Abstract
Nutraceuticals are a category of products more often associated with food but having pharmaceuticals property and characteristics. However, there is still no internationally accepted concept of these food-pharmaceutical properties, and their interpretation can differ from country to country. Nutraceuticals are used as part of dietary supplements in most countries. They can be phytochemicals which are biologically active and have health benefits. These can be supplied as a supplement and/or as a functional food to the customer. For human health and longevity, these materials are likely to play a vital role. Consumption of these items is typical without a therapeutic prescription and/or supervision by the vast majority of the public. The development of nutraceuticals can be achieved through many bioresources and organisms. This review article will discuss the current research on nutraceuticals from different biological sources and their potential use as an agent for improving human health and well-being, as well as the gaps and future perspective of research related to nutraceutical development.
1. Introduction to Nutraceuticals
The term “nutraceutical” has been used since the late 1980s to describe medicinally or nutritionally functional food [1]. A nutraceutical can also be described as an all-natural product that is known to have biological advantages against one or more illnesses. The term “nutraceuticals” is an amalgamation of the words “nutrient” and “ceutical”—where “ceutical” means therapy—and it is referring to food, supplements, and/or products that provide therapeutical medical value besides main nutrients [2]. Generally, nutraceuticals are claimed to possess pharmaceutical and/or anti-ageing properties, be capable of reducing symptoms or prevent the development of some long-term diseases, improve overall wellness, or show other specific physiological benefits, such as boosting immunity [3]. Nutraceuticals may be referred to as superfoods, health supplements such as vitamins that are derived from natural sources, phytochemicals, nutrient-rich food such as fortified cereals, dietary supplements such as functional herbal pills, and food from a genetically modified organism that serve other purposes besides nutrition (Figure 1) [4].
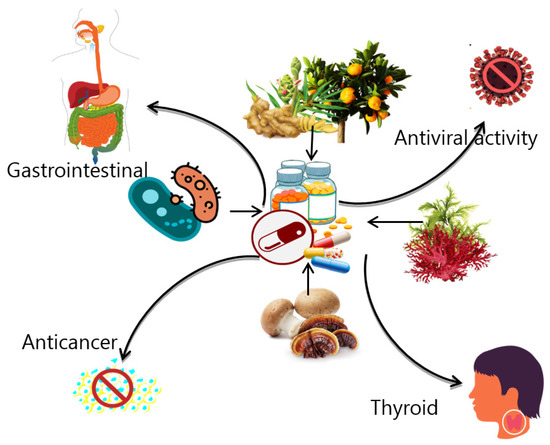
Figure 1.
An overview of different types of nutraceuticals and their uses.
Nutraceuticals could be very useful and effective on the condition that they are used properly or as advised by a physician [5]. Nutraceuticals are generally safe to be consumed with very few reported side effects and contraindication [5]. The daily consumptions of nutraceuticals may help in some diseases to avoid or reduce the intake of traditional medication especially for those to be taken for the whole life [6,7]. Health supplements include products that have micro- and macro-nutrient elements such as organic acids, vitamins, flavonoids, phenolic compounds, glucosinolates, polysaccharides, amino acids and protein, and fibres [8] and are normally classified in the market according to their types of nutritional functions such as dietary fibre, pro- and pre-biotics, and vitamins [9]. It has been proposed that plants, animals, and even mushrooms can be a good source for nutraceuticals/pharmaceutical factories which have proven to be more economically efficient [10]. They are regarded as sources for the avoidance of potentially fatal illnesses such as kidney and gut disorders, as well as prevention of microbial disease [11]. On the other side, the industry has its definition for nutraceuticals as “any nontoxic food component that has potential health benefits (Figure 2) [12]. It depends on each country regulation to define and regulate; for instance, in the United States, the Food and Drug Administration (FDA) regulates nutraceuticals as a dietary supplement, while in Malaysia there is no definition for nutraceuticals, and the National Pharmaceutical Regulatory Agency (NPRA) approves health supplements depending on the claim made by the manufacturer [13].
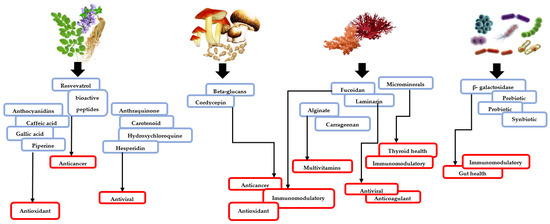
Figure 2.
Flow chart illustrating different sources of potential molecules to be used in nutraceutical applications. Natural nutraceuticals could be obtained from higher plants, fungi, bacteria, and algae.
The industry related to nutraceutical and supplement production earns more than 200 billion USD per year globally, and it is expected to double in the upcoming five years [14,15]. Strict regulation is required for the use of these nutraceuticals supported by evidence that those supplement meet minimum standards of safety and quality before approving the production for that supplement, considering that those product are getting more popular and the potential risk from any possible risk or side effect [16]. For that reason, understanding the information regarding nutraceuticals, their proven benefits, and possible side effects is essential for both the manufacturer and consumers. The prospective of a superfood, supplement products to aid health, and the development of substitute remedies for different illness is supported by relatively many in-depth studies and research; however, the mode of action of the active compound is still unknown in many cases and needs further investigations [17]. The goal of this review article is to introduce, define, and briefly discuss the types of potential molecules that can be used in the production of nutraceuticals that are derived from bacterial, fungal, algal, cyanobacterial, and plant origins while highlighting their potential impacts on the consumers’ health. The food industry has drastically also adjusted to accomplish customers’ necessities with the help of modern food manufacturing technologies. Due to the wide connection between contemporary nourishment and the emergence of a pandemic disease, superfood may be the answer to boost immunity and prevent or treat epidemic as well as pandemic diseases.
2. Nutraceuticals from Bacterial Origin
Several nonpathogenic microorganisms have demonstrated therapeutic properties that are beneficial for gut and immune health, and they are collectively known as probiotics. A probiotic is known as “valuable live microorganisms”, that benefit to the host when given in suitable amounts. Food industries such as vegetables, milk, meat products are wide applications of probiotic bacteria; to extend their positive effects to the host, they must be taken in sufficient quantities [18], that of 106–109 CFU/g upon intake. The positive effect of probiotics is linked to a specific strain of bacteria, not species only [19]. These strains of bacterial probiotics are alleged to grant health benefits to the human when consumed adequately [20]. Functional food which carrying probiotic bacteria exhibit determinations in connection with the viability of the bacterial cells, correlating with criteria such as food processing and storage conditions, and the acidity of the food matrix [21,22]. The gut normal flora can be moderated to recover host healthiness through the administration of probiotic organisms [23]. The encapsulation of mRNA, siRNA, and proteins into extracellular vesicles of Bacillus subtilis, for instance, could have functional participation in food- and clinical-related therapy through the communication between host and the bacteria, reaching the bloodstream and consequently to the intestine and organs [24]. Examples of daily consumed commercially probiotics that are relatively affordable are Lactobacillus casei and Lactobacillus brevis, suitable for curing children in 6–12 years old suffering from early carious lesions [25]. In this case, the bacteria slow down the development of cariogenic germs and complete the function of fluoride to provide a better oral habitat, and such food raises the buffer solution and enhances the dental remineralisation [26]. Lactobacillus probiotics have anticolorectal cancer activity, Lactobacillus plantarum, Pediococcus pentosaceus, and Lactobacillus rhamnosus ability to modulate the metabolism and decreasing faecal pH, leading to the elimination of carcinogens and toxins [27]. Yoghurt is the famous example of at-hand’s-reach probiotics, these products usually contain one or a mixture of Lactobacillus delbrueckii subsp bulgaricus and Streptococcus thermophiles, and some types of additions of Bifidobacterium and Lactobacillus to enhance health benefits [28,29]. Lactobacillus reuteri and L. rhamnosus were recently reported for their antioxidant properties with more than 38% scavenging activities [30]. On the other hand, yoghurt consumption is related to β-galactosidase enzyme produced by yoghurt bacteria with the ability to enhance lactose digestion [31]. The Scheers et al. (2016) study on vegetables consumed by humans concluded that iron bioavailability increased in vegetables fermented with L. plantarum, when compared to nonfermented groups [32]. Lactic acid bacteria (LAB) are known to be used for food preservation and safety. Their phytate-degrading ability is of importance in food processing as it digests phytates that are known to inhibit the absorption of some minerals such as iron, calcium, and zinc [33].
Probiotics are potential factors in the handling of obesity, leading to a reduction in body weight, body mass index, and total fat [34]. Quantitative and qualitative disorders in gut microbiota lead to dysbiosis [35]. Studies showed an enhancement of gut morphology leads to productive modification of the GI metabolome [36,37]. Intestinal bacteria have enzymes with the ability of deconjugation, dehydrogenation, dihydroxylation, and sulfation of primary bile acids which control detoxification [38]. To avoid dysbiosis in the gut of humans, it is favourable to consume LAB, Bifidobacteria spp. fermented food. Moreover, compositional alterations to the gut microbiota accompanied by changes in immune or metabolic factors in the host indicate that microbiota can serve as a mediators between health outcomes and consumption of fermented food [39]. An aqueous extract mixed with L. casei culture was co-encapsulated and mixed with inulin, chitosan, and whey protein isolate, with very high encapsulation efficiency for anthocyanins and lactic bacteria with high efficiency of release in the human intestine [40]. Utilizing onion peel fermented with LAB to produce functional food revealed some potential to regulate blood pressure [41]. Commercially available L. plantarum fermented juices have adequate antioxidants, antimicrobials, and anti-inflammatory properties [42]. Recently, phylogenetic analysis revealed that strains of Lactobacillus fermentum and L. plantarum, with two strains of Pediococcus acidilactici, exhibited different degrees of cell surface hydrophobicity and cholesterol-lowering activity [42]. Consequently, O22, O24, L. rhamnosus K3, and L. plantarum O20 are four tested strains that revealed the ability to induce anti-inflammatory mediators, balancing between regulatory and pro-inflammatory cytokines, with the potential to improve the host immunity [43].
LAB has shown promising antifungal activities. In vitro study of selected L. plantarum LO3 with the maximum antifungal activity was shown to prevent aflatoxin production and fungal growth [44]. The supernatant of this strain showed the ability to inhibit the growth of aflatoxigenic mould in tomato without modifying its physical and chemical properties
The protective effect of milk against cancer with a synergistic combination of Lactobacillus gasseri 505 and melon berry leaf extract that have potential anticancer characteristic against colorectal cancer [45]. L. casei and L. plantarum were utilized as a functional food and ingredient exhibited the most potent anticancer effect on human colon adenocarcinoma by regulating apoptotic proteins [46].Prebiotics are those molecules/fibres which help probiotics grow in the human gut. Mixing Leuconostoc mesenteroides and Lactobacillus sakei in kimchi fermentation produced variable percentages of mannitol, acetic acid, and lactic acid [47]. Fermentation of pasta with a mixed culture of L. plantarum CRL 1964 and L. plantarum CRL 2107 resulted in increased vitamin B2 and B9 levels and haemoglobin in blood [48]. Previously, it was usual that prebiotics and probiotics used separately, but in the last 30 years, a combination between them increased their activity and efficiency [49]. More future research is needed for modulating probiotics, prebiotics, or synbiotics to treat overweight and other metabolic disorders besides the intestinal degradation of non-digestible carbohydrates. Besides that, more in vitro prebiotic expansion studies with model animals are needed to see the changes in their gut microbiota, before clinical trials. For safety and specific targeting, it is advised to involve bioinformatics approaches for precise probing of the bacterial–probiotic mode of function. Complete analysis and full sequencing for the entire genome are necessary with aggressive quality control that should be carried out before introducing the new strains into the market. Future research could include not only prebiotics restricted to Bifidobacteria species which are common in the gut but also expanding to other prebiotics targeting species in the lung and skin. Poor societies should have the chance to obtain probiotics and bacterial fermented food, to reduce the risk of infections, malnutrition, and diabetes. Some probiotics such as L. casei can interact with receptors on the epithelial cells, stimulating the production of cytokines which play an important role in immune cell activation. Due to the powerful capabilities of probiotic bacteria in various body systems and immune system stimulation, competition with pathogens may direct future studies for examining the probiotics activity with focus on boosting immune system.
3. Nutraceuticals from Fungal Origin
Certain types of fungi are used as traditional medicine across various parts of the world. For example, in Africa, fungi are consumed as food and medicine, for recreational purposes, and for performing certain rituals [50]. Some popular fungi used by the Chinese in traditional medicine include the Chinese Cordyceps [51] and Ganoderma lucidum [52]. Ganoderma lucidum has been touted as having anti-ageing properties in addition to promoting other health benefits, and it is even named as “mannentake” in Japanese, which can be translated into “10,000-year-old mushroom” [52].
In recent years, modern science has uncovered fungi as rich sources of nutraceuticals [53]. Mushrooms are rich in various types of bioactive compounds with functional properties as antioxidants [54,55], anticancer compounds [56], antibiotics [57], immunomodulators [58], hypocholesterolemics [59], and hypoglycemics [60]. The Cordyceps fungi that were used as traditional medicine contain many bioactive molecules, with cordycepin considered the most important due to its beneficial potentials [61]. Cordycepin is known for various nutraceutical abilities, such as fighting diabetic, cancer cardiovascular-related diseases which makes it a valued remedial mushroom [62]. Other fungi like Ganoderma are usually used as a traditional medicine and are now one of the main components in the nutraceutical and pharmaceutical industries, recently characterised by spectroscopy and mass spectrometry, showing that it has multiple bioactive compounds important for biological reactions [63]. Ganoderma was also shown to be very active in preventing and treating various kidney diseases [64]. A recent study also showed that certain medicinal mushrooms-derived metabolites are potential candidates for easing neurological disorders such as ataxia and mental dysfunction (dementia) [65]. Ophiocordyceps sinensis cultured mycelia showed antibacterial potential when the extracts were tested against a group of pathogenic bacteria. These antibacterial properties highlight the potential utilisation of fungi for use in therapeutics and the nutraceutical industry [66]. One of the common components used in the nutraceutical industry, beta-glucans, is found in many fungal species. Beta-glucan is a natural polysaccharide that can be found in plant cell walls such as barley and oats [67]. It is also present in yeast cells, generally fungal cell walls, and some other microorganisms. The biological activity of this molecule depends on the molecular weight, bond frequency, and changes in structure. It is known to lower cholesterol levels in the blood by binding cholesterol molecules in the intestines [68] and promoting beneficial metabolic effects in high-fat diet individuals [69]. Beta-glucans were also shown to promote apoptosis in certain cancer cell lines [70]. It is also known to be an immunostimulant by binding to specific receptors such as CR3 and Dectin-1, which activates macrophages [71]. Beta-glucan extracted from yeast used as supplements have shown antidiabetic properties, reducing insulin resistance, and regulating gut microbiota in patients, to the extent of potentially ameliorating neuronal damage in brain tissue [72,73]. Apart from polysaccharides such as beta-glucans, other macromolecules found in fungi also contribute to the advantages of fungi as a source for nutraceutical products [74]. Extracellular polysaccharopeptides from Sanghuang mushrooms in Eastern Asia showed antioxidant and anti-inflammatory activities in inhibiting ulcerative colitis (UC) when tested in mice, possessing the potential to be developed as nutraceuticals or incorporated in functional food development [75].
Prospects of Fungi for Novel Nutraceutical Discovery
The vast diversity of fungi on Earth can be taken advantage of as a potential source for novel discoveries. It is reported that many fungal species are yet to be discovered, and only less than 7% are screened for bioactive compounds [76]. Even with fungi residing in residential properties, a crowdsourcing program found fungal strains with anticancer metabolites [77]. Another study that screened more than 10,000 fungi for bioactive compounds found that 1526 of those fungi extracts had certain biological activity against developing zebrafish [78]. Efforts have been made to search for novel fungi living in marine environments [79] and the Antarctic [80], which could lead to new potential fungal nutraceuticals. Recent surveys in the Arctic and Antarctic discovered that potential lichen-forming fungi within the Parmeliaceae family produced a wide array of metabolites and proteins with promising nutraceutical activities [81]. This further enunciates the potential for discovering novel nutraceuticals and bioactive compounds from fungi living in remote or extreme ecological areas. Fungi are also known to adapt rapidly to environmental changes and can produce a variety of secondary metabolites in response to different ecological interactions such as protection from invertebrates; the explanation for this adaptation lies in the complexity of their biochemical pathways and the richness of gene clusters [82]. Future research could highlight nutraceuticals from traditional remedies and allows the modern pharmaceutical/nutraceuticals industry to reintroduce those metabolites as new health supplements [83].
4. Nutraceuticals from the Algal and Cyanobacterial Origin
Seaweed bioactive metabolites have been consumed for many years in different cuisines [84]. Technologies that may improve the production of seaweed molecules are also previously discussed [85]. The brown seaweed polysaccharide fucoidan has immunomodulatory properties by modulating T cells and decreasing cytokines like interleukin 6 (IL6) [86]. A promising functional drink product from the brown seaweed Sargassum binderi tea contains fucoidan exhibits antioxidant activity [87]. Fucoidan from other species like wakame Undaria pinnatifida showed anticancer and antioxidant properties [88], anticoagulant [88], and antioxidant properties [89]. Generally, seaweed is known as a great source of some essential elements like iron and iodine [90]. High concentrations of minerals such as iron, cadmium, and zinc were found in a group of different Mediterranean seaweed species like Gracilaria verrucosa, Codium tomentosum, and Sargassum linifolium with high potential to be used in the nutritional supplement industry [91]. However, the content of those minerals depends not only on the species but also on the geographical distributions and environmental conditions [92]; therefore, the assessment of the content and potential human toxicity should be a routine procedure for those species selected for health supplements and the food processing industry. Brown seaweed Dictyota friabilis showed potential antiviral properties against HIV [93]. Besides Dictoyta spp., Turbinaria decurrens using an in vitro metabolomics study also revealed anti-HIV activity for polysaccharides extracted from these species [94]. Dictyota menstrualis showed inhibition activity against Zika virus in Brazil [95]. This property of antiviral activity of the seaweed extracts/products related to bioactive secondary metabolites has promising applications to fight different viruses, making these different species of seaweeds a target for the supplement/drug development process and thus increasing their values.
Another group of brown seaweeds showed their ability to be a source of functional food and nutraceuticals products such as fucoidan, laminarin, and alginate [96]. Carrageenan production from red/brown seaweed showed promising abilities to contribute as nutraceuticals in many food industry applications [97]. It also shows some medical applications against obesity in mice by interaction and affecting lipid metabolism, which could potentially use carrageenan as healthy additives in the food industry [98]. Alaria esculenta and Saccharina latissima are brown seaweed algal species with high nutritional value for the food and feed industry value [99]. Both species can be a good natural source of iron and iodine in the food industry [100,101]. Pyropia vietnamensis showed a high value of protein content which also could be a good substitute for protein sources for vegetarian and vegan diets [102].
Spirulina or Arthrospira is a blue-green alga considered one of the famous examples of beneficial cyanobacteria, with many products already in the market for many years now [103]. This alga and its extracts are believed to have many functions as antioxidant and anti-inflammatory properties [104]. Many Spirulina products, now available as superfood/herbal supplements to reduce cholesterol concentration by reducing triacylglycerols level [105], through research on rabbit confirm this characteristic [106], while more recent research on a human claim no changes were made after consuming Spirulina on cholesterol profile [107]. More research on human lipid profile behaviour after spirulina consumption is highly recommended. Chlorella, on the other hand, is another group of microalgae that is widely known for producing nutraceutical compounds with potential used in nutraceutical/pharmaceuticals applications [108].
Seaweed, cyanobacteria, and other algal sources of these food supplements and additives draw the attention of researchers pharma/nutraceuticals manufacturers in recent years [109]. Chlorella and Spirulina have been relatively well studied compared to other algae in terms of their antioxidant, anticancer, and anti-hyperlipidemia properties besides being natural sources of many macro- and micro-nutrients. Despite some reports related to a possible side effect of its related products [110], the seaweed industry is expanding tremendously [111]. Algae, and to be specific seaweed, is considered a good source of multivitamins [112]. Saccharina latissima (sugar kelp) for instance is considered a good source of vitamin C in its dried and fermented form [113,114]. Undaria pinnatifida and Porphyra purpurea have high contents of vitamin A besides having the known activity of lowering the bad cholesterol (LDL) and being a good natural source of proteins and carbohydrates [115]. Red seaweeds Gracilaria spp. (Gracilaria edulis and Gracilaria corticata) revealed multivitamin contents including different forms of vitamin B and vitamin C in addition to their content of minerals, polysaccharides, and fibre [116]. Considering the number of market shares of these algae and the increased popularity of their related products, more targeted researches should be encouraged to focus on (1) understanding the algal and cyanobacterial mode of action in exhibiting their nutraceutical properties and clinical verification of the claims related to their health benefits and (2) exploring and identifying more potential bioactive compounds from different underutilized species of algae and cyanobacteria.
5. Nutraceuticals from Plant Origin
Medicinal plants have an ancient history of usage as local remedies for many illnesses. Today, this principle is the same as using those plant molecules as nutraceuticals and pharmaceuticals for human well-being [117]. The nutraceutical industry has continued supply from nature with the vast majority of registered products in the US FDA from natural sources [118]. Traditional plant-based nutraceutical is considered the source of nutritional elements, phytochemicals, and functional food to more than three quarters of the world’s population [119]. The most universally known plant-based nutraceuticals are vitamins [120]. Phytonutrients are considered as a source of a bioactive raw unprocessed compound which can be counted as a primitive form of nutraceuticals [121]. For instance, fruits and vegetables give us a group of nutrients such as vitamins, minerals, dietary fibre, water, and healthful phytochemicals [122]. Amongst known phytochemicals are flavonoids, isothiocyanate, carotenoids, polyphenols, anthocyanidins, allyl sulfides, and catechins [123]. It is believed that grains, fruit, and vegetables contain more than 4000 phytochemicals with potential nutraceutical application [124]. In addition to the macronutrients such as proteins, fats, carbohydrates, necessary for normal metabolism, and plant-based micronutrients such as trace elements, vitamins also have significant importance in health improvement [125,126]. Several nutraceuticals bioactive peptides, vitamins, are proposed to be able to prevent against microbial bowel diseases [127]. Anthocyanidins, for instance, are promising anti-inflammatory and antioxidant flavonoids. A previous study demonstrated that ulcerative colitis patients who were giving an anthocyanidin extract daily for 9 weeks show high improvement [128]. Epigallocatechin gallate (present in green tea, nuts, pomegranates, and berries) has shown anti-inflammatory against microbial bowel disease and gut microbiome revival abilities [129,130]. Resveratrol has also been shown to have a protecting role in gastric inflammation and reduce the progression to colorectal cancer. Few species of Lamiaceae, have been linked with a reduction of inflammatory bowel disease [131]. The anti-inflammatory effect of Lavender was exerted through the nuclear kappa factor signalling pathway [131,132]. Moringa is one of the well-studied plants with a big number of identified phytochemicals, vitamins, minerals (sodium, potassium, magnesium, phosphorus, iron, zinc, copper, calcium, and manganese), proteins, and amino acids [133]. Moringa oleifera leaves contain plenty of antimicrobial molecules natural antioxidant with more moringa based-supplements available in the market [134]. A recent study used in silico-peptide docking with phytochemicals extracted from M. oleifera, proposed that hydroxychloroquine, kaempferol, and anthraquinone can be used in clinical trials as effective antiviral agents against COVID-19 [135]. Basil leave callus on the other hand revealed recently antioxidant activity linked to the production of chicoric acid and rosmarinic acid by the inhibition of reactive oxygen species (ROS) [136]. Cinnamon has been used to relieve many disease complications of humans [137]. Recent works show cinnamon’s ability to produce compounds with root canal disinfection capabilities in dental applications [138]. Recent research proposed antiviral phytochemicals compounds extracted from Cinnamon against the main protease enzyme of COVID-19 and to reveal the in silico activity against the virus [139]. However, in vitro studies have to be achieved performed 1st to build upon such claims [140]. Garlic and its phyto-components strongly inhibit angiotensin-converting enzyme activity as well as alter the production of Prostaglandin E2 and thromboxane to alter the vascular function to control blood pressure [141]. Also, garlic was shown to display strong anti-inflammatory and antioxidant activities thereby lowering the vascular oxidative stress and exhibiting antihypertensive activity [142,143]. Recent investigation on active compounds found in garlic essential oil was identified by mass spectrometry analysis with potential coronavirus resistance specifically against Angiotensin-converting enzyme protein and the main protease PDB6LU7 of COVID-19 [144]. One of the commonly phenolic acids present in a wide variety of plants such as coffee caffeic acid [145]. A recent study indicates that there is a powerful in vitro antagonist activity between human coronavirus NL63 and caffeic acid resulting in stopping the interaction of the virus with human angiotensin-converting enzyme 2 [146]. Research on the antioxidant activity of equisetum debile extracts ethyl acetate extract was suggested as the most attractive ingredients for functional food and nutraceuticals because of the high inhibitory activity against IL-6 secretion, 5α-reductase, and lipid peroxidation inhibition [147]. A study on the plant data revealed the presence of beneficial individual phenolic and sugar compounds. Antioxidants such as caffeicacid, gallic acid, and ρ-coumaric acid were present. Surpassingly, not many nutraceuticals/supplement products developed yet from the plant [148]. Therefore, as high antioxidant and promising nutritional components, Phoenix dactylifera can be well adopted as source of nutraceuticals product which will have respective share of their market value [149]. Citrus sinensis (sweet orange) contains fats, proteins, carbohydrates (soluble sugars) [150]. Other active ingredients include carotenoids, and flavanones and hesperidin [151]. In sweet orange, there more than 60 mg of hesperidin in 100 mL of fruit juice [152]. Recent research showed interestingly antiviral activity for hesperidin against viral protease. Despite coronavirus sequence variations, the main protease backbone structure and active site conformation are conserved, it is proposed that the inhibitory effect of hesperidin previously detected in SARS virus can be confirmed also in SARS-CoV-2 [153].
A study showed that the use of polyphenol extracted from Camellia sinensis (Black tea) results in increased Bacteroidetes compared to Firmicutes in a simulated intestinal microbiota [154]. Cardamom showed anti-inflammatory ability by acting as a cytokines inhibition agent [155,156]. Curcuma longa root (Turmeric) is excessively used as a major condiment in Asian food as well as a traditional therapy in Indian and Chinese traditional medicine. Curcuminoids are metabolized by colonic normal flora, changing bacterial populations and their metabolic activity [157]. Piperine that present in Piper nigrum L. (black pepper) is considered a high bioactive compound with anti-inflammatory, antibacterial, and antioxidant properties, also composed of 40% nutritional fibre; these characteristics make this species a considerable prebiotic candidate [158]. Low molecular weight phenolic metabolism of white bran catalyzed by the contents of gut microbiota, such as Eubacterium spp., Lactobacillus spp., Escherichia coli, Bacteroides spp., and Bifidobacterium spp., which may exhibit health effects on the host [159]. Sago starch is one of the remarkable sources of functional foods, that extracted from Metroxylon sagu (palm) which is considered a Southeast Asia indigenous [160]. It is called “lemantak” in its native form and resistant starches compose about 60% of its contents. In vivo and in vitro studies have proved the significance of sago starch to increase numbers of Bifidobacterium and Lactobacillus spp. with a high potential to develop gut health supplements [161]. The role of resistant starch as a functional food had been demonstrated due to its effects on insulin responses, glycaemic index, and satiety [162]. Lentils are another proposed prebiotic crop that grown abundantly in Asia, especially in India, because 13% of its content is nondigestible carbohydrate [163]. Some plants can be utilized both as whole foods added to the diet with monitoring the potential prebiotic activity and a source of prebiotic compounds. Kiwifruit is considered a rich source of insoluble and soluble fibre, composed of cellulose, polysaccharides, and hemicellulose [164,165]. Many in vivo and in vitro studies detected the capability of the whole, fresh kiwifruit to generate metabolites such as short-chain fatty acids (SCFA) and the modulation of the colonic microbiota [166,167]. Similarly, β-glucan, a nonstarch water-soluble polysaccharide found in cereal plants such as barley and oats, exerts resistance to digestion in the upper mammalian gastrointestinal tract and then is fermented in the large intestine by gut microbiota. β-glucan comprised 2–20% and 3–8% in barley and oats respectively while barley β-glucan contain lower water-soluble fraction (65%) compared with oats (80%) [168]. High solubility levels make oats bran a preferred source of β-glucan for gastrointestinal microbiota fermentation. In vivo and in vitro studies suggest that oat β-glucan could selectively enhance the growth of bifidobacteria and lactobacilli, causing the production of lactic and acetic acids [169]. These characteristics qualify oats as a candidate for prebiotic development [170]. Ginger is a famous plant with its medicinal value that has been used for a long time as a therapeutic plant, metabolites isolate from ginger revealed antioxidant, antimicrobial, and anticancer which were discussed in detail recently [171].
Future Prospects of Nutraceuticals Derived from Plants
Plant-derived molecules have been recognized based on the presence of different biological characteristics which contribute to their positive effect on human health. Classes of phytochemicals such as saponins, flavonoids, alkaloids, tannins, terpenoids, steroids, and glycosides are frequently isolated from different plant organs with an important role in the maintenance of human health hence they can be used in functional food and supplement development. Plant-derived nutraceuticals could be further used to improve human health, treating chronic diseases, and support other human metabolomics activities. The interest in plant nutraceuticals-related research gaining more attention due to the therapeutic effects especially in the pandemic era considering the supplement manufacturer’s interest to replace the synthetic component with natural plant-based molecules due to the high demand for natural products. Increasing research in the field of safety and effectiveness of these novel products encourages further investment in the application and technology. Promising molecular biology technologies such as nutrigenomics, nutriproteomics, and nutrigenetics are highly encouraged in nutraceutical development research. The innovation in plant-based industries technology might also participate in the further progress in developing food products that can enhance optimum human health.
6. Improving the Yields of Nutraceuticals Using Fungal Elicitors
A common issue in the production of natural compounds—nutraceuticals included—is production time and costs. Extraction of nutraceuticals is usually resource-intensive due to multiple solvent extraction processes. This problem can be greatly alleviated by the employment of genetic and metabolic engineering techniques [172]. While plants are also sources of nutraceuticals, it was found that plant cell cultures have increased yields after coculturing with their respective native endophytic fungi. Therefore, besides fungi being the source of the nutraceutical and therapeutical products, fungi can also be used as elicitors that can influence metabolic changes in other organisms for enhancing the yields of the intended target product. For example, the production of paclitaxel increased when plant cell cultures of Taxus chinensis were cocultured with Aspergillus niger [173]. Other fungal elicitors have been studied in the production of other compounds such as glycyrrhizin [174] and ajmalicine [175]. A more recent example related to nutraceutical elicitors is the use of fungal suspension homogenate of the pathogenic fungi, Alternaria panax Whetz. as an elicitor for the mass and rapid production of Panax ginseng [176]. Fungal elicitors have also been studied to increase the P. ginseng root’s growth. In a recent study, Penicillium sp. YJM-2013 activated the expression of transcription factors and by this promoted the increment in ginsenosides production [177]. From this observation, it is important to take into consideration the role of fungi that is not only commonly used as a source of nutraceutical products but also as a mediator in eliciting nutraceutical production in plants.
7. Issues in Nutraceutical Efficacy
Concerns have been raised regarding the efficacy of nutraceuticals. While nutraceuticals are found in many commonly consumed foods, their beneficial effects may not translate to any meaningful effect. This includes nutraceuticals that have been sold as a supplement. Optimising dosage and delivery systems is vital for the efficacy of nutraceuticals. More studies need to be carried out specifically to find out whether long-term, frequent dosing of nutraceuticals has an actual positive health impact. Due to the chemical complexity of those supplements, it can be difficult to pinpoint the exact nutrient responsible for the acclaimed health benefits. It is also possible that certain health benefits are garnered not by one single nutrient but by a group of nutrients with intertwining interactions.
8. Conclusions and Future Perspective of Nutraceuticals Development
The rapid population growth and the growth in health challenges due to the rising of different diseases made the availability of health-related nutraceuticals or supplements from natural sources an urgent necessity. The need for nutraceuticals may further increase with the changing in the demographic with an elevated trend in an ageing population which will lead to higher incidences of chronic diseases. It is also projected that the workforce will consist of more and more people above the age of 60 [178]. Common diseases associated with ageing are hypertension, congestive heart failure, dementia, osteoporosis, breathing problems, cataract, diabetes, and pathologies associated with decreased immunity [179]. Invertebrate models have shown that certain nutraceuticals can be beneficial in promoting healthy ageing, reducing cancer incidence, and reducing inflammation [180]. The field of nutraceuticals can be advanced from many perspectives. Production-wise, the costs and time can be greatly reduced through enzyme engineering, coupled with gene/protein discovery and characterisation to avoid purely synthetic supplement manufacturing [181]. The advancement in DNA sequencing and gene-editing technologies also opens up avenues for biosynthetic pathway engineering, reducing the number of steps for the synthesis of nutraceuticals [182]. For peptide- and protein-based nutraceuticals, better artificial intelligence and deep-learning technologies can help better understand protein-folding mechanisms in engineering more potent nutraceuticals [183]. Increasing the bioavailability of nutraceuticals is also an active area of research. Due to the diverse chemical nature of nutraceuticals, their bioavailability profiles differ due to the difference in composition and chemical structure [184]. Developing better delivery systems for nutraceuticals to survive the harsh acidic environments of the stomach and to increase absorption through the intestinal wall is one way to increase bioavailability. Specialised nanodelivery systems have been used for the delivery of nutraceuticals for cancer treatment [185]. These function by increasing the cellular uptake of nutraceuticals through mediated endocytosis and other similar mechanisms [186].
Author Contributions
Conceptualization, J.R.A.-O.; writing—original draft preparation J.R.A.-O., E.I.A.-K., K.H.A., B.S.A.-T., D.H.-S.W., H.H., N.N.J. and N.S.A.; figures conceptualization and preparation, J.R.A.-O. and E.I.A.-K. All authors have read and agreed to the published version of the manuscript.
Funding
This work received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- DeFelice, S.L. The nutraceutical revolution: Its impact on food industry R&D. Trends Food Sci. Technol. 1995, 6, 59–61. [Google Scholar] [CrossRef]
- Borodina, I.; Kenny, L.C.; McCarthy, C.M.; Paramasivan, K.; Pretorius, E.; Roberts, T.J.; van der Hoek, S.A.; Kell, D.B. The biology of ergothioneine, an antioxidant nutraceutical. Nutr. Res. Rev. 2020, 33, 190–217. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R. Nutraceuticals and nutraceutical supplementation criteria in cancer: A literature survey. Open Nutraceuticals J. 2009, 2, 92–106. [Google Scholar] [CrossRef]
- Shao, A.; Drewnowski, A.; Willcox, D.; Krämer, L.; Lausted, C.; Eggersdorfer, M.; Mathers, J.; Bell, J.; Randolph, R.; Witkamp, R. Optimal nutrition and the ever-changing dietary landscape: A conference report. Eur. J. Nutr. 2017, 56, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J. Nutraceuticals, nutritional therapy, phytonutrients, and phytotherapy for improvement of human health: A perspective on plant biotechnology application. Recent Pat. Biotechnol. 2007, 1, 75–97. [Google Scholar] [CrossRef] [PubMed]
- Santini, A.; Cammarata, S.M.; Capone, G.; Ianaro, A.; Tenore, G.C.; Pani, L.; Novellino, E. Nutraceuticals: Opening the debate for a regulatory framework. Br. J. Clin. Pharmacol. 2018, 84, 659–672. [Google Scholar] [CrossRef]
- Elhassan, G.; Khan, M.; Khalilullah, H.; Eid, E. Nutraceuticals–an opportunity of healthcare: A review. Trop. J.Pharm.Res. 2016, 5, 229–239. [Google Scholar]
- Xiang, H.; Sun-Waterhouse, D.; Waterhouse, G.I.; Cui, C.; Ruan, Z. Fermentation-enabled wellness foods: A fresh perspective. Food. Sci. Hum. Wellness 2019, 8, 203–243. [Google Scholar] [CrossRef]
- Chanda, S.; Tiwari, R.K.; Kumar, A.; Singh, K. Nutraceuticals Inspiring the Current Therapy for Lifestyle Diseases. Adv. Pharmacol. Pharm. Sci. 2019, 2019, 6908716. [Google Scholar] [CrossRef]
- Pandey, N.; Meena, R.; Rai, S.; Rai, S.P. Medicinal plants derived nutraceuticals: A Re-emerging health aid. Int. J. Pharma Bio Sci. 2011, 2, 420–441. [Google Scholar]
- Meštrović, T. What Are Nutraceuticals? Available online: https://www.news-medical.net/health/What-are-Nutraceuticals.aspx (accessed on 18 July 2020).
- Das, L.; Bhaumik, E.; Raychaudhuri, U.; Chakraborty, R. Role of nutraceuticals in human health. J. Food Sci. Technol. 2012, 49, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Bull, E.; Rapport, L.; Lockwood, B. Nutraceuticals: 1. What is a nutraceutical? Pharm. J. 2000, 265, 57–58. [Google Scholar]
- Torabally, N.B.; Rahmanpoor, H.A. Nutraceuticals: Nutritionally Functional Foods –an Overview. Biomed. J. Sci. Tech. Res. 2019, 15, 1–3. [Google Scholar]
- Market Data Forecast. Nutraceuticals Market by Application (Functional Food, Functional Beverages, Dietary Supplements, Personal Care, Animal Nutrition and Others), and Region—Global Forecast to 2025. Available online: https://www.marketsandmarkets.com/Market-Reports/nutraceutical-ingredient-market-1319.html (accessed on 20 July 2020).
- Gupta, R.C. Introduction. In Nutraceuticals; Gupta, R.C., Ed.; Academic Press: Boston, MA, USA, 2016; pp. xv–xvii. [Google Scholar] [CrossRef]
- Tapsell, L.C.; Neale, E.P.; Satija, A.; Hu, F.B. Foods, Nutrients, and Dietary Patterns: Interconnections and Implications for Dietary Guidelines. Adv. Nutr. 2016, 7, 445–454. [Google Scholar] [CrossRef]
- Kehinde, B.A.; Panghal, A.; Garg, M.K.; Sharma, P.; Chhikara, N. Vegetable milk as probiotic and prebiotic foods. In Advances in Food and Nutrition Research; Academic Press: Cambridge, MA, USA, 2020. [Google Scholar] [CrossRef]
- Zielińska, D.; Ołdak, A.; Rzepkowska, A.; Zieliński, K. Chapter 6—Enumeration and Identification of Probiotic Bacteria in Food Matrices. In Advances in Biotechnology for Food Industry; Holban, A.M., Grumezescu, A.M., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 167–196. [Google Scholar] [CrossRef]
- Sanders, M.E.; Merenstein, D.; Merrifield, C.A.; Hutkins, R. Probiotics for human use. Nutr. Bull. 2018, 43, 212–225. [Google Scholar] [CrossRef]
- de Brito Alves, J.L.; de Oliveira, Y.; de Sousa, V.P.; de Souza, E.L. Chapter 16—Probiotics for humans: Current status and future prospects. In New and Future Developments in Microbial Biotechnology and Bioengineering; Rastegari, A.A., Yadav, A.N., Yadav, N., Eds.; Elsevier: Asterdam, The Netherlands, 2020; pp. 243–254. [Google Scholar] [CrossRef]
- Frakolaki, G.; Giannou, V.; Kekos, D.; Tzia, C. A review of the microencapsulation techniques for the incorporation of probiotic bacteria in functional foods. Crit. Rev. Food Sci. Nutr. 2020, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Indira, M.; Venkateswarulu, T.C.; Abraham Peele, K.; Nazneen Bobby, M.; Krupanidhi, S. Bioactive molecules of probiotic bacteria and their mechanism of action: A review. 3 Biotech 2019, 9, 306. [Google Scholar] [CrossRef] [PubMed]
- Rubio, A.P.D.; Martínez, J.; Palavecino, M.; Fuentes, F.; López, C.M.S.; Marcilla, A.; Pérez, O.E.; Piuri, M. Transcytosis of Bacillus subtilis extracellular vesicles through an in vitro intestinal epithelial cell model. Sci. Rep. 2020, 10, 3120. [Google Scholar] [CrossRef]
- Lin, Y.-T.J.; Chou, C.-C.; Hsu, C.-Y.S. Effects of Lactobacillus casei Shirota intake on caries risk in children. J. Dent. Sci. 2017, 12, 179–184. [Google Scholar] [CrossRef]
- Angarita-Díaz, M.d.P.; Arias, J.C.; Bedoya-Correa, C.; Cepeda, M.J.; Arboleda, M.F.; Chacón, J.M.; Leal, Y. The effect of commercial functional food with probiotics on microorganisms from early carious lesions. Sci. Rep. 2020, 10, 10732. [Google Scholar] [CrossRef]
- Chandel, D.; Sharma, M.; Chawla, V.; Sachdeva, N.; Shukla, G. Isolation, characterization and identification of antigenotoxic and anticancerous indigenous probiotics and their prophylactic potential in experimental colon carcinogenesis. Sci. Rep. 2019, 9, 14769. [Google Scholar] [CrossRef]
- Hill, D.; Ross, R.P.; Arendt, E.; Stanton, C. Microbiology of yogurt and bio-yogurts containing probiotics and prebiotics. In Yogurt in Health and Disease Prevention; Elsevier: Amsterdam, The Netherlands, 2017; pp. 69–85. [Google Scholar] [CrossRef]
- Savaiano, D.A.; Hutkins, R.W. Yogurt, cultured fermented milk, and health: A systematic review. Nutr. Rev. 2020. [Google Scholar] [CrossRef]
- Kim, H.; Kim, J.-S.; Kim, Y.; Jeong, Y.; Kim, J.-E.; Paek, N.-S.; Kang, C.-H. Antioxidant and Probiotic Properties of Lactobacilli and Bifidobacteria of Human Origins. Biotechnol. Bioprocess Eng. 2020, 25, 421–430. [Google Scholar] [CrossRef]
- Chen, P.; Liu, L.; Zhang, X.; Massounga Bora, A.F.; Li, X.; Zhao, M.; Hao, X.; Wang, Y. Antioxidant activity of Cheddar cheese during its ripening time and after simulated gastrointestinal digestion as affected by probiotic bacteria. Int. J. Food Prop. 2019, 22, 218–229. [Google Scholar] [CrossRef]
- Scheers, N.; Rossander-Hulthen, L.; Torsdottir, I.; Sandberg, A.S. Increased iron bioavailability from lactic-fermented vegetables is likely an effect of promoting the formation of ferric iron (Fe(3+)). Eur. J. Nutr. 2016, 55, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Angural, S.; Rana, M.; Puri, N.; Kondepudi, K.K.; Gupta, N. Phytase producing lactic acid bacteria: Cell factories for enhancing micronutrient bioavailability of phytate rich foods. Trends Food Sci. Technol. 2020, 96, 1–12. [Google Scholar] [CrossRef]
- John, G.K.; Wang, L.; Nanavati, J.; Twose, C.; Singh, R.; Mullin, G. Dietary Alteration of the Gut Microbiome and Its Impact on Weight and Fat Mass: A Systematic Review and Meta-Analysis. Genes 2018, 9, 167. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Tomar, S.; Hati, S. Isolation and identification of phytate-degrading lactobacilli from indian cereal-based fermented milk product-“Raabadi”. Int. J. Fermented Food. 2015, 4, 49–59. [Google Scholar] [CrossRef]
- Forte, C.; Manuali, E.; Abbate, Y.; Papa, P.; Vieceli, L.; Tentellini, M.; Trabalza-Marinucci, M.; Moscati, L. Dietary Lactobacillus acidophilus positively influences growth performance, gut morphology, and gut microbiology in rurally reared chickens. Poult. Sci. 2018, 97, 930–936. [Google Scholar] [CrossRef]
- De Cesare, A.; Sala, C.; Castellani, G.; Astolfi, A.; Indio, V.; Giardini, A.; Manfreda, G. Effect of Lactobacillus acidophilus D2/CSL (CECT 4529) supplementation in drinking water on chicken crop and caeca microbiome. PLoS ONE 2020, 15, e0228338. [Google Scholar] [CrossRef] [PubMed]
- Stacchiotti, V.; Rezzi, S.; Eggersdorfer, M.; Galli, F. Metabolic and functional interplay between gut microbiota and fat-soluble vitamins. Crit. Rev. Food Sci. Nutr. 2020, 1–22. [Google Scholar] [CrossRef]
- Stiemsma, L.T.; Nakamura, R.E.; Nguyen, J.G.; Michels, K.B. Does Consumption of Fermented Foods Modify the Human Gut Microbiota? J. Nutr. 2020, 150, 1680–1692. [Google Scholar] [CrossRef]
- Vasile, M.A.; Milea, Ș.A.; Enachi, E.; Barbu, V.; Cîrciumaru, A.; Bahrim, G.E.; Râpeanu, G.; Stănciuc, N. Functional Enhancement of Bioactives from Black Beans and Lactic Acid Bacteria into an Innovative Food Ingredient by Comicroencapsulation. Food Bioprocess Technol. 2020, 13, 978–987. [Google Scholar] [CrossRef]
- Kimoto-Nira, H.; Ohashi, Y.; Amamiya, M.; Moriya, N.; Ohmori, H.; Sekiyama, Y. Fermentation of onion (Allium cepa L.) peel by lactic acid bacteria for production of functional food. J. Food Meas. Charact. 2020, 14, 142–149. [Google Scholar] [CrossRef]
- Sharma, K.; Attri, S.; Goel, G. Selection and Evaluation of Probiotic and Functional Characteristics of Autochthonous Lactic Acid Bacteria Isolated from Fermented Wheat Flour Dough Babroo. Probiotics Antimicrob. Proteins 2019, 11, 774–784. [Google Scholar] [CrossRef]
- Zielińska, D.; Długosz, E.; Zawistowska-Deniziak, A. Functional Properties of Food Origin Lactobacillus in the Gastrointestinal Ecosystem—In Vitro Study. Probiotics Antimicrob. Proteins 2019, 11, 820–829. [Google Scholar] [CrossRef] [PubMed]
- Marie, K.P.; Ngoufack François, Z.; Edith Marius, F.K.; Ciobotaru, O.; Matei, F.; Cornea, C.P.; Israel-Roming, F. Antifungal Activity of Lactic Acid Bacteria Isolated from Peanuts, Gari, and Orange Fruit Juice against Food Aflatoxigenic Molds. Food Biotechnol. 2018, 32, 237–256. [Google Scholar] [CrossRef]
- Oh, N.S.; Lee, J.Y.; Kim, Y.T.; Kim, S.H.; Lee, J.H. Cancer-protective effect of a synbiotic combination between Lactobacillus gasseri 505 and a Cudrania tricuspidata leaf extract on colitis-associated colorectal cancer. Gut Microbes 2020, 12, 1785803. [Google Scholar] [CrossRef] [PubMed]
- Lizardo, R.C.M.; Cho, H.-D.; Lee, J.-H.; Won, Y.-S.; Seo, K.-I. Extracts of Elaeagnus multiflora Thunb. fruit fermented by lactic acid bacteria inhibit SW480 human colon adenocarcinoma via induction of cell cycle arrest and suppression of metastatic potential. J. Food Sci. 2020. [Google Scholar] [CrossRef]
- Lee, J.-J.; Choi, Y.-J.; Jung Lee, M.; Jin Park, S.; Jin Oh, S.; Yun, Y.-R.; Gi Min, S.; Seo, H.-Y.; Park, S.-H.; Lee, M.-A. Effects of combining two lactic acid bacteria as a starter culture on model kimchi fermentation. Food Res. Int. 2020. [Google Scholar] [CrossRef]
- Carrizo, S.L.; de Moreno de LeBlanc, A.; LeBlanc, J.G.; Rollán, G.C. Quinoa pasta fermented with lactic acid bacteria prevents nutritional deficiencies in mice. Food Res. Int. 2020, 127, 108735. [Google Scholar] [CrossRef]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef] [PubMed]
- Milenge Kamalebo, H.; Nshimba Seya Wa Malale, H.; Masumbuko Ndabaga, C.; Degreef, J.; De Kesel, A. Uses and importance of wild fungi: Traditional knowledge from the Tshopo province in the Democratic Republic of the Congo. J. Ethnobiol. Ethnomed. 2018, 14, 13. [Google Scholar] [CrossRef]
- Paterson, R.R.M. Cordyceps: A traditional Chinese medicine and another fungal therapeutic biofactory? Phytochemistry 2008, 69, 1469–1495. [Google Scholar] [CrossRef] [PubMed]
- Wachtel-Galor, S.; Yuen, J.; Buswell, J.A.; Benzie, I.F. Ganoderma lucidum (Lingzhi or Reishi). In Herbal Medicine: Biomolecular and Clinical Aspects, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2011. [Google Scholar]
- Al-Obaidi, J.R. Proteomics of edible mushrooms: A mini-review. Electrophoresis 2016, 37, 1257–1263. [Google Scholar] [CrossRef] [PubMed]
- Kalaras, M.D.; Richie, J.P.; Calcagnotto, A.; Beelman, R.B. Mushrooms: A rich source of the antioxidants ergothioneine and glutathione. Food Chem. 2017, 233, 429–433. [Google Scholar] [CrossRef]
- Sánchez, C. Bioactives from Mushroom and Their Application. In Food Bioactives: Extraction and Biotechnology Applications; Puri, M., Ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 23–57. [Google Scholar] [CrossRef]
- Ajith, T.A.; Janardhanan, K.K. Indian medicinal mushrooms as a source of antioxidant and antitumor agents. J. Clin. Biochem. Nutr. 2007, 40, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Kaushik, N. Metabolites of endophytic fungi as novel source of biofungicide: A review. Phytochem. Rev. 2012, 11, 507–522. [Google Scholar] [CrossRef]
- Mallard, B.; Leach, D.N.; Wohlmuth, H.; Tiralongo, J. Synergistic immuno-modulatory activity in human macrophages of a medicinal mushroom formulation consisting of Reishi, Shiitake and Maitake. PLoS ONE 2019, 14, e0224740. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-Y.; Chung, S.-I.; Nam, S.-H.; Kang, M.-Y. Cholesterol lowering action and antioxidant status improving efficacy of noodles made from unmarketable oak mushroom (Lentinus edodes) in high cholesterol fed rats. J. Korean Soc. Coast. Ocean Eng. 2009, 52, 207–212. [Google Scholar] [CrossRef]
- Giavasis, I. Bioactive fungal polysaccharides as potential functional ingredients in food and nutraceuticals. Curr. Opin. Biotechnol. 2014, 26, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Soltani, M.; Malek, R.A.; Elmarzugi, N.A.; Mahomoodally, M.F.; Uy, D.; Leng, O.M.; El-Enshasy, H.A. Cordycepin: A Biotherapeutic Molecule from Medicinal Mushroom. In Biology of Macrofungi; Singh, B.P., Lallawmsanga, Passari, A.K., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 319–349. [Google Scholar] [CrossRef]
- Ashraf, S.A.; Elkhalifa, A.E.O.; Siddiqui, A.J.; Patel, M.; Awadelkareem, A.M.; Snoussi, M.; Snoussi, M.; Ashraf, M.S.; Adnan, M.; Hadi, S. Cordycepin for Health and Wellbeing: A Potent Bioactive Metabolite of an Entomopathogenic Medicinal Fungus Cordyceps with Its Nutraceutical and Therapeutic Potential. Molecules 2020, 25, 2735. [Google Scholar] [CrossRef]
- Garuba, T.; Olahan, G.; Lateef, A.; Alaya, R.; Awolowo, M.; Sulyman, A. Proximate Composition and Chemical Profiles of Reishi Mushroom (Ganoderma lucidum (Curt: Fr.) Karst). J. Sci. Res. 2020, 12, 103–110. [Google Scholar] [CrossRef]
- Geng, X.; Zhong, D.; Su, L.; Yang, B. Preventive and Therapeutic Effect of Ganoderma (Lingzhi) on Renal Diseases and Clinical Applications. Adv. Exp. Med. Biol. 2019, 1182, 243–262. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.K.; Ir, R.; Jeewon, R.; Doble, M.; Hyde, K.D.; Kaliappan, I.; Jeyaraman, R.; Reddi, R.N.; Krishnan, J.; Li, M.; et al. A Mechanistic Review on Medicinal Mushrooms-Derived Bioactive Compounds: Potential Mycotherapy Candidates for Alleviating Neurological Disorders. Planta Med. 2020, 86, 1161–1175. [Google Scholar] [CrossRef]
- Kaushik, V.; Arya, A.; Sindhu, A.; Singh, A. Identification, Optimization of Culture Conditions, and Bioactive Potential of Chinese Caterpillar Mushroom Ophiocordyceps sinensis (Ascomycetes) Mycelium Isolated from Fruiting Body. Int. J. Med. Mushrooms 2019, 21, 931–942. [Google Scholar] [CrossRef] [PubMed]
- Ciecierska, A.; Drywień, M.E.; Hamulka, J.; Sadkowski, T. Nutraceutical functions of beta-glucans in human nutrition. Rocz Panstw Zakl Hig 2019, 70, 315–324. [Google Scholar] [CrossRef]
- Rondanelli, M.; Opizzi, A.; Monteferrario, F. The biological activity of beta-glucans. Minerva Med. 2009, 100, 237–245. [Google Scholar]
- de Araújo, T.V.; Andrade, E.F.; Lobato, R.V.; Orlando, D.R.; Gomes, N.F.; de Sousa, R.V.; Zangeronimo, M.G.; Pereira, L.J. Effects of beta-glucans ingestion (Saccharomyces cerevisiae) on metabolism of rats receiving high-fat diet. J. Anim. Physiol. Anim. Nutr. 2017, 101, 349–358. [Google Scholar] [CrossRef]
- Kobayashi, H.; Yoshida, R.; Kanada, Y.; Fukuda, Y.; Yagyu, T.; Inagaki, K.; Kondo, T.; Kurita, N.; Suzuki, M.; Kanayama, N.; et al. Suppressing effects of daily oral supplementation of beta-glucan extracted from Agaricus blazei Murill on spontaneous and peritoneal disseminated metastasis in mouse model. J. Cancer Res. Clin. Oncol. 2005, 131, 527–538. [Google Scholar] [CrossRef]
- Vetvicka, V. Glucan-immunostimulant, adjuvant, potential drug. World J. Clin. Oncol. 2011, 2, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Mengdai, X.; Xiaoxing, M.; Hao, H.; Liegang, L.; Wei, Y. Yeast beta-Glucan effects on gut microbiota for regulating insulin signaling system on alzheimer’s disease. Proc. Nutr. Soc. 2020, 79, E263. [Google Scholar] [CrossRef]
- Xu, M.; Mo, X.; Huang, H.; Chen, X.; Liu, H.; Peng, Z.; Chen, L.; Rong, S.; Yang, W.; Xu, S.; et al. Yeast β-glucan alleviates cognitive deficit by regulating gut microbiota and metabolites in Aβ1–42-induced AD-like mice. Int. J. Biol. Macromol. 2020, 161, 258–270. [Google Scholar] [CrossRef] [PubMed]
- Valverde, M.E.; Hernández-Pérez, T.; Paredes-López, O. Edible mushrooms: Improving human health and promoting quality life. Int. J. Microbiol. 2015, 2015, 376387. [Google Scholar] [CrossRef] [PubMed]
- Zuo, K.; Tang, K.; Liang, Y.; Xu, Y.; Sheng, K.; Kong, X.; Wang, J.; Zhu, F.; Zha, X.; Wang, Y. Purification and antioxidant and anti-Inflammatory activity of extracellular polysaccharopeptide from sanghuang mushroom, Sanghuangporus lonicericola. J. Sci. Food Agric. 2021, 101, 1009–1020. [Google Scholar] [CrossRef] [PubMed]
- Hawksworth, D.L. Fungal diversity and its implications for genetic resource collections. Stud. Mycol. 2004, 50, 9–18. [Google Scholar]
- Du, L.; Robles, A.J.; King, J.B.; Powell, D.R.; Miller, A.N.; Mooberry, S.L.; Cichewicz, R.H. Crowdsourcing Natural Products Discovery to Access Uncharted Dimensions of Fungal Metabolite Diversity. Angew. Chem. Int. Ed. 2014, 53, 804–809. [Google Scholar] [CrossRef] [PubMed]
- Hoeksma, J.; Misset, T.; Wever, C.; Kemmink, J.; Kruijtzer, J.; Versluis, K.; Liskamp, R.M.J.; Boons, G.J.; Heck, A.J.R.; Boekhout, T.; et al. A new perspective on fungal metabolites: Identification of bioactive compounds from fungi using zebrafish embryogenesis as read-out. Sci. Rep. 2019, 9, 17546. [Google Scholar] [CrossRef] [PubMed]
- Raghukumar, C. Marine fungal biotechnology: An ecological perspective. Fungal Divers. 2008, 31, 19–35. [Google Scholar]
- Nichols, D.S.; Sanderson, K.; Buia, A.; Kamp, J.V.D.; Holloway, P.; Bowman, J.P.; Smith, M.; Mancuso, C.; Nichols, P.D.; McMeekin, T.A. Bioprospecting and Biotechnology in Antarctica. The Antarctic: Past, Present and Future. In Proceedings of the Conference Celebrating the 40th Anniversary of the Entry into Force of the Antarctic Treaty on 23rd June 1961, Hobart, Australia, 22 June 2001; pp. 85–103. [Google Scholar]
- González-Burgos, E.; Fernández-Moriano, C.; Gómez-Serranillos, M.P. Current knowledge on Parmelia genus: Ecological interest, phytochemistry, biological activities and therapeutic potential. Phytochemistry 2019, 165, 112051. [Google Scholar] [CrossRef]
- Naranjo-Ortiz, M.A.; Gabaldón, T. Fungal evolution: Cellular, genomic and metabolic complexity. Biol. Rev. 2020, 95, 1198–1232. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Fasoyin, O.; Molnár, I.; Yuquan, X. Secondary metabolites from hypocrealean entomopathogenic fungi: Novel bioactive compounds. Nat. Prod. Rep. 2020, 37. [Google Scholar] [CrossRef]
- Tiwari, B.K.; Troy, D.J. Chapter 1—Seaweed sustainability—food and nonfood applications. In Seaweed Sustainability; Tiwari, B.K., Troy, D.J., Eds.; Academic Press: San Diego, CA, USA, 2015; pp. 1–6. [Google Scholar] [CrossRef]
- Hayes, M. Chapter 14—Seaweeds: A nutraceutical and health food. In Seaweed Sustainability; Tiwari, B.K., Troy, D.J., Eds.; Academic Press: San Diego, CA, USA, 2015; pp. 365–387. [Google Scholar] [CrossRef]
- Tanna, B.; Mishra, A. Nutraceutical Potential of Seaweed Polysaccharides: Structure, Bioactivity, Safety, and Toxicity. Compr. Rev. Food Sci. Food Saf. 2019, 18, 817–831. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.J.; Wan mustapha, W.; Maskat, M. Seaweed Tea: Fucoidan-Rich Functional Food Product Development from Malaysian Brown Seaweed, Sargassum binderi. Sains Malays. 2017, 46, 1573–1579. [Google Scholar] [CrossRef]
- Faggio, C.; Pagano, M.; Morabito, M.; Minicante, S.A.; Arfuso, F.; Genovese, G. In vitro assessment of the effect of Undaria pinnatifida extracts on erythrocytes membrane integrity and blood coagulation parameters of Equus caballus. J. Coast. Life Med. 2014, 2, 614–616. [Google Scholar]
- Neri, T.A.N.; Rohmah, Z.; Ticar, B.F.; Palmos, G.N.; Choi, B.-D. Evaluation of sea mustard (Undaria pinnatifida) sporophylls from South Korea as fucoidan source and its corresponding antioxidant activities. Fish. Aquat. Sci. 2019, 22, 24. [Google Scholar] [CrossRef]
- Mišurcová, L.; Machů, L.; Orsavová, J. Chapter 29—Seaweed Minerals as Nutraceuticals. In Advances in Food and Nutrition Research; Kim, S.-K., Ed.; Academic Press: Boston, MA, USA, 2011; Volume 64, pp. 371–390. [Google Scholar]
- El-Said, G.F.; El-Sikaily, A. Chemical composition of some seaweed from Mediterranean Sea coast, Egypt. Environ. Monit. Assess. 2013, 185, 6089–6099. [Google Scholar] [CrossRef]
- Chen, Q.; Pan, X.-D.; Huang, B.-F.; Han, J.-L. Distribution of metals and metalloids in dried seaweeds and health risk to population in southeastern China. Sci. Rep. 2018, 8, 3578. [Google Scholar] [CrossRef]
- Stephens, P.R.S.; Cirne-Santos, C.C.; de Souza Barros, C.; Teixeira, V.L.; Carneiro, L.A.D.; Amorim, L.d.S.C.; Ocampo, J.S.P.; Castello-Branco, L.R.R.; de Palmer Paixão, I.C.N. Diterpene from marine brown alga Dictyota friabilis as a potential microbicide against HIV-1 in tissue explants. J. Appl. Phycol. 2017, 29, 775–780. [Google Scholar] [CrossRef]
- Sanniyasi, E.; Venkatasubramanian, G.; Anbalagan, M.M.; Raj, P.P.; Gopal, R.K. In vitro anti-HIV-1 activity of the bioactive compound extracted and purified from two different marine macroalgae (seaweeds) (Dictyota bartayesiana J.V.Lamouroux and Turbinaria decurrens Bory). Sci. Rep. 2019, 9, 12185. [Google Scholar] [CrossRef]
- Cirne-Santos, C.C.; Barros, C.d.S.; Gomes, M.W.L.; Gomes, R.; Cavalcanti, D.N.; Obando, J.M.C.; Ramos, C.J.B.; Villaça, R.C.; Teixeira, V.L.; Paixão, I.C.N.d.P. In Vitro Antiviral Activity Against Zika Virus From a Natural Product of the Brazilian Brown Seaweed Dictyota menstrualis. Nat. Prod. Commun. 2019, 14. [Google Scholar] [CrossRef]
- Múzquiz de la Garza, A.R.; Tapia-Salazar, M.; Maldonado-Muñiz, M.; de la Rosa-Millán, J.; Gutiérrez-Uribe, J.A.; Santos-Zea, L.; Barba-Dávila, B.A.; Ricque-Marie, D.; Cruz-Suárez, L.E. Nutraceutical Potential of Five Mexican Brown Seaweeds. BioMed Res. Int. 2019, 2019, 3795160. [Google Scholar] [CrossRef] [PubMed]
- Usuldin, S.R.A.; Al-Obaidi, J.R.; Razali, N.; Junit, S.M.; Ajang, M.J.; Hussin, S.N.I.S.; Hamid, S.S.; Hanafi, N.M.; Roni, A.N.H.M.; Saleh, N.M. Molecular investigation of carrageenan production in Kappaphycus alvarezii in different culture conditions: A proteomic approach. J. Appl. Phycol. 2017, 29, 1989–2001. [Google Scholar] [CrossRef]
- Chin, Y.X.; Mi, Y.; Cao, W.X.; Lim, P.E.; Xue, C.H.; Tang, Q.J. A Pilot Study on Anti-Obesity Mechanisms of Kappaphycus Alvarezii: The Role of Native κ-Carrageenan and the Leftover Sans-Carrageenan Fraction. Nutrients 2019, 11, 1133. [Google Scholar] [CrossRef]
- Stévant, P.; Marfaing, H.; Rustad, T.; Sandbakken, I.; Fleurence, J.; Chapman, A. Nutritional value of the kelps Alaria esculenta and Saccharina latissima and effects of short-term storage on biomass quality. J. Appl. Phycol. 2017, 29, 2417–2426. [Google Scholar] [CrossRef]
- Schiener, P.; Black, K.D.; Stanley, M.S.; Green, D.H. The seasonal variation in the chemical composition of the kelp species Laminaria digitata, Laminaria hyperborea, Saccharina latissima and Alaria esculenta. J. Appl. Phycol. 2015, 27, 363–373. [Google Scholar] [CrossRef]
- Circuncisão, A.R.; Catarino, M.D.; Cardoso, S.M.; Silva, A.M.S. Minerals from Macroalgae Origin: Health Benefits and Risks for Consumers. Mar. Drugs 2018, 16, 400. [Google Scholar] [CrossRef] [PubMed]
- Kavale, M.; Kazi, M.; Bagal, P.; Singh, V.; Behera, D.P. Food value of Pyropia vietnamensis (Bangiales, Rhodophyta) from India. Indian J. Geo-Mar. Sci. 2018, 46, 402–408. [Google Scholar]
- Karkos, P.D.; Leong, S.C.; Karkos, C.D.; Sivaji, N.; Assimakopoulos, D.A. Spirulina in clinical practice: Evidence-based human applications. Evid. Based Complement. Alternat. Med. 2011, 2011, 531053. [Google Scholar] [CrossRef]
- Deng, R.; Chow, T.-J. Hypolipidemic, Antioxidant, and Antiinflammatory Activities of Microalgae Spirulina. Cardiovasc. Ther. 2010, 28, e33–e45. [Google Scholar] [CrossRef]
- Colla, L.M.; Muccillo-Baisch, A.L.; Costa, J.A.V. Spirulina platensis effects on the levels of total cholesterol, HDL and triacylglycerols in rabbits fed with a hypercholesterolemic diet. Braz. Arch. Biol. Technol. 2008, 51, 405–411. [Google Scholar] [CrossRef]
- Cheong, S.H.; Kim, M.Y.; Sok, D.-E.; Hwang, S.-Y.; Kim, J.H.; Kim, H.R.; Lee, J.H.; Kim, Y.-B.; Kim, M.R. Spirulina Prevents Atherosclerosis by Reducing Hypercholesterolemia in Rabbits Fed a High-Cholesterol Diet. J. Nutr. Sci. Vitaminol. 2010, 56, 34–40. [Google Scholar] [CrossRef] [PubMed]
- van den Driessche, J.J.; Plat, J.; Konings, M.C.J.M.; Mensink, R.P. Effects of spirulina and wakame consumption on intestinal cholesterol absorption and serum lipid concentrations in non-hypercholesterolemic adult men and women. Eur. J. Nutr. 2019. [Google Scholar] [CrossRef]
- Silva, J.; Alves, C.; Pinteus, S.; Reboleira, J.; Pedrosa, R.; Bernardino, S. Chapter 3.10—Chlorella. In Nonvitamin and Nonmineral Nutritional Supplements; Nabavi, S.M., Silva, A.S., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 187–193. [Google Scholar] [CrossRef]
- García, J.L.; de Vicente, M.; Galán, B. Microalgae, old sustainable food and fashion nutraceuticals. Microb. Biotechnol. 2017, 10, 1017–1024. [Google Scholar] [CrossRef]
- Weiner, M.L.; McKim, J.M. Comment on “Revisiting the carrageenan controversy: Do we really understand the digestive fate and safety of carrageenan in our foods?” by S. David, CS Levi, L. Fahoum, Y. Ungar, EG Meyron-Holtz, A. Shpigelman and U. Lesmes, Food Funct. 2018, 9, 1344–1352. Food. Funct. 2019, 10, 1760–1762. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I.; Shin, J.H.; Kim, J.D. The promising future of microalgae: Current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb. Cell Fact. 2018, 17, 36. [Google Scholar] [CrossRef] [PubMed]
- Škrovánková, S. Chapter 28—Seaweed Vitamins as Nutraceuticals. In Advances in Food and Nutrition Research; Kim, S.-K., Ed.; Academic Press: Cambridge, MA, USA, 2011; Volume 64, pp. 357–369. [Google Scholar]
- Sappati, P.K.; Nayak, B.; VanWalsum, G.P.; Mulrey, O.T. Combined effects of seasonal variation and drying methods on the physicochemical properties and antioxidant activity of sugar kelp (Saccharina latissima). J. Appl. Phycol. 2019, 31, 1311–1332. [Google Scholar] [CrossRef]
- Bruhn, A.; Brynning, G.; Johansen, A.; Lindegaard, M.S.; Sveigaard, H.H.; Aarup, B.; Fonager, L.; Andersen, L.L.; Rasmussen, M.B.; Larsen, M.M.; et al. Fermentation of sugar kelp (Saccharina latissima)—effects on sensory properties, and content of minerals and metals. J. Appl. Phycol. 2019, 31, 3175–3187. [Google Scholar] [CrossRef]
- Taboada, M.C.; Millán, R.; Miguez, M.I. Nutritional value of the marine algae wakame (Undaria pinnatifida) and nori (Porphyra purpurea) as food supplements. J. Appl. Phycol. 2013, 25, 1271–1276. [Google Scholar] [CrossRef]
- Rosemary, T.; Arulkumar, A.; Paramasivam, S.; Mondragon-Portocarrero, A.; Miranda, J.M. Biochemical, micronutrient and physicochemical properties of the dried red seaweeds Gracilaria edulis and Gracilaria corticata. Molecules 2019, 24, 2225. [Google Scholar] [CrossRef]
- Howes, M.-J.R.; Perry, N.S.L.; Vásquez-Londoño, C.; Perry, E.K. Role of phytochemicals as nutraceuticals for cognitive functions affected in ageing. Br. J. Pharmacol. 2020, 177, 1294–1315. [Google Scholar] [CrossRef] [PubMed]
- Thomford, N.E.; Senthebane, D.A.; Rowe, A.; Munro, D.; Seele, P.; Maroyi, A.; Dzobo, K. Natural Products for Drug Discovery in the 21st Century: Innovations for Novel Drug Discovery. Int. J. Mol. Sci. 2018, 19, 1578. [Google Scholar] [CrossRef]
- Blades, M.; Kasbia, G.S. Functional foods and nutraceuticals in the management of obesity. Nutr. Food Sci. 2005, 35, 344–352. [Google Scholar]
- Stipanuk, M.H.; Caudill, M.A. Biochemical, Physiological, and Molecular Aspects of Human Nutrition-E-Book; Elsevier Health Science: Amsterdan, The Netherlands, 2018. [Google Scholar]
- Khan, M.A. Study of Phyto-Nutrients from Apricot and In Vivo Assessment of Their Bioactivity. Ph.D. Thesis, PMAS-Arid Agriculture University, Rawalpindi, Pakistan, 2019. [Google Scholar]
- Sarker, U.; Oba, S.; Daramy, M.A. Nutrients, minerals, antioxidant pigments and phytochemicals, and antioxidant capacity of the leaves of stem amaranth. Sci. Rep. 2020, 10, 3892. [Google Scholar] [CrossRef] [PubMed]
- Septembre-Malaterre, A.; Remize, F.; Poucheret, P. Fruits and vegetables, as a source of nutritional compounds and phytochemicals: Changes in bioactive compounds during lactic fermentation. Food Res. Int. 2018, 104, 86–99. [Google Scholar] [CrossRef]
- Alamgir, A. Therapeutic Use of Medicinal Plants and Their Extracts; Springer: New York, NY, USA, 2018; Volume 1. [Google Scholar]
- Cena, H.; Calder, P.C. Defining a healthy diet: Evidence for the role of contemporary dietary patterns in health and disease. Nutrients 2020, 12, 334. [Google Scholar] [CrossRef]
- Al-Obaidi, J.R. Micro- and Macronutrient Signalling in Plant Cells: A Proteomic Standpoint Under Stress Conditions. In Plant Micronutrients: Deficiency and Toxicity Management; Aftab, T., Hakeem, K.R., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 241–255. [Google Scholar] [CrossRef]
- Uranga, J.A.; López-Miranda, V.; Lombó, F.; Abalo, R. Food, nutrients and nutraceuticals affecting the course of inflammatory bowel disease. Pharmacol. Rep. 2016, 68, 816–826. [Google Scholar] [CrossRef]
- Biedermann, L.; Mwinyi, J.; Scharl, M.; Frei, P.; Zeitz, J.; Kullak-Ublick, G.A.; Vavricka, S.R.; Fried, M.; Weber, A.; Humpf, H.U.; et al. Bilberry ingestion improves disease activity in mild to moderate ulcerative colitis—An open pilot study. J. Crohns Colitis 2013, 7, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Martin, H.; Burgess, E.J.; Smith, W.A.; McGhie, T.K.; Cooney, J.M.; Lunken, R.C.; de Guzman, E.; Trower, T.; Perry, N.B. JAK2 and AMP-kinase inhibition in vitro by food extracts, fractions and purified phytochemicals. Food. Funct. 2015, 6, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, L.R. Dietary interactions with the bacterial sensing machinery in the intestine: The plant polyphenol case. Front. Genet. 2014, 5, 64. [Google Scholar]
- Algieri, F.; Rodriguez-Nogales, A.; Vezza, T.; Garrido-Mesa, J.; Garrido-Mesa, N.; Utrilla, M.P.; González-Tejero, M.R.; Casares-Porcel, M.; Molero-Mesa, J.; del Mar Contreras, M. Anti-inflammatory activity of hydroalcoholic extracts of Lavandula dentata L. and Lavandula stoechas L. J. Ethnopharmacol. 2016, 190, 142–158. [Google Scholar] [CrossRef] [PubMed]
- Rufino, A.T.; Ferreira, I.; Judas, F.; Salgueiro, L.; Lopes, M.C.; Cavaleiro, C.; Mendes, A.F. Differential effects of the essential oils of Lavandula luisieri and Eryngium duriaei subsp. juresianum in cell models of two chronic inflammatory diseases. Pharm. Biol. 2015, 53, 1220–1230. [Google Scholar] [CrossRef] [PubMed]
- Vergara-Jimenez, M.; Almatrafi, M.M.; Fernandez, M.L. Bioactive components in Moringa oleifera leaves protect against chronic disease. Antioxidants 2017, 6, 91. [Google Scholar] [CrossRef]
- El Sohaimy, S.A.; Hamad, G.M.; Mohamed, S.E.; Amar, M.H.; Al-Hindi, R.R. Biochemical and functional properties of Moringa oleifera leaves and their potential as a functional food. Glob. Adv. Res. J. Agric. Sci. 2015, 4, 188–199. [Google Scholar]
- Hamza, M.; Ali, A.; Khan, S.; Ahmed, S.; Attique, Z.; Ur Rehman, S.; Khan, A.; Ali, H.; Rizwan, M.; Munir, A. nCOV-19 peptides mass fingerprinting identification, binding, and blocking of inhibitors flavonoids and anthraquinone of Moringa oleifera and hydroxychloroquine. J. Biomol. Struct. Dyn. 2020, 1–11. [Google Scholar] [CrossRef]
- Nazir, S.; Jan, H.; Tungmunnithum, D.; Drouet, S.; Zia, M.; Hano, C.; Abbasi, B.H. Callus Culture of Thai Basil Is an Effective Biological System for the Production of Antioxidants. Molecules 2020, 25, 4859. [Google Scholar] [CrossRef]
- Ose, R.; Tu, J.; Schink, A.; Maxeiner, J.; Schuster, P.; Lucas, K.; Saloga, J.; Bellinghausen, I. Cinnamon extract inhibits allergen-specific immune responses in human and murine allergy models. Clin. Exp. Allergy 2020, 50, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Marcoux, E.; Lagha, A.B.; Gauthier, P.; Grenier, D. Antimicrobial activities of natural plant compounds against endodontic pathogens and biocompatibility with human gingival fibroblasts. Arch. Oral Biol. 2020, 116, 104734. [Google Scholar] [CrossRef]
- Prasanth, D.; Murahari, M.; Chandramohan, V.; Panda, S.P.; Atmakuri, L.R.; Guntupalli, C. In silico identification of potential inhibitors from Cinnamon against main protease and spike glycoprotein of SARS CoV-2. J. Biomol. Struct. Dyn. 2020, 1–15. [Google Scholar] [CrossRef]
- Cicero, A.F.; Ferroni, A.; Ertek, S. Tolerability and safety of commonly used dietary supplements and nutraceuticals with lipid-lowering effects. Expert Opin. Drug Saf. 2012, 11, 753–766. [Google Scholar] [CrossRef]
- Chiu, H.-F.; Venkatakrishnan, K.; Wang, C.-K. Nutraceuticals and functional foods in the prevention of hypertension induced by excessive intake of dietary salt. In Dietary Sugar, Salt and Fat in Human Health; Elsevier: Amsterdam, The Netherlands, 2020; pp. 423–450. [Google Scholar]
- Ried, K.; Fakler, P. Potential of garlic (Allium sativum) in lowering high blood pressure: Mechanisms of action and clinical relevance. Integr. Blood Press. Control 2014, 7, 71–82. [Google Scholar] [CrossRef]
- Shouk, R.; Abdou, A.; Shetty, K.; Sarkar, D.; Eid, A.H. Mechanisms underlying the antihypertensive effects of garlic bioactives. Nutr. Res. 2014, 34, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Thuy, B.T.P.; My, T.T.A.; Hai, N.T.T.; Hieu, L.T.; Hoa, T.T.; Thi Phuong Loan, H.; Triet, N.T.; Anh, T.T.V.; Quy, P.T.; Tat, P.V. Investigation into SARS-CoV-2 resistance of compounds in garlic essential oil. ACS Omega 2020, 5, 8312–8320. [Google Scholar] [CrossRef] [PubMed]
- Meinhart, A.D.; Damin, F.M.; Caldeirão, L.; de Jesus Filho, M.; da Silva, L.C.; da Silva Constant, L.; Filho, J.T.; Wagner, R.; Godoy, H.T. Chlorogenic and caffeic acids in 64 fruits consumed in Brazil. Food Chem. 2019, 286, 51–63. [Google Scholar] [CrossRef]
- Weng, J.-R.; Lin, C.-S.; Lai, H.-C.; Lin, Y.-P.; Wang, C.-Y.; Tsai, Y.-C.; Wu, K.-C.; Huang, S.-H.; Lin, C.-W. Antiviral activity of Sambucus FormosanaNakai ethanol extract and related phenolic acid constituents against human coronavirus NL63. Virus Res. 2019, 273, 197767. [Google Scholar] [CrossRef]
- Chaiyana, W.; Punyoyai, C.; Somwongin, S.; Leelapornpisid, P.; Ingkaninan, K.; Waranuch, N.; Srivilai, J.; Thitipramote, N.; Wisuitiprot, W.; Schuster, R. Inhibition of 5α-reductase, IL-6 secretion, and oxidation process of Equisetum debile Roxb. ex vaucher extract as functional food and nutraceuticals ingredients. Nutrients 2017, 9, 1105. [Google Scholar] [CrossRef] [PubMed]
- Younas, A.; Naqvi, S.A.; Khan, M.R.; Shabbir, M.A.; Jatoi, M.A.; Anwar, F.; Inam-Ur-Raheem, M.; Saari, N.; Aadil, R.M. Functional food and nutra-pharmaceutical perspectives of date (Phoenix dactylifera L.) fruit. J. Food Biochem. 2020, 44, e13332. [Google Scholar] [CrossRef]
- Siddiqi, S.A.; Rahman, S.; Khan, M.M.; Rafiq, S.; Inayat, A.; Khurram, M.S.; Seerangurayar, T.; Jamil, F. Potential of dates (Phoenix dactylifera L.) as natural antioxidant source and functional food for healthy diet. Sci. Total Environ. 2020, 748, 141234. [Google Scholar] [CrossRef] [PubMed]
- Rampersaud, G.C.; Valim, M.F. 100% citrus juice: Nutritional contribution, dietary benefits, and association with anthropometric measures. Crit. Rev. Food Sci. Nutr. 2017, 57, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Chakravarty, T.; Chen, P.; Akhmerov, A.; Falk, J.; Friedman, O.; Zaman, T.; Ebinger, J.E.; Gheorghiu, M.; Marbán, L.; et al. Allogeneic cardiosphere-derived cells (CAP-1002) in critically ill COVID-19 patients: Compassionate-use case series. Basic Res. Cardiol. 2020, 115, 36. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-W.; Tsai, F.-J.; Tsai, C.-H.; Lai, C.-C.; Wan, L.; Ho, T.-Y.; Hsieh, C.-C.; Chao, P.-D.L. Anti-SARS coronavirus 3C-like protease effects of Isatis indigotica root and plant-derived phenolic compounds. Antivir. Res. 2005, 68, 36–42. [Google Scholar] [CrossRef]
- Joshi, R.S.; Jagdale, S.S.; Bansode, S.B.; Shankar, S.S.; Tellis, M.B.; Pandya, V.K.; Chugh, A.; Giri, A.P.; Kulkarni, M.J. Discovery of potential multi-target-directed ligands by targeting host-specific SARS-CoV-2 structurally conserved main protease. J. Biomol. Struct. Dyn. 2020, 1–16. [Google Scholar] [CrossRef]
- Kemperman, R.A.; Gross, G.; Mondot, S.; Possemiers, S.; Marzorati, M.; Van de Wiele, T.; Doré, J.; Vaughan, E.E. Impact of polyphenols from black tea and red wine/grape juice on a gut model microbiome. Food Res. Int. 2013, 53, 659–669. [Google Scholar] [CrossRef]
- Kandikattu, H.K.; Rachitha, P.; Jayashree, G.V.; Krupashree, K.; Sukhith, M.; Majid, A.; Amruta, N.; Khanum, F. Anti-inflammatory and anti-oxidant effects of Cardamom (Elettaria repens (Sonn.) Baill) and its phytochemical analysis by 4D GCXGC TOF-MS. Biomed. Pharmacother. 2017, 91, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Souissi, M.; Azelmat, J.; Chaieb, K.; Grenier, D. Antibacterial and anti-inflammatory activities of cardamom (Elettaria cardamomum) extracts: Potential therapeutic benefits for periodontal infections. Anaerobe 2020, 61, 102089. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, R.S.; Hussain, M.B.; Sultan, M.T.; Arshad, M.S.; Waheed, M.; Shariati, M.A.; Plygun, S.; Hashempur, M.H. Biochemistry, Safety, Pharmacological Activities, and Clinical Applications of Turmeric: A Mechanistic Review. Evid. Based Complement. Alternat. Med. 2020, 2020, 7656919. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.Y.; Summanen, P.H.; Lee, R.P.; Huang, J.; Henning, S.M.; Heber, D.; Finegold, S.M.; Li, Z. Prebiotic potential and chemical composition of seven culinary spice extracts. J. Food Sci. 2017, 82, 1807–1813. [Google Scholar] [CrossRef]
- Duncan, S.H.; Russell, W.R.; Quartieri, A.; Rossi, M.; Parkhill, J.; Walker, A.W.; Flint, H.J. Wheat bran promotes enrichment within the human colonic microbiota of butyrate-producing bacteria that release ferulic acid. Environ. Microbiol. 2016, 18, 2214–2225. [Google Scholar] [CrossRef]
- Hussain, H.; Mustafa Kamal, M.; Al-Obaidi, J.R.; Hamdin, N.E.; Ngaini, Z.; Mohd-Yusuf, Y. Proteomics of Sago Palm Towards Identifying Contributory Proteins in Stress-Tolerant Cultivar. Protein J. 2020, 39, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Arshad, N.; Zaman, S.; Rawi, M.; Sarbini, S. Resistant starch evaluation and in vitro fermentation of lemantak (native sago starch), for prebiotic assessment. Int. Food Res. J. 2018, 25, 951–957. [Google Scholar]
- Zaman, S.A.; Sarbini, S.R. The potential of resistant starch as a prebiotic. Crit. Rev. Biotechnol. 2016, 36, 578–584. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.E.; Sjövall, H.; Hansson, G.C. The gastrointestinal mucus system in health and disease. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Carnachan, S.M.; Bootten, T.J.; Mishra, S.; Monro, J.A.; Sims, I.M. Effects of simulated digestion in vitro on cell wall polysaccharides from kiwifruit (Actinidia spp.). Food Chem. 2012, 133, 132–139. [Google Scholar] [CrossRef]
- Mulcahy, E. Preparation, Characterisation and Functional Applications of Whey Protein-Carbohydrate Conjugates as Food Ingredients. Ph.D. Thesis, University College Cork, Cork, Ireland, 2017. [Google Scholar]
- Paturi, G.; Butts, C.A.; Bentley-Hewitt, K.L.; Ansell, J. Influence of Green and Gold Kiwifruit on Indices of Large Bowel Function in Healthy Rats. J. Food Sci. 2014, 79, H1611–H1620. [Google Scholar] [CrossRef] [PubMed]
- Blatchford, P.; Stoklosinski, H.; Walton, G.; Swann, J.; Gibson, G.; Gearry, R.; Ansell, J. Kiwifruit fermentation drives positive gut microbial and metabolic changes irrespective of initial microbiota composition. Bioact. Carbohydr. Diet. Fibre 2015, 6, 37–45. [Google Scholar] [CrossRef]
- El Khoury, D.; Cuda, C.; Luhovyy, B.L.; Anderson, G.H. Beta Glucan: Health Benefits in Obesity and Metabolic Syndrome. J. Nutr. Metab. 2012, 2012, 851362. [Google Scholar] [CrossRef]
- Metzler-Zebeli, B.U.; Zijlstra, R.T.; Mosenthin, R.; Gänzle, M.G. Dietary calcium phosphate content and oat β-glucan influence gastrointestinal microbiota, butyrate-producing bacteria and butyrate fermentation in weaned pigs. FEMS Microbiol. Ecol. 2011, 75, 402–413. [Google Scholar] [CrossRef] [PubMed]
- Marotti, I.; Bregola, V.; Aloisio, I.; Di Gioia, D.; Bosi, S.; Di Silvestro, R.; Quinn, R.; Dinelli, G. Prebiotic effect of soluble fibres from modern and old durum-type wheat varieties on Lactobacillus and Bifidobacterium strains. J. Sci. Food Agric. 2012, 92, 2133–2140. [Google Scholar] [CrossRef]
- Mao, Q.-Q.; Xu, X.-Y.; Cao, S.-Y.; Gan, R.-Y.; Corke, H.; Beta, T.; Li, H.-B. Bioactive Compounds and Bioactivities of Ginger (Zingiber officinale Roscoe). Foods 2019, 8, 185. [Google Scholar] [CrossRef]
- Liu, L.; Guan, N.; Li, J.; Shin, H.D.; Du, G.; Chen, J. Development of GRAS strains for nutraceutical production using systems and synthetic biology approaches: Advances and prospects. Crit. Rev. Biotechnol. 2017, 37, 139–150. [Google Scholar] [CrossRef]
- Wang, C.; Wu, J.; Mei, X. Enhancement of Taxol production and excretion in Taxus chinensis cell culture by fungal elicitation and medium renewal. Appl. Microbiol. Biotechnol. 2001, 55, 404–410. [Google Scholar] [CrossRef] [PubMed]
- Karwasara, V.S.; Jain, R.; Tomar, P.; Dixit, V. Elicitation as yield enhancement strategy for glycyrrhizin production by cell cultures of Abrus precatorius Linn. In Vitro Cell. Dev. Biol. Plant 2010, 46, 354–362. [Google Scholar] [CrossRef]
- Namdeo, A.; Patil, S.; Fulzele, D.P. Influence of fungal elicitors on production of ajmalicine by cell cultures of Catharanthus roseus. Biotechnol. Prog. 2002, 18, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.-J.; An, X.-L.; Sun, H.-D.; Piao, X.-C.; Gao, R.; Lian, M.-L. Ginsenoside synthesis of adventitious roots in Panax ginseng is promoted by fungal suspension homogenate of Alternaria panax and regulated by several signaling molecules. Ind. Crops Prod. 2020, 150, 112414. [Google Scholar] [CrossRef]
- Wang, S.-h.; Liang, W.-x.; Lu, J.; Yao, L.; Wang, J.; Gao, W.-y. Penicillium sp. YJM-2013 induces ginsenosides biosynthesis in Panax ginseng adventitious roots by inducing plant resistance responses. Chin. Herb. Med. 2020, 12, 257–264. [Google Scholar] [CrossRef]
- Burtless, G. The Impact of Population Aging and Delayed Retirement on Workforce Productivity. 2013. Available online: https://crr.bc.edu/working-papers/the-impact-of-population-aging-and-delayed-retirement-on-workforce-productivity/ (accessed on 20 July 2020).
- Gupta, C.; Prakash, D. Nutraceuticals for geriatrics. J. Tradit. Complement. Med. 2014, 5, 5–14. [Google Scholar] [CrossRef]
- Dong, Y.; Guha, S.; Sun, X.; Cao, M.; Wang, X.; Zou, S. Nutraceutical Interventions for Promoting Healthy Aging in Invertebrate Models. Oxid. Med. Cell. Longev. 2012, 2012, 718491. [Google Scholar] [CrossRef] [PubMed]
- Wiltschi, B.; Cernava, T.; Dennig, A.; Galindo Casas, M.; Geier, M.; Gruber, S.; Haberbauer, M.; Heidinger, P.; Herrero Acero, E.; Kratzer, R.; et al. Enzymes revolutionize the bioproduction of value-added compounds: From enzyme discovery to special applications. Biotechnol. Adv. 2020, 40, 107520. [Google Scholar] [CrossRef]
- Goss, R.J.; Shankar, S.; Fayad, A.A. The generation of “unnatural” products: Synthetic biology meets synthetic chemistry. Nat. Prod. Rep. 2012, 29, 870–889. [Google Scholar] [CrossRef]
- Campos, K.R.; Coleman, P.J.; Alvarez, J.C.; Dreher, S.D.; Garbaccio, R.M.; Terrett, N.K.; Tillyer, R.D.; Truppo, M.D.; Parmee, E.R. The importance of synthetic chemistry in the pharmaceutical industry. Science 2019, 363. [Google Scholar] [CrossRef]
- Santini, A.; Tenore, G.C.; Novellino, E. Nutraceuticals: A paradigm of proactive medicine. Eur. J. Pharm. Sci. 2017, 96, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Nair, H.B.; Sung, B.; Yadav, V.R.; Kannappan, R.; Chaturvedi, M.M.; Aggarwal, B.B. Delivery of antiinflammatory nutraceuticals by nanoparticles for the prevention and treatment of cancer. Biochem. Pharmacol. 2010, 80, 1833–1843. [Google Scholar] [CrossRef] [PubMed]
- Dima, C.; Assadpour, E.; Dima, S.; Jafari, S.M. Nutraceutical nanodelivery; an insight into the bioaccessibility/bioavailability of different bioactive compounds loaded within nanocarriers. Crit. Rev. Food Sci. Nutr. 2020, 1–35. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).