Abstract
Critically ill patients in the intensive care unit (ICU) have a high risk of developing malnutrition, and this is associated with poorer clinical outcomes. In clinical practice, nutrition, including enteral nutrition (EN), is often not prioritized. Resulting from this, risks and safety issues for patients and healthcare professionals can emerge. The aim of this literature review, inspired by the Rapid Review Guidebook by Dobbins, 2017, was to identify risks and safety issues for patient safety in the management of EN in critically ill patients in the ICU. Three databases were used to identify studies between 2009 and 2020. We assessed 3495 studies for eligibility and included 62 in our narrative synthesis. Several risks and problems were identified: No use of clinical assessment or screening nutrition assessment, inadequate tube management, missing energy target, missing a nutritionist, bad hygiene and handling, wrong time management and speed, nutritional interruptions, wrong body position, gastrointestinal complication and infections, missing or not using guidelines, understaffing, and lack of education. Raising awareness of these risks is a central aspect in patient safety in ICU. Clinical experts can use a checklist with 12 identified top risks and the recommendations drawn up to carry out their own risk analysis in clinical practice.
1. Introduction
In intensive care units (ICUs) critically ill patients have a high risk of developing malnutrition which is associated with a poorer clinical outcome [1]. Therefore, enteral nutrition (EN) has become an increasingly important research topic in recent years. In 2017, Arabi et al. (2017) reported that the intensive care medicine research agenda in nutrition and metabolism includes topics like optimal protein dose combined with standardized active and passive mobilization during the acute and post-acute phases of critical illness, nutritional assessment and nutritional strategies in critically obese patients. Moreover, the effects of continuous versus intermittent EN were classified as a hot topic [2]. The European Society for Clinical Nutrition and Metabolism (ESPEN), established standard operating procedures (SOPs) and guidelines for the provision of the best nutritional therapy for critically ill patients. They are regularly updated and cover various aspects of medical nutritional therapy such as duration, timing, vulnerable patient populations with, for example, dysphagia or frailty and provide clinicians with practical procedures [3]. However, in clinical practice, EN is often not given highest priority due to other symptomatic problems such as cardiovascular status or the need of ventilation. Moreover, ICU patients are mostly very heterogeneous in terms of their illness, resulting in multiple risks and safety issues for patients. The literature offers possible solutions to this problem, but many unresolved questions still cannot be answered conclusively. However, there is some degree of consensus. For ventilated patients in ICUs, if possible, it is crucial to prefer EN over parenteral nutrition (PN) [3]. Other topics like the microbiome or recommendations on additives such as micronutrients and vitamins are still being discussed, for example, optimal vitamin D levels. Many critically ill patients suffer from vitamin D deficiency (serum 25-hydroxyvitamin D [25(OH)D] < 20 ng/mL), with levels lower than 12 ng/dL [4].
In addition to the clinical impact there are further uncertainties that affect the outcome of patients. Overall, human errors are the third leading cause why patients die in a hospital [5]. ICUs are in general a high-risk area, where critical ill patients receive a highly sophisticated care. Patients receive a lot of drugs; medical devices are used for administration of drugs and for ventilation. To increase patient safety, clinical risk management focuses on improving the quality and safety of health care services by identifying the circumstances and opportunities that expose patients to risk of harm, and on acting to prevent or control those risks [6]. In fact, clinical risk management is a key element of clinical governance and management [7]. Risks must be handled appropriately with a bundle of measures and they need to be audited regularly to ascertain whether risk-reducing measures are being used sufficiently [8]. If risks are unknown, a risk management audit is needed to identify potential risks. Especially in EN, there are several risks such as a failure in reaching the nutrition target (CIRSmedical® no. 26 (V1)). The aforementioned error can lead to hypoglycemia and malnutrition [9]. Furthermore, EN and aspiration [10], which can occur when a patient was not placed in a head-up position or when post-pyloric feeding (nasojejunal tube) was not administered to patients with a high risk of aspiration [11]. On top, the worldwide introduction of the DIN EN ISO 80369-3 and the use of ENFit™-technology was a decisive step to increase patient safety in EN and can be seen as an important contribution in terms of risk management. However, the implementation of this standard still has shortcomings as not all security gaps were eliminated. It is still possible that enteral medication in Luer injections or tube feeding can be administered intravenously [12]. In addition to ICU-related risks there are further general risks such as an incorrect patient identification, nosocomial infections, medication errors, overlooked allergies, insufficient pain management, and failures in communication and documentation or failures in handling medical devices [6]. A consequent risk management in ICUs can help to increase patient safety; therefore, the aim of this rapid literature review was to identify risks for patient safety in the management and handling of EN in critically ill patients in ICU.
2. Materials and Methods
We conducted a literature review guided by the Rapid Review Guidebook by Dobbins (2017) [13] and the PRISMA Checklist [14]. As the first step in the EN risk identification process, we (MH, CMS, SF, CS, EL) used feedback from ICU staff (physicians, nurses, dieticians), reports from the Critical Incident Reporting System (CIRS) of the University Hospital of Graz, Austria, CIRSmedical® and, online, the “berrypicking” method [15]. We structured these risks in a process: admission, prescription, verification, preparation, administration, monitoring, discharge, and general risks in EN in the ICU. We then systematically identified and screened the literature for these risks.
Search Strategy
Keywords, related MeSH terms and Boolean operators (AND, OR) were used. The main terms in the first search were: enteral nutrition, risks and intensive care and the identified risks. For example, “enteral nutrition” AND “risks” OR safety AND “intensive care unit” AND “aspiration”. At least two different terms were used for each risk. The primary search was limited to systematic reviews, meta-analyses, and guidelines. Next, studies, including randomized controlled trials (RCTs), retrospective studies and prospective observational studies were included to detect possible risks in EN in the ICU. The search was done with a language restriction to German and English and available full-text articles. Three different scientific databases, PubMed, Cochrane Library and Web of Science, were used to identify studies between 2009 and 2020.
Four independent reviewers (CMS, SF, CS, EL) searched in scientific databases between June and August 2020. The search was always carried out by two independent researchers in order to reduce a possible selection bias. The risks and the related literature were reviewed by independent researchers (CMS, SF, CS, EL and senior researcher (MMJ) with research experience >10 years). After the decision for inclusion of studies, the results were viewed and discussed by two researchers (MH, CMS). All identified risks were thematically summarized in a narrative synthesis by one senior researcher (MMJ) and checked by another researcher (MH). Data were prepared for a summary in a narrative synthesis and a checklist.
3. Results
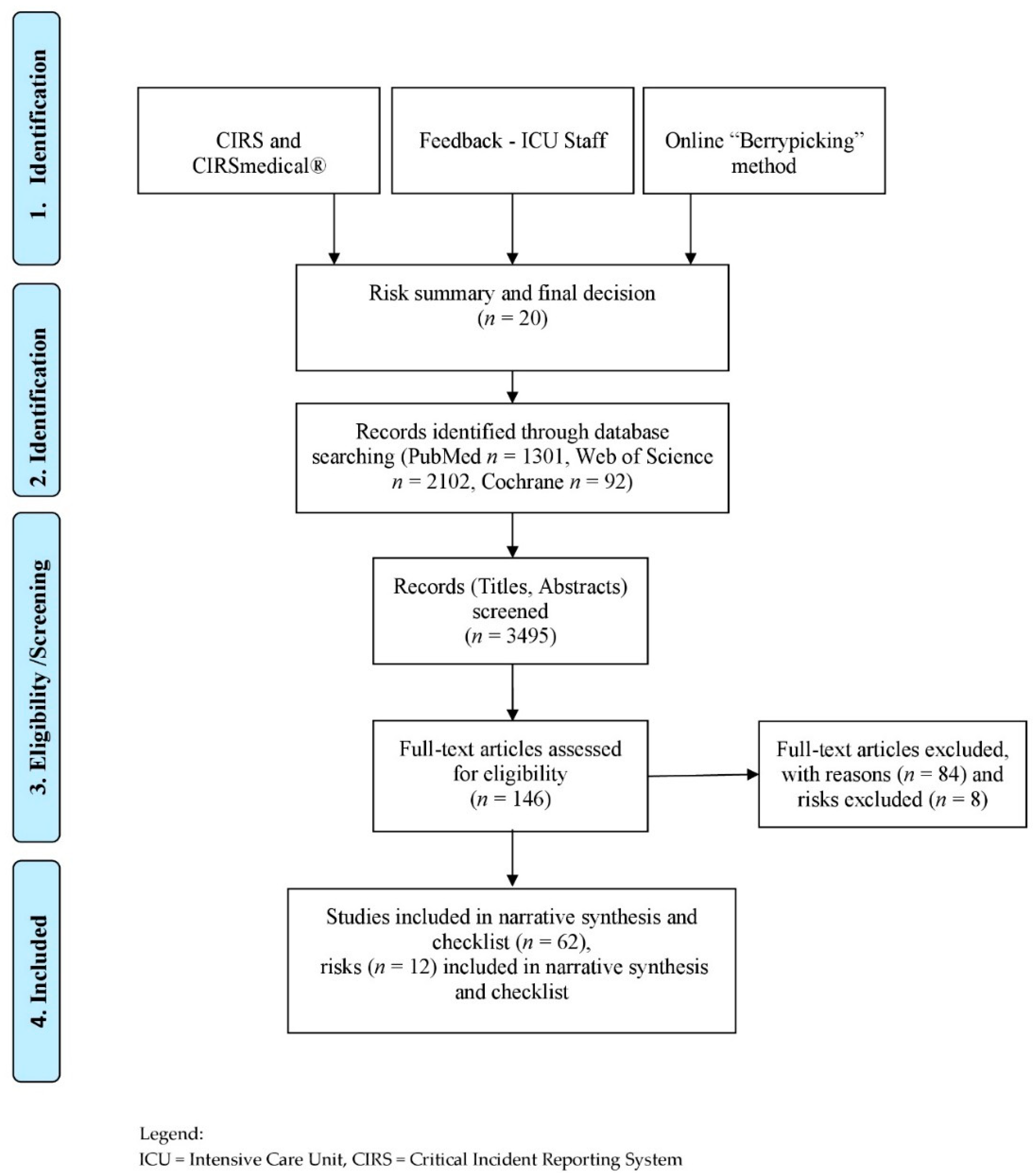
In total we found 20 risks. We included 12 risks in our top risk list, which we could verify in the literature. We had 3495 hits in scientific databases (PubMed n = 1301, Web of Science n = 2102, Cochrane n = 92) and found 146 relevant studies and included 62 in our narrative synthesis. A detailed presentation of the included literature can be found in the following Table 1.

Table 1.
Literature, terms, and hits.
The identified studies focused primarily on assessments or interventions and less on risks or safety issues. These risks and safety issues were derived from the studies directly and indirectly. A detailed description of the search process can be found in Figure 1 (by Moher et al. (2009) [14] adapted by Hoffmann).
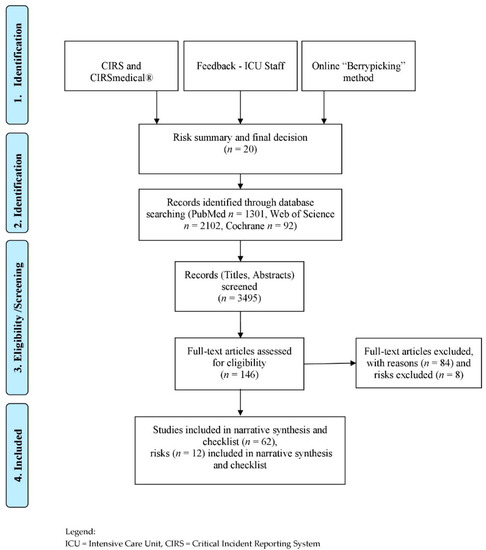
Figure 1.
Flow Diagram—Risk identification process and narrative synthesis.
3.1. Admission
3.1.1. No Use of Clinical Assessment or Screening Nutrition Assessment
Singer et al. (2019) primarily recommended a general clinical assessment, including a history, report of weight loss or decrease in physical performance before ICU admission, physical examination, etc., in addition to screening and assessment instruments. Presence of frailty is considered to be significant in ICU patients, and should therefore be considered in nutritional management. Recording of muscle mass is also used as a parameter for assessing the nutritional status [3]. In a systematic review, Lew et al. (2017) investigated nutrition assessment tools such as the Subjective Global Assessment (SAG) and Mini Nutritional Assessment (MNA) on the one hand and, on the other hand, nutrition screening instruments like the Nutrition Risk Screening 2002 (NRS-2002) and the Malnutrition Universal Screening Tool (MUST) [1]. Further studies focused on instruments such as the NUTRIC score and the mNUTRIC score [16,17,18,19,20,21]. Given that the studies investigated different illnesses, different instruments, various concepts such as nutrition screening or assessment, and that there is no clear definition of critical illness-associated malnutrition [3], various risks can arise in practice. However, studies and guidelines recommended a clinical assessment supplemented by an easy-to-use validated screening instrument such as the NRS-2002 (NRS-2002) and the NUTRIC score—determine both nutrition status and disease severity, while the use of a frailty scale can be helpful for elderly patients.
3.1.2. Inadequate Tube Management and Position
Singer et al. (2019) recommended the use of gastric access as the standard approach to initiate EN. Post-pyloric, mainly jejunal, feeding is possible for patients deemed to be at high risk of aspiration [3]. Ultrasonography, camera-assisted technology with real-time video guidance and X-rays are used to check the position of the tube after insertion and thus obtain a positive outcome [22,23,24]. In clinical practice further tube placement testing methods are used such as aspirate appearance, aspirate pH or auscultation, although X-rays and real-time video guidance are considered to be the most adequate methods. Different types of tubes designed for use with imaging procedures should also be considered. However, certain questions remain unanswered with regard to risk management, for example how long the tubes remain in the correct position, or after what period of time or type of intervention a further positional check is needed, including for jejunal tubes. It is important for individual ICUs to develop a protocol to guide their tube management policy.
3.2. Prescription
Missing Energy Target
For mechanically ventilated patients, guidelines and studies recommended that EN should be determined by indirect calorimetry [25,26,27]. In the absence of indirect calorimetry, VO2, or VCO2 measurements and simple weight-based equations (such as 20–25 kcal/kg/d) should be used. In order to prevent risks, it is important that the prescribed quantity should match the calorie requirement, and that this should be re-evaluated in regular intervals [28], as the measured energy expenditure increased in the course of time with great individual variation [29]. De Waele et al. (2012) recommended a dedicated nutrition support team for a more systematic use of indirect calorimetry in long-term mechanically ventilated patients [30]. However, in order to determine patients’ energy target, their nutritional status before admission to ICU should not be used [3].
3.3. Verification
Missing a Nutritionist at the ICU
Two studies, examining the clinical impact of a two-step interdisciplinary nutrition program and enteral feeding protocols in the ICU, have found that interventions by a dietician significantly improved patient energy balances by day 7 [31,32]. Additionally, the presence of a dietician in the ICU has been associated with better nutrition performance, with a multi-professional approach reducing risks through shared responsibility, interdisciplinary quality programs, and re-evaluations of EN [33]. A systematic review by Mistiaen et al. (2020), stated, that there is weak evidence that Nutrition Support Teams increase appropriate EN use in ICU patients. A decrease of the duration of PN could not be shown [34].
3.4. Preparation
Insufficient Hygiene and Handling
In their study, Perry et al. (2015) compared open systems, “ready-to-hang”-systems (RTH), and modular hospital-built tube feeding systems (MTF), in a normothermic (23 °C) and hypothermal ICU environment. The contamination in both environments/systems does not differentiate between open and closed feeding systems for up to 8 h. However, adding modules to open systems can lead to an unacceptable risk of contamination in hyperthermic (i.e., particularly warm) environments [35]. Training in preparing the setups, maintaining constant temperatures before and after preparation, as well as storage were important factors.
3.5. Administering
3.5.1. Wrong Time Management, Speed and Route
Medical nutrition therapy should be considered for all ICU patients, mainly for those staying for more than 48 h [3]. An oral diet is preferable to EN or PN for critically ill patients who are able to eat. If oral intake is not possible, early EN (within 48 h) in critically ill adult patients should be performed/initiated without delay [3,36,37,38]. To avoid overfeeding, early full EN and PN should not be used in critically ill patients but should be prescribed within three to seven days. Singer et al. (2019) suggest to be caution in critically ill patients with uncontrolled shock, uncontrolled hypoxemia and acidosis, uncontrolled upper gastrointestinal bleeding, gastric aspirate >500 mL/6 h, bowel ischemia, bowel obstruction, abdominal compartment syndrome, and high-output fistula without distal feeding access. A systematic review and a meta-analysis showed that, when given within 48 h after admission, EN itself is efficient and safe for those patients with predicted severe acute pancreatitis [39]. An episode of vomiting was observed in patients with sepsis [40]. Blaser et al. (2017) and Zheng et al. (2019) confirmed that early EN reduced infectious complications in unselected critically ill patients, and in traumatic brain injury, severe acute pancreatitis, gastrointestinal (GI) surgery and abdominal trauma [41,42]. However, their recommendations are weak because of the poor quality of the evidence, with several information based only on expert opinion. In order to actively counter the risks, early EN should be monitored like a vital sign. Though the implementation of the ENFit™-standard it is still possible that enteral medication in Luer injections or tube feeding can be administered intravenously. A systematic review and meta-analysis by Alkhawaja et al. (2015) evaluated the effectiveness and safety of post-pyloric feeding versus gastric feeding. There was no difference in mortality or duration of mechanical ventilation but post-pyloric feeding is associated with lower rates of pneumonia compared with gastric tube feeding [43].
3.5.2. Nutritional Interruptions
Lee et al. (2018) reported 332 episodes of feeding interruptions, this means 12.8% (4190 h) of the total 1367 nutrition days. Each ICU patient experienced feeding interruptions for a median of three days. Total duration of feeding interruptions for the entire ICU stay: 24.5 h, which resulted in an energy and protein deficit. They therefore recommended an evidence-based feeding protocol and a nutrition support team [44]. Williams et al. (2013) investigated the number of nutritional interruptions. They cited education, audit, leadership support, interprofessional collaboration and the use of guidelines as starting points for reducing these interruptions [45]. Based on a chart review, Uozumi et al. (2017) also proposed the development of a protocol for nutritional interruptions, this could possibly reveal deficits in the administration of the EN at an early stage [46]. Prolonged fasting before and after surgery, airway procedures, dressing changes, feed intolerance, and tube malfunction were identified as the most important causes of delays by Segaran et al. (2016) [47,48].
3.5.3. Wrong Body Position
To reduce gastric residual volume in ICU patients, Farsi et al. (2020) recommended positioning patients in the right lateral and supine, semi recumbent positions rather than in the supine position [49]; however, this remains contradictory. There is insufficient literature on this subject. The use of a protocol based on the elevation of the patient’s head, the use of fixed prokinetics and reduced speed of the diet allowed the application of early EN and faster attainment of the planned energy target in prone position, this was found by Regnier et al. (2009) [50]. However, the literature regarding the effect of EN while in the prone position is also sparse and of limited quality [51]. No studies on agitated patients, whose position can change continuously, have been found. Therefore, to prevent risks, ongoing clinical observations are needed.
3.6. Monitoring
Gastrointestinal Complication and Infections
Digestive complications in EN were found. Most often described as vomiting, diarrhea, bowel ischemia, and acute colonic pseudo-obstruction [52,53,54,55,56] these complications were considered as risk factors for extended ICU stay and prolonged mechanical ventilation [55,56,57]. Further complications included refeeding hypophosphatemia and aspiration [11,58,59,60]. However, comparing early EN versus early PN, Singer et al. (2019) showed in their results, a reduction of infectious complications, shorter ICU and hospital stay in EN. They recommended the use of EN over PN in patients with an intact gastrointestinal tract.
3.7. General Risk
3.7.1. Missing or Not Using Existing Guidelines, Standards or Protocols
Singer et al. (2019) recommended the implementation of evidence based protocols [3]. Studies showed significant improvement in EN delivery and reduced duration of feed breaks when using a protocol [39,61,62]. EN is even started earlier [32]. The main barriers to EN guideline compliance were delays, unpredictable timing of procedures, and differing guidance from senior staff and non-ICU teams [3,32,61,63].
3.7.2. Understaffing
Risk factors for inadequate nutrition support as described by Honda et al. (2013), Darawad et al. (2018), Cahill et al., (2012), and Huang et al., (2019) included fewer nursing professionals per bed, and a lack of dietitian coverage during weekends and holidays [42,64,65,66,67].
3.7.3. Lack of Education
Studies show that multifaceted nutritional education programs and protocols are able to improve the knowledge of the healthcare professionals. These tools and training programs should be versatile, easy to use, and can be web-based [68,69,70,71,72,73,74,75].
4. Discussion
Our narrative synthesis highlights the risks of EN in ICU. Each process step, from admission to discharge, demonstrated certain risks, like no use of clinical assessment or screening nutrition tools, inadequate tube management and position, missing energy target, missing a nutritionist at the ICU, bad hygiene and handling, wrong time management and speed, nutritional interruptions, wrong body position, gastrointestinal complication and infections, missing or not using guidelines, standards or protocols, understaffing, and lack of education which are often intertwined and mutually dependent. Due to the heterogeneity of the ICU patient population, no consensual evidence-based protocols about EN were found. However, studies indicated, that for all the aforementioned risks, safety measures exist.
In health care systems in recent years, patient safety has become a priority issue [6]. In addition to individual measures, national and international strategies and protocols have attempted to overcome the most prominent hazards. In clinical risk management it is important to identify, analyze, and manage potential risks. The implementation of measures into routine procedures within complex hospital organizations like ICUs is challenging and have to be monitored regularly as adherence or compliance can be lacking [8].
Based on our findings we developed a short checklist (see Table S1) which can be used by key groups like ICU staff to detect risks in ICUs. Diverse reviewers, with different years of work experience, should use the checklist independently (e.g., nurses, physicians, and dieticians), because a person’s knowledge is not the department’s knowledge. Application of the checklist should be followed by a discussion of the results, implementation of relevant measures and ways of how a distinctive measure can be checked in the routine.
In our results, we did not describe all identified risk which were reported by staff and CIRS, such as interaction between medication or other additives and EN, involvement of relatives and missing discharge plans. This does not imply that these risks and safety issues do not exist, but reflects the lack of adequate literature. Due to a very high turnover in ICU staff [76] and a very complex setting, the implementation of multifaceted nutritional education programs and protocols based on the newest guidelines [3] are necessary to improve and secure the knowledge of healthcare professionals. These tools and training programs should be versatile, easy to use, can be web based [68,69,70,71,72,73,74,75] and trained at regular intervals, followed by internal and external audits [8].
Strengths and Limitations
Our narrative synthesis highlighted risks of EN in ICU. These risks can be observed, and institutional approaches exist for minimizing these risks, for example, by raising awareness, evidence-based protocols, guidelines, consulting professionals, and education. The narrative synthesis should prompt the readers to reflect on their own way of working and on the actual and potential risks. However, our study has several limitations. First, due to the lack of systematic reviews, we also included other studies with different methods and missing high-quality evidence. Depending on the study type, bias is possible. Many of these previous nutrition trials were open to bias because they were unblinded, very small or had other confounders. Second, our literature languages were restricted to German and English and the search terms were also limited due to a large number of studies. Published guideline recommendations for the management of nutrition in ICU patients remain largely supported by expert opinion and only a minority of the studies and reports includes high-quality evidence [77]. Finally, few of the identified studies addressed the risks and safety issues of EN directly, therefore further research is needed.
5. Conclusions
The aim was to identify risks in the management of EN in critically ill patients in ICU. Based on our results, numerous risks related to the management of EN in the ICU were discovered. Clinical experts can use the risk checklist and the recommendations drawn up to carry out their own risk analysis in clinical practice. Once risks have been identified, appropriate measures can be taken. From the authors’ point of view, risk management with tools such as checklists or other risk analysis tools are important for improving patient safety in the ICU and secure knowledge. Further research is needed on how risk management can be implemented in the daily routine, so that the staff reflects on and reviews their own EN management.
Supplementary Materials
The following are available online at https://www.mdpi.com/2072-6643/13/1/82/s1, Table S1: Risk-Checklist.
Author Contributions
Conceptualization, M.H. and C.M.S.; methodology, M.H., C.M.S., and M.-M.J.; validation, M.H., C.M.S., C.S., S.F., E.L., and M.-M.J.; formal analysis, M.H., C.M.S., C.S., S.F., E.L., M.-M.J., and G.S.; data curation, M.H., E.L., C.M.S., C.S., S.F., G.S., and M.-M.J.; writing—original draft preparation, M.H., G.S., and M.-M.J.; writing—review and editing, all authors; visualization, M.H., and C.M.S.; supervision, M.-M.J. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Acknowledgments
We would like to thank Julia Grossenbacher for her help with professional literature management and everyone who contributed to the study. Especially, we would like to thank the Medical University of Graz and the Executive Department for Quality and Risk Management at the University Hospital Graz, Austria.
Conflicts of Interest
SF received speaking honoraria from Takeda. All other authors have declared no conflict of interests.
Abbreviations
| ASPEN | American Society for Parenteral and Enteral Nutrition |
| CIRS | Critical Incident Reporting System |
| DIN EN ISO 80369-3 | Deutsches Institut für Normung and designation of the standard |
| e.g., | exempli gratia: for example |
| EN | Enteral Nutrition |
| ENFit™-technology | Connector for safe feeding |
| ESPEN | European Society for Clinical Nutrition and Metabolism |
| GI | gastrointestinal |
| i.e., | id est: that is |
| ICU | Intensive Care Unit |
| MeSH | Medical Subject Headings |
| MNA | Mini Nutritional Assessment |
| mNUTRIC | modified NUTRIC score |
| MTF | modular hospital-built tube feeding systems |
| MUST | Malnutrition Universal Screening Tool (MUST) |
| NRS-2002 | Nutrition Risk Screening 2002 |
| PN | Parenteral Nutrition |
| RCTs | Randomized Controlled Trials |
| RHT systems | “ready-to-hang”-systems |
| SAG | Subjective Global Assessment |
| SOPs | Standard Operating Procedures (SOPs) |
| VCO2 | volume of carbon dioxide |
| VO2 | volume of oxygen |
References
- Lew, C.C.H.; Yandell, R.; Fraser, R.J.L.; Chua, A.P.; Chong, M.F.F.; Miller, M. Association between Malnutrition and Clinical Outcomes in the Intensive Care Unit: A Systematic Review. J. Parenter. Enter. Nutr. 2017, 41, 744–758. [Google Scholar] [CrossRef] [PubMed]
- Arabi, Y.M.; Casaer, M.P.; Chapman, M.; Heyland, D.K.; Ichai, C.; Marik, P.E.; Martindale, R.G.; McClave, S.A.; Preiser, J.C.; Reignier, J.; et al. The intensive care medicine research agenda in nutrition and metabolism. Intensive Care Med. 2017, 43, 1239–1256. [Google Scholar] [CrossRef] [PubMed]
- Singer, P.; Blaser, A.R.; Berger, M.M.; Alhazzani, W.; Calder, P.C.; Casaer, M.P.; Hiesmayr, M.; Mayer, K.; Montejo, J.C.; Pichard, C.; et al. ESPEN guideline on clinical nutrition in the intensive care unit. Clin. Nutr. 2019, 38, 48–79. [Google Scholar] [CrossRef] [PubMed]
- Amrein, K.; Scherkl, M.; Hoffmann, M.; Neuwersch-Sommeregger, S.; Köstenberger, M.; Tmava Berisha, A.; Martucci, G.; Pilz, S.; Malle, O. Vitamin D deficiency 2.0: An update on the current status worldwide. Eur. J. Clin. Nutr. 2020, 74, 1498–1513. [Google Scholar] [CrossRef] [PubMed]
- Makary, M.A.; Daniel, M. Medical Error-the Third Leading Cause of Death in the US. Available online: https://www.bmj.com/content/353/bmj.i2139 (accessed on 20 October 2020).
- Sendlhofer, G.; Brunner, G.; Tax, C.; Falzberger, G.; Smolle, J.; Leitgeb, K.; Kober, B.; Kamolz, L.P. Systematische Einführung vom Klinischen Risikomanagement in einem Universitätsklinikum: Bedeutung von Risikomanagern. Wien. Klin. Wochenschr. 2015, 127, 1–11. [Google Scholar] [CrossRef]
- Chiozza, M.L.; Ponzetti, C. FMEA: A model for reducing medical errors. Clin. Chim. Acta 2009, 404, 75–78. [Google Scholar] [CrossRef]
- Sendlhofer, G.; Leitgeb, K.; Kober, B.; Brunner, G.; Tax, C.; Kamolz, L.P. Neue Wege zur Evaluierung von patientensicherheitsrelevanten Aspekten: Feedback-Patientensicherheit. Z. Evid. Fortbild. Qual. Gesundhwes. 2016, 114, 13–27. [Google Scholar] [CrossRef]
- Kim, H.; Stotts, N.A.; Froelicher, E.S.; Engler, M.M.; Porter, C.; Kwak, H. Adequacy of early enteral nutrition in adult patients in the intensive care unit. J. Clin. Nurs. 2012, 21, 2860–2869. [Google Scholar] [CrossRef]
- Nseir, S.; Le Gouge, A.; Lascarrou, J.-B.; Lacherade, J.-C.; Jaillette, E.; Mira, J.-P.; Mercier, E.; Declercq, P.-L.; Sirodot, M.; Piton, G.; et al. Impact of nutrition route on microaspiration in critically ill patients with shock: A planned ancillary study of the NUTRIREA-2 trial. Crit. Care 2019, 23, 111. [Google Scholar] [CrossRef]
- Sajid, M.S.; Harper, A.; Hussain, Q.; Forni, L.; Singh, K.K. An integrated systematic review and meta-analysis of published randomized controlled trials evaluating nasogastric against postpyloris (nasoduodenal and nasojejunal) feeding in critically ill patients admitted in intensive care unit. Eur. J. Clin. Nutr. 2014, 68, 424–432. [Google Scholar] [CrossRef]
- ISO Small-Bore Connectors for Liquids and Gases in Healthcare Applications—Part 1: General Requirements. Available online: https://www.iso.org/standard/64419.html (accessed on 20 October 2020).
- Dobbins, M. Rapid Review Guidebook. Natl. Collab. Cent. Method Tools 2017, 13, 25. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Altman, D.; Antes, G.; Atkins, D.; Barbour, V.; Barrowman, N.; Berlin, J.A.; et al. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Kleibel, V.; Mayer, H. Literaturrecherche Für Gesundheitsberufe, 2nd ed.; Maudrich Wien: Wien, Austria, 2011; ISBN 978-3-7089-0309-5. [Google Scholar]
- Canales, C.; Elsayes, A.; Yeh, D.D.; Belcher, D.; Nakayama, A.; McCarthy, C.M.; Chokengarmwong, N.; Quraishi, S.A. Nutrition Risk in Critically Ill Versus the Nutritional Risk Screening 2002: Are They Comparable for Assessing Risk of Malnutrition in Critically Ill Patients? J. Parenter. Enter. Nutr. 2019, 43, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Coruja, M.K.; Cobalchini, Y.; Wentzel, C.; Fink, J.D. Nutrition Risk Screening in Intensive Care Units: Agreement Between NUTRIC and NRS 2002 Tools. Nutr. Clin. Pract. 2020, 35, 567–571. [Google Scholar] [CrossRef]
- Hollis, G.; Robins, E.; Powlesand, A.; Duff, A.; Ziegenfuss, M.; Shekar, K. Evaluation of the nutrition risk in critically ill score against a dietitian led nutrition triage tool in a tertiary ICU. Aust. Crit. Care 2018, 31, 123–124. [Google Scholar] [CrossRef]
- Kenworthy, S.; Agarwal, E.; Farlow, L.; Angus, R.; Marshall, A.P. Feasibility of using the “modified NUTrition Risk In the Critically ill” nutritional risk screening tool to identify nutritionally at-risk patients in an Australian intensive care unit. Aust. Crit. Care 2020, 33, 259–263. [Google Scholar] [CrossRef]
- Rahman, A.; Hasan, R.M.; Agarwala, R.; Martin, C.; Day, A.G.; Heyland, D.K. Identifying critically-ill patients who will benefit most from nutritional therapy: Further validation of the “modified NUTRIC” nutritional risk assessment tool. Clin. Nutr. 2016, 35, 158–162. [Google Scholar] [CrossRef]
- Zhang, P.; Bian, Y.; Tang, Z.; Wang, F. Use of Nutrition Risk in Critically Ill (NUTRIC) Scoring System for Nutrition Risk Assessment and Prognosis Prediction in Critically Ill Neurological Patients: A Prospective Observational Study. J. Parenter. Enter. Nutr. 2020, 1–10. [Google Scholar] [CrossRef]
- Li, Y.; Ye, Y.; Mei, Y.; Ruan, H.; Yu, Y. Semi-automated ultrasound guidance applied to nasogastrojejunal tube replacement for enteral nutrition in critically ill adults. Biomed. Eng. Online 2018, 17, 21. [Google Scholar] [CrossRef]
- Gao, X.; Zhang, L.; Zhao, J.; Tian, F.; Sun, H.; Wang, P.; Wang, J.; Wang, Z.; Wang, X. Bedside electromagnetic-guided placement of nasoenteral feeding tubes among critically Ill patients: A single-centre randomized controlled trial. J. Crit. Care 2018, 48, 216–221. [Google Scholar] [CrossRef]
- Wischmeyer, P.E.; McMoon, M.M.; Waldron, N.H.; Dye, E.J. Successful Identification of Anatomical Markers and Placement of Feeding Tubes in Critically Ill Patients via Camera-Assisted Technology with Real-Time Video Guidance. J. Parenter. Enter. Nutr. 2019, 43, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Sabatino, A.; Theilla, M.; Hellerman, M.; Singer, P.; Maggiore, U.; Barbagallo, M.; Regolisti, G.; Fiaccadori, E. Energy and protein in critically Ill patients with AKI: A prospective, multicenter observational study using indirect calorimetry and protein catabolic rate. Nutrients 2017, 9, 802. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.M.; Reintam-Blaser, A.; Calder, P.C.; Casaer, M.; Hiesmayr, M.J.; Mayer, K.; Montejo, J.C.; Pichard, C.; Preiser, J.C.; van Zanten, A.R.H.; et al. Monitoring nutrition in the ICU. Clin. Nutr. 2019, 38, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Frankenfield, D.C.; Ashcraft, C.M.; Galvan, D.A. Longitudinal prediction of metabolic rate in critically ill patients. J. Parenter. Enter. Nutr. 2012, 36, 700–712. [Google Scholar] [CrossRef] [PubMed]
- Singer, P.; Anbar, R.; Cohen, J.; Shapiro, H.; Shalita-Chesner, M.; Lev, S.; Grozovski, E.; Theilla, M.; Frishman, S.; Madar, Z. The tight calorie control study (TICACOS): A prospective, randomized, controlled pilot study of nutritional support in critically ill patients. Intensive Care Med. 2011, 37, 601–609. [Google Scholar] [CrossRef] [PubMed]
- Ridley, E.J.; Tierney, A.; King, S.; Ainslie, E.; Udy, A.; Scheinkestel, C.; Nyulasi, I. Measured Energy Expenditure Compared With Best-Practice Recommendations for Obese, Critically Ill Patients—A Prospective Observational Study. J. Parenter. Enter. Nutr. 2020, 44, 1144–1149. [Google Scholar] [CrossRef]
- De Waele, E.; Spapen, H.; Honoré, P.M.; Mattens, S.; Rose, T.; Huyghens, L. Bedside calculation of energy expenditure does not guarantee adequate caloric prescription in long-term mechanically ventilated critically ill patients: A quality control study. Sci. World J. 2012, 2012, 909564. [Google Scholar] [CrossRef]
- Soguel, L.; Revelly, J.P.; Schaller, M.D.; Longchamp, C.; Berger, M.M. Energy deficit and length of hospital stay can be reduced by a two-step quality improvement of nutrition therapy: The intensive care unit dietitian can make the difference. Crit. Care Med. 2012, 40, 412–419. [Google Scholar] [CrossRef]
- Heyland, D.K.; Cahill, N.E.; Dhaliwal, R.; Sun, X.; Day, A.G.; McClave, S.A. Impact of enteral feeding protocols on enteral nutrition delivery: Results of a multicenter observational study. J. Parenter. Enter. Nutr. 2010, 34, 675–684. [Google Scholar] [CrossRef]
- Arney, B.D.; Senter, S.A.; Schwartz, A.C.; Meily, T.; Pelekhaty, S. Effect of Registered Dietitian Nutritionist Order-Writing Privileges on Enteral Nutrition Administration in Selected Intensive Care Units. Nutr. Clin. Pract. 2019, 34, 899–905. [Google Scholar] [CrossRef]
- Mistiaen, P.; Van den Heede, K. Nutrition Support Teams: A Systematic Review. J. Parenter. Enter. Nutr. 2020, 44, 1004–1020. [Google Scholar] [CrossRef] [PubMed]
- Perry, J.; Stankorb, S.M.; Salgueiro, M. Microbial contamination of enteral feeding products in thermoneutral and hyperthermal ICU environments. Nutr. Clin. Pract. 2015, 30, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Elke, G.; van Zanten, A.R.H.; Lemieux, M.; McCall, M.; Jeejeebhoy, K.N.; Kott, M.; Jiang, X.; Day, A.G.; Heyland, D.K. Enteral versus parenteral nutrition in critically ill patients: An updated systematic review and meta-analysis of randomized controlled trials. Crit. Care 2016, 20, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Doig, G.S.; Heighes, P.T.; Simpson, F.; Sweetman, E.A. Early enteral nutrition reduces mortality in trauma patients requiring intensive care: A meta-analysis of randomised controlled trials. Injury 2011, 42, 50–56. [Google Scholar] [CrossRef]
- Tian, F.; Heighes, P.T.; Allingstrup, M.J.; Doig, G.S. Early enteral nutrition provided within 24 hours of ICU admission: A meta-analysis of randomized controlled trials. Crit. Care Med. 2018, 46, 1049–1056. [Google Scholar] [CrossRef]
- Song, J.; Zhong, Y.; Lu, X.; Kang, X.; Wang, Y.; Guo, W.; Liu, J.; Yang, Y.; Pei, L. Enteral nutrition provided within 48 hours after admission in severe acute pancreatitis: A systematic review and meta-analysis. Medicine (United States) 2018, 97, e11871. [Google Scholar] [CrossRef]
- Patel, J.J.; Kozeniecki, M.; Peppard, W.J.; Peppard, S.R.; Zellner-Jones, S.; Graf, J.; Szabo, A.; Heyland, D.K. Phase 3 Pilot Randomized Controlled Trial Comparing Early Trophic Enteral Nutrition With “No Enteral Nutrition” in Mechanically Ventilated Patients With Septic Shock. J. Parenter. Enter. Nutr. 2020, 44, 866–873. [Google Scholar] [CrossRef]
- Reintam Blaser, A.; Starkopf, J.; Alhazzani, W.; Berger, M.M.; Casaer, M.P.; Deane, A.M.; Fruhwald, S.; Hiesmayr, M.; Ichai, C.; Jakob, S.M.; et al. Early enteral nutrition in critically ill patients: ESICM clinical practice guidelines. Intensive Care Med. 2017, 43, 380–398. [Google Scholar] [CrossRef]
- Zheng, X.-X.; Jiang, L.-X.; Huang, M. Early Versus Delayed Enteral Nutrition in Critically Ill Patients: A Meta-Analysis of Randomized Controlled Trials. Int. J. Clin. Exp. Med. 2019, 12, 4755–4763. [Google Scholar]
- Alkhawaja, S.; Martin, C.; Butler, R.J.; Gwadry-Sridhar, F. Post-pyloric versus gastric tube feeding for preventing pneumonia and improving nutritional outcomes in critically ill adults. Cochrane Database Syst. Rev. 2015, 1–72. [Google Scholar] [CrossRef]
- Lee, Z.-Y.; Ibrahim, N.A.; Mohd-Yusof, B.-N. Prevalence and duration of reasons for enteral nutrition feeding interruption in a tertiary intensive care unit. Nutrition 2018, 53, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Williams, T.A.; Leslie, G.D.; Leen, T.; Mills, L.; Dobb, G.J. Reducing interruptions to continuous enteral nutrition in the intensive care unit: A comparative study. J. Clin. Nurs. 2013, 22, 2838–2848. [Google Scholar] [CrossRef] [PubMed]
- Uozumi, M.; Sanui, M.; Komuro, T.; Iizuka, Y.; Kamio, T.; Koyama, H.; Mouri, H.; Masuyama, T.; Ono, K.; Lefor, A.K. Interruption of enteral nutrition in the intensive care unit: A single-center survey. J. Intensive Care 2017, 5, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Segaran, E.; Barker, I.; Hartle, A. Optimising enteral nutrition in critically ill patients by reducing fasting times. J. Intensive Care Soc. 2016, 17, 38–43. [Google Scholar] [CrossRef]
- Sudenis, T.; Hall, K.; Cartotto, R. Enteral nutrition: What the dietitian prescribes is not what the burn patient gets! J. Burn Care Res. 2015, 36, 297–305. [Google Scholar] [CrossRef]
- Farsi, Z.; Kamali, M.; Butler, S.; Zareiyan, A. The Effect of Semirecumbent and Right Lateral Positions on the Gastric Residual Volume of Mechanically Ventilated, Critically Ill Patients. J. Nurs. Res. 2020, 28, e108. [Google Scholar] [CrossRef]
- Reignier, J.; Dimet, J.; Martin-Lefevre, L.; Bontemps, F.; Fiancette, M.; Clementi, E.; Lebert, C.; Renard, B. Before-after study of a standardized ICU protocol for early enteral feeding in patients turned in the prone position. Clin. Nutr. 2010, 29, 210–216. [Google Scholar] [CrossRef]
- Machado, L.D.S.; Rizzi, P.; Silva, F.M. Administration of enteral nutrition in the prone position, gastric residual volume and other clinical outcomes in critically ill patients: A systematic review. Rev. Bras. Ter. Intensiva 2020, 32, 133–142. [Google Scholar] [CrossRef]
- Fabiani, A.; Sanson, G.; Bottigliengo, D.; Dreas, L.; Zanetti, M.; Lorenzoni, G.; Gatti, G.; Sacilotto, M.; Pappalardo, A.; Gregori, D. Impact of a natural versus commercial enteral-feeding on the occurrence of diarrhea in critically ill cardiac surgery patients. A retrospective cohort study. Int. J. Nurs. Stud. 2020, 108, 103605. [Google Scholar] [CrossRef]
- Reignier, J.; Boisramé-Helms, J.; Brisard, L.; Lascarrou, J.B.; Ait Hssain, A.; Anguel, N.; Argaud, L.; Asehnoune, K.; Asfar, P.; Bellec, F.; et al. Enteral versus parenteral early nutrition in ventilated adults with shock: A randomised, controlled, multicentre, open-label, parallel-group study (NUTRIREA-2). Lancet 2018, 391, 133–143. [Google Scholar] [CrossRef]
- Arabi, Y.M.; Aldawood, A.S.; Haddad, S.H.; Al-Dorzi, H.M.; Tamim, H.M.; Jones, G.; Mehta, S.; McIntyre, L.; Solaiman, O.; Sakkijha, M.H.; et al. Permissive Underfeeding or Standard Enteral Feeding in Critically Ill Adults. N. Engl. J. Med. 2015, 372, 2398–2408. [Google Scholar] [CrossRef] [PubMed]
- Atasever, A.G.; Ozcan, P.E.; Kasali, K.; Abdullah, T.; Orhun, G.; Senturk, E. The frequency, risk factors, and complications of gastrointestinal dysfunction during enteral nutrition in critically ill patients. Ther. Clin. Risk Manag. 2018, 14, 385–391. [Google Scholar] [CrossRef]
- Jakob, S.M.; Bütikofer, L.; Berger, D.; Coslovsky, M.; Takala, J. A randomized controlled pilot study to evaluate the effect of an enteral formulation designed to improve gastrointestinal tolerance in the critically ill patient-the SPIRIT trial. Crit. Care 2017, 21, 140. [Google Scholar] [CrossRef] [PubMed]
- Gungabissoon, U.; Hacquoil, K.; Bains, C.; Irizarry, M.; Dukes, G.; Williamson, R.; Deane, A.M.; Heyland, D.K. Prevalence, Risk Factors, Clinical Consequences, and Treatment of Enteral Feed Intolerance During Critical Illness. J. Parenter. Enter. Nutr. 2015, 39, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, E.; Yeh, D.D.; Quraishi, S.A.; Johnson, E.A.; Kaafarani, H.; Lee, J.; King, D.R.; Demoya, M.; Fagenholz, P.; Butler, K.; et al. Hypophosphatemia in Enterally Fed Patients in the Surgical Intensive Care Unit: Common but Unrelated to Timing of Initiation or Aggressiveness of Nutrition Delivery. Nutr. Clin. Pract. 2017, 32, 252–257. [Google Scholar] [CrossRef]
- Coşkun, R.; Gündoǧan, K.; Baldane, S.; Güven, M.; Sungur, M. Refeeding hypophosphatemia: A potentially fatal danger in the intensive care unit. Turk. J. Med. Sci. 2014, 44, 369–374. [Google Scholar] [CrossRef]
- Ralib, A.M.; Nor, M.B.M. Refeeding hypophosphataemia after enteral nutrition in a Malaysian intensive care unit: Risk factors and outcome. Asia Pac. J. Clin. Nutr. 2018, 27, 329–335. [Google Scholar] [CrossRef]
- Jenkins, B.; Calder, P.C.; Marino, L.V. Evaluation of implementation of fasting guidelines for enterally fed critical care patients. Clin. Nutr. 2019, 38, 252–257. [Google Scholar] [CrossRef]
- Jiang, L.; Huang, X.; Wu, C.; Tang, J.; Li, Q.; Feng, X.; He, T.; Wang, Z.; Gao, J.; Ruan, Z.; et al. The effects of an enteral nutrition feeding protocol on critically ill patients: A prospective multi-center, before-after study. J. Crit. Care 2020, 56, 249–256. [Google Scholar] [CrossRef]
- Friesecke, S.; Schwabe, A.; Stecher, S.S.; Abel, P. Improvement of enteral nutrition in intensive care unit patients by a nurse-driven feeding protocol. Nurs. Crit. Care 2014, 19, 204–210. [Google Scholar] [CrossRef]
- Honda, C.K.Y.; Freitas, F.G.R.; Stanich, P.; Mazza, B.F.; Castro, I.; Nascente, A.P.M.; Bafi, A.T.; Azevedo, L.C.P.; Machado, F.R. Nurse to bed ratio and nutrition support in critically ill patients. Am. J. Crit. Care 2013, 22, e71–e78. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Darawad, M.W.; Alfasfos, N.; Zaki, I.; Alnajar, M.; Hammad, S.; Samarkandi, O.A. ICU Nurses’ Perceived Barriers to Effective Enteral Nutrition Practices: A Multicenter Survey Study. Open Nurs. J. 2018, 12, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Cahill, N.E.; Murch, L.; Cook, D.; Heyland, D.K. Barriers to feeding critically ill patients: A multicenter survey of critical care nurses. J. Crit. Care 2012, 27, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Yang, L.; Zhuang, Y.; Qi, H.; Chen, X.; Lv, K. Current status and influencing factors of barriers to enteral feeding of critically ill patients: A multicenter study. J. Clin. Nurs. 2019, 28, 677–685. [Google Scholar] [CrossRef]
- Spear, S.; Sim, V.; Moore, F.A.; Todd, S.R. Just say no to intensive care unit starvation: A nutrition education program for surgery residents. Nutr. Clin. Pract. 2013, 28, 387–391. [Google Scholar] [CrossRef]
- Morphet, J.; Clarke, A.B.; Bloomer, M.J. Intensive care nurses’ knowledge of enteral nutrition: A descriptive questionnaire. Intensive Crit. Care Nurs. 2016, 37, 68–74. [Google Scholar] [CrossRef]
- Kim, H.; Chang, S.J. Implementing an educational program to improve critical care nurses’ enteral nutritional support. Aust. Crit. Care 2019, 32, 218–222. [Google Scholar] [CrossRef]
- Castro, M.G.; Pompilio, C.E.; Horie, L.M.; Verotti, C.C.G.; Waitzberg, D.L. Education program on medical nutrition and length of stay of critically ill patients. Clin. Nutr. 2013, 32, 1061–1066. [Google Scholar] [CrossRef]
- Gonya, S.; Baram, M. Do we really know how much we are feeding our patients? Hosp. Pract. (1995) 2015, 43, 277–283. [Google Scholar] [CrossRef]
- Hurt, R.T.; McClave, S.A.; Evans, D.C.; Jones, C.; Miller, K.R.; Frazier, T.H.; Minhas, M.A.; Lowen, C.C.; Stout, A.; Edakkanambeth Varayil, J.; et al. Targeted Physician Education Positively Affects Delivery of Nutrition Therapy and Patient Outcomes: Results of a Prospective Clinical Trial. J. Parenter. Enter. Nutr. 2015, 39, 948–952. [Google Scholar] [CrossRef]
- Lee, J.M.; Fernandez, F.; Staff, I.; Mah, J.W. Web-based teaching module improves success rates of postpyloric positioning of nasoenteric feeding tubes. J. Parenter. Enter. Nutr. 2012, 36, 323–329. [Google Scholar] [CrossRef] [PubMed]
- McCall, M.; Cahill, N.; Murch, L.; Sinuff, T.; Bray, T.; Tanguay, T.; Heyland, D.K. Lessons learned from implementing a novel feeding protocol: Results of a multicenter evaluation of educational strategies. Nutr. Clin. Pract. 2014, 29, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, B.A.; Gillum, D.R.; Kelly, J.M. Burnout, Moral Distress, and Job Turnover in Critical Care Nurses. Int. J. Stud. Nurs. 2018, 3, 108. [Google Scholar] [CrossRef]
- Xu, E.; Tejada, S.; Solé-Lleonart, C.; Campogiani, L.; Valenzuela-Sanchez, F.; Koulenti, D.; Rello, J. Evaluation of the quality of evidence supporting guideline recommendations for the nutritional management of critically ill adults. Clin. Nutr. ESPEN 2020, 39, 144–149. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).