Abstract
Background/Objectives: Gut microbiota has been implicated in the pathogenesis of metabolic dysfunction-associated steatotic liver disease (MASLD) and cardiovascular disease (CVD). This study aimed to identify associations between gut dysbiosis and MASLD, regarding body mass index (BMI) and subclinical coronary atherosclerosis (SCA). Methods: We conducted a cross-sectional study of 202 patients with MASLD who had no previous history of CVD. The severity of MASLD was evaluated using a magnetic resonance imaging-based method, and SCA was measured by assessing coronary artery calcification (CAC). Gut microbiota was assessed in fecal specimens using sequencing targeting the V4 region of the 16S rRNA gene. Results: Our results demonstrated that gut microbial profiles between low- and high-BMI groups (<30 vs. ≥30 kg/m2) differed significantly in beta-diversity, but not in alpha-diversity indices. At the genus level, we identified Megamonas, Sutterella, Catenibacterium, and Odoribacter, enriched in the high BMI group. Compared to the low CAC group (<100 AU), MASLD patients with high CAC scores (≥100 AU) exhibited enrichment in Ruminococcus gnavus, Bacteroides, and Lachnoclostridium, along with depletion of several short-chain fatty acid (SCFA)-producing bacteria, such as Faecalibacterium. Multivariate analysis demonstrated that older age, the presence of diabetes, high BMI, fibrosis stage F3-F4, and high plasma trimethylamine oxide (TMAO) levels were independently associated with a high CAC score in patients with MASLD. Conclusions: These data indicated that gut dysbiosis and related metabolites, in association with advanced liver disease, were potential contributors to the progression of SCA in obese patients with MASLD.
1. Introduction
Metabolic dysfunction-associated steatotic liver disease (MASLD), previously termed non-alcoholic fatty liver disease (NAFLD), has been recognized as one of the most common chronic liver diseases, affecting approximately 30–35% of the global adult population []. MASLD is mainly characterized by a combination of steatotic liver disease and phenotypic features of cardiometabolic risk factors []. Regarding hepatic manifestations, MASLD can lead to advanced fibrosis, cirrhosis, and hepatocellular carcinoma (HCC) []. As a high proportion of patients with MASLD will ultimately develop advanced liver disease, it is necessary to assess the severity of liver disease in managing patients with MASLD. Currently, liver biopsy is the gold standard for determining the extent of steatosis and fibrosis. However, this procedure is invasive, has potential complications, and is prone to sampling errors []. As a result, non-invasive methods such as magnetic resonance imaging-proton density fat fraction (MRI-PDFF) and magnetic resonance elastography (MRE) are now considered the best options for evaluating steatosis and fibrosis, respectively [].
In addition to liver-related complications, accumulating data demonstrate that MASLD is strongly linked to an increased risk of extrahepatic disorders, such as cancers, chronic kidney disease (CKD), and cardiovascular disease (CVD). Indeed, CVD, particularly coronary artery disease, is a significant cause of morbidity and mortality in patients with MASLD []. Subclinical coronary atherosclerosis (SCA), an early indicator of atherosclerotic burden, is defined by coronary arterial plaques with or without thrombosis in the absence of overt clinical manifestations []. A recent meta-analysis has indicated that MASLD is significantly associated with SCA, particularly among patients with advanced fibrosis []. Indeed, MASLD is also closely associated with type 2 diabetes mellitus (T2DM) and obesity. According to the Asian-Pacific guideline, persons with body mass index (BMI) of 18.5–22.9, 23–24.9, 25–29.9, and ≥30 kg/m2 were classified as lean, overweight, obese class I, and obese class II, respectively []. However, approximately 10–20% of Asian individuals with MASLD are classified as having normal BMI []. A recent meta-analysis has emphasized the weight-dependent effect, as obese MASLD individuals are significantly more metabolically unhealthy than those with normal BMI []. Although BMI may have some shortcomings, including its inability to differentiate body composition, it provides a good indicator for both sexes and all age groups within the adult population. Given the close connection among obesity, MASLD, and CVD risk, it is necessary to detect SCA early and understand the mechanisms that interact among these cardiometabolic diseases beyond traditional risk factors.
Emerging evidence suggests that gut microbiota may influence the development and severity of MASLD through the gut–liver axis [,]. Notably, gut dysbiosis is characterized by an imbalance in the microbial community, with an increased abundance of potentially harmful bacterial taxa and a reduced abundance of beneficial bacterial taxa, which contributes to the pathogenesis of MASLD. An altered intestinal barrier and translocation of bacterial components and metabolites, including lipopolysaccharides (LPS) and other endotoxins, could induce systemic inflammation and potentially drive MASLD progression []. Likewise, several reports have demonstrated that alterations in gut microbial composition and increased intestinal permeability, or leaky gut, may play a crucial role in the development of clinical CVD []. Additionally, gut dysbiosis affects CVD by producing related metabolites, particularly trimethylamine oxide (TMAO). This metabolite is produced by converting choline, betaine, and other precursor compounds into trimethylamine (TMA) by gut bacteria, which is then oxidized to TMAO in the liver []. TMAO can accelerate atherosclerosis through several mechanisms, including modulating lipid metabolism, increasing reactive oxygen species (ROS) production, promoting platelet aggregation and vascular inflammation, and ultimately leading to the formation of atherogenic plaques []. However, current information linking gut dysbiosis to subclinical coronary atherosclerosis in patients with MASLD remains inadequate and thus needs further investigation.
In this cross-sectional study, we aimed to investigate the diversity and composition of gut microbiota in a well-characterized cohort of Thai individuals with MASLD, focusing on BMI and SCA. In this respect, we used MRE and MRI-PDFF measurements to evaluate the severity of fibrosis and steatosis, respectively. To quantify subclinical coronary atherosclerosis, we employed coronary artery calcification (CAC), a reliable and non-invasive tool for assessing the severity of CVD in asymptomatic individuals []. It has been shown that CAC is closely linked to the severity of atherosclerosis and its prognosis, irrespective of conventional CVD risk factors []. Moreover, we identified various clinical and laboratory parameters that may be independently associated with the severity of CAC using logistic regression analyses. Together, our findings might provide additional valuable data to better understand how gut dysbiosis affects the pathogenesis of MASLD and SCA.
2. Materials and Methods
2.1. Participants and Study Design
The participants in this report were selected from a subgroup of Thai patients aged ≥18 years, who were undergoing follow-up in the outpatient clinic in the King Chulalongkorn Memorial Hospital in Bangkok, Thailand. These patients had evidence of liver steatosis based on vibration-controlled transient elastography (FibroScan with controlled attenuation parameter, CAP > 248 dB/m) or findings on ultrasound examination. Among them, we further confirmed the diagnosis of liver steatosis by using MRI-PDFF (MRI-PDFF ≥ 5.4%) [], and there were 202 patients enrolled in this study. Exclusion criteria were (1) associated with other chronic liver diseases, such as chronic viral hepatitis (HBsAg, anti-HCV positive), autoimmune hepatitis (anti-nuclear antibodies, anti-smooth muscle antibodies positive), and hemochromatosis (high transferrin saturation and ferritin levels); (2) human immunodeficiency virus (HIV) infection; (3) the presence of decompensated cirrhosis and complications, including ascites, variceal bleeding, and HCC, (4) self-report of excessive alcohol intake (>10 g/day in women and >20 g/day in men), and (5) previous history of CVD such as coronary artery disease or stroke.
The patients were advised to avoid herbal or nutritional supplements, proton pump inhibitors, antibiotics, prebiotics, and probiotics for at least 4 weeks before recruitment. Clinical data and anthropometric measurements were documented at enrollment. The protocols (IRB No. 981-64 and No. 769/66) were approved by the Institutional Review Board of the Faculty of Medicine, Chulalongkorn University, Bangkok, Thailand. The study adhered to the Declaration of Helsinki and the principles of good clinical practice, following the provision of written informed consent by the patients.
2.2. Liver Stiffness and Steatosis Measurement
MRE and MRI-PDFF were used to assess liver stiffness and steatosis by the MR imaging system Philips Ingenia at 3.0 T (Philips Healthcare, Best, The Netherlands). According to a systematic review and meta-analysis in MASLD, the cut-off values for fibrosis stages ≥F1, ≥F2, ≥F3, and F4 by MRE measurement were 2.6, 3.0, 3.6, and 4.7 kPa, respectively []. As to MRI-PDFF, the cut-off points for steatosis grades ≥1, ≥2, and ≥3 were 5.4%, 15.4%, and 20.4%, respectively []. A radiologist evaluated and interpreted the acquired imaging information without being aware of the participant’s clinical and laboratory data.
2.3. CAC Quantification Using a Multisection CT Scan
Participants underwent CAC quantification using a multisection CT scan (Somatom Sensation 64; Siemens Medical Systems), as described previously []. This process was used to quantify coronary calcium by multiplying the weighted density score by the pixel area of the calcification speck []. The standard scan parameters consisted of a 3 mm section thickness, 1.2 × 24 mm collimation, a 0.37 s rotation, and spiral mode with 120 kVp at 80 mAs, with reconstruction operating at 60% of the R-R interval. The Agatston scores assessing the severity of CAC were confirmed by a single experienced observer blinded to the patient’s clinical and laboratory data. The presence of SCA is established by a CAC score of >0 Agatston units (AU), while a CAC score of ≥100 and ≥400 AU characterizes intermediate and severe risk, respectively. In this study, 97 (48%) patients were evaluated for the CAC score.
2.4. Fecal Collection, DNA Extraction, and Sequencing
Fecal samples collected from the participants in the DNA/RNA Shield™-Fecal Collection tubes (Zymo Research Corp, Irvine, CA, USA) were shaken and stored at −80 °C until further analysis. The collected specimens were then extracted for DNA by the ZymoBIOMICS™ DNA Miniprep Kit (Zymo Research Corp, Irvine, CA, USA), following the manufacturer’s instructions. The purity and concentration of total DNA were then measured using a DeNovix™ UV-Vis spectrophotometer (DeNovix Inc., Wilmington, DE, USA), and the DNA was stored at −20 °C. Subsequently, amplicon-based 16S rRNA gene sequencing located in the V4 hypervariable regions was amplified based on forward primer 515F (5′-GTGCCAGCMGCCGCGGTAA-3′) and reverse primer 806R (5′-GGACTACHVGGGTWTCTAAT-3′). Additionally, paired-end sequencing was conducted on the Illumina MiSeq 2 × 300 bp platform (San Diego, CA, USA) at Mod Gut Co., Ltd. (Bangkok, Thailand).
2.5. Gut Microbiota Data Processing and Analysis
The gut microbiome analysis was examined using QIIME 2 48 (version 2024.2) as the raw sequence data was demultiplexed using the q2-demux plugin. Next, reads with expected errors (maxEE) higher than 3.0 were discarded using denoising software DADA2 (via q2-dada2), and the mitochondria and chloroplast 16S sequences were then eliminated. A phylogeny was constructed from representative sequences using the align_to_tree_mafft_fasttree command of the q2-phylogeny plugin. Alpha-diversity indexes and beta-diversity metrics were assessed using the q2-diversity plugin after rarefying the samples to 12,000 reads. Next, Principal Coordinate Analysis (PCoA) was analyzed on the beta-diversity distance metrics. Permutational Multivariate Analysis of Variance (PERMANOVA) was conducted to assess differences in microbial community composition between groups. Statistical significance was defined as p < 0.05. The amplicon sequence variants (ASVs) were taxonomically assigned using the classify-sklearn naive Bayes taxonomy classifier against the Silva (version 138) 99% reference sequences. The Spearman Rank Coefficient was applied to assess the relation between clinical parameters and microbial genera. The clustermap function from the seaborn library in Python version 3.10.13 was employed to yield a correlation graph representing clusters.
2.6. Fecal BCoAT Gene Assessment
The butyryl-CoA: acetate CoA transferase (BCoAT) was assessed to estimate the concentration of butyrate in fecal specimens by all butyrate-producing bacteria. Briefly, the quantification was tested by using qPCR based on 4X CAPITAL™ qPCR Green Master Mix (Biotechrabbit GmbH, Berlin, Germany) with the following degenerate primers: (forward primer) 5′-GCIGAICATTTCACITGGAAYWSITGGCAYATG-3′ and (reverse primer) 5′-CCTGCCTTTGCAATRTCIACRAANGC-3′ as previously described []. The qPCR conditions began with a DNA-denaturation step at 95 °C for 15 min, followed by 40 cycles of denaturation at 95 °C for 15 s, annealing at a primer-specific temperature for 20 s, and extension at 72 °C for 30 s. The quantification of the BCoAT gene was examined by normalizing its assay using the V3-V4 gene as the representative of total bacteria.
2.7. Plasma Biomarker Measurement
Peripheral blood samples were collected from the patients, handled within two hours to separate the plasma component, and preserved at −80 °C until analysis. Plasma intestinal fatty acid binding protein (I-FABP), a biomarker of intestinal permeability [], was determined using an ELISA kit (Hycult Biotech, Uden, The Netherlands) after being diluted at 1:2, following the manufacturer’s protocol. Plasma lipopolysaccharide-binding protein (LBP) concentrations, an accurate biomarker of LPS and endotoxemia [], were analyzed using ELISA kits (Hycult Biotech, Uden, The Netherlands) after being diluted at 1:1000. Plasma choline, betaine, and TMAO levels were assessed using ultra-high performance liquid chromatography-tandem spectrometry (UHPLC-MS/MS), as previously described []. In this study, the limits of detection were 47 pg/mL for I-FABP, 0.94 pg/mL for LPS, and 0.05 µM for TMAO. The intra-assay coefficients of variation (CVs) ranged from 3.2 to 6.4% for I-FABP, 4.13 to 6.7% for LPS, and 1.40 to 7.62% for TMAO, whereas the inter-assay CVs of the respective tests ranged from 0.2 to 1.6%, 3.13 to 4.62%, and 1.65 to 7.15%, respectively.
2.8. Statistical Analysis
The statistical evaluation of the variables was performed using SPSS (version 22.0.0, SPSS Inc., Chicago, IL, USA) and GraphPad Prism (version 9.5.0, GraphPad Software, Inc., San Diego, CA, USA) as appropriate. Categorical data were analyzed using the Chi-square test and one-way ANOVA. The Mann–Whitney test was applied to compare the distributions of two unmatched groups. Spearman’s rank test was applied to analyze correlations between parameters. Univariate and multivariable analyses were performed using SPSS binary logistic regression to identify the parameters associated with a CAC greater than 100. A p-value < 0.05 was considered statistically significant.
3. Results
3.1. Clinical Characteristics of Patients
The clinical characteristics of the patients in this study, categorized by BMI (<30 kg/m2, n = 151 and ≥30 kg/m2, n = 51), are presented in Table 1. Compared to patients with low BMI, the high BMI group was significantly younger but had a significantly higher MRI-PDFF value and average CAC score. There was no difference in gender distribution, as well as in the frequencies of T2DM, hypertension (HT), and dyslipidemia (DLP) between the groups. Additionally, the biochemical blood tests, including liver function and renal function tests, as well as the average MRE measurement, were similar between groups.

Table 1.
Characteristics of patients with MASLD according to BMI.
3.2. The Alpha and Beta Diversities of Gut Microbiota
The alpha diversities of bacteria, as measured by the Chao1, Observed, and Shannon indices, were compared between patients in low- and high-BMI categories (<30 vs. ≥30 kg/m2). Our results demonstrated that there were no significant differences in any of the indices between the two studied groups (Mann–Whitney test, p = 0.247, 0.298, and 0.556, respectively). These results indicated that bacterial richness and evenness did not differ according to the patients’ BMI (Figure 1a–c).
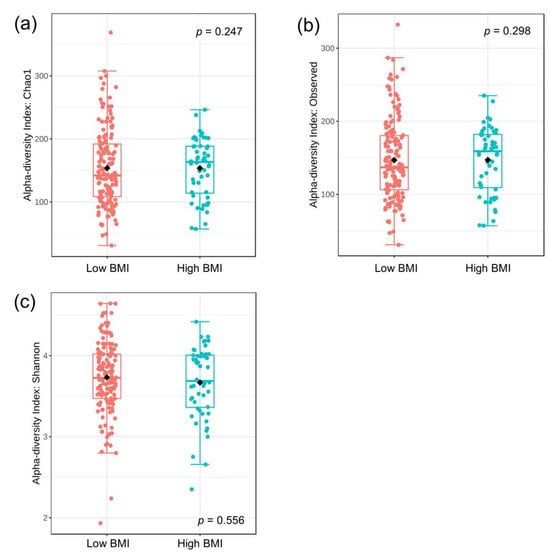
Figure 1.
Alpha diversities between the low and high BMI groups: (a) Chao1 index, (b) observed index, and (c) Shannon index. Statistical measures were median, interquartile range, and outliers. The statistical analysis was performed using the Mann–Whitney test.
To measure the compositional dissimilarity of bacteria between the low and high BMI groups, the Bray–Curtis, Jaccard, Unweighted Unifrac, and Weighted Unifrac indices were applied in conjunction with PERMANOVA tests. The results were then visualized in a Principal Coordinate Analysis (PCoA) plot, as shown in Figure 2a–d. The data demonstrated a significant difference in beta diversity between the low and high BMI groups, as indicated by the Bray–Curtis and Jaccard indices (PERMANOVA, p = 0.007 and p = 0.006, respectively). However, there was no such difference regarding Unweighted Unifrac and Weighted Unifrac indices (p = 0.216 and p = 0.419, respectively).
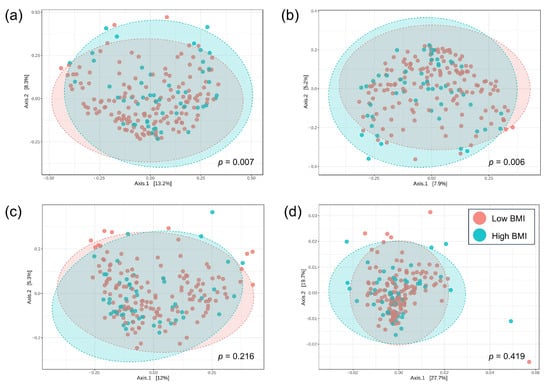
Figure 2.
The principal coordinate analysis (PCoA) of beta-diversity profiling between the low and high BMI groups: (a) Bray–Curtis index, (b) Jaccard index, (c) Unweighted Unifrac index, (d) Weighted Unifrac index. The differences in beta diversity were tested PERMANOVA.
We also compared the diversities of gut microbiota in association with CAC scores (<100 vs. ≥100 AU). Regarding alpha diversities, only the Shannon index showed a significant difference between groups (Mann–Whitney test, p = 0.025). However, the Chao1, Observed, and Simpson indices did not differ significantly (p = 0.103, p = 0.087, and p = 0.066, respectively) (Figure S1). For beta diversities, only the Bray–Curtis displayed a significant difference between the low and high CAC score groups (PERMANOVA, p = 0.049). In contrast, no significant difference was found for Jaccard, Unweighted Unifrac, and Weighted Unifrac indices (p = 0.065, p = 0.120, and p = 0.387, respectively) (Figure S2).
3.3. Gut Bacterial Compositional Analysis
To determine whether the difference in gut microbial composition was associated with obesity, we compared the top 20 relative abundances of bacteria at the genus level according to patients’ BMI category (Figure 3 and Table S1).
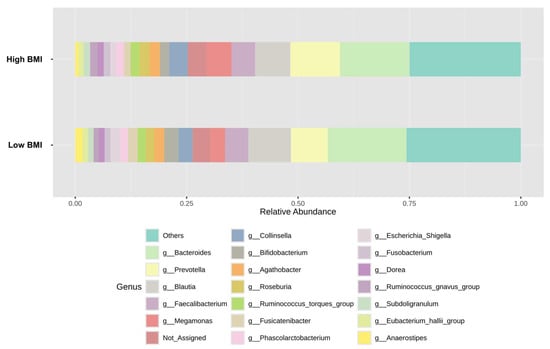
Figure 3.
Top 20 of relative bacterial composition at the genus level of the low and high BMI groups.
To further evaluate the significant differences in microbiota composition between low-and high-BMI groups, the LEfSe method was performed (Figure 4). Our results showed that the enriched Blautia, Bifidobacterium, Anaerostipes, and Butyricicoccus, among others, were observed in patients with low BMI. Additionally, several bacterial genera, including Megamonas, Sutterella, Catenibacterium, and Odoribacter, were found to be increased in the high BMI subjects. These data indicated a significant distinction in gut microbiota composition among patients with MASLD based on the severity of obesity.
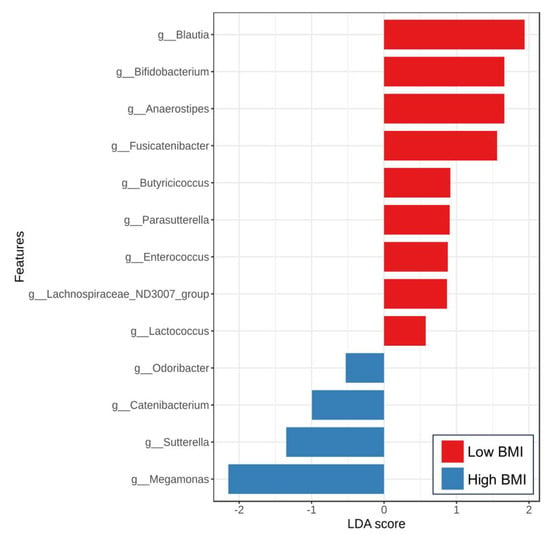
Figure 4.
Linear discriminant analysis (LDA) effect size (LEfSe) analysis of gut microbiota between the low and high BMI groups at the genus level (LAD > 2, p < 0.05).
Regarding CAC severity, the analysis using the LEfSe method also demonstrated the discriminating bacterial abundance between groups (Figure 5). Among the low CAC group, the enrichment of short-chain fatty acid (SCFA)-producing bacteria, including Faecalibacterium, Agathobacter, Fusicatenibacter, Eubacterium, Lachnospiraceae, and Christensenella, was detected. Among patients with high CAC scores above 100 AU, an increased abundance of Ruminococcus gnavus, Bacteroides, and Lachnoclostridium was observed compared to those with less severe CAC scores. Notably, the CAC score was positively correlated with the relative abundance of Ruminococcus gnavus (r = 0.232, p = 0.022) and Bacteroides (r = 0.221, p = 0.029). In contrast, the CAC score was negatively correlated with the abundance of several genera, including Faecalibacterium (r = −0.345, p = 0.001), Agathobacter (r = −0.252, p = 0.012), Fusicatenibacter (r = −0.408, p < 0.001), and Lachnospira (r = −0.289, p = 0.004).
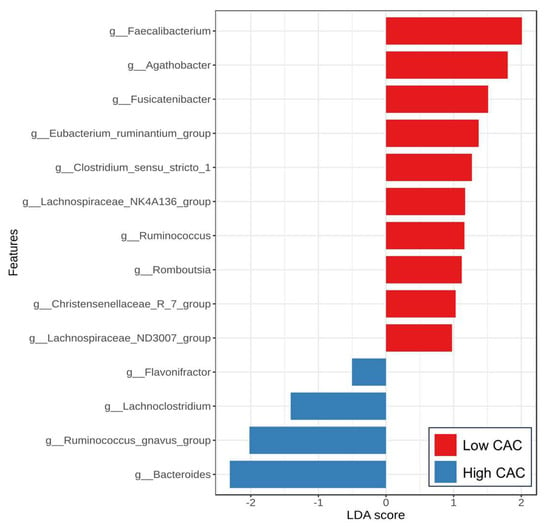
Figure 5.
Linear discriminant analysis (LDA) effect size (LEfSe) analysis of gut microbiota between the low and high CAC score groups at the genus level (LAD > 2, p < 0.05).
3.4. Fecal BCoAT Level
We also measured the levels of BCoAT in fecal specimens to evaluate the production of butyrate by gut microbiota. The results showed no difference in BCoAT expression between the low and high BMI groups (0.030 ± 0.040 vs. 0.035 ± 0.063, p = 0.539). However, patients with a high CAC score (≥100 AU) had significantly lower fecal BCoAT levels than those with a low CAC score <100 AU (0.020 ± 0.024 vs. 0.041 ± 0.068, p = 0.046). Fecal BCoAT had a weak negative correlation with age (r = −0.177, p = 0.012), MRE (r = −0.153, p = 0.031), and CAC score (r = −0.265, p = 0.008), but displayed no correlation with PDFF (r = 0.041, p = 0.571).
3.5. Plasma Biomarker Levels
Plasma concentrations of I-FABP and LBP, representing gut epithelial permeability and bacterial translocation, respectively, were further evaluated. Our results demonstrated that the low BMI group had similar I-FABP levels compared with the high BMI group (778.5 ± 887.3 vs. 648.2 ± 611.1 ng/mL, p = 0.337) but showed significantly lower LBP levels (15,174.8 ± 6449.8 vs. 18,577.5 ± 7363.7 ng/mL, p = 0.002). Comparing groups regarding CAC scores, there was a significant increase in I-FABP levels in patients with high CAC compared to those with low CAC scores (1137.0 ± 1306.8 vs. 560.0 ± 445.4 ng/mL, p = 0.002). Regarding LBP, the high CAC group tended to have higher levels than the low CAC group, but no significant difference was found (17,654.8 ± 8053.6 vs. 15,688.6 ± 6450.0 ng/mL, p = 0.187).
Plasma TMAO and related biomarkers were further investigated. Comparing the low and high BMI groups, there was no significant difference regarding choline (13.1 ± 6.4 vs. 13.6 ± 5.1 µM, p = 0.641), betaine (41.3 ± 26.4 vs. 41.4 ± 25.1 µM, p = 0.983), and TMAO levels (4.1 ± 7.5 vs. 3.0 ± 2.6 µM, p = 0.339). Comparing the low and high CAC groups, there was no significant difference regarding choline (12.6 ± 5.8 vs. 14.5 ± 7.6 µM, p = 0.177) and betaine (35.1 ± 25.9 vs. 39.8 ± 26.4 µM, p = 0.381). However, patients with a high CAC score had significantly elevated TMAO levels than those with a low CAC score (6.0 ± 8.0 vs. 2.2 ± 2.1 µM, p = 0.002).
Plasma I-FABP was significantly correlated with patients’ age (r = 0.338, p < 0.001), LBP (r = 0.202, p = 0.004), MRE (r = 0.395, p < 0.001), and CAC score (r = 0.347, p < 0.001), but showed a negative correlation with fecal BCoAT (r = −0.234, p = 0.001). Plasma LBP level showed a positive correlation with BMI (r = 0.211, p = 0.003) and CAC score (r = 0.405, p < 0.001) but displayed a negative correlation with fecal BCoAT (r = −0.268, p < 0.001). The significant correlations of selected parameters are shown in Figure 6.
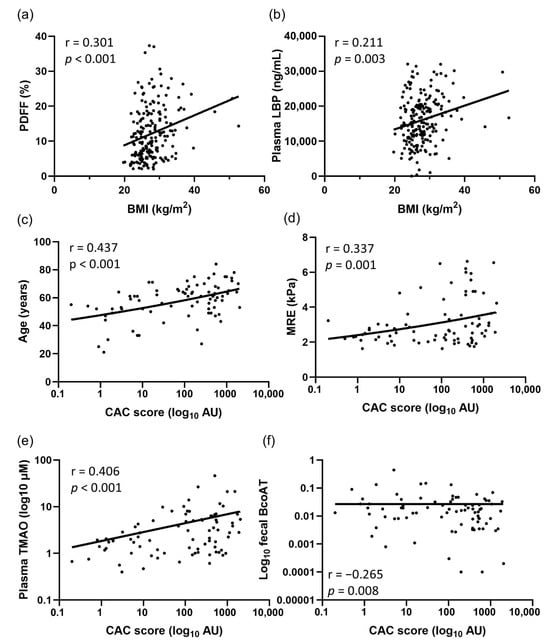
Figure 6.
The correlations of selected parameters regarding BMI and CAC scores: (a) BMI vs. PDFF, (b) BMI vs. LBP, (c) CAC score vs. age, (d) CAC score vs. MRE, (e) CAC score vs. TMAO, and (f) CAC score vs. BCoAT. The statistical analysis was performed using Spearman’s rank test.
Plasma biomarkers in correlation with the abundance of bacterial genera were also analyzed. TMAO levels showed a weakly positive correlation with Bacteroides (r = 0.145, p = 0.040) and Escherichia_Shigella (r = 0.157, p = 0.025). However, they exhibited a negative correlation with the abundance of several SCFA-producing bacteria, including Faecalibacterium (r = −0.321, p < 0.001), Agathobacter (r = −0.322, p < 0.001), Fusicatenibacter (r = −0.217, p = 0.002), Eubacterium (r = −0.237, p = 0.001), and Lachnospiraceae (r = −0.211, p = 0.003). Additionally, plasma I-FABP concentrations were positively correlated with Bacteroides (r = 0.147, p = 0.039) and Escherichia_Shigella (r = 0.172, p = 0.015) but negatively correlated with Faecalibacterium (r = −0.161, p = 0.024). For LBP, the marker was negatively correlated with the abundance of Blautia (r = −0.163, p = 0.021).
3.6. Univariate and Multivariate Analyses
We further examined whether any of the parameters studied were independently associated with a high CAC score greater than 100 AU. The variables entered into univariate and multivariate analyses included age, gender, BMI, T2DM, HT, DLP, smoking, serum aspartate aminotransferase (AST), alanine aminotransferase (ALT), serum albumin, platelet count, estimated glomerular filtration rate (eGFR) liver steatosis, and liver fibrosis, as well as biomarkers associated with gut dysbiosis, including fecal BCoAT, plasma I-FABP, LBP, choline, betaine and TMAO. Multivariate analysis demonstrated that older age, the presence of T2DM, high BMI, Fibrosis stage F3-F4, and high plasma TMAO levels were independently associated with a high CAC score in patients with MASLD (Table 2).

Table 2.
Factors associated with high CAC scores (≥100 AU).
4. Discussion
MASLD is currently the most common chronic liver disease associated with several systemic metabolic disorders, particularly CVD consequences [,]. MASLD has become an increasing public health concern in Asian populations due to the obesity epidemic related to Westernized dietary patterns and lifestyle changes. A meta-analysis has demonstrated that the severity of metabolic dysregulation is weight-dependent, and obese individuals typically display more metabolic dysfunction compared with non-obese patients []. Additionally, obese individuals with MASLD commonly exhibit worse clinical outcomes, including end-stage liver disease and extrahepatic complications, compared to those with normal weight []. In this study, we also demonstrated that MASLD with advanced fibrosis/cirrhosis (F3-F4) was independently associated with high CAC scores, aligning with the findings in a recent report from eastern China []. These results are also similar to those of the meta-analysis in MASLD, indicating an association between fibrosis severity and subclinical atherosclerosis []. Moreover, current evidence suggests that progressive MASLD is an independent risk for developing CVD, particularly in patients with obesity and advanced fibrosis []. In this study, our multivariate analysis revealed that a BMI of ≥30, the presence of T2DM, along with the patient’s age, fibrosis severity, and high plasma TMAO levels, were independent factors associated with CAC scores exceeding 100 AU. Indeed, this cut-off value of CAC is considered a high risk for future development of CVD events in a systematic review, indicating that lifestyle modification and therapeutic intervention, such as statins, are required to prevent further complications []. Together, these results suggest that the intersection of MASLD severity, metabolic disorders, including obesity and T2DM, and gut-derived metabolites displays a complex clinical scenario challenging healthcare in the management and prevention of CVD.
Obesity and associated metabolic disorders, including T2DM and MASLD, have markedly increased over the last decades. Recent advances suggest that gut microbiota play a crucial role in obesity and its related diseases []. Gut dysbiosis, characterized by an altered intestinal microbial balance, is a disruption of microbial composition and function that affects several pathophysiological conditions, including MASLD and CVD [,]. In this report, one of the main findings was the changes in gut microbiota, particularly altered beta-diversities but not alpha-diversities. These data suggested that the overall bacterial richness and evenness were comparable between groups according to the BMI of patients; however, the specific gut bacterial genera presented in each group were not the same. Regarding gut microbial community at the genus level, we identified several bacterial genera enriched in the obese population, including Megamonas, Sutterella, Catenibacterium, and Odoribacter. In contrast, various SCFA-producing bacteria, such as Blautia, Bifidobacterium, Anaerostipes, and Butyricicoccus, had a higher abundance in individuals with lower BMI categories. Aligned with our data, a higher abundance of Megamonas has been reported to be associated with the development of obesity and MASLD in Chinese children and adolescents []. Similarly, Megamonas was shown to be associated with obesity in Mexican populations []. Interestingly, a recent large-scale study involving over 1000 Chinese individuals identified potential obesity-related gut microbial enterotypes, specifically the Megamonas cluster, which is enriched in obese individuals []. In a mouse model and functional analysis, the bacteria in this cluster may contribute to the development of obesity by inhibiting fatty acid transport, degrading myoinositol, and increasing dietary lipid absorption []. Collectively, these data indicate that Megamonas is a potential obesity-related gut microbiota and establish its clinical importance through human studies, animal models, and cell experiments.
Additionally, a meta-analysis demonstrated that the relative abundances of Sutterella and Catenibacterium were significantly higher in obese adults compared to non-obese adults, findings consistent with our results []. Sutterella is a genus of bacteria associated with several human diseases, including inflammatory bowel disease (IBD) []. Recent data suggested a potential link between its abundance and obesity, particularly in children and adolescents, associated with high-fat diets and inflammation []. Moreover, Sutterella was associated with fibrosis in morbidly obese patients with MASLD undergoing bariatric surgery []. Catenibacterium was shown to be markedly enriched in patients with end-stage renal disease []. The bacterium was also found to be increased in the obese population residing in areas with transitions towards Westernized diets and inactive lifestyles []. Together, our report and previous data supported a significant distinction in gut microbiota profiles in obese individuals with MASLD. Understanding the specific microbial composition associated with obese MASLD may lead to more personalized and targeted therapy.
Accumulating evidence also suggests that gut dysbiosis plays a role in influencing the development and severity of CVD and SCA. Here, we demonstrated the significant alterations of gut microbial composition in our MASLD cohort according to SCA severity. This study is one of the first to report on the characteristics of gut dysbiosis in patients with MASLD, regarding SCA. The CAC score was positively correlated with the relative abundance of potentially harmful bacteria such as Ruminococcus gnavus and Bacteroides. In contrast, the CAC score was negatively correlated with the abundance of several SCFA-producing genera, including Faecalibacterium, Agathobacter, among others. Additionally, in patients with high SCA scores of above 100 AU, enrichment in Ruminococcus gnavus, Bacteroides, and Lachnoclostridium, together with depletion of several SCFA-producing bacteria, including Faecalibacterium, Agathobacter, Fusicatenibacter, Eubacterium, Lachnospiraceae, and Christensenella, was observed compared to those with less severe CAC scores.
Recent data have suggested a crucial role of Lachnoclostridium in promoting the development of atherosclerosis via its production of TMA []. Other reports have also demonstrated that Ruminococcus gnavus is consistently associated with various disorders, such as inflammatory bowel disease (IBD), neurological diseases, malignancies, and cardiometabolic disorders, including MASLD, T2DM, and obesity []. Moreover, the enriched abundance of Ruminococcus gnavus was also found in patients with CVD after adjusting for conventional risk factors, such as dyslipidemia and T2DM [,]. A large population-based cohort also indicated a strong association between the relative abundance of Ruminococcus gnavus and the amount of body fat, after adjustment for multiple confounders []. In this context, recent data have provided mechanistic insights suggesting that bacterium-derived tryptamine and phenethylamine may play a pathogenic role in connection with cardiometabolic disorders [].
SCFAs, principally including acetate, propionate, and butyrate, are the main metabolites generated by microbial fermentation of dietary fiber. SCFAs are implicated in mediating various physiological functions, including intestinal epithelial proliferation, gut barrier permeability, energy modulation, and immune response []. Increasing interest is being shown in the role of butyrate in cardio-protective effects against atherosclerotic complications. As demonstrated in both in vitro and in vivo studies, butyrate regulates gene expression related to lipid and glucose metabolism, thereby suppressing the oxidative stress of endothelial cells and enhancing vascular integrity, which may reduce the occurrence and progression of atherosclerosis []. In a longitudinal study of overweight/obese people, for example, increased serum or fecal butyrate is associated with lowered systolic blood pressure and the prevalence of HT, thereby reducing the risk of CVD []. In a large-scale study of Chinese populations, several major SCFA-producing bacteria, including Roseburia and Faecalibacterium, significantly declined in patients with atherosclerotic CVD compared to healthy controls, indicating a potential protective role of these bacteria and their metabolites in atherosclerosis []. Moreover, other cross-sectional studies have confirmed the reduced abundance of SCFA-producing bacteria in patients with subclinical atherosclerosis [,]. Similarly, our study demonstrated that several SCFA-producing bacteria were depleted in patients with high CAC scores. Among these bacterial taxa, Faecalibacterium has emerged as an independent predictor for CVD occurrence and progression, potentially impacting reduced LPS synthesis and enhanced intestinal mucosal barrier integrity [].
Growing evidence has revealed that gut dysbiosis plays a detrimental role in the development of atherosclerosis by increasing intestinal permeability and producing injurious metabolites []. Indeed, this so-called ‘leaky gut’ phenotype has been demonstrated in several animal and human studies of CVD []. Disrupted intestinal permeability facilitates the translocation of microbes and related metabolites into the systemic circulation, which induces the production of proinflammatory mediators, immune responses, and vascular dysfunction, contributing to the development of atherosclerosis []. In this context, our study demonstrated that plasma levels of I-FABP, a surrogate marker of intestinal damage, were significantly higher in univariate analysis among patients with CAC ≥ 100 AU than those with CAC < 100 AU. Our report also showed that plasma levels of I-FABP were positively correlated with liver fibrosis measured by MRE and total CAC scores, reflecting the potential role of disrupted intestinal permeability in promoting adverse liver and cardiovascular events. Elevated circulating I-FABP levels were also shown in a previous study demonstrating a positive association with adverse clinical outcomes in patients with CVD [].
TMAO, a metabolite derived from gut microbiota, originates from TMA, which is produced by consuming dietary precursors such as choline, carnitine, and betaine, abundantly found in meat and eggs []. After TMA is generated, it is absorbed and subsequently converted into TMAO by hepatic flavin-containing monooxygenase 3 (FMO3) enzyme before being released into the circulation []. Although its exact role in cardiometabolic disorders is not fully clarified, it has been constantly shown that TMAO is directly or indirectly involved in the pathogenesis of CVD, as well as representing a significant risk factor affecting the prognosis of CVD []. In this study, elevated circulating TMAO levels were independently associated with increased SCA scores in multivariate analysis, consistent with previous results from a multicenter study in Thailand []. Of note, we also demonstrated that TMAO levels had a positive correlation with the abundance of Bacteroides and Escherichia_Shigella. In contrast, TMAO exhibited a negative correlation with the abundance of several SCFA-producing bacteria, including Faecalibacterium and Agathobacter, among others. These results indicated that enriched inflammatory bacteria and depleted SCFA-producing genera might play a role in the production of TMAO and be connected to the severity of SCA, as demonstrated in this study.
TMAO is considered an independent risk factor for CVD events in a meta-analysis, and elevated TMAO levels have been related to an increased risk of CVD events, including coronary atherosclerosis and heart failure []. For example, the analysis of a large cohort revealed a significant correlation between elevated circulating TMAO levels and increased atherosclerotic risk []. Preclinical and clinical data have also suggested mechanistic links between TMAO and the development of various cardiometabolic events, including the promotion of platelet aggregation and the induction of vascular inflammation []. To strengthen these findings, the selective targeting of the TMAO signaling pathway via small molecules that directly inhibit microbial-derived TMA production has been shown to improve the clinical consequences of atherosclerosis and vascular thrombosis [].
Despite the valuable results of this study, some limitations should be acknowledged. First, this study identified associations between gut microbiota and clinical variables in patients with MASLD and SCA, although these findings do not establish a cause-and-effect relationship. The study also included a relatively small sample size of patients with MASLD, which might be susceptible to selection bias; for instance, our data showed a higher average BMI among younger than older individuals, contrary to most reports. Moreover, the report was designed as a cross-sectional study conducted in a single institute in Thailand, and longitudinal cohorts among other populations should further confirm our findings. Additionally, the complexity of the gut microbiota is modulated by several factors, including lifestyle and dietary patterns. Thus, these factors may influence the gut microbial profiling analysis in our cohort, given the limited dietary information available. Finally, the gut microbial composition was characterized by 16S rRNA sequencing, which may be inadequate for providing species-level information and comprehensive functional insights. Accordingly, whole-genome or shotgun metagenomics would offer additional details regarding microbial communities and functional assessment among strain-level differences. Considering these limitations, our strength was the inclusion of a well-characterized cohort of patients with MASLD at various stages of fibrosis. Moreover, our analysis using multiple logistic regression to examine the association with CAC scores allowed us to evaluate multiple factors and minimize their confounding effects.
5. Conclusions
In conclusion, our study provided significant evidence of gut dysbiosis and metabolites, particularly TMAO, in association with not only obesity but also SCA severity in patients with MASLD. The altered gut microbiota in patients with a high SCA burden was characterized by an increased abundance of pathogenic bacterial taxa, particularly Ruminococcus gnavus, and a reduction in SCFA-producing genera, such as Faecalibacterium, among others. These data lead to a better understanding of the mechanistic role of gut dysbiosis concerning obese MASLD and SCA. Given the growing evidence linking TMAO to CVD, the role of this metabolite as a complementary biomarker in identifying high-risk individuals within the MASLD population warrants further investigation. Additionally, our results may support personalized interventions, such as modulating gut dysbiosis through probiotic or prebiotic supplements, which could be beneficial in slowing CVD progression in patients with MASLD [].
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/nu17172759/s1, Figure S1: Alpha diversities between the low and high CAC score groups. Figure S2: The principal coordinate analysis (PCoA) of beta-diversity profiling between the low and high CAC score groups. Table S1: Top 20 of relative bacterial composition at the genus level according to BMI groups.
Author Contributions
Conceptualization, K.I., P.K. and P.T.; methodology and participants recruitment, K.I., P.K. and P.T.; Investigation, K.I., P.K., M.C., M.T., P.C., P.P. and P.T.; data curation, P.P.; formal analysis, K.I., P.K., M.C., M.T., P.C., P.P. and P.T.; writing—original draft preparation, K.I. and P.K.; writing—review and editing, P.T.; supervision, P.T.; funding acquisition, P.T. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the NSRF via the Program Management Unit for Human Resources & Institutional Development, Research and Innovation (PMU-B, grant number B36G660010), and the National Research Council of Thailand (grant number N84H670030). Kittiya Islam was supported by the Second Century Fund (C2F), Chulalongkorn University for the postdoctoral fellowship.
Institutional Review Board Statement
The study was conducted in compliance with the Declaration of Helsinki and the principles of Good Clinical Practice. The study protocol was approved by the Institutional Review Board of the Faculty of Medicine, Chulalongkorn University (IRB nos. 981-64 (approval date: 1 February 2022) and 769/66 (approval date: 21 May 2024)).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgments
We would like to thank all participants for providing the clinical specimens and data, as well as the staff at the Center of Excellence in Hepatitis and Liver Cancer, Faculty of Medicine, Chulalongkorn University, Bangkok, Thailand. We would like to thank ModGut Co., Ltd., Bangkok, Thailand, for providing the sequencing of gut microbial profiles.
Conflicts of Interest
Author Pinidphon Prombutara was employed by the company Mod Gut Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Rinella, M.E.; Lazarus, J.V.; Ratziu, V.; Francque, S.M.; Sanyal, A.J.; Kanwal, F.; Romero, D.; Abdelmalek, M.F.; Anstee, Q.M.; Arab, J.P.; et al. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. J. Hepatol. 2023, 79, 1542–1556. [Google Scholar] [CrossRef]
- Younossi, Z.; Tacke, F.; Arrese, M.; Chander Sharma, B.; Mostafa, I.; Bugianesi, E.; Wai-Sun Wong, V.; Yilmaz, Y.; George, J.; Fan, J.; et al. Global Perspectives on Nonalcoholic Fatty Liver Disease and Nonalcoholic Steatohepatitis. Hepatology 2019, 69, 2672–2682. [Google Scholar] [CrossRef]
- Chalasani, N.; Younossi, Z.; Lavine, J.E.; Charlton, M.; Cusi, K.; Rinella, M.; Harrison, S.A.; Brunt, E.M.; Sanyal, A.J. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology 2018, 67, 328–357. [Google Scholar] [CrossRef]
- Hsu, C.; Caussy, C.; Imajo, K.; Chen, J.; Singh, S.; Kaulback, K.; Le, M.D.; Hooker, J.; Tu, X.; Bettencourt, R.; et al. Magnetic Resonance vs Transient Elastography Analysis of Patients With Nonalcoholic Fatty Liver Disease: A Systematic Review and Pooled Analysis of Individual Participants. Clin. Gastroenterol. Hepatol. 2019, 17, 630–637.e638. [Google Scholar] [CrossRef]
- Lazarus, J.V.; Mark, H.E.; Anstee, Q.M.; Arab, J.P.; Batterham, R.L.; Castera, L.; Cortez-Pinto, H.; Crespo, J.; Cusi, K.; Dirac, M.A.; et al. Advancing the global public health agenda for NAFLD: A consensus statement. Nat. Rev. Gastroenterol. Hepatol. 2022, 19, 60–78. [Google Scholar] [CrossRef]
- Kawai, K.; Finn, A.V.; Virmani, R.; Subclinical Atherosclerosis, C. Subclinical Atherosclerosis: Part 1: What Is it? Can it Be Defined at the Histological Level? Arterioscler. Thromb. Vasc. Biol. 2024, 44, 12–23. [Google Scholar] [CrossRef]
- Jamalinia, M.; Zare, F.; Lankarani, K.B. Systematic review and meta-analysis: Association between liver fibrosis and subclinical atherosclerosis in nonalcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2023, 58, 384–394. [Google Scholar] [CrossRef]
- Tham, K.W.; Abdul Ghani, R.; Cua, S.C.; Deerochanawong, C.; Fojas, M.; Hocking, S.; Lee, J.; Nam, T.Q.; Pathan, F.; Saboo, B.; et al. Obesity in South and Southeast Asia-A new consensus on care and management. Obes. Rev. 2023, 24, e13520. [Google Scholar] [CrossRef]
- Ye, Q.; Zou, B.; Yeo, Y.H.; Li, J.; Huang, D.Q.; Wu, Y.; Yang, H.; Liu, C.; Kam, L.Y.; Tan, X.X.E.; et al. Global prevalence, incidence, and outcomes of non-obese or lean non-alcoholic fatty liver disease: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2020, 5, 739–752. [Google Scholar] [CrossRef]
- Tang, A.; Ng, C.H.; Phang, P.H.; Chan, K.E.; Chin, Y.H.; Fu, C.E.; Zeng, R.W.; Xiao, J.; Tan, D.J.H.; Quek, J.; et al. Comparative Burden of Metabolic Dysfunction in Lean NAFLD vs Non-lean NAFLD—A Systematic Review and Meta-analysis. Clin. Gastroenterol. Hepatol. 2023, 21, 1750–1760. [Google Scholar] [CrossRef]
- Tilg, H.; Cani, P.D.; Mayer, E.A. Gut microbiome and liver diseases. Gut 2016, 65, 2035–2044. [Google Scholar] [CrossRef]
- Jayakumar, S.; Loomba, R. Review article: Emerging role of the gut microbiome in the progression of nonalcoholic fatty liver disease and potential therapeutic implications. Aliment. Pharmacol. Ther. 2019, 50, 144–158. [Google Scholar] [CrossRef]
- Kobayashi, T.; Iwaki, M.; Nakajima, A.; Nogami, A.; Yoneda, M. Current Research on the Pathogenesis of NAFLD/NASH and the Gut-Liver Axis: Gut Microbiota, Dysbiosis, and Leaky-Gut Syndrome. Int. J. Mol. Sci. 2022, 23, 11689. [Google Scholar] [CrossRef]
- Witkowski, M.; Weeks, T.L.; Hazen, S.L. Gut Microbiota and Cardiovascular Disease. Circ. Res. 2020, 127, 553–570. [Google Scholar] [CrossRef]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; Dugar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.M.; et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011, 472, 57–63. [Google Scholar] [CrossRef]
- Kim, M.; Huda, M.N.; Bennett, B.J. Sequence meets function-microbiota and cardiovascular disease. Cardiovasc. Res. 2022, 118, 399–412. [Google Scholar] [CrossRef]
- Folsom, A.R.; Kronmal, R.A.; Detrano, R.C.; O’Leary, D.H.; Bild, D.E.; Bluemke, D.A.; Budoff, M.J.; Liu, K.; Shea, S.; Szklo, M.; et al. Coronary artery calcification compared with carotid intima-media thickness in the prediction of cardiovascular disease incidence: The Multi-Ethnic Study of Atherosclerosis (MESA). Arch. Intern. Med. 2008, 168, 1333–1339. [Google Scholar] [CrossRef]
- Gu, Q.; Cen, L.; Lai, J.; Zhang, Z.; Pan, J.; Zhao, F.; Yu, C.; Li, Y.; Chen, C.; Chen, W.; et al. A meta-analysis on the diagnostic performance of magnetic resonance imaging and transient elastography in nonalcoholic fatty liver disease. Eur. J. Clin. Investig. 2021, 51, e13446. [Google Scholar] [CrossRef]
- Chattranukulchai, P.; Vassara, M.; Siwamogsatham, S.; Buddhari, W.; Tumkosit, M.; Ketloy, C.; Shantavasinkul, P.; Apornpong, T.; Lwin, H.M.S.; Kerr, S.J.; et al. High-Sensitivity Troponins and Subclinical Coronary Atherosclerosis Evaluated by Coronary Calcium Score Among Older Asians Living With Well-Controlled Human Immunodeficiency Virus. Open Forum Infect. Dis. 2023, 10, ofad234. [Google Scholar] [CrossRef]
- Agatston, A.S.; Janowitz, W.R.; Hildner, F.J.; Zusmer, N.R.; Viamonte, M., Jr.; Detrano, R. Quantification of coronary artery calcium using ultrafast computed tomography. J. Am. Coll. Cardiol. 1990, 15, 827–832. [Google Scholar] [CrossRef]
- Louis, P.; Flint, H.J. Development of a Semiquantitative Degenerate Real-Time PCR-Based Assay for Estimation of Numbers of Butyryl-Coenzyme A (CoA) CoA Transferase Genes in Complex Bacterial Samples. Appl. Environ. Microbiol. 2007, 73, 2009–2012. [Google Scholar] [CrossRef]
- Vanuytsel, T.; Tack, J.; Farre, R. The Role of Intestinal Permeability in Gastrointestinal Disorders and Current Methods of Evaluation. Front. Nutr. 2021, 8, 717925. [Google Scholar] [CrossRef]
- Schumann, R.R.; Leong, S.R.; Flaggs, G.W.; Gray, P.W.; Wright, S.D.; Mathison, J.C.; Tobias, P.S.; Ulevitch, R.J. Structure and function of lipopolysaccharide binding protein. Science 1990, 249, 1429–1431. [Google Scholar] [CrossRef]
- Ocque, A.J.; Stubbs, J.R.; Nolin, T.D. Development and validation of a simple UHPLC-MS/MS method for the simultaneous determination of trimethylamine N-oxide, choline, and betaine in human plasma and urine. J. Pharm. Biomed. Anal. 2015, 109, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Behari, J.; Wang, R.; Luu, H.N.; McKenzie, D.; Molinari, M.; Yuan, J.M. Severe obesity is associated with worse outcomes than lean metabolic dysfunction-associated steatotic liver disease. Hepatol. Commun. 2024, 8, e0468. [Google Scholar] [CrossRef]
- Ma, G.; Xu, G.; Huang, H. Correlation between metabolic dysfunction-associated steatotic liver disease and subclinical coronary atherosclerosis in eastern China. Diabetol. Metab. Syndr. 2025, 17, 16. [Google Scholar] [CrossRef]
- Simon, T.G.; Roelstraete, B.; Hagstrom, H.; Sundstrom, J.; Ludvigsson, J.F. Non-alcoholic fatty liver disease and incident major adverse cardiovascular events: Results from a nationwide histology cohort. Gut 2022, 71, 1867–1875. [Google Scholar] [CrossRef]
- Doshi, A.; Gandhi, H.; Patel, K.N.; Majmundar, M.; Doshi, R. Aspirin for Primary Prevention in Patients With Elevated Coronary Artery Calcium Score: A Systematic Review of Current Evidences. Am. J. Cardiol. 2024, 220, 9–15. [Google Scholar] [CrossRef]
- Sasidharan Pillai, S.; Gagnon, C.A.; Foster, C.; Ashraf, A.P. Exploring the Gut Microbiota: Key Insights Into Its Role in Obesity, Metabolic Syndrome, and Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2024, 109, 2709–2719. [Google Scholar] [CrossRef]
- Zhou, J.; Zhang, Q.; Zhao, Y.; Zou, Y.; Chen, M.; Zhou, S.; Wang, Z. The relationship of Megamonas species with nonalcoholic fatty liver disease in children and adolescents revealed by metagenomics of gut microbiota. Sci. Rep. 2022, 12, 22001. [Google Scholar] [CrossRef]
- Maya-Lucas, O.; Murugesan, S.; Nirmalkar, K.; Alcaraz, L.D.; Hoyo-Vadillo, C.; Pizano-Zarate, M.L.; Garcia-Mena, J. The gut microbiome of Mexican children affected by obesity. Anaerobe 2019, 55, 11–23. [Google Scholar] [CrossRef]
- Wu, C.; Yang, F.; Zhong, H.; Hong, J.; Lin, H.; Zong, M.; Ren, H.; Zhao, S.; Chen, Y.; Shi, Z.; et al. Obesity-enriched gut microbe degrades myo-inositol and promotes lipid absorption. Cell Host Microbe 2024, 32, 1301–1314. [Google Scholar] [CrossRef]
- Pinart, M.; Dotsch, A.; Schlicht, K.; Laudes, M.; Bouwman, J.; Forslund, S.K.; Pischon, T.; Nimptsch, K. Gut Microbiome Composition in Obese and Non-Obese Persons: A Systematic Review and Meta-Analysis. Nutrients 2021, 14, 12. [Google Scholar] [CrossRef]
- Kaakoush, N.O. Sutterella Species, IgA-degrading Bacteria in Ulcerative Colitis. Trends Microbiol. 2020, 28, 519–522. [Google Scholar] [CrossRef]
- Squillario, M.; Bonaretti, C.; La Valle, A.; Di Marco, E.; Piccolo, G.; Minuto, N.; Patti, G.; Napoli, F.; Bassi, M.; Maghnie, M.; et al. Gut-microbiota in children and adolescents with obesity: Inferred functional analysis and machine-learning algorithms to classify microorganisms. Sci. Rep. 2023, 13, 11294. [Google Scholar] [CrossRef]
- Cornejo-Pareja, I.; Amiar, M.R.; Ocana-Wilhelmi, L.; Soler-Humanes, R.; Arranz-Salas, I.; Garrido-Sanchez, L.; Gutierrez-Repiso, C.; Tinahones, F.J. Non-alcoholic fatty liver disease in patients with morbid obesity: The gut microbiota axis as a potential pathophysiology mechanism. J. Gastroenterol. 2024, 59, 329–341. [Google Scholar] [CrossRef]
- Vaziri, N.D.; Wong, J.; Pahl, M.; Piceno, Y.M.; Yuan, J.; DeSantis, T.Z.; Ni, Z.; Nguyen, T.H.; Andersen, G.L. Chronic kidney disease alters intestinal microbial flora. Kidney Int. 2013, 83, 308–315. [Google Scholar] [CrossRef]
- Oduaran, O.H.; Tamburini, F.B.; Sahibdeen, V.; Brewster, R.; Gomez-Olive, F.X.; Kahn, K.; Norris, S.A.; Tollman, S.M.; Twine, R.; Wade, A.N.; et al. Gut microbiome profiling of a rural and urban South African cohort reveals biomarkers of a population in lifestyle transition. BMC Microbiol. 2020, 20, 330. [Google Scholar] [CrossRef]
- Cai, Y.Y.; Huang, F.Q.; Lao, X.; Lu, Y.; Gao, X.; Alolga, R.N.; Yin, K.; Zhou, X.; Wang, Y.; Liu, B.; et al. Integrated metagenomics identifies a crucial role for trimethylamine-producing Lachnoclostridium in promoting atherosclerosis. NPJ Biofilm. Microbiomes 2022, 8, 11. [Google Scholar] [CrossRef]
- Crost, E.H.; Coletto, E.; Bell, A.; Juge, N. Ruminococcus gnavus: Friend or foe for human health. FEMS Microbiol. Rev. 2023, 47, fuad014. [Google Scholar] [CrossRef]
- Jie, Z.; Xia, H.; Zhong, S.L.; Feng, Q.; Li, S.; Liang, S.; Zhong, H.; Liu, Z.; Gao, Y.; Zhao, H.; et al. The gut microbiome in atherosclerotic cardiovascular disease. Nat. Commun. 2017, 8, 845. [Google Scholar] [CrossRef] [PubMed]
- Toya, T.; Corban, M.T.; Marrietta, E.; Horwath, I.E.; Lerman, L.O.; Murray, J.A.; Lerman, A. Coronary artery disease is associated with an altered gut microbiome composition. PLoS ONE 2020, 15, e0227147. [Google Scholar] [CrossRef]
- Grahnemo, L.; Nethander, M.; Coward, E.; Gabrielsen, M.E.; Sree, S.; Billod, J.M.; Engstrand, L.; Abrahamsson, S.; Langhammer, A.; Hveem, K.; et al. Cross-sectional associations between the gut microbe Ruminococcus gnavus and features of the metabolic syndrome. Lancet Diabetes Endocrinol. 2022, 10, 481–483. [Google Scholar] [CrossRef]
- Zhai, L.; Xiao, H.; Lin, C.; Wong, H.L.X.; Lam, Y.Y.; Gong, M.; Wu, G.; Ning, Z.; Huang, C.; Zhang, Y.; et al. Gut microbiota-derived tryptamine and phenethylamine impair insulin sensitivity in metabolic syndrome and irritable bowel syndrome. Nat. Commun. 2023, 14, 4986. [Google Scholar] [CrossRef]
- Nogal, A.; Valdes, A.M.; Menni, C. The role of short-chain fatty acids in the interplay between gut microbiota and diet in cardio-metabolic health. Gut Microbes 2021, 13, 1897212. [Google Scholar] [CrossRef]
- Amiri, P.; Hosseini, S.A.; Ghaffari, S.; Tutunchi, H.; Ghaffari, S.; Mosharkesh, E.; Asghari, S.; Roshanravan, N. Role of Butyrate, a Gut Microbiota Derived Metabolite, in Cardiovascular Diseases: A comprehensive narrative review. Front. Pharmacol. 2021, 12, 837509. [Google Scholar] [CrossRef]
- Tilves, C.; Yeh, H.C.; Maruthur, N.; Juraschek, S.P.; Miller, E.; White, K.; Appel, L.J.; Mueller, N.T. Increases in Circulating and Fecal Butyrate are Associated With Reduced Blood Pressure and Hypertension: Results From the SPIRIT Trial. J. Am. Heart Assoc. 2022, 11, e024763. [Google Scholar] [CrossRef]
- Baragetti, A.; Severgnini, M.; Olmastroni, E.; Dioguardi, C.C.; Mattavelli, E.; Angius, A.; Rotta, L.; Cibella, J.; Caredda, G.; Consolandi, C.; et al. Gut Microbiota Functional Dysbiosis Relates to Individual Diet in Subclinical Carotid Atherosclerosis. Nutrients 2021, 13, 304. [Google Scholar] [CrossRef]
- Zhu, S.; Xu, K.; Jiang, Y.; Zhu, C.; Suo, C.; Cui, M.; Wang, Y.; Yuan, Z.; Xue, J.; Wang, J.; et al. The gut microbiome in subclinical atherosclerosis: A population-based multiphenotype analysis. Rheumatology 2021, 61, 258–269. [Google Scholar] [CrossRef]
- Yang, H.T.; Jiang, Z.H.; Yang, Y.; Wu, T.T.; Zheng, Y.Y.; Ma, Y.T.; Xie, X. Faecalibacterium prausnitzii as a potential Antiatherosclerotic microbe. Cell Commun. Signal 2024, 22, 54. [Google Scholar] [CrossRef]
- Lewis, C.V.; Taylor, W.R. Intestinal barrier dysfunction as a therapeutic target for cardiovascular disease. Am. J. Physiol. Heart Circ. Physiol. 2020, 319, H1227–H1233. [Google Scholar] [CrossRef]
- Kitai, T.; Kim, Y.H.; Kiefer, K.; Morales, R.; Borowski, A.G.; Grodin, J.L.; Tang, W.H.W. Circulating intestinal fatty acid-binding protein (I-FABP) levels in acute decompensated heart failure. Clin. Biochem. 2017, 50, 491–495. [Google Scholar] [CrossRef]
- Thomas, M.S.; Fernandez, M.L. Trimethylamine N-Oxide (TMAO), Diet and Cardiovascular Disease. Curr. Atheroscler. Rep. 2021, 23, 12. [Google Scholar] [CrossRef]
- Senthong, V.; Kiatchoosakun, S.; Wongvipaporn, C.; Phetcharaburanin, J.; Tatsanavivat, P.; Sritara, P.; Phrommintikul, A. Gut microbiota-generated metabolite, trimethylamine-N-oxide, and subclinical myocardial damage: A multicenter study from Thailand. Sci. Rep. 2021, 11, 14963. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Lu, Y.; Yuan, S.; Cai, X.; He, Y.; Chen, J.; Wu, Q.; He, D.; Fang, A.; Bo, Y.; et al. Gut microbiota-derived metabolite trimethylamine-N-oxide and multiple health outcomes: An umbrella review and updated meta-analysis. Am. J. Clin. Nutr. 2022, 116, 230–243. [Google Scholar] [CrossRef]
- Bogiatzi, C.; Gloor, G.; Allen-Vercoe, E.; Reid, G.; Wong, R.G.; Urquhart, B.L.; Dinculescu, V.; Ruetz, K.N.; Velenosi, T.J.; Pignanelli, M.; et al. Metabolic products of the intestinal microbiome and extremes of atherosclerosis. Atherosclerosis 2018, 273, 91–97. [Google Scholar] [CrossRef]
- Roberts, A.B.; Gu, X.; Buffa, J.A.; Hurd, A.G.; Wang, Z.; Zhu, W.; Gupta, N.; Skye, S.M.; Cody, D.B.; Levison, B.S.; et al. Development of a gut microbe-targeted nonlethal therapeutic to inhibit thrombosis potential. Nat. Med. 2018, 24, 1407–1417. [Google Scholar] [CrossRef]
- Wu, H.; Chiou, J. Potential Benefits of Probiotics and Prebiotics for Coronary Heart Disease and Stroke. Nutrients 2021, 13, 2878. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).