The Body Status of Manganese and Activity of This Element-Dependent Mitochondrial Superoxide Dismutase in a Rat Model of Human Exposure to Cadmium and Co-Administration of Aronia melanocarpa L. Extract
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Experimental Animals
2.3. Experimental Protocol
2.4. Assay of the Concentration of Mn in Biological Fluids, Tissues, and Faeces, as Well as in the Labofeed Diets, Redistilled Water, and 0.1% AE
2.5. Evaluation of the Bioavailability and Retention of Mn in the Body
2.6. Estimation of the Total Content of Mn in Internal Organs
2.7. Determination of the Activity of MnSOD and the Concentrations of Mn and Cd in the Mitochondria of the Liver, Kidney, and Brain
2.7.1. Preparation of the Mitochondrial Fraction
2.7.2. Determination of the Activity of MnSOD
2.7.3. Determination of the Concentrations of Mn and Cd
2.7.4. Estimation of the Concentration of Total Protein
2.8. Statistical Analysis
3. Results
3.1. Daily Intake of Mn via the Labofeed Diet and Drinking Fluids
3.2. Effect of Cd and/or AE on the Body Status of Mn
3.2.1. Apparent Absorption and the Body Retention of Mn, and Its Faecal and Urinary Excretion
3.2.2. The Concentration of Mn in the Stomach and Duodenum
3.2.3. The Concentration of Mn in the Serum and Tissues
3.2.4. Total Content of Mn in Internal Organs
3.3. Effect of Cd and/or AE on the Activity of MnSOD, and the Concentrations of Mn and Cd in the Mitochondrial Fraction of the Liver, Kidney, and Brain
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Berger, M.M.; Shenkin, A.; Schweinli, A.; Amrein, K.; Augsburger, M.; Biesalski, H.K.; Bischoff, S.C.; Casaer, M.P.; Gundogan, K.; Lepp, H.L.; et al. ESPEN micronutrient guideline. Clin. Nutr. 2022, 41, 1357–1424. [Google Scholar] [CrossRef] [PubMed]
- Bornhorst, J.; Kipp, A.P.; Haase, H.; Meyer, S.; Schwerdtle, T. The crux of inept biomarkers for risks and benefits of trace elements. Trends Anal. Chem. 2018, 104, 183–190. [Google Scholar] [CrossRef]
- Mehri, A. Trace elements in human nutrition (II)—An update. Int. J. Prev. Med. 2020, 11, 2. [Google Scholar] [CrossRef]
- Balachandran, R.C.; Mukhopadhyay, S.; McBride, D.; Veevers, J.; Harrison, F.E.; Aschner, M.; Haynes, E.N.; Bowman, A.B. Brain manganese and the balance between essential roles and neurotoxicity. J. Biol. Chem. 2020, 295, 6312–6329. [Google Scholar] [CrossRef] [PubMed]
- Scher, D.P.; Goeden, H.M.; Klos, K.S. Potential for manganese-induced neurologic harm to formula-fed infants: A risk assessment of total oral exposure. Environ. Health Persp. 2021, 129, 047011. [Google Scholar] [CrossRef] [PubMed]
- Farina, M.; Avila, D.S.; Rocha, J.B.; Aschner, M. Metals, oxidative stress and neurodegeneration: A focus on iron, manganese and mercury. Neurochem. Int. 2013, 62, 575–594. [Google Scholar] [CrossRef]
- Liu, X.; Song, J.; Zheng, Z.; Guan, H.; Nan, X.; Zhang, N. Effects of excess manganese on the oxidative status, and the expression of inflammatory factors and heat shock proteins in cock kidneys. Biol. Trace Elem. Res. 2020, 197, 639–650. [Google Scholar] [CrossRef]
- Fan, X.M.; Luo, Y.; Cao, Y.M.; Xiong, T.W.; Song, S.; Liu, J.; Fan, Q.Y. Chronic manganese administration with longer intervals between injections produced neurotoxicity and hepatotoxicity in rats. Neurochem. Res. 2020, 45, 1941–1952. [Google Scholar] [CrossRef]
- Eybl, V.; Kotyzová, D.; Leseticky, L.; Bludovská, M.; Koutensky, J. The influence of curcumin and manganese complex of curcumin on cadmium-induced oxidative damage and trace elements status in tissues of mice. J. Appl. Toxicol. 2006, 26, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Eybl, V.; Kotyzová, D. Protective effect of manganese in cadmium-induced hepatic oxidative damage, changes in cadmium distribution and trace elements level in mice. Interdisc. Toxicol. 2010, 3, 68–72. [Google Scholar] [CrossRef]
- Chaudhary, S.; Iram, S.; Raisuddin, S.; Parvez, S. Manganese pre-treatment attenuates cadmium induced hepatotoxicity in Swiss albino mice. J. Trace Elem. Med. Biol. 2015, 29, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Casalino, E.; Calzaretti, G.; Sblano, C.; Landriscina, C. Molecular inhibitory mechanisms of antioxidant enzymes in rat liver and kidney by cadmium. Toxicology 2002, 179, 37–50. [Google Scholar] [CrossRef]
- Ansenberger-Fricano, K.; Ganini, D.; Mao, M.; Chatterjee, S.; Dallas, S.; Mason, R.P.; Stadler, K.; Santos, J.H.; Bonini, M.G. The peroxidase activity of mitochondrial superoxide dismutase. Free Rad. Biol. Med. 2013, 54, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Candas, D.; Li, J.J. MnSOD in oxidative stress response-potential regulation via mitochondrial protein influx. Antioxid. Redox Signal. 2014, 20, 1599–1617. [Google Scholar] [CrossRef] [PubMed]
- Thévenod, F.; Lee, W.K.; Garrick, M.D. Iron and cadmium entry into renal mitochondria: Physiological and toxicological implications. Front. Cell. Dev. Biol. 2020, 8, 848. [Google Scholar] [CrossRef]
- Satarug, S.; Kikuchi, M.; Wisedpanichkij, R.; Li, B.; Takeda, K.; Na-Bangchang, K.; Moore, M.R.; Hirayama, K.; Shibahara, S. Prevention of cadmium accumulation in retinal pigment epithelium with manganese and zinc. Exp. Eye Res. 2008, 87, 587–593. [Google Scholar] [CrossRef]
- Mężyńska, M.; Brzóska, M.M. Environmental exposure to cadmium—A risk for health of the general population in industrialized countries and preventive strategies. Environ. Sci. Pollut. Res. Int. 2018, 25, 3211–3232. [Google Scholar] [CrossRef]
- Zhang, H.; Reynolds, M. Cadmium exposure in living organisms: A short review. Sci. Total Environ. 2019, 678, 761–767. [Google Scholar] [CrossRef]
- Nordberg, G.F.; Bernard, A.; Diamond, G.L.; Duffus, J.H.; Illing, P.; Nordberg, M.; Bergdahl, I.A.; Jin, T.; Skerfving, S. Risk assessment of effects of cadmium on human health (IUPAC Technical Report). Pure Appl. Chem. 2018, 90, 755–808. [Google Scholar] [CrossRef]
- Genchi, G.; Sinicropi, M.S.; Lauria, G.; Carocci, A.; Catalano, A. The effects of cadmium toxicity. Int. J. Environ. Res. Public Health 2020, 17, 3782. [Google Scholar] [CrossRef]
- Ruczaj, A.; Brzóska, M.M. Environmental exposure of the general population to cadmium as a risk factor of the damage to the nervous system—A critical review of current data. J. Appl. Toxicol. 2022, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Brzóska, M.M.; Rogalska, J.; Roszczenko, A.; Gałażyn-Sidorczuk, M.; Tomczyk, M. The mechanism of the osteoprotective action of a polyphenol-rich Aronia melanocarpa extract during chronic exposure to cadmium is mediated by the oxidative defense system. Planta Med. 2016, 82, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Mężyńska, M.; Brzóska, M.M.; Rogalska, J.; Piłat-Marcinkiewicz, B. Extract from Aronia melanocarpa L. berries prevents cadmium-induced oxidative stress in the liver: A study in a rat model of low-level and moderate lifetime human exposure to this toxic metal. Nutrients 2019, 11, 21. [Google Scholar] [CrossRef] [PubMed]
- Mężyńska, M.; Brzóska, M.M.; Rogalska, J.; Galicka, A. Extract from Aronia melanocarpa L. berries protects against cadmium-induced lipid peroxidation and oxidative damage to proteins and DNA in the liver: A study in a rat model of environmental human exposure to this xenobiotic. Nutrients 2019, 11, 758. [Google Scholar] [CrossRef]
- Dąbrowski, A.; Onopiuk, B.M.; Car, H.; Onopiuk, P.; Dąbrowska, Z.N.; Rogalska, J.; Brzóska, M.M.; Dąbrowska, E. Beneficial impact of an extract from the berries of Aronia melanocarpa L. on the oxidative-reductive status of the submandibular gland of rats exposed to cadmium. Antioxidants 2020, 9, 185. [Google Scholar] [CrossRef]
- Borowska, S.; Brzóska, M.M.; Gałażyn-Sidorczuk, M.; Rogalska, J. Effect of an extract from Aronia melanocarpa L. berries on the body status of zinc and copper under chronic exposure to cadmium: An in vivo experimental study. Nutrients 2017, 9, 1374. [Google Scholar] [CrossRef]
- Mouro, V.G.S.; Martins, A.L.P.; Silva, J.; Menezes, T.P.; Gomes, M.L.M.; Oliveira, J.A.; Melo, F.C.S.A.; Matta, S.L.P. Subacute testicular toxicity to cadmium exposure intraperitoneally and orally. Oxid. Med. Cell. Longev. 2019, 2019, 3429635. [Google Scholar] [CrossRef]
- Song, Y.F.; Luo, Z.; Pan, Y.X.; Liu, X.; Huang, C.; Chen, Q.L. Effects of copper and cadmium on lipogenic metabolism and metal element composition in the javelin goby (Synechogobius hasta) after single and combined exposure. Arch. Environ. Contam. Toxicol. 2014, 67, 167–180. [Google Scholar] [CrossRef]
- Fujishiro, H.; Yano, Y.; Takada, Y.; Tanihara, M.; Himeno, S. Roles of ZIP8, ZIP14, and DMT1 in transport of cadmium and manganese in mouse kidney proximal tubule cells. Metallomics 2012, 4, 700–708. [Google Scholar] [CrossRef]
- Goering, P.L.; Klaassen, C.D. Mechanism of manganese-induced tolerance to cadmium lethality and hepatotoxicity. Biochem. Pharmacol. 1985, 34, 1371–1379. [Google Scholar] [CrossRef]
- Brzóska, M.M.; Gałażyn-Sidorczuk, M.; Jurczuk, M.; Tomczyk, M. Protective effect of Aronia melanocarpa polyphenols on cadmium accumulation in the body: A study in a rat model of human exposure to this metal. Curr. Drug Targets 2015, 16, 1470–1487. [Google Scholar] [CrossRef] [PubMed]
- Brzóska, M.M.; Rogalska, J.; Gałażyn-Sidorczuk, M.; Jurczuk, M.; Roszczenko, A.; Tomczyk, M. Protective effect of Aronia melanocarpa polyphenols against cadmium-induced disorders in bone metabolism: A study in a rat model of lifetime human exposure to this heavy metal. Chem. Biol. Interact. 2015, 229, 132–146. [Google Scholar] [CrossRef] [PubMed]
- Brzóska, M.M.; Roszczenko, A.; Rogalska, J.; Gałażyn-Sidorczuk, M.; Mężyńska, M. Protective effect of chokeberry (Aronia melanocarpa L.) extract against cadmium impact on the biomechanical properties of the femur: A study in a rat model of low and moderate lifetime women exposure to this heavy metal. Nutrients 2017, 9, 543. [Google Scholar] [CrossRef]
- Kozłowska, M.; Brzóska, M.M.; Rogalska, J.; Galicka, A. The impact of a polyphenol-rich extract from the berries of Aronia melanocarpa L. on collagen metabolism in the liver: A study in an in vivo model of human environmental exposure to cadmium. Nutrients 2020, 12, 2766. [Google Scholar] [CrossRef]
- Borowska, S.; Brzóska, M.M.; Tomczyk, M. Complexation of bioelements and toxic metals by polyphenolic compounds—Implications for health. Curr. Drug Targets 2018, 19, 1612–1638. [Google Scholar] [CrossRef] [PubMed]
- Ekere, N.R.; Okoye, C.O.B.; Ihedioha, J.N. Novel simultaneous spectrophotometric determinations of Bi (III), Sn (II), Mn (II), V (III) and Se (II) ions in mixed aqueous solutions. J. Chem. Pharm. Res. 2014, 6, 5–8. [Google Scholar]
- Chang, K.C.; Hsu, C.C.; Liu, S.H.; Su, C.C.; Yen, C.C.; Lee, M.J.; Chen, K.L.; Ho, T.J.; Hung, D.Z.; Wu, C.C.; et al. Cadmium induces apoptosis in pancreatic β-cells through a mitochondria-dependent pathway: The role of oxidative stress-mediated c-Jun N-terminal kinase activation. PLoS ONE 2013, 8, e54374. [Google Scholar] [CrossRef]
- Brzóska, M.M.; Kozłowska, M.; Rogalska, J.; Gałażyn-Sidorczuk, M.; Roszczenko, A.; Smereczański, N.M. Enhanced zinc intake protects against oxidative stress and its consequences in the brain: A study in an in vivo rat model of cadmium exposure. Nutrients 2021, 13, 478. [Google Scholar] [CrossRef]
- Borowska, S.; Brzóska, M.M. Chokeberries (Aronia melanocarpa) and their products as a possible means for the prevention and treatment of noncommunicable diseases and unfavorable health effects due to exposure to xenobiotics. Compr. Rev. Food Sci. Food Saf. 2016, 15, 982–1017. [Google Scholar] [CrossRef]
- Zujko, M.E.; Witkowska, A.M.; Waskiewicz, A.; Sygnowska, E. Estimation of dietary intake and patterns of polyphenol consumption in Polish adult population. Adv. Med. Sci. 2012, 57, 375–384. [Google Scholar] [CrossRef]
- Grosso, G.; Stepaniak, U.; Topor-Madry, R.; Szafraniec, K.; Pajak, A. Estimated dietary intake and major food sources of polyphenols in the Polish arm of the HAPIEE study. Nutrition 2014, 30, 1398–1403. [Google Scholar] [CrossRef] [PubMed]
- Ziauddeen, N.; Rosi, A.; del Rio, D.; Amoutzopoulos, B.; Nicholson, S.; Page, P.; Scazzina, F.; Brighenti, F.; Ray, S.; Mena, P. Dietary intake of (poly)phenols in children and adults: Cross-sectional analysis of UK National Diet and Nutrition Survey Rolling Programme (2008–2014). Eur. J. Nutr. 2019, 58, 3183–3198. [Google Scholar] [CrossRef] [PubMed]
- del Bo’, C.; Bernardi, S.; Marino, M.; Porrini, M.; Tucci, M.; Guglielmetti, S.; Cherubini, A.; Carrieri, B.; Kirkup, B.; Kroon, P.; et al. Systematic review on polyphenol intake and health outcomes: Is there sufficient evidence to define a health-promoting polyphenol-rich dietary pattern? Nutrients 2019, 11, 1355. [Google Scholar] [CrossRef]
- Martin, P.; Fareh, M.; Poggi, M.C.; Boulukos, K.E.; Pognonec, P. Manganese is highly effective in protecting cells from cadmium intoxication. Biochem. Biphys. Res. Commun. 2006, 351, 294–299. [Google Scholar] [CrossRef]
- Okuda, B.; Iwamoto, Y.; Tachibana, H.; Sugita, M. Parkinsonism after acute cadmium poisoning. Clin. Neurol. Neurosurg. 1997, 99, 263–265. [Google Scholar] [CrossRef]
- Min, J.; Min, K. Blood cadmium levels and Alzheimer’s disease mortality risk in older US adults. Environ. Health 2016, 15, 69. [Google Scholar] [CrossRef]
- Carmona, A.; Devès, G.; Roudeau, S.; Cloetens, P.; Bohic, S.; Ortega, R. Manganese accumulates within Golgi apparatus in dopaminergic cells as revealed by synchrotron x-ray fluorescence nanoimaging. ACS Chem. Neurosci. 2010, 1, 194–203. [Google Scholar] [CrossRef]
- Seth, T.D.; Agarwal, L.N.; Satija, N.K.; Hasan, M.Z. The effect of lead and cadmium on liver, kidney, and brain levels of cadmium, copper, lead, manganese, and zinc, and on erythrocyte ALA-D activity in mice. Bull. Environ. Contam. Toxicol. 1976, 16, 190–196. [Google Scholar] [CrossRef]
- Brzóska, M.M.; Borowska, S.; Tomczyk, M. Antioxidants as a potential and therapeutic strategy for cadmium. Curr. Drug Targets 2016, 17, 1350–1384. [Google Scholar] [CrossRef]
- Mężyńska, M.; Brzóska, M.M. Review of polyphenol-rich products as potential protective and therapeutic factors against cadmium hepatotoxicity. J. Appl. Toxicol. 2019, 39, 117–145. [Google Scholar] [CrossRef]
- Xu, Z.; Jin, X.; Pan, T.; Liu, T.; Wan, N.; Li, S. Antagonistic effects of selenium on cadmium-induced apoptosis by restoring the mitochondrial dynamic equilibrium and energy metabolism in chicken spleens. Oncotarget 2017, 8, 52629–52641. [Google Scholar] [CrossRef][Green Version]
- Cao, X.; Fu, M.; Bi, R.; Zheng, X.; Fu, B.; Tian, S.; Liu, C.; Li, Q.; Liu, J. Cadmium induced BEAS-2B cells apoptosis and mitochondria damage via MAPK signaling pathway. Chemosphere 2021, 263, 128346. [Google Scholar] [CrossRef] [PubMed]
- Flynn, J.M.; Melov, S. SOD2 in mitochondrial dysfunction and neurodegeneration. Free Radic. Biol. Med. 2013, 62, 4–12. [Google Scholar] [CrossRef] [PubMed]
- Keller, J.N.; Kindy, M.S.; Holtsberg, F.W.; St Clair, D.K.; Yen, H.C.; Germeyer, A.; Steiner, S.M.; Bruce-Keller, A.J.; Hutchins, J.B.; Mattson, M.P. Mitochondrial manganese superoxide dismutase prevents neural apoptosis and reduces ischemic brain injury: Suppression of peroxynitrite production, lipid peroxidation, and mitochondrial dysfunction. J. Neurosci. 1998, 18, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Serino, A.; Salazar, G. Protective role of polyphenols against vascular inflammation, aging and cardiovascular disease. Nutrients 2019, 11, 53. [Google Scholar] [CrossRef]
- Khan, N.; Mukhtar, H. Tea polyphenols in promotion of human health. Nutrients 2019, 11, 39. [Google Scholar] [CrossRef] [PubMed]
- Savici, J.; Cristina, R.T.; Brezovan, D.; Radulov, I.; Balta, C.; Boldura, O.M.; Muselin, F. Aronia melanocarpa ameliorates adrenal cytoarchitecture against the hexavalent chromium-induced injury. Biol. Trace Elem. Res. 2021, 199, 2936–2944. [Google Scholar] [CrossRef] [PubMed]
- Mitra, E.; Ghosh, A.K.; Ghosh, D.; Mukherjee, D.; Chattopadhyay, A.; Dutta, S.; Pattari, S.K.; Bandyopadhyay, D. Protective effect of aqueous Curry leaf (Murraya koenigii) extract against cadmium-induced oxidative stress in rat heart. Food Chem. Toxicol. 2012, 50, 1340–1353. [Google Scholar] [CrossRef]
- Elmallah, M.I.Y.; Elkhadragy, M.F.; Al-Olayan, E.M.; Abdel Moneim, A.E. Protective effect of Fragaria ananassa crude extract on cadmium-induced lipid peroxidation, antioxidant enzymes suppression, and apoptosis in rat testes. Int. J. Mol. Sci. 2017, 18, 957. [Google Scholar] [CrossRef]
- Merra, E.; Calzaretti, G.; Bobba, A.; Storelli, M.M.; Casalino, E. Antioxidant role of hydroxytyrosol on oxidative stress in cadmium-intoxicated rats: Different effect in spleen and testes. Drug Chem. Toxicol. 2014, 37, 420–426. [Google Scholar] [CrossRef]
 , decrease;
, decrease;  , increase), respectively.
, increase), respectively.
 , decrease;
, decrease;  , increase), respectively.
, increase), respectively.
 , decrease;
, decrease;  , increase), respectively.
, increase), respectively.
 , decrease;
, decrease;  , increase), respectively.
, increase), respectively.
 , decrease;
, decrease;  , increase), respectively.
, increase), respectively.
 , decrease;
, decrease;  , increase), respectively.
, increase), respectively.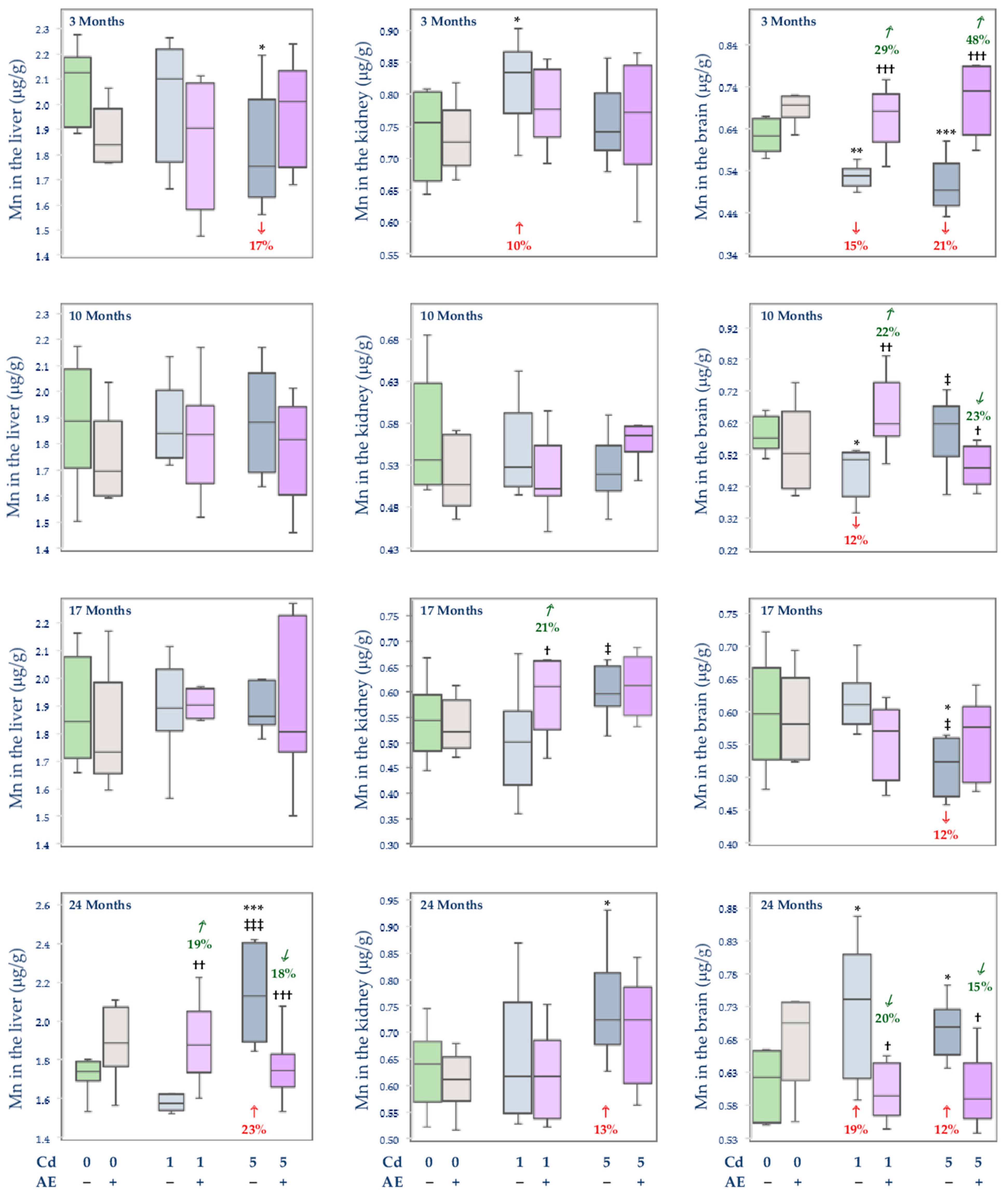
 , decrease;
, decrease; 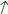 , increase), respectively.
, increase), respectively.
 , decrease;
, decrease;  , increase), respectively.
, increase), respectively.
 , decrease;
, decrease;  , increase), respectively.
, increase), respectively.
 , decrease;
, decrease;  , increase), respectively.
, increase), respectively.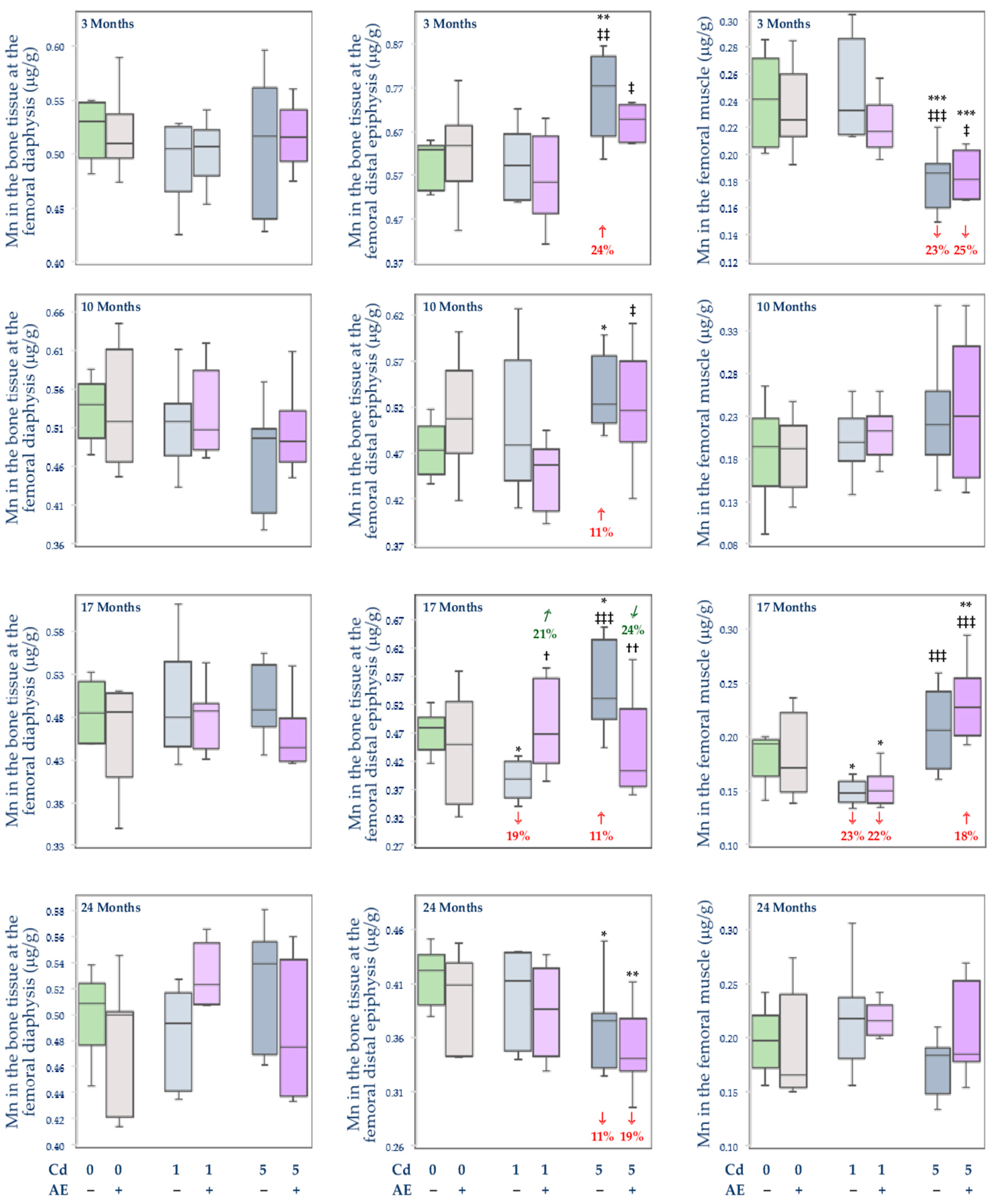
 , decrease), respectively.
, decrease), respectively.
 , decrease), respectively.
, decrease), respectively.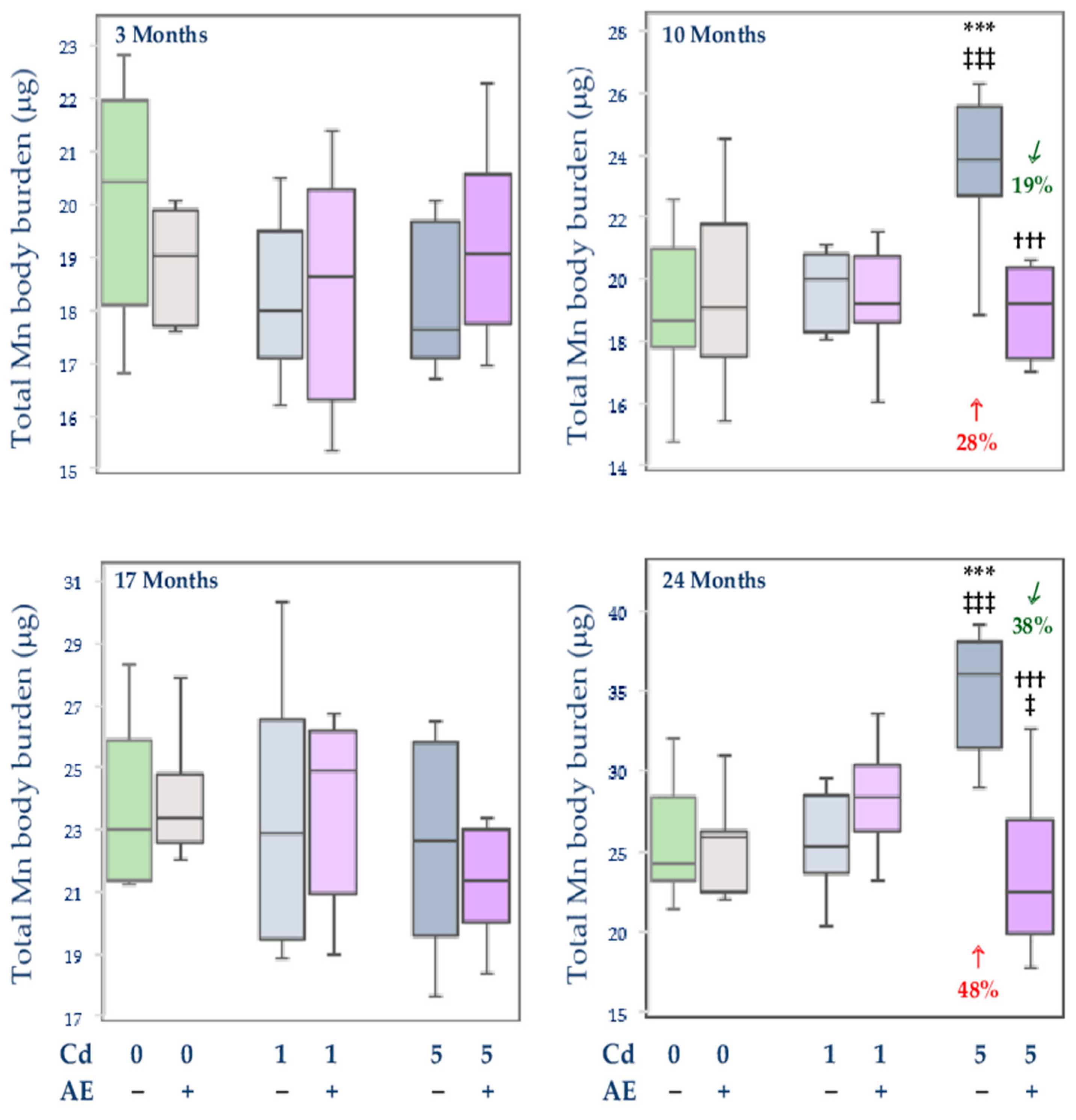
 , decrease;
, decrease;  , increase).
, increase).
 , decrease;
, decrease;  , increase).
, increase).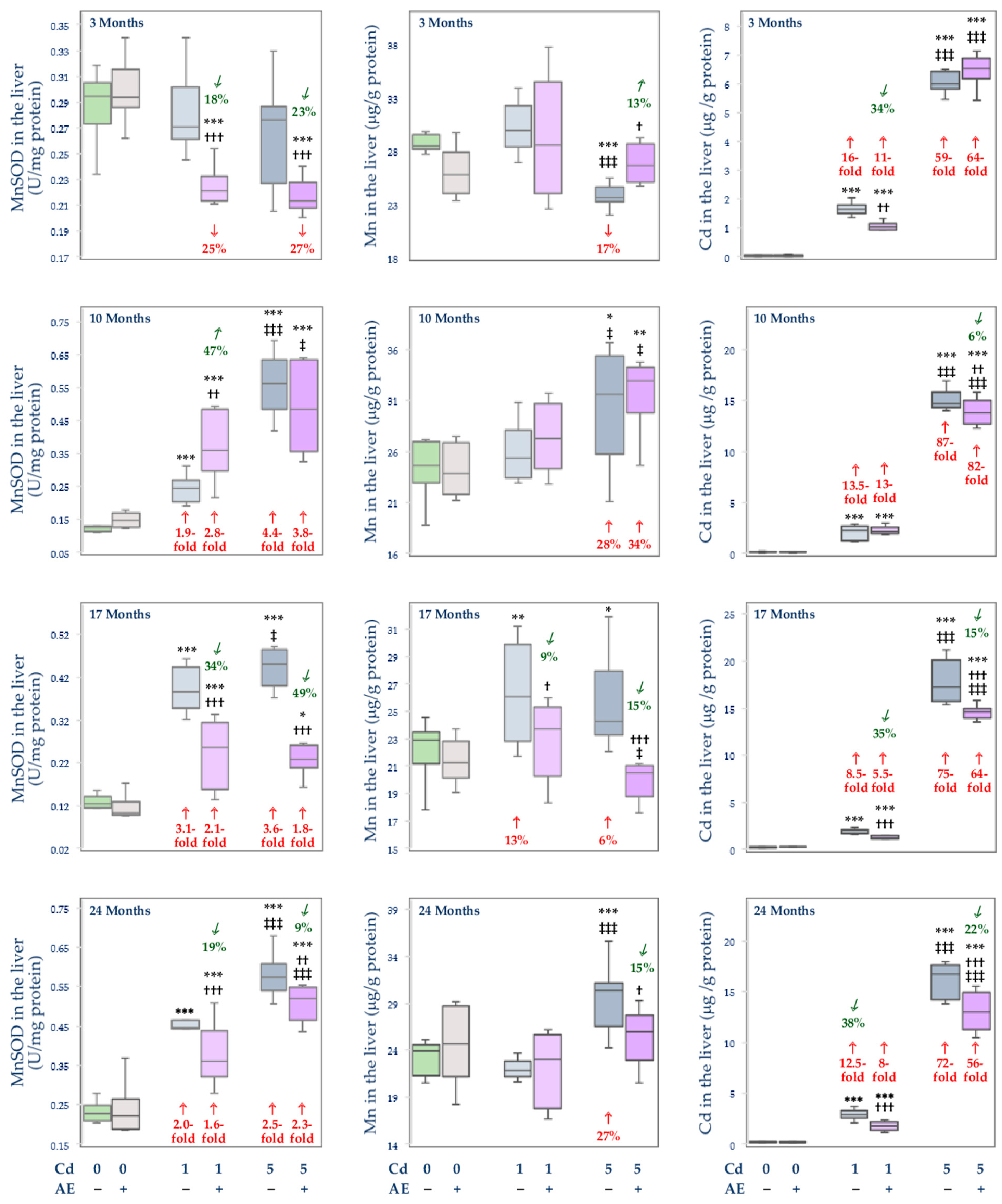
 , decrease;
, decrease;  , increase).
, increase).
 , decrease;
, decrease;  , increase).
, increase).
 , decrease;
, decrease;  , increase), respectively.
, increase), respectively.
 , decrease;
, decrease;  , increase), respectively.
, increase), respectively.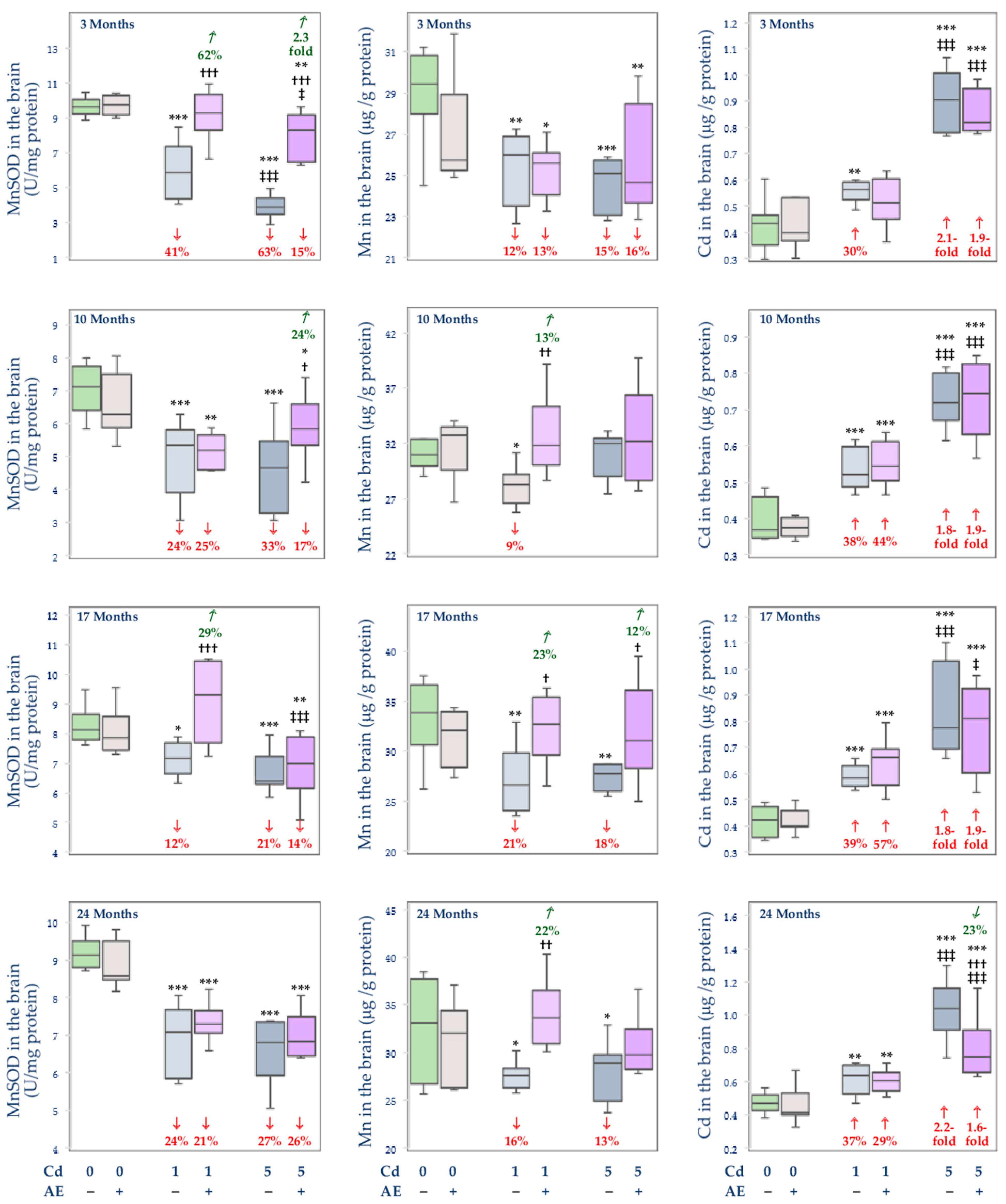
| Group | Daily Intake during the 24-Month Administration 1 | |
|---|---|---|
| AE [Polyphenols] (mg/kg b.w.) | Cd (μg/kg b.w.) | |
| Control | - | 2.30–4.98 |
| 0.1% AE (AE) | 67.4–146.6 [44.3–96.4] | 2.25–4.95 |
| 1 mg Cd/kg (Cd1) | - | 39.2–83.8 |
| 1 mg Cd/kg + 0.1% AE (Cd1 + AE) | 67.2–154.7 [44.2–101.7] | 37.5–84.9 |
| 5 mg Cd/kg (Cd5) | - | 210.1–403.2 |
| 5 mg Cd/kg + 0.1% AE (Cd5 + AE) | 63.1–150.3 [41.5–98.8] | 200.2–401.9 |
| Group | Mn Intake (mg/kg b.w./24 h) | |||
|---|---|---|---|---|
| 3 Months | 10 Months | 17 Months | 24 Months | |
| Control | 11.479 10.830–12.038 | 5.347 *** 5.111–6.278 | 5.182 *** 4.523–5.409 | 5.652 *** 5.397–6.276 |
| AE | 11.545 10.748–12.294 | 5.805 *** 5.278–5.917 | 5.002 *** 4.619–5.408 | 5.641 *** 5.336–6.106 |
| Cd1 | 11.565 10.804–12.320 | 5.591 *** 5.291–6.027 | 5.095 *** 4.762–5.429 | 5.744 *** 5.499–6.296 |
| Cd1 + AE | 11.538 10.983–11.900 | 5.587 *** 5.114–6.169 | 4.988 *** 4.762–5.655 | 5.895 *** 5.268–6.878 |
| Cd5 | 11.141 10.679–11.740 | 5.620 *** 5.336–5.959 | 5.020 *** 4.542–5.211 | 5.853 *** 5.559–6.944 |
| Cd5 + AE | 11.335 10.785–11.655 | 5.720 *** 5.620–5.964 | 4.956 *** 4.743–5.289 | 5.870 *** 5.329–6.931 |
| Group | Mn Content (μg) | ||||
|---|---|---|---|---|---|
| Liver | Kidneys | Spleen | Brain | Heart | |
| 3 Months | |||||
| Control | 17.203 14.007–19.909 | 1.4302 1.1793–1.5344 | 0.1108 0.1030–0.1272 | 1.0769 0.9949–1.2456 | 0.4666 0.3760–0.5078 |
| AE | 15.641 14.325–16.757 | 1.4062 1.3373–1.5276 | 0.1124 0.0894–0.1448 | 1.1863 * 1.0755–1.3064 ↑ 10% | 0.5008 0.3637–0.6853 |
| Cd1 | 14.922 13.440–17.265 | 1.5174 1.2470–1.6783 | 0.1367 * 0.1113–0.1539 ↑ 23% 4 | 0.9457 ** 0.8961–0.9945 ↓ 12% | 0.4612 0.3266–0.5053 |
| Cd1 + AE | 15.434 12.265–18.272 | 1.4570 1.3324–1.5486 | 0.1212 0.1005–0.1480 | 1.1308 ††† 1.0169–1.3611  20% 20% | 0.4016 0.3746–0.5880 |
| Cd5 | 14.861 13.726–17.315 | 1.3960 1.1715–1.8040 | 0.1336 * 0.1010–0.1640 ↑ 21% | 0.8530 *** 0.7591–0.9941 ↓ 21% | 0.4650 0.3731–0.5843 |
| Cd5 + AE | 15.757 13.524–18.984 | 1.3219 1.2104–1.6073 | 0.1355 * 0.1213–0.1560 ↑ 22% | 1.3779 *** ††† ‡‡‡ 1.2279–1.5017 ↑ 28%  61% 61% | 0.4770 0.3574–0.6427 |
| 10 Months | |||||
| Control | 15.968 12.280–19.566 | 0.9707 0.8403–1.2758 | 0.0885 0.0777–0.1260 | 1.0730 0.9609–1.2735 | 0.4818 0.4029–0.5079 |
| AE | 16.516 13.132–21.623 | 0.9972 0.8799–1.1366 | 0.0906 0.0728–0.1045 | 0.9985 0.6901–1.4094 | 0.4307 0.3573–0.6381 |
| Cd1 | 17.523 15.420–18.591 | 1.0433 0.8891–1.2831 | 0.0960 0.0656–0.1057 | 0.9387 * 0.6052–1.0903 ↓ 12% | 0.5099 0.3833–0.5852 |
| Cd1 + AE | 16.465 13.524–18.433 | 0.9824 0.9333–1.0921 | 0.0901 0.07373–0.1465 | 1.2195 †† 0.9981–1.4677  30% 30% | 0.4877 0.3532–0.6908 |
| Cd5 | 21.047 *** ‡‡‡ 16.154–23.227 ↑ 32% | 1.1004 1.0737–1.1784 | 0.1007 0.0779–0.1376 | 1.2202 ‡ 0.8001–1.4410 | 0.5897 * 0.4779–0.6411 ↑ 22% |
| Cd5 + AE | 16.355 ††† 13.607–17.993  22% 22% | 1.0725 0.9453–1.1966 | 0.0910 0.0725–0.1204 | 0.9244 † ‡‡ 0.7660–1.1211  24% 24% | 0.4705 0.4010–0.6262 |
 , decrease;
, decrease; 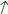 , increase).
, increase).| Group | Mn Content (μg) | ||||
|---|---|---|---|---|---|
| Liver | Kidneys | Spleen | Brain | Heart | |
| 17 Months | |||||
| Control | 19.986 17.953–24.879 | 1.1942 1.1215–1.4911 | 0.1492 0.1239–0.1849 | 1.1898 0.9564–1.6245 | 0.5696 0.4657–0.7360 |
| AE | 19.854 18.933–24.663 | 1.3243 1.1184–1.5192 | 0.1919 * 0.1411–0.2750 ↑ 29% 4 | 1.1590 1.0233–1.4371 | 0.6174 0.5222–0.7383 |
| Cd1 | 19.610 16.339–27.141 | 1.2306 0.6885–1.4514 | 0.1390 0.1094–0.1982 | 1.1355 0.8848–1.5478 | 0.4915 0.4441–0.5860 |
| Cd1 + AE | 21.565 16.131–23.630 | 1.3745 0.8566–1.5348 | 0.1780 0.1411–0.2330 | 1.1292 0.9455–1.1983 | 0.5181 0.4236–0.7106 |
| Cd5 | 19.194 15.017–24.167 | 1.3954 ‡ 1.2463–1.5796 | 0.1597 0.1413–0.1960 | 0.9732 ** ‡ 0.7127–1.1645 ↓ 18% | 0.5680 0.3973–0.7040 |
| Cd5 + AE | 18.230 15.648–29.830 | 1.4082 1.2262–1.5270 | 0.1473 ‡ 0.1160–0.1893 | 1.1166 0.9020–1.3074 | 0.5094 0.4084–0.6568 |
| 24 Months | |||||
| Control | 20.502 18.387–26.985 | 1.7338 1.1643–1.9272 | 0.1882 0.1527–0.2158 | 1.2136 1.1322–1.3682 | 0.8048 0.5802–0.8828 |
| AE | 21.714 18.427–26.752 | 1.5159 1.1668–2.2083 | 0.1823 0.1333–0.3042 | 1.4236 0.8137–1.4560 | 0.6920 0.5522–0.9004 |
| Cd1 | 21.173 16.252–25.473 | 1.6397 1.3793–2.2194 | 0.1771 0.1200–0.2821 | 1.4519 ** 1.2269–1.8314 ↑ 20% | 0.6980 0.6137–0.7732 |
| Cd1 + AE | 24.604 19.232–29.978 | 1.8263 1.3935–2.0252 | 0.2148 0.1565–0.2889 | 1.1785 †† 0.8500–1.6496  19% 19% | 0.6550 0.5887–0.7678 |
| Cd5 | 29.789 *** ‡‡‡ 24.711–34.724 ↑ 45% | 2.1949 ** ‡ 1.6741–2.3331 ↑ 27% | 0.2125 0.1854–0.2678 | 1.3887 1.1759–1.6099 | 0.6304 * 0.5377–0.6879 ↓ 22% |
| Cd5 + AE | 20.619 ††† 13.679–28.991  31% 31% | 1.7185 † 1.2205–2.0305  22% 22% | 0.1852 0.1513–2800 | 1.2137 1.0994–1.3173 |
0.5343 *** † ‡ 0.4607–0.6713 ↓ 34%  15% 15% |
 , decrease).
, decrease).Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brzóska, M.M.; Gałażyn-Sidorczuk, M.; Kozłowska, M.; Smereczański, N.M. The Body Status of Manganese and Activity of This Element-Dependent Mitochondrial Superoxide Dismutase in a Rat Model of Human Exposure to Cadmium and Co-Administration of Aronia melanocarpa L. Extract. Nutrients 2022, 14, 4773. https://doi.org/10.3390/nu14224773
Brzóska MM, Gałażyn-Sidorczuk M, Kozłowska M, Smereczański NM. The Body Status of Manganese and Activity of This Element-Dependent Mitochondrial Superoxide Dismutase in a Rat Model of Human Exposure to Cadmium and Co-Administration of Aronia melanocarpa L. Extract. Nutrients. 2022; 14(22):4773. https://doi.org/10.3390/nu14224773
Chicago/Turabian StyleBrzóska, Małgorzata M., Małgorzata Gałażyn-Sidorczuk, Magdalena Kozłowska, and Nazar M. Smereczański. 2022. "The Body Status of Manganese and Activity of This Element-Dependent Mitochondrial Superoxide Dismutase in a Rat Model of Human Exposure to Cadmium and Co-Administration of Aronia melanocarpa L. Extract" Nutrients 14, no. 22: 4773. https://doi.org/10.3390/nu14224773
APA StyleBrzóska, M. M., Gałażyn-Sidorczuk, M., Kozłowska, M., & Smereczański, N. M. (2022). The Body Status of Manganese and Activity of This Element-Dependent Mitochondrial Superoxide Dismutase in a Rat Model of Human Exposure to Cadmium and Co-Administration of Aronia melanocarpa L. Extract. Nutrients, 14(22), 4773. https://doi.org/10.3390/nu14224773






