V. New Gastropod Vectors and Tetrodotoxin Potential Expansion in Temperate Waters of the Atlantic Ocean. Mar. Drugs 2012, 10, 712–726. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.; Rodriguez, I.; Barreiro, A.; Kaufmann, M.; Neto, A.I.; Hassouani, M.; Sabour, B.; Alfonso, A.; Botana, L.M.; Vasconcelos, V. First Report of Ciguatoxins in Two Starfish Species: Ophidiaster ophidianus and Marthasterias glacialis. Toxins 2015, 7, 3740–3757. [Google Scholar] [CrossRef] [PubMed]
- Hallegraeff, G.M. Algal blooms are not a simple toxic broth. Search 1993, 24, 179. [Google Scholar]
- Hallegraeff, G.M. Ocean climate change, phytoplankton community responses, and harmful algal blooms: A formidable predictive challenge. J. Phycol. 2010, 46, 220–235. [Google Scholar] [CrossRef]
- Anderson, D.M.; Glibert, P.M.; Burkholder, J.M. Harmful Algal blooms and eutrophication: Nutrient sources, composition, and consequences. Estuaries 2002, 25, 704–726. [Google Scholar] [CrossRef]
- Burkholder, J.M.; Glibert, P.M.; Skelton, H.M. Mixotrophy, a major mode of nutrition for harmful algal species in eutrophic waters. Harmful Algae 2008, 8, 77–93. [Google Scholar] [CrossRef]
- Glibert, P.; Seitzinger, S.; Heil, C.A.; Burkholder, J.M.; Parrow, M.W.; Codispoti, L.A.; Kelly, V. The role of eutrophication in coastal proliferation of harmful algal blooms: New perspectives and new approaches. Oceanography 2005, 18, 198–209. [Google Scholar] [CrossRef]
- Heisler, J.P.; Gilbert, J.; Burkholder, J.; Anderson, D.; Cochlan, W.; Dennison, W.; Dortch, Q.; Gobler, C.J.; Heil, C.; Humphries, E.; et al. Eutrophication and harmful algal blooms: Scientific consensus. Harmful Algae 2008, 8, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Oshima, Y. Postcolumn derivatization liquid chromatographic method for paralytic shellfish toxins. J. AOAC Int. 1995, 78, 528–532. [Google Scholar]
- Asakawa, M.; Takagi, M.; Iida, A.; Oishi, K. Studies on the conversion of paralytic shellfish poison (Psp) components by biochemical reducing agents. Eisei Kagaku 1987, 33, 50–55. [Google Scholar] [CrossRef]
- Oshima, Y. Chemical and Enzymatic Transformation of Paralytic Shellfish Toxins in Marine Organisms. In Harmful Marine Algal Blooms; Lassus, P., Arzul, G., Erard, E., Gentien, P., Marcaillou, C., Eds.; Lavoisier Intercept Ltd.: Paris, France, 1995; p. 475. [Google Scholar]
- Regulation (EC) No. 853/2004 of the European Parliament and of the Council of 29 April 2004 Laying Down Specific Hygiene Rules for Food of Animal Origin. OJ L 139. Available online: http://eur-lex.europa.eu (accessed on 17 December 2010).
- Asakawa, M.; Nishimura, F.; Miyazaki, K.; Noguchi, T. Occurrence of Paralytic Shellfish Poison in the starfish Asteria amurensis in Kure Bay, Hiroshima prefecture, Japan. Toxicon 1997, 35, 1081–1087. [Google Scholar] [CrossRef]
- Ito, K.; Asakawa, M.; Sida, Y.; Miyazaki, K. Occurrence of paralytic shellfish poison (PSP) in the starfish Asterina pectinifera collected from the Kure Bay, Hiroshima Prefecture, Japan. Toxicon 2003, 41, 291–295. [Google Scholar] [CrossRef]
- Lin, S.J.; Tsai, Y.H.; Lin, H.P.; Hwang, D.F. Paralytic toxins in Taiwanese starfish Astropecten scoparious. Toxicon 1998, 36, 799–803. [Google Scholar] [CrossRef]
- Van de Riet, J.M.; Gibbs, R.S.; Chou, F.W.; Muggah, P.M.; Rourke, W.A.; Burns, G.; Thomas, K.; Quilliam, M.A. Liquid chromatographic post-column oxidation method for analysis of paralytic shellfish toxins in mussels, clams, scallops, and oysters: Single-laboratory validation. J. AOAC Int. 2009, 92, 1690–1704. [Google Scholar] [PubMed]
- Anon, A. Official Method 2011.02 Determination of Paralytic Shellfish Poisoning Toxins in Mussels, Clams, Oysters and Scallops; Post-column Oxidation Method (PCOX); First Action 2011; AOAC International: Gaithersburg, MD, USA, 2011. [Google Scholar]
- Vale, C.; Alfonso, A.; Vieytes, M.R.; Romarís, X.M.; Arévalo, F.; Botana, A.M.; Botana, L.M. In vitro and in vivo evaluation of paralytic shellfish poisoning toxin potency and the influence of the pH of extraction. Anal. Chem. 2008, 80, 1770–1776. [Google Scholar] [CrossRef] [PubMed]
- Rourke, W.A.; Murphy, C.J.; Pitcher, G.; van de Riet, J.M.; Burns, B.G.; Thomas, K.M.; Quilliam, M.A. Rapid postcolumn methodology for determination of paralytic shellfish toxins in shellfish tissue. J. AOAC Int. 2008, 91, 589–597. [Google Scholar] [PubMed]
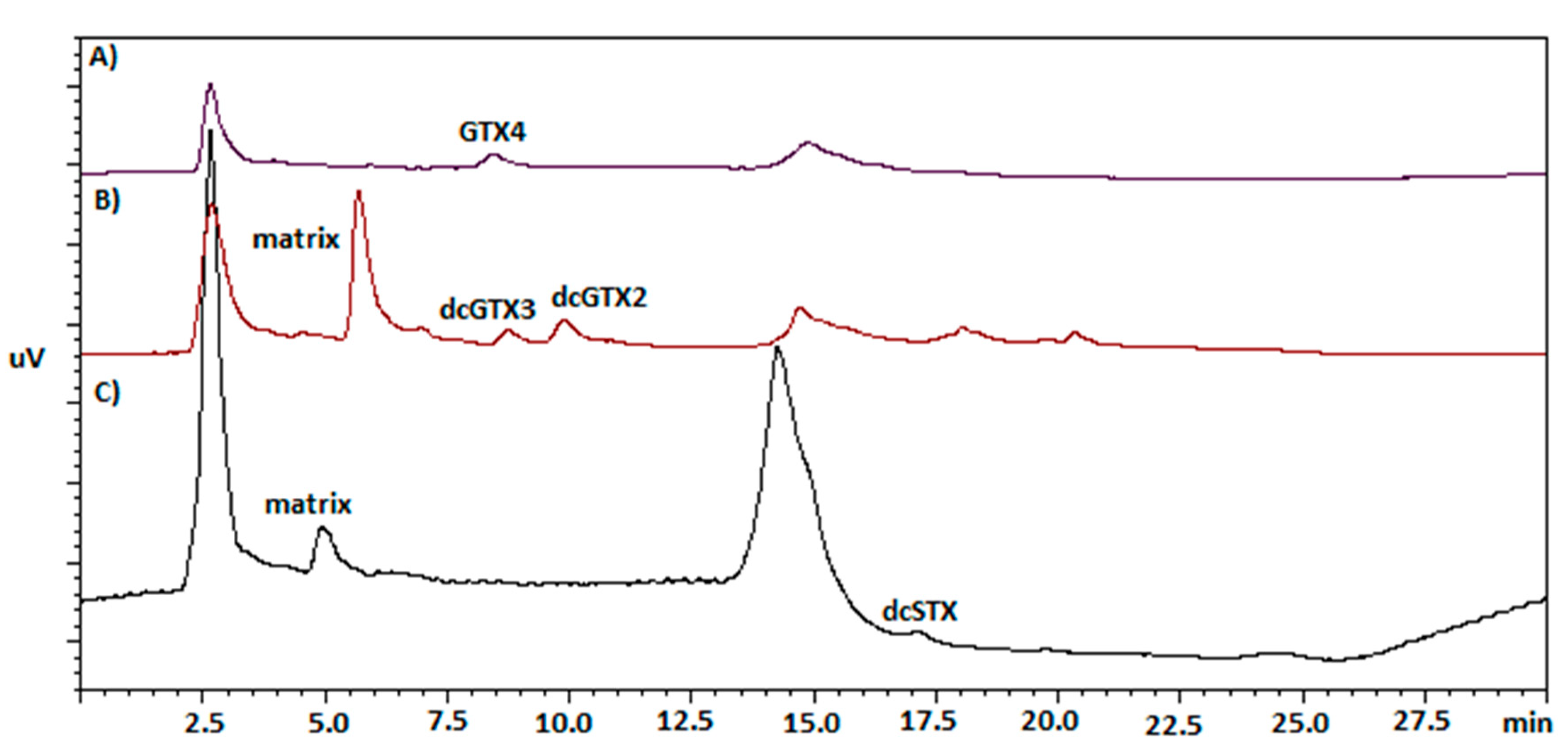
| Sampling Data | Sampling Site | Sample | Species | Code | µg STX.diHCleq/Kg SM |
|---|---|---|---|---|---|
| 8 August 2012 | Northern coast of Madeira | Limpet | Patella ordinaria | 336 | 1123.3 |
| Limpet | Patella aspera | 337 | 122.4 | ||
| 16 September 2012 | Reis Magos | Sea urchin | Paracentrotus lividus | 339#1 | <LOQ |
| Starfish | Ophidiaster ophidianus | 341#1 | 2071 | ||
| Starfish | O. ophidianus | 341#2 | 2224.1 | ||
| Starfish | O. ophidianus | 341#3 | 4625.4 | ||
| Limpet | P. aspera | 344 | 866.4 | ||
| Sea urchin | Arbacia lixula | 345 | <LOQ | ||
| Sea snail | Stramonita haemastoma | 346 | 964.5 | ||
| 18 September 2012 | Caniçal | Limpet | P. aspera | 350 | 12.2 |
| Limpet | Umbraculum umbraculum | 351 | 536.8 | ||
| Starfish | Echinaster sepositus | 353 | 668 | ||
| Sea snail | Charonia lampas | 354 | 1423.4 | ||
| Sea urchin | Diadema africanum | 355#1 | 276.3 | ||
| Sea urchin | D. africanum | 355#2 | 227.9 |
| Sampling Data | Sampling Site | Sample | Species | Code | µg STX.diHCleq/Kg SM |
|---|---|---|---|---|---|
| 7 June 2013 | Lagoa | Sea urchin | Sphaerechinus granularis | 409#3 | 43.4 |
| Sea urchin | S. granularis | 409#4 | 42.5 | ||
| Starfish | O. ophidianus | 412 | 1689.6 | ||
| Sea snail | S. haemastoma | 413 | 939.4 | ||
| Limpet | Patella gomesii | 415 | 1192.4 | ||
| Mosteiros | Limpet | P. gomesii | 420 | 902.3 | |
| 8 June 2013 | Etar | Sea urchin | A. lixula | 421 | <LOQ |
| Ilhéu S. Roque | Sea urchin | A. lixula | 423 | 111.7 | |
| Starfish | O. ophidianus | 424 | 2588.4 | ||
| Sea urchin | S. granularis | 425#1 | <LOQ | ||
| Sea urchin | S. granularis | 425#2 | <LOQ | ||
| Sea urchin | S. granularis | 425#3 | <LOQ | ||
| Starfish | Marthasterias glacialis | 426#2 | 3.8 | ||
| 9 June 2013 | Cruzeiro | Starfish | M. glacialis | 428 | 7744.3 |
| Sea snail | S. haemastoma | 431 | 678.3 | ||
| Caloura | Sea urchin | A. lixula | 432 | <LOQ | |
| Starfish | M. glacialis | 433#1 | 47.5 | ||
| Starfish | M. glacialis | 433#2 | 24.9 | ||
| Sea snail | S. haemastoma | 434 | 544.7 | ||
| Starfish | O. ophidianus | 435 | 920.3 | ||
| 10 June 2013 | Caloura | Starfish | O. ophidianus | 440 | 245 |
| 10 June 2013 | Caloura | Sea snail | S. haemastoma | 443 | 128.6 |
| Sampling Data | Sampling Site | Sample | Species | Code | µg STX.diHCleq/Kg SM |
|---|---|---|---|---|---|
| 22 July 2013 | Casablanca Corniche | Bivalve | Mytilus sp. | 447 | 1376.9 |
| Sea snail | Phorcus lineatus | 448 | 929.4 | ||
| 23 July 2013 | Sidi Bouzid | Sea snail | P. lineatus | 449 | 1404.5 |
| Limpet | Patella sp. | 450 | 1090.5 | ||
| Sea slug | Aplysia depilans | 451 | <LOQ | ||
| Bivalve | Mytilus sp. | 453 | 2266.4 | ||
| Sea snail | Cerithium vulgatum | 454 | 158.8 | ||
| El Jadida Sâada | Sea snail | C. vulgatum | 455 | 2556 | |
| Sea cucumber | Holothuria (Platyperona) sanctori | 458#1 | 2.3 | ||
| Limpet | Patella sp. | 459 | 8.6 | ||
| Starfish | M. glacialis | 463 | 1852.4 | ||
| 24 July 2013 | El Jadida Haras | Barnacle | Pollicipes pollicipes | 464 | 17.7 |
| Mrizika | Bivalve | Mytilus sp. | 465 | 1140.4 | |
| Barnacle | P. pollicipes | 466 | 17.6 | ||
| Limpet | Patella sp. | 467 | 3622.5 | ||
| Bivalve | Mytilus sp. | 468 | 1080.9 | ||
| Sea snail | Gibbula umbilicalis | 469 | 1.6 | ||
| Sea snail | P. lineatus | 470 | 1043.9 | ||
| Starfish | M. glacialis | 473 | 1325.4 | ||
| Sea slug | Onchidella celtica | 474 | 38.8 | ||
| Oualidia | Sea snail | C. lampas | 475 | 0.02 | |
| Sea slug | A. depilans | 476 | 0.6 | ||
| Sea snail | S. haemastoma | 477 | 384 | ||
| Sea snail | P. lineatus | 482 | 85.7 | ||
| Sea snail | G. umbilicalis | 483 | 12.1 | ||
| Barnacle | P. pollicipes | 484 | 17.4 | ||
| Bivalve | Mytilus sp. | 485 | 2708.9 |
| Date | Location | Sampling Site | Geographic Coordinates |
|---|---|---|---|
| September 2012 | Madeira Island | Reis Magos | 32°39′16.21″ N; 16°49′05.29″ W |
| Caniçal | 32°44′20.08″ N; 16°44′17.55″ W | ||
| June 2013 | São Miguel Island | Cruzeiro | 37° 50′31.19″ N; 25° 41′33.61″ W |
| Étar | 37°44′19.31″ N; 25°39′38.84″ W | ||
| São Roque | 37°45′15.35″ N; 25°38′31.60″ W | ||
| Mosteiros | 37°53′25.57″ N; 25°49′14.72″ W | ||
| Lagoa | 37°44′42.38″ N; 25°19′.47″ W | ||
| Caloura | 37°42′49.34″ N; 25°29′54.54″ W | ||
| July 2013 | Morocco Coast | Casablanca corniche | 33°36′01.2″ N; 7°39′57.5″ W |
| El Jadida Haras | 33°14′42.0″ N; 8°28′37.5″ W | ||
| El Jadida Sâada | 33°14′42.4″ N; 8°32′26.9″ W | ||
| Sidi Bouzid | 33°13′57.1″ N; 8°33′20.9″ W | ||
| Mrizika | 32°57′21.8″ N; 8°46′53.2″ W | ||
| Oualidia | 32°43′55.8″ N; 9°02′57.6″ W |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, M.; Rey, V.; Barreiro, A.; Kaufmann, M.; Neto, A.I.; Hassouani, M.; Sabour, B.; Botana, A.; Botana, L.M.; Vasconcelos, V. Paralytic Shellfish Toxins Occurrence in Non-Traditional Invertebrate Vectors from North Atlantic Waters (Azores, Madeira, and Morocco). Toxins 2018, 10, 362. https://doi.org/10.3390/toxins10090362
Silva M, Rey V, Barreiro A, Kaufmann M, Neto AI, Hassouani M, Sabour B, Botana A, Botana LM, Vasconcelos V. Paralytic Shellfish Toxins Occurrence in Non-Traditional Invertebrate Vectors from North Atlantic Waters (Azores, Madeira, and Morocco). Toxins. 2018; 10(9):362. https://doi.org/10.3390/toxins10090362
Chicago/Turabian StyleSilva, Marisa, Verónica Rey, Aldo Barreiro, Manfred Kaufmann, Ana Isabel Neto, Meryem Hassouani, Brahim Sabour, Ana Botana, Luis M. Botana, and Vitor Vasconcelos. 2018. "Paralytic Shellfish Toxins Occurrence in Non-Traditional Invertebrate Vectors from North Atlantic Waters (Azores, Madeira, and Morocco)" Toxins 10, no. 9: 362. https://doi.org/10.3390/toxins10090362
APA StyleSilva, M., Rey, V., Barreiro, A., Kaufmann, M., Neto, A. I., Hassouani, M., Sabour, B., Botana, A., Botana, L. M., & Vasconcelos, V. (2018). Paralytic Shellfish Toxins Occurrence in Non-Traditional Invertebrate Vectors from North Atlantic Waters (Azores, Madeira, and Morocco). Toxins, 10(9), 362. https://doi.org/10.3390/toxins10090362







