Coagulotoxic Cobras: Clinical Implications of Strong Anticoagulant Actions of African Spitting Naja Venoms That Are Not Neutralised by Antivenom but Are by LY315920 (Varespladib)
Abstract
1. Introduction
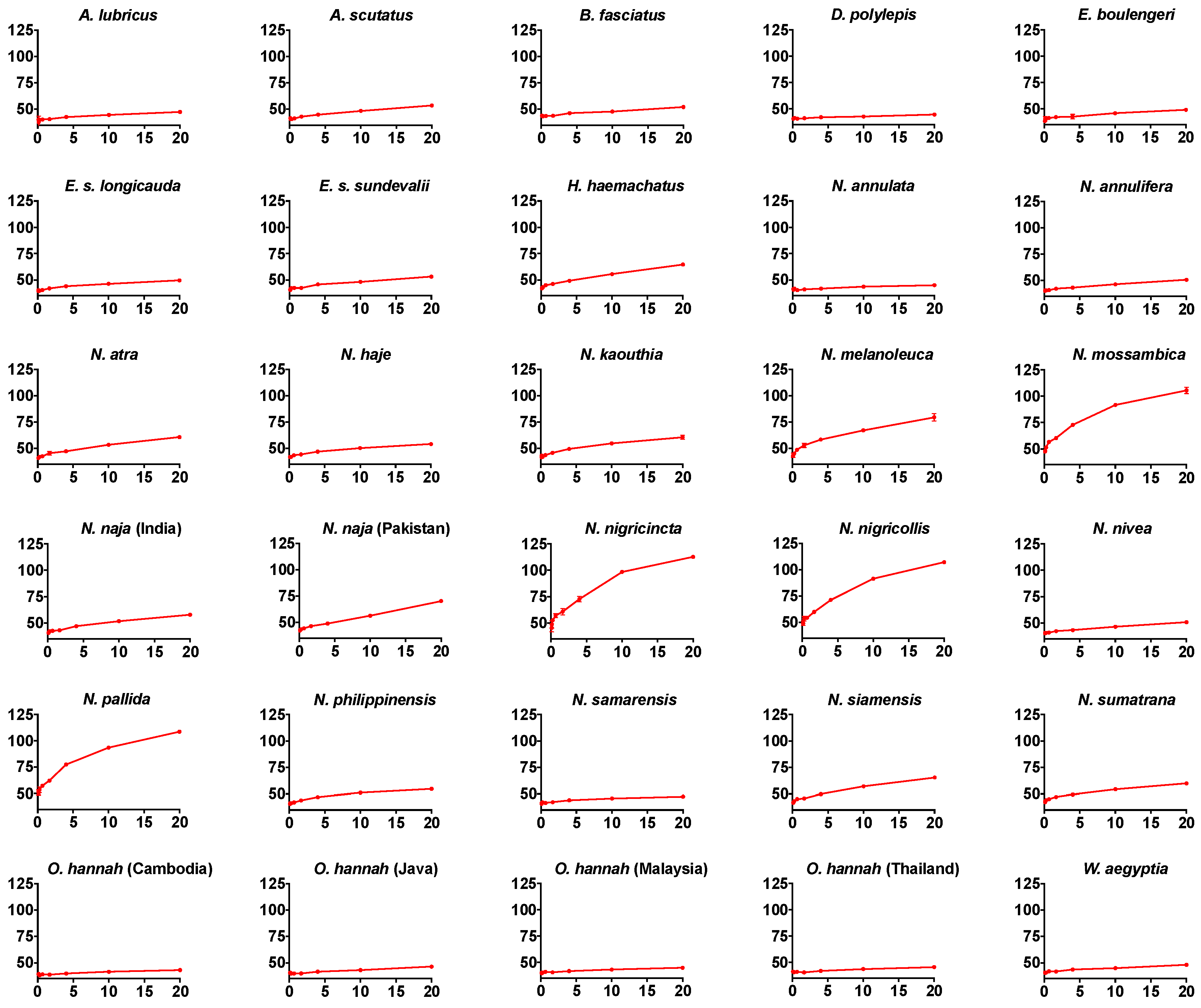
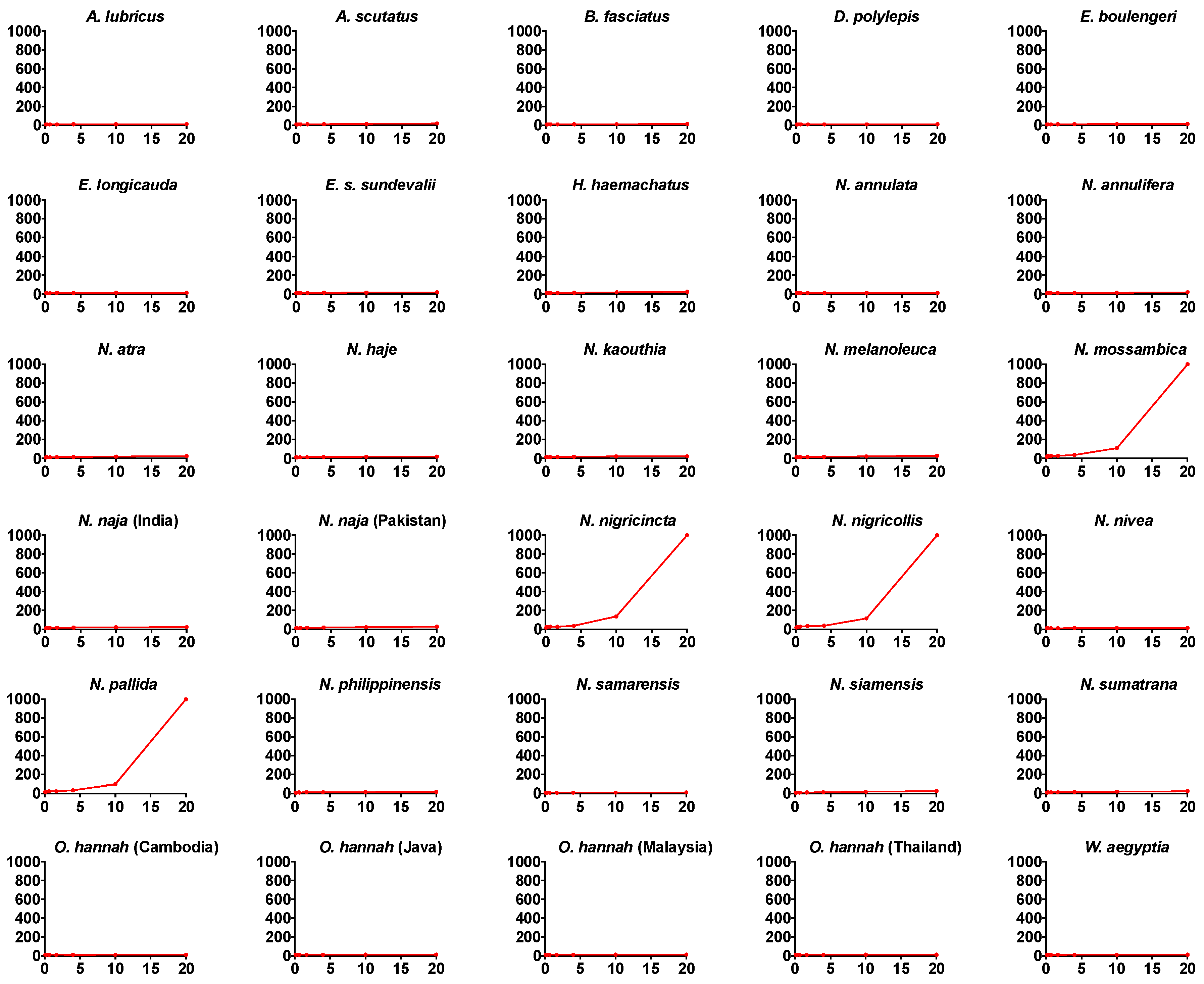
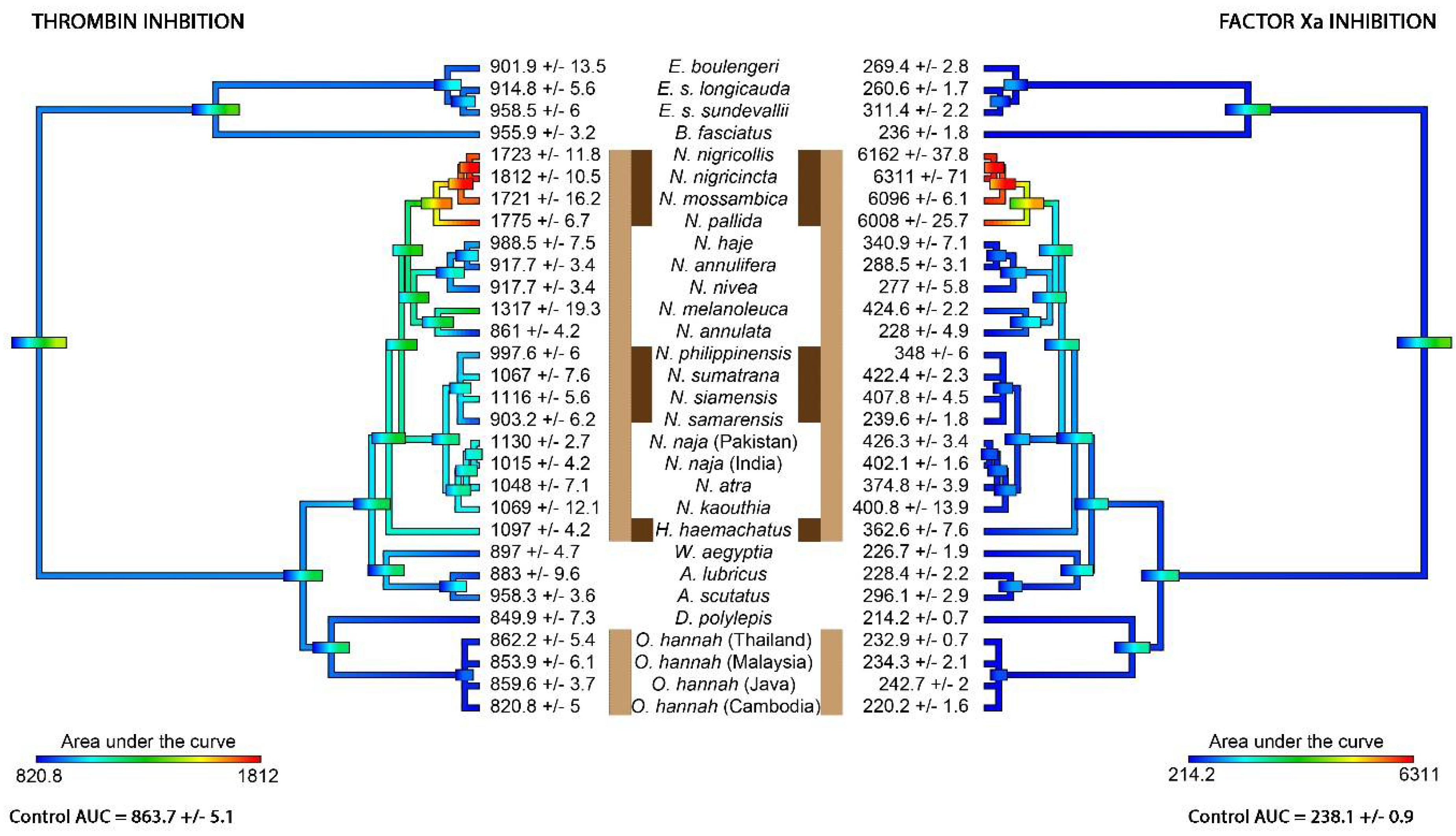
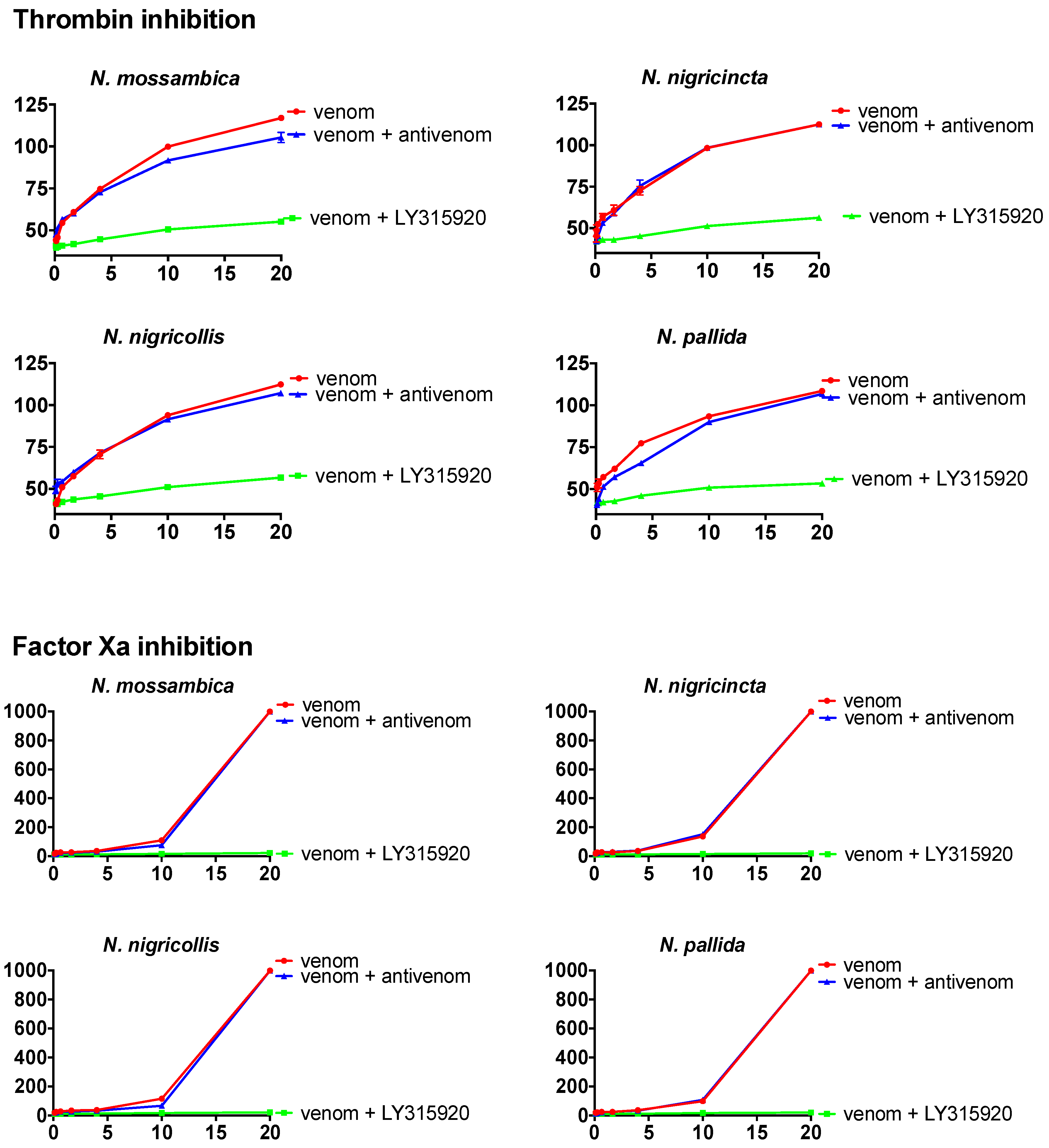
2. Results
3. Discussion
4. Materials and Methods
4.1. Antivenom
4.2. Human Plasma
4.3. Human Fibrinogen
4.4. Coagulation Assays Using the Stago STA-R Max
4.5. Phylogenetic Comparative Analyses
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chippaux, J. Snakebite envenomation turns again into a neglected tropical disease! J. Venom. Anim. Toxins Incl. Trop. Dis. 2017, 23, 38. [Google Scholar] [CrossRef]
- Fry, B.G. Snakebite: When the human touch becomes a bad touch. Toxins 2018, 10, 170. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, J.M.; Theakston, R.D.; Warrell, D.A. Confronting the neglected problem of snake bite envenoming: The need for a global partnership. PLoS Med. 2006, 3, e150. [Google Scholar] [CrossRef]
- Kasturiratne, A.; Wickremasinghe, A.R.; de Silva, N.; Gunawardena, N.K.; Pathmeswaran, A.; Premaratna, R.; Savioli, L.; Lalloo, D.G.; de Silva, H.J. The global burden of snakebite: A literature analysis and modelling based on regional estimates of envenoming and deaths. PLoS Med. 2008, 5, e218. [Google Scholar] [CrossRef] [PubMed]
- Longbottom, J.; Shearer, F.M.; Devine, M.; Alcoba, G.; Chappuis, F.; Weiss, D.J.; Ray, S.E.; Ray, N.; Warrell, D.A.; Ruiz de Castaneda, R.; et al. Vulnerability to snakebite envenoming: A global mapping of hotspots. Lancet 2018, 392, 673–684. [Google Scholar] [CrossRef]
- Boyer, L.; Alagón, A.; Fry, B.G.; Jackson, T.N.W.; Sunagar, K.; Chippaux, J.P. Signs, Symptoms and Treatment of Envenomation. In Venomous Reptiles and Their Toxins: Evolution, Pathophysiology and Biodiscovery; Fry, B.G., Ed.; Oxford University Press: New York, NY, USA, 2015; pp. 32–60. [Google Scholar]
- Harrison, R.A.; Hargreaves, A.; Wagstaff, S.C.; Faragher, B.; Lalloo, D.G. Snake envenoming: A disease of poverty. PLoS Negl. Trop. Dis. 2009, 3, e569. [Google Scholar] [CrossRef] [PubMed]
- Bénard-Valle, M.; Neri-Castro, E.E.; Fry, B.G.; Boyer, L.; Cochran, C.; Alam, M.; Jackson, T.N.W.; Paniagua, D.; Olvera-Rodríguez, F.; Koludarov, I.; et al. Antivenom research and development. In Venomous Reptiles and Their Toxins: Evolution, Pathophysiology and Biodiscovery; Fry, B.G., Ed.; Oxford University Press: New York, NY, USA, 2015; pp. 61–72. [Google Scholar]
- Fry, B.G.; Roelants, K.; Champagne, D.E.; Scheib, H.; Tyndall, J.D.; King, G.F.; Nevalainen, T.J.; Norman, J.A.; Lewis, R.J.; Norton, R.S.; et al. The toxicogenomic multiverse: Convergent recruitment of proteins into animal venoms. Ann. Rev. Genomics Hum. Genet. 2009, 10, 483–511. [Google Scholar] [CrossRef]
- Osipov, A.V.; Filkin, S.Y.; Makarova, Y.V.; Tsetlin, V.I.; Utkin, Y.N. A new type of thrombin inhibitor, noncytotoxic phospholipase A2, from the Naja haje cobra venom. Toxicon 2010, 55, 186–194. [Google Scholar] [CrossRef]
- Mukherjee, A.K.; Kalita, B.; Thakur, R. Two acidic, anticoagulant PLA2 isoenzymes purified from the venom of monocled cobra Naja kaouthia exhibit different potency to inhibit thrombin and factor Xa via phospholipids independent, non-enzymatic mechanism. PloS ONE 2014, 9, e101334. [Google Scholar] [CrossRef]
- Kerns, R.T.; Kini, R.M.; Stefansson, S.; Evans, H.J. Targeting of venom phospholipases: The strongly anticoagulant phospholipase A(2) from Naja nigricollis venom binds to coagulation factor Xa to inhibit the prothrombinase complex. Arch. Biochem. Biophys. 1999, 369, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Kini, R.M. Structure-function relationships and mechanism of anticoagulant phospholipase A2 enzymes from snake venoms. Toxicon 2005, 45, 1147–1161. [Google Scholar] [CrossRef] [PubMed]
- Stefansson, S.; Kini, R.M.; Evans, H.J. The basic phospholipase A2 from Naja nigricollis venom inhibits the prothrombinase complex by a novel nonenzymatic mechanism. Biochemistry 1990, 29, 7742–7746. [Google Scholar] [CrossRef] [PubMed]
- Mladic, M.; Zietek, B.M.; Iyer, J.K.; Hermarij, P.; Niessen, W.M.; Somsen, G.W.; Kini, R.M.; Kool, J. At-line nanofractionation with parallel mass spectrometry and bioactivity assessment for the rapid screening of thrombin and factor Xa inhibitors in snake venoms. Toxicon 2016, 110, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Panagides, N.; Jackson, T.N.; Ikonomopoulou, M.P.; Arbuckle, K.; Pretzler, R.; Yang, D.C.; Ali, S.A.; Koludarov, I.; Dobson, J.; Sanker, B.; et al. How the cobra got its flesh-eating venom: Cytotoxicity as a defensive innovation and its co-evolution with hooding, aposematic marking, and spitting. Toxins 2017, 9, 103. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.S.; Sanders, K.L.; King, B.; Palci, A. Diversification rates and phenotypic evolution in venomous snakes (Elapidae). R. Soc. Open Sci. 2016, 3, 150277. [Google Scholar] [CrossRef]
- Wuster, W.; Crookes, S.; Ineich, I.; Mane, Y.; Pook, C.E.; Trape, J.F.; Broadley, D.G. The phylogeny of cobras inferred from mitochondrial DNA sequences: Evolution of venom spitting and the phylogeography of the African spitting cobras (Serpentes: Elapidae: Naja nigricollis complex). Mol. Phylogenet. Evol. 2007, 45, 437–453. [Google Scholar] [CrossRef] [PubMed]
- Warrell, D.A.; Greenwood, B.M.; Davidson, N.M.; Ormerod, L.D.; Prentice, C.R. Necrosis, haemorrhage and complement depletion following bites by the spitting cobra (Naja nigricollis). Q. J. Med. 1976, 45, 1–22. [Google Scholar] [PubMed]
- Lewin, M.; Samuel, S.; Merkel, J.; Bickler, P. Varespladib (LY315920) appears to be a potent, broad-spectrum, inhibitor of snake venom phospholipase A2 and a possible pre-referral treatment for envenomation. Toxins 2016, 8, 248. [Google Scholar] [CrossRef] [PubMed]
- Saaiman, E.; Buys, C.; Fry, B.G. (Windhoek, Namibia). Haemorrhage following Naja nigricincta envenomation. Personal communication, 2018. [Google Scholar]
- Kandiwa, E.; Mushonga, B.; Samkange, A.; Fabiano, E. Quantitative characterization of the hemorrhagic, necrotic, coagulation-altering properties and edema-forming effects of zebra snake (Naja nigricincta nigricincta) venom. J. Toxicol. 2018, 2018, 6940798. [Google Scholar] [CrossRef]
- Mukherjee, A.K.; Maity, C.R. Biochemical composition, lethality and pathophysiology of venom from two cobras—Naja naja and N. kaouthia. Comp. Biochem. Physiol. Part B Biochem. Molecular Biol. 2002, 131, 125–132. [Google Scholar] [CrossRef]
- Sunagar, K.; Jackson, T.N.W.; Reeks, T.; Fry, B.G. Group I Phospholipase A2 Enzymes. In Venomous Reptiles and Their Toxins: Evolution, Pathophysiology and Biodiscovery; Fry, B.G., Ed.; Oxford University Press: New York, NY, USA, 2015; pp. 324–327. [Google Scholar]
- Lewin, M.R.; Gutierrez, J.M.; Samuel, S.P.; Herrera, M.; Bryan-Quiros, W.; Lomonte, B.; Bickler, P.E.; Bulfone, T.C.; Williams, D.J. Delayed Oral LY333013 Rescues Mice from Highly Neurotoxic, Lethal Doses of Papuan Taipan (Oxyuranus scutellatus) Venom. Toxins 2018, 10, 380. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, J.; Zhang, D.; Xiao, H.; Xiong, S.; Huang, C. Exploration of the inhibitory potential of varespladib for snakebite envenomation. Molecules 2018, 23, 391. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Li, H.; Zhang, D.; Li, Y.; Sun, S.; Huang, C. Inactivation of Venom PLA(2) Alleviates myonecrosis and facilitates muscle regeneration in envenomed mice: A time course observation. Molecules 2018, 23, 1911. [Google Scholar] [CrossRef] [PubMed]
- Tilbury, C.R. Observations on the bite of the Mozambique spitting cobra (Naja mossambica mossambica). S. Afr. Med. J. 1982, 61, 308–313. [Google Scholar] [PubMed]
- Vermaak, S.S. A deadly bed partner: M’fess (Mozambique spitting cobra). SA Orthop. J. 2010, 58–62. [Google Scholar]
- Pattinson, J.P.; Kong, V.Y.; Bruce, J.L.; Oosthuizen, G.V.; Bekker, W.; Laing, G.L.; Wood, D.; Brysiewicz, P.; Clarke, D.L. Defining the need for surgical intervention following a snakebite still relies heavily on clinical assessment: The experience in Pietermaritzburg, South Africa. S. Afr. Med. J. 2017, 107, 1082–1085. [Google Scholar] [CrossRef]
- Müller, G.J. Snake bite in southern Africa: Diagnosis and Management. CME 2012, 30, 362–391. [Google Scholar]
- Rivel, M.; Solano, D.; Herrera, M.; Vargas, M.; Villalta, M.; Segura, A.; Arias, A.S.; Leon, G.; Gutierrez, J.M. Pathogenesis of dermonecrosis induced by venom of the spitting cobra, Naja nigricollis: An experimental study in mice. Toxicon 2016, 119, 171–179. [Google Scholar] [CrossRef]
- Mendez, I.; Gutierrez, J.M.; Angulo, Y.; Calvete, J.J.; Lomonte, B. Comparative study of the cytolytic activity of snake venoms from African spitting cobras (Naja spp., Elapidae) and its neutralization by a polyspecific antivenom. Toxicon 2011, 58, 558–564. [Google Scholar] [CrossRef]
- Paradis, E.; Claude, J.; Strimmer, K. APE: Analyses of phylogenetics and evolution in R language. Bioinformatics 2004, 20, 289–290. [Google Scholar] [CrossRef]
- Revell, L.J. phytools: An R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol. 2012, 3, 217–223. [Google Scholar] [CrossRef]
- Symonds, M.R.E.; Blomberg, S.P. A Primer on Phylogenetic Generalised Least Squares. In Modern Phylogenetic Comparative Methods and Their Application in Evolutionary Biology; Garamszegi, L.Z., Ed.; Springer: Berlin/Heidelberg, Germay, 2014; pp. 105–130. [Google Scholar]
- Orme, D.; Freckleton, R.; Thomas, G.; Petzoldt, T.; Fritz, S.; Isaac, N.; Pearse, W. Caper: Comparative Analyses of Phylogenetics and Evolution in R. R Package Version 0.5.2. Methods Ecol. Evol. 2013, 3, 145–151. [Google Scholar]
- Lister, C.; Arbuckle, K.; Jackson, T.N.W.; Debono, J.; Zdenek, C.N.; Dashevsky, D.; Dunstan, N.; Allen, L.; Hay, C.; Bush, B.; et al. Catch a tiger snake by its tail: Differential toxicity, co-factor dependence and antivenom efficacy in a procoagulant clade of Australian venomous snakes. Comp. Biochem. Physiol. Toxicol. Pharmacol. CBP 2017, 202, 39–54. [Google Scholar] [CrossRef] [PubMed]
- Rogalski, A.; Soerensen, C.; Op den Brouw, B.; Lister, C.; Dashevsky, D.; Arbuckle, K.; Gloria, A.; Zdenek, C.N.; Casewell, N.R.; Gutierrez, J.M.; et al. Differential procoagulant effects of saw-scaled viper (Serpentes: Viperidae: Echis) snake venoms on human plasma and the narrow taxonomic ranges of antivenom efficacies. Toxicol. Lett. 2017, 280, 159–170. [Google Scholar] [CrossRef] [PubMed]
| Thrombin inhibition assay | Step 1. 25 µL of 0.2 mg/mL venom (1 mg/mL 50% glycerol stock diluted with Owren Koller (OK) Buffer (Stago Catalog # 00360) + 75 µL of [50 µL 0.025 M calcium (Stago catalog # 00367 + 25 µL of OK buffer] + 50 µL of phospholipid (Stago catalog #00597) + 25 µL of thrombin (Stago catalog # 00611). Step 2. 120 second incubation. Step 3. Addition of 75 µL of 4 mg/mL fibrinogen. |
| Thrombin inhibition assay (antivenom neutralisation tests) | Step 1. 25 µL of 0.2 mg/mL venom (1 mg/mL 50% glycerol stock diluted with OK Buffer + 75 µL of [50 µL 0.025 M calcium + 25 µL of 5% concentration antivenom] + 50 µL phospholipid + 25 µL of thrombin. Step 2. 120 s incubation. Step 3. Addition of 75 µL of 4 mg/mL fibrinogen. |
| Thrombin inhibition assay (LY315920 [varespladib] neutralisation tests) | Step 1. 25 µL of 0.2 mg/mL venom (1 mg/mL 50% glycerol stock diluted with OK Buffer + 75 µL of [50 µL 0.025 M calcium + 25 µL of 0.025 mg/mL LY315920] + 50 µL phospholipid + 25 µL of thrombin. Step 2. 120 s incubation. Step 3. Addition of 75 µL of 4 mg/mL fibrinogen. |
| FXa inhibition assay | Step 1. 25 µL 0.2 mg/mL venom (1 mg/mL 50% glycerol stock diluted with OK Buffer + 75 μl of [50 µL 0.025 M calcium +25 µL OK buffer] + 50 µL of phospholipid + 25 µL of FXa (Stago catalog # 00311). Step 2. 120 s incubation. Step 3. Addition of 75 µL plasma. |
| FXa inhibition assay (antivenom neutralisation tests) | Step 1. 25 µL 0.2 mg/mL of venom (1 mg/mL 50% glycerol stock diluted with OK Buffer + 75 µL of [50 µL 0.025 M calcium + 25 µL of 5% concentration antivenom] + 50 µL of phospholipid + 50 µL of FXa. Step 2. 120 s incubation. Step 3. Addition of 75 µL of plasma. |
| FXa inhibition assay (LY315920 neutralisation tests) | Step 1. 25 µL of 0.2 mg/mL venom (1 mg/mL 50% glycerol stock diluted with OK Buffer + 75 µL of [50 µL 0.025 M calcium + 25 µL of 0.025 mg/mL LY315920] + 50 µL of phospholipid + 25 µL of FXa. Step 2. 120 s incubation. Step 3. Addition of 75 µL of plasma. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bittenbinder, M.A.; Zdenek, C.N.; Op den Brouw, B.; Youngman, N.J.; Dobson, J.S.; Naude, A.; Vonk, F.J.; Fry, B.G. Coagulotoxic Cobras: Clinical Implications of Strong Anticoagulant Actions of African Spitting Naja Venoms That Are Not Neutralised by Antivenom but Are by LY315920 (Varespladib). Toxins 2018, 10, 516. https://doi.org/10.3390/toxins10120516
Bittenbinder MA, Zdenek CN, Op den Brouw B, Youngman NJ, Dobson JS, Naude A, Vonk FJ, Fry BG. Coagulotoxic Cobras: Clinical Implications of Strong Anticoagulant Actions of African Spitting Naja Venoms That Are Not Neutralised by Antivenom but Are by LY315920 (Varespladib). Toxins. 2018; 10(12):516. https://doi.org/10.3390/toxins10120516
Chicago/Turabian StyleBittenbinder, Mátyás A., Christina N. Zdenek, Bianca Op den Brouw, Nicholas J. Youngman, James S. Dobson, Arno Naude, Freek J. Vonk, and Bryan G. Fry. 2018. "Coagulotoxic Cobras: Clinical Implications of Strong Anticoagulant Actions of African Spitting Naja Venoms That Are Not Neutralised by Antivenom but Are by LY315920 (Varespladib)" Toxins 10, no. 12: 516. https://doi.org/10.3390/toxins10120516
APA StyleBittenbinder, M. A., Zdenek, C. N., Op den Brouw, B., Youngman, N. J., Dobson, J. S., Naude, A., Vonk, F. J., & Fry, B. G. (2018). Coagulotoxic Cobras: Clinical Implications of Strong Anticoagulant Actions of African Spitting Naja Venoms That Are Not Neutralised by Antivenom but Are by LY315920 (Varespladib). Toxins, 10(12), 516. https://doi.org/10.3390/toxins10120516






