A Graded Approach for Evaluating Health Claims about Plant-Based Food Supplements: Application of a Case Study Methodology
Abstract
:1. Introduction
1.1. Context of Plant-Based Food Supplements
1.2. The Graded Health Claim System: Principles
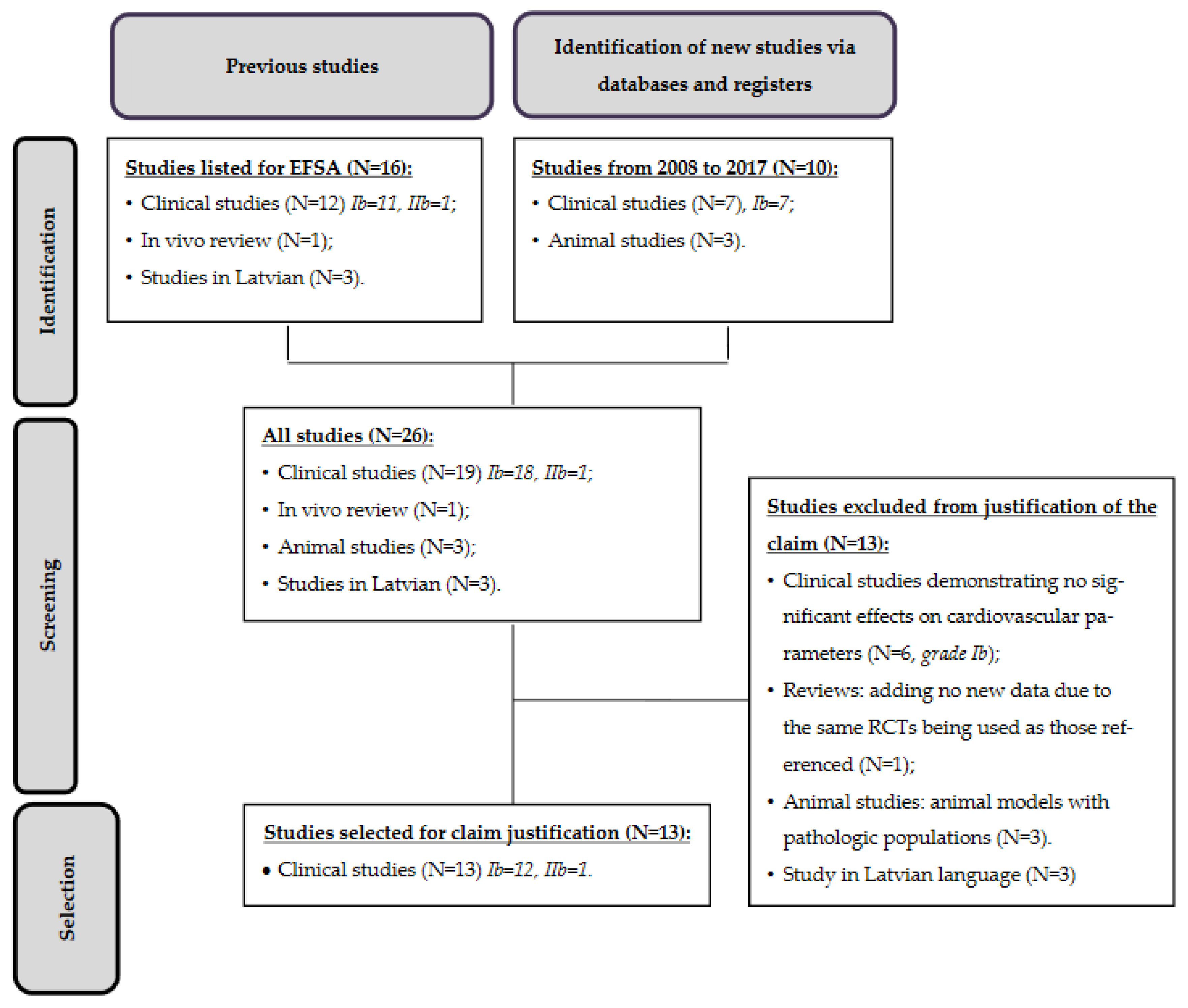
2. Materials and Methods
3. Results
Study Characteristics
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- European Union Herbal Monograph on Aesculus hippocastanum L., Semen:9. Committee on Herbal Medicinal Products (HMPC). 15 May 2019. EMA/HMPC/638242/2018. Available online: https://www.ema.europa.eu/en/documents/herbal-monograph/european-union-herbal-monograph-aesculus-hippocastanum-l-semen-final-revision-1_en.pdf (accessed on 31 March 2021).
- European Union Herbal Monograph on Ginkgo biloba L., Folium:8. Committee on Herbal Medicinal Products (HMPC). 28 January 2015. EMA/HMPC/321097/2012. Available online: https://www.ema.europa.eu/en/documents/herbal-monograph/final-european-union-herbal-monograph-ginkgo-biloba-l-folium_en.pdf (accessed on 31 March 2021).
- European Union Herbal Monograph on Prunus africana (Hook f.) Kalkm., Cortex:6. Committee on Herbal Medicinal Products (HMPC). 24 November 2015. EMA/HMPC/680626/2013. Available online: https://www.ema.europa.eu/en/documents/herbal-monograph/draft-european-union-herbal-monograph-prunus-africana-hook-f-kalkm-cortex_en.pdf (accessed on 31 March 2021).
- Commission Regulation (EU) No 432/2012 of 16 May 2012. Establishing A List of Permitted Health Claims Made on Foods, Other than Those Referring to the Reduction of Disease Risk and to Children’s Development and Health Text with EEA Relevance. Off. J. Eur. Union 2012, L136, 1–40. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32012R0432 (accessed on 9 April 2021).
- Anton, R.; Serafini, M.; Delmulle, L. The Substantiation of Claims for Botanical Food Supplements in Relation to Traditional Use. Eur. Food Feed Law Rev. 2013, 8, 321–328. [Google Scholar]
- Anton, R.; Mathioudakis, B.; Pramono, S.; Sezik, E.; Sharma, S. Traditional Use of Botanicals and Botanical Preparations. Eur. Food Feed Law Rev. 2019, 14, 132–141. [Google Scholar]
- U.S. Food and Drug Administration (FDA). Center for Food Safety and Applied Nutrition. Guidance for Industry: Evidence-Based Review System for the Scientific Evaluation of Health Claims; 2020. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-evidence-based-review-system-scientific-evaluation-health-claims (accessed on 31 March 2021).
- Williams, B.; Mancia, G.; Spiering, W.; Rosei, E.A.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; De Simone, G.; Dominiczak, A.; et al. ESC/ESH Guidelines for the management of arterial hypertension: The Task Force for the management of arterial hypertension of the European Society of Cardiology (ESC) and the European Society of Hypertension (ESH). Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef]
- Allaert, F.A.; Vandermander, J.; Herpin, F.; Ventura, G. Grading health claims by weight of evidence would make for a better-informed public. Minerva Gastroenterol. Dietol. 2017, 63, 293–303. [Google Scholar] [CrossRef] [PubMed]
- EFSA Guidance for Those Carrying out Systematic Reviews European Food Safety Authority. Application of Systematic Review Methodology to Food and Feed Safety Assessments to Support Decision Making. Eur. Food Saf. Auth. J. 2010, 8, 1637. Available online: https://www.efsa.europa.eu/en/efsajournal/pub/1637 (accessed on 9 April 2021).
- EFSA. Consolidated List of Article 13 Health Claims. List of References Received by EFSA. Part 3, IDs 2001—3000; 2011. Available online: https://www.efsa.europa.eu/sites/default/files/topic/ndaart13ref03.pdf (accessed on 9 April 2021).
- Atkinson, C.; Oosthuizen, W.; Scollen, S.; Loktionov, A.; Day, N.E.; Bingham, S.A. Modest protective effects of isoflavones from a red clover-derived dietary supplement on cardiovascular disease risk factors in perimenopausal women, and evidence of an interaction with ApoE genotype in 49–65 year-old women. J. Nutr. 2004, 134, 1759–1764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blakesmith, S.J.; Lyons-Wall, P.M.; George, C.; Joannou, G.E.; Petocz, P.; Samman, S. Effects of supplementation with purified red clover (Trifolium pratense) isoflavones on plasma lipids and insulin resistance in healthy premenopausal women. Br. J. Nutr. 2003, 89, 467–474. [Google Scholar] [CrossRef]
- Howes, J.B.; Tran, D.; Brillante, D.; Howes, L.G. Effects of dietary supplementation with isoflavones from red clover on ambulatory blood pressure and endothelial function in postmenopausal type 2 diabetes. Diabetes Obes. Metab. 2003, 5, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Campbell, M.J.; Woodside, J.V.; Honour, J.W.; Morton, M.S.; Leathem, A.J.C. Effect of red clover-derived isoflavone supplementation on insulin-like growth factor, lipid and antioxidant status in healthy female volunteers: A pilot study. Eur. J. Clin. Nutr. 2004, 58, 173–179. [Google Scholar] [CrossRef]
- Clifton-Bligh, P.B.; Baber, R.J.; Fulcher, G.R.; Nery, M.L.; Moreton, T. The effect of isoflavones extracted from red clover (Rimostil) on lipid and bone metabolism. Menopause 2001, 8, 259–265. [Google Scholar] [CrossRef]
- Geller, S.E.; Studee, L. Soy and red clover for mid-life and aging. Climacteric 2006, 9, 245–263. [Google Scholar] [CrossRef] [Green Version]
- Hidalgo, L.A.; Chedraui, P.A.; Morocho, N.; Ross, S.; Miguel, G.S. The effect of red clover isoflavones on menopausal symptoms, lipids and vaginal cytology in menopausal women: A randomized, double-blind, placebo-controlled study. Gynecol. Endocrinol. 2005, 21, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Howes, J.B.; Sullivan, D.; Lai, N.; Nestel, P.; Pomeroy, S.; West, L.; Eden, J.A.; Howes, L.G. The effects of dietary supplementation with isoflavones from red clover on the lipoprotein profiles of post menopausal women with mild to moderate hypercholesterolaemia. Atherosclerosis 2000, 152, 143–147. [Google Scholar] [CrossRef]
- Schult, T.M.K.; Ensrud, K.; Blackwell, T.; Ettinger, B.; Wallace, R.; Tice, J. Effect of isoflavones on lipids and bone turnover markers in menopausal women. Maturitas 2004, 48, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Nestel, P.J.; Pomeroy, S.; Kay, S.; Komesaroff, P.; Behrsing, J.; Cameron, J.D.; West, L. Isoflavones from red clover improve systemic arterial compliance but not plasma lipids in menopausal women. J. Clin. Endocrinol. Metab. 1999, 84, 895–898. [Google Scholar] [CrossRef]
- Nestel, P.; Cehun, M.; Chronopoulos, A.; DaSilva, L.; Teede, H.; McGrath, B. A biochanin-enriched isoflavone from red clover lowers LDL cholesterol in men. Eur. J. Clin. Nutr. 2004, 58, 403–408. [Google Scholar] [CrossRef]
- Squadrito, F.; Altavilla, D.; Morabito, N.; Crisafulli, A.; D’Anna, R.; Corrado, F.; Ruggeri, P.; Campo, G.M.; Calapai, G.; Caputi, A.P.; et al. The effect of the phytoestrogen genistein on plasma nitric oxide concentrations, endothelin-1 levels and endothelium dependent vasodilation in postmenopausal women. Atherosclerosis 2002, 163, 339–347. [Google Scholar] [CrossRef]
- Teede, H.J.; McGrath, B.P.; DeSilva, L.; Cehun, M.; Fassoulakis, A.; Nestel, P.J. Isoflavones reduce arterial stiffness: A placebo-controlled study in men and postmenopausal women. Arter. Thromb. Vasc. Biol. 2003, 23, 1066–1071. [Google Scholar] [CrossRef] [Green Version]
- Chen, T.; Zhong, F.-J.; Hong, Y.-M.; Su, W.-J.; Zhuang, L.-L.; Qiu, L.-X. Effect of Trifolium pratense extract on methionine-choline-deficient diet-induced steatohepatitis in C57BL/6 mice. Chin. J. Nat. Med. 2014, 12, 194–198. [Google Scholar] [CrossRef]
- Qiu, L.; Chen, T.; Zhong, F.; Hong, Y.; Chen, L.; Ye, H. Red clover extract exerts antidiabetic and hypolipidemic effects in db/db mice. Exp. Ther. Med. 2012, 4, 699–704. [Google Scholar] [CrossRef]
- Qiu, L.; Ye, H.; Chen, L.; Hong, Y.; Zhong, F.; Zhang, T. Red clover extract ameliorates dyslipidemia in streptozotocin-induced diabetic C57BL/6 mice by activating hepatic PPARα. Phytother. Res. 2012, 26, 860–864. [Google Scholar] [CrossRef]
- Chedraui, P.; San Miguel, G.; Hidalgo, L.; Morocho, N.; Ross, S. Effect of Trifolium pratense-derived isoflavones on the lipid profile of postmenopausal women with increased body mass index. Gynecol. Endocrinol. 2008, 24, 620–624. [Google Scholar] [CrossRef]
- Clifton-Bligh, P.B.; Nery, M.-L.; Clifton-Bligh, R.; Visvalingam, S.; Fulcher, G.; Byth, K.; Baber, R. Red clover isoflavones enriched with formononetin lower serum LDL cholesterol-a randomized, double-blind, placebo-controlled study. Eur. J. Clin. Nutr. 2015, 69, 134–142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lambert, M.N.T.; Thorup, A.C.; Hansen, E.S.S.; Jeppesen, P.B. Combined Red Clover isoflavones and probiotics potently reduce menopausal vasomotor symptoms. PLoS ONE 2017, 12, e0176590. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mainini, G.; Torella, M.; Di Donna, M.C.; Esposito, E.; Ercolano, S.; Correa, R.; Cucinella, G.; Stradella, L.; Luisi, A.; Basso, A.; et al. Nonhormonal management of postmenopausal women: Effects of a red clover based isoflavones supplementation on climacteric syndrome and cardiovascular risk serum profile. Clin. Exp. Obstet. Gynecol. 2013, 40, 337–341. [Google Scholar]
- Terzic, M.M.; Dotlic, J.; Maricic, S.; Mihailovic, T.; Tosic-Race, B. Influence of red clover-derived isoflavones on serum lipid profile in postmenopausal women. J. Obstet. Gynaecol. Res. 2009, 35, 1091–1095. [Google Scholar] [CrossRef] [PubMed]
- Terzic, M.; Micic, J.; Dotlic, J.; Maricic, S.; Mihailovic, T.; Knezevic, N. Impact of Phytoestrogens on Serum Lipids in Postmenopausal Women. Geburtshilfe Frauenheilkd. 2012, 72, 527–531. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thorup, A.C.; Lambert, M.N.; Kahr, H.S.; Bjerre, M.; Jeppesen, P.B. Intake of Novel Red Clover Supplementation for 12 Weeks Improves Bone Status in Healthy Menopausal Women. Evid. Based Complement Altern. Med. 2015, 2015, 689138. [Google Scholar] [CrossRef] [Green Version]
- Turck, D.; Bresson, J.-L.; Burlingame, B.; Dean, T.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.I.; Mangelsdorf, I.; McArdle, H.J.; Naska, A.; et al. Guidance for the scientific requirements for health claims related to antioxidants, oxidative damage and cardiovascular health. EFSA J. Eur. Food Saf. Auth. 2018, 16, e05136. [Google Scholar]

| Grade | C | B | A |
|---|---|---|---|
| Traditionally Used for… | Can Contribute to… | Decreases... Relieves… or Improves… | |
| General health claim “General function” claims refer to the role of a nutrient or substance in growth, development, body functions, biological activities, body structure, psychological and behavioral functions, weight control to maintain good health, and comfort. Reduction in disease risk factors Any health claim that states, suggests or implies that the consumption of a food category, a food, or one of its constituents significantly reduces a risk factor in the development of a human disease, including for example:
| Authoritative reference texts:
| Convergent body of evidence with at least:
| Two convergent studies with at least:
|
| Grade | Type of Evidence |
|---|---|
| Ia | Evidence from a meta-analysis of randomized controlled trials |
| Ib | Evidence from at least one randomized controlled trial |
| IIa | Evidence from at least one controlled study without randomization |
| IIb | Evidence from at least one other type of quasi-experimental study |
| III | Evidence from observational studies |
| IV | Evidence from expert committee reports or experts |
| Grade | Recommendation |
|---|---|
| A (Evidence levels quality Ia, Ib) | Requires at least one randomized controlled trial as part of the body of literature of overall value and consistency addressing the specific recommendation. |
| B (Evidence levels IIa, IIb, III) | Requires the availability of well-conducted clinical studies but no randomized clinical trials on the topic of recommendation. |
| C (Evidence Level IV) | Requires evidence from expert committee reports or opinions and/or clinical experience of respected authorities. Indicates an absence of directly applicable studies of good quality. |
| Author_Date | Type of Evidence Classification | Inclusion or Exclusion in the Analysis in Favor of the Pending Claim |
|---|---|---|
| Studies from EFSA’s list | ||
| Atkinson_2004 [12] | Ib | Excluded, no significant results on cardiovascular parameters |
| Blakesmith_2003 [13] | Ib | Excluded, no significant results on cardiovascular parameters |
| Campbell_2004 [15] | Ib | Included |
| Clifton-Bligh_2001 [16] | IIb | Included |
| Geller_2006 [17] | Review | Excluded, utilization of articles already present in the analysis |
| Hidalgo_2005 [18] | Ib | Included |
| Howes_2000 [19] | Ib | Excluded, no significant results on cardiovascular parameters |
| Howes_2003 [14] | Ib | Included |
| Knudson Shult_2004 [20] | Ib | Included |
| Nestel_1999 [21] | Ib | Included |
| Nestel_2004 [22] | Ib | Included |
| Rubine_2001 | / | Excluded, Latvian language only, classification not possible. Not referenced on classical database. |
| Rubine_2004 | / | Excluded, Latvian language only, classification not possible. Not referenced on classical database. |
| Skutelis_2005 | / | Excluded, Latvian language only, classification not possible. Not referenced on classical database. |
| Squadrito_2002 [23] | Ib | Included |
| Teede_2003 [24] | Ib | Included |
| Studies from the literature search for the period 2008–2017 | ||
| Chen_2014 [25] | Animal study | Excluded, animal models not relevant (disease) |
| Qiu_2012a [26] | Animal study | Excluded, animal models not relevant (disease) |
| Qiu_2012b [27] | Animal study | Excluded, animal models not relevant (disease) |
| Chedraui_2008 [28] | Ib | Included |
| Clifton-Bligh_2015 [29] | Ib | Included |
| Lambert_2007 [30] | Ib | Excluded, no significant results on cardiovascular parameters |
| Mainini_2013 [31] | Ib | Excluded, no significant results on cardiovascular parameters |
| Terzic_2009 [32] | Ib | Included |
| Terzic_2012 [33] | Ib | Included |
| Thorup_2015 [34] | Ib | Excluded, no significant results on cardiovascular parameters |
| Authors | Number of Subjects | Population | Primary Endpoint | Evaluation Criteria of the Cardiovascular System (Observed Significant Effect) | Statistical Comparisons for the Significant Results | Doses (Expressed in Isoflavones) | Duration |
|---|---|---|---|---|---|---|---|
| Campbell, 2004 [15] | N = 23 | Pre-menopausal (n = 16) and postmenopausal women (n = 7) | IGF-1 | HDL-c: significant increase for postmenopausal women vs. placebo groups only (p = 0.02) | Between-group comparisons | 86 mg/day or placebo | 1 month |
| Clifton-Bligh, 2001 [16] | N = 46 | Menopausal women | Unspecified in the article | HDL-c: significant increase for all doses (p = 0.007, p = 0.002, and p = 0.027) ApoB: significant decrease for all doses (p = 0.005, p = 0.043, and p = 0.007) | Within-group comparisons | 28.5 mg/day, 57 mg/day or 85.5 mg/day | 6 months |
| Hidalgo, 2005 [18] | N = 60 | Menopausal women (>40 years old) | Unspecified in the article | Triglycerides: Significant decrease (pvalue unspecified) | Between-group comparisons | 80 mg/day or placebo | 187 days |
| Howes, 2003 [14] | N = 16 | Menopausal women with type 2 diabetes | Unspecified in the article | Systolic and diastolic blood pressure: significant decrease (p < 0.05) Vascular resistance (forearm): significant increase (p < 0.05) | Between-group comparisons | 50 mg/day or placebo | 4 weeks |
| Knudson Shult, 2004 [20] | N = 252 | Menopausal women (45–60 years old) | HDL-c, osteocalcin, and urinary N-telopeptide | Triglycerides: significant decrease for all doses (p = 0.02) for 57.2 mg/day and (p = 0.05) for 82 mg/day | Between-group comparisons | 57.2 mg/day, 82 mg/day or placebo | 12 weeks |
| Nestel, 1999 [21] | N = 17 | Menopausal women (<70 years old) | Unspecified in the article | Systemic Arterial Compliance (SAC): significant increase (p < 0.05) | Between-group comparisons | 40 mg/day then 80 mg/day or placebo | 2 periods of 5 weeks |
| Nestel, 2004 [22] | N = 80 | 46 men and 34 menopausal women (aged 45–75 years old) | LDL-c | LDL-c: significant decrease observed in men only (p < 0.05) | Between-group comparisons | 40 mg/day or placebo | 6 weeks |
| Squadrito, 2002 [23] | N = 60 | Menopausal women (52–60 years old) | Unspecified in the article | Endothelium-dependent and flow-mediated vasodilation (FMD) of the brachial artery, ratio of oxide nitric/endothelin-1: significant improvement (p < 0.05 and (p < 0.01) | Between-group comparisons | 54 mg/day or placebo | 6 months |
| Teede, 2003 [24] | N = 80 | 46 men and 34 menopausal women (aged 45–75 years old) | Unspecified in the article | Systemic Arterial Compliance: significant increase (p = 0.04) Central Pulse Wave Velocity (PWV): significant reduction (p = 0.02) Effects observed for biochanin form only. | Between-group comparisons | 80 mg/day (enriched in biochanin or formononetin) or placebo | 15 weeks |
| Clifton-Bligh, 2015 [29] | N = 97 | Menopausal women (average age 54 years old) | Bone Mineral Density (BMD) and LDL-c | LDL-c: significant decrease (p = 0.005) | Within-group comparisons | 50 mg/day or placebo | >1 year |
| Terzic, 2012 [33] | N = 74 | Menopausal women | Unspecified in the article | Total cholesterol, LDL-c, triglycerides: significant reduction (p < 0.001) HDL-c: significant increase (p < 0.001) | Between-group comparisons | 40 mg/day or control (no supplementation) | 18 months |
| Terzic, 2009 [32] | N = 40 | Menopausal women (mean age 56 years old) | Unspecified in the article | Total cholesterol, LDL-c: significant reduction (p < 0.005) HDL-c: significant increase (p < 0.005) | Between-group comparisons | 40 mg/day or control (no supplementation) | 12 months |
| Chedraui 2008 [28] | N = 60 | Menopausal women (>40 years old) | Unspecified in the article | Total cholesterol, LDL-c, and Lipoprotein (a): significant decrease in women with BMI ≥ 25 kg/m2. | Unspecified in the article | 80 mg/day or placebo | 90 days |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chevallier, H.; Herpin, F.; Kergosien, H.; Ventura, G.; Allaert, F.-A. A Graded Approach for Evaluating Health Claims about Plant-Based Food Supplements: Application of a Case Study Methodology. Nutrients 2021, 13, 2684. https://doi.org/10.3390/nu13082684
Chevallier H, Herpin F, Kergosien H, Ventura G, Allaert F-A. A Graded Approach for Evaluating Health Claims about Plant-Based Food Supplements: Application of a Case Study Methodology. Nutrients. 2021; 13(8):2684. https://doi.org/10.3390/nu13082684
Chicago/Turabian StyleChevallier, Hélène, Florent Herpin, Hélène Kergosien, Gabrielle Ventura, and François-André Allaert. 2021. "A Graded Approach for Evaluating Health Claims about Plant-Based Food Supplements: Application of a Case Study Methodology" Nutrients 13, no. 8: 2684. https://doi.org/10.3390/nu13082684
APA StyleChevallier, H., Herpin, F., Kergosien, H., Ventura, G., & Allaert, F.-A. (2021). A Graded Approach for Evaluating Health Claims about Plant-Based Food Supplements: Application of a Case Study Methodology. Nutrients, 13(8), 2684. https://doi.org/10.3390/nu13082684






