Abstract
Environmental health is at the intersection between health and the environment. However, it still has a recent (and narrow) history as a scientific area, mainly addressing human biomonitoring and toxicological issues. Only recently additional environmental ‘layers’, other than the traditional chemical, biological and physical environmental determinants, have been considered. This broader perspective of environmental health also encompasses digital, psychosocial, political, socioeconomic and cultural determinants, all of them relevant when considering human health from a planetary health paradigm. This reflects the progressive adoption of a systemic perspective regarding the impact of gains for human health and well-being towards a sustainable environment. It also implies a multi-method and participatory approach to understand the intertwined relationship between environmental changes and human health. In this paper, the broader approach to environmental health is discussed in order to ‘set the stage’ for introducing the Institute of Environmental Health (ISAMB) of the Lisbon School of Medicine, Portugal. Each of the research groups and labs that compose ISAMB are presented, as well as their main lines of research. Present and planned contributions of ISAMB to advance knowledge on environmental health and for promoting human health gains in an environmentally sustainable way are also discussed.
1. Introduction
Human health is now better than ever before. In a relatively short period of time, we have witnessed an impressive increase of life expectancy and an accelerated growth of the world’s population (from 1 billion in 1800 to an estimated 7.7 billion in 2019 [1,2]). The prosperity of humankind has been supported by our planet’s ecological and biophysical systems, and involved the deprivation of natural ecosystems, some of them hardly recovered by future generations (e.g., Amazon deforestation). Under this scenario, ‘being green’ has become the epitome of post-modern and developed societies. As such, there has been a shift from poor or no environmental awareness (i.e., affective, cognitive, dispositional and behaviour factors that make individuals engage in pro-environmental behaviours [3]) to a generalized concern about environmental issues and the implementation of pro-environment actions at individual, community and political levels [4]. Data from the Special Eurobarometer 340: Science and Technology support the increased importance given to environmental issues by revealing that these are at the top of the list of interests of citizens from the European Union (EU-27; 37%), followed by medical and scientific discoveries (32%) and technological developments (30%) [5]. In some cases, being green and adopting an eco-friendly lifestyle become a source of stress and anxiety at the individual level, which can induce unhealthy behaviours (e.g., stop visiting relatives to avoid spending fuel, food habits that may result in malnutrition stata) that may indicate some degree of psychological dysfunction. Indeed, the term ecorexia was first used in 2008–2009 to refer to those pursuing “green perfection”, even though that would cause them some level of physical and/or psychological discomfort [6].
Our current knowledge about the consequences of human-induced environmental changes is unprecedented, and so it is the acknowledgement that our health and well-being depend on the natural environment. However, the link between the two—human health and natural environment—has been mainly considered from a narrow perspective. In this context, a limited set of environmental determinants (physical, chemical and biological) has been considered in relation to human health [7,8,9]. However, this is not only insufficient to capture the multitude of environmental health determinants to which we are exposed to in our daily lives, but it also disregards the urgent necessity to secure a balance between human health and the health of the planet [2,10]. So, there is space, and need, for a paradigm shift in environmental health involving the progressive abandonment of the traditional and narrow-focused “source-exposure-receptor” model and the adoption of a biopsychosocial model of environmental health, permeable to the introduction of additional layers, while simultaneously fostering environmental sustainability [11].
Grasping a Broader Perspective of Environmental Health
In a changing world that calls for attention to human health and well-being gains in relation to environmental sustainability [10,12], the Institute of Environmental Health (ISAMB) of the Lisbon School of Medicine was created. This is a young research institute that is devoted to the study of environmental determinants of human health broadly considered, and contributes to inequities’ reduction while considering environmental sustainability (Figure 1) within a planetary health paradigm [10]. ISAMB conceptualizes ‘environmental health’ as the assessment and management of ‘modifiable’ environmental factors (physical, biological, chemical, digital, psychosocial, political, socioeconomic and cultural), from both multidimensional natural and built environments that determine human health and well-being, as well as related behaviours, in a lifespan perspective [13]. As such, human behavioural change, both at the individual and community levels, is assumed as essential to achieve sustainable health supportive environments.
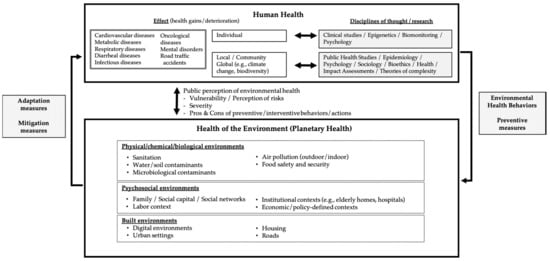
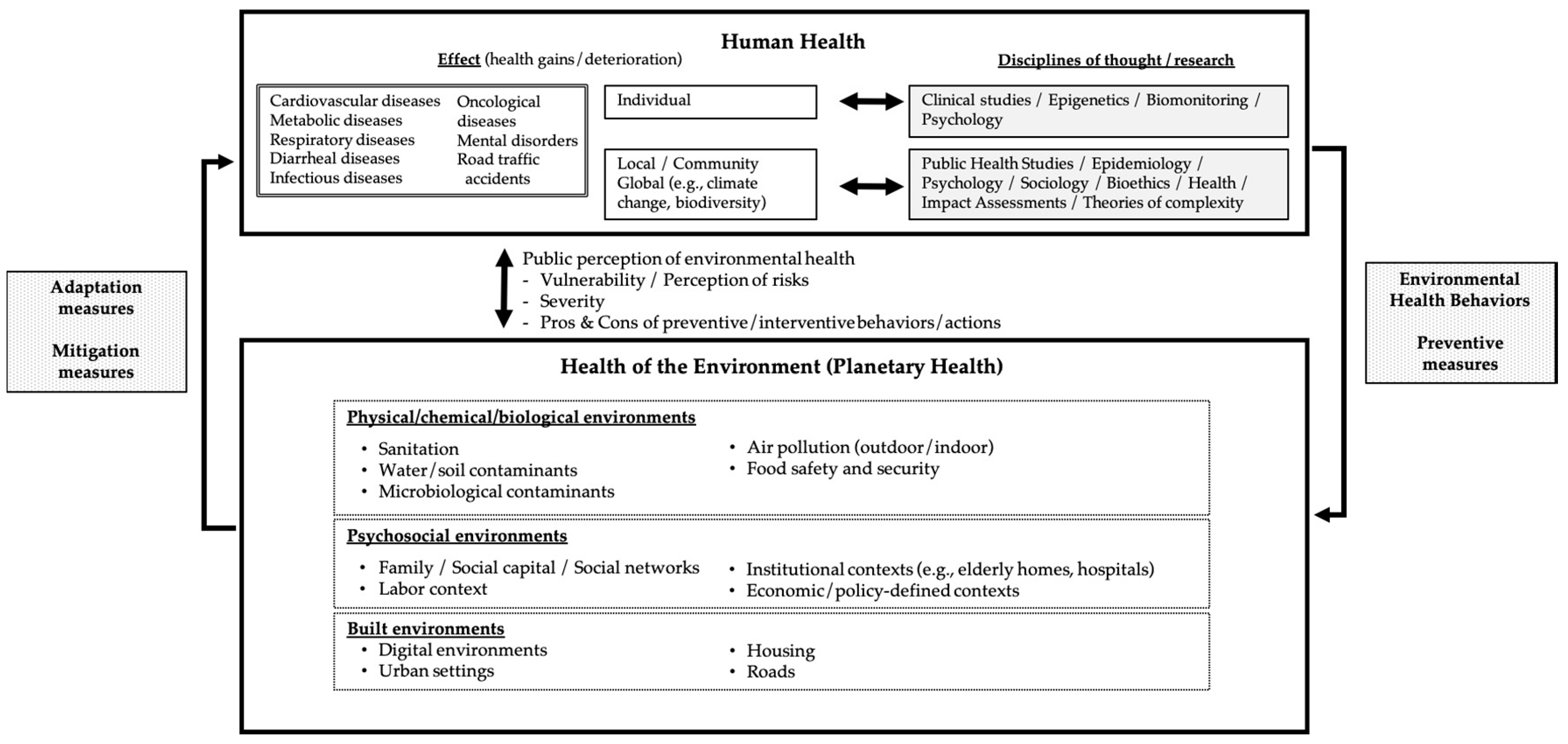
Figure 1.
A broad perspective of Environmental Health regarding the interplay between human health and the health of the environment.
ISAMB is embedded in a very particular ecosystem. It is located within the facilities of the largest medical school and university hospital in Portugal and right next door to the Preventive Medicine and Public Health Institute from the Lisbon School of Medicine. Thus, research being conducted at ISAMB benefits from the interconnections between the clinical and academic settings, namely concerning: (a) healthy ageing as a priority for health and socioeconomic policy agendas; (b) health promotion and disease treatment accounting for modifiable environmental health determinants, from a lifespan perspective; and (c) improvement of human health while assuring environmental sustainability.
In-house research is led by five independent, though complementary in nature, research groups (RGs) (Table 1). In a rapidly changing environment, RG “Environment and infectious diseases” and RG “Environment and non-communicable diseases” investigate the prevalence and aetiology of infectious and non-communicable diseases (NCDs), respectively, by considering a broad set of environmental health determinants. These two RGs make use of sound epidemiological surveillance systems, which allow (a) the timely detection of disease outbreaks in the case of infectious diseases, and (b) the design of patient-tailored rehabilitation programs for individuals diagnosed with NCDs.

Table 1.
Research groups and labs of the Institute of Environmental Health (ISAMB).
Health promotion and disease prevention through the creation of health supportive environments and empowerment of individuals to control their own health are the guiding principles of two other RGs; the RG “Supportive environments for Public Health and Health Promotion” and the RG “Supportive environments for individuals’ lifespan development”. Whereas the former RG is much focused on health promotion at the community by acting on modifiable physical, psychosocial and digital environmental determinants, the latter one focuses on empowering individuals to take control of their health within their contextual environments (from schools to elderly homes).
Finally, the RG “Ecogenetics and human health” addresses the pathophysiology of infectious and non-communicable diseases by considering the interplay between genetic, epigenetic and (modifiable and non-modifiable) environmental determinants.
Research being conducted at each of the five RGs is complemented and integrated by two research labs: the “Environmental Health Behaviour (EnviHeB) Lab” and the “Environmental Health Microbiology (EnviHealthMicro) Lab”. Each lab is devoted to specific research objectives with cross-sectional relevance for the five RGs. Furthermore, whereas the five RGs pursue distinct though complementary research lines under a broad concept of environmental health—occupational settings, health promotion, lifespan development and active ageing, infectious and non-communicable diseases, ecogenetics—each lab aims at novel knowledge production and integration of the environmental health phenomena addressed across the five RGs.
In the following sections, the five RGs and the two labs of ISAMB are described in more detail, with a particular focus on their main research endeavours and on how these contribute to better human health while fostering environmental sustainability.
2. ISAMB’s Research Groups: Environmental Health Research Domains and Contributions
2.1. Research Group “Environment and Infectious Diseases”
A hot environmental health topic in the last few years has been the changes that have occurred in the geographical and seasonal distribution range of several species, including infective pathogens (e.g., viruses, bacteria and parasites), as a consequence of habitat fragmentation, and more recently, as a response to climate change [14]. This situation implies consequences for the natural ecosystems, including their equilibrium, but also for human health, especially regarding infectious diseases [15]. Indeed, the life cycles of infective pathogens are temperature-dependent, which means that climate change, combined with globalization and facilitated human transportation, can create opportunities for these organisms to spread out of their ecological distribution range to areas that may provide them, among other favourable conditions, with the optimal temperatures for their development, growth and reproduction [16,17].
In Portugal, the growing concern regarding vector-borne diseases is supported by two factors. First, the Mediterranean region, where Portugal is located, is considered to be a climate change hotspot and current projections indicate that this region will be hit by dramatic changes in temperature and precipitation in the upcoming years [18,19]. Second, historical outbreaks of relevant vector-borne diseases (i.e., malaria, mediterranean spotted fever, Lyme borreliosis, and dengue) have been detected in the country [20]. Against this background, the National Network for Vector Surveillance (REVIVE) was created in 2008 to tackle this issue (see Table S1 in Supplementary Materials for more information regarding RG “Environment and infectious diseases” research lines/projects). REVIVE is a collaborative network of infectious diseases specialists (medical doctors and researchers), health professionals and staff from the Portuguese Regional Health Authorities (Mainland, Azores and Madeira Islands) and the Directorate-General of Health, which is coordinated by the National Institute of Health Doutor Ricardo Jorge. Researchers from ISAMB’s “Environment and infectious diseases” research group also take part in the REVIVE network. Since its establishment, REVIVE plays a key role in gathering information concerning (a) the distribution, abundance and ecology of species of hematophagous arthropods, (b) the identity and seasonal occurrence of vector-borne agents, and (c) the identification of public health-relevant pathogenic agents. Data collected within the REVIVE network follow two main paths: (a) scientific knowledge on animal vectors and pathogens (e.g., ecology, occurrence) that is shared with peers, and (b) regular reports to inform health policies by the Portuguese Public Health Authorities. REVIVE was designed as a monitoring program that allows the timely detection of any changes in abundance and species diversity of mosquitoes (REVIVE-Mosquitoes) and ticks (REVIVE-Ticks) that may cause harm to human health, thus supporting timely and effective control measures rather than emergency responses by health authorities. For example, within REVIVE-Mosquitoes, 24 species of mosquitoes were identified for the period 2008–2014 from a pool of 500,000 mosquitoes trapped across the country, and no pathogenic viral activity for humans was detected in these mosquitoes’ populations [21]. Apart from agents with human pathogenic activity already reported for Portugal, human pathogenic agents reported from other countries were also punctually identified within REVIVE-Ticks for the period 2011–2015 [17], which strengthens the need for regular and detailed control actions in order to prevent disease outbreaks.
Within the context of infectious diseases, food safety has also grown to be relevant in recent years [22]. Warmer temperatures in association with decreased rainfall are expected to influence the distribution, survival and transmission of pathogenic microorganisms (e.g., bacteria, virus and fungi) with consequences for the patterns of their corresponding foodborne diseases. In addition to the impacts for human and animal health of food contamination by microorganisms, this issue is anticipated to aggravate health and social inequities between developed and developing countries [23]. While research in this topic is flourishing in Europe and North America, less is known from African countries [24,25]. First of all, this happens because many food-borne diseases lack specific symptoms and the incubation period between infection and symptoms is generally long, which cumulatively challenges the adequate diagnosis and identification of the food vehicle that resulted in exposure. This is a common difficulty to the proper diagnosis of a foodborne disease, which is aggravated in developing countries by the long distances between communities, inexistence of (adequate) communication infrastructures, and the lack of human and material resources ensuring proper surveillance programs and responses [26]. Under this context, researchers from this RG are collaborating with Mozambique’s local health authorities and researchers in order to detect foodborne parasites (Cryptosporidium spp., Giardia duodenalis and microsporidia) in vegetables for sale in the farmer markets in Maputo city. This is an ongoing project involving the local community (i.e., food vendors), which results from a longstanding successful collaboration between researchers from the two countries under the topic of infectious diseases. The outputs of this project are expected to support the development of (a) health literacy actions tailored for food vendors, (b) a reliable and sustainable system for provision of drinking water and sewage disposal at the community, and (c) food safety guidelines and tools, tailored to the specific characteristics of each local farmer markets, to be implemented by the local health authorities, thus reducing the changes for food contamination and disease outbreaks (e.g., [27]).
Environmental changes alone, namely climate change, cannot explain the emergence or re-emergence of infectious pathogens. Instead, multiple factors have been implicated in the process, and these include technology and industry-related determinants, namely referring to changes in food processing or to the widespread use of antibiotics, inadequate public health control measures, demographic processes (e.g., migration), and human behaviour at individual or societal levels, such as risky sexual behaviour, intravenous drug use and human migrations, among others [28]. An environmental health currently relevant issue is human infection by thermotolerant fungi, namely by Candida auris, in hospital wards [29,30]. This fungi is expected to be favoured over less thermotolerant species in a context of climate change towards increasingly warmer temperatures, which potentially leads to changes in the distribution range of heat-tolerant vs. susceptible fungal species [31,32,33]. In the particular case of C. auris, this pathogenic fungi is easily confounded with less aggressive species of the same genus, which makes it harder for the researchers to identify its environmental reservoirs, ecological niches outside hospital wards, and selective pressures responsible for its emergence [34,35,36]. Several hypothesis have been pointed out to explain its successful widespread, including rising temperatures [34,37,38] and the widespread use of antifungal drugs [39]. This RG is highly committed to contribute further knowledge to disentangle the environmental variables favouring the emergence of fungi infections among in-patients, and the outputs from the ongoing project about the hospital mycobiome will assist the design of (cost and time) effective control measures for reducing fungi exposure to be implemented in the hospital setting. To accomplish this, a multidisciplinary team of researchers is working in close association with the hospital community to (a) characterize the hospital mycobiome, namely by identifying the fungal species (pathogenic and non-pathogenic) that colonize this setting, their frequency and distribution, and to (b) study the ecology of high concern pathogenic fungi, including its environmental reservoirs inside and outside the hospital setting, and the interactions with other microorganisms that might facilitate or halt their successful spread.
As previously mentioned, multiple factors are involved in the spread of infectious diseases, including human behaviour and lifestyle. This is particularly relevant in the case of sexually transmitted infections, but also concerning the spread of infections among intravenous drug users through the sharing of contaminated injection material, such as needles. Intravenous drug users are a high-risk population for viral hepatitis C (HCV): almost one quarter of the patients infected with HCV inject drugs, and more than half of the intravenous drug users were diagnosed with chronic HCV infection [40,41]. Given the high burden and increasing mortality of viral hepatitis (mortality globally increased 22% between 2010 and 2015) [40], responses at all scales—local, national and global—are urgently needed. To start, further knowledge on the psychosocial environment where drug injection takes place will shed light on the route(s) of transmission. The available evidence indicates that injecting drugs per se does not seem to be the leading cause of HCV infection [42,43]. On the contrary, engaging in unsafe injecting practices, such as sharing contaminated injection material (e.g., needles, syringes, ‘cookers’, and filters) mostly contribute to the spread of the virus [43,44]. In the same way, social contexts favouring limited access to sterile injecting equipment, such as prisons, tend to be associated with a higher risk for HCV infection [44]. In this context, this RG is gathering epidemiological data on HCV prevalence among intravenous drug users in Portugal and producing a detailed characterization of the psychosocial contexts where injection activities occur (e.g., prisons, syringe exchange programs), which will contribute, in the first place, to define a national health strategy to tackle HCV infection. Apart from contributing for the design and implementation of health policies that maintain and, if possible, increase the quality of life of people living with HCV, outputs from this project will also assist the development of prevention programs targeted to at-risk populations in order to break the chain of infection and decrease the prevalence of HCV in Portugal.
2.2. Research Group “Environment and Non-Communicable Diseases”
The combined action of a longer life expectancy and environmental exposure to risk factors throughout the lifespan has motivated increased attention to non-communicable diseases (NCDs) and associated disability [45]. This type of diseases constitutes a major challenge to sustainable development [46] as they disproportionately tend to affect people from developing countries, thus contributing to increased health inequities at the global scale. In line with this, the third Sustainable Development Goal defined in 2015 within the 2030 Agenda for Sustainable Development has sought to address these concerns by establishing an ambitious objective—to reduce by one-third premature mortality from NCDs through prevention and treatment by 2030 [47]. This is a demanding challenge, because numerous genetic, physiological, behavioural and environmental factors, with different weights, might be involved in the aetiology of disease. Prevention and treatment strategies for NCDs have been mainly focused on clinical and/or public health interventions, targeting behavioural risk factors, such as tobacco use, physical inactivity, or unhealthy diet, or recurring to the use of drugs or other health technologies. Most of these human health-oriented actions impact environmental sustainability (mainly in a negative manner through increased waste production, carbon emissions, etc.), but also seem to be not enough to tackle NCDs, which is mostly due to the multitude of NCDs’ determinants and their complex interactions. Therefore, environmentally-based public health and clinical approaches potentially provide more informative data concerning the prevalence of NCDs, corresponding risk factors, and the environmental impact of NCDs prevention and treatment strategies.
Uneven exposure of the world’s population to environmental risk factors for NCDs is indisputable. For example, exposure to air pollution is expected to be more prevalent in urban (vs. rural) areas. Moreover, even in rural areas, exposure to outdoor polluted air is expected to be increased in communities living in the vicinity of industrial plants or close to highways. As a consequence, differences in the prevalence of NCDs, namely chronic respiratory diseases, are expected. The lack of environmental exposure data motivated the creation of the Burden of Obstructive Lung Disease (BOLD) Initiative, an international consortium that aims to estimate the prevalence of chronic obstructive pulmonary disease (COPD) and to identify environmental risk factors for COPD across developed and developing countries [48]. This RG takes part in the BOLD Initiative and is responsible for conducting data collection in Portugal, one of the participating countries in the project, by using cross-sectional surveys following standardized methods and meeting high levels of quality control. Baseline data collection covered more than 30,000 individuals from 42 countries and provided relevant information concerning the prevalence of COPD across countries and exposure to environmental risk factors for COPD [49], thus allowing for the estimation of COPD-related social and economic burden, namely the health care costs associated. By mapping the worldwide situation concerning COPD and supporting informed public health policy decisions [50,51,52,53], which are tailored to the specific situation of each country, BOLD Initiative will ultimately contribute to reduce health and social inequities between developed and developing countries.
Another main issue regarding further understanding and effective treatment of NCDs at the individual and community levels concerns the access and integration of real-world data, aiming to the generation of real-world evidence. For example, electronic health records contain retrospective (quantitative and qualitative) information on physical and/or mental health conditions of the patients, prescriptions, sociodemographic and lifestyle data, and other relevant environmental health determinants. The potential of these data for the report on quality indicators of healthcare providers, epidemiological surveillance (e.g., disease outbreaks), and the translation of personalized medicine into clinical practice, has attracted much attention in recent years. However, electronic health records are mainly text-based and thus, the analysis of such large amounts of unstructured big data to obtain clinical-relevant and meaningful information has been based on the implementation of artificial intelligence algorithms [54,55]. Researchers from the “Environment and non-communicable diseases” RG teamed up with software developers, who are highly experienced in applying artificial intelligence algorithms to the analysis of biomedical data, in order to identify (a) environmental health determinants of NCDs, with a particular focus on respiratory diseases, and (b) healthy ageing determinants (protection profiles), as opposed to disability and premature death determinants (risk profiles) of Portuguese adults. This is an ongoing project that makes use of the national health databases to address the mentioned aims. Apart from the academic interest of this project, expected outcomes will contribute for a cost-efficient and timely answer from the health providers concerning NCDs; it will also produce relevant information to support healthy ageing policies in the country.
In the age of personalized medicine, the integrated analysis of data from the electronic health records (i.e., individual phenotypical data) and individual molecular information (e.g., multi-omics data) benefits from recent advances in big data management and analysis, and provides an unprecedented opportunity for individual-tailored diagnosis and therapy (e.g., [54,56]). Within this context, researchers from this RG have been involved in several projects aimed at identifying the specific biomarkers for the obstructive sleep apnoea, which has been also associated to negative cardiovascular outcomes [57], and lung cancer. Putative biomarkers for obstructive sleep apnoea [58,59] and for the association between obstructive sleep apnoea and lung cancer [58,59] were identified through biochemical determination. The exploratory approach using molecular genetics and proteomics have already produced relevant results concerning the identification of biomarkers for these two health conditions [60,61,62,63,64] (Table S2). Once the identification of biomarkers is validated, these data can be translated into a proteomics-based non-invasive testing for screening, diagnosis, and/or treatment of obstructive sleep apnoea and lung cancer. The following step is to use machine learning algorithms to integrate molecular genetics and proteomics data with information on environmental exposure to risk factors (e.g., second-hand smoke exposure, occupational exposure, exposure to heat waves, eating habits), thus increasing the ability of health providers to administer safer and more effective health treatments to their patients.
Finally, the contribution of RG “Environment and non-communicable diseases” to individual-tailored healthcare solutions include the design of a patient-tailored pulmonary telerehabilitation program, which enable patients to receive individual healthcare regardless of where they live [65], thus avoiding unnecessary hospital consultations. This program has relevant benefits for both the patients (e.g., reduced exposure to environmental determinants of nosocomial disease) and the environment (e.g., reduced carbon emission associated to regular home-hospital-home commuting). Telerehabilitation has benefit from the technological developments of recent years and has been increasingly used to remotely monitor patients during treatment or rehabilitation [66]. SmartReab, the innovative telemedicine solution developed by this RG, combines a smartphone telemonitoring system with a mobile software application connected to an accelerometer sensor and to an oximeter [67,68]. It has proven successful in measuring the intensity of physical activity by patients and, at the same time, tissue oxygenation levels. Physical inactivity is the fourth most important risk factor worldwide for ill health [69], and decreasing the sedentary behaviour of individuals diagnosed with chronic diseases is crucial to maintain optimal health. An interesting feature of this method is the possibility for using regular monitoring data collected to design a tailored intervention for patient pulmonary rehabilitation, while potentially increasing treatment adherence and long-term compliance with the program [68].
2.3. Research Group “Supportive Environments for Public Health and Health Promotion”
As already pointed out, the world is going through unprecedented demographic changes. An inversion of the age pyramid [70,71] has been associated with a dramatic increase in life expectancy [72] and a decrease in fertility rates [73]. This has brought several economic and social challenges [74], namely concerning the development of public health policies aimed at healthy ageing. A central aspect to public health policies and health promotion actions is the environment in which individuals are embedded, as acknowledged in the Ottawa Charter for Health Promotion [75]. As such, a great emphasis of RG “Supportive environments for Public Health and Health Promotion” is placed upon the creation of sustainable health supportive environments—safer, enjoyable, all-inclusive settings that make healthier choices the easiest ones for the local communities at various levels, including the neighbourhood, health units and the workplace, while fostering individuals’ active participation in health and promoting the contact with the natural environment in a sustainable fashion.
A key aspect in health promotion, as argued for in the Ottawa Charter for Health Promotion, is the combined action of intersectoral measures targeting opportunities for the individuals to efficiently use information in order to make healthier choices and improve their health and wellbeing throughout the life course [75]. A good example of this situation is food purchasing. How to increase consumer’s awareness of food nutritional quality at the time of purchasing? Front-of-pack nutrition labels have been pointed out as a key determinant of consumer healthier food choices (e.g., [76]). However, no front-of-pack nutrition label is consensual as the most effective one and thus, consumers are faced with different front-of-pack nutrition label schemes at the time of purchasing, which hinders easy and fast comparability between products and, ultimately, precludes a conscious healthy choice. This leaves the decision on which scheme to be adopted by the food retail operators, within a specific cultural context, to the health authorities. Under this context, researchers from RG “Supportive environments for Public Health and Health Promotion” used a prospective health impact assessment approach to formally examine the potential health impacts of endorsing one front-of-pack nutrition label scheme to be adopted by Portuguese food retailers [77,78] (Table S3). This project resulted from the collaboration of relevant stakeholders—researchers, health authorities, food retailers and consumers—and the outcomes will inform health policies concerning nutritional labelling in Portugal, a subject that is currently under discussion by the competent national authorities.
Another topic of increased interest in recent years has been social prescribing as an avenue to provide patients with individualized healthcare while promoting health and well-being [79,80]. This approach assumes that tackling social determinants of health and well-being are as important as physiological determinants, and thus, it makes uses of the supportive services in the community to increase health and well-being gains while empowering individuals to take control over their health [79]. For example, referrals by general practitioners to community-based green exercise, i.e., physical activity in natural environments, have proven successful in promoting health and well-being across all ages, although much emphasis has been placed on older people and at-risk groups of the population for NCDs [81]. Exercising in contact with nature offers an excellent opportunity for promoting health gains in a bidirectional fashion, i.e., health gains at both human and ecosystem levels. Indeed, it is possible to contribute to environmental sustainability while exercising and reducing sedentary behaviours by electing soft mobility alternatives (e.g., walking, cycling) or improving green spaces through tree plantation [82], among others. This RG is leading an ongoing project aimed at implementing and assessing the effectiveness of a social prescribing intervention for Portuguese individuals aged 55 and over. This project involves a network of social prescribers (health professionals from primary health care units), social users (individuals who will engage in the community-based activities) and social providers (non-clinically trained individuals who work at supportive services in the community). Expected outcomes will (a) contribute knowledge on the impacts of social prescribing as a health promotion strategy as a whole, namely concerning the factors promoting and/or hindering adherence to the program, and (b) provide policy makers with relevant data that support intersectoral informed decision aimed at improved health, social and health equity.
This RG is also concerned about challenges to health equity imposed by global issues. As a matter of fact, health promotion actions are defied by the current global patterns of increased migration [83]. For example, during their inclusion process in the host country, migrant women often take low-skilled, poorly paid jobs, which greatly determines their increased risk of poverty, which combined with their limited access to healthcare (not properly provided by the host countries during the initial phase of their integration or not searched for by the women due to financial difficulties) exposes them to increased health vulnerability. However, social determinants of health in migrant populations have been seldom explored [84], which motivated our RG to design a project aimed at exploring intersectoral inequalities, at the socioeconomic and political systems, and facets of the acculturation process that might determine sexual and reproductive inequalities in health between migrants living in Portugal and Portuguese individuals [85]. This is an ongoing study that targets the Cape Verdean community living in Portugal, which is the second-largest foreign community residing in the country according to recent demographic data [86]. Expected outcomes will support informed policy making concerning (a) the social determinants of sexual and reproductive health of a very specific vulnerable population, (b) the actions needed to create supportive environments that promote their healthier and safer integration in the host communities, namely regarding their kids in school, access to healthcare and family planning services, a smooth acculturation process while maintaining their cultural identity and practices, among others, and (c) the reduction of the social stigma towards migrant communities.
2.4. Research Group “Supportive Environments for Individuals’ Lifespan Development”
It is widely recognized that health and health behaviours in adolescence and adulthood are strongly associated. Despite much attention being initially placed on the social contexts and determinants during childhood that affect health and health behaviours during adulthood [87], the relation between social determinants during adolescence and health outcomes during adulthood has received increased attention in recent years (e.g., [88,89]). During adolescence, family is not the single primary environmental health determinant. At this stage, the school environment, the neighbourhood and peers collectively determine also the adoption of health behaviours concerning physical activity, healthy eating, drugs misuse and sex, these being major determinants of later life health outcomes and inequities [87]. Under this scenario, the meaningful involvement of the target population, adolescents in this case, throughout the whole process of designing and implementing health promotion and disease prevention programs potentially scales up their chances of success [90,91,92]. One of the great challenges of such a participatory approach to adolescents’ health is to make them feel competent, motivated and to create opportunities for environmental behaviour change to take place [93]—this perspective emphasizes the relation between supportive environments (i.e., the ‘opportunities’) and health and wellbeing throughout the life course.
The scarcity of data on Portuguese adolescents’ social behaviour, namely at the school environment, and later health outcomes motivated the design and implementation of the Social Adventure Project (SAP), back in 1987. Several researchers from the RG “Supportive environments for individuals’ lifespan development” have been actively involved in the project since its inception. The SAP has been used as a regular monitoring initiative of the adolescents’ health behaviours in their environmental contexts, but also as a privileged space to motivate youngsters to engage in community-based approaches to health and wellbeing promotion. Importantly, youngsters are challenged to focus on the environmental factors that maintain and sustain health and wellbeing rather than devoting much attention to risk factors [94,95,96]. The SAP later integrated the Health Behaviour in School-Aged Children (HBSC) study [97,98], a World Health Organization collaborative survey implemented in 1994 that now includes 50 countries across Europe and North America. The HBSC study collects information about youngsters’ health behaviours and well-being in their environmental contexts—at their home, at school, at their neighbourhood, and during leisure time. This regular survey (conducted every four years) provides a relevant dataset on several behavioural and health-related domains, as well as on health and well-being determinants, such as digital contexts and climate change. Data from the SAP and the HBSC have been serving the following purposes: (a) to advance knowledge on health behaviour among adolescents, (b) to support the design and implementation of cost-effective and sustainable participatory research projects involving youngsters aimed at health promotion and environmental sustainability [99,100,101,102], and (c) to inform health policies concerning relevant environmental determinants of health behaviours among adolescents.
Youth meaningful engagement within their communities in a broadest sense (i.e., family, peer groups, school, neighbourhood) builds on youth strengths in order to develop health promotion programs centred on youth happiness and well-being, thus strengthening their competences in a positive and constructive manner instead of focusing on disease [94,95]. This approach is particularly relevant in a changing world—we are witnessing profound changes in natural environments, political and socioeconomic systems, psychosocial environment, among others—that call for youth prosocial involvement in a lifespan perspective. This was the trigger needed for this RG to go further the SAP and implement the Dream Teens project, which aimed to empower Portuguese youth and to provide them with the opportunities for playing an active role in decision making concerning issues affecting their lives and the communities where they live [90]. Youth involved in Dream Teens project developed and strengthened positive assets, namely leadership, interpersonal and action-research skills, which contributed to empowering them to identify problems in their life contexts and propose corresponding solving strategies. Moreover, a communication channel with policy makers was also opened during the project, and youth involved had the opportunity to directly discuss their concerns with decision makers [103], and to some degree, to influence political decisions concerning relevant health issues for them. Dream Teens is a successful project that already has two ongoing spin-off projects: Dream Teens powered by Cascais Jovem and #Generation with a Voice. While the original project is much concerned with health issues, the latter two go further and aim at awareness raising and discussion around intergenerational justice, climate change and sustainability, thus assuming a lifespan perspective in the creation of environments that ease healthier choices (Table S4).
It is widely recognized that social participation is important in all ages, this being associated with better health and well-being outcomes and the promotion of active ageing [104,105]. However, social participation tends to decrease as people grow older (e.g., [106,107]), which can be mitigated by developing intergenerational programs that put together younger and older generations. Intergenerational programs for promoting social participation are associated with enhanced self-esteem, improved social skills, better overall health and well-being, and connectedness to the community among those involved [108]. Under this context, this RG considers that intergenerational projects aimed at increased social participation are fundamental to contribute to healthy ageing in a world growing older. The first steps in this area are now being given by this RG with the project Inter-help, which aims at promoting individual skills and resources, such as leadership and interpersonal skills, as well as to strengthen social networks in the elderly population through their active involvement and participation in activities and organizations from the community in an intergenerational perspective. A great challenge to this approach is that community engagement, of both youth and old populations, is not exclusively dependent on the individuals; it is also determined by environmental factors that should be taken into account in the design and implementation of such health promotion strategies [109].
As previously mentioned, a more complete picture of youngsters’ health behaviours and well-being should consider them in their various environmental contexts—at their home, at school, at their neighbourhood, and during leisure time. This is also true when it comes to the design and implementation of action-research participatory programs for health promotion across all ages. As such, this RG is currently leading intervention programs at the family, school and hospital environmental contexts. A common goal to these projects is to strengthen competences, and to motivate and create the opportunities for environmental health behaviour change of the involved parties. Briefly, Adventure in Parenting adopts an intergenerational perspective by involving young people, parents and grandparents around the strengthening of nurturing relationships within the family, empowerment of the individuals concerning their health and well-being and their engagement in social participation activities that foster sustainable health and lifestyle. By involving all levels within the school organization, from the students to the principal director, school projects led by this RG aim to promote educational organizations as friendly and sheltering environments for health and well-being, while strengthening students and staff leadership skills in health. Finally, a multidimensional approach involving health and non-health professionals, as well as patients, is currently being implemented in the hospital setting, which aims at promoting health organizations as sustainable, friendly and sheltering environments for health and well-being. This project assumes a particular relevance in the current COVID-19 pandemic, given the high distress levels that health professionals in the frontline are experiencing [110], patients without COVID-19 who are afraid to seek help [111], postponed surgical procedures [112], among other factors that challenge the provision of healthcare and the sustainability of health organizations.
2.5. Research Group “Ecogenetics and Human Health”
Genetic factors determine the susceptibility of individuals to environmentally-induced adverse health outcomes. As such, health effects driven by the exposure to environmental factors result from the intricate links between the underlying characteristics of the environmental agent to which the individual is exposed and the biochemical response of the individual, which is known to be genetically determined [113,114]. The advent of modern molecular techniques (e.g., real-time PCR, next-generation DNA sequencing, metagenomics and other “omics”) in combination with biochemical, clinical and epidemiological studies allow the detailed assessment of (a) the interaction(s) between environmental factors and genetic and epigenetic backgrounds, and (b) the expression of intermediate (i.e., phenotypes positioned between genetic variation and disease phenotypes) and distant (i.e., disease) phenotypes and disease risk and how this is modulated by the interaction between environmental and genetic/epigenetic factors. This approach reduces the risk of bias introduced by behavioural, social and unknown physiological factors to our knowledge on the real extent of the effect(s) of a suspected environmental exposure on infectious and non-communicable diseases risk.
As previously mentioned, the creation of supportive environments to which the individual is exposed during the entire lifespan, starting from pre-conception until elderly, also assuming an intergenerational approach, is fundamental for better health. Under this context, further understanding on the pathophysiology of diseases, namely through the identification of genetic, epigenetic and (modifiable and non-modifiable) environmental factors involved in the aetiology of multifactorial diseases, is crucial for health promotion and disease prevention programs, but also for disease early screening and treatment. As such, this RG has been leading projects (Table S5) aimed at identifying (a) susceptibility genes for the development of a given pathology (distant genotype-distant phenotype) under specific environmental conditions, (b) biomarkers (biochemical and cellular) associated to disease (intermediate phenotype-distant phenotype), and (c) candidate genes modulating the expression of those biomarkers (genotype-intermediate phenotype). Relevant contributions, at both academic and clinical practice levels, include the genetic profiling and identification of biomarkers for infectious diseases (human papillomavirus infection and cervix cancer, viral hepatitis C), NCDs (obesity, hypertension, diabetes, asthma, polycystic kidney disease, alcoholism-related sickle cell disease), and longevity-related NCDs (osteoporosis and sarcopenia) [114,115,116,117,118,119,120]. These studies cover a wide range of metabolic (e.g., cellular iron homeostasis, lipids, glucose, neurotransmitters) and signalling pathways (e.g., amino acid signalling), and inflammation biomarkers (e.g., acute-phase proteins), among others known to be involved in the pathophysiology of disease.
A comprehensive understanding of the natural history of diseases, including those resulting from mutations in a single (set of) gene(s), such as sickle cell disease or polycystic kidney disease, will benefit from assessing the environmental factors potentially involved. Towards this end, the close collaboration between researchers of this RG and medical doctors allows to examine the association between patient’s behavioural data and the development and progression of a given pathology, as well as their role in modelling biochemical markers and genes’ impact [121,122]. The consideration of environmental factors is much facilitated by the multidisciplinary nature of ISAMB, which sets the ground for valuable collaborations among researchers with different skills and expertise to be successfully established. As such, the set of environmental determinants now considered for the discriminant analysis of intermediate and distant phenotypes based on the distribution of genetic polymorphic variants is much broader and include physical (e.g., light and noise), nutritional (i.e., diet), behavioural (e.g., physical exercise, alcohol consumption, smoking habits and coffee drinking), microbiological (e.g., microbiota, viruses, bacteria and parasites) and natural environment determinants.
3. ISAMB’s Research Labs: Environmental Health Research Domains and Contributions
3.1. Environmental Health Behaviour Lab (EnviHeB Lab)
Reduction of red meat consumption, active commuting, visiting green and blue spaces, and using clean energy are good examples of synergic health and pro-environmental behaviours [123]. If activated and maintained as habits, these behaviours act as relevant health and sustainability promotion agents. Indeed, behaviours like these have recently attracted much attention, not only due to their potential for human health promotion, but also because of their contribution to environmental sustainability throughout the reduction of greenhouse gas emissions, water and energy saving, biodiversity protection and reduction of the ecological footprint. In both cases, human health and environmental sustainability positive outcomes can only be achieved if these behaviours are activated and maintained as long-standing habits at the individual and community levels, respectively. However, habit formation is a complex process that initiates when one makes a decision to take action, for example, getting to work by bike every day, willing to get exercised and/or to contribute to the reduction in the emission of greenhouse gases. The translation of this behavioural intention into action takes some planning and self-regulation. For example, after cycling to work several times in a planned fashion, at some point, this behaviour becomes a habit, i.e., an automatic response that is activated by environmental cues [124]. As such, the identification of the determinants involved in habit formation is essential in order to promote behavioural change as part of any effective health promotion program. In addition, the implementation of cost-effective and sustainable interventions for environmental health behavioural change, at both individual and community levels, lies in the application of different theories of health behaviour change, focusing on cognitive processes (e.g., beliefs, attitudes, intentions and decision-making [125,126]), motivational and self-regulatory processes [127,128], nudging processes [129,130] and value-based decision processes [131]. Finally, the effectiveness assessment of such health promotion interventions, i.e., the impact of behavioural change on relevant health outcomes, usually requires the application of suitable epidemiological tools.
The Environmental Health Behaviour (EnviHeB) Lab is a multidisciplinary research lab devoted to the study of human behaviour modification, with a particular focus on the formation of habits that might impact human health as well as environmental sustainability. A guiding principle of the research being conducted at the EnviHeB Lab is that the concurrent promotion of human health and well-being, and Earth’s sustainability implies the effective modification of pathogenic habits (e.g., sedentary behaviour, unhealthy eating behaviour, and excessive waste production) at both individual and societal levels. Given that a multiplicity of individual (e.g., own personality, cognitive, volitional and emotional factors), cultural, socioeconomic and political determinants have been implicated in behavioural choices, environmental health behaviour promotion should be framed within an ecological model [132,133]. This motivated the EnviHeB Lab to design a cohort (Health Behaviours and Attitudes, HABITUS) aimed at identifying environmental determinants involved in the aetiology of health and pro-environmental behaviours, such as smoking cessation, healthy eating, and exercising, among others (Table S6). Data collection at baseline was recently conducted and mainly focused on behaviours (and behavioural change) during social containment due to COVID-19 pandemics. Areas to be covered within the HABITUS questionnaires are not fixed and each RG within ISAMB contributes according to their specific research interests—this was already the case for the baseline questionnaire, which was developed in close collaboration with researchers from RG “Supportive environments for Public Health and Health Promotion”.
A main challenge to habit formation concerns the change of values as organizing principles, supporting the adoption of behaviours that, though less gratifying in the short-term, are recognized to improve human health and potentially contribute to environmental sustainability in the medium- to long-term. This implies gaining a better understanding on the mechanisms/processes that underlie the successful repetition of consistent decisions, which ultimately lead to the formation of health and pro-environmental habits [124,134]. A good example is treatment adherence—what are the individual and environmental determinants underlying treatment adherence? This question motivated the EnviHeB Lab, in collaboration with researchers from different RGs within ISAMB, to design the GamInMind project (Gaming against chronic diseases: A quest for crossroads between joy and cognitive health) aimed at developing and assessing the effectiveness of a videogame’s platform using game difficulty balancing technology for training executive cognitive function. Expected outputs from this project will shed light on the individual’s competences that are involved in treatment adherence, with promising implications for clinical practice.
As previously discussed within the subsection concerning the RG “Supportive environments for individuals’ lifespan development”, empowering individuals in relation to their health and supporting their meaningful involvement in health promotion programs implies making them feel competent, motivated and creating the opportunities for environmental behaviour change to take place [93]. This is the underlying principle for the MAMH@WORK project and the Health Promotion Observatory in Cascais, two projects led by the EnviHeB Lab. MAMH@WORK project was designed to promote the mental health and well-being of women during the postpartum period, including the return to work moment, by empowering them in relation to their health and well-being, not only by providing women with the information and the tools to do it, but also by involving organizational leaders in the creation of healthy workplaces. Expected outcomes go far beyond merely academic and clinical practice ones, and will also contribute to (a) set maternal perinatal mental health as a priority in the political and organizational agendas, (b) to empower women and promote their participation in the labour market, with a potential positive impact in fertility rates, and (c) to support smart, sustainable and inclusive economic growth. The Health Promotion Observatory in Cascais was created within a community-based participatory approach for health promotion concerning healthy eating, physical activity, and mental health and well-being in Cascais municipality, Lisbon Metropolitan Area. Researchers from the EnviHeB Lab are responsible for monitoring the health promotion actions implemented by Cascais municipality and to provide expert advice to their various initiatives. The outputs from this project have been used to inform political decisions concerning strategic actions to be implemented for promoting health and well-being of Cascais community members across different settings—schools, community organizations, health units, neighbourhoods, among others.
Finally, a main ambition of the EnviHeB Lab is to get strategic knowledge about how to better inform communities about the interconnection between human health and environmental sustainability. The role of science communication for developing strategies to effectively reach the target population(s) of a given health promotion program has been increasingly recognized [135,136]. Strategic health communication is a powerful tool to support the development of effective and insightful health promotion interventions at different levels of the ecological model applied to health, i.e., a multitude of individual and social factors that shape behaviours and, consequently, health outcomes [137,138]. As such, a national-based and representative survey aimed to assess perceptions, attitudes and behavioural intentions related to environmental and human health, with a particular focus on blue environments (e.g., lakes, rivers, sea, and urban blue spaces) as determinants of human health and well-being, was designed and is currently being implemented. This project also aims to (a) measure pro-environmental health behaviours, and (b) create audiences from segments, which will be used to develop audience-tailored communication strategies for promoting public engagement in mitigation and adaptation actions to climate change. Strategic health communication is a recent area of research within the EnviHeB Lab, which directly and indirectly connects the Lab to the five RGs of ISAMB.
3.2. EnviHealthMicro Lab
Microorganisms (bacteria, virus, and fungi) are increasingly, and alarmingly, becoming resistant to the medicines used to treat the infectious diseases they cause, which poses significant threats to human and animal health at the global scale [139,140,141]. Under a scenario of widespread antimicrobial resistance (AMR), the burden of drug-resistant infections in the community, and both associated morbidity and mortality, are expected to increase, which potentially means longer and more severe illnesses, prolonged lengths of stay in hospitals, increased risk of hospital-acquired infections, increased mortality and increased costs of treatments. On top of the direct impacts from AMR to health, illness-related losses of productivity and increased investment in scientific and technological development (e.g., development of new diagnostic tools and treatments, strengthening of the national health systems, among others) by the governments to tackle AMR poses several challenges to the economic growth and sustainable development of each country [140]. The widespread use of antibiotics [140,142,143] and the spread of drug-resistant microorganisms and genes encoding for resistance have been pointed out as the main factors driving the rapid bacterial evolution of resistance [143], and much emphasis has been put into the reduction of antibiotics prescription as an effective public health measure to tackle AMR [140,143].
Gram-negative bacteria are ubiquitous and cause serious infections, such as pneumonia, bloodstream infections, wound or surgical site infections, and meningitis, among others. These have evolved several mechanisms to degrade and/or inactivate antibiotics [144]; therefore, further understanding on how these mechanisms work and their epidemiology is critical to shed light on how AMR evolves and is transferred from bacteria to animal hosts, humans included [145]. As such, the Environmental Health Microbiology (EnviHealthMicro) Lab is highly committed to address the molecular epidemiology and genetic determinants involved in carbapenem resistance by Enterobacteriaceae isolates in the hospital setting. These are described to frequently produce enzymes degrading carbapenems, which reduces the chances of success in the treatment of infections caused by these pathogens, because carbapenems are large spectrum antibiotics frequently used to treat infectious caused by multidrug-resistant strains [146]. This is an ongoing study that will allow the identification of environmental factors determining the virulence and infectivity of Enterobacteriaceae isolates in Portuguese hospital settings (Table S7). A ‘sister’ project is currently ongoing for the characterization of the genetic diversity and environmental determinants involved in AMR by the Klebsiella pneumoniae species complex in both hospital and community settings, in Portugal. Data available from the hospital setting show that carbapenem-resistant isolates of K. pneumoniae are able to successfully adapt to the host environment and sustain virulence via different pathways [147]. The integrated analysis of genetic determinants for carbapenem resistance (currently ongoing), hospital-related conditions (e.g., hygiene and disinfection practices, ventilation, health professionals’ behaviour, food handling practices) and host individual characteristics (e.g., general health status and comorbidities, sociodemographic characteristics, genetic background) will contribute to the design and implementation of cost-effective and sustainable guidelines for decreasing infection rates in the hospital (and the community) setting(s).
AMR is a multifactorial issue with negative impacts at various levels, as previously mentioned. As such, it demands an integrated answer that brings together medical doctors, microbiologists, veterinarians, epidemiologists, health economists, professionals from the pharmaceutical industry, social scientists, ecologists, policy makers, among others. We know almost nothing concerning the environmental determinants of AMR broadly considered. There is some evidence suggesting the role of educational level, income, public-health spending, and countries governance structure in driving AMR [148]. However, what is the relative contribution of each of these factors? Does their relative weight vary between high- and low-income countries? What about within-countries differences between rural and urban areas? Relevant questions to be addressed also concern the transfer of pathogens between hosts from different species. For example, workers in livestock farming are at an increased risk of getting in contact with resistant pathogens via meat consumption, exposure to crops contaminated by manure or even through groundwater contamination [140]. The emergence of resistant pathogens also affects soil biota with consequences for a broad array of ecosystems services [149]. AMR ultimately threatens global health and sustainability [141]. A main endeavour of the EnviHealthMicro Lab is to contribute to put AMR in the research and political agendas. The next steps will be to gain further understanding on the environmental determinants broadly considered that are involved in AMR—sociodemographic characteristics of the hosts determining higher susceptibility to infection by resistant pathogens. Future studies addressing health macroeconomics are also planned.
Preventing exposure to environments where the risk of infection is higher is an obvious healthcare strategy, most especially for vulnerable groups. In recent years, the number of patients receiving home respiratory therapy (HRT) for chronic respiratory failure (e.g., COPD, neuromuscular diseases, and obstructive sleep apnoea) has greatly increased. Although moving healthcare from hospitals/health units to home might decrease the associated burden for health providers, less is known about how the patients perceive home healthcare [150]. Moreover, the impact of HRT on healthcare associated infections is not well characterized for the Portuguese population. For example, patients who are on a ventilator are at an increased risk of lung infection by the bacteria causing pneumonia. As such, researchers from the EnviHealthMicro Lab are currently working with HRT providers in order to gather information on the quality, effectiveness and safety of HRT programs, as well as on patients’ experience, expectations and attitudes towards receiving respiratory therapy at home. Expected outcomes will assist the design of guidelines for HRT providers, as well as a set of patient-tailored recommendations, in order to decrease the risk for healthcare associated infections. This project will ultimately contribute to provide safer and better home respiratory healthcare to the Portuguese population.
The EnviHealthMicro Lab is the youngest Research & Development unit created within ISAMB. As such, only the first steps were taken in establishing a research agenda that simultaneously meets EnviHealthMicro Lab members’ research interests and is relevant for the five RG and the EnviHeB Lab. Nevertheless, collaborative research with the RG “Environment and non-communicable diseases” concerning the monitoring of patients in a telerehabilitation program has already been established, namely regarding further understanding of patients’ expectations and the interplay between NCDs and healthcare associated infections.
4. Conclusions
The interconnections between environment and health have never before been a hot topic as they are now. We are witnessing unprecedented global environmental changes, such as climate change, human migrations, a digital and robotic revolution, and (on top of it) a pandemic with unpredictable health, economic and political consequences. These accelerated-paced changes encompass major challenges for both human health and environmental sustainability, which calls for a paradigm shift regarding the way we relate to ourselves, others (humans and animals) and the environment broadly considered. A wide perspective of environmental health has been sparsely advocated for, and much action has been mainly focused on environmental needs, such as basic sanitation, the improvement of agricultural systems and productivity and the reduction of plastic use, among others. We are convinced that these will continue to drive international and national agendas, and we argue for the harmonious combination of local and global environmental health needs.
ISAMB is a seven-year-old research institute that, since its inception, has been in the frontline arguing for a multidimensional approach to environmental health that considers a broad set of environmental health determinants (e.g., psychosocial, digital, economic and political determinants of health), from both natural and human-made environments, under a Planetary Health paradigm. Much of the research conducted by ISAMB’s researchers is within the healthcare sector. However, this is not a naïve approach motivated by its privileged positioning right next door to the largest Portuguese University Hospital; instead, the deleterious effects to the environmental sustainability arising from healthcare have been increasingly recognized. The first global estimate of health care’s climate footprint indicates that, in 2014, it contributed 4.4% to the global net emissions of CO2 [151]. In line with this, an emerging research interest in ISAMB relates to the climate footprint of the healthcare sector, namely concerning the relative contributions of sources of energy, supply chain, healthcare units’ operational emissions, among others, to the footprint of the healthcare sector.
A pertinent question at this stage is where is ISAMB positioned in relation to its main research goal (i.e., to study environmental determinants of human health broadly considered and fostering to contribute to inequities’ reduction; also considering environmental sustainability within a Planetary Health paradigm), and what are the next steps towards getting closer to fulfil it. ISAMB undoubtedly assumed a broad perspective of ‘environment’ and the research conducted across RGs and Labs targeted multiple environmental factors, as described in this paper and summarized in Tables S1–S7. An interesting feature of ISAMB is its multidisciplinary character—the main disciplines involved in the research being conducted in each RG and Lab somehow reflect the academic/professional background of the team. Moreover, the translation of research findings into informed political decisions, also by involving policy makers in the research process whenever possible (e.g., NutrHIA project by “Supportive environments for Public Health and Health Promotion” RG, together with the EnviHeB Lab; Health Promotion Observatory in Cascais by EnviHeB Lab) has been a main endeavour across RGs and Labs. Despite these efforts, much can be done concerning the meaningful involvement of the political and socioeconomic systems towards a coordinated action across sectors of human activity. One avenue to ease this process is by the creation of an Environmental Health Politics Lab (EnviHealth Politics Lab), which would be devoted to the investigation in health and environmental politics, within a Planetary Health paradigm, and would also contribute to ease the translation of research findings into sustainable health and environmental health policies. Health inequalities have also been a concern of the researchers in ISAMB. For example, FEMINA project led by “Supportive environments for Public Health and Health Promotion” RG addresses health inequalities between Cape Verdean immigrants and Portuguese individuals, with potential impacts concerning public health policies and inequalities reduction. However, this is not the only one. Research conducted by the “Environment and infectious diseases” RG on food safety in Maputo farmers markets, Mozambique, also has the potential to contribute to reduce health inequities between low- and high-income countries. Finally, ISAMB is giving encouraging steps in the study of how human behaviours affect environmental sustainability. A good example is the project on the climate footprint of the healthcare sector mentioned above (currently under peers’ revision). In the near future, ISAMB is highly committed to get its research closer to its main goal, to continuously foster synergies between RGs and Labs, as well as to foster scientific maturity and to grow as a research centre.
Supplementary Materials
The following materials are available online at https://www.mdpi.com/2071-1050/12/21/8963/s1, Table S1: Flagship projects of the “Environment and infectious diseases” research group, Table S2: Flagship projects of the “Environment and non-communicable diseases” research group, Table S3: Flagship projects of the “Supportive environments for Public Health and Health Promotion” research group, Table S4: Flagship projects of the “Supportive environments for individuals’ lifespan development” research group, Table S5: Flagship projects of the “Ecogenetics and human health” research group, Table S6: Flagship projects of the EnviHeB Lab, Table S7: Flagship projects of the EnviHealthMicro Lab.
Author Contributions
F.A., A.V., and O.S. conceptualized the whole paper. A.V. wrote the first draft of the introduction section, with the contribution of O.S., A.V., F.A. and O.S. put together and revised the texts from each R.G. and lab. J.C.: did an overall review of the manuscript. A.V.C. did a final checking of the manuscript. F.A. (Research Group coordinator), R.S., M.S.N. and O.M. wrote the first draft of the text describing the “Environment and infectious diseases” Research Group; C.B. (Research Group coordinator) did the same for the “Environment and non-communicable diseases” Research Group; A.C. (Research Group coordinator) and V.A. for “Supportive environments for Public Health and Health Promotion” Research Group; M.G.d.M. (Research Group coordinator) and T.G. for “Supportive environments for individuals’ lifespan development” Research Group; M.B. (Research Group coordinator) and J.F. for the “Ecogenetics and human health” Research Group; O.S. (Lab coordinator) for “Environmental Health Behavior” Lab, with the contribution of A.V., J.C. and R.R.S.; and C.C. (Lab coordinator) wrote the first draft of the section regarding “Environmental Health Microbiology” Lab. J.C. wrote the first draft of the conclusions, with contributions from A.V. and O.S. All the authors revised and approved the final version of the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Fundação para a Ciência e a Tecnologia, grant number UIDB/04295/2020 and UIDP/04295/2020.
Acknowledgments
The authors would like to thank to José Pereira Miguel and Maria de Fátima Reis as former and first Director and Executive Director of ISAMB (respectively). Without their insight, vision and perseverance, ISAMB would not exist. The authors also would like to thank Ana Sequeira, Hugo Caldeira and Rodrigo Feteira-Santos for the technical support given to this work.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Roser, M.; Ritchie, H.; Ortiz-Ospina, E. World Population Growth. Available online: https://ourworldindata.org/world-population-growth (accessed on 18 October 2020).
- Panorama. Panorama Perspectives: Conversations on Planetary Health—Planetary Health 101: Information and Resources, Report I. 2017. Available online: https://www.rockefellerfoundation.org/wp-content/uploads/Planetary-Health-101-Information-and-Resources.pdf (accessed on 18 October 2020).
- Jiménez Sánchez, M.; Lafuente, R. Defining and measuring environmental consciousness. Rev. Int. Sociol. 2010, 68, 731–755. [Google Scholar] [CrossRef]
- Rosa, C.D.; Collado, S. Experiences in nature and environmental attitudes and behaviors: Setting the ground for future research. Front. Psychol. 2019, 10, 763. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Special EUROBAROMETER 340: Science and Technology. 2010. Available online: https://ec.europa.eu/commfrontoffice/publicopinion/archives/eb_special_359_340_en.htm#340 (accessed on 26 July 2020).
- Ek, R.; Johansson, N. (Eds.) Perspectives on Waste from the Social Sciences and Humanities: Opening the Bin; Cambridge Scholars Publishing: Newcastle, UK, 2020. [Google Scholar]
- National Environmental Health Association. Definitions of Environmental Health. Available online: https://www.neha.org/about-neha/definitions-environmental-health (accessed on 10 May 2020).
- Gordon, J.L. The future of environmental health, part 1. J. Environ. Health 1992, 55, 28–32. [Google Scholar]
- American Public Health Association. Environmental Health. Available online: https://www.apha.org/topics-and-issues/environmental-health (accessed on 10 May 2020).
- Whitmee, S.; Haines, A.; Beyrer, C.; Boltz, F.; Capon, A.G.; De Souza Dias, B.F.; Ezeh, A.; Frumkin, H.; Gong, P.; Head, P.; et al. Safeguarding human health in the Anthropocene epoch: Report of the Rockefeller Foundation-Lancet Commission on planetary health. Lancet 2015, 386, 1973–2028. [Google Scholar] [CrossRef]
- Guidotti, T.L. Environmental health needs a new paradigm, I. getting back in focus. Arch. Environ. Occup. Health 2018, 73, 267–269. [Google Scholar] [CrossRef]
- Haines, A.; Hanson, C.; Ranganathan, J. Planetary Health Watch: Integrated monitoring in the Anthropocene epoch. Lancet Planet. Health 2018, 2, e141–e143. [Google Scholar] [CrossRef]
- Santos, O.; Virgolino, A.; Santos, R.R.; Costa, J.; Rodrigues, A.; Vaz-Carneiro, A. Environmental health: An overview on the evolution of the concept and its definitions. In Encyclopedia of Environmental Health; Nriagu, J., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 2, pp. 466–474. ISBN 9780124095489. [Google Scholar]
- Semenza, J.; Suk, J. Vector-borne diseases and climate change: A European perspective. FEMS Microbiol. Lett. 2018, 365, fnx244. [Google Scholar] [CrossRef]
- Lee, V.J.; Aguilera, X.; Heymann, D.; Wilder-Smith, A.; Lee, V.J.; Heymann, D.L.; Bausch, D.G.; Briand, S.; Bruschke, C.; Carmo, E.H.; et al. Preparedness for emerging epidemic threats: A Lancet Infectious Diseases Commission. Lancet Infect. Dis. 2020, 20, 17–19. [Google Scholar] [CrossRef]
- World Health Organization. Global Vector Control Response 2017–2030; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Centro de Estudos de Vetores e Doenças Infeciosas Doutor Francisco Cambournac. Relatório REVIVE 2011–2015—Culicídeos e Ixodídeos: Rede de Vigilância de Vetores; Instituto Nacional de Saúde Doutor Ricardo Jorge, IP: Lisboa, Portugal, 2016. [Google Scholar]
- Tuel, A.; Eltahir, E.A.B. Why is the Mediterranean a climate change hot spot? J. Clim. 2020, 33, 5829–5843. [Google Scholar] [CrossRef]
- Giorgi, F.; Lionello, P. Climate change projections for the Mediterranean region. Glob. Planet. Chang. 2008, 63, 90–104. [Google Scholar] [CrossRef]
- Núncio, M.S.; Alves, M.J. (Eds.) Doenças Associadas a Artrópodes Vetores e Roedores – 2ª Edição; Instituto Nacional de Saúde Doutor Ricrado Jorge, IP: Lisboa, Portugal, 2019; Available online: http://www.insa.min-saude.pt/wp-content/uploads/2019/09/Doencas_artropodes_vetores_roedores.pdf (accessed on 16 October 2020).
- Osório, H.C.; Zé-Zé, L.; Amaro, F.; Alves, M.J. Mosquito surveillance for prevention and control of emerging mosquito-borne diseases in Portugal—2008–2014. Int. J. Environ. Res. Public Health 2014, 11, 11583–11596. [Google Scholar] [CrossRef] [PubMed]
- Akeda, Y. Food safety and infectious diseases. J. Nutr. Sci. Vitaminol. 2015, 61, S95. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Food Safety: Climate Change and the Role of WHO; WHO: Geneva, Switzerland, 2018; Available online: https://www.who.int/foodsafety/_Climate_Change.pdf (accessed on 17 October 2020).
- Uyttendaele, M.; Franz, E.; Schlüter, O. Food safety, a global challenge. Int. J. Environ. Res. Public Health 2016, 13, 67. [Google Scholar] [CrossRef]
- Bisholo, K.Z.; Ghuman, S.; Haffejee, F. Food-borne disease prevalence in rural villages in the Eastern Cape, South Africa. Afr. J. Prim. Health Care Fam. Med. 2018, 10, 1796. [Google Scholar] [CrossRef]
- Robertson, L.J.; van der Giessen, J.W.B.; Batz, M.B.; Kojima, M.; Cahill, S. Have foodborne parasites finally become a global concern? Trends Parasitol. 2013, 29, 101–103. [Google Scholar] [CrossRef]
- Salamandane, C.; Fonseca, F.; Afonso, S.; Lobo, M.L.; Antunes, F.; Matos, O. Handling of fresh vegetables: Knowledge, hygienic behavior of vendors, public health in Maputo markets, Mozambique. Int. J. Environ. Res. Public Health 2020, 17, 6302. [Google Scholar] [CrossRef]
- Morse, S.S. Factors in the emergence of infectious diseases. Emerg. Infect. Dis. 1995, 1, 7–15. [Google Scholar] [CrossRef]
- Rhodes, J.; Abdolrasouli, A.; Farrer, R.A.; Cuomo, C.A.; Aanensen, D.M.; Armstrong-James, D.; Fisher, M.C.; Schelenz, S. Genomic epidemiology of the UK outbreak of the emerging human fungal pathogen Candida auris. Emerg. Microbes Infect. 2018, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.G.; Shin, J.H.; Uh, Y.; Kang, M.G.; Kim, S.H.; Park, K.H.; Jang, H.C. First three reported cases of nosocomial fungemia caused by Candida auris. J. Clin. Microbiol. 2011, 49, 3139–3142. [Google Scholar] [CrossRef] [PubMed]
- Greer, A.; Ng, V.; Fisman, D. Climate change and infectious diseases in North America: The road ahead. CMAJ 2008, 178, 715–722. [Google Scholar] [PubMed]
- Barrozo, L.V.; Mendes, R.P.; Marques, S.A.; Benard, G.; Siqueira Silva, M.E.; Bagagli, E. Climate and acute/subacute paracoccidioidomycosis in a hyper-endemic area in Brazil. Int. J. Epidemiol. 2009, 38, 1642–1649. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Solache, M.A.; Casadevall, A. Global warming will bring new fungal diseases for mammals. MBio 2010, 1, e00061-10. [Google Scholar] [CrossRef] [PubMed]
- Casadevall, A.; Kontoyiannis, D.P.; Robert, V. On the emergence of Candida auris: Climate change, azoles, swamps, and birds. MBio 2019, 10, e01397-19. [Google Scholar] [CrossRef] [PubMed]
- Ruiz Gaitán, A.C.; Moret, A.; López Hontangas, J.L.; Molina, J.M.; Aleixandre López, A.I.; Cabezas, A.H.; Mollar Maseres, J.; Arcas, R.C.; Gómez Ruiz, M.D.; Chiveli, M.Á.; et al. Fungemia nosocomial por Candida auris: Primeros cuatro casos en Europa continental. Rev. Iberoam. Micol. 2017, 34, 23–27. [Google Scholar] [CrossRef]
- Schelenz, S.; Hagen, F.; Rhodes, J.L.; Abdolrasouli, A.; Chowdhary, A.; Hall, A.; Ryan, L.; Shackleton, J.; Trimlett, R.; Meis, J.F.; et al. First hospital outbreak of the globally emerging Candida auris in a European hospital. Antimicrob. Resist. Infect. Control 2016, 5, 35. [Google Scholar] [CrossRef]
- Misseri, G.; Ippolito, M.; Cortegiani, A. Global warming “heating up” the ICU through Candida auris infections: The climate changes theory. Crit. Care 2019, 23, 416. [Google Scholar] [CrossRef]
- Jackson, B.R.; Chow, N.; Forsberg, K.; Litvintseva, A.P.; Lockhart, S.R.; Welsh, R.; Vallabhaneni, S.; Chiller, T. On the origins of a species: What might explain the rise of Candida auris? J. Fungi 2019, 5, 58. [Google Scholar] [CrossRef]
- Rudramurthy, S.M.; Chakrabarti, A.; Paul, R.A.; Sood, P.; Kaur, H.; Capoor, M.R.; Kindo, A.J.; Marak, R.S.K.; Arora, A.; Sardana, R.; et al. Candida auris candidaemia in Indian ICUs: Analysis of risk factors. J. Antimicrob. Chemother. 2017, 72, 1794–1801. [Google Scholar] [CrossRef]
- World Health Organization. Global Hepatitis Report 2017; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- UNODC. World Drug Report 2018: Executive Summary, Conclusions and Policy Implications; UNODC: Vienna, Austria, 2018. [Google Scholar]
- Smyth, B.P.; Barry, J.; Keenan, E. Irish injecting drug users and hepatitis C: The importance of the social context of injecting. Int. J. Epidemiol. 2005, 34, 166–172. [Google Scholar] [CrossRef]
- van Beek, I.; Buckley, R.; Stewart, M.; MacDonald, M.; Kaldor, J. Risk factors for hepatitis C infection among injecting drug users in Sydney. Genitourin. Med. 1994, 70, 321–324. [Google Scholar] [CrossRef]
- Kåberg, M.; Karlsson, N.; Discacciati, A.; Widgren, K.; Weiland, O.; Ekström, A.M.; Hammarberg, A. Significant decrease in injection risk behaviours among participants in a needle exchange programme. Infect. Dis. 2020, 52, 336–346. [Google Scholar] [CrossRef]
- Lunenfeld, B. An Aging World—Demographics and challenges. Gynecol. Endocrinol. 2008, 24, 1–3. [Google Scholar] [CrossRef] [PubMed]
- United Nations. Transforming Our World: The 2030 Agenda for Sustainable Development; United Nations: New York, NY, USA, 2015; Available online: https://sustainabledevelopment.un.org/post2015/transformingourworld (accessed on 16 October 2020).
- United Nations. SDG 3—Ensure Healthy Lives and Promote Well-Being for All at All Ages. Available online: https://sdgs.un.org/goals/goal3 (accessed on 20 October 2020).
- Buist, A.S.; Vollmer, W.M.; Sullivan, S.D.; Weiss, K.B.; Lee, T.A.; Menezes, A.M.B.; Crapo, R.O.; Jensen, R.L.; Burney, P.G.J.B. The Burden of Obstructive Lung Disease Initiative (BOLD): Rationale and design. COPD 2005, 2, 277–283. [Google Scholar] [CrossRef] [PubMed]
- BOLD Study. Burden of Obstructive Lung Disease. Scientific Objectives. Available online: https://www.boldstudy.org/scientific-objectives (accessed on 15 October 2020).
- Sator, L.; Horner, A.; Studnicka, M.; Lamprecht, B.; Kaiser, B.; McBurnie, M.A.; Buist, A.S.; Gnatiuc, L.; Mannino, D.M.; Janson, C.; et al. Overdiagnosis of COPD in subjects with unobstructed spirometry: A BOLD analysis. Chest 2019, 156, 277–288. [Google Scholar] [CrossRef]
- Burney, P.; Jithoo, A.; Kato, B.; Janson, C.; Mannino, D.; Nizankowska-Mogilnicka, E.; Studnicka, M.; Tan, W.; Bateman, E.; KoÇabas, A.; et al. Chronic obstructive pulmonary disease mortality and prevalence: The associations with smoking and poverty-A BOLD analysis. Thorax 2014, 69, 465–473. [Google Scholar] [CrossRef]
- Townend, J.; Minelli, C.; Mortimer, K.; Obaseki, D.O.; Al Ghobain, M.; Cherkaski, H.; Denguezli, M.; Gunesekera, K.; Hafizi, H.; Koul, P.A.; et al. The association between chronic airflow obstruction and poverty in 12 sites of the multinational BOLD study. Eur. Respir. J. 2017, 49, 1601880. [Google Scholar] [CrossRef] [PubMed]
- Bárbara, C.; Rodrigues, F.; Dias, H.; Cardoso, J.; Almeida, J.; Matos, M.J.; Simão, P.; Santos, M.; Ferreira, J.R.; Gaspar, M.; et al. Chronic obstructive pulmonary disease prevalence in Lisbon, Portugal: The Burden of Obstructive Lung Disease study. Rev. Port. Pneumol. 2013, 19, 96–105. [Google Scholar] [CrossRef]
- Dash, S.; Shakyawar, S.K.; Sharma, M.; Kaushik, S. Big data in healthcare: Management, analysis and future prospects. J. Big Data 2019, 6, 54. [Google Scholar] [CrossRef]
- Murdoch, T.B.; Detsky, A.S. The inevitable application of big data to health care. JAMA 2013, 309, 1351–1352. [Google Scholar] [CrossRef]
- Cirillo, D.; Valencia, A. Big data analytics for personalized medicine. Curr. Opin. Biotechnol. 2019, 58, 161–167. [Google Scholar] [CrossRef]
- Floras, J.S. Sleep apnea and cardiovascular disease an enigmatic risk factor. Circ. Res. 2018, 122, 1741–1764. [Google Scholar] [CrossRef]
- Staats, R.; Barros, I.; Fernandes, D.; Grencho, D.; Reis, C.; Matos, F.; Valença, J.; Marôco, J.; de Almeida, A.B.; Bárbara, C. The importance of sleep fragmentation on the hemodynamic dipping in obstructive sleep apnea patients. Front. Physiol. 2020, 11, 104. [Google Scholar] [CrossRef] [PubMed]
- Staats, R.; Rodrigues, R.; Barros, A.; Bacelar-Nicolau, L.; Aguiar, M.; Fernandes, D.; Moreira, S.; Simões, A.; Silva-Santos, B.; Rodrigues, J.V.; et al. Role of CD3 + γδ-T cells in the association of obstructive sleep-disordered breathing and cancer. Sleep Breath. 2019. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, A.S.; Cuco, C.M.; Lavareda, C.; Miguel, F.; Ventura, M.; Almeida, S.; Pinto, P.; De Abreu, T.T.; Rodrigues, L.V.; Seixas, S.; et al. Bronchoalveolar lavage proteomics in patients with suspected lung cancer. Sci. Rep. 2017, 7, 42190. [Google Scholar] [CrossRef]
- Feliciano, A.; Vaz, F.; Torres, V.M.; Valentim-Coelho, C.; Silva, R.; Prosinecki, V.; Alexandre, B.M.; Carvalho, A.S.; Matthiesen, R.; Malhotra, A.; et al. Evening and morning peroxiredoxin-2 redox/oligomeric state changes in obstructive sleep apnea red blood cells: Correlation with polysomnographic and metabolic parameters. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 621–629. [Google Scholar] [CrossRef] [PubMed]
- Moreira, S.; Rodrigues, R.; Barros, A.B.; Pejanovic, N.; Neves-Costa, A.; Pedroso, D.; Pereira, C.; Fernandes, D.; Rodrigues, J.V.; Barbara, C.; et al. Changes in expression of the CLOCK gene in obstructive sleep apnea syndrome patients are not reverted by continuous positive airway pressure treatment. Front. Med. 2017, 4, 187. [Google Scholar] [CrossRef]
- Staats, R.; Rodrigues, R.; Barros, A.; Bacelar-Nicolau, L.; Aguiar, M.; Fernandes, D.; Moreira, S.; Simões, A.; Silva-Santos, B.; Rodrigues, J.V.; et al. Decrease of perforin positive CD3+γδ-T cells in patients with obstructive sleep disordered breathing. Sleep Breath. 2018, 22, 211–221. [Google Scholar] [CrossRef]
- Gomes, S.; Cavadas, B.; Ferreira, J.C.; Marques, P.I.; Monteiro, C.; Sucena, M.; Sousa, C.; Vaz Rodrigues, L.; Teixeira, G.; Pinto, P.; et al. Profiling of lung microbiota discloses differences in adenocarcinoma and squamous cell carcinoma. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Brooks, D. Telehealth technology: an emerging method of delivering pulmonary rehabilitation to patients with chronic obstructive pulmonary disease. Can Respir J. 2011, 18, 196. [Google Scholar] [CrossRef]
- Peretti, A.; Amenta, F.; Tayebati, S.K.; Nittari, G.; Mahdi, S.S. Telerehabilitation: Review of the state-of-the-art and areas of application. JMIR Rehabil. Assist. Technol. 2017, 4, e7. [Google Scholar] [CrossRef]
- Faria, I.; Gaspar, C.; Zamith, M.; Matias, I.; César Das Neves, R.; Rodrigues, F.; Bárbara, C. TELEMOLD project: Oximetry and exercise telemonitoring to improve long-term oxygen therapy. Telemed. e-Health 2014, 20, 626–632. [Google Scholar] [CrossRef] [PubMed]
- Santos, C.D.; Rui, C.; Ribeiro, R.M.; Caneiras, C. Novel input for designing patient-tailored pulmonary rehabilitation: Telemonitoring physical activity as a vital sign—SMARTREAB Study. J. Clin. Med. 2020, 9, 2450. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Physical Activity Strategy for the WHO European Region 2016–2025; WHO: Copenhagen, Denmark, 2015. [Google Scholar]
- European Comission. Demography Report—Short Analytical Web Note; Publications Office of the European Union; European Comission: Louxembourg, 2015. [Google Scholar]
- UNECE/European Comission. Active Ageing Index 2014: Analytical Report; 2015. Available online: https://ec.europa.eu/eip/ageing/library/2014-active-ageing-index-aai-analytical-report_en (accessed on 16 October 2020).
- World Health Organization. World Report on Ageing and Health; WHO: Geneva, Switzerland, 2015. [Google Scholar]
- Zaidi, B.; Morgan, S.P. The second demographic transition theory: A review and appraisal. Annu. Rev. Sociol. 2017, 43, 473–492. [Google Scholar] [CrossRef] [PubMed]
- Reher, D.S. Economic and social implications of the demographic transition. Popul. Dev. Rev. 2011, 37, 11–33. [Google Scholar] [CrossRef]
- World Health Organization. The Ottawa Charter for Health Promotion. In Proceedings of the First International Conference on Health Promotion, Ottawa, ON, Canada, 21 November 1986. [Google Scholar]
- World Health Organization. European Food and Nutrition Action Plan 2015–2020; WHO: Copenhagen, Denmark, 2015. [Google Scholar]
- Santos, O.; Alarcão, V.; Feteira-Santos, R.; Fernandes, J.; Virgolino, A.; Sena, C.; Vieira, C.P.; Gregório, M.J.; Nogueira, P.; Graça, P.; et al. Impact of different front-of-pack nutrition labels on online food choices. Appetite 2020, 154, 104795. [Google Scholar] [CrossRef]
- Graça, P.; Silva, A.; Vieira, C.P.; Sena, C.; Gregório, M.J.; Nogueira, P.; Virgolino, A.; Fernandes, J.; Santos, O.; Santos, R.; et al. NUTR-HIA: Improving Nutrition Labelling in Portugal—Health Impact Assessment Final Report; Direção-Geral da Sáude: Lisboa, Portugal, 2019. [Google Scholar]
- Drinkwater, C.; Moffatt, S. Social prescribing. BMJ 2019, 364, l1285. [Google Scholar] [CrossRef]
- Husk, K.; Elston, J.; Gradinger, F.; Callaghan, L.; Asthana, S. Social prescribing: Where is the evidence? Br. J. Gen. Pract. 2019, 69, 6–7. [Google Scholar] [CrossRef]
- Isaacs, A.J.; Critchley, J.A.; Tai, S.S.; Buckingham, K.; Westley, D.; Harridge, S.D.R.; Smith, C.; Gottlieb, J.M. Exercise Evaluation Randomised Trial (EXERT): A randomised trial comparing GP referral for leisure centre-based exercise, community-based walking and advice only. Health Technol. Assess. 2007, 11, 1–165. [Google Scholar] [CrossRef]
- The Conservation Volunteers. Green Gym. Available online: https://www.tcv.org.uk/greengym/ (accessed on 14 October 2020).
- World Health Organization; Gobierno de España; IOM International Organization for Migration. Health of Migrants—The Way forward: Report of a Global Consultation; WHO: Madrid, Spain, 2010. [Google Scholar]
- Castañeda, H.; Holmes, S.M.; Madrigal, D.S.; Young, M.E.D.T.; Beyeler, N.; Quesada, J. Immigration as a social determinant of health. Annu. Rev. Public Health 2015, 36, 375–392. [Google Scholar] [CrossRef]
- Alarcão, V.; Stefanovska-Petkovska, M.; Virgolino, A.; Santos, O.; Ribeiro, S.; Costa, A.; Nogueira, P.; Pascoal, P.M.; Pintassilgo, S.; Machado, F.L. Fertility, Migration and Acculturation (FEMINA): A research protocol for studying intersectional sexual and reproductive health inequalities. Reprod. Health 2019, 16, 140. [Google Scholar] [CrossRef]
- Reis Oliveira, C.; Gomes, N. Indicadores de Integração de Imigrantes-Relatório Estatístico Anual 2018; Alto Comissariado para as Migrações: Lisboa, Portugal, 2018. [Google Scholar]
- Viner, R.M.; Ozer, E.M.; Denny, S.; Marmot, M.; Resnick, M.; Fatusi, A.; Currie, C. Adolescence and the social determinants of health. Lancet 2012, 379, 1641–1652. [Google Scholar] [CrossRef]
- Yang, T.C.; Chen, I.C.; Choi, S.W.; Kurtulus, A. Linking perceived discrimination during adolescence to health during mid-adulthood: Self-esteem and risk-behavior mechanisms. Soc. Sci. Med. 2019, 232, 434–443. [Google Scholar] [CrossRef] [PubMed]
- Landstedt, E.; Hammarstrom, A.; Winefield, H. How well do parental and peer relationships in adolescence predict health in adulthood? Scand. J. Public Health 2015, 43, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Matos, M.G.; Simões, C. From positive youth development to youth’s engagement: The Dream Teens. Int. J. Emot. Educ. 2016, 8, 4–17. [Google Scholar]
- Israel, B.A.; Schulz, A.J.; Parker, E.A.; Becker, A.B. Review of community-based research: Assessing partnership approaches to improve public health. Annu. Rev. Public Health 1998, 19, 173–202. [Google Scholar] [CrossRef]
- Horn, K.; McCracken, L.; Dino, G.; Brayboy, M. Applying community-based participatory research principles to the development of a smoking-cessation program for American Indian teens: “Telling our story. ” Health Educ. Behav. 2008, 35, 44–69. [Google Scholar] [CrossRef]
- Michie, S.; van Stralen, M.M.; West, R. The behaviour change wheel: A new method for characterising and designing behaviour change interventions. Implement. Sci. 2011, 6, 42. [Google Scholar] [CrossRef]
- Morgan, A. Social capital as a health asset for young people’s health and well-being. Rev. Psicol. Criança Adolesc. 2013, 1, 19–42. [Google Scholar]
- Morgan, A.; Davies, M.; Ziglio, E. Health Assets in a Global Context: Theory, Methods, Action; Springer: New York, NY, USA, 2010. [Google Scholar]
- Morgan, A.; Ziglio, E. Revitalising the evidence base for public health: An assets model. Promot. Educ. 2007, 14, 17–22. [Google Scholar] [CrossRef]
- Matos, M.G.; Equipa Aventura Social. Relatório do Estudo HBSC 2018: A Saúde Dos Adolescentes Portugueses Após a Recessão; Equipa Aventura Social: Lisboa, Portugal, 2018. [Google Scholar]
- World Health Organization Regional Office for Europe. Spotlight on Adolescent Health and Well-Being: Findings from the 2017/2018 Health Behaviour in School-Aged Children (HBSC) Survey in Europe and Canada; WHO Regional Office for Europe: Copenhagen, Denmark, 2018; Volume 1. [Google Scholar]
- Matos, M.G.; Wainwright, T.; Brebels, L.; Craciun, B.; Gabrhelík, R.; Schjodt, B.H.; Plantade-Gipch, A.; Poštuvan, V.; Stojadinovic, I.; Richards, J. Looking ahead: Challenges and opportunities for applied psychology in prevention and promotion. Eur. Psychol. 2019, 24, 1–12. [Google Scholar] [CrossRef]
- Matos, M.G.; Santos, T.; Reis, M.; Marques, A. Positive youth development: interactions between healthy lifestyle behaviours and psychosocial variables. Glob. J. Health Sci. 2018, 10, 68. [Google Scholar] [CrossRef][Green Version]
- Matos, M.G.; Camacho, I.; Reis, M.; Costa, D.; Galvão, D. Worries, coping strategies and well-being in adolescence: Highlights from HBSC study in Portugal. Vulnerable Child. Youth Stud. 2016, 11, 274–280. [Google Scholar] [CrossRef]
- Matos, M.G.; Camacho, I.; Reis, M.; Tomé, G.; Branquinho, C.; Ramiro, L. Is truth in the eyes of the beholder? Or are Portuguese schools, as viewed by Portuguese pupils, mismatching with what the educational system offers? Vulnerable Child. Youth Stud. 2018, 13, 116–126. [Google Scholar] [CrossRef]
- Branquinho, C.; Matos, M.G. The “Dream Teens” Project: After a two-year participatory action-research program. Child Indic. Res. 2019, 12, 1243–1257. [Google Scholar] [CrossRef]
- Levasseur, M.; Richard, L.; Gauvin, L.; Raymond, É. Inventory and analysis of definitions of social participation found in the aging literature: Proposed taxonomy of social activities. Soc. Sci. Med. 2010, 71, 2141–2149. [Google Scholar] [CrossRef] [PubMed]
- Jerliu, N.; Burazeri, G.; Toçi, E.; Kempen, G.I.J.M.; Jongen, W.; Ramadani, N.; Brand, H. Social networks, social participation and self-perceived health among older people in transitional Kosovo. Eur. J. Public Health 2014, 24, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Bukov, A.; Maas, I.; Lampert, T. Social participation in very old age: Cross-sectional and longitudinal findings from BASE. J. Gerontol. Ser. B Psychol. Sci. Soc. Sci. 2002, 57, 510–517. [Google Scholar] [CrossRef]
- Lee, H.Y.; Jang, S.N.; Lee, S.; Cho, S., II; Park, E.O. The relationship between social participation and self-rated health by sex and age: A cross-sectional survey. Int. J. Nurs. Stud. 2008, 45, 1042–1054. [Google Scholar] [CrossRef]
- Aroogh, M.D.; Shahboulaghi, F.M. Social participation of older adults: A concept analysis. Int. J. Community Based Nurs. Midwifery 2020, 8, 55–72. [Google Scholar]
- Teater, B. Intergenerational programs to promote active aging: the experiences and perspectives of older adults. Act. Adapt. Aging 2016, 40, 1–19. [Google Scholar] [CrossRef]
- Lai, J.; Ma, S.; Wang, Y.; Cai, Z.; Hu, J.; Wei, N.; Wu, J.; Du, H.; Chen, T.; Li, R.; et al. Factors associated with mental health outcomes among health care workers exposed to coronavirus disease 2019. JAMA Netw. Open 2020, 3, e203976. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, L. The untold toll—The pandemic’s effects on patients without covid-19. N. Engl. J. Med. 2020, 382, 2368–2371. [Google Scholar] [CrossRef] [PubMed]
- Søreide, K.; Hallet, J.; Matthews, J.B.; Schnitzbauer, A.A.; Line, P.D.; Lai, P.B.S.; Otero, J.; Callegaro, D.; Warner, S.G.; Baxter, N.N.; et al. Immediate and long-term impact of the COVID-19 pandemic on delivery of surgical services. Br. J. Surg. 2020, 107, 1250–1261. [Google Scholar] [CrossRef] [PubMed]
- Costa, L.G.; Eaton, D.L. (Eds.). Gene-Environment Interactions: Fundamentals of Ecogenetics; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2006. [Google Scholar]
- Mendes, A.I.; Mascarenhas, M.R.; Matos, S.; Sousa, I.; Ferreira, J.; Barbosa, A.P.; Bicho, M.; Jordan, P. A WNK4 gene variant relates to osteoporosis and not to hypertension in the Portuguese population. Mol. Genet. Metab. 2011, 102, 465–469. [Google Scholar] [CrossRef]
- Guerra, A.; Rego, C.; Laires, M.J.; Castro, E.M.; Silva, D.; Monteiro, C.; Silva, Z.; Lebre, A.; Bicho, M. Lipid profile and redox status in high performance rhythmic female teenagers gymnasts. J. Sports Med. Phys. Fit. 2001, 41, 505–512. [Google Scholar]
- Marinho, C.; Alho, I.; Arduíno, D.; Falcão, L.M.; Brás-Nogueira, J.; Bicho, M. GST M1/T1 and MTHFR polymorphisms as risk factors for hypertension. Biochem. Biophys. Res. Commun. 2007, 353, 344–350. [Google Scholar] [CrossRef]
- Laires, M.J.; Monteiro, C.P.; Bicho, M. Role of cellular magnesium in health and human disease. Front. Biosci. 2004, 9, 262–276. [Google Scholar] [CrossRef]
- Pombo, S.; Levy, P.; Bicho, M.; Ismail, F.; Neves Cardoso, J.M. Neuropsychological function and platelet monoamine oxidase activity levels in type I alcoholic patients. Alcohol. Alcohol. 2008, 43, 423–430. [Google Scholar] [CrossRef]
- Alho, I.; Costa, L.; Bicho, M.; Coelho, C. The role of low-molecular-weight protein tyrosine phosphatase (LMW-PTP ACP1) in oncogenesis. Tumor Biol. 2013, 34, 1979–1989. [Google Scholar] [CrossRef]
- Apelt, N.; Pereira da Silva, A.; Ferreira, J.; Alho, I.; Monteiro, C.; Marinho, C.; Teixeira, P.; Sardinha, L.; Laires, M.J.; Mascarenhas, M.R.; et al. ACP1 genotype, glutathione reductase activity, and riboflavin uptake affect cardiovascular risk in the obese. Metabolism 2009, 58, 1415–1423. [Google Scholar] [CrossRef]
- Almeida, E.; Alho, I.; Marques, F.; Thiran, C.; Bicho, M.; Prata, M. Haemoglobin and erythropoietin levels in polycystic kidney disease. Nephrol. Dial. Transplant. 2008, 23, 412–413. [Google Scholar] [CrossRef][Green Version]
- Aguiar, L.; Matos, A.; Gil, Â.; Afonso, C.; Almeida, S.; Braga, L.; Lavinha, J.; Kjollerstrom, P.; Faustino, P.; Bicho, M.; et al. Sickle cell anemia—Nitric oxide related genetic modifiers of hematological and biochemical parameters. Clin. Hemorheol. Microcirc. 2016, 64, 957–963. [Google Scholar] [CrossRef] [PubMed]
- Swinburn, B.A.; Kraak, V.I.; Allender, S.; Atkins, V.J.; Baker, P.I.; Bogard, J.R.; Brinsden, H.; Calvillo, A.; De Schutter, O.; Devarajan, R.; et al. The global syndemic of obesity, undernutrition, and climate change: The Lancet Commission report. Lancet 2019, 393, 791–846. [Google Scholar] [CrossRef]
- Lally, P.; Gardner, B. Promoting habit formation. Health Psychol. Rev. 2013, 7, S137–S158. [Google Scholar] [CrossRef]
- Armitage, C.J.; Conner, M. Social cognition models and health behaviour: A structured review. Psychol. Health 2000, 15, 173–189. [Google Scholar] [CrossRef]
- Hofmann, W.; Friese, M.; Wiers, R.W. Impulsive versus reflective influences on health behavior: A theoretical framework and empirical review. Health Psychol. Rev. 2008, 2, 111–137. [Google Scholar] [CrossRef]
- Baumeister, R.F.; Bratslavsky, E.; Muraven, M.; Tice, D.M. Ego depletion: Is the active self a limited resource? J. Pers. Soc. Psychol. 1998, 74, 1252–1265. [Google Scholar] [CrossRef]
- Deci, E.L.; Ryan, R.M. Intrinsic Motivation and Self-Determination in Human Behavior; Springer Science & Business Media: New York, NY, USA, 1985. [Google Scholar]
- Thaler, R. Toward a positive theory of consumer choice. J. Econ. Behav. Organ. 1980, 1, 39–60. [Google Scholar] [CrossRef]
- Thaler, R.H.; Sunstein, C.R. Nudge: Improving Decisions about Health, Wealth, and Happiness; Yale University Press: New Haven, USA, 2008; ISBN 978-0-300-12223-7. [Google Scholar]
- Schwartz, S.H. Are there universal aspects in the structure and contents of human values? J. Soc. Issues 1994, 50, 19–45. [Google Scholar] [CrossRef]
- Michie, S.; Thomas, J.; Johnston, M.; Mac Aonghusa, P.; Shawe-Taylor, J.; Kelly, M.P.; Deleris, L.A.; Finnerty, A.N.; Marques, M.M.; Norris, E.; et al. The Human Behaviour-Change Project: Harnessing the power of artificial intelligence and machine learning for evidence synthesis and interpretation. Implement. Sci. 2017, 12, 121. [Google Scholar] [CrossRef] [PubMed]
- Bronfenbrenner, U. The Ecology of Human Development: Experiments by Nature and Design; Harvard University Press: Cambridge, MA, USA, 1979. [Google Scholar]
- Berkman, E.T. Value-based choice: An integrative, neuroscience-informed model of health goals. Psychol. Health 2018, 33, 40–57. [Google Scholar] [CrossRef] [PubMed]
- Holtzhausen, D.; Zerfass, A. The Routledge Handbook of Strategic Communication; Routledge: New York, NY, USA, 2015. [Google Scholar]
- Parvanta, C. A Public Health Communication Planning Framework. In Essentials of Public Health Communication; Parvanta, C., Nelson, D.E., Parvanta, S.A., Harner, R.N., Eds.; Jones & Bartlet Learning LLC: Sudbury, MA, USA, 2011; pp. 19–38. [Google Scholar]
- Richard, L.; Gauvin, L.; Raine, K. Ecological models revisited: Their uses and evolution in health promotion over two decades. Annu. Rev. Public Health 2011, 32, 307–326. [Google Scholar] [CrossRef]
- Mcleroy, K.R.; Bibeau, D.; Steckler, A.; Glanz, K. An ecological perspective on health promotion programs. Health Educ. Behav. 1988, 15, 351–377. [Google Scholar] [CrossRef]
- Thomas, S. (Ed.) Antimicrobial Resistance; Springer: Singapore, 2020; ISBN 978-981-15-3657-1. [Google Scholar]
- World Health Organization. Global Action Plan on Antimicrobial Resistance; WHO: Geneva, Switzerland, 2015. [Google Scholar]
- Asaduzzaman, M. Antimicrobial resistance: An urgent need for a planetary and ecosystem approach. Lancet Planet. Health 2018, 2, e99–e100. [Google Scholar] [CrossRef]
- Laxminarayan, R.; Sridhar, D.; Blaser, M.; Wang, M.; Woolhous, M. Achieving global targets for antimicrobial resistance. Policy Forum 2016, 353, 874–875. [Google Scholar] [CrossRef]
- Collignon, P. Antibiotic resistance: Are we all doomed? Intern. Med. J. 2015, 45, 1109–1115. [Google Scholar] [CrossRef]
- Kapoor, G.; Saigal, S.; Elongavan, A. Action and resistance mechanisms of antibiotics: A guide for clinicians. J. Anaesthesiol. Clin. Pharmacol. 2017, 33, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Eichenberger, E.M.; Thaden, J.T. Epidemiology and mechanisms of resistance of extensively drug resistant gram-negative bacteria. Antibiotics 2019, 8, 37. [Google Scholar] [CrossRef] [PubMed]
- Papp-Wallace, K.M.; Endimiani, A.; Taracila, M.A.; Bonomo, R.A. Carbapenems: Past, present, and future. Antimicrob. Agents Chemother. 2011, 55, 4943–4960. [Google Scholar] [CrossRef] [PubMed]
- Caneiras, C.; Lito, L.; Mayoralas-Alises, S.; Díaz-Lobato, S.; Melo-Cristino, J.; Duarte, A. Virulence and resistance determinants of Klebsiella pneumoniae isolated from a Portuguese tertiary university hospital centre over a 31-year period. Enferm. Infecc. Microbiol. Clin. 2019, 37, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Collignon, P.; Beggs, J.J.; Walsh, T.R.; Gandra, S.; Laxminarayan, R. Anthropological and socioeconomic factors contributing to global antimicrobial resistance: A univariate and multivariable analysis. Lancet Planet. Health 2018, 2, e398–e405. [Google Scholar] [CrossRef]
- Zhu, Y.G.; Zhao, Y.; Zhu, D.; Gillings, M.; Penuelas, J.; Ok, Y.S.; Capon, A.; Banwart, S. Soil biota, antimicrobial resistance and planetary health. Environ. Int. 2019, 131, 105059. [Google Scholar] [CrossRef]
- Caneiras, C.; Jácome, C.; Mayoralas-Alises, S.; Ramon Calvo, J.; Almeida Fonseca, J.; Escarrabill, J.; Winck, J.C. Patient experience in home respiratory therapies: Where we are and where to go. J. Clin. Med. 2019, 8, 555. [Google Scholar] [CrossRef]
- Karliner, J.; Slotterback, S.; Boyd, R.; Ashby, B.; Steele, K. Health Care’s Climate Footprint: How the Health Sector Contributes to the Global Climate Crisis and Opportunities for Action. 2019. Available online: https://noharm-uscanada.org/ClimateFootprintReport (accessed on 16 October 2020).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).