Evolution of the Dietary Patterns across Nutrition Transition in the Sardinian Longevity Blue Zone and Association with Health Indicators in the Oldest Old
Abstract
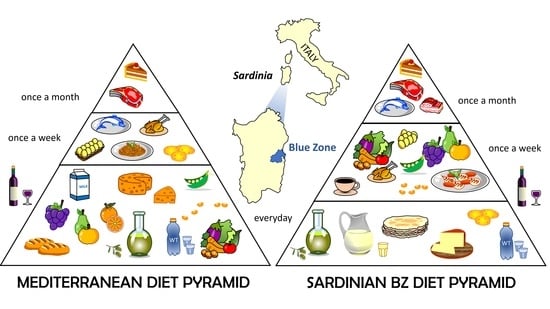
1. Introduction
2. Materials and Methods
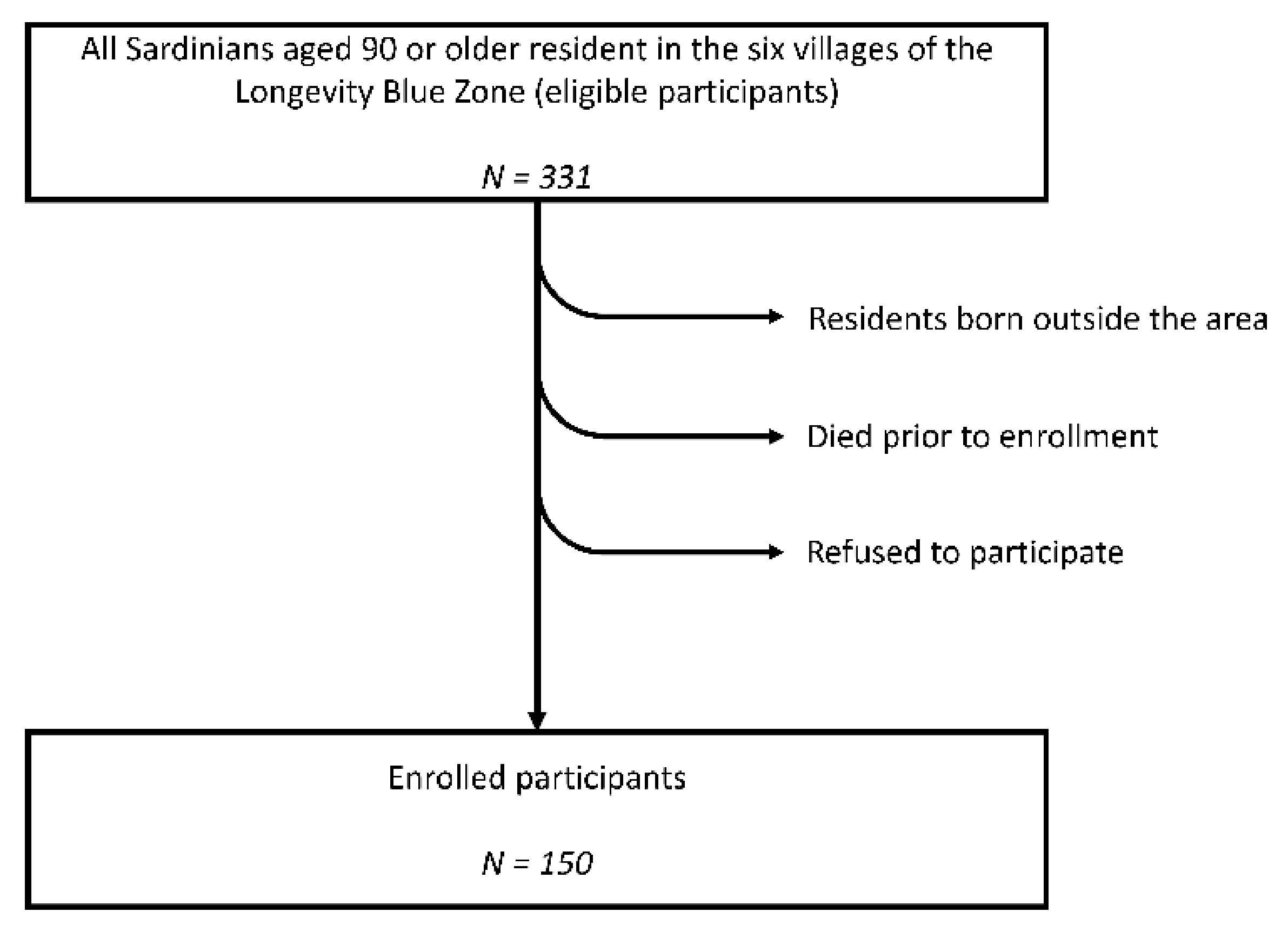
2.1. Study Design and Participants
2.2. Data Collection
2.2.1. Anthropometric Measures
2.2.2. Multidimensional Geriatric Examination
2.2.3. Performance-Based Functional Capacity
2.2.4. Cognitive and Affective Indicators
2.2.5. Physical Performance Tests
2.2.6. Assessment of Dietary Habits
2.3. Ethical Considerations
2.4. Statistical Analysis
3. Results
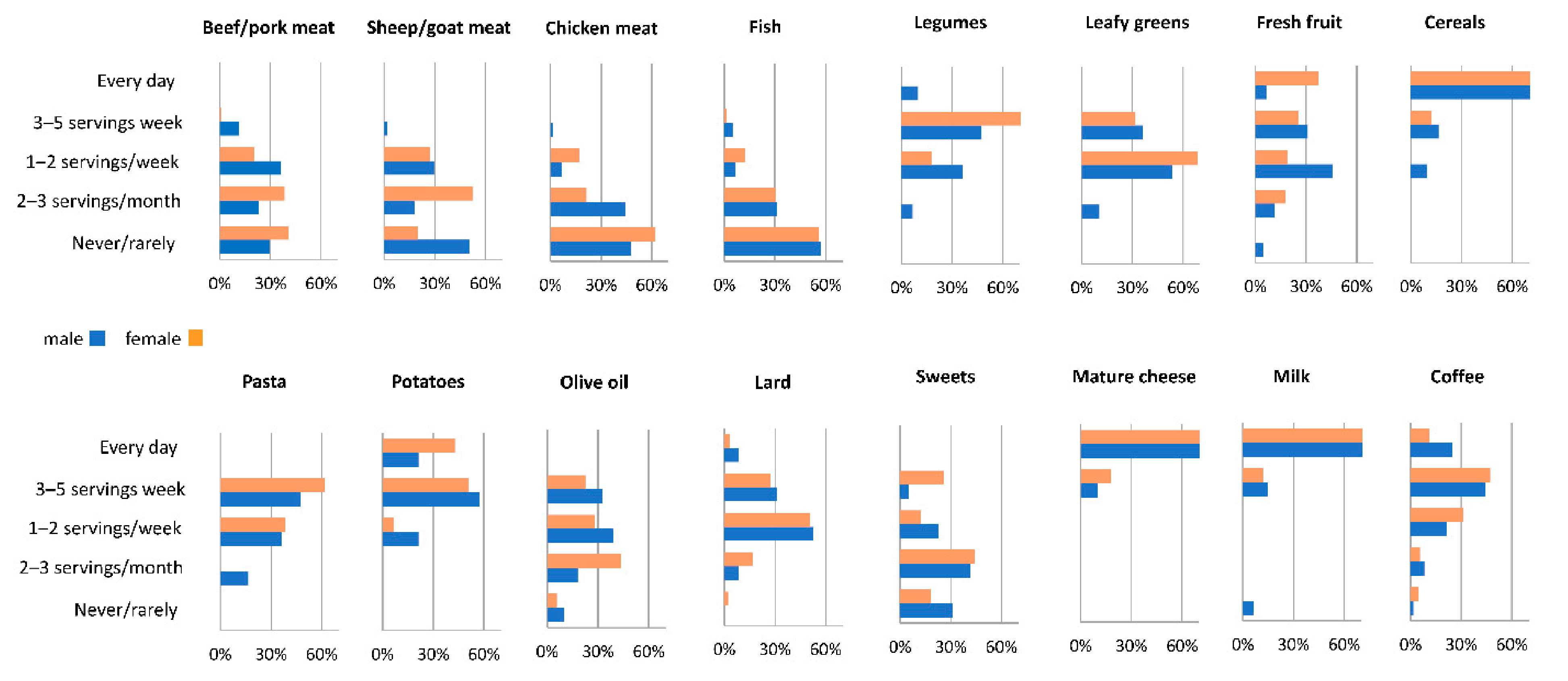
3.1. Assessment of the Current Diet
3.2. Dietary Intake at Age 30 and Changes across Nutrition Transition
3.3. Health Indicators
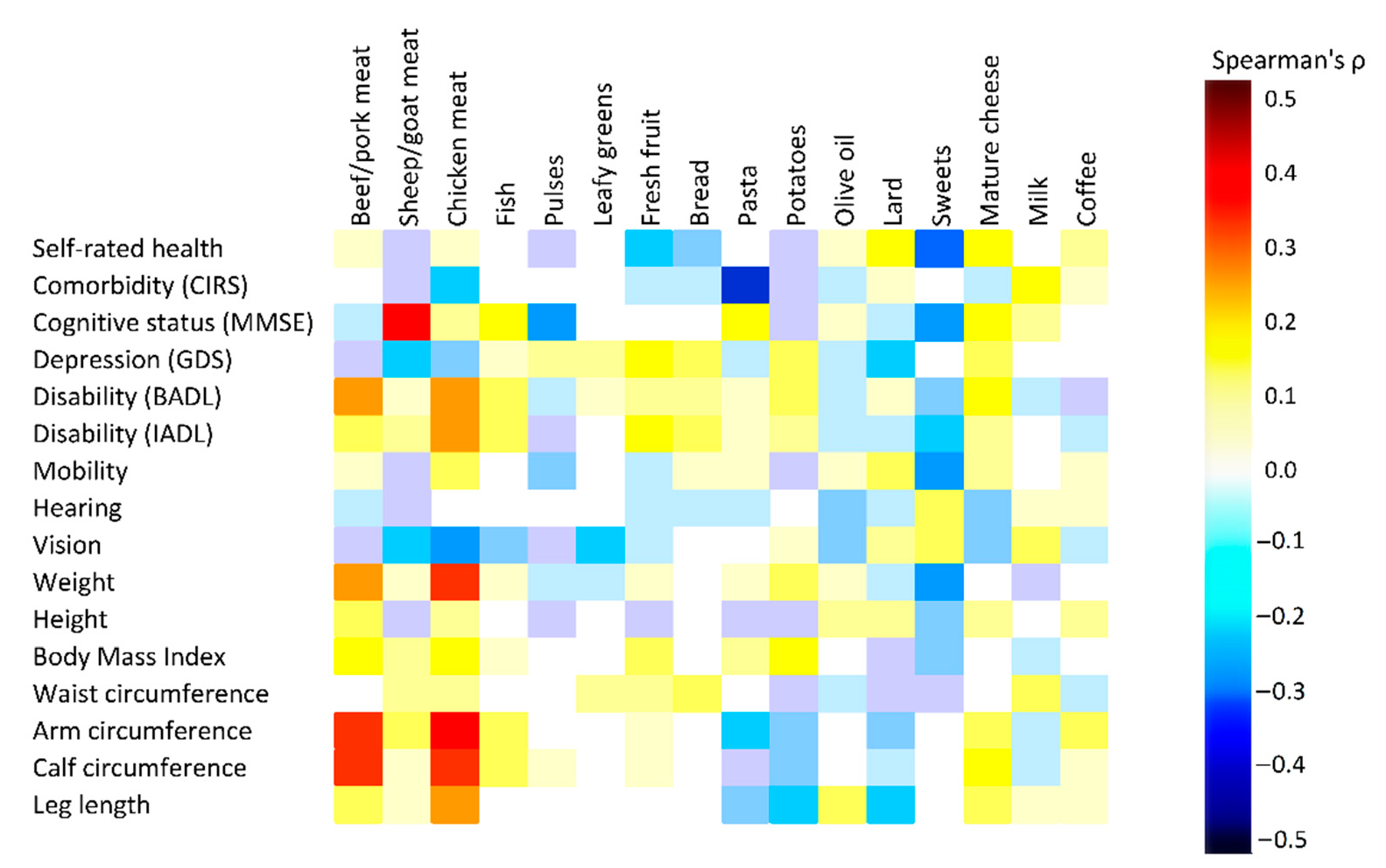
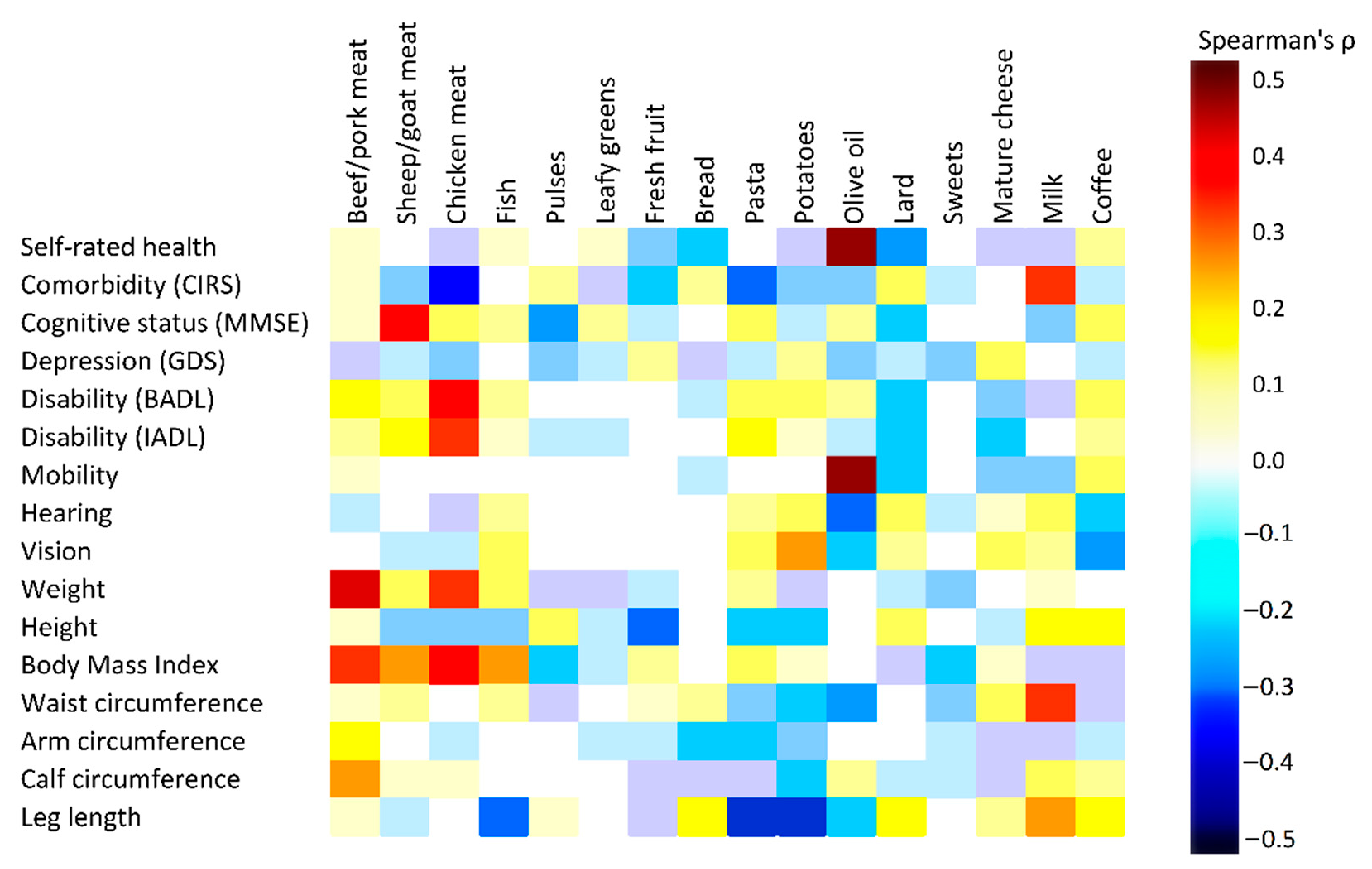
3.4. Association between Current Food Frequencies and Health Indicators
3.5. Association of Dietary Changes Across NT with Health Indicators
4. Discussion
5. Strengths and Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Prince, M.J.; Wu, F.; Guo, Y.; Gutierrez Robledo, L.M.; O’Donnell, M.; Sullivan, R.; Yusuf, S. The burden of disease in older people and implications for health policy and practice. Lancet 2015, 385, 549–562. [Google Scholar] [CrossRef]
- World Population Ageing 2019 Highlights; United Nations: New York, NY, USA, 2019.
- Dowd, J.B.; Andriano, L.; Brazel, D.M.; Rotondi, V.; Block, P.; Ding, X.; Liu, Y.; Mills, M.C. Demographic science aids in understanding the spread and fatality rates of COVID-19. Proc. Natl. Acad. Sci. USA 2020, 117, 9696–9698. [Google Scholar] [CrossRef] [PubMed]
- Ettinger, W.H., Jr.; Fried, L.P.; Harris, T.; Shemanski, L.; Schulz, R.; Robbins, J. Self-reported causes of physical disability in older people: The Cardiovascular Health Study. CHS Collaborative Research Group. J. Am. Geriatr. Soc. 1994, 42, 1035–1044. [Google Scholar] [CrossRef] [PubMed]
- Fontana, L.; Partridge, L. Promoting health and longevity through diet: From model organisms to humans. Cell 2015, 161, 106–118. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Kouris-Blazos, A.; Wahlqvist, M.L.; Gnardellis, C.; Lagiou, P.; Polychronopoulos, E.; Vassilakou, T.; Lipworth, L.; Trichopoulos, D. Diet and overall survival in elderly people. BMJ 1995, 311, 1457–1460. [Google Scholar] [CrossRef]
- León-Muñoz, L.M.; García-Esquinas, E.; López-García, E.; Banegas, J.R.; Rodríguez-Artalejo, F. Major dietary patterns and risk of frailty in older adults: A prospective cohort study. BMC Med. 2015, 13, 11. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Vasilopoulou, E. Mediterranean diet and longevity. Br. J. Nutr. 2000, 84 (Suppl. 2), S205–S209. [Google Scholar] [CrossRef]
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean diet pyramid: A cultural model for healthy eating. Am. J. Clin. Nutr. 1995, 61 (Suppl. 6), S1402–S1406. [Google Scholar] [CrossRef]
- Trajkovska Petkoska, A.; Trajkovska-Broach, A. Mediterranean diet: A nutrient-packed diet and a healthy lifestyle for a sustainable world. J. Sci. Food. Agric. 2020. [Google Scholar] [CrossRef]
- Mantzioris, E.; Villani, A. Translation of a Mediterranean-style diet into the Australian dietary guidelines: A nutritional, ecological and environmental perspective. Nutrients 2019, 11, 2507. [Google Scholar] [CrossRef]
- Appel, L.J. Dietary patterns and longevity: Expanding the blue zones. Circulation 2008, 118, 214–215. [Google Scholar] [CrossRef]
- Poulain, M.; Pes, G.M.; Grasland, C.; Carru, C.; Ferrucci, L.; Baggio, G.; Franceschi, C.; Deiana, L. Identification of a geographic area characterized by extreme longevity in the Sardinia island: The AKEA study. Exp. Gerontol. 2004, 39, 1423–1429. [Google Scholar] [CrossRef]
- Poulain, M.; Pes, G.; Salaris, L. A population where men live as long as women: Villagrande Strisaili Sardinia. J. Aging Res. 2011, 2011, 153756. [Google Scholar] [CrossRef]
- Poulain, M.; Herm, A.; Pes, G.M. The blue zones: Areas of exceptional longevity around the world. Vienna Yearb. Popul. Res. 2013, 11, 83–102. [Google Scholar] [CrossRef]
- Cavalli-Sforza, L.L.; Menotti, P.; Piazza, A. The History and Geography of Human Genes; Princeton University: Princeton, NJ, USA, 1994. [Google Scholar]
- Caselli, G.; Pozzi, L.; Vaupel, J.W.; Deiana, L.; Pes, G.; Carru, C.; Franceschi, C.; Baggio, G. Family clustering in Sardinian longevity: A genealogical approach. Exp. Gerontol. 2006, 41, 727–736. [Google Scholar] [CrossRef]
- Vona, G. The peopling of Sardinia (Italy): History and effects. Int. J. Anthropol. 1997, 12, 71–87. [Google Scholar] [CrossRef]
- Poulain, M. Individual longevity versus population longevity. In Centenarians: An Example of Positive Biology; Caruso, C., Ed.; Springer Nature: Cham, Switzerland, 2019. [Google Scholar]
- Popkin, B.M. Understanding the nutrition transition. Urban Health Newsl. 1996, 30, 3–19. [Google Scholar]
- Bodirsky, B.L.; Dietrich, P.; Martinelli, E.; Stenstad, A.; Pradhan, P.; Gabrysch, S.; Mishra, A.; Weindl, I.; Le Mouël, C.; Rolinski, S.; et al. The ongoing nutrition transition thwarts long-term targets for food security, public health and environmental protection. Sci. Rep. 2020, 10, 19778. [Google Scholar] [CrossRef]
- Popkin, B.M. Technology, transport, globalization and the nutrition transition food policy. Food Policy 2006, 31, 554–569. [Google Scholar] [CrossRef]
- Buettner, D.; Skemp, S. Blue Zones: Lessons from the world’s longest lived. Am. J. Lifestyle Med. 2016, 5, 318–321. [Google Scholar] [CrossRef]
- Nieddu, A.; Vindas, L.; Errigo, A.; Vindas, J.; Pes, G.M.; Dore, M.P. Dietary habits, anthropometric features and daily performance in two independent long-lived populations from Nicoya Peninsula (Costa Rica) and Ogliastra (Sardinia). Nutrients 2020, 6, 1621. [Google Scholar] [CrossRef]
- Tessier, S.; Gerber, M. Comparison between Sardinia and Malta: The Mediterranean diet revisited. Appetite 2005, 45, 121–126. [Google Scholar] [CrossRef]
- Tessier, S.; Gerber, M. Factors determining the nutrition transition in two Mediterranean islands: Sardinia and Malta. Public Health Nutr. 2005, 8, 1286–1292. [Google Scholar] [CrossRef]
- Pes, G.M.; Tolu, F.; Dore, M.P.; Sechi, G.P.; Errigo, A.; Canelada, A.; Poulain, M. Male longevity in Sardinia, a review of historical sources supporting a causal link with dietary factors. Eur. J. Clin. Nutr. 2015, 69, 411–418. [Google Scholar] [CrossRef]
- Salinari, G.; Ruiu, G. The effect of disease burden on the speed of aging: An analysis of the Sardinian mortality transition. Genus 2018, 1, 9. [Google Scholar] [CrossRef]
- Pes, G.M.; Errigo, A.; Tedde, P.; Dore, M.P. Sociodemographic, clinical and functional profile of nonagenarians from two areas of Sardinia characterized by distinct longevity levels. Rejuvenation Res. 2020, 4, 341–348. [Google Scholar] [CrossRef]
- Norton, K.; Whittingham, N.; Carter, L.; Kerr, D.; Gore, C.; Marfell-Jones, M. Measurement Techniques in Anthropometry. Anthropometrica 1996, 1, 25–76. [Google Scholar]
- World Health Organization. Physical Status: The Use and Interpretation of Anthropometry; World Health Organization: Geneva, Switzerland, 1995. [Google Scholar]
- Wang, J.; Thornton, J.C.; Kolesnik, S.; Pierson, R.N. Anthropometry in body composition: An overview. Ann. N. Y. Acad. Sci. 2000, 904, 317–326. [Google Scholar] [CrossRef]
- Linn, B.S.; Linn, M.W.; Gurel, L. Cumulative illness rating scale. J. Am. Geriatr. Soc. 1968, 16, 622–626. [Google Scholar] [CrossRef]
- Vischer, A.S.; Burkard, T. Principles of blood pressure measurement—Current techniques; office vs ambulatory blood pressure measurement. Adv. Exp. Med. Biol. 2017, 956, 85–96. [Google Scholar] [CrossRef]
- Whelton, P.K.; Carey, R.M.; Aronow, W.S.; Casey, D.E.; Collins, K.J.; Dennison Himmelfarb, C.; DePalma, S.M.; Gidding, S.; Jamerson, K.A.; Jones, D.W.; et al. ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation and management of high blood pressure in adults. J. Am. Coll. Cardiol. 2018, 71, e127–e248. [Google Scholar] [CrossRef] [PubMed]
- Idler, E.L.; Benyamini, Y. Self-rated health and mortality: A review of twenty-seven community studies. J. Health. Soc. Behav. 1997, 38, 21–37. [Google Scholar] [CrossRef] [PubMed]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Molloy, D.W.; Standish, T.I. A guide to the standardized minimental state examination. Int. Psychogeriatr. 1997, 9 (Suppl. 1), 87–94. [Google Scholar] [CrossRef]
- Yesavage, J.A.; Brink, T.L.; Rose, T.L.; Lum, O.; Huang, V.; Adey, M.; Leirer, V.O. Development and validation of a geriatric depression screening scale: A preliminary report. J. Psychiatr. Res. 1982–1983, 17, 37–49. [Google Scholar] [CrossRef]
- Sheikh, J.I.; Yesavage, J.A. 9/Geriatric Depression Scale (GDS): Recent evidence and development of a shorter version. Clin. Gerontol. 1986, 5, 165–173. [Google Scholar] [CrossRef]
- Guralnik, J.M.; Simonsick, E.M.; Ferrucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of mortality and nursing home admission. J. Gerontol. 1994, 49, M85–M94. [Google Scholar] [CrossRef]
- Cohen, J. A power primer. Psychol. Bull. 1992, 112, 155–159. [Google Scholar] [CrossRef]
- Bujang, M.A.; Baharum, N. Sample Size Guideline for Correlation Analysis. WJSS 2016, 3, 37–46. [Google Scholar] [CrossRef]
- Brotzu, G. Food conditions. In Proceedings of the International Congress for the Study of the Problem of Underdeveloped Areas, Milan, Italy, 10–15 October 1954. [Google Scholar]
- Willcox, D.C.; Willcox, B.J.; Hsueh, W.C.; Suzuki, M. Genetic determinants of exceptional human longevity: Insights from the Okinawa centenarian study. Age 2006, 4, 313–332. [Google Scholar] [CrossRef]
- Cengarle, L.; Tilloca, G.; Carta, A.; Manca, R. Study on some nutritional parameters of cold meat and salami from Sardinia and Corsica. Chemistry 2001, 30, 133–143. [Google Scholar]
- Bouvard, V.; Loomis, D.; Guyton, K.Z.; Loomis, D.; Guyton, K.Z.; Grosse, Y.; Ghissassi, F.E.; Benbrahim-Tallaa, L.; Guha, N.; Mattock, H.; et al. International Agency for Research on Cancer Monograph Working Group. Carcinogenicity of consumption of red and processed meat. Lancet Oncol. 2015, 16, 1599–1600. [Google Scholar] [CrossRef]
- Pes, G.M.; Fanciulli, G.; Delitala, A.P.; Piana, A.F.; Dore, M.P. Spatial association between gastric cancer mortality and goiter in sardinia. Asian. Pac. J. Cancer Prev. 2021, 1, 105–110. [Google Scholar] [CrossRef]
- Dore, M.P.; Sepulveda, A.R.; El-Zimaity, H.; Yamaoka, Y.; Osato, M.S.; Mototsugu, K.; Nieddu, A.M.; Realdi, G.; Graham, D.Y. Isolation of Helicobacter pylori from sheep-implications for transmission to humans. Am. J. Gastroenterol. 2001, 5, 1396–1401. [Google Scholar] [CrossRef]
- Dore, M.P.; Bilotta, M.; Vaira, D.; Manca, A.; Massarelli, G.; Leandro, G.; Atzei, A.; Pisanu, G.; Graham, D.Y.; Realdi, G. High prevalence of Helicobacter pylori infection in shepherds. Dig. Dis. Sci. 1999, 6, 1161–1164. [Google Scholar] [CrossRef]
- Bernstein, D.M.; Pernat, N.L.M.; Loftus, E.F. The false memory diet: False memories alter food preferences. In Handbook of Behaviour, Food and Nutrition; UC Irvine School of Law Research Paper No. 2011-34; Preedy, V.R., Watson, R.R., Martin, C.R., Eds.; Springer: New York, NY, USA, 2011; pp. 1645–1663. [Google Scholar]
- Garcia-Closas, R.; Berenguer, A.; González, C.A. Changes in food supply in Mediterranean countries from 1961 to 2001. Public Health Nutr. 2006, 1, 53–60. [Google Scholar] [CrossRef]
- Peretti, G. Rapporti tra alimentazione e caratteri antropometrici. Studio statistico-biometrico in Sardegna. Quad. Nutr. 1943, 9, 69–130. [Google Scholar]
- Cuesta-Triana, F.; Verdejo-Bravo, C.; Fernández-Pérez, C.; Martín-Sánchez, F.J. Effect of milk and other dairy products on the risk of frailty, sarcopenia, and cognitive performance decline in the elderly: A systematic review. Adv. Nutr. 2019, 10 (Suppl. 2), S105–S119. [Google Scholar] [CrossRef]
- Larsson, S.C.; Orsini, N. Red meat and processed meat consumption and all-cause mortality: A meta-analysis. Am. J. Epidemiol. 2014, 179, 282–289. [Google Scholar] [CrossRef]
- Marangoni, F.; Corsello, G.; Cricelli, C.; Ferrara, N.; Ghiselli, A.; Lucchin, L.; Poli, A. Role of poultry meat in a balanced diet aimed at maintaining health and wellbeing: An Italian consensus document. Food. Nutr. Res. 2015, 59, 2760. [Google Scholar] [CrossRef]
| Variables | Male (n = 61) | Female (n = 89) |
|---|---|---|
| Age at recruitment (years) | ||
| Mean and standard deviation | 94.2 ± 3.3 | 93.4 ± 4.3 |
| Range | 90–101 | 90–101 |
| Marital status, n (%) | ||
| Single | 8 (13.1) | 13 (14.6) |
| Married | 33 (54.1) | 13 (14.6) |
| Widowed | 19 (31.1) | 63 (70.8) |
| Divorced | 1 (1.6) | 0 (0.0) |
| Education (years) | 3.2 ± 1.7 | 5.4 ± 2.1 |
| Living conditions, n (%) | ||
| Home | 60 (98.4) | 88 (98.8) |
| Nursing home | 1 (1.6) | 1 (1.2) |
| Body mass index (kg/m2) | ||
| <18 | 3 (4.9) | 4 (4.5) |
| 18–24.9 | 31 (50.8) | 49 (55.1) |
| 25–29.9 | 18 (29.5) | 30 (33.7) |
| ≥30 | 9 (14.8) | 6 (6.7) |
| Activities of daily living | ||
| Severe disability | 9 (14.7) | 20 (22.5) |
| Moderate or no disability | 52 (85.2) | 69 (77.5) |
| GDS1score | ||
| <6 | 49 (80.3) | 59 (66.3) |
| ≥6 | 12 (19.7) | 30 (33.7) |
| MMSE2score | ||
| <24 | 32 (52.5) | 52 (58.4) |
| ≥24 | 29 (47.5) | 37 (42.6) |
| Foods | Diet at Age 30 (n = 98) | Current Diet (n = 98) | p Value |
|---|---|---|---|
| Beef/pork meat | 1.38 | 2.01 | <0.00011 |
| Sheep/goat meat | 1.84 | 1.97 | <0.0001 |
| Chicken meat | 1.55 | 1.58 | 0.001 |
| All meat | 1.79 | 1.85 | 0.012 |
| Fish | 1.33 | 1.59 | 0.004 |
| Pulses | 4.42 | 3.73 | <0.0001 |
| Greens | 4.28 | 3.29 | <0.0001 |
| Vegetables | 4.68 | 3.51 | <0.0001 |
| Fresh fruit | 2.43 | 3.58 | <0.0001 |
| Bread | 4.99 | 4.78 | 0.157 |
| Pasta | 3.00 | 3.49 | 0.012 |
| Cereals | 4.99 | 4.14 | <0.0001 |
| Potatoes | 4.33 | 4.21 | 0.956 |
| Olive oil | 2.51 | 2.79 | 0.810 |
| Lard | 3.88 | 3.23 | <0.0001 |
| Sweets | 1.88 | 2.29 | 0.007 |
| Cheese | 4.88 | 4.85 | 0.705 |
| Milk | 4.13 | 4.76 | <0.0001 |
| Dairy | 4.92 | 4.81 | 0.003 |
| Coffee | 3.87 | 3.66 | 0.310 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pes, G.M.; Poulain, M.; Errigo, A.; Dore, M.P. Evolution of the Dietary Patterns across Nutrition Transition in the Sardinian Longevity Blue Zone and Association with Health Indicators in the Oldest Old. Nutrients 2021, 13, 1495. https://doi.org/10.3390/nu13051495
Pes GM, Poulain M, Errigo A, Dore MP. Evolution of the Dietary Patterns across Nutrition Transition in the Sardinian Longevity Blue Zone and Association with Health Indicators in the Oldest Old. Nutrients. 2021; 13(5):1495. https://doi.org/10.3390/nu13051495
Chicago/Turabian StylePes, Giovanni Mario, Michel Poulain, Alessandra Errigo, and Maria Pina Dore. 2021. "Evolution of the Dietary Patterns across Nutrition Transition in the Sardinian Longevity Blue Zone and Association with Health Indicators in the Oldest Old" Nutrients 13, no. 5: 1495. https://doi.org/10.3390/nu13051495
APA StylePes, G. M., Poulain, M., Errigo, A., & Dore, M. P. (2021). Evolution of the Dietary Patterns across Nutrition Transition in the Sardinian Longevity Blue Zone and Association with Health Indicators in the Oldest Old. Nutrients, 13(5), 1495. https://doi.org/10.3390/nu13051495